Abstract
Angiogenesis, or the formation of new blood vessels from the preexisting vasculature, is a key component in numerous physiologic and pathologic responses and has broad impact in many medical and surgical specialties. In this review, we discuss the key cellular steps which lead to the neovascularization of tissues, and highlight the main molecular mechanisms and mediators in this process. We include discussions on proteolytic enzymes, cell/matrix interactions, pertinent cell signaling pathways, and end with a survey of the mechanisms which lead to the stabilization and maturation of neovasculatures.
Keywords: angiogenesis, proteases, pericytes, growth factors, extracellular matrix
I. Introduction
The study of angiogenesis has clinical applications in numerous fields including peripheral and coronary vascular disease, oncology, hematology, wound healing, dermatology, ophthalmology, and many others. Experimental evidence demonstrates that the promotion of angiogenic processes can have potential therapeutic benefit in ischemic tissue, while the study of anti-angiogenic therapies have yielded several FDA approved additions to chemotherapeutic regimens in the field of oncology.1 In wound physiology, neovascularization is a key component in the adaptive repair response, promotes prompt coverage of thermal and traumatic wounds, and is required for the survival of grafts and flaps. In addition, inadequately vascularized tissue increases the risk for bacterial and fungal infections which complicate wound management, increase hospital admissions and clinic visits, prolong hospital stays, and add to the cost of health care for patients. For these reasons, a clear and fundamental understanding of the molecular mechanisms which regulate angiogenesis should allow for the development of targeted pro- and anti-angiogenesis therapies which would provide for benefits in the management of complicated wounds, the development of biologic tissue substitutes, and the treatment of pathologies across numerous medical and surgical specialties.
This review focuses on the relevant growth factors, cell-matrix interactions, matrix remodeling enzymes, and pertinent intracellular signaling pathways involved in modulating angiogenesis. We will also discuss the cellular and molecular processes which contribute to the prevention of uncontrolled growth and regression of newly developed vessels.
II. The Process of Angiogenesis
Overview
New blood vessel formation, or neovascularization, occurs either by angiogenesis or by vasculogenesis. Vasculogenesis is the creation of new vessels de novo from precursor cells, such as angioblasts, which differentiate into endothelial cells (ECs), form lumens, and create primitive blood vessels. In contrast, angiogenesis is the formation of new capillaries from the preexisting vasculature. Arteriogenesis, or collateralization, results from the hypertrophy and luminal distention of preexisting vessels in response to mechanical stresses caused by redirected blood flow from occluded or stenosed distal vessels, mediated in part by mechanosensitive signaling in vascular wall cells and by macrophage-derived biomolecular signals.2 While a clinically important process, arteriogenesis is not the formation of new blood vessels and will not be discussed.
The process of angiogenesis involves a complex and dynamic interaction between ECs and the corresponding extracellular environment. In vivo, angiogenesis occurs either by the sprouting of vascular ECs from pre-existing capillary endothelia into the surrounding tissues, or by intussusception (aka non-sprouting angiogenesis), which involves the division of capillaries by tissue pillars into two or more daughter vessels.3, 4 This review will focus primarily on sprouting angiogenesis, to which our usage of the term “angiogenesis” will refer. However, the distinctions made between sprouting and non-sprouting angiogenesis, angiogenesis and vasculogenesis, and the individual processes described below, may be somewhat arbitrary, as they all likely occur in concert and are regulated by related cellular and molecular mechanisms.
Endothelial Cell Sprouting
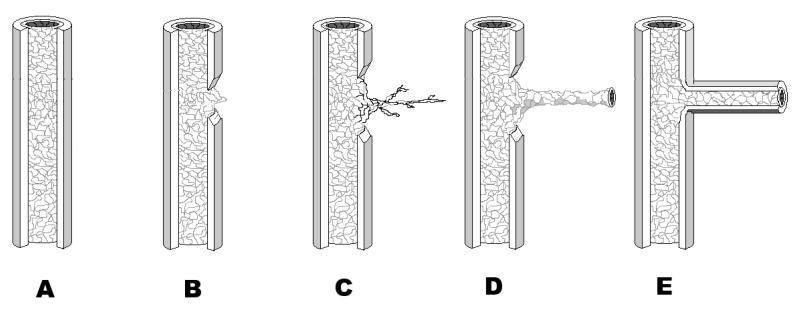
In a normal blood vessel, a 100-200 ηm thick basement membrane (BM) lies immediately deep to the EC monolayer in the arterial intima. Composed mainly of laminins, type-IV collagen, type-VIII collagen, and proteoglycans, the BM must be degraded prior to EC invasion into the surrounding extracellular matrix (ECM), which itself is comprised of fibrillar collagens, elastins, and various other ECM proteins.5 ECs, in response to angiogenic stimuli, convert from a quiescent to a synthetically active phenotype characterized by a high mitotic index and increased capacity for migration and matrix proteolysis. These activated ECs are capable of disrupting the tight junctions, adherens junctions, and gap junctions which exist between neighboring intimal ECs and perivascular cells, and invade into the BM and surrounding ECM.3, 6 Once freed from the capillary intima and in the extravascular space, ECs proliferate and migrate towards chemotactic and angiogenic stimuli in a 3-D extracellular environment and form new angiogenic sprouts (Fig 1). 7
Figure 1.
Diagrammatic description of the steps involved in angiogenesis. A) A stable vascular structure comprised of an endothelial monolayer surrounded by mural tissue (i.e. basement membrane, pericytes, and adventitia); B) Destabilized vessel demonstrating the disruption of mural tissue by endothelial cells; C) Endothelial cell invasion into perivascular tissue and subsequent sprout formation; D) Endothelial cell lumenogenesis and tube formation with inosculation (not shown) to other pre-existing vascular structures; E) Stabilization of the capillary sprout by recruitment of pericytes and deposition of basement membrane.
Lumenogenesis and Tubulogenesis
Lumen formation (and subsequent tube formation) can be considered a hallmark of angiogenesis as it is a relatively specific behavior attributed mainly to cells of the epithelium and endothelium. It is the genetically programmed capacity of ECs to create luminal compartments within multicellular chains which allows for the flow of blood from the pre-existing vasculature to the neovasculature, without which new capillary networks would be unable to perform their central function of oxygen and nutrient transport to normal or pathologic tissues. Indeed, one of the earliest demonstrations of angiogenesis in vitro by Folkman and Haudenschild provided evidence that, in the presence of “direct” angiogens, ECs in culture form capillary-like structures with rudimentary luminal compartments.8
While several mechanisms for lumen formation have been suggested, the most widely investigated mechanism is the process of intracellular vacuolization (or intracellular canalization). The earliest observations of angiogenesis described the presence of “seamless” EC lined capillary lumens in vivo which were lacking in apparent cross-sectional EC-EC junctions.9, 10 Since then, several in vitro and in vivo studies have provided for a mechanistic model of lumen formation consistent with this observation. Mediated by the α2β1 integrin and members of the Rho GTPase family, ECs undergo pinocytosis, leading to the formation of intracellular vacuoles which coalesce and form one larger intracellular lumen.11-16 This would explain the appearance of “seamless” EC lined capillary lumens lacking apparent junctional contacts among several lumen lining cells. The association of caveolin-1 expression, a protein involved in receptor mediated endocytosis and the formation of caveolae (cellular invaginations which often precede vacuole formation), with EC lumenogenesis is an interesting observation in light of this mechanistic hypothesis.17 Concurrently, cytoplasmic projections have been suggested to sense and form junctional contacts with neighboring ECs to thereby form more complex multicellular capillary tubes.15
While intracellular vacuolization is the most widely studied model of lumen formation in ECs, there are descriptions of other mechanisms. These include intussusception, or the insertion of tissue pillars into the newly forming capillaries,18 autophagy by lysosomes within individual ECs which leave behind luminal structures,19 trans-luminal ingrowth of cytoplasmic filopodial projections which create a network of luminal structures,16 as well as the exocytosis of vacuoles between two (or more) ECs which are sprouting in close apposition to one another to form intercellular lumens (intercellular canalization).11, 13 Other groups have proposed that apoptosis of centrally located ECs within a cluster of many ECs can lead to tubular neovascular structures.20, 21 This is consistent with data that demonstrates deficiencies in embryologic lumen formation and microvascular development in transgenic mice which conditionally express the anti-apoptotic protein Bcl-2 in ECs.22
It is important to note that these processes are often described or observed using in vitro assays of angiogenesis, and that the translation of these finding to the modeling of physiologic lumen formation in vivo is challenging.23 Nonetheless, these models of angiogenesis are invaluable in attempting to understand the basic cellular and molecular mechanisms regulating lumenogenesis and angiogenesis in general. It is also important to note that while the above mechanisms for lumen formation have been proposed as separate entities, they may in fact all be contributing to EC lumen formation concurrently, and the relative importance of any one particular mechanism may be dependent on factors such as the location of the cells undergoing lumenogenesis (i.e. the sprouting tip vs. the trunk/stalk of the sprout).24 It has also been proposed that heterogeneous populations of ECs with different functional characteristics can contribute to lumen and tube formation simultaneously. Meyer et. al. suggested a model of angiogenic tube formation which relies on three different populations of ECs: 1) those with phagocytic functions which create matrix channels and later undergo apoptosis, 2) those which form vacuoles which coalesce and get exocytosed to the intercellular space leading to a primitive lumen which is subsequently remodeled, and 3) those which are added into the capillary tube at the final stage of angiogenesis and aid in lumen growth and remodeling.25
A further consideration is the temporal relationship of lumen formation to sprouting. Whether one precedes the other, or, as suggested above, these processes occur simultaneously, is not entirely clear. There is experimental evidence to suggest both possibilities. Davis and Camarillo have demonstrated that vacuoles can be visible in ECs which are actively sprouting,14 while other studies demonstrate that branching and new sprout formation can occur in the absence of lumen formation.26, 27
Inosculation
Inosculation is the fusion or anastomosis of two vascular lumens or luminal segments to form one continuous lumen. Although an understudied process in angiogenesis, some literature from the field of skin graft vascularization can be extrapolated to provide some insights into this later step of angiogenesis. In 1980, Tsukada demonstrated that the preservation of the subcutaneous vascular network of a skin graft facilitated free tissue transfer, and that this was associated with both the formation of a neovasculature, as well as the inosculation between the preexisting host and donor vasculature.28 By analyzing the establishment of donor/host vascular continuity in grafted skin, studies have demonstrated that skin neovascularization with complete inosculation occurs between three to seven post-operative days following skin grafting.29, 30 Immunohistochemical staining of human skin transplanted onto athymic mice has documented recipient EC invasion into the graft between days 3 and 21 via neovascularization with blood flow commencing on days 3-7. Of note, recipient beds were not seen to have donor EC ingrowth.31 The molecular mechanisms involved in inosculation, however, are still relatively unknown, and further study of this phase of angiogenesis is required.
