Abstract
Background
Previous studies have identified sarcopenia as a significant risk factor for cardiovascular disease (CVD). However, these studies primarily focused on sarcopenia status at baseline, without considering changes in sarcopenia status during follow-up. The aim of this study is to investigate the association between changes in sarcopenia status and the incidence of new-onset cardiovascular disease.
Methods
This study utilized prospective cohort data from the China Health and Retirement Longitudinal Study (CHARLS). Sarcopenia status was assessed using the 2019 Asian Working Group for Sarcopenia (AWGS) algorithm and categorized as non-sarcopenia, possible sarcopenia, or sarcopenia. Changes in sarcopenia status were evaluated based on assessments at baseline and at the second follow-up survey 2 years later. CVD was identified through self-reported physician diagnoses of heart disease, including angina, myocardial infarction, congestive heart failure, and other heart problems, or stroke. Cox proportional hazards models were employed to calculate hazard ratios (HRs) and 95% confidence intervals (CIs), adjusting for potential confounding factors.
Results
Based on the inclusion and exclusion criteria, a total of 7499 CHARLS participants were included in the analysis, with 50.8% being female and an average age of 58.5 years. Compared to participants with stable non-sarcopenia status, those who progressed from non-sarcopenia to possible sarcopenia or sarcopenia exhibited a significantly increased risk of new-onset CVD (HR 1.30, 95% CI 1.06–1.59). Conversely, participants who recovered from sarcopenia to non-sarcopenia or possible sarcopenia had a significantly reduced risk of new-onset CVD compared to those with stable sarcopenia status (HR 0.61, 95% CI 0.37–0.99). Among participants with baseline possible sarcopenia, those who recovered to non-sarcopenia had a significantly lower risk of new-onset CVD compared to those with stable possible sarcopenia status (HR 0.67, 95% CI 0.52–0.86).
Conclusions
Changes in sarcopenia status are associated with varying risks of new-onset CVD. Progression in sarcopenia status increases the risk, while recovery from sarcopenia reduces the risk of developing cardiovascular disease.
Graphical Abstract

Supplementary Information
The online version contains supplementary material available at 10.1186/s12916-024-03841-x.
Keywords: Sarcopenia, Cardiovascular disease, Dynamic nature, Epidemiology
Background
Sarcopenia is a skeletal muscle disorder commonly observed in middle-aged and older adults, characterized by a decline in muscle strength, mass, and function due to factors such as inflammation, insulin resistance, and mitochondrial dysfunction [1–3]. The prevalence of sarcopenia varies by region and age, ranging from 1 to 29% in community-dwelling populations and from 14 to 33% in long-term care facilities [4]. Sarcopenia leads to a range of adverse clinical outcomes, including increased risk of falls and fractures, progression to physical disability, more frequent hospitalization, reduced quality of life, and elevated mortality risk [5–7]. As the prevalence and incidence of cardiovascular disease (CVD) also rise with age, sarcopenia has garnered significant attention from cardiologists [8–11]. Understanding the association between sarcopenia and CVD could provide new insights into the prevention and management of CVD in the aging population.
Previous studies have found that possible sarcopenia and sarcopenia increase the risk of new-onset CVD, but these studies primarily focused on sarcopenia status at baseline without considering changes in sarcopenia status during follow-up [10, 12, 13]. Investigating changes in sarcopenia status, rather than assessing sarcopenia only at baseline, could reveal a more comprehensive biological link, such as the association between sarcopenia progression and CVD. Importantly, growing evidence suggests that sarcopenia can be reversed with appropriate interventions [14, 15]. Evaluating the risk of new-onset CVD in individuals who recover from sarcopenia could provide critical evidence for incorporating sarcopenia interventions into cardiovascular practice. Therefore, it is imperative to study the association between changes in sarcopenia status and the risk of new-onset CVD.
In this study, we utilized prospective cohort data from the China Health and Retirement Longitudinal Study (CHARLS) to explore the association between changes in sarcopenia status and the risk of new-onset CVD. We hypothesize that the progression of sarcopenia status increases the risk of new-onset CVD, while the recovery from sarcopenia reduces this risk.
Methods
Study design and population
This study utilized data from the China Health and Retirement Longitudinal Study (CHARLS), a prospective, nationally representative cohort study conducted in China. The detailed study design is summarized in the methods section of the supplementary data. In this study, the first wave of CHARLS (2011) was considered the baseline, and the second wave (2013) served as the follow-up survey (Additional file 2: Fig. S1). Data from these two waves were used to assess the dynamic changes in sarcopenia status, with subsequent surveys used to track outcomes until the final fifth wave (2020). The study was approved by the Peking University Institutional Review Board, and informed consent was obtained from each participant.
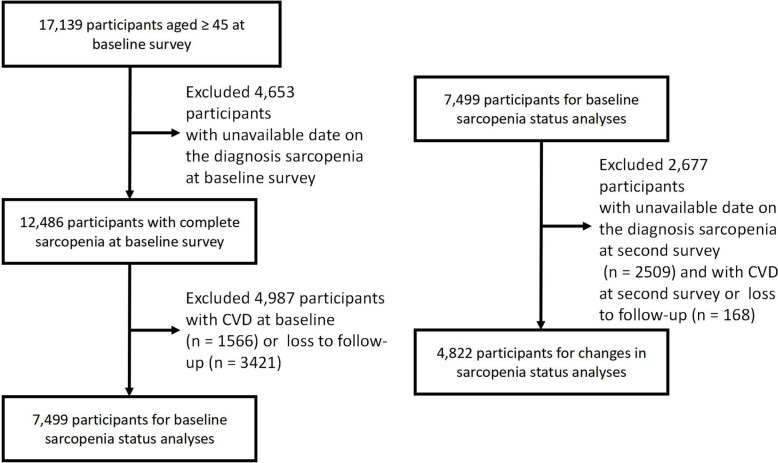
Figure 1 illustrates the selection process of the study population. Among the 17,139 CHARLS participants, 4653 individuals were excluded due to missing sarcopenia status data at baseline (including appendicular skeletal muscle mass(ASM), grip strength, and physical performance). Additionally, 4987 participants who had CVD at baseline or were lost to follow-up were also excluded. Ultimately, 7499 eligible participants were included in the baseline sarcopenia status analysis. For the analysis of changes in sarcopenia status, a further 2677 participants were excluded based on similar criteria, leaving 4822 participants in the final analysis.
