Abstract
Anterior eye disorders including dry eye syndrome, keratitis, chemical burns, and trauma have varying prevalence rates in the world. Classical dosage forms based-topical ophthalmic drugs are popular treatments for managing corneal diseases. However, current dosage forms of ocular drugs can be associated with major challenges such as the short retention time in the presence of ocular barriers. Developing alternative therapeutic methods is required to overcome drug bioavailability from ocular barriers. Nanocarriers are major platforms and promising candidates for the administration of ophthalmic drugs in an adjustable manner. This paper briefly introduces the advantages, disadvantages, and characteristics of delivery systems for the treatment of corneal diseases. Additionally, advanced technologies such as 3D printing are being considered to fabricate ocular drug carriers and determine drug dosages for personalized treatment. This comprehensive review is gathered through multiple databases such as Google Scholar, PubMed, and Web of Science. It explores information around "ocular drug delivery systems'', "nano-based drug delivery systems'', "engineered nanocarriers'', and "advanced technologies to fabricate personalized drug delivery systems''.
Keywords: Corneal disorders, Drug delivery systems, Personalized medicine, 3D printing
Graphical abstract
Highlights
-
•
The delivery systems for the treatment of corneal diseases are introduced.
-
•
Nanocarriers are promising platforms for customizable ophthalmic medication and treatment.
-
•
3D and 4D printing can be used to design delivery platforms based on corneal damage.
-
•
Personalized platforms offer an attractive route to study and improve therapeutic precision and effectiveness.
1. Introduction
The normal cornea is a connective tissue with dome-shaped, transparent avascular, and compact multilayer structure in the eye's front portion [1]. This tissue consists of five layers including epithelium, Bowman's layer, stroma, Descemet's membrane, and endothelium [[1], [2], [3]]. The cornea serves important functions such as protecting the eye, contributing to the eye's refractive power, focusing light rays on the retina with minimum scatter, and preventing optical degradation [4]. Infectious keratitis, corneal perforation, melting, corneal scarring, vascularization, haze, and corneal dystrophies can be significant causes of visual impairments resulting in reduced corneal transparency, vision loss, and blindness. However, there is also a variation in the prevalence and epidemiology of corneal diseases according to region [[5], [6], [7]]. For example, the environmental risk factors such as UV light exportation during corneal collagen cross-linking (CXL), contact lens wear, and refractive surgeries (e.g., LASIK (laser-assisted in situ keratomileusis, small incision lenticule extraction (SMILE)) or Photoreactive keratectomy (PRK) can contribute to the development of corneal disorders [[8], [9], [10], [11]]. For example, spontaneous persistent epithelial defects (PEDs) can appear following surgical approaches such as PRK. After surgery approaches, transforming growth factor beta isoforms (TGFβ-1, TGFβ-2, or TGFβ-3) can localize in the corneal stroma at PEDs and promote myofibroblasts development from precursor cells [[12], [13], [14]]. The above findings favor previous pre-clinical studies conducted on rabbit models with PRK [15,16]. According to the experimental observations, spontaneous PED signs with the presence of myofibroblasts were indicated 7 days post-surgery in the superficial stroma at PED. Some myofibroblast marker alpha-smooth muscle actin (α-SMA) with TGFβ-3 localized to the stroma at PEDs. After PRK, all three TGFβ isoforms could be detected in the stroma at PEDs. This pathological mechanism likely prompts corneal scarring, opacity, and visual impairment.
Current therapeutic methods include non-invasive (e.g. topical eye drops, ocular emulsion, suspension, ointment, and polymeric gels). However, these approaches may be associated with limitations due to intricate anatomical and physiological barriers [[17], [18], [19], [20]]. There are physical methods for improving ocular bioavailability, such as formulations in suspensions (Zigran(®)), solutions (Zymaxid(™)), and gels (Akten®), as well as chemical methods such as prodrugs (Xalatan™), and soft drugs [21]. Soft drugs are referred as chemical compounds like anti-inflammatory corticosteroids that can be administrated locally at the desired eye sites [22].
Nano-based systems have been investigated for disease treatment [23]. Various delivery systems have been applied precisely to encapsulate, transport, and deliver chemical drugs to reduce complications. Based on the type of ocular diseases, delivery systems can be administrated as eye drops, injections, etc [[24], [25], [26]]. Several kinds of delivery systems for ophthalmic diseases are available, including nanoparticles, liposomes, dendrimers, niosomes, hydrogels, contact lenses, punctal plugs, and microneedles [17,27]. These carrier systems have the potential to penetrate the corneal membrane and deliver cargos to the defective parts of the cornea [28]. Therefore, to minimize side effects, it is important to improve drug efficiency and reduce the amount of precorneal drugs [29].
Moreover, drug delivery systems based on nanocarriers or ocular devices are considered for managing ocular diseases. Notably, developing advanced and customized ocular delivery systems would also be beneficial. This review examines the applications of nanocarriers and ocular devices for treating anterior segment diseases of the eye, particularly the cornea. Previous studies-based delivery systems for managing anterior ocular disorders are also evaluated. Finally, advanced technologies, such as 3D printing, may be used to administer drug dosages more precisely, as well as fabricate delivery systems based on an individual's damaged corneal properties [[30], [31], [32]].
2. Managing corneal disorders with topical drugs
The eye's surface can be affected by corneal diseases or environmental factors, potentially impairing vision or leading to blindness in some cases [33,34]. Current topical ocular drug-based non-invasive methods include eye drops, such as non-steroidal anti-inflammatory drugs (NSAIDs), steroidal (SAID) drugs, and autologous serum. These drugs are effective in healing ocular surface disorders such as burnt corneas, infectious inflammations, and neovascularization [35,36].
In (Table 1) some examples of current topical drugs for treating other corneal disorders and diseases are mentioned.
Table 1.
Some of the current therapeutic methods for treating corneal disorders and diseases.
| Corneal diseases | Corneal impairments | Current non-invasive medications | Ref. |
|---|---|---|---|
| Corneal trauma | Ocular trauma is caused by an injury due to corneal abrasions and foreign bodies commonly. The severity and symptoms of ocular trauma disorders can include pain, corneal perforation, corneal laceration, recurrent erosion, corneal opacity, and even pose a threat to vision. Bacterial keratitis, corneal ulcer, and PED may be the other complications of corneal injuries. |
- To alleviate inflammation and corneal wound healing, topical NSAIDs such as diclofenac eye drops may be effective. | [37,38] |
| Microbial Keratitis | It is a leading cause of localized pain, corneal scarring, neovascularization and can ultimately result in blindness due to various infectious factors. For example, NK is a rare degenerative disease that can be caused by HSV or herpes zoster infection. It is diagnosed by a reduction or absence of corneal sensitivity. The symptoms are dysfunction of corneal healing process, stromal ulcers, corneal epithelium disruption, impaired healing, corneal opacity and, the risk of melting and perforation. These symptoms can result in an irreversible deficit of vision. |
- Antiviral therapy containing systemic drugs as well as systemic and topical drugs such as trifluridine and ganciclovir gel are demonstrated. | [39] |
| Ocular chemical burns | Chemical burns vary based on the range of penetration of the chemical substances in the front surface of the eye and the severity of their symptoms. Causing LSCD may occur as a result of an alkali burn. This disorder may occur opacification and neovascularization of the cornea. |
- Biological fluids such as autologous serum, umbilical cord blood serum, PRP, and amniotic membrane suspension with a rich source of growth factors and angiogenic anti‐fibrotic effects to promote wound healing may be effective in diminishing opacity and corneal neovascularization. - Corticosteroids such as triamcinolone can prescription too |
[[40], [41], [42], [43], [44], [45], [46], [47], [48]] |
| DED or KCS | DES or KCS is a chronic and a multifactorial disease caused by insufficient production of tears in the eye. Common ocular symptoms of DES include eye redness, ocular discomfort, blurred vision and instability of the tear membrane which causes to the damaged ocular surface. Inflammation and oxidative stress play crucial role in the pathogenesis of this disease. Prolonged wear of contact lenses and exposure to weather conditions with low quality and humidity are risk factors for this disease. Additionally, DED can be caused by medical problems such as lupus, scleroderma, and Sjogren's syndrome. |
- CsA is known as an FDA-approved drug with immunosuppressive and anti-inflammatory properties. - Topical corticosteroids to reduce inflammation in a short time are also used. - Autologous serum tears have similar properties of biochemical and mechanical with normal aqueous tears which can improve symptoms and signs of severe or refractory in patients. |
[[49], [50], [51], [52], [53], [54], [55]] |
Abbreviations: PED: persistent corneal epithelial defects, NSAIDs: non-steroidal anti-inflammatory drugs, NK: Neurotrophic keratitis, HSV: herpes simplex virus, LSCD: limbal stem cell deficiency, PRP: Platelet-rich plasma, DED: dry eye disease, KCS: keratoconjunctivitis sicca, CsA: Cyclosporin A.
Some topical drugs are listed in Table 1. However, topical drugs may present challenges, including corneal epithelial injuries, ulceration, and perforation [37]. Moreover, it is important to note that biological mechanisms, such as eyelid movement, tear turnover, and drainage can significantly affect the rapid elimination of topical drugs from the eye surface [17,35,[56], [57], [58]]. Furthermore, topical drug administration may be associated with low bioavailability, poor solubility, limited shelf life (insufficient in vitro stability), short half-life (insufficient in vivo stability), lack of large-scale production, and necessity for targeted delivery due to ocular barriers [59,60]. In commercial formulations, mucoadhesive properties and contact duration with corneal and conjunctival tissues are diminished. The steroid responses of these agents can increase the risk of cataracts and intraocular pressure [42,43,[61], [62], [63]]. Lack of selectivity, short retention time, and low corneal permeability are the other challenges of topical drugs [20,64,65]. Additional side effects may include the presence of cytotoxic or antigenic/immunogenic agents in patients [65]. Thus, current topical approaches may not be appropriate for the long-term treatment of moderate to severe cases [42,51,62].
3. Ocular barriers
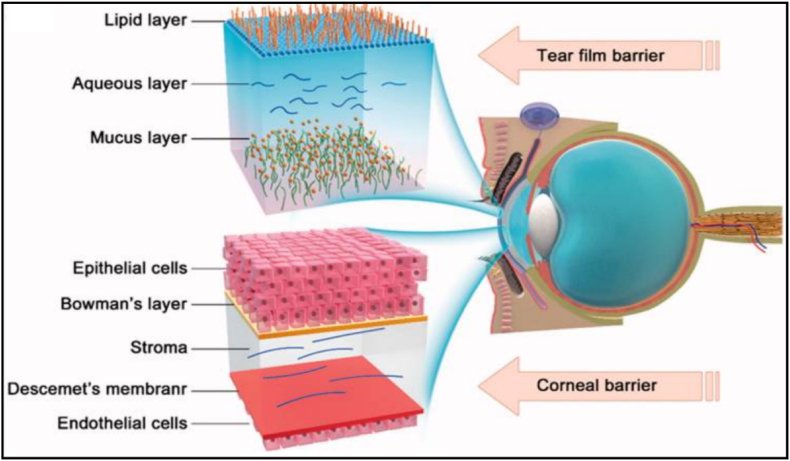
The eye stands as a well-preserved structure that effectively hinders the penetration of drugs to their intended targets by biological barriers including precorneal, corneal, and blood-aqueous barrier [57]. The cornea, a highly specialized multilayer tissue with a thickness of approximately 0.5 mm, is characterized by its transparency and avascular nature. Three main corneal layers including epithelium-stroma-endothelium are formed equivalent to a fat-water-fat structure. Matured corneal epithelium, serving as a tight diffusion barrier for drug delivery, is equipped with paracellular pores measuring 2.0 nm in diameter. The lipophilic structure of the epithelium layer is a barrier for 90 % of hydrophilic drugs. Conversely, this layer acts as an obstacle for 10 % of hydrophobic drugs [57,58,[66], [67], [68]]. Similarly, the corneal endothelium layer, comprising a monolayer of the cells, can serve as a hindrance to the entry of hydrophilic drug molecules. However, the transfer of hydrophilic drug molecules into these layers is restricted by the presence of the epithelial and endothelial layers. On the other hand, diffusion of hydrophobic drug molecules is inhibited by the corneal stroma regarding their hydrophilic structure. The blood-aqueous barrier is formed by uveal capillary endothelium and ciliary epithelium, effectively restricting the passage of medications from the systemic circulation to the anterior chamber [69]. The structure of important ocular surface bio-barriers including tear film and cornea is shown in (Fig. 1) [70].
Fig. 1.
Key bio-barriers of the anterior segment of the eye (Reprinted with some modifications from Ref. [70]).
4. Ocular devices
Ophthalmic devices for ocular disorders have been employed. For instance, punctal plugs are miniature non-invasive medical implants, ranging in size from 2 to 5 mm that prevent the drainage of tears placed in the tear ducts for patients with dry eye syndrome [71]. Punctual plugs as a safe and effective treatment to deliver the drug in a sustained manner are also suggested [72,73].
Using mild electric charges, iontophoresis is a non-invasive method that effectively enhances delivery of drugs through biological barriers. They can be used for drug permeation for anterior and posterior ocular disorders. For example, trans-corneal iontophoresis can be utilized to treat anterior segment disorders, including corneal ulcers, dry eye disease, ocular inflammation, keratitis, and ocular uveitis. However, there may be potential complications associated with this technique, including discomfort and a burning sensation [71,74].
Contact lenses are discs with curved shapes that cover the cornea. Based on material components they have various types, including hard lenses, soft lenses, and silicon lenses. In general, increasing the bioavailability of these lenses is suitable for controlling drug release. Prolonged use of contact lenses can lead to corneal toxicity [[75], [76], [77], [78], [79], [80]]. Prosthetic Replacement of the Ocular Surface Ecosystem (PROSE) lens is an effective drug delivery device. This lens is efficient in improving vision in cases of corneal irregularities and chronic ocular surface diseases associated with corneal neovascularization, Stevens-Johnson syndrome/toxic epidermal necrolysis [[81], [82], [83]].
Collagen corneal shields are similar to ocular lenses and are composed of collagen sourced from bovine or porcine origins [84]. Drugs can be placed into collagen shields, which dissolve and form a lubricating layer to promote epithelial repair and progressive drug release [85]. Drugs can reversibly attach to these reservoirs for regulated release, lowering the risk of systemic toxicity [84]. However, because of their opacity, they may result in impaired vision [86,87]. A straightforward application approach for treating corneal problems such as neovascularization, injuries, dry eye, and infections is provided by nanowafers, which are tiny circular discs or membranes [[88], [89], [90]]. Nanowafers are transparent and can be applied to the eye with the fingertip, withstand continuous blinking. Gradual drug release from the nanowafers prolongs drug retention on the ocular surface and enhances drug absorption [91].
