Abstract
NHE3−/− mice display a profound defect in proximal tubule bicarbonate reabsorption but are only mildly acidotic owing to reduced glomerular filtration rate and enhanced H+ secretion in distal nephron segments. In vivo microperfusion of rat distal tubules suggests that a significant fraction of bicarbonate reabsorption in this nephron segment is mediated by NHE2. Two approaches were used to evaluate the role of distal tubule NHE2 in compensating for the proximal defect of H+ secretion in NHE3−/− mice. First, renal clearance experiments were used to assess the impact of HOE694, an inhibitor with significant affinity for NHE2, on excretion of bicarbonate in NHE3−/− and NHE2−/− mice. Second, in vivo micropuncture and microperfusion were employed to measure the concentration of bicarbonate in early distal tubule fluid and to measure distal bicarbonate reabsorption during a constant bicarbonate load. Our data show that HOE694 had no effect on urinary bicarbonate excretion in NHE3+/+ mice, whereas bicarbonate excretion was higher in NHE3−/− mice receiving HOE694. HOE694 induced a significant increase in bicarbonate excretion in mice given an acute bicarbonate load, but there was no effect during metabolic acidosis. Bicarbonate excretion was not affected by HOE694 in bicarbonate-loaded NHE2−/− mice. In vivo micropuncture revealed that early distal bicarbonate concentration was elevated in both bicarbonate-loaded and NHE3−/− mice. Further, microperfusion experiments showed that HOE694-sensitive bicarbonate reabsorption capacity was higher in acidotic and NHE3 null animals. We conclude that NHE2 contributes importantly to acidification in the distal tubule, and that it plays a major role in limiting urinary bicarbonate losses in states in which a high luminal bicarbonate load is presented to the distal tubule, such as in NHE3 null mice.
Several isoforms of the sodium–hydrogen exchanger (NHE) have been identified in the mammalian kidney (Biemesderfer et al. 1992, 1997; Amemiya et al. 1995; Chambrey et al. 1998; Peti-Peterdi et al. 2000; Goyal et al. 2002). NHE3 is recognized as the predominant NHE in the apical membrane of the proximal tubule and thick ascending limb of Henle (TAL), accounting for the major part of bicarbonate reabsorption in these segments (Capasso et al. 1991; Wang et al. 2001). The distal tubule reabsorbs between 5 and 8% of the filtered bicarbonate load (Gottschalk et al. 1960; Malnic et al. 1972; Capasso et al. 1987), and in vivo microperfusion studies suggest that about half of this is mediated by sodium–hydrogen exchange located in the early distal tubule (Wang et al. 1993). Immunohistochemical studies identify NHE2 in the apical membrane of the rat distal convoluted tubule and connecting tubule (Chambrey et al. 1998). Studies using HOE694, an agent that inhibits NHE2 but less so NHE3 (Counillon et al. 1993), confirm functional expression of NHE2 in the rat distal tubule (Wang et al. 2001). Proximal tubule reabsorption of bicarbonate and fluid is reduced by ∼60% in mice with a non-functional mutation of NHE3 (Schultheis et al. 1998b; Wang et al. 1999). In absolute terms, however, fluid (and by inference, sodium) delivery into and out of the loop of Henle is similar in NHE3 null and wild-type mice (Lorenz et al. 1999). This reflects a compensatory response involving reduction in single-nephron glomerular filtration rate mediated by activation of tubulo-glomerular feedback.
Bicarbonate excretion has not been measured in NHE3−/− mice. We hypothesize that compromised reabsorption in both the proximal tubule and loop of Henle results in delivery of bicarbonate-rich fluid into the early distal tubule. Since bicarbonate reabsorption in the distal tubule is load dependent (Capasso et al. 1987), manoeuvres that increase delivery, such as bicarbonate loading (Malnic et al. 1972; Capasso et al. 1987) and administration of carbonic anhydrase inhibitors (Vieira & Malnic, 1968; Malnic et al. 1972; Capasso et al. 1987), promote increased distal reabsorption. Enhancement of the activity of H+-ATPase and H+,K+-ATPase (Nakamura et al. 1999) in isolated collecting tubules of NHE3−/− mice removed from the animal is consistent with increased intrinsic capacity for H+ ion secretion possibly as a consequence of metabolic acidosis. However, the possible adaptive response of the distal convoluted tubule in NHE3−/− mice has not yet been explored.
In the present study we measured the effect of HOE694, an inhibitor with strong specificity for NHE2-mediated H+ ion secretion, on renal bicarbonate excretion. We find that NHE2 contributes importantly to acidification in the distal tubule, that it is up-regulated by metabolic acidosis and in NHE3 null mice, and that it has a major role in limiting urinary bicarbonate excretion in states where distal bicarbonate delivery is increased above normal.
Methods
Animals
Experiments were performed on three groups of animals. Mice with a null mutation in NHE3 were bred locally, as previously described (Schultheis et al. 1998b). Wild-type littermates (Black Swiss background) were used as controls. NHE2 null mice (Schultheis et al. 1998a) and wild-type littermates (Non-Swiss Albino background) were provided by Dr G. E. Shull (Molecular Genetics, University of Cincinnati). The chronic acidosis experiments and those involving bicarbonate loading were performed on C57/Bl6 mice (The Jackson Laboratory, Bar Harbour, ME, USA). Most mice were maintained on a regular diet and tap water ad libitum until the day of the experiment. Metabolic acidosis in mice was induced by giving drinking water containing 1.5% ammonium chloride and 5% sucrose for 10 days prior to experimentation. All data reported were obtained under anaesthesia and animals were killed by an overdose of anaesthetic in accordance with Yale University guidelines.
Surgical protocols
The methods for preparing mice for renal clearance measurements, for monitoring mean arterial blood pressure (MABP), and for collecting urine samples have been previously described (Wang et al. 1999). Mice were anaesthetized by intraperitoneal injection of [5-ethyl-5-(l-methylpropyl)-2-thiobarbituric acid (Inactin, 100 mg kg−1; Sigma, USA). A tracheotomy was performed and the jugular vein and carotid artery were catheterized for intravenous infusion and blood sampling. Following surgery, a saline solution (see below) was infused at 0.5 ml h−1. For the clearance experiments only, [3H]inulin was included in the infusate (10 μCi ml−1; 10 μCi primer). For the microperfusion experiments, the left kidney was exposed by flank incision and immobilized in a Perspex cup clamped to the operating table. During this period, mice were given an additional bolus of saline to compensate for surgical losses.
