Abstract
Obesity is a prevalent disease that also contributes to the incidence and severity of many other chronic diseases and health conditions. Treatment approaches include lifestyle intervention, bariatric surgery, and pharmacological approaches, with glucagon-like peptide 1 (GLP-1) receptor agonists approved specifically for weight loss having changed the treatment landscape significantly in the last 5 years. Targeting the glucose-dependent insulinotropic polypeptide (GIP) receptor may enhance the metabolic benefits of GLP-1 receptor agonism. These beneficial effects are seen with both GIP receptor antagonism and GIP receptor agonism, although the mechanisms underlying this apparent paradox remain unknown. Here, we summarize the physiologic, genetic, and clinical evidence for pursuing GIP receptor antagonism to achieve metabolic and weight benefits. Both global and central nervous system knockout of GIP receptors protects mice fed a high-fat diet from obesity and insulin resistance. Genome-wide association studies in humans support this notion, correlating lower BMI with GIP receptor genetic variants with reduced function. Pharmacologic approaches in mice and monkeys confirm that GIP receptor antagonism enhances GLP-1–induced weight reduction and other metabolic benefits, and a phase 1 study provides proof of principle that beneficial effects extend to humans. GIP receptor antagonism may represent an important new mechanism to expand the treatment options available to individuals living with obesity.
UNMET Needs of People Living with Obesity
Glucagon-like peptide 1 (GLP-1) receptor agonists (GLP-1RAs) have transformed the care of people living with obesity and may alleviate the public health burden of obesity (1). Nevertheless, while efficacy has steadily improved from early GLP-1RAs such as exenatide and liraglutide, additional therapeutic options would likely help more individuals reach their weight goals. Similarly, the administration frequency for GLP-1RAs has decreased from twice daily (exenatide) to once daily (liraglutide) to once weekly (semaglutide), but longer-acting agents are desirable. Finally, people rapidly regain weight after discontinuation of current antiobesity medications (1,2), and some individuals experience persistent gastrointestinal adverse events. Therefore, exploration of additional mechanisms of action that complement GLP-1RAs is relevant.
One approach to complement GLP-1 receptor agonism is glucose-dependent insulinotropic polypeptide (GIP) receptor antagonism. Here, we briefly review the physiologic role of GIP in fat deposition, the human genetic rationale for GIP receptor antagonism, and empirical data demonstrating that reductions in GIP receptor signaling promote leanness and improve metabolism. Recent human experimental data with maridebart cafraglutide (MariTide), a peptide-antibody conjugate combining a GIP receptor antagonist antibody with two GLP-1 analog agonist peptides, provide clinical support for GIP antagonism as a complementary approach to GLP-1 receptor agonism (3). This concept is further supported by recent data from nonhuman primates (NHPs) that showed that using the peptide-based GIP receptor antagonist AT-7687 in combination with GLP-1 receptor agonism resulted in additive reduction in weight relative to either agent alone (4).
Physiologic Role of GIP
The physiologic role of GIP has been investigated over many decades, and a combination of human and mouse data, reinforced by the resistance of GIP receptor knockout (KO) mice to diet-induced obesity (DIO), resulted in GIP being regarded as the so-called obesity hormone (5,6). GIP is secreted from intestinal K-cells, with high-lipid meals being the most effective stimulants (7). Infusion of GIP increases intestinal blood flow and increases nutrient absorption (8). Moreover, GIP enhances adipose tissue blood flow and lipid deposition in lean people (9). In individuals with obesity and type 2 diabetes, GIP increases adipose tissue triglycerides (10). These obesogenic effects of GIP are negated by the GIP-derived GIP receptor antagonist GIP(3–30)NH2, the first GIP receptor antagonist administered to humans (9). These patient data collectively paint a picture of GIP as an obesogenic factor that enhances lipid uptake and storage.
Human Genetics
Importantly, three missense GIP receptor variants (Arg190Gln [rs139215588], Glu288Gly [rs143430880], and Glu354Gln [rs1800437]) with impaired GIP receptor function have been reported to associate with decreased BMI (11,12). G protein-coupled receptors (GPCRs), like the GIP receptor, induce cAMP and stimulate other effectors primarily through a family of G-proteins that associate with the receptor (13). In addition, GPCRs often undergo ligand-mediated arrestin recruitment, internalization, and transient desensitization (13). A recent study of 47 missense variants of the human GIP receptor supported an essential role of GIP receptor desensitization for overall GIP receptor function, as carriers of variants with impaired Gs G-protein activation and arrestin recruitment had lower levels of adiposity-related traits, while those solely impaired in Gs-mediated signaling did not show these changes (12). Including more loss-of-function variants, the same association with lower BMI was confirmed, and meta-analysis regression using single-variant estimates revealed a significant effect of solely impaired β-arrestin recruitment (below 50% of wild-type levels) on BMI and hip circumference but no impact of impaired cAMP production (12). In line with the essential role of arrestins in internalization, the variants with impaired arrestin recruitment also displayed decreased endosomal signaling (12). In summary, the preponderance of human genetic data indicates that inactivating GIP receptor mutations are associated with lower BMI (11,12).
