Abstract
Purpose
Norepinephrine (NE) has been implicated in epithelial–mesenchymal transition (EMT) of cancer cells. However, the underlying mechanism is poorly understood. The goal of this study was to explore the effect of NE on cancer cell EMT and to investigate the potential mechanism.
Methods
HT-29 and A549 cells were treated with NE, β-adrenergic receptor (β-AR) antagonist (propranolol) or inhibitor of transforming growth factor-β (TGF-β) receptor type I kinase (Ly2157299). Morphology of cells was observed with optical and electron microscope and immunofluorescence staining. Cellular migration and invasion were tested with transwell migration assay and Matrigel invasion assay, respectively. TGF-β1 and cyclic adenosine monophosphate (cAMP) were quantified. EMT markers and signaling pathway were measured by RT-PCR and western blot.
Results
NE stimulated TGF-β1 secretion and intracellular cAMP synthesis, induced morphological alterations in HT-29 and A549 cells, and enhanced their ability of migration and invasion. EMT markers induction was observed in NE-treated cancer cells. The effect of NE could be inhibited by propranolol or Ly2157299. β-AR/TGF-β1 signaling/p-Smad3/Snail and β-AR/TGF-β1 signaling/HIF-1α/Snail were two signaling pathways.
Conclusion
These findings demonstrated that TGF-β1 signaling pathway was a significant factor of NE-induced cancer cells EMT. The data also suggested that psychological stress might be a risk factor which enhances the ability of migration or invasion of cancer cells.
Keywords: Chronic stress, Norepinephrine, Adrenergic receptor, Transforming growth factor-β1, Epithelial–mesenchymal transition
Introduction
Epithelial–mesenchymal transition (EMT), occurring in embryonic development and tumor progression (Thiery et al. 2009), is a process during which epithelial cells acquire the characteristics of mesenchymal cells (Thiery 2002). This process has been shown to be an important step for tumor invasion and metastasis (Thiery et al. 2009). During EMT, tumor cells gradually lose intercellular contacts and undergo morphological changes from epithelial cells to mesenchymal-appearing cells (Xie et al. 2010). Increased microvilli and invasiveness, upregulated mesenchymal markers such as vimentin, and reduced adhesive ability are hallmarks for EMT (Xie et al. 2010; Shan et al. 2014). The decreased E-cadherin and the overexpressed vimentin are believed to be key characters of EMT (Shan et al. 2014; Xu et al. 2009; Xie et al. 2010). Besides, as a repressor of E-cadherin, Snail is considered as another critical EMT marker (Thiery et al. 2009; Moreno-Bueno et al. 2009).
Transforming growth factor-beta1 (TGF-β1), hepatocyte growth factor (HGF), epidermal growth factor (EGF), and placental-derived growth factor (PDGF) are common inducers of EMT in various cancers (Zlobec and Lugli 2010). Recently, it was reported that norepinephrine (NE) induced ovarian cancer cells EMT through human telomerase reverse transcriptase (hTERT) mediation (Choi et al. 2014). Shan et al. (2014) also found that NE induced gastric adenocarcinoma cell lines EMT in vitro through β2-AR (adrenergic receptor)/HIF-1α/Snail signaling pathway. Moreover, after NE treatment, EMT occurred in prostate cancer cells as well (Barbieri et al. 2015). Increasing evidence showed that cancer cells EMT was associated with NE.
Suffering from cancer, the majority of cancer patients might experience long-term negative emotions, including anxiety and depression (Reiche et al. 2004; Deng et al. 2014b; Wang et al. 2013). It is well known that chronic stress could lead to increased stress-related hormones (SRH), including epinephrine, norepinephrine, glucocorticoid. (Spiegel 2012; Thornton et al. 2010). Chronic stress is associated with tumor recurrence, progression, and poor overall survival (Glaser and Kiecolt-Glaser 2005; Reiche et al. 2004). However, NE is regarded as the most important tumor-related SRH (Entschladen et al. 2004; Kiecolt-Glaser et al. 2005), and the effect of chronic stress could be mimicked by NE (Yang et al. 2009). In various cancer cells such as melanoma, ovarian, prostate, colon, and nasopharyngeal cancer cells, NE has been demonstrated to upregulate IL-6, IL-8, VEGF (vascular epithelial growth factor), or MMP (matrix metalloproteinase) expression by β-AR activation (Yang et al. 2006a, 2009; Thaker et al. 2006; Hassan et al. 2013; Liu et al. 2014a). The activation of β-AR increases the synthesis of cAMP which further activates cAMP-dependent kinase PKA, resulting in the activation of downstream pathways (Yang et al. 2014; Sloan et al. 2010). Propranolol, an antagonist of β-AR, was found to be able to abrogate NE effect (Yang et al. 2009; Thaker et al. 2006; Liu et al. 2014a). Furthermore, propranolol was observed to be able to inhibit NE-induced prostate cancer cells EMT (Barbieri et al. 2015). Clinical research also revealed that propranolol reduced cancer progression and mortality and improved relapse-free survival (Barron et al. 2011; Melhem-Bertrandt et al. 2011).
In most malignancies, the TGF-β1 autocrine is considered to be one of the ways promoting cancer cells migration and invasion (Hasina et al. 1999; Akhurst and Derynck 2001; Jia et al. 2013). TGF-β1 plasma level is often elevated in cancer patients (Akhurst and Derynck 2001), and the elevated TGF-β1 is associated with poor prognosis in many cancer sites, such as colorectal cancer (Tsushima et al. 2001), breast cancer (de Jong et al. 1998), and prostate cancer (Wikstrom et al. 1998). A research has shown that TGF-β1 could remarkably increase the frequency of oncogenesis and shorten the latency period, representing an independent predictor of cancer recurrence (Calon et al. 2012). After TGF-β1 binding to TGF-β receptor I (TGF-βRI), Smad2, and Smad3 were phosphorylated. In company with Smad4, the Smad transcriptional complexes translocate into nucleus where they control the expression of Snail (Massague and Chen 2000; Neuzillet et al. 2015), and further induce cancer cells EMT through TGF-β1/p-Smad3/Snail signaling pathway (Liu et al. 2014b). Blocking TGF-β1 signaling seems to be associated with cancer inhibition. Nowadays, Ly2157299, a TGF-β receptor type I kinase inhibitor, is being investigated in a phase II clinical trial (NCT01246986, http://clinicaltrials.gov) (Giannelli et al. 2014). Interestingly, NE was supposed to stimulate TGF-β1 secretion (Elenkov and Chrousos 2002; Sigala et al. 2013). Then, the role of TGF-β1 signaling playing in NE-induced cancer cells EMT is worthy of exploration.
This study aimed at verifying the effect of NE-induced EMT in other cancer cells. Another goal is to investigate the potential mechanisms of TGF-β1 signaling involved in this phenomenon.
Materials and methods
Reagents, antibodies and instruments
Norepinephrine, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), and DMSO (dimethylsulfoxide) were purchased from Sigma-Aldrich (USA); propranolol from Enzo (Germany); Ly2157299 from Selleck (USA). TRIzol was from Life Technologies (USA) and One Step SYBR® PrimeScript™ RT-PCR Kit from TaKaRa (Japan). Rabbit monoclonal antibodies against human E-cadherin, vimentin, TGF-βRI, HIF-1α, p-Smad3, Smad3, Snail were obtained from Cell Signaling (USA). Rabbit anti-human GAPDH (glyceraldehyde-3-phosphate dehydrogenase), HRP-conjugated secondary antibody, CY3-labeled goat anti-rabbit, and FITC-labeled goat anti-rabbit antibodies were from Boster (China). Matrigel and 24-well transwell (8-μm pore size, 6.5 mm diameter) were purchased from BD Biosciences (USA) and Corning Costar Corporation (USA), respectively. Human TGF-β1 ELISA kit (Neobioscience Technology Co., ltd, China) and cAMP immunoassay kit (R&D Systems, Inc, USA) were used.
