Take Home Message
New multiport surgical robots have the potential to become viable alternatives to the Da Vinci surgical system, offering comparable feasibility, safety, functional, and oncological outcomes. However, the limited literature, which includes only one randomized controlled trial and several nonrandomized retrospective studies primarily based on robot-assisted prostatectomy and robot-assisted partial nephrectomy, must be considered when interpreting these results. New multiport surgical robots represent a potential opportunity to spread the use of robot-assisted surgery globally. Further research, particularly large-scale randomized controlled trials, is needed to validate these findings and fully understand the potential of new multiport surgical robots in clinical practice.
Keywords: Hinotori, Hugo robot-assisted surgery, KangDuo, Partial nephrectomy, Prostatectomy, Robotic surgery, Senhance, Toumai, Urology, Versius
Abstract
Background and objective
Robot-assisted surgery (RAS) has steadily become more prevalent in urology. The Da Vinci multiport surgical robot (DVM-SR) continues to lead the field. In recent years, new multiport surgical robots (NM-SRs) have been introduced to the market; however, their safety and efficacy remain unassessed. This study aims to give a comprehensive evaluation of the perioperative, oncological, and functional outcomes of NM-SRs and a comparison with the DVM-SR.
Methods
A systematic search was performed in PubMed, Scopus, Web of Science, Embase, and clinicaltrial.gov to identify studies that evaluate NM-SRs in major urological surgeries assessing perioperative, functional, and oncological endpoints. A meta-analysis was performed comparing NM-SRs with the DVM-SR for safety, and functional and oncological outcomes.
Key findings and limitations
Seventy-four studies involving 5487 patients were included in the review. Nine platforms were studied: Hinotori, Hugo RAS, Revo-I, Versius, Avatera, Senhance, KangDuo Surgical Robot-01, Dexter, and Toumai. NM-SRs were used to perform robot-assisted radical prostatectomy (RARP; 41 studies), partial nephrectomy (RAPN; 14 studies), radical nephrectomy (RARN; four studies), adrenalectomy (four studies), nephroureterectomy (two studies), RARN and thrombectomy (one study), colpopexy (four studies), pyeloplasty (seven studies), simple nephrectomy (four studies), simple prostatectomy (three studies), and ureteral surgery (four studies). Cystectomies with NM-SRs were described only in case reports and were excluded. The comparative analysis between NM-SRs and the DVM-SR showed similar outcomes in terms of intraoperative SATAVA grade ≥2 complications (odds ratio [OR] 0.89, 95% confidence interval [CI] 0.25, 3.1, p = 0.9 for RARP and OR 0.59, 95% CI 0.11, 3.3, p = 0.5 for RAPN), postoperative high-grade complications (Clavien-Dindo ≥IIIa, OR 0.85, 95% CI 0.4, 2, p = 0.7 for RARP and OR 0.59, 95% CI 0.1, 3.3, p = 0.6 for RAPN), and positive surgical margins (OR 0.90, 95% CI 0.72, 1.1, p = 0.3 for RARP and OR 1.65, 95% CI 0.3, 9.1, p = 0.6 for RAPN). For patients undergoing RARP, biochemical recurrence and urinary continence rates at 3 mo were comparable (OR 0.99 [95% CI 0.5, 1.8, p = 1] and OR 0.99 [95% CI 0.77, 1.3, p = 0.9], respectively). The achievement of the trifecta for RAPN appeared to be similar between the included studies on NM-SRs and the DVM-SR (OR 1.3, 95% CI 0.4, 4.4, p = 0.7). The small sample size of the included studies and the preliminary nature of the results represent the major limitations.
Conclusions and clinical implications
When compared with the DVM-SR, NM-SRs may offer similar safety, and oncological and functional outcomes across most surgeries for both benign and malignant diseases. Further research is needed to explore the potential of NM-SRs, given the promising initial findings.
Patient summary
New multiport surgical robots (NM-SRs) appear to be safe and effective compared with the Da Vinci surgical robotic system. However, further research is required to thoroughly assess their long-term outcomes and cost effectiveness. NM-SRs represent an opportunity to spread the use of robot-assisted surgery globally.
1. Introduction
Over the past decade, robot-assisted surgery (RAS) has revolutionized urology, by providing surgeons with enhanced visualization, precision, and control during complex procedures [1]. The introduction of the groundbreaking Da Vinci (Intuitive Surgical, Sunnyvale, California, US) multiport surgical robot (DVM-SR) in 2000 marked a pivotal moment in this evolution, establishing the DVM-SR as the cornerstone of robotic-assisted urological surgery.
The DVM-SR, equipped with seven degrees of freedom, tremor filtration, three-dimensional visualization, and precise camera control, has mitigated the historical challenges associated with laparoscopy, particularly the steep learning curve related to intricate tasks such as suturing and anastomosis [2].
As RAS gains widespread adoption, development of new robotic systems has gained momentum. In recent years, companies are competing to advance the field with new multiport surgical robots (NM-SRs), such as the Hugo RAS (Medtronic, Minneapolis, Minnesota, US), Versius (CMR Surgical, Cambridge, UK), and Senhance (TransEnterix, Durham, North Carolina, US), each claiming to surpass existing standards in safety and feasibility for urological procedures [3]. However, the safety, and functional and oncological outcomes of these NM-SRs remain largely unproven. Given the substantial costs associated with RAS and the shifting market landscape as the Intuitive DVM-SR loses its monopoly, it is crucial to determine whether hospitals should invest in these NM-SRs.
For health care providers, recognizing the importance of allocating time and resources to master these NM-SRs is critical. This understanding is also essential for making informed decisions about adopting alternative robotic systems beyond the well-established DVM-SR platform.
While individual studies have appraised these platforms separately, one systematic review (SR) focused only on robot-assisted radical prostatectomy (RARP) performed with Hugo RAS has been published [4]. However, a comprehensive and systematic evaluation of the operative times, complications, and early oncological and functional outcomes associated with the use of NM-SRs in urology remains lacking in the current literature.
To address the existing gap in the literature, this SR aims to offer a comprehensive understanding of the perioperative, oncological, and functional outcomes associated with NM-SRs, also comparing these outcomes with those of the established DVM-SR through a meta-analysis.
2. Methods
2.1. Search strategy
This SR was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement [5].
The review protocol was registered on the International Prospective Registry of Systematic Review (PROSPERO; ID “CRD42023451138”). A systematic search in Medline/PubMed, Scopus, Web of Science, Embase through OVID, and clinicaltrial.gov was performed in August 2024 for relevant studies including NM-SRs. Details on the search strategy can be found in the Supplementary material.
2.2. Study inclusion and exclusion criteria, and outcomes
We used the PICO framework to formulate the research question and inclusion criteria for the review [6]. We included studies on patients with a urological disease (population) treated with one of the NM-SRs (intervention). The comparison group, when applicable, was using the DVM-SR (comparator). The primary outcome of the study was the evaluation of operative times, estimated blood loss (EBL), complications, system failures, functional and oncological endpoints of the major urological surgeries (RARP, robot-assisted partial nephrectomy [RAPN], and other RASs for malignant or benign diseases). Studies were screened based on title and abstract; a full-text review was limited to those considered appropriate for the SR. Reference lists from the identified publications were also evaluated. The exclusion criteria comprised papers focused only on the DVM-SR, studies on single-port surgical robots, studies on animal models and/or dry lab and/or cadavers, manuscripts on the use of a surgical robot in fields other than urology, letters, editorials, conference abstracts, and reviews. Duplicate works were also discarded. Covidence (Veritas Health Innovation, Melbourne, Australia) was used to screen and organize all the studies. Manuscripts identified through the screening were based on the following multiport robotic platforms defined a priori after an extensive literature search: Hinotori surgical robot system (Medicaroid, Kobe, Japan), Hugo RAS, Revo-I surgical robot (Meerecompany, Hwaseong, Gyeonggi, South Korea), Versius surgical robot, Avatera robotic surgical system (Avateramedical GmbH, Jena, Germany), Senhance surgical system, KangDuo Surgical Robot-01 system (SuZhou KangDuo Robot Co., Suzhou, China), Dexter surgical robot (Distalmotion, Épalinges, Vaud, Switzerland), and Toumai laparoscopic surgical robot (MedBot, Shanghai, China).
Four reviewers (G.R. A.Ta., A.Tu., and G.S.) selected and screened abstracts and full-text articles in pairs. Conflicts were reviewed independently and arbitrated by a senior author (F.Z.).
2.3. Data abstraction
Three reviewers (A. Tu., F.M., and G.S.) performed the data extraction, which was verified by two authors (G.R. and F.Z.) to minimize the intraexaminer variability. The following variables were recorded for each eligible paper: primary author, year of publication, number of patients, type of surgery, baseline, pre- and intraoperative features, and postoperative complications categorized based on the Clavien-Dindo (CD) score. For oncological surgeries, pathological information and recurrences during follow-up were also collected. Finally, continence and potency rates after RARP, and success rate for RAPN (indicated as the achievement of the trifecta) were also abstracted. When it was not specified or determined in the manuscripts concerning RAPN, the standard definition used for the trifecta was as follows: negative surgical margins, warm ischemia time <20 min, and no urological complications [7].
2.4. Risk of bias assessment
The risk of bias (RoB) assessment was completed by two authors (G.R. and U.G.). Cochrane RoB2 [8] and ROBINS-I [9] tools were used, respectively, for randomized and nonrandomized studies.
The quality of single-arm studies was ascertained using the European Association of Urology (EAU) guidelines for systematic case series review [10].
2.5. Data synthesis and analysis
Owing to heterogeneity among the single-arm studies, pooled estimates were not obtained. Continuous variables were summarized as median and interquartile range (IQR), median and range, or mean and standard deviation (SD). The number of events with percentages was estimated for categorical, binary, and discrete variables. When raw data were available, mean and SD were calculated [11].
A quantitative analysis of comparative studies of NM-SRs versus the DVM-SR was performed when methodologically appropriate. If patient populations overlapped across two or more included studies, those with longer follow-up periods and larger cohort of patients were preferred to studies with shorter follow-up periods and smaller sample size. Data that were not available in the most recent studies were retrieved from the older manuscripts. A random-effect or fixed-effect model was chosen based on the heterogeneity assumption. Mean and SD were estimated when these were not available using the methods of Luo et al [12] and Wan et al [13]. Mean differences (MDs) were calculated, and the restricted maximum-likelihood method was used to determine the heterogeneity variance in the random-effect model. When a fixed-effect model was used for continuous data, the inverse variance method was applied. The Mantel-Haenszel method was used in the fixed-effect model to estimate odds ratio (OR) for dichotomous variables. Forest plots were generated for the visualization of the results. A subgroup analysis was carried out when appropriate to explore heterogeneity among subgroups.
The p values were considered statistically significant for the overall effect size when these were below 0.05. All the analyses were performed with STATA version 18.0 (2023; StataCorp., College Station, TX, USA).
3. Results
3.1. Study selection
Supplementary Fig. 1 summarizes the flowchart of the SR.
A total of 1375 publications were retrieved. After selection, a total of 74 studies were identified through the literature search. Of these, 45 were single-arm studies, 27 were double-arm comparative studies, and two were ongoing three-arm clinical trials. Among the published comparative studies, three compared the new multiport robotic systems with conventional laparoscopic surgery [14], [15], [16], and 24 involved the Da Vinci system as a comparator, with one of these being a randomized controlled trial (RCT). The SR included a total of 5487 patients. The studies were distributed as follows: ten focused on the Hinotori surgical robot system, 31 on the Hugo RAS, two on the Revo-I surgical robot, eight on the Versius surgical robot, two on the Avatera robotic surgical system, ten on the Senhance surgical system, seven on the KangDuo Surgical Robot-01 system, one on the Dexter surgical robot, and one on the Toumai laparoscopic surgical robot. Fig. 1 summarizes the main characteristics, prices, and availability of the robots included in this SR.
Fig. 1.
Key elements, costs, and distribution of Da Vinci Xi and new multiport surgical robots.
3.2. RoB assessment
The only RCT included in the review is considered to have a low RoB (Supplementary Fig. 2). Most of the double-arm studies are nonrandomized, with six of these employing methods to adjust for relevant confounders (Supplementary Fig. 3). However, only three of these studies can be considered to have a moderate RoB. Twenty-two single-arm studies are classified as having a low RoB according to the EAU guidelines for systematic case series (Supplementary Fig. 4).
3.3. Qualitative data synthesis for single-arm studies
Table 1, Table 2, and Supplementary Tables 1 and 2 provide a descriptive analysis and a narrative synthesis for single-arm studies.
Table 1.
