Summary
Background
This single-arm study evaluates the feasibility, safety, and preliminary effects of two senolytic agents, Dasatinib and Quercetin (DQ), in older adults at risk of Alzheimer’s disease.
Methods
Participants took 100 mg of Dasatinib and 1250 mg of Quercetin for two days every two weeks over 12 weeks. Recruitment rate, adverse events, absolute changes in functional outcomes, and percent changes in biomarkers were calculated. Spearman correlations between functional and biomarker outcomes were performed.
Findings
Approximately 10% of telephone-screened individuals completed the intervention (n = 12). There were no serious adverse events related to the intervention. Mean Montreal Cognitive Assessment (MoCA) scores non-significantly increased following DQ by 1.0 point (95% CI: −0.7, 2.7), but increased significantly by 2.0 points (95% CI: 0.1, 4.0) in those with lowest baseline MoCA scores. Mean percent change in tumour necrosis factor-alpha (TNF-α), a key product of the senescence-associated secretory phenotype (SASP), non-significantly decreased following DQ by −3.0% (95% CI: −13.0, 7.1). Changes in TNF-α were significantly and inversely correlated with changes in MoCA scores (r = −0.65, p = 0.02), such that reductions in TNF- α were correlated with increases in MoCA scores.
Interpretation
This study suggests that intermittent DQ treatment is feasible and safe; data hint at potential functional benefits in older adults at risk of Alzheimer’s disease. The observed reduction in TNF-α and its correlation with increases in MoCA scores suggests that DQ may improve cognition by modulating the SASP. However, there was not an appropriate control group. Data are preliminary and must be interpreted cautiously.
Funding
National Institute on Ageing grants R21AG073886 and R33AG061456 funded this research.
Keywords: Senolytics, Ageing, Cognitive impairment, Gait, Flavonoids, Geroscience
Research in context.
Evidence before this study
Cellular senescence is one of the hallmarks of ageing that theoretically contributes to the development of age-related diseases. Senolytic agents such as Dasatinib and Quercetin promote the elimination of senescent cells and may provide a viable strategy for the prevention or treatment of diseases of ageing.
Studies have demonstrated that co-administration of Dasatinib and Quercetin improved aspects of both physical and cognitive function in mice. However, to our knowledge, the safety, feasibility, and preliminary efficacy of Dasatinib and Quercetin to improve function in humans with mild cognitive impairment (MCI) and slow gait speed is unknown.
Added value of this study
As a pivotal step, the feasibility, safety, and preliminary efficacy of Dasatinib and Quercetin needs to be established before conducting a large-scale study to evaluate definitive efficacy for improving mobility and cognition. Thus far, only a few studies administering Dasatinib and Quercetin have been conducted in humans. One study of 14 patients with idiopathic pulmonary fibrosis showed improvements in 6-min walk distance, 4-m gait speed, and chair-stand time after 12 weeks of intermittent treatment. Another study of 5 patients with early Alzheimer’s disease treated intermittently with Dasatinib and Quercetin for 12 weeks, showed a favourable safety profile, penetrance of Dasatinib into the cerebral spinal fluid (CSF), increased levels of CSF interleukin-6 (IL-6) and glial fibrillary acidic protein (GFAP), non-significant decreases in senescence-related cytokines and chemokines, and non-significant increases in Aβ42 levels. This small study supported the safety, tolerability and feasibility of Dasatinib and Quercetin treatment in patients with Alzheimer’s disease. Our study is an evaluation of Dasatinib and Quercetin specifically in a population of older adults with slow walking speed and MCI who are at risk of developing Alzheimer’s disease. This is a particularly common group of people who often suffer from falls, frailty, dementia, and the subsequent loss of independent physical function, and for whom preventive interventions are critically needed.
The emerging field of geroscience, which has discovered fundamental mechanisms of ageing such as the accumulation of senescent cells and their toxic by-products, offers exciting promise to extend health longevity through senolytic therapies. Such therapy has been highly successful in rodent models of diseases related to ageing processes where it has improved physical function and reduced vascular stiffness, fatty liver, osteoporosis, and neurodegenerative changes in models of Alzheimer’s disease. Our study is testing the geroscience hypothesis in humans. Additionally, senolytics do not need to be present continuously to reduce senescent cell abundance because: 1) Preclinical studies have shown that Dasatinib and Quercetin appear to be as or more effective if administered intermittently as continuously1,2; 2) Dasatinib has an elimination half-life <4 h3 and Quercetin <11 h4; 3) Senescent cells are in a state of replicative arrest; 4) It generally takes cells 1 to >6 weeks to become senescent and develop a pro-inflammatory secretory phenotype5,6; and 5) A brief exposure (<3 h) of susceptible senescent cells to senolytics is sufficient to initiate irreversible apoptosis.7 Hence, intermittent dosing schedules of short-acting senolytics, as in the current study, may reduce toxic off-target effects related to continuous drug presence when, for example, Dasatinib is administered frequently for treating leukaemias or lymphomas.
Implications of all the available evidence
Our study findings suggest that administering Dasatinib and Quercetin to older adults with slow walking speed and MCI appears feasible and safe. Furthermore, the preliminary data suggest that Dasatinib and Quercetin may improve memory/cognition by modulating specific markers of senescence (i.e., tumour necrosis factor alpha). However, the sample size of the study is very small and there was not a control group for the assessment of functional outcomes. Thus, these results are preliminary and should be interpreted with caution. Future studies of larger size are needed to confirm our initial findings.
Introduction
Abnormalities in cognition and mobility are common accompaniments of ageing and predictors for the development of Alzheimer’s disease.8, 9, 10 Recent evidence suggests that the accumulation of senescent cells, which can release a variety of pro-inflammatory, apoptotic, and toxic by-products, called the senescence-associated secretory phenotype (SASP), is one of the drivers of deficits in both cognition11, 12, 13, 14, 15, 16 and mobility.7,17, 18, 19, 20, 21, 22 Therefore, targeting the process of cellular senescence may be a viable strategy to improve aspects of cognition and mobility, possibly preventing the development of Alzheimer’s disease and related disorders.
One way to target the process of cellular senescence is through the use of senolytic compounds, which are able to selectively eliminate senescent cells and their toxic SASP components. Senolytic compounds include Dasatinib, a chemotherapeutic Src/tyrosine kinase inhibitor, and Quercetin, a plant-based flavonol.13,23 In preclinical studies, co-administration of Dasatinib and Quercetin (DQ) improved aspects related to physical function (e.g., endurance and speed) and cognition (e.g., reduced neurofibrillary tangle density/neuron loss).7,15,24 Given that senolytic administration has demonstrated physical and cognitive benefits in mouse models,15,16,24, 25, 26 these effects may translate to humans.14,27,28
To date, four human studies administering DQ as a combination senolytic therapy have been completed in various populations, including idiopathic pulmonary fibrosis,29 diabetic kidney disease,30 age-related osteoporosis,31 and Alzheimer’s disease.32 Yet, no studies have evaluated the impact of DQ on individuals with age-related deficits in mobility and cognition unrelated to specific disease states. These conditions are very common precursors of frailty, dementia, and functional decline that may in part be related to the accumulation of senescent cells and their toxic by-products. Confirmation of feasibility and safety in new populations is paramount when applying an existing treatment (i.e., DQ) for a new indication before a large-scale efficacy trial can be considered. Furthermore, pilot research is essential to identify potential intermediate outcomes and biomarkers that may provide targets for future large-scale therapeutic trials.
Thus, this pilot study aimed to evaluate feasibility, safety, and the preliminary effect of a DQ intervention in older adults with slow gait and mild cognitive impairment (MCI), who are at risk of Alzheimer’s disease. The study aimed to generate preliminary data on functional (i.e., cognitive outcomes such as the Montreal Cognitive Assessment (MoCA) and mobility outcomes such as gait speed) and blood biomarker outcomes necessary to help motivate a larger, subsequent trial of DQ. We hypothesised that the intervention is feasible, safe, and demonstrates preliminary efficacy.
Methods
Study design
The STAMINA (Senolytics To Alleviate Mobility Issues and Neurological Impairments in Ageing) study is a 12-week, single-arm, open-label, pre-post pilot study implementing intermittent doses of DQ in 12 older adults aged ≥65 years with slow gait speed and MCI, which are both precursors to the development of Alzheimer’s disease.8, 9, 10 A full description of the study rationale and design has been published elsewhere.33 All participants in this study provided informed consent. Participants were asked to take 100 mg of Dasatinib and 1250 mg of Quercetin orally once a day for two consecutive days every two weeks, for six cycles over 12 weeks. DQ was administered under Investigational New Drug (IND) 157920. The rationale for the dose and treatment schedule were based on previous studies.29,30 The study was conducted from 2022 to 2024 in the greater Boston area in Massachusetts, USA.