III. Molecular Mediators of Angiogenesis
Growth Factors and Cytokines
Among the most commonly described angiogenic growth factors and cytokines include vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), tumor necrosis factor-alpha (TNF-α), transforming growth factor-beta (TGF-β), and the angiopoietins (Ang). Sources of these growth factors include endothelial cells, fibroblasts, smooth muscle cells, platelets, inflammatory cells, and cancer cells. Some growth factors, including fibroblast growth factor-2 (FGF-2, or basic FGF), are sequestered in the ECM, and serve as reservoirs for sustained cell-demanded growth factor release. The local concentrations of specific growth factors in the extracellular milieu may be impacted by physiologic or pathologic responses such as those seen during injury response, chronic inflammatory states or cancer. Platelet derived growth factor (PDGF), also an important mediator of angiogenesis, will be discussed later in the context of neovessel stabilization and maturation.
Vascular endothelial growth factor
VEGF is one of the most well studied growth factors involved in EC migration, mitogenesis, sprouting, and tube formation. Upregulated VEGF and VEGF-receptor (VEGF-R) mRNA has been detected in the tips of invasive angiogenic sprouts, and antibody blockade of VEGF significantly decreases microvessel outgrowth.32, 33 This heparan sulfate binding molecule exists in several isoforms resulting from alternative splice variants from a single gene product. The soluble or membrane bound form binds most commonly to 2 transmembrane receptor tyrosine kinases, VEGFR-1 (Flt-1) and VEGFR-2 (KDR/Flk-1).34, 35 A third transmembrane receptor neuropilin-1 (Nrp-1) binds the VEGF-A splice variant of VEGF165, is involved in capillary morphogenesis, and appears to be required for EC filopodial tip directionality during angiogenesis.36 VEGF also promotes von Willebrand factor release, integrin expression, interstitial collagenase expression, plasminogen activator (PA) and plasminogen activator receptor (PA-R) expression, and increases both vascular permeability and fenestration, which accounts for its original name, vascular permeability factor. Heparin and heparan sulfate proteoglycan binding contributes to VEGF/VEGF-R binding, stabilizes the active conformation of VEGF, prevents its degradation, and has been shown to greatly increase the angiogenic response of ECs to VEGF in a type-I collagen matrix.37
Its role in angiogenesis has made VEGF a popular target in both pro- and anti-angiogenesis therapies, and anti-VEGF/VEGF-R antibodies and receptor tyrosine kinase inhibitors have become agents of chemotherapeutic regimens in lung and colon cancer. 1, 38 However, its utility as a targeted drug or gene therapy has also been associated with some potential and observed side effects including gastro-intestinal toxicity, hypothyroidism, proteinuria, coagulation disorders, neurotoxicity, impaired wound healing, and excessive chaotic neovascularization.39, 40 It is probable that these potential side effects are partially attributable to a potentially narrow therapeutic range, making accurate therapeutic concentrations for both pro- and anti-angiogenesis therapy critical.41 Thus, the clinical applicability of VEGF (or any angiogen) therapy can likely greatly improve with a better understanding of its absorbance and release kinetics from ECM.42
The above concept is especially true for therapeutic neovascularization strategies. For example, the attachment of VEGF to ECM scaffolds, such as fibrin or collagen, has been shown to promote sustained and controlled release of growth factor, and is associated with more organized vascular networks as compared to the chaotic and highly branching structures that commonly result from the local administration of their soluble counterparts.43, 44 Recently, VEGF-R binding and vascular ingrowth induced by a fibrin binding variant of VEGF121 demonstrated low continuous receptor binding and increased subcutaneous vascular invasion with eventual pericyte recruitment compared to wild type controls.45
Fibroblast Growth Factor
FGF is another pro-angiogenic growth factor, which is both stored in the vascular basement membrane to serve as a reservoir supply, and is upregulated during active angiogenesis. The two most commonly studied forms are FGF-2 or basic FGF (bFGF) and FGF-1 or acidic FGF (aFGF) which bind most commonly to the receptor tyrosine kinases FGFR-1 or FGFR-2.46 In vitro, FGF binding of FGFR-1 increases EC migration and promotes capillary morphogenesis when cultured on collagen gels,47 and activates signaling pathways mediated in part by protein kinase-C (PKC), phospholipase A2, 48, 49 and numerous others. The observation that FGF-2 enhances endogenous VEGF production, and that VEGF is required for the FGF-2-induced expression of placental growth factor demonstrates the existence of cross talk and synergism between FGF and other growth factor pathways.50-55 In addition, FGF-2-mediated proteolysis of matrix components (via up-regulation of urokinase receptors), and induction of the synthesis of collagen, fibronectin and proteoglycans by ECs, demonstrate its effects on ECM remodeling during angiogenesis.47, 56
The successful maintenance of microvasculatures engineered by the exogenous delivery of FGF, or VEGF, is likely dependent on the local concentration of total growth factor present in the culture environment. 57 Proof of this concept is provided by studies by Uriel, et. al. which demonstrated that persistent low levels of exogenously delivered FGF-1 promoted long-term survival of morphologically healthy appearing microvasculatures in vitro compared to higher levels of FGF-1.58 The observed regression of engineered microvasculatures created by the sustained delivery of angiogens (i.e. VEGF, FGF) in the absence of physiologic demand highlights a potential cautionary paradigm in growth factor therapy of angiogen supply and cellular demand.45 Unlike VEGF165, however, FGF binds with a high degree of affinity to ECM proteins such as fibrin and with a high degree of saturation without a cross-linking molecule (i.e. heparan sulfate proteoglycan), which can potentiate its angiogenic potential.59, 60 This observation, coupled with the ability of ECs to produce and release FGF during angiogenesis,61 makes controlling local concentrations of FGF for therapeutic neovascularization purposes challenging. Strategies for sustained or targeted delivery of FGF and designer FGF mutants with biologic properties relatively more beneficial than those of wild type FGF have been investigated to partially address these challenges.62-65
Angiopoietin
The angiopoietins are a family of extracellular ligands that bind to the primarily EC-specific Tie receptors. Similar to other growth factor receptors, they represent a group of receptor tyrosine kinases which function primarily during vascular remodeling and angiogenesis.66, 67 Ang-1 and Ang-2 bind the same receptor, Tie-2, but elicit very different responses. Ang-1 acts as an agonist, causing rapid receptor transphosphorylation, leading to subsequent downstream activation of the protein kinase B/Akt/FKHR (FOX01) pathway to promote EC survival and to inhibit Ang-2 expression.68
This latter effect is significant, as Ang-2 is perhaps the most well studied signaling molecule involved in EC activation and the initiation of angiogenesis. The early role of Ang-2 in angiogenesis is evidenced by its dramatic upregulation in ECs after exposure to angiogenic stimuli such as hypoxia and VEGF.69-75 The release of Ang-2 from Weibel-Palade bodies of vessel ECs induces an autocrine response, which, in the presence of persistent pro-angiogenic signals such as VEGF, acts as a competitive antagonist of Ang-1 to prevent Tie-2 receptor phosphorylation and signaling, is involved in the disruption of the endothelial monolayer, and promotes EC survival, vessel sprouting and angiogenesis.66, 76-78 Ang-2 has been shown to both promote the formation of neovessels during cerebral ischemia and to act as a critical regulator of postnatal vessel remodeling in the ischemic mouse hind limb.78-80 Elevated Ang-2 levels in some tumors and direct Ang-2 retinal injections are associated with decreased pericyte coverage of vessel capillaries, which is consistent with a model of Ang-2-mediated dynamic vessel instability which can lead to increased neovascularization in the presence of other angiogenic stimuli.79, 81-83 A clear understanding of the role of angiopoietins remains elusive and difficult to define however, as their highly context dependent functions vary with the relative levels of Ang-1, Ang-2 , VEGF, and cellular sources of angiogens under specific physiologic and pathologic conditions.
Transforming Growth Factor-β
The TGF-β superfamily is comprised of over 30 structurally similar growth factors and include the three TGF-β isotypes 1-3. They are secreted in an inactive latent form which requires cleavage of the latency associated peptide domain by proteases, often under acidic conditions. In vivo, TGF-β promotes ECM deposition and integrin receptor upregulation,84 is one of the most important cytokines for granulation tissue formation,85 and modulates wound EC proliferation, migration and capillary tube formation.86 While in vitro results have demonstrated that TGF-β can directly inhibit angiogenesis in a receptor specific manner,87 its ability to recruit inflammatory cells in vivo, which act as a source of pro-angiogenic signals, or its ability to modulate other angiogenesis pathways, may account in part for its pro-angiogenic effects.86, 88 Its more well established role in mediating EC-pericyte interactions and vessel stabilization will be discussed below.
Tumor Necrosis Factor-alpha
TNF-α is an inflammatory cytokine, secreted primarily by activated macrophages during inflammation and immune response, but has also been associated with mast cells, ECs, fibroblasts, myocytes, adipose tissue, and neuronal tissue. It has a broad array of functions ranging from stimulation of granulocyte-macrophage-colony stimulating factor (GM-CSF) and interleukin-1 (IL-1), and has been suggested to act on ECs both directly, by inducing cell differentiation, and indirectly in vivo, by stimulating the production of angiogenic factors from other cells.89
Cell-matrix interactions
The proteolysis and remodeling of the ECM is critical in all phases of angiogenesis, affecting EC migration, invasion into the perivascular tissue, and the morphologic formation of luminal structures. The regulation of the activity of these enzymes is highly complex and is impacted by the micro-environmental context within which it occurs. We will discuss the main tools of matrix remodeling, and attempt to highlight the bidirectional nature of cell-matrix interactions.