Renal clearance infusion protocol
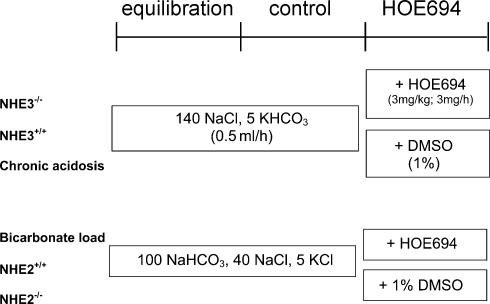
Throughout the experiment, each animal received an intravenous infusion of 140 mm NaCl and 5 mm KHCO3. The acute increase of bicarbonate load was induced by i.v. infusion of 100 mm NaHCO3 with 40 mm NaCl and 5 mm KCl. These protocols are shown in Fig. 1. After an equilibration period, renal clearance measurements were initiated for a 60-min control period and a 60-min experimental period during which the animals received either HOE694 (3 mg kg−1 bolus, 3 mg kg−1 h−1 infusion) or the vehicle alone (time control: 1% DMSO in saline). Urine was collected under mineral oil and a 30 μl blood sample was taken at hourly intervals. At the start and end of the experiment a 40 μl blood sample was taken for blood-gas analysis. Blood pressure was measured at the beginning, middle and end of each clearance period.
Figure 1. Infusion protocols for renal clearance studies.
All concentrations expressed in mmol l−1. Infusions were maintained for 3 h. After a 60 min equilibration period, baseline measurements were made during the control period followed by infusion of either the NHE2 inhibitor HOE694 or the time control vehicle (1% DMSO) during the experimental period.
Microperfusion infusion protocol
Throughout the experiment, each animal received an intravenous infusion of 140 mm NaCl and 5 mm KHCO3 at a rate of 0.5 ml h−1. For the bicarbonate loading experiments, 100 mm NaHCO3, 40 mm NaCl and 5 mm KCl was infused at a similar rate. Mannitol (2%) was added to all infusates.
Stationary microperfusion of the distal tubule
The technique employed in these experiments has been previously described (Fernandez et al. 1994). Briefly, a proximal tubule, chosen at random, was impaled with a micropipette (perfusing) containing an FD & C green-coloured solution of 100 mm NaCl and 25 mm NaHCO3. In order to minimize fluid reabsorption, this solution had been rendered isotonic through the addition of raffinose pentahydrate (Riedel-de Haën, Hannover, Germany). A small bolus of fluid was injected into the tubule lumen to identify downstream loops of the proximal and early distal tubule. An H+-selective microelectrode (see below) was lowered onto the kidney surface for zero voltage measurement before being inserted into the tubule lumen of the early distal tubule to obtain free-flow recordings of pH. A second (blocking) micropipette containing Sudan black-coloured heavy mineral oil was then inserted into a proximal tubular segment and the original perfusion pipette moved to the last superficial segment of the proximal tubule.
For stationary measurements of tubular H+ secretion, the control solution was injected into the late proximal tubule thereby perfusing orthogradely the loop of Henle and distal tubule. Perfusion was continued until the early distal microelectrode recorded a large and stable voltage deflection equal to that of the perfusate pH (pH 8.0). At this point, perfusion was stopped and a column of heavy oil injected into the proximal tubule to prevent downstream flow of native tubule fluid or perfusate. Voltage recordings were continued until a stable reading was once again obtained. Following control measurements, the first perfusion pipette was replaced by a second containing HOE694 (100 μm) and again recordings were made. The average from three measurements each of control and HOE694 perfusions was taken to generate one value per tubule thereby allowing for paired comparisons to be made.
Ion-selective microelectrodes
Double-barrelled microelectrodes were pulled and treated as previously described (Bailey, 2004). The mouse was electrically grounded through the tail. The tip of the ion-selective barrel was filled with H+ ionophore (cocktail B; Fluka, Buchs, Switzerland) and the electrode was calibrated before and after impalement by superfusion onto the kidney surface of standard solutions kept at 37°C. Standards of pH 6.0, 7.0 and 8.0 were used to measure the mean voltage difference per 10-fold change in H+ activity. This was 47.6 ± 0.8 mV (n = 39 electrodes). The reference barrel was used to measure transepithelial potential difference. The voltages from each barrel were measured by a high-impedance electrometer (Model 223, World Precision Instruments, USA). An AD converter (model 104, iWorx, USA), sampling at a rate of 5 Hz, allowed voltages to be recorded onto a PC using LabScribe software (iWorx, USA).
The voltage difference between the reference and ion-selective barrels, representing lumen H+ activity, was converted to pH, and bicarbonate concentration was calculated from this value and from measured arterial PCO2. For each perfusion, pH fell from the initial value of 8, to a stationary value of ∼6.6. A one-phase exponential was fitted to this fall, allowing the half-time (t½) of acidification to be generated. Bicarbonate reabsorption (JHCO3) was then calculated using the following equation (Fernandez et al. 1994):
 |
where JHCO3 is in nmol cm−2 s−1, t½ is the acidification half-time, initial [HCO3−] is 25 mmol l−1, stationary [HCO3−] is calculated from pHstat and arterial PCO2, and r is the lumen radius (μm).
Analysis
Blood gas analysis was performed on freshly drawn arterial blood using a Corning Blood Gas Analyser. Glomerular filtration rate (GFR) was estimated from the clearance of [3H]inulin, measured in plasma and urine samples by β-emission spectroscopy following dispersal in scintillation cocktail. Sodium and potassium concentrations in plasma and urine were measured by flame photometry (Model 480, Corning, USA). Bicarbonate concentration in urine was measured by a microcalorimetric method (picapnotherm).
Statistics
All data are expressed as the mean ± s.e.m. For the renal clearance studies, Student's t test was used for comparisons between groups. For multiple comparisons a two-way ANOVA was used with the Bonferroni post hoc test for point-to-point comparisons. For the micropuncture data, a paired t test was used to assess the impact of HOE694 on bicarbonate reabsorption within a given group. For comparisons between groups, the non-parametric Kruskal–Wallis test was used since Bartlett's test indicated unequal variances; Dunn's multiple comparison test was used for post hoc analysis. Differences were considered statistically significant at P < 0.05.
Results
Blood electrolytes and kidney function
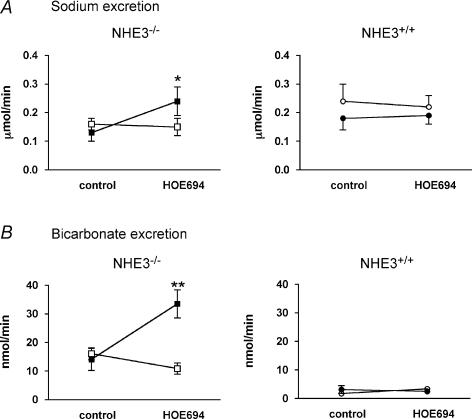
Blood-gas analysis, summarized in Table 1, indicates that NHE3−/− mice have a moderate metabolic acidosis with a reduction in arterial blood bicarbonate concentration and pH, confirming previous results (Schultheis et al. 1998b; Lorenz et al. 1999; Wang et al. 1999; Brooks et al. 2001). GFR and mean arterial blood pressure were significantly lower in NHE3−/− mice than in control mice (Table 1), the latter suggesting volume depletion. The reduction in GFR reduced the filtered sodium load in NHE3−/− mice (44.3 ± 0.3 versus 28.6 ± 2.9 mmol min−1; P < 0.05). However, sodium excretion was significantly lower (P < 0.05) in these animals (Fig. 2A) and fractional sodium excretion was thus similar in both groups (0.54 ± 0.10 versus 0.51 ± 0.09%; NS). In contrast, despite the reduction in the filtered bicarbonate load in NHE3−/− mice (6.6 ± 0.4 versus 3.5 ± 0.4 mmol min−1; P < 0.05), the absolute excretion of bicarbonate was approximately 8 times higher (P < 0.01) than in controls (Fig. 2B); ∼0.6% of the filtered bicarbonate load thus escapes reabsorption. The rate of potassium excretion was similar in both groups of mice resulting in a significantly greater fractional excretion in NHE3−/− than in control mice (17.1 ± 2.5 versus 35.8 ± 8.6%; P < 0.05).
