Abstract
Introduction
There has been increasing interest in the use of medications that affect the dopamine receptor in the treatment of alcoholism. Aripiprazole has the unique pharmacology of being a partial dopamine agonist serving to stabilize brain dopamine systems in both frontal cortical and sub-cortical areas. As such, it might act to dampen alcohol reinforcement and craving and/or alter control over alcohol use. The current clinical laboratory study was conducted to evaluate the safety and efficacy of aripiprazole as a potential agent to alter drinking and objective effects of alcohol.
Methods
Thirty non-treatment seeking alcoholics were enrolled in a sub-acute human laboratory study and received double-blind treatment with up to 15mg of aripiprazole (n=15) or identical placebo (n=15) for eight days. Tolerability and utility of aripiprazole was monitored during natural drinking over the first six days of medication treatment and also during a free-choice limited-access alcohol consumption paradigm following an initial drink of alcohol in a bar-lab setting on day 8.
Results
Aripiprazole was well-tolerated and reduced drinking in non-treatment seeking alcoholics over 6 days of natural drinking – especially in those with lower self control (more impulsive). It also reduced drinks in the bar-lab after a priming drink and broke the link between priming drink induced stimulation and further drinking. During the bar-lab drinking session, there were no differences in subjective high, intoxication, or craving between subjects treated with aripiprazole or placebo.
Discussion
This study joins several others in demonstrating the utility of sub-acute dosing laboratory paradigms for evaluating medication effects in alcoholics. Aripiprazole was well-tolerated and lowered alcohol use, especially in those with lower impulse control. Further study is needed to determine the safety and utility of aripiprazole in the treatment of alcoholism and if sub-groups of alcoholics are more likely to respond.
Keywords: Alcohol, Aripiprazole, Laboratory, Treatment
INTRODUCTION
A basic assumption is that the mechanisms by which medications reduce drinking have a neurobehavioral basis (Anton and Swift, 2003), which is underpinned by brain chemistry (Koob and Roberts, 1999); (Koob and Le Moal, 2001); (Weiss and Porrino, 2002). One of the most consistent and compelling alcohol effects on the brain is stimulation of dopamine release in the nucleus accumbens/ventral striatum (Gonzales and Weiss, 1998); (Katner and Weiss, 1999); (Middaugh et al., 2003). Of interest, not only does alcohol itself stimulate dopamine release but dopamine is also elevated by stimuli (cues) associated with alcohol consumption (as modeled by place conditioning in rodents). This effect might underlie alcohol expectancy or cue-induced craving in humans. Furthermore, naltrexone has been found to reduce this cue-induced dopamine release and drinking in mice (Middaugh and Bandy, 2000), leading to the hypothesis that blunting of nucleus accumbens dopamine release could form a neural basis for the effectiveness of medications that reduce craving and prevent relapse.
More recently, there has been increasing focus on the role of frontal cortical brain areas in relationship to alcohol cue salience and craving. Data from our group have shown that the anterior cingulate as well as ventral striatum is markedly activated by alcohol cues in non-treatment seeking alcoholics (Myrick et al., 2004; Myrick et al., 2008). Others have found similar cortical activation by alcohol cues in treatment seeking alcoholics (Grüsser et al., 2004). Koob and Le Moal (Koob and Le Moal, 2001) have postulated that a hypo-dopaminergic state might underlie an allostatic shift in brain function caused by chronic heavy alcohol use. This allostatic shift might manifest during the early abstinence period, leading to craving and the drive to normalize brain dopamine levels by drinking. Basic scientists have begun to focus on alcohol/dopamine effects in the frontal cortex (Lapish et al., 2006). It appears certain that dopamine levels regulate gating of impulses that can be affected by both GABA and glutamate (both of which are very sensitive to alcohol). It is generally thought that the frontal cortex, including the anterior cingulate, is involved with both assessment of the salience of environmental stimuli (cues) but also in modulation of behavioral and emotional response to those cues (Bechara, 2005). For instance, it has been found that in several conditions where impulse control is limited, such as in attention deficit disorder (Seidman et al., 2005), sociopathy (Anckarsater, 2006) depression (Rogers et al., 2004), and schizophrenia (Pantelis et al., 2005) that there is a general reduction in activation/functioning of the frontal lobe. It is intriguing to speculate that in alcoholics, either by predisposition (Tapert et al., 2003)or secondary to chronic alcohol effects (Rosenbloom et al., 2003), that frontal lobe functioning is subnormal, leading to a heightened awareness of alcohol cues, a reduction of impulse control (Lyvers, 2000)and a decisional balance biased towards drinking in response to those cues (Bechara, 2005). Reduced frontal lobe function could lead to increased relapse risk during the initial stages of “recovery”. Given that dopamine plays a crucial part in the regulation and focus of the frontal lobes, a medication that affects dopamine in this area might be novel and useful in the treatment for alcohol dependence. Aripiprazole appears to possess a unique pharmacology on the dopamine system that might offer such a benefit.
Aripiprazole, a drug that has been approved by the FDA for the treatment of schizophrenia (DeLeon et al., 2004); (Marder et al., 2003) and which shows good efficacy in bipolar disorder (Keck et al., 2006), has a very unique pharmacology. A number of studies have confirmed that aripiprazole acts as a dopamine stabilizer (Lawler et al., 1999) alone, and also may act to “stabilize” dopamine-serotonin balance (Burris et al., 2002). Its pharmacology is vastly different than other dopamine antagonists that have been found to be useful as antipsychotics. Aripiprazole is thought to be a partial dopamine agonist in that it blocks dopamine at higher dopamine concentrations and augments/releases pre-frontal cortex dopamine at lower dopamine concentrations. This might occur through a unique mechanism of post-synaptic antagonism (reducing dopamine effects) and pre-synaptic antagonism (removing the inhibition to dopamine release). Either through this mechanism, or through direct effects on dopamine receptors conditioned on the amount of endogenous dopamine binding, the net effect is to keep dopamine “balanced”. The resultant clinical utility is that aripiprazole allows for specific beneficial behavioral effects without causing non-specific adverse effects such as extrapyramidal (EPS) effects, or tardive dyskinesia (Marder et al., 2003). In schizophrenia, it has effects both on positive symptoms (ostensibly through dopamine-lowering effects in the meso-limbic system) and negative symptoms (ostensibly through dopamine elevating effects in the meso-cortical system) while not affecting the nigro-striatal system (minimal to no EPS effects) or the hypothalamic pituitary endocrine system (no prolactin elevation). In animals, at lower doses, aripiprazole increases dopamine release in pre-cortical areas but, at slightly higher doses, reduces dopamine release in the nucleus accumbens (Li et al., 2004). This is a unique pharmacological profile for an antipsychotic agent. Moreover, this unique combination lends itself for potential use in addiction. Other, less-novel, dopamine antagonists (especially D2 receptor acting agents) have had too many adverse effects (e.g. EPS, weight gain etc.) to be useful in addictive disorders. The relative freedom from the more troubling adverse effects makes aripiprazole a medication worth considering on both theoretical and practical grounds.
