Abstract
Mitochondrial transfer is highly significant under physiological as well as pathological states given the emerging recognition of mitochondria as cellular “processors” akin to microchip processors that control the operation of a mobile device. Mitochondria play indispensable roles in healthy functioning of the brain, the organ with the highest energy demand in the human body and therefore, loss of mitochondrial function plays a causal role in multiple brain diseases. In this review, we will discuss various aspects of extracellular vesicle (EV)-mediated mitochondrial transfer and their effects in increasing recipient cell/tissue bioenergetics with a focus on these processes in brain cells. A subset of EVs with particle diameters >200 nm, referred to as medium-to-large EVs (m/lEVs), are known to entrap mitochondria during EV biogenesis. The entrapped mitochondria are likely a combination of either polarized, depolarized mitochondria or a mixture of both. We will also discuss engineering approaches to control the quality and quantity of mitochondria entrapped in the m/lEVs. Controlling mitochondrial quality can allow for optimizing/maximizing the therapeutic potential of m/lEV mitochondria—a novel drug with immense potential to treat a wide range of disorders associated with mitochondrial dysfunction.
Keywords: Extracellular vesicles (EVs), medium-to-large EVs, microvesicles, mitochondria, small EVs
Introduction
An emerging body of literature demonstrate that a subset of extracellular vesicles (EVs) with particle diameters >200 nm (also referred to as medium-to-large EVs (m/lEVs), ectosomes or microvesicles) incorporate mitochondria during their biogenesis. We and others have demonstrated the selective presence of mitochondria in cell-derived m/lEVs but not in the smaller EVs (sEVs)/exosomes.10–16 The transfer of m/lEV mitochondria resulted in a variety of responses in the recipient cells and tissues (depending on the disease/pathological model that was used) that primarily stemmed from the bioenergetic increases as a result of the EV-associated mitochondria. This review will discuss the premise of developing m/lEV mitochondria as a novel drug by discussing (1) hypotheses and mechanisms involved in the entrapment of mitochondria into the m/lEVs, (2) the effects of m/lEV mitochondria transfer into the recipient cells/tissues and (3) the promise of m/lEV-mediated mitochondria delivery as a next-generation drug to treat a variety of diseases associated with mitochondrial dysfunction. Given this niche focus on EV-mediated mitochondria delivery to brain cells, we had to extensively refer to our prior works while placing these findings in context with published literature as fit.
Terminology
Recommendations from the International Society of Extracellular Vesicles (ISEV) suggest that EVs such as exosomes and microvesicles be classified on the basis of particle diameters into either small EVs (sEVs)/exosomes (<200 nm in particle diameter) or medium-to-large EVs (m/lEVs)/microvesicles (>200 nm in particle diameter) unless the studies demonstrate evidence of real-time biogenesis of the EV subtype under investigation. 17 In accordance with the ISEV guidelines, we refer to exosomes that do not carry mitochondria as sEVs and the mitochondria-containing larger EVs as m/lEVs throughout this manuscript. To avoid any confusion upfront, our discussion will focus on mitochondria entrapment in the m/lEVs that originate from plasma membrane buds and not mitochondria-derived vesicles ( MDVs )/mitovesicles. MDVs originate from mitochondrial membranes, are released via endolysosomal pathways and are known to have functions related to disposal of damaged mitochondria. These damaged mitochondria contain mitochondrial damage-associated molecular patterns with proinflammatory activity and function as biomarkers in relevant experimental models/disease conditions.18,19 Readers interested in MDVs should refer to these excellent works.19–24
Rationale to develop m/lEV mitochondria as a novel drug
Two pivotal studies published by Hayawaka et al. 25 and Islam et al. 26 demonstrated the process of endogenous cell-to-cell mitochondrial transfer and the outcomes of such transfer in two different disease models: a CNS/brain disease model (transient ischemic stroke) and in a non-CNS/lung disease model, respectively. Hayakawa et al. demonstrated that the addition of astrocyte conditioned medium containing mitochondrial particles to oxygen-glucose deprived (OGD) neurons increased neuronal ATP levels and cell viability. In an in vivo mouse model middle artery occlusion of stroke, fluorescently labeled astrocytic mitochondrial particles (likely m/lEVs) were detected within the peri-infarct neurons at 24 h-post infusion into the cerebral cortex. These cortical neurons showed upregulation of pro-survival-related signals including phosphorylated AKT and BCL-XL concomitant with increased TOM40 expression, a mitochondrial marker. Downregulation of CD38 signaling via CD38 gene silencing experiments suppressed the endogenous mitochondrial astrocyte-to-neuron transfer resulting in decreased neuronal mitochondria that were accompanied with worsened neurological outcomes. These findings revealed a previously unknown mechanism of endogenous neuroprotection mediated by astrocytes via transfer of functional mitochondria—laying a strong foundation to our work in the pursuit of exogenous mitochondria delivery via m/lEVs as an approach to achieve therapeutic outcomes in stroke.14,15,27
Islam et al. 26 demonstrated that exogenously instilled bone-marrow-derived stromal cells (BMSCs) transfer mitochondria-containing microvesicles to alveolar epithelial cells via gap junction channels in mice. BMSC-mitochondrial transfer resulted in increased alveolar ATP levels and protected mouse lungs during the acute phase of lung injury by inducing protection against leukocytosis, albumin leakage and increased overall mouse survival. The protective effects of this mitochondrial transfer were contingent upon functional formation of connexin 43-containing gap junction channels between the instilled BMSCs and the alveolar epithelial cells. Collectively, the findings reported by Hayakawa et al. 25 and Islam et al. 26 demonstrate that endogenous intercellular mitochondrial transfer in vivo can overall increase recipient cell bioenergetics leading to improved therapeutic outcomes in two discrete, acute pathological models. Two recent works have documented the protective effects of endogenous mitochondrial transfer among cell types of the neurovascular unit (NVU), especially from astrocytes to BECs and from astrocytes to pericytes.28,29 In particular, this mitochondrial transfer plays key roles in maintaining the BBB structural integrity, with aging affecting the levels of mitochondrial transfer based on the experimental model that was employed.28,29 As previously reported by Hayakawa et al. 25 , naïve/endogenous EVs have shown to been involved in such mitochondrial transfer. Transfer of mitochondria from astrocytes to endothelial cells of the BBB sustained endothelial homeostasis, protected against endothelial dysfunction, and promoted angiogenesis. 28 These findings on the transfer of mitochondria via endogenous EVs among different cell types of the NVU and the resulting protective effects are encouraging and reinforce the paradigm of EV-mediated mitochondria delivery as a novel therapeutic approach. Defining the threshold of mitochondrial transfer that can lead to protective outcomes during the chronic stages of such disease conditions is a challenge and it is fair to hypothesize that exogenous delivery of mitochondria-containing vesicles at pre-defined doses is a promising approach to achieve optimal therapeutic responses in such disease models and beyond.
Biogenesis of m/lEVs and incorporation of mitochondria in m/lEVs
In contrast to the widely investigated mechanistic basis of exosome/sEV biogenesis pathways, the pathways of m/lEV biogenesis have been studied only to a very small extent. It was proposed that m/lEV biogenesis involves secretion of plasma membrane buds that carry cargo including nucleic acids, lipids and proteins. 40 In terms of molecular pathways, m/lEV formation in breast cancer cells involves the actin-myosin machinery and small GTPases, such as ADP-ribosylation factor 6 (ARF6).41,42 ARF6-GTP-dependent activation of phospholipase D promoted recruitment of extracellular signal-regulated kinase (ERK) to the plasma membrane followed by ERK-mediated phosphorylation and activation of myosin light-chain kinase (MLCK). Activation of MLCK caused actomyosin-based contractions at the necks of these structures that likely resulted in the pinching/budding off and release of microvesicles/m/lEVs from the plasma membrane into the extracellular spaces. The released m/lEVs were found to be an independent fraction compared to exosomes/sEVs that did not express ARF6. Importantly, the larger m/lEVs showed a dynamic range of particle diameters ranging from 300 to 900 nm in comparison to the much smaller sEVs that ranged from 50–70 nm as determined using transmission electron microscopy analysis. m/lEVs were sedimented at 10,000 × g while sEVs were sedimented at 100,000 × g to collect these two EV subsets from cell culture supernatants.