Baseline characteristics and intraoperative outcomes of robot-assisted surgeries for malignant diseases
| Author | Year | Design | New robot | Comparator | Cases | Age (yr) |
PSA (ng/ml) |
cT stage n (%) |
ASA n (%) or median |
OT (min) |
DT (min) |
CT (min) |
EBL (ml) |
IOC SATAVA grade ≥2 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RARP—single-arm studies | ||||||||||||||
| Hinata [26] (TP) |
2022 | Single-arm clinical trial | Hinotori | No | 30 | Median: 72 (range: 52–77) | Mean: 10 (SD: 9.2) | cT1: 1 (3) cT2: 28 (93) cT3: 1 (3) |
– | Mean: 231.8 (SD: 58.12) | – | Mean: 167.8 (SD: 52.8) | Mean: 199 (SD: 157.94) | 0/30 (0) |
| Alfano [17] (TP) |
2023 | Case series | Hugo RAS | No | 15 | Median: 62 (IQR: 59–67) | Median: 7.3 (IQR: 4.3 – 8.1) | cT1: 8 (53) cT2a: 4 (27) cT2b: 2 (13) cT2c: 1 (7) |
– | Median: 235 (IQR: 213–271) | Median: 7 | – | Median: 300 (IQR: 100–310) | 0/15 (0) |
| Bravi [19] (TP) |
2023 | Case series | Hugo RAS | No | 112 | Median: 65 (IQR: 60–70) | Median: 7.9 (IQR: 5.8–10.7) | cT1: 71 (63) cT2–3: 41 (37) |
– | Median: 180 (IQR: 145–200) | – | Median: 150 (IQR: 145–175) | Median: 400 (IQR: 250–575) | 0/112 (0) |
| Totaro [39] (TP) |
2024 | Single-arm clinical trial | Hugo RAS | No | 132 | Mean: 66.1 (SD: 6.8) | Mean: 10.72 (SD: 10.81) | cT1: 105 (80) cT2-cT3: 27 (20) |
Median: 2 (IQR: 0) | Mean: 189.3 (SD: 57.3) | Mean: 10 (SD: 2) | Mean: 124 (SD: 48) | Mean: 100 (SD: 100) | 0/132 (0) |
| Ragavan [33] | 2022 | Case series | Hugo RAS | No | 3 | Mean: 67.7 (SD: 8) | Mean: 18 (SD: 8.18) | – | – | Mean: 185 (SD: 10.53) | Mean: 10.67 (SD: 4.2) | – | Mean: 133.3 (SD: 23.57) | 0/3 (0) |
| Marques-Monteiro [29] (EP) |
2023 | Cross sectional | Hugo RAS | No | 16 | Median: 68 (IQR: 57–65) | Median: 10.8 (IQR: 5.6–14.2) | – | – | Median: 211 (IQR: 145–200) | Median: 4.5 (IQR: 4.1–5.2) | Median: 152 (IQR: 110–175) | Median: 200 (IQR: 150–400) | 0/16 (0) |
| Veccia [40] (TP) |
2024 | Case series | Hugo RAS | No | 10 | Median: 64.5 (SD: 5.8) | Mean:13 (SD: 17.75) | – | – | – | Median: 12.5 (IQR: 12–15) | Median: 229 (IQR: 174–235) | Median: 425 (IQR: 350–650) | 0/10 (0) |
| Territo [37] (TP and EP) |
2023 | Single-arm clinical trial | Hugo RAS | No | TP: 17 | TP: median: 64 (IQR: 59–69) | TP: median: 6.4 (IQR: 5.1–9.4) | TP: cT1: 10 (59) cT2: 7 (41) |
– | – | TP: mean: 12 (IQR: 7–16) | TP: median: 185 (IQR: 177–192) | TP: median: 200 (IQR: 150–250) | TP: 0/17 (0) |
| EP: 3 | EP: median: 61 (SD: 4.6) | EP: mean: 13 (SD: 14.5) | EP: cT1: 2 (67) cT2: 1 (33) |
– | – | EP: mean: 15 (SD: 4.1) | EP: mean: 175 (SD: 18.7) | EP: mean: 166.7 (SD: 62.4) | EP: 0/3 (0) | |||||
| Andrade [18] (TP) |
2024 | Case series | Hugo RAS | No | 19 | Mean: 62.1 (SD: 8.5) | Mean: 10 (SD 17.16) | – | I: 2 (11) II: 16 (84) III: 1 (5) |
– | Median: 10 (IQR: 5–20) | Median: 127 (IQR: 110–150) | – | 0/19 (0) |
| Dell'Oglio [22] (TP) |
2025 | Case series | Hugo RAS | No | 26 | Median: 64 (IQR: 61–68) | Median: 6.3 (IQR: 3.9–9.2) | cT1c: 12 (46) cT2a: 9 (35) cT2b: 3 (11) cT2c: 2 (8) |
– | Median: 215 (IQR: 181–262) | Median: 9 (IQR: 8–10) | Median: 156 (IQR: 143–197) | Median: 90 (IQR: 54–100) | 0/26 (0) |
| Ou [30] | 2023 | Case series | Hugo RAS | No | 12 | Median: 71.5 (SD: 13.2) | Median: 9.67 (IQR: 7.96) | – | II: 5 (42) III: 7 (58) |
– | Mean: 27.6 (SD: 9.6) | Mean: 145 (SD: 20.9) | Mean: 193 (SD: 226) | 0/12 (0) |
| Takahara [36] (TP) |
2024 | Case series | Hugo RAS | No | 13 | Median: 68 (IQR: 66–74) | Median: 5.8 (IQR: 4.8–9.9) | cT1c: 1 (8) cT2a: 8 (62) cT2b: 2 (15) cT2c: 2 (15) |
– | Median: 197 (IQR: 187–228) | Median: 7 (IQR: 6–10) | Median: 134 (IQR: 125–157) | Median: 150 (IQR: 80–250) | 0/13 (0) |
| Fan [24] (EP) |
2022 | Single-arm clinical trial | KangDuo | No | 16 | Median: 66 (range: 58–72) | Median: 6.67 (IQR: 0.88–17.98) | cT2b: 5 (31) cT2c: 11 (69) |
– | – | Median: 5.9 (range: 2.5–11.5) | Median: 87 (range: 70–120) | Median: 50 (range: 10–200) | – |
| Xiong [42] (TP and EP) |
2023 | Case series | KangDuo | No | TP: 25 EP: 16 |
Median: 53 (range: 16–75) | TP: median: 11.20 (range: 5.59–50.00) EP: median: 9.58 (range: 4.68–18.68) |
– | – | TP: median: 147 (range: 111–197) EP: median: 127 (range: 107–159) |
– | – | TP: mean: 50 (range: 20–200) EP: mean: 50 (range: 10–200) |
0/41 (0) |
| Chang [20] (TP) |
2018 | Single-arm clinical trial | Revo-I | No | 17 | Median: 72 (IQR: 62.5–75) | Median: 8 (SD: 7.68) | cT1c: 6 (35) cT2: 8 (47) cT3a: 3 (18) |
I: 1 (6) II: 7 (41) III: 9 (53) |
– | Median: 8 (IQR: 7–11) | Median: 92 (IQR: 85.5–133) | Median: 200 (IQR: 200–300) | 0/17 (0) |
| Gkeka [25] (EP) |
2024 | Single arm clinical trial | Senhance | No | 14 | Median: 66.5 (range: 57–73) | Median: 7.8 (range: 3.3–15.1) | cT2: 5 (36) cT3: 9 (64) |
– | – | Median: 10 (range: 5–40) | Median: 104 (range: 90–121) | – | 0/14 (0) |
| Venckus [41] | 2021 | Single-arm clinical trial | Senhance | No | 127 | Median: 61 (IQR: 37–73) | Median: 5.5 (IQR: 2.0–26.8) | cT1: 42 (33) cT2: 66 (52) cT3: 19 (15) |
Median: 180 (IQR: 150–215) | – | – | Median: 250 (IQR: 175–400) | – | |
| Hudolin [27] (EP) |
2023 | Case series | Senhance | No | 200 | Median: 65 (range: 41–79) | Median: 6.9 (range: 1–29.8) | cT1: 115 (58) cT2: 79 (40) |
– | Median: 190 (IQR: 120–315) | Median: 3 (IQR: 1–14) | Median: 130 (IQR: 110–155) | Median: 250 (IQR: 15–200) | 2/200 (2) |
| Kulis [28] (EP and TP) |
2024 | Single-arm clinical trial | Senhance | No | 375 | Mean: 62.8 (SD: 6.6) | – | – | – | Median: 190 (IQR: 167.5–215) | Median: 3 (IQR: 2–5) | – | – | 4/375 (1) |
| Reeves [34] | 2022 | Case series | Versius | No | 4 | Mean: 66 | – | – | Mean: 335 (range: 258–440) | – | Mean: 272 (range: 195–377) | – | 0/4 (0) | |
| Sighinolfi [35] | 2024 | Case series | Versius | No | 22 | – | – | – | – | – | – | Median: 201 (IQR: 130–242) | Median: 140 (IQR: 100–550) | 0/22 (0) |
| De Maria [21] | 2024 | Case series | Versius | No | 18 | Mean: 70 (IQR: 55–76) | Mean: 15 (range: 7–25) | cT1: 6 (33) cT2: 12 (67) |
– | Median: 213 (IQR: 186–226) | – | Median: 201 (IQR: 170–242) | Median: 140 (IQR: 100–550) | 0/18 (0) |
| Polom [32] (TP) |
2024 | Case series | Versius | No | 58 | Median: 66.9 (range: 52–75) | Mean: 9.8 (range: 1.9 –29.4) | cT1: 43 (74) a cT2: 15 (26) a |
– | Median: 213 (range: 128–348) | – | Median: 150.9 (range: 62–279) | Mean: 437 (range: 210–2050) | 3/58 (5) |
| Dibitetto [23] (EP) |
2024 | Case series | Versius | No | 53 | Mean: 67 (range: 48–73) | Mean: 6.6 (range: 4.3–20.5) | – | – | Mean: 130 (range: 80–260) | Median: 15 (range: 8–30) | Mean: 100 (range: 63–240) | Mean: 100 (range: 30–300) |
0/53 (0) |
| Thillou [38] (TP) |
2024 | Case series | Avatera | No | 10 | Median: 67 (IQR: 62–71) | Median: 8.5 (IQR: 5.0–12.9) | cT1: 4 (40) cT2a: 1 (10) cT2b: 4 (40) cT2c: 1 (10) |
II: 7 (70) III: 3 (30) |
Median: 230 (IQR: 226–235) | – | – | Median: 655 (IQR: 425–788) | 0/10 (0) |
| Pokhrel [31] (TP) |
2024 | Single-arm clinical trial | Toumai | No | 3 | Median: 68 (range: 60–75) | – | – | Median: 210 (range: 154–245) | Median: 20 (range: 17–29) | – | Median: 50 (range: 25–100) | 0/3 (0) | |
| RARP—double-arm studies | ||||||||||||||
| Sasaki [79] (TP) |
2024 | Cohort retrospective | Hinotori | 48 | Median: 72 (IQR: 68–75) |
Median: 8.6 (IQR: 6.0–14.0) |
– | – | Median: 260 (IQR: 235–308) | – | Median: 205 (IQR: 180–226) | – | – | |
| Da Vinci | 46 | Median: 72 (IQR: 69–76) | Median: 8.7 (IQR: 6.9–11.4) | – | – | Median: 239 (IQR: 207–283) | – | Median: 172 (IQR: 150–219) | – | – | ||||
| Kohjimoto [72] (TP) |
2024 | Cohort retrospective | Hinotori | 60 | Median: 70 (IQR: 65–74) | Median: 7.0 (IQR: 5.6–10.4) | cT1: 1 (2) cT2a-b: 18 (30) cT2c: 36 (60) cT3: 5 (8) |
– | Median: 236 (IQR: 217–267) | – | Median: 178 (IQR: 210–256) | Median: 285 (IQR: 151–455) | 0/60 (0) | |
| Da Vinci | 100 | Median: 71 (IQR: 65–74) | Median: 9.6 (IQR: 6.4–13.9) | cT1: 4 (4) cT2a-b: 27 (27) cT2c: 48 (48) cT3: 21 (21) |
– | Median: 249 (IQR: 213–295) | – | Median: 202 (IQR: 152–237) | Median: 205 (IQR: 115–408) | 0/100 (0) | ||||
| Nakayama [75] (TP) |
2024 | Cohort retrospective | Hinotori | 97 | Median: 69 (IQR: 63.5–73) | Median: 7.2 (IQR:5.4–10.8) | cT1: 28 (29) cT2: 59 (61) cT3: 10 (10) |
I: 19 (19.6) II: 74 (76.3) III: 4 (4.1) |
– | – | Median: 173 (IQR: 141–207) | Median: 20 (IQR: 10–50) | 0/97 (0) | |
| Da Vinci | 246 | Median: 69 (IQR: 65–72) | Median: 7.4 (IQR 5.4–11.3) | cT1: 42 (17) cT2: 160 (65) cT3: 44 (18) |
I: 53 (21.5) II: 176 (71.5) III: 17 (6.9) |
– | – | Median: 144 (IQR: 112–190) | Median: 25 (IQR: 10–90) | 0/246 (0) | ||||
| Ragavan [78] (TP) |
2023 | Cohort prospective | Hugo RAS | 17 | Median: 68 (IQR: 66–72) | Median: 12.39 (IQR: 8.8–27) | cT2: 11 (65) cT3: 6 (35) |
– | Median: 195 (IQR: 180–240) | Median: 10 | Median: 170 (IQR: 160–205) | – | 0/17 (0) | |
| Da Vinci | 17 | Median: 68 (IQR 65–73) | Median: 22 (IQR 7.38–42) | cT2: 9 (53) cT3: 8 (47) |
– | Median: 210 (IQR: 210–240) | – | Median: 190 (IQR: 180–210) | – | 0/17 (0) | ||||
| Antonelli [67] (TP) |
2024 | Double-arm nonrandomized clinical trial | Hugo RAS | 50 | Mean: 65.9 (SD: 5.9) | Median: 7.7 (IQR: 5.9–11.0) | – | I: 0 (0) II: 39 (78) III: 11 (22) |
Median: 180 (IQR: 150–200) | Median: 11 (IQR: 8–14) | – | Median: 300 (IQR: 150–400) | 0/50 (0) | |
| Da Vinci | 50 | Mean: 66.4 (SD: 5.5) | Median: 5.9 (IQR: 4.8–8.7) | – | I: 3 (6) II: 38 (76) III: 9 (18) |
Median: 165 (IQR: 130–200) | Median: 6 (IQR: 4–7) | – | Median: 200 (IQR: 150–300) | 1/50 (2) | ||||
| Bravi [68] | 2024 | Cohort retrospective | Hugo RAS | 164 | Median: 65 (IQR: 60–70) | Median: 8.0 (IQR: 5.7–11.1) | cT1: 109 (66) cT2–3: 55 (34) |
– | Median: 180 (IQR: 150–200) | – | – | Median: 400 (IQR: 250–500) | – | |
| Da Vinci | 378 | Median: 66 (IQR: 61–71) | Median: 7.6 (IQR: 5.1–11.3) | cT1: 283 (75) cT2–3: 95 (25) |
– | Median: 165 (IQR: 130–200) | – | – | Median: 350 (IQR: 200–500) | – | ||||
| Olsen [76] | 2024 | Cohort prospective | Hugo RAS | 19 | Median: 66 (IQR: 63–73) | ≤10:12 (63) 10–20: 3 (16) ≥20: 4 (21) |
≤cT2a: 10 (53) cT2b: 4 (21) ≥cT2c: 5 (26) |
Median: 2 (IQR: 2–2) | – | Median: 8 (IQR: 6–11) | Median: 97 (IQR: 87–120) | Median: 300 (IQR: 150–400) | 0/19 (0) | |
| Da Vinci | 11 | Median: 59 (IQR: 58–62) | ≤10: 7 (64) 10–20: 4 (36) ≥20: 0 (0) |
≤cT2a: 8 (73) cT2b: 2 (18) ≥cT2c: 1 (9) |
Median: 2 (IQR: 1–2) | – | Median: 3 (IQR: 2–5) | Median: 89 (IQR: 77–94) | Median: 200 (IQR: 100–350) | 0/11 (0) | ||||
| Ou [77] | 2024 | Cohort retrospective | Hugo RAS | 30 | Median: 66.5 (SD: 10) | Median: 8.81 (IQR: 7.66) | cT2a: 16 (53) cT2b: 5 (17) cT2c: 9 (30) |
II: 14 (46.7) III: 16 (53.3) |
– | – | Mean: 146.5 (SD: 36.5) | Mean: 187.5 (SD: 242.5) | 0/30 (0) | |
| Da Vinci | 30 | Median: 67.5 (SD: 11.5) | Median: 9.46 (IQR: 9.53) | cT2a: 13 (43) cT2b: 11 (37) cT2c: 6 (20) |
II: 14 (46.7) III: 16 (53.3) |
– | – | Mean: 133 (SD: 36) | Mean: 200 (SD: 177.5) | 0/30 (0) | ||||
| Gandi [71] | 2024 | Cohort retrospective | Hugo RAS | 103 | Median: 68 (IQR: 62–72) | Median: 7.89 (IQR: 5.50–11.82) |
– | – | Median: 170 (IQR: 147.5–195.5) | – | – | Median: 100 (IQR: 100–150) | – | |