Cell cultures and treatments
The human colorectal adenocarcinoma HT-29 cells and human lung adenocarcinoma A549 cells, kindly gifted from State Key Laboratory of Biotherapy (Sichuan University, Chengdu), were maintained in DMEM (Gibco) and RPMI 1640 complete medium containing 10 % fetal bovine serum (Hyclone), penicillin (100 U/mL), and streptomycin (10 mg/L) at 37 °C with 5 % CO2 in humidified atmosphere, respectively. Prior to treatment, cells reached 80 % confluence and were serum-starved overnight. Subsequently, cells were treated for 4 days according to the groups as follows (all of the groups were treated with serum-free culture media to avoid possible interference of serum ingredients): (1) culture medium (control); (2) 10 μM NE; (3) 10 μM Prop; (4) 10 μM NE + 10 μM Prop (NE + P); (5) 10 μM Ly2157299 (Ly); and (6) 10 μM NE + 10 μM Ly2157299 (NE + Ly). Propranolol and Ly2157299 were added in the culture media 1 h before adding norepinephrine.
Cell morphological observation
The cells treated or untreated with NE (culture media containing 10 μM NE without serum) were observed and photographed under optical microscope (magnification 100× and 200×).
To further observe the extracellular microvilli or pseudopodium, scanning electron microscope was used. Briefly, the cells were washed with PBS (phosphate-buffered saline) and fixed in 1 % glutaraldehyde in 0.1 M PBS (pH 7.4) for 2 h. Washed again in PBS, the cells were progressively dehydrated in ethanol and dried in acetonitrile solution. The cells were sprayed with gold and then were observed under a scanning electron microscope.
Immunofluorescence staining
The cells of six treated groups were fixed in 4 % formaldehyde for 30 min, permeabilized with 0.2 % Triton X-100 solution for 15 min, and then blocked with 2 % BSA for 30 min. Thereafter, cells were incubated overnight with rabbit anti-human E-cadherin (1:100) and vimentin (1:100) at 4 °C. The next day, after washing in PBS, cells were incubated with CY3-labeled (1:400) and FITC-labeled (1:400) goat anti-rabbit antibodies for 2 h at 37 °C, respectively. Washed again in PBS, DAPI was used to counterstain cell nuclei. The cells were examined under a confocal laser scanning microscope (magnification 200×).
Measurement of cAMP
The intracellular cAMP concentration of the cells in the six groups was measured by cAMP immunoassay kit according to the manufacturer’s instructions. Briefly, cells were washed three times in cold PBS and resuspended in its cell lysis buffer. The mixture was centrifuged at 600×g for 10 min at 2–8 °C to remove cellular debris. The supernatant was immediately added in the 96-well polystyrene microplate and was treated with reagents following the instructional steps. The optical density of each well was determined using a microplate reader (PerkinElmer, USA) set to 450 nm. Each sample was tested at least in triplicate.
ELISA
HT-29 and A549 cells were treated according to the 6 groups described above (serum-free media used as before) for 72 and 48 h, respectively. The concentration of TGF-β1 in the culture supernatant was tested using human TGF-β1 ELISA kit according to the manufacturer’s protocol. The 450-nm absorbance was read with a luminescence plate reader (PerkinElmer, USA). From the standard curve, the concentrations of each group were calculated. Each sample was detected at least in triplicate.
Real-time quantitative PCR (RT-PCR)
Total RNA from the cells in the six groups was isolated using TRIzol reagent following the RNA extraction protocols. RT-PCR was performed with One Step SYBR® PrimeScript™ RT-PCR Kit and CFX 96™ Real-Time System in C1000™ Thermal Cycler (Bio-Rad, USA). GAPDH was used as an internal positive control. The sequences of the primers were as follows:
E-cadherin sense 5′-AGCCTCAGGTCATAAACATCATTG-3′,
anti-sense 5′-GATAGATTCTTGGGTTGGGTCG-3′;
Vimentin sense 5′-ATCTGGATTCACTCCCTCTGGTT-3′,
anti-sense 5′-GTGATGCTGAGAAGTTTCGTTGATA-3′;
TGF-βRI sense 5′-GCTGTGAAGCCTTGAGAGTAATGG-3′,
anti-sense 5′-TTCCTGTTGACTGAGTTGCGATAA-3′;
HIF-1α sense 5′-ACTGCACAGGCCACATTCACG-3′,
anti-sense 5′-GGTTCACAAATCAGCACCAAGC-3′;
Smad3 sense 5′-ATGAACCACAGCATGGACGC-3′,
anti-sense 5′-AGGAGATGGAGCACCAGAAGG-3′;
Snail sense 5′-GTCTCTTCCTTGGAGGCCGA-3′,
anti-sense 5′-AGGCTGAGGTATTCCTTGTTGC-3′;
GAPDH sense 5′-CTTTGGTATCGTGGAAGGACTC-3′,
anti-sense 5′-GTAGAGGCAGGGATGATGTTCT-3′.
The RT-PCR program was performed according to the manufacturer’s protocol. Each sample was run at least in triplicate, and the mean C t was used in the ΔC t equation. The relative mRNA expression of interested gene was calculated using the following formula: relative expression = , ΔC t = (C t gene of interest − C t GAPDH).
Western blot analysis
The cells in the six groups were washed with PBS, incubated on ice for 30 min in RIPA buffer (150 mM NaCl, 50 mM Tris base, 1 % Triton X-100, 0.1 % SDS, 1 mM sodium orthovanadate, 10 mM sodium fluoride, 0.5 % sodium deoxycholate, 1 % protease inhibitor cocktail, and 1 % phosphatase inhibitor cocktail) and centrifuged to remove debris. Total proteins of 10–30 μg were resuspended in SDS-PAGE loading buffer, separated on polyacrylamide gel, and electro-transferred into nitrocellulose membranes (Millipore, USA). After being blocked with 5 % non-fat milk in TBST (Tris-buffered saline containing 0.1 % Tween 20) for 1 h at room temperature, the membranes were incubated at 4 °C overnight with primary antibodies in appropriate dilution, followed by extensive washing with TBST. Then, the membranes were incubated with HRP-conjugated secondary antibody (1:3000–1:5000) for 2 h at room temperature. Washing again with TBST, the immunoreactive bands were detected using Chemiluminescent HRP Substrate (Millipore, USA) and analyzed by Image-Pro Plus 6.0.
Cell migration and invasion assay
The transwell migration assay was described as previous (Liu et al. 2014a). Briefly, the cells were serum-starved overnight and suspended in serum-free medium (2.5 × 106/mL HT-29 cells and 5 × 105/mL A549 cells). A total of 500 μL medium containing 10 % fetal bovine serum was added in the lower chambers as a chemoattractant. For upper wells, 100 μL cell suspension (2.5 × 105 HT-29 cells and 5 × 104 A549 cells) was added in each chamber and was treated with NE, Prop, or Ly2157299, as previously described in six groups. After incubation for 12 h at 37 °C in a humidified tissue culture incubator, the remained cells were carefully removed from the upper surface of the separating membranes using a cotton swab. The separating membranes were fixed with 4 % paraformaldehyde for 30 min and were stained with 0.1 % crystal violet (Beyotime, China) for 20 min. The cell migration was examined and evaluated by counting five random views under the microscope at 200× magnification.
For the Matrigel invasion assay, 100 μL of 1.0 mg/mL Matrigel was coated on the upper chamber of the transwell beforehand. Then, serum-starved 5 × 105/100 μL HT-29 cells and 1 × 105/100 μL A549 cells were seeded in each upper chamber and treated with NE, Prop, or Ly2157299. The lower chambers were added in 500 μL medium containing 10 % fetal bovine serum. The cells were incubated for 24 h at 37 °C in a humidified tissue culture incubator. The remaining procedure is described as transwell migration assay.