Experimental evidence for the direct plasma membrane budding process was reported by Nabhan et al. in HEK293T cells where m/lEVs/microvesicles containing arrestin domain-containing protein 1 (ARRDC1) and TSG101 were released from cell membranes and importantly, were devoid of exosomal markers—demonstrating m/lEVs as a unique subset of EVs. 43 These ARRDC1 pathway-mediated m/lEVs were speculated to carry plasma membrane proteins and also load RNA cargo, similar to exosomes. 43 While the classical tetraspannin markers CD9, CD63 and CD81 are widely documented in the sEVs, the corresponding evolution of markers for m/lEVs has been rather limited. Based on the involvement of Arf6 in m/lEV biogenesis, we have demonstrated the presence of Arf6 in the m/lEVs. 15
Entrapment of mitochondria into secreted m/lEVs
There are no published studies that report the mechanism of packaging of healthy/polarized mitochondria into m/lEVs. However, Phinney et al. reported mechanisms of packaging of partially depolarized mitochondria into the secreted m/lEVs from cultured mesenchymal stem cells (MSCs). MSCs subjected to oxidative damage showed the presence of partially depolarized mitochondria in secreted m/lEVs via an ARRDC1-mediated m/lEV release mechanism. The m/lEV mitochondria were reported to undergo fusion with recipient macrophages allowing regeneration of macrophage bioenergetics via increased oxygen consumption rates. 10
An unexplored hypothesis of mitochondria entrapment into m/lEVs
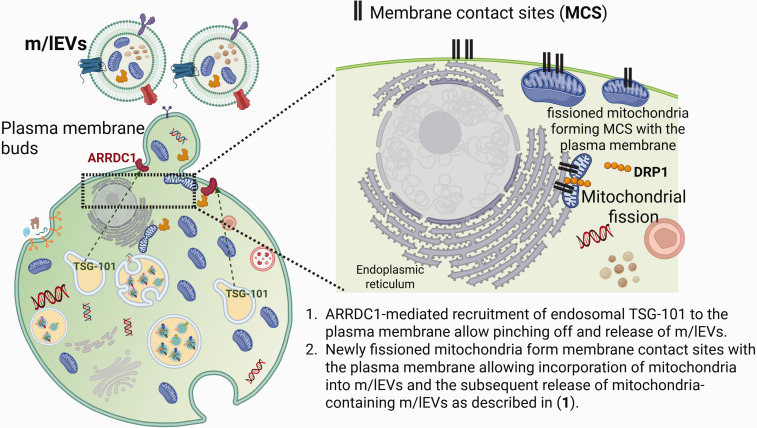
We have previously reported the presence of electron-dense mitochondria structures in sectioned EVs derived from basal/healthy mouse and human BECs using transmission electron microscopy analysis.14,15,39 A key question that arises upon observing mitochondria structures in the m/lEVs is that how do mitochondria that have reported lengths ranging from 0.5 to 3 µm fit into vesicles that have diameters ranging from 200 to 400 nm? As there are no studies that have described the mechanistic basis of entrapment of healthy mitochondria into m/lEVs, the following section discusses a possible hypothesis in context with our group’s findings. We focused our studies on EV mitochondria using BECs as a EV donor cells as the BBB capillary endothelial cells contain nearly a five-fold greater volume of mitochondria compared to peripheral and non-BBB endothelial cells. 44 We propose that the active dynamics of mitochondrial fission and fusion processes in mitochondria-rich cells such as BECs will allow incorporation of a greater amount of mitochondria into “cell buds” that may further mature into m/lEVs. 45 Membrane contact sites (MCS) between the endoplasmic reticulum (ER) and mitochondria are reported sites of mitochondrial fission and fusion.46,47 In addition to predominantly forming MCS with the ER, the outer mitochondrial membrane forms MCS with several intracellular organelles and membranes including plasma membrane, lysosomes, peroxisomes, endosomes, and lipid droplets. A key player in mitochondria fission is the dynamin-related protein 1 (DRP1).48,49 Briefly, DRP1 is dephosphorylated by calcineurin followed by which DRP1 is recruited to the mitochondrial surface where it binds to the DRP1 receptors—this DRP1-mediated fission is known to occur at sites where the ER wraps mitochondria (“fission hotspots”). This step is followed by oligomerization of DRP1 and GTP hydrolysis-mediated mitochondrial membrane constriction—leading to mitochondrial fission. It is likely that MCS at the mitochondria fission hotspots between the mitochondria and ER release smaller fragments of mitochondria that further form secondary MCS with the nearby plasma membrane followed by their constitutive release into cell buds/m/lEVs. 45 Mitochondria tether to the plasma membranes via the binding of mitofusin 1, a member of the DRP family of proteins, to the plasma membrane-associated pool of protein kinase C isoform-ζ in mammary stem cells but it is unknown if these interactions may happen in BECs as well. An alternate/parallel scenario to consider is related to the metabolic status of the BECs and how that may affect changes in mitochondrial morphology and subsequent incorporation of mitochondria in the m/lEVs.
Our EV isolation protocol involves exposing confluent BEC monolayers to serum-free medium 48 h prior to collecting EV-conditioned medium. The EV-conditioned medium is then subjected to a series of sequential centrifugation steps in order to first remove the cell debris/apoptotic bodies followed by separation of m/lEVs from the smaller EVs/exosomes.13–15,50 Apart from the regular mitochondrial dynamics that involves fission and fusion processes in cells under basal conditions, nutrient deprivation such as serum starvation that is used in our EV isolation protocol is also known to trigger DRP1-mediated mitochondrial fission. The DRP1-mediated fission occurs prior to elongation/mitochondrial fusion as a strategy to inhibit mitophagy/autophagy-mediated degradation to promote cell’s metabolic/mitochondrial fitness and ultimately avoid cell death. It is likely that a fraction of the “freshly-fissioned” mitochondria that have already formed MCS with the plasma membrane prior to the elongation/mitochondrial fusion may be incorporated into cell buds and released as m/lEVs. Figure 1 is a graphical representation of this proposed hypothesis on the biogenesis of m/lEVs incorporating mitochondria from healthy donor cells.
Figure 1.
Membrane contact sites formed between freshly-fissioned mitochondria and plasma membrane may result in the incorporation of those mitochondrial segments into the membrane buds that are then likely released as m/lEVs into the extracellular spaces via an ARRDC1-regulated mechanism described in the text.
Activation of DRP1 is also known to mediate the formation of ER-mitochondria MCS leading to mitochondrial fission to promote mitophagy, the selective degradation of damaged mitochondria via autophagosomes.51,52 If the proposed hypothesis for the incorporation of mitochondria via DRP1 and formation of ER-mitochondria and mitochondria-plasma membrane contact sites will be tested (in the future) and determined to be true, the next major question to be answered is the degree of overlap between the fraction of mitochondria that will be entrapped in the m/lEVs vs. the fraction of mitochondria that will be marked as ready for mitophagy. However, the observed pro-bioenergetic responses of the delivered m/lEV mitochondria (see Figure 2) seems to suggest that the mitochondria entrapped into m/lEVs are indeed healthy and functional excluding the possibility that m/lEV mitochondria are mitophagic. Given the central role of DRP1 in maintaining mitochondrial health and homeostasis via mediating mitochondrial fission, it is reasonable to speculate that DRP1 is involved in multiple aspects of mitochondrial dynamics in cells—including the trafficking and incorporation of mitochondria into m/lEVs.
Figure 2.
DLS Z-Average particle diameter, zeta potential and NTA particle concentration (a). Detection of EV marker proteins using western blotting. Each lane was loaded with 50 μg total protein, electrophoresed, immunoblotted and scanned on an Odyssey imager (b). EVs were cross sectioned as described by us previously [14–15]. m/lEVs contained one or more mitochondria noted as electron dense structures while sEV cross sections lacked electron dense structures. Images are representative sections obtained from a JEOL JEM 1400 Plus TEM (c). Oxygen consumption rates (OCR) and extracellular acidification rates (ECAR) were measured by treating cells with the indicated samples (EV particles) in OGD medium. We used a standard Mitochondrial Stress Test protocol on an Agilent Seahorse XFe24 Analyzer. All rates were normalized to cellular protein content measured using MicroBCA assay. Data are mean ± SEM, n = 3 (d). Quantification of total hemispheric infarct volume and neurological deficit scoring (NDS) at 24 h post-stroke (e). Data are mean ± SEM (n = 8). OGD primary HBMECs were incubated with the indicated samples for 24 h and were washed once in 1x PBS prior to measuring relative cellular ATP levels using a Cell Glo luminescence assay (f and g). Data represent average ± SD (n=3). Donor BECs were treated with 0.5 or 1 µM rotenone (RTN) to inhibit mitochondrial complex I prior to isolating EVs from mitochondria-impaired donor BECs (RTN-EVs). OGD HBMECs were then treated at the medium dose of 225,000 EV particles (g). Figures from our previous publications have been reproduced with permission from Elsevier (b to e) and Springer (g).