| Da Vinci | 276 | Median: 68 (IQR: 63–72) | Median: 7.50 (IQR: 5.60–10.03) | – | – | Median: 170 (IQR: 147–206) | – | – | Median: 100 (IQR: 100–155) | – | ||||
| Menendez [74] (TP) |
2024 | Cohort prospective | Hugo RAS | 75 | Mean: 65.8 (SD: 8.1) | Mean: 6.41 (SD: 1.90) | cT1: 51 (68) cT2–3: 24 (32) |
– | Mean: 138.95 (SD: 45.5) | Mean: 18.6 (SD: 8) | – | Mean: 251.5 (SD: 77) | 0/75 (0) | |
| Da Vinci | 75 | Mean: 66.3 (SD: 7) | Mean: 6.88 (SD: 2.76) | cT1: 55 (73) cT2–3: 20 (23) |
– | Mean: 145.34 (SD: 43.1) | Mean: 10.45 (SD: 3.1) | – | Mean: 328.3 (SD: 56.9) | 0/75 (0) | ||||
| Fan [70] (EP) |
2023 | Cohort retrospective | KangDuo | 16 | Median: 66 (range: 58–75) | Median: 6.67 (range: 0.88–17.98) |
cT2b: 5 (31) cT2c: 11 (69) |
– | Median: 127 (range: 107–159) | – | – | Median: 50 (range: 10–200) | 0/16 (0) | |
| Da Vinci | 16 | Median: 69 (range: 57–78) | Median: 6.28 (range: 0.03–26.18) | cT2a: 3 (19) cT2b: 4 (25) cT2c: 9 (56) |
– | Median: 70.5 (range: 54–90) | – | – | Median: 50 (range: 0–200) | 0/16 (0) | ||||
| Alip [66] | 2022 | Cohort retrospective | Revo-I |
33 | Median: 71 (SD: 6) | Median: 6.64 (SD: 6.35) | ≤cT2: 25 (74) ≥cT3: 8 (24) |
I: 3 (9) II: 16 (48) III: 14 (43) |
Mean: 126.18 (SD: 55.2) | – | Mean: 89.45 (SD: 31.3) | Mean: 284.2 (SD: 262.3) | 0/33 (0) | |
| Da Vinci | 33 | Median: 72 (SD: 9) | Median: 6.0 (SD: 3.20) | ≤cT2: 23 (70) ≥ cT3: 10 (30) |
I: 2 (6) II: 16 (48) III: 15 (46) |
Mean: 92.42 (SD: 26.12) | – | Mean: 49.57 (SD: 14.29) | Mean: 206.4 (SD: 165.9) | 0/33 (0) | ||||
| Lin [73] (TP) |
2024 | Single-arm clinical trial and cohort retrospective matching | Senhance | 63 | Median: 66 (IQR: 64–71 | Median: 11.3 (IQR: 7.4–19.0 | cT1: 23 (36.5) cT2: 23 (36.5) cT3: 17 (27) |
Median: 231 (IQR: 198–272.5) | – | – | Median: 180 (IQR: 100–285) | – | ||
| Da Vinci | 63 | Median: 66 (IQR: 62–68.5) | Median: 11.9 (IQR: 7.4–19.4) | cT1: 23 (36.5) cT2: 23 (36.5) cT3: 17 (27) |
Median: 265 (IQR: 234–306) | – | – | Median: 180 (IQR: 92.5–285) | – | |||||
| Kulis [15] (EP) |
2022 | Cohort prospective | Senhance | 107 | Median: 65 (IQR: 60–68) | Median: 6.85 (IQR: 5.13–8.8) | cT1: 63 (59) cT2: 44 (61) |
– | Median: 195 (IQR: 180–218) | Median: 5 (IQR: 3–5) | – | Median: 300 (IQR: 200–500) | 0/107 (0) | |
| Laparoscopy | 61 | Median: 64 (IQR: 59–68) | Median: 5.97 (IQR: 4.67–7.82) | cT1: 31 (51) cT2: 30 (49) |
– | – | – | – | Median: 350 (IQR: 280–400) | 0/61 (0) | ||||
| Dibitetto [69] (EP) |
2024 | Cohort retrospective | Versius | 53 | Median: 66 (IQR: 61–72) | Median: 7.8 (IQR: 5.2–10.4) | cT1: 45 (85) ≥cT2: 8 (15) |
– | Median: 170 (IQR: 158–202) | – | – | Median: 350 (range: 100–600) | – | |
| Da Vinci | 53 | Median: 65 (IQR: 57–74) | Median: 8.1 (IQR: 5.1–12.7) | cT1: 40 (75) ≥cT2: 13 (25) |
– | Median: 96 (IQR: 88–109) | – | – | Median: 250 (range: 70–550) | – | ||||
| RAPN—single-arm studies | ||||||||||||||
| Miyake [46] (TP and RP) |
2023 | Case series | Hinotori | No | 30 | Median: 62 (range: 46–84) | – | cT1a: 23 (76) cT1b: 7 (24) |
– | Median: 179 (IQR: 122–268) | – | Median: 106 (range: 73–191) | Median: 39 (range: 5–312) | 0/30 (0) |
| Gallioli [44] (TP) |
2023 | Single-arm clinical trial | Hugo RAS | No | 10 | Mean: 65.8 (SD: 15) | – | cT1a: 8 (80) cT1b: 2 (20) |
– | Mean: 141.2 (SD: 32.5) | Mean: 12.1 (SD: 5.6) | – | Mean: 101 (SD: 43) | 1/10 (10) |
| Prata [48] (TP) |
2023 | Single-arm clinical trial | Hugo RAS | No | 25 | Median: 69 (IQR: 60–73) | – | cT1a: 19 (76) cT1b: 4 (16) cT2: 2 (8) |
I: 1(4) II: 18 (72) III: 5 (20) IV: 1 (4) |
– | Median: 5 (IQR: 5–6) | Median: 90 (IQR: 68–135.75) | Median: 175 (IQR: 100–400) | 0/25 (0) |
| Prata [47] (TP) |
2024 | Case series | Hugo RAS | No | 27 | Median: 68 (IQR: 57–73) | – | cT1a: 21 (77.8) cT1b: 3 (11.1) cT2a: 2 (7.4) cT2b: 1 (3.7) |
I: 1 (3.7) II: 17 (63) III: 9 (33.3) |
Median: 91 (IQR: 50–149) | – | – | Median: 150 (IQR: 50–450) | 0/27 (0) |
| Andrade [18] (TP) |
2024 | Case series | Hugo RAS | No | 4 | Median: 60 (IQR: 58.5–63.5) | – | cT1a: 3 (75) cT1b: 1 (25) |
I: 1 (25) II: 3 (75) |
– | Median: 12 (IQR: 7–19) | Median: 82 (IQR: 80–130) | – | 0/4 (0) |
| Meneghetti [45] (TP) |
2024 | Case series | Versius | No | 13 | Median: 64 (IQR: 55–69) | – | – | – | Mean: 100.6 (SD: 15.4) | – | Mean: 71.1 (SD: 15.5) | Mean: 178.5 (SD: 83.4) | 0/13 (0) |
| Xu [49] (TP and RP) |
2022 | Single-arm clinical trial | KangDuo | No | 17 | Median: 61 (range: 36–72) | – | cT1a: 17/17 (100) | – | Mean: 110 (SD: 37.6) | Mean: 3.3 (range: 2.2–6.3) | Mean: 68.6 (SD: 26) | Median: 50 (range: 50–200) | 0/17 (0) |
| Xiong [42] (TP and RP) |
2023 | Case series | KangDuo | No | TP: 17 RP: 11 |
TP: median: 55 (range: 38–69) RP: median: 52 (range: 33–74) |
– | cT1a: 28 (100) | – | TP: median: 117 (IQR: 75–184) RP: median: 103 (IQR: 88–192) |
TP: median: 3.7 (range: 2.3–9.9) RP: median: 4.8 (range: 3.7–8.8) |
TP: median: 117 (range: 75–184) RP: median: 103 (range: 88–192) |
TP: median: 20 (range: 0–450) EP: median: 10 (range: 0–50) |
0/28 (0) |
| Pokhrel [31] (TP) |
2024 | Single-arm clinical trial | Toumai | No | 4 | Median: 58 (range: 49–76) | – | – | – | Median: 120 (range: 113–145) | – | – | Median: 75 (range: 50–100) | 0/4 (0) |
| RAPN—double-arm studies | ||||||||||||||
| Motoyama [83] (TP and RP) |
2023 | Cohort retrospective | Hinotori | 40 | Median: 63 (range: 37–84) | – | – | – | Median: 170 (range: 106–268) | – | Median: 102 (range: 53–191) | Median: 35 (range: 1–312) | 0/40 (0) | |
| Da Vinci | 303 | Median: 65 (range: 18–90) | – | – | – | Median: 170 (range: 97–446) | – | Median: 106 (range: 50–202) | Median: 50 (range: 0–4195) | 4/303 (1.3) | ||||
| Prata [16] (TP) |
2024 | Cohort prospective | Hugo RAS | 27 | Median: 68 (IQR: 57–73) | – | cT1a: 21 (77.8) cT1b: 3 (11.1) cT2a: 2 (7.4) cT2b: 1 (3.7) |
I: 1 (3.7) II: 17 (63) III: 9 (33.3) IV: 0 |
Median: 91 (IQR: 50–149) | – | – | Median: 150 (IQR: 50–450) | 0/27 (0) | |
| Laparoscopy | 62 | Median: 65.5 (IQR: 54–72) | – | cT1a: 41 (66.2) cT1b: 16 (25.8) cT2a: 3 (4.8) cT3: 2 (3.2) |
I: 0 II: 52 (83.9) III: 9 (14.5) IV: 1 (1.6) |
Median: 149.5 (IQR: 83–203) | – | – | Median: 100 (IQR: 50–200) | 0/62 (0) | ||||
| Rojo [84] (TP) |
2024 | Cohort prospective | Hugo RAS | 25 | Mean: 61.48 (SD: 9.71) | – | – | I: 2 (8) II: 15 (60) III: 8 (32) |
– | Mean: 20.1 (SD: 6.3) | Mean: 103.15 (SD: 46.5) | Mean: 142.4 (SD: 85.25) | 0/25 (0) | |
| Da Vinci | 25 | Mean: 63.56 (SD: 9.31) | – | – | I: 4 (16) II: 18 (72) III: 3 (12) |
– | Mean: 12.6 (SD: 3) | Mean: 102.3 (SD: 45.1) | Mean: 233.8 (SD: 265.75) | 0/25 (0) | ||||
| Hussein [81] | 2023 | Cohort retrospective | Versius | 6 | Median: 45 (IQR: 26–50) |
– | – | – | Median: 170 (IQR: 140–180) | – | – | Median: 450 (IQR: 150–500) | 0/6 (0) | |
| Da Vinci | 4 | Median: 50 (IQR: 34–58) | – | – | – | Median: 185 (IQR: 180–258) | – | – | Median: 400 (IQR: 275–1050) | 1/4 (25) | ||||
| Li [82] (TP and RP) |
2023 | Double-arm randomized controlled clinical trial | KangDuo | 49 | Mean: 54.36 (SD: 10.25) | – | cT1: 49 (100) | – | – | Mean: 4.3 (SD: 1.7) | Mean: 69.55 (SD: 30.9) | Median: 50 (IQR: 10–50) | 0/49 (0) | |
| Da Vinci | 50 | Mean: 52.14 (SD: 12.38) | cT1: 50 (100) | – | – | Mean: 3.4 (SD: 1.3) | Mean: 61.45 (SD: 19.7) | Median: 30 (IQR: 20–30) | 0/50 (0) | |||||
| RAA—single- and double-arm studies | ||||||||||||||
| Raffaelli [53] | 2023 | Case series | Hugo RAS | No | 5 | Mean: 60.6 (SD: 18.9) | – | – | – | Median: 119 (IQR: 99–139) | Mean: 5.8 (SD: 1.2) | Mean: 61.4 (SD: 25.8) | – | 0/5 (0) |
| Dong [52] | 2023 | Case series | KangDuo | No | 23 | Median: 49 (range: 22–67) | – | – | Median: 2 (range: 1–3) | Median: 86.5 (IQR: 60.0–112.5) | Median: 4.3 (IQR: 3.4–5.6) | Median: 46.5 (IQR: 35.1–59.0) | Median: 50 (range: 20–400) | – |
| Motoyama [86] | 2023 | Cohort retrospective | Hinotori | 6 | Median: 48 (range: 40–64) | – | – | II: 4 (66.6) III: 2 (33.3) |
Mean: 119 (SD: 8.7) | – | Mean: 53.5 (SD: 9) | Mean: 21.2 (SD: 29.8) | 0/6 (0) | |
| Da Vinci | 5 | Median: 63 (range: 30–79) | – | – | I: 2 (40) II: 2 (40) III: 1 (20) |
Mean: 104.8 (SD: 22.3) | – | Mean: 47.2 (SD: 14.8) |
Mean: 8.8 (SD: 8.7) | 0/5 (0) | ||||
| Knežević [14] | 2022 | Case series and retrospective comparison with laparoscopic experience | Senhance | 12 | Mean: 48.3 (IQR: 42.5–51.5) | – | – | – | Mean: 165.1 (IQR: 146.2–188.7) | Mean: 11.6 (IQR: 9.5–14.3) | Mean: 98.6 (IQR: 85–112.5) | Mean: 47 (IQR: 8.75–62.5) | 0/12 (0) | |
| Laparoscopy | 306 | – | – | – | – | Mean: 95 (IQR: 45–145) | – | – | – | 0/306 (0) | ||||
| RARN—single- and double-arm studies | ||||||||||||||
| Kaneko [51] | 2021 | Case series | Senhance | No | 2 | Median: 59.5 (range: 52–67) | – | cT1: 1 (50) cT2: 1 (50) |
– | – | – | Mean: 132.5 (SD: 14.8) | Mean: 27 (SD: 23) | 0/2 (0) |
| Pokhrel [31] | 2024 | Single-arm clinical trial | Toumai | No | 4 | Median: 56 (range: 48–77) | – | – | – | Median: 140 (range: 87–170) | Median: 19 (range: 14–25) | – | Median: 75 (range: 25–300) | 0/4 (0) |
| Motoyama [85] (TP +1 RP Da Vinci) |
2023 | Cohort retrospective | Hinotori | 13 | Median: 65 (range: 46–85) | – | cT1: 4 (31) cT2: 3 (23) cT3: 6 (46) |
– | Median: 157 (range: 129–232) | – | Median: 83 (range: 65–166) | Median: 11 (range: 5–66) | 0/13 (0) | |
| Da Vinci | 21 | Median: 64 (range: 18–82) | – | cT1: 13 (62) cT2: 1 (5) cT3: 7 (33) |
– | Median: 160 (range: 118–237) | – | Median: 75 (range: 55–129) | Median: 20 (range: 5–630) | 0/21 (0) | ||||
| Hussein [81] | 2023 | Cohort retrospective | Versius | 10 | Median: 56 (range: 47–60) | – | – | – | Median: 167 (IQR: 160–190) | – | – | Median: 200 (IQR: 100–600) | 0/10 (0) | |
| Da Vinci | 19 | Median: 50 (range: 37–65) | – | – | – | Median: 180 (IQR: 145–200) | – | – | Median: 150 (IQR: 100–300) | 0/19 (0) |
||||
| RANU—single-arm studies | ||||||||||||||
| Motoyama [54] | 2023 | Case series | Hinotori | No | 8 | Median: 76 (IQR: 69.75–79.5) | – | cT1: 6 (75) cT3: 1 (12.5) |
– | Mean: 232.6 (SD: 18.87) | – | Mean: 145 (SD: 26.3) | Mean: 24.4 (SD: 18.9) | 0/8 (0) |
| Pokhrel [31] | 2024 | Single-arm clinical trial | Toumai | No | 5 | Median: 64 (range: 58–75) | – | – | – | Median: 186 (range: 136–330) | Median: 23 (range: 21–36) | – | Median: 50 (range: 30–200) | 0/5 (0) |
| RARN and caval thrombectomy—single-arm study | ||||||||||||||
| Motoyama [55] | 2023 | Case series | Hinotori | No | 2 | Mean: 73 (SD: 8.48) | – | cT3: 2 (100) | I: 1 (50) II: 1 (50) |
Mean: 221 (SD: 7) | – | Mean: 157 (SD: 1) | Mean: 367.5 (SD: 167.5) | 0/2 (0) |
ASA = American Society of Anesthesiology; CT = console time; DT = docking time; EBL = estimated blood loss; EP = extraperitoneal; IOC = intraoperative complications; IQR = interquartile range; MRI = magnetic resonance imaging; OT = total operative time; PSA = prostate-specific antigen; RAA = robot-assisted adrenalectomy; RANU = robot-assisted radical nephroureterectomy; RAPN = robot-assisted partial nephrectomy; RARN = robot-assisted radical nephrectomy; RARP = robot-assisted radical prostatectomy; RAS = robot-assisted surgery; RP = retroperitoneal; SD = standard deviation; TP = transperitoneal.
cT stage at MRI.
