Abstract
Background
Adoptive transfer of chimeric antigen receptor (CAR)-expressing natural killer (NK) cells has demonstrated success against hematological malignancies. Efficacy against solid tumors has been limited by poor NK cell survival and function in the suppressive tumor microenvironment (TME). To enhance efficacy against solid tumors, stimulatory cytokines have been incorporated into CAR-NK cell therapeutic approaches. However, current cytokine strategies have limitations, including systemic toxicities, exogenous dependencies, and unwanted TME bystander effects. Here, we aimed to overcome these limitations by modifying CAR-NK cells to express a constitutively active interleukin (IL)-7 receptor, termed C7R, capable of providing intrinsic CAR-NK cell activation that does not rely on or produce exogenous signals nor activate bystander cells.
Methods
We examined persistence, antitumor function, and transcriptional profiles of CAR-NK cells coexpressing C7R in a novel tumor immune microenvironment (TiME) co-culture system and against hematologic and solid tumor xenografts in vivo.
Results
Peripheral blood NK cells expressing a CAR directed against the solid tumor antigen GD2 and modified with C7R demonstrated enhanced tumor killing and persistence in vitro compared with CAR-NK cells without cytokine support and similar functions to CAR-NK cells supplemented with recombinant IL-15. C7R.CAR-NK cells exhibited enhanced survival and proliferation within neuroblastoma TiME xenografts in vivo but produced poor long-term tumor control compared with CAR-NK cells supplemented with IL-15. Similar results were seen using C7R-expressing CD19.CAR-NK cells against CD19+leukemia xenografts. Gene expression analysis revealed that chronic signaling via C7R induced a transcriptional signature consistent with intratumor stressed NK cells with blunted effector function. We identified gene candidates associated with chronic cytokine-stressed NK cells that could be targeted to reduce CAR-NK cell stress within the solid TME.
Conclusion
C7R promoted CAR-NK cell survival in hostile TMEs independent of exogenous signals but resulted in poor antitumor function in vivo. Our data reveals the detrimental role of continuous IL-7 signaling in CAR-NK cells and provides insights into proper application of cytokine signals when attempting to enhance CAR-NK cell antitumor activity.
Keywords: Adoptive cell therapy - ACT, Chimeric antigen receptor - CAR, Cytokine, Tumor microenvironment - TME, Natural killer - NK
WHAT IS ALREADY KNOWN ON THIS TOPIC
Adoptively transferred natural killer (NK) cells for the treatment of cancer require cytokine support to promote survival and function. Current approaches rely on the addition of exogenous cytokines or genetically modifying NK cells to secrete cytokines, both of which can produce toxicity and unwanted bystander cell activation. Novel approaches to cytokine supplementation are needed to bypass current limitations while maintaining efficacy.
WHAT THIS STUDY ADDS
Our study explores the use of a novel interleukin (IL-7) cytokine receptor, termed C7R, that confers to chimeric antigen receptor (CAR)-NK cells intrinsic and persistent signal transducer and activator of transcription 5 (STAT5) activity, an essential signaling node for NK cell function and survival. C7R bypasses current limitations by removing exogenous cytokine dependency and providing a solely NK-intrinsic means of activation. However, while C7R promotes NK survival, its chronic signaling induces a transcriptional phenotype associated with stressed NK cells and weakens NK cell cytotoxicity compared with stimulation with IL-15.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE, OR POLICY
Our study highlights the consequences of persistent IL-7 signaling in CAR-NK cells and informs future cytokine design strategies to improve the persistence and function of adoptively transferred NK cells. We additionally identify candidate genes that could overcome cytokine-induced dysfunction and improve NK efficacy.
Introduction
Natural killer (NK) cells expressing chimeric antigen receptors (CARs) to treat cancer have been reported over the last decade. Despite feasibility and impressive objective responses in CD19+leukemia/lymphoma,1 antitumor efficacy remains limited in solid tumors due in part to low NK cell durability. The short half-life of adoptively transferred NK cells and the hostile tumor microenvironment (TME) that is devoid of NK cell activating signals necessitate cytokine support to maintain NK cell viability.2 Cytokines that signal through the common gamma chain receptor, such as interleukin (IL)-15, play a critical role in the development and maintenance of NK cells.3 Early clinical trials using adoptively transferred NK cells provided cytokine support by doses of IL-2 or IL-15.4 5 However, systemic administration was associated with significant toxicities, long inpatient times or multiple clinic visits due to the necessity of repeated administration, and the expansion of regulatory T-cell populations.6 Novel IL-15 agonists engineered to improve safety and stability have paradoxically limited the durability, and thus efficacy, of adoptively transferred allogeneic NK cells through activation of host allorejection.7 Genetic modification of NK cells to secrete IL-15 has been especially promising for CD19 CAR-modified NK cells.8 While safe, the increased IL-15 secreted by CAR-NK cells has the potential for trans-presentation and activation of unwanted bystander cells such as suppressive myeloid populations within the TME. Indeed, glioblastoma-directed CAR-NK cells engineered to secrete IL-15 resulted in activation of local microglia populations and subsequent neurotoxicity.9 Other studies have demonstrated that an IL-15 receptor/IL-15 fusion or an IL-15 /Granulocyte-macrophage colony stimulating factor (GM-CSF) fusion molecule paradoxically limits antitumor immunity through IL-15 trans-presentation and signal transducer and activator of transcription (STAT)3 activation in suppressive macrophages.10 Thus, novel cytokine strategies that can bypass these limitations are needed to improve CAR-NK cell efficacy.
Cytokines converge on the Janus-kinase (JAK)-STAT intracellular signaling pathway. Specifically, STAT5 is a critical signaling node for NK cell development and survival.11 12 A strategy that efficiently and safely modulates STAT5 signaling specifically within adoptively transferred CAR-NK cells would be an attractive opportunity to promote persistence. Our center has previously described a synthetic constitutively-active IL-7 receptor, termed C7R,13 that contains mutations in the IL-7Rα transmembrane domain resulting in IL-7Rα homodimerization and sustained JAK1/STAT5 activation within modified cells independent of the common cytokine gamma chain. To avoid any additional exogenous modulation of the receptor and to prevent trans-presentation of signals to bystander cells, the IL7Rα extracellular domain was replaced by ectodomains derived from CD34. Thus, C7R bypasses the limitations of conventional cytokine augmentation strategies by conferring cell-intrinsic cytokine signaling irrespective of exogenous signals.
A role for IL-7 signaling in survival of early NK cell progenitors within the bone marrow and CD56bright NK cells in peripheral compartments has been established as expression of the IL-7R and its components are primarily limited to these NK subsets.14 In these settings, IL-7 provides potent signals for NK cell survival without increased activation or terminal maturation. Thus, investigating whether IL-7 signaling would benefit CAR-NK cells, in which hyperactivation or maturation could lead to rapid exhaustion within the TME, is intriguing as it would provide needed NK viability without compromising long-term function. In addition, cytokine co-stimulation of CAR-NK cells with IL-15 via canonical means, either via administration of recombinant IL-15, IL-15 super-agonists or transgenic expression of IL-15R or IL-15R/IL-15 complexes, has broader effects on NK viability, maturation, and activation.5 Given that these broader effects could increase the risk for hyperactivation in NK cells bearing a CAR (that already has potent co-stimulatory endodomains), we wanted to examine whether the unique IL-7 signaling derived from C7R could produce different results than the canonical cytokine support previously investigated. Given that C7R converges specifically on the STAT5 signaling node within NK cells without broader effects on PI-3 kinase or MAP kinase signaling seen with IL-15, we hypothesized beneficial effects on CAR-NK viability without risk of hyperstimulation or exhaustion.
Here, we demonstrate that C7R promotes survival of CAR-NK cells in the harsh solid TME but reduces their long-term cytotoxic potential. Chronic signaling via C7R in CARNK cells confers a unique transcriptional phenotype with significant overlap with recently reported stressed NK cells. This study provides valuable insights into the consequences of differential cytokine signaling in CAR-NK cells, identifies candidate target genes for maintenance of IL-7-driven viability while reversing cytokine-driven dysfunction, and guides future efforts in optimizing cytokine augmentation strategies to promote CAR-NK cell efficacy for solid tumors.
Methods
Cytokines, cell lines, and antibodies
Recombinant human IL-2 was obtained from National Cancer Institute Biologic Resources Branch (Frederick, Maryland, USA). Recombinant human M-CSF was purchased from PeproTech (Rocky Hill, New Jersey, USA). Recombinant human IL-15 was purchased from R&D Systems (Minneapolis, Minnesota, USA). The human neuroblastoma cell line LAN-1 was purchased from American Type Culture Collection (Manassas, Virginia, USA) and cultured in Dulbecco’s Modified Eagle Medium (DMEM) culture medium supplemented with 2 mM L-Glutamine (Gibco-BRL) and 10% Fetal Bovine Serum (FBS; Gibco-BRL). The human neuroblastoma cell line CHLA255 was kindly provided by Dr Leonid Metelitsa at Baylor College of Medicine and was cultured in Roswell Park Memorial Institute (RPMI)-1640 medium (Gibco-BRL) supplemented with 2 mM L-Glutamine and 10% FBS (C-RPMI). The human acute lymphoblastic leukemia cell line Nalm6.ffluc was cultured in C-RPMI. HLA-negative K562 cells genetically modified to express a membrane-bound version of IL-15 and 4-1BB ligand, K562-mb15-4-1BB-L, were kindly provided by Dr Dario Campana (National University of Singapore). All cell lines were authenticated and tested for Mycoplasma contamination monthly via MycoAlert (Lonza) mycoplasma enzyme detection kit.