MTT assay
The MTT assay was used to estimate the viability of the cells after Prop or Ly treatment. HT-29 and A549 cells were suspended in complete culture media and seeded in 96-well plates at a density of 5 × 103/200 μL/well. After overnight incubation, the cells were serum-starved for 12 h and then the culture supernatant was replaced by serum-free media containing 10 μM Prop or 10 μM Ly or not. The cells were incubated at 37 °C in humidified 5 % CO2 for 24 h. Subsequently, 20 μL of 5 mg/mL MTT was added to each well and the plates were incubated for 4 h at 37 °C. Then, the supernate was carefully discarded and 150 μL DMSO was added to each well. Finally, the light absorbance was measured in a microplate reader (PerkinElmer, USA) at 490 nm.
Statistical analysis
Data from all experiments are expressed as mean ± standard deviation (SD). Differences between groups were assessed using Student’s t test and one-way ANOVA. All P values were two-sided, and P < 0.05 was considered statistically significant. All the statistical analyses were performed using SPSS 18.0 software.
Results
Norepinephrine induced cell morphological changes
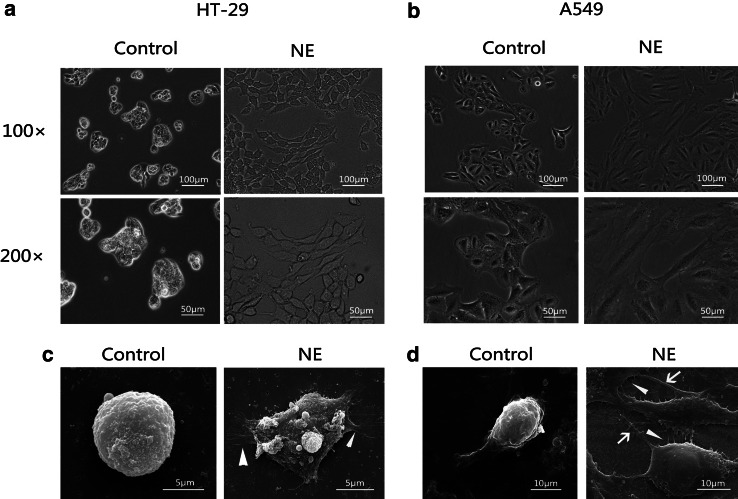
NE has been implicated in gastric, ovarian, and prostate cancer cells EMT; we questioned whether NE could induce EMT in other cells. After being exposed to NE for 4 days, the morphological changes of HT-29 and A549 cells were observed. Under the optical microscope, the cells lost intercellular contacts and showed a spindle-shaped, fibroblast-like phenotype which is consistent with a mesenchymal phenotype (Fig. 1a, b). However, the untreated cells maintained the classic epithelial morphology. The figures of scanning electron microscope showed that microvilli and pseudopodium increased in some NE-treated cells (Fig. 1c, d).
Fig. 1.
Norepinephrine induced EMT morphological alterations in HT-29 and A549 cells. Cells were cultured in serum-free medium (control) and serum-free medium containing 10 μM NE (NE) for 4 days. Cellular morphology was observed using optical microscope (×100 and ×200) and scanning electron microscope (×5000–10,000). Cells treated with 10 μM NE showed a spindle-shaped, fibroblast-like phenotype (a, b), increased microvilli (c, d white triangle), and pseudopodium (d white arrows), while the control cells maintained the classic epithelial morphology
Norepinephrine increased cancer cells migration and invasion which was blocked by propranolol or Ly2157299
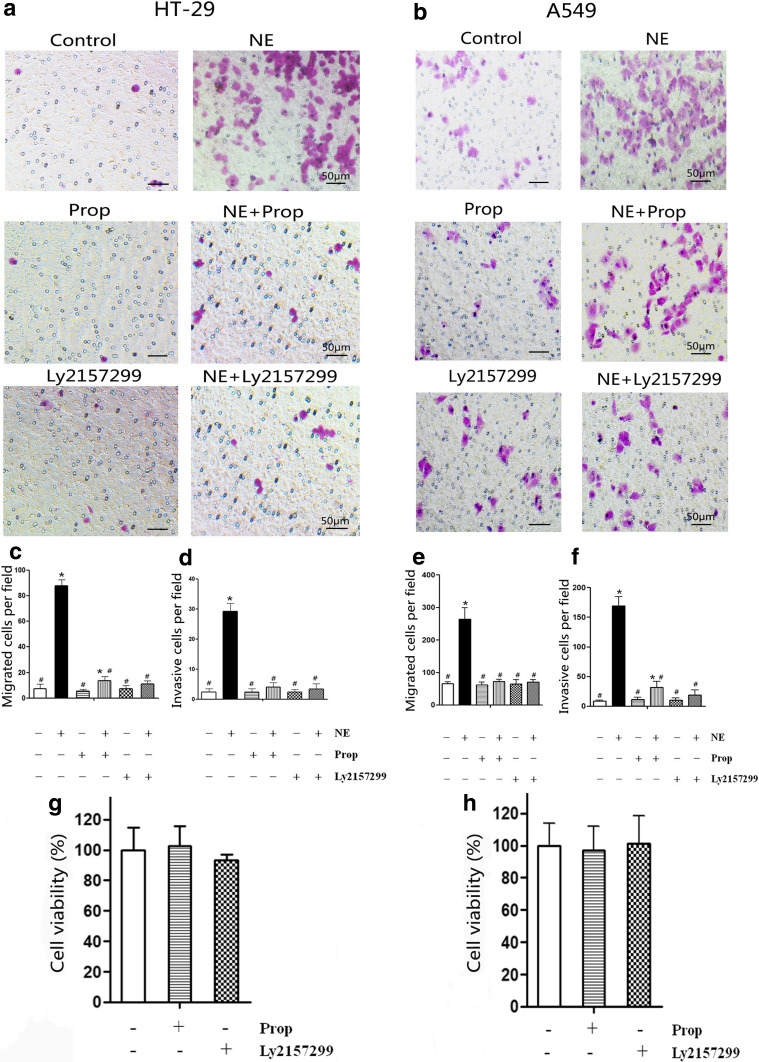
EMT process is a key step in the migratory or invasive cascade. It was demonstrated that NE could promote cancer cells migration and invasion. To testify this effect and further explore whether Prop or Ly2157299 could inhibit this effect, the transwell migration assay and the Matrigel invasion assay were used (Fig. 2). In terms of migration (Fig. 2a–c, e), a significant difference was observed between the NE-treated and control groups. Both Prop and Ly2157299 were able to decrease the migratory numbers of NE-treated cells, while they did not affected cells viability (Fig. 2g, h). However, the number of HT-29 cells in the NE + Prop group was still more than in the control group, suggesting that Prop partially inhibited the migration effect by NE. It was also found that NE could induce cancer cells invasion (Fig. 2d, f), and this effect could be inhibited by Prop or Ly2157299. However, similar to HT-29 cells, the invasive number of A549 cells in the NE + Prop group was more than in the control group. Prop seemed to partially inhibit the NE-induced cancer cells invasion.
Fig. 2.