In summary, the above hypotheses on the incorporation of mitochondria into m/lEVs remain to be evaluated experimentally. Insights into the biogenesis of m/lEVs containing shorter (not full-length) pieces yet healthy/functional pieces of mitochondria may allow for further bioengineering approaches to increase the quality and quantity of mitochondria in the m/lEVs and ultimately increase the therapeutic index of these carriers while allowing for administration of lower doses/lower m/lEV numbers.
Entrapment of mitochondrial proteins and mitochondrial DNA (mtDNA) in the smaller EVs (sEVs/exosomes)
In addition to mitochondria being released into m/lEVs, the sEVs that are derived from endosomal biogenesis pathways are known to entrap mitochondrial proteins and mtDNA due to the interactions of endosomes and mitochondria 53 likely via the formation of MCS as described in the above section. Mitochondria that are uncoupled from respiratory chain (and therefore do not produce ATP anymore) recruit phosphatase and tensin homolog-induced putative kinase 1 (PINK1) onto the outer mitochondrial membranes following which PINK1-expressing mitochondria interacted with late endosomes/multivesicular bodies (MVBs) that progressed to release sEVs upon fusion with the plasma membrane. While silencing the cellular expression of PINK1 via siPINK1 transfection of the donor MDA-MB-231 breast cancer cells or primary mammary tumor cells and PINK1 knockout cell lines showed decreased release of mtDNA-containing EVs, exogenous transfection of PINK1 restored the release of mtDNA-containing EVs. These mtDNA-containing cancer cell-derived EVs promoted metastatic invasion in the recipient MDA-MB-231 cells. It is important to recognize that the nature and quality of the mitochondrial load that is entrapped within EVs strongly reflects the donor cell’s metabolic/pathological status (see more in section ‘m/lEV characteristics, transfer, and functional activity of m/lEV mitochondria in recipient cells and mice’).
Rationale to develop BEC-derived mitochondria-containing m/lEVs
Given our longstanding interest in protecting the post-ischemic blood-brain barrier (BBB), our laboratory has developed m/lEVs derived from both mouse and human brain endothelial cell lines (BEC) as a carrier for the delivery of innate m/lEV mitochondria to the BBB.13–15,27,38,39,45 While neuroprotection has largely dominated the development of stroke therapies, the importance of protecting brain BECs lining the BBB is increasingly recognized.30–34 Loss of BEC mitochondria increases BBB permeability and worsens post-stroke outcomes.35–37 Given the focus of our studies on delivery of mitochondria-containing m/lEVs to treat the ischemic BBB post-stroke,13–15,27,38,39 the rest of the discussion in this review will stay focused with respect to mitochondrial transfer to brain cell models. One key advantage of the process of exogenous delivery of mitochondria via EVs is based on the rationale that this delivery paradigm can model the natural/endogenous repair mechanisms used by the brain post-stroke, for example, the astrocyte to neuron mitochondrial transfer discovered by Hayakawa et al. 25
m/lEV characteristics, transfer, and functional activity of m/lEV mitochondria in recipient cells and mice
Particle diameters and protein markers of BEC-derived sEVs vs. m/lEVs were studied using dynamic light scattering and western blotting, respectively.13–15,27 Mitochondria-containing m/lEVs showed particle diameters ca. 400 nm, likely reflective of the increased vesicle size as well as volume due to mitochondria entrapment in contrast to sEVs that showed particle diameters ca. 130 nm. Both sEVs and m/lEVs showed weakly anionic surface charges (reported as ζ-potential) indicative of the phospholipid membranes surrounding the EVs. EV particle concentrations measured using nanoparticle tracking analysis revealed comparable particle concentrations for both sEVs and m/lEVs (Figure 2(a)). While both sEVs and m/lEVs showed expression of ATP5A: a subunit of mitochondrial ATP synthase (a mitochondrial matrix protein), the mitochondrial membrane protein TOMM20 (translocase of the outer mitochondrial membrane protein 20) was detected only in the larger, mitochondria-containing m/lEVs (Figure 2(b)).14,15 CD63 was used as a tetraspannin marker for the sEVs/exosomes and was not detected in m/lEVs. 54 The presence of electron-dense cristae structures in the m/lEV mitochondria (Figure 2(c)) detected using transmission electron microscopy images of ultrathin EV sections suggest that these mitochondria are capable of participating in oxidative phosphorylation (OXPHOS) as cristae organization is important to allow optimal conditions for ATP production while minimizing the diffusion of protons, ADP and metabolites during mitochondrial respiration. 55 Indeed, recipient oxygen-glucose deprived (OGD) BECs treated with BEC-derived m/lEVs show increased rates of oxygen consumption rates (OCR) measured using Seahorse analysis (Figure 2(d)).14,15,27 Hypoxic BECs treated with m/lEVs showed a significant, dose-dependent increase in the maximal OCR compared to untreated cells and cells treated with sEVs. The basal and maximal OCR increased by 179 and 269% and 147 and 224% in the groups treated with m/lEVs at doses of 30,000 and 150,000 m/lEVs (compared to untreated and sEV-treated cells), respectively. Concomitant with the OCR increases, the maximum glycolytic capacity of cells treated with 150,000 m/lEVs also increased by ∼200% compared to untreated BECs. We noted small, non-significant changes in proton leak among the groups suggesting that the mitochondrial respiration is linked to ATP production via oxidative phosphorylation (OXPHOS). In addition to the effects of the delivered m/lEV mitochondria on recipient BEC mitochondrial function, we also studied the potential therapeutic effects of BEC-derived m/lEVs that were injected two hours post stroke-onset in a transient mouse model of middle cerebral artery occlusion (MCAo). Mice were injected with an equivalent of 2*109 particles via an intravenous ( i.v. ) tail vein injection and 24 h-post injection, brain slices stained with 2,3,5-triphenyltetrazolium chloride revealed that m/lEV-injected mice showed a 50% lower cerebral infarct volume and a 33% improvement in neurological functions (measured as changes in neurological disease score (NDS)) in comparison to control mice injected with saline (Figure 2(e)). Importantly, no acute toxicities were noted in mice injected with m/lEVs—indicating the potential safety of i.v. injected m/lEVs containing mitochondria. While we observed the above therapeutic effects, further whole body/serum pharmacokinetic studies will need to be done to determine the % injected dose that reached the BBB/brain tissue and mechanistic studies measuring mitochondrial function in the isolated BBB microvessels/brain tissue will reveal if m/lEV mitochondria are responsible for any of the observed effects. These studies are currently underway in our laboratory.
In addition to Seahorse assays, we studied the effect of BEC-derived m/lEVs on recipient OGD BECs by measuring changes in relative cellular ATP levels using a luminescence assay. The amount of oxyluciferin (measured here as the luminescent output) produced from a luciferin-luciferase oxidation reaction is correlated with the amount of ATP present in the cells. 56 It is important to note that while this assay does not allow to measure absolute concentrations of intracellular ATP, the readout can be expressed as relative cellular ATP levels—with the use of appropriate experimental controls such an untreated OGD BECs and normoxic BECs. OGD primary human brain microvessel endothelial cells (HBMECs) treated with sEVs and m/lEVs resulted in a 140 to 200-fold increase in relative ATP levels compared to control, untreated cells. While both sEVs and m/lEVs increased ATP levels, the m/lEV-mediated increases were greater in magnitude and demonstrated a dose-dependent increase compared to sEV-treated cells and control untreated OGD BECs (Figure 2(f)).14,15 The increases noted with sEV-treated cells is likely explained by the presence of cytoprotective heat shock proteins (HSP) in the EVs detected using proteomic analysis. 27 Our proteomic analysis of BEC-EVs revealed the presence of several endogenous HSPs (HSP70, HSP71A, HSPA8, HSPA5, HSP105, HSP90A/B and HSPβ1) 27 that may have contributed to the observed ATP increases (Figure 2(f)). HSPs contribute to the preservation of ATP levels during stress conditions by interrupting cytochrome c translocation from the mitochondria to the nucleus.57,58 It is important to note that while both subtypes of EVs studied in our works contained HSPs, the greater magnitude of increases noted with the m/lEV-treated cells is likely reflective of their mitochondrial content.
