Abstract
The purpose of the study was to determine the overall risk of a permanent stoma in patients with complicated perianal Crohn’s disease, and to identify risk factors predicting stoma carriage. A total of 102 consecutive patients presented with the first manifestation of complicated perianal Crohn’s disease in our outpatient department between 1992 and 1995. Ninety-seven patients (95%) could be followed up at a median of 16 years after first diagnosis of Crohn’s disease. Patients were sent a standardized questionnaire and patient charts were reviewed with respect to the recurrence of perianal abscesses or fistulas and surgical treatment, including fecal diversion. Factors predictive of permanent stoma carriage were determined by univariate and multivariate analysis. Thirty of 97 patients (31%) with complicated perianal Crohn’s disease eventually required a permanent stoma. The median time from first diagnosis of Crohn’s disease to permanent fecal diversion was 8.5 years (range 0–23 years). Temporary fecal diversion became necessary in 51 of 97 patients (53%), but could be successfully removed in 24 of 51 patients (47%). Increased rates of permanent fecal diversion were observed in 54% of patients with complex perianal fistulas and in 54% of patients with rectovaginal fistulas, as well as in patients that had undergone subtotal colon resection (60%), left-sided colon resection (83%), or rectal resection (92%). An increased risk for permanent stoma carriage was identified by multivariate analysis for complex perianal fistulas (odds ratio [OR] 5; 95% confidence interval [CI] 2–18), temporary fecal diversion (OR 8; 95% CI 2–35), fecal incontinence (OR 21, 95% CI 3–165), or rectal resection (OR 30; 95% CI 3–179). Local drainage, setons, and temporary stoma for deep and complicated fistulas in Crohn’s disease, followed by a rectal advancement flap, may result in closing of the stoma in 47% of the time. The risk of permanent fecal diversion was substantial in patients with complicated perianal Crohn’s disease, with patients requiring a colorectal resection or suffering from fecal incontinence carrying a particularly high risk for permanent fecal diversion. In contrast, patients with perianal Crohn’s disease who required surgery for small bowel disease or a segmental colon resection carried no risk of a permanent stoma.
Keywords: Fecal diversion, Crohn’s disease, Perianal abscesses, Fistulas
Introduction
Crohn’s disease was initially described as a nonspecific inflammatory bowel disease, affecting mainly the terminal ileum and characterized by a subacute or chronic necrotizing and cicatrizing inflammation.1 Eventually, gastrointestinal Crohn’s disease became recognized as a full-thickness disease of the gastrointestinal wall that may affect the entire gastrointestinal tract, including the perianal region.2–6
Perianal lesions are common in patients with Crohn’s disease.7–13 Clinical manifestations vary from asymptomatic skin tags to severe, debilitating perianal destruction and sepsis. Asymptomatic perianal lesions require no treatment, but because they become painful and disabling, they may require surgical treatment. Surgical management needs to be conservative and should focus on the drainage of septic sites, preserving sphincter function and palliating symptoms.10–13 Medical management has had some success in improving symptoms, but as yet, it has not been able to eliminate most perianal complaints permanently.14,15
Fecal diversion was successfully used to achieve remission in colonic Crohn’s disease. Moreover, it was utilized to allow severe perianal disease to settle, thereby avoiding proctectomy.16–19 However, restoring the intestinal passage carries the risk of recurrent perianal disease activity, possibly resulting in a decreased quality of life compared to the situation with fecal diversion.
We investigated the overall risk of a permanent stoma in patients with severe perianal Crohn’s disease and tried to identify risk factors predicting permanent stoma carriage.
Methods
Patients
A total of 102 consecutive patients with Crohn’s disease presented with the first manifestation of a perianal fistula or a perianal abscess in our outpatient department between 1992 and 1995. Patients were investigated in Trendelenburg’s position by perianal inspection, proctoscopy, rectoscopy, and rigid sigmoidoscopy. Endoanal ultrasound was performed in case of suspected perianal abscess formation, and MRI was conducted of the pelvic floor in case of complicated fistulizing disease or intrapelvic abscess formation. All patients were documented prospectively. Follow-up data of 97 patients (95%) were available by a standardized questionnaire mailed to the patients and by a standardized chart review. There were 50 female and 47 male patients (ratio 1.06:1) with a median age of 23 years (range 8–51 years). Patients were evaluated with respect to the recurrence of perianal abscesses, fistulas, or surgical treatment of Crohn’s disease over the years. The median interval between the first diagnosis of Crohn’s disease and last follow-up was 16 years (range 8–37 years). Four patients had isolated small intestinal disease, 11 patients had isolated colonic disease, and 82 patients had small intestinal and colonic disease.
The abscess location was categorized as subcutaneous, intersphincteric, deep perianal, ischiorectal, and above the pelvic floor. Abscess formations were categorized into simple (subcutaneous, intersphincteric, deep perianal, and ischiorectal, circular extension less than 90°, pelvic floor not involved) and complicated (circular extension more than 90° [horse shoe abscess] or pelvic floor involved). Fistulas were classified according to Parks et al.20 into subcutaneous, intersphincteric, extrasphincteric, transsphincteric, rectovaginal, and suprasphincteric, as described previously.21 We divided fistulas into simple fistulas (no more than two perianal openings) and complex fistulas (rectovaginal, three or more perianal openings).