Matrix metalloproteinases (MMPs) and other proteases
Also call matrixins, MMPs are a family of proteases that degrade ECM proteins and are critical in vascular remodeling, cellular migration, and sprout formation.90 The observation that there is significant upregulation of MMP activity in ECs during inflammation, wound healing and tumor growth reflect their important role in both physiologic and pathologic angiogenesis.91 Protein inhibitors of MMP-2 and MMP-9 have been shown to greatly attenuate the migration of both ECs and tumor cells and have been proposed as therapeutic targets in oncology.92 Knockout mice deficient in MMP-2 demonstrate greatly reduced melanoma tumor angiogenesis and tumor progression.93 The anti-tumor effects of targeted MMP inhibition is likely due to the dual benefits of inhibiting tumor angiogenesis and metastasis, as these processes both require significant ECM degradation.94, 95 Disappointing clinical results with MMP inhibition in cancer therapy, however, highlights the complex nature of MMP activity in tumor and angiogenesis progression.96
In humans, these zinc-containing endopeptidases may be either secreted or membrane bound (MT-MMP).91 The secreted forms are typically secreted as inactive pro-peptides which are often activated by a cascade of cell surface protease activity, including that of the MT-MMPs, which cleaves the pro-peptide to the active form. Increased expression of surface urokinase receptors in activated ECs, for example, bind the inactive proenzyme of urokinase plasminogen activator (uPA) (aka urokinase) and convert it to the active form. Active urokinase then promotes the conversion of plasminogen to plasmin.97 Plasmin then activates MMPs that degrade the extracellular matrix.98, 99 As such, the inhibition of uPA by plasminogen activator inhibitors (PAI-1 and 2) can affect not only the direct degradation of ECM but also of the activation of latent MMPs,100 and PAI expression in ECs or pericytes can act to modulate angiogenesis.101
In addition to contributing to the proteolytic activation of MMPs, other proteases like uPA, tPA, and plasmin contribute to significant ECM degradation with relative specificity. Plasmin, a serine protease, cleaves both ECM (i.e. fibronectin and laminin), as well as coagulation cascade proteins, and is the enzyme primarily responsible for fibrin degradation, a significant component of the injury response which often is a signal for neovascularization.102 Plasmin transversely cuts fibrin fibers and after prolonged exposure results in a broad range of progressively smaller fragments, which may themselves modulate the angiogenic response.103-105 uPA is a secreted enzyme that binds with high affinity to its surface receptor, uPA receptor (uPAR), on a variety of cell types, and upregulated expression of both uPA and uPAR is seen in actively migrating ECs.56, 99, 106 In vitro, TNF-α potentiates VEGF- or FGF-2- induced EC tube formation in fibrin matrices by the mediation of cell surface u-PA expression.107 Blocking uPA or uPAR, or the presence of anti-plasmin compounds such as ε-aminocaproic acid or aprotinin reduces cellular invasion and matrix degradation,108 and the upregulation of PAI in an athymic mouse prostate cancer model has been shown to decrease both the density of tumor-associated microvasculatures and the rates of liver and lung metastases.109
While the presence of growth factors can modulate the activity and expression of ECM proteolytic pathways, it is clear that the interaction between growth factors and proteases is bidirectional. Proteolytic growth factor activation or release from ECM reservoirs can propagate the angiogenic response by allowing for a sustained cell-demanded local concentration of active pro- (or anti-) angiogenic stimuli within the tissue. In addition, MMP-2 has been shown to directly cleave the extracellular domain of the FGF-1 receptor, which releases a soluble extracellular receptor domain that binds FGF-1 and may modulate angiogenesis in vivo.110 Pathologic dysregulation of this intrinsic positive feedback loop between protease and growth factor activity can have significant clinical consequences as seen during the neovascularization of tumors.111
Matrix remodeling proteins also appear to be involved in the formation of luminal and tubular structures during the morphogenic differentiation of ECs in a manner that is specific to the type of MMP involved, with the membrane-bound forms appearing to be more critical in these events than secreted MMPs.112, 113 This has been supported by experiments utilizing specific tissue inhibitors of metalloproteinases (TIMPs) which demonstrate relative substrate specificity. The inhibition of MT1-MMP, but not MT3-MMP, for example, by TIMP-2 or by siRNA knockdown, inhibits EC tube formation in a 3-D model of angiogenesis.15, 114 This is consistent with results of an experiment in which aortic rings from MT1-MMP knockout mice displayed attenuated neovessel formation within a 3-D collagen matrix.115 These results should be viewed cautiously, however, as numerous factors (e.g. EC source), can produce vastly different responses from study to study.116 The mechanistic explanation for how MMPs contribute to lumenogenesis is unclear, but it has been proposed that MMPs create “vascular guidance tunnels”, which serve as molds in the ECM that aid in vascular morphogenesis, pericyte recruitment, and basement membrane assembly.15
The extracellular matrix
The ECM is a highly important modulator of angiogenesis during oncogenesis, wound healing, and development. For example, the differentiation of ECs grown as a monolayer in vitro into branching tubule structures has been shown to require the coverage of cells with collagen and the establishment of appropriate cell-matrix interactions.117 Angiostatic molecules like thrombospondin-1 (TSP-1) and interferon-inducible protein-10 have been shown to be less effective in inhibiting VEGF-induced EC proliferation when cultured on type-I collagen, an observation that has intuitive implications in the clinical use of pro- or anti-angiogenesis drugs.118 Laminin, a non-collagen hetero-trimeric (3 chains: α, β, and γ) glycoprotein found in basement membrane and granulation tissue, has been shown to increase EC-ECM attachment and migration in vitro,86 is produced throughout the length of EC sprouts and has been shown to limit proliferation and lumen size,119 and is associated with breast and brain cancer neovascularization and membrane invasion (i.e. laminin-8 and laminin-10).120, 121 Fibronectin, often an injury response protein, is actively unfolded by ECs and serves as an ECM scaffold for cellular matrix deposition and neovascularization,122 while combinations of other healing proteins like hyaluronic acid, type I collagen and fibrin can have differential angiogenic effects compared to when they are present alone.123, 124
While the composition of the ECM is clearly an important factor in the regulation of angiogenesis, the structure of the ECM is also key. Studies have demonstrated in numerous cell types that cell culture in 3-D environments vs. 2-D environments can significantly impact the expression of differentiation markers, diminish cell proliferation, and affect the expression of proteins like MMP-2.125, 126 Physical characteristics such as fiber thickness, density, orientation, cross-linking, and pore size have all been shown to affect the number, length, and cross-sectional area of angiogenic sprouts.5, 127, 128 In vitro, ECs have been shown to demonstrate decreased cell migration and invasion with increased capillary morphogenesis in fibrin as the rigidity is decreased by altering pH, NaCl concentration, and thrombin content. 108, 129, 130 In chick chorioallantoic membrane (CAM) angiogenesis assays, significantly more sprout invasion is associated with a reduction in fibrin substrate concentration, and the physical characteristics of the ECM were found to be as important as the presence or absence of growth factors such as FGF and VEGF.123
Integrins
Among the most important cell surface receptors involved in cell-matrix interactions, the integrins are transmembrane heterodimeric receptors composed of an α and β subunit which engage ECM ligands to transduce environmental signals to cytoskeletal alterations and intracellular signaling pathways which affect phenotypic modulation, motility, mechanotransduction, and cell proliferation,131-135 and thus impact a broad range of angiogenic processes.26, 136-145 In addition, downstream integrin signaling events mediate the cellular processes involved in angiogenesis, as exemplified by focal adhesion kinase (FAK) knockout mice which demonstrate altered EC morphogenesis during blood vessel assembly.146
These receptors have the capacity to act in a ligand-specific manner producing specific cellular responses depending on the particular ECM or integrin subunits engaged,131, 147 with the αv and β1 subunits, specifically, proposed to be particularly important in morphologic differentiation.86 Some evidence suggests that the disruption of cell-matrix interactions by αvβ3 and αvβ1 integrin receptor blockade actually tends to decrease EC proliferation and to promote the differentiation of ECs to large and multicellular tubules in a matrix specific manner.148
Not surprisingly, the expression and activity of the EC proteolytic machinery is mediated by integrin-dependent signaling pathways. Observed colocalization of active cell surface MMP-2 with the αVβ3 integrin in angiogenic blood vessels and melanoma cells likely reflects the tight coordination between motility and matrix degradation in invasive cells, once again highlighting the bidirectional nature of cell-matrix interactions.149 The α2β1 integrin receptor, but not α3β1integrin receptor, has been shown to be required for type I collagen-induced gelatinase A (MMP-2) activation, in a mechanism that also requires MT1-MMP.105 This, and other studies, demonstrate the highly complex proteolytic cellular responses which are dependent on the nature of the ECM-integrin interaction identified by the specific ECM and corresponding integrin subtype engaged.150
In addition to its direct effects on cellular responses, engagement and activation of integrin-dependent pathways mediate responses to growth factors via significant cross-talk with growth factor pathways, allowing for coordination of cell-matrix-growth factor interactions during angiogenesis.151-153 FGF-2-induced EC migration seems to be mediated in part by the upregulation of the αVβ3 integrin. 154 Similarly, VEGF has been shown to upregulate the expression of the collagen binding integrins α1β1 and α2β1, and blockade of these receptors attenuates the angiogenic response to VEGF.141 Recently, it was shown that VEGF recruits c-src to the EC membrane, which subsequently tyrosine phosphorylates the β3 integrin and thereby mediates interaction between VEGF-R2 and integrin β3 after VEGF stimulation.155
Intracellular Signaling Pathways
A detailed and extensive review of the intracellular signaling pathways involved in angiogenesis is well beyond the scope of this review. We will attempt to introduce some of the most pertinent pathways here, and have cited recent reviews for further study.7, 156
Rho GTPases
The Rho family of guanine nucleotide (GTP) binding proteins (including Rho, Rac, and Cdc42), which are members of the Ras superfamily of GTPases, are intimately involved in growth factor signaling, cytoskeletal dynamics, cell motility, cell shape and spreading, cell phenotype and other processes which significantly impact angiogenesis.157, 158 VEGF-induced EC chemotaxis has been shown to require activated Rac, and type I collagen-induced migration requires Rac and Cdc42 activity.159 Interestingly, while both Rac and Rho induce the formation of stress fibers, special actinomyosin filaments arranged to generate the force required for movement, Rho activity may not be involved in either VEGF- or collagen-induced motility, and may even inhibit the migratory response to extracellular matrix.159 Intracellular signaling molecules phosphatidylinositol 3-kinase (PI3-K), Akt, protein kinase B (PKB), p38 MAPK, FAK, and Rho-associated-kinase (ROCK) all provide molecular linkages among VEGF-R2-mediated Rho GTPase signal transduction pathways in EC migration.7, 157
In addition to its effects on EC migration, several studies have demonstrated that Cdc42 plays a crucial role in EC lumenogenesis.12 Consistent with this, recent evidence demonstrated that overexpression of Cdc42 GTPase activating protein (Cdc42 GAP), a protein which catalyzes GTP hydrolyzation and consequently decreases the active conformation of Cdc42, attenuates EC lumen formation while siRNA knockdown of Cdc42 GAP increased lumenogenesis.160 In vitro, ECs with lower levels of Cdc42 GAP expression have been shown to be more likely to form tubes and lumens in comparison to ECs with a higher level of expression.160 Although the exact mechanistic role of Cdc42 in EC lumen formation is still unclear, this and other downstream GTPase pathway proteins demonstrate potentially promising therapeutic targets for pro or anti-angiogenesis therapies.161
Protein Kinase C (PKC)
PKC is an intracellular kinase that is an important mediator of endothelial cell migration, proliferation and capillary tube formation. Decreasing PKC activity has been shown to attenuate EC attachment to and migration along ECM coated surfaces, without significantly affecting cell spreading.162 Its multifaceted role in VEGF signaling is demonstrated by the inhibition of VEGF-induced cell proliferation, MAPK phosphorylation, migration, endothelial tube formation, matrix adhesion, and vessel formation in vitro and in vivo in cells treated with antisense PKC-α oligonucleotides.163
Notch
Signaling through Notch, a cell membrane receptor, is an evolutionarily conserved pathway which impacts cell fate and differentiation determination,164 and is involved in regulating angiogenesis.165, 166 Notch receptors and their Delta-like-4 (Dll4) ligand appear to be central components in restricting the formation of tip cells in response to VEGF within angiogenic sprouts, which limits the branching and sprouting patterns of angiogenic networks167 and limits tube diameter by inhibiting EC proliferation.168 Blockade of Dll4 in tumor-associated ECs leads to the increased vascularity and branching of tumors, although these microvasculatures demonstrate poor function and are associated with decreased tumor growth.169, 170
Cell-Cell Interactions
Perivascular cells such as pericytes (aka mural cells), SMCs, and adventitial fibroblasts, are associated with the endothelium of capillaries and are known to be involved in the later stages of angiogenesis during vessel maturation and stabilization (discussed below). However, these primarily mesenchymally derived cells surrounding the endothelium also appear to have a significant role in mediating the early events of active angiogenesis.171-174 The co-culture of ECs with pericytes, SMCs and fibroblasts in vitro have demonstrated that the secretion of soluble factors such as VEGF or the deposition of ECM proteins such as fibronectin elicit angiogenic activities in ECs such as increased plasminogen activity, proliferation, motility, and cordlike structure formation.175-180 Evidence that these paracrine interactions may be regulated by the local extracellular environment is provided by the observed endothelin-1-mediated activation of EC mitogenesis by fibroblasts,176 and the production of Ang-2 and VEGF in mesangial cells (renal pericytes) under hypoxic conditions.69 In vivo observations of pericytes found “leading” angiogenic sprout tips and lining vascular tubes connected to tumor vasculatures which lack ECs in vivo suggest a more direct role for pericytes as guiding cells during angiogenesis.181, 182 These data are consistent with observations that under hypoxic stimulation, VEGF-secreting perivascular cells appear to be the first vascular cells to invade the corpus luteum parenchyma during ovulatory angiogenesis.183 In our own lab, we have observed that ECs in both direct and indirect co-culture with SMCs form sprouts which extend to greater lengths, form more robust networks, and persist over longer time periods in fibrin hydrogels compared to ECs cultured alone (unpublished results). Thus, while the later stabilization and maturation of the microvasculature by pericytes is well established, the likelihood that they promote angiogenesis given the proper pro-angiogenic environmental context suggests a more broad role for these cells which warrants further study.184
Gene Expression
An emerging strategy for understanding the molecular regulators governing many aspects of angiogenic differentiation is the study of genetic expression patterns in activated angiogenic ECs. An analysis of genes expressed in ECs which invade and form tubules in a fibrin rich matrix vs. ECs which remain in a monolayer demonstrated differential regulation of a number of genes.160 In vivo, genetic analyses of tumor-associated ECs, which tend to be more angiogenic than ECs from normal vessels, have identified potential angiogenesis markers, termed tumor endothelial markers (TEMs).160, 185, 186 The identity and function of many of these genes, are largely still unknown and under study, although their roles in EC survival, integrin signaling, migration, proliferation, and matrix remodeling have been implicated.160, 187-189 The analysis of TEM expression patterns, and genetic expression patterns in general, offers a unique and exciting methodology which may provide for real translational and therapeutic benefits in a wide variety of patients, and may even provide potential utility in the assessment of cancer prognosis.190
IV. Stabilization and Maturation of the Neovasculature
Molecular and cellular feedback is critical in preventing pathologic angiogenesis. This is achieved by the preferential upregulation of anti-angiogenic stimuli and downregulation of the above pro-angiogenic processes. The net result is a final product of stable capillary tubes lined with a quiescent, functional monolayer of ECs demonstrating junctional contacts, low in both mitotic index and apoptotic turnover, supported by surrounding pericytes.