C7R and CAR-encoding retroviral vectors
The construction of the SFG-retroviral vector encoding C7R was previously described.13 The SFG-retroviral vectors encoding the CD19-CAR.4-1BB.ζ and the GD2-CAR.4-1BB.ζ have been previously described.15 RD114-pseudotyped viral particles for all constructs were produced by transient transfection in 293 T cells, as previously described.16
Expansion and retroviral transduction of human NK cells
Human peripheral blood mononuclear cells (PBMCs) were obtained from healthy donors under Baylor College of Medicine IRB-approved protocols and were depleted of CD3+ T-cells using the EasySep Human CD3 Positive Selection Kit II (STEMCELL technologies). CD3-depleted PBMCs were co-cultured with irradiated (100 Gy) K562-mb15-4-1BB-L at a 1:10 (NK cell:feeder cell) ratio in G-Rex cell culture devices (Wilson Wolf, St. Paul, Minnesota, USA) for 4–5 days in STEMCELL Growth Medium (CellGenix) supplemented with 10% FBS and 10 IU/mL IL-2 and 10 ng/mL IL-15.17 NK cells were sequentially transduced on retronectin-coated plates with SFG-based retroviral vectors expressing C7R on day 5 for 16–24 hours followed by the CAR retroviral vector for an additional 48 hours. To further modify cells to express the GFP.ffluc for in vivo bioluminescence imaging, NK cells were transduced with a 1:1 mixture of GFP.ffluc and CAR retroviral vector supernatants first, and then transduced the following day with C7R retroviral vectors. To generate adequate cell numbers for experiments, transduced cell populations were subjected to a secondary expansion in G-Rex devices at a 1:1 ratio of NK cell:irradiated K562-mb15-4-1BB-L supplemented with 10 IU/mL IL-2 and 10 ng/mL IL-15 for an additional 4–7 days.17 This 15–18 days human genemodified NK cell protocol resulted in 90.0±5.6%, 89.7±5.7%, and 90.2±6.1% for untransduced (UTD), CAR, and C7R.CAR NK cells, respectively.
Flow cytometry
Fluorochrome-conjugated antibodies and the manufacturer are listed in table 1. For surface staining, cells were incubated with antibodies for 15–30 min at 4°C in the dark. For intracellular cytokine staining, cells were surface stained and then stained intracellularly using the Transcription Factor Buffer Set (BD Sciences, Franklin Lakes, New Jersey, USA) according to the manufacturer’s instructions. For phosphorylated STAT (pSTAT)5 intracellular staining, cells were surface stained and then stained intracellularly using the BD Pharmingen Transcription Factor Phospho Buffer Set (BD Sciences, Franklin Lakes, New Jersey, USA) according to the manufacturer’s instructions. For exogenous IL-15 stimulation, manufactured NK cells were rested overnight then cultured in the presence of 10 ng/mL rhIL-15 for 30 min before fixing and staining for flow cytometry. Cells were acquired on Beckman Coulter Gallios or BD FACSymphony A5, and analysis was performed using FlowJo V.10.9.0.
Table 1. Flow cytometry antibodies.
| Marker | Fluorochrome | Source | Clone |
|---|---|---|---|
| GD2-CAR | AF647 | Conjugated in-house via Abcam Alexa Fluor Conjugation Kit | 1A7 |
| 2B4 | BV605 | BD Biosciences | C1.7 |
| CD107a | APC-Cy7 | BioLegend | H4A3 |
| CD14 | PE-Cy7 | BioLegend | HCD14 |
| CD16 | PerCP-Cy5.5 | BD Biosciences | 3G8 |
| CD163 | APC-Cy7 | BioLegend | GHI/61 |
| CD3 | BUV496 | BD Biosciences | UCHT1 |
| CD33 | PE | BD Biosciences | P67.6 |
| CD34 | BUV737 | BD Biosciences | 563 |
| CD45 | BUV805 | BD Biosciences | HI30 |
| CD56 | APC-Cy7 | BioLegend | HCD56 |
| DNAM-1 | PE-Cy7 | BioLegend | 4B4-1 |
| Eomes | BV421 | BD Biosciences | X4-83 |
| GD2 | PE | BioLegend | 14G2a |
| HLA-DR | FITC | BD Biosciences | L243 |
| IFN-γ | BV421 | BD Biosciences | 4S.B3 |
| Live/Dead | FVS620 | BD Biosciences | N/A |
| NKG2D | BV786 | BD Biosciences | 1D11 |
| NKp44 | BUV615 | BD Biosciences | p44-8 |
| pSTAT5 | PE | BD Biosciences | 47/Stat5(pY694) |
| T-bet | PE | BD Biosciences | O4-46 |
| TIM-3 | AF488 | BD Biosciences | 7D3 |
Cytotoxicity assay
A 4-hour flow-cytometry-based assay was performed to determine NK cell activation via CD107a and interferon (IFN)-γ and specific cytotoxicity after stimulation against the LAN-1 neuroblastoma cell line. Briefly, 2×104 LAN-1 cells were plated per well in a 96-well tissue-culture treated flat-bottomed plate. 24 hours later, UTD or CAR-NK cells were added at an effector-to-target ratio (E:T) ratio of 10:1. The absolute viable number of LAN-1 cells per well was determined via CountBright Absolute Counting Beads (Thermo Fisher, Waltham, Massachusetts, USA) according to the manufacturer’s instructions 4 hours later. The formula used to calculate the percent specific cytotoxicity is as follows: (cell number in untreated well – cell number in assay well) / (cell number in untreated well). For CD107a and IFN-γ intracellular staining, UTD or CAR-NK cells were incubated with LAN-1 cells at a 2:1 E:T ratio for 4 hours. At 1-hour post-NK cell addition, cells were incubated in the presence of 1X Brefeldin A Solution (BioLegend, San Diego, California, USA), 1X eBioscience Monensin Solution, and anti-human APC-Cy7 CD107a (LAMP-1; BioLegend) for an additional 3 hours.
In vitro TME assay: CD14+ PBMCs were obtained from healthy donors and positively selected using CD14+ magnetic bead separation (Miltenyi Biotec, Bergisch Gladbach, Germany), frozen in 50% FBS, 40% RPMI, and 10% dimethyl sulfoxide (DMSO). Thawed CD14+ PBMCs were plated at a 1:1 ratio (2.5×105 cells) with CHLA255 cells and plated in a tissue-culture treated 24 well plate in 1 mL of C-RPMI supplemented with 100 ng/mL M-CSF to promote macrophage differentiation. 3 days later, an additional 1 mL of C-RPMI was plated on top of the existing cells and supplemented with fresh M-CSF, followed by the addition of 1×105 UTD or CAR NK cells. Four days later, supernatants were harvested for cytokine analysis and cells were harvested to assess specific cytotoxicity as described above. Absolute CAR+NK cell counts were quantified via CountBright Absolute Counting Beads. Cell culture supernatants were analyzed via MILLIPLEX Multiplex custom cytokine panel, as listed in table 2.
Table 2. Custom MILLIPLEX panel analytes.
| Bead/analyte name | Luminex magnetic bead region | Cat # |
|---|---|---|
| Anti-Human GM-CSF Bead | 21 | HGMCSF-MG |
| Anti-Human IFN-γ Bead | 26 | HIFNG-MG |
| Anti-Human IL-10 Bead | 42 | HIL10-MG |
| Anti-Human IL-1α Bead | 27 | HIL1A-MG |
| Anti-Human IL-1β Bead | 28 | HIL1B-MG |
| Anti-Human IL-6 Bead | 36 | HIL6-MG |
| Anti-Human IL-8 Bead | 38 | HIL8-MG |
| Anti-Human MIP-1α Bead | 66 | HMIP1A-MG |
| Anti-Human MIP-1β Bead | 67 | HMIP1B-MG |
| Anti-Human RANTES Bead | 74 | HRANTES-MG |
| Anti-Human TNFα Bead | 76 | HTNFA |
CAR-stimulation via 1A7
The GD2.CAR anti-idiotype antibody 1A7 was plated in a 24-well non-tissue culture plate the day before CAR-NK cell addition at a concentration of 1 µg/mL in phosphate-buffered saline (PBS) and placed at 4°C overnight. The following day, wells were aspirated and washed once with C-RPMI followed by the addition of 5×105 CAR+ NK cells to each well. 16 hours later, cell culture supernatants were harvested and analyzed for granzyme B using ELISA (R&D Systems) and cytokines via MILLIPLEX Multiplex custom panel (table 2).
IncuCyte cytotoxicity assay
Day 14 (D14) C7R.CAR-NK cells or D14 CAR-NK cells supplemented with IL-15 (10 ng/mL) were challenged at a 1:1 (1×105 cells) ratio with CHLA255.GFP.FFluc plated the day before in a 96-well tissue-cultured treated plate. Tumor growth was monitored by IncuCyte live cell image analysis by quantifying the total green area using IncuCyte imaging software as previously described.18 Data are presented as the average of triplicate wells±SEM. Area under the curve was calculated using GraphPad Prism.