In order to further understand the specific contribution of m/lEV mitochondria towards these bioenergetic responses, we isolated EVs from donor BECs that were pretreated either with rotenone (RTN) or oligomycin (OGM), inhibitors of mitochondrial complex I or complex V.13–15 Interestingly, recipient OGD BECs treated with RTN-m/lEVs showed sharp decline in relative ATP levels as a function of the RTN concentration used to block mitochondrial complex I in the donor BECs (in contrast to cells treated with naïve m/lEVs generated from healthy BECs not treated with RTN)—suggesting that the mitochondrial content in naïve m/lEVs is responsible for the ATP-increasing functionality of the m/lEVs. More interestingly, recipient BECs treated with both naïve sEVs or RTN-sEVs showed comparable levels of relative cellular ATP levels, suggesting no apparent effects of RTN on sEV functionality (Figure 2(g)).13–15 OGM-m/lEVs on the other hand completely abrogated cell survival in contrast to the RTN concentration dependent loss of m/lEV activity in the recipient BECs (not shown, see our paper 13 ). In addition to total loss of the ATP-increasing functionality, OGM-m/lEVs showed particle diameters measured using dynamic light scattering that were nearly 200% greater than that of naïve m/lEVs while naïve sEV and OGM-sEV diameters remained comparable—indicating that the OGM-treated donor BECs packaged likely larger aggregates of depolarized/damaged mitochondria. 14 Collectively, these results demonstrating the selective loss of m/lEV (when m/lEVs were derived from donor BECs with compromised mitochondrial function) but not sEV functionality in the recipient BECs reconcile well with the presence of mitochondria in m/lEVs but not sEVs (see sections ‘Biogenesis of m/lEVs and incorporation of mitochondria in m/lEVs’ and ‘m/lEV characteristics, transfer, and functional activity of m/lEV mitochondria in recipient cells and mice’). Combining our observations from physicochemical characterization and biological assays, we suggest that the donor cell mitochondrial status (healthy vs. damaged, polarized vs. depolarized) determines the quality of mitochondrial load that is packaged into the m/lEVs (Figure 3).
Figure 3.
Mitochondrial status in the donor cells (healthy (a) vs. damaged (b)/polarized vs. partially or fully depolarized) determines the quality of mitochondrial load that is packaged into the m/lEVs and ultimately, the resulting ATP levels in the recipient BECs.
Transfer of m/lEV mitochondria into recipient cells and tissues
The tracking of m/lEV mitochondria in the recipient cells is helpful to determine (a) if the EV mitochondria do enter the recipient cells and (b) the subcellular localization of the EV mitochondria in the recipient cells. Multiple studies (including ones that used experimental models other than brain cells) commonly use mitochondria labeling dyes such as MitoTracker (MitoT) Deep Red and MitoT Green that label polarized mitochondria via binding to thiol‐reactive chloromethyl groups in the mitochondrial membrane and mitochondrial proteins via covalent binding of the dye to cysteine thiols in the inner mitochondrial membrane, respectively. 59 A widely used protocol is to first label donor cells with MitoT dyes followed by subsequent isolation of MitoT-labeled EVs for use in subsequent experiments using flow cytometry and fluorescence microscopy techniques (Figure 4(a)).13–15,27,60 Recipient BECs treated with MitoT-m/lEVs showed strong intracellular puncta after 24 h of incubation suggesting efficient cellular uptake (Figure 4(b)). Both normoxic as well as hypoxic recipient BECs treated with MitoT-EVs for 24 h showed a dose-dependent increase in %+MitoT-recipient BECs (Figure 4(c)) albeit hypoxic BECs showed a 10% lower MitoT-EV uptake. In addition, recipient BECs treated with MitoT-m/lEVs showed a greater degree of %+MitoT-recipient BECs compared to recipient BECs treated with MitoT-sEVs at the same doses. It is likely that MitoT-Deep Red used in this experiment also likely stains thiol-reactive mitochondrial proteins that are known to be present in sEVs.61–63 MitoT-m/lEV mitochondria seemed to undergo fusion with the endogenous mitochondria in recipient primary human brain microvascular endothelial cells (HBMECs) (Figure 4(d)). Recipient HBMECs were first transduced with CellLight Mitochondria-GFP BacMam that labeled the alpha pyruvate mitochondrial matrix protein in the recipient HBMECs followed by the which the cells were treated with MitoT-EVs. 14 The overlap of MitoT-EV signals and the endogenous mitochondria in recipient HBMECs (Figure 4(c)) combined with the increase in mitochondrial function and relative ATP levels (Figures 2(f) and (g)) seems to suggest that the delivered m/lEV mitochondria contribute to recipient BEC OXPHOS (Figure 2(d)).
Figure 4.
MitoTracker Red labeling of donor BECs allows tracking and intracellular uptake of mitoT-EVs into recipient BECs (a) using fluorescence microscopy (b) and flow cytometry (c). MitoT-EVs colocalized with recipient HBMEC mitochondria pre-transduced with CellLight Mitochondria-GFP BacMam to tag the alpha pyruvate mitochondrial matrix protein (d). MitoT-EVs were also internalized by cells in cortical and hippocampal mouse brain slices (e and f). Slice insets show MitoT signals without DAPI for clarity. Mean intensity values were normalized to control slices. Figures have been reproduced from our previous publications with permission from Elsevier (b to f).
Control of mitochondrial quality and quantity packaged into m/lEVs
As discussed in the above section, one of the factors that affects the quality of mitochondria entrapped into m/lEVs seems to be the metabolic fitness of the donor cell mitochondria. Other factors such as cell culture conditions, growth medium composition (presence/absence of serum), metabolic health/fitness of the donor cells may also determine the quality of the m/lEV mitochondria. As discussed in section ‘m/lEV characteristics, transfer, and functional activity of m/lEV mitochondria in recipient cells and mice’, nutrient starvation can be one of the triggers for DRP1-mediated mitochondrial fission, 46 leading to changes in the cell’s mitochondrial dynamics likely altering the rate and frequency of fission/fusion. It can be anticipated that a combination of the above-listed factors will affect the cell’s metabolic/mitochondrial fitness and therefore, the quality of mitochondria entrapped and secreted in the m/lEVs. Overall, the quality of m/lEV mitochondria will determine the extent of functional/metabolic activity in the recipient cells and tissues.
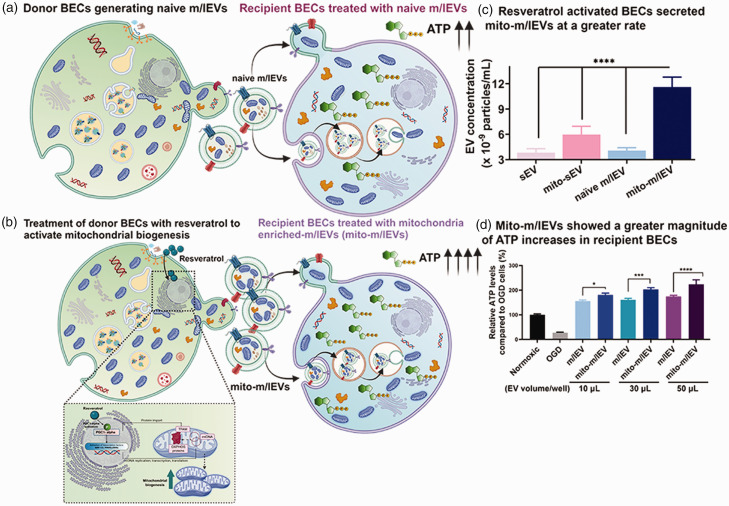
Apart from the mitochondrial quality, the quantity of mitochondria entrapped in the m/lEVs is a significant determinant of the extent of mitochondrial function in the recipient cells/tissues. We have detected anywhere from one to five electron-dense mitochondria structures in the sectioned EVs under transmission electron microscopy analysis.14,15 We developed a simple bioengineering-based approach to control the quality and quantity of m/lEV mitochondria by increasing the mitochondrial biogenesis in the donor BECs—we refer to these EVs as mitochondria-enriched EVs (mito-EVs) (Figure 5). 13
Figure 5.
Activation of PGC-1α using resveratrol increased mitochondrial biogenesis in the donor BECs (a-b) followed by increased rates of m/lEV secretion into the extracellular spaces (c) compared to naïve m/lEVs collected from non-activated/basal cells. Mitochondria-enriched EVs (mito-m/lEVs) showed a greater magnitude of relative ATP increases compared to naïve m/lEVs in the recipient OGD BECs. Figures (c and d) have been reproduced from our previous publications with permission from Springer.