Fig. 1.
Selection process of the study population. CVD, cardiovascular disease
Assessment of sarcopenia status
Sarcopenia status was evaluated using the 2019 Asian Working Group for Sarcopenia (AWGS) algorithm, which includes three components: muscle strength, ASM, and physical performance [16]. Sarcopenia is diagnosed when low muscle mass is detected along with either low muscle strength or low physical performance. Severe sarcopenia is diagnosed when there is low muscle mass combined with low muscle strength and low physical performance. Possible sarcopenia is diagnosed when there is low muscle strength or low physical performance.
Grip strength (kg) was measured in both the dominant and non-dominant hands using a Yuejian™ WL-1000 dynamometer (Nantong Yuejian Physical Measurement Instrument Co., Ltd., China), with participants instructed to squeeze as hard as possible [17]. The cutoff values for low grip strength were < 28 kg for men and < 18 kg for women.
ASM was estimated using a validated anthropometric equation suitable for the Chinese population [18, 19]. Multiple studies have demonstrated strong concordance between ASM equation models and dual-energy X-ray absorptiometry (DXA) [18–21]. The cutoff values for low muscle mass were based on the sex-specific values in the lowest 20% of the study population, adjusted for height (ASM/Ht2) [19, 22]. In this study, weight and height were measured to the nearest 0.1 cm and 0.1 kg using a stadiometer and digital scale, respectively [23]. The final ASM/Ht2 cutoff values for men were < 7.01, < 7.05, and < 7.07 kg/m2 in 2011, 2013, and 2015, respectively, and for women, the cutoff values were < 5.31, < 5.38, and < 5.39 kg/m2.
For physical performance, gait speed and chair stand tests were conducted following the methods described by Wu et al. [23]. Detailed definitions of the sarcopenia components in CHARLS have been previously described [16]. In our study population, only 180 participants (2.4%) were classified as having severe sarcopenia at baseline. Therefore, participants with severe sarcopenia were merged into the sarcopenia group, resulting in three baseline groups: non-sarcopenia (n = 4860), possible sarcopenia (n = 1874), and sarcopenia (n = 765).
Ascertainment of covariates
Covariates were chosen based on their established relevance in the existing literature on sarcopenia and cardiovascular disease, as well as data availability. The covariates considered in the analysis included age, sex, marital status, education level, residence location, smoking status, drinking status, body mass index (BMI), diastolic blood pressure (DBP), C-reactive protein (CRP), glycated hemoglobin (HbA1c), triglycerides (TG), non-high-density lipoprotein cholesterol (NonHDL-C), and common comorbidities such as diabetes mellitus (DM) and hypertension. Marital status was categorized as married or other (which included separated, divorced, never married, or widowed). Education level was divided into two categories: junior high school or below, and senior high school or above. Residence location was classified as rural or urban. Smoking status was categorized as never smokers and ever smokers, with the latter including former and current smokers. Similarly, drinking status was divided into never drinkers and ever drinkers.
Assessment of CVD events
The study outcome was CVD events, including heart disease and stroke. Consistent with previous studies, CVD events were assessed through the following questions: “Have you been told by a doctor that you have been diagnosed with a heart attack, angina, coronary heart disease, heart failure, or other heart problems?” and “Have you been told by a doctor that you have been diagnosed with a stroke?”. Participants who reported having heart disease or stroke were classified as having CVD [24, 25].
Statistical analysis
For descriptive statistics, continuous variables were presented as mean (standard deviation) or median (interquartile range), while categorical variables were expressed as count (percentage). To analyze the association between baseline sarcopenia status and the risk of new-onset CVD, hazard ratios (HRs) and their 95% confidence intervals (95% CIs) were calculated using Cox proportional hazards regression models. Four models were estimated: Model 0 was an unadjusted model to estimate crude HRs; Model 1 adjusted for age and sex; Model 2 further adjusted for marital status, residence location, and education level; and Model 3 additionally adjusted for alcohol consumption, smoking status, BMI, DBP, CRP, HbA1c, TG, NonHDL-C, and histories of diabetes and hypertension. The missing rates of covariates were summarized in Additional file 1: Tables S1 and S2. Missing data for covariates were imputed using multiple imputation by chained equations, with the specific methods described in the Additional file 2: Supplemental Methods [13, 26–31]. Similar methods were used to analyze the association with changes in sarcopenia status. The proportional hazards assumption of the Cox regression models was confirmed using Schoenfeld residuals [32].
For the analysis of changes in sarcopenia status, stratified analyses by sex and age (middle-aged: < 65 years; older adults: ≥ 65 years) were also conducted. The statistical significance of interactions was assessed using likelihood ratio tests. Several sensitivity analyses were performed regarding changes in sarcopenia status: (i) to reduce bias from potentially unstable changes in sarcopenia status, sarcopenia status was reassessed at the third survey to ensure stability of the changes (Additional file 1: Fig. S2); (ii) the primary analysis was repeated with additional adjustments for the use of antihypertensive, antidiabetic and lipid-lowering medications; (iii) the possible sarcopenia/sarcopenia group was separated from the non-sarcopenia/possible sarcopenia group.
All statistical analyses were conducted using R software (version 4.4.1). All P-values were two-sided, with P < 0.05 considered statistically significant.
Results
Baseline characteristics of the study population
Based on the inclusion and exclusion criteria, a total of 7499 participants (50.8% female, average age 58.5 years) were included in the baseline sarcopenia status analysis. The baseline characteristics of these participants are detailed in Table 1. Compared to participants without sarcopenia, those with sarcopenia were generally older, less likely to be married, had lower educational attainment, and were more likely to reside in rural areas. Additionally, sarcopenic participants had lower BMI, DBP, TG, and non-HDL cholesterol levels, but higher CRP levels.
Table 1.