Propranolol and Ly2157299 attenuated norepinephrine-stimulated cancer cells migration and invasion. Serum-starved HT-29 (a, c, d) and A549 (b, e, f) cells were treated with 10 μM NE, 10 μM Prop, or 10 μM Ly2157299 as the figure labeled. Representative photographs are shown as cells that migrated to the lower membrane through filter pores (a, b ×200). The number of migratory (c, e) or invasive cells (d, f) was counted in five random views and is presented in columns as mean ± SD. NE increased cells migration and invasion. Prop and Ly2157299 blocked NE effect (c–f), and they not affected cells viability (g, h). *P < 0.05 versus control group; # P < 0.05 versus NE group
Norepinephrine regulated EMT markers expressions in HT-29 and A549 cells
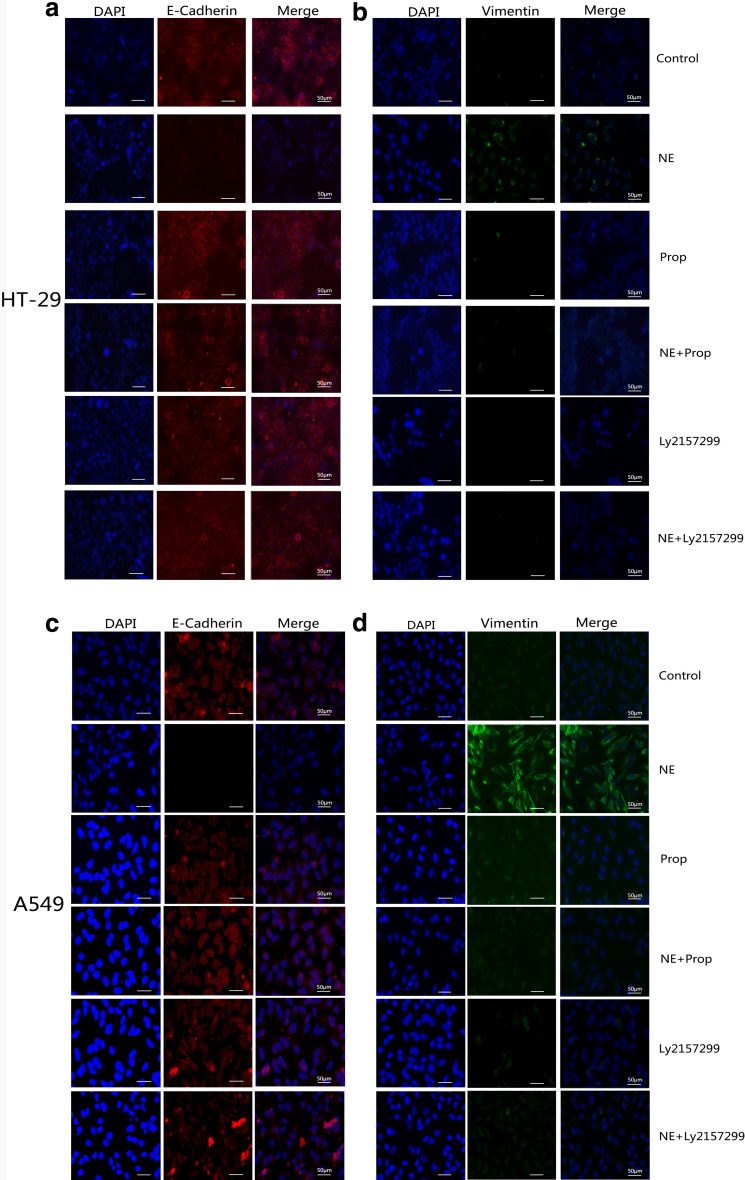
To testify the EMT phenomenon, EMT markers such as E-cadherin and vimentin were confirmed from confocal laser scanning microscope, mRNA and protein expressions. As shown in HT-29 and A549 cells immunofluorescence staining (Fig. 3), E-cadherin fluorescence signal (red) in the NE-treated group was weaker than in the control group, while vimentin fluorescence signal (green) was increased. However, both Prop and Ly2157299 could inhibit this phenomenon.
Fig. 3.
Immunofluorescence staining of E-cadherin and vimentin in HT-29 and A549 cells. HT-29 (a, b) and A549 (c, d) cells were treated with 10 μM NE, 10 μM Prop, or 10 μM Ly2157299 for 4 days as the figure labeled. After then, cells were incubated with rabbit anti-human E-cadherin and vimentin and then observed under confocal laser scanning microscope. DAPI was used to counterstain cell nuclei. In the NE-treated group, E-cadherin fluorescence signal (a, c red) was weaker than in other groups, while vimentin fluorescence signal (b, d green) was stronger compared with others
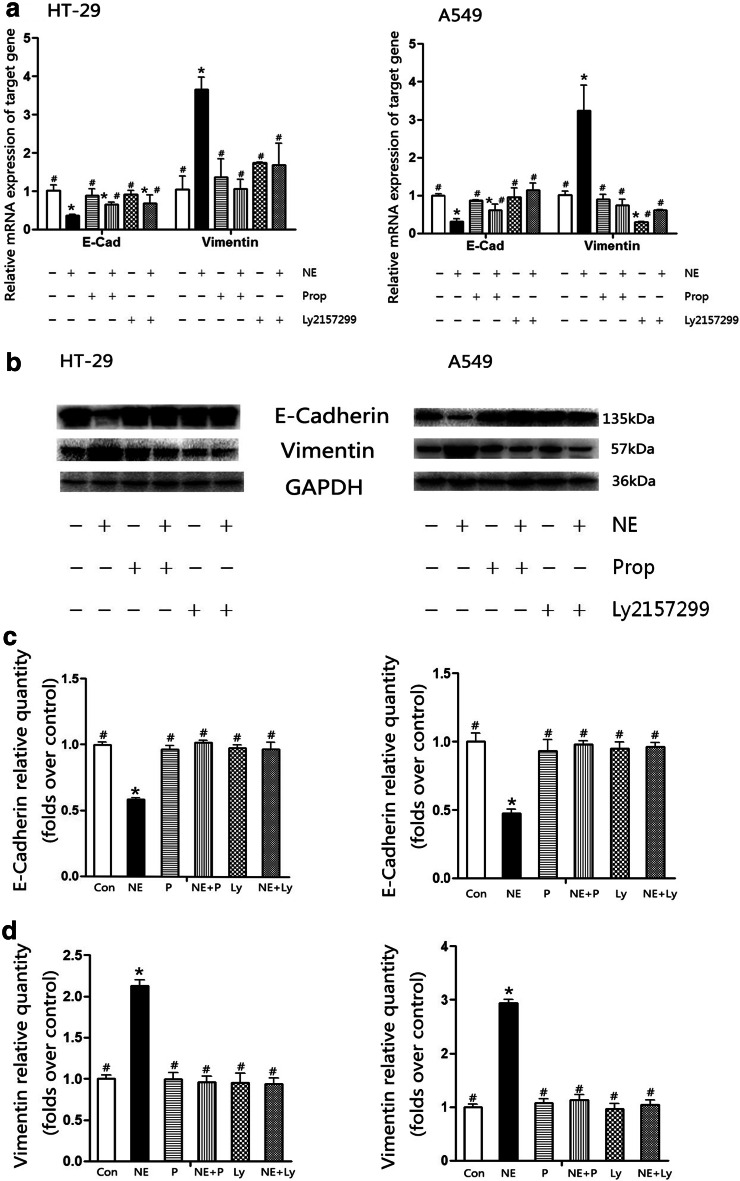
To further confirm the ability of NE-induced EMT markers and the potential block ability of Prop or Ly2157299, the mRNA and protein of E-cadherin and vimentin were detected (Fig. 4). The results of RT-PCR and western blot indicated that NE repressed E-cadherin mRNA and protein expressions, but increased vimentin mRNA and protein expressions, both in HT-29 and in A549 cells. Prop and Ly2157299 were able to block the effect of NE. However, the mRNA expressions of E-cadherin in the NE + Prop groups (HT-29 and A549) were still lower than in the controls. For HT-29 cell, even in the NE + Ly group, the mRNA expression of E-cadherin was lower than in controls.
Fig. 4.