The extracellular matrix
A primary mediator of this final stage of angiogenesis is the composition of the ECM surrounding the endothelial sprouts.191 Type IV collagen, a major basement membrane protein, has been shown to both promote the lengthening of neovessels and to prevent their regression and continued growth in a dose-dependent manner.192 It has been shown that the degradation of basement membranes rich in type-IV collagen releases several anti-angiogenic proteins such as arresten (from collagen’s α1 chain), and canstatin (α2 chain), which suggests a mechanism for the almost immediate negative regulation of angiogenesis after its induction.193, 194 Alpha-statin and kinostatin, products of MMP mediated fibrinogen and HMW-kininogen degradation, respectively, possess anti-angiogenic activities and likely regulate the angiogenic response to injury.91, 96 Thrombospondin is a platelet and inflammatory cell derived protein found within the ECM that is secreted in response to injury195 which has been shown to inhibit EC proliferation and migration, induce apoptosis, and downregulate MMP activity.102, 196 For these reasons, thrombospondin and other endogenous inhibitors of angiogenesis, like endostatin, have become attractive targets for cancer treatment, but have unfortunately met with limited clinical success.197, 198
Regulation of proteolysis
Given the importance of proteolytic activity and the impact of ECM degradation in modulating angiogenesis, the regulation of MMPs and other proteases is a critical mode of angiogenesis control. Tissue inhibitors of metalloproteinases (TIMPs) are proteins that regulate MMP activity and can act to both activate and inhibit angiogenesis, reflecting the complicated pro- and anti-angiogenic nature of protease activity during angiogenesis.90, 91, 199 In addition to directly inhibiting MMPs, TIMP-3 prevents VEGF binding to VEGF-R2 and serves to inhibit VEGF induced signaling and angiogenesis.200
Pericytes
The recruitment and incorporation of pericytes are key events which mediate the final steps required for the stabilization and maturation of the neovasculature.6 Pericytes are primarily thought to be derived from cells of mesenchymal origins such as SMCs, fibroblasts or other progenitor cells and possess markers such as α-actin, desmin, PDGF-β receptor, and NG2 which are common to those cells.6, 173, 201 Several lines of evidence support the role of pericyte incorporation for the stabilization of neovessels and the regulation of uncontrolled angiogenic growth. Early studies showed that mice lacking the ability to recruit pericytes during embryonic vascular development demonstrated increased vessel microaneurysm formation and rupture.202 Generally speaking, abnormal pericyte coverage and function in the microvasculatures of certain types of tumors is associated with increased EC sensitivity to VEGF withdrawal203-207 and microvasculatures with disrupted EC/pericyte interactions are associated with more dynamic remodeling.208
Direct contact via gap junctions, adhesion plaques, and adherens junctions, as well as indirect paracrine mechanisms between ECs and pericytes, allow for significant interactions which impact cellular differentiation, migration, proliferation, and quiescence.209-212 Among the paracrine mediators, PDGF is thought to be critical in the recruitment and proliferation of pericytes during angiogenesis. PDGF is a mitogen and chemoattractant for fibroblasts, SMCs, and other mesenchymal cells which is present in four known isoforms A, B, C, and D. Two known PDGF transmembrane receptor tyrosine kinases known as the α and β subunit bind each chain with relative specificity (the α receptor binds the A, B, and C chains primarily while the β receptor binds the B and D chains). Because PDGF exists as a hetero- or homodimer, ligand binding causes receptor hetero- or homodimerization (i.e. PDGF-BB and PDGF-AB can bind α/α, α/β, or β/β, while PDGF AA binds α/α exclusively) and subsequent cell signaling.213-215 As PDGF-β is the primary receptor present on SMCs and other pericytes, PDGF-B/β signaling is especially critical in the proper recruitment of pericytes to capillaries. Disrupted PDGF-B/β signaling is associated with the attenuated ability to recruit PDGF-β receptor positive pericytes and precursors from the perivascular tissue and the bone marrow and is associated with significant vascular abnormalities in physiologic and pathologic angiogenesis.202, 205, 216-221
The source of PDGF during this process is likely from actively angiogenic ECs themselves, which suggests an inherent negative feedback mechanism to prevent uncontrolled neovascularization.6 Proliferative ECs in vitro are potent stimulators of pericyte/SMC growth and migration, in part mediated by PDGF-BB, although this effect is dependent on the time in co-culture, the presence or absence of direct contact, and the deposition of endothelial-specific ECM proteins.222-230 The importance of EC-specific PDGF-B production in pericyte recruitment in vivo was demonstrated in studies which showed that EC-specific knockout of PDGF-B led to significant vascular abnormalities with pericyte loss in mice.231-233
Clinically, the simultaneous targeting of pericytes and ECs by pro- or anti-PDGF and VEGF therapy, has been investigated in tissue engineering and oncologic research. Combination therapy has been shown in some studies to be more effective in disrupting tumor vascularity than either agent alone and, as such, combinatorial antitumor therapy is under investigation.234-237 It is unclear, however, if the apparent improved anti-angiogenic effect observed in combination therapy is specifically attributable to PDGF blockade or to the broad non-specific effects of the receptor tyrosine kinase inhibitors used in most studies of combination therapy. Recently, it was shown that adenoviral expression of soluble anti-PDGF-β receptor provided added inhibition of tumor growth and angiogenesis only when sub-optimal levels of VEGF blockade were present.238 Clearly, however, PDGF signaling does impact angiogenesis in vivo, as the spatiotemporal delivery of PDGF with VEGF has been shown to induce denser and more mature appearing vascular networks within tissue engineered constructs, which is in part associated with the recruitment of α-actin positive (indicative of pericytes) cells into the neovascular networks.239, 240
Although the expansion, spreading, and migration of the SMC/pericyte population on newly formed capillaries may be dependent on PDGF-B/β signaling, it does not appear to be sufficient or necessary for the phenotypic differentiation of undifferentiated mesenchymal cells to pericytes. Instead, this appears to be regulated by TGF-β,218, 241 as evidenced by observations that ECs direct 10T1/2 (a cell line commonly used to model pericyte precursor cells in vitro) to an SMC/pericyte phenotype via a TGF-β-mediated mechanism,228, 242 and that the phenotypic differentiation of SMCs in co-culture with ECs involves activation of the SMC PI3-K/Akt pathway, which may be mediated by TGF-β.243, 244
With these above observation taken together, it has been proposed that two modes of pericyte incorporation occur simultaneously: 1) the differentiation of undifferentiated mesenchymal and progenitor cells into pericytes; and 2) the recruitment and co-migration of differentiated mesenchymal cells such as SMCs from a pool of available cells in the perivascular tissue into neovessels.218
It is believed that once incorporated, a major function of pericytes is to mediate the quiescence of ECs. In vitro, ECs in direct contact with pericytes have lower levels mitosis compared to ECs cultured alone, a process which may require Rho GTPase activity in pericytes.229, 245, 246 Similarly, ECs in close contact with SMCs in a spherical aggregate of SMCs in collagen gels demonstrated decreased responsiveness to VEGF (in the absence of Ang-2), increased inter-endothelial junctions, decreased PDGF-B growth factor chain expression, and reduced apoptosis.247 ECs in co-culture with 10T1/2 cells also displayed a significantly higher resistance to the permeability of a tracer molecule biotin-dextran which was associated with tightening of EC-EC tight junctions and with the localization of tight junction proteins plakoglobin, ZO-1, ZO-2, and occludin.248
It has also been suggested that pericytes induce vessel stabilization by the production of survival factors. This is supported by several lines of evidence: 1) Neonatal vessels which are resistant to oxygen-induced degeneration are associated with pericytes which express TGF-β1, which induces VEGFR-1 expression in ECs;249 2) 10T1/2 precursor cells produce VEGF upon TGF-β-mediated conversion to pericytes when in direct contact with ECs;250 3) Pericytes express Ang-1 in vitro and in vivo, a growth factor which is associated with basement membrane formation, EC quiescence and endothelial leak resistance, and which can mature vessels lacking pericytes;67, 251-253 and 4) Pericyte loss caused by PDGF inhibition in a rat model of retinopathy results in overexpression of VEGF/VEGFR2 in ganglion cells and other supporting cells.254
A final mechanistic hypothesis for the pericyte-mediated regulation of angiogenesis is that pericytes significantly modulate the ECM remodeling capacity of ECs by directly influencing the local ECM composition. Pericyte-conditioned media has been shown to limit both EC migration and branching in vitro by inducing the upregulation of PAI-1 in ECs.255 In addition, pericytes have been shown to produce TIMP-3 and induce TIMP-2 production in ECs, which specifically targets MT1-MMP, MT2-MMP and ADAM proteinases, with the net effect of decreasing angiogenic potential and tube regression.112, 256, 257 In addition, pericyte-mediated ECM deposition, specifically the deposition and synthesis of basement membrane, has also been proposed to promote EC quiescence and vessel stability.191, 258, 259 The importance of the basement membrane in vascular stability is demonstrated in laminin-8 knockout animals which demonstrate poorly developed microvasculature with defective basement membranes, noticeable deformities, and increased responsiveness to pro-angiogenic stimuli.260
While the mechanisms discussed above in the maturation and stabilization of existing and newly formed vessels are still topics of current study, it is becoming clear that this final stage of angiogenesis is a significant contributor to the persistence and function of pathologic and physiologic neovasculatures, and will likely provide for future therapeutic strategies.