To reduce the side effects of topical cysteamine (Cys) eye drops, such as redness, ocular pain, and inflammation due to frequent administration, a clinically translatable Cys nanowafer (Cys-NW) was fabricated [92]. In vivo experiments in cystinosin knockout mice revealed that Cys-NW with 10 μg Cys, administered once daily, had more therapeutic efficacy than 44 μg Cys delivered as topical eye drops twice daily. Moreover, Cys-NW maintained Cys stability for up to 4 months at room temperature, whereas topical Cys eye drops require freezing or refrigeration and remain effective for only one week. Due to its safety profile, prolonged drug stability, and superior therapeutic efficacy at room temperature, Cys-NW for human application in clinical trials can be suggested.
Through minimally invasive patches, microneedles can be useful for localized drug delivery. Microneedles can deliver both hydrophilic and hydrophobic drugs in precision injections by overcoming the ocular barriers [[93], [94], [95], [96], [97]].
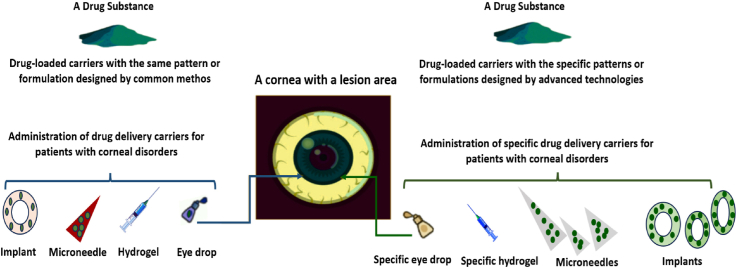
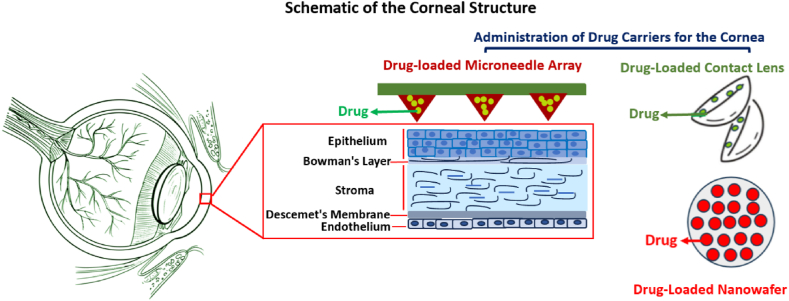
Fig. 2, presents a schematic of drug delivery carriers such as contact lenses, nanowafers, and microneedles and their application in corneal disorder treatment.
Fig. 2.
Administration of drug delivery carriers to be used as the corneal segments and provide suitable therapeutic approaches for corneal disorders. (www.vecteezy.com).
Some of the ophthalmic drug delivery systems for managing ocular disorders are depicted in (Table 2).
Table 2.
A summary of evaluated ophthalmic systems for managing ocular surface disorders.
| Ocular systems | Drug | Role of drug | Administration | Purpose of Study | Pre-clinical/clinical studies | Advantages | Potential issues/limitations | Ref. |
|---|---|---|---|---|---|---|---|---|
| Drug-depository contact lens (DDCL) | Moxifloxacin | An antimicrobial drug | Insertion | -Increasing the time of drug interaction with corneal damaged site - Evaluating the therapeutic efficiency of DDCLs combined with an antimicrobial drug to reduce bacterial keratitis in patients. |
Patients with bacterial keratitis In two groups (18 cases with topical antimicrobial drug & 17 cases with DDLC-antimicrobial drug) |
- Greater reduction in pain score in patients treated with DDCL-antimicrobial drug compared to group with topical drug treatment during the follow-up period (p < 0.05) - Faster disease healing with DDLC-drug due to more drug availability in the lesion site of the cornea compared to topical drug. - Possible reduction in drug consumption by patients treated with DDLC. |
More investigations needed for ulcer patients. - Evaluation of drug availability in damaged corneal sites for other keratitis (e,g. protozoal and mycotic). |
[98] |
| Collagen punctal plug | Insertion | Administration of a temporal collagen punctal plug to improve dry eye disease and reduce intraocular pressure | Human patients with dry eye disease and primary open-angle glaucoma (Case group: 33 patients that received glaucoma drugs in the presence of a temporary collagen plug) and (control group with 33 patients that used glaucoma drugs received without punctal plug) |
- Improvement in dry eye disease in the case group in comparison with the control group (p < 0.001). - Significant reduction of intraocular pressure in the case group compared to the control group. |
Short time follow-up - The counterpart eye of each patient should have been considered as control group. |
[99] | ||
| Collagen shields | Voriconazole | A drug to treatment of fungal keratitis | Insertion | An optimized drug delivery system production | Rabbit eyes | - Higher antifungal efficacy and trans-corneal permeation with the optimized formula than drug solution. | N/A | [100] |
| Double-layered microneedles | DC101/diclofenac | An anti-angiogenic monoclonal antibody/anti-inflammatory compound | Self-implantable | Evaluating the efficiency and safety of an eye patch fabricated of micro-drug-reservoirs to control drug release into the ocular surface tissue. | Mouse eyes | - Efficient non-invasive home-based treatment for various eye diseases. - Improved corneal neovascularization healing in comparison with topical eye drops. -Providing therapeutic efficacy by fast and sustained release of medications. |
N/A | [101] |
| Polyvinyl alcohol (PVA)- Polyvinyl pyrrolidone (PVP)- Microneedles | Besifloxacin | An antimicrobial drug | Insertion | Evaluation of the polymeric microneedles to deliver besifloxacin | In vitro/Ex vivo infection models | - Higher permeation and deposition of besifloxacin-loaded microneedles (for 5 min) in the cornea compared to free besifloxacin solution (p < 0.05). - Superior antibacterial activity in infected corneas with Staphylococcus aureus than free besifloxacin. |
The reduction of besifloxacin dosage through microneedles should be investigated. | [102] |
5. The significance of nanotechnology-based drug delivery systems for ocular applications
Traditional ocular dosage forms, such as eye drops, may not be well-suited for treating ocular disorders [55]. Moreover, administrating drugs through the conventional dosage might lead to inactivation or have adverse effects on non-targeted tissues [60,103]. In general, drug delivery systems have shown remarkable efficacy in the treatment of eye diseases [[104], [105], [106]]. Notably, nanotechnology-based drug delivery systems offer alternative strategies to improve drug permeation, optimize and control drug delivery to specific ocular tissue sites, and prolong drug release [107,108].
Substances such as drugs can be loaded into nanocarriers via physical encapsulation, adsorption, or covalent attachment [60,109,110]. Nanocarriers have shown significant potential in the treatment of glaucoma, dry eye, and fungal keratitis [111]. In general, nano-based delivery systems improve therapeutic properties, increase drug solubility, and absorption. For instance, the utilization of drug-loaded carriers to enhance solubility and protect against rapid degradation has been proven. Furthermore, these systems diminish required dosages by enhancing the concentration of drugs in target tissues [[112], [113], [114]]. Essential attributes of effective drug delivery systems consist of favorable biocompatibility, considerable biodegradability, minimal cytotoxicity, and non-immunogenicity. Besides, features like controlled drug release, lysosome escape, swift endocytosis, and high drug-carrying capacity are the other advantageous aspects of nano-based drug delivery systems [[115], [116], [117]]. Regarding the intended design of nanocarriers, it is important to consider their physicochemical attributes, such as particle size, particle shape, surface properties, and chemical composition [60]. A nanoparticle within the range of 5–200 nm can easily evade renal clearance and the reticuloendothelial system during blood circulation. Thus, this size range contributes to their longer retention time in the bloodstream and higher blood concentration [118]. Factors like biological activity, biodistribution, speed of internalization into target cells, and clearance may be influenced by nanomaterial shapes (e.g., spheres, rods, tubes, fibers, and disks). In this case, comparing rod nanoparticles and those that are like fiber has indicated that endocytosis of the spherical shape of nanoparticles is easier and quicker [119]. Furthermore, the surface characteristics of nanomaterials, encompassing surface area, pores, and charge can impact processes like phagocytosis, blood circulation, and biodistribution [120]. In the case of these systems, electrostatic interactions between the positively charged surface and the negatively charged surface of the corneal epithelium prolong drug retention [57].
5.1. Solid-lipid-nanoparticles (SLNs)
SLNs are colloidal delivery systems consisting of a solid lipid matrix with a perfect crystal lattice structure, ranging in size from 10 to 500 nm. These nanosystems can encapsulate both hydrophilic and hydrophobic drugs, enabling them to effectively traverse biological barriers. Notable properties of SLNs include controlled drug release, targeted drug delivery, long-term stability, and biocompatibility. Moreover, their large-scale production is feasible. Due to their lipophilic nature, SLNs can cross the epithelial barrier, blood-aqueous barrier, and blood-retinal barriers of the eye [[121], [122], [123]].
Patel et al. [63] developed SLNs containing besifloxacin hydrochloride (BSF@SLN) and optimized to improve antibiotic absorption via passive diffusion in the eye. Compared to Besix eye drops (a commercialized drug), the improved ophthalmic nanoformulation proved to be 2.76 times more effective against bacteria. BSF@SLN ophthalmic nanoformulations were also compared to Besix in an in vivo eye irritation study and demonstrated substantial pharmacological effects.
5.2. Nanostructured lipid carriers (NLCs)
NLCs are the second generation of the SLN with non-immunogenic, compatible, and biocompatible properties, ranging in size from 50 to 1000 nm. NLCs are formed from a mix of liquid lipids with solid lipids, preventing crystallization of lipids. Their drug-loading capacity is higher and more efficient in comparison with the SLNs. The existence of liquid lipids in NLCs may be the underlying reason for this feature [58,[124], [125], [126]].
Kumari et al. [127] evaluated an eye drop formulation of dexamethasone-loaded NLC for ophthalmic applications during in vitro and ex vivo studies. Based on observations from superior tolerability and internalization capability in human corneal epithelial cells and ex vivo porcine cornea experiments, this ophthalmic formulation may be applicable to the eye surface distribution. Besides, the biomarkers of inflammation in dexamethasone-loaded NLC were assessed using ELISA. As showed a five-fold decrease in TNF-α production compared to the free dexamethasone solution. To sum up, NLC formulation may be a viable alternative for the topical management of dry eye disease due to its simplicity, scalability, and efficacy.
5.3. Niosomes
Niosomes are composed of non-ionic surfactant vesicles. They are suitable for delivering both hydrophilic and lipophilic drugs. The significant properties of these vesicles are biodegradability, biocompatibility, and non-immunogenicity. In addition, they can enhance corneal permeability while reducing drug clearance in the aqueous humor. However, it is important to note that they have some limitations, such as aggregation, fusion, limited shelf life, and potential eye irritation [[128], [129], [130]].
In a prior study by Zeng et al. [128] Tacrolimus (FK506) was administered to patients unresponsive to steroids and cyclosporine to prevent the rejection of corneal allograft. The hydrophobicity, molecular weight, and physiological and anatomical limitations hampered FK506's ocular distribution. By combining niosomes with mucoadhesive hyaluronic acid (HA), FK506HA-coated niosomes were developed. FK506HA-coated niosomes were compared to HA solution and non-coated niosomes using surface plasmon resonance to investigate the mucoadhesion. In rabbits, HA-coated niosomes significantly extended the residence time of FK506 in the precorneal area compared to suspensions or non-coated niosomes. Pharmacokinetic studies of aqueous humor revealed a greater area under the curve for HA-coated niosomes than for suspensions and non-coated niosomes. Using a confocal laser scanning microscope, the hybrid delivery system for FK506 demonstrated synergistic improvement in corneal permeability. By improving mucoadhesion, aqueous humor pharmacokinetics, precorneal retention, and transcorneal permeability, the hybrid approach was shown to enhance FK506 ocular delivery. Collectively, HA-coated niosomes may offer a potential method for FK506 administration in ocular applications.
5.4. Liposomes
Liposomes are in the form of either nanovesicular or microvesicular structures of different sizes. Their structure comprises one or more phospholipid bilayers that enclose an aqueous core. Due to their chemical structures, liposomes are capable of encapsulating both hydrophobic and hydrophilic drugs. They have excellent biocompatibility and a cell membrane-like structure. However, concerning the mononuclear phagocytic system or reticuloendothelial system as clearance mechanisms, they have some limitations, such as limited capacity for drug loading, sterilization issues, and short half-life [121].
Liposomes have considerable potential for administering ophthalmic medications since they are small, biocompatible, and capable of delivering drugs with multiple polarities simultaneously. The physicochemical characteristics of a liposome formulation encapsulating Bevacizumab and Dexamethasone were investigated for ocular delivery topically [131]. In the preparation of liposomes, thin-film hydration and extrusion with an optimized formulation were used. The formulation was assessed in wound healing, and sustained release of Bevacizumab from the liposomes was observed. A topical eye drops drug delivery system capable of co-loading Dexamethasone and Bevacizumab was developed and evaluated for delivering ocular medications.
5.5. Cubosomes
Cubosomes comprise amphiphilic structures of the carbon chain and are typically less than 500 nm in size. They have a lipid bilayer with a high surface area, which can enhance their efficiency by improving the residence time and bioavailability of hydrophilic and hydrophobic ocular medications with different solubilities [[132], [133], [134], [135], [136]].
In a previous investigation [137], cubosomes containing the antibiotic moxifloxacin hydrochloride were produced using glycerol monooleate and poloxamer 407 to treat conjunctivitis. To prepare moxifloxacin cubogels, optimized cubosomes were dispersed in a cold in-situ gelling system. Compared to a traditional formulation, moxifloxacin cubogel absorbed 81 % through the goat cornea in an in vitro corneal permeation study. Antibacterial and histological studies demonstrated the safety of the formulations selected for ocular application. Based on its enhanced permeability and prolonged release properties, moxifloxacin hydrochloride cubogel can be suggested to serve as a viable alternative to traditional eye drops.
5.6. Electrospun nanofibers
Electrospun nanofibers, as an alternative delivery system, offer the advantage of adapting to the eye surface while facilitating sustained release [138,139]. Sangole et al. [140] produced a polymeric ocular insert of levofloxacin using electrospinning technology. Electrospun nanofiber sheets were fabricated using hydroxypropyl cellulose and polycaprolactone polymer. In vitro examination of drug release demonstrated an 8-h release of the medication, with 99 % efficiency. Nanocomposite sheets were formulated to reduce dosing frequency and improve patient compliance through the prolonged release of levofloxacin in ocular diseases.