Table 1.
Plasma data, mean arterial blood pressure (MABP) and glomerular filtration rate (GFR) in NHE3+/+ (n = 12) and NHE3−/− (n = 13) mice
| NHE3+/+ | P | NHE3−/− | |
|---|---|---|---|
| PNa (mmol l−1) | 147.2 ± 1.5 | NS | 148.6 ± 1.8 |
| PK (mmol l−1) | 4.54 ± 0.14 | NS | 4.52 ± 0.22 |
| PHCO3 (mmol l−1) | 22.4 ± 0.7 | < 0.05 | 18.9 ± 1.3 |
| pH | 7.36 ± 0.01 | < 0.05 | 7.28 ± 0.02 |
| MABP (mmHg) | 108 ± 3 | < 0.01 | 97 ± 2 |
| GFR (ml min−1 (100 g)−1) | 1.05 ± 0.04 | < 0.01 | 0.62 ± 0.06 |
Data are means ± s.e.m. Statistical comparisons were made using Student's t test.
Figure 2. Urinary sodium excretion (A) and bicarbonate excretion (B) in NHE3−/− (squares, n = 13) and NHE3+/+ mice (circles, n = 12) during infusion of either the NHE2 inhibitor HOE694 (filled symbols) or vehicle (open symbols).
The drug had no significant effect in NHE3+/+ mice. Two-way ANOVA indicated a significant effect of HOE694 for both sodium and bicarbonate excretion in NHE3−/− mice (see text). Point-to-point comparisons were made using Bonferroni's post hoc test; *P < 0.05, **P < 0.01.
Effect of i.v. HOE694
Systemic administration of HOE694 affected neither MABP nor GFR in NHE3−/− or NHE3+/+ mice (Table 2A). In NHE3+/+ animals, HOE694 was without effect on sodium or bicarbonate reabsorption (Fig. 2A and B). This indicates that under normal physiological conditions, HOE-sensitive, NHE2-mediated hydrogen ion secretion is either negligible or can be effectively compensated by other mechanisms of distal tubule H+ secretion, such as electrogenic H+ ion transport and/or H+,K+-ATPase. As shown in Fig. 2B, HOE694 caused a significant increase in the renal excretion of both bicarbonate (P < 0.01) and sodium (P < 0.05) in NHE3−/− mice. These results suggest that NHE2-mediated H+ ion secretion contributes importantly to bicarbonate reabsorption following loss of functional activity of NHE3 in the proximal tubule and TAL.
Table 2.
Mean arterial blood pressure (MABP) and glomerular filtration rate (GFR) in NHE3+/+ and NHE3−/− mice during the control period and during a 60-min period of infusion with either HOE694 (3 mg kg−1 h−1) or vehicle (time control 1% DMSO)
| Control | HOE694 | |
|---|---|---|
| A. MABP (mmHg) | ||
| NHE+/+ Time control | 107 ± 3 | 108 ± 3 |
| NHE+/+ HOE694 | 110 ± 5 | 112 ± 6 |
| NHE−/− Time control | 99 ± 4 | 103 ± 2 |
| NHE−/− HOE694 | 96 ± 4 | 97 ± 3 |
| B. GFR (ml min−1 (100 g)−1) | ||
| NHE+/+ time control | 1.10 ± 0.09 | 1.16 ± 0.09 |
| NHE+/+ HOE694 | 1.00 ± 0.04 | 0.92 ± 0.11 |
| NHE−/− time control | 0.65 ± 0.10 | 0.59 ± 0.09 |
| NHE−/− HOE694 | 0.59 ± 0.10 | 0.46 ± 0.09 |
Data are means ± s.e.m. Statistical comparisons were made using ANOVA.
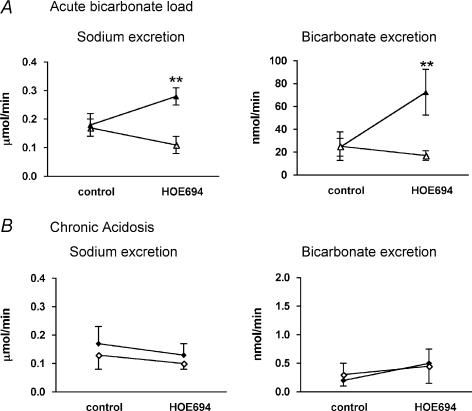
NHE3 null mice differ from wild-type mice in having high distal delivery of bicarbonate as well as metabolic acidosis. We therefore tested whether these parameters would alter the effects of HOE694 in wild-type mice. Mice were either acutely loaded with bicarbonate (blood [HCO3−] = 30.1 ± 2.6 mmol l−1; n= 14) or rendered chronically acidotic (blood [HCO3−]= 16.9 ± 0.7 mmol l−1; n = 8). Inspection of Fig. 3A shows that HOE694 increased the excretion of sodium bicarbonate in those animals receiving an infusion of sodium bicarbonate. In acidotic mice, bicarbonate excretion was minimal and not significantly different from zero. Thus, HOE694 was without effect on sodium bicarbonate excretion in wild-type mice with acidosis.
Figure 3. Sodium and bicarbonate excretion in bicarbonate-loaded mice (A) (triangles; n = 7 in each group) and acidotic mice (B) (diamonds; n = 5 for HOE694 and 4 for time controls) during infusion of either the NHE2 inhibitor HOE694 (filled symbols) or vehicle (open symbols).
Two-way ANOVA indicated a significant effect of HOE694 on both variables in bicarbonate-loaded mice only (see text). Point-to-point comparisons were made using Bonferroni's post hoc test; **P < 0.01.