Baseline characteristics of participants for baseline sarcopenia status analyses
| Characteristics | Total (n = 7499) | Non sarcopenia (n = 4860) | Possible sarcopenia (n = 1874) | Sarcopenia (n = 765) | P value |
|---|---|---|---|---|---|
| Age, mean (SD), years | 58.46 ± 9.20 | 56.13 ± 7.88 | 60.31 ± 9.12 | 68.72 ± 9.11 | < 0.0001 |
| Sex, n (%) | < 0.0001 | ||||
| Female | 3812 (50.83) | 2311 (47.55) | 1080 (57.63) | 421 (55.03) | |
| Male | 3687 (49.17) | 2549 (52.45) | 794 (42.37) | 344 (44.97) | |
| Marital status, n (%) | < 0.0001 | ||||
| Married | 6657 (88.77) | 4480 (92.18) | 1628 (86.87) | 549 (71.76) | |
| Others | 842 (11.23) | 380 ( 7.82) | 246 (13.13) | 216 (28.24) | |
| Education, n (%) | < 0.0001 | ||||
| Junior and below | 6609 (88.16) | 4129 (84.98) | 1733 (92.53) | 747 (97.65) | |
| Senior and above | 890 (11.84) | 731 (15.02) | 141 ( 7.47) | 18 ( 2.35) | |
| Residence, n (%) | < 0.0001 | ||||
| Rural | 4793 (63.92) | 3003 (61.79) | 1199 (63.98) | 591 (77.25) | |
| Urban | 2706 (36.08) | 1857 (38.21) | 675 (36.02) | 174 (22.75) | |
| Drinking status, n (%) | < 0.0001 | ||||
| Ever drinkers | 3105 (41.41) | 2167 (44.59) | 670 (35.75) | 268 (35.03) | |
| Never drinkers | 4394 (58.59) | 2693 (55.41) | 1204 (64.25) | 497 (64.97) | |
| Smoking status, n (%) | < 0.0001 | ||||
| Ever smokers | 3052 (40.70) | 2083 (42.86) | 663 (35.40) | 306 (40.00) | |
| Never smokers | 4446 (59.30) | 2777 (57.14) | 1210 (64.60) | 459 (60.00) | |
| BMI, mean (SD), kg/m2 | 23.50 ± 3.85 | 23.71 ± 3.72 | 24.73 ± 3.54 | 19.12 ± 1.84 | < 0.0001 |
| DBP, mean (SD), mmHg | 75.75 ± 11.90 | 75.93 ± 11.66 | 76.61 ± 12.07 | 72.51 ± 12.45 | < 0.0001 |
| CRP median (IQR), mg/L | 1.01 (0.54,2.07) | 0.99 (0.53,2.00) | 1.07 (0.57,2.14) | 0.98 (0.53,2.36) | 0.01 |
| HbA1c, mean (SD), % | 5.24 ± 0.79 | 5.24 ± 0.78 | 5.28 ± 0.84 | 5.17 ± 0.69 | < 0.01 |
| Triglycerides, mean (SD), mmol/L | 1.52 ± 1.13 | 1.55 ± 1.18 | 1.56 ± 1.13 | 1.26 ± 0.76 | < 0.0001 |
| NonHDL cholesterol, mean (SD), mmol/L | 3.69 ± 1.01 | 3.70 ± 1.03 | 3.74 ± 0.97 | 3.56 ± 0.99 | < 0.001 |
| DM, n (%) | < 0.001 | ||||
| No | 6607 (88.21) | 4293 (88.48) | 1613 (86.12) | 701 (91.63) | |
| Yes | 883 (11.79) | 559 (11.52) | 260 (13.88) | 64 ( 8.37) | |
| Hypertension, n (%) | < 0.0001 | ||||
| No | 4679 (62.39) | 3208 (66.01) | 1021 (54.48) | 450 (58.82) | |
| Yes | 2820 (37.61) | 1652 (33.99) | 853 (45.52) | 315 (41.18) |
BMI body mass index, DBP diastolic blood pressure, CRP C-reactive protein, HbA1c glycated hemoglobin, HDL-C high-density lipoprotein cholesterol, DM diabetes
For the analysis of changes in sarcopenia status, 4822 participants (51.6% female, average age 58.4 years) were included based on the relevant criteria, with their baseline characteristics presented in Table 2. We also described the baseline characteristics of participants using non-imputed data (Additional file 1: Tables S3 and S4), which yielded results similar to those in Tables 1 and 2.
Table 2.
Baseline characteristics of participants for changes in sarcopenia status analyses
| Characteristics | Total (n = 4822) | Non sarcopenia (n = 3213) | Possible sarcopenia (n = 1162) | Sarcopenia (n = 447) | P value |
|---|---|---|---|---|---|
| Age, mean (SD), years | 58.37 ± 8.78 | 56.50 ± 7.88 | 59.95 ± 8.63 | 67.75 ± 8.48 | < 0.0001 |
| Sex, n (%) | < 0.0001 | ||||
| Female | 2490 (51.64) | 1559 (48.52) | 686 (59.04) | 245 (54.81) | |
| Male | 2332 (48.36) | 1654 (51.48) | 476 (40.96) | 202 (45.19) | |
| Marital status, n (%) | < 0.0001 | ||||
| Married | 4350 (90.21) | 2987 (92.97) | 1024 (88.12) | 339 (75.84) | |
| Others | 472 ( 9.79) | 226 ( 7.03) | 138 (11.88) | 108 (24.16) | |
| Education, n (%) | < 0.0001 | ||||
| Junior and below | 4277 (88.70) | 2767 (86.12) | 1076 (92.60) | 434 (97.09) | |
| Senior and above | 545 (11.30) | 446 (13.88) | 86 ( 7.40) | 13 ( 2.91) | |
| Residence, n (%) | < 0.0001 | ||||
| Rural | 3246 (67.32) | 2097 (65.27) | 790 (67.99) | 359 (80.31) | |
| Urban | 1576 (32.68) | 1116 (34.73) | 372 (32.01) | 88 (19.69) | |
| Drinking status, n (%) | < 0.0001 | ||||
| Ever drinkers | 1974 (40.94) | 1412 (43.95) | 402 (34.60) | 160 (35.79) | |
| Never drinkers | 2848 (59.06) | 1801 (56.05) | 760 (65.40) | 287 (64.21) | |
| Smoking status, n (%) | < 0.001 | ||||
| Ever smokers | 1917 (39.76) | 1339 (41.67) | 405 (34.85) | 173 (38.70) | |
| Never smokers | 2905 (60.24) | 1874 (58.33) | 757 (65.15) | 274 (61.30) | |
| BMI, mean (SD), kg/m2 | 23.49 ± 3.81 | 23.67 ± 3.73 | 24.69 ± 3.45 | 19.09 ± 1.71 | < 0.0001 |
| DBP, mean (SD), mmHg | 75.44 ± 11.91 | 75.60 ± 11.69 | 76.40 ± 12.26 | 71.76 ± 11.94 | < 0.0001 |
| CRP, median (IQR), mg/L | 0.99 (0.53,1.95) | 0.97 (0.52,1.95) | 1.06 (0.56,1.96) | 0.93 (0.52,1.94) | 0.15 |
| HbA1c, mean (SD), % | 5.24 ± 0.78 | 5.24 ± 0.79 | 5.28 ± 0.83 | 5.13 ± 0.56 | < 0.01 |
| Triglycerides, mean (SD), mmol/L | 1.51 ± 1.17 | 1.55 ± 1.24 | 1.52 ± 1.12 | 1.22 ± 0.68 | < 0.0001 |
| NonHDL cholesterol, mean (SD), mmol/L | 3.69 ± 1.01 | 3.69 ± 1.04 | 3.72 ± 0.96 | 3.57 ± 0.96 | 0.03 |
| DM, n (%) | < 0.001 | ||||
| No | 4249 (88.17) | 2835 (88.32) | 998 (85.89) | 416 (93.06) | |
| Yes | 570 (11.83) | 375 (11.68) | 164 (14.11) | 31 ( 6.94) | |
| Hypertension, n (%) | < 0.0001 | ||||
| No | 3065 (63.56) | 2137 (66.51) | 642 (55.25) | 286 (63.98) | |
| Yes | 1757 (36.44) | 1076 (33.49) | 520 (44.75) | 161 (36.02) |
BMI body mass index, DBP diastolic blood pressure, CRP C-reactive protein, HbA1c glycated hemoglobin, HDL-C high-density lipoprotein cholesterol, DM diabetes
In the baseline sarcopenia status analysis, the median follow-up period in CHARLS was 9 years, during which 1,345 participants developed CVD. In the analysis of changes in sarcopenia status, the median follow-up period was 7 years, during which 929 participants developed CVD.