A variety of factors, such as sex of the patient, perianal fistula, rectovaginal fistula, abscess formation, anal stricture, fecal incontinence, or abdominal surgery were evaluated with regard to their predictive character for permanent stoma carriage by univariate and multivariate analysis.
Statistical Analysis
Kaplan–Meier analysis using a log-rank test was used for comparing risk rates over time. Factors that might influence permanent fecal diversion were analyzed using the chi-square test. Fisher’s exact test was used for univariate analysis and multiple logistic regression and Wald’s test for multivariate analysis. Subgroup analysis was performed for rectovaginal fistulas, as present in female patients only. Data are given as numbers of cases and percentages or median and interquartile ranges. A P < 0.05 was considered as significant.
Results
Overall Risk of Permanent Fecal Diversion
Thirty of 97 patients (31%) with perianal Crohn’s disease eventually required a permanent stoma. Nineteen patients were female and 11 male (ratio 1.73:1). The median time from first diagnosis of Crohn’s disease to permanent fecal diversion was 8.5 years (range 0–23 years; Fig. 1). Increased rates of permanent fecal diversion were observed in 53% of patients with previous temporary fecal diversion, in 54% of patients with complex perianal fistulas, in 54% of female patients with rectovaginal fistulas, and in 83% of patients with fecal incontinence (Table 1). Patients who required subtotal colon resection (60%), left-sided colon resection (83%), or rectal resection (93%) needed a permanent stoma at high rates (Table 2).
Figure 1.
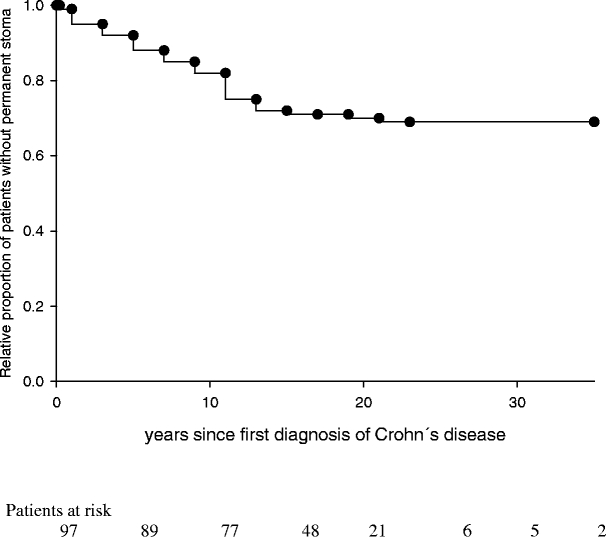
Relative proportion of patients without permanent stoma in complicated perianal Crohn’s disease (n = 97) during follow-up.
Table 1.
Patients with First Manifestation of Complicated Perianal Crohn’s Disease (n = 97) and the Rate of Fecal Diversion During Follow-up
| Patients | Temporary Stoma | Permanent Stoma | |
|---|---|---|---|
| Overall | 97 | 51 (53%) | 30 (31%) |
| Abscess formation | 75 | 32 (43%) | 21 (28%) |
| Simple abscess formation | 33 (44%) | 13 (39%) | 7 (21%) |
| Complex abscess formation | 42 (56%) | 23 (55%) | 15 (36%) |
| Fistulas | 88 | 51 (58%) | 26 (30%) |
| Simple perianal fistulas | 42 (48%) | 17 (40%) | 8 (19%) |
| Complex perianal fistulas | 46 (52%) | 34 (74%) | 23 (50%) |
| Rectovaginal fistulas | 26 (54% of ♀) | 18 (69% of 26) | 14 (54% of 26) |
Overlap between abscess formation and presence of fistulas exists, and abscesses led to fecal diversion in combination with fistulas only.
Table 2.
Abdominal Procedures and the Rate of Fecal Diversion During Follow-up in Patients with Complicated Perianal Crohn’s Disease (n = 97)
| Patient Number | Permanent Stoma (% of Patients) | |
|---|---|---|
| Small Bowel Procedures | ||
| Stricturplasty | 7 | 0 |
| Small bowel resection because of stenosis | 23 | 0 |
| Small bowel resection because of enteroenteric fistula | 7 | 0 |
| Anastomosis resection because of inflammatorystenosis | 30 | 0 |
| Stoma revision | 4 | 0 |
| Large bowel procedures | ||
| Segmental colon resection | 14 | 0 |
| Right-sided colon resection | 20 | 4 (20) |
| Ileocecal resection | 68 | 24 (35) |
| Subtotal colon resection | 35 | 21 (60) |
| Left-sided colon resection | 6 | 5 (83) |
| Rectal resection | 13 | 12 (92) |
Patients that required small bowel resections carried no risk of fecal diversion, whereas patients with colon resections carried an increased risk of fecal diversion.