In the present study, a total of 0.2 mg HOE694 was administered intravenously. Assuming free distribution in total body water, a blood concentration of about 30 μmol l−1 would be expected. It is reasonable to assume that NHE2 would be inhibited extensively under this condition (Ki= 5μmol l−1), whereas the effect on NHE3 would be small (Ki= 650 μmol l−1) (Counillon et al. 1993). However, the possibility must be considered that NHE1 (Ki= 0.16 μmol l−1) also contributes to the renal effects of HOE694. It would be expected that inhibition by HOE694 of NHE1, which is present in the basolateral membrane of several nephron segments (Biemesderfer et al. 1992), should acidify the cytosol and enhance bicarbonate reabsorption. In the rat TAL, however, inhibition of basolateral NHE1 inhibits HCO3− reabsorption (Good et al. 1995). Moreover, reduced bicarbonate reabsorption is observed in the TAL of NHE1 null mice (Good et al. 2004). Accordingly, it is possible that the observed inhibitory effects of HOE694 include a significant contribution of basolateral effects on NHE1-mediated Na+–H+ exchange.
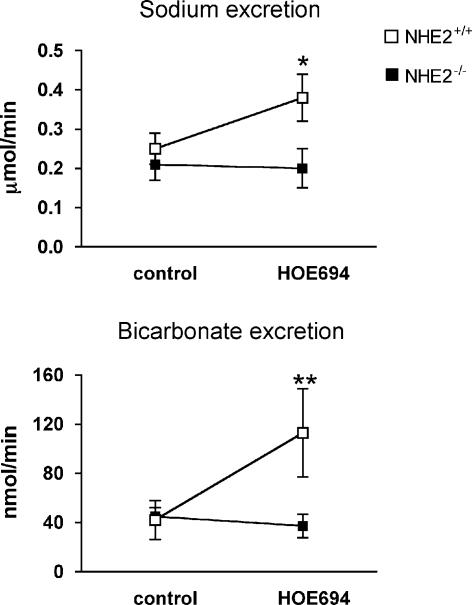
In order to further demonstrate that the effects of HOE694 reported above were due to inhibition of NHE2, the effect of the drug on sodium and bicarbonate excretion was also assessed in NHE2−/− mice. Inspection of Fig. 4 indicates that sodium and bicarbonate excretion rates in bicarbonate-loaded wild-type mice were again sharply increased by HOE694 (Fig. 4), as previously shown in Fig. 3. Such an effect, however, was totally absent in NHE2 null mice. These findings verify that the main effect of HOE694 under the conditions of our experiments is to inhibit NHE2-mediated H+ secretion and bicarbonate reabsorption.
Figure 4. Sodium and bicarbonate excretion in bicarbonate-loaded NHE2+/+ mice (open squares, n = 5) and NHE2−/− mice (filled squares, n = 6) during infusion of the NHE2 inhibitor HOE694.
Point-to-point comparisons between groups were made using Bonferroni's post hoc test; *P < 0.05, **P < 0.01.
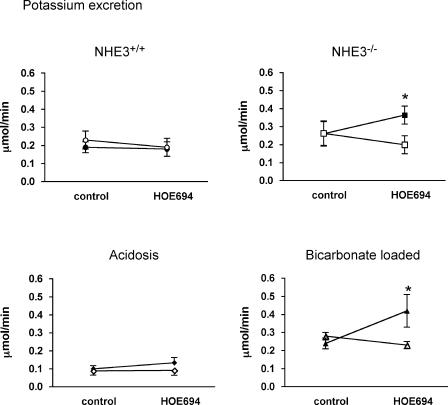
Urinary potassium excretion was affected in a predictable manner by acid–base perturbations, being suppressed by acidosis and stimulated by alkalosis in wild-type mice (Fig. 5) (Giebisch, 1998). As also shown in Fig. 5, under conditions in which HOE694 had been shown to increase sodium and bicarbonate excretion, namely in NHE3 null mice or with bicarbonate loading in wild-type mice, HOE694 also increased potassium excretion.
Figure 5. The effect of HOE694 on renal potassium excretion in four groups of mice. Infusion of the NHE2 inhibitor (black symbols) increased potassium excretion compared to time controls (open symbols) in NHE3−/− mice and bicarbonate-loaded animals.
Point-to-point comparisons were made using Bonferroni's post hoc test; *P < 0.05. Data are means ± s.e.m.
Free-flow bicarbonate concentration in the early distal tubule
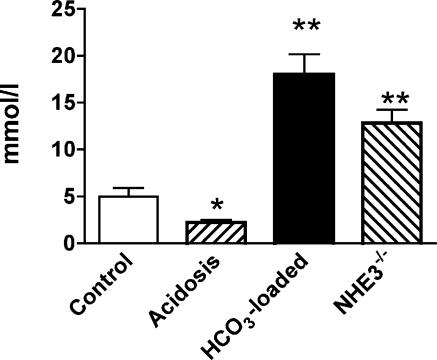
The free-flow transepithelial potential differences in the four groups of mice are summarized in Table 3. These values, being in the range of −10 mV, confirm the positioning of the electrode in the early to mid, rather than late, distal tubule (De Mello-Aires & Malnic, 2002). The bicarbonate concentrations in early distal tubule fluid, estimated from tubule fluid pH and arterial PCO2, are summarized in Fig. 6. In control animals, the mean bicarbonate concentration was ∼5 mmol l−1, similar to that obtained from the rat using direct measurement of tubule fluid bicarbonate (Capasso et al. 1986). Chronic acidosis reduced early distal bicarbonate concentration by ∼50% (P < 0.05), whereas acute bicarbonate loading elicited a 3-fold increase (P < 0.01) in distal bicarbonate concentration. In NHE3−/− mice, tubule fluid bicarbonate concentration was significantly elevated, ∼13 mmol l−1, supporting the interpretation that impaired reabsorption in the proximal tubule and thick ascending limb of Henle leads to delivery of a bicarbonate-rich fluid into the distal nephron.
Table 3.
Transepithelial potential difference (Vte), measured under free flow conditions, in NHE3+/+ (n = 5), NHE3−/− (n = 2), chronically acidotic (n = 7) and acutely bicarbonate loaded (n = 3) C57/Bl6 mice
| Vte (mV) | n (tubules) | |
|---|---|---|
| NHE3+/+ | −10.7 ± 0.8 | 18 |
| NHE3−/− | −8.9 ± 1.2 | 9 |
| Acidotic | −10.3 ± 1.4 | 8 |
| Bicarbonate-loaded | −10.3 ± 1.5 | 14 |
Data are means ± s.e.m. of individual tubules. Statistical comparisons indicated no difference between the groups (Kruskal–Wallis test).
Figure 6. Bicarbonate concentration in the early distal tubule of control (n = 18 tubules/5 mice), chronically acidotic mice (n = 14/7), bicarbonate loaded (n = 8/3) or NHE3−/− (n = 9/2) mice.
The Kruskal–Wallis test indicated a significant difference between the 4 groups (P < 0.001). Point-to-point comparisons were made using Dunn's post hoc test; *P < 0.05, **P < 0.01 versus control. Data are means ± s.e.m.