Xenograft mouse models
All animal experiments followed a protocol approved by the Baylor College of Medicine Institutional Animal Care and Use Committee. Subcutaneous CHLA255 TME model. 8–12 weeks old female NSG mice were implanted subcutaneously in the dorsal right flank with 1×106 CHLA255 mixed with 0.5×105 autologous CD14+ monocytes in 100 µL of basement membrane Matrigel (Corning, Corning, New York, USA). 14 days later, when tumors became palpable, mice were randomized by tumor size to ensure equal size and variance between each group and 5×106 CAR-NK cells were administered intravenously or intratumorally, depending on the experiment. 3–5 days later, a second dose of CAR-NK cells was administered via the same route as the first dose. PBS or 1 µg of recombinant hIL-15 (R&D Systems) was administered intraperitoneally (i.p.) three times weekly. Tumors were measured two to three times per week with calipers. For in vivo bioluminescence imaging of NK cells, mice were administered 100 µL of 15 mg/mL Dluciferin (Xenogen Corporation, Hopkinton, Massachusetts, USA) injected i.p. and 7 min later were imaged using the IVIS system (Xenogen Corporation). The mice were euthanized when the tumor volume was ≥1,500 mm3. For intratumor analysis of NK cells via flow cytometry, tumors were mechanically digested and red blood cells (RBC) lysed via eBioscience RBC Lysis Buffer (Thermo Fisher). Live cells were counted via trypan blue staining, and 2×106 cells were stained for flow analysis. Intravenous Nalm6 minimal residual disease model.19 10-week-old female NSG mice were intravenously injected with 5×104 Nalm6.GFP.ffluc, and the following day, dosed with 4×106 NK cells intravenous. 7 days later, mice received a second dose of 4×106 NK cells. Tumor growth was monitored via bioluminescence imaging using the same protocol as described above, and mice were imaged two times weekly. Mice were euthanized when they became moribund.
Gene expression analysis
Total RNA was isolated using the RNeasy Micro Kit (Qiagen, Venlo, the Netherlands). Gene expression analysis used the nCounter Human Immune Exhaustion Panel (NanoString, Seattle, Washington, USA) and was acquired on an nCounter MAX Analysis System (Baylor College of Medicine Genomic and RNA Profiling Core, Houston, Texas, USA). Data was analyzed using the nSolver V.4.0 software (NanoString). Raw and processed files have been uploaded to the Gene Expression Omnibus (GEO) repository and GEO accession number is GSE277499.20
Statistical analysis
All numerical data are represented as mean±SD of either number of donors or experimental replicates, as indicated. Paired two-tailed t-test or one-way analysis of variance with Tukey’s multiple comparison test was used to determine significance of differences between means with p<0.05 indicating a significant difference. All statistical analyses were performed with GraphPad Prism V.10 Software (GraphPad).
Results
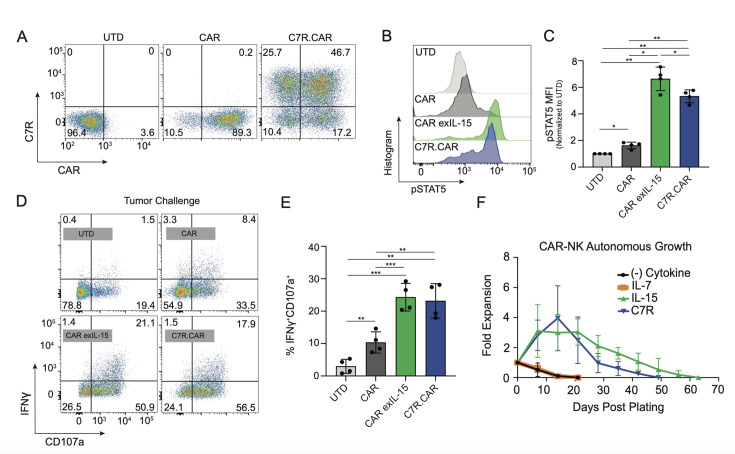
C7R promotes CAR-NK cell survival and enhances activity against tumor targets in vitro. We first investigated the effect of C7R in NK cells co-expressing a CAR directed against the GD2 antigen (C7R.CAR-NK cells) in eliciting pSTAT5 signaling, improved persistence, and increased effector function. To generate C7R.CAR-NK cells, primary human NK cells were expanded for 4–5 days and sequentially transduced with retroviral vectors encoding C7R and the GD2-CAR as detailed in Methods. We achieved 90.3±6.1% pure NK populations (online supplemental figure 1A). No significant differences in NK purity were found between UTD, CAR, or C7R.CAR-NK cells. NK cells were able to express both C7R and CAR at rates of 43.0±10.2% (n=11 donors) (figure 1A, online supplemental figure 1B, C). Sequential transduction of C7R followed by the CAR resulted in a slight but significant decrease in the overall total proportions of CAR-expressing NK cells (online supplemental figure 1D). No differences in secondary expansion rates or cell-surface phenotype were observed between CAR-NK cells with or without C7R due to the presence of IL-2 and IL-15 in the expansion system (online supplemental figure E-M). The decreased expansion exhibited by CAR-expressing NK cells compared with their unmodified counterparts in post-generation expansion cultures that do not contain the CAR antigen (online supplemental fig 1E) is a noted response to viral transduction.17 21 To determine if C7R increased pSTAT5 signaling, we performed intracellular flow cytometry in CAR-NK cells after 24 hours of cytokine starvation. C7R.CAR-NK cells exhibited a 3.4±0.7-fold increase in pSTAT5 compared with unstimulated CAR-NK cells (n=4 donors) (figure 1B, C). Stimulating CAR-NK cells with exogenous IL-15 for 30 min resulted in a slight but significant increase in pSTAT5 compared with C7R.CAR-NK cells. In short-term co-culture with GD2+ LAN-1 tumor cells, C7R.CAR-NK cells demonstrated similar proportions of IFN-γ+CD107a+ double-positive cells to CAR-NK cells activated with exogenous IL-15 (figure 1D, E). To assess if C7R promoted NK survival and expansion, we plated C7R.CAR-NK cells in cytokine-free media and compared fold expansion over time to that of CAR-NK cells with or without two times weekly supplementation of IL-7 or IL-15 (figure 1E). Without cytokine, CAR-NK cell numbers declined steeply over 1–2 weeks. IL-15 supplemented CAR-NK cells and C7R.CAR-NK cells proliferated in the first 3 weeks followed by CAR-NK cell contraction, confirming no C7R-mediated autonomous growth. The vast majority of peripheral blood NK cells do not express IL-7Rα,14 and because our CAR-NK cell product is derived from peripheral blood NK cells, we did not expect IL-7 to improve CAR-NK survival. Indeed, CAR-NK cells supplemented with IL-7 did not promote CAR-NK cell survival over no cytokine (figure 1E).
Figure 1. C7R transduction increases CAR-NK persistence and short-term function. (A) Representative flow plots demonstrating transduction efficiencies in untransduced (UTD), CAR, and C7R.CAR-NK cells. (B) Representative flow plot of pSTAT5 after overnight cytokine starvation. For exIL-15, cytokine-starved CAR-NK cells were stimulated for 30 min with 10 ng/mL IL-15. (C) Pooled data from (B), n=4 independent donors. Data presented as mean±SD; p*<0.05; p**<0.01. (D) Representative flow plots of intracellular IFN-γ and CD107a in 4-hour flow co-culture assays with LAN1 tumor cells at a 2:1 effector-to-target ratio ratios. (E) Pooled data from (D), n=4 independent donors. presented as mean±SD; p*<0.05; p**<0.01; p***<0.001. (F) CAR-NK fold expansion in the absence (black) or presence of IL-7 (10 ng/mL; orange), IL-15 (10 ng/mL; green), or transduced with C7R. CAR, chimeric antigen receptor; exIL, exogenous interleukin; IFN, interferon; NK, natural killer; pSTAT, phosphorylated signal transducer and activator of transcription.
C7R.CAR-NK cells mediate antitumor activity without cytokine support in a novel tumor immune microenvironment co-culture
To test our hypothesis that C7R would enhance the survival and function of CAR-NK cells in TMEs, we developed a tumor immune microenvironment (TiME) co-culture assay that models extratumoral components of solid tumors by incorporating suppressive myeloid infiltrates. To induce M2-like polarization of monocytes, we co-cultured CD14+ monocytes for 3 days with the CHLA255 neuroblastoma cell line previously shown to secrete high amounts of transforming growth factor-beta (TGF-β)1 and induce robust monocyte-mediated production of IL-6.22 Both cytokines have been implicated in inhibiting NK function.23 After 3 days of co-culture, the monocytes were skewed to an inhibitory M2 macrophage-like phenotype with lower cell surface levels of HLA-DR and increased CD163, and consisted of immunosuppressive factors including IL-1α, IL-4, IL-6, IL-8, and IL-10 (online supplemental figure 2). To this established TiME co-culture, we added C7R.CAR-NK cells (autologous to macrophages to prevent alloreactivity) at a tumor-favoring E:T ratio of 1:5. In this model, we included exogenous IL-15 supplementation (exIL-15.CAR-NK) as a cytokine control. Because our novel TiME co-culture assay is 4 days in duration, it serves as an indirect representation of serial killing capacity, as CAR-NK cells can kill their first targets encountered within hours and likely kill multiple rounds during this 4-day period. Given tumor cells and suppressive tumor-associated macrophages are both adherent to NK cells in co-cultures, we chose to use our 4-day TiME assay instead of performing the more traditional serial co-culture assay which would result in transfer of dead tumor and NK cell doublets from the previous co-cultures into the new one, and yield difficult-to-interpret data. As seen in figure 2 using our novel TME co-culture, CAR-NK cells co-expressing C7R exhibited improved cytotoxicity, expansion, and proinflammatory cytokine secretion compared with CAR-NK cells without cytokine, while demonstrating similar function to CAR-NK supplemented with exIL-15.