Peroxisome proliferator-activated receptor-gamma coactivator -1 alpha (PGC-1α) is the master modulator of mitochondrial biogenesis in cells 64 and we hypothesized that activation of PGC-1α in the donor cells/BECs may increase the mitochondrial biogenesis and thereby increase the packaging of new/healthy mitochondria into the secreted m/lEVs. 13 To test this hypothesis, we chose resveratrol, a natural anti-inflammatory compound found in dietary substances such as grapes, berries, peanuts, etc. 65 EV-conditioned medium collected from donor BECs pre-treated with resveratrol (10 µM) were subjected to the standard sequential centrifugation and mito-EVs (mito-m/lEVs or mito-sEVs) were isolated as described by us previously. 13 EVs derived from basal/non-activated BECs were referred to as naïve EVs (naïve m/lEVs or naïve sEVs) and were used as a control in all of the experiments to determine the effect of PGC-1α activation in the donor BECs and the resulting changes in the mitochondrial load in the secreted mito-m/lEVs. Mito-m/lEVs were secreted at particle concentrations that were 2.8-fold greater compared to naïve-m/lEVs and the particle concentrations of mito-sEVs were about 1.6-fold greater compared to naïve-sEVs (Figure 5(c)). 13 These increased particle concentrations likely reflect that PGC-1α activation of the donor BECs increased mitochondrial biogenesis followed by a greater rate of secretion of m/lEVs containing mitochondria. The particle diameters of naïve sEVs vs. mito-sEVs and naïve m/lEVs vs. mito-m/lEVs seemed to comparable suggesting that similar amounts of mitochondrial content was packaged by EVs released from both the basal/non-activated as well as the PGC-1α-activated donor BECs—however, the rate of EV secretion/particle concentrations were increased (as described previously) in the case of mito-m/lEVs suggesting that the PGC-1α-activated donor BECs contained an increased mitochondrial load.
As noted earlier in this section, we have noted anywhere from one to five electron-dense/mitochondria structures in the naïve m/lEVs 14 and noted similar ranges of electron dense structures in the mito-m/lEVs as well. 13 In addition to the increased particle concentrations, mito-sEVs showed a 14-fold enrichment in PGC-1α mRNA levels compared to naïve sEVs—demonstrating the potential of mito-sEVs to stimulate de novo mitochondria biogenesis in recipient cells/tissues (unpublished, Manickam laboratory). OGD BECs treated with mito-m/lEVs showed a 16 to 40% increase in relative ATP levels in a dose-dependent manner compared to cells treated naïve m/lEVs at equivalent volumes (Figure 5(d)). This finding suggests that the increased mitochondrial quantity/quality in the mito-m/lEVs compared to naïve m/lEVs impact the functional activity of the delivered EVs in the recipient BECs. 13 In summary, our approach on increasing donor cell mitochondrial biogenesis via PGC-1α activation using resveratrol resulted in both increased numbers of m/lEVs with mitochondria, likely improved the quality of mitochondria in those mito-m/lEVs as well increased amounts PGC-1α mRNA in the mito-sEVs. In addition to resveratrol, we have identified a list of pharmacological compounds that can increase PGC-1α that will be investigated in our future works as an approach to identify optimal activation parameters to increase the mitochondrial quality and quantity of the secreted m/lEVs. 66 We anticipate that the mitochondria-enriched m/lEVs will mediate a greater extent of cerebroprotective effects in experimental models of stroke as well in other disease models where increasing mitochondrial function can have therapeutic effects.1–9
Mitochondrial transplantation vs. EV-mediated mitochondria delivery
In this section, we compare considerations for the translation of EV-mediated mitochondria delivery vs. mitochondrial transplantation for clinical applications. Mitochondrial transplantation involves a procedure in which mitochondria are isolated from a biofluid/cellular/tissue source and delivered to elicit a therapeutic response. 67 Mitochondrial transplantation has demonstrated improved therapeutic outcomes in experimental models of ischemic-reperfusion injury, stroke, traumatic brain injury, lung injury and skeletal muscle injury.68–75 Sustained outcomes of mitochondrial transplantation relies on the successful engraftment and retention of mitochondria in the target cells. There are ongoing clinical trials on mitochondrial transplantation and the outcomes of these trials are not published yet. 76 EV-mediated mitochondrial delivery is still in its nascent stages compared to the advances made in the area of mitochondrial transplantation and the clinical promise of the EV-based approach to deliver mitochondria is expected to evolve in the upcoming years. One advantage of EV-mediated mitochondrial transfer relies on tapping into an optimal cell source of EVs: homotypic interactions between the EV membranes and recipient cells may allow increased transfer of EV mitochondria into the recipient cells. We have previously demonstrated that BEC-derived EVs transferred mitochondria to recipient BECs and concurrently increased relative ATP levels in the recipient BECs to a greater extent compared to cells treated with macrophage-derived EVs. 27 Jiang et al. reported an interesting finding that EVs isolated from mesenchymal stromal cells (MSC-EVs) stabilized free mitochondria collected from fibroblasts in cold storage (2°C–6°C). 77 A mixture of MSC-EV+mitochondria (1:9 vol:vol) retained the mitochondrial membrane potential (MMP) and the ATP content of the mitochondria whereas fibroblast mitochondria stored in solution showed a 70% loss in MMP and concurrent losses in ATP levels. Despite reporting these interesting findings, this study did not examine the nature of the interactions between the MSC-EVs and the isolated mitochondria that likely translated to the reported improvements in stability and function of the mitochondria.
Open questions, perspectives, and future directions
Mitochondria entrapment into m/lEVs, isolation and characterization of m/lEVs containing mitochondria
There are no published reports that report the mechanistic basis of mitochondria entrapment into m/lEVs and we have presented a speculative hypothesis related to the incorporation of freshly-fissioned mitochondria at the membrane contact sites between the plasma membrane cell buds and the mitochondria into the secreted m/lEVs (Figure 1). Understanding of the mechanism may allow for improved control over the quality of mitochondria that is entrapped into the m/lEVs. Currently, sequential centrifugation allows separating the sEVs vs. m/lEVs from EV-conditioned cell culture medium and this technique works quite well resulting in nominal yields of both EV fractions. 78 Although there are a variety of EV isolation methods that are applicable to improving the yields and purification of sEVs/exosomes, only density gradient centrifugation allows further purification of m/lEVs from non-vesicular extracellular particles.79–81 Development of affinity-based chromatography protocols that can specifically isolate mitochondria-containing m/lEVs will be a significant advance that will allow rapid isolation and further purification of mitochondria-containing m/lEVs. In addition to developing methods to isolate mitochondria-m/lEVs, it will be beneficial to further refine such methods to specifically isolate m/lEVs containing polarized mitochondria while avoiding depolarized mitochondria in the m/lEVs that may beyond a certain threshold cause detrimental effects in the recipient cells. Our findings to date suggest that dosing cells/mice in the range between 104–109 m/lEVs do not cause acute toxicities in recipient BECs or MCAo mice.13–15,27,50 However, further studies are required to determine if dose escalation of >109 EVs or repeated dosing in mice will cause any toxicities—both acute and chronic.
As discussed in earlier in section ‘Biogenesis of m/lEVs and incorporation of mitochondria in m/lEVs’, Arf6 seems to be the sole marker of m/lEVs and identification and validation of additional markers will improve the rigor and reproducibility of independent isolations of m/lEVs. We and others have reported TOMM20 as a marker of mitochondria-containing m/lEVs and TOMM20 has been reported in mitochondria-derived vesicles as well14,15,61,82 Ikeda et al. 11 have reported the presence of multiple other mitochondrial electron transport chain (ETC) proteins such as ATP5A (V), UQCRC2 (III), SDHB(II), COXII (IV) and NDUFB8 (I) in large/mitochondria-containing EVs from cardiomyocytes. It will be important to determine if those ETC proteins are also present in BEC-derived m/lEVs. If the speculated hypothesis on incorporation of ER-fissioned mitochondria into m/lEVs holds up to be true (section ‘Biogenesis of m/lEVs and incorporation of mitochondria in m/lEVs’), it will allow the further development of lipids involved in the ER-mitochondria contact sites 83 as potential markers of mitochondria-containing m/lEVs.