Given the effects of addictive substances, including alcohol, on ventral striatum/nucleus accumbens dopamine release and frontal cortical dopamine effects, a drug like aripiprazole could hold great promise as a potential agent to reverse or block these effects. In fact, there have been a few small open label studies which suggest that aripiprazole was efficacious in reducing cocaine (Beresford et al., 2005) use, attenuating the effects of amphetamine challenge (Lile et al., 2005) and reducing alcohol use (Warsi et al., 2005) in humans. Animal studies have also shown that it could markedly reduce cocaine reinstatement (Feltenstein et al., 2007) and to a lesser degree, alcohol use (Ingman et al., 2006). A multisite trial of aripiprazole in treatment-seeking alcohol dependent individuals suggested that its side effect profile, especially at doses above 15mg per day, may have limited its effectiveness on maintaining abstinence but that it might be useful in reducing drinking if a person did initiate drinking (Anton et al., 2008). A recent clinical lab study reported that in “moderate drinkers” a one-time dose of 10 mg of aripiprazole reduced stimulation and euphoria after alcohol consumption but increased sedation (Kranzler et al., 2008).
The current study attempted to understand aripiprazole's tolerability and explore its possible mechanisms of action on alcohol consumption in a well-defined clinical laboratory paradigm (Anton et al., 2004b; Drobes et al., 2004) in non-treatment seeking alcoholics. We also wished to explore the hypothesis that those with low self-control, as an aspect of trait impulsivity, might have a greater aripiprazole response in this paradigm.
MATERIALS AND METHODS
Participants
Thirty non-treatment seeking alcoholics between the ages of 21 and 65 participated in this study. Potential participants, recruited through newspaper and community ads based on drinking at least 20 drinks per week, were told that the study was investigating effects of a medication that may have beneficial effects for alcoholics in treatment. All participants met DSM-IV criteria for alcohol dependence (APA, 1994), including loss of control drinking or an inability to cut down or quit, but they denied any active involvement in, or desire for, alcohol treatment. All participants stated that they mostly drink in bars but have, in the past, also drank on their own.
Exclusion criteria were as follows: current DSM-IV criteria for drug dependence (excluding nicotine) by verbal report and urine drug screens, other major DSM-IV Axis I disorders, psychoactive medication or substance use (except marijuana) in the past 30 days or a positive urine drug screen, current suicidal or homicidal ideation, past history of alcohol-related medical illness, liver enzymes ≥ 2.5 times above normal, or significant health problems. All DSM-IV criteria were assessed by the Structured Clinical Interview for the DSM-IV Axis 1 Disorders (SCID) (First et al., 1997).
Procedures
Upon arrival for the first session, the study was described in detail to the participant and Informed Consent was obtained using a form and procedures approved by the Investigational Review Board at our institution. Each participant was then evaluated with a number of standard interview, questionnaire, and medical diagnostic procedures similar to those in other studies reported by our group (Drobes et al., 2003);(Anton et al., 2004a). Interview procedures included a demographic form, the alcohol and drug section of the SCID (First et al., 1997) administered by a trained physician, and a timeline follow-back interview to quantify drinking during the preceding 90 days (Sobell and Sobell, 1992). The Obsessive-Compulsive Drinking Scale (OCDS); (Anton RF et al., 1996), the Self-Administered Alcohol Screening Test (SAAST); (Davis et al., 1987), The Barratt Impulsiveness Scale (BIS); (Patton et al., 1995) and the Alcohol Dependence Scale (ADS); (Skinner and Allen, 1982) were administered. Finally, a urine specimen was collected to screen for abused drugs, and a blood sample collected for liver function and general health screening. Additional assessments were conducted at a second session (conducted within one week of the first session), including psychiatric sections of the SCID, the Alcohol Craving Questionnaire (ACQ); (Singleton et al., 1999). In addition, a physical exam was conducted by a physician assistant and reviewed by a physician.
As one aim of the study was to assess the safety and tolerability of the interaction between aripiprazole and alcohol, several scales were employed to measure such effects. We were primarily interested in the negative clinical effects of aripiprazole and alcohol on the central nervous system that included impaired cognition, confusion, fatigue, and sedation. The following scales allowed for an assessment of these dimensions and others. The Profile of Mood States (POMS); (McNair et al., 1981) were administered at baseline and on Day 6. Side effects (physical and psychological) were rated at baseline and Day 6 using a symptom checklist listing 21 symptoms (e.g., nausea/vomiting, headache, irritability, rash, energy, and sex drive) used in prior research trials by our group (Anton et al., 1999); (Drobes et al., 2003); (Anton et al., 2005). The symptom checklist monitors the presence (yes or no) and severity of symptoms (mild, moderate or severe). In addition, several scales were employed during the bar-lab setting to identify effects of aripiprazole on acute alcohol effects and craving: the Biphasic Alcohol Effects Scale (BAES; (Martin et al., 1993), Subjective High Assessment Scale (SHAS); (Schuckit, 1984)), and Alcohol Urge Questionnaire (AUQ); (Bohn et al., 1995). The BAES assesses sedation and stimulation whereas the SHAS assesses untoward effects such as intoxication, clumsiness, confusion, dizziness, nausea, sedation, and difficulty concentrating. All of the above assessments are administered as paper and pencil questionnaires with the oversight of study staff.