Figure 2. C7R augments CAR-NK function in in vitro TME mimics. (A) Specific cytotoxicity of indicated NK cells after 4-day co-culture with CHLA255 and autologous CD14+monocytes. Briefly, 2.5×105 CHLA255 and 2.5×105 autologous CD14+ monocytes were cultured in a 24-well plate for 3 days prior to the addition of NK cells after which 1×105 UTD or CAR-NK cells were added directly into the cell culture. For exIL-15, IL-15 was supplemented at a concentration of 10 ng/mL and was replenished each day. (B) CAR+ NK fold-expansion after 4 days of TME co-culture. CAR+ NK fold expansion was quantitated via absolute CAR+ NK counts calculated from counting beads added to flow cytometry samples prior to acquisition. Each unique shape represents a different donor. (C) Cell culture supernatants were analyzed for IFN-γ, TNF-α, and GM-CSF after 4 days of TME co-culture in indicated CAR-NK conditions. Data presented as mean±SD of independent donors (n=6, A; n=4, B, C). *p<0.05, **p<0.01, ****p<0.001 (one-way paired analysis of variance, A, B, C). CAR, chimeric antigen receptor; exIL, exogenous interleukin; GM-CSF, granulocyte-macrophage colony stimulating factor; IFN, interferon; NK, natural killer; TME, tumor microenvironment; TNF, tumor necrosis factor; UTD, untransduced.
C7R.CAR-NK cells fail to control tumor in solid TiME xenografts
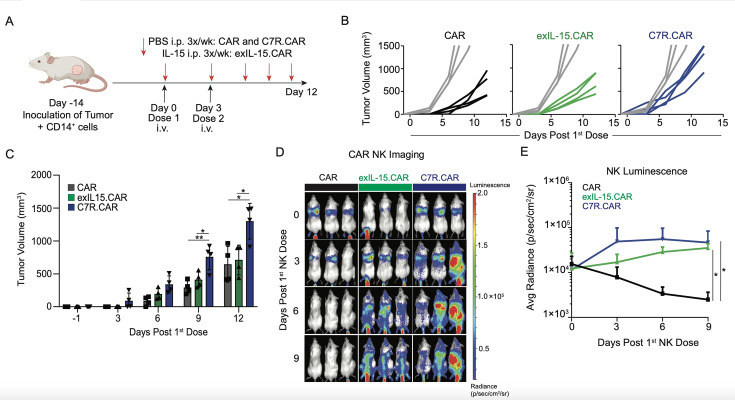
Our collective in vitro data demonstrated that C7R augmented CAR-NK cell function and survival without the need for exogenous cytokine support. We next assessed the in vivo survival and function of C7R.CAR-NK cells compared with that of CAR-NK cells with or without exIL-15. To simulate a human myeloid-rich TME similar to our TiME in vitro co-culture, we subcutaneously inoculated CHLA255 tumor cells admixed with CD14+ PBMCs in a Matrigel matrix into NSG mice (figure 3A), resulting in accelerated tumor growth compared with CHLA255 tumors engrafted alone (online supplemental figure 3). On day 14, when tumors reached~50 mm3, C7R.CAR-NK cells were infused intravenously followed by a second dose 3 days later. All in vivo experiments used the same NK cell donor. When compared with a group of independent donors used in our studies overall (online supplemental figure 4), the in vivo donor exhibited low endogenous cytotoxicity and moderate cytotoxicity with CAR expression that was further enhanced with cytokine support. We used this criteria to select the in vivo donor for these studies because we wanted an NK donor in which most of the cytotoxicity was conferred by the CAR, not endogenous NK cell receptors such as KIR and NCR, thus truly assessing effects of cytokine modulation on CAR-specific activity in the NK cells. As seen in figure 3B, C, CAR-NK cells delayed tumor growth in all groups. However, by day 6 post the second NK cell dose, C7R.CAR-NK cells lost tumor control relative to CAR-NK cells with exIL15. To confirm that the observed differences in efficacy were not due to differences in NK cell survival, we performed a separate in vivo study in which we modified CAR-NK or C7R.CAR-NK cells with firefly luciferase to track NK cells via bioluminescence. As seen in figure 3D, E, either C7R or exIL-15 cytokine stimulation was necessary to maintain CAR-NK cell bioluminescence signal in vivo. Importantly, C7R.CAR-NK cells demonstrated similar bioluminescence to CAR-NK cells with exIL15, suggesting that the waning antitumor efficacy seen with C7R.CAR-NK cells was not due to poor NK cell survival.
Figure 3. C7R.CAR-NK fail to control tumor long-term in in vivo xenograft. (A) In vivo schematic. Briefly, 1×106 CHLA255 and 0.5×106 autologous CD14+peripheral blood mononuclear cells were implanted subcutaneously in the right flank of NSG mice. 2 weeks later, 5×106 CAR-NK cells were administered intravenously followed by a second dose 3 days later. PBS (CAR, C7R.CAR) or IL-15 (CAR exIL-15) was administered i.p. three times per week. (B) Spider plots of tumor volume in each indicated NK condition overlaid with control PBS (gray) condition. Tumor volumes were determined by caliper measurement. (C) Tumor volumes from (B) plotted at respective timepoints in indicated CAR-NK condition. (D). Sequential bioluminescent imaging of NK cells administered intravenously. (E) Quantitated bioluminescent signal of NK cells over time from (D). Four mice were used for each NK group and three for control in B, C. Three mice were used for each NK group in D, E. Data presented as averages between mice±SD (n=4, C; n=3, E). *p<0.05, **p<0.01 (one-way analysis of variance, C, E). CAR, chimeric antigen receptor; exIL, exogenous interleukin; i.p., intraperitoneal; NK, natural killer; PBS, phosphate-buffered saline.
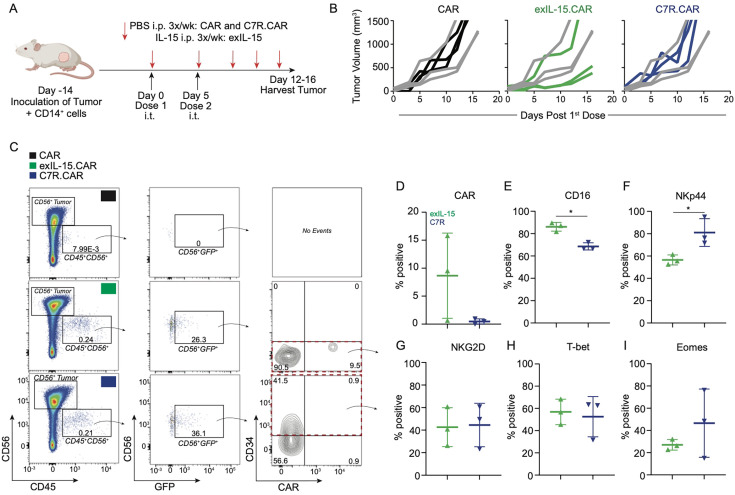
To control for potential differences in CAR-NK cell trafficking and viability within the TME conferred by NK cell-internal C7R versus systemically administered IL-15, we performed a separate study in which CAR-NK cells were administered directly into the subcutaneous tumor (figure 4A). In this intratumor treatment setting, neither CAR-NK cells without exogenous cytokine supplementation nor C7R.CAR-NK controlled tumor, while CAR-NK cells with exIL-15 delayed tumor growth in the majority of mice (figure 4B and online supplemental figure 5). In this experiment, CAR-NK cells were also transduced with firefly luciferase to determine CAR-NK cell survival within the TME. While CAR-NK cells without cytokine exhibited poor survival in the TME, C7R and exIL-15 supported CAR-NK survival to the same extent (online supplemental figure 6A, B). We harvested tumors 9–13 days post-1st NK cell dose to confirm CAR-NK cell survival in the TME and to determine any potential differences in phenotype. Given that virtually all green fluorescent protein (GFP)+ cells were also CAR+ at time of injection (not shown), gating on GFP+ human NK cells prior to examining CAR and C7R expression ensured we were examining all modified NK cells injected irrespective of downregulation of CAR or C7R expression after TME exposure (shown in figure 4C) when comparing tumors from different treatment groups. We detected NK cells in the exIL-15 and C7R groups, but no NK cells in the CAR-NK cell group without cytokine supplementation. While NK cells from both groups showed loss of surface CAR staining within the TME, tumors from two of the three mice supplemented with exIL-15 had detectable CAR expression on NK cells, while NK cells in the C7R.CAR-NK treated tumors had virtually no surface CAR expression (figure 4D). C7R.CAR-NK also demonstrated decreasing and increasing proportions of CD16+ and NKp44+ cells, respectively (figure 4E, F). In contrast, the levels of NKG2D and the transcription factor, T-bet, were similar between groups while Eomes was trending towards increased in C7R.CAR-NK cell (figure 4G–I). This data demonstrates that C7R confers signals to keep NK cells alive within the TME and that C7R.CARNK cells adopt a distinct phenotype compared with exIL15.CAR-NK cells.