Recruitment and eligibility
Inclusion and exclusion criteria
The majority (∼60%) of individuals were recruited through a recruitment firm that utilises online targeted advertisements based on an individual’s browsing history as previously described.34 Additional recruitment methods included senior housing presentations, study flyers at clinical practices, and invitations to previous research volunteers who agreed to be in our participant registries. Older adults (age ≥65 years) were first screened by phone to confirm the presence of MCI (defined as telephone MoCA scores between 10 and 20 points, but able to perform all Katz Activities of Daily Living [ADLs]).35 If they passed this screen, they were invited to an in-person screening where they were confirmed eligible for this study if they had slow gait speed (<1 m/s). At any point in the screening process, individuals were excluded if they had reported being non-ambulatory, had unstable heart disease, heart failure, stroke, or hospitalization within the last six months, liver disease, neurodegenerative disease, advanced renal disease, pulmonary hypertension, fluid retention, self-reported drug or alcohol use disorder, QTc prolongation (>450 ms), anaemia, thrombocytopaenia, neutropenia, prothrombin time >13 s or International Normalized Ratio >1.1, use of anti-arrhythmic medications, antipsychotics and anxiolytics, anti-platelet or anti-coagulant medications other than aspirin, quinolone antibiotics, or drugs metabolised by the same liver enzymes as Dasatinib or Quercetin. The majority of the exclusion criteria were proposed out of an abundance of caution to ensure safety. Volunteers were also excluded if we were unable to insonate the middle cerebral artery through a temporal bone window on at least one side of the head using transcranial Doppler ultrasound (TCD), which was necessary for another aspect of the study not reported here. There were 3 individuals excluded for this reason and 6 for minor deviations from our inclusion criteria, including fast gait speed (n = 2), compliance concerns (n = 1), low white blood cells and high cystatin C levels (n = 2), or due to closed enrolment (n = 1). These 9 individuals and one other individual who was unwilling to take the study medications were invited to participate in a control biomarker assessment (total n = 10, referred to as Biomarker Control) described below. A full list of exclusion criteria and corresponding definitions is provided elsewhere.33
Study procedure
A thorough description of the assessments that were performed at each study visit is provided elsewhere.33 Safety assessments, including vital signs, electrocardiogram, self-reported symptoms, and safety blood tests were also performed before each drug administration. Participants were then asked to take the first day dose of the study medications in the study clinic, while being monitored for any acute effects. They were then provided with their second day dose to take at home, which was confirmed by telephone the following day and return of pill bottles at the subsequent visit. Symptoms were evaluated by telephone on the day of their at-home dosage and 3–5 days after their visit. Functional outcomes were measured after 6 and 12 weeks of DQ. However, to minimise participant burden, urine and blood were only collected at screening (approximately 2 weeks prior to baseline) and 2 weeks after all 12 doses of DQ, for a total time of 14 weeks between blood draws.
Feasibility and safety outcomes
Feasibility
Throughout the study, the ratio of the number of participants who were eligible for and/or completed the study to the number screened was carefully tracked. The protocol deviations that were reported to the IRB were logged, counted, and documented. Compliance with study medication was monitored for each dose. Based on the number of pills that were returned in the medication bottles, a compliance score (i.e., the number of taken doses/the number of dispensed doses) was calculated. If a participant returned an empty pill bottle, it was assumed that the dose was taken.
Drug safety and tolerance
The frequency of adverse events (AEs) was evaluated by symptom questionnaires and blood safety values during each visit. The blood tests included a complete blood count with differential, prothrombin time/International Normalized Ratio, and a comprehensive metabolic panel (including glucose, calcium, sodium, potassium, bicarbonate, chloride, blood urea nitrogen, creatinine, albumin, total protein, alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase, total and direct bilirubin, glomerular filtration rate, and cystatin C), which were all measured by Quest Diagnostics. The QTc interval, body temperature, heart rate, breathing rate, oxygen saturation, seated blood pressure, postural change of blood pressure, and heart rate were also measured at each visit. Laboratory results or vital signs that deviated from a pre-determined safety range (described in full elsewhere33) were considered AEs if they appeared for the first time at any visit following the screening visit or if they had more than a 20% change from baseline.
Functional outcomes
Cognitive assessments
The cognitive assessments included the Trail Making Test (TMT)36,37 to assess executive function and the MoCA38 to assess global cognition. These assessments were measured at baseline, mid-point, and follow-up by a trained research coordinator. A different version of the TMT and MoCA was used at each visit to avoid a learning effect.
Physical assessments
The physical assessments included the short physical performance battery,39 the timed up and go test,40 and maximum grip strength (by handheld dynamometer). These assessments were measured at baseline, mid-point, and follow-up.
Dual task assessment
To assess the impact of a cognitive task on mobility and balance while walking, a dual task assessment was performed. Participants underwent walking trials of 32 m during the following two conditions in a randomised order: (1) Single task (ST): walking without a cognitive distraction and (2) Dual task (DT): walking while performing a serial subtraction task. Dual-task cost in gait speed was calculated as the absolute difference in gait speed while performing the DT compared to the ST (i.e., [VelocityST - VelocityDT]). Similar calculations were performed for stride length. Dual task outcomes were measured at baseline, mid-point, and follow-up.
Biomarker outcomes
Blood biomarkers included cytokines and chemo-attractants, matrix metalloproteinases (MMP), tissue inhibitors of MMPs (TIMPs), adhesion molecules, growth factors, and their binding proteins.20,23,29 Expression of senescence markers (e.g., ARF, p16, p21, p16INK4a)41, 42, 43, 44 in CD3 positive T lymphocytes was measured by rt-PCR. All markers that were measured are listed in Supplementary Table S1. All immunoassay kits were purchased from R&D Systems (Bio-Techne, Minneapolis, MN) and the corresponding catalogue numbers are provided in Supplementary Table S2, while catalogue numbers and suppliers of primers are provided in Supplementary Table S3. The markers originally hypothesised to be affected by DQ are described in the main body of this report. Although this was a single-arm study, we compared biomarker changes to those in a small comparison group of similar subjects with slow gait and MCI (n = 10) who were excluded from the trial for the reasons above, in order to determine the variation in biomarkers over a similar time period (∼14 weeks). This biomarker control group did not complete any of the follow-up physical or cognitive assessments.
CD3 T cell isolation
Blood draws were conducted at screening and 2 weeks after their last dose of DQ (i.e., a total of 14 weeks in between blood draws). T cell populations expressing CD3 as a surface marker were isolated by negative depletion using RosetteSep Human T cell enrichment cocktail (STEMCELL Technologies, Cat#: 15061). Viability was measured by Trypan blue staining all samples to ensure cell viability was in the range of 95–100%. Purity was evaluated in the first 5 samples and every new bottle of RosetteSep Human T cell enrichment cocktail to ensure purity of CD3+ cells was >90%. The PE-conjugated anti-CD3 monoclonal antibody, RRID: AB_395740, was used to confirm purity of cell isolation. RNA was extracted using TRIzol (Invitrogen, Ref#: 15596018). RNA quantity and purity were confirmed with a Nanodrop spectrophotometer (Thermo Scientific, Wilmington, DE). One μg of RNA was reverse transcribed using the High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems by Life Technologies, Foster City, CA). PCR reactions were run using the ABI Prism 7900HT Real Time System (Applied Biosystems, Carlsbad, CA) with TaqMan primer-probe sets (Applied Biosystems, Carlsbad, CA). See Supplementary Table S3 for information on primers. Expression of target genes was normalised to the expression of TATA-box binding protein. The absolute and percent change of the expression of target genes between screening and final follow-up values were calculated.