Preclinical Data
Murine evidence supporting GIP receptor antagonism emerged from studies using mice with a global, germline GIP receptor KO (6). These animals have body weights similar to those of wild-type controls on a standard chow diet, but they are protected from DIO when placed on a high-fat diet (HFD). This resistance to DIO was associated with a short-lived shift toward lipid catabolism with no change in food intake (6), consistent with the obesogenic role of GIP during overnutrition (5). The changes in lipid catabolism and deposition in global GIP receptor KO mice implicate an adipocyte-centric mechanism. However, inducible, adipocyte-specific GIP receptor deletion only partially recapitulated the global KO phenotype (14), while transgenic expression of human GIP receptor in adipocytes exacerbated HFD-induced weight gain but not adiposity (15). Mice with β-cell–specific GIP receptor deletion do not display overall protection from DIO (16,17), although the evidence is mixed on whether adiposity is reduced (16) or not (17). However, normalizing deficient prandial insulin in these animals reduced adiposity to control levels (16).
Deletion of broad central nervous system (CNS) (18) and GABAergic, neuron-specific (19) GIP receptor populations in mice produced a DIO protective phenotype along with reduced fat mass and improved glucose homeostasis. Unlike the global GIP receptor KO, these two mouse lines exhibit decreased food intake, which has not been fully explored. Collectively, these rodent genetics models have yet to provide clarity on the mechanisms of action by which adipose tissue, β-cell, and CNS GIP receptor populations regulate lipid accumulation, food intake, and, ultimately, body weight. Regardless, the data do provide a rationale for GIP receptor antagonism as a likely potentiator for current obesity pharmacotherapies by reducing overall body weight with a specific reduction in adipose mass.
Pharmacologic exploration of GIP receptor antagonism for treatment of obesity and diabetes began >20 years ago (20). It is important to emphasize differences between rodents, particularly mice, and humans regarding the GIP receptor system, which necessitates the development of mouse-specific GIP receptor antagonists for in vivo use in mice (21). A recently presented mouse anti-murine GIP receptor–antagonizing antibody nearly abolished HFD-induced weight gain in mice (17). This activity was associated with decreased fat mass and fasting insulin, phenocopying the global GIP receptor KO mice. Minor reductions in lean mass were observed compared with controls. A similar effect of resistance to weight gain has been observed with other GIP receptor antagonists (22,23). Of particular note, a novel GIP analog optimized for chronic use in murine studies, mGIPAnt-1, also demonstrated a reduction in gain of fat mass in ovariectomized mice (23). This finding was corroborated recently with the GIP-derived GIP receptor antagonist AT-7687 that, when given as monotherapy, prevented weight gain in HFD-fed NHPs (4). GIP receptor antagonists and agonists have consistently demonstrated an ability to prevent body weight gain in both rodents and NHPs, but neither is as effective as GLP-1RAs in reducing body weight in obese animals. Despite this, compelling preclinical data demonstrate that GIP receptor antagonists (or agonists) induce substantial additional weight loss when combined with a GLP-1RA. The mouse anti-murine GIP receptor–antagonizing antibody mentioned above prevented weight gain in DIO mice when used alone (14,17), while combining it with liraglutide nearly doubled the weight loss seen with liraglutide alone (approximately 30% body weight loss compared with vehicle). This was associated with additional fat mass reduction and improved oral glucose control but not food intake reduction, hinting at a novel mechanism compared with GIP receptor agonists (14,17–19,24). It was further demonstrated that the effect of GIP receptor antagonists to induce additional weight loss beyond the maximal effect of GLP-1RAs translates to NHPs (4,17). Others have reported similar outcomes using a GIP receptor–antagonizing peptide (25) and a different antibody antagonist (26). A single report on mice showed no additional weight loss when combining GIP receptor antagonism with GLP-1 receptor agonism; however, the interpretations of these data are confounded by the short half-life of the GIP receptor antagonist (24). In total, these data suggest that the metabolic benefits of GIP receptor–targeting agents rely at least partially on interactions with GLP-1 receptor in cell types expressing both receptors. Further research will be required to understand if there is a subset of cellular targets that express only one of the two receptors relevant to the weight reduction phenotype compared with target tissues where the interactions of coexpressed GIP and GLP-1 receptors are required for metabolic benefits.