Abscess Formation
Seventy-five of 97 patients (77%) had at least one perianal abscess at first presentation or during follow-up. Surgical therapy for abscesses consisted of seton drainage, mushroom catheter drainage, or incision and drainage, as described previously.21 A permanent fecal diversion because of recurrent abscess formations, always in combination with fistula problems and perianal sepsis, became necessary in 21 of 75 cases (28%). Simple abscess formations occurred in 33 patients, and 7 of those patients (21%) required permanent fecal diversion (Fig. 2). Complex abscess formations were present in 42 patients, with permanent fecal diversion being necessary in 14 cases (33%; Table 1).
Figure 2.
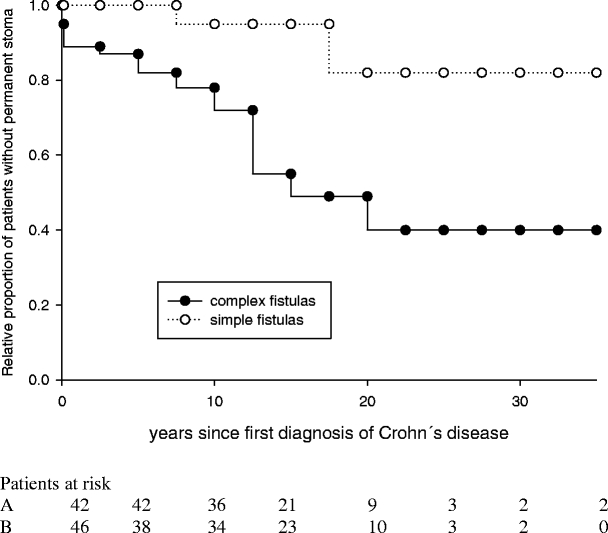
Relative proportion of patients without permanent stoma in complicated perianal Crohn’s disease with simple (n = 42) and complex (n = 46) fistulas during follow-up. The presence of a complex fistula significantly increased the risk of permanent fecal diversion (P < 0.001).
Perianal and Rectovaginal Fistulas
Perianal fistulas affected 88 of 97 patients (91%), including 26 female patients with rectovaginal fistulas. Symptomatic fistulas with abscess formation were treated by simple drainage procedures and provided with a seton. Azathioprine therapy was tried if purulent secretion persisted. Infliximab was rarely used in recent years at the discretion of the referring gastroenterologist. Overall, permanent fecal diversion became necessary in 26 of 88 patients (30%). Forty-two patients (48%) had simple fistulas, whereas 46 patients (52%) had complex fistulas. Of these, permanent fecal diversion was documented in 14 patients (54%), whereas simple fistulas eventually required a permanent stoma in only 8 cases (16%). Twenty-six of 48 female patients had rectovaginal fistulas, of which 14 (54%) eventually required permanent fecal diversion (Table 1).
Thirty-four of 97 patients (35%) developed anal strictures, whereas fecal incontinence was documented in 12 patients (12%) during the follow-up. In 14 of 34 patients (41%) with anal stricture, a permanent fecal diversion became necessary, whereas 10 of 12 patients (83%) suffering from fecal incontinence required a permanent stoma.
Abdominal Surgery
Eighty-three of 97 patients with perianal Crohn’s disease (86%) underwent abdominal surgery at least once during follow-up because of Crohn’s disease activity, with a total of 227 abdominal procedures being performed. In 68 of 227 operations (29%), intestinal stenosis required surgery. Segmental colon resection was performed 108 times (46%), whereas subtotal colectomy for fulminant colitis was performed 35 times (15%). One patient required surgery because of a fistula carcinoma. Patients with complicated perianal Crohn’s disease who required a small bowel resection carried no risk of permanent fecal diversion, whereas the majority of patients with left-sided colon resection, subtotal colon resection, or rectal resection needed permanent fecal diversion (Table 2). Patients that needed abdominal surgery repeatedly carried an increased risk for a permanent stoma (permanent stoma rate: three or more abdominal operations, n = 20, 50%; less than two abdominal operations, n = 20, 15%; P < 0.043). Patients with permanent fecal diversion had undergone previous abdominal surgery three times as often as those patients without (P < 0.05; Table 3). Eventually, 17 patients (18%) needed proctectomy, of which 13 were female patients with rectovaginal fistulas. In 14 patients with a permanent stoma, a rectal stump remained in place and was controlled at yearly intervals. Patients with complicated perianal Crohn’s disease and large bowel resection carried a significantly increased risk of permanent fecal diversion compared to patients with complicated perianal Crohn’s disease and small bowel resection (48 vs 0%, P < 0.001) (Fig. 3).
Table 3.