Participants who passed all screening and eligibility criteria were randomly assigned to receive aripiprazole (n=15) in 5mg or identical placebo capsules (n=15) in a double blind fashion for eight days. The following dose titration for aripiprazole or matching placebo was employed: 5 mg/day for the first day, 10mg/day for the next two days, and 15 mg/day for days 4th through 8th. Medication ingestion was witnessed on the first day and days 7 and 8. All medications, including inactive placebo, were blister packed and administered in standard gel caps with 25 mg riboflavin added to assess for compliance via a laboratory based urinary fluorescence assay. Urine samples were obtained and assessed for riboflavin at baseline and Day 7. Samples showing greater than 1500 ng/ml of riboflavin on day 7 were considered compliant (Anton, 1996). All but one subject were deemed compliant by these measures and all subjects were used in the analysis.
Participants were given no explicit instructions regarding use of alcohol or modification of their drinking behavior for Days 1 − 6. However, they were required to abstain completely from drinking on Days 7 and the morning of day 8. On Day 7, several assessments were completed. Participants were clinically evaluated for alcohol withdrawal using the Clinical Institute Withdrawal Assessment for Alcohol - Revised (CIWA-Ar); (Sullivan et al., 1989), and a urine sample was collected to ascertain riboflavin levels for medication compliance. A 6-Day version of the timeline follow-back interview (in which they reported their alcohol consumption since the outset of the medication period) was also completed. The symptom checklist, the POMS, and AUQ were repeated. Participants were also familiarized with the bar-lab room in which the alcohol consumption assessment would be conducted the following day. However, alcohol-specific cues (bar, barstools, alcohol bottles on shelves, signs, etc.) were shielded from the participants' view by a curtain.
On Day 8, each participant completed a session in the bar-lab setting, including a standard alcohol administration and an alcohol choice self-administration period in the presence of only a research assistant. Participants arrived at the study site at 11:30 am and ingested the last dose of study medication. They were provided with a standard caloric lunch (weight and gender adjusted). At 2:00 pm, the curtain was pulled back to reveal the bar-like cues described above (indicating the bar “was open”) and they consumed a standard dose of spirits (vodka, gin, rum or bourbon) calculated to achieve a breath alcohol concentration (BAC) level of about 30mg% (0.3 gm/L) using a formula which takes into account each participant's total body water derived from calculating the individual's gender, age, height and weight (Watson et al. 1989). The alcohol was diluted in a fruit juice of the subject's choosing at a dilution of 3 parts fruit juice to 1 part spirits. The beverage was consumed in the bar-like setting over a 5-minute interval. All subjects rinsed their mouth with water several times after consuming the initial drink and before serial BAC measurements at 10, 20, and 30 minutes after the end of the initial drink consumption. Subjects also filled out the Biphasic Alcohol Effects Scale (BAES), Subjective High Assessment Scale (SHAS), and Alcohol Urge Questionnaire (AUQ) immediately prior to the BAC measurements. At 40 minutes after the initial drink participants were brought the first tray of 4 “mini-drinks” (each consisting of 1/2 the alcohol consumed in the initial drink) to which they had free access and were told that they could consume as many as they desired over the next hour period. A second tray of 4 “mini-drinks” was then provided to which they again had free access and they could drink freely over the second hour. A BAC assessment was done at the end of the hour of free access. The intent was to try to keep the 2 one-hour blocks of alcohol consumption as natural as possible.
As per our previous paradigms (Drobes et al., 2003); (Anton et al., 2004b) and that of O'Malley (O'Malley et al., 2002), we chose to assess alcohol consumption in the context of an alternative reinforcer. Therefore, each subject was told that they had a “bar-credit” of $24 that they could keep (payment at the end of the session) if they did not drink at all, but they would be “charged” $3 for each “mini-drink” they consumed during the free-access period. A “tally-sheet” was laid on the bar and they moved a pushpin for each drink they consumed. The participant also had snacks and water available during this period. The snacks available to subjects included potato chips (28.3 gram individual bags). These snacks were provided to simulate a natural bar setting and provided face validity in our past studies (Drobes et al., 2003), (Anton et al., 2004b). Snack ingestion was limited, participants could have only two bags of snacks total or one bag per tray and snacks were only available during the free choice drinking period and not during the priming drink assessments.
Following the alcohol choice self-administration period, all participants were required to remain in the laboratory until 10:00 pm. Prior to dismissal, the participant was given dinner and permitted to listen to music, watch videos, or read magazines. Participants were also given educational and self-help materials regarding alcohol effects and motivation to change drinking behavior. All subjects had a breathalyzer reading below 20 mg% at the time of departure and either were driven home by a friend or family member, or a taxi was provided. On the day following the laboratory session, the participant attended an individual feedback and debriefing session designed to educate participants on the negative physical and social effects of drinking as well as increase motivation to reduce drinking or seek treatment. This semi-structured session, conducted by a trained physician/counselor, utilized aspects of Motivational Enhancement Therapy (MET); (Miller et al., 1992) and the Guided Self-Change approach e.g., (Sobell et al., 1996) that has been found previously by us to decrease drinking in the months after subject participation (Drobes and Anton, 2000). Participants were paid $300 by check approximately one week after study completion.
Data Analysis
Analysis of covariance (ANCOVA) was used to examine differences in the number of drinks consumed during the natural environment drinking period, as well as the number of drinks consumed during the limited access alcohol consumption portion of the bar-lab session, as a function of medication status adjusting for the number of drinks consumed in the 5 days before medication. One-way ANOVA and a multivariate analysis of variance were used to examine differences in the Profile of Mood States total and subscale scores by medication condition. A two-way ANOVA was used to evaluate the interaction of “self-control” (below or above the median BIS score) and medication group (aripiprazole and placebo) on drinks per day during the natural environment drinking period. Repeated measures analysis of variance (ANOVA) was used to examine medication (aripiprazole vs. placebo) effects on blood alcohol levels achieved, the SHAS total scores, AUQ and BAES subscales, with Time following the priming drink during the bar-lab procedure used as the repeated measure. Finally, χ2 analyses were used to evaluate potential differences in side effects between medication groups.