ELISAs for exploratory blood biomarkers of senescence
Biomarkers were measured in blood samples collected in serum separator, EDTA, and heparin tubes before and after 14 weeks in the STAMINA and Biomarker Control groups. Blood was centrifuged, aliquoted, and serum/plasma was temporarily stored in a minus 80-degree freezer until batch analysis of all samples. Circulating SASP associated proteins in plasma or serum were measured at the Facility of Geoscience Analysis, an analytical lab of the NIA funded Translational Geroscience Network. Duplicate samples were analysed using either the FLEXMAP3D Machine (Luminex) or the Ella Automated Immunoassays (Protein Simple, Bio-Techne) platforms with commercially available immunoassay kits (R&D Systems, Bio-Techne, Minneapolis, MN). Based on the abundance of the targeted proteins bead region and antibody compatibility, the targets were organised into 18, 10, or 6 plex plates. To ensure linearity, very low abundance proteins were measured using the ELLA Automated Immunoassay (Protein Simple/Bio-Techne) with cartridges purchased from Protein Simple/Bio-Techne. Values below the assay’s detection limit, resulting in the absence of a matching paired sample, were excluded from the paired t-test analyses. The range and specificity of the assays are described in Supplementary Table S2. The absolute and percent change of blood biomarkers between screening and final follow-up values were calculated.
Statistics
Feasibility and safety outcomes were tabulated. Additionally, the frequency of clinical safety and symptom AEs were tabulated by participant. Participant demographics, functional outcomes, and biomarker outcomes are reported as mean ± standard deviation or as n (%), as appropriate. Within-person changes are reported as mean with a corresponding measure of spread (e.g., standard deviation or 95% confidence interval [95% CI]).
For the functional outcomes, mixed linear models were estimated to evaluate the change in the outcomes over multiple visits. Within and between-group differences in biomarker concentrations were assessed using paired and unpaired Student’s t-tests, respectively. Markers considered a priori in our statistical analysis plan (i.e., interleukin-1 alpha, IL-1α; interlekin-6, IL-6; matrix metalloproteinase-9, MMP9; and cyclin-dependent kinase inhibitor 2A/alternate splice variant – exons 1 alpha-2, p16INK4a)23,44 and those identified post hoc for their published roles in inflammation, cognition, and senescence (tumour necrosis factor alpha; TNF-α,45 matrix metalloproteinase-3, MMP346 and programmed death-ligand 2, PDL-247; respectively) are highlighted here, while others are included in the supplement. Spearman correlations were performed to assess the association between percent changes in biomarker concentrations and changes in functional outcomes. Subgroup analyses were also performed based on stratifications by age (pre-specified: < and ≥ median age of the study population; data not shown), lower MoCA scores at baseline (i.e., 18–25 points on MoCA, n = 8), and the median expression of the senescent cell marker p16INK4a at baseline in the study population (i.e., “Low Senescent Cell Burden” vs. “High Senescent Cell Burden,” respectively). The sample size was pre-determined and motivated by the need to assess resource needs for a subsequent randomized controlled trial of DQ. Unfortunately, due to the limited sample diversity and small sample size (n = 12), we were unable to analyse the data by race. All analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC).
Ethics
This study was conducted at the Marcus Institute for Ageing Research in Boston, MA, approved by the Advarra IRB (#Pro00053594), registered at Clinicaltrials.gov (Identifier: NCT05422885), and monitored by the FDA. The biomarker control arm was also approved by the Advarra IRB (#Pro00066895). All participants provided informed consent prior to engaging in research activities.
Role of funders
The funding sources had no role in study design, conduct, analysis, interpretation, or writing of the manuscript or in the decision to submit the manuscript.
Results
Study participant Flow Chart and characteristics
The process of participant recruitment is shown in Fig. 1. A total of 332 individuals expressed interest in the STAMINA study. One hundred and fifteen individuals were screened over the telephone, of which 60 were found ineligible due to not meeting the definition of MCI (n = 31), a medication contraindication (n = 11), an exclusionary health condition (n = 6), or multiple reasons (n = 12). In addition, 24 eligible individuals were not enroled due to loss of interest (n = 12), loss to follow-up (n = 10), or closed enrolment (n = 2). A total of 31 participants were screened in-person, of which 16 were excluded due to inability or unwillingness to participate in the study protocol (n = 5), fast gait speed (n = 5), exclusionary health conditions (n = 4), a medication contraindication (n = 1), or multiple reasons (n = 1). In addition, after confirmation of study eligibility, one individual was lost to follow-up (n = 1) before starting DQ, while two participants withdrew consent during medication administration due to pre-existing anxiety (n = 1) and concerns about taking study medications (n = 1). Thus, 12 participants completed the intervention.
Fig. 1.
Flow chart of recruitment for the STAMINA Study. MCI, mild cognitive impairment.
The characteristics of those individuals who completed the study (n = 12) are presented in Table 1. STAMINA participants had a mean (SD) age of 77 (±8) years and 58% self-reported as female (n = 7). They primarily identified as white (n = 10) and were relatively highly educated. Although not displayed in the table, participants were on a variety of medications, but these remained unchanged throughout the study. The characteristics of the Biomarker Control participants are in Supplementary Table S4.
Table 1.
Characteristics of completed STAMINA participants (n = 12) at time of screening.
| Mean ± SD or n (%) | |
|---|---|
| Age (y) | 77 ± 8 |
| Sex (female) | 7 (58) |
| Ethnicity | |
| White | 10 (83) |
| African American | 2 (17) |
| Highest level of education | |
| Associate’s degree | 1 (8) |
| Bachelor’s degree | 5 (42) |
| Graduate/Doctoral/Law Degree | 6 (50) |
| Gait speed (m/s) | 0.8 ± 0.1 |
| Telephone MoCA score (range of instrument: 0–22 points, range of inclusion criteria: 10–20 points) | 17 ± 2 |
| BMI (kg/m2) | 28 ± 4 |
| Major adverse cardiovascular eventsa | 1 (8) |
| High blood pressure | 5 (42) |
| High cholesterol | 6 (50) |
| Osteo- or degenerative arthritis | 6 (50) |
| Depression | 3 (25) |
| Diabetes | 2 (17) |
| Kidney disease | 2 (17) |
| Systolic seated blood pressure (mmHg) | 124 ± 11 |
| Diastolic seated blood pressure (mmHg) | 68 ± 7 |
| QTc interval (ms) | 412 ± 24 |
| Haemoglobin (g/dL) | 12.9 ± 1.3 |
| White blood cells (cells/mcL) | 6.4 ± 1.6 |
| Platelets (count/mcL) | 251 ± 40 |
| Haematocrit (%) | 38.5 ± 3.4 |
| ALT (U/L) | 16.9 ± 7.2 |
| AST (U/L) | 21.9 ± 6.0 |
| Alkaline phosphatase (U/L) | 71.0 ± 12.2 |
| Glucose (mg/dL) | 97.9 ± 32.8 |
| Estimated GFR (mL/min/1.73 m2) | 61.9 ± 14.9 |
| Total bilirubin (mg/dL) | 0.5 ± 0.1 |
| Creatinine (mg/dL) | 1.1 ± 0.4 |
| Blood urea nitrogen (mg/dL) | 21.3 ± 9.4 |
| Cystatin C (mg/dL) | 1.4 ± 0.4 |
| Prothrombin time (sec) | 10.5 ± 0.5 |
ALT, Alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; GFR, glomerular filtration rate; MoCA, Montreal Cognitive Assessment.
Defined as having a myocardial infarction within 6 months, a pacemaker, atrial fibrillation, or chronic heart failure.
Feasibility and safety
Table 2 shows the recruitment rate, the number of protocol deviations, the number of AEs, and the participants’ adherence, which are all indicators of feasibility. Approximately 10% of all the individuals who were screened by telephone and 39% who were screened in-person completed the study. Twenty-one protocol deviations were primarily for minor schedule changes and none necessitated reporting to the IRB or resulted in AEs. Of the 81 AEs, only five (7%) were deemed “possibly” related to the study medications and only one (∼1%) was “probably” related to the study medications. One serious adverse event (SAE) was reported (accidental injury) during the intervention, however, it was not related to the study. Of the 12 individuals who completed the study, participants were 99% compliant with Dasatinib and 100% compliant with Quercetin.
Table 2.