Abdominal Procedures per Patient and Percentage of Patients with Abdominal Surgery
| Abdominal Procedures | Median | (25–75%) | Patients with Abdominal Surgery (%) | P Value |
|---|---|---|---|---|
| Permanent fecal diversion | 3 | (2–4) | 100 | P < 0.05 |
| Without permanent fecal diversion | 1 | (1–3) | ||
| Temporary fecal diversion | 3 | (2–4.25) | 80 | P < 0.05 |
| Without temporary fecal diversion | 1 | (0–2.5) | ||
| Anal stricture | 2 | (1–4) | 60 | n.s. |
| Without anal stricture | 2 | (1–3) | ||
| Incontinence | 2 | (2–4.5) | 10 | n.s. |
| Without incontinence | 2 | (1–3) |
Patients with Crohn’s disease and complicated perianal fistulas who required temporary or permanent stoma had more abdominal procedures than patients who did not need a stoma, whereas patients with anal stricture or fecal incontinence had no increased abdominal procedure rate (the number of abdominal procedures per patient is given as median with interquartile ranges). Abdominal surgery and fecal diversion correlated on univariate analysis (P < 0.05).
Figure 3.
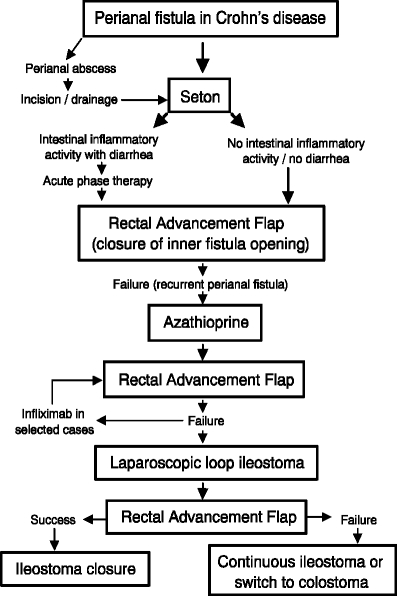
Treatment algorithm for patients with complicated perianal Crohn’s disease.
Temporary Fecal Diversion
Temporary fecal diversion, done mostly by loop ileostomy, became necessary in 51 of 97 patients (53%). Increased rates of temporary fecal diversion were observed in 55% of patients with complex abscess formations, including those with severe perianal sepsis, in 77% of patients with complex perianal fistulas, and in 69% of female patients with rectovaginal fistulas. In 24 of 51 patients (47%) the temporary stoma could be removed after perianal disease had subsided. Fistulas were closed in these patients by rectal advancement flaps,23 whereas cutting setons, fistulectomy, or infliximab infusions were not employed in these patients.
Risk Factors Predictive of a Permanent Fecal Diversion
Univariate Analysis
Complex perianal fistulas (P < 0.04), fecal incontinence (P < 0.001), and rectovaginal fistulas in female patients (P < 0.001) carried an increased risk for a permanent stoma. Patients with high rates of abdominal surgery had a significantly increased risk for fecal diversion, whereas the frequency of abdominal surgery was not influenced by the presence of anal stricture or fecal incontinence (Table 3). The need for subtotal colon resection (P < 0.001), rectal resection (P < 0.001), or temporary fecal diversion (P = 0.001) also resulted in an increased risk of permanent fecal diversion. A variety of factors did not increase the risk of a permanent stoma, such as sex of the patient, anal stenosis, perianal abscesses, and abdominal surgery, excluding subtotal colon and rectal resection (Table 4).
Table 4.
Risk Factors for Permanent Fecal Diversion by Univariate and Multivariate Analysis in Patients with Complicated Perianal Crohn’s Disease
| Risk Factors | Univariate Analysis | Multivariate Analysis | |||
|---|---|---|---|---|---|
| Permanent Fecal Diversion (%) | P Value | OR | 95% CI | P Value | |
| Rectal resection | 92 | P < 0.001 | 30 | 5–179 | P < 0.002 |
| Fecal incontinence | 83 | P < 0.001 | 21 | 3–165 | P < 0.02 |
| Subtotal colectomy | 60 | P < 0.001 | |||
| Rectovaginal fistulas | 54 | P < 0.001 | |||
| Temporary fecal diversion | 51 | p = 0.001 | 8 | 2–35 | P < 0.02 |
| Complex perianal fistulas | 54 | P < 0.04 | 5 | 2–18 | P < 0.03 |
Multivariate Analysis
According to multivariate analysis, complex perianal fistulas (P < 0.03), fecal incontinence (P < 0.02), temporary fecal diversion (P < 0.02), or rectal resection (P < 0.002) still carried a significantly increased risk for permanent fecal diversion (Table 4).