RESULTS
Demographics and Baseline Drinking Parameters
Thirty non-treatment seeking alcoholics received either aripiprazole (n= 15, 13 males and 2 females, mean age 26) or identical placebo (n=15, 12 males 3 females, mean age 29). As can be seen in Table 1 there were no significant baseline differences in demographics or alcohol use parameters between the medication groups. Subjects drank an average of 9.8 drinks per drinking day on 72% of days in the past 90 days prior to randomization.
Table 1.
Demographics, drinking history and severity of non-treatment seeking alcoholics*. (No significant differences between groups).
| Aripiprazole N=15 | Placebo N=15 | |
|---|---|---|
| Mean Age (SD) | 29.0 (±10.6) | 25.5 (±8.9) |
| Gender (%Male) | 80% | 86% |
| Race: | ||
| Caucasian | 46% | 44% |
| African-American | 6.7% | 0% |
| Native-American | 0% | 3.3% |
| Years of Education | 15.47 (±1.46) | 14.73 (±1.71) |
| Marital Status (% Married) | 20% | 6.6% |
| Drinks per Day-previous 90 (SD) | 6.0 (±1.8) | 7.7 (±4.6) |
| Drinks/Drinking Day-previous 90 (SD) | 9.7 (±5.2) | 10.0 (±4.6) |
| OCDS (SD) | 17.5 (±4.4) | 19.2 (±9.8) |
| Alcohol Dependence Scale (SD) | 10.9 (±3.5) | 13.6 (±7.8) |
| Percent Days Abstinent (SD) | 32% (±20) | 24% (±19) |
Adverse Events, Mood and Sleep
All subjects completed the 8-day study. Aripiprazole subjects experienced more insomnia (p=0.017) and nervousness (p=0.042), mostly of a mild to moderate nature (see Table 2). These were expected findings and consistent with observations in clinical trials. There were no overall significant differences between the groups across the POMS scales except for a tendency for aripiprazole-treated individuals to have a higher tension subscale score (F=3.99 and sig. =.056).
Table 2.
Adverse events reported.
| ARIPIPRAZOLE | PLACEBO | |||||||
|---|---|---|---|---|---|---|---|---|
| N=15 | N=15 | |||||||
| Mild | Moderate | Severe | Mild | Moderate | Severe | P1 | P2 | |
| Trouble sleeping | 5 | 7 | 1 | 6 | 0 | 1 | 0.003 | 0.017 |
| Daytime Sleepiness | 5 | 6 | 4 | 11 | 2 | 0 | 0.005 | 0.088 |
| Nervousness | 6 | 1 | 0 | 0 | 2 | 0 | 0.007 | 0.042 |
| Feeling Depressed | 0 | 0 | 0 | 2 | 1 | 0 | 0.106 | 0.034 |
| Constipation | 3 | 1 | 0 | 0 | 0 | 0 | 0.046 | 0.013 |
P1=Overall chi-squared for unequal distribution among the 4 alternative responses
P2=Overall chi-squared for presence or absence of symptom
Effects During the 6-Day Natural Observation Period
During the 6 days under natural drinking conditions, prior to the lab experiment, aripiprazole treated subjects had fewer drinks per day (5.4 aripiprazole versus 7.7 placebo p=0.06) and greater percent days of total abstinence (30% aripiprazole versus 17% placebo p=0.045) (Figure 1).
Figure 1.
Drinks per day and percent days abstinent during the 6-Day natural observation period.
Bar Lab Paradigm Data
We did not find a significant difference in peak blood alcohol concentration (BAC) after the first (priming) drink between the medication groups. The aripiprazole group had a peak BAC 0.022 (SD=0.009) and placebo group a peak BAC of 0.021 (SD=0.009) (F=.306, sig. = 0.585). We did not find any difference on overall BAES stimulation or sedation between the aripiprazole and placebo groups after the first (priming) drink of alcohol in the bar-lab paradigm on day 8. The aripiprazole group had BAES stimulation and sedation scores of 9.7 (SD=10.7) and 7.6 (SD=9.1), while the placebo group had scores of 9.4 (SD=10.3) (p=0.93) and 9.0 (SD=11) (p=0.71).
After the priming drink in the bar lab, there was no significant difference between placebo (mean score 47.2 (SD=50.9)) and aripiprazole (mean score 51.4 (SD=32.2)) groups on the Subjective High Assessment Scale (SHAS) which measures various parameters of intoxication and alcohol induced adverse effects (p=0.79). This supports data that drinking alcohol while ingesting aripiprazole does not seem to intensify clinically meaningful effects of alcohol.
We also did not find any difference on overall Alcohol Urge Questionnaire (AUQ) scores between the aripiprazole and placebo groups after the first (priming) drink of alcohol in the bar-lab paradigm. The aripiprazole group had AUQ scores of 23.6 (SD=8.1), while the placebo group had scores of 23.1(SD=14.6) (p=0.91). However there was a significant difference in AUQ scores on the follow-up (day 8) visit. The AUQ score on Day 8 for the aripiprazole and placebo groups were 12.2 (SD=5.2) and 19.1 (SD=11.5) (p=0.047) respectively.
After the first/priming drink in the bar-lab paradigm, 53% of placebo-treated subjects who opted to drink, drank an average of 5.4 drinks while the 60% of aripiprazole treated subjects who opted to drink, drank 3.2 drinks (p=0.12).