5.7. In-situ gelling systems
These systems involve smart hydrogels that respond to environmental stimuli such as temperature, pH, ions, and ultrasound waves. This responsiveness triggers a sol-gel phase transition, resulting in the formation of a viscoelastic gel. Notably, these systems offer advantages including sustained drug release, prolonged drug contact with the cornea, and enhanced bioavailability compared to conventional eye drops. However, it's worth mentioning that these systems are associated with limitations such as potential blurred vision and the possibility of eyelid adhesion [36,80,141].
Chitosan-carbomer nanoparticles were loaded with timolol and incorporated into a carbomer-based pH-responsive in-situ gel system using the polyelectrolyte complexation method previously [142]. Nanoparticle-laden in-situ gels were analyzed under physiological conditions. Briefly, in-situ gels containing nanoparticles exhibited more mucoadhesive characteristics. In-situ gels containing nanoparticles significantly reduced intraocular pressure compared with timolol solution and nanoparticles alone. Furthermore, the in-situ gelling system significantly reduced intraocular pressure over a longer duration than nanoparticles and timolol solution. Accordingly, the formulated in-situ nanoparticle gel was an effective carrier for ocular drugs. In-situ nanoparticle gelling systems may enhance therapeutic effectiveness by slowly releasing nanoparticles, while their mucoadhesive properties and acquisition of high viscosities at precorneal sites make them remarkable for delivering drugs to the eyes.
5.8. Nanomicelles
Nanomicelles are colloidal delivery systems with a hydrophobic core and a hydrophilic shell, ranging in size from 10 to 100 nm. Easy preparation, low toxicity, monodispersity, and high bioavailability are advantages of this system. Nanomicelles are water-soluble and have a high drug-coating capacity. They are capable of increasing the solubility of ocular drugs with limited water solubility in their hydrophobic core. This feature can lead to more efficient drug delivery in ocular tissues. Additionally, they efficiently enhance tissue permeation and minimize drug degradation [17,[143], [144], [145], [146], [147]].
Commercial suspensions (5 % Natamycin) exhibit quick precorneal clearance, limited corneal permeability, increased administration frequency, and corneal irritation due to undissolved particles. However, in a valuable study [148], natamycin-loaded nanomicelles (1 % Natcel) were formulated with transparency and stability properties. Compared with the 5 % suspension, 1 % Natcel showed superior corneal permeability. The formulation of 1 % Natcel provided a sustained release for up to 24 h. It was also biocompatible and had a longer mean residence time (MRT) in tears than the 5 % suspension. Consequently, 1 % Natcel may be a viable alternative treatment for fungal keratitis.
5.9. Nanoemulsion
Nanoemulsion is a colloidal system consisting of oil(s), water, surfactant(s), and co-surfactant(s) within the size range of 20–500 nm. The advantages of small-sized droplets include a higher active surface area, which results in more efficient ophthalmic drug delivery, improved ocular permeability, and higher bioavailability. Nanoemulsions possess the added advantage of reduced surface tension and superior drug dispersion over the cornea, facilitating effective integration with precorneal components. These attributes contribute to the prolonged drug contact duration on the corneal epithelium [[149], [150], [151], [152]].
Yeu et al. [153] evaluated the clinical effectiveness and safety of the propylene glycol/hydroxypropyl-guar (PG-HPG)-based nanoemulsion (Systane® Complete) for lubricating eye drops in patients with dry eye disease. In an IV phase study, adult dry eye cases were classified into three subgroups: aqueous deficient, evaporative, and mixed. A drop of PG-HPG was applied to each eye twice a day for 28 days. Briefly, the data demonstrated the efficacy of PG-HPG nanoemulsion eye drops for the treatment of dry eye disease.
5.10. Dendrimers
Dendrimers are artificial nanoconstructs with highly branched, star-shaped polymeric structures ranging from 5 to 20 nm in size. Considering the physicochemical properties of these carriers, such as molecular weight, size, surface charge, molecular geometry, and functional groups, they are considered a significant category in the concept of drug delivery. Dendrimers are suitable for delivering both hydrophobic and hydrophilic drugs due to their highly branched structure. Additionally, they enhance drug solubility and demonstrate high drug-loading capacity, along with sustained drug release characteristics. However, limitations such as blurred vision and potential loss of eyesight are associated with them [36,80,[154], [155], [156], [157], [158]].
In a prior study by Lin et al. [159], free dexamethasone (Free-Dex), dendrimer-dexamethasone (D-Dex), or saline were administered through a single subconjunctival injection to animals with induced autoimmune dacryoadenitis. A significant reduction in the number of pro-inflammatory genes (IL6, IL8, MMP9, TNFα) expression was observed in the D-Dex group compared to the Free-Dex and saline groups. This nanomedicine study for managing ophthalmic diseases (such as induced autoimmune dacryoadenitis) could be efficient for clinical applications.
5.11. Cyclodextrins
Cyclodextrins represent a group of cyclic oligosaccharides capable of forming inclusion complexes with various drugs. These complexes can adhere to the eye's surface, enhancing drug permeation and enabling sustained release. In addition, they can enhance drug solubility and mitigate drug irritation by controlling drug concentration. However, potential toxicity concerns at higher concentrations may limit their applicability [80,[160], [161], [162], [163], [164]].
Sohani et al. [165] developed an ophthalmic drop hydrogel utilizing curcumin nanoparticles encapsulated with β-cyclodextrin and hyaluronic acid to enhance corneal healing. Curcumin, which exhibits significant pharmacological qualities, including anti-cancer, anti-inflammatory, and antioxidant effects, is utilized in numerous medicinal formulations. The nanoparticles were assessed by in vitro investigations such as zeta potential, Fourier-transform infrared (FTIR), and scanning electron microscopy (SEM) analyses. Additionally, rabbit models of ulcerative keratitis were individually treated with topical drugs and assessed via various methods (such as fluorescein dye staining, corneal clarity score, and pathological assessments). In comparison with traditional treatment, the frequency of medication administration was reduced. Furthermore, improved tissue quality and a reduction in ulcerative keratitis (p < 0.05) were demonstrated. This study indicates the potential of novel ophthalmic topical medications for treating ulcerative keratitis.
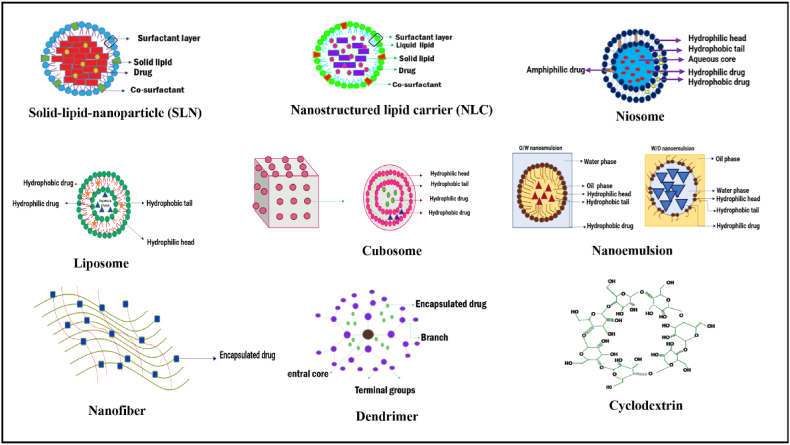
The chemical structure of nano-delivery systems is represented in (Fig. 3).
Fig. 3.
The chemical structures of drug carriers.
6. Engineered nanosystems for improved drug delivery
Various delivery systems have been used in numerous investigations to treat corneal disorders, frequently in modified or combined designs for drug loading in both laboratory and clinical situations [166]. These delivery systems can be manufactured using traditional technologies such as freeze drying, spray coating, micro milling, micromachining, photolithography, etching, deposition, mold replication, and assembly [167]. Alternatives include composite nanosystems that can simultaneously load and release medications or bioactive compounds over an extended time [168].
6.1. Engineered nanovesicles
Nanovesicles can be included in hydrogels (Exosome-loaded hydrogels, liposome-loaded hydrogels) for the benefit of protecting them against quick clearance, which improves their membrane integrity and mechanical toughness. Additionally, the physical, mechanical, and biological properties of hydrogels can be improved and customized by using these nanovesicle-infused hydrogels [168,169]. Piffoux et al. [170] fabricated personalized biosynthetic hybrid vectors by loading extracellular vesicles (EVs) into liposomes, thereby developing an advanced delivery system. Through this approach, fused exosomes were obtained using functionalized liposomes containing biological constituents such as membrane and soluble cargoes, alongside polyethylene glycol. These hybrid structures, enriched with biogenic molecules within their lipid bilayer and inner compartments, exhibited more therapeutic efficiency compared to either free drug or the drug-loaded liposome precursors. Another study conducted by Chang et al. [166]. administered the development of liposome-loaded hydrogels as a dual drug delivery system. Their formulation encapsulated antibiotics moxifloxacin (MFX) and dexamethasone (DEX) using nanostructured lipid carriers, which were integrated with a biodegradable material composed of collagen, gelatin, and alginate (CGA).
The utilization of the CGA-Lipo-MFX/DEX complex in an animal model yielded positive outcomes without observed side effects or toxicities. Moreover, this complex demonstrated the inhibition of pathogenic microorganism growth and enhancement of corneal wound healing. The evaluated results suggest that this anti-inflammatory formulation holds promise as a potential treatment for the ophthalmological disease.
Additionally, the integration of hybrid exosome-liposome particles into smart hydrogel systems, responsive to different stimuli such as chemical, biological, electrical, photo, thermo, or pH changes, presents a viable avenue for achieving more sustained and controlled drug delivery [168].
6.2. Metal-based nanosystems
Ceria-based nanosystems have been also developed to alleviate dry eye symptoms. Yang et al. [171] synthesized an engineered multifunctional nanoceria to deliver gabapentin in the presence of gelatin and glutaraldehyde. The structure exhibited antioxidative, antiangiogenic, anti-inflammatory, antiapoptotic, and biocompatible properties. Gelatin's abundant thiol groups enhanced cellular uptake by 2.3-fold and efficient mucin binding (10-fold), increasing ocular retention and therapeutic activity. A rabbit model of dry eye showed that the nanoformulation improved tear production. It also repaired the corneal epithelium defect, maintained corneal nerve density, and was more effective than free drugs. In summary, a potent and safe nanoformulation was proposed to reduce dry eye symptoms.
Nguyen et al. [172] investigated amination levels in chitosan coatings to enhance the penetration of ceria nanocapsules through ocular barriers to effective drug release. In addition to enhancing the carriers' burst release capabilities, high amination levels increased permeability through corneal cell layers by approximately 43-fold. Pilocarpine-loaded chitosan-coated ceria nanocapsules were used to treat an experimental rabbit model of acute glaucoma. Within 4 h, the treatment successfully reduced high intraocular pressures to normal levels, prevented retinal degeneration, opened the angle closure, and inhibited endothelium damage. This solution for the treatment of acute glaucoma and other urgent medical conditions may be available in the form of nano eye drops.
A nanomedicine approach was conducted to modify surface roughness-controlled ceria nanocages (SRCNs) and poly(l-histidine) surface coatings to activate various bioactive functions of intrinsically therapeutic nanocarriers, facilitate transport across the barriers of corneal epithelium, and release of dual drugs [acetylcholine chloride (ACh) and SB431542] at the injury site [173]. Briefly, surface modifications enhanced corneal penetration and enabled the smart release of drugs in damaged tissue. In vivo observations confirmed the efficiency of a single-dose topical nanoformulation, as shown by a reduction in corneal wound sites and approximately 93 % inhibition of abnormal blood vessel formation in a rat model of alkali burn (19-fold better than a commercial eye drop). Furthermore, corneal transparency was almost fully restored four days after administration. This nanoformulation was suggested to provide multifunctional metallic nanotherapeutics for tissue regeneration and ocular pharmacology.
In the management of ocular hypertension, an enormously common ocular condition, Y-27632 is an effective ophthalmic agent [174]. However, continuous administration of Y-27632 via a therapeutic carrier to damaged sites in the eye's inner segments remains a challenge. In a prior study, shell thickness influenced Y-27632 release patterns. This research showed that hollow mesoporous ceria nanoparticles (HMCNs) with thick (∼40 nm) and thin (∼10 nm) shells exhibited burst release properties (within 2 days) or had less drug-loading capacity. The longest drug release occurred in HMCNs with a shell thickness of approximately 20 nm, lasting 10 days. In a rabbit model of glaucoma treated with Y-27632-loaded HMCNs, about 86 % of compromised photoreceptor cells were restored. It was suggested that HMCN shell thickness is significant in the development of long-acting nano eye drops for glaucoma management. Nanobiomaterial structural engineering may play a significant role in pharmacologically managing intraocular diseases by developing long-lasting eye drops.
6.3. Metal-organic frameworks (MOFs)
Metal-organic frameworks (MOFs) are novel drug delivery systems comprising crystalline inorganic-organic hybrids. They offer potential encapsulating and delivering imaging agents and small-molecule drugs. However, challenges include potential linker toxicity, synthesis conditions, and pore size limitations. Huang et al. addressed these by developing nanoassembly carriers through coordinative interactions between imidazole of His6 and zinc ions, eliminating traditional ligands [115]. His6-metal assemblies (HmA) exhibited benefits like high loading capacity, pH-dependent release, low cytotoxicity, rapid endocytosis, and intracellular release. In treating anterior segment diseases, Wang et al. [65] utilized HmA in eye drops. For instance, they encapsulated dexamethasone sodium phosphate (Dexp) within nanosized particles (Dexp@HmA) to treat alkali burn inflammation. In vitro and in vivo results showed Dexp@HmA features like high encapsulation efficiency, high drug payload, pH-responsive drug release, and low cytotoxicity. Furthermore, its capability to control sustained drug release is associated with increased therapeutic efficiency in both in vitro and in vivo models. Notably, HmA emerges as a promising approach for addressing diverse anterior segment eye diseases.
6.4. Polymersomes
Polymersomes are nanocarriers to the load and release of hydrophilic and hydrophobic drugs. these nanostructures contain hydrophilic lumens which are encased with membranes of hydrophobic bilayer. Their low permeability, however, may make payload release difficult. To overcome this obstacle, redesigned polymersomes with improved applicability, such as stimuli-responsive versions, must be designed [175,176]. As an illustration, photoresponsive polymersomes with excellent efficiency have been used to accelerate corneal wound healing [176].
