Abstract
An adequate supply of taurine during fetal life is important for normal beta-cell development and insulin action. An altered availability of taurine may programme glucose metabolism in utero and result in type 2 diabetes in adult age. We examined whether maternal taurine supplementation in late pregnant rats affects postnatal growth, adult body composition, insulin sensitivity and endogenous insulin secretion in intrauterine growth restricted (IUGR) and normal offspring. Uterine artery ligation or sham operations were performed on gestational day (GD) 19. Taurine supplementation was given to half of the dams from GD 18 until term, resulting in four groups of offspring: sham (n = 22), sham/taurine (n = 22), IUGR (n = 22) and IUGR/taurine (n = 24). The offspring were studied at 12 weeks of age. In offspring with normal birth weight, fetal taurine supplementation markedly stimulated postnatal growth. In sham/taurine females, fat depots, plasma free fatty acid and leptin concentrations were increased, and insulin sensitivity was reduced. Insulin sensitivity was unaltered in IUGR and IUGR/taurine offspring. However, whereas IUGR offspring showed little catch-up growth, 50% of IUGR/taurine animals displayed complete catch-up at 12 weeks of age, and these animals had increased fat depots and reduced insulin sensitivity. In conclusion, taurine supplementation in late gestation resulted in accelerated postnatal growth, which was associated with adult obesity and insulin resistance in both IUGR and normal offspring. This effect was particularly evident in females. These data suggest that fetal taurine availability is an important determinant for postnatal growth, insulin sensitivity and fat accumulation.
The prevalence of type 2 diabetes is increasing at an alarming rate and it is estimated that 200–300 million people around the world will be afflicted by the end of the decade (Zimmet et al. 2001). A perinatal origin of adult disease has been suggested to contribute to the rapid increase of metabolic syndrome, since intrauterine growth restriction (IUGR) has been identified as an important risk factor for development of insulin resistance, obesity, hyperlipidaemia and type 2 diabetes in adult age (Hofman et al. 1997). A lively debate has also evolved as to whether catch-up growth following early growth restriction may add to the risk of developing obesity and cardiovascular disease later in life. Epidemiological studies have suggested that high postnatal growth rates in subjects with low birth weights are associated with an increased risk for central obesity (Ong et al. 2004), type 2 diabetes (Forsen et al. 2000), and death from cardiovascular disease (Eriksson & Forsen, 2002). Recently, it was reported that adults with ischaemic heart disease were, on average, small at birth, thin at 2 years of age, and subsequently displayed a rapid weight gain (Barker et al. 2005), suggesting that catch up-growth in weight before 2 years of age may not be linked to later cardiovascular disease. In experimental studies of prenatal growth restriction induced by maternal food restriction, postnatal cross-fostering to normal lactating dams and subsequent weight catch-up growth correlated with increased percentage body fat in the adult rat offspring (Desai et al. 2005).
Maternal bilateral uterine artery ligation in the rat represents a model of IUGR caused by placental insufficiency. We have previously found a gender-specific response in IUGR offspring where female rats were associated with increased fasting blood glucose levels and impaired glucose tolerance, whereas male offspring had normal glucose metabolism (Jansson & Lambert, 1999). Subsequently, Simmons and coworkers demonstrated that IUGR offspring following maternal bilateral uterine artery ligation displayed insulin resistance and reduced beta-cell mass compared with controls at 15 weeks (Simmons et al. 2001).
Taurine (2-aminoethanesulphonic acid) is a sulphur-containing amino acid that is present in the diet, and which can also be synthesized from cysteine and methionine in postnatal life. Although it is not incorporated into proteins, taurine has been shown and proposed to have a number of essential biological functions (Huxtable, 1992). It is best known for its role in lipid metabolism, where taurine enhances the absorption of fat in the intestine by stimulating bile acid synthesis, and the degradation of cholesterol. In addition, taurine plays important roles in cellular osmoregulation and in inhibition of apoptosis and protein phosphorylation (Huxtable, 1992; Hansen, 2001). Animal studies have shown that taurine also seems to have plasma lipid-lowering effects (Murakami et al. 2002) and improves insulin sensitivity (Nakaya et al. 2000; Anuradha & Balakrishnan, 1999).
The endogenous synthesis of taurine is inadequate in the fetus, and taurine is therefore considered to be an essential amino acid during fetal development (Hibbard et al. 1990). Consequently, the fetus is dependent on placental transport, mediated by high-affinity Na+- and Cl−-dependent transporters (Ramamoorthy et al. 1994), for an adequate supply of taurine. We have previously shown a reduced activity of the placental taurine transporters in human IUGR (Norberg et al. 1998), which may explain the lower fetal plasma concentrations of taurine often observed in this pregnancy complication (Cetin et al. 1990). Previous studies have demonstrated an association between fetal taurine deficiency and abnormal pancreas development and function. In dams fed a low-protein diet, insulin secretion in vitro from islets of Langerhans obtained from the offspring was reduced. When taurine was added to the drinking water of low-protein-fed dams, the release of insulin from fetal islets was restored to normal levels (Cherif et al. 1998). Further, taurine supplementation in low-protein-fed dams has been reported to normalize the vascularization and beta-cell mass in the fetal endocrine pancreas (Boujendar et al. 2003). Recently, it was reported that taurine supplementation throughout pregnancy and lactation in rats fed a low-protein diet restored, a least in part, beta-cell function in vivo in the 12-week-old offspring (Merezak et al. 2004).
Maternal protein restriction may not represent an optimal model for programming of diabetes in human IUGR in Western societies where placental insufficiency probably is the most common cause of restricted fetal growth. This study was undertaken to investigate the effects of prenatal taurine supplementation on postnatal growth, adult body composition, insulin sensitivity and endogenous insulin secretion in normal offspring as well as in offspring with IUGR due to placental insufficiency.