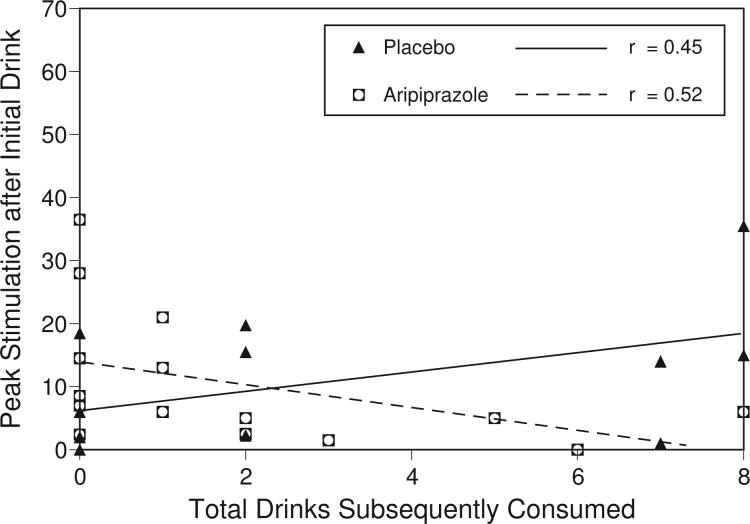
We observed a disassociation between priming drink-induced stimulation and subsequent drinking (see Figure 2). Aripiprazole-treated subjects had a negative correlation between amount of priming drink-induced stimulation and the number of drinks subsequently consumed. This is in marked contrast to the placebo-treated subjects who had a strong positive correlation between priming drink-induced stimulation and subsequent drinks consumed (p=0.008 for the interaction of maximum stimulation and medication group on amount of drinks consumed).
Figure 2.
Relationship between maximum BAES stimulation after the initial (priming) drink and subsequent drinking.
Baseline Impulsivity: Interaction with Aripiprazole
Given aripiprazole's unique effects on frontal lobe dopamine, we wished to examine the differential effects of aripiprazole on alcohol consumption based on a individual's reported level of self-control /impulsivity. Therefore, individuals were categorized based on whether they scored above (n=15) or below (n=15) the median on the self-control subscale of the Barratt's Impulsiveness Scale (Patton et al, 1995) at entry into the study. Ratings on the self-control subscale significantly interacted with medication effect on heavy drinking during the 6-day natural observation period. Those who had lower self-control (more impulsivity) compared to those who had higher self-control (less impulsivity) were more likely to have fewer drinks per day during the natural-observation period when on aripiprazole compared to placebo (p=0.03) (see Figure 3). This suggests that aripiprazole might be particularly useful in those who have high impulsivity/low self control especially in real-world/natural drinking conditions.
Figure 3.
Drinks per day during a natural observation period depending on trait “self-control” (as measured by the Barratt Impulsiveness Scale). Interaction between medications and level of “self-control” (p=0.03). Note: Pre-study drinking was similar. Sample size was 15 subjects in the low and 15 individuals in high “impulsivity” groups.
DISCUSSION
Aripiprazole appeared to be safe and well-tolerated in the current study. The fact that subjects reported minimal side effects despite a rather rapid titration of dose, and that no one discontinued the medication, further supports the safety and tolerability of aripiprazole in heavily drinking individuals. The side effects that were reported here were similar to those reported in a clinical trial of aripiprazole in treatment-seeking alcoholics (Anton et al., 2008), but perhaps at a lower intensity in this study since a maximum daily dose of 15 mg was used versus a maximum daily dose of 30 mg in the clinical trial.
The main findings of this study were that aripiprazole reduced the amount of drinking (reducing drinks per day and increasing days abstinent) during a natural observation period and tended to reduce drinks in the bar-lab after an initial priming drink. Interestingly, while placebo-treated subjects had a positive relationship between the amount of stimulation engendered by a priming drink and subsequent drinking, aripiprazole-treated subjects had the opposite effect, the more stimulation after the priming drink the less alcohol subsequently consumed. This is very similar to a finding previously reported by our group with naltrexone (Anton et al., 2004a). However, we did not find any overall aripiprazole-induced decrease in stimulation, or increase in sedation, as reported by Kranzler and colleagues (Kranzler et al., 2008). Kranzler also reported a reduction in alcohol-induced euphoria after alcohol but did not relate the reduction in stimulation to this effect. Also, in our study BAC levels (0.02 g/L range) were quite a bit lower than those of Kranzler et. al. (0.06 g/L range). Other differences between the studies include our use of heavier drinking non-treatment seeking alcoholics versus “moderate drinkers”, a higher dose of aripiprazole (15mg) over 8 days versus a single effective dose (10mg) given one time. Any of these differences could account for differential effects of aripiprazole on alcohol-induced stimulation or sedation between the studies.
Of interest was our finding that aripiprazole reduced drinking in those with lower self-control during the natural observation period. Since dopamine abnormalities, particularly in the frontal lobe, might underlie impulsive responses, a feature of attention deficit disorder, schizophrenia, and also addiction, the potential ability of aripiprazole to stabilize dopamine, particularly in the frontal cortex, might underlie its enhanced effectiveness in those with less self-control. It is possible that those individuals with lower self-control when confronted with a choice to drink or to engage in other behaviors will more easily give in to drinking or to continue drinking. Aripiprazole could alter this balance in favor of a more realistic appraisal, leading to a delay and/or a reduction of drinking as suggested by our data. However, since we did not randomize individuals to treatment based on their level of self-control/ impulsivity, the relationship between low self-control and aripiprazole response should be considered a tentative finding needing further exploration and replication.
Similar to naltrexone, we observed that aripiprazole had the ability to break the link between cognitive stimulation induced by low BAC and the drive to consume more alcohol. Interestingly, similar to naltrexone, we did not observe this effect for sedation. It is tempting to speculate that breaking the link between alcohol-induced stimulation and subsequent alcohol ingestion is related to the reported “dopamine lowering or stabilizing effects” of these medications. It is also tempting, but speculative, to suggest that the stimulatory effect of alcohol might drive further drinking (perhaps by sensitized mechanisms) and breaking the link between stimulation and drinking might be a potentially useful construct to predict clinical effectiveness of medications to treat alcohol dependence.