Different drug delivery systems for the treatment of corneal diseases are evaluated in (Table 3).
Table 3.
Chemical drug delivery systems for healing corneal diseases.
| Drug carriers | Drug | Role of Drug | Administration | Outcomes | In vitro/in vivo | Ref. |
|---|---|---|---|---|---|---|
| mPEG-b-PLGA NPs | TAC | A hydrophobic drug with low corneal penetrability that inhibits corneal allograft rejection after transplantation by reducing the IL-2, IL-17, and VEGF expression levels in the cornea and aqueous humor. | Eye drops and subconjunctival injection | - This study aimed to enhance transcorneal permeation and absorption of drugs. - This system had the potential to treat ocular diseases, by increasing the efficacy of hydrophobic drugs. |
Rabbits and rats | [177] |
| SLNs | NAT | An agent against Fungal keratitis | - NAT-SLNs in a prolonged drug release rate improved corneal penetration and enhanced antifungal activity without cytotoxic effects on corneal tissues. - The designed system as a promising ocular delivery system for treating deep corneal keratitis could be considered. |
Ex vivo | [178] | |
| NLCs | Curcumin | It is a potent agent with antioxidant, anti-inflammatory, and anti-infectious effects. Curcumin has poor bioavailability due to its low aqueous solubility and stability which limits its clinical efficacy. | These results demonstrated curcumin permeation enhancement in excised corneas. | In vitro and ex vivo | [179] | |
| Niosomes in ketorolac tromethamine gels | NAT | It is useful for healing fungal keratitis | Topical | To improve the corneal permeability and enhance the prolonged delivery of NAT is considered. | Rabbit model (keratitis) | [180] |
| Cubosomal NPs | Fluconazole | It is a bistriazole antifungal agent to treat ocular mycoses. However, it has poor permeation into ocular tissues and has a short half-life. | Topical | An enhanced antifungal activity of this drug for the treatment of fungal keratitis was observed. | Rat eyes (fungal keratitis) | [181] |
| Hyalugel-integrated liposomes | Fluconazole | It is a bistriazole antifungal agent for the treatment of ocular mycoses as an eye drop. However, it has poor permeation into ocular tissues and has a short half-life. | Topical | - An enhanced corneal permeability and increased antifungal activity were reported. - It would be a good candidate for ocular applications. |
Ex vivo/in vivo (Rabbit eyes) | [182] |
| Electrospun nanofibers | CsA | An immunosuppressive drug to control the rejection of organ transplants and to treat inflammatory and various autoimmune conditions. | Suture | This carrier system was effective in suppressing corneal inflammation and corneal neovascularization. | Rabbit model (corneal alkali injury) | [138] |
| PLGA-PEG-PLGA thermosensitive hydrogel | MET- LFH | Inhibition factors for corneal neovascularization. | Subconjunctival injection | Co-polymers which showed great potential for ophthalmic anti-angiogenic therapy. | Mouse model (corneal alkali burn) | [183] |
| Nanomicelles based on PVCL-PVA-PEG | Curcumin | It is a potent agent with antioxidant, anti-inflammatory, and anti-infectious effects. Curcumin has poor bioavailability due to its low aqueous solubility and stability which limits its clinical efficacy. |
Topical and/or intranasal | Nanomicelle curcumin was used for the treatment of corneal epithelial/nerve wound healing in diabetic keratopathy. | Mice (diabetic model) | [184] |
| Dendrimer | DEX | A synthetic corticosteroid, with anti-inflammatory effect to treat eye injuries. | Injectable gel | - This platform demonstrated the ability to improve the retention time of the drugs for the treatment of many inflammatory ocular surface disorders such as dry eye, auto-immune keratitis, and post-surgical complications. | Rat model (mild alkali burn) | [185] |
| HPβCD | TAC | To reduce inflammatory activity in different ocular diseases such as dry eye, uveitis, and corneal graft rejection is suggested. | Topical | A topical delivery system with a high potential for ophthalmic diseases was considered. | Rat eyes | [186] |
Abbriviations: mPEG-b-PLGA NPs: methoxy poly (ethylene glycol-block-poly (D, L)-lactic-co-glycolic acid) nanoparticles, TAC: tacrolimus, IL2: interlucin 2, IL17:interlucin 17, VEGF: vascular endothelial growth factor, 2-HP-β-cyclodextrin- Polylactic-co-glycolic acid nanoparticles, TA: Triamcinolone Acetonide, SLNs: solid lipid nanoparticles, NAT: natamycin, NLCs: nanostructured lipid carriers, CsA: Cyclosporine A, PLGA-PEG-PLGA: poly(D,L-lactic-co-glycolic acid)-block-poly(ethylene glycol)-block-poly(D,L-lactic-co-glycolic acid), MET: Metformin, LFH: Levofloxacin Hydrochloride, PVCL-PVA-PEG: polyvinyl caprolactam-polyvinyl acetate-polyethylene glycol, DEX: Dexamethasone, HPβCD: Hydroxypropyl-β-cyclodextrin.
7. Drug-free systems
Various modification techniques have been applied to improve the pharmacokinetic properties of nanocarriers, aiming to enhance their efficiency in encapsulation, conjugation, transportation, and drug delivery [187]. However, limitations like inadequate effectiveness due to cellular barriers, multidrug resistance mechanisms, burst release, and potential organ toxicity, such as kidney damage, may be accompanied by drug-loaded carriers [[188], [189], [190]].
The intricate process of loading drugs often involves complex synthesis methods and the use of specific materials, increasing costs and diminishing the stability of drug delivery systems. Additionally, challenges such as batch-to-batch inconsistencies can hinder their clinical application [[191], [192], [193], [194]]. Despite these issues, many drug delivery systems still suffer from limited drug loading capacities, leading to unsatisfactory therapeutic outcomes [195]. Thus, the development of drug-free systems has emerged as a viable option for disease management, including inflammation and cancer therapy [187,196]. Luo et al. [197] employed a bioadhesive metallic nanotherapeutic system, utilizing gelatin-capped silver nanoparticles (G-Ag NPs), which exhibited dual antimicrobial and antiangiogenic properties for treating Staphylococcus aureus-induced keratitis. The in vitro and in vivo studies demonstrated a potent autoantigenicity efficiency of G-Ag NPs. Also, the system exhibited excellent corneal stromal residence, stability, antibacterial activity, biocompatibility, and significant bioactivity.
Several drug-free hydrogels are marketed for use as artificial tears or sealants. Various artificial tears have been used to treat dry eyes, such as Viscotears®, Liquivisc®, Clinitas Gel®, GelTears®, and Xailin Gel® [198,199]. Lin et al. [200] developed a long-lasting eye drop formulation for topical dry eye disease therapy using a drug-free carbonized nanogel (CNG) by pyrolysis of lysine hydrochloride (Lys). Lys-CNGs enhanced ocular bioavailability and precorneal retention. Additionally, in vitro and in vivo biocompatibility evaluations of Lys-CNGs demonstrated their safety as ocular drops. A single dose of Lys-CNGs (50 μg mL⁻1) in a rabbit dry eye model significantly reduced symptoms within 4 days. However, compared to cyclosporine A, multiple doses with 10 times higher content were required to achieve the same therapeutic effects (one dose every 12 h; 500 μg mL⁻1). Compared to commercial cyclosporine A eye drops, Lys-CNGs exhibited enhanced therapeutic efficacy due to reduced therapeutic dosages and prolonged dosing intervals. Lys-CNGs possess strong anti-inflammatory, free radical-scavenging, biocompatibility, antioxidant, and ocular bioadhesive properties, suggesting they could be used to develop long-lasting eye drops for treating dry eye disease.
In another study by Jiao et al. [201] an innovative antibacterial and antioxidant contact lens was fabricated using a polyacrylamide semi-interpenetrating network hydrogel comprising quaternized chitosan and tannic acid (PAM-QCS-TA). The synthesized hydrogels exhibited swelling resistance, high water content, light transmittance, and tunable mechanical properties. In antibacterial assays against Staphylococcus aureus and Escherichia coli (E. coli), great sterilization was demonstrated (particularly against E. coli). The presence of tannic acid in the structure conferred high antioxidant properties, protecting cells from reactive oxygen species cytotoxicity. Animal studies showed a reduced treatment duration for bacterial keratitis (within 3 days) and eye tissue preservation. Consequently, a contact lens with drug-free antibacterial and antioxidant properties and emergence of drug resistance prevention was developed. This efficient contact lens as a promising option for managing inflammatory diseases and ocular infections was suggested.
8. Designing drug delivery systems by emerging technologies
Administering dosage specifically at pathological sites can significantly enhance the efficiency of drug delivery systems [167,202]. Therefore, the utilization of advanced platforms such as microfluidic devices and 3D printing to optimize drug delivery processes with minimal side effects, achieving reproducible, on-demand, and tunable dosing at the required timing for patients while avoiding repetition, holds great promise [203]. 3D printing is one of the revolutionary formulation-preparation processes needed to enable the development of customized medicine. To create 3D objects, printing materials are deposited layer by layer using a computer-aided design method, which may be efficient in developing personalized medications [204].
The development of recent microfluidic lab-on-a-chip technology has provided unprecedented opportunities for precise drug delivery through the integration, implantation, localization, automation, and meticulous control of various microdevice parameters. An illustrative study showed [205], the sub-tenon microfluidic system (SMS) in regulating the pharmacokinetics of plasma and ocular dexamethasone (DEX) for delivering dexamethasone sodium phosphate (DEXP) in rabbits with experimental autoimmune uveitis (EAU). Employing a miniature pump connected to a tube implanted within the sub-tenon's sac of EAU rabbits facilitated the sub-tenon microfluidic system (SMS) to exhibit sustained DEX release and a notable drug loading capacity. Comparative to the conventional approach of combined medication (subconjunctival injection combined with intravenous injection) in EAU rabbits, the SMS showcased an enhanced distribution of DEX throughout ocular tissues. Notably, this method limited drug entry into the bloodstream, thereby minimizing systemic adverse effects. With its potential to potentially replace or eliminate the need for systemic medications in managing acute ocular inflammatory conditions, the SMS presents itself as a viable choice for ocular clinical trials.
Moreover, the utilization of 3D printing for dosage form manufacturing has garnered substantial interest in the realm of pharmaceutical technology. The field of pharmaceutical technology has seen significant interest in the utilization of 3D printing for manufacturing dosage forms [206]. The mechanism of this technology is the fabrication of 3D objects from digital models by deposing layer-by-layer printed materials.
Sophisticated 3D printing methodologies hold substantial promise in the fabrication of intricate dosage forms with diverse profiles of drug release, thereby facilitating the tailoring of pharmaceutical formulations [207]. The expedited production of personalized pharmaceutical formulations, encompassing patient-specific variables such as age, weight, organ functionality, and disease severity, is a notable feature of 3D printing technology. The execution of this process is facilitated through the application of computer-aided design (CAD) files [202,208]. Stereolithography, selective laser sintering, inkjet-based 3DP, fused deposition modeling, and pressure-assisted microsyringe are among the 3D printing methods utilized in the pharmaceutical domain [202,207]. These techniques have been explored for enhancing drug delivery efficiency and achieving sustained release profiles.
In a prior study [209], 3D printing technology has been employed in the pharmaceutical realm. In this investigation, innovative ophthalmic patches for controlled drug release are developed using a semi-solid material extrusion-type 3D printer. Printer inks based on hydrogel consisting of hypromellose, sugar alcohols (mannitol, xylitol), and drugs, were utilized to construct patch-shaped structures through 3D printing, followed by a freeze-drying process. 3D-printed structures were characterized, based on physical properties, surface structure, water uptake, antimicrobial activity, and drug release profile of lyophilized patches. Afterward, based on patient-tailored dosages, different dosages, and patterns were obtained containing multiple drugs for clinical applications.
Digital light processing (DLP) 3D printing was used to fabricate dexamethasone-loaded punctal plugs previously [210]. A semi-interpenetrating network (semi-IPN) was established using polyethylene glycol diacrylate (PEGDA) and polyethylene glycol 400 (PEG 400). An evaluation of drug-loaded punctal plugs was conducted for cytocompatibility, drug release profile, potential drug-photopolymer interactions (FTIR), and physical properties (XRD and DSC). According to the results, 20 % w/w PEG 400 and 80 % w/w PEGDA punctal plugs released dexamethasone continuously for up to seven days, whereas 100 % PEGDA punctal plugs released it continuously for over 21 days. Collectively, DLP 3D printing was offered as a viable manufacturing platform to fabricate customized punctal plugs with sustained medication release for ocular administration.
In another study, 3D-printed inserts containing liposomal moxifloxacin (SL: MOX) to improve drug penetration and ocular contact time were fabricated [211]. Through dynamic light scattering, moxifloxacin liposomes were measured at 150 nm. This study showed that 80 % of moxifloxacin could be encapsulated within lecithin liposomes (SL: MOX). An ex vivo investigation was conducted to assess the antibacterial susceptibility of MOX, SL: MOX, and a commercial product. The ocular inserts were then designed and optimized using sodium hyaluronate (NaH) through an extrusion 3D printing method. Liposomes improved the solubility of moxifloxacin, and the 3D-printed insert released the medication noticeably slower than the control. A 3D printer produced ocular inserts (moxifloxacin and lecithin liposomes) with stability and superior content uniformity by incorporating liposomal moxifloxacin. Based on 3D printing, customized eye medications can be developed specifically for each patient with eye diseases.
Besides, a dissolving ocular microneedle patch using an optimized stereolithography (SLA) 3D printing method was produced [212]. The microneedle architecture and printing precision were analyzed in terms of layer thickness, aspect ratio, mold geometry, length, and printing orientation. For a variety of fundamental shapes, including pyramidal, conical, and triangular pyramidal, the effects of the printing angle on needle fidelity were also evaluated. In vitro mechanical and penetration tests were conducted on polymeric polyvinyl pyrrolidone and polyvinyl alcohol (PVP/PVA) microneedle patches, fabricated from reverse molds in a variety of heights and shapes, and aspect ratios. An ex vivo penetration test was further performed on excised ocular tissues (cornea and sclera). Through optimization, the parameters required to produce microneedles with the highest dimensional fidelity and sharpest tips were determined. According to ex vivo studies, these optimized systems to penetrate ocular tissue with minimal pressure and easy self-administration for patients have potential.
In general, designing structures using biomaterials and tissue engineering has been studied extensively. Moreover, the fabrication of mimicked microenvironments has garnered significant interest in tissue regeneration [[213], [214], [215]]. In drug delivery system development, a polymer biomaterial's interaction with a drug enhances therapeutic efficacy, improves drug stability, and reduces local immune responses [92].