Feasibility outcomes of the STAMINA study.
| Outcome | Brief description | Rate or n |
|---|---|---|
| Telephone Screen Recruitment Rate (# of participants completed study/# of telephone screened participants) | The proportion of all of the volunteers screened by telephone that completed the study. | 10% of all individuals telephone screened, eventually completed the study. |
| In-person Screen Recruitment Rate (# of participants completed study/# of in-person screened participants/) | The proportion of all of the volunteers screened in-person that completed the study. | 39% of all individuals screened in person, eventually completed the study. |
| Protocol Deviations | The frequency of protocol deviations that were reported to the IRB and/or resulted in AEs. | 0 |
| Total Adverse Events | The frequency of all adverse events during the study. | 81 |
| Clinical Safety Adverse Events | The frequency of adverse events specifically defined by our study clinical safety labs. | 21 |
| Adverse Events Possibly Related to Study Activities | The frequency of adverse events related or possibly related to the study activities. | 6 |
| Serious Adverse Events | The frequency of serious adverse events. | 1 |
| Serious Adverse Events Possibly Related to Study Activities | The frequency of serious adverse events related or possibly related to the study activities. | 0 |
| Dasatinib Medication Compliance of Completed STAMINA Participants (n = 12) (% of intended doses consumed) | Compliance with Dasatinib medication administration. Includes clinic-administered and self-administered at-home intake of study drugs (assessed by self-report and returned pill containers) divided by the number of intended pills over the period of study participation. | 143/144 = 99% |
| Quercetin Medication Compliance of Completed STAMINA Participants (n = 12) (% of intended doses consumed) | Compliance with Quercetin medication administration. Includes clinic-administered and self-administered at-home intake of study drugs (assessed by self-report and returned pill containers) divided by the number of intended pills over the period of study participation. | 720/720 = 100% |
AE, adverse event; IRB, institutional review board.
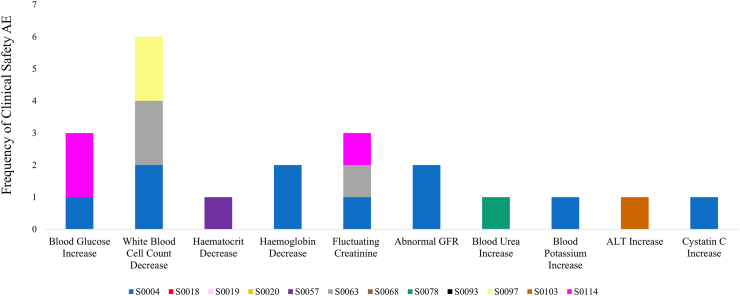
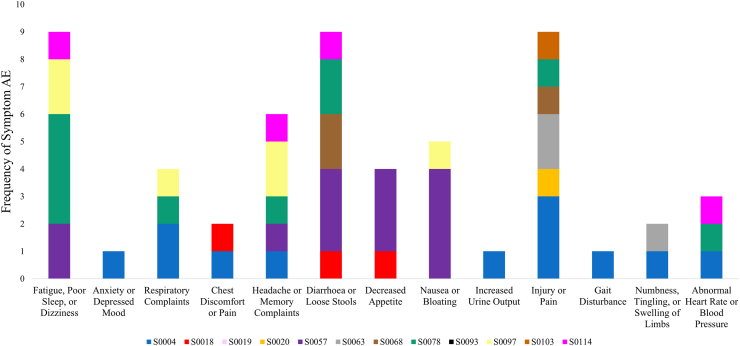
The frequencies of both clinical blood safety and self-reported symptom AEs are displayed in Fig. 2, Fig. 3, respectively. The most common clinical blood safety AE was a mild reduction in white blood cell count (n = 6; Fig. 2), but not to dangerous levels. Of the self-reported symptom AEs, there were 9 AEs of “injury or pain,” 9 AEs of “fatigue/poor sleep/dizziness,” and 9 AEs of “diarrhoea/loose stools” (Fig. 3).
Fig. 2.
The frequency of clinical safety adverse events in the STAMINA study. Clinical blood safety measures were evaluated prior to each administration of the 2-day dose of Dasatinib and Quercetin. If the Laboratory results deviated from a pre-determined safety range (described in full elsewhere33) by more than 20% and/or newly appeared, they were considered adverse events. Each colour represents a different participant (n = 12). AE, adverse events; ALT, alanine-aminotransferase; GFR, glomerular filtration rate.
Fig. 3.
The frequency of self-reported symptoms adverse events in the STAMINA study. Self-reported symptoms were evaluated once before and two times after each 2-day drug treatment of Dasatinib and Quercetin. If the symptom newly appeared or was reported to be more severe than usual, then it was considered an adverse event. Each colour represents a different participant (n = 12). AE, adverse events.
Functional outcomes
The primary cognitive and mobility functional outcomes during the STAMINA study are presented in Table 3. As expected, due to the small sample size of the study, most measures did not change significantly over time. Nevertheless, 12-week change in mean scores of the cognitive measures MoCA (mean change = 1.0 point, 95% CI: −0.7, 2.7) and TMT B minus A (mean change = −17.2 s, 95% CI: −53.4, 19.0) appeared to non-significantly improve after treatment. Individual level pre- and post-data for change in MoCA for all participants is shown in Supplementary Fig. S1.
Table 3.
Phenotypic outcomes before, during, and after STAMINA.
| Baseline (Week 2) |
Mid-point (Week 8) |
Follow-up (Week 14) |
|
|---|---|---|---|
| Mean ± SD | |||
| Cognitive outcomes | |||
| MoCA (range: 0–30 points) | 23 ± 2 | 25 ± 3a | 24 ± 2 |
| MoCA in participants with low baseline MoCA scores (≤25 points, n = 8) | 22 ± 3 | 25 ± 3a | 24 ± 2a |
| Trail Making Test: A (sec) | 52.0 ± 29.0 | 45.6 ± 23.1 | 43.1 ± 12.7 |
| Trail Making Test: B (sec) | 132.6 ± 86.6 | 120.6 ± 79.2 | 106.5 ± 51.9 |
| Trail Making Test: B – A (sec) | 80.6 ± 80.2 | 75.0 ± 61.4 | 63.4 ± 44.6 |
| Mobility outcomes | |||
| SPPB (range: 0–12 points) | 9 ± 1 | 9 ± 2 | 9 ± 2 |
| TUG (seconds) | 14.2 ± 2.6 | 13.9 ± 3.1 | 13.6 ± 2.4 |
| Maximum grip strength (kg) | 23.5 ± 7.2 | 22.7 ± 5.8 | 23.6 ± 5.3 |
| Normal walk stride length (m) | 1.01 ± 0.13 | 1.00 ± 0.12 | 1.02 ± 0.14 |
| Dual task stride length (m) | 0.93 ± 0.13 | 0.93 ± 0.13 | 0.96 ± 0.14b |
| Dual task cost of stride length (m) | −0.08 ± 0.08 | −0.07 ± 0.06 | −0.06 ± 0.05b |
| Normal walk gait speed (m/s) | 0.87 ± 0.15 | 0.85 ± 0.16 | 0.87 ± 0.14 |
| Dual task gait speed (m/s) | 0.76 ± 0.13 | 0.75 ± 0.16 | 0.78 ± 0.14 |
| Dual task cost of gait speed (m/s) | −0.11 ± 0.11 | −0.11 ± 0.09 | −0.09 ± 0.08 |
MoCA, Montreal Cognitive Assessment; SPPB, short physical performance battery; TUG, timed up and go.
Indicates a p-value < 0.05 when compared to baseline.
Indicates a p-value between 0.05 and 0.10 when compared to baseline.
In the mobility outcomes, 12-week change in mean scores of dual task gait speed (mean change = 0.03 m/s, 95% CI: −0.03, 0.07) and stride length (mean change = 0.031 m, 95% CI: −0.003, 0.066) non-significantly increased. There was also a non-significant reduction in dual task cost of gait speed by 0.02 (95% CI −0.03, 0.06) and dual task cost of stride length by 0.02 (95% CI: −0.01, 0.05) after treatment when comparing baseline to follow-up values (p-range: 0.05–0.10). Individual level pre- and post-data for change in stride length during dual task are shown in Supplementary Fig. S2.
The pre-specified sub-group analyses showed that those with lower baseline MoCA scores (i.e., 18–25 points) appeared to have a significant increase in MoCA score by 2.0 points (95% CI 0.1, 4.0) at follow-up when compared to baseline (p = 0.04). The group with lower MoCA scores had similar mobility outcomes as the entire STAMINA cohort (data not shown). An exploratory sub-group analysis was performed in High vs. Low Senescent Cell Burden. Within the High Senescent Cell Burden sub-group, there were non-significant improvements in dual task costs of stride length at the mid-point (p = 0.08) and dual task gait speed at follow-up (p = 0.07; Supplementary Table S5). The Low Senescent Cell Burden sub-group demonstrated a non-significant improvement in the TMT at mid-point (p = 0.07), particularly on the B portion (p = 0.06; Supplementary Table S5). The sub-group analyses by age did not yield meaningful results in any of our measured outcomes (data not shown).