Discussion
Perianal disease is a feature of Crohn’s disease that afflicts approximately one third of patients, but prevalence rates vary between 8 and 90%.19 Patients with colonic involvement will have perianal disease in more than 50% of cases, whereas patients with small bowel disease are affected in less than 20%.24 Perianal Crohn’s disease can present as minor lesions, such as skin tags, skin excoriations, and fissures, which rarely need surgical treatment. In contrast, perianal fistulas often result in abscesses or perianal sepsis, requiring urgent surgery, and fistulas and abscesses are considered as suppurative complications of perianal Crohn’s disease.25 We conducted surgery as conservatively as possible because sphincter function should be preserved as much as possible.19,22,23 Throughout the study period, 77% of patients developed abscess formations, which is considerably more than the 48% rate reported previously with a shorter mean follow-up of 32 months.22
The incidence of perianal fistulas in Crohn’s disease is somewhat lower, affecting 10–34% of patients.26 Healing rates of 60–70% in patients with Crohn’s disease and perianal fistulas were reported in earlier series.21,26–28 However, these series contained a large proportion of simple fistulas, whereas most of our patients had complex transsphincteric or rectovaginal fistulas and fistulas that did not heal through conservative treatment. Perianal fistulas that resulted in abscess formation were drained surgically, and in case of massive purulent secretion or perianal sepsis, oral antibiotics were used additionally. Setons were put into place to prevent recurrent abscess formation, a treatment that is well known as a possible means for controlling perianal sepsis and preventing recurrent abscess formation.19,21–23,29 We did not use cutting setons nor fistulotomy for transsphincteric fistulas, as fecal incontinence may succeed.19,22,23 Infliximab was successfully shown to reduce fistula secretion, and outer fistula openings may heal.30 However, fistula tracks persist with varying degrees of residual inflammation, which may cause recurrent fistulas and perianal abscesses.31
Rectal advancement flaps can be used to close the inner opening of transsphincteric or rectovaginal fistulas when perianal disease has abated and the rectal mucosa is not inflamed. However, fistulas eventually recurred in about one third of patients after a mean follow-up of 19 months, and some patients developed new fistulas.23 Hyman32 reported an initial 79% healing rate in a series of 14 patients with Crohn’s disease and perianal fistulas, but observed longer term success in only 50% of patients. Our current approach is to use rectal advancement flaps for transsphincteric fistulas while delaying surgery if there is an acute flare of Crohn’s disease, in particular, if proctitis is present.23,33 Perianal fistulas in Crohn’s disease closed by a rectal advancement flap have a recurrence rate of 50–60%, but can be attempted repeatedly.23,34 If a second rectal advancement flap fails, the failure rate increases up to 75%,34 and a temporary stoma is suggested to the patient before a third attempt is made. By this approach, we were able to heal perianal fistulas in 24 out of 51 patients (47%) who required a temporary stoma. In a previous study, diversion was the most powerful factor influencing healing according to multiple regression analysis.23 The chances of a symptomatic fistula recurrence were increased without a stoma (52% vs 14%, 21), and patients with Crohn’s colitis carried an increased risk of fistula recurrence.23
A particular problem is rectovaginal fistulas, which almost always open at the dentate line.19 They occur in 3–10% of patients with Crohn’s disease.19 In our series, 54% of female patients had rectovaginal fistulas, indicating that a large patient proportion had complex perianal fistulas; referral bias might have contributed to this high proportion of rectovaginal fistulas, as our outpatient clinic for Crohn’s disease is well known regionally. Rectovaginal fistulas carry a poor prognosis,18 and a 70% recurrence rate was observed 24 months after a rectal advancement flap was performed.23 In the experience of Keighley et al.35, 11 of 13 patients with rectovaginal fistulas required proctectomy with a permanent stoma. In our series, 8 out of 26 rectovaginal fistulas healed by a rectal advancement flap. Eighteen patients required a temporary stoma, of which four could be removed after a rectal advancement flap was tried again successfully (overall healing rate 12 of 26, 46%), resulting in a permanent stoma rate of 14 of 26 patients (54%). Healing rates of rectovaginal fistulas associated with Crohn’s disease vary widely, and low patient numbers are usually reported.18 In the Cleveland clinic, 16 of 37 rectovaginal fistulas (43%) healed using an endorectal advancement flap, but patients not having Crohn’s disease were included in the study.36 In low anovaginal fistulas, higher healing rates of up to 68% were reported in patients with Crohn’s disease.37 Recurrent rectovaginal fistulas were shown to heal after repeated rectal advancement flap procedures were conducted at about the same rate even in Crohn’s disease, but surgery should be delayed for at least 3 months after a previous repair,38 which is standard policy at our clinic. It is interesting to note that Halverson et al.38 reported that the presence of a diverting stoma significantly increased the fistula recurrence rate (67 vs 50%).
After years of perianal or transvaginal pus secretion and recurrent abscess formations, patients are often tired of the suffering involved. Antibiotics, azathioprine, or 6-mercaptopurine may be used successfully in suppurative perianal Crohn’s disease.15,18,38,39 In our department, this is routinely tried if perianal disease does not settle after surgical drainage is provided. However, azathioprine or 6-mercaptopurine are not tolerated by all patients and may also fail to relieve perianal Crohn’s disease. In these cases, fecal diversion can be offered as a relatively minor procedure with low morbidity,18 and might be performed laparoscopically in selected cases.40 However, overall healing rates of perianal disease are only around 40%, with 87% of those being diverted eventually retaining their stoma.18 We diverted 51 patients, of which 27 retained there stoma (53%), which compares favorably, but indicates a considerable risk for a permanent stoma if diversion is tried, and being a significant risk factor according to multivariate analysis in our series.