Methods
Animals
Dams
Pregnant rats (Sprague-Dawley, 10–12 weeks of age, 230–300 g; B-K, Sweden) were delivered to the animal facility 3–5 days prior to uterine artery ligation or sham operation. At day 18 of gestation (day 1 is defined as the day immediately following the night during which males were present), supplementation of taurine (Sigma-Aldrich, CA, USA) started in half of the animals. Taurine 2% (w/v) was given until term (day 23 of gestation) as an addition to the drinking water, and it was prepared fresh daily. At day 19 of gestation, uterine artery ligation or sham operation was performed. General anaesthesia was induced by intraperitoneal injection (0.1 ml (100 g rat)−1) of a mixture consisting of 3 ml xylazine (Rompun vet, 20 mg ml−1; Bayer) and 10 ml ketamine hydrochloride (Ketalar, 50 mg ml−1; Parke-Davis). A 2 cm lower abdominal midline incision was performed and the uterine arteries were localized. Both uterine arteries were ligated by sutures and the incision was closed. Sham-operated dams underwent an identical procedure without ligation. After surgery, dams were returned to individual cages. Approximately 2–4 h after the completion of the surgery, animals were walking around normally, drinking water and exhibiting the characteristic exploratory behaviour. The water and food intake was measured the day after surgery in a subgroup of animals and found to be normal.
Offspring
After parturition, pups with a birth weight of less than 5 g were defined as IUGR. Thus, there were four groups of offspring: IUGR (offspring, with a birth weight less than 5 g, of dams in which uterine artery ligation was performed), IUGR/taurine (offspring, with a birth weight less than 5 g, of dams that received taurine supplementation and in which uterine artery ligation was performed), sham (offspring of dams in which sham operation was performed) and sham/taurine (offspring of dams that received taurine supplementation and in which sham operation was performed). Approximately two or three of the pups from mothers that were subjected to uterine artery ligation had a birth weight less than 5 g. However, the majority of IUGR pups were either dead at birth or died during the first days after birth. Thus, only 12% of the pups of dams undergoing uterine artery ligation were IUGR that survived until adult age. The number of animals studied in the different groups and the number of litters from which they originated are given in Table 1. In order to normalize litter size, the number of pups in litters larger than eight were reduced to eight, and no litters with less than six pups were included, resulting in 6–8 pups in all litters, with approximately the same number of females and males (Jansson & Lambert, 1999). All pups were raised with their mothers until 4 weeks of age, and subsequently offspring were separated by sex, and housed in cages of 4–5 animals. The offspring were fed standard rat chow (2016; Harlan-Orlac, Holland) and provided with tap water. Food intake was measured in offspring as described (Dahlgren et al. 2001). No significant difference in food intake was found between experimental groups. Pup weight was recorded at birth and once a week thereafter, starting at day 7. Nose–anus length was measured during the first 4 weeks and at clamp experiment. Blood samples for determination of serum leptin and IGF-1 were collected at 10 weeks of age using tail-vein bleeds, and blood samples for analysis of plasma free fatty acids (FFA), cholesterol and triglycerides were collected at experiment at 12 weeks of age in offspring. The term ‘catch-up growth’ was defined as an accelerated growth, after a transient period of growth restriction, resulting in a final weight within mean body weight ±2 s.d. of sham group. The study was approved by the Animal Ethics Committee of Gothenburg University.
Table 1.
Number of litters and animals studied
| Female | Male | |||
|---|---|---|---|---|
| Group | N | n | N | n |
| Sham | 5 | 10 | 8 | 11 |
| Sham/taurine | 7 | 11 | 6 | 11 |
| IUGR | 8 | 11 | 6 | 11 |
| IUGR/taurine | 7 | 12 | 9 | 12 |
IUGR intrauterine growth restricted; N number of litters; n number of offspring.
Euglycaemic–hyperinsulinaemic and hyperglycaemic clamps
Rats were subjected to a euglycemic–hyperinsulinaemic clamp at 12 weeks of age, as previously described (Holmang et al. 1990). The animals were anaesthetized with 125 mg kg−1 body weight of thiobutabarbital sodium (Inactin; RBI, Natick, MA, USA). Subsequently, catheters were inserted into the left carotid artery for blood sampling and into the right jugular vein for infusion of glucose and insulin. Thirty minutes after insertion of the catheters, a bolus injection of [3H-3]glucose (254 μCi kg−1, 1 mCi ml−1; Amersham, Arlington Heights, IL, USA) was given, followed by a continuous infusion of 1.5 μCi (kg)−1 (min)−1 throughout the 150 min study. At t = 0 min, 30 min after the bolus injection of [3H-3]glucose, and just before the initiation of the clamp, a blood sample for determination of insulin and [3H-3]glucose was obtained. After a bolus injection of insulin (100 U ml−1 human Actrapid; Novo Nordisk, Bagsværd, Denmark), insulin was continuously infused at a rate of 8 mU kg−1 min−1. A 20% glucose solution in physiological saline was administered to maintain plasma glucose concentration at 6 mmol l−1. Glucose was measured in 10 μl blood samples at regular intervals (every 5 min during the first 40 min and then every 10 min). Blood samples were also obtained at 80 and 90 min for determination of insulin concentration and [3H-3]glucose. The total blood volume withdrawn from each animal was 1.5 ml. The mean glucose infusion rate was calculated on the basis of values from the last 60 min of the clamp. Glucose turnover rate was calculated from the radioactivity of the [3H-3]glucose infusion (d.p.m. μl−1) times the glucose infusion rate (μl min−1) divided by the specific radioactivity of glucose (d.p.m. mg−1) and body weight (kg). Hepatic glucose production was calculated by subtracting the glucose infusion rate from the glucose turnover rate. In a separate series of experiments, rats were subjected to a hyperglycaemic clamp, as previously described (Terrettaz & Jeanrenaud, 1983; Nilsson et al. 1998), in order to determine the endogenous insulin secretion. At completion of the clamps, the anaesthetized animals were killed by decapitation, and parametrial, retroperitoneal, mesenteric (constituting abdominal fat depots) and inguinal adipose tissues were dissected out and weighed.
Analytical methods
Plasma insulin concentrations were measured by enzyme-linked immunosorbent assay (ELISA) (Mercordia, Uppsala, Sweden). For samples obtained from the euglycaemic–hyperinsulinaemic clamp, antibodies cross-reacting with human insulin (Insulin ELISA; Mercordia) were used, and for samples from the hyperglycaemic clamp, rat anti-insulin antibodies (Rat insulin ELISA; Mercordia) were used. The assay had a mean intra-assay coefficient of variation (CV) of 2.8–4%, and mean interassay CV was 2.6–3.6%. For estimation of endogenous glucose production, the activity of plasma [3H-3]glucose was determined. A liquid scintillation cocktail (Beckman Counter, CA, USA) was added to samples prior to analysis. The β radiation of each sample was detected by a liquid scintillation counter (LKB Wallac, Sweden). For determination of plasma FFA, cholesterol and triglycerides, blood samples were collected in EDTA-coated microvette tubes (Sarstedt AB, Nümbrecht, Germany) centrifuged, and the plasma was frozen at −20°C until analysis. Plasma FFA were analysed using a NEFA C kit (Wako Chemicals, Neuss, Germany), and this analysis had a mean intra-assay CV of 2.1%, and interassay CV was 2.7%. Triglyceride levels were determined by an enzymatic technique (Thermo Electron Corporation, Vantaa, Finland) and analyses were performed on a Konelab 20 autoanalyser (Thermo Electron Corporation), with an interassay coefficient of variation of 2.9%. RIA kits were used to analyse serum IGF-1 (IGF-R20; Mediagnost, Reutlingen, Germany) and leptin concentrations (RL-83K; Linco Research, Hampshire, UK; with a mean intra-assay CV of 2–4.6% and mean interassay CV of 3–5.7%).