Figure 4. C7R.CAR-NK cells fail to control tumor despite localization and survival in the tumor microenvironment. (A) In vivo schematic. Briefly, 1×106 CHLA255 and 0.5×106 autologous CD14+ peripheral blood mononuclear cells were implanted subcutaneously in the right flank of NSG mice. 2 weeks later, 5×106 CAR-NK cells were administered intratumorally (i.t.) followed by a second dose 3 days later. PBS (CAR, C7R.CAR) or IL-15 (CAR exIL-15) was administered i.p. three times per week. (B) Spider plots of tumor volume in each indicated CAR-NK condition overlaid with control PBS (gray) condition. Tumor volumes were determined by caliper measurement. (C) Gating strategy to determine GFP+ NK phenotype in digested tumors, red boxes indicate gated population for downstream analysis in (D), (E), (F), (G), (H), (I). Cell surface receptors and intracellular transcription factors expression from cell populations gated in red from (C). Three mice were used for each group in B, C. Data presented as averages between mice±SD (n=3, B, C, D, E, F, G, H, I). *p<0.05, **p<0.01 (two-tailed t-test, D, E, F, G, H, I). CAR, chimeric antigen receptor; exIL, exogenous interleukin; GFP, green fluorescent protein; i.p., intraperitoneal; NK, natural killer; PBS, phosphate-buffered saline.
A potential explanation for the surprizing low in vivo activity of C7R.CAR-NK cells despite their durability was that the constitutive C7R signal combined with the known tonic signaling of the second generation GD2.CAR led to early dysfunction of the CAR-NK cells.24 While there have been no studies definitively reporting the effects of tonic signaling in CAR-NK cells, observations reported in the literature extrapolated from data or observations in CAR-T cells assume similar mechanisms. We wanted to determine if a combination of C7R with a first-generation GD2.CAR with lower tonic signaling reported in T cells would reduce NK exhaustion and result in enhanced in vivo antitumor efficacy. We performed an in vivo study in which we compared NK cells that co-expressed C7R and a first generation GD2.CAR with only the cytotoxic ζ-chain (CAR.ζ) to the second generation C7R.CAR.4-1BBζ cells used in previous experiments. CAR.ζ and CAR.4-1BBζ NK cells with exIL-15 were used as controls (online supplemental figure 7A). While both first and second generation CARs with exIL-15 exhibited tumor control (online supplemental figure 7B, C), neither first nor second generation C7R containing CAR-NK cells controlled tumor. Thus, the signaling domain of the CAR in the context of a tonic-signaling GD2.CAR does not explain the reduced efficacy of C7R.CAR-NK cells.
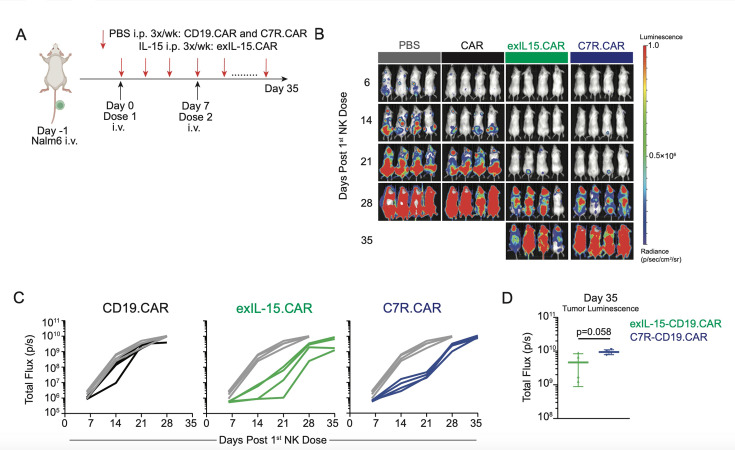
Treating established GD2+ subcutaneous tumors in immunodeficient mice with NK cell therapies has proven challenging.25 While we demonstrated modest tumor control in our CAR-NK groups supplemented with IL-15, all tumors eventually progressed and mice succumbed to tumor growth. To clarify if C7R was detrimental to the long-term cytotoxic potential of CAR-NK cells in general, we used an established CD19-targeting CAR with demonstrated clinical efficacy.26 We treated the CD19+ Nalm6 tumor in a systemic model of leukemia19 with CD19.CAR-NK cells with or without exIL-15 or C7R.CD19.CAR-NK cells (figure 5A). Nalm6 quickly escaped CD19.CAR-NK cells given without cytokine support, while CD19.CAR-NK cells with exIL-15 and C7R.CAR-NK cells demonstrated early control (figure 5B, C). However, by day 35, mice treated with CAR-NK cells with exIL-15 demonstrated improved tumor control compared with C7R.CAR-NK (figure 5D), providing further support that C7R and IL-15 signaling were resulting in different propensities for long-term tumor control.
Figure 5. C7R-CD19.CAR-NK fail to control tumor long-term in B-cell leukemia. (A) In vivo schematic. Briefly, on day −1, 5×104 Nalm6 were administered intravenously in NSG mice. On day 0, 4×106 CAR-NK cells were administered intravenously followed by a second dose 7 days later. IL-15 (1 µg/dose) was administered i.p. only in the CAR exIL-15 treated mice two to three times per week. (B) Representative imaging of Nalm6 growth over time. (C) Quantitated bioluminescent signal of Nalm6 over time from (C). (D) Quantitated Nalm6 bioluminescence on day 35. Four mice were used for each group in B, C. Data presented as averages between mice±SD (n=4, D; two-way t-test). CAR, chimeric antigen receptor; exIL, exogenous interleukin; i.p., intraperitoneal; NK, natural killer; PBS, phosphate-buffered saline.
Chronic C7R signaling induces a stressed NK cell phenotype
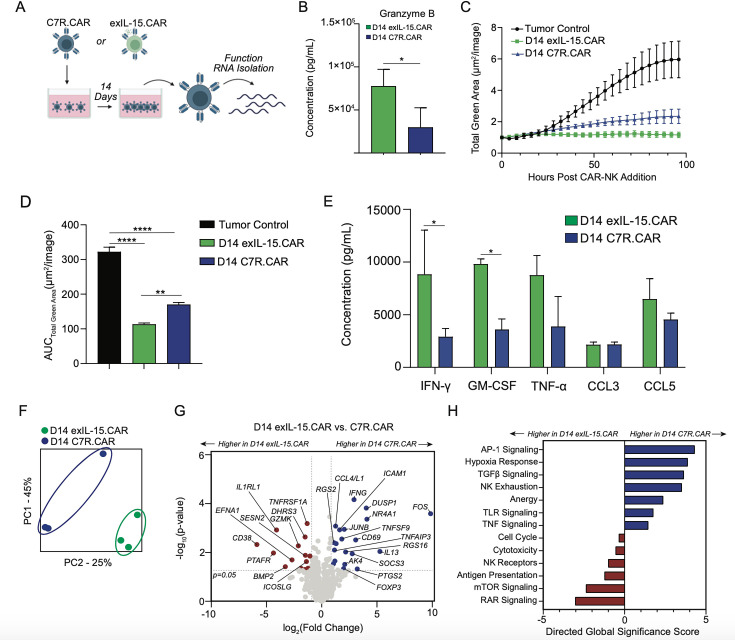
Given that C7R.CAR-NK cells demonstrated early efficacy both in vitro and in some in vivo experiments, we posited that differences between C7R and IL-15 signaling are reflective of chronic signaling conditions. To assess CAR-NK cell function after chronic C7R or IL-15 signaling, we cultured CAR-NK cells co-expressing C7R or supplemented with exogenous IL-15 for 14 days (a biologically significant time point to capture potential differences based on our in vivo data) and then activated the resultant CAR-NK cells via plate-bound anti-CAR single-chain variable fragment (scFv) idiotype antibody (figure 6A). Day 14 (D14) C7R.CAR-NK cells demonstrated 63.5±19.6% lower levels of granzyme B compared with D14 exIL-15.CAR-NK cells (figure 6B). This difference could be explained by lower CAR expression on the surface of the NK cells since D14 C7R.CAR-NK cells demonstrated 19.8±5.1% lower CAR mean fluorescent intensity (MFI) relative to exIL-15 (online supplemental figure 8A, B). However, the mildly lowered CAR expression was disproportional to the vastly decreased granzyme B, suggesting additional factors that would reduce granzyme B release. In 96-hour co-cultures at a 1:1 ratio with CHLA255 tumor cells, D14 CAR-NK cells with exIL-15 demonstrated better tumor control than D14 C7R.CAR-NK cells (figure 6C, D). We also examined cytokine and chemokine production after 48 hours of CAR activation via plate-bound anti-CAR scFv. D14 exIL-15 CAR-NK cells produced more IFN-γ and GM-CSF than D14 C7R.CAR after stimulation while secreting similar levels of tumor necrosis factor (TNF)-α and the chemokines C-C motif ligand (CCL)3/5 (figure 6E). This data demonstrated that constitutive C7R signaling in CAR-NK cells was associated with blunted effector function compared with stimulation with IL-15 but was able to maintain some similar functions such as cytokine and chemokine secretion.