Biomarker outcomes
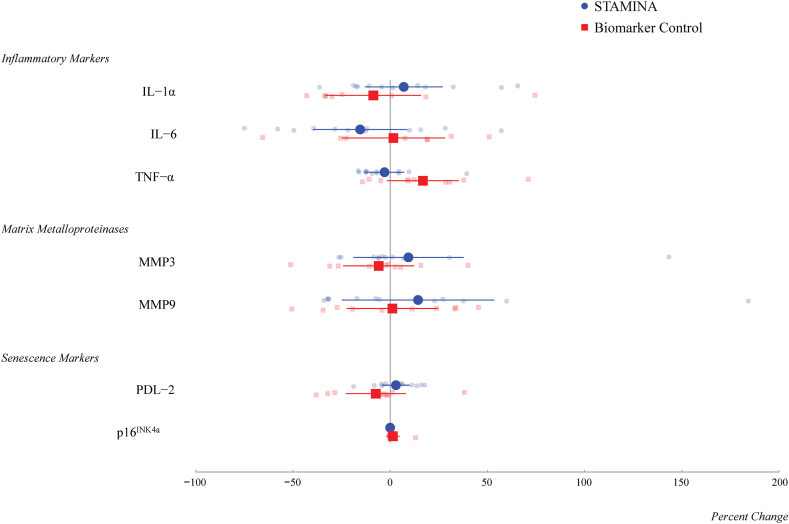
The mean percent change of select biomarkers of senescence for the STAMINA and Biomarker Control groups are displayed in Table 4 and Fig. 4. Data for all biomarkers are in Supplementary Tables S6 and S7 Baseline values of the SASP markers were relatively similar between the STAMINA and Biomarker Control groups. Although mean baseline p16INK4a expression appeared lower in STAMINA participants (10.6 ± 5.2), this was not significantly different from the Biomarker Control group (20.4 ± 23.0) (Supplementary Table S6). After DQ treatment, mean percent change in IL-6 and TNF-α, which have prominent roles in inflammation, declined by −15.5% (95% CI: −39.8, 8.8) and −3.0% (95% CI: −13.0, 7.1), respectively. The mean percent reduction in TNF-α was significantly different (p = 0.04) than the mean percent change in the Biomarker Control group (mean percent change = 16.8, 95% CI: −1.7, 35.4). Compared to all STAMINA participants, those with lower MoCA scores had larger percent changes in most of the select biomarkers (Table 4). There were no statistical differences in the percent change of select biomarkers between the Low and the High Senescent Cell Burden groups (Table 4).
Table 4.
Percent change of select markers of senescence.
| Biomarker | Function | Mean % change ± SD |
||||
|---|---|---|---|---|---|---|
| STAMINA (n = 12) | Low Baseline MoCA Scores (n = 8) | High senescent cell burden (n = 5) | Low senescent cell burden (n = 5) | Biomarker controls (n = 10) | ||
| Inflammatory markers | ||||||
| IL-1α | Pro-inflammatory cytokine | 7.0 ± 31.6 | 13.0 ± 36.6 | −4.8 ± 40.2 | 20.2 ± 25.1 | −8.7 ± 34.7 |
| IL-6a | Pro-inflammatory cytokine | −15.5 ± 38.3 | −24.6 ± 38.0 | −22.5 ± 33.4 | −8.7 ± 43.2 | 1.6 ± 35.0 |
| TNF-α | Central role in immune and inflammatory responses | −3.0 ± 15.8b | −7.4 ± 9.6b | −4.8 ± 11.2 | 1.5 ± 22.6 | 16.8 ± 25.9 |
| Matrix metalloproteinases | ||||||
| MMP3 | Degrades extracellular matrix proteins | 9.4 ± 44.8 | 17.6 ± 53.2 | 0.50 ± 20.8 | 22.2 ± 68.7 | −5.9 ± 25.7 |
| MMP9 | Degrades ECM proteins and is upregulated during inflammation | 14.3 ± 62.0 | 16.0 ± 73.3 | −16.1 ± 24.7 | 37.5 ± 89.3 | 1.1 ± 33.1 |
| Other markers of senescence | ||||||
| PDL-2 | Regulates immune responses and triggers tumorigenesis/progression | 2.9 ± 10.9 | 0.97 ± 10.8 | 1.7 ± 4.7 | 4.8 ± 15.9 | −7.5 ± 21.8 |
| p16INK4aa | Marker of senescent cells | −4.1 ± 25.9 | −13.7 ± 27.1 | −14.2 ± 21.0 | 6.0 ± 28.4 | −6.6 ± 24.5 |
IL, interleukin; MMP, matrix metalloproteinase; p16INK4a, a genetic marker of senescent cells; PDL-2, programmed cell death-ligand 2; TNF-α, tumour necrosis factor alpha.
Individuals with extreme percent changes were excluded from this table (i.e., n = 1 for IL-6, % change = 600; n = 1 for p16INK4a, % change = 1306).
Indicates a p-value < 0.05 when compared to the Biomarker Control group.
Fig. 4.
Percent change of select senescence biomarkers. The percent change between screening and final follow-up values (∼14 weeks) was calculated for each biomarker in STAMINA (n = 12, blue circle) and Biomarker Control (n = 10, red square) participants. All data points are displayed on the figure with the mean expressed as the larger shape and 80% confidence intervals expressed as the solid line. IL, interleukin; MMP, matrix metalloproteinase; p16INK4a, a genetic marker of senescent cells; PDL-2, programmed cell death-ligand 2; TNF-α, tumour necrosis factor alpha.
Percent changes in TNF-α and PDL-2 were negatively correlated with change in MoCA scores such that increases in MoCA scores correlated with reductions in these biomarkers (r = −0.65, 95% CI: −0.88, −0.09, p = 0.02 and r = −0.78, 95% CI: −0.93, −0.34, p < 0.01, respectively) (Fig. 5a and b). Additionally, reductions in PDL-2 was non-significantly correlated with increases in SPPB scores (r = −0.51, 95% CI: −0.84, 0.09, p = 0.08).
Fig. 5.
(a and b) Spearman correlations between change in cognitive scores and biomarkers. The absolute change of MoCA scores after treatment was correlated with percent change of TNF-α (a) and PDL-2 (b) in 12 participants. MoCA, Montreal Cognitive Assessment; PDL-2, programmed cell death-ligand 2; TNF-α, tumour necrosis factor alpha.
Discussion
The goal of this study was to assess the feasibility and safety of a senolytic DQ intervention and to provide preliminary estimates for functional and biomarker outcomes of interest in older adults with slow gait and MCI. To establish feasibility and to reach our goal of 12 completed participants, we screened 115 people by telephone and among these, 31 older adults were screened in-person. Adherence to intended doses of the study medications was excellent (≥99%) and there were no serious adverse effects. Although this study was underpowered to determine efficacy, DQ treatment appeared to improve some aspects of cognition and mobility, and elicited hypothesised improvements in some biomarkers of senescence, particularly in participants with the lowest MoCA scores. However, it is important to note that the estimates of change in the functional and biomarker data are intended strictly as hypothesis generating.
Senolytic trials may be burdened by significant recruitment challenges due to the added complications of concomitant medication use and comorbid health conditions, both of which may exclude individuals from participation. Thus, before implementing a large-scale trial to determine efficacy of DQ in older adults with slow gait and MCI, it is crucial to determine the recruitment rate to help inform the necessary duration and cost of the study. In our trial, approximately 10% of individuals who were telephone screened actually completed the study. Although this percentage is in range with estimates from other studies of older adults,48,49 more than half of the volunteers screened by telephone were ineligible due to medication contraindications, not meeting entry criteria, exclusionary health conditions, or a combination of those reasons. The exclusion criteria for this study were relatively stringent in order to satisfy FDA requirements that ensure the safety of participants who were receiving an established chemotherapeutic agent (i.e., Dasatinib) for a new indication. Therefore, the lengthy list of exclusions likely contributed to a large proportion of individuals being deemed ineligible. Furthermore, a substantial portion of the telephone screens did not meet our definition for MCI or slow gait. In our recent paper describing our recruitment methods and results, we reported that the study cohort was predominately white and well-educated,34 which is a limitation of this work. Therefore, the study cohort is not representative of the larger population of older adults with MCI and slow gait, and generalizability of the results of our exploratory study for this new indication is limited.