Propranolol and Ly2157299 attenuated norepinephrine-induced EMT makers’ changes in HT-29 and A549 cells. After 4-day incubation with 10 μM NE, 10 μM Prop, or 10 μM Ly2157299, mRNA (a) and protein (b) expressions of E-cadherin and vimentin in HT-29 and A549 cells were examined by RT-PCR and western blot, respectively. Quantification of E-cadherin (c) and vimentin (d) protein is presented in columns as mean ± SD. NE decreased E-cadherin expression and increased vimentin expression, while Prop and Ly2157299 reversed this effect. *P < 0.05 versus control group; # P < 0.05 versus NE group
Norepinephrine increased intracellular cAMP and induced TGF-β1 elevation in culture supernatant
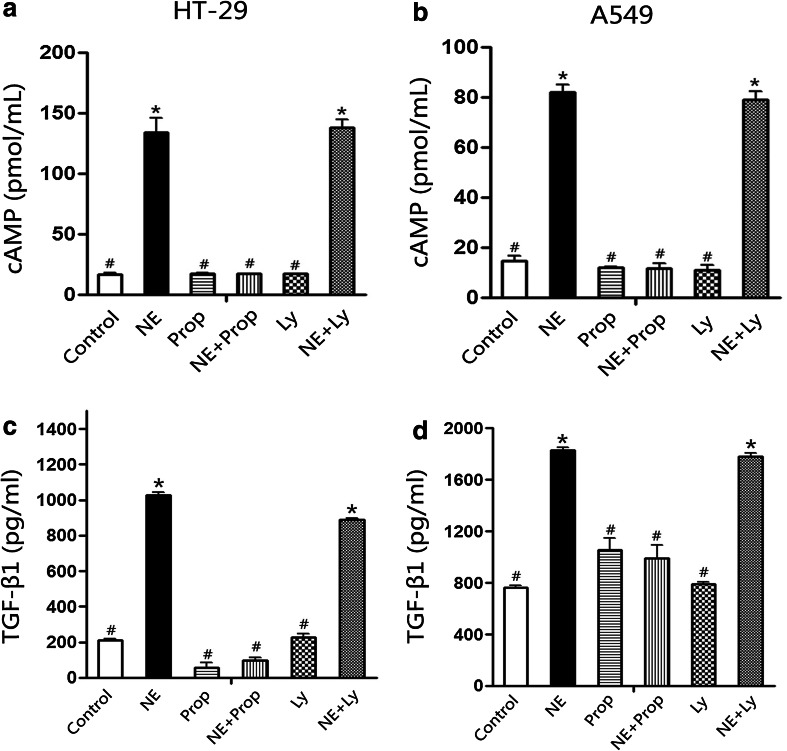
The activation of β-AR is associated with the synthesis of cAMP. After NE treatment, the intracellular cAMP of HT-29 and A549 cells was increased, while Prop completely suppressed NE-induced cAMP synthesis. However, Ly2157299 could not induce cAMP elevation and could not inhibit NE-induced cAMP synthesis neither (Fig. 5a, b). This result might stand for the β-AR activation of NE and the blocking effect of Prop.
Fig. 5.
Norepinephrine stimulated cAMP synthesis and TGF-β1 secretion. Intracellular cAMP and TGF-β1 in culture supernatant were determined. Data are presented in columns as mean ± SD. NE triggered HT-29 (a) and A549 (b) cells cAMP synthesis. TGF-β1 in supernatant from HT-29 (c) and A549 cells (d) was increased after incubating with NE for 72 and 48 h, respectively. β-Adrenoceptor antagonist Prop could inhibit this effect, but Ly2157299 could not. *P < 0.05 versus control group; # P < 0.05 versus NE group
Studies have found that TGF-β1 played a critical role in EMT induction. To understand whether TGF-β1 is involved in the process of NE-induced EMT, the culture supernatant was tested using ELISA. As shown in Fig. 5c, d, it was found that TGF-β1 was significantly increased in HT-29 cells and A549 cells after 10 μM NE treatment for 72 h and 48 h, respectively. Prop could inhibit the effect of NE, but Ly2157299 could not. TGF-β1 remained at relatively low level when cells were treated with 10 μM Prop or 10 μM Ly2157299 1 h before NE treatment. It indicated that the augmentation of TGF-β1 was regulated by β-AR.
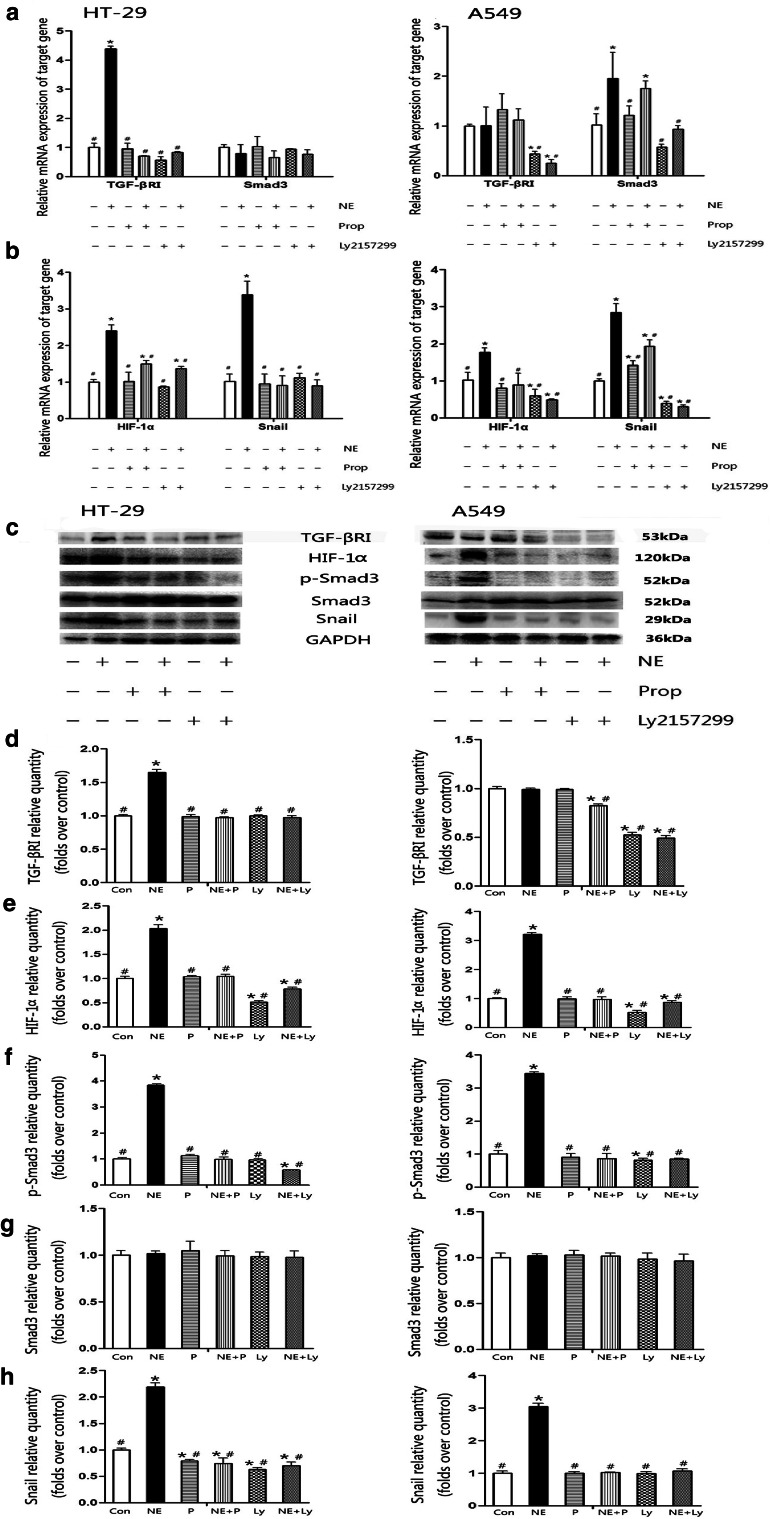
We further detected the mRNA and protein expression of TGF-βRI. After exposing to NE for 4 days, the mRNA and protein expressions of TGF-βRI were increased in HT-29 cells while staying in baseline level in A549 cells. Details are shown in Fig. 6a, c, d.