Perianal disease in combination with fecal incontinence might necessitate fecal diversion as well. Factors that may contribute to fecal incontinence in patients with Crohn’s disease are reduced stool consistency, sphincter injuries after abscess incisions, and keyhole deformities after laying open perianal fistulas. The latter was reported to result in fecal incontinence in 5 of 27 patients with perianal fistulas in Crohn’s disease,41 which is why we did not use this technique or cutting setons in transsphincteric fistulas. We always incise perianal abscesses along a circular perianal line, meticulously preserving sphincter integrity. Nevertheless, fecal incontinence became prevalent in 12% of our patients during follow-up and was a significant predictor of permanent fecal diversion by multivariate analysis. Rectal advancement flap procedures may influence continence,22 and a 9% rate of worsened continence was reported thereafter.33 In particular, the risk of fecal incontinence was increased after flap repairs in patients with previous surgical fistula repairs,33 but only 2 out of 12 patients with fecal incontinence had a previous rectal advancement flap repair in our study. A high rate of abdominal procedures was associated with an increased risk for a temporary or a permanent stoma, possibly reflecting high intestinal disease activity of Crohn’s disease in these patients. All 30 patients that ended with a permanent stoma had frequently undergone abdominal procedures (median of three procedures).
Because of the suffering involved and unsuccessful previous attempts to heal perianal fistulas, about half of our patients eventually opted for temporary ileostomy, being brought into contact with a stoma nurse and stoma carriers with Crohn’s disease before, and 31% of all patients remained with a permanent stoma. In general, the need for a stoma was reduced during recent decades in Crohn’s disease,43 but the overall long-term risk for a stoma in patients with Crohn’s disease who require abdominal surgery was reported between 30 and 40%.44,45 Fecal diversion remains an option to subside perianal disease activity, with an early response rate of about 70–80%.46–48 Unfortunately, 75% of those patients eventually experienced a relapse, and the restoration of intestinal continuity was achieved in 10% only.48 In 130 patients with surgically treated perianal Crohn’s disease, a permanent stoma became necessary in 24%, and this rate increased to 53% in patients with Crohn’s colitis.45 In this light, considering that all of our patients had complicated perianal disease and almost all (93 of 97 patients, 96%) had colonic involvement, we judge a 31% permanent stoma rate as a success in a subset of patients carrying a particularly high risk for a stoma. Local drainage, setons, and, if not successful, a temporary stoma after a rectal advancement flap, resulted in closure of the stoma in 47% of the time, which is an improvement compared to previous studies where restoration of intestinal continuity was reported in only 10–40% of patients, with most studies providing the lower end percentages.44,46–48 If perianal Crohn’s disease was the indication for creating a stoma, stoma closure was successful in 2 out of 15 patients only (13%).47
Eventually, proctectomy became necessary in 18% of patients, which is in the range others have reported.24,44,49,50 However, if the presence of colonic disease was considered separately, seven out of eight patients with complicated perianal Crohn’s disease needed proctectomy, again indicating that an 18% proctectomy rate in our high-risk population is rather low. All of our patients who needed proctectomy had colonic disease, and 13 of 17 had a rectovaginal fistula.
In a recent study investigating the quality of life in patients with Crohn’s proctocolitis, patients in remission had a health-related quality of life similar to controls. Patients with active disease had a reduced quality of life, and only the symptom index negatively predicted a reduced quality of life, whereas neither previous colonic surgery nor the presence of a stoma did.51 Accordingly, carrying a stoma does not necessarily mean a reduced quality of life, although a slight reduction in two out of eight domains (physical and emotional role) was detected.51 However, in patients with perianal disease, the cumulative abscess rate during 3 years of follow-up and the symptomatic fistula recurrence rate with a mean follow-up of 22 months were both greatly reduced with a stoma (76 vs 13% and 52 vs 14%), indicating reduced suffering and need for surgery.21,22
In summary, patients with complicated perianal Crohn’s disease, colonic involvement, and a high rate of abdominal procedures carried a significant risk for a permanent stoma. However, if the risk factors identified were taken into account, the rate of patients eventually requiring a permanent stoma seemed low and showed a decrease compared to previous studies, indicating that multiple treatment episodes and complex surgery, including temporary fecal diversion, might eventually heal at least some patients in this high-risk population, as was also observed by others.50
References
- 1.Crohn BB, Ginzburg L, Oppenheimer GD. Regional ileitis: A pathologic and clinical entity. JAMA 1932;99:1323–1329. [DOI] [PubMed]