In summary we have shown that the dopamine stabilizer aripiprazole at doses up to 15 mg reduced drinking over a 6 Day natural observation period – especially in those with lower self control (more impulsive traits) as well as a tendency to reduce drinks in the bar lab after a priming drink. This medication also broke the link between priming drink induced stimulation and further drinking and was safe and well tolerated in non-treatment seeking alcoholics. This study joins several others in demonstrating the utility of sub-acute dosing laboratory paradigms for evaluating medication effects in alcoholics. Taken together, our data suggest that aripiprazole may find utility in the treatment of alcohol dependence, either alone or in combination with other medications, and perhaps especially in those with lower impulse control.
Acknowledgements
Abraham Tiffany assisted in collection of data. Nancy White and Mary Evelyn Boyd assisted in preparation of this manuscript.
Supported by grant P50 AA010761
References
- Anckarsater H. Central nervous changes in social dysfunction: autism, aggression, and psychopathy. Brain Res Bull. 2006;69:259–265. doi: 10.1016/j.brainresbull.2006.01.008. [DOI] [PubMed] [Google Scholar]
- Anton RF. New methodologies for pharmacologic treatment trials for alcohol dependence. Alcohol Clin Exp Res. 1996;20:3A–9A. doi: 10.1111/j.1530-0277.1996.tb01183.x. [DOI] [PubMed] [Google Scholar]
- Anton RF, Moak DH, Waid LR, Latham PK, Malcolm RJ, Dias JK. Naltrexone and cognitive behavioral therapy for the treatment of outpatient alcoholics: results of a placebo-controlled trial. Am J Psychiatry. 1999;156:1758–1764. doi: 10.1176/ajp.156.11.1758. [DOI] [PubMed] [Google Scholar]
- Anton RF, Swift RM. Current pharmacotherapies of alcoholism: a U.S. perspective. Am J Addict. 2003;12:S52–S68. doi: 10.1111/j.1521-0391.2003.tb00496.x. [DOI] [PubMed] [Google Scholar]
- Anton RF, Drobes DJ, Voronin K, Durazo-Avizu R, Moak D. Naltrexone effects on alcohol consumption in a clinical laboratory paradigm: temporal effects of drinking. Psychopharmacology (Berl) 2004a;173:32–40. doi: 10.1007/s00213-003-1720-7. [DOI] [PubMed] [Google Scholar]
- Anton RF, Voronin K, Drobes DJ, Moak DH, Myrick H. A short-term dosing and bar-lab alcohol administration paradigm for evaluating medications (naltrexone, gabapentin, ondansetron) for alcoholism treatment. Alcohol Clin Exp Res. 2004b;28:177A. [Google Scholar]
- Anton RF, Moak DH, Latham PK, Waid LR, Myrick H, Voronin K, Thevos A, Wang W, Woolson R. Naltrexone combined with either cognitive behavioral or motivational enhancement therapy for alcohol dependence. J Clin Psychopharmacol. 2005;25:1–9. doi: 10.1097/01.jcp.0000172071.81258.04. [DOI] [PubMed] [Google Scholar]
- Anton RF, Kranzler H, Breder C, Marcus RN, Carson WH, Han J. A randomized, multicenter, double-blind, placebo-controlled study of the efficacy and safety of aripiprazole for the treatment of alcohol dependence. J Clin Psychopharmacol. 2008;28:5–12. doi: 10.1097/jcp.0b013e3181602fd4. [DOI] [PubMed] [Google Scholar]
- Anton RF, Moak DH, Latham PK. The obsessive compulsive drinking scale (OCDS): a new method of assessing outcome in alcoholism treatment studies. Arch Gen Psychiatry. 1996;53:225–231. doi: 10.1001/archpsyc.1996.01830030047008. [DOI] [PubMed] [Google Scholar]
- Bechara A. Decision making, impulse control and loss of willpower to resist drugs: a neurocognitive perspective. Nat Neurosci. 2005;8:1458–1463. doi: 10.1038/nn1584. [DOI] [PubMed] [Google Scholar]
- Beresford TP, Clapp L, Martin B, Wiberg JL, Alfers J, Beresford HF. Aripiprazole in schizophrenia with cocaine dependence: a pilot study. J Clin Psychopharmacol. 2005;25:363–6. doi: 10.1097/01.jcp.0000169419.38899.5b. [DOI] [PubMed] [Google Scholar]
- Bohn MJ, Krahn DD, Staehler BA. Development and initial validation of a measure of drinking urges in abstinent alcoholics. Alcohol Clin Exp Res. 1995;19:600–606. doi: 10.1111/j.1530-0277.1995.tb01554.x. [DOI] [PubMed] [Google Scholar]
- Burris KD, Molski TF, Xu C, Ryan E, Tottori K, Kikuchi T, Yocca FD, Molinoff PB. Aripiprazole, a novel antipsychotic, is a high-affinity partial agonist at human dopamine D2 receptors. J Pharmacol Exp Ther. 2002;302:381–389. doi: 10.1124/jpet.102.033175. [DOI] [PubMed] [Google Scholar]
- Davis LJ, Jr., Hurt RD, Morse RM, O'Brien PC. Discriminant analysis of the self-administered alcoholism screening test. Alcohol Clin Exp Res. 1987;11:269–273. doi: 10.1111/j.1530-0277.1987.tb01306.x. [DOI] [PubMed] [Google Scholar]
- DeLeon A, Patel NC, Crismon ML. Aripiprazole: A comprehensive review of its pharmacology, clinical efficacy, and tolerability. Clin Ther. 2004;26:649–666. doi: 10.1016/s0149-2918(04)90066-5. [DOI] [PubMed] [Google Scholar]
- Drobes DJ, Anton RF. Drinking in alcoholics following an alcohol challenge research protocol. J Stud Alcohol. 2000;61:220–224. doi: 10.15288/jsa.2000.61.220. [DOI] [PubMed] [Google Scholar]