Fig. 6.
Propranolol and Ly2157299 blocked norepinephrine-induced HIF-1α, p-Smad3, and Snail expressions. NE induced TGF-βRI mRNA and protein expression in HT-29 cells (a, c, d) and Smad3 mRNA expression in A549 cells (a). NE stimulated transcripts and protein expressions of HIF-1α (b, c, e) and Snail (b, c, h) expressions, and increased p-Smad3 protein expressions (c, f) in HT-29 and A549 cells. Prop and Ly2157299 could block NE effect. Quantification of proteins is presented in columns as mean ± SD (d–g). *P < 0.05 versus control group; # P < 0.05 versus NE group
β-AR/TGF-β1 signaling/HIF-1α/Snail and β-AR/TGF-β1 signaling/p-Smad3/Snail signaling pathways were required for NE-induced EMT
It was reported that β-AR/HIF-1α/Snail signaling was required for NE-mediated EMT and p-Smad3 (phosphorylated Smad3)/Snail signaling was involved in TGF-β1-induced EMT. Moreover, HIF-1α and p-Smad3 were both demonstrated to be the downstream signal of TGF-β1. Therefore, more and detailed mechanisms seem to be related to NE-induced EMT. Our results showed that NE promoted transcription and protein expressions of HIF-1α and Snail expressions and increased p-Smad3 protein expressions (Fig. 6). The mRNA expression of Smad3 was upregulated in A549 cells, but remained at baseline level in HT-29 cells. However, protein expressions did not exhibit difference.
It was found that β-AR antagonist Prop and TGF-βRI inhibition Ly2157299 could also reverse NE-induced HIF-1α, p-Smad3, and Snail expressions. The results showed the significant roles of β-AR/TGF-β1 signaling/HIF-1α/Snail and β-AR/TGF-β1 signaling/p-Smad3/Snail in NE-induced EMT.
Discussion
Accumulating data demonstrate that EMT is a key step throughout epithelial cells acquiring malignant phenotype (Brabletz et al. 2005; Thiery et al. 2009). During EMT, various changes occur in cellular morphology, architecture, adhesion, migration, and invasion (Xie et al. 2010). In this study, NE-treated HT-29 and A549 cells showed decreased intercellular adhesion and spindle-shaped, fibroblast-like phenotype under the optical microscope. Meanwhile, increased microvilli and pseudopodium were observed under the scanning electron microscope. These changes in morphology were similar to previous reports (Shan et al. 2014; Shankar et al. 2010; Jia et al. 2014). Next, we found that NE significantly decreased the expression of epithelial marker (E-cadherin) and increased the expression of mesenchymal marker (vimentin) and EMT marker (Snail) in the meantime, consistent with previous studies as well (Shan et al. 2014; Barbieri et al. 2015). Moreover, our previous work (Liu et al. 2014a) and the present results suggested that the ability of migration and invasion was remarkably enhanced with NE treatment in cancer cells. The results described above indicated that NE induced EMT in HT-29 and A549 cells.
It was reported that stress hormones, such as glucocorticoid and catecholamine, could stimulate the secretion of TGF-β1 (Elenkov and Chrousos 2002). As shown in Fig. 5, the level of TGF-β1 in supernatant was increased after HT-29 and A549 cells being treated with NE. However, propranolol, the inhibitor of β-AR, could block this effect. It meant that NE promoted the secretion of TGF-β1 through β-AR. We also found that Ly2157299 reversed the NE-induced HT-29 and A549 cells EMT. Similar results have been shown in another study, which found that the use of Ly2157299 to block TGF-β1 signaling appears to reduce EMT in hepatocellular carcinoma (Giannelli et al. 2014). We subsequently speculated that TGF-β1 signaling was a key pathway during NE-induced EMT and it was extremely likely to be the downstream signal of β-AR.
Qian et al. (2004) and Yang et al. (2006b) reported that TGF-β1, as the upstream signal of HIF-1α, has the ability to increase the expression of HIF-1α, which further mediates VEGF secretion. We found that NE could also induce the expression of HIF-1α, while propranolol or Ly2157299 blocked the induction, indicating that HIF-1α was the downstream signal of TGF-β1 signaling. On the basis of previous research which β2-AR/HIF-1α/Snail signaling pathway was involved in NE-induced EMT, we supplemented the signaling pathway to β-AR/TGF-β1 signaling/HIF-1α/Snail. Meanwhile, TGF-β1/p-Smad3/Snail was proved to be another signaling pathway participating in TGF-β1-induced EMT. In present study, NE stimulated the expression of p-Smad3 and this stimulation could be blocked by Prop or Ly2157299. It suggested that β-AR/TGF-β1 signaling/p-Smad3/Snail might be another signaling pathway involved in NE-induced EMT.
Referring to previous reports, after being exposed to NE for 24 h, ovarian cancer cells would exhibit EMT phenomenon, while the gastric adenocarcinoma cell lines needed 48 h. The sensitivity might be different in various cancer cells. We found that the morphological alteration was not obvious in HT-29 and A549 cells until they were exposed to NE for 4 days. When probed about the changes of NE-induced TGF-β1, there was a time discrepancy between HT-29 and A549 cells as regards NE treatment (HT-29 for 72 h while A549 for 48 h). Although TGF-β1 exhibited an elevated tendency in HT-29 cells supernatant at 48 h, there was no statistical significance (data not shown). In addition, NE increased the expression of TGF-βRI in HT-29 cells, whereas we did not observe similar results in A549 cells. All of these results indicated that various cancer cells might exhibit different responses to NE treatment because of the cell specificity.
Some studies reported that IL-6 and IL-8 were able to induce cancer cells EMT (Yadav et al. 2011; Fernando et al. 2011). Furthermore, the effect of TGF-β1-induced EMT was enhanced by IL-6 via JAK/STAT3/Snail signaling pathway (Liu et al. 2014b; Yadav et al. 2011). According to our previous reports, NE could upregulate IL-6 and IL-8 protein levels in culture supernatant of cancer cells (Liu et al. 2014a; Deng et al. 2014a). IL-6 and IL-8 seemed to be involved in NE-induced cancer cells EMT. Therefore, we believed that other potential mechanisms might be involved in this process. Moreover, what is the connection or crosstalk between p-Smad3 and HIF-1α? More detailed studies need to be further explored.
Taken together, our results supported the hypothesis that NE could induce cancer cells EMT, and indicated the significant role of TGF-β1 signaling. We further discussed two potential signaling pathways (β-AR/TGF-β1 signaling/HIF-1α/Snail and β-AR/TGF-β1 signaling/p-Smad3/Snail) involved in this process.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (81172202). We thank Lei Deng, M.D. and Mr Di Luo for the English editing.
Compliance with ethical standards
Conflict of interest
We declare that no conflict of interest exists in this manuscript.
Informed consent and ethical standard
There is not any informed consent or any ethical issue involved in this study.
Footnotes
Jie Zhang and Yao-tiao Deng have contributed equally to this work.