Another important aspect of study feasibility is safety, which is necessary to establish prior to initiating a larger-scale clinical trial. The most common clinical safety event in our pilot study was a reduction in white blood cell count, which was reported six times across all possible blood draws (∼90 draws). Regardless, there were only six AEs that were possibly/probably related to the intervention, and no SAEs. Other trials with DQ also reported relative safety of the treatment.29,30,32 Our data add to the existing literature and support the tolerance and safety of DQ in older adults with slow gait and MCI.
Our study design was not adequately powered to confidently detect statistically significant changes in the functional and biomarker outcomes. Rather, the goal of the current trial was to collect data on the variation of these measures, which would be used to inform a power analysis for a larger trial. Thus, an emphasis was placed on directionality of the change in the outcome measures. After treatment with DQ, the cognitive outcomes (i.e., MoCA and TMT scores) appeared to improve (MoCA, higher scores indicate better cognition and lower TMT scores indicate better executive function). In all participants, MoCA scores increased by only 1 point, which may not be clinically meaningful. However, the TMT scores improved by 17 s, which was primarily driven by a reduction in the Trails B time by ∼25 s. Changes in Trails B by 20 s was suggested to be a clinically meaningful change in a cohort of older adults with MCI.50 Furthermore, those with lower cognitive scores at baseline and thus with the most potential to improve, had a significant increase of approximately 2 points in MoCA scores at final follow-up compared to baseline. Although a 2-point increase in MoCA scores is considered clinically meaningful,51 other studies have suggested that larger changes may be required for clinically meaningful outcomes.52 Additionally, we cannot exclude the possibility that the changes in MoCA scores or TMT are simply a regression to the mean. To unequivocally determine if DQ improves cognition, future trials will need a larger sample size and the inclusion of a placebo control group.
Although not statistically significant, our results indicate that participants experienced improved mobility following DQ as reflected by non-significant reduction in time (approximately 1 s) to perform the TUG test, which included several aspects of mobility (e.g., walking, turning, rising from a chair). However, such a small magnitude of change is unlikely to be clinically meaningful. Furthermore, dual task gait speed appeared to improve after DQ by approximately 0.02 m/s. However, changes of 0.05 m/s are reported to represent clinically significant changes.53 Interestingly, we saw a non-significant reduction in dual task cost of stride length and gait speed (i.e., less shortening of stride length and less slowing of gait speed while performing a serial subtraction task), which suggests that executive control of gait was improved. The improvement in stride length during dual tasking is potentially of clinical value, since short stride length has been reported as a particularly robust predictor of life-threatening events (defined as major adverse events, physical disability, and mortality) in older adults.54 Furthermore, participants with the highest burden of senescent CD3+ cells in the peripheral circulation had the largest change from baseline in dual task gait speed and dual task cost of stride length. Future trials should consider recruiting participants with a high burden of senescent cells and examine whether the effects of reducing senescent circulating CD3+ cells are limited to the peripheral musculature, rather than central nervous system. Although these findings suggest that DQ may improve mobility, they are preliminary and mainly hypothesis generating in nature. Therefore, they must be confirmed in a future larger trial.
Single-arm pilot studies administering DQ have shown reductions in IL-1α, IL-6, and MMP9 after DQ treatment.29,30 Despite a similar percent reduction in IL-6 following DQ treatment in the current study, this change was not statistically different from changes in the Biomarker Control group. Interestingly, the STAMINA participants experienced a significant reduction in TNF-α compared to Biomarker Controls. TNF-α is a prominent immune and inflammatory regulator involved in cellular senescence.55,56 However, it is unclear whether this magnitude of change in TNF-α seen in the STAMINA participants is of biological relevance. We did not observe a similar trend in p16INK4a, which is the most widely used marker of senescent cells. Our study examined expression of a splice variant of p16INK4a in CD3+ T cell populations isolated from peripheral blood. Genetic or pharmacological clearance of p16 positive cells in different tissues has shown efficacy in more than forty disease models, including models of neurodegenerative diseases.1 Although, circulating CD3+ T cells expressing p16INK4a was identified as one the first markers linked to senescent cells that correlates with chronological ageing,44,57 sensitivity of it to senolytics has not been tested previously. We observed mean percent reduction of p16INK4a expression in the STAMINA participants. However, data were highly variable, which could be due to the heterogeneity of the cell population, chance, small numbers, medications, metabolic status, or the study time window between DQ treatment and when blood samples were collected. It is also possible that p16INK4a CD3+ senescent cells in circulation are not sensitive to DQ due to the absence of anti-apoptotic pathways that DQ targets. Unexpectedly, there was a larger reduction in p16INK4a in the Biomarker Controls, suggesting that changes in this marker may not have been a result of DQ. Other studies have shown reductions in p16INK4a in adipose tissue following senolytic treatment.20,30 Unfortunately, we were not able to obtain tissue from study subjects. The percent changes in IL-6, TNF-α, and p16INK4a were larger in magnitude in the individuals with the lowest MoCA scores, suggesting that these may be the individuals most likely to benefit from a senolytic intervention.
Further supporting the potential clinical implications of our study is the observation that changes in MoCA scores were negatively correlated with changes in TNF-α and PDL-2 (a recently proposed serum marker of senescence47), such that larger reductions in these biomarkers were strongly and significantly correlated with larger improvements in MoCA scores. Additionally, changes in the SPPB scores were negatively correlated with changes in PDL-2, such that improvements in SPPB were correlated with reductions in PDL-2. A similar observation was found by Justice et al. who reported that select SASP markers (e.g., IL-10 and IL-18) correlated with aspects of physical function (e.g., chair stands and 4 m gait speed) after an intervention with DQ.29 However, there was inconsistency in the specific biomarkers that correlated with physical function between our study and those by Justice et al.29 Together these results help support our hypothesis that DQ targets senescence and may mediate aspects of cognition and physical function.
The findings of our study must be considered in light of several limitations and results must be interpreted with caution, particularly our sub-group analyses. As a pilot study aimed at determining feasibility and safety, we did not include a placebo control group and are largely underpowered to determine efficacy of DQ. Therefore, we cannot unequivocally determine whether the functional changes were a result of regression to the mean, chance, or DQ treatment. Future studies that include a placebo control group are critical to confirm our findings. Furthermore, the generalizability of our results is limited to primarily white, highly educated, older adults with slow gait and MCI. Unfortunately, our small study has very limited racial diversity. Additionally, the preliminary impact on functional and biomarker outcomes could be due to chance and/or regression to the mean. Furthermore, it is important to note that 14 weeks is a relatively short time to determine changes in physical function or cognition. Given the limitations of a single arm trial, we included the Biomarker Control group to provide a reference for the changes in biomarkers over approximately 14 weeks. Since this group was recruited from individuals excluded from the STAMINA study, it may not be representative of the actual STAMINA study participants. Finally, both functional and biomarker outcomes were evaluated two weeks after the last dose of DQ, which may have impacted our estimates and leaves us unable to discern whether the changes seen were a result of the intervention or discontinuation of DQ. However, since changes in cognition and mobility typically take time to occur, we surmise it is more likely that changes are a result of DQ rather than its discontinuation. Future studies may benefit from a longer DQ treatment period and follow-up visits after DQ discontinuation.
This preliminary study provides evidence that administering the combined DQ senolytic intervention is feasible and well tolerated in a select population of older adults who are at risk of developing Alzheimer’s disease. It further supports continued investigation of DQ as a potential treatment for individuals with slow gait and MCI.
Contributors
Conceptualization: Tamara Tchkonia, James L. Kirkland, Thomas Travison, and Lewis Lipsitz. Data curation: Courtney Millar, Ike Iloputaife, and Thomas Travison. Formal analysis: Courtney Millar. Funding acquisition: Lewis Lipsitz. Investigation: Courtney Millar, Ike Iloputaife, Kathryn Baldyga, Afroditi Boulougoura, Theodoros Vichos, Tamara Tchkonia, Aaron Deisinger, Tamar Pirtskhalava, and Lewis Lipsitz. Methodology: Tamara Tchkonia, James L. Kirkland, Thomas Travison, and Lewis Lipsitz. Project administration: Courtney Millar, Ike Iloputaife, Kathryn Baldyga, and Lewis Lipsitz. Resources: Afroditi Boulougoura, Theodoros Vichos, Aaron Deisinger, Tamar Pirtskhalava, Tamara Tchkonia, James L. Kirkland, Thomas Travison, and Lewis Lipsitz. Supervision: Courtney Millar and Lewis Lipsitz. Visualization: Courtney Millar, Ike Iloputaife, Kathryn Baldyga, Amani Norling, and Thomas Travison. Writing: Courtney Millar. All authors have directly accessed and verified the underlying data reported in the manuscript. All authors have reviewed, edited, and approved the final version of the manuscript.