Analysis of taurine plasma concentration by HPLC
In order to assess the effect of taurine supplementation on maternal and fetal taurine concentrations, a separate series of experiments was carried out in which dams were anaesthetized at day 22 of gestation using 125 mg (kg body weight)−1 thiobutabarbital sodium, and subsequently killed by decapitation. Fetal (mixed arteriovenous blood after decapitation) and maternal blood samples (cardiac puncture) were collected in heparinized tubes, centrifuged at 500g for 10 min at 4°C and stored at −70°C. Plasma samples were deproteinized with 4 vols methanol, incubated for 10 min at 4°C, and centrifuged at 8000g for 5 min. Amino acids were determined using o-phtaldialdehyde derivatization and fluorescence detection, essentially as described earlier (Lindroth, 1979; Sandberg et al. 1986).
Statistical analysis
Analysis of variance was performed by ANOVA (Fischer's PLSD as post hoc test) or Mann-Whitney. Correlation analysis was performed by simple regression. Only significances of differences between experimental and control groups (i.e. sham/taurine versus sham and IUGR/taurine versus IUGR) and differences between IUGR versus sham and IUGR/taurine versus sham are presented. All results are presented as means ± standard error of the mean (s.e.m.). A P value of 0.05 or less was considered statistically significant. StatView statistics software (SAS Institute, Inc.) for the Macintosh system was used for all statistical calculations.
Results
Fetal and maternal plasma taurine concentrations
Taurine supplementation did not affect food or water intake in dams (data not shown). Taurine supplementation resulted in a marked elevation of maternal taurine concentrations and a twofold increase in fetal plasma taurine concentrations in both IUGR/taurine and sham/taurine groups (Table 2). Taurine concentrations were approximately 30% lower in IUGR compared with sham fetal plasma; however, this difference did not reach statistical significance.
Table 2.
Concentration of taurine on day 22 of gestation in maternal and fetal plasma
| Group | n | Taurine (μm) |
|---|---|---|
| Maternal | ||
| Sham | 4 | 15.7 ± 4.0 |
| Sham/taurine | 3 | 55.8 ± 4.8** |
| IUGR | 3 | 13.6 ± 2.0 |
| IUGR/taurine | 5 | 215.4 ± 55.7* |
| Fetal | ||
| Sham | 4 | 60.7 ± 7.2 |
| Sham/taurine | 3 | 120.9 ± 11.6** |
| IUGR | 3 | 43.6 ± 4.6 |
| UGR/taurine | 4 | 97.9 ± 12.1* |
Fetal samples from each litter were pooled prior to analysis; n = number of dams and litters. Values are means ±s.e.m. Statistical analysis was performed comparing the taurine-supplemented group with its corresponding control (i.e. sham/taurine versus sham and IUGR/taurine versus IUGR).
P < 0.05 versus IUGR
P < 0.01 versus sham (Mann-Whitney).
Animal weight and length development
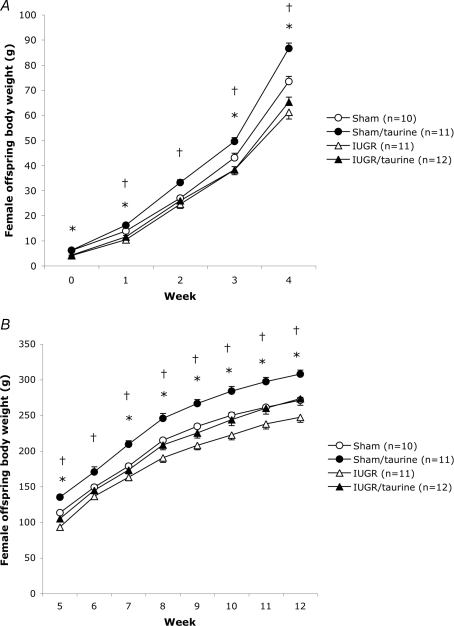
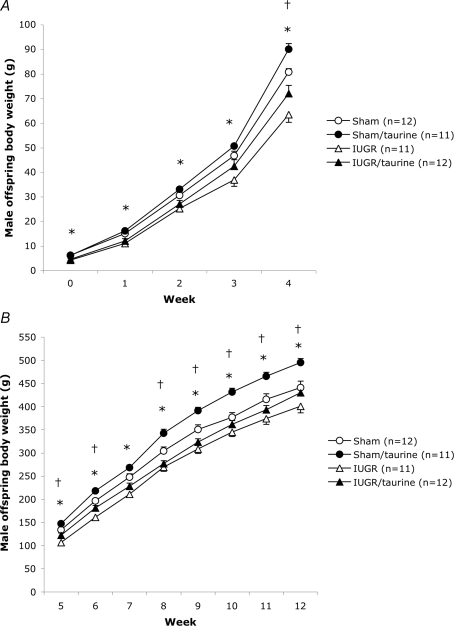
Weight development for female offspring from birth until 4 weeks of age, and from 5 weeks until 12 weeks of age, is shown in Fig. 1A and B, respectively, and the corresponding data for males is given in Fig. 2A and B. At birth, the weight of female and male IUGR and IUGR/taurine offspring were significantly lower than in sham and sham/taurine groups. However, there were no significant differences in birth weight between groups with and without taurine supplementation. As early as week 1 in females (+17%) and week 4 in males (+13%), body weights of sham/taurine animals were significantly higher than in sham groups, and sham/taurine animals remained heavier throughout the study period. Both female and male IUGR groups displayed significantly lower weights from birth until 12 weeks of age compared with sham groups (although not fully significant for females at 2 and 6 weeks of age). Thus, IUGR offspring did not display complete body weight catch-up growth at 12 weeks of age (Figs 1B and 2B). Female IUGR/taurine offspring had a 7% mean increase in body weight by 6 weeks of age as compared with IUGR females, and they were significantly heavier (+11%) than this group at 12 weeks of age. Male IUGR/taurine offspring displayed an accelerated weight gain compared with the IUGR group at 3–6 weeks of age, and remained heavier than the IUGR males, although this apparent difference failed to reach statistical significance at 12 weeks of age (Fig. 2B). Studying the growth curves of individual animals in the female and male IUGR/taurine groups (data not shown), it was apparent that whereas approximately half of the animals showed no or only little catch-up growth, the other half of the animals attained body weights at 12 weeks of age that were similar or greater than the those of the sham groups. Typically, the catch-up growth started 5–7 week of age. In order to address whether animals with different degrees of catch-up growth in the IUGR/taurine groups differed in any of the variables measured in the study, we subsequently used body weight at 12 weeks of age as one of the variables in simple linear regression analysis. It is important to note that most animals showing a particular postnatal growth pattern originated from different litters. For example, there were six female and five male IUGR/taurine animals displaying complete catch-up growth, and these animals originated from five (females) and four (males) different litters.