Association of baseline sarcopenia status with incident cardiovascular disease
The association between baseline sarcopenia status and the risk of new-onset CVD is presented in Additional file 1: Table S5. After adjusting for confounding factors, participants with possible sarcopenia had a significantly increased risk of developing CVD compared to those without sarcopenia (HR 1.25, 95% CI 1.11–1.42). Although participants with sarcopenia also showed a trend toward an increased risk of CVD compared to those without sarcopenia, this finding did not reach statistical significance (HR 1.01, 95% CI 0.81–1.26). Additionally, the relationship between baseline sarcopenia status and the risks of heart disease and stroke is detailed in Additional file 1: Table S5.
Association of changes in sarcopenia status with incident cardiovascular disease
Table 3 presents the number and percentage of participants whose sarcopenia status changed over the 2-year follow-up period. Among participants without sarcopenia at baseline, 615 (19.1%) progressed to possible sarcopenia or sarcopenia. Conversely, among those with sarcopenia at baseline, 219 (49.0%) recovered to a non-sarcopenia or possible sarcopenia status.
Table 3.
Number and percentage of the changes in sarcopenia status
The time interval between baseline and the second survey was two years in the CHARLS
Table 4 shows the association between changes in sarcopenia status and the risk of new-onset CVD. Compared to participants with stable non-sarcopenia status, those who progressed to possible sarcopenia/sarcopenia exhibited a significantly increased risk of new-onset CVD (HR 1.30, 95% CI 1.06–1.59). In contrast, participants with sarcopenia at baseline who recovered to a non-sarcopenia/possible sarcopenia status had a significantly reduced risk of new-onset CVD compared to those with stable sarcopenia status (HR 0.61, 95% CI 0.37–0.99). For participants with possible sarcopenia at baseline, those who recovered to a non-sarcopenia status had a significantly lower risk of new-onset CVD compared to those who remained in the possible sarcopenia status (HR 0.67, 95% CI 0.52–0.86). Additionally, participants with baseline possible sarcopenia who progressed to sarcopenia did not show a statistically significant increase in risk.
Table 4.
Association of changes in sarcopenia status with risks of incident cardiovascular disease
| Crude model | Model 1 | Model 2 | Model 3 | ||
|---|---|---|---|---|---|
| Character | Events/n | HR (95%CI) | HR (95%CI) | HR (95%CI) | HR (95%CI) |
| Stable non-sarcopenia | 420/2598 | Reference | Reference | Reference | Reference |
| Non-sarcopenia to possible sarcopenia/sarcopenia | 130/615 | 1.36 (1.11,1.65) | 1.24 (1.01,1.52) | 1.26 (1.03,1.54) | 1.30 (1.06,1.59) |
| Stable possible sarcopenia | 141/447 | Reference | Reference | Reference | Reference |
| Possible sarcopenia to non-sarcopenia | 148/670 | 0.65 (0.52,0.82) | 0.65 (0.51,0.82) | 0.66 (0.51,0.83) | 0.67 (0.52,0.86) |
| Possible sarcopenia to sarcopenia | 8/45 | 0.54 (0.26,1.09) | 0.55 (0.27,1.13) | 0.56 (0.27,1.14) | 0.61 (0.29,1.25) |
| Stable sarcopenia | 47/228 | Reference | Reference | Reference | Reference |
| Sarcopenia to non-sarcopenia/possible sarcopenia | 35/219 | 0.75 (0.48,1.15) | 0.73 (0.46,1.15) | 0.73 (0.46,1.16) | 0.61 (0.37,0.99) |
Model 1 included adjustments for age and sex; Model 2 further adjusted for marriage, residence, and education level; and Model 3 additionally adjusted for drinking, smoking, BMI, DBP, CRP, HbA1c, TG, NonHDL-C, as well as history of DM, and hypertension
CVD cardiovascular disease, BMI body mass index, DBP diastolic blood pressure, CRP C-reactive protein, HbA1c glycated hemoglobin, TG triglycerides, HDL-C high-density lipoprotein cholesterol, DM diabetes
Additional file 1: Table S6 provides a detailed description of the association between changes in sarcopenia status and the risk of new-onset heart disease and stroke.