V. Summary
We have attempted to describe the major cellular processes involved in the angiogenic development of new blood vessels and the molecular mediators involved in regulating these events. It should be noted that while we do not cover the biomechanical environment within which these events occur, the effects of shear stresses and cyclic distention resulting from pulsatile flow is an important consideration.261, 262
In summary, the relationships among phenotypically variable vascular (and progenitor and stem) cells, ECM and ECM receptors, growth factors and growth factor receptors, proteases, and intracellular signaling pathways during blood vessel formation represent an infinitely complex set of orchestrated events which are tightly regulated by a myriad of macro and micro-environmental factors in an apparently perpetual state of dynamic interdependent flux. It is a challenge for investigators of such complex biologic systems to develop more intricate and sophisticated models in order to paradoxically apply a more precise and reductionistic investigatory approach. The ultimate benefit of this work will likely be to facilitate the discovery of new anti- or pro-angiogenic therapies that can specifically target pathologic tissues to impact the health of patients in numerous medical and surgical specialties.
Acknowledgments
This work was supported by grants from the National Institutes of Health (NIH R01-HL41272), the Department of Veterans’ Affairs, the American Heart Association (AHA 0815674G), and the Falk Medical Research Trust.
References
- 1.Carmeliet P. Angiogenesis in life, disease and medicine. Nature. 2005 Dec 15;438(7070):932. doi: 10.1038/nature04478. [DOI] [PubMed] [Google Scholar]
- 2.Scholz D, Cai WJ, Schaper W. Arteriogenesis, a new concept of vascular adaptation in occlusive disease. Angiogenesis. 2001;4(4):247–257. doi: 10.1023/a:1016094004084. [DOI] [PubMed] [Google Scholar]
- 3.Risau W. Mechanisms of angiogenesis. Nature. 1997 Apr 17;386(6626):671. doi: 10.1038/386671a0. [DOI] [PubMed] [Google Scholar]
- 4.Burri PH, Djonov V. Intussusceptive angiogenesis--the alternative to capillary sprouting. Mol Aspects Med. 2002 Dec;23(6S):S1–27. doi: 10.1016/s0098-2997(02)00096-1. [DOI] [PubMed] [Google Scholar]
- 5.Francis ME, Uriel S, Brey EM. Endothelial cell-matrix interactions in neovascularization. Tissue Eng Part B Rev. 2008 Mar;14(1):19–32. doi: 10.1089/teb.2007.0115. [DOI] [PubMed] [Google Scholar]
- 6.Armulik A, Abramsson A, Betsholtz C. Endothelial/Pericyte Interactions. Circ Res. 2005 September 16;97(6):512–523. doi: 10.1161/01.RES.0000182903.16652.d7. 2005. [DOI] [PubMed] [Google Scholar]
- 7.Lamalice L, Le Boeuf F, Huot J. Endothelial cell migration during angiogenesis. Circ Res. 2007 Mar 30;100(6):782–794. doi: 10.1161/01.RES.0000259593.07661.1e. [DOI] [PubMed] [Google Scholar]
- 8.Folkman J, Haudenschild C. Angiogenesis in vitro. Nature. 1980 Dec 11;288(5791):551. doi: 10.1038/288551a0. [DOI] [PubMed] [Google Scholar]
- 9.Wolff JR, Bar T. ‘Seamless’ endothelia in brain capillaries during development of the rat’s cerebral cortex. Brain Res. 1972 Jun 8;41(1):17. doi: 10.1016/0006-8993(72)90613-0. [DOI] [PubMed] [Google Scholar]
- 10.Guldner FH, Wolff JR. Seamless endothelia as indicators of capillaries developed from sprouts. Bibl.Anat. 1973;12:120. [PubMed] [Google Scholar]
- 11.Davis GE, Bayless KJ. An integrin and Rho GTPase-dependent pinocytic vacuole mechanism controls capillary lumen formation in collagen and fibrin matrices. Microcirculation. 2003 Jan;10(1):27. doi: 10.1038/sj.mn.7800175. [DOI] [PubMed] [Google Scholar]
- 12.Bayless KJ, Davis GE. The Cdc42 and Rac1 GTPases are required for capillary lumen formation in three-dimensional extracellular matrices. J.Cell.Sci. 2002 Mar 15;115(Pt 6):1123. doi: 10.1242/jcs.115.6.1123. [DOI] [PubMed] [Google Scholar]
- 13.Kamei M, Saunders WB, Bayless KJ, Dye L, Davis GE, Weinstein BM. Endothelial tubes assemble from intracellular vacuoles in vivo. Nature. 2006 Jul 27;442(7101):453. doi: 10.1038/nature04923. [DOI] [PubMed] [Google Scholar]
- 14.Davis GE, Camarillo CW. An alpha 2 beta 1 integrin-dependent pinocytic mechanism involving intracellular vacuole formation and coalescence regulates capillary lumen and tube formation in three-dimensional collagen matrix. Exp.Cell Res. 1996 Apr 10;224(1):39. doi: 10.1006/excr.1996.0109. [DOI] [PubMed] [Google Scholar]
- 15.Davis GE, Koh W, Stratman AN. Mechanisms controlling human endothelial lumen formation and tube assembly in three-dimensional extracellular matrices. Birth Defects Res.C.Embryo.Today. 2007 Dec;81(4):270. doi: 10.1002/bdrc.20107. [DOI] [PubMed] [Google Scholar]
- 16.Egginton S, Gerritsen M. Lumen formation: in vivo versus in vitro observations. Microcirculation. 2003 Jan;10(1):45. doi: 10.1038/sj.mn.7800174. [DOI] [PubMed] [Google Scholar]
- 17.Liu J, Wang XB, Park DS, Lisanti MP. Caveolin-1 expression enhances endothelial capillary tubule formation. J.Biol.Chem. 2002 Mar 22;277(12):10661. doi: 10.1074/jbc.M110354200. [DOI] [PubMed] [Google Scholar]
- 18.Djonov VG, Galli AB, Burri PH. Intussusceptive arborization contributes to vascular tree formation in the chick chorio-allantoic membrane. Anat.Embryol.(Berl) 2000 Nov;202(5):347. doi: 10.1007/s004290000126. [DOI] [PubMed] [Google Scholar]
- 19.Stromhaug PE, Klionsky DJ. Approaching the molecular mechanism of autophagy. Traffic. 2001 Aug;2(8):524. doi: 10.1034/j.1600-0854.2001.20802.x. [DOI] [PubMed] [Google Scholar]
- 20.Peters K, Troyer D, Kummer S, Kirkpatrick CJ, Rauterberg J. Apoptosis causes lumen formation during angiogenesis in vitro. Microvasc.Res. 2002 Sep;64(2):334. doi: 10.1006/mvre.2002.2438. [DOI] [PubMed] [Google Scholar]
- 21.Duval H, Harris M, Li J, Johnson N, Print C. New insights into the function and regulation of endothelial cell apoptosis. Angiogenesis. 2003;6(3):171. doi: 10.1023/B:AGEN.0000021390.09275.bc. [DOI] [PubMed] [Google Scholar]
- 22.Duval H, Johnson N, Li J, et al. Vascular development is disrupted by endothelial cell-specific expression of the anti-apoptotic protein Bcl-2. Angiogenesis. 2007;10(1):55. doi: 10.1007/s10456-006-9057-6. [DOI] [PubMed] [Google Scholar]
- 23.Ucuzian AA, Greisler HP. In vitro models of angiogenesis. World J.Surg. 2007 Apr;31(4):654. doi: 10.1007/s00268-006-0763-4. [DOI] [PubMed] [Google Scholar]
- 24.Egginton S, Zhou AL, Brown MD, Hudlicka O. Unorthodox angiogenesis in skeletal muscle. Cardiovasc.Res. 2001 Feb 16;49(3):634. doi: 10.1016/s0008-6363(00)00282-0. [DOI] [PubMed] [Google Scholar]
- 25.Meyer GT, Matthias LJ, Noack L, Vadas MA, Gamble JR. Lumen formation during angiogenesis in vitro involves phagocytic activity, formation and secretion of vacuoles, cell death, and capillary tube remodelling by different populations of endothelial cells. Anat.Rec. 1997 Nov;249(3):327. doi: 10.1002/(SICI)1097-0185(199711)249:3<327::AID-AR3>3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- 26.Davis GE, Camarillo CW. Regulation of endothelial cell morphogenesis by integrins, mechanical forces, and matrix guidance pathways. Exp.Cell Res. 1995 Jan;216(1):113. doi: 10.1006/excr.1995.1015. [DOI] [PubMed] [Google Scholar]
- 27.Vernon RB, Angello JC, Iruela-Arispe ML, Lane TF, Sage EH. Reorganization of basement membrane matrices by cellular traction promotes the formation of cellular networks in vitro. Lab.Invest. 1992 May;66(5):536. [PubMed] [Google Scholar]
- 28.Tsukada S. Transfer of free skin grafts with a preserved subcutaneous vascular network. Ann.Plast.Surg. 1980 Jun;4(6):500. doi: 10.1097/00000637-198006000-00009. [DOI] [PubMed] [Google Scholar]
- 29.O’Ceallaigh S, Herrick SE, Bluff JE, McGrouther DA, Ferguson MW. Quantification of total and perfused blood vessels in murine skin autografts using a fluorescent double-labeling technique. Plast.Reconstr.Surg. 2006 Jan;117(1):140. doi: 10.1097/01.prs.0000185611.87601.b8. [DOI] [PubMed] [Google Scholar]
- 30.Okada T. Revascularisation of free full thickness skin grafts in rabbits: a scanning electron microscope study of microvascular casts. Br.J.Plast.Surg. 1986 Apr;39(2):183. doi: 10.1016/0007-1226(86)90080-9. [DOI] [PubMed] [Google Scholar]
- 31.Plenat F, Vignaud JM, Guerret-Stocker S, Hartmann D, Duprez K, Duprez A. Host-donor interactions in healing of human split-thickness skin grafts onto nude mice: in situ hybridization, immunohistochemical, and histochemical studies. Transplantation. 1992 May;53(5):1002. doi: 10.1097/00007890-199205000-00008. [DOI] [PubMed] [Google Scholar]
- 32.Brown KJ, Maynes SF, Bezos A, Maguire DJ, Ford MD, Parish CR. A novel in vitro assay for human angiogenesis. Lab.Invest. 1996 Oct;75(4):539. [PubMed] [Google Scholar]
- 33.Gerhardt H, Golding M, Fruttiger M, et al. VEGF guides angiogenic sprouting utilizing endothelial tip cell filopodia. J.Cell Biol. 2003 Jun 23;161(6):1163. doi: 10.1083/jcb.200302047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003 Jun;9(6):669–676. doi: 10.1038/nm0603-669. [DOI] [PubMed] [Google Scholar]