3D (bio)printing, by fabricating mimicked structures similar to native tissues, is also highly applicable for regenerative medicine applications. For instance, in a prior study by Nie et al. [216] 3D printing was used to fabricate mimicked scaffolds of natural polymer biodegradable hydrogel to promote corneal tissue regeneration. A polymer hydrogel ink was made from concentrated aqueous solutions of gelatin and carbohydrazide-modified alginate (Gel/Alg-CDH). Extrusion printing with hydrogel Gel/Alg-CDH exhibited appropriate viscosity thickening, shear thinning, and improved mechanical properties. Moreover, superior transparency, swelling stability in physiological conditions, suturability, and rapid degradation by enzymes were demonstrated in Gel-Alg-CDH-Ca2+-carbodiimide hydrochloride (EDC) hydrogels. With multi-nozzle printing, the scaffold for a biomimetic bilayer hydrogel can be personalized to carry growth factors and drugs. Through rabbit corneal keratoplasty, this scaffold induced corneal epithelium regeneration and inhibited corneal scarring. Fig. 4 (I) shows the application of extrusion printing for development of bilayer scaffolds using Gel/Alg-CDH inks. A hydrogel bilayer scaffold loaded with recombinant human epidermal growth factor layer (rhEGF)/Trichostatin A (TSA) in rabbit corneal models is also presented in this figure.
Fig. 4.
(I) Schematic representation of the Gel/Alg-CDH inks used to 3D-printed hydrogel bilayer scaffolds loaded with rhEGF/TSA for rabbits with corneal ALK models. (II) The evaluation of 3D-printed hydrogel scaffolds containing rhEGF/TSA in rabbit ALK model. (A) The ALK procedure involves the removal of the corneal epithelium and partial of the corneal stroma, followed by hydrogel scaffold implantation and suturing. (B–E) The slit lamp images (left) and the AS-OCT images (right) show the normal cornea, the defected cornea (control group), the implantation of hydrogel group, and the implantation of rhEGF/TSA bilayer hydrogel group, respectively.
Fig. 4 (II) shows the efficiency of 3D-printed bilayer scaffolds containing rhEGF/TSA to repair corneal defects in the rabbit model with anterior lamellar keratoplasty (ALK) [216].
ALK: anterior lamellar keratoplasty; rhEGF: recombinant human epidermal growth factor layer; TSA: Trichostatin A (Reprinted with some modifications from Ref. [216]).
By combining artificial intelligence and advanced technologies (such as 3D printing), the recent transformation of the medical industry is expected to facilitate the design and development of customized medications. Hu et al. [217]. presented the design and manufacturing of personalized capsules incorporating AI and 3D printing technology. After discretizing and encoding the capsule geometry, a traditional drug dissolution model was used to simulate the capsule's disintegration.
A genetic algorithm was then employed to investigate the geometric structure space of the capsules and produce a sophisticated multi-layer configuration that meets the specified drug release characteristics. The AI-designed capsule was precisely printed using fused deposition modeling technology, employing two model medications, isoniazid and acetaminophen. By altering the capsule's form, it was possible to design an accurate and independent release curve for the drug. An in vitro comparison between the printed capsules' release curve and the target curve confirmed the method's reliability. The method is expected to provide individualized dosage forms for various medications.
9. Conclusions
The topical prescription of conventional systemic medications is often challenged by rapid excretion and degradation processes, leading to inadequate drug accumulation at desired anatomical sites. Innovative drug delivery systems, encompassing nanoparticles, liposomes, niosomes, and dendrimers, exhibit remarkable potential in addressing these limitations by facilitating controlled and sustained drug release, thereby minimizing untoward effects and optimizing therapeutic outcomes.
Moreover, the pursuit of tailored drug delivery systems with specific release kinetics for localized medication administration, achieved through strategies such as integration, implantation, and localization, holds substantial appeal. The emergence of such personalized platforms offers an intriguing avenue for exploration, allowing for enhanced precision and effectiveness in therapeutic interventions. For instance, 3D printing technologies enable customization of drug delivery systems in terms of size, shape, and release profile, catering to specific patient populations such as pediatrics, pregnant women, and geriatrics based on their genetic and genomic attributes. These versatile structures, including tablets, scaffolds, implants, microneedles, capsules, films, hydrogels, mouthguards, tubes, stents, and rings, hold promise for applications in personalized medicine.
CRediT authorship contribution statement
Mahsa Fallah Tafti: Writing – original draft, Investigation, Data curation, Conceptualization. Zeinab Fayyaz: Writing – review & editing, Investigation. Hossein Aghamollaei: Writing – review & editing, Validation, Project administration, Conceptualization. Khosrow Jadidi: Writing – review & editing, Validation, Project administration, Conceptualization. Shahab Faghihi: Writing – review & editing, Supervision, Project administration.
Data availability statement
Data will be made available on request.
Ethical statement
This work does not need an ethical statement.
Funding
No funding was received for this study.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Fuest M., et al. Prospects and challenges of translational corneal bioprinting. Bioengineering. 2020;7(3):71. doi: 10.3390/bioengineering7030071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fallah Tafti M., et al. An inspired microenvironment of cell replicas to induce stem cells into keratocyte-like dendritic cells for corneal regeneration. Sci. Rep. 2023;13(1) doi: 10.1038/s41598-023-42359-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tafti M.F., et al. Emerging tissue engineering strategies for the corneal regeneration. J Tissue Eng Regen Med. 2022;16(8):683–706. doi: 10.1002/term.3309. [DOI] [PubMed] [Google Scholar]
- 4.Sridhar M.S. Anatomy of cornea and ocular surface. Indian J. Ophthalmol. 2018;66(2):190. doi: 10.4103/ijo.IJO_646_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mobaraki M., et al. Corneal repair and regeneration: current concepts and future directions. Front. Bioeng. Biotechnol. 2019;7:135. doi: 10.3389/fbioe.2019.00135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lucas S.E., Burdon K.P. Genetic and environmental risk factors for keratoconus. Annu Rev Vis Sci. 2020;6:25–46. doi: 10.1146/annurev-vision-121219-081723. [DOI] [PubMed] [Google Scholar]
- 7.Whitcher J.P., Srinivasan M., Upadhyay M.P. Corneal blindness: a global perspective. Bull. World Health Organ. 2001;79:214–221. [PMC free article] [PubMed] [Google Scholar]
- 8.Farrokhpour H., et al. A case series of infectious keratitis after corneal cross-linking. J. Refract. Surg. 2023;39(8):564–572. doi: 10.3928/1081597X-20230717-03. [DOI] [PubMed] [Google Scholar]
- 9.Zhao L., et al. Comprehensive management of post‐LASIK ectasia: from prevention to treatment. Acta Ophthalmol. 2023;101(5):485–503. doi: 10.1111/aos.15636. [DOI] [PubMed] [Google Scholar]
- 10.Fleiszig S.M., et al. Contact lens-related corneal infection: intrinsic resistance and its compromise. Prog. Retin. Eye Res. 2020;76 doi: 10.1016/j.preteyeres.2019.100804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yahalomi T., et al. Dry eye disease following LASIK, PRK, and LASEK: an observational cross-sectional study. J. Clin. Med. 2023;12(11):3761. doi: 10.3390/jcm12113761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sampaio L.P., et al. Topical losartan decreases myofibroblast generation but not corneal opacity after surface blast-simulating irregular PTK in rabbits. Translational Vision Science & Technology. 2023;12(9) doi: 10.1167/tvst.12.9.20. 20-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.de Oliveira R.C., et al. TGFβ1 and TGFβ2 proteins in corneas with and without stromal fibrosis: delayed regeneration of apical epithelial growth factor barrier and the epithelial basement membrane in corneas with stromal fibrosis. Exp. Eye Res. 2021;202 doi: 10.1016/j.exer.2020.108325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mohan R.R., et al. A novel method for generating corneal haze in anterior stroma of the mouse eye with the excimer laser. Exp. Eye Res. 2008;86(2):235–240. doi: 10.1016/j.exer.2007.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Villabona-Martinez V., et al. Corneal stromal localization of TGF beta isoforms in spontaneous persistent epithelial defects after PRK in rabbits. Exp. Eye Res. 2024;239 doi: 10.1016/j.exer.2024.109794. [DOI] [PubMed] [Google Scholar]
- 16.Martinez V.V., et al. TGF beta-3 localization in rabbit corneas with spontaneous persistent epithelial defects after Photorefractive keratectomy (PRK) Invest. Ophthalmol. Vis. Sci. 2024;65(7) 504-504. [Google Scholar]
- 17.Bachu R.D., et al. Ocular drug delivery barriers—role of nanocarriers in the treatment of anterior segment ocular diseases. Pharmaceutics. 2018;10(1):28. doi: 10.3390/pharmaceutics10010028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Peh G.S., et al. Functional evaluation of two corneal endothelial cell-based therapies: tissue-engineered construct and cell injection. Sci. Rep. 2019;9(1):1–13. doi: 10.1038/s41598-019-42493-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Del Barrio J.L.A., et al. Corneal transplantation after failed grafts: options and outcomes. Surv. Ophthalmol. 2020;66(1):20–40. doi: 10.1016/j.survophthal.2020.10.003. [DOI] [PubMed] [Google Scholar]
- 20.Rozi M.F., Sabere A.S.M. A review on conventional and novel topical ocular drug delivery system. J. Pharm. (Lahore) 2021;1(1):19–26. [Google Scholar]
- 21.Yellepeddi V.K., Palakurthi S. Recent advances in topical ocular drug delivery. J. Ocul. Pharmacol. Therapeut. 2016;32(2):67–82. doi: 10.1089/jop.2015.0047. [DOI] [PubMed] [Google Scholar]
- 22.Bodor N., Buchwald P. Ophthalmic drug design based on the metabolic activity of the eye: soft drugs and chemical delivery systems. AAPS J. 2005;7:E820–E833. doi: 10.1208/aapsj070479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nguyen D.D., Lai J.-Y. Synthesis, bioactive properties, and biomedical applications of intrinsically therapeutic nanoparticles for disease treatment. Chem. Eng. J. 2022;435 [Google Scholar]
- 24.Weng Y., et al. Nanotechnology-based strategies for treatment of ocular disease. Acta Pharm. Sin. B. 2017;7(3):281–291. doi: 10.1016/j.apsb.2016.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nguyen D.D., Lai J.-Y. Advancing the stimuli response of polymer-based drug delivery systems for ocular disease treatment. Polym. Chem. 2020;11(44):6988–7008. [Google Scholar]
- 26.Shafiq M., et al. An insight on ophthalmic drug delivery systems: focus on polymeric biomaterials-based carriers. J. Contr. Release. 2023;362:446–467. doi: 10.1016/j.jconrel.2023.08.041. [DOI] [PubMed] [Google Scholar]
- 27.Mobaraki M., et al. Biodegradable nanoparticle for cornea drug delivery: focus review. Pharmaceutics. 2020;12(12):1232. doi: 10.3390/pharmaceutics12121232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mobaraki M., Ghaffari M., Mozafari M. Self-Healing Composite Materials. Elsevier; 2020. Potential self-healing functionality in a composite structure: methodology and applications; pp. 53–70. [Google Scholar]
- 29.Xiong S., et al. Engineering topography: effects on corneal cell behavior and integration into corneal tissue engineering. Bioact. Mater. 2019;4:293–302. doi: 10.1016/j.bioactmat.2019.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lim S.H., et al. 3D printed drug delivery and testing systems—a passing fad or the future? Adv. Drug Deliv. Rev. 2018;132:139–168. doi: 10.1016/j.addr.2018.05.006. [DOI] [PubMed] [Google Scholar]
- 31.Beg S., et al. 3D printing for drug delivery and biomedical applications. Drug Discov. Today. 2020;25(9):1668–1681. doi: 10.1016/j.drudis.2020.07.007. [DOI] [PubMed] [Google Scholar]
- 32.Willemen N.G., et al. From oral formulations to drug-eluting implants: using 3D and 4D printing to develop drug delivery systems and personalized medicine. BIO-DES MANUF. 2022;5(1):85–106. [Google Scholar]
- 33.Ong H.S., Ang M., Mehta J. Evolution of therapies for the corneal endothelium: past, present and future approaches. Br. J. Ophthalmol. 2021;105(4):454–467. doi: 10.1136/bjophthalmol-2020-316149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Aghamollaei H., et al. Applications of SMILE-extracted lenticules in ophthalmology. Int. J. Ophthalmol. 2024;17(1):173. doi: 10.18240/ijo.2024.01.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mazet R., et al. Recent advances in the design of topical ophthalmic delivery systems in the treatment of ocular surface inflammation and their biopharmaceutical evaluation. Pharmaceutics. 2020;12(6):570. doi: 10.3390/pharmaceutics12060570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Patel A., et al. Ocular drug delivery systems: an overview. World J. Pharmacol. 2013;2(2):47. doi: 10.5497/wjp.v2.i2.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Iwamoto S., et al. Non-steroidal anti-inflammatory drug delays corneal wound healing by reducing production of 12-hydroxyheptadecatrienoic acid, a ligand for leukotriene B4 receptor 2. Sci. Rep. 2017;7(1):1–10. doi: 10.1038/s41598-017-13122-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Comez A.T., Ozbas M. 2022. Evaluation and Management of Ocular Traumas. [Google Scholar]