Table 2.
Complications and postoperative, pathological, and functional outcomes of robot-assisted surgeries for malignant diseases
| Author | New robot/comparator | PO CD grade ≥III | LoS (d) |
Conversion to LS | Conversion to open | Failure or malfunctions | pT stage n (%) |
PSM | Recurrences | Continence (RARP) | Definition of continence | Potency (RARP) | Trifecta (RAPN) | Definition of trifecta |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RARP—single-arm studies | ||||||||||||||
| Hinata [26] (TP) |
Hinotori | 3/30 (10) | – | 0/30 (0) | 0/30 (0) | 4/30 (13) | pT2: 26 (87) pT3a: 4 (13) |
1/30 (3) | – | – | – | – | – | – |
| Alfano [17] (TP) |
Hugo RAS | 0/15 (0) | Median: 2 (IQR: 2–2) | 0/15 (0) | 0/15 (0) | 0/15 (0) | pT2: 11 (73) pT3: 4 (27) |
5/15 (33) | 0/15 (0) | 9/15 (61) at 4 wk | Capacity to hold urine without pads or patients using one security pad |
– | – | – |
| Bravi [19] (TP) |
Hugo RAS | 2/112 (2) | Median: 3 (IQR: 3–4) | 0/112 (0) | 0/112 (0) | 0/112 (0) | pT2: 78 (70) pT3a: 22 (20) pT3b-pT4: 12 (10) |
10/112 (9) | 8/68 (12) | 91/112 (81) at 3 mo | The use of no/one safety pad (3 mo) | – | – | – |
| Totaro [39] (TP) |
Hugo RAS | 2/132 (2) | Median: 3 (IQR: 1–3) |
0/132 (0) | 0/132 (0) | 52/132 (39) | pT2: 98 (74) pT3: 34 (26) |
54/132 (41) | 7/132 (5) at 3 mo 27/132 (20) at 6 mo |
100/132 (75.7) at 3 mo 98/132 (86) at 6 mo |
Use of no more than one pad per day | IIEF5: Median: 9 (SD: 11) at 3 mo Median: 10 (SD: 12) at 6 mo |
– | – |
| Ragavan [33] | Hugo RAS | 0/3 (0) | Mean: 1 (SD: 0) | 0/3 (0) | 0/3 (0) | 0/3 (0) | pT3: 3 (100) | – | – | – | – | – | – | |
| Marques-Monteiro [29] (EP) |
Hugo RAS | 0/16 (0) | Median: 2 (IQR: 2–3) | 0/16 (0) | 0/16 (0) | 0/16 (0) | – | – | – | – | – | – | – | – |
| Veccia [40] (TP) |
Hugo RAS | 0/10 (0) | – | 0/10 (0) | 0/10 (0) | 0/10 (0) | – | – | – | – | – | – | – | – |
| Territo [37] (TP and EP) |
Hugo RAS | TP: 1/17 (6) | TP: median: 3 (IQR: 2–4) | TP: 0/17 (0) | TP: 0/17 (0) | TP: 0/17 (0) | TP: pT2: 14 (82) pT3a: 3 (18) |
TP: 5/17 (29) |
TP: 0/17 (0) |
– | – | – | – | – |
| EP: 0/3 (0) |
EP: mean: 3.33 (SD: 0.47) | EP: 0/3 (0) | EP: 0/3 (0) | EP: 0/3 (0) | EP: pT2: 2 (67) pT3a: 1 (33) |
EP: 1/3 (33) |
EP: 0/3 (0) | – | – | – | – | – | ||
| Andrade [18] (TP) |
Hugo RAS | 0/19 (0) | – | 0/19 (0) | 0/19 (0) | 1/19 (5) | pT2: 14 (74) pT3a: 4 (21) pT3b: 1 (5) |
1/19 (5) | – | 18/19 (95) at 3 mo | 1 or 0 pads | 17/18 (94) at 3 mo | – | – |
| Dell'Oglio [22] (TP) |
Hugo RAS | 1/26 (4) | Median: 3 (IQR: 2–3) | 0/26 (0) | 0/26 (0) | 4/26 (15) | pT2: 22 (85) pT3: 4 (15) |
8/26 (31) | 3/26 (12) | 24/26 (92) at 2 mo | Use of no/1 safety pad at catheter removal and at 2 mo | – | – | – |
| Ou [30] | Hugo RAS | 0/12 (0) | Mean: 7 (SD: 0) | 0/12 (0) | 0/12 (0) | 6/12 (50) | pT2: 9 (75) pT3a: 1 (8) pT3b: 2 (17) |
3/12 (25) | 0/12 (0) | – | – | – | – | – |
| Takahara [36] (TP) |
Hugo RAS | 0/13 (0) | Median: 8 (IQR: 8–8.5) | 0/13 (0) | 0/13 (0) | – | pT1-pT2: 9 (69) pT3a: 2 (15) pT3b: 2 (15) |
1/13 (8) | – | – | – | – | – | – |
| Fan [24] (EP) |
KangDuo | 0/16 (0) | Median: 5 (range: 4–10) | 0/16 (0) | 0/16 (0) | 0/16 (0) | pT2: 8 (50) pT3a: 7 (44) pT3b: 1 (6) |
4/16 (25) | 0/16 (0) | 14/16 (87.5) at 1 mo | No more than 1 pad daily or urine leakage of ≤20 g by the 24-h pad weight test | – | – | – |
| Xiong [42] (TP and EP) |
KangDuo | TP: median: 5 (range: 3–13) EP: median: 5 (range: 4–10) |
0/41 (0) | 0/41 (0) | TP: pT2: 10 (40) pT3: 14 (56) pT4:1 (4) EP: pT2: 8 (50) pT3:8 (50) |
TP: 12/25 (48) EP: 4/16 (25) |
0/41 (0) | TP: 22/25 (88) at 1 mo EP: 14/16 (88) at 1 month |
Rate of urinary continence recovery at 1 mo after catheter removal | – | – | – | ||
| Chang [20] (TP) |
Revo-I | 0/17 (0) | Median: 4 (IQR: 4–7) | 0/17 (0) | 0/17 (0) | 0/17 (0) | pT2: 16 (94) pT3: 1 (6) |
4/17 (24) | 1/17 (6) | 15/17 (88.2) at 3 mo | Use of no pad or one safety pad | – | – | – |
| Gkeka [25] (EP) |
Senhance | 0/14 (0) | Median: 2 (IQR: 2–2) | 0/14 (0) | 0/14 (0) | 0/14 (0) | pT2: 5 (36) pT3a: 5 (36) pT3b: 4 (28) |
2/14 (14) | – | 11/14 (71.4) at 3 mo | Use of no or one security pad (without significant urine leakage) daily | 12/14 (77) | – | – |
| Venckus [41] | Senhance | 3/127 (2) | – | – | – | – | pT2: 108 (85) pT3a: 12 (9) pT3b: 7 (6) |
43/127 (34) | – | – | – | – | – | – |
| Hudolin [27] (EP) |
Senhance | 1/200 (1) | Median: 5 (range: 3–12) | 6/200 (3) | 2/200 (1) | 2/200 (1) | pT2: 156 (78) pT3: 43 (22) |
55/200 (28) | 13/200 (7) | 140/150 (93.3) | ≤2 urinary pads | – | – | – |
| Kulis [28] (EP and TP) |
Senhance | – | – | 18/375 (5) | 2/375 (1) | 18/375 (5) | – | – | – | – | – | – | – | – |
| Reeves [34] | Versius | 0/4 (0) | – | 0/4 (0) | 0/4 (0) | 0/4 (0) | 0/4 (0) | – | 2/4 (50) at 1 wk | Early within week continence | – | – | – | |
| Sighinolfi [35] | Versius | 1/22 (5) | Median: 4 (IQR: 3.75–7) | 0/22 (0) | 0/22 (0) | 0/22 (0) | – | – | 1/22 (5) | – | – | – | – | – |
| De Maria [21] | Versius | 0/18 (0) | Median: 4 (IQR: 3.75–5) | 0/18 (0) | 0/18 (0) | 0/18 (0) | pT2: 7 (39) pT3: 11 (61) |
15/18 (83) | – | 13/18 (72) at 2 mo | Use of none/1 pad per day | – | – | – |
| Polom [32] (TP) |
Versius | 4/58 (7) | Median: 4.5 (range: 4–12) | – | 1/58 (2) | 0/58 (0) | pT2: 31 (53) pT3: 27 (47) |
15/58 (26) | 2/58 (4) | 52/58 (89.7) at 6 wk | – | – | – | – |
| Dibitetto [23] (EP) |
Versius | 0/53 (0) | Median: 3 (range: 2–6) | 4/53 (8) | 0/53 (0) | 6/53 (11) | – | – | 0/53 (0) | – | – | – | – | – |
| Thillou [38] (TP) |
Avatera | 1/10 (10) | Median: 3 (IQR: 3–4) | 0/10 (0) | 0/10 (0) | 3/10 (30) | – | – | – | – | – | – | – | – |
| Pokhrel [31] (TP) |
Toumai | – | Median: 9 (range: 7–11) | 0/3 (0) | 0/3 (0) | 0/3 (0) | – | 1/5 (20) | – | – | – | – | – | – |
| RARP—double-arm studies | ||||||||||||||
| Sasaki [79] (TP) |
Hinotori | 1/48 (2; <30 d) 2/48 (4; late >30 d) |
Median: 8 (IQR: 7–8) | – | – | – | pT2: 43 (90) ≥pT3: 5 (10) |
9/48 (19) | 2/48 (4) | 24/48 (50) at 3 mo | Use of 0–1 incontinence pad per day | – | – | – |
| Da Vinci | 2/46 (4; late >30 d) | Median: 8 (IQR: 7–8) | – | – | – | pT2: 41 (90) ≥ pT3: 5 (10) |
7/46 (15) | 2/46 (4) | 25/46 (54) at 3 mo | – | ||||
| Kohjimoto [72] (TP) |
Hinotori | 0/60 (0) | Median: 8 (IQR: 8–10) | – | 0/60 (0) | – | pT2: 35 (58) pT3a: 22 (37) pT3b: 3 (5) |
11/60 (18) | – | 15/32 (47) at 3 mo 17/23 (74) at 6 mo 10/13 (77) at 12 mo |
Need for 0–1 incontinence pads per day | – | – |
– |
| Da Vinci | 3/100 (3) | Median: 9 (IQR: 8–10) | – | 0/100 (0) | – | pT2: 44 (44) pT3a: 44 (44) pT3b: 10 (10) |
17/100 (17) | – | 19/38 (50) at 3 mo 19/33 (58) at 6 mo 13/21 (62) at 12 mo |
– | ||||
| Nakayama [75] (TP) |
Hinotori | 0/97 (0) | – | 0/97 (0) | 0/97 (0) | 0/97 (0) | pT2: 58 (60) pT3: 36 (37) |
35/97 (36) | – | – | – | – | – |
– |
| Da Vinci | 4/246 (2) | – | 0/246 (0) | 0/246 (0) | 0/246 (0) | pT2: 183 (75) pT3: 60 (24) pT4: 2 (1) |
93/244 (38) | – | – | – | – | |||
| Ragavan [78] (TP) |
Hugo RAS | 0/17 (0) | Median: 1 (IQR: 1–2) | 0/17 (0) | 0/17 (0) | 0/17 (0) | – | 4/17 (24) | 0/17 (0) | 17/17 (100) at 3 mo | One protective pad | – | – |
– |
| Da Vinci | 0/17 (0) | Median: 1 (IQR: 1–2) | 0/17 (0) | 0/17 (0) | 0/17 (0) | – | 4/17 (24) | 0/17 (0) | 17/17 (100) at 3 mo | |||||
| Antonelli [67] (TP) |
Hugo RAS |
– | – | 0/50 (0) | 20/50 (40) | – | – | – | – | – | – | – | – | |
| Da Vinci | – | – | 1/50 (2) | 4/50 (8) | – | – | – | – | – | – | ||||
| Bravi [68] | Hugo RAS | – | Median: 3 (IQR: 3–4) | – | – | – | pT2: 110 (67) pT3a: 37 (23) pT3b: 17 (10) |
20/164 (12) | – | 108/164 (66) at 1 mo 133/164 (81) at 3 mo |
Use of no/one safety pad | – | – | – |
| Da Vinci | – | Median: 3 (IQR: 2–4) | – | – | – | pT2: 250 (61) pT3a: 84 (22) pT3b: 44 (12) |
55/378 (15) | – | 238/378 (63) at 1 mo 297/378 (79) at 3 mo |
– | ||||
| Olsen [76] | Hugo RAS | 1/19 (5) | Median: 1 (IQR: 1–2) | 0/19 (0) | 0/19 (0) | 0/19 (0) | pT2: 11 (58) pT3a: 4 (21) pT3b: 4 (21) |
7/19 (37) | 0/19 (0) | 11/18 (62) at 3 mo | Continence is 0 pads per day | 9/16 (56.25) | – | – |
| Da Vinci | 0/11 (0) | Median: 1 (IQR: 1–2) | 0/11 (0) | 0/11 (0) | 0/11 (0) | pT2: 9 (82) pT3a: 1 (9) pT3b: 1 (9) |
3/11 (27) | 0/11 (0) | 9/10 (81) at 3 mo | 5/9 (55.5) | ||||
| Ou [77] | Hugo RAS | 0/30 (0) | Median: 7 (IQR: 7–7) | 0/30 (0) | 0/30 (0) | 0/30 (0) | pT2: 27 (90) pT3: 3 (10) |
6/30 (20) | 12/30 (40) | 8/30 (26.7) at 1 mo 23/30 (76.7) at 3 mo |
0–1 pad used per day was taken to indicate continence recovery | 7/30 (23.3) after 1 mo 22/30 (73.3) after 3 mo |
– | – |
| Da Vinci | 0/30 (0) | Median: 7 (IQR: 7–7) | 0/30 (0) | 0/30 (0) | 0/30 (0) | pT2: 26 (87) pT3a: 4 (13) |
9/30 (30) | 11/30 (37) | 6/30 (20) at 1 mo 16/30 (53.3) at 3 mo |
6/30 (20) after 1 mo 16/30 (53.3) after 3 mo |
||||
| Gandi [71] | Hugo RAS |
– | ‘ | – | – | – | pT2: 78 (76) pT3a: 13 (13) pT3b: 12 (12) |
23/103 (22) | – | 78/103 (75.7) at 3 mo | No more than one pad per day | – | – |
– |
| Da Vinci | – | Median: 4 (IQR: 4–5) | – | – | – | pT2: 2 (1) pT2a: 6 (2) pT2b: 2 (1) pT2c: 198 (72) pT3a: 34 (12) pT3b: 34 (12) |
58/276 (21) | – | 202/276 (73.2) at 3 mo | – | ||||
| Menendez [74] (TP) |
Hugo RAS | 0/75 (0) | Mean: 2.2 (SD: 0.4) | 0/75 (0) | 0/75 (0) | 0/75 (0) | pT2: 55 (73) pT3a: 14 (19) pT3b: 6 (8) |
15/75 (20) | 66/75 (88) at 6 mo |
Any experienced urine leakage or social incontinence that refers to requiring more than one pad per day | – | – |
– |
|
| Da Vinci | 0/75 (0) | Mean: 2.1 (SD: 0.3) | 0/75 (0) | 0/75 (0) | 0/75 (0) | pT2: 52 (69) pT3a: 15 (20) pT3b: 8 (11) |
8/75 (11) | 69/75 (92) at 6 mo | – | |||||
| Fan [70] (EP) |
KangDuo | 0/16 (0) | Median: 5 (range: 4–10) | 0/16 (0) | 0/16 (0) | 0/16 (0) | pT2: 8 (50) pT3a: 7 (44) pT3b: 1 (6) |
4/16 (25) | 0/16 (0) | 15/16 (93.8) at 3 mo |
The use of no more than one pad daily or urine leakage of <20 g as determined by a 24-h pad weight test | – | – |