- 35.Neufeld G, Tessler S, Gitay-Goren H, Cohen T, Levi BZ. Vascular endothelial growth factor and its receptors. Prog Growth Factor Res. 1994;5(1):89–97. doi: 10.1016/0955-2235(94)90019-1. [DOI] [PubMed] [Google Scholar]
- 36.Gerhardt H, Ruhrberg C, Abramsson A, Fujisawa H, Shima D, Betsholtz C. Neuropilin-1 is required for endothelial tip cell guidance in the developing central nervous system. Dev Dyn. 2004 Nov;231(3):503–509. doi: 10.1002/dvdy.20148. [DOI] [PubMed] [Google Scholar]
- 37.Steffens GC, Yao C, Prevel P, et al. Modulation of angiogenic potential of collagen matrices by covalent incorporation of heparin and loading with vascular endothelial growth factor. Tissue Eng. 2004 Sep-Oct;10(910):1502–1509. doi: 10.1089/ten.2004.10.1502. [DOI] [PubMed] [Google Scholar]
- 38.Hurwitz H. Integrating the anti-VEGF-A humanized monoclonal antibody bevacizumab with chemotherapy in advanced colorectal cancer. Clin Colorectal Cancer. 2004 Oct;4(Suppl 2):S62–68. doi: 10.3816/ccc.2004.s.010. [DOI] [PubMed] [Google Scholar]
- 39.Roodhart JM, Langenberg MH, Witteveen E, Voest EE. The molecular basis of class side effects due to treatment with inhibitors of the VEGF/VEGFR pathway. Curr Clin Pharmacol. 2008 May;3(2):132–143. doi: 10.2174/157488408784293705. [DOI] [PubMed] [Google Scholar]
- 40.Drake CJ, Little CD. Exogenous vascular endothelial growth factor induces malformed and hyperfused vessels during embryonic neovascularization. Proc Natl Acad Sci U S A. 1995 Aug 15;92(17):7657–7661. doi: 10.1073/pnas.92.17.7657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ozawa CR, Banfi A, Glazer NL, et al. Microenvironmental VEGF concentration, not total dose, determines a threshold between normal and aberrant angiogenesis. J Clin Invest. 2004 Feb;113(4):516–527. doi: 10.1172/JCI18420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sahni A, Francis CW. Vascular endothelial growth factor binds to fibrinogen and fibrin and stimulates endothelial cell proliferation. Blood. 2000 Dec 1;96(12):3772. [PubMed] [Google Scholar]
- 43.Ehrbar M, Djonov VG, Schnell C, et al. Cell-demanded liberation of VEGF121 from fibrin implants induces local and controlled blood vessel growth. Circ Res. 2004 Apr 30;94(8):1124–1132. doi: 10.1161/01.RES.0000126411.29641.08. [DOI] [PubMed] [Google Scholar]
- 44.Ishikawa T, Eguchi M, Wada M, et al. Establishment of a functionally active collagen-binding vascular endothelial growth factor fusion protein in situ. Arterioscler.Thromb.Vasc.Biol. 2006 Sep;26(9):1998. doi: 10.1161/01.ATV.0000233359.74484.77. [DOI] [PubMed] [Google Scholar]
- 45.Ehrbar M, Zeisberger SM, Raeber GP, Hubbell JA, Schnell C, Zisch AH. The role of actively released fibrin-conjugated VEGF for VEGF receptor 2 gene activation and the enhancement of angiogenesis. Biomaterials. 2008 Apr;29(11):1720–1729. doi: 10.1016/j.biomaterials.2007.12.002. [DOI] [PubMed] [Google Scholar]
- 46.Gerwins P, Skoldenberg E, Claesson-Welsh L. Function of fibroblast growth factors and vascular endothelial growth factors and their receptors in angiogenesis. Crit.Rev.Oncol.Hematol. 2000 Jun;34(3):185. doi: 10.1016/s1040-8428(00)00062-7. [DOI] [PubMed] [Google Scholar]
- 47.Kanda S, Landgren E, Ljungstrom M, Claesson-Welsh L. Fibroblast growth factor receptor 1-induced differentiation of endothelial cell line established from tsA58 large T transgenic mice. Cell Growth Differ. 1996 Mar;7(3):383–395. [PubMed] [Google Scholar]
- 48.Sa G, Murugesan G, Jaye M, Ivashchenko Y, Fox PL. Activation of cytosolic phospholipase A2 by basic fibroblast growth factor via a p42 mitogen-activated protein kinase-dependent phosphorylation pathway in endothelial cells. J Biol Chem. 1995 Feb 3;270(5):2360–2366. doi: 10.1074/jbc.270.5.2360. [DOI] [PubMed] [Google Scholar]
- 49.Kent KC, Mii S, Harrington EO, Chang JD, Mallette S, Ware JA. Requirement for protein kinase C activation in basic fibroblast growth factor-induced human endothelial cell proliferation. Circ Res. 1995 Aug;77(2):231–238. doi: 10.1161/01.res.77.2.231. [DOI] [PubMed] [Google Scholar]
- 50.Landgren E, Eriksson A, Wennstrom S, Kanda S, Claesson-Welsh L. Induction of fibroblast growth factor receptor-1 mRNA and protein by platelet-derived growth factor BB. Exp.Cell Res. 1996 Mar 15;223(2):405. doi: 10.1006/excr.1996.0095. [DOI] [PubMed] [Google Scholar]
- 51.Couper LL, Bryant SR, Eldrup-Jorgensen J, Bredenberg CE, Lindner V. Vascular endothelial growth factor increases the mitogenic response to fibroblast growth factor-2 in vascular smooth muscle cells in vivo via expression of fms-like tyrosine kinase-1. Circ.Res. 1997 Dec;81(6):932. doi: 10.1161/01.res.81.6.932. [DOI] [PubMed] [Google Scholar]
- 52.Vernon RB, Sage EH. A novel, quantitative model for study of endothelial cell migration and sprout formation within three-dimensional collagen matrices. Microvasc.Res. 1999 Mar;57(2):118. doi: 10.1006/mvre.1998.2122. [DOI] [PubMed] [Google Scholar]
- 53.Xue L, Greisler HP. Angiogenic effect of fibroblast growth factor-1 and vascular endothelial growth factor and their synergism in a novel in vitro quantitative fibrin-based 3-dimensional angiogenesis system. Surgery. 2002 Aug;132(2):259–267. doi: 10.1067/msy.2002.125720. [DOI] [PubMed] [Google Scholar]
- 54.Bonello MR, Khachigian LM. Fibroblast growth factor-2 represses platelet-derived growth factor receptor-alpha (PDGFR-alpha) transcription via ERK1/2-dependent Sp1 phosphorylation and an atypical cis-acting element in the proximal PDGFR-alpha promoter. J.Biol.Chem. 2004 Jan 23;279(4):2377. doi: 10.1074/jbc.M308254200. [DOI] [PubMed] [Google Scholar]
- 55.Faraone D, Aguzzi MS, Ragone G, Russo K, Capogrossi MC, Facchiano A. Heterodimerization of FGF-receptor 1 and PDGF-receptor-alpha: a novel mechanism underlying the inhibitory effect of PDGF-BB on FGF-2 in human cells. Blood. 2006 Mar 1;107(5):1896. doi: 10.1182/blood-2005-04-1524. [DOI] [PubMed] [Google Scholar]
- 56.Pepper MS, Sappino AP, Stocklin R, Montesano R, Orci L, Vassalli JD. Upregulation of urokinase receptor expression on migrating endothelial cells. J.Cell Biol. 1993 Aug;122(3):673. doi: 10.1083/jcb.122.3.673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Satake S, Kuzuya M, Ramos MA, Kanda S, Iguchi A. Angiogenic stimuli are essential for survival of vascular endothelial cells in three-dimensional collagen lattice. Biochem.Biophys.Res.Commun. 1998 Mar 27;244(3):642. doi: 10.1006/bbrc.1998.8313. [DOI] [PubMed] [Google Scholar]
- 58.Uriel S, Brey EM, Greisler HP. Sustained low levels of fibroblast growth factor-1 promote persistent microvascular network formation. Am.J.Surg. 2006 Nov;192(5):604. doi: 10.1016/j.amjsurg.2006.08.012. [DOI] [PubMed] [Google Scholar]
- 59.Sahni A, Odrljin T, Francis CW. Binding of basic fibroblast growth factor to fibrinogen and fibrin. J Biol Chem. 1998 Mar 27;273(13):7554–7559. doi: 10.1074/jbc.273.13.7554. [DOI] [PubMed] [Google Scholar]
- 60.Sahni A, Khorana AA, Baggs RB, Peng H, Francis CW. FGF-2 binding to fibrin(ogen) is required for augmented angiogenesis. Blood. 2006 Jan 1;107(1):126–131. doi: 10.1182/blood-2005-06-2460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Schweigerer L, Neufeld G, Friedman J, Abraham JA, Fiddes JC, Gospodarowicz D. Capillary endothelial cells express basic fibroblast growth factor, a mitogen that promotes their own growth. Nature. 1987 Jan 15-21;325(6101):257. doi: 10.1038/325257a0. [DOI] [PubMed] [Google Scholar]
- 62.Zhao W, Chen B, Li X, et al. Vascularization and cellularization of collagen scaffolds incorporated with two different collagen-targeting human basic fibroblast growth factors. J Biomed Mater Res A. 2007 Sep 1;82(3):630–636. doi: 10.1002/jbm.a.31179. [DOI] [PubMed] [Google Scholar]
- 63.Shireman PK, Xue L, Maddox E, Burgess WH, Greisler HP. The S130K fibroblast growth factor-1 mutant induces heparin-independent proliferation and is resistant to thrombin degradation in fibrin glue. J.Vasc.Surg. 2000 Feb;31(2):382. doi: 10.1016/s0741-5214(00)90168-x. [DOI] [PubMed] [Google Scholar]
- 64.Erzurum VZ, Bian JF, Husak VA, et al. R136K fibroblast growth factor-1 mutant induces heparin-independent migration of endothelial cells through fibrin glue. J.Vasc.Surg. 2003 May;37(5):1075. doi: 10.1067/mva.2003.177. [DOI] [PubMed] [Google Scholar]
- 65.Brewster LP, Washington C, Brey EM, et al. Construction and characterization of a thrombin-resistant designer FGF-based collagen binding domain angiogen. Biomaterials. 2008 Jan;29(3):327. doi: 10.1016/j.biomaterials.2007.09.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Maisonpierre PC, Suri C, Jones PF, et al. Angiopoietin-2, a natural antagonist for Tie2 that disrupts in vivo angiogenesis. Science. 1997 Jul 4;277(5322):55. doi: 10.1126/science.277.5322.55. [DOI] [PubMed] [Google Scholar]
- 67.Suri C, Jones PF, Patan S, et al. Requisite role of angiopoietin-1, a ligand for the TIE2 receptor, during embryonic angiogenesis. Cell. 1996 Dec 27;87(7):1171. doi: 10.1016/s0092-8674(00)81813-9. [DOI] [PubMed] [Google Scholar]