Mechanisms of mitochondria transfer from m/lEVs to the recipient cells
We and others have demonstrated that the colocalization of m/lEV mitochondria with recipient cell mitochondria in BECs as well as non-BEC models (cardiomyocytes).11,13,14 However, the exact mechanism of this transfer is not known yet – how do the m/lEVs release the mitochondrial load and do the released mitochondria diffuse and integrate with the recipient cell mitochondrial network? The overlap of fluorescent signals between fluorescently tagged EV mitochondria and recipient cell mitochondria (Figure 4(d)) does not indicate if the overlay/fusion occurs between mitochondria within the m/lEVs and the recipient cell mitochondria or if the signals indicate mitochondria released from the m/lEVs that then subsequently undergo fusion with the recipient cell mitochondria.
Mitochondria-containing m/lEVs appear to enter recipient low pinocytic cells such as BECs primarily via membrane fusion as endocytic inhibition of m/lEV uptake did not dramatically decrease uptake into the recipient BECs. 84 Interestingly, the route of m/lEV uptake into phagocytic cell models such as macrophages seemed to be sensitive to endocytic inhibition of m/lEV uptake suggesting that the cellular uptake pathway of m/lEVs depends on the pinocytic capabilities of the recipient cells. 84 These findings suggest that the subcellular trafficking of m/lEVs and the mechanism of mitochondria release within recipient cells may too differ based on the experimental models. m/lEVs internalized via membrane fusion into recipient BECs may release their mitochondrial load via fusion-driven lipid exchange that can release the mitochondria into cytosolic spaces that may subsequently diffuse towards the recipient cell mitochondrial network—this hypothesis however remains to be evaluated. Dual labeling of m/lEV mitochondria and recipient BEC’s endogenous mitochondria using fluorescence resonance energy transfer-amenable probes may allow testing of the mitochondrial fusion hypothesis. 85
Delivery challenges: Biodistribution and pharmacokinetics of m/lEV mitochondria for brain delivery
The two most significant challenges for the delivery of mitochondria-containing EVs to the brain or to the BBB are related to EV pharmacokinetics and biodistribution upon systemic injection. To date, there are no published reports that have systematically measured the whole body biodistribution and pharmacokinetics (PK) of m/lEVs containing mitochondria. In addition to whole body PK, it is important to measure the brain PK parameters of m/lEVs considering their applications in delivery to the BBB and the brain tissue. Robust experimental tools thankfully exist to measure the whole body PK parameters such as circulation t1/2, clearance, blood-to-brain unidirectional influx constant, and brain-to-blood efflux rate using radiolabeled EVs. 86 These studies are currently underway in our laboratory to support the further development of m/lEVs as a delivery carrier for mitochondria in preclinical models of stroke.
Similar to most nanoparticles, EVs too tend to accumulate in large quantities in the “filtering” organs of the reticuloendothelial system such as the liver and spleen. Uptake into the liver and spleen will dramatically lower the amounts of EVs that will stay in circulation for subsequent uptake into the target tissues, for example, the BBB or the brain. Therefore, the biodistribution and pharmacokinetic processes are key aspects that will have to be studied and optimized for the delivery of mitochondria-containing EVs to the brain. EVs have been engineered to express targeting ligands on EV membranes to facilitate the uptake of EVs into the brain tissue. The seminal study by Alvarez-Erviti et al. was one of the earliest works that reported the modification of exosome membranes to express a fusion construct: Lamp2b, an exosomal membrane protein was fused to the neuron-specific RVG peptide to target delivery of siRNA-loaded exosomes to neurons. 87 Mice i.v.-injected with RVG-exosomes demonstrated selective GAPDH mRNA knockdown in the cortex, striatum and midbrain regions while peripheral tissues such as the liver, spleen, muscle and heart showed none-to-little signs of gene knockdown. We had previously reviewed a handful of small EV/exosome modification strategies for delivery to the brain. 39 It remains to be investigated if engineering EV membranes of the larger, mitochondria-containing EVs, will also yield similar outcomes in improving targeted delivery to the brain.
Potential safety concerns associated with m/lEV delivery
Small EVs derived from HEK293FT cells did not show signs of toxicity and immunogenicity upon repeated dosing via intravenous and intraperitoneal routes in C57BL/6 mice. 88 However, it is not known if large EVs with mitochondria may show a similar safety profile in injected animals. In our studies, C57BL/6 mice i.v. injected with mitochondria containing-BEC-derived EVs were well-tolerated for up to 24 hours post-injection suggesting the acute safety of large EVs in injected mice. 15 Future studies will have to determine the potential safety of large EVs by assessing changes in hematology, blood chemistry, and immune markers after repeated administration. It is not known if the transfer of mitochondria via EVs will result induce tumorigenicity and/or mitochondrial DNA-induced heteroplasmy. These are indeed very critical questions that will have to be addressed in future studies. For example, mtDNA sequencing in the recipient cells and tissues should be performed to determine if the transfer of EV mitochondria induces mtDNA heteroplasmy.
Conclusions
We have described the scientific premise of developing m/lEVs containing as a natural, cell-derived carrier for the delivery of mitochondria based on our published studies on BEC-derived m/lEVs. There are several important gaps that will have to be addressed such as understanding the mechanistic basis of mitochondria entrapment into secreted m/lEVs from healthy donor cells, subcellular trafficking and release of mitochondria from the m/lEVs prior to the fusion of m/lEV mitochondria with the recipient cell’s mitochondrial network and determination of whole body PK, biodistribution as well as brain PK parameters of injected m/lEVs in experimental/preclinical models. Addressing the above challenges will propel the preclinical development of m/lEV mitochondria as a novel drug to treat a wide range of diseases associated with mitochondrial dysfunction.
Abbreviations
- ATP
adenosine triphosphate
- BBB
blood-brain barrier
- BECs
brain endothelial cells
- EVs
extracellular vesicles
- EXOs
exosomes
- MCAo
middle cerebral artery occlusion
- MDV
mitochondria-derived vesicles
- m/lEVs
medium-to-large EVs
- mtDNA
mitochondrial DNA
- MVs
microvesicles
- OGD
oxygen-glucose deprivation
- OXPHOS
oxidative phosphorylation
- sEVs
small extracellular vesicles
Acknowledgements
The authors gratefully acknowledge the excellent contribution of all past members of the Manickam laboratory, especially Drs. Anisha D’Souza and Kandarp M. Dave and many undergraduate trainees whose tireless efforts advanced these studies in the Manickam laboratory. Devika S Manickam is deeply grateful to a cross-institutional team of wonderful collaborators/mentors, Professors Louise D. McCullough, Venugopal R. Venna, Sruti S. Shiva, Donna B. Stolz, Carol Milligan, Osvaldo Delbono and Rehana K. Leak who have made important contributions to the development of m/lEVs as a novel delivery carrier for mitochondria. Devika S Manickam would like to consider this work as a small tribute to her long time mentor-collaborator, Dr. Prof. Louise D. McCullough, whose kind and powerful mentoring has made an immense and long lasting impact on Manickam’s career in academia.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Manickam group’s works referred to in this review have been supported by the Charles Henry Leach II Fund for Duquesne University, Faculty Development Fund, and Manickam laboratory start-up funds from Duquesne University and the Department of Defense ALS Therapeutic Idea Award (HT9425-23-1-0218).
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Devika S Manickam is a named inventor on a non-provisional US patent application related to mitochondria-enriched extracellular vesicles.