– |
| Da Vinci | 0/16 (0) | Median: 6.5 (Range: 5–10) |
0/16 (0) | 0/16 (0) | 0/16 (0) | pT2: 8 (50) pT3a: 7 (44) pT3b: 1 (6) |
2/16 (13) | 0/16 (0) | 14/16 (87.5) at 3 months | – | ||||
| Alip [66] | Revo-I | 0/33 (0) | Mean: 5 (SD: 1.9) | 0/33 (0) | 0/33 (0) | 0/33 (0) | pT2: 27 (82) ≥pT3: 6 (18) |
16/33 (48) | 4/33 (12) | – | – | – | – |
– |
| Da Vinci | 1/33 (3) | Mean: 5.9 (SD: 2.15) | 0/33 (0) | 0/33 (0) | 0/33 (0) | pT2:25 (76) ≥pT3: 8 (24) |
15/33 (45) | 0/33 (0) | – | – | – | |||
| Lin [73] (TP) |
Senhance | 2/63 (3) | Median: 9 (range: 9–10.5) | – | – | – | pT2: 39 (62) pT3: 24 (48) |
23/63 (37) | 20/63 (32) | 54/63 (85.7) | Daily pad use fewer than two | – | – | – |
| Da Vinci | 3/63 (5) | Median: 9 (range: 9–9) | – | – | – | pT2: 34 (54) pT3: 28 (44) pT4: 1 (2) |
26/63 (41) | 21/63 (33) | 53/63 (84.1) | |||||
| Kulis [15] (EP) |
Senhance | 1/107 (1) | Mean: 5 (IQR: 5–5) | 9/107 (8) | 0/107 (0) | 0/107 (0) | pT2: 87 (81) pT3: 20 (19) |
30/107 (28) | 5/107 (5; 3 BCR and 2 BCF) | 79/107(79) | 0–1 pad per day | – | – | – |
| Laparoscopy | 0/61 (0) | Mean: 5 (IQR: 5–6) | – | 0/61 (0) | 0/61 (0) | pT2: 53 (87) pT3: 8 (13) |
14/61(23) | 5/61 (8; 2 BCR and 3 BCF) | 46/61(75.4) | |||||
| Dibitetto [69] (EP) |
Versius | 2/53 (4) | Median: 3 (range: 2–6) | – | – | – | pT2: 38 (72) pT3a: 14 (26) pT3b: 1 (2) |
9/53 (17) | – | 25/53 (47) at 1 mo 29/53 (55) at 3 mo 36/53 (68) at 6 mo |
0–1 pad per day | – | – | – |
| Da Vinci | 4/53 (8) | Median: 3 (range: 2–5) | – | – | – | pT2: 32 (60) pT3a: 15 (28) pT3b: 6 (11) |
6/53 (11) | – | 28/53 (53) at 1 mo 39/53 (74) at 3 mo 43/53 (81) at 6 mo |
|||||
| RAPN—single-arm studies | ||||||||||||||
| Miyake [46] (TP and RP) |
Hinotori | 0/30 (0) | Median: 7 (IQR: 4–23) | 0/30 (0) | 0/30 (0) | 0/30 (0) | – | 0/30 (0) | – | – | – | – | 30/30 (100) | – |
| Gallioli [44] (TP) |
Hugo RAS | 1/10 (10) | Mean: 5 (SD: 3) | 1/10 (10) | 0/10 (0) | 0/10 (0) | pT1a: 4 (40) pT3a: 1 (10) |
0/10 (0) | – | – | – | – | 9/10 (90) | – |
| Prata [48] (TP) |
Hugo RAS | 0/25 (0) | Median: 3 (IQR: 3–4) | 0/25 (0) | 0/25 (0) | 0/25 (0) | pT1a: 21 (84) pT1b: 2 (8) pT2a: 2 (8) |
0/25 (0) | – | – | – | – | 25/25 (100) | – |
| Prata [47] (TP) |
Hugo RAS | 1/27 (3.7) | Median: 3 (IQR: 3–4) | 0/27 (0) | 0/27 (0) | 0/27 (0) | pT1a: 20 (74) pT1b: 3 (11.1) pT2a: 2 (7.4) pT2b: 1 (3.7) pT3a: 1 (3.7) |
1/27 (3.7) | 0/27 (0) | – | – | – | 20/27 (74) | Coexistence of negative surgical margin status, no Clavien-Dindo grade ≥3 complications, and eGFR decline ≤30% |
| Andrade [18] (TP) |
Hugo RAS | – | – | 0/4 (0) | 0/4 (0) | 1/4 (25) | – | – | – | – | – | – | – | – |
| Meneghetti [45] (TP) |
Versius | 0/13 (0) | Mean: 3.7 (SD: 0.49) | – | – | – | pT1a: 6 (46) pT1b: 5 (38) pT2a: 1 (8) |
1/13 (8) | – | – | – | – | 12/13 (92) | – |
| Xu [49] (TP and RP) |
KangDuo | 0/17 (0) | Median: 5 (range: 4–9) | 0/17 (0) | 0/17 (0) | 0/17 (0) | pT1a: 17 (100) | 0/17 (0) | – | – | – | – | – | – |
| Xiong [42] (TP and RP) |
KangDuo | 0/28 (0) | Median: 4 (range: 4–5) | 0/28 (0) | 0/28 (0) | 0/28 (0) | pT1a: 28 (100) | 0/28 (0) | 0/28 (0) | – | – | – | – | – |
| Pokhrel [31] (TP) |
Toumai | 0/4 (0) | Median: 6.5 (range:6–8) | 0/4 (0) | 0/4 (0) | 0/4 (0) | – | 0/4 (0) | – | – | – | – | – | – |
| RAPN—double-arm studies | ||||||||||||||
| Motoyama [83] (TP and RP) |
Hinotori | 0/40 (0) | Median: 6 (IQR: 4–23) |
– | 0/40 (0) | – | – | 0/40 (0) | – | – | – | – | 40/40 (100) | – |
| Da Vinci | 9/303 (3) | Median: 8 (IQR: 4–31) | – | 2/303 (0.7) | – | – | 2/303 (0.7) | – | – | – | – | 289/303 (95.4) | – | |
| Prata [16] (TP) |
Hugo RAS | 0/27 (0) | Median: 3 (IQR: 3–4) | 0/27 (0) | 0/27 (0) | 0/27 (0) | pT1a: 20 (74.1) pT1b: 3 (11.1) pT2a: 2 (7.4) pT2b: 1 (3.7) pT3a: 1 (3.7) |
1/27 (3.7) | – | – |
– |
– |
25/27 (92.6) | Negative surgical margin, decline of eGFR ≤30%, and Clavien-Dindo grade <2 complications |
| Laparoscopy | 0/62 (0) | Median: 5 (IQR: 4–5) | – | 0/62 (0) | 0/62 (0) | pT1a: 39 (62.9) pT1b: 14 (22.6) pT2a: 1 (1.6) pT3a: 8 (12.9) |
3/62 (4.8) | – | 51/62 (82.3) | |||||
| Rojo [84] (TP) |
Hugo RAS | 0/25 (0) | – | 0/25 (0) | 0/25 (0) | 0/25 (0) | pT1a: 16 (64) pT1b: 7 (28) pT2: 1 (4) pT3a: 1 (4) |
2/25 (8) | – |
– |
– |
– |
21/25 (84) | Negative surgical margins, no Clavien-Dindo >3 complications, and ≤30% postoperative eGFR |
| Da Vinci | 0/25 (0) | – | 0/25 (0) | 0/25 (0) | 0/25 (0) | pT1a: 17 (68) pT1b: 7 (28) pT3a: 1 (4) |
1/25 (4) | – | 22/25 (88) | |||||
| Hussein [81] | Versius | 0/6 (0) | – | 0/6 (0) | 0/6 (0) | 0/6 (0) | – | – | – |
– |
– |
– |
– | – |
| Da Vinci | 0/3 (0) | – | 0/4 (0) | 1/4 (25) | 1/4 (25) | – | – | – | – | |||||
| Li [82] (TP and RP) |
KangDuo | 0/49 (0) | Median: 4 (IQR: 3–10) | 0/49 (0) | 0/49 (0) | 0/49 (0) | – | 0/49 (0) | – |
– | – | – | 49/49 (100) | – |
| Da Vinci | 0/50 (0) | Median: 4 (IQR: 4–8) | 0/50 (0) | 0/50 (0) | 0/50 (0) | – | 0/50 (0) | – | 50/50 (100) | |||||
| RAA—single- and double-arm studies | ||||||||||||||
| Raffaelli [53] | Hugo RAS | 0/5 (0) | Mean: 3.2 (SD: 2.4) | 0/5 (0) | 0/5 (0) | 0/5 (0) | – | – | – | – | – | – | – | – |
| Dong [52] | KangDuo | 0/23 (0) | Median: 10 (IQR: 8–12) | 0/23 (0) | 0/23 (0) | – | – | 0/23 (0) | – | – | – | – | – | – |
| Motoyama [86] | Hinotori | 0/6 (0) | Mean: 10 (SD: 9.9) | 0/6 (0) | 0/6 (0) | 0/6 (0) | – | – | – | – | – | – | – | – |
| Da Vinci | 0/5 (0) | Mean: 7.3 (SD: 3.7) | 0/5 (0) | 0/5 (0) | 0/5 (0) | – | – | – | ||||||
| Knežević [14] | Senhance | 1/12 (8) | Mean: 4.5 (IQR: 4–5) | 1/12 (8) | 0/12 (0) | 0/12 (0) | – | 0/12 (0) | 0/12 (0) | – | – | – | – | – |
| Laparoscopy | – | – | – | – | – | – | – | – | ||||||
| RAA—single- and double-arm studies | ||||||||||||||
| Raffaelli [53] | Hugo RAS | 0/5 (0) | Mean: 3.2 (SD: 2.4) | 0/5 (0) | 0/5 (0) | 0/5 (0) | – | – | – | – | – | – | – | – |
| Dong [52] | KangDuo | 0/23 (0) | Median: 10 (IQR: 8–12) | 0/23 (0) | 0/23 (0) | – | – | 0/23 (0) | – | – | – | – | – | – |
| Motoyama [86] | Hinotori | 0/6 (0) | Mean: 10 (SD: 9.9) | 0/6 (0) | 0/6 (0) | 0/6 (0) | – | – | – | – | – | – | – | – |
| Da Vinci | 0/5 (0) | Mean: 7.3 (SD: 3.7) | 0/5 (0) | 0/5 (0) | 0/5 (0) | – | – | – | ||||||
| Knežević [14] | Senhance | 1/12 (8) | Mean: 4.5 (IQR: 4–5) | 1/12 (8) | 0/12 (0) | 0/12 (0) | – | 0/12 (0) | 0/12 (0) | – | – | – | – | – |
| Laparoscopy | – | – | – | – | – | – | – | – | ||||||
| RANU—single-arm studies | ||||||||||||||
| Motoyama [54] | Hinotori | 0/8 (0) | Median: 8 (IQR: 6–8.25) | 0/8 (0) | 0/8 (0) | – | Papilloma: 1 (12.5) pTIS: 1 (12.5) pT1: 4 (50) pT3: 2 (25) |
1/8 (12.5) | – | – | – | – | – | – |
| Pokhrel [31] | Toumai | 1/5 (20) | Median: 8 (range: 7–15) | 0/5 (0) | 0/5 (0) | 0/5 (0) | – | 0/5 (0) | – | – | – | – | – | – |
| RARN and caval thrombectomy—single-arm study | ||||||||||||||
| Motoyama [55] | Hinotori | 0/2 (0) | Median: 9 (IQR: 8.5–9.5) | 0/2 (0) | 0/2 (0) | 0/2 (0) | pT3: 2 (100) | – | – | |||||
BCF = biochemical failure; BCR = biochemical recurrence; eGFR = estimated glomerular filtration rate; EP = extraperitoneal; IIEF5 = International Index of Erectile Function; IQR = interquartile range; LoS = length of stay; LS = laparoscopy; PO CD = postoperative Clavien-Dindo; PSM = positive surgical margin; RAA = robot-assisted adrenalectomy; RANU = robot-assisted radical nephroureterectomy; RAPN = robot-assisted partial nephrectomy; RARN = robot-assisted radical nephrectomy; RARP = robot-assisted radical prostatectomy; RAS = robot-assisted surgery; RP = retroperitoneal; SD = standard deviation; TP = transperitoneal.
3.3.1. Robot-assisted radical prostatectomy
Twenty-six single-arm studies involving 1366 patients discussed the use of one of the NM-RSs to perform RARP [17], [18], [19], [20], [21], [22], [23], [24], [25], [26], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42]. The most common NM-SR used was the Hugo RAS robot in 11 single-arm studies [17], [18], [19], [22], [29], [30], [33], [36], [37], [39], [43]. Baseline information, perioperative outcomes, and oncological and functional endpoints for each study are summarized in Table 1, Table 2. Among the largest series (n ≥ 100) [19], [27], [28], [39], [41], the most common approach was transperitoneal, and the total operative time was similar between different NM-SRs; the median operative time ranged between 180 min (Hugo RAS) and 190 min (Senhance). Intraoperative SATAVA ≥2 and postoperative CD ≥IIIa complications ranged between 0 and 5% and 0 and 10%, respectively, among all the single-arm studies. The highest proportion of positive surgical margins (PSMs) that occurred in a large series was obtained with the use of Senhance (28%, 55/200) [27], while the lowest proportion of PSMs in a large experience was obtained through Hugo RAS (9%, 10/112) [19]. Urinary continence rates at 3 mo ranged from 71.4% to 95%.
3.3.2. Robot-assisted partial nephrectomy
Nine single-arm papers focused on 158 RAPNs performed with one of the NM-SRs [18], [31], [42], [44], [45], [46], [47], [48], [49]. The most common NM-SR used was Hugo RAS, with four reports from three different centers [18], [44], [47], [50].
Table 1, Table 2 summarize the surgical and clinical endpoints of the studies retrieved.
The median operative time ranged between 91 min (Hugo RAS) and 179 min (Hinotori). Only one intraoperative SATAVA ≥ 2 complication occurred in a series involving Hugo RAS [44]. Two postoperative CD ≥IIIa complications related to Hugo RAS were registered among all the single-arm studies [44], [47]. One PSM was reported in the Hugo RAS series [47] and one for Versius [45]. No PSMs were observed in the studies involving Hinotori, KangDuo, and Toumai. Trifecta achievement for Hugo RAS ranged between 74% and 100%. Hinotori and Versius RAPN obtained a trifecta of 100% and 92%, respectively.