- 68.Daly C, Wong V, Burova E, et al. Angiopoietin-1 modulates endothelial cell function and gene expression via the transcription factor FKHR (FOXO1) Genes Dev. 2004 May 1;18(9):1060. doi: 10.1101/gad.1189704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Yuan HT, Yang SP, Woolf AS. Hypoxia up-regulates angiopoietin-2, a Tie-2 ligand, in mouse mesangial cells. Kidney Int. 2000 Nov;58(5):1912. doi: 10.1111/j.1523-1755.2000.00363.x. [DOI] [PubMed] [Google Scholar]
- 70.Pichiule P, LaManna JC. Angiopoietin-2 and rat brain capillary remodeling during adaptation and deadaptation to prolonged mild hypoxia. J.Appl.Physiol. 2002 Sep;93(3):1131. doi: 10.1152/japplphysiol.00318.2002. [DOI] [PubMed] [Google Scholar]
- 71.Mandriota SJ, Pepper MS. Regulation of angiopoietin-2 mRNA levels in bovine microvascular endothelial cells by cytokines and hypoxia. Circ.Res. 1998 Oct 19;83(8):852. doi: 10.1161/01.res.83.8.852. [DOI] [PubMed] [Google Scholar]
- 72.Krikun G, Schatz F, Finlay T, et al. Expression of angiopoietin-2 by human endometrial endothelial cells: regulation by hypoxia and inflammation. Biochem.Biophys.Res.Commun. 2000 Aug 18;275(1):159. doi: 10.1006/bbrc.2000.3277. [DOI] [PubMed] [Google Scholar]
- 73.Mandriota SJ, Pyke C, Di Sanza C, Quinodoz P, Pittet B, Pepper MS. Hypoxia-inducible angiopoietin-2 expression is mimicked by iodonium compounds and occurs in the rat brain and skin in response to systemic hypoxia and tissue ischemia. Am.J.Pathol. 2000 Jun;156(6):2077. doi: 10.1016/S0002-9440(10)65079-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Oh H, Takagi H, Suzuma K, Otani A, Matsumura M, Honda Y. Hypoxia and vascular endothelial growth factor selectively up-regulate angiopoietin-2 in bovine microvascular endothelial cells. J.Biol.Chem. 1999 May 28;274(22):15732. doi: 10.1074/jbc.274.22.15732. [DOI] [PubMed] [Google Scholar]
- 75.Ray PS, Estrada-Hernandez T, Sasaki H, Zhu L, Maulik N. Early effects of hypoxia/reoxygenation on VEGF, ang-1, ang-2 and their receptors in the rat myocardium: implications for myocardial angiogenesis. Mol.Cell.Biochem. 2000 Oct;213(12):145. doi: 10.1023/a:1007180518474. [DOI] [PubMed] [Google Scholar]
- 76.Holash J, Maisonpierre PC, Compton D, et al. Vessel cooption, regression, and growth in tumors mediated by angiopoietins and VEGF. Science. 1999 Jun 18;284(5422):1994. doi: 10.1126/science.284.5422.1994. [DOI] [PubMed] [Google Scholar]
- 77.Scharpfenecker M, Fiedler U, Reiss Y, Augustin HG. The Tie-2 ligand angiopoietin-2 destabilizes quiescent endothelium through an internal autocrine loop mechanism. J.Cell.Sci. 2005 Feb 15;118(Pt 4):771. doi: 10.1242/jcs.01653. [DOI] [PubMed] [Google Scholar]
- 78.Post S, Peeters W, Busser E, et al. Balance between angiopoietin-1 and angiopoietin-2 is in favor of angiopoietin-2 in atherosclerotic plaques with high microvessel density. J.Vasc.Res. 2008;45(3):244. doi: 10.1159/000112939. [DOI] [PubMed] [Google Scholar]
- 79.Beck H, Acker T, Wiessner C, Allegrini PR, Plate KH. Expression of angiopoietin-1, angiopoietin-2, and tie receptors after middle cerebral artery occlusion in the rat. Am.J.Pathol. 2000 Nov;157(5):1473. doi: 10.1016/S0002-9440(10)64786-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Reiss Y, Droste J, Heil M, et al. Angiopoietin-2 impairs revascularization after limb ischemia. Circ.Res. 2007 Jul 6;101(1):88. doi: 10.1161/CIRCRESAHA.106.143594. [DOI] [PubMed] [Google Scholar]
- 81.Lobov IB, Brooks PC, Lang RA. Angiopoietin-2 displays VEGF-dependent modulation of capillary structure and endothelial cell survival in vivo. Proc.Natl.Acad.Sci.U.S.A. 2002 Aug 20;99(17):11205. doi: 10.1073/pnas.172161899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hammes HP, Lin J, Wagner P, et al. Angiopoietin-2 causes pericyte dropout in the normal retina: evidence for involvement in diabetic retinopathy. Diabetes. 2004 Apr;53(4):1104–1110. doi: 10.2337/diabetes.53.4.1104. [DOI] [PubMed] [Google Scholar]
- 83.Zhang L, Yang N, Park JW, et al. Tumor-derived vascular endothelial growth factor up-regulates angiopoietin-2 in host endothelium and destabilizes host vasculature, supporting angiogenesis in ovarian cancer. Cancer Res. 2003 Jun 15;63(12):3403–3412. [PubMed] [Google Scholar]
- 84.Collo G, Pepper MS. Endothelial cell integrin alpha5beta1 expression is modulated by cytokines and during migration in vitro. J Cell Sci. 1999 Feb;112(Pt 4):569–578. doi: 10.1242/jcs.112.4.569. [DOI] [PubMed] [Google Scholar]
- 85.Roberts AB, Sporn MB, Assoian RK, et al. Transforming growth factor type beta: rapid induction of fibrosis and angiogenesis in vivo and stimulation of collagen formation in vitro. Proc Natl Acad Sci U S A. 1986 Jun;83(12):4167–4171. doi: 10.1073/pnas.83.12.4167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Li J, Zhang YP, Kirsner RS. Angiogenesis in wound repair: angiogenic growth factors and the extracellular matrix. Microsc Res Tech. 2003 Jan 1;60(1):107–114. doi: 10.1002/jemt.10249. [DOI] [PubMed] [Google Scholar]
- 87.Sankar S, Mahooti-Brooks N, Bensen L, McCarthy TL, Centrella M, Madri JA. Modulation of transforming growth factor beta receptor levels on microvascular endothelial cells during in vitro angiogenesis. J Clin Invest. 1996 Mar 15;97(6):1436–1446. doi: 10.1172/JCI118565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kim KY, Jeong SY, Won J, Ryu PD, Nam MJ. Induction of angiogenesis by expression of soluble type II transforming growth factor-beta receptor in mouse hepatoma. J Biol Chem. 2001 Oct 19;276(42):38781–38786. doi: 10.1074/jbc.M104944200. [DOI] [PubMed] [Google Scholar]
- 89.Klagsbrun M, D’Amore PA. Regulators of angiogenesis. Annu Rev Physiol. 1991;53:217–239. doi: 10.1146/annurev.ph.53.030191.001245. [DOI] [PubMed] [Google Scholar]
- 90.Visse R, Nagase H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function, and biochemistry. Circ Res. 2003 May 2;92(8):827–839. doi: 10.1161/01.RES.0000070112.80711.3D. [DOI] [PubMed] [Google Scholar]
- 91.van Hinsbergh VW, Koolwijk P. Endothelial sprouting and angiogenesis: matrix metalloproteinases in the lead. Cardiovasc Res. 2008 May 1;78(2):203–212. doi: 10.1093/cvr/cvm102. [DOI] [PubMed] [Google Scholar]
- 92.Koivunen E, Arap W, Valtanen H, et al. Tumor targeting with a selective gelatinase inhibitor. Nat.Biotechnol. 1999 Aug;17(8):768. doi: 10.1038/11703. [DOI] [PubMed] [Google Scholar]
- 93.Itoh T, Tanioka M, Yoshida H, Yoshioka T, Nishimoto H, Itohara S. Reduced angiogenesis and tumor progression in gelatinase A-deficient mice. Cancer Res. 1998 Mar 1;58(5):1048. [PubMed] [Google Scholar]
- 94.Wilson CL, Heppner KJ, Labosky PA, Hogan BL, Matrisian LM. Intestinal tumorigenesis is suppressed in mice lacking the metalloproteinase matrilysin. Proc.Natl.Acad.Sci.U.S.A. 1997 Feb 18;94(4):1402. doi: 10.1073/pnas.94.4.1402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Montesano R, Pepper MS, Vassalli JD, Orci L. Phorbol ester induces cultured endothelial cells to invade a fibrin matrix in the presence of fibrinolytic inhibitors. J.Cell.Physiol. 1987 Sep;132(3):509. doi: 10.1002/jcp.1041320313. [DOI] [PubMed] [Google Scholar]
- 96.Egeblad M, Werb Z. New functions for the matrix metalloproteinases in cancer progression. Nat.Rev.Cancer. 2002 Mar;2(3):161. doi: 10.1038/nrc745. [DOI] [PubMed] [Google Scholar]
- 97.Mignatti P, Rifkin DB. Biology and biochemistry of proteinases in tumor invasion. Physiol.Rev. 1993 Jan;73(1):161. doi: 10.1152/physrev.1993.73.1.161. [DOI] [PubMed] [Google Scholar]
- 98.Murphy G, Stanton H, Cowell S, et al. Mechanisms for pro matrix metalloproteinase activation. Apmis. 1999 Jan;107(1):38. doi: 10.1111/j.1699-0463.1999.tb01524.x. [DOI] [PubMed] [Google Scholar]
- 99.Mazar AP, Henkin J, Goldfarb RH. The urokinase plasminogen activator system in cancer: implications for tumor angiogenesis and metastasis. Angiogenesis. 1999;3(1):15. doi: 10.1023/a:1009095825561. [DOI] [PubMed] [Google Scholar]
- 100.Collen D. The plasminogen (fibrinolytic) system. Thromb.Haemost. 1999 Aug;82(2):259. [PubMed] [Google Scholar]
- 101.Pepper MS, Montesano R, Mandriota SJ, Orci L, Vassalli JD. Angiogenesis: a paradigm for balanced extracellular proteolysis during cell migration and morphogenesis. Enzyme Protein. 1996;49(13):138. doi: 10.1159/000468622. [DOI] [PubMed] [Google Scholar]
- 102.Pepper MS. Role of the matrix metalloproteinase and plasminogen activator-plasmin systems in angiogenesis. Arterioscler.Thromb.Vasc.Biol. 2001 Jul;21(7):1104. doi: 10.1161/hq0701.093685. [DOI] [PubMed] [Google Scholar]
- 103.Veklich Y, Francis CW, White J, Weisel JW. Structural studies of fibrinolysis by electron microscopy. Blood. 1998 Dec 15;92(12):4721. [PubMed] [Google Scholar]