Figure 1.
Body weight development in female offspring A, body weight (g) development from birth (day 1) to 4 weeks of age in female sham, sham/taurine, (IUGR) and IUGR/taurine groups. B, body weight (g) development from 5 to 12 weeks of age in female sham, sham/taurine, IUGR and IUGR/taurine groups. Values are means ±s.e.m.*P < 0.05 versus sham. †P < 0.05 versus IUGR (ANOVA).
Figure 2.
Body weight development in male offspring A, body weight (g) development from birth (day 1) to 4 weeks of age in male sham, sham/taurine, IUGR and IUGR/taurine groups. B body weight (g) development from 5 to12 weeks of age in male sham, sham/taurine, IUGR and IUGR/taurine groups. Values are means ±s.e.m.*P < 0.05 versus sham. †P < 0.05 versus IUGR (ANOVA).
Nose–anus length of female and male groups is shown in Table 3. At birth, nose–anus length in IUGR and IUGR/taurine offspring was significantly lower than in sham and sham/taurine groups. However, there were no significant differences in nose–anus length at birth between groups with and without taurine supplementation. Female IUGR rats were significantly shorter than sham rats at 1, 4 and 12 weeks of age. Male IUGR group was significantly shorter at birth and at week 1, but did not show any significant difference as compared with the sham group at weeks 2–4 or at 12 weeks of age. Further, female sham/taurine rats were significantly longer than sham rats at week 1 until week 4 and at 12 weeks of age. Male sham/taurine rats were significantly longer than sham rats at week 1 until week 4, but did not differ significantly from this group at 12 weeks of age. Length of IUGR/taurine animals at 12 weeks of age was not significantly different from sham (Table 3), and IUGR/taurine females were significantly longer that IUGR female rats (Table 3). In the IUGR/taurine groups, length at 12 weeks of age was significantly correlated to weight at 12 weeks of age (females r = 0.84, P < 0.001; and males r = 0.88, P < 0.001), demonstrating that the IUGR animals responding to prenatal taurine supplementation with postnatal catch-up in weight also caught up in nose–anus length.
Table 3.
Nose–anus length development from birth (day 1) until 12 weeks of age in female and male offspring
| Nose–anus length (mm) | |||||||
|---|---|---|---|---|---|---|---|
| Group | n | Birth | Week 1 | Week 2 | Week 3 | Week 4 | Week 12 |
| Female offspring | |||||||
| Sham | 10 | 50.4 ± 0.5 | 67.4 ± 0.8 | 80.6 ± 1.5 | 102.2 ± 2.1 | 127.8 ± 1.7 | 223 ± 1.7 |
| Sham/taurine | 11 | 49.3 ± 0.3 | 76.7 ± 1.8*** | 86.8 ± 1.1** | 109 ± 1.9* | 136.3 ± 1.3*** | 230.1 ± 2.7* |
| IUGR | 11 | 46.4 ± 0.8*** | 62.7 ± 1.2* | 79.4 ± 1.3 | 98.3 ± 1.1 | 122.2 ± 1.8* | 216.1 ± 2.7* |
| IUGR/taurine | 12 | 45.5 ± 0.4*** | 63.3 ± 1.2* | 83 ± 1.6 | 103 ± 2.3 | 125.5 ± 1.2 | 225.2 ± 1.6† |
| Male offspring | |||||||
| Sham | 12 | 50.3 ± 0.5 | 66.2 ± 0.6 | 82.6 ± 0.8 | 100.4 ± 1.5 | 129.3 ± 0.9 | 253.4 ± 5.4 |
| Sham/taurine | 11 | 50.6 ± 0.4 | 69.7 ± 0.8*** | 87.9 ± 1.3*** | 106.3 ± 1.3* | 134.2 ± 1.5* | 262.8 ± 2.3 |
| IUGR | 11 | 46.4 ± 0.7*** | 62.3 ± 0.6** | 83.0 ± 1.4 | 100.4 ± 2.8 | 127.4 ± 2.5 | 252.1 ± 2.6 |
| IUGR/taurine | 12 | 45.5 ± 0.4*** | 65.5 ± 1.1† | 84.4 ± 1.5 | 100.6 ± 1.9 | 129.3 ± 2.1 | 255.4 ± 2.5 |
Values are means ±s.e.m.
P < 0.05
P < 0.01
P < 0.001 versus sham
P < 0.05 versus IUGR; n = number of offspring (ANOVA).