ORCID iD
Devika S Manickam https://orcid.org/0000-0002-6363-3679
References
- 1.Currais A. Ageing and inflammation – a central role for mitochondria in brain health and disease. Age Res Rev 2015; 21: 30–42. [DOI] [PubMed] [Google Scholar]
- 2.López-Lluch G, Irusta PM, Navas P, de Cabo R. Mitochondrial biogenesis and healthy aging. Exp Gerontol 2008; 43: 813–819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Komen JC, Thorburn DR. Turn up the power – pharmacological activation of mitochondrial biogenesis in mouse models. Br J Pharmacol 2014; 171: 1818–1836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Whitaker RM, Corum D, Beeson CC, et al. Mitochondrial biogenesis as a pharmacological target: a new approach to acute and chronic diseases. Annu Rev Pharmacol Toxicol 2016; 56: 229–249. [DOI] [PubMed] [Google Scholar]
- 5.Moreira PI, Carvalho C, Zhu X, et al. Mitochondrial dysfunction is a trigger of Alzheimer's disease pathophysiology. Biochim Biophys Acta 2010; 1802: 2–10. [DOI] [PubMed] [Google Scholar]
- 6.Andreux PA, Houtkooper RH, Auwerx J. Pharmacological approaches to restore mitochondrial function. Nature Rev Drug Discov 2013; 12: 465–483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Duchen MR. Mitochondria in health and disease: perspectives on a new mitochondrial biology. Mol Aspects Med 2004; 25: 365–451. [DOI] [PubMed] [Google Scholar]
- 8.Fiskum G, Murphy AN, Beal MF. Mitochondria in neurodegeneration: acute ischemia and chronic neurodegenerative diseases. J Cereb Blood Flow Metab 1999; 19: 351–369. [DOI] [PubMed] [Google Scholar]
- 9.Wallace DC. A mitochondrial paradigm of metabolic and degenerative diseases, aging, and cancer: a dawn for evolutionary medicine. Annu Rev Genet 2005; 39: 359–407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Phinney DG, Di Giuseppe M, Njah J, et al. Mesenchymal stem cells use extracellular vesicles to outsource mitophagy and shuttle microRNAs. Nature Commun 2015; 6: 8472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ikeda G, Santoso MR, Tada Y, et al. Mitochondria-rich extracellular vesicles from autologous stem cell-derived cardiomyocytes restore energetics of ischemic myocardium. J Am Coll Cardiol 2021; 77: 1073–1088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Puhm F, Afonyushkin T, Resch U, et al. Mitochondria are a subset of extracellular vesicles released by activated monocytes and induce type I IFN and TNF responses in endothelial cells. Circ Res 2019; 125: 43–52. [DOI] [PubMed] [Google Scholar]
- 13.Dave KM, Dobbins DX, Farinelli MN, et al. Engineering extracellular vesicles to modulate their innate mitochondrial load. Cellular Mol Bioeng 2022; 15: 367–389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dave KM, Stolz DB, Venna VR, et al. Mitochondria-containing extracellular vesicles (EV) reduce mouse brain infarct sizes and EV/HSP27 protect ischemic brain endothelial cultures. J Control Rel 2023; 354: 368–393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dave KM, Venna VR, Rao KS, et al. Mitochondria-containing extracellular vesicles from mouse vs. human brain endothelial cells for ischemic stroke therapy. J Control Rel 2024; 373: 803–822. [DOI] [PubMed] [Google Scholar]
- 16.O’Brien Connor G, Ozen Mehmet O, et al. Mitochondria-rich extracellular vesicles rescue patient-specific cardiomyocytes from doxorubicin injury. JACC: CardioOncol 2021; 3: 428–440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Théry C, Witwer KW, Aikawa E, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicl 2018; 7: 1535750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.D’Acunzo P, Pérez-González R, Kim Y, et al. Mitovesicles are a novel population of extracellular vesicles of mitochondrial origin altered in Down syndrome. Sci Adv 2021; 7: eabe5085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.König T, Nolte H, Aaltonen MJ, et al. MIROs and DRP1 drive mitochondrial-derived vesicle biogenesis and promote quality control. Nat Cell Biol 2021; 23: 1271–186. [DOI] [PubMed] [Google Scholar]
- 20.Hazan R, Pines O, Saada A. Mitochondrial derived vesicles – Quo Vadis? FEBS J 2024; 291: 4660–4669. [DOI] [PubMed] [Google Scholar]
- 21.McLelland GL, Soubannier V, Chen CX, et al. Parkin and PINK1 function in a vesicular trafficking pathway regulating mitochondrial quality control. EMBO J 2014; 33: 282–295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Neuspiel M, Schauss AC, Braschi E, et al. Cargo-selected transport from the mitochondria to peroxisomes is mediated by vesicular carriers. Curr Biol 2008; 18: 102–108. [DOI] [PubMed] [Google Scholar]
- 23.Soubannier V, McLelland G-L, Zunino R, et al. A vesicular transport pathway shuttles cargo from mitochondria to lysosomes. Curr Biol 2012; 22: 135–141. [DOI] [PubMed] [Google Scholar]
- 24.König T, McBride HM. Mitochondrial-derived vesicles in metabolism, disease, and aging. Cell Metab 2024; 36: 21–35. [DOI] [PubMed] [Google Scholar]
- 25.Hayakawa K, Esposito E, Wang X, et al. Transfer of mitochondria from astrocytes to neurons after stroke. Nature 2016; 535: 551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Islam MN, Das SR, Emin MT, et al. Mitochondrial transfer from bone-marrow–derived stromal cells to pulmonary alveoli protects against acute lung injury. Nat Med 2012; 18: 759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.D'Souza A, Burch A, Dave KM, et al. Microvesicles transfer mitochondria and increase mitochondrial function in brain endothelial cells. J Control Rel 2021; 338: 505–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Liu D, Liao P, Li H, Tong S, et al. Regulation of blood-brain barrier integrity by Dmp1-expressing astrocytes through mitochondrial transfer. Sci Adv 2024; 10: eadk2913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Velmurugan GV, Vekaria HJ, Patel SP, et al. Astrocytic mitochondrial transfer to brain endothelial cells and pericytes in vivo increases with aging. J Cereb Blood Flow Metab 2024:0271678X241306054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sweeney MD, Zhao Z, Montagne A, et al. Blood-brain barrier: from physiology to disease and back. Physiol Rev 2019; 99: 21–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.del Zoppo GJ. Inflammation and the neurovascular unit in the setting of focal cerebral ischemia. Neuroscience 2009; 158: 972–982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shi Y, Jiang X, Zhang L, et al. Endothelium-targeted overexpression of heat shock protein 27 ameliorates blood–brain barrier disruption after ischemic brain injury. Proc Natl Acad Sci 2017; 114: E1243–E1252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shi Y, Zhang L, Pu H, et al. Rapid endothelial cytoskeletal reorganization enables early blood–brain barrier disruption and long-term ischaemic reperfusion brain injury. Nat Commun 2016; 7: 10523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hayakawa K, Chan SJ, Mandeville ET, et al. Protective effects of endothelial progenitor cell-derived extracellular mitochondria in brain endothelium. Stem Cells 2018; 36: 1404–1410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Doll DN, Hu H, Sun J, et al. Mitochondrial crisis in cerebrovascular endothelial cells opens the blood-brain barrier. Stroke 2015; 46: 1681–1689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Razmara A, Sunday L, Stirone C, et al. Mitochondrial effects of estrogen are mediated by estrogen receptor alpha in brain endothelial cells. J Pharmacol Exp Ther 2008; 325: 782–790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wen Y, Li W, Poteet EC, et al. Alternative mitochondrial electron transfer as a novel strategy for neuroprotection. J Biol Chem 2011; 286: 16504–16515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.D'Souza A, Dave KM, Stetler RA, et al. Targeting the blood-brain barrier for the delivery of stroke therapies. Adv Drug Deliv Rev 2021; 171: 332–351. [DOI] [PubMed] [Google Scholar]
- 39.Dave KM, Stolz DB, Manickam DS. Delivery of mitochondria-containing extracellular vesicles to the BBB for ischemic stroke therapy. Expert Opin Drug Deliv 2023; 20: 1769–1788. [DOI] [PubMed] [Google Scholar]
- 40.Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol 2013; 200: 373–383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Muralidharan-Chari V, Clancy J, Plou C, et al. ARF6-regulated shedding of tumor cell-derived plasma membrane microvesicles. Curr Biol 2009; 19: 1875–1885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Muralidharan-Chari V, Clancy JW, Sedgwick A, et al. Microvesicles: mediators of extracellular communication during cancer progression. J Cell Sci 2010; 123: 1603–1611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nabhan JF, Hu R, Oh RS, et al. Formation and release of arrestin domain-containing protein 1-mediated microvesicles (ARMMs) at plasma membrane by recruitment of TSG101 protein. Proc Natl Acad Sci 2012; 109: 4146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Oldendorf WH, Cornford ME, Brown WJ. The large apparent work capability of the blood-brain barrier: a study of the mitochondrial content of capillary endothelial cells in brain and other tissues of the rat. Ann Neurol 1977; 1: 409–417. [DOI] [PubMed] [Google Scholar]
- 45.Manickam DS. Delivery of mitochondria via extracellular vesicles – a new horizon in drug delivery. J Control Rel 2022; 343: 400–7. [DOI] [PubMed] [Google Scholar]
- 46.Giacomello M, Pyakurel A, Glytsou C, et al. The cell biology of mitochondrial membrane dynamics. Nat Rev Mol Cell Biol 2020; 21: 204–224. [DOI] [PubMed] [Google Scholar]
- 47.Abrisch RG, Gumbin SC, Wisniewski BT, et al. Fission and fusion machineries converge at ER contact sites to regulate mitochondrial morphology. J Cell Biol 2020; 219: e201911122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kalia R, Wang RY-R, Yusuf A, et al. Structural basis of mitochondrial receptor binding and constriction by DRP1. Nature 2018; 558: 401–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Smirnova E, Griparic L, Shurland DL, et al. Dynamin-related protein Drp1 is required for mitochondrial division in mammalian cells. Mol Biol Cell 2001; 12: 2245–2256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Dave KM, Zhao W, Hoover C, et al. Extracellular vesicles derived from a human brain endothelial cell line increase cellular ATP levels. AAPS PharmSciTech 2021; 22: 18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tong M, Zablocki D, Sadoshima J. The role of Drp1 in mitophagy and cell death in the heart. J Mol Cell Cardiol 2020; 142: 138–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Duan C, Liu R, Kuang L, et al. Activated Drp1 initiates the formation of endoplasmic reticulum-mitochondrial contacts via Shrm4-mediated actin bundling. Adv Sci 2023; 10: 2304885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Rabas N, Palmer S, Mitchell L, et al. PINK1 drives production of mtDNA-containing extracellular vesicles to promote invasiveness. J Cell Biol 2021; 220: e202006049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Mathieu M, Névo N, Jouve M, et al. Specificities of exosome versus small ectosome secretion revealed by live intracellular tracking of CD63 and CD9. Nat Commun 2021; 12: 4389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Demongeot J, Glade N, Hansen O, et al. An open issue: the inner mitochondrial membrane (IMM) as a free boundary problem. Biochimie 2007; 89: 1049–1057. [DOI] [PubMed] [Google Scholar]
- 56.CellTiter-Glo® 2.0 Assay, www.promega.com/products/cell-health-assays/cell-viability-and-cytotoxicity-assays/celltiter_glo-2_0-assay/?tabset0=0 (3 December 2024).