Subgroup analyses and sensitivity analyses
In the subgroup analysis, compared to participants with stable non-sarcopenia status, women and participants aged 65 years and older who progressed from non-sarcopenia to possible sarcopenia or sarcopenia had a significantly increased risk of new-onset CVD (women: HR 1.45, 95% CI 1.12–1.88; ≥ 65 years: HR 1.54, 95% CI 1.04–2.27). However, this increased risk did not reach statistical significance in men or participants younger than 65 years. Among participants who recovered from sarcopenia to non-sarcopenia or possible sarcopenia, those under 65 years of age showed a reduced risk of new-onset CVD compared to those with stable sarcopenia (HR 0.42, 95% CI 0.21–0.83). For participants with possible sarcopenia at baseline, recovery to a non-sarcopenia status was associated with a significantly lower risk of new-onset CVD compared to those who remained in possible sarcopenia across all subgroups (women: HR 0.69, 95% CI 0.52–0.93; men: HR 0.59, 95% CI 0.41–0.87; < 65 years: HR 0.64, 95% CI 0.49–0.84; ≥ 65 years: HR 0.62, 95% CI 0.39–0.98). Subgroup analysis results related to CVD, heart disease, and stroke are also provided in Additional file 1: Tables S7–S9.
When sarcopenia status changes were reassessed using data from the third survey, consistent results were observed, indicating that progression to sarcopenia continued to be associated with an increased risk of new-onset CVD, while recovery from sarcopenia was associated with a reduced risk (Additional file 1: Tables S10–11). These findings remained consistent even after further adjustment for the use of antihypertensive, antidiabetic, and lipid-lowering medications (Additional file 1: Tables S12–13). Additionally, when analyzing the possible sarcopenia/sarcopenia group separately from the non-sarcopenia/possible sarcopenia group, the results were consistent: participants who progressed from non-sarcopenia to possible sarcopenia or sarcopenia had an increased risk of new-onset CVD, while those who recovered from sarcopenia to non-sarcopenia or possible sarcopenia had a reduced risk (Additional file 1: Table S14).
Discussion
In this study, we investigated the association between baseline sarcopenia status and changes in sarcopenia status with the risk of new-onset CVD using prospective cohort data. The findings indicate that participants with possible sarcopenia had a significantly higher risk of developing CVD compared to those without sarcopenia. While participants with sarcopenia also showed an increased risk, this association was not statistically significant after adjusting for confounding factors. Additionally, participants who progressed from non-sarcopenia to possible sarcopenia or sarcopenia exhibited a significantly increased risk of CVD. Conversely, participants with possible sarcopenia who recovered to a non-sarcopenia status and those with sarcopenia who recovered to a non-sarcopenia or possible sarcopenia status had a significantly reduced risk of new-onset CVD (Structured Graphical Abstract).
Sarcopenia is becoming increasingly prevalent among the global aging population. Previous research has demonstrated a close relationship between sarcopenia status and cardiovascular health [8, 10, 11, 33–35]. A meta-analysis of 38 studies revealed that the prevalence of sarcopenia in patients with CVD is approximately twice that of the general population, suggesting a positive correlation between CVD and sarcopenia [10]. Similarly, Ke et al. [13] found that both possible sarcopenia and sarcopenia were associated with CVD in the general population. A study using data from CHARLS from 2015 to 2018, with a follow-up period of 3.6 years, reported that participants with possible sarcopenia (HR 1.22, 95% CI 1.05–1.43) and sarcopenia (HR 1.33, 95% CI 1.04–1.71) had a higher risk of new-onset CVD compared to their peers without sarcopenia. Our study corroborates these findings, showing a significant increase in CVD risk among participants with possible sarcopenia and sarcopenia in a prospective cohort setting. The biological mechanisms underlying the association between sarcopenia and CVD may involve shared physiological pathways, such as systemic inflammation, oxidative stress, overactivation of the ubiquitin–proteasome system, and impaired glucose tolerance [9, 36–38]. However, our results remained significant even after adjusting for traditional cardiovascular risk factors, further supporting the notion that sarcopenia should be considered an independent risk factor for CVD.
In addition to the baseline sarcopenia status, our study also investigated the associations of changes in sarcopenia status with incident CVD, which were not examined previously. In a previous study comprised of 4395 individuals with a total of 10,778 records of sarcopenia state assessment, a total of 60.3% remained possible sarcopenia, 24.5% of individuals with a current state of possible sarcopenia returned to non-sarcopenia, 6.7% progressed to sarcopenia, and 8.5% died by the next follow-up [39]. Our study confirmed previous findings on the dynamic nature of sarcopenia status in the CHARLS cohort. Importantly, compared to participants with stable non-sarcopenia status, those who progressed to possible sarcopenia or sarcopenia exhibited a significantly increased risk of developing CVD. This finding underscores the adverse impact of sarcopenia progression on the incidence of CVD. Conversely, participants who recovered to a non-sarcopenia or possible sarcopenia status from sarcopenia, as well as those who recovered to a non-sarcopenia status from possible sarcopenia, demonstrated a significantly reduced risk of new-onset CVD. These results suggest that reversing sarcopenia has substantial benefits for the prevention of CVD. When sarcopenia status changes were reassessed using data from the third survey, consistent results were observed: progression to sarcopenia remained associated with an increased risk of new-onset CVD, while recovery from sarcopenia was linked to a decreased risk of CVD. These findings persisted even after further adjustments, highlighting the robustness of the associations. However, these results need to be further replicated and validated in other cohorts to confirm their generalizability. Changes in sarcopenia status may influence CVD risk through several mechanisms. First, sarcopenia is linked to chronic inflammation and oxidative stress, which contribute to endothelial dysfunction—a key factor in CVD [40]. Worsening sarcopenia may increase these inflammatory pathways and heighten CVD risk, while recovery could help reduce inflammation. Second, sarcopenia is associated with insulin resistance and metabolic dysfunction; muscle loss decreases insulin sensitivity, elevating cardiovascular risk, whereas improved muscle mass can help restore insulin sensitivity [41]. Additionally, changes in sarcopenia affect blood pressure regulation and are often associated with levels of physical activity and fitness, both of which have a direct impact on CVD outcomes [42, 43].
This study has significant clinical and public health implications. First, integrating sarcopenia assessment into routine cardiovascular practice is crucial, particularly in older people. Individuals with sarcopenia or possible sarcopenia should be prioritized as key target groups for the prevention of adverse cardiovascular events. Additionally, it is important to assess sarcopenia-related risk factors in individuals without sarcopenia to identify those at risk early. By implementing appropriate preventive measures, the progression of sarcopenia can be delayed, thereby reducing the incidence of CVD in these patients. Notably, sarcopenia is a reversible condition, and the study found that individuals who reversed their sarcopenia status had a significantly lower risk of CVD. Therefore, more efforts are needed to implement effective interventions aimed at reversing sarcopenia. Possible sarcopenia might present a better intervention window for CVD prevention than sarcopenia, as individuals with possible sarcopenia are more likely to recover to a non-sarcopenia status (as shown in Table 3). Further research, including real-world data and clinical trials, is needed to explore the best interventions for reversing sarcopenia and to evaluate their effectiveness and safety in cardiovascular practice.