While the metabolic benefits of GIP receptor antagonism and agonism clearly intersect, especially when used in combination with GLP-1 receptor agonism, it is possible that other pharmacodynamic effects and safety profiles do not exhibit the same overlap.
Clinical Data on Concurrent GIP Receptor Antagonism and GLP-1 Receptor Agonism
In a phase 1, randomized, double-blind, placebo-controlled study (NCT04478708, clinicaltrials.gov) in participants with obesity, the combined GIP receptor antagonist-GLP-1RA (MariTide) displayed an acceptable safety and tolerability profile with no notable differences between treatment groups for clinical safety laboratory parameters (3). The most common adverse events were gastrointestinal, including nausea and vomiting, and were generally mild and resolved within 48 h. Dose-dependent weight reduction of up to –14.5% was observed with the highest dose studied, and weight reductions appeared to be maintained for up to 150 days after the last dose. MariTide reached maximum concentration 4–7 days postdose, and mean half-life was 14–16 days for intact MariTide and 21–24 days for total MariTide (3). MariTide reduced fasting glucose relative to placebo. The full potential of MariTide is being investigated in an ongoing clinical development program.
How could GIP Receptor Agonism and Antagonism Both be Clinically Meaningful?
We have thus far laid out the rationale for GIP receptor antagonism as a pharmacologic strategy to treat obesity. GIP and GIP receptor agonism had long been viewed as a therapeutic dead end due to the observation that the insulinotropic effect of GIP in most patients with type 2 diabetes is much reduced compared with that observed in healthy individuals (27). However, GIP resistance can be partially overcome by agents that lower circulating glucose levels, like GLP-1, suggesting a complementary activity (27). The clinical efficacy of dual receptor agonists of GLP-1 and GIP receptors has now been clearly demonstrated (2). With both GIP receptor agonism and antagonism emerging as effective pharmacological approaches to couple with GLP-1 receptor agonism, there is now intense focus on elucidating the mechanism(s) underlying this apparent paradox.
One hypothesis is that GIP receptor agonism does not play a significant role in weight loss mediated by the approved dual agonist tirzepatide, which may elicit superior efficacy through the GLP-1 receptor due to its biased agonism profile (13). Biased agonism at the GLP-1 receptor by tirzepatide, characterized by disproportionately reduced β-arrestin recruitment and internalization relative to G protein coupling, has been demonstrated to confer superior body weight reduction in preclinical models (28).
It has also been suggested that chronic GIP receptor activation results in functional antagonism (14). Chronic GIP receptor stimulation of primary adipocytes with an acylated GIP receptor agonist induces substantial desensitization to subsequent ligand-mediated stimulation, resulting in functional antagonism (14). Chronic GIP receptor agonism and antagonists of GIP receptor both reduce body weight or prevent weight gain and also inhibit adipose tissue fatty acid uptake in response to a GIP analog ([D-Ala2]-GIP), but only agonists enhance insulin secretion (14).
A third hypothesis is that GIP receptor agonists and antagonists act on different populations of receptors in the CNS to elicit their effects. Acylated peptide GIP receptor agonists appear to act on GABAergic neurons in the CNS to additively reduce food intake and body weight when combined with GLP-1RAs in mice (18,19). This, along with reports that liraglutide acts on glutamatergic CNS neurons to reduce body weight (29), suggests GIP receptor agonism serves to disinhibit GLP-1 receptor signaling on food intake regulation centers of the rodent brain; it remains to be seen whether similar CNS pathways are activated in humans.
A fourth mechanism suggests that dual targeting of GLP-1 and GIP receptors in cells expressing both receptors can enhance cAMP response and insulin secretion, potentially through receptor heteromerization (30). However, in humans, GIP infusions during GLP-1 exposure did not increase insulin secretion (31).
Although evidence exists for all the above hypotheses, further research will be needed before true consensus can be gained.