- Drobes DJ, Anton RF, Thomas SE, Voronin K. A clinical laboratory paradigm for evaluating medication effects on alcohol consumption: naltrexone and nalmefene. Neuropsychopharmacology. 2003;28:755–764. doi: 10.1038/sj.npp.1300101. [DOI] [PubMed] [Google Scholar]
- Drobes DJ, Anton RF, Thomas SE, Voronin K. Effects of naltrexone and nalmefene on subjective response to alcohol among non-treatment seeking alcoholics and social drinkers. Alcohol Clin Exp Res. 2004;28:1362–1370. doi: 10.1097/01.alc.0000139704.88862.01. [DOI] [PubMed] [Google Scholar]
- Feltenstein MW, Altar CA, See RE. Aripiprazole Blocks Reinstatement of Cocaine Seeking in an Animal Model of Relapse. Biol Psychiatry. 2007;61:582–590. doi: 10.1016/j.biopsych.2006.04.010. [DOI] [PubMed] [Google Scholar]
- First MB, Spitzer RL, Gibbon M, Williams JBW. Structured Clinical Interview for DSM-IV Axis I Disorders. American Psychiatric Press, Inc.; Washington, DC: 1997. [Google Scholar]
- Gonzales RA, Weiss F. Suppression of ethanol-reinforced behavior by naltrexone is associated with attenuation of the ethanol-induced increase in dialysate dopamine levels in the nucleus accumbens. J Neurosci. 1998;18:10663–10671. doi: 10.1523/JNEUROSCI.18-24-10663.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grüsser SM, Wrase J, Klein S, Hermann D, Smolka MN, Ruf M, Weber-Fahr W, Flor H, Mann K, Braus DF, Heinz A. Cue-induced activation of the striatum and medial prefrontal cortex is associated with subsequent relapse in abstinent alcoholics. Psychopharmacology (Berl) 2004;175:296–302. doi: 10.1007/s00213-004-1828-4. [DOI] [PubMed] [Google Scholar]
- Ingman K, Kupila J, Hyytia P, Korpi ER. Effects of aripiprazole on alcohol intake in an animal model of high-alcohol drinking. Alcohol Alcohol. 2006;41:391–398. doi: 10.1093/alcalc/agl037. [DOI] [PubMed] [Google Scholar]
- Katner SN, Weiss F. Ethanol-associated olfactory stimuli reinstate ethanol-seeking behavior after extinction and modify extracellular dopamine levels in the nucleus accumbens. Alcohol Clin Exp Res. 1999;23:1751–1760. [PubMed] [Google Scholar]
- Keck PE, Calabrese JR, McQuade RD, Carson WH, Carlson BX, Rollin LM, Marcus RN, Sanchez R, Group AS. A randomized, double-blind, placebo-controlled 26-week trial of aripiprazole in recently manic patients with bipolar I disorder. J Clin Psychiatry. 2006;67:626–37. doi: 10.4088/jcp.v67n0414. [DOI] [PubMed] [Google Scholar]
- Koob GF, Roberts AJ. Brain reward circuits in alcoholism. CNS Spectrums. 1999;4:23–37. [Google Scholar]
- Koob GF, Le Moal M. Drug addiction, dysregulation of reward, and allostasis. Neuropharmacology. 2001;24:97–129. doi: 10.1016/S0893-133X(00)00195-0. [DOI] [PubMed] [Google Scholar]
- Kranzler HR, Covault J, Pierucci-Lagha A, Chan G, Douglas K, Arias A, Oncken C. Effects of aripiprazole on subjective and physiological responses to alcohol. Alcohol Clin Exp Res. 2008;32:573–579. doi: 10.1111/j.1530-0277.2007.00608.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lapish CC, Seamans JK, Chandler LJ. Glutamate-dopamine cotransmission and reward processing in addiction. Alcohol Clin Exp Res. 2006;30:1451–65. doi: 10.1111/j.1530-0277.2006.00176.x. [DOI] [PubMed] [Google Scholar]
- Lawler CP, Prioleau C, Lewis MM, Mak C, Jiang D, Schetz JA, Gonzalez AM, Sibley DR, Mailman RB. Interactions of the novel antipsychotic aripiprazole (OPC-14597) with dopamine and serotonin receptor subtypes. Neuropsychopharmacology. 1999;20:612–627. doi: 10.1016/S0893-133X(98)00099-2. [DOI] [PubMed] [Google Scholar]
- Li Z, Ichikawa J, Dai J, Meltzer HY. Aripiprazole, a novel antipsychotic drug, preferentially increases dopamine release in the prefrontal cortex and hippocampus in rat brain. Eur J Pharmacol. 2004;493:75–83. doi: 10.1016/j.ejphar.2004.04.028. [DOI] [PubMed] [Google Scholar]
- Lile JA, Stoops WW, Vansickel AR, Glaser PEA, Hays LR, Rush CR. Aripiprazole attenuates the discriminative-stimulus and subject-rated effects of D-amphetamine in humans. Neuropsychopharmacology. 2005;30:2103–2114. doi: 10.1038/sj.npp.1300803. [DOI] [PubMed] [Google Scholar]
- Lyvers M. “Loss of control” in alcoholism and drug addiction: a neuroscientific interpretation. Experimental and Clinical Psychopharmacology. 2000;8:225–245. doi: 10.1037//1064-1297.8.2.225. [DOI] [PubMed] [Google Scholar]
- Marder SR, McQuade RD, Stock E, Kaplita S, Marcus R, Safferman AZ, Saha A, Ali M, Iwamoto T. Aripiprazole in the treatment of schizophrenia: safety and tolerability in short-term, placebo-controlled trials. Schizophr Res. 2003;61:123–136. doi: 10.1016/s0920-9964(03)00050-1. [DOI] [PubMed] [Google Scholar]
- Martin CS, Earleywine M, Musty RE, Perrine MW, Swift RM. Development and validation of the biphasic alcohol effects scale. Alcohol Clin Exp Res. 1993;17:140–146. doi: 10.1111/j.1530-0277.1993.tb00739.x. [DOI] [PubMed] [Google Scholar]
- McNair DM, Loor M, Droppleman LF. Manual for the Profile of Mood States. Educational and Industrial Testing Service; San Diego, CA: 1981. [Google Scholar]