Bicarbonate reabsorption in the early distal tubule
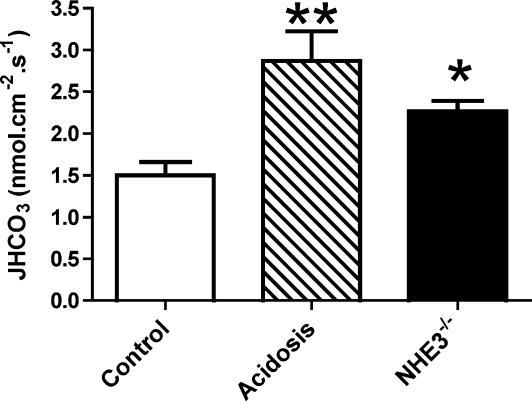
H+-selective microelectrodes were used to measure HCO3− reabsorption in the early distal tubule of control, acidotic and NHE3 null mice. Since the concentration of HCO3− perfused into the tubule was the same in all groups of mice, this technique allowed the evaluation of the reabsorptive capacity of the distal tubule independent of variations in the luminal bicarbonate load. Inspection of Fig. 7 shows that JHCO3 was different in the three groups of mice (P < 0.01). Specifically, JHCO3 was increased by ∼90% in the acidotic mice (P < 0.01) and increased by ∼50% (P < 0.05) in NHE3 null mice. These findings thus indicate that there is up-regulation of distal tubule H+ secretion in NHE3 null mice, as in metabolic acidosis.
Figure 7. Bicarbonate reabsorption during perfusion of the early distal tubule with a control perfusate (25 mm HCO3) in either control (n = 11 tubules/5 mice), chronically acidotic (n = 14 /7) or NHE3−/− (n = 6/2) mice.
The Kruskal–Wallis test indicated a significant difference between the 3 groups (P < 0.01). Point-to-point comparisons were made using Dunn's post hoc test; *P < 0.05, **P < 0.01versus control. Data are means ± s.e.m.
Impact of HOE694 on JHCO3 in the early distal tubule
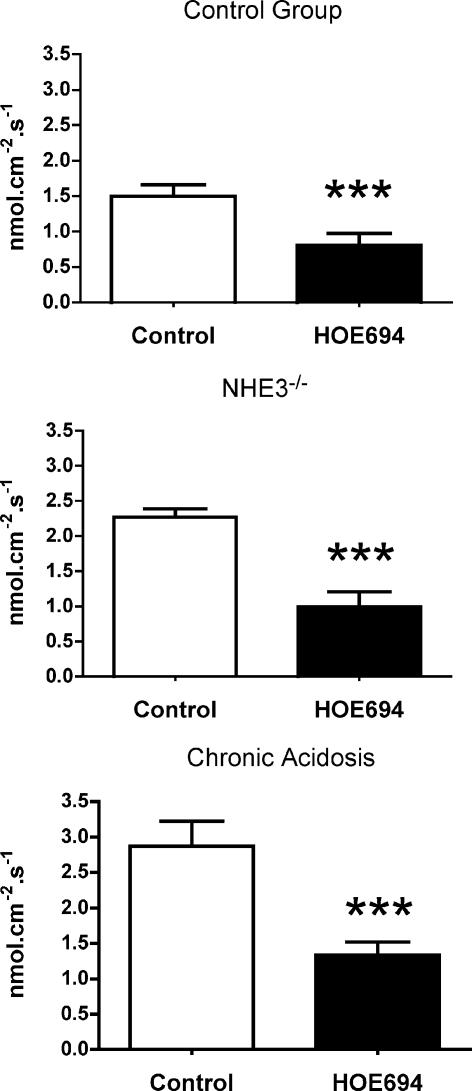
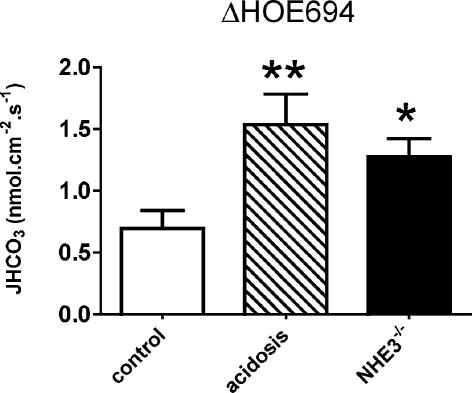
As shown in Fig. 8, HOE694 significantly reduced JHCO3 in the three groups of mice. The net effect of this inhibitor, shown in Fig. 9, was significantly greater in both acidotic (P < 0.01) and NHE3 null mice (P < 0.05) than in control animals. These findings demonstrate that administering bicarbonate directly into the distal tubule induces appreciable HOE694-sensitive, NHE2-mediated bicarbonate absorption even under control conditions when an effect of the drug on urinary sodium and bicarbonate excretion in the intact animal was not detected. Importantly, the capacity of the distal tubule for HOE694-sensitive, NHE2-mediated bicarbonate absorption was up-regulated both in NHE3 null mice and in wild-type mice with metabolic acidosis. Under the latter condition, the effect of HOE694 on urinary sodium and bicarbonate excretion was not detected, presumably because of the low distal bicarbonate load in metabolic acidosis. Taken together, these findings demonstrate that NHE2 contributes importantly to acidification in the distal tubule, that it is up-regulated by metabolic acidosis and in NHE3 null mice, but that it only affects urinary bicarbonate excretion in states where distal bicarbonate delivery is increased above normal.
Figure 8. Bicarbonate reabsorption during perfusion of the early distal tubule with a control perfusate (25 mm HCO3−) and the same solution to which HOE694 (100 μmol l−1) had been added in either control (n = 11 tubules/4 mice), chronically acidotic (n = 14/7) or NHE3−/− (n = 6/2) mice.
The drug evoked a significant inhibition of bicarbonate reabsorption in each of the 3 groups, as indicated. Comparisons were made using Student's paired t test.
Figure 9. The net effect of HOE694 (100 μmol l−1) on early distal bicarbonate reabsorption in either control (n = 11 tubules/5 mice), chronically acidotic (n = 14/7) or NHE3−/− (n = 6/2) mice.
The Kruskal–Wallis test indicated a significant difference between the 3 groups (P < 0.01). Point-to-point comparisons were made using Dunn's post hoc test; *P < 0.05, **P < 0.01 versus control. Data are means ± s.e.m.
Discussion
The main finding of the present study is the emergence of NHE2 as a mechanism of renal bicarbonate reabsorption in circumstances of elevated delivery to the early distal tubule. This is based upon the observation that systemic HOE694 produced a significant enhancement of bicarbonate excretion in (1) NHE3 null mice having sharply impaired proximal bicarbonate transport and (2) in wild-type mice receiving an acute bicarbonate load. The increased bicarbonate reabsorption capacity occurring in response to elevated bicarbonate delivery to the early distal tubule is best explained by activation of NHE2 since HOE694 was without effect in bicarbonate-loaded NHE2 null mice.