Euglycaemic–hyperinsulinaemic and hyperglycaemic clamp
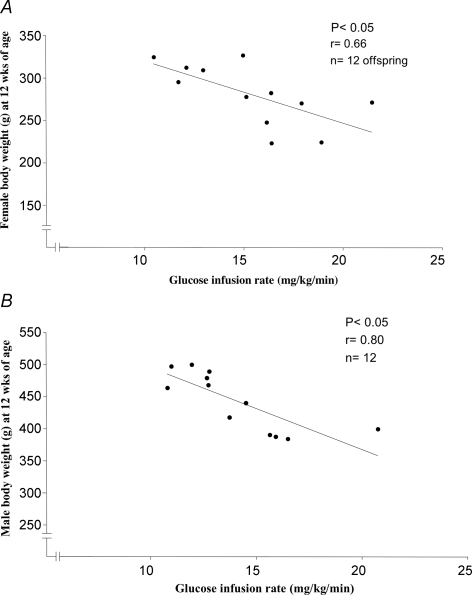
In the euglycaemic–hyperinsulinaemic clamp experiments, female sham/taurine rats showed a significantly lower glucose uptake compared with the sham group, indicating insulin resistance (sham 17.8 ± 1.3 mg kg−1 min−1, n = 10, and sham/taurine 14.0 ± 0.6 mg kg−1 min−1, n = 11, P < 0.01). Insulin sensitivity was not significantly altered in female IUGR (16.1 ± 1.4 mg kg−1 min−1) or in the IUGR/taurine group (16.2 ± 1.5 mg kg−1 min−1) as compared with sham. No significant differences in glucose uptake were found between male groups (sham 13.8 ± 1.0 mg kg−1 min−1, sham/taurine 12.3 ± 1.0 mg kg−1 min−1, IUGR 12.9 ± 1.3 mg kg−1 min−1, IUGR/taurine 14.8 ± 1.5 mg kg−1 min−1). However, in the IUGR/taurine groups the glucose infusion rate was inversely correlated to weight at 12 weeks of age in both females (r = 0.66, P < 0.05, Fig. 3A) and males (r = 0.80, P < 0.01, Fig. 3B), suggesting that catch-up growth is associated with insulin resistance. At clamp, the plasma glucose during steady state was held at approximately 6 mmol l−1. The mean plasma levels of insulin at steady-state were not significantly different between experimental groups (female sham, 257 ± 11 mU l−1; sham/taurine, 240 ± 20 mU l−1; IUGR, 244 ± 16 mU l−1; IUGR/taurine, 262 ± 18 mU l−1; male sham, 253 ± 10 mU l−1; sham/taurine, 245 ± 15 mU l−1; IUGR, 242 ± 22 mU l−1; IUGR/taurine, 258 ± 19 mU l−1). Values for the hepatic glucose production were obtained from the euglycaemic–hyperinsulinaemic clamp. No differences were found between female or male experimental groups (data not shown). Further, there were no significant differences between groups in endogenous insulin response measured during the hyperglycaemic clamp (data not shown).
Figure 3.
Glucose infusion rate inversely correlates to weight in IUGR/taurine offspring A, correlation between body weight at 12 weeks of age (g) and glucose infusion rate (mg kg−1 min−1), P < 0.05, r = 0.66 (n = 12) in IUGR/taurine female offspring (simple regression). B, correlation between body weight at 12 weeks of age (g) and glucose infusion rate (mg kg−1 min−1), P < 0.05, r = 0.80 (n = 12) in IUGR/taurine male offspring (simple regression).
Fat depots
Parametrial and retroperitoneal fat depots (which together with mesenteric fat constitute abdominal fat depots) were significantly larger in female sham/taurine group compared with the sham group at 12 weeks of age (Table 4). Male sham/taurine rats displayed a significant increase in epididymal fat compared with sham rats (Table 4). No differences were found in inguinal (subcutaneous fat depot) or mesenteric fat in female or male groups (data not shown). The weights of parametrial/epididymal and retroperitoneal fat depots were not significantly different in IUGR/taurine animals as compared with IUGR groups (Table 4). However, parametrial fat pad weight (g body weight) was positively correlated to adult body weight in IUGR/taurine females (r = 0.73, P < 0.01) and epididymal fat pad weight was positively correlated to body weight at 12 weeks in IUGR/taurine males (r = 0.86, P < 0.001), indicating that catch-up growth was associated with increased abdominal fat deposition.
Table 4.
Weights of parametrial/epididymal and retroperitoneal fat depots in offspring at 12 weeks of age
| Adipose tissue (g (kg body weight)−1) | |||
|---|---|---|---|
| Group | n | Parametrial/epididymal | Retroperitoneal |
| Female offspring | |||
| Sham | 10 | 14.7 ± 0.8 | 16.9 ± 1 |
| Sham/taurine | 11 | 23.7 ± 1.6*** | 24.4 ± 2.3** |
| IUGR | 11 | 18.4 ± 1.8 | 19.3 ± 1.8 |
| IUGR/taurine | 12 | 18.6 ± 1.4 | 20.5 ± 1.4 |
| Male offspring | |||
| Sham | 12 | 16.5 ± 1.2 | 20.3 ± 1.4 |
| Sham/taurine | 11 | 20.9 ± 0.9* | 23.0 ± 1.3 |
| IUGR | 11 | 17.8 ± 1.3 | 21.4 ± 1.5 |
| IUGR/taurine | 12 | 18.1 ± 1.4 | 23.2 ± 1.6 |
Parametrial and epididymal fat depots were measured in female and male offspring, respectively. Values are means ±s.e.m.
P < 0.05
P < 0.01
P < 0.001 versus sham; n = number of offspring (ANOVA).
Lipid metabolism, leptin and IGF-1
Plasma concentrations of FFA were significantly higher in female sham/taurine group compared with the sham group (1.07 ± 0.1 and 0.75 ± 0.1 mmol l−1, respectively, P < 0.05, n = 8). In contrast, circulating levels of FFA were not significantly different in the other study groups or between male groups (data not shown). No differences were observed in plasma cholesterol, triglycerides or serum IGF-1 between the experimental groups (data not shown). In females, serum leptin levels were significantly increased in the sham/taurine group (2.17 ± 0.40 ng ml−1, P < 0.05, n = 8) and the IUGR/taurine group (2.27 ± 0.80 ng ml−1, n = 8) as compared with the sham group (1.16 ± 0.19 ng ml−1, n = 9). Although serum leptin concentrations in IUGR/taurine females did not differ significantly from IUGR females (2.70 ± 1.50 ng ml−1, n = 11), serum leptin was positively correlated to body weight at 12 weeks of age in this group (r = 0.74, P < 0.01). Serum leptin concentrations did not differ significantly in males (sham, 3.0 ± 0.8 ng ml−1; sham/taurine, 3.8 ± 1.7 ng ml−1; IUGR, 4.1 ± 1.0 ng ml−1; IUGR/taurine, 3.4 ± 0.7 ng ml−1). In male IUGR/taurine animals, serum leptin was not significantly correlated to body weight at 12 weeks of age (data not shown).
Discussion
Two important novel findings are reported in this work. First, taurine supplementation in late gestation results in accelerated postnatal weight and length development, especially in female offspring. Second, an increased postnatal growth rate is associated with adult abdominal obesity and insulin resistance, both in IUGR animals displaying catch-up growth, and in offspring with normal birth weight.