Data sharing statement
De-identified data presented in this manuscript, data dictionary, and analytic plan will be made available by secure transfer upon request to the corresponding author under conditions of appropriate ethical oversight, upon investigator approval and execution of a data use agreement. Banked biospecimens (serum, plasma, urine, and/or cDNA of CD3 cells) that are stored at Mayo Clinic and/or Cedars Sinai Medical Center will also be made available under the same conditions as stated above.
Declaration of interests
J.L.K. and T.T. have a financial interest related to this research. Patents on senolytic drugs are held by Mayo Clinic where they were employed before moving to Cedars-Sinai. T.P was also employed by Mayo Clinic. This research has been reviewed by the Mayo Clinic Conflict of Interest Review Board and was conducted in compliance with Mayo Clinic Conflict of Interest policies. J.L.K, T.T., L.A.L., T.G.T., are members of the NIH-funded Translational Geroscience Network. C.L.M has received study support from the US Highbush Blueberry Council. All other authors have no declarations.
Acknowledgements
This study was funded by the NIA grant R21AG073886 awarded to the Hebrew SeniorLife Hinda and Arthur Marcus Institute for Ageing Research (PI Lewis Lipsitz), with a subcontract awarded to Mayo Clinic where James L. Kirkland and Tamara Tchkonia were employed. The NIA Translational Geroscience Network grant R33 AG061456 also provided support for this study. Support for J.L.K. and T.T. was received from the Connor Fund, Robert J. and Theresa W. Ryan, and the Noaber Foundation. C.L.M and A.M.N. were supported by NIA T32-AG023480. C.L.M., I.I., and K.B. were also supported by R21AG073886. Supplemental funding was provided by philanthropic donations to the Marcus Institute for this project.
Footnotes
Supplementary data related to this article can be found at https://doi.org/10.1016/j.ebiom.2025.105612.
Appendix ASupplementary data
References
- 1.Chaib S., Tchkonia T., Kirkland J.L. Cellular senescence and senolytics: the path to the clinic. Nat Med. 2022;28(8):1556–1568. doi: 10.1038/s41591-022-01923-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Khosla S. Senescent cells, senolytics and tissue repair: the devil may be in the dosing. Nat Aging. 2023;3(2):139–141. doi: 10.1038/s43587-023-00365-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Levêque D., Becker G., Bilger K., Natarajan-Amé S. Clinical pharmacokinetics and pharmacodynamics of dasatinib. Clin Pharmacokinet. 2020;59(7):849–856. doi: 10.1007/s40262-020-00872-4. [DOI] [PubMed] [Google Scholar]
- 4.Graefe E.U., Wittig J., Mueller S., et al. Pharmacokinetics and bioavailability of quercetin glycosides in humans. J Clin Pharmacol. 2001;41(5):492–499. doi: 10.1177/00912700122010366. [DOI] [PubMed] [Google Scholar]
- 5.Ito T., Teo Y.V., Evans S.A., Neretti N., Sedivy J.M. Regulation of cellular senescence by polycomb chromatin modifiers through distinct DNA damage- and histone methylation-dependent pathways. Cell Rep. 2018;22(13):3480–3492. doi: 10.1016/j.celrep.2018.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Coppe J.P., Desprez P.Y., Krtolica A., Campisi J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol. 2010;5:99–118. doi: 10.1146/annurev-pathol-121808-102144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Xu M., Pirtskhalava T., Farr J.N., et al. Senolytics improve physical function and increase lifespan in old age. Nat Med. 2018;24(8):1246–1256. doi: 10.1038/s41591-018-0092-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Verghese J., Wang C., Lipton R.B., Holtzer R. Motoric cognitive risk syndrome and the risk of dementia. J Gerontol A Biol Sci Med Sci. 2013;68(4):412–418. doi: 10.1093/gerona/gls191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Verghese J., Wang C., Bennett D.A., Lipton R.B., Katz M.J., Ayers E. Motoric cognitive risk syndrome and predictors of transition to dementia: a multicenter study. Alzheimers Dement. 2019;15(7):870–877. doi: 10.1016/j.jalz.2019.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ayers E., Verghese J. Gait dysfunction in Motoric cognitive risk syndrome. J Alzheimers Dis. 2019;71(s1):S95–S103. doi: 10.3233/JAD-181227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lin Y.F., Wang L.Y., Chen C.S., Li C.C., Hsiao Y.H. Cellular senescence as a driver of cognitive decline triggered by chronic unpredictable stress. Neurobiol Stress. 2021;15 doi: 10.1016/j.ynstr.2021.100341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sikora E., Bielak-Zmijewska A., Dudkowska M., et al. Cellular senescence in brain aging. Front Aging Neurosci. 2021;13 doi: 10.3389/fnagi.2021.646924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tchkonia T., Kirkland J.L. Aging, cell senescence, and chronic disease: emerging therapeutic strategies. JAMA. 2018;320(13):1319–1320. doi: 10.1001/jama.2018.12440. [DOI] [PubMed] [Google Scholar]
- 14.Wissler Gerdes E.O., Zhu Y., Weigand B.M., et al. Cellular senescence in aging and age-related diseases: implications for neurodegenerative diseases. Int Rev Neurobiol. 2020;155:203–234. doi: 10.1016/bs.irn.2020.03.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhang P., Kishimoto Y., Grammatikakis I., et al. Senolytic therapy alleviates Abeta-associated oligodendrocyte progenitor cell senescence and cognitive deficits in an Alzheimer's disease model. Nat Neurosci. 2019;22(5):719–728. doi: 10.1038/s41593-019-0372-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang X., Pearsall V.M., Carver C.M., et al. Rejuvenation of the aged brain immune cell landscape in mice through p16-positive senescent cell clearance. Nat Commun. 2022;13(1):5671. doi: 10.1038/s41467-022-33226-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Farr J.N., Xu M., Weivoda M.M., et al. Targeting cellular senescence prevents age-related bone loss in mice. Nat Med. 2017;23(9):1072–1079. doi: 10.1038/nm.4385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fielding R.A., Atkinson E.J., Aversa Z., et al. Associations between biomarkers of cellular senescence and physical function in humans: observations from the lifestyle interventions for elders (LIFE) study. GeroScience. 2022;44(6):2757–2770. doi: 10.1007/s11357-022-00685-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fielding R.A., Atkinson E.J., Aversa Z., et al. Biomarkers of cellular senescence predict the onset of mobility disability and are reduced by physical activity in older adults. J Gerontol A Biol Sci Med Sci. 2024;79(3) doi: 10.1093/gerona/glad257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Justice J.N., Gregory H., Tchkonia T., et al. Cellular senescence biomarker p16INK4a+ cell burden in thigh adipose is associated with poor physical function in older women. J Gerontol A Biol Sci Med Sci. 2018;73(7):939–945. doi: 10.1093/gerona/glx134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Roos C.M., Zhang B., Palmer A.K., et al. Chronic senolytic treatment alleviates established vasomotor dysfunction in aged or atherosclerotic mice. Aging Cell. 2016;15(5):973–977. doi: 10.1111/acel.12458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xu M., Tchkonia T., Ding H., et al. JAK inhibition alleviates the cellular senescence-associated secretory phenotype and frailty in old age. Proc Natl Acad Sci USA. 2015;112(46):E6301–E6310. doi: 10.1073/pnas.1515386112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kirkland J.L., Tchkonia T. Cellular senescence: a translational perspective. EBioMedicine. 2017;21:21–28. doi: 10.1016/j.ebiom.2017.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Musi N., Valentine J.M., Sickora K.R., et al. Tau protein aggregation is associated with cellular senescence in the brain. Aging Cell. 2018;17(6) doi: 10.1111/acel.12840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ogrodnik M., Zhu Y., Langhi L.G.P., et al. Obesity-induced cellular senescence drives anxiety and impairs neurogenesis. Cell Metab. 2019;29(5):1061–1077.e8. doi: 10.1016/j.cmet.2018.