This study has several strengths. To our knowledge, it is the first to investigate the association between changes in sarcopenia status and the risk of new-onset CVD. Additionally, the study included a large, nationally representative sample, enhancing the generalizability of the findings to the middle-aged and older adult population in China. The robustness of the results was also ensured through various sensitivity analyses.
This study has several limitations. First, similar to previous studies, the identification of CVD was based on self-reported physician diagnoses [24, 44, 45]. Due to the absence of medical records in the CHARLS dataset, this approach may introduce classification bias. However, Xie et al. [46] reported that 77.5% of self-reported new-onset coronary heart disease cases were confirmed by medical records in the English Longitudinal Study of Ageing, suggesting reasonable reliability. Second, changes in sarcopenia status were assessed based on two surveys. While theoretically, using more surveys could provide a more accurate assessment of sarcopenia status changes, our sensitivity analysis, which reassessed sarcopenia status using data from the third survey, yielded consistent results, indicating the reliability of our findings. Third, due to the lack of detailed classification of CVD, we were unable to explore the associations between sarcopenia and specific types of heart disease. Fourth, although we adjusted for multiple confounding factors, residual confounding may still exist, such as genetic predisposition and dietary habits. Future research should explore these important factors in greater detail. Fifth, using the Appendicular Skeletal Muscle Mass (ASM) formula may yield some differences compared to directly assessing muscle mass with DXA. Additionally, physical activity, a crucial factor in sarcopenia, was not included in our analysis due to considerable missing data in the database. Future analyses should consider more comprehensive methods for assessing muscle mass and incorporate physical activity as a key variable when feasible. Additionally, certain types of selection bias, such as potential volunteer bias, should be considered when interpreting and generalizing our results. Despite these limitations, this study contributes to our understanding of the significance of changes in sarcopenia status in cardiometabolic health.
Conclusions
Changes in sarcopenia status are associated with varying risks of new-onset CVD. Progression of sarcopenia status increases the risk of developing CVD, while recovery from sarcopenia reduces this risk. Future research should focus on developing precise preventive strategies to delay the progression of sarcopenia and implementing targeted interventions to reverse sarcopenia within cardiovascular practice.
Supplementary Information
Additional file 1: Table S1. The missing number and rate of covariates for baseline sarcopenia status analyses. Table S2. The missing number and rate of covariates for changes in sarcopenia status analyses. Table S3. Baseline characteristics of participants for baseline sarcopenia status analyses using data not being imputed. Table S4. Baseline characteristics of participants for changes in sarcopenia status analyses using data not being imputed. Table S5. Association of baseline sarcopenia status with risks of incident CVD. Table S6. Association of changes in sarcopenia status with risks of incident heart diseases and stroke. Table S7. Subgroup of association of changes in non-sarcopenia group with risks of incident cardiovascular disease. Table S8. Subgroup of association of changes in possible-sarcopenia group with risks of incident cardiovascular disease. Table S9. Subgroup of association of changes in sarcopenia group with risks of incident cardiovascular disease. Table S10. Baseline characteristics of participants for changes in sarcopenia status in sensitivity analyses using data from the third survey. Table S11. Association of changes in sarcopenia status with risks of incident cardiovascular disease in sensitivity analyses using data from the third survey. Table S12. Baseline characteristics of participants for changes in sarcopenia status in sensitivity analyses using of antihypertensive, antidiabetic, and lipid-lowering medications. Table S13. Association of changes in sarcopenia status with risks of incident cardiovascular disease in sensitivity analyses using of antihypertensive, antidiabetic, and lipid-lowering medications. Table S14. Association of changes in sarcopenia status with risks of incident cardiovascular disease in sensitivity analyses separating the possible sarcopenia/sarcopenia group from the non-sarcopenia/possible sarcopenia group.
Additional file 2: Supplemental Methods. Figure S1. Change trajectory of sarcopenia status. Figure S2. Stable change trajectory of sarcopenia status when using the third survey.
Acknowledgements
This study is based on the baseline of the China Health and Retirement Longitudinal Study (CHARLS). We would like to thank the CHARLS research team, the field team, and every respondent for their time and efforts that they have devoted to the CHARLS project.
Abbreviations
- CVD
Cardiovascular diseases
- CHARLS
China Health and Retirement Longitudinal Study
- ASM
Appendicular skeletal muscle mass
- AWGS
Asian Working Group for Sarcopenia
- DXA
Dual X-ray absorptiometry
- ASM/Ht2
Height-adjusted muscle mass
- BMI
Body mass index
- DBP
Diastolic blood pressure
- CRP
C-reactive protein
- HbA1c
Glycated hemoglobin
- TG
Triglycerides
- NonHDL-C
Non-high-density lipoprotein cholesterol
- DM
Diabetes
- HR
Hazard ratio
- CI
Confidence intervals
Authors' contributions
Conceptualization: QZ1 (Qingyue Zeng), LZ, ZA, and SL. Data curation: QZ1 (Qingyue Zeng) and QZ2 (Qian Zhong). Formal analysis: QZ1 (Qingyue Zeng) and QZ2 (Qian Zhong). Writing – original draft: QZ1 (Qingyue Zeng). Writing – review and editing: LZ, ZA, and SL. Supervision: ZA and SL. All authors actively participated in the research process, made substantial contributions to manuscript revisions, and carefully reviewed and approved the final version. All authors read and approved the final manuscript.
Funding
This work was supported by the This study was supported by Community Health Association of China (Grant number 2021–2-045) and the National Clinical Research Center for Geriatrics, West China Hospital, Sichuan University (Grant number Z2021JC005).
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
The ethical review board of Peking University meticulously examined and subsequently sanctioned the CHARLS project (IRB 00001052–11014). Informed consent was obtained from all subjects prior to their participation of this study.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Qingyue Zeng, Lijun Zhao and Qian Zhong are the first co-authors.