Conclusions
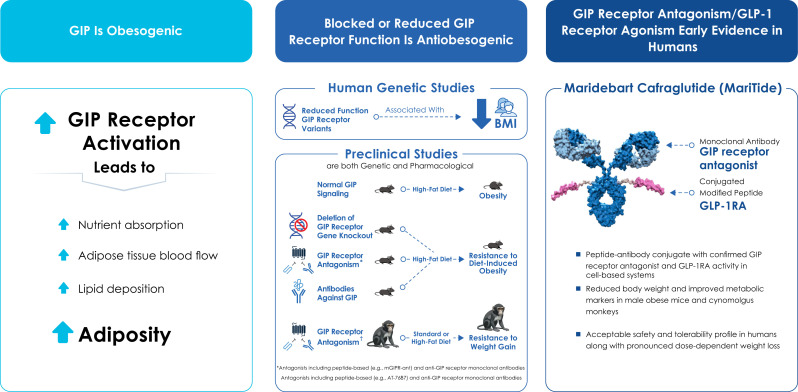
Together, the obesogenic role of GIP, human genetics, preclinical data, and emerging clinical trial data all point to the potential of GIP receptor antagonism to complement GLP-1 receptor agonism in helping people living with obesity achieve healthy weight goals (Fig. 1).
Figure 1.
Rationale for GIP receptor antagonism in the treatment of obesity.
Article Information
Acknowledgments. Editorial support was provided by Eugene Gillespie, PhD (Amgen).
Duality of Interest. M.M.R. and J.J.H. are cofounders and minority shareholders in Antag Therapeutics. J.J.H. is a board member of Antag Therapeutics and has served as a consultant or advisor to Novo Nordisk, Roche, Novartis Pharmaceuticals, and Merck Sharp & Dohme and has received fees for lectures from Merck Sharp & Dohme and Novo Nordisk. M.M.R. is a consultant for Antag Therapeutics. J.T.G. and M.M.V. are employees and stockholders of Amgen. Graphical support was provided by Vault Bioventures and funded by Amgen.
Author Contributions. M.M.R., J.T.G., M.M.V., and J.J.H. researched data, contributed to discussion, wrote the manuscript, and reviewed and edited the manuscript.
Footnotes
References
- 1. Rubino D, Abrahamsson N, Davies M, et al.; STEP 4 Investigators . Effect of continued weekly subcutaneous semaglutide vs placebo on weight loss maintenance in adults with overweight or obesity: the STEP 4 randomized clinical trial. JAMA 2021;325:1414–1425 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Aronne LJ, Sattar N, Horn DB, et al.; SURMOUNT-4 Investigators . Continued treatment with tirzepatide for maintenance of weight reduction in adults with obesity: the SURMOUNT-4 randomized clinical trial. JAMA 2024;331:38–48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Véniant MM, Lu S-C, Atangan L, et al. A GIPR antagonist conjugated to GLP-1 analogues promotes weight loss with improved metabolic parameters in preclinical and phase 1 settings. Nat Metab 2024;6:290–303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Jensen MH, Sanni SJ, Riber D, Holst JJ, Rosenkilde MM, Sparre-Ulrich AH. AT-7687, a novel GIPR peptide antagonist, combined with a GLP-1 agonist, leads to enhanced weight loss and metabolic improvements in cynomolgus monkeys. Mol Metab 2024;88:102006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Marks V. The early history of GIP 1969-2000: from enterogastrone to major metabolic hormone. Peptides 2020;125:170276. [DOI] [PubMed] [Google Scholar]
- 6. Miyawaki K, Yamada Y, Ban N, et al. Inhibition of gastric inhibitory polypeptide signaling prevents obesity. Nat Med 2002;8:738–742 [DOI] [PubMed] [Google Scholar]
- 7. Yamane S, Harada N, Hamasaki A, et al. Effects of glucose and meal ingestion on incretin secretion in Japanese subjects with normal glucose tolerance. J Diabetes Investig 2012;3:80–85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Singh SK, Bartoo AC, Krishnan S, Boylan MO, Schwartz JH, Michael Wolfe M. Glucose-dependent insulinotropic polypeptide (GIP) stimulates transepithelial glucose transport. Obesity (Silver Spring) 2008;16:2412–2416 [DOI] [PubMed] [Google Scholar]
- 9. Asmar M, Asmar A, Simonsen L, et al. The gluco- and liporegulatory and vasodilatory effects of glucose-dependent insulinotropic polypeptide (GIP) are abolished by an antagonist of the human GIP receptor. Diabetes 2017;66:2363–2371 [DOI] [PubMed] [Google Scholar]
- 10. Thondam SK, Daousi C, Wilding JPH, et al. Glucose-dependent insulinotropic polypeptide promotes lipid deposition in subcutaneous adipocytes in obese type 2 diabetes patients: a maladaptive response. Am J Physiol Endocrinol Metab 2017;312:E224–E233 [DOI] [PubMed] [Google Scholar]