- 39.Lobo A.-M., Agelidis A.M., Shukla D. Pathogenesis of herpes simplex keratitis: the host cell response and ocular surface sequelae to infection and inflammation. Ocul. Surf. 2019;17(1):40–49. doi: 10.1016/j.jtos.2018.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yoeruek E., et al. Safety, penetration and efficacy of topically applied bevacizumab: evaluation of eyedrops in corneal neovascularization after chemical burn. Acta Ophthalmol. 2008;86(3):322–328. doi: 10.1111/j.1600-0420.2007.01049.x. [DOI] [PubMed] [Google Scholar]
- 41.Dohlman C.H., Robert M.-C., Paschalis E.I. Foundations of Corneal Disease. Springer; 2020. Treatment of chemical burn to the eye: a changing picture; pp. 109–119. [Google Scholar]
- 42.Bakunowicz-Łazarczyk A., Urban B. Assessment of therapeutic options for reducing alkali burn-induced corneal neovascularization and inflammation. Adv. Med. Sci. 2016;61(1):101–112. doi: 10.1016/j.advms.2015.10.003. [DOI] [PubMed] [Google Scholar]
- 43.Abdelfattah N.S., et al. Clinical correlates of common corneal neovascular diseases: a literature review. Int. J. Ophthalmol. 2015;8(1):182. doi: 10.3980/j.issn.2222-3959.2015.01.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Shanbhag S.S., et al. Boston keratoprosthesis type 1 for limbal stem cell deficiency after severe chemical corneal injury: a systematic review. Ocul. Surf. 2018;16(3):272–281. doi: 10.1016/j.jtos.2018.03.007. [DOI] [PubMed] [Google Scholar]
- 45.Lee R., et al. Long-term visual outcomes and complications of Boston keratoprosthesis type II implantation. Ophthalmology. 2017;124(1):27–35. doi: 10.1016/j.ophtha.2016.07.011. [DOI] [PubMed] [Google Scholar]
- 46.Singh D., et al. Outcomes of deep anterior lamellar keratoplasty following autologous simple limbal epithelial transplant in pediatric unilateral severe chemical injury. Indian J. Ophthalmol. 2017;65(3):217. doi: 10.4103/ijo.IJO_880_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Haagdorens M., et al. Limbal stem cell deficiency: current treatment options and emerging therapies. Stem Cell. Int. 2016;2016 doi: 10.1155/2016/9798374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sharma N., et al. Treatment of acute ocular chemical burns. Surv. Ophthalmol. 2018;63(2):214–235. doi: 10.1016/j.survophthal.2017.09.005. [DOI] [PubMed] [Google Scholar]
- 49.Muz O.E., et al. A novel integrated active herbal formulation ameliorates dry eye syndrome by inhibiting inflammation and oxidative stress and enhancing glycosylated phosphoproteins in rats. Pharmaceuticals. 2020;13(10):295. doi: 10.3390/ph13100295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Semba C.P., Gadek T.R.J.C.O. Development of lifitegrast: a novel T-cell inhibitor for the treatment of dry eye disease. Clin. Ophthalmol. 2016;10:1083. doi: 10.2147/OPTH.S110557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Foulks G.N., et al. Clinical guidelines for management of dry eye associated with Sjögren disease. Ocul. Surf. 2015;13(2):118–132. doi: 10.1016/j.jtos.2014.12.001. [DOI] [PubMed] [Google Scholar]
- 52.Joossen C., et al. A novel serine protease inhibitor as potential treatment for dry eye syndrome and ocular inflammation. Sci. Rep. 2020;10(1):1–14. doi: 10.1038/s41598-020-74159-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lemp M.A. Management of dry eye disease. Am. J. Manag. Care. 2008;14(3 Suppl):S88–S101. [PubMed] [Google Scholar]
- 54.Şimşek C., et al. Current management and treatment of dry eye disease. Turk J Ophthalmol. 2018;48(6):309. doi: 10.4274/tjo.69320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Solanki A., Solanki K.P. 2018. OCCULAR DRUG DELIVERY SYSTEM: A RECENT CHALLENGES AND ADVANCES. [Google Scholar]
- 56.Boddu S.H., et al. Ocular microdialysis: a continuous sampling technique to study pharmacokinetics and pharmacodynamics in the eye. Bioanalysis. 2010;2(3):487–507. doi: 10.4155/bio.10.2. [DOI] [PubMed] [Google Scholar]
- 57.Sánchez-López E., et al. Lipid nanoparticles (SLN, NLC): overcoming the anatomical and physiological barriers of the eye–Part I–Barriers and determining factors in ocular delivery. Eur. J. Pharm. Biopharm. 2017;110:70–75. doi: 10.1016/j.ejpb.2016.10.009. [DOI] [PubMed] [Google Scholar]
- 58.Lakhani P., Patil A., Majumdar S.J.T.d. Recent advances in topical nano drug-delivery systems for the anterior ocular segment. Ther. Deliv. 2018;9(2):137–153. doi: 10.4155/tde-2017-0088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Chang T.-L., et al. Nanocrystal technology for drug formulation and delivery. Front. Chem. Sci. Eng. 2015;9(1):1–14. [Google Scholar]
- 60.Himri I., Guaadaoui A. Nanostructures for the Engineering of Cells, Tissues and Organs. Elsevier; 2018. Cell and organ drug targeting: types of drug delivery systems and advanced targeting strategies; pp. 1–66. [Google Scholar]
- 61.Wilhelmus K., et al. Risk factors for herpes simplex virus epithelial keratitis recurring during treatment of stromal keratitis or iridocyclitis. Herpetic Eye Disease Study Group. Br. J. Ophthalmol. 1996;80(11):969–972. doi: 10.1136/bjo.80.11.969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Pflugfelder S.C. Antiinflammatory therapy for dry eye. Am. J. Ophthalmol. 2004;137(2):337–342. doi: 10.1016/j.ajo.2003.10.036. [DOI] [PubMed] [Google Scholar]
- 63.Patel R., et al. An antibiotic efficacy comparative study between market-prescribed and solid-lipid fluoroquinolone ophthalmic nanoformulation against conjunctivitis. Materials. 2024:39–55. [Google Scholar]
- 64.Ameeduzzafar Colloidal drug delivery system: amplify the ocular delivery. Drug Deliv. 2016;23(3):700–716. doi: 10.3109/10717544.2014.923065. [DOI] [PubMed] [Google Scholar]
- 65.Wang L., et al. Evaluation of his6-metal assemblies as a drug delivery vehicle in the treatment of anterior segment disease using a corneal inflammation model. ACS Biomater. Sci. Eng. 2020;6(7):4012–4023. doi: 10.1021/acsbiomaterials.0c00218. [DOI] [PubMed] [Google Scholar]
- 66.Ghate D., Edelhauser H.F. Ocular drug delivery. Expet Opin. Drug Deliv. 2006;3(2):275–287. doi: 10.1517/17425247.3.2.275. [DOI] [PubMed] [Google Scholar]
- 67.Gonzalez-Mira E., et al. Improved and safe transcorneal delivery of flurbiprofen by NLC and NLC-based hydrogels. J. Pharmaceut. Sci. 2012;101(2):707–725. doi: 10.1002/jps.22784. [DOI] [PubMed] [Google Scholar]
- 68.Cheng K.-J., et al. Ocular disease therapeutics: design and delivery of drugs for diseases of the eye. J. Med. Chem. 2020;63(19):10533–10593. doi: 10.1021/acs.jmedchem.9b01033. [DOI] [PubMed] [Google Scholar]
- 69.Klaassen I., et al. Molecular basis of the inner blood-retinal barrier and its breakdown in diabetic macular edema and other pathological conditions. Prog. Retin. Eye Res. 2013;34:19–48. doi: 10.1016/j.preteyeres.2013.02.001. [DOI] [PubMed] [Google Scholar]
- 70.Li J., et al. Fabrication of a drug delivery system that enhances antifungal drug corneal penetration. Drug Deliv. 2018;25(1):938–949. doi: 10.1080/10717544.2018.1461278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Gote V., et al. Ocular drug delivery: present innovations and future challenges. J. Pharmacol. Exp. Therapeut. 2019;370(3):602–624. doi: 10.1124/jpet.119.256933. [DOI] [PubMed] [Google Scholar]
- 72.Bertens C.J., et al. Topical drug delivery devices: a review. Exp. Eye Res. 2018;168:149–160. doi: 10.1016/j.exer.2018.01.010. [DOI] [PubMed] [Google Scholar]
- 73.Yellepeddi V.K., et al. Punctal plug: a medical device to treat dry eye syndrome and for sustained drug delivery to the eye. Drug Discov. Today. 2015;20(7):884–889. doi: 10.1016/j.drudis.2015.01.013. [DOI] [PubMed] [Google Scholar]
- 74.Molokhia S.A., et al. Anterior eye segment drug delivery systems: current treatments and future challenges. J. Ocul. Pharmacol. Therapeut. 2013;29(2):92–105. doi: 10.1089/jop.2012.0241. [DOI] [PubMed] [Google Scholar]
- 75.Gupta H., Aqil M.J.D.d.t. Contact lenses in ocular therapeutics. Drug Discov. Today. 2012;17(9–10):522–527. doi: 10.1016/j.drudis.2012.01.014. [DOI] [PubMed] [Google Scholar]
- 76.Tieppo A., et al. In vitro controlled release of an anti-inflammatory from daily disposable therapeutic contact lenses under physiological ocular tear flow. Eur. J. Pharm. Biopharm. 2012;81(1):170–177. doi: 10.1016/j.ejpb.2012.01.015. [DOI] [PubMed] [Google Scholar]
- 77.Kim J., Chauhan A. Dexamethasone transport and ocular delivery from poly (hydroxyethyl methacrylate) gels. Int. J. Pharm. 2008;353(1–2):205–222. doi: 10.1016/j.ijpharm.2007.11.049. [DOI] [PubMed] [Google Scholar]
- 78.Peng C.-C., Chauhan A. Extended cyclosporine delivery by silicone–hydrogel contact lenses. J. Contr. Release. 2011;154(3):267–274. doi: 10.1016/j.jconrel.2011.06.028. [DOI] [PubMed] [Google Scholar]
- 79.Dumbleton K.J.C.L., Eye A. Adverse events with silicone hydrogel continuous wear. Contact Lens Anterior Eye. 2002;25(3):137–146. doi: 10.1016/s1367-0484(02)00009-7. [DOI] [PubMed] [Google Scholar]
- 80.Singh M., et al. Therapeutic nanoemulsions in ophthalmic drug administration: concept in formulations and characterization techniques for ocular drug delivery. J. Contr. Release. 2020;10(328):895–916. doi: 10.1016/j.jconrel.2020.10.025. [DOI] [PubMed] [Google Scholar]
- 81.Parra A.S., et al. Assessment of the Prosthetic Replacement of Ocular Surface Ecosystem (PROSE) scleral lens on visual acuity for corneal irregularity and ocular surface disease. Ocul. Surf. 2018;16(2):254–258. doi: 10.1016/j.jtos.2018.01.003. [DOI] [PubMed] [Google Scholar]
- 82.Wang Y., et al. Prosthetic replacement of the ocular surface ecosystem treatment for ocular surface disease in pediatric patients with Stevens-Johnson syndrome. Am. J. Ophthalmol. 2019;201:1–8. doi: 10.1016/j.ajo.2019.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Yin J., Jacobs D.S. Long-term outcome of using Prosthetic Replacement of Ocular Surface Ecosystem (PROSE) as a drug delivery system for bevacizumab in the treatment of corneal neovascularization. Ocul. Surf. 2019;17(1):134–141. doi: 10.1016/j.jtos.2018.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Willoughby C., Batterbury M., Kaye S. Collagen corneal shields. Surv. Ophthalmol. 2002;47(2):174–182. doi: 10.1016/s0039-6257(01)00304-6. [DOI] [PubMed] [Google Scholar]
- 85.Poland D.E., Kaufman H.E. Clinical uses of collagen shields. J. Cataract Refract. Surg. 1988;14(5):489–491. doi: 10.1016/s0886-3350(88)80003-8. [DOI] [PubMed] [Google Scholar]
- 86.Dave V.S. Formulation approaches for ocular drug delivery. Nano-Biomaterials For Ophthalmic Drug Delivery. 2016:147–175. [Google Scholar]
- 87.Agban Y., et al. Nanoparticle cross-linked collagen shields for sustained delivery of pilocarpine hydrochloride. Int. J. Pharm. 2016;501(1–2):96–101. doi: 10.1016/j.ijpharm.2016.01.069. [DOI] [PubMed] [Google Scholar]
- 88.Yuan X., et al. Ocular drug delivery nanowafer with enhanced therapeutic efficacy. ACS Nano. 2015;9(2):1749–1758. doi: 10.1021/nn506599f. [DOI] [PubMed] [Google Scholar]
- 89.Coursey T.G., et al. Dexamethasone nanowafer as an effective therapy for dry eye disease. J. Contr. Release. 2015;213:168–174. doi: 10.1016/j.jconrel.2015.07.007. [DOI] [PubMed] [Google Scholar]
- 90.Mehra N.K., et al. Safety and toxicity of nanomaterials for ocular drug delivery applications. Nanotoxicology. 2016;10(7):836–860. doi: 10.3109/17435390.2016.1153165. [DOI] [PubMed] [Google Scholar]
- 91.Coursey T.G., et al. Dexamethasone nanowafer as an effective therapy for dry eye disease. J. Contr. Release. 2015;213:168–174. doi: 10.1016/j.jconrel.2015.07.007. [DOI] [PubMed] [Google Scholar]
- 92.Marcano D.C., et al. Synergistic cysteamine delivery nanowafer as an efficacious treatment modality for corneal cystinosis. Mol. Pharm. 2016;13(10):3468–3477. doi: 10.1021/acs.molpharmaceut.6b00488. [DOI] [PubMed] [Google Scholar]
- 93.Donnelly R.F., et al. 2012. Microneedle-mediated Transdermal and Intradermal Drug Delivery. [Google Scholar]
- 94.Quinn H.L., et al. The role of microneedles for drug and vaccine delivery. Expet Opin. Drug Deliv. 2014;11(11):1769–1780. doi: 10.1517/17425247.2014.938635. [DOI] [PubMed] [Google Scholar]
- 95.Thakur Singh R.R., et al. Minimally invasive microneedles for ocular drug delivery. Expet Opin. Drug Deliv. 2017;14(4):525–537. doi: 10.1080/17425247.2016.1218460. [DOI] [PubMed] [Google Scholar]
- 96.Jamaledin R., et al. Advances in antimicrobial microneedle patches for combating infections. Adv. Mater. 2020;32(33) doi: 10.1002/adma.202002129. [DOI] [PubMed] [Google Scholar]
- 97.Khandan O., et al. Fenestrated microneedles for ocular drug delivery. Sensor. Actuator. B Chem. 2016;223:15–23. [Google Scholar]
- 98.Ponniah L.R.D.R., et al. Novel drug-repository contact lens for prolonging the antimicrobial-cornea interaction for bacterial keratitis treatment: randomised controlled trial results. BMJ Open Ophthalmology. 2022;7(1) [Google Scholar]