Figure 6. Long-term C7R signaling induces phenotype associated with NK dysfunction. (A) Experimental schematic. Briefly, C7R.CAR or exIL-15 CAR-NK cells were cultured for 14 days followed by total RNA isolation. RNA was also isolated from day 0 samples. Gene expression analysis was subsequently performed using the Human Immune Exhaustion and nCounter Analysis System (NanoString). (B) On day 14, CAR exIL-15 or C7R.CAR-NK cells were stimulated with the 1A7 anti-idiotype antibody for 16 hours to stimulate CAR-mediated production of granzyme B measured via ELISA. (C) Day 14 CAR-NK cells were cultured at a 1:1 ratio to assess CHLA255 tumor growth analyzed via live cell imaging using IncuCyte. (D) Area under the curve (AUC) from (C). (E) Soluble cytokine concentrations in different CAR-NK conditions after 48 hours anti-idiotype 1A7 stimulation. (F) Principal component analysis in day 14 CAR.exIL-15 versus day 14 C7R.CAR. (G) Volcano plot demonstrating differentially expressed genes in day 0 versus day 14 C7R.CAR NK cells. Horizontal dashed line indicates p value<0.05; vertical dashed lines indicate genes that are <log2 fold change (−1) or >log2 fold change. (H) Directed global significance scores of indicated pathways at day 14 RNA. Data presented as mean±SD of different donors. (n=3, B–H). p*<0.05, p**<0.01, p****<0.001 (paired two-sided t-test, B, E; one-way analysis of variance, D). CAR, chimeric antigen receptor; CCL, C-C motif ligand; exIL, exogenous interleukin; GM-CSF, granulocyte-macrophage colony stimulating factor; IFN, interferon; NK, natural killer; TNF, tumor necrosis factor.
To assess the transcriptomic consequences of chronic C7R and IL-15 signaling, we isolated bulk RNA from D14 C7R and exIL-15.CAR-NK cells. Principal component analysis revealed distinct transcriptomes in D14 C7R and exIL-15.CAR-NK cells (figure 6F). Relative to IL-15, C7R.CAR-NK cells upregulated genes associated with AP-1 signaling including FOS and JUNB (figure 5G). This resulted in a high global pathway significance score for AP-1 signaling (figure 6H). Interestingly, C7R.CAR-NK cells also demonstrated significant upregulation of IFNG (figure 6G), despite their blunted capacity to secrete IFN-γ in response to CAR stimulation (figure 6E). Additional genes upregulated in C7R.CAR-NK cells included NR4A1 and RGS16 (figure 6G), proteins that have been implicated in both NK- and T-cell exhaustion.27 28 Overall, transcriptomic differences between C7R and IL-15 revealed higher pathway scores for NK exhaustion, anergy, TGF-β signaling, and hypoxia response with C7R (figure 6H). Conversely, IL-15 treated cells scored higher for NK activating receptors, antigen presentation, and cytotoxicity. Taken together, these data suggest that chronic C7R signaling drives a distinct “stressed” NK cell transcriptional profile recently identified in intratumor NK cells.29 Because stressed cells can externalize phosphatidylserine while remaining viable,30 we probed D14 C7R.CAR-NK cells and exIL15.CAR-NK cells for Annexin-V and 7-aminoactinomycin D (AAD) expression to determine the proportion of cells that displayed features of stress (Annexin-V+7-AAD−) versus apoptosis (Annexin-V+7-AAD+). D14 C7R.CAR-NK cells demonstrated higher proportions of stressed, early apoptotic cells compared with exIL-15 but similar proportions of apoptotic cells (online supplemental figure 8C, D), further supporting the hypothesis that chronic C7R signaling induces a stressed NK phenotype in CAR-NK cells while maintaining viability.
Discussion
Here, we describe the generation and functional evaluation of CAR-NK cells modified to express a constitutively active IL-7 receptor, termed C7R. CAR-NK cells co-expressing C7R demonstrated improved cytotoxicity and survival in vitro in an exogenous cytokine-independent manner. However, C7R.CAR-NK cells demonstrated poor long-term tumor control in our TME models in vivo when compared with CAR-NK cells supplemented exogenously with recombinant IL-15. The difference in tumor control was not a result of decreased C7R.CAR-NK persistence in vivo as C7R and IL-15 resulted in similar CAR-NK survival. Given these observations, we reason that long-term C7R signaling induces an NK cell phenotype with enhanced survival capabilities but diminished CAR-mediated cytotoxicity compared with IL-15. Transcriptomic profiling in C7R.CAR-NK cells identified gene signatures typically associated with NK cell metabolic stress. Our data reveal important consequences of continuous IL-7 signaling within CAR-NK cells and provide novel insights for improving cytokine stimulation of CAR-NK cells for optimal function within the TME of solid tumors.
Given that C7R converges on the STAT5 signaling pathway considered essential for NK cell survival and function,31 testing whether C7R enhanced survival and function in CAR-NK cells seemed reasonable. Surprisingly, while improvement in CAR-NK cell survival within the solid TME was noted with C7R, tumor control was diminished. A potential explanation for the effect of C7R in CAR-NK cells may reside in the natural biology of IL-7 in lymphocyte lineages. IL-7 signaling initiates when it binds to its receptor heterodimer consisting of the IL-7Rα (CD127) and the common cytokine gamma chain.32 The common lymphoid progenitor (CLP) gives rise to all lymphoid lineages, and IL-7 plays a critical role in the maintenance of the CLP pool.32 However, as both T and NK cells progress through maturation, the expression of IL-7Rα changes. In naïve and memory T cells, IL-7 functions as a survival factor33 and its overexpression can increase memory T-cell numbers.34 Given its role in supporting memory T cells, strategies to incorporate IL-7 signaling into CAR-T therapies have resulted in promising preclinical and clinical results.35 36 In contrast, NK cells lose expression of the IL-7Rα as they mature, and its expression is largely confined to the immature CD56bright NK cell compartment, a small subset of blood NK cells.14 37 The more prominent CD56dim NK cell subset in blood does not express IL-7Rα, and treatment of blood NK cells with IL-7 functions only to augment CD56bright NK cell survival and does not increase NK cell cytotoxicity or IFN-γ secretion.14 38 Our data suggest that conferring IL-7 signaling to a peripheral blood-derived engineered CAR-NK cell population results in similar effects, augmenting CAR-NK cell survival without promoting cytotoxicity.
As our CAR-NK manufacturing protocol includes IL-15 (both supplemented in culture and transpresented by feeder cells) that promotes the maturation of CD56bright NK to CD56dim,39 the minority of CD56bright blood NK cells at the start of manufacture converts to a CD56dim population by manufacturing end. Thus, when we transduce peripheral blood-derived CAR-NK cells with C7R, we are inducing IL-7 signaling that would otherwise not occur naturally in this CD56dim population. Given the role IL-7 plays in maintaining the survival of CD56bright NK cells, it is plausible that forcing IL-7 signaling in the CD56dim mature subset could polarize CAR-NK cells to a CD56bright-like phenotype capable of viability and robust cytokine secretion, but with poor cytotoxicity. Our data support this contention. First, chronic C7R signaling in CAR-NK cells resulted in increased expression of DUSP1, FOS, JUN, and CD69, a recently identified gene signature of a terminally differentiated blood NK cell subset with features of CD56bright NK cells.40 Other high-dimensional analyses have revealed that the CD56bright NK subset is associated with the high relative expression of FOS compared with CD56dim subsets.41 Lastly, we saw that C7R.CAR-NK cells secreted equivalent amounts of TNF-α, CCL3, and CCL5 to CAR-NK stimulated with IL-15. This suggests that C7R.CAR-NK cells remain in a state equipped to secrete cytokines and chemokines, in line with the canonical functions of CD56bright NK cells. Our data highlights a few unanswered questions: (1) whether forced expression of IL-7Rα in CD56dim CAR-NK cells and subsequent treatment with IL-7 would result in a similar NK cell phenotype; and (2) whether isolating CD56dim versus CD56bright NK cells from PBMCs initially, then conferring C7R via non-viral transduction methods that circumvent the need for NK activation/expansion (ie, direct genome editing) to each of the NK subsets independently would result in differential durability, phenotypic and transcriptomic characteristics, and CAR-based killing of tumor. These experiments require additional technical innovation for adequate application to primary human CAR-NK cells, are out of the scope of the current study, but are part of ongoing investigations.