The association between low birth weight and increased risk of developing insulin resistance in adulthood has been the subject of numerous epidemiological studies (Hales et al. 1991; Phipps et al. 1993). This hypothesis has been further supported by animal studies using different experimental models of IUGR (Nyirenda et al. 1998; Ozanne et al. 2005). We employed bilateral uterine artery ligation, a model in which a reduced placental blood flow and transport of nutrients to the fetus results in IUGR with characteristics similar to the majority of clinical cases of IUGR seen in Western countries (Bernstein, 1996).
In the present study, taurine supplementation to the dams increased fetal plasma taurine concentrations twofold and had marked effects on postnatal growth in the offspring. At birth, no weight difference was found between taurine-supplemented and unsupplemented offspring. However, as early as week 1 for females and week 4 for males, animals in sham/taurine groups were significantly heavier than controls and body weight remained elevated (by 14% for females and 13% for males) at 12 weeks of age. Interestingly, a subgroup of IUGR offspring of dams receiving taurine supplementation in late gestation showed complete catch-up growth, in contrast to IUGR animals. The effect of prenatal taurine supplementation was not restricted to increased body weight since sham/taurine animals also were significantly longer. Further, IUGR/taurine rats displaying catch-up growth in weight also demonstrated catch-up in length at 12 weeks of age. These findings are compatible with an important role for fetal taurine levels in determining growth after birth. To the best of our knowledge, this is the first report demonstrating a stimulation of postnatal growth by prenatal taurine supplementation. The effect of maternal taurine supplementation on beta-cell function in the 12-week-old offspring was recently studied in dams fed either a control or low-protein diet (Merezak et al. 2004), and no postnatal growth stimulating effect of taurine supplementation was observed. These differences may be due to the markedly different protocols used for taurine supplementation, which was given throughout pregnancy and lactation in the study of Merezak and coworkers (Merezak et al. 2004), but only during the last 5 days of pregnancy in the current study. Breast milk during the initial stage of lactation, and in particular colostrum, contains high amounts of taurine. Interestingly, rat pups suckling breast milk with reduced concentrations of taurine for the first 5 postnatal days exhibit a slower growth during the first 3 weeks of life (Hu et al. 2000), demonstrating a link between early postnatal taurine availability and growth.
The reason why some of the IUGR/taurine offspring catch-up in weight and others do not is unclear. It is unlikely that the pattern of postnatal growth, i.e. catch-up growth or not, was determined primarily by the dam since the majority of the offspring displaying a particular pattern of growth originated from different litters. We speculate that genetic differences between littermates, as well as between offspring of different litters, may be responsible for the different ability to catch-up in weight postnatally. Also, even offspring in the same litter will be subjected to differences in the intrauterine environment, where, for example, different positions in the uterine horn are associated with varying placental blood flow and transport, which may confer differences in the postnatal growth trajectory. Finally, littermates may exhibit distinct behaviour, which may have consequences for the ability to obtain milk during lactation, which can determine the postnatal growth trajectory.
The mechanisms by which taurine supplementation late in gestation programme the fetus and increase postnatal growth remain to be established. Since the stimulating effect on postnatal growth was evident also for length development, prenatal taurine supplementation may have an effect on GH/IGF-1 axis, the primary endocrine determinant for postnatal longitudinal growth. Indeed, taurine has been shown to stimulate GH secretion in rats (Ikuyama et al. 1988) and in the human (Mantovani & DeVivo, 1979), and the decreased postnatal growth rate in response to low milk taurine levels in the rat is associated with decreased serum levels of IGF-I (Hu et al. 2000). No significant differences were found in plasma IGF-1 levels between the experimental groups in our study at 10 weeks of age; however, it is possible that offspring supplemented with taurine in late gestation had higher IGF-1 levels earlier during postnatal development.
Several epidemiological studies have shown an association between obesity, especially increased abdominal fat, and insulin resistance (Bosello & Zamboni, 2000). In our study, the female sham/taurine group and IUGR/taurine animals showing catch-up growth displayed increased fat depots and insulin resistance, which is consistent with this association. Further, we found increased plasma concentrations of FFA in the female sham/taurine group compared with the sham group. Thus, female sham/taurine offspring had an increased postnatal growth rate and displayed three of the key components of the metabolic syndrome (Alberti et al. 2006) at 12 weeks of age. The mechanisms underlying the insulin resistance in female sham/taurine and IUGR/taurine animals with catch-up growth remain to be fully established, but may be related to the increased levels of plasma FFA and leptin, alterations that are well established to be associated with insulin resistance (Randle, 1998; Wannamethee et al. 2006).
Few experimental studies have specifically addressed the role of postnatal growth rate after IUGR and the risk of developing disease later in life. The impact of early catch-up growth on adult obesity has recently been examined in a rat model of IUGR induced by maternal food restriction (Desai et al. 2005). In that study, growth-restricted offspring cross fostered to normal lactating dams exhibited rapid postnatal growth, increased body weight and percentage body fat in adult life. These results correspond well to our study where rats with high postnatal growth rates possessed significantly larger fat depots. It may be argued that it is taurine supplementation per se, rather than rapid postnatal growth, that causes insulin resistance and increased fat depots in the female sham/taurine and in IUGR/taurine animals showing catch-up growth. However, IUGR/taurine animals that did not display catch-up growth had normal insulin sensitivity and fat depots. These findings support the interpretation that it is the accelerated postnatal growth, rather than the taurine supplementation per se, that may be important for the development of insulin resistance and obesity.
In a previous study, we induced IUGR by uterine artery ligation at gestation day 18 in rat pregnancy, and measured plasma glucose and insulin concentrations prior to and during an intravenous glucose load in the offspring at 12 weeks of age in the awake animal (Jansson & Lambert, 1999). We found that IUGR females, but not males, showed signs of insulin resistance and dysregulation of insulin secretion. However, in the present study when using more advanced clamp techniques to assess insulin secretion and insulin sensitivity in the anaesthetized animal, adult IUGR rats did not differ from control rats. The reasons for this discrepancy in outcome in these two studies from the same laboratory are intriguing but may be due to slight differences in the timing of uterine artery ligation (1 day difference) or be related to the different methodology used to assess insulin sensitivity. Furthermore, Simmons and coworkers have reported that IUGR offspring following uterine artery ligation were obese and insulin resistant at 12 weeks of age (Simmons et al. 2001). Again, the different results between our current study and the report of Simmons et al. may be methodological, but it is interesting to note that IUGR offspring in the Simmons study showed catch-up growth, indirectly supporting the importance of rapid postnatal growth in the association between IUGR and later type 2 diabetes.