3.3.3. Other oncological surgeries
The use of NM-SRs was described in two experiences with robot-assisted radical nephrectomy (RARN) [31], [51], two single-arm studies for robot-assisted adrenalectomy (RAA) [52], [53], two manuscripts for robot-assisted nephroureterectomy (RANU) [31], [54], and one paper for RARN and thrombectomy [55]. The first RAA performed with Hugo RAS and KangDuo is described in Table 1, Table 2. Longer operative time was required for the cases performed with Hugo RAS (median 119 min, IQR 99–139), while the shortest operative time was derived from KangDuo in the largest series included (median 86.5, IQR 60–113). No postoperative CD ≥IIIa complications were recorded in the single-arm studies [52], [53]. All the margins were negative in the only single-arm study that explored this outcome using the KangDuo robot [52].
Table 1, Table 2 also show the perioperative and main oncological outcomes related to RARN, RANU, and two cases of RARN and thrombectomy performed with Hinotori. Radical cystectomy was discussed in only a few case reports [37], [56], [57], which are not shown. One CD ≥IIIa complication occurred during a RANU performed with Toumai. One PSM was identified after a RANU performed with Hinotori.
3.3.4. Other nononcological surgeries
Perioperative outcomes for robot-assisted colpopexy (RACP, two single-arm studies), pyeloplasty (five single-arm studies), simple nephrectomy (RASN; three single-arm studies), simple prostatectomy (RASP; two single-arm studies), and ureteral surgery are available in Supplementary Tables 1 and 2 [18], [31], [33], [34], [42], [58], [59], [60], [61], [62], [63], [64], [65]. Pyeloplasty was the most common surgery performed with five single-arm experiences. Hugo RAS was the most employed NM-SR among nononcological surgeries. No intraoperative SATAVA ≥2 complications happened in the single-arm series for benign diseases. No postoperative CD ≥IIIa complications were reported among 65 patients who underwent RACP in the two single-arm experiences, and only one complication (CD ≥IIIa) happened among those undergoing pyeloplasty, which was related to the use of Hugo RAS [65]. No CD ≥IIIa complications occurred when NM-SRs were used to complete RASN, RASP, and ureteral surgery.
3.4. Data synthesis and meta-analysis for double-arm studies (NM-SRs vs DVM-SR)
3.4.1. Robot-assisted radical prostatectomy
A total of ten retrospective cohort studies, three prospective cohort studies, and one nonrandomized clinical trial concerning RARP were included [66], [67], [68], [69], [70], [71], [72], [73], [74], [75], [76], [77], [78], [79]. Transperitoneal approach was most used for both arms (Table 1, Table 2).
Overall, the MD in total operative time between NM-SRs and the DVM-SR was 14 min (95% confidence interval [CI] –6.2, 34, p = 0.2; Fig. 2A).
Fig. 2.
Prostatectomy: (A) total operative time; (B) console time; (C) docking time; and (D) estimated blood loss. CI = confidence interval; diff. = difference; RAS = robot-assisted surgery; SD = standard deviation.
The MD in docking time (DT) could be calculated only for studies comparing Hugo RAS with the DVM-SR and was 6.2 min (95% CI 4.2, 8.2, p < 0.001; Fig. 2C). The MD in console time (CT) was 18.2 min (95% CI 6.2, 30, p < 0.001; Fig. 2B). Finally, the MD in EBL was 8.6 ml (95% CI –19.3, 36.4, p = 0.6; Fig. 2D). A high level of heterogeneity was found for all these outcomes, and this must be considered when interpreting the results. Intraoperative SATAVA grade ≥2 complications (OR 0.89, 95% CI 0.3, 3.1, p = 0.9; Fig. 2D) and postoperative CD ≥ IIIa complications (OR 0.85, 95% CI 0.37, 2.0, p = 0.7; Supplementary Fig. 7C) were similar between the two groups, with no heterogeneity detected.
In the quantitative analysis for oncological outcomes, the proportion of PSMs and the biochemical recurrence (BCR) rate at 3 mo seemed similar and did not show any heterogeneity between the two groups (respectively, OR 0.9, 95% CI 0.72, 1.1, p = 0.3 and OR 0.99, 95% CI 0.55, 1.8, p = 1; Fig. 3B and 3C).
Fig. 3.
Prostatectomy: (A) complications Clavien-Dindo ≥IIIa; (B) surgical margins; (C) biochemical recurrences at 3 mo; and (D) continence at 3 mo. CI = confidence interval; diff. = difference; RAS = robot-assisted surgery.
Urinary continence at 3 mo was reported in 11 studies, with an estimated OR of 0.99 (95% CI 0.77–1.3, p = 0.9) [24], [69], [71], [72], [73], [74], [76], [77], [78], [79], [80]. Only a small amount of heterogeneity was found among the studies assessing urinary continence (I2: 15.2%; Fig. 3D). Percentages of potency were provided by only two studies (Table 2), both focused on Hugo RAS. Potency rates at 3 mo for Hugo RAS and the DVM-SR ranged from 56% to –73% and 53% to 56%, respectively.
3.4.2. Robot-assisted partial nephrectomy
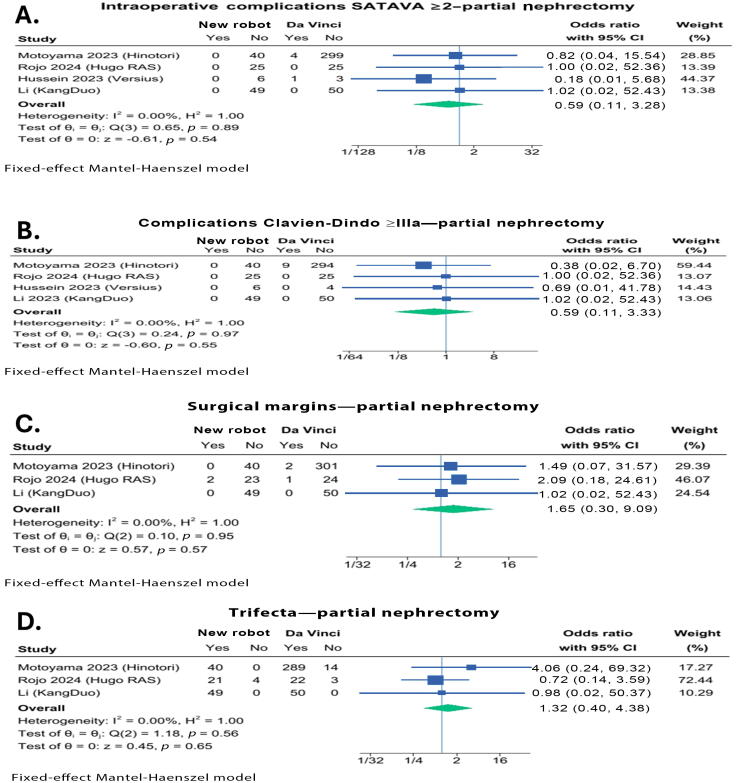
Three retrospective cohort studies and one RCT were included for RAPN [81], [82], [83], [84]. The majority of patients in these studies underwent a transperitoneal approach, although two authors included cases treated with a retroperitoneal approach [82], [83]. NM-SRs and the DVM-SR showed MDs of, respectively, 1.3 min (95% CI –5.3, 7.8, p = 0.7 in terms of CT; Supplementary Fig. 7A) and –35 ml (95% CI –118, 48, p = 0.4; Supplementary Fig. 7B) for EBL; both endpoints were associated with significant heterogeneity. Similar outcomes between NM-SRs and the DVM-SR were observed for SATAVA grade ≥2 (OR 0.59, 95% CI 0.11, 3.3, p = 0.5; Fig. 4A) and CD ≥IIIa (OR 0.59, 95% CI 0.11, 3.3, p = 0.6; Fig. 4B) complications.
Fig. 4.
Partial nephrectomy: (A) intraoperative complications SATAVA grade ≥2; (B) complications Clavien-Dindo ≥IIIa; (C) surgical margins; and (D) trifecta. CI = confidence interval; RAS = robot-assisted surgery.
PSMs and trifecta achievement were apparently comparable among the two groups (respectively, OR 1.65 [95% CI 0.3, 9.1, p = 0.6]; Fig. 4C and OR 1.32 [95% CI 0.4, 4.4, p = 0.7]; Fig. 4D).
3.4.3. Other oncological surgeries
Two comparative experiences explored the role of Hinotori and Versius for RARN [81], [85], while only one comparative study examined the role of Hinotori and compare it with the DVM-SR in the context of RAA [86]. The only experience with RAA reported similar operative time (mean: 119 min, SD 8.7) to the DVM-SR (mean: 105 min, SD 22). Hinotori and Versius RARN presented similar operative time to the DVM-SR. In these three experiences, both arms completed all the surgeries without severe complications. More details on these studies can be found in Table 1, Table 2.
3.4.4. Other nononcological surgeries
Comparable total operative time between NM-SRs and the DVM-SR can also be seen for RACP, pyeloplasty, RASP, and RASN. High levels of safety have been demonstrated in these comparative studies even if these represent initial experiences [81], [87], [88], [89], [90].
Details on the comparative studies concerning benign diseases can be reviewed in Supplementary Tables 1 and 2.
3.5. Subgroup meta-analysis based on robot type for double-arm studies (NM-SR RARP vs DVM-SR RARP)
Owing to high heterogeneity for total operative time, CT, and EBL, a subgroup analysis based on the type of NM-SRs used was undertaken. Results of the analyses can be reviewed in Supplementary Figs. 5A, 5B, and 6.
4. Discussion
In this SR, we summarize initial experiences with NM-SRs and compare these emerging technologies with the current gold standard, the DVM-SR, which has dominated the market until recently and in which most urologists have considerable confidence and experience.
Recently, Ficarra et al [91] published an SR assessing the IDEAL framework of the published literature on NM-SRs. Most of the included studies were classified as those in the developmental or exploratory stages according to IDEAL, with few comparative studies. Only one randomized trial was included, showing similar outcomes to the Da Vinci system. However, the study has some limitations: granular perioperative outcomes were not evaluated, some studies within the timespan of the inclusion criteria were missing, case reports were included, and the RoB for single-arm studies was not assessed. Furthermore, a meta-analysis comparing NM-SRs with the DVM-SR was not performed.
Currently, no studies directly compared different NM-SRs; two ongoing trials aim to compare the perioperative outcomes of RARP (NCT05766163) and RAPN (NCT06290427) performed with the DVM-SR, Hugo RAS, and Versius [92], [93]. Their results are awaited eagerly.
We assessed the safety profile of NM-SRs during RARP and RAPN. Overall, both systems exhibited low percentages of intraoperative SATAVA grade 2 or superior complications, with comparable proportions of conversion to open and laparoscopic surgery. System failures and errors are common among NM-SRs. Most of the technical issues that occurred with Hugo RAS, such as arm conflicts, battery alarms, and instrument malfunctions, were minor and resolved easily. Antonelli et al [67] reported a significantly higher number of errors in the NM-SR arm than in the DVM-SR arm, with over 5 min required to resolve issues. On the contrary, Totaro et al [39] found that most of the problems occurred during the initial procedures, and a software update helped reduce the number of errors. Kulis et al [28] described a 5% of malfunctions in a cohort of 375 patients undergoing RARP in two institutions using the Senhance platform. System failures were also more common during initial procedures with Versius, with most issues related to arm conflicts [23].
Our analysis suggests that the total operative time may be similar between NM-SRs and the DVM-SR. However, the high heterogeneity observed in the analysis could compromise the result and may be attributed to factors such as patient selection, differences in robotic design, surgical approach, and the learning curve associated with navigating these new systems. By contrast, the familiarity of surgeons and staff with the DVM-SR, which has been in use for many years, likely contributes to faster operative times. NM-SRs introduce new techniques, workflows, and control systems that require a learning curve, particularly evident in larger series, where operative times are longer during initial procedures [27], [28], [47], [69], [75].
The learning curve associated with NM-SRs seems to be eased by prior experience with the DVM-SR for similar surgeries. Indeed, surgeons proficient with the DVM-SR appear to adapt swiftly to the controls and workflow of the Hugo RAS system, maintaining high efficiency [67], [76]. However, DT is longer with Hugo RAS, likely due to its design, which requires individually moving and docking each robotic arm and cart around the patient. In contrast, the DVM-SR is more centralized, with all robotic arms connected to a single console, facilitating quicker docking (Fig. 1).
RARP performed with the Hinotori system demonstrated a longer CT than that performed with the DVM-SR (I2: 0%; Supplementary Fig. 5B). This may be attributed to Hinotori’s design, which does not require docking of robotic arms, thereby reducing setup time and compensating for the longer CT related to the initial cases. The only comparative study focusing on the Revo-I system reported shorter CT in favor of the new robot [66]. Although this finding comes from a small cohort and the study has a moderate RoB, it is an important early result that warrants further exploration through well-structured RCTs.
Despite these results, the significant heterogeneity among studies on RARP and RAPN regarding total operative time, CT, DT, and EBL limits the interpretability of the results and prevents definitive conclusions.
Serious postoperative complications (CD ≥IIIa) were infrequent. These findings indicate that an NM-SR offers a safety level comparable with the DVM-SR, aligning with the high safety standards expected in modern RAS.
A similar result in terms of PSMs indicates that NM-SRs may be clinically equivalent to the DVM-SR in terms of precise tumor resection, although more granular analyses based on the type of nerve-sparing approach and the surgeon's overall surgical experience were not possible due to the nature of the data. Oncological safety is further supported by the apparent overlap in BCR rates at 3 mo between NM-SRs and the DVM-SR. Additionally trifecta achievement—a key outcome for conservative kidney surgery—was comparable between the two systems, as supported by the only one RCT [82]. Another important functional indicator of efficacy is urinary continence 3 mo after RARP, where NM-SRs demonstrated performance equal to that of the DVM-SR, showcasing their ability to preserve continence. Oncological and functional outcomes are not associated with significant heterogeneity.
Only a few studies explored the costs associated with these platforms [15], [73], [90]. Preliminary data suggest that NM-SRs may be a valid alternative for centers with limited budgets. For example, the CMR Versius, the smallest robot available, is priced between $800 000 and $1 000 000, which is significantly less than the price of the DVM-SR system [94]. Additionally, platforms such as Senhance and Dexter are compatible with the standard laparoscopic tools, increasing their versatility. However, the introduction of an NM-SR increases the workload and requires specific training for surgeons and nurses, which may limit their adoption in smaller hospitals.
Studies on the feasibility of surgeries performed with Senhance and Hugo RAS reported a comfortable seated position for the surgeon, without neck strain [51], [78], [94]. A study on the KangDuo system also found high levels of surgeon comfort using the National Aeronautics and Space Administration Task Load Index [24]. The docking-free design of Hinotori provides more space and a cleaner operative field, enhancing the efficiency of bedside assistants [79]. Finally, enhanced communication between the surgeon and the surgical staff has been reported for those NM-SRs with an open console such as Hugo RAS, Versius, and KangDuo.
Different NM-SRs offer several advantages. Health care institutions have the flexibility to choose platforms that best meet their specific clinical needs and budget. For example, modular platforms are often more compact, making them ideal for smaller operating rooms, and these are typically more affordable than systems such as Da Vinci Xi, Hinotori, or Avatera (Fig. 1). This increased accessibility can facilitate broader integration of NM-SRs into clinical practice worldwide.
The availability of multiple robots also fosters healthy competition, driving continuous innovation and improvements in both technology and cost effectiveness. As NM-SRs are introduced, manufacturers are incentivized to enhance their systems, leading to advancements in surgical capabilities and, ultimately, better patient outcomes. The future of NM-SRs looks promising, with rapid technological advancements and increasing adoption of platforms such as Versius and Dexter, especially following their recent clearances by the US Food and Drug Administration. As these systems continue to evolve, the focus may shift from comparing different robotic platforms based on clinical outcomes to evaluating their cost effectiveness relative to one another.