Contributor Information
Zhenmei An, Email: azmhxnfm@163.com.
Shuangqing Li, Email: 1259594471@qq.com.
References
- 1.Cho MR, Lee S, Song SK. A review of sarcopenia pathophysiology, diagnosis, treatment and future direction. J Korean Med Sci. 2022;37(18):e146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zeng QY, Qin Y, Shi Y, Mu XY, Huang SJ, Yang YH, et al. Systemic immune-inflammation index and all-cause and cause-specific mortality in sarcopenia: a study from National Health and Nutrition Examination Survey 1999–2018. Front Immunol. 2024;15:1376544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Beaudart C, Zaaria M, Pasleau F, Reginster JY, Bruyère O. Health outcomes of sarcopenia: a systematic review and meta-analysis. PLoS ONE. 2017;12(1):e0169548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cruz-Jentoft AJ, Landi F, Schneider SM, Zúñiga C, Arai H, Boirie Y, et al. Prevalence of and interventions for sarcopenia in ageing adults: a systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing. 2014;43(6):748–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sayer AA, Cruz-Jentoft A. Sarcopenia definition, diagnosis and treatment: consensus is growing. Age Ageing. 2022;51(10):afac220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 2019;48(1):16–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sepúlveda-Loyola W, Osadnik C, Phu S, Morita AA, Duque G, Probst VS. Diagnosis, prevalence, and clinical impact of sarcopenia in COPD: a systematic review and meta-analysis. J Cachexia Sarcopenia Muscle. 2020;11(5):1164–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Damluji AA, Alfaraidhy M, AlHajri N, Rohant NN, Kumar M, Al Malouf C, et al. Sarcopenia and cardiovascular diseases. Circulation. 2023;147(20):1534–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim D, Lee J, Park R, Oh CM, Moon S. Association of low muscle mass and obesity with increased all-cause and cardiovascular disease mortality in US adults. J Cachexia Sarcopenia Muscle. 2024;15(1):240–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zuo X, Li X, Tang K, Zhao R, Wu M, Wang Y, et al. Sarcopenia and cardiovascular diseases: a systematic review and meta-analysis. J Cachexia Sarcopenia Muscle. 2023;14(3):1183–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mirzai S, Carbone S, Batsis JA, Kritchevsky SB, Kitzman DW, Shapiro MD. Sarcopenic obesity and cardiovascular disease: an overlooked but high-risk syndrome. Curr Obes Rep. 2024;13(3):532–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Boonpor J, Pell JP, Ho FK, Celis-Morales C, Gray SR. In people with type 2 diabetes, sarcopenia is associated with the incidence of cardiovascular disease: a prospective cohort study from the UK Biobank. Diabetes Obes Metab. 2024;26(2):524–31. [DOI] [PubMed] [Google Scholar]
- 13.Gao K, Cao LF, Ma WZ, Gao YJ, Luo MS, Zhu J, et al. Association between sarcopenia and cardiovascular disease among middle-aged and older adults: findings from the China health and retirement longitudinal study. EClinicalMedicine. 2022;44: 101264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Beaudart C, Dawson A, Shaw SC, Harvey NC, Kanis JA, Binkley N, et al. Nutrition and physical activity in the prevention and treatment of sarcopenia: systematic review. Osteoporos Int. 2017;28(6):1817–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lu L, Mao L, Feng Y, Ainsworth BE, Liu Y, Chen N. Effects of different exercise training modes on muscle strength and physical performance in older people with sarcopenia: a systematic review and meta-analysis. BMC Geriatr. 2021;21(1):708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chen LK, Woo J, Assantachai P, Auyeung TW, Chou MY, Iijima K, et al. Asian Working Group for Sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J Am Med Dir Assoc. 2020;21(3):300-7.e2. [DOI] [PubMed] [Google Scholar]
- 17.Zhao Y, Hu Y, Smith JP, Strauss J, Yang G. Cohort profile: the China Health and Retirement Longitudinal Study (CHARLS). Int J Epidemiol. 2014;43(1):61–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wen X, Wang M, Jiang CM, Zhang YM. Anthropometric equation for estimation of appendicular skeletal muscle mass in Chinese adults. Asia Pac J Clin Nutr. 2011;20(4):551–6. [PubMed] [Google Scholar]
- 19.Yang M, Hu X, Wang H, Zhang L, Hao Q, Dong B. Sarcopenia predicts readmission and mortality in elderly patients in acute care wards: a prospective study. J Cachexia Sarcopenia Muscle. 2017;8(2):251–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu Y, Cui J, Cao L, Stubbendorff A, Zhang S. Association of depression with incident sarcopenia and modified effect from healthy lifestyle: the first longitudinal evidence from the CHARLS. J Affect Disord. 2024;344:373–9. [DOI] [PubMed] [Google Scholar]
- 21.Xiong L, Liao T, Guo T, Zeng Z, Wang S, Yang G, et al. The relationship between sarcopenia and mortality in Chinese community-dwelling adults: a 7-year cohort study with propensity score matching and Mendelian randomization. Front Endocrinol (Lausanne). 2023;14:1215512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Alexandre Tda S, Duarte YA, Santos JL, Wong R, Lebrão ML. Sarcopenia according to the European Working Group on Sarcopenia in Older People (EWGSOP) versus dynapenia as a risk factor for mortality in the elderly. J Nutr Health Aging. 2014;18(8):751–6. [DOI] [PubMed] [Google Scholar]
- 23.Wu X, Li X, Xu M, Zhang Z, He L, Li Y. Sarcopenia prevalence and associated factors among older Chinese population: findings from the China Health and Retirement Longitudinal Study. PLoS ONE. 2021;16(3):e0247617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.He D, Wang Z, Li J, Yu K, He Y, He X, et al. Changes in frailty and incident cardiovascular disease in three prospective cohorts. Eur Heart J. 2024;45(12):1058–68. [DOI] [PubMed] [Google Scholar]