Although most of the filtered bicarbonate is reabsorbed along the proximal tubule, significant reabsorption is also observed in the thick ascending limb of Henle (Good, 1993; Capasso et al. 2002); in both sites, NHE3 is the predominant apical mechanism of H+ secretion. The superficial distal tubule reabsorbs ∼5% of the filtered bicarbonate under physiological conditions (De Mello-Aires & Malnic, 2002). Reabsorption is load dependent and rises sharply with increased bicarbonate delivery (Capasso et al. 1987; Chan et al. 1989). Both electrogenic H+ secretion and Na+–H+ exchange (Wang et al. 1993; Fernandez et al. 1994) have been shown to mediate bicarbonate transport in the distal tubule. The role of NHE2 is further underscored by immunological studies demonstrating NHE2 expression in the apical membrane of the distal tubule (Chambrey et al. 1998). In vivo microperfusion studies provided pharmacological evidence to suggest that it is this isoform, not NHE3, that mediates an important fraction of distal tubule bicarbonate reabsorption (Wang et al. 2001).
The clearance experiments reported herein suggest that NHE2-mediated bicarbonate reabsorption is, under physiological circumstances, of small magnitude. It is not surprising therefore that NHE2 knockout mice have normal acid–base status (Ledoussal et al. 2001a,b). Given that ∼50% of distal tubule bicarbonate reabsorption is mediated by NHE-dependent H+ secretion, demonstrated here in the mouse and previously in the rat (Wang et al. 1993, 2001; Fernandez et al. 1994; Wesson, 1998), the failure of HOE694 to increase bicarbonate excretion in control mice appears to be counterintuitive. This discrepancy can be accounted for on the basis of variations in bicarbonate load. In the rat, perfusion with a solution containing 25 mm bicarbonate results in a JHCO3 of ∼70 pmol min−1 mm−1 (Wang et al. 2001), about ∼50% of which is mediated by Na+–H+ exchange. With lower bicarbonate concentrations (7.5 mm), JHCO3 is 15 pmol min−1 mm−1 (Wesson, 1998), yet the proportion effected by Na+–H+ exchange remains about the same. In the present study therefore the concentration of luminal bicarbonate observed in control mice under free-flow conditions (5 mm) would limit the absolute magnitude of NHE2-mediated reabsorption and thus render a systemic effect of HOE694 too small to discern.
Systemic administration of HOE694 does, however, evoke significant bicarbonate excretion under conditions in which bicarbonate delivery to the early distal tubule (confirmed by free-flow micropuncture) was elevated, as observed in mice receiving an acute bicarbonate load and in NHE3 null mice. Taken together, our data suggest that NHE2 reclaims normally only a small proportion of the filtered bicarbonate load but is part of the compensatory up-regulation of apical transporters that guard against base loss under circumstances in which proximal reabsorption is overwhelmed or compromised.
It is of interest that we did not observe increased urinary bicarbonate excretion in acidotic animals in response to systemic HOE694. This may be taken to suggest that acidosis does not stimulate distal tubule activity of NHE2. Yet previous reports of microperfusion studies in which high bicarbonate concentrations were used suggest that distal tubule bicarbonate reabsorption rises in metabolic acidosis (Wesson, 1996, 1997). Similarly, the intrinsic reabsorptive capacity of the early distal tubule, measured in the present study during perfusion with a high (25 mm) bicarbonate solution, was almost doubled in acidotic mice compared to control conditions. Importantly, the absolute effect of luminal HOE694 was also found to be significantly greater in acidotic mice than in untreated control animals. Since our free-flow measurements show that metabolic acidosis markedly reduced the delivery of bicarbonate into the distal tubule, it is likely that limiting bicarbonate delivery under free-flow conditions countervails the stimulatory effects of metabolic acidosis on bicarbonate reabsorption in general, and NHE2 activity in particular.
Our micropuncture analysis also showed that early distal tubule bicarbonate reabsorption was higher in NHE3−/− mice, as was the absolute inhibitory effect of HOE694. Thus, it is reasonable to suggest that NHE2-mediated bicarbonate reabsorption is stimulated by the moderate systemic acidosis observed in NHE3 null mice. Moreover, impaired bicarbonate reabsorption in the proximal tubule (Schultheis et al. 1998b; Wang et al. 1999) ensures that delivery to the early distal tubule is not limiting and that an effect of HOE694 on whole-kidney bicarbonate excretion can be observed. A note of caution should be mentioned in evaluating the relative importance of compensation by NHE2 in NHE3 null mice. Studies of NHE2/NHE3 double knockout mice (Ledoussal et al. 2001b) revealed no additional disturbance of acid–base balance beyond that observed in NHE3−/− mice. In fact, conscious NHE2/NHE3 double knockouts maintained relatively normal blood pH and bicarbonate concentrations. In light of the current studies, which clearly demonstrate the loss of a significant bicarbonate absorptive capacity in the distal tubule of NHE2−/− mice, it seems likely that other renal compensatory mechanisms for maintenance of acid–base balance were activated in the double knockout. In addition to sodium–hydrogen exchange activity, both H+-ATPase and H+,K+-ATPase-mediated bicarbonate reabsorption are also augmented in outer-medullary and cortical collecting ducts harvested from NHE3−/− mice (Nakamura et al. 1999). However, as evidenced by elevated urinary bicarbonate excretion rates in NHE3−/− mice prior to HOE694 administration, the combined effect of these three acidification mechanisms does not prevent the spilling of bicarbonate in the final urine. The continued loss of bicarbonate both through renal and gastrointestinal routes (Schultheis et al. 1998b) is thus responsible for the mild metabolic acidosis in these animals.
We have confirmed effects of acid–base derangements upon potassium secretion. Thus, induction of metabolic acidosis curtailed potassium excretion and metabolic alkalosis induced kaliuresis (Giebisch, 1998). NHE3−/− mice maintained renal potassium excretion at a level comparable to that of the wild-type mice despite a substantial reduction in GFR. This dissociation of potassium excretion from filtered load has long been recognized (Berliner & Kennedy, 1948) and is a central tenet of the maintenance of potassium balance. Since sodium delivery to the early distal tubule is adequate in both groups of mice (Lorenz et al. 1999), it can be assumed that potassium secretion is elevated in the knockout animals. These data contrast with those of a previous investigation in which NHE3 null mice were hyperkalaemic and had reduced potassium excretion (Brooks et al. 2001). It is possible that the net effect on potassium in the present study may reflect the balance between countervailing parameters. Elevated aldosterone (Schultheis et al. 1998b) and increased bicarbonate to chloride ratio of tubule fluid are known to stimulate potassium secretion (Carlisle et al. 1991; Amorim et al. 2003). Metabolic acidosis and increased buffer delivery would, on the other hand, be expected to activate potassium reabsorption by H+,K+-ATPase (Silver et al. 1996; Schultheis et al. 1998b; Nakamura et al. 1999). HOE694 induced a significant kaliuresis in both NHE3 null mice and in alkalotic wild type mice. This may reflect both the diuretic effect of the drug and the inhibition of NHE2-mediated bicarbonate reabsorption in the early distal tubule: increased flow rate and luminal bicarbonate concentration stimulate potassium secretion in the distal tubule (Malnic et al. 1989; Amorim et al. 2003).