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ogrodnik M., Evans S.A., Fielder E., et al. Whole-body senescent cell clearance alleviates age-related brain inflammation and cognitive impairment in mice. Aging Cell. 2021;20(2) doi: 10.1111/acel.13296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Orr M., Gonzales M., Garbarino V., et al. Senolytic therapy to modulate the progression of Alzheimer's Disease (SToMP-AD) - outcomes from the first clinical trial of senolytic therapy for Alzheimer's disease. Res Sq. 2023;9(1):22–29. doi: 10.14283/jpad.2021.62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gonzales M.M., Garbarino V.R., Marques Zilli E., et al. Senolytic therapy to modulate the progression of alzheimer's disease (SToMP-AD): a pilot clinical trial. J Prev Alzheimers Dis. 2022;9(1):22–29. doi: 10.14283/jpad.2021.62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Justice J.N., Nambiar A.M., Tchkonia T., et al. Senolytics in idiopathic pulmonary fibrosis: results from a first-in-human, open-label, pilot study. EBioMedicine. 2019;40:554–563. doi: 10.1016/j.ebiom.2018.12.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hickson L.J., Langhi Prata L.G.P., Bobart S.A., et al. Senolytics decrease senescent cells in humans: preliminary report from a clinical trial of dasatinib plus Quercetin in individuals with diabetic kidney disease. EBioMedicine. 2019;47:446–456. doi: 10.1016/j.ebiom.2019.08.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Farr J.N., Atkinson E.J., Achenbach S.J., et al. Effects of intermittent senolytic therapy on bone metabolism in postmenopausal women: a phase 2 randomized controlled trial. Nat Med. 2024;30(9):2605–2612. doi: 10.1038/s41591-024-03096-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gonzales M.M., Garbarino V.R., Kautz T.F., et al. Senolytic therapy in mild Alzheimer's disease: a phase 1 feasibility trial. Nat Med. 2023;29:2481–2488. doi: 10.1038/s41591-023-02543-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Millar C.L., Iloputaife I., Baldyga K., et al. Rationale and design of STAMINA: senolytics to alleviate mobility issues and neurological impairments in aging, a geroscience feasibility study. Transl Med Aging. 2023;7:109–117. [Google Scholar]
- 34.Baldyga K., Iloputaife I., Taffet G., et al. Comparison of targeted web-based advertising versus traditional methods for recruiting older adults into clinical trials. J Am Geriatr Soc. 2024;73(1):182–192. doi: 10.1111/jgs.19225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Katz S., Downs T.D., Cash H.R., Grotz R.C. Progress in development of the index of ADL. Gerontol. 1970;10(1):20–30. doi: 10.1093/geront/10.1_part_1.20. [DOI] [PubMed] [Google Scholar]
- 36.Trailmaking Tests A and B. 1944. In: Washington DWDAGsO. [Google Scholar]
- 37.Tombaugh T.N. Trail Making Test A and B: normative data stratified by age and education. Arch Clin Neuropsychol. 2004;19(2):203–214. doi: 10.1016/S0887-6177(03)00039-8. [DOI] [PubMed] [Google Scholar]
- 38.Nasreddine Z.S., Phillips N.A., Bedirian V., et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695–699. doi: 10.1111/j.1532-5415.2005.53221.x. [DOI] [PubMed] [Google Scholar]
- 39.Guralnik J.M., Simonsick E.M., Ferrucci L., et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49(2):M85–M94. doi: 10.1093/geronj/49.2.m85. [DOI] [PubMed] [Google Scholar]
- 40.Steffen T.M., Hacker T.A., Mollinger L. Age-and gender-related test performance in community-dwelling elderly people: six-minute walk test, berg balance scale, timed up & go test, and gait speeds. Phys Ther. 2002;82(2):128–137. doi: 10.1093/ptj/82.2.128. [DOI] [PubMed] [Google Scholar]
- 41.Wang B., Wang L., Gasek N.S., et al. Intermittent clearance of p21-highly-expressing cells extends lifespan and confers sustained benefits to health and physical function. Cell Metab. 2024;36(8):1795–1805.e6. doi: 10.1016/j.cmet.2024.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Krishnamurthy J., Torrice C., Ramsey M.R., et al. Ink4a/Arf expression is a biomarker of aging. J Clin Investig. 2004;114(9):1299–1307. doi: 10.1172/JCI22475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wagner K.D., Wagner N. The senescence markers p16INK4A, p14ARF/p19ARF, and p21 in organ development and homeostasis. Cells. 2022;11(12):1966:1–196626. doi: 10.3390/cells11121966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Liu Y., Sanoff H.K., Cho H., et al. Expression of p16(INK4a) in peripheral blood T-cells is a biomarker of human aging. Aging Cell. 2009;8(4):439–448. doi: 10.1111/j.1474-9726.2009.00489.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.van Loo G., Bertrand M.J.M. Death by TNF: a road to inflammation. Nat Rev Immunol. 2023;23(5):289–303. doi: 10.1038/s41577-022-00792-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Aksnes M., Capogna E., Vidal-Piñeiro D., et al. Matrix metalloproteinases are associated with brain atrophy in cognitively unimpaired individuals. Neurobiol Aging. 2023;131:11–23. doi: 10.1016/j.neurobiolaging.2023.05.012. [DOI] [PubMed] [Google Scholar]
- 47.Chaib S., López-Domínguez J.A., Lalinde-Gutiérrez M., et al. The efficacy of chemotherapy is limited by intratumoral senescent cells expressing PD-L2. Nat Cancer. 2024;5(3):448–462. doi: 10.1038/s43018-023-00712-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bracken K., Hague W., Keech A., et al. Recruitment of men to a multi-centre diabetes prevention trial: an evaluation of traditional and online promotional strategies. Trials. 2019;20(1):366. doi: 10.1186/s13063-019-3485-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ory M.G., Lipman P.D., Karlen P.L., et al. Recruitment of older participants in frailty/injury prevention studies. Prev Sci. 2002;3(1):1–22. doi: 10.1023/a:1014610325059. [DOI] [PubMed] [Google Scholar]
- 50.Borland E., Edgar C., Stomrud E., Cullen N., Hansson O., Palmqvist S. Clinically relevant changes for cognitive outcomes in preclinical and prodromal cognitive stages: implications for clinical alzheimer trials. Neurology. 2022;99(11):e1142–e1153. doi: 10.1212/WNL.0000000000200817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lindvall E., Abzhandadze T., Quinn T.J., Sunnerhagen K.S., Lundström E. Is the difference real, is the difference relevant: the minimal detectable and clinically important changes in the Montreal Cognitive Assessment. Cereb Circ Cogn Behav. 2024;6 doi: 10.1016/j.cccb.2024.100222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jehu D.A., Davis J.C., Madden K., Parmar N., Liu-Ambrose T. Minimal clinically important difference of executive function performance in older adults who fall: a secondary analysis of a randomized controlled trial. Gerontology. 2021;68(7):771–779. doi: 10.1159/000518939. [DOI] [PubMed] [Google Scholar]
- 53.Brach J.S., Perera S., Studenski S., Katz M., Hall C., Verghese J. Meaningful change in measures of gait variability in older adults. Gait Posture. 2010;31(2):175–179. doi: 10.1016/j.gaitpost.2009.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bytyçi I., Henein M.Y. Stride length predicts adverse clinical events in older adults: a systematic review and meta-analysis. J Clin Med. 2021;10(12):2670. doi: 10.3390/jcm10122670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Huang W., Hickson L.J., Eirin A., Kirkland J.L., Lerman L.O. Cellular senescence: the good, the bad and the unknown. Nat Rev Nephrol. 2022;18(10):611–627. doi: 10.1038/s41581-022-00601-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kandhaya-Pillai R., Miro-Mur F., Alijotas-Reig J., Tchkonia T., Kirkland J.L., Schwartz S. TNFα-senescence initiates a STAT-dependent positive feedback loop, leading to a sustained interferon signature, DNA damage, and cytokine secretion. Aging (Albany NY) 2017;9(11):2411–2435. doi: 10.18632/aging.101328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ressler S., Bartkova J., Niederegger H., et al. p16INK4A is a robust in vivo biomarker of cellular aging in human skin. Aging Cell. 2006;5(5):379–389. doi: 10.1111/j.1474-9726.2006.00231.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.