Our data revealed that C7R.CAR-NK cells demonstrated rapid functional impairment in vivo. Recent reports have similarly revealed that NK cells rapidly lose their effector function on entry into tumor,42 43 and that intratumor NK cells adopt a distinct transcriptional signature when compared with their circulating counterparts.29 Although we were not able to transcriptionally interrogate C7R.CAR-NK cells isolated from the TME of our in vivo studies, our in vitro data from chronically stimulated C7R.CAR-NK cells revealed shared transcriptional features with intratumor NK cells, including increased CD69, DUSP1, and NR4A1, a gene implicated in NK cell exhaustion.27 44 C7R.CAR-NK cells also shared higher pathway signaling scores for immunosuppressive pathways, including TGF-β signaling and hypoxic responses.37 Further, a recent study identified a “stress” score associated with intratumor CD56bright NK cells characterized by high expression of DUSP1 and genes associated with TGF-β and hypoxia.29 Our data showing reduced secretion of IFN-γ, GM-CSF, TNF-α, and the chemokines CCL3 and CCL5 (figure 6E), an activating cytokine profile expressed by highly functional NK cells,45 46 by CAR-NK chronically exposed to C7R compared with exIL15-stimulated NK cells support the notion of a stressed CAR-NK phenotype. Thus, taken together, these data suggest that chronic C7R signaling results in a stressed NK phenotype capable of persistence but not cytotoxicity. The capacity of C7R to promote a stressed NK phenotype likely exacerbates the inhibitory effects of the TME, resulting in poor tumor control in vivo demonstrated by C7R.CAR-NK cells.
The constitutive nature of the C7R signal likely has detrimental effects on the long-term fitness of CAR-NK cells. Recent studies showing that chronic treatment of non-CAR NK cells with IL-15 or novel IL-15 agonists resulted in phenotypic and functional NK cell alterations detrimental to NK cytotoxicity are consistent with our findings.47 48 Our data implies that providing cytokine “bursts” as co-stimulation may be best for CAR-NK cells. Indeed, intermittent IL-15 exposure of non-CAR NK cells resulted in improved cytotoxicity compared with continuous IL-15 stimulation.48 Accordingly, pulsing C7R.CAR-NK cells with JAK inhibitors, such as ruxolitinib, could intermittently turn off C7R and potentially provide discontinuous cytokine signaling. However, these drugs are not C7R-specific and concerns about inhibiting all JAK proteins, which would likely have consequences for NK cell viability and overall function, remain unaddressed. Whether C7R can be modulated to intermittently signal remains an area of ongoing investigation.
Successfully translating CAR-NK therapies to the solid tumor setting will likely require multiple modifications to the CAR-NK cell, and genetic modifications to knockout pro-stress genes could play a part in improving the efficacy of adoptive CAR-NK cell therapy. Our data identifies potential gene candidates that could be targeted to reduce NK cell stress within the solid TME. In particular, the NR4A family of proteins represent promising initial targets, given the upregulation of NR4A1 seen in stressed C7R.CAR-NK cells. The NR4A family of orphan nuclear receptors consists of NR4A1, NR4A2, and NR4A3 that function in a ligand-independent manner.49 NR4A1 has been shown to be upregulated in poorly cytotoxic tumor-infiltrating NK cells in hepatocellular carcinoma (HCC), and CRISPR/Cas9 knockout of NR4A1 improved the efficacy of adoptively transferred murine NK cells in an in vivo model of HCC.27 Furthermore, triple knockout of all NR4A proteins improved CAR-T efficacy due to enhanced metabolic fitness.50 However, this approach has yet to be reported for CAR-NK cells.
In conclusion, we have demonstrated that constitutively active IL-7 signaling from an engineered IL-7 receptor significantly enhanced NK survival in vitro and in vivo but failed to result in long-term tumor control. Our data revealed the differential effects of IL-7 versus IL-15 signaling in CAR-NK cells. Chronic C7R signaling was associated with a phenotype resembling characteristics of poorly cytotoxic CD56bright NK cells and stressed intratumor NK cells with inferior long-term antitumor function. This study lends important insights into the benefits and limitations of cytokine-based CAR-NK co-stimulation and informs future directions in the development of effective cytokine-modified NK cells for the adoptive immunotherapy of solid tumors.
Research ethics approval
This study does not directly involve human subjects but does use human materials that require IRB approval for acquisition and use. Human PBMCs used in this study were obtained from healthy donors under Institutional Review Board for Baylor College of Medicine and Affiliated Hospitals-approved “Healthy Participant Study H45017: Studies of immune responses to tumor and viruses in healthy and immunocompromised individuals”. The current study also involved animal subjects and was approved by the Baylor College of Medicine Institutional Animal Care and Use Committee, Protocol AN-8703: Engineered NK-based and T cell-based immunotherapies.
Supplementary material
Acknowledgements
Authors acknowledge support of the Baylor College of Medicine Comprehensive Cancer Training Program (CPRIT RP210027; PI: J. Rosen, Co-PI V. Hoyos) for trainee, Dayenne van Leeuwen.
Footnotes
Funding: Funding for work was provided by Alex’s Lemonade Stand Foundation (grant # 521‑23536) and American Cancer Society (grant # MRSG-16-196-01-LIB)
Provenance and peer review: Not commissioned; externally peer reviewed.
Patient consent for publication: Not applicable.
Ethics approval: This study does not directly involve human subjects but does use human materials that require institutional review board approval for acquisition and use. Human peripheral blood mononuclear cells (PBMCs) used in this study were obtained from healthy donors under Institutional Review Board for Baylor College of Medicine and Affiliated Hospitals-approved “Healthy Participant Study H-45017: Studies of immune responses to tumor and viruses in healthy and immunocompromised individuals.”
Data availability free text: Dysthe, M. and Parihar, R. Data from: Human Immune Exhaustion Gene Expression Profile in C7R-CAR and CAR NK cells. NCBI GEO Repository, September 30, 2024. GEO accession # GSE277499. https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE277499.
Data availability statement
Data are available in a public, open access repository. All data relevant to the study are included in the article or uploaded as supplementary information.
References
- 1.Marin D, Li Y, Basar R, et al. Safety, efficacy and determinants of response of allogeneic CD19-specific CAR-NK cells in CD19+ B cell tumors: a phase 1/2 trial. Nat Med. 2024;30:772–84. doi: 10.1038/s41591-023-02785-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kong JC, Sa’ad MA, Vijayan HM, et al. Chimeric antigen receptor-natural killer cell therapy: current advancements and strategies to overcome challenges. Front Immunol. 2024;15:1384039. doi: 10.3389/fimmu.2024.1384039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lodolce JP, Boone DL, Chai S, et al. IL-15 receptor maintains lymphoid homeostasis by supporting lymphocyte homing and proliferation. Immunity. 1998;9:669–76. doi: 10.1016/s1074-7613(00)80664-0. [DOI] [PubMed] [Google Scholar]
- 4.Geller MA, Cooley S, Judson PL, et al. A phase II study of allogeneic natural killer cell therapy to treat patients with recurrent ovarian and breast cancer. Cytotherapy. 2011;13:98–107. doi: 10.3109/14653249.2010.515582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cooley S, He F, Bachanova V, et al. First-in-human trial of rhIL-15 and haploidentical natural killer cell therapy for advanced acute myeloid leukemia. Blood Adv. 2019;3:1970–80. doi: 10.1182/bloodadvances.2018028332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Burns LJ, Weisdorf DJ, DeFor TE, et al. IL-2-based immunotherapy after autologous transplantation for lymphoma and breast cancer induces immune activation and cytokine release: a phase I/II trial. Bone Marrow Transplant. 2003;32:177–86. doi: 10.1038/sj.bmt.1704086. [DOI] [PubMed] [Google Scholar]
- 7.Berrien-Elliott MM, Becker-Hapak M, Cashen AF, et al. Systemic IL-15 promotes allogeneic cell rejection in patients treated with natural killer cell adoptive therapy. Blood. 2022;139:1177–83. doi: 10.1182/blood.2021011532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liu E, Tong Y, Dotti G, et al. Cord blood NK cells engineered to express IL-15 and a CD19-targeted CAR show long-term persistence and potent antitumor activity. Leukemia. 2018;32:520–31. doi: 10.1038/leu.2017.226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shanley M, Daher M, Dou J, et al. Interleukin-21 engineering enhances NK cell activity against glioblastoma via CEBPD. Cancer Cell. 2024;42:1450–66. doi: 10.1016/j.ccell.2024.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rafei M, Wu JH, Annabi B, et al. A GMCSF and IL-15 fusokine leads to paradoxical immunosuppression in vivo via asymmetrical JAK/STAT signaling through the IL-15 receptor complex. Blood. 2007;109:2234–42. doi: 10.1182/blood-2006-07-037473. [DOI] [PubMed] [Google Scholar]
- 11.Gotthardt D, Putz EM, Grundschober E, et al. STAT5 Is a Key Regulator in NK Cells and Acts as a Molecular Switch from Tumor Surveillance to Tumor Promotion. Cancer Discov. 2016;6:414–29. doi: 10.1158/2159-8290.CD-15-0732. [DOI] [PubMed] [Google Scholar]