In conclusion, we find that NHE2 contributes importantly to acidification in the distal tubule, and that it plays a major role in limiting urinary bicarbonate losses in states in which a high luminal bicarbonate load is presented to the distal tubule. Up-regulation of NHE2-mediated bicarbonate reabsorption occurs in response to metabolic acidosis. Thus, up-regulated NHE2-mediated bicarbonate absorption in the distal tubule is one of the compensatory mechanisms that are activated in the kidneys of NHE3 null mice.
Acknowledgments
We would like to thank Drs Max Hropot and Hans-Jochen Lang for providing HOE694. This work was supported by The Wellcome Trust (M.B.) and by National Institutes of Health grants DK62289 (T.W.), DK17433 (G.G.), DK33793 (P.S.A.), DK50594 (G.E.S.).
References
- Amemiya M, Loffing J, Lotscher M, Kaissling B, Alpern RJ, Moe OW. Expression of NHE3 in the apical membrane of rat renal proximal tubule and thick ascending limb. Kidney Int. 1995;48:1206–1215. doi: 10.1038/ki.1995.404. [DOI] [PubMed] [Google Scholar]
- Amorim JB, Bailey MA, Musa-Aziz R, Giebisch G, Malnic G. Role of luminal anion and pH in distal tubule potassium secretion. Am J Physiol Renal Physiol. 2003;284:F381–F388. doi: 10.1152/ajprenal.00236.2002. [DOI] [PubMed] [Google Scholar]
- Bailey MA. Inhibition of bicarbonate reabsorption in the rat proximal tubule by activation of luminal P2Y1 receptors. Am J Physiol Renal Physiol. 2004;287:F789–F796. doi: 10.1152/ajprenal.00033.2004. [DOI] [PubMed] [Google Scholar]
- Berliner RW, Kennedy TJ. Renal tubular secretion of potassium in the normal dog. Proc Soc Exper Biol Medical. 1948;67:542–545. [PubMed] [Google Scholar]
- Biemesderfer D, Reilly RF, Exner M, Igarashi P, Aronson PS. Immunocytochemical characterization of Na+-H+ exchanger isoform NHE-1 in rabbit kidney. Am J Physiol. 1992;263:F833–F840. doi: 10.1152/ajprenal.1992.263.5.F833. [DOI] [PubMed] [Google Scholar]
- Biemesderfer D, Rutherford PA, Nagy T, Pizzonia JH, Abu-Alfa AK, Aronson PS. Monoclonal antibodies for high-resolution localization of NHE3 in adult and neonatal rat kidney. Am J Physiol. 1997;273:F289–F299. doi: 10.1152/ajprenal.1997.273.2.F289. [DOI] [PubMed] [Google Scholar]
- Brooks HL, Sorensen AM, Terris J, Schultheis PJ, Lorenz JN, Shull GE, Knepper MA. Profiling of renal tubule Na+ transporter abundances in NHE3 and NCC null mice using targeted proteomics. J Physiol. 2001;530:359–366. doi: 10.1111/j.1469-7793.2001.0359k.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capasso G, Jaeger P, Giebisch G, Guckian V, Malnic G. Renal bicarbonate reabsorption in the rat. II. Distal tubule load dependence and effect of hypokalemia. J Clin Invest. 1987;80:409–414. doi: 10.1172/JCI113087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capasso G, Kinne R, Malnic G, Giebisch G. Renal bicarbonate reabsorption in the rat. I. Effects of hypokalemia and carbonic anhydrase. J Clin Invest. 1986;78:1558–1567. doi: 10.1172/JCI112748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capasso G, Unwin R, Agulian S, Giebisch G. Bicarbonate transport along the loop of Henle. I. Microperfusion studies of load and inhibitor sensitivity. J Clin Invest. 1991;88:430–437. doi: 10.1172/JCI115322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capasso G, Unwin R, Rizzo M, Pica A, Giebisch G. Bicarbonate transport along the loop of Henle: molecular mechanisms and regulation. J Nephrol. 2002;15(Suppl. 5):S88–S96. [PubMed] [Google Scholar]
- Carlisle EJ, Donnelly SM, Ethier JH, Quaggin SE, Kaiser UB, Vasuvattakul S, Kamel KS, Halperin ML. Modulation of the secretion of potassium by accompanying anions in humans. Kidney Int. 1991;39:1206–1212. doi: 10.1038/ki.1991.152. [DOI] [PubMed] [Google Scholar]
- Chambrey R, Warnock DG, Podevin RA, Bruneval P, Mandet C, Belair MF, Bariety J, Paillard M. Immunolocalization of the Na+/H+ exchanger isoform NHE2 in rat kidney. Am J Physiol. 1998;275:F379–F386. doi: 10.1152/ajprenal.1998.275.3.F379. [DOI] [PubMed] [Google Scholar]
- Chan YL, Malnic G, Giebisch G. Renal bicarbonate reabsorption in the rat. III. Distal tubule perfusion study of load dependence and bicarbonate permeability. J Clin Invest. 1989;84:931–938. doi: 10.1172/JCI114255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Counillon L, Scholz W, Lang HJ, Pouyssegur J. Pharmacological characterization of stably transfected Na+/H+ antiporter isoforms using amiloride analogs and a new inhibitor exhibiting anti-ischemic properties. Mol Pharmacol. 1993;44:1041–1045. [PubMed] [Google Scholar]
- De Mello-Aires M, Malnic G. Distal tubule bicarbonate transport. J Nephrol. 2002;15(Suppl. 5):S97–S111. [PubMed] [Google Scholar]
- Fernandez R, Lopes MJ, De Lira RF, Dantas WF, Cragoe Junior EJ, Malnic G. Mechanism of acidification along cortical distal tubule of the rat. Am J Physiol. 1994;266:F218–F226. doi: 10.1152/ajprenal.1994.266.2.F218. [DOI] [PubMed] [Google Scholar]
- Giebisch G. Renal potassium transport: mechanisms and regulation. Am J Physiol. 1998;274:F817–F833. doi: 10.1152/ajprenal.1998.274.5.F817. [DOI] [PubMed] [Google Scholar]
- Good DW. The thick ascending limb as a site of renal bicarbonate reabsorption. Semin Nephrol. 1993;13:225–235. [PubMed] [Google Scholar]