In summary, our findings demonstrate that taurine supplementation in late gestation causes accelerated postnatal growth, which results in adult obesity, as evidenced by increased fat depots and insulin resistance, both in IUGR and normal offspring. This effect was particularly evident in females. We propose that the insulin resistance is related to the increased levels of plasma FFA and leptin. Once the mechanisms underlying the programming effects of fetal taurine availability have been identified in animal models, they can be compared with analogous processes in humans. This knowledge will be important in efforts to design effective perinatal interventions to prevent obesity and type 2 diabetes.
Acknowledgments
Supported by grants from the Swedish Research Council (10838 and 12206), the Swedish Diabetes Association, the NovoNordisk foundation, the Åhlens Foundation, the Sven Jerring Foundation, the Frimurare-Barnhus direktionen and the Willhelm and Martina Lundgren Foundation. The authors thank Britt-Marie Larsson for excellent laboratory work throughout the study.
References
- Alberti KG, Zimmet P, Shaw J. Metabolic syndrome – a new world-wide definition. A Consensus Statement from the International Diabetes Federation. Diabet Med. 2006;23:469–480. doi: 10.1111/j.1464-5491.2006.01858.x. [DOI] [PubMed] [Google Scholar]
- Anuradha CV, Balakrishnan SD. Taurine attenuates hypertension and improves insulin sensitivity in the fructose-fed rat, an animal model of insulin resistance. Can J Physiol Pharmacol. 1999;77:749–754. [PubMed] [Google Scholar]
- Barker DJ, Osmond C, Forsen TJ, Kajantie E, Eriksson JG. Trajectories of growth among children who have coronary events as adults. N Engl J Med. 2005;353:1802–1809. doi: 10.1056/NEJMoa044160. [DOI] [PubMed] [Google Scholar]
- Bernstein IGS. Obstetrics. Intrauterine Growth Restriction. New York: Churchill Livingstone; 1996. [Google Scholar]
- Bosello O, Zamboni M. Visceral obesity and metabolic syndrome. Obesity Rev. 2000;1:47–56. doi: 10.1046/j.1467-789x.2000.00008.x. [DOI] [PubMed] [Google Scholar]
- Boujendar S, Arany E, Hill D, Remacle C, Reusens B. Taurine supplementation of a low protein diet fed to rat dams normalizes the vascularization of the fetal endocrine pancreas. J Nutr. 2003;133:2820–2825. doi: 10.1093/jn/133.9.2820. [DOI] [PubMed] [Google Scholar]
- Cetin I, Corbetta C, Sereni LP, Marconi AM, Bozzetti P, Pardi G, Battaglia FC. Umbilical amino acid concentrations in normal and growth-retarded fetuses sampled in utero by cordocentesis. Am J Obstet Gynecol. 1990;162:253–261. doi: 10.1016/0002-9378(90)90860-a. [DOI] [PubMed] [Google Scholar]
- Cherif H, Reusens B, Ahn MT, Hoet JJ, Remacle C. Effects of taurine on the insulin secretion of rat fetal islets from dams fed a low-protein diet. J Endocrinol. 1998;159:341–348. doi: 10.1677/joe.0.1590341. [DOI] [PubMed] [Google Scholar]
- Dahlgren J, Nilsson C, Jennische E, Ho HP, Eriksson E, Niklasson A, Bjorntorp P, Albertsson Wikland K, Holmang A. Prenatal cytokine exposure results in obesity and gender-specific programming. Am J Physiol Endocrinol Metab. 2001;281:E326–E334. doi: 10.1152/ajpendo.2001.281.2.E326. [DOI] [PubMed] [Google Scholar]
- Desai M, Gayle D, Babu J, Ross MG. Programmed obesity in intrauterine growth-restricted newborns: modulation by newborn nutrition. Am J Physiol Regul Integr Comp Physiol. 2005;288:R91–R96. doi: 10.1152/ajpregu.00340.2004. [DOI] [PubMed] [Google Scholar]
- Eriksson JG, Forsen TJ. Childhood growth and coronary heart disease in later life. Ann Med. 2002;34:157–161. [PubMed] [Google Scholar]
- Forsen T, Eriksson J, Tuomilehto J, Reunanen A, Osmond C, Barker D. The fetal and childhood growth of persons who develop type 2 diabetes. Ann Intern Med. 2000;133:176–182. doi: 10.7326/0003-4819-133-3-200008010-00008. [DOI] [PubMed] [Google Scholar]
- Hales CN, Barker DJ, Clark PM, Cox LJ, Fall C, Osmond C, Winter PD. Fetal and infant growth and impaired glucose tolerance at age 64. BMJ. 1991;303:1019–1022. doi: 10.1136/bmj.303.6809.1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen SH. The role of taurine in diabetes and the development of diabetic complications. Diabetes Metab Res Rev. 2001;17:330–346. doi: 10.1002/dmrr.229. [DOI] [PubMed] [Google Scholar]
- Hibbard JU, Pridjian G, Whitington PF, Moawad AH. Taurine transport in the in vitro perfused human placenta. Pediatr Res. 1990;27:80–84. doi: 10.1203/00006450-199001000-00021. [DOI] [PubMed] [Google Scholar]
- Hofman PL, Cutfield WS, Robinson EM, Bergman RN, Menon RK, Sperling MA, Gluckman PD. Insulin resistance in short children with intrauterine growth retardation. J Clin Endocrinol Metab. 1997;82:402–406. doi: 10.1210/jcem.82.2.3752. [DOI] [PubMed] [Google Scholar]
- Holmang A, Svedberg J, Jennische E, Bjorntorp P. Effects of testosterone on muscle insulin sensitivity and morphology in female rats. Am J Physiol Endocrinol Metab. 1990;259:E555–E560. doi: 10.1152/ajpendo.1990.259.4.E555. [DOI] [PubMed] [Google Scholar]
- Hu JM, Rho JY, Suzuki M, Nishihara M, Takahashi M. Effect of taurine in rat milk on the growth of offspring. J Vet Med Sci. 2000;62:693–698. doi: 10.1292/jvms.62.693. [DOI] [PubMed] [Google Scholar]
- Huxtable RJ. Physiological actions of taurine. Physiol Rev. 1992;72:101–163. doi: 10.1152/physrev.1992.72.1.101. [DOI] [PubMed] [Google Scholar]