- 99.Chen M., Choi S.Y.J.M.H., Discovery, Ophthalmology I.i. Preliminary outcomes of temporary collagen punctal plugs for patients with dry eye and glaucoma. Discov Innov Ophthalmol. 2020;9(1):56. [PMC free article] [PubMed] [Google Scholar]
- 100.Abouelatta S.M., et al. Optimized molecular imprints in gamma-irradiated collagen shields of an antifungal drug: in vitro characterization, in-vivo bioavailability enhancement. Eur. J. Pharm. Biopharm. 2021;1(166):135–143. doi: 10.1016/j.ejpb.2021.06.008. [DOI] [PubMed] [Google Scholar]
- 101.Than A., et al. Self-implantable double-layered micro-drug-reservoirs for efficient and controlled ocular drug delivery. Nat. Commun. 2018;9(1):1–12. doi: 10.1038/s41467-018-06981-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Bhatnagar S., et al. Corneal delivery of besifloxacin using rapidly dissolving polymeric microneedles. Drug Deliv. Transl. Res. 2018;8(3):473–483. doi: 10.1007/s13346-017-0470-8. [DOI] [PubMed] [Google Scholar]
- 103.Wilczewska A.Z., et al. Nanoparticles as drug delivery systems. Pharmacol. Rep. 2012;64(5):1020–1037. doi: 10.1016/s1734-1140(12)70901-5. [DOI] [PubMed] [Google Scholar]
- 104.Nguyen D.D., Luo L.J., Lai J.Y. Dendritic effects of injectable biodegradable thermogels on pharmacotherapy of inflammatory glaucoma‐associated degradation of extracellular matrix. Adv. Healthcare Mater. 2019;8(24) doi: 10.1002/adhm.201900702. [DOI] [PubMed] [Google Scholar]
- 105.Lai J.-Y., Luo L.-J., Nguyen D.D. Multifunctional glutathione-dependent hydrogel eye drops with enhanced drug bioavailability for glaucoma therapy. Chem. Eng. J. 2020;402 [Google Scholar]
- 106.Luo L.-J., et al. Therapeutic hydrogel sheets programmed with multistage drug delivery for effective treatment of corneal abrasion. Chem. Eng. J. 2022;429 [Google Scholar]
- 107.Nguyen D.D., et al. Highly retina-permeating and long-acting resveratrol/metformin nanotherapeutics for enhanced treatment of macular degeneration. ACS Nano. 2022;17(1):168–183. doi: 10.1021/acsnano.2c05824. [DOI] [PubMed] [Google Scholar]
- 108.Luo L.-J., et al. Targeting nanocomposites with anti-oxidative/inflammatory/angiogenic activities for synergistically alleviating macular degeneration. Appl. Mater. Today. 2021;24 [Google Scholar]
- 109.ud Din F., et al. Effective use of nanocarriers as drug delivery systems for the treatment of selected tumors. Int. J. Nanomed. 2017;12:7291. doi: 10.2147/IJN.S146315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sánchez A., Mejía S.P., Orozco J.J.M. Recent advances in polymeric nanoparticle-encapsulated drugs against intracellular infections. Molecules. 2020;25(16):3760. doi: 10.3390/molecules25163760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Onugwu A.L., et al. Nanotechnology based drug delivery systems for the treatment of anterior segment eye diseases. J. Contr. Release. 2023;354:465–488. doi: 10.1016/j.jconrel.2023.01.018. [DOI] [PubMed] [Google Scholar]
- 112.Rani K., Paliwal S. A review on targeted drug delivery: its entire focus on advanced therapeutics and diagnostics. Scholars J. Appl. Med. Sci. 2014;2(1C):328–331. [Google Scholar]
- 113.Muller R.H., Keck C.M. Challenges and solutions for the delivery of biotech drugs–a review of drug nanocrystal technology and lipid nanoparticles. J. Biotechnol. 2004;113(1–3):151–170. doi: 10.1016/j.jbiotec.2004.06.007. [DOI] [PubMed] [Google Scholar]
- 114.Vasir J.K., Reddy M.K., Labhasetwar V.D.J.C.N. Nanosystems in drug targeting: opportunities and challenges. Curr. Nanosci. 2005;1(1):47–64. [Google Scholar]
- 115.Huang W., et al. Hexahistidine-metal assemblies: a promising drug delivery system. Acta Biomater. 2019;90:441–452. doi: 10.1016/j.actbio.2019.03.058. [DOI] [PubMed] [Google Scholar]
- 116.Battaglia L., et al. Application of lipid nanoparticles to ocular drug delivery. Expet Opin. Drug Deliv. 2016;13(12):1743–1757. doi: 10.1080/17425247.2016.1201059. [DOI] [PubMed] [Google Scholar]
- 117.Kwon S., et al. Potential therapeutic usage of nanomedicine for glaucoma treatment. Int. J. Nanomed. 2020;15:5745. doi: 10.2147/IJN.S254792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Zhang X., et al. Tumor targeting strategies for chitosan-based nanoparticles. Colloids Surf. B Biointerfaces. 2016;148:460–473. doi: 10.1016/j.colsurfb.2016.09.020. [DOI] [PubMed] [Google Scholar]
- 119.Gatoo M.A., et al. Physicochemical properties of nanomaterials: implication in associated toxic manifestations. BioMed Res. Int. 2014;2014 doi: 10.1155/2014/498420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Duan X., Li Y.J.S. Physicochemical characteristics of nanoparticles affect circulation, biodistribution, cellular internalization, and trafficking. Small. 2013;9(9‐10):1521–1532. doi: 10.1002/smll.201201390. [DOI] [PubMed] [Google Scholar]
- 121.Qamar Z., et al. Nano-based drug delivery system: recent strategies for the treatment of ocular disease and future perspective. Recent Pat. Drug Deliv. Formulation. 2019;13(4):246–254. doi: 10.2174/1872211314666191224115211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Khatak S., Dureja H. Structural composition of solid lipid nanoparticles for invasive and non-invasive drug delivery. Current Nanomaterials. 2017;2(3):129–153. [Google Scholar]
- 123.Wang F.-z., et al. Preparation, optimization, and characterization of chitosan-coated solid lipid nanoparticles for ocular drug delivery. J Biomed Res. 2018;32(6):411. doi: 10.7555/JBR.32.20160170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Salvi V.R. P.J.J.o.D.D.S. Pawar, and Technology, Nanostructured lipid carriers (NLC) system: a novel drug targeting carrier. J. Drug Deliv. Sci. Technol. 2019;51:255–267. [Google Scholar]
- 125.Shen J., et al. Thiolated nanostructured lipid carriers as a potential ocular drug delivery system for cyclosporine A: improving in vivo ocular distribution. Int. J. Pharm. 2010;402(1–2):248–253. doi: 10.1016/j.ijpharm.2010.10.008. [DOI] [PubMed] [Google Scholar]
- 126.Gorantla S., et al. Nanocarriers for ocular drug delivery: current status and translational opportunity. RSC Adv. 2020;10(46):27835–27855. doi: 10.1039/d0ra04971a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Kumari S., et al. Dexamethasone-loaded nanostructured lipid carriers for the treatment of dry eye disease. Pharmaceutics. 2021;13(6):905. doi: 10.3390/pharmaceutics13060905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Zeng W., et al. Hyaluronic acid-coated niosomes facilitate tacrolimus ocular delivery: mucoadhesion, precorneal retention, aqueous humor pharmacokinetics, and transcorneal permeability. Colloids Surf. B Biointerfaces. 2016;141:28–35. doi: 10.1016/j.colsurfb.2016.01.014. [DOI] [PubMed] [Google Scholar]
- 129.Durak S., et al. Niosomal drug delivery systems for ocular disease—recent advances and future prospects. Nanomaterials. 2020;10(6):1191. doi: 10.3390/nano10061191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Sahoo S.K., Dilnawaz F., Krishnakumar S. Nanotechnology in ocular drug delivery. Drug Discov. Today. 2008;13(3–4):144–151. doi: 10.1016/j.drudis.2007.10.021. [DOI] [PubMed] [Google Scholar]
- 131.Farooq U., et al. Design of liposomal nanocarriers with a potential for combined dexamethasone and bevacizumab delivery to the eye. Int. J. Pharm. 2024;654 doi: 10.1016/j.ijpharm.2024.123958. [DOI] [PubMed] [Google Scholar]
- 132.Gan L., et al. Self-assembled liquid crystalline nanoparticles as a novel ophthalmic delivery system for dexamethasone: improving preocular retention and ocular bioavailability. Int. J. Pharm. 2010;396(1–2):179–187. doi: 10.1016/j.ijpharm.2010.06.015. [DOI] [PubMed] [Google Scholar]
- 133.Huang J., et al. Ocular cubosome drug delivery system for timolol maleate: preparation, characterization, cytotoxicity, ex vivo, and in vivo evaluation. AAPS PharmSciTech. 2017;18(8):2919–2926. doi: 10.1208/s12249-017-0763-8. [DOI] [PubMed] [Google Scholar]
- 134.Karami Z., Hamidi M.J.D.d.t. Cubosomes: remarkable drug delivery potential. Drug Discov. Today. 2016;21(5):789–801. doi: 10.1016/j.drudis.2016.01.004. [DOI] [PubMed] [Google Scholar]
- 135.Siekmann B., et al. Preparation and structural investigations of colloidal dispersions prepared from cubic monoglyceride–water phases. Int. J. Pharm. 2002;244(1–2):33–43. doi: 10.1016/s0378-5173(02)00298-3. [DOI] [PubMed] [Google Scholar]
- 136.Singhvi G., Banerjee S., Khosa A. Organic Materials as Smart Nanocarriers for Drug Delivery. Elsevier; 2018. Lyotropic liquid crystal nanoparticles: a novel improved lipidic drug delivery system; pp. 471–517. [Google Scholar]
- 137.Bhageerathy A., Prasanth V. Formulation and evaluation of moxifloxacin hydrochloride loaded cubosomal gel for ocular delivery. J. Drug Deliv. Therapeut. 2024;14(3):88–99. [Google Scholar]
- 138.Cejkova J., et al. Treatment of alkali-injured cornea by cyclosporine A-loaded electrospun nanofibers–An alternative mode of therapy. Exp. Eye Res. 2016;147:128–137. doi: 10.1016/j.exer.2016.04.016. [DOI] [PubMed] [Google Scholar]
- 139.Grimaudo M.A., Concheiro A., Alvarez-Lorenzo C.J.P. Crosslinked hyaluronan electrospun nanofibers for ferulic acid ocular delivery. Pharmaceutics. 2020;12(3):274. doi: 10.3390/pharmaceutics12030274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Sangole S., et al. Electrospun nanofiber composite for levofloxacin in ocular drug delivery. Pharm. Nanotechnol. 2022;10(5):393–400. [Google Scholar]
- 141.Rajoria G., Gupta A.J.A. In-situ gelling system: a novel approach for ocular drug delivery. AJPTR. 2012;2:24–53. [Google Scholar]
- 142.Rahbar N., et al. Chitosan/carbomer nanoparticles-laden in situ gel for improved ocular delivery of timolol: in vitro, in vivo, and ex vivo study. Drug Delivery and Translational Research. 2024:1–11. doi: 10.1007/s13346-024-01663-1. [DOI] [PubMed] [Google Scholar]
- 143.Cholkar K., et al. Novel nanomicellar formulation approaches for anterior and posterior segment ocular drug delivery. Recent Pat. Nanomed. 2012;2(2):82–95. doi: 10.2174/1877912311202020082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Wang X., Wang S., Zhang Y.J.D.d. Advance of the application of nano-controlled release system in ophthalmic drug delivery. Drug Deliv. 2016;23(8):2897–2901. doi: 10.3109/10717544.2015.1116025. [DOI] [PubMed] [Google Scholar]
- 145.Vadlapudi A.D., Mitra A.K.J.T.d. Nanomicelles: an emerging platform for drug delivery to the eye. Thea Deliv. 2013;4(1):1–3. doi: 10.4155/tde.12.122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Civiale C., et al. Polyhydroxyethylaspartamide-based micelles for ocular drug delivery. Int. J. Pharm. 2009;378(1–2):177–186. doi: 10.1016/j.ijpharm.2009.05.028. [DOI] [PubMed] [Google Scholar]
- 147.Vadlapudi A., et al. Ocular drug delivery. Drug Deliv. Jones Bartlett Learn. Burlingt. 2015;1:219–263. Ma USA. [Google Scholar]
- 148.Sathe P., et al. Nanomicelles empower natamycin in treating fungal keratitis: an in vitro, ex vivo and in vivo study. Int. J. Pharm. 2024;656 doi: 10.1016/j.ijpharm.2024.124118. [DOI] [PubMed] [Google Scholar]
- 149.Khiev D., et al. Emerging nano-formulations and nanomedicines applications for ocular drug delivery. Nanomaterials. 2021;11(1):173. doi: 10.3390/nano11010173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Mahboobian M.M., et al. Development of thermosensitive in situ gel nanoemulsions for ocular delivery of acyclovir. J. Drug Deliv. Sci. Technol. 2020;55 [Google Scholar]
- 151.Dasgupta N., Ranjan S., Gandhi M.J.E.C.L. Nanoemulsion ingredients and components. Environ. Chem. Lett. 2019;17(2):917–928. [Google Scholar]
- 152.McClements D.J., et al. Recent advances in the utilization of natural emulsifiers to form and stabilize emulsions. Annu. Rev. Food Sci. Technol. 2017;8:205–236. doi: 10.1146/annurev-food-030216-030154. [DOI] [PubMed] [Google Scholar]
- 153.Yeu E., et al. Efficacy and safety of phospholipid nanoemulsion-based ocular lubricant for the management of various subtypes of dry eye disease: a phase IV, multicenter trial. Clin. Ophthalmol. 2020:2561–2570. doi: 10.2147/OPTH.S261318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Araújo R.V.d., et al. New advances in general biomedical applications of PAMAM dendrimers. Molecules. 2018;23(11):2849. doi: 10.3390/molecules23112849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Abdelkader H., Alany R.G. Controlled and continuous release ocular drug delivery systems: pros and cons. Curr. Drug Deliv. 2012;9(4):421–430. doi: 10.2174/156720112801323125. [DOI] [PubMed] [Google Scholar]
- 156.Vandamme T.F., Brobeck L. Poly (amidoamine) dendrimers as ophthalmic vehicles for ocular delivery of pilocarpine nitrate and tropicamide. J. Contr. Release. 2005;102(1):23–38. doi: 10.1016/j.jconrel.2004.09.015. [DOI] [PubMed] [Google Scholar]