- Good DW, George T, Watts BA., III Basolateral membrane Na+/H+ exchange enhances HCO3 absorption in rat medullary thick ascending limb: evidence for functional coupling between basolateral and apical membrane Na+/H+ exchangers. Proc Natl Acad Sci U S A. 1995;92:12525–12529. doi: 10.1073/pnas.92.26.12525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Good DW, Watts IB, George T, Meyer JW, Shull GE. Transepithelial HCO3 absorption is defective in renal thick ascending limbs from NHE1 Na+/H+ exchanger null mutant mice. Am J Physiol Renal Physiol. 2004;287:F1244–F1249. doi: 10.1152/ajprenal.00176.2004. 10.1152/ajprenal.00176.2004. [DOI] [PubMed] [Google Scholar]
- Gottschalk LW, Lassiter WE, Mylle M. Localization of urine acidification in the mammalian kidney. Am J Physiol. 1960;206:719–724. doi: 10.1152/ajplegacy.1960.198.3.581. [DOI] [PubMed] [Google Scholar]
- Goyal S, Vanden Heuvel G, Aronson PS. Renal expression of novel Na+-H+ exchanger isoform NHE8. Am J Physiol Renal Physiol. 2002;284:F467–F473. doi: 10.1152/ajprenal.00352.2002. [DOI] [PubMed] [Google Scholar]
- Ledoussal C, Lorenz JN, Nieman ML, Soleimani M, Schultheis PJ, Shull GE. Renal salt wasting in mice lacking NHE3 Na+/H+ exchanger but not in mice lacking NHE2. Am J Physiol Renal Physiol. 2001a;281:F718–F727. doi: 10.1152/ajprenal.2001.281.4.F718. [DOI] [PubMed] [Google Scholar]
- Ledoussal C, Woo AL, Miller ML, Shull GE. Loss of the NHE2 Na+/H+ exchanger has no apparent effect on diarrheal state of NHE3-deficient mice. Am J Physiol Gastrointest Liver Physiol. 2001b;281:G1385–G1396. doi: 10.1152/ajpgi.2001.281.6.G1385. [DOI] [PubMed] [Google Scholar]
- Lorenz JN, Schultheis PJ, Traynor T, Shull GE, Schnermann J. Micropuncture analysis of single-nephron function in NHE3-deficient mice. Am J Physiol. 1999;277:F447–F453. doi: 10.1152/ajprenal.1999.277.3.F447. [DOI] [PubMed] [Google Scholar]
- Malnic G, Berliner RW, Giebisch G. Flow dependence of K+ secretion in cortical distal tubules of the rat. Am J Physiol. 1989;256:F932–F941. doi: 10.1152/ajprenal.1989.256.5.F932. [DOI] [PubMed] [Google Scholar]
- Malnic G, De Mello Aires M, Giebisch G. Micropuncture study of renal tubular hydrogen ion transport in the rat. Am J Physiol. 1972;222:147–158. doi: 10.1152/ajplegacy.1972.222.1.147. [DOI] [PubMed] [Google Scholar]
- Nakamura S, Amlal H, Schultheis PJ, Galla JH, Shull GE, Soleimani M. HCO3 reabsorption in renal collecting duct of NHE-3-deficient mouse: a compensatory response. Am J Physiol. 1999;276:F914–F921. doi: 10.1152/ajprenal.1999.276.6.F914. [DOI] [PubMed] [Google Scholar]
- Peti-Peterdi J, Chambrey R, Bebok Z, Biemesderfer D, St John PL, Abrahamson DR, Warnock DG, Bell PD. Macula densa Na+/H+ exchange activities mediated by apical NHE2 and basolateral NHE4 isoforms. Am J Physiol Renal Physiol. 2000;278:F452–F463. doi: 10.1152/ajprenal.2000.278.3.F452. [DOI] [PubMed] [Google Scholar]
- Schultheis PJ, Clarke LL, Meneton P, Harline M, Boivin GP, Stemmermann G, Duffy JJ, Doetschman T, Miller ML, Shull GE. Targeted disruption of the murine Na+/H+ exchanger isoform 2 gene causes reduced viability of gastric parietal cells and loss of net acid secretion. J Clin Invest. 1998a;101:1243–1253. doi: 10.1172/JCI1249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultheis PJ, Clarke LL, Meneton P, Miller ML, Soleimani M, Gawenis LR, Riddle TM, Duffy JJ, Doetschman T, Wang T, Giebisch G, Aronson PS, Lorenz JN, Shull GE. Renal and intestinal absorptive defects in mice lacking the NHE3 Na+/H+ exchanger. Nat Genet. 1998b;19:282–285. doi: 10.1038/969. 10.1038/969. [DOI] [PubMed] [Google Scholar]
- Silver RB, Mennitt PA, Satlin LM. Stimulation of apical H-K-ATPase in intercalated cells of cortical collecting duct with chronic metabolic acidosis. Am J Physiol. 1996;270:F539–F547. doi: 10.1152/ajprenal.1996.270.3.F539. [DOI] [PubMed] [Google Scholar]
- Vieira FL, Malnic G. Hydrogen ion secretion by rat renal cortical tubules as studied by an antimony microelectrode. Am J Physiol. 1968;214:710–718. doi: 10.1152/ajplegacy.1968.214.4.710. [DOI] [PubMed] [Google Scholar]
- Wang T, Hropot M, Aronson PS, Giebisch G. Role of NHE isoforms in mediating bicarbonate reabsorption along the nephron. Am J Physiol Renal Physiol. 2001;281:F1117–F1122. doi: 10.1152/ajprenal.2001.281.6.F1117. [DOI] [PubMed] [Google Scholar]
- Wang T, Malnic G, Giebisch G, Chan YL. Renal bicarbonate reabsorption in the rat. IV. Bicarbonate transport mechanisms in the early and late distal tubule. J Clin Invest. 1993;91:2776–2784. doi: 10.1172/JCI116519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang T, Yang CL, Abbiati T, Schultheis PJ, Shull GE, Giebisch G, Aronson PS. Mechanism of proximal tubule bicarbonate absorption in NHE3 null mice. Am J Physiol. 1999;277:F298–F302. doi: 10.1152/ajprenal.1999.277.2.F298. [DOI] [PubMed] [Google Scholar]
- Wesson DE. Reduced bicarbonate secretion mediates increased distal tubule acidification induced by dietary acid. Am J Physiol. 1996;271:F670–F678. doi: 10.1152/ajprenal.1996.271.3.F670. [DOI] [PubMed] [Google Scholar]
- Wesson DE. Endogenous endothelins mediate increased distal tubule acidification induced by dietary acid in rats. J Clin Invest. 1997;99:2203–2211. doi: 10.1172/JCI119393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wesson DE. Na/H exchange and H-K ATPase increase distal tubule acidification in chronic alkalosis. Kidney Int. 1998;53:945–951. doi: 10.1111/j.1523-1755.1998.00838.x. 10.1046/j.1523-1755.1998.00838.x. [DOI] [PubMed] [Google Scholar]