- Ikuyama S, Okajima T, Kato K, Ibayashi H. Effect of taurine on growth hormone and prolactin secretion in rats: possible interaction with opioid peptidergic system. Life Sci. 1988;43:807–812. doi: 10.1016/0024-3205(88)90506-1. [DOI] [PubMed] [Google Scholar]
- Jansson T, Lambert GW. Effect of intrauterine growth restriction on blood pressure, glucose tolerance and sympathetic nervous system activity in the rat at 3–4 months of age. J Hypertens. 1999;17:1239–1248. doi: 10.1097/00004872-199917090-00002. [DOI] [PubMed] [Google Scholar]
- Lindroth PMK. High performance liquid chromatographic determination of subpicomole amounts of amino acids by precolumn fluorescence derivatization with o-phthaldialdehyde. Anal Chem. 1979;51:1667–1674. [Google Scholar]
- Mantovani J, DeVivo DC. Effects of taurine on seizures and growth hormone release in epileptic patients. Arch Neurol. 1979;36:672–674. doi: 10.1001/archneur.1979.00500470042006. [DOI] [PubMed] [Google Scholar]
- Merezak S, Reusens B, Renard A, Goosse K, Kalbe L, Ahn MT, Tamarit-Rodriguez J, Remacle C. Effect of maternal low-protein diet and taurine on the vulnerability of adult Wistar rat islets to cytokines. Diabetologia. 2004;47:669–675. doi: 10.1007/s00125-004-1357-z. [DOI] [PubMed] [Google Scholar]
- Murakami S, Kondo Y, Toda Y, Kitajima H, Kameo K, Sakono M, Fukuda N. Effect of taurine on cholesterol metabolism in hamsters: up-regulation of low density lipoprotein (LDL) receptor by taurine. Life Sci. 2002;70:2355–2366. doi: 10.1016/s0024-3205(02)01507-2. [DOI] [PubMed] [Google Scholar]
- Nakaya Y, Minami A, Harada N, Sakamoto S, Niwa Y, Ohnaka M. Taurine improves insulin sensitivity in the Otsuka Long-Evans Tokushima Fatty rat, a model of spontaneous type 2 diabetes. Am J Clin Nutr. 2000;71:54–58. doi: 10.1093/ajcn/71.1.54. [DOI] [PubMed] [Google Scholar]
- Nilsson C, Niklasson M, Eriksson E, Bjorntorp P, Holmang A. Imprinting of female offspring with testosterone results in insulin resistance and changes in body fat distribution at adult age in rats. J Clin Invest. 1998;101:74–78. doi: 10.1172/JCI1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norberg S, Powell TL, Jansson T. Intrauterine growth restriction is associated with a reduced activity of placental taurine transporters. Pediatr Res. 1998;44:233–238. doi: 10.1203/00006450-199808000-00016. [DOI] [PubMed] [Google Scholar]
- Nyirenda MJ, Lindsay RS, Kenyon CJ, Burchell A, Seckl JR. Glucocorticoid exposure in late gestation permanently programs rat hepatic phosphoenolpyruvate carboxykinase and glucocorticoid receptor expression and causes glucose intolerance in adult offspring. J Clin Invest. 1998;101:2174–2181. doi: 10.1172/JCI1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ong KK, Petry CJ, Emmett PM, Sandhu MS, Kiess W, Hales CN, Ness AR, Dunger DB. Insulin sensitivity and secretion in normal children related to size at birth, postnatal growth, and plasma insulin-like growth factor-I levels. Diabetologia. 2004;47:1064–1070. doi: 10.1007/s00125-004-1405-8. [DOI] [PubMed] [Google Scholar]
- Ozanne SE, Jensen CB, Tingey KJ, Storgaard H, Madsbad S, Vaag AA. Low birthweight is associated with specific changes in muscle insulin-signalling protein expression. Diabetologia. 2005;48:547–552. doi: 10.1007/s00125-005-1669-7. [DOI] [PubMed] [Google Scholar]
- Phipps K, Barker DJ, Hales CN, Fall CH, Osmond C, Clark PM. Fetal growth and impaired glucose tolerance in men and women. Diabetologia. 1993;36:225–228. doi: 10.1007/BF00399954. [DOI] [PubMed] [Google Scholar]
- Ramamoorthy S, Leibach FH, Mahesh VB, Han H, Yang-Feng T, Blakely RD, Ganapathy V. Functional characterization and chromosomal localization of a cloned taurine transporter from human placenta. Biochem J. 1994;300:893–900. doi: 10.1042/bj3000893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Randle PJ. Regulatory interactions between lipids and carbohydrates: the glucose fatty acid cycle after 35 years. Diabetes Metab Rev. 1998;14:263–283. doi: 10.1002/(sici)1099-0895(199812)14:4<263::aid-dmr233>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- Sandberg M, Butcher SP, Hagberg H. Extracellular overflow of neuroactive amino acids during severe insulin-induced hypoglycemia: in vivo dialysis of the rat hippocampus. J Neurochem. 1986;47:178–184. doi: 10.1111/j.1471-4159.1986.tb02847.x. [DOI] [PubMed] [Google Scholar]
- Simmons RA, Templeton LJ, Gertz SJ. Intrauterine growth retardation leads to the development of type 2 diabetes in the rat. Diabetes. 2001;50:2279–2286. doi: 10.2337/diabetes.50.10.2279. [DOI] [PubMed] [Google Scholar]
- Terrettaz J, Jeanrenaud B. In vivo hepatic and peripheral insulin resistance in genetically obese (fa/fa) rats. Endocrinology. 1983;112:1346–1351. doi: 10.1210/endo-112-4-1346. [DOI] [PubMed] [Google Scholar]
- Wannamethee SG, Tchernova J, Whincup P, Lowe GD, Kelly A, Rumley A, Wallace AM, Sattar N. Plasma leptin: associations with metabolic, inflammatory and haemostatic risk factors for cardiovascular disease. Atherosclerosis. 2006 doi: 10.1016/j.atherosclerosis.2006.04.012. in press. [DOI] [PubMed] [Google Scholar]
- Zimmet P, Alberti KG, Shaw J. Global and societal implications of the diabetes epidemic. Nature. 2001;414:782–787. doi: 10.1038/414782a. [DOI] [PubMed] [Google Scholar]