- 157.Peng Y., et al. Research and development of drug delivery systems based on drug transporter and nano-formulation. Asian J. Pharm. Sci. 2020;15(2):220–236. doi: 10.1016/j.ajps.2020.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Lancina III, M.G. and H.J.C.j.o.c. Yang, Dendrimers for ocular drug delivery. Can. J. Chem. 2017;95(9):897–902. doi: 10.1139/cjc-2017-0193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Lin H., et al. Subconjunctival dendrimer-drug therapy for the treatment of dry eye in a rabbit model of induced autoimmune dacryoadenitis. Ocul. Surf. 2018;16(4):415–423. doi: 10.1016/j.jtos.2018.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Liu C.-H., et al. Encapsulating curcumin in ethylene diamine-β-cyclodextrin nanoparticle improves topical cornea delivery. Colloids Surf. B Biointerfaces. 2020;186 doi: 10.1016/j.colsurfb.2019.110726. [DOI] [PubMed] [Google Scholar]
- 161.Karim S., et al. 2018. A GLIMPSE OF MINI REVIEW ON OCULAR DRUG DELIVERY SYSTEM. [Google Scholar]
- 162.Loftsson T., Brewster M.E. Cyclodextrins as functional excipients: methods to enhance complexation efficiency. J. Pharmaceut. Sci. 2012;101(9):3019–3032. doi: 10.1002/jps.23077. [DOI] [PubMed] [Google Scholar]
- 163.Loftsson T. Brewster, and Pharmacology, Pharmaceutical applications of cyclodextrins: effects on drug permeation through biological membranes. J. Pharm. Pharmacol. 2011;63(9):1119–1135. doi: 10.1111/j.2042-7158.2011.01279.x. [DOI] [PubMed] [Google Scholar]
- 164.Meza-Rios A., et al. Therapies based on nanoparticles for eye drug delivery. Ophthalmol Ther. 2020;9:1–14. doi: 10.1007/s40123-020-00257-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Sohani Z., et al. The Effect of Topical Curcumin Nanoparticles Encapsulated with β-Cyclodextrin into Hyaluronic Acid-Based Hydrogel Complex on Experimental Ulcerative Keratitis in Comparison with N-Acetylcysteine in Rabbit. Model. 2024 [Google Scholar]
- 166.Chang M.-C., et al. Liposomal dexamethasone–moxifloxacin nanoparticle combinations with collagen/gelatin/alginate hydrogel for corneal infection treatment and wound healing. Biomed. Mater. 2020;15(5) doi: 10.1088/1748-605X/ab9510. [DOI] [PubMed] [Google Scholar]
- 167.Sanjay S.T., et al. Recent advances of controlled drug delivery using microfluidic platforms. Adv. Drug Deliv. Rev. 2018;128:3–28. doi: 10.1016/j.addr.2017.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Elkhoury K., et al. Engineering smart targeting nanovesicles and their combination with hydrogels for controlled drug delivery. Pharmaceutics. 2020;12(9):849. doi: 10.3390/pharmaceutics12090849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Elkhoury K., et al. Soft‐Nanoparticle functionalization of natural hydrogels for tissue engineering applications. Adv. Healthcare Mater. 2019;8(18) doi: 10.1002/adhm.201900506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Piffoux M., et al. Modification of extracellular vesicles by fusion with liposomes for the design of personalized biogenic drug delivery systems. ACS Nano. 2018;12(7):6830–6842. doi: 10.1021/acsnano.8b02053. [DOI] [PubMed] [Google Scholar]
- 171.Yang C.-J., et al. Unveiling the power of gabapentin-loaded nanoceria with multiple therapeutic capabilities for the treatment of dry eye disease. ACS Nano. 2023;17(24):25118–25135. doi: 10.1021/acsnano.3c07817. [DOI] [PubMed] [Google Scholar]
- 172.Nguyen D.D., et al. Amination-mediated nano eye-drops with enhanced corneal permeability and effective burst release for acute glaucoma treatment. Chem. Eng. J. 2023;451 [Google Scholar]
- 173.Yang C.J., Nguyen D.D., Lai J.Y. Poly (l‐Histidine)‐mediated on‐demand therapeutic delivery of roughened ceria nanocages for treatment of chemical eye injury. Adv. Sci. 2023;10(26) doi: 10.1002/advs.202302174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Luo L.-J., Nguyen D.D., Lai J.-Y. Harnessing the tunable cavity of nanoceria for enhancing Y-27632-mediated alleviation of ocular hypertension. Theranostics. 2021;11(11):5447. doi: 10.7150/thno.54525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Hou W., et al. Photo-responsive polymersomes as drug delivery system for potential medical applications. Molecules. 2020;25(21):5147. doi: 10.3390/molecules25215147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Duan Y., et al. Light-triggered nitric oxide (NO) release from photoresponsive polymersomes for corneal wound healing. Chem. Sci. 2020;11(1):186–194. doi: 10.1039/c9sc04039k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Wu Q., et al. Development and effects of tacrolimus-loaded nanoparticles on the inhibition of corneal allograft rejection. Drug Deliv. 2019;26(1):290–299. doi: 10.1080/10717544.2019.1582728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Khames A., et al. Natamycin solid lipid nanoparticles–sustained ocular delivery system of higher corneal penetration against deep fungal keratitis: preparation and optimization. Int. J. Nanomed. 2019;14:2515. doi: 10.2147/IJN.S190502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Lakhani P., et al. Curcumin-loaded Nanostructured Lipid Carriers for ocular drug delivery: design optimization and characterization. J. Drug Deliv. Sci. Technol. 2018;47:159–166. doi: 10.1016/j.jddst.2018.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.El-Nabarawi M.A., et al. Natamycin niosomes as a promising ocular nanosized delivery system with ketorolac tromethamine for dual effects for treatment of candida rabbit keratitis; in vitro/in vivo and histopathological studies. Drug Dev. Ind. Pharm. 2019;45(6):922–936. doi: 10.1080/03639045.2019.1579827. [DOI] [PubMed] [Google Scholar]
- 181.Nasr M., et al. In vitro and in vivo evaluation of cubosomal nanoparticles as an ocular delivery system for fluconazole in treatment of keratomycosis. Drug Deliv. Transl. Res. 2020;10(6):1841–1852. doi: 10.1007/s13346-020-00830-4. [DOI] [PubMed] [Google Scholar]
- 182.Moustafa M.A., et al. Hyalugel-integrated liposomes as a novel ocular nanosized delivery system of fluconazole with promising prolonged effect. Int. J. Pharm. 2017;534(1–2):14–24. doi: 10.1016/j.ijpharm.2017.10.007. [DOI] [PubMed] [Google Scholar]
- 183.Liu D., et al. Co-delivery of metformin and levofloxacin hydrochloride using biodegradable thermosensitive hydrogel for the treatment of corneal neovascularization. Drug Deliv. 2019;26(1):522–531. doi: 10.1080/10717544.2019.1609623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Guo C., et al. Intranasal delivery of nanomicelle curcumin promotes corneal epithelial wound healing in streptozotocin-induced diabetic mice. Sci. Rep. 2016;6(1):1–13. doi: 10.1038/srep29753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Soiberman U., et al. Subconjunctival injectable dendrimer-dexamethasone gel for the treatment of corneal inflammation. Biomaterials. 2017;125:38–53. doi: 10.1016/j.biomaterials.2017.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.García-Otero X., et al. Development and characterization of a tacrolimus/hydroxypropyl-β-cyclodextrin eye drop. Pharmaceutics. 2021;13(2):149. doi: 10.3390/pharmaceutics13020149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Wei C., et al. A drug‐free therapeutic system for cancer therapy by diselenide‐based polymers themselves. Adv. Healthcare Mater. 2021;10(2) doi: 10.1002/adhm.202001471. [DOI] [PubMed] [Google Scholar]
- 188.Ganoth A., Merimi K.C., Peer D. Overcoming multidrug resistance with nanomedicines. Expet Opin. Drug Deliv. 2015;12(2):223–238. doi: 10.1517/17425247.2015.960920. [DOI] [PubMed] [Google Scholar]
- 189.Wang Q., et al. Multifunctional shell–core nanoparticles for treatment of multidrug resistance hepatocellular carcinoma. Adv. Funct. Mater. 2018;28(14) [Google Scholar]
- 190.Torchilin V.P. Multifunctional, stimuli-sensitive nanoparticulate systems for drug delivery. Nat. Rev. Drug Discov. 2014;13(11):813–827. doi: 10.1038/nrd4333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Lu Y., et al. Bioresponsive materials. Nat. Rev. Mater. 2016;2(1):1–17. [Google Scholar]
- 192.Wang A.Z., Langer R., Farokhzad O.C. Nanoparticle delivery of cancer drugs. Annu. Rev. Med. 2012;63:185–198. doi: 10.1146/annurev-med-040210-162544. [DOI] [PubMed] [Google Scholar]
- 193.Ye M., et al. A tumor‐specific cascade amplification drug release nanoparticle for overcoming multidrug resistance in cancers. Adv. Mater. 2017;29(38) doi: 10.1002/adma.201702342. [DOI] [PubMed] [Google Scholar]
- 194.Wang Y., et al. A step‐by‐step multiple stimuli‐responsive nanoplatform for enhancing combined chemo‐photodynamic therapy. Adv. Mater. 2017;29(12) doi: 10.1002/adma.201605357. [DOI] [PubMed] [Google Scholar]
- 195.Greenwald R.B., et al. Effective drug delivery by PEGylated drug conjugates. Adv. Drug Deliv. Rev. 2003;55(2):217–250. doi: 10.1016/s0169-409x(02)00180-1. [DOI] [PubMed] [Google Scholar]
- 196.Saito E., et al. Designing drug-free biodegradable nanoparticles to modulate inflammatory monocytes and neutrophils for ameliorating inflammation. J. Contr. Release. 2019;300:185–196. doi: 10.1016/j.jconrel.2019.02.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Luo L.-J., et al. Dual-functional gelatin-capped silver nanoparticles for antibacterial and antiangiogenic treatment of bacterial keratitis. J. Colloid Interface Sci. 2019;536:112–126. doi: 10.1016/j.jcis.2018.10.041. [DOI] [PubMed] [Google Scholar]
- 198.Fang G., et al. Hydrogels-based ophthalmic drug delivery systems for treatment of ocular diseases. Mater. Sci. Eng. C. 2021;127 doi: 10.1016/j.msec.2021.112212. [DOI] [PubMed] [Google Scholar]
- 199.Al-Kinani A.A., et al. Ophthalmic gels: past, present and future. Adv. Drug Deliv. Rev. 2018;126:113–126. doi: 10.1016/j.addr.2017.12.017. [DOI] [PubMed] [Google Scholar]
- 200.Lin P.-H., et al. Alleviation of dry eye syndrome with one dose of antioxidant, anti-inflammatory, and mucoadhesive lysine-carbonized nanogels. Acta Biomater. 2022;141:140–150. doi: 10.1016/j.actbio.2022.01.044. [DOI] [PubMed] [Google Scholar]
- 201.Jiao Z., et al. Drug-free contact lens based on quaternized chitosan and tannic acid for bacterial keratitis therapy and corneal repair. Carbohydr. Polym. 2022;286 doi: 10.1016/j.carbpol.2022.119314. [DOI] [PubMed] [Google Scholar]
- 202.Elkasabgy N.A., Mahmoud A.A., Maged A. 3D printing: an appealing route for customized drug delivery systems. Int. J. Pharm. 2020;588 doi: 10.1016/j.ijpharm.2020.119732. [DOI] [PubMed] [Google Scholar]
- 203.Parhi R., Jena G.K. An updated review on application of 3D printing in fabricating pharmaceutical dosage forms. Drug Deliv Transl Res. 2021:1–35. doi: 10.1007/s13346-021-01074-6. [DOI] [PubMed] [Google Scholar]
- 204.Peng H., et al. 3D printing processes in precise drug delivery for personalized medicine. Biofabrication. 2024;16(3) doi: 10.1088/1758-5090/ad3a14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Huang X., et al. Dexamethasone pharmacokinetics characteristics via sub-tenon microfluidic system in uveitis rabbits. J. Drug Deliv. Sci. Technol. 2020;57 [Google Scholar]
- 206.Trenfield S.J., et al. 3D printed drug products: non-destructive dose verification using a rapid point-and-shoot approach. Int. J. Pharm. 2018;549(1–2):283–292. doi: 10.1016/j.ijpharm.2018.08.002. [DOI] [PubMed] [Google Scholar]
- 207.Elkasabgy N.A., Mahmoud A.A. Fabrication strategies of scaffolds for delivering active ingredients for tissue engineering. AAPS PharmSciTech. 2019;20(7):1–18. doi: 10.1208/s12249-019-1470-4. [DOI] [PubMed] [Google Scholar]
- 208.Alhnan M.A., et al. Emergence of 3D printed dosage forms: opportunities and challenges. Pharm. Res. (N. Y.) 2016;33(8):1817–1832. doi: 10.1007/s11095-016-1933-1. [DOI] [PubMed] [Google Scholar]
- 209.Tagami T., et al. Lyophilized ophthalmologic patches as novel corneal drug formulations using a semi-solid extrusion 3D printer. Int. J. Pharm. 2022;617 doi: 10.1016/j.ijpharm.2022.121448. [DOI] [PubMed] [Google Scholar]
- 210.Xu X., et al. 3D printed punctal plugs for controlled ocular drug delivery. Pharmaceutics. 2021;13(9):1421. doi: 10.3390/pharmaceutics13091421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Duman G., et al. Development and evaluation of 3D-printed ocular insert containing liposomal moxifloxacin. J. Drug Deliv. Sci. Technol. 2024;92 [Google Scholar]
- 212.Fitaihi R., et al. Optimization of stereolithography 3D printing of microneedle micro-molds for ocular drug delivery. Int. J. Pharm. 2024;658 doi: 10.1016/j.ijpharm.2024.124195. [DOI] [PubMed] [Google Scholar]
- 213.Nosrati H., et al. Directed differentiation of adipose-derived stem cells using imprinted cell-like topographies as a growth factor-free approach. Stem Cell Reviews and Reports. 2024:1–30. doi: 10.1007/s12015-024-10767-7. [DOI] [PubMed] [Google Scholar]
- 214.Jadidi K., et al. Acellular human amniotic membrane: a safe and stable matrix for corneal stromal regeneration. Colloids Surf. A Physicochem. Eng. Asp. 2024;690 [Google Scholar]
- 215.Fallah Tafti M., Faghihi S. Cartilage: from Biology to Biofabrication. Springer; 2023. Cartilage tissue engineering: advances and frontiers; pp. 255–287. [Google Scholar]
- 216.Nie X., et al. 3D printing sequentially strengthening high-strength natural polymer hydrogel bilayer scaffold for cornea regeneration. Regenerative Biomaterials. 2024;11:rbae012. doi: 10.1093/rb/rbae012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Hu J., et al. AI-driven design of customized 3D-printed multi-layer capsules with controlled drug release profiles for personalized medicine. Int. J. Pharm. 2024;656 doi: 10.1016/j.ijpharm.2024.124114. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data will be made available on request.