- 11. Speliotes EK, Willer CJ, Berndt SI, et al.; Procardis Consortium . Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nat Genet 2010;42:937–948 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Kizilkaya HS, Sørensen KV, Madsen JS, et al. Characterization of genetic variants of GIPR reveals a contribution of β-arrestin to metabolic phenotypes. Nat Metab 2024;6:1268–1281 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Willard FS, Douros JD, Gabe MB, et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight 2020;5:e140532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Killion EA, Chen M, Falsey JR, et al. Chronic glucose-dependent insulinotropic polypeptide receptor (GIPR) agonism desensitizes adipocyte GIPR activity mimicking functional GIPR antagonism. Nat Commun 2020;11:4981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ugleholdt R, Pedersen J, Bassi MR, et al. Transgenic rescue of adipocyte glucose-dependent insulinotropic polypeptide receptor expression restores high fat diet-induced body weight gain. J Biol Chem 2011;286:44632–44645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Campbell JE, Ussher JR, Mulvihill EE, et al. TCF1 links GIPR signaling to the control of beta cell function and survival. Nat Med 2016;22:84–90 [DOI] [PubMed] [Google Scholar]
- 17. Killion EA, Wang J, Yie J, et al. Anti-obesity effects of GIPR antagonists alone and in combination with GLP-1R agonists in preclinical models. Sci Transl Med 2018;10:eaat3392. [DOI] [PubMed] [Google Scholar]
- 18. Zhang Q, Delessa CT, Augustin R, et al. The glucose-dependent insulinotropic polypeptide (GIP) regulates body weight and food intake via CNS-GIPR signaling. Cell Metab 2021;33:833–844.e5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Liskiewicz A, Khalil A, Liskiewicz D, et al. Glucose-dependent insulinotropic polypeptide regulates body weight and food intake via GABAergic neurons in mice. Nat Metab 2023;5:2075–2085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Gault VA, O’Harte FPM, Flatt PR. Glucose-dependent insulinotropic polypeptide (GIP): anti-diabetic and anti-obesity potential? Neuropeptides 2003;37:253–263 [DOI] [PubMed] [Google Scholar]
- 21. Gasbjerg LS, Rasmussen RS, Dragan A, et al. Altered desensitization and internalization patterns of rodent versus human glucose-dependent insulinotropic polypeptide (GIP) receptors. An important drug discovery challenge. Br J Pharmacol. 1 July 2024. [Epub ahead of print]. DOI: 10.1111/bph.16478 [DOI] [PubMed] [Google Scholar]
- 22. Nakamura T, Tanimoto H, Mizuno Y, et al. Gastric inhibitory polypeptide receptor antagonist, SKL-14959, suppressed body weight gain on diet-induced obesity mice. Obes Sci Pract 2018;4:194–203 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Boer GA, Hunt JE, Gabe MBN, et al. Glucose-dependent insulinotropic polypeptide receptor antagonist treatment causes a reduction in weight gain in ovariectomised high fat diet-fed mice. Br J Pharmacol 2022;179:4486–4499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Mroz PA, Finan B, Gelfanov V, et al. Optimized GIP analogs promote body weight lowering in mice through GIPR agonism not antagonism. Mol Metab 2019;20:51–62 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Yang B, Gelfanov VM, El K, et al. Discovery of a potent GIPR peptide antagonist that is effective in rodent and human systems. Mol Metab 2022;66:101638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Svendsen B, Capozzi ME, Nui J, et al. Pharmacological antagonism of the incretin system protects against diet-induced obesity. Mol Metab 2020;32:44–55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Højberg PV, Vilsbøll T, Rabøl R, et al. Four weeks of near-normalisation of blood glucose improves the insulin response to glucagon-like peptide-1 and glucose-dependent insulinotropic polypeptide in patients with type 2 diabetes. Diabetologia 2009;52:199–207 [DOI] [PubMed] [Google Scholar]
- 28. Jones B, Buenaventura T, Kanda N, et al. Targeting GLP-1 receptor trafficking to improve agonist efficacy. Nat Commun 2018;9:1602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Adams JM, Pei H, Sandoval DA, et al. Liraglutide modulates appetite and body weight through glucagon-like peptide 1 receptor-expressing glutamatergic neurons. Diabetes 2018;67:1538–1548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lu S-C, Chen M, Atangan L, et al. GIPR antagonist antibodies conjugated to GLP-1 peptide are bispecific molecules that decrease weight in obese mice and monkeys. Cell Rep Med 2021;2:100263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Bergmann NC, Gasbjerg LS, Heimbürger SM, et al. No acute effects of exogenous glucose-dependent insulinotropic polypeptide on energy intake, appetite, or energy expenditure when added to treatment with a long-acting glucagon-like peptide 1 receptor agonist in men with type 2 diabetes. Diabetes Care 2020;43:588–596 [DOI] [PubMed] [Google Scholar]