- 2.Price AB, Morson BC. The surgical pathology of Crohn’s disease and ulcerative colitis. Human Pathol 1975;6:7–29. [DOI] [PubMed]
- 3.Johnson OA, Hoskins DW, Todd J, Thorbjarnarson B. Crohn’s disease of the stomach. Gastroenterology 1996;50:571–577. [PubMed]
- 4.Gelfand MD, Krone CL. Dysphagia and esophageal ulceration in Crohn’s disease. Gastroenterology 1968;55:510–514. [PubMed]
- 5.Dudeney TP. Crohn’s disease of the mouth. Proc R Soc Med 1969;62:1237. [DOI] [PMC free article] [PubMed]
- 6.Wolff BG. Crohn’s disease: the role of surgical treatment. Mayo Clin Proc 1986;61:191–195. [DOI] [PubMed]
- 7.Fielding JF. Perianal lesions in Crohn’s disease. J R Coll Surg Edinb 1971;17:32–37. [PubMed]
- 8.Lennard-Jones JE, Ritchie JK, Zohrab WJ. Proctocolitis and Crohn’s disease of the colon: A comparison of the clinical course. Gut 1976;17:477–482. [DOI] [PMC free article] [PubMed]
- 9.Allan A, Keighley MRB. Management of perianal Crohn’s disease. World J Surg 1988;12:198–202. [DOI] [PubMed]
- 10.Alexander-Williams J. The place of surgery in Crohn’s disease. Gut 1971;12:739. [DOI] [PMC free article] [PubMed]
- 11.Buchmann P, Keighley MRB, Allan RN, Thompson H, Alexander-Williamy J. Natural history of perianal Crohn’s disease: Ten year follow up: A plea for conservatism. Am J Surg 1980;140:642–644. [DOI] [PubMed]
- 12.Keighley MRB, Allan RN. Current status and influence of operation on perianal Crohn’s disease. Int J Colorectal Dis 1986;1:104–107. [DOI] [PubMed]
- 13.Alexander-Williams J, Buchmann P. Perianal Crohn’s disease. World J Surg 1980;4:203–208. [DOI] [PubMed]
- 14.Poritz LS, Rowe WA, Koltun WA. Remicade does not abolish the need for surgery in fistulizing Crohn’s disease. Dis Colon Rectum 2002;45:771–775. [DOI] [PubMed]
- 15.Dejaco C, Harrer M, Waldhoer T, Miehsler W, Vogelsang H, Reinisch W. Antibiotics and azathioprine for the treatment of perianal fistulas in Crohn’s disease. Aliment Pharmacol Ther 2003;18:1113–1120. [DOI] [PubMed]
- 16.Oberhelman HA, Kohatsu S, Taylor KB, Kivel RM. Diverting ileostomy in the surgical management of anal and perianal Crohn’s disease of the colon. Am J Surg 1968;115:231–240. [DOI] [PubMed]
- 17.Burman JM, Thompson M, Cooke WT, Alexander-Williams J. The effects of diversion of intestinal contents on the progress of Crohn’s disease of the large bowel. Gut 1971;2:11. [DOI] [PMC free article] [PubMed]
- 18.Frizelle FA, Santoro GA, Pemberton JH. The management of perianal Crohn’s disease. Int J Colorectal Dis 1996;11:227–237. [DOI] [PubMed]
- 19.McClane, Rombeau JL. Anorectal Crohn’s disease. Surg Clin North Am 2001;81:169–183. [DOI] [PubMed]
- 20.Parks AG, Gordon PH, Hardcastle JD. A classification of fistula-in-ano. Br J Surg 1976;63:1–12. [DOI] [PubMed]
- 21.Makowiec F, Jehle EC, Starlinger M. Clinical course of perianal fistulas in Crohn’s disease. Gut 1995;37:696–701. [DOI] [PMC free article] [PubMed]
- 22.Makowiec F, Jehle EC, Becker HD, Starlinger M. Perianal abscess in Crohn’s disease. Dis Colon Rectum 1997;40:443–450. [DOI] [PubMed]
- 23.Makowiec F, Jehle EC, Becker HD, Starlinger M. Clinical course after transanal advancement flap repair of perianal fistula in patients with Crohn’s disease. Br J Surg 1995;82:603–606. [DOI] [PubMed]
- 24.Williams DR, Collier JA, Corman ML, Nugent FW, Veidenheimer MC. Anal complications in Crohn’s disease. Dis Colon Rectum 1981;24:22–24. [DOI] [PubMed]
- 25.Williamson PR, Hellinger MD, Larach SW, Ferrara A. Twenty-year review of the surgical management of perianal Crohn’s disease. Dis Colon Rectum 1995;38:389–392. [DOI] [PubMed]
- 26.Williams JG, Rothenberger DA, Nemer FD, Goldberg SM. Fistula-in-ano in Crohn’s disease: Results of aggressive surgical treatment. Dis Colon Rectum 1991;34:378–384. [DOI] [PubMed]
- 27.Sohn N, Korelitz BI, Weinstein MA. Anorectal Crohn’s disease: Definitive surgery for fistulas and recurrent abscesses. Am J Surg 1980;139:394–397. [DOI] [PubMed]
- 28.Bernard D, Morgan S, Tasse D. Selective surgical management of Crohn’s disease of the anus. Can J Surg 1986;29:318–321. [PubMed]
- 29.Scott HJ, Northover JMA. Evaluation of surgery for perianal Crohn’s disease. Dis Colon Rectum 1996;39:1039–1043. [DOI] [PubMed]
- 30.Present DH, Rutgeerts P, Targan S, et al. Infliximab for the treatment of fistulas in patients with Crohn’s disease. N Engl J Med 1999;340:1398–1405. [DOI] [PubMed]
- 31.Van Assche G, Vanbeckevoort D, Bielen D, Coremans G, Aerden I, Noman M, D’Hoore A, Penninckx F, Marchal g, Cornillie F, Rutgeerts P. Magnetic resonance imaging of the effects of infliximab on perianal fistulizing Crohn’s disease. Am J Gastroenterol 2003;98:332–339. [DOI] [PubMed]
- 32.Hyman N. Endoanal advancement flap repair for complex anorectal fistulas. Am J Surg 1999;178:337–340. [DOI] [PubMed]
- 33.Radcliffe AG, Ritchie JK, Hawely PR, Lennard-Jones JE, Northover JMA. Anovaginal and rectovaginal fistulas in Crohn’s disease. Dis Colon Rectum 1988;31:94–99. [DOI] [PubMed]
- 34.Mizrahi N, Wexner SD, Zmora O, Da Silva G, Efron J, Weiss EG, Vernava AM, Nogueras JJ. Endorectal advancement flaps. Are there predictors of failure? Dis Colon Rectum 2002;45:1616–1621. [DOI] [PubMed]
- 35.Keighley MR, Williams NS. Surgical treatment of perianal Crohn’s disease. In Keighley MR, Williams NS, Eds. Surgery of the Anus, Rectum, and Colon, 2nd ed. New York: WB Saunders, 1999, pp 2135.