- 25.Ji JJ, Zhao MJ, Xiao ML, Zhang HE, Tan Q, Cheng YR, et al. Association between relative muscle strength and cardiovascular disease among middle-aged and older adults in China. BMC Public Health. 2024;24(1):1928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bu F, Steptoe A, Fancourt D. Relationship between loneliness, social isolation and modifiable risk factors for cardiovascular disease: a latent class analysis. J Epidemiol Community Health. 2021;75(8):749–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Madley-Dowd P, Hughes R, Tilling K, Heron J. The proportion of missing data should not be used to guide decisions on multiple imputation. J Clin Epidemiol. 2019;110:63–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sato K, Noguchi H, Inoue K, Kawachi I, Kondo N. Retirement and cardiovascular disease: a longitudinal study in 35 countries. Int J Epidemiol. 2023;52(4):1047–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Skoblow HF, Proulx CM. C-reactive protein, subjective aging, and incident cardiovascular disease: a mediation model. J Gerontol B Psychol Sci Soc Sci. 2022;77(9):1654–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.van Es N, Takada T, Kraaijpoel N, Klok FA, Stals MAM, Büller HR, et al. Diagnostic management of acute pulmonary embolism: a prediction model based on a patient data meta-analysis. Eur Heart J. 2023;44(32):3073–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.White IR, Royston P, Wood AM. Multiple imputation using chained equations: issues and guidance for practice. Stat Med. 2011;30(4):377–99. [DOI] [PubMed] [Google Scholar]
- 32.Wang X, Ma H, Li X, Heianza Y, Fonseca V, Qi L. Joint association of loneliness and traditional risk factor control and incident cardiovascular disease in diabetes patients. Eur Heart J. 2023;44(28):2583–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fang M, Liu C, Liu Y, Tang G, Li C, Guo L. Association between sarcopenia with incident cardio-cerebrovascular disease: a systematic review and meta-analysis. Biosci Trends. 2023;17(4):293–301. [DOI] [PubMed] [Google Scholar]
- 34.Cho HW, Chung W, Moon S, Ryu OH, Kim MK, Kang JG. Effect of sarcopenia and body shape on cardiovascular disease according to obesity phenotypes. Diabetes Metab J. 2021;45(2):209–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Han P, Chen X, Yu X, Zhang Y, Song P, Cai M, et al. The predictive value of sarcopenia and its individual criteria for cardiovascular and all-cause mortality in suburb-dwelling older Chinese. J Nutr Health Aging. 2020;24(7):765–71. [DOI] [PubMed] [Google Scholar]
- 36.Sasaki KI, Fukumoto Y. Sarcopenia as a comorbidity of cardiovascular disease. J Cardiol. 2022;79(5):596–604. [DOI] [PubMed] [Google Scholar]
- 37.Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol. 2018;15(9):505–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Barbalho SM, Flato UAP, Tofano RJ, Goulart RA, Guiguer EL, Detregiachi CRP, et al. Physical exercise and myokines: relationships with sarcopenia and cardiovascular complications. Int J Mol Sci. 2020;21(10):3607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Luo YX, Zhou XH, Heng T, Yang LL, Zhu YH, Hu P, et al. Bidirectional transitions of sarcopenia states in older adults: The longitudinal evidence from CHARLS. J Cachexia Sarcopenia Muscle. 2024;15(5):1915-29. [DOI] [PMC free article] [PubMed]
- 40.Inoue DS, Janini GM. Integrative insights into PNI: low-grade chronic inflammation, skeletal muscle wasting, and brain impairments. Brain Behav Immun Health. 2024;40:100838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Guo C, He L, Tu Y, Xu C, Liao C, Lai H, et al. Insulin resistance and sarcopenia: a prognostic longitudinal link to stroke risk in middle-aged and elderly Chinese population. BMC Public Health. 2024;24(1):2757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mogi M. Pulse pressure tells the story of sarcopenia. Hypertens Res. 2024;47(9):2583–5. [DOI] [PubMed] [Google Scholar]
- 43.Mayakrishnan V, Kannappan P, Balakarthikeyan J, Kim CY. Rodent model intervention for prevention and optimal management of sarcopenia: a systematic review on the beneficial effects of nutrients & non-nutrients and exercise to improve skeletal muscle health. Ageing Res Rev. 2024;102:102543. [DOI] [PubMed] [Google Scholar]
- 44.Jiang M, Ren X, Han L, Zheng X. Associations between sarcopenic obesity and risk of cardiovascular disease: a population-based cohort study among middle-aged and older adults using the CHARLS. Clin Nutr. 2024;43(3):796–802. [DOI] [PubMed] [Google Scholar]
- 45.Li F, Wang Y, Shi B, Sun S, Wang S, Pang S, et al. Association between the cumulative average triglyceride glucose-body mass index and cardiovascular disease incidence among the middle-aged and older population: a prospective nationwide cohort study in China. Cardiovasc Diabetol. 2024;23(1):16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Xie W, Zheng F, Yan L, Zhong B. Cognitive decline before and after incident coronary events. J Am Coll Cardiol. 2019;73(24):3041–50. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. The missing number and rate of covariates for baseline sarcopenia status analyses. Table S2. The missing number and rate of covariates for changes in sarcopenia status analyses. Table S3. Baseline characteristics of participants for baseline sarcopenia status analyses using data not being imputed. Table S4. Baseline characteristics of participants for changes in sarcopenia status analyses using data not being imputed. Table S5. Association of baseline sarcopenia status with risks of incident CVD. Table S6. Association of changes in sarcopenia status with risks of incident heart diseases and stroke. Table S7. Subgroup of association of changes in non-sarcopenia group with risks of incident cardiovascular disease. Table S8. Subgroup of association of changes in possible-sarcopenia group with risks of incident cardiovascular disease. Table S9. Subgroup of association of changes in sarcopenia group with risks of incident cardiovascular disease. Table S10. Baseline characteristics of participants for changes in sarcopenia status in sensitivity analyses using data from the third survey. Table S11. Association of changes in sarcopenia status with risks of incident cardiovascular disease in sensitivity analyses using data from the third survey. Table S12. Baseline characteristics of participants for changes in sarcopenia status in sensitivity analyses using of antihypertensive, antidiabetic, and lipid-lowering medications. Table S13. Association of changes in sarcopenia status with risks of incident cardiovascular disease in sensitivity analyses using of antihypertensive, antidiabetic, and lipid-lowering medications. Table S14. Association of changes in sarcopenia status with risks of incident cardiovascular disease in sensitivity analyses separating the possible sarcopenia/sarcopenia group from the non-sarcopenia/possible sarcopenia group.
Additional file 2: Supplemental Methods. Figure S1. Change trajectory of sarcopenia status. Figure S2. Stable change trajectory of sarcopenia status when using the third survey.
Data Availability Statement
No datasets were generated or analysed during the current study.