- Middaugh LD, Bandy A-LE. Naltrexone effects on ethanol consumption and response to ethanol conditioned cues in C57BL/6 mice. Psychopharmacology (Berl) 2000;151:321–327. doi: 10.1007/s002130000479. [DOI] [PubMed] [Google Scholar]
- Middaugh LD, Szumlinski KK, Patten YV, Marlowe A-LB, Kalivas PW. Chronic ethanol consumption by C57BL/6 mice promotes tolerance to its interoceptive cues and increases extracellular dopamine, an effect blocked by naltrexone. Alcohol Clin Exp Res. 2003;27:1892–1900. doi: 10.1097/01.ALC.0000099264.36220.48. [DOI] [PubMed] [Google Scholar]
- Miller WR, Zweben A, DiClemente CC, Rychtarik RG. Motivational Enhancement Therapy Manual: a Clinical Research Guide for Therapists Treating Individuals with Alcohol Abuse and Dependence. (Series 2).Project MATCH Monograph. 1992 [Google Scholar]
- Myrick H, Anton RF, Li X, Henderson S, Drobes D, Voronin K, George MS. Differential brain activity in alcoholics and social drinkers to alcohol cues: relationship to craving. Neuropsychopharmacology. 2004;29:393–402. doi: 10.1038/sj.npp.1300295. [DOI] [PubMed] [Google Scholar]
- Myrick H, Anton RF, Li X, Henderson S, Randall PK, Voronin K. Effect of naltrexone and ondansetron on alcohol cue-induced activation of the ventral striatum in alcohol-dependent people. Arch Gen Psychiatry. 2008;65:466–475. doi: 10.1001/archpsyc.65.4.466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Malley SS, Krishnan-Saria S, Farren C, Sinha R, Kreek MJ. Naltrexone decreases craving and alcohol self-administration in alcohol-dependent subjects and activates the hypothalamo-pituitary-adrenocortical axis. Psychopharmacology (Berl) 2002;160:19–29. doi: 10.1007/s002130100919. [DOI] [PubMed] [Google Scholar]
- Pantelis C, Yucel M, Wood SJ, Velakoulis D, Sun D, Berger G, Stuart GW, Yung A, Phillips L, McGorry PD. Structural brain imaging evidence for multiple pathological processes at different stages of brain development in schizophrenia. Schizophr Bull. 2005;31:672–696. doi: 10.1093/schbul/sbi034. [DOI] [PubMed] [Google Scholar]
- Patton JH, Stanford MS, Barratt ES. Factor Structure of the Barratt Impulsiveness Scale. J Clin Psychol. 1995;5:768–774. doi: 10.1002/1097-4679(199511)51:6<768::aid-jclp2270510607>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]
- Rogers MA, Kasai K, Koji M, Fukuda R, Iwanami A, Nakagome K, Fukuda M, Kato N. Executive and prefrontal dysfunction in unipolar depression: a review of neuropsychological and imaging evidence. Neurosci Res. 2004;50:1–11. doi: 10.1016/j.neures.2004.05.003. [DOI] [PubMed] [Google Scholar]
- Rosenbloom M, Sullivan EV, Pfefferbaum A. Using magnetic resonance imaging and diffusion tensor imaging to assess brain damage in alcoholics. Alcohol Research & Health: the Journal of the National Institute on Alcohol Abuse and Alcoholism. 2003;27(7):146. [PMC free article] [PubMed] [Google Scholar]
- Schuckit M. Subjective responses to alcohol in sons of alcoholics and control subjects. Arch Gen Psychiatry. 1984;41:879–884. doi: 10.1001/archpsyc.1984.01790200061008. [DOI] [PubMed] [Google Scholar]
- Seidman LJ, Valera EM, Makris N. Structural brain imaging of attention-deficit/hyperactivity disorder. Biol Psychiatry. 2005;57:1263–1272. doi: 10.1016/j.biopsych.2004.11.019. [DOI] [PubMed] [Google Scholar]
- Singleton EG, Tiffany ST, Henningfield JE. Alcohol Craving Questionnaire (ACQ-NOW): Background, scoring, and administration (Revised) Intramural Research Program NIDA; Baltimore, MD: 1999. [Google Scholar]
- Skinner HA, Allen BA. Alcohol dependence syndrome: measurement and validation. J Abnorm Psychol. 1982;91:199–209. doi: 10.1037//0021-843x.91.3.199. [DOI] [PubMed] [Google Scholar]
- Sobell LC, Sobell MB. Timeline Followback: A technique for assessing self-reported ethanol consumption. In: Allen J, Litten RZ, editors. Measuring Alcohol Consumption: Psychosocial and Biological Methods. Humana Press; Totowa: 1992. pp. 41–72. [Google Scholar]
- Sobell LC, Cunningham JA, Sobell MB. Recovery for alcohol problems with and without treatment: prevalence in two population surveys. Am J Public Health. 1996;86:966–972. doi: 10.2105/ajph.86.7.966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan JT, Sykora K, Schneiderman J, Naranjo CA, Sellers EM. Assessment of alcohol withdrawal: the revised clinical institute withdrawal assessment for alcohol scale (CIWA-Ar). Br J Addict. 1989;84:1353–1357. doi: 10.1111/j.1360-0443.1989.tb00737.x. [DOI] [PubMed] [Google Scholar]
- Tapert SF, Cheung EH, Brown GG, Frank LR, Paulus MP, Schweinsburg AD, Meloy MJ, Brown SA. Neural Response to Alcohol Stimuli in Adolescents With Alcohol Use Disorder. Arch Gen Psychiatry. 2003;60:727–735. doi: 10.1001/archpsyc.60.7.727. [DOI] [PubMed] [Google Scholar]
- Warsi M, Sattar SP, Bhatia SC, Petty F. Aripiprazole reduces alcohol use. Canadian Journal of Psychiatry. 2005;50:244. doi: 10.1177/070674370505000415. [DOI] [PubMed] [Google Scholar]
- Weiss F, Porrino LJ. Behavioral neurobiology of alcohol addiction: recent advances and challenges. J Neurosci. 2002;22:3332–3337. doi: 10.1523/JNEUROSCI.22-09-03332.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]