References
- Akhurst RJ, Derynck R (2001) TGF-β signaling in cancer—a double-edged sword. Trends Cell Biol 11(11):S44–S51. doi:10.1016/S0962-8924(01)02130-4 [DOI] [PubMed] [Google Scholar]
- Barbieri A, Bimonte S, Palma G, Luciano A, Rea D, Giudice A, Scognamiglio G, La Mantia E, Franco R, Perdona S, De Cobelli O, Ferro M, Zappavigna S, Stiuso P, Caraglia M, Arra C (2015) The stress hormone norepinephrine increases migration of prostate cancer cells in vitro and in vivo. Int J Oncol. doi:10.3892/ijo.2015.3038 [DOI] [PubMed] [Google Scholar]
- Barron TI, Connolly RM, Sharp L, Bennett K, Visvanathan K (2011) Beta blockers and breast cancer mortality: a population-based study. J Clin Oncol 29(19):2635–2644. doi:10.1200/JCO.2010.33.5422 [DOI] [PubMed] [Google Scholar]
- Brabletz T, Hlubek F, Spaderna S, Schmalhofer O, Hiendlmeyer E, Jung A, Kirchner T (2005) Invasion and metastasis in colorectal cancer: epithelial–mesenchymal transition, mesenchymal–epithelial transition, stem cells and β-catenin. Cells Tissues Organs 179(1–2):56–65. doi:10.1159/000084509 [DOI] [PubMed] [Google Scholar]
- Calon A, Espinet E, Palomo-Ponce S, Tauriello DV, Iglesias M, Cespedes MV, Sevillano M, Nadal C, Jung P, Zhang XH, Byrom D, Riera A, Rossell D, Mangues R, Massague J, Sancho E, Batlle E (2012) Dependency of colorectal cancer on a TGF-β-driven program in stromal cells for metastasis initiation. Cancer Cell 22(5):571–584. doi:10.1016/j.ccr.2012.08.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi MJ, Cho KH, Lee S, Bae YJ, Jeong KJ, Rha SY, Choi EJ, Park JH, Kim JM, Lee JS, Mills GB, Lee HY (2014) hTERT mediates norepinephrine-induced Slug expression and ovarian cancer aggressiveness. Oncogene. doi:10.1038/onc.2014.270 [DOI] [PubMed] [Google Scholar]
- de Jong JS, van Diest PJ, van der Valk P, Baak JP (1998) Expression of growth factors, growth-inhibiting factors, and their receptors in invasive breast cancer. II: correlations with proliferation and angiogenesis. J Pathol 184(1):53–57. doi:10.1002/(SICI)1096-9896(199801)184:1%3c53:AID-PATH6%3e3.0.CO;2-7 [DOI] [PubMed] [Google Scholar]
- Deng GH, Liu J, Zhang J, Wang Y, Peng XC, Wei YQ, Jiang Y (2014a) Exogenous norepinephrine attenuates the efficacy of sunitinib in a mouse cancer model. J Exp Clin Cancer Res 33:21. doi:10.1186/1756-9966-33-21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng YT, Zhong WN, Jiang Y (2014b) Measurement of distress and its alteration during treatment in patients with nasopharyngeal carcinoma. Head Neck 36(8):1077–1086. doi:10.1002/hed.23412 [DOI] [PubMed] [Google Scholar]
- Elenkov IJ, Chrousos GP (2002) Stress hormones, proinflammatory and antiinflammatory cytokines, and autoimmunity. Ann N Y Acad Sci 966:290–303 [DOI] [PubMed] [Google Scholar]
- Entschladen F, Drell TL, Lang K, Joseph J, Zaenker KS (2004) Tumour-cell migration, invasion, and metastasis: navigation by neurotransmitters. Lancet Oncol 5(4):254–258. doi:10.1016/S1470-2045(04)01431-7S1470204504014317 [DOI] [PubMed] [Google Scholar]
- Fernando RI, Castillo MD, Litzinger M, Hamilton DH, Palena C (2011) IL-8 signaling plays a critical role in the epithelial–mesenchymal transition of human carcinoma cells. Cancer Res 71(15):5296–5306. doi:10.1158/0008-5472.CAN-11-0156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giannelli G, Villa E, Lahn M (2014) Transforming growth factor-β as a therapeutic target in hepatocellular carcinoma. Cancer Res 74(7):1890–1894. doi:10.1158/0008-5472.CAN-14-0243 [DOI] [PubMed] [Google Scholar]
- Glaser R, Kiecolt-Glaser JK (2005) Stress-induced immune dysfunction: implications for health. Nat Rev Immunol 5(3):243–251. doi:10.1038/nri1571 [DOI] [PubMed] [Google Scholar]
- Hasina R, Matsumoto K, Matsumoto-Taniura N, Kato I, Sakuda M, Nakamura T (1999) Autocrine and paracrine motility factors and their involvement in invasiveness in a human oral carcinoma cell line. Br J Cancer 80(11):1708–1717. doi:10.1038/sj.bjc.6690587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hassan S, Karpova Y, Baiz D, Yancey D, Pullikuth A, Flores A, Register T, Cline JM, D’Agostino R Jr, Danial N, Datta SR, Kulik G (2013) Behavioral stress accelerates prostate cancer development in mice. J Clin Investig 123(2):874–886. doi:10.1172/JCI63324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia L, Jin H, Zhou J, Chen L, Lu Y, Ming Y, Yu Y (2013) A potential anti-tumor herbal medicine, Corilagin, inhibits ovarian cancer cell growth through blocking the TGF-β signaling pathways. BMC Complement Altern Med 13:33. doi:10.1186/1472-6882-13-33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia Y, Wu D, Yun F, Shi L, Luo N, Liu Z, Shi Y, Sun Q, Jiang L, Wang S, Du M (2014) Transforming growth factor-β1 regulates epithelial–mesenchymal transition in association with cancer stem-like cells in a breast cancer cell line. Int J Clin Exp Med 7(4):865–872 [PMC free article] [PubMed] [Google Scholar]
- Kiecolt-Glaser JK, Loving TJ, Stowell JR, Malarkey WB, Lemeshow S, Dickinson SL, Glaser R (2005) Hostile marital interactions, proinflammatory cytokine production, and wound healing. Arch Gen Psychiatry 62(12):1377–1384. doi:10.1001/archpsyc.62.12.1377 [DOI] [PubMed] [Google Scholar]
- Liu J, Deng GH, Zhang J, Wang Y, Xia XY, Luo XM, Deng YT, He SS, Mao YY, Peng XC, Wei YQ, Jiang Y (2014a) The effect of chronic stress on anti-angiogenesis of sunitinib in colorectal cancer models. Psychoneuroendocrinology 52C:130–142. doi:10.1016/j.psyneuen.2014.11.008 [DOI] [PubMed] [Google Scholar]
- Liu RY, Zeng Y, Lei Z, Wang L, Yang H, Liu Z, Zhao J, Zhang HT (2014b) JAK/STAT3 signaling is required for TGF-β-induced epithelial–mesenchymal transition in lung cancer cells. Int J Oncol 44(5):1643–1651. doi:10.3892/ijo.2014.2310 [DOI] [PubMed] [Google Scholar]
- Massague J, Chen YG (2000) Controlling TGF-β signaling. Genes Dev 14(6):627–644 [PubMed] [Google Scholar]
- Melhem-Bertrandt A, Chavez-Macgregor M, Lei X, Brown EN, Lee RT, Meric-Bernstam F, Sood AK, Conzen SD, Hortobagyi GN, Gonzalez-Angulo AM (2011) Beta-blocker use is associated with improved relapse-free survival in patients with triple-negative breast cancer. J Clin Oncol 29(19):2645–2652. doi:10.1200/JCO.2010.33.4441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moreno-Bueno G, Peinado H, Molina P, Olmeda D, Cubillo E, Santos V, Palacios J, Portillo F, Cano A (2009) The morphological and molecular features of the epithelial-to-mesenchymal transition. Nat Protoc 4(11):1591–1613. doi:10.1038/nprot.2009.152 [DOI] [PubMed] [Google Scholar]