- 57.Voloboueva LA, Duan M, Ouyang Y, et al. Overexpression of mitochondrial Hsp70/Hsp75 protects astrocytes against ischemic injury in vitro. J Cereb Blood Flow Metab 2008; 28: 1009–1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kim JY, Kim JW, Yenari MA. Heat shock protein signaling in brain ischemia and injury. Neurosci Lett 2020; 715: 134642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Chazotte B. Labeling mitochondria with MitoTracker dyes. Cold Spring Harb Protoc 2011; 2011: 990–992. [DOI] [PubMed] [Google Scholar]
- 60.Hough KP, Trevor JL, Strenkowski JG, et al. Exosomal transfer of mitochondria from airway myeloid-derived regulatory cells to T cells. Redox Biol 2018; 18: 54–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Todkar K, Chikhi L, Desjardins V, et al. Selective packaging of mitochondrial proteins into extracellular vesicles prevents the release of mitochondrial DAMPs. Nat Commun 2021; 12: 1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jang SC, Crescitelli R, Cvjetkovic A, et al. Mitochondrial protein enriched extracellular vesicles discovered in human melanoma tissues can be detected in patient plasma. J Extracell Vesicles 2019; 8: 1635420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Pallet N, Sirois I, Bell C, et al. A comprehensive characterization of membrane vesicles released by autophagic human endothelial cells. PROTEOMICS 2013; 13: 1108–1120. [DOI] [PubMed] [Google Scholar]
- 64.Puigserver P, Spiegelman BM. Peroxisome proliferator-activated receptor-gamma coactivator 1 alpha (PGC-1 alpha): transcriptional coactivator and metabolic regulator. Endocr Rev 2003; 24: 78–90. [DOI] [PubMed] [Google Scholar]
- 65.Burns J, Yokota T, Ashihara H, et al. Plant foods and herbal sources of resveratrol. J Agri Food Chem 2002; 50: 3337–3340. [DOI] [PubMed] [Google Scholar]
- 66.Manickam DS. InventorMitochondria-enriched extracellular vesicles, US Patent application 18227026. 2023.
- 67.McCully JD, Levitsky S, Del Nido PJ, et al. Mitochondrial transplantation for therapeutic use. Clin Transl Med 2016; 5: 16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Norat P, Sokolowski JD, Gorick CM, et al. Intraarterial transplantation of mitochondria after ischemic stroke reduces cerebral infarction. Stroke: Vasc Interven Neurol 2023; 3: e000644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Cloer CM, Givens CS, Buie LK, et al. Mitochondrial transplant after ischemia reperfusion promotes cellular salvage and improves lung function during ex-vivo lung perfusion. J Heart Lung Transplant 2023; 42: 575–584. [DOI] [PubMed] [Google Scholar]
- 70.Alway SE, Paez HG, Pitzer CR, et al. Mitochondria transplant therapy improves regeneration and restoration of injured skeletal muscle. J Cachexia Sarcopenia Muscle 2023; 14: 493–507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zhu Z, Li X, Wang X, et al. Photobiomodulation augments the effects of mitochondrial transplantation in the treatment of spinal cord injury in rats by facilitating mitochondrial transfer to neurons via Connexin 36. Bioeng Transl Med 2023; 8: e10473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.McCully JD, Cowan DB, Pacak CA, et al. Injection of isolated mitochondria during early reperfusion for cardioprotection. Am J Physiol Heart Circ Physiol. 2009; 296: H94–H105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Guariento A, Piekarski BL, Doulamis IP, et al. Autologous mitochondrial transplantation for cardiogenic shock in pediatric patients following ischemia-reperfusion injury. J Thorac Cardiovasc Surg 2021; 162: 992–1001. [DOI] [PubMed] [Google Scholar]
- 74.Emani SM, Piekarski BL, Harrild D, et al. Autologous mitochondrial transplantation for dysfunction after ischemia-reperfusion injury. J Thorac Cardiovasc Surg 2017; 154: 286–289. [DOI] [PubMed] [Google Scholar]
- 75.Masuzawa A, Black KM, Pacak CA, et al. Transplantation of autologously derived mitochondria protects the heart from ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol 2013; 304: H966–H982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Brestoff JR, Singh KK, Aquilano K, et al. Recommendations for mitochondria transfer and transplantation nomenclature and characterization. Nat Metab 2025; 7: 53–67. [DOI] [PubMed] [Google Scholar]
- 77.Jiang XR, Braesch-Andersen K, Baucom CC, et al. Extracellular vesicles from mesenchymal stromal cells (imEVs) improve cold preservation of isolated mitochondria J Biosci Med 2024; 12: 52–63. [Google Scholar]
- 78.Brennan K, Martin K, FitzGerald SP, et al. A comparison of methods for the isolation and separation of extracellular vesicles from protein and lipid particles in human serum. Scient Rep 2020; 10: 1039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zhao X, Lei Y, Zheng J, et al. Identification of markers for migrasome detection. Cell Discov 2019; 5. DOI: 10.1038/s41421-019-0093-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Zhang Q, Jeppesen DK, Higginbotham JN, et al. Comprehensive isolation of extracellular vesicles and nanoparticles. Nat Protoc 2023; 18: 1462–1487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Jeppesen DK, Zhang Q, Franklin JL, et al. Extracellular vesicles and nanoparticles: emerging complexities. Trends Cell Biol 2023; 33: 667–681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Liang W, Sagar S, Ravindran R, et al. Mitochondria are secreted in extracellular vesicles when lysosomal function is impaired. Nat Commun 2023; 14: 5031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Flis VV, Daum G. Lipid transport between the endoplasmic reticulum and mitochondria. Cold Spring Harb Perspect Biol 2013; 5: a013235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Jhaveri JR, Khare P, Paul Pinky P, et al. Low pinocytic brain endothelial cells primarily utilize membrane fusion to internalize extracellular vesicles. Eur J Pharm Biopharm 2024; 204: 114500. [DOI] [PubMed] [Google Scholar]
- 85.Feng R, Guo L, Fang J, et al. Construction of the FRET pairs for the visualization of mitochondria membrane potential in dual emission colors. Anal Chem 2019; 91: 3704–3709. [DOI] [PubMed] [Google Scholar]
- 86.Banks WA, Sharma P, Bullock KM, et al. Transport of extracellular vesicles across the blood-brain barrier: brain pharmacokinetics and effects of inflammation. Int J Mol Sci 2020; 21: 4407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Alvarez-Erviti L, Seow Y, Yin H, Betts C, et al. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotech 2011; 29: 341–345. [DOI] [PubMed] [Google Scholar]
- 88.Zhu X, Badawi M, Pomeroy S, et al. Comprehensive toxicity and immunogenicity studies reveal minimal effects in mice following sustained dosing of extracellular vesicles derived from HEK293T cells. J Extracell Vesicle 2017; 6: 1324730. [DOI] [PMC free article] [PubMed] [Google Scholar]