Future research could prioritize the standardization of surgical training for NM-SRs. This could help reduce variability across platforms and facilitate the adoption of NM-SRs, even in smaller hospitals, by ensuring consistent training and improving the overall efficiency and effectiveness of these technologies.
Our study has several limitations, the most notable being the heterogeneity among the included studies, most of which are retrospective and in the developmental or exploratory stages of using new devices, as classified by the IDEAL framework. Heterogeneity primarily arises from differences in patients’ selection, characteristics, robotic platforms used, surgical approach, and volume of the center. However, the heterogeneity associated with oncological and functional outcomes assessed through the meta-analysis was very low. This is a promising finding, potentially reflecting the accumulated experience of urologists with the DVM-SR over the past years. The lack of high-level evidence from well-designed RCTs with adequate follow-up restricts our ability to draw definitive conclusions.
A selection bias must be considered in studies examining initial experience. Surgeons, particularly those early in their experience with NM-SRs, might preferentially select cases they expect to be less complex or more likely to yield favorable outcomes, which can skew the results. Two other population-related biases are referral bias and healthy volunteer bias. Patients who are more likely to benefit from robotic surgery may be directed to centers that offer NM-SRs, resulting in a patient population that does not necessarily represent the broader cohort of individuals requiring surgery. At the same time, in nonrandomized trials, healthier and younger patients may be more likely to participate.
Given these potential biases, it is essential to interpret the findings of studies involving NM-SRs with caution. Only three studies included in the cumulative analyses are at a moderate RoB, while the rest are at a high risk, primarily due to unassessed confounders and missing selection criteria. This can lead to an overestimation of results in favor of NM-SRs, driven by confounders, selection bias, missing data, and short follow-up duration. While these studies provide valuable insights, they must be evaluated carefully in light of their methodological limitations and the potential influence of these biases. In the future, adequately powered RCTs may further strengthen the evidence gathered from our study. Single-port robotic surgery was excluded from our analysis due to its fundamentally different design, making comparisons with multiport systems, including the Da Vinci or other platforms, conceptually inappropriate and potentially misleading. Despite these challenges, this study provides the first comprehensive comparative synthesis on this rapidly evolving topic, offering valuable insights into the performance of new multisystem robotic platforms in urology.
5. Conclusions
NM-SRs have the potential to become viable alternatives to the DVM-SR, offering comparable feasibility, safety, and functional and oncological outcomes. However, the limited literature, which includes only one RCT and several nonrandomized retrospective studies primarily based on RARP and RAPN, must be considered when interpreting these results. Further research, particularly large-scale RCTs, is needed to validate these findings and fully understand the potential of NM-SRs in clinical practice.
Author contributions: Fabio Zattoni had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Study concept and design: Reitano, Zattoni, Novara.
Acquisition of data: Reitano, Tumminello, Ghaffar, Saggionetto, Taverna, Mangiacavallo.
Analysis and interpretation of data: Reitano, Ghaffar, Novara.
Drafting of the manuscript: Reitano, Tumminello, Zattoni, Novara.
Critical revision of the manuscript for important intellectual content: Basourakos, Ahmed, Carletti, Minardi, Morlacco, Betto, Iafrate, Karnes, Dal Moro.
Statistical analysis: Reitano.
Obtaining funding: None.
Administrative, technical, or material support: None.
Supervision: Zattoni, Novara.
Other: None.
Financial disclosures: Fabio Zattoni certifies that all conflicts of interest, including specific financial interests and relationships and affiliations relevant to the subject matter or materials discussed in the manuscript (eg, employment/affiliation, grants or funding, consultancies, honoraria, stock ownership or options, expert testimony, royalties, or patents filed, received, or pending), are the following: None.
Funding/Support and role of the sponsor: None.
Associate Editor: Roderick van den Bergh
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.euros.2025.02.003.
Appendix A. Supplementary material
The following are the Supplementary data to this article:
References
- 1.Franco A., Ditonno F., Manfredi C., et al. Robot-assisted surgery in the field of urology: the most pioneering approaches 2015–2023. Res Rep Urol. 2023;15:453–470. doi: 10.2147/RRU.S386025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ma J., Xu W., Chen R., et al. Robotic-assisted versus laparoscopic radical prostatectomy for prostate cancer: the first separate systematic review and meta-analysis of randomised controlled trials and non-randomised studies. Int J Surg. 2023;109:1350–1359. doi: 10.1097/JS9.0000000000000193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Koukourikis P., Rha K.H. Robotic surgical systems in urology: what is currently available? Investig Clin Urol. 2021;62:14–22. doi: 10.4111/icu.20200387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Marino F., Moretto S., Rossi F., et al. Robot-assisted radical prostatectomy performed with the novel Hugo™ RAS system: a systematic review and pooled analysis of surgical, oncological, and functional outcomes. J Clin Med. 2024;13:2551. doi: 10.3390/jcm13092551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Page M.J., McKenzie J.E., Bossuyt P.M., et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372 doi: 10.1136/bmj.n71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Munn Z., Moola S., Lisy K., Riitano D., Tufanaru C. Methodological guidance for systematic reviews of observational epidemiological studies reporting prevalence and cumulative incidence data. Int J Evid Based Healthc. 2015;13:147–153. doi: 10.1097/XEB.0000000000000054. [DOI] [PubMed] [Google Scholar]
- 7.Hung A.J., Cai J., Simmons M.N., Gill I.S. “Trifecta” in partial nephrectomy. J Urol. 2013;189:36–42. doi: 10.1016/j.juro.2012.09.042. [DOI] [PubMed] [Google Scholar]
- 8.Sterne J.A.C., Savović J., Page M.J., et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366 doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 9.Sterne J.A., Hernán M.A., Reeves B.C., et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355 doi: 10.1136/bmj.i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Knoll T., Omar M.I., Maclennan S., et al. Key steps in conducting systematic reviews for underpinning clinical practice guidelines: methodology of the European Association of Urology. Eur Urol. 2018;73:290–300. doi: 10.1016/j.eururo.2017.08.016. [DOI] [PubMed] [Google Scholar]
- 11.Sarkar J., Rashid M. Visualizing mean, median, mean deviation, and standard deviation of a set of numbers. Am Stat. 2016;70:304–312. [Google Scholar]
- 12.Luo D., Wan X., Liu J., Tong T. Optimally estimating the sample mean from the sample size, median, mid-range, and/or mid-quartile range. Stat Methods Med Res. 2018;27:1785–1805. doi: 10.1177/0962280216669183. [DOI] [PubMed] [Google Scholar]
- 13.Wan X., Wang W., Liu J., Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014;14:135. doi: 10.1186/1471-2288-14-135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Knežević N., Penezić L., Kuliš T., et al. Senhance robot-assisted adrenalectomy: a case series. Croat Med J. 2022;63:197–201. doi: 10.3325/cmj.2022.63.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kulis T., Hudolin T., Penezic L., et al. Comparison of extraperitoneal laparoscopic and extraperitoneal Senhance radical prostatectomy. Int J Med Robot. 2022;18 doi: 10.1002/rcs.2344. [DOI] [PubMed] [Google Scholar]
- 16.Prata F., Ragusa A., Tedesco F., et al. Trifecta outcomes of robot-assisted partial nephrectomy using the new Hugo™ RAS system versus laparoscopic partial nephrectomy. J Clin Med. 2024;13:2138. doi: 10.3390/jcm13072138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Alfano C.G., Moschovas M.C., Montagne V., et al. Implementation and outcomes of Hugo(TM) RAS system in robotic-assisted radical prostatectomy. Int Braz J Urol. 2023;49:211–220. doi: 10.1590/S1677-5538.IBJU.2023.9902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Andrade G.M., Lau C., Olivares R., et al. Implementation of robot-assisted urologic surgeries using Hugo™ RAS system in a high-volume robotic “Da Vinci Xi” center: outcomes and initial experience. Urology. 2024;192:44–51. doi: 10.1016/j.urology.2024.06.052. [DOI] [PubMed] [Google Scholar]
- 19.Bravi C.A., Paciotti M., Balestrazzi E., et al. Outcomes of robot-assisted radical prostatectomy with the Hugo RAS surgical system: initial experience at a high-volume robotic center. Eur Urol Focus. 2023;9:642–644. doi: 10.1016/j.euf.2023.01.008. [DOI] [PubMed] [Google Scholar]
- 20.Chang K.D., Abdel Raheem A., Choi Y.D., Chung B.H., Rha K.H. Retzius-sparing robot-assisted radical prostatectomy using the Revo-i robotic surgical system: surgical technique and results of the first human trial. BJU Int. 2018;122:441–448. doi: 10.1111/bju.14245. [DOI] [PubMed] [Google Scholar]
- 21.De Maria M., Meneghetti I., Mosillo L., Collins J.W., Catalano C. Versius robotic surgical system: case series of 18 robot-assisted radical prostatectomies. BJU Int. 2024;133:197–205. doi: 10.1111/bju.16156. [DOI] [PubMed] [Google Scholar]
- 22.Dell'Oglio P., Chierigo F., Cellini V., et al. Retzius-sparing robot-assisted radical prostatectomy with the Hugo™ robot-assisted surgery system: feasibility, operative setup and surgical outcomes. BJU Int. 2025;135:166–170. doi: 10.1111/bju.16455. [DOI] [PubMed] [Google Scholar]
- 23.Dibitetto F., Fede Spicchiale C., Castellucci R., et al. Extraperitoneal robot assisted laparoscopic prostatectomy with Versius system: single centre experience. Prostate Cancer Prostatic Dis. 2024;27:323–326. doi: 10.1038/s41391-024-00810-6. [DOI] [PubMed] [Google Scholar]
- 24.Fan S., Zhang Z., Wang J., et al. Robot-assisted radical prostatectomy using the KangDuo Surgical Robot-01 system: a prospective, single-center, single-arm clinical study. J Urol. 2022;208:119–127. doi: 10.1097/JU.0000000000002498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gkeka K., Kallidonis P., Peteinaris A., et al. Robot-assisted radical prostatectomy using the Avatera system™: a prospective pilot study. Minerva Urol Nephrol. 2024;76:52–59. doi: 10.23736/S2724-6051.23.05545-3. [DOI] [PubMed] [Google Scholar]
- 26.Hinata N., Yamaguchi R., Kusuhara Y., et al. Hinotori Surgical Robot System, a novel robot-assisted surgical platform: preclinical and clinical evaluation. Int J Urol. 2022;29:1213–1220. doi: 10.1111/iju.14973. [DOI] [PubMed] [Google Scholar]
- 27.Hudolin T., Kuliš T., Penezić L., et al. Senhance robotic radical prostatectomy: a single-centre, 3-year experience. Int J Med Robot Comput Assist Surg. 2023;19 doi: 10.1002/rcs.2549. [DOI] [PubMed] [Google Scholar]
- 28.Kulis T., Samalavicius N.E., Hudolin T., et al. Robotic-assisted radical prostatectomy: a multicenter experience with the Senhance surgical system. World J Urol. 2024;42:39. doi: 10.1007/s00345-023-04732-1. [DOI] [PubMed] [Google Scholar]
- 29.Marques-Monteiro M., Teixeira B., Mendes G., et al. Extraperitoneal robot-assisted radical prostatectomy with the Hugo™ RAS system: initial experience of a tertiary center with a high background in extraperitoneal laparoscopy surgery. World J Urol. 2023;41:2671–2677. doi: 10.1007/s00345-023-04571-0. [DOI] [PubMed] [Google Scholar]
- 30.Ou Y.C., Ou H.C., Juan Y.S., et al. Robot-assisted radical prostatectomy using Hugo RAS system: the pioneer experience in Taiwan and Northeast Asia. Int J Med Robot. 2023;20 doi: 10.1002/rcs.2577. [DOI] [PubMed] [Google Scholar]
- 31.Pokhrel G., Zheng H., Tao J., et al. Assessing the feasibility and safety of the Toumai® robotic system in urologic surgery: initial experience. J Endourol. 2024;38:552–558. doi: 10.1089/end.2024.0028. [DOI] [PubMed] [Google Scholar]
- 32.Polom W., Matuszewski M. Initial experience of the Versius robotic system in robot-assisted radical prostatectomy: a study of 58 cases. Cent Eur J Urol. 2024;77:30–36. doi: 10.5173/ceju.2023.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ragavan N., Bharathkumar S., Chirravur P., Sankaran S., Mottrie A. Evaluation of Hugo RAS system in major urologic surgery: our initial experience. J Endourol. 2022;36:1029–1035. doi: 10.1089/end.2022.0015. [DOI] [PubMed] [Google Scholar]
- 34.Reeves F., Challacombe B., Ribbits A., Ourselin S., Dasgupta P. Idea, Development, Exploration, Assessment, Long-term follow-up study (IDEAL) stage 1/2a evaluation of urological procedures with the Versius robot. BJU Int. 2022;130:441–443. doi: 10.1111/bju.15829. [DOI] [PubMed] [Google Scholar]
- 35.Sighinolfi M.C., De Maria M., Meneghetti J., et al. The use of Versius CMR for pelvic surgery: a multicentric analysis of surgical setup and early outcomes. World J Urol. 2024;42:31. doi: 10.1007/s00345-023-04730-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Takahara K., Motonaga T., Nakamura W., et al. Robot-assisted radical prostatectomy with the Hugo™ robot-assisted surgery system: a single-center initial experience in Japan. Asian J Endosc Surg. 2024;17 doi: 10.1111/ases.13342. [DOI] [PubMed] [Google Scholar]
- 37.Territo A., Uleri A., Gallioli A., et al. Robot-assisted oncologic pelvic surgery with Hugo™ robot-assisted surgery system: a single-center experience. Asian J Urol. 2023;10:461–466. doi: 10.1016/j.ajur.2023.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Thillou D., Robin H., Ricolleau C., et al. Robot-assisted radical prostatectomy with the Dexter robotic system: initial experience and insights into on-demand robotics. Eur Urol. 2024;85:185–189. doi: 10.1016/j.eururo.2023.05.034. [DOI] [PubMed] [Google Scholar]
- 39.Totaro A., Scarciglia E., Marino F., et al. Robot-assisted radical prostatectomy performed with the novel surgical robotic platform Hugo™ RAS: monocentric first series of 132 cases reporting surgical, and early functional and oncological outcomes at a tertiary referral robotic center. Cancers (Basel) 2024;16:1602. doi: 10.3390/cancers16081602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Veccia A., Malandra S., Montanaro F., Pettenuzzo G., De Marco V., Antonelli A. Mirrored port placement for robotic radical prostatectomy with the Hugo RAS™ System: initial experience. Minerva Urol Nephrol. 2024;76:235–240. doi: 10.23736/S2724-6051.23.05597-0. [DOI] [PubMed] [Google Scholar]