- 36.Sonoda T, Hull T, Piedmonte MR, Fazio VW. Outcomes of primary repair of anorectal and rectovaginal fistulas using the endorectal advancement flap. Dis Colon Rectum 2002;45:1622–1628. [DOI] [PubMed]
- 37.Hull TL, Fazio VW. Surgical approaches to low anovaginal fistulas in Crohn’s disease. Am J Surg 1997;173:95–98. [DOI] [PubMed]
- 38.Halverson AL, Hull TL, Fazio VW, Church J, Hammel J, Floruta C. Repair of recurrent rectovaginal fistulas. Surgery 2001:30:753–758. [DOI] [PubMed]
- 39.Korelitz BI, Present DH. Favorable effect of 6-mercaptopurine on fistulae in Crohn’s disease. Dig Dis Sci 1985;30:58–64. [DOI] [PubMed]
- 40.Pearson DC, May GR, Fick GH, Sutherland LR. Azathioprine and 6-mercaptopurine in Crohn’s disease: A meta-analysis. Ann Intern Med 1995;123:132–142. [DOI] [PubMed]
- 41.Weiss UL, Jehle E, Becker HD, Buess GF, Starlinger M. Laparoscopic ileostomy. Br J Surg 1995;82:1648. [DOI] [PubMed]
- 42.Scott HJ, Northover JMA. Evaluation of surgery for perianal Crohn’s fistulas. Dis Colon Rectum 1996;39:1039–1043. [DOI] [PubMed]
- 43.Andersson P, Olaison G, Bendtsen P, Myrelid P, Sjödahl R. Health related quality of life in Crohn’s proctocolitis does not differ from a general population when in remission. Colorectal Dis 2003;5:56–62. [DOI] [PubMed]
- 44.Post S, Herfarth C, Schumacher H, Golling M, Schürmann G, Timmermanns G. Experience with ileostomy and colostomy in Crohn’s disease. Br J Surg 1995;82:1629–1633. [DOI] [PubMed]
- 45.Hurst RD, Molinari M, Chung TP, Rubin M, Michelassi F. Prospective study of the features, indications, and surgical treatment in 513 consecutive patients affected by Crohn’s disease. Surgery 1997;122:661–668. [DOI] [PubMed]
- 46.Harper PH, Kettlewell MGW, Lee ECG. The effect of split ileostomy on perianal Crohn’s disease. Br J Surg 1982;69:608–610. [DOI] [PubMed]
- 47.Edwards CM, George BD, Jewell DP, Warren BF, Mc Mortensen NJ, Kettlewell MGW. Role of a defunctioning stoma in the management of large bowel Crohn’s disease. Br J Surg 2000;87:1063–1066. [DOI] [PubMed]
- 48.Yamamoto T, Allan RN, Keighley MRB. Effect of fecal diversion alone on perianal Crohn’s disease. World J Surg 2000;24:1258–1263. [DOI] [PubMed]
- 49.Pescatori M, Interisano A, Basso L, Arcana F, Buffatti P, Di Bella F, Doldi A, Forcheri V, Gaetini R, Pera A, Mattana C, Prantera C, Pulvirenti A, Segre D, Simi M, Tonelli F.. Management of perianal Crohn’s disease. Results of a multicenter study in Italy. Dis Colon Rectum 1995;38:121–124. [DOI] [PubMed]
- 50.Bell SJ, Williams AB, Wiesel P, Wilkinson K, Cohen RCG, Kamm MA. The clinical course of fistulating Crohn’s disease. Aliment Pharmacol Ther 2003;17:1145–1151. [DOI] [PubMed]
- 51.Andersson P, Olaison G, Bodemar G, Nyström P-O, Sjödahl R. Surgery for Crohn colitis over a twenty-eight-year period: Fewer stomas and the replacement of total colectomy by segmental resection. Scand J Gastroenterol 2002;37:68–73. [DOI] [PubMed]