- 12.Eckelhart E, Warsch W, Zebedin E, et al. A novel Ncr1-Cre mouse reveals the essential role of STAT5 for NK-cell survival and development. Blood. 2011;117:1565–73. doi: 10.1182/blood-2010-06-291633. [DOI] [PubMed] [Google Scholar]
- 13.Shum T, Omer B, Tashiro H, et al. Constitutive Signaling from an Engineered IL7 Receptor Promotes Durable Tumor Elimination by Tumor-Redirected T Cells. Cancer Discov. 2017;7:1238–47. doi: 10.1158/2159-8290.CD-17-0538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Michaud A, Dardari R, Charrier E, et al. IL-7 Enhances Survival of Human CD56bright NK Cells. J Immunother. 2010;33:382–90. doi: 10.1097/CJI.0b013e3181cd872d. [DOI] [PubMed] [Google Scholar]
- 15.Pule MA, Savoldo B, Myers GD, et al. Virus-specific T cells engineered to coexpress tumor-specific receptors: persistence and antitumor activity in individuals with neuroblastoma. Nat Med. 2008;14:1264–70. doi: 10.1038/nm.1882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kelly PF, Carrington J, Nathwani A, et al. RD114‐Pseudotyped Oncoretroviral Vectors. Ann N Y Acad Sci. 2001;938:262–77. doi: 10.1111/j.1749-6632.2001.tb03596.x. [DOI] [PubMed] [Google Scholar]
- 17.Lapteva N, Parihar R, Rollins LA, et al. Large-Scale Culture and Genetic Modification of Human Natural Killer Cells for Cellular Therapy. Methods Mol Biol. 2016;1441:195–202. doi: 10.1007/978-1-4939-3684-7_16. [DOI] [PubMed] [Google Scholar]
- 18.Cichocki F, Bjordahl R, Goodridge JP, et al. Quadruple gene-engineered natural killer cells enable multi-antigen targeting for durable antitumor activity against multiple myeloma. Nat Commun. 2022;13:7341. doi: 10.1038/s41467-022-35127-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mo F, Watanabe N, McKenna MK, et al. Engineered off-the-shelf therapeutic T cells resist host immune rejection. Nat Biotechnol. 2021;39:56–63. doi: 10.1038/s41587-020-0601-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dysthe M M, Parihar R R. NCBI GEO Repository; 2024. Data from: Human Immune Exhaustion Gene Expression Profile in C7R-CAR and CAR NK cells.https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE277499 Available. [Google Scholar]
- 21.Sutlu T, Nyström S, Gilljam M, et al. Inhibition of intracellular antiviral defense mechanisms augments lentiviral transduction of human natural killer cells: implications for gene therapy. Hum Gene Ther. 2012;23:1090–100. doi: 10.1089/hum.2012.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xu Y, Sun J, Sheard MA, et al. Lenalidomide overcomes suppression of human natural killer cell anti-tumor functions by neuroblastoma microenvironment-associated IL-6 and TGFβ1. Cancer Immunol Immunother. 2013;62:1637–48. doi: 10.1007/s00262-013-1466-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wu J, Gao F, Wang C, et al. IL-6 and IL-8 secreted by tumour cells impair the function of NK cells via the STAT3 pathway in oesophageal squamous cell carcinoma. J Exp Clin Cancer Res. 2019;38 doi: 10.1186/s13046-019-1310-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chen J, Qiu S, Li W, et al. Tuning charge density of chimeric antigen receptor optimizes tonic signaling and CAR-T cell fitness. Cell Res. 2023;33:341–54. doi: 10.1038/s41422-023-00789-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kailayangiri S, Altvater B, Spurny C, et al. Targeting Ewing sarcoma with activated and GD2-specific chimeric antigen receptor-engineered human NK cells induces upregulation of immune-inhibitory HLA-G. Oncoimmunology. 2017;6:e1250050. doi: 10.1080/2162402X.2016.1250050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Marin D, Li Y, Basar R, et al. Safety, efficacy and determinants of response of allogeneic CD19-specific CAR-NK cells in CD19+ B cell tumors: a phase 1/2 trial. Nat Med. 2024;30:772–84.:38238616. doi: 10.1038/s41591-023-02785-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yu W, He J, Wang F, et al. NR4A1 mediates NK-cell dysfunction in hepatocellular carcinoma via the IFN-γ/p-STAT1/IRF1 pathway. Immunology. 2023;169:69–82. doi: 10.1111/imm.13611. [DOI] [PubMed] [Google Scholar]
- 28.Weisshaar N, Wu J, Ming Y. Rgs16 promotes antitumor CD8(+) T cell exhaustion. Sci Immunol. 2022 doi: 10.1126/sciimmunol.abh1873. [DOI] [PubMed] [Google Scholar]
- 29.Netskar H, Pfefferle A, Goodridge JP, et al. Pan-cancer profiling of tumor-infiltrating natural killer cells through transcriptional reference mapping. Nat Immunol. 2024;25:1445–59. doi: 10.1038/s41590-024-01884-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shlomovitz I, Speir M, Gerlic M. Flipping the dogma - phosphatidylserine in non-apoptotic cell death. Cell Commun Signal. 2019;17:139. doi: 10.1186/s12964-019-0437-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gotthardt D, Sexl V. STATs in NK-Cells: The Good, the Bad, and the Ugly. Front Immunol. 2016;7:694. doi: 10.3389/fimmu.2016.00694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.ElKassar N, Gress RE. An overview of IL-7 biology and its use in immunotherapy. J Immunotoxicol. 2010;7:1–7. doi: 10.3109/15476910903453296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tan JT, Dudl E, LeRoy E, et al. IL-7 is critical for homeostatic proliferation and survival of naive T cells. Proc Natl Acad Sci U S A. 2001;98:8732–7. doi: 10.1073/pnas.161126098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kieper WC, Tan JT, Bondi-Boyd B, et al. Overexpression of interleukin (IL)-7 leads to IL-15-independent generation of memory phenotype CD8+ T cells. J Exp Med. 2002;195:1533–9. doi: 10.1084/jem.20020067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kim MY, Jayasinghe R, Devenport JM, et al. A long-acting interleukin-7, rhIL-7-hyFc, enhances CAR T cell expansion, persistence, and anti-tumor activity. Nat Commun. 2022;13:3296. doi: 10.1038/s41467-022-30860-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lei W, Zhao A, Liu H, et al. Safety and feasibility of anti-CD19 CAR T cells expressing inducible IL-7 and CCL19 in patients with relapsed or refractory large B-cell lymphoma. Cell Discov. 2024;10:5. doi: 10.1038/s41421-023-00625-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tang F, Li J, Qi L, et al. A pan-cancer single-cell panorama of human natural killer cells. Cell. 2023;186:4235–51. doi: 10.1016/j.cell.2023.07.034. [DOI] [PubMed] [Google Scholar]
- 38.Decot V, Voillard L, Latger-Cannard V, et al. Natural-killer cell amplification for adoptive leukemia relapse immunotherapy: comparison of three cytokines, IL-2, IL-15, or IL-7 and impact on NKG2D, KIR2DL1, and KIR2DL2 expression. Exp Hematol. 2010;38:351–62. doi: 10.1016/j.exphem.2010.02.006. [DOI] [PubMed] [Google Scholar]
- 39.Wang X, Zhao XY. Transcription Factors Associated With IL-15 Cytokine Signaling During NK Cell Development. Front Immunol. 2021 doi: 10.3389/fimmu.2021.610789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Smith SL, Kennedy PR, Stacey KB, et al. Diversity of peripheral blood human NK cells identified by single-cell RNA sequencing. Blood Adv. 2020;4:1388–406. doi: 10.1182/bloodadvances.2019000699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rebuffet L, Melsen JE, Escalière B, et al. High-dimensional single-cell analysis of human natural killer cell heterogeneity. Nat Immunol. 2024;25:1474–88. doi: 10.1038/s41590-024-01883-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dean I, Lee CYC, Tuong ZK, et al. Rapid functional impairment of natural killer cells following tumor entry limits anti-tumor immunity. Nat Commun. 2024;15:683. doi: 10.1038/s41467-024-44789-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Gill S, Vasey AE, De Souza A, et al. Rapid development of exhaustion and down-regulation of eomesodermin limit the antitumor activity of adoptively transferred murine natural killer cells. Blood. 2012;119:5758–68. doi: 10.1182/blood-2012-03-415364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Liu X, Wang Y, Lu H, et al. Genome-wide analysis identifies NR4A1 as a key mediator of T cell dysfunction. Nature New Biol. 2019;567:525–9. doi: 10.1038/s41586-019-0979-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sullivan MR, Finocchiaro M, Yang Y. An innovative single-cell approach for phenotyping and functional genotyping of CAR NK cells. J Immunother Cancer. 2014 doi: 10.1136/jitc-2024-008912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Valeri A, García-Ortiz A, Castellano E, et al. Overcoming tumor resistance mechanisms in CAR-NK cell therapy. Front Immunol. 2022;13:953849. doi: 10.3389/fimmu.2022.953849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bou-Tayeh B, Laletin V, Salem N, et al. Chronic IL-15 Stimulation and Impaired mTOR Signaling and Metabolism in Natural Killer Cells During Acute Myeloid Leukemia. Front Immunol. 2021;12:730970. doi: 10.3389/fimmu.2021.730970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Felices M, Lenvik AJ, McElmurry R, et al. Continuous treatment with IL-15 exhausts human NK cells via a metabolic defect. JCI Insight. 2018;3:e96219. doi: 10.1172/jci.insight.96219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Odagiu L, May J, Boulet S, et al. Role of the Orphan Nuclear Receptor NR4A Family in T-Cell Biology. Front Endocrinol (Lausanne) 2020;11:624122. doi: 10.3389/fendo.2020.624122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nakagawara K, Ando M, Srirat T, et al. NR4A ablation improves mitochondrial fitness for long persistence in human CAR-T cells against solid tumors. J Immunother Cancer. 2024;12:e008665. doi: 10.1136/jitc-2023-008665. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data are available in a public, open access repository. All data relevant to the study are included in the article or uploaded as supplementary information.