- Neuzillet C, Tijeras-Raballand A, Cohen R, Cros J, Faivre S, Raymond E, de Gramont A (2015) Targeting the TGFβ pathway for cancer therapy. Pharmacol Ther 147C:22–31. doi:10.1016/j.pharmthera.2014.11.001 [DOI] [PubMed] [Google Scholar]
- Qian D, Lin HY, Wang HM, Zhang X, Liu DL, Li QL, Zhu C (2004) Involvement of ERK1/2 pathway in TGF-β1-induced VEGF secretion in normal human cytotrophoblast cells. Mol Reprod Dev 68(2):198–204. doi:10.1002/mrd.20061 [DOI] [PubMed] [Google Scholar]
- Reiche EM, Nunes SO, Morimoto HK (2004) Stress, depression, the immune system, and cancer. Lancet Oncol 5(10):617–625. doi:10.1016/S1470-2045(04)01597-9 [DOI] [PubMed] [Google Scholar]
- Shan T, Cui X, Li W, Lin W, Li Y, Chen X, Wu T (2014) Novel regulatory program for norepinephrine-induced epithelial–mesenchymal transition in gastric adenocarcinoma cell lines. Cancer Sci 105(7):847–856. doi:10.1111/cas.12438 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shankar J, Messenberg A, Chan J, Underhill TM, Foster LJ, Nabi IR (2010) Pseudopodial actin dynamics control epithelial–mesenchymal transition in metastatic cancer cells. Cancer Res 70(9):3780–3790. doi:10.1158/0008-5472.CAN-09-4439 [DOI] [PubMed] [Google Scholar]
- Sigala B, McKee C, Soeda J, Pazienza V, Morgan M, Lin CI, Selden C, Vander Borght S, Mazzoccoli G, Roskams T, Vinciguerra M, Oben JA (2013) Sympathetic nervous system catecholamines and neuropeptide Y neurotransmitters are upregulated in human NAFLD and modulate the fibrogenic function of hepatic stellate cells. PLoS ONE 8(9):e72928. doi:10.1371/journal.pone.0072928PONE-D-13-20898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sloan EK, Priceman SJ, Cox BF, Yu S, Pimentel MA, Tangkanangnukul V, Arevalo JM, Morizono K, Karanikolas BD, Wu L, Sood AK, Cole SW (2010) The sympathetic nervous system induces a metastatic switch in primary breast cancer. Cancer Res 70(18):7042–7052. doi:10.1158/0008-5472.CAN-10-0522 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiegel D (2012) Mind matters in cancer survival. Psychooncology 21(6):588–593. doi:10.1002/pon.3067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thaker PH, Han LY, Kamat AA, Arevalo JM, Takahashi R, Lu C, Jennings NB, Armaiz-Pena G, Bankson JA, Ravoori M, Merritt WM, Lin YG, Mangala LS, Kim TJ, Coleman RL, Landen CN, Li Y, Felix E, Sanguino AM, Newman RA, Lloyd M, Gershenson DM, Kundra V, Lopez-Berestein G, Lutgendorf SK, Cole SW, Sood AK (2006) Chronic stress promotes tumor growth and angiogenesis in a mouse model of ovarian carcinoma. Nat Med 12(8):939–944. doi:10.1038/nm1447 [DOI] [PubMed] [Google Scholar]
- Thiery JP (2002) Epithelial–mesenchymal transitions in tumour progression. Nat Rev Cancer 2(6):442–454. doi:10.1038/nrc822 [DOI] [PubMed] [Google Scholar]
- Thiery JP, Acloque H, Huang RY, Nieto MA (2009) Epithelial–mesenchymal transitions in development and disease. Cell 139(5):871–890. doi:10.1016/j.cell.2009.11.007 [DOI] [PubMed] [Google Scholar]
- Thornton LM, Andersen BL, Blakely WP (2010) The pain, depression, and fatigue symptom cluster in advanced breast cancer: covariation with the hypothalamic-pituitary-adrenal axis and the sympathetic nervous system. Health Psychol 29(3):333–337. doi:10.1037/a0018836 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsushima H, Ito N, Tamura S, Matsuda Y, Inada M, Yabuuchi I, Imai Y, Nagashima R, Misawa H, Takeda H, Matsuzawa Y, Kawata S (2001) Circulating transforming growth factor β1 as a predictor of liver metastasis after resection in colorectal cancer. Clin Cancer Res 7(5):1258–1262 [PubMed] [Google Scholar]
- Wang Y, Zou L, Jiang M, Wei Y, Jiang Y (2013) Measurement of distress in Chinese inpatients with lymphoma. Psychooncology 22(7):1581–1586. doi:10.1002/pon.3170 [DOI] [PubMed] [Google Scholar]
- Wikstrom P, Stattin P, Franck-Lissbrant I, Damber JE, Bergh A (1998) Transforming growth factor β1 is associated with angiogenesis, metastasis, and poor clinical outcome in prostate cancer. Prostate 37(1):19–29. doi:10.1002/(SICI)1097-0045(19980915)37:1%3c19:AID-PROS4%3e3.0.CO;2-3 [DOI] [PubMed] [Google Scholar]
- Xie D, Gore C, Liu J, Pong RC, Mason R, Hao G, Long M, Kabbani W, Yu L, Zhang H, Chen H, Sun X, Boothman DA, Min W, Hsieh JT (2010) Role of DAB2IP in modulating epithelial-to-mesenchymal transition and prostate cancer metastasis. Proc Natl Acad Sci USA 107(6):2485–2490. doi:10.1073/pnas.0908133107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J, Lamouille S, Derynck R (2009) TGF-β-induced epithelial to mesenchymal transition. Cell Res 19(2):156–172. doi:10.1038/cr.2009.5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yadav A, Kumar B, Datta J, Teknos TN, Kumar P (2011) IL-6 promotes head and neck tumor metastasis by inducing epithelial–mesenchymal transition via the JAK-STAT3-SNAIL signaling pathway. Mol Cancer Res 9(12):1658–1667. doi:10.1158/1541-7786.MCR-11-0271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang EV, Sood AK, Chen M, Li Y, Eubank TD, Marsh CB, Jewell S, Flavahan NA, Morrison C, Yeh PE, Lemeshow S, Glaser R (2006a) Norepinephrine up-regulates the expression of vascular endothelial growth factor, matrix metalloproteinase (MMP)-2, and MMP-9 in nasopharyngeal carcinoma tumor cells. Cancer Res 66(21):10357–10364. doi:10.1158/0008-5472.CAN-06-2496 [DOI] [PubMed] [Google Scholar]
- Yang QC, Zeng BF, Shi ZM, Dong Y, Jiang ZM, Huang J, Lv YM, Yang CX, Liu YW (2006b) Inhibition of hypoxia-induced angiogenesis by trichostatin A via suppression of HIF-1α activity in human osteosarcoma. J Exp Clin Cancer Res 25(4):593–599 [PubMed] [Google Scholar]
- Yang EV, Kim SJ, Donovan EL, Chen M, Gross AC, Webster Marketon JI, Barsky SH, Glaser R (2009) Norepinephrine upregulates VEGF, IL-8, and IL-6 expression in human melanoma tumor cell lines: implications for stress-related enhancement of tumor progression. Brain Behav Immun 23(2):267–275. doi:10.1016/j.bbi.2008.10.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang R, Lin Q, Gao HB, Zhang P (2014) Stress-related hormone norepinephrine induces interleukin-6 expression in GES-1 cells. Braz J Med Biol Res 47(2):101–109. doi:10.1590/1414-431X20133346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zlobec I, Lugli A (2010) Epithelial mesenchymal transition and tumor budding in aggressive colorectal cancer: tumor budding as oncotarget. Oncotarget 1(7):651–661 [DOI] [PMC free article] [PubMed] [Google Scholar]