- 41.Venckus R., Jasenas M., Telksnys T., et al. Robotic-assisted radical prostatectomy with the Senhance(®) robotic platform: single center experience. World J Urol. 2021;39:4305–4310. doi: 10.1007/s00345-021-03792-5. [DOI] [PubMed] [Google Scholar]
- 42.Xiong S.W., Fan S.B., Chen S.L., et al. Robotic urologic surgery using the KangDuo-Surgical Robot-01 system: a single-center prospective analysis. Chin Med J. 2023;136:2960–2966. doi: 10.1097/CM9.0000000000002920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Paciotti M., Bravi C.A., Mottaran A., et al. Nerve-sparing robot-assisted radical prostatectomy with the HUGO™ robot-assisted surgery system using the 'Aalst technique'. BJU Int. 2023;132:227–230. doi: 10.1111/bju.16084. [DOI] [PubMed] [Google Scholar]
- 44.Gallioli A., Uleri A., Gaya J.M., et al. Initial experience of robot-assisted partial nephrectomy with Hugo™ RAS system: implications for surgical setting. World J Urol. 2023;41:1085–1091. doi: 10.1007/s00345-023-04336-9. [DOI] [PubMed] [Google Scholar]
- 45.Meneghetti I., Sighinolfi M.C., Dibitetto F., et al. Partial nephrectomy series using Versius robotic surgical system: technique and outcomes of an initial experience. J Robot Surg. 2024;18:73. doi: 10.1007/s11701-024-01843-8. [DOI] [PubMed] [Google Scholar]
- 46.Miyake H., Motoyama D., Matsushita Y., et al. Initial experience of robot-assisted partial nephrectomy using Hinotori surgical robot system: single institutional prospective assessment of perioperative outcomes in 30 cases. J Endourol. 2023;37:531–534. doi: 10.1089/end.2022.0775. [DOI] [PubMed] [Google Scholar]
- 47.Prata F., Basile S., Tedesco F., et al. Skill transfer from laparoscopic partial nephrectomy to the Hugo™ RAS system: a novel proficiency score to assess surgical quality during the learning curve. J Clin Med. 2024;13:2226. doi: 10.3390/jcm13082226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Prata F., Raso G., Ragusa A., et al. Robot-assisted renal surgery with the New Hugo RAS system: trocar placement and docking settings. J Pers Med. 2023;13:1372. doi: 10.3390/jpm13091372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Xu W., Dong J., Xie Y., et al. Robot-assisted partial nephrectomy with a new robotic surgical system: feasibility and perioperative outcomes. J Endourol. 2022;36:1436–1443. doi: 10.1089/end.2022.0140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Prata F., Ragusa A., Civitella A., et al. Robot-assisted partial nephrectomy using the novel Hugo™ RAS system: feasibility, setting and perioperative outcomes of the first off-clamp series. Urologia. 2024;91:372–378. doi: 10.1177/03915603231220109. [DOI] [PubMed] [Google Scholar]
- 51.Kaneko G., Shirotake S., Oyama M., Koyama I. Initial experience of laparoscopic radical nephrectomy using the Senhance(®) robotic system for renal cell carcinoma. Int Cancer Conf J. 2021;10:228–232. doi: 10.1007/s13691-021-00487-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dong J., Ji R., Liu G., et al. Feasibility, safety and effectiveness of robot-assisted retroperitoneal partial adrenalectomy with a new robotic surgical system: a prospective clinical study. Front Surg. 2023;10 doi: 10.3389/fsurg.2023.1071321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Raffaelli M., Gallucci P., Voloudakis N., et al. The new robotic platform Hugo™ RAS for lateral transabdominal adrenalectomy: a first world report of a series of five cases. Updates Surg. 2023;75:217–225. doi: 10.1007/s13304-022-01410-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Motoyama D., Matsushita Y., Watanabe H., et al. Robot-assisted radical nephroureterectomy for upper urinary tract tumor: initial experience with the use of novel surgical robot system, Hinotori. Transl Cancer Res. 2023;12:3522–3529. doi: 10.21037/tcr-23-853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Motoyama D., Matsushita Y., Watanabe H., et al. Robot-assisted radical nephrectomy and inferior vena cava tumor thrombectomy using the novel surgical robot platform, Hinotori: initial experience with two cases. IJU Case Rep. 2023;7:96–99. doi: 10.1002/iju5.12673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hayashi T., Kitano H., Hieda K., Hinata N. First case report of robot-assisted radical cystectomy and intracorporeal urinary diversion using the Hinotori surgical robot system. Transl Cancer Res. 2024;13:471–479. doi: 10.21037/tcr-23-991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rocco B., Sighinolfi M.C., Sarchi L., et al. First case of robot-assisted radical cystectomy and intracorporeal neobladder reconstruction with the Hugo RAS system: step-by-step surgical setup and technique. J Robot Surg. 2023;17:2247–2251. doi: 10.1007/s11701-023-01629-4. [DOI] [PubMed] [Google Scholar]
- 58.Elorrieta V., Villena J., Kompatzki Á., Velasco A., Salvadó J.A. Robot assisted laparoscopic surgeries for nononcological urologic disease: initial experience with Hugo RAS system. Urology. 2023;174:118–125. doi: 10.1016/j.urology.2023.01.042. [DOI] [PubMed] [Google Scholar]
- 59.Kallidonis P., Tatanis V., Peteinaris A., et al. Robot-assisted pyeloplasty for ureteropelvic junction obstruction: initial experience with the novel Avatera system. World J Urol. 2023;41:3155–3160. doi: 10.1007/s00345-023-04586-7. [DOI] [PubMed] [Google Scholar]
- 60.Kastelan Z., Hudolin T., Kulis T., et al. Upper urinary tract surgery and radical prostatectomy with Senhance(®) robotic system: single center experience—first 100 cases. Int J Med Robot. 2021;17 doi: 10.1002/rcs.2269. [DOI] [PubMed] [Google Scholar]
- 61.Mottaran A., Bravi C.A., Sarchi L., et al. Robot-assisted sacropexy with the novel HUGO robot-assisted surgery system: initial experience and surgical setup at a tertiary referral robotic center. J Endourol. 2023;37:35–41. doi: 10.1089/end.2022.0495. [DOI] [PubMed] [Google Scholar]
- 62.Mottaran A., Paciotti M., Bravi C.A., et al. Robot-assisted simple prostatectomy with the novel HUGO™ RAS System: feasibility, setting, and perioperative outcomes. Minerva Urol Nephrol. 2023;75:235–239. doi: 10.23736/S2724-6051.22.05031-5. [DOI] [PubMed] [Google Scholar]
- 63.Panico G., Vacca L., Campagna G., et al. The first 60 cases of robotic sacrocolpopexy with the novel HUGO RAS system: feasibility, setting and perioperative outcomes. Fron Surg. 2023;10 doi: 10.3389/fsurg.2023.1181824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Piro A., Piramide F., Balestrazzi E., et al. Initial experience of robot-assisted simple prostatectomy with Hugo robot-assisted surgery system: step-by-step description of two different techniques. J Endourol. 2023;37:1021–1027. doi: 10.1089/end.2023.0277. [DOI] [PubMed] [Google Scholar]
- 65.Rebuffo S., Ticonosco M., Ruvolo C.C., et al. Robot-assisted pyeloplasty with HUGO™ robotic system: initial experience and optimal surgical set-up at a tertiary referral robotic center. J Endourol. 2024;38:323–330. doi: 10.1089/end.2023.0598. [DOI] [PubMed] [Google Scholar]
- 66.Alip S., Koukourikis P., Han W.K., Rha K.H., Na J.C. Comparing Revo-i and da Vinci in Retzius-sparing robot-assisted radical prostatectomy: a preliminary propensity score analysis of outcomes. J Endourol. 2022;36:104–110. doi: 10.1089/end.2021.0421. [DOI] [PubMed] [Google Scholar]
- 67.Antonelli A., Veccia A., Malandra S., et al. Intraoperative performance of DaVinci versus Hugo RAS during radical prostatectomy: focus on timing, malfunctioning, complications, and user satisfaction in 100 consecutive cases (the COMPAR-P trial) Eur Urol Open Sci. 2024;63:104–112. doi: 10.1016/j.euros.2024.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bravi C.A., Balestrazzi E., De Loof M., et al. Robot-assisted Radical prostatectomy performed with different robotic platforms: first comparative evidence between Da Vinci and HUGO robot-assisted surgery robots. Eur Urol Focus. 2024;10:107–114. doi: 10.1016/j.euf.2023.08.001. [DOI] [PubMed] [Google Scholar]
- 69.Dibitetto F., Castellucci R., Russo P., et al. Extraperitoneal robot-assisted radical prostatectomy by the da Vinci and Versius system: first comparative analysis. Minerva Urol Nephrol. 2024;76:570–577. doi: 10.23736/S2724-6051.24.05792-6. [DOI] [PubMed] [Google Scholar]
- 70.Fan S.B., Hao H., Chen S.L., et al. Robot-assisted laparoscopic radical prostatectomy using the KangDuo surgical robot system vs the da Vinci Si robotic system. J Endourol. 2023;37:568–574. doi: 10.1089/end.2022.0739. [DOI] [PubMed] [Google Scholar]
- 71.Gandi C., Marino F., Totaro A., et al. Perioperative outcomes of robotic radical prostatectomy with Hugo™ RAS versus daVinci surgical platform: propensity score-matched comparative analysis. J Clin Med. 2024;13:3157. doi: 10.3390/jcm13113157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kohjimoto Y., Yamashita S., Iwagami S., Muraoka S., Wakamiya T., Hara I. Hinotori(TM) vs. da Vinci(®): propensity score-matched analysis of surgical outcomes of robot-assisted radical prostatectomy. J Robot Surg. 2024;18:130. doi: 10.1007/s11701-024-01877-y. [DOI] [PubMed] [Google Scholar]
- 73.Lin Y.C., Yuan L.H., Tseng C.S., et al. Comparison of Senhance and da Vinci robotic radical prostatectomy: short-term outcomes, learning curve, and cost analysis. Prostate Cancer Prostatic Dis. 2024;27:116–121. doi: 10.1038/s41391-023-00717-8. [DOI] [PubMed] [Google Scholar]
- 74.Menendez R.B., Rojo E.G., Palacios V.H., et al. Da Vinci vs. Hugo RAS for robot-assisted radical prostatectomy: a prospective comparative single-center study. World J Urol. 2024;42:336. doi: 10.1007/s00345-024-05045-7. [DOI] [PubMed] [Google Scholar]
- 75.Nakayama A., Izumi K., Ikezoe E., et al. Robot-assisted radical prostatectomy using the novel HinotoriTM surgical robot system: initial experience and operation learning curve at a single institution. Transl Cancer Res. 2024;13:57–64. doi: 10.21037/tcr-23-1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Olsen R.G., Karas V., Bjerrum F., et al. Skills transfer from the DaVinci® system to the Hugo™ RAS system. Int Urol Nephrol. 2024;56:389–397. doi: 10.1007/s11255-023-03807-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ou H.C., Marian L., Li C.C., et al. Robot-assisted radical prostatectomy by the Hugo robotic-assisted surgery (RAS) system and the da Vinci system: a comparison between the two platforms. Cancers (Basel) 2024;16:1207. doi: 10.3390/cancers16061207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ragavan N., Bharathkumar S., Chirravur P., Sankaran S. Robot-assisted laparoscopic radical prostatectomy utilizing Hugo RAS platform: initial experience. J Endourol. 2023;37:147–150. doi: 10.1089/end.2022.0461. [DOI] [PubMed] [Google Scholar]
- 79.Sasaki Y., Kusuhara Y., Oyama T., et al. Radical prostatectomy using the Hinotori robot-assisted surgical system: docking-free design may contribute to reduction in postoperative pain. Int J Med Robot Comput Assist Surg. 2024;20 doi: 10.1002/rcs.2648. [DOI] [PubMed] [Google Scholar]
- 80.Bravi C.A., Paciotti M., Sarchi L., et al. Robot-assisted radical prostatectomy with the novel Hugo robotic system: initial experience and optimal surgical set-up at a tertiary referral robotic center. Eur Urol. 2022;82:233–237. doi: 10.1016/j.eururo.2022.04.029. [DOI] [PubMed] [Google Scholar]
- 81.Hussein A.A., Mohsin R., Qureshi H., et al. Transition from da Vinci to Versius robotic surgical system: initial experience and outcomes of over 100 consecutive procedures. J Robot Surg. 2023;17:419–426. doi: 10.1007/s11701-022-01422-9. [DOI] [PubMed] [Google Scholar]
- 82.Li X., Xu W., Fan S., et al. Robot-assisted partial nephrectomy with the newly developed KangDuo surgical robot versus the da Vinci Si surgical system: a double-center prospective randomized controlled noninferiority trial. Eur Urol Focus. 2023;9:133–140. doi: 10.1016/j.euf.2022.07.008. [DOI] [PubMed] [Google Scholar]
- 83.Motoyama D., Matsushita Y., Watanabe H., et al. Perioperative outcomes of robot-assisted partial nephrectomy using Hinotori versus da Vinci surgical robot system: a propensity score-matched analysis. J Robot Surg. 2023;17:2435–2440. doi: 10.1007/s11701-023-01614-x. [DOI] [PubMed] [Google Scholar]
- 84.Rojo E.G., Palacios V.H., Menendez R.B., et al. Da Vinci and Hugo RAS platforms for robot-assisted partial nephrectomy: a preliminary prospective comparative analysis of the outcomes. Minerva Urol Nephrol. 2024;76:303–311. doi: 10.23736/S2724-6051.24.05623-4. [DOI] [PubMed] [Google Scholar]
- 85.Motoyama D., Matsushita Y., Watanabe H., et al. Robot-assisted radical nephrectomy using novel surgical robot platform, Hinotori: report of initial series of 13 cases. Int J Urol. 2023;30:1175–1179. doi: 10.1111/iju.15292. [DOI] [PubMed] [Google Scholar]
- 86.Motoyama D., Matsushita Y., Watanabe H., et al. Robot-assisted adrenalectomy using a Hinotori surgical robot system: report of first series of six cases. Asian J Endosc Surg. 2023;16:489–495. doi: 10.1111/ases.13212. [DOI] [PubMed] [Google Scholar]
- 87.Balestrazzi E., Paciotti M., Piro A., et al. Comparative analysis of robot-assisted simple prostatectomy: the HUGO™ RAS system versus the DaVinci® Xi system. Prostate Cancer Prostatic Dis. 2024;27:122–128. doi: 10.1038/s41391-023-00726-7. [DOI] [PubMed] [Google Scholar]
- 88.Collà Ruvolo C., Afonina M., Balestrazzi E., et al. A comparative analysis of the HUGO(TM) robot-assisted surgery system and the Da Vinci(®) Xi surgical system for robot-assisted sacrocolpopexy for pelvic organ prolapse treatment. Int J Med Robot. 2023;20 doi: 10.1002/rcs.2587. [DOI] [PubMed] [Google Scholar]
- 89.Fan S.B., Xiong S.W., Li Z.H., et al. Pyeloplasty with the KangDuo surgical robot vs the da Vinci Si robotic system: preliminary results. J Endourol. 2022;36:1538–1544. doi: 10.1089/end.2022.0366. [DOI] [PubMed] [Google Scholar]
- 90.Glass Clark S., Shepherd J.P., Sassani J.C., Bonidie M. Surgical cost of robotic-assisted sacrocolpopexy: a comparison of two robotic platforms. Int Urogynecol J. 2023;34:87–91. doi: 10.1007/s00192-022-05391-3. [DOI] [PubMed] [Google Scholar]
- 91.Ficarra V., Rossanese M., Giannarini G., et al. Evaluation of clinical research on novel multiport robotic platforms for urological surgery according to the IDEAL framework: a systematic review of the literature. Eur Urol Open Sci. 2024;67:7–25. doi: 10.1016/j.euros.2024.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.National Library of Medicine (USA). Comparative evaluation of new surgical robotic platforms for the radical prostatectomy procedure. https://clinicaltrials.gov/study/NCT05766163.
- 93.National Library of Medicine (USA). Comparison of outcomes of multiple platforms for assisted robotic-kidney. https://clinicaltrials.gov/study/NCT06290427.
- 94.Adhikari K., Penmetsa G.K., Krishnappa D., Taori R., Raghunath S.K. Revolutionizing urology: the advancements and applications of robotic platforms. J Robot Surg. 2024;18:106. doi: 10.1007/s11701-023-01758-w. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.