- 104.Pilcher BK, Dumin JA, Sudbeck BD, Krane SM, Welgus HG, Parks WC. The activity of collagenase-1 is required for keratinocyte migration on a type I collagen matrix. J.Cell Biol. 1997 Jun 16;137(6):1445. doi: 10.1083/jcb.137.6.1445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Nguyen M, Arkell J, Jackson CJ. Three-dimensional collagen matrices induce delayed but sustained activation of gelatinase A in human endothelial cells via MT1-MMP. Int.J.Biochem.Cell Biol. 2000 Jun;32(6):621. doi: 10.1016/s1357-2725(00)00013-3. [DOI] [PubMed] [Google Scholar]
- 106.Yebra M, Parry GC, Stromblad S, et al. Requirement of receptor-bound urokinase-type plasminogen activator for integrin alphavbeta5-directed cell migration. J Biol Chem. 1996 Nov 15;271(46):29393–29399. doi: 10.1074/jbc.271.46.29393. [DOI] [PubMed] [Google Scholar]
- 107.Koolwijk P, van Erck MG, de Vree WJ, et al. Cooperative effect of TNFalpha, bFGF, and VEGF on the formation of tubular structures of human microvascular endothelial cells in a fibrin matrix. Role of urokinase activity. J.Cell Biol. 1996 Mar;132(6):1177. doi: 10.1083/jcb.132.6.1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Collen A, Koolwijk P, Kroon M, van Hinsbergh VW. Influence of fibrin structure on the formation and maintenance of capillary-like tubules by human microvascular endothelial cells. Angiogenesis. 1998;2(2):153. doi: 10.1023/a:1009240522808. [DOI] [PubMed] [Google Scholar]
- 109.Soff GA, Sanderowitz J, Gately S, et al. Expression of plasminogen activator inhibitor type 1 by human prostate carcinoma cells inhibits primary tumor growth, tumor-associated angiogenesis, and metastasis to lung and liver in an athymic mouse model. J.Clin.Invest. 1995 Dec;96(6):2593. doi: 10.1172/JCI118323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Levi E, Fridman R, Miao HQ, Ma YS, Yayon A, Vlodavsky I. Matrix metalloproteinase 2 releases active soluble ectodomain of fibroblast growth factor receptor 1. Proc.Natl.Acad.Sci.U.S.A. 1996 Jul 9;93(14):7069. doi: 10.1073/pnas.93.14.7069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Chang C, Werb Z. The many faces of metalloproteases: cell growth, invasion, angiogenesis and metastasis. Trends Cell Biol. 2001 Nov;11(11):S37. doi: 10.1016/s0962-8924(01)02122-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Saunders WB, Bohnsack BL, Faske JB, et al. Coregulation of vascular tube stabilization by endothelial cell TIMP-2 and pericyte TIMP-3. J.Cell Biol. 2006 Oct 9;175(1):179. doi: 10.1083/jcb.200603176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Bayless KJ, Davis GE. Sphingosine-1-phosphate markedly induces matrix metalloproteinase and integrin-dependent human endothelial cell invasion and lumen formation in three-dimensional collagen and fibrin matrices. Biochem.Biophys.Res.Commun. 2003 Dec 26;312(4):903. doi: 10.1016/j.bbrc.2003.11.017. [DOI] [PubMed] [Google Scholar]
- 114.Davis GE, Saunders WB. Molecular balance of capillary tube formation versus regression in wound repair: role of matrix metalloproteinases and their inhibitors. J.Investig.Dermatol.Symp.Proc. 2006 Sep;11(1):44. doi: 10.1038/sj.jidsymp.5650008. [DOI] [PubMed] [Google Scholar]
- 115.Chun TH, Sabeh F, Ota I, et al. MT1-MMP-dependent neovessel formation within the confines of the three-dimensional extracellular matrix. J.Cell Biol. 2004 Nov 22;167(4):757. doi: 10.1083/jcb.200405001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Plaisier M, Kapiteijn K, Koolwijk P, et al. Involvement of membrane-type matrix metalloproteinases (MT-MMPs) in capillary tube formation by human endometrial microvascular endothelial cells: role of MT3-MMP. J.Clin.Endocrinol.Metab. 2004 Nov;89(11):5828. doi: 10.1210/jc.2004-0860. [DOI] [PubMed] [Google Scholar]
- 117.Montesano R. Cell-extracellular matrix interactions in morphogenesis: an in vitro approach. Experientia. 1986 Sep 15;42(9):977–985. doi: 10.1007/BF01940700. [DOI] [PubMed] [Google Scholar]
- 118.Addison CL, Nor JE, Zhao H, Linn SA, Polverini PJ, Delaney CE. The response of VEGF-stimulated endothelial cells to angiostatic molecules is substrate-dependent. BMC Cell Biol. 2005;6:38. doi: 10.1186/1471-2121-6-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Jakobsson L, Domogatskaya A, Tryggvason K, Edgar D, Claesson-Welsh L. Laminin deposition is dispensable for vasculogenesis but regulates blood vessel diameter independent of flow. Faseb J. 2008 May;22(5):1530. doi: 10.1096/fj.07-9617com. [DOI] [PubMed] [Google Scholar]
- 120.Fujita M, Khazenzon NM, Bose S, et al. Overexpression of beta1-chain-containing laminins in capillary basement membranes of human breast cancer and its metastases. Breast Cancer Res. 2005;7(4):R411–421. doi: 10.1186/bcr1011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Ljubimova JY, Fujita M, Khazenzon NM, Ljubimov AV, Black KL. Changes in laminin isoforms associated with brain tumor invasion and angiogenesis. Front Biosci. 2006;11:81–88. doi: 10.2741/1781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Zhou X, Rowe RG, Hiraoka N, et al. Fibronectin fibrillogenesis regulates three-dimensional neovessel formation. Genes Dev. 2008 May 1;22(9):1231. doi: 10.1101/gad.1643308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Smith JD, Melhem ME, Magge KT, Waggoner AS, Campbell PG. Improved growth factor directed vascularization into fibrin constructs through inclusion of additional extracellular molecules. Microvasc.Res. 2007 Mar;73(2):84. doi: 10.1016/j.mvr.2006.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Kroon ME, van Schie ML, van der Vecht B, van Hinsbergh VW, Koolwijk P. Collagen type 1 retards tube formation by human microvascular endothelial cells in a fibrin matrix. Angiogenesis. 2002;5(4):257–265. doi: 10.1023/a:1024540701634. [DOI] [PubMed] [Google Scholar]
- 125.Haas TL, Davis SJ, Madri JA. Three-dimensional type I collagen lattices induce coordinate expression of matrix metalloproteinases MT1-MMP and MMP-2 in microvascular endothelial cells. J.Biol.Chem. 1998 Feb 6;273(6):3604. doi: 10.1074/jbc.273.6.3604. [DOI] [PubMed] [Google Scholar]
- 126.Stegemann JP, Nerem RM. Altered response of vascular smooth muscle cells to exogenous biochemical stimulation in two- and three-dimensional culture. Exp.Cell Res. 2003 Feb 15;283(2):146. doi: 10.1016/s0014-4827(02)00041-1. [DOI] [PubMed] [Google Scholar]
- 127.Ghajar CM, Blevins KS, Hughes CC, George SC, Putnam AJ. Mesenchymal stem cells enhance angiogenesis in mechanically viable prevascularized tissues via early matrix metalloproteinase upregulation. Tissue Eng. 2006 Oct;12(10):2875. doi: 10.1089/ten.2006.12.2875. [DOI] [PubMed] [Google Scholar]
- 128.Yao C, Markowicz M, Pallua N, Noah EM, Steffens G. The effect of cross-linking of collagen matrices on their angiogenic capability. Biomaterials. 2008 Jan;29(1):66–74. doi: 10.1016/j.biomaterials.2007.08.049. [DOI] [PubMed] [Google Scholar]
- 129.van Hinsbergh VW, Collen A, Koolwijk P. Role of fibrin matrix in angiogenesis. Ann.N.Y.Acad.Sci. 2001;936:426. doi: 10.1111/j.1749-6632.2001.tb03526.x. [DOI] [PubMed] [Google Scholar]
- 130.Nehls V, Herrmann R. The configuration of fibrin clots determines capillary morphogenesis and endothelial cell migration. Microvasc.Res. 1996 May;51(3):347. doi: 10.1006/mvre.1996.0032. [DOI] [PubMed] [Google Scholar]
- 131.Moiseeva EP. Adhesion receptors of vascular smooth muscle cells and their functions. Cardiovasc.Res. 2001 Dec;52(3):372. doi: 10.1016/s0008-6363(01)00399-6. [DOI] [PubMed] [Google Scholar]
- 132.Romer LH, Birukov KG, Garcia JG. Focal adhesions: paradigm for a signaling nexus. Circ.Res. 2006 Mar 17;98(5):606. doi: 10.1161/01.RES.0000207408.31270.db. [DOI] [PubMed] [Google Scholar]
- 133.Coppolino MG, Dedhar S. Bi-directional signal transduction by integrin receptors. Int.J.Biochem.Cell Biol. 2000 Feb;32(2):171. doi: 10.1016/s1357-2725(99)00043-6. [DOI] [PubMed] [Google Scholar]
- 134.Parsons JT. Focal adhesion kinase: the first ten years. J.Cell.Sci. 2003 Apr 15;116(Pt 8):1409. doi: 10.1242/jcs.00373. [DOI] [PubMed] [Google Scholar]
- 135.Wiesner S, Legate KR, Fassler R. Integrin-actin interactions. Cell Mol.Life Sci. 2005 May;62(10):1081. doi: 10.1007/s00018-005-4522-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Bayless KJ, Salazar R, Davis GE. RGD-dependent vacuolation and lumen formation observed during endothelial cell morphogenesis in three-dimensional fibrin matrices involves the alpha(v)beta(3) and alpha(5)beta(1) integrins. Am.J.Pathol. 2000 May;156(5):1673. doi: 10.1016/s0002-9440(10)65038-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Risau W, Lemmon V. Changes in the vascular extracellular matrix during embryonic vasculogenesis and angiogenesis. Dev.Biol. 1988 Feb;125(2):441. doi: 10.1016/0012-1606(88)90225-4. [DOI] [PubMed] [Google Scholar]
- 138.Stupack DG, Cheresh DA. Integrins and angiogenesis. Curr.Top.Dev.Biol. 2004;64:207. doi: 10.1016/S0070-2153(04)64009-9. [DOI] [PubMed] [Google Scholar]
- 139.Francis SE, Goh KL, Hodivala-Dilke K, et al. Central roles of alpha5beta1 integrin and fibronectin in vascular development in mouse embryos and embryoid bodies. Arterioscler.Thromb.Vasc.Biol. 2002 Jun 1;22(6):927. doi: 10.1161/01.atv.0000016045.93313.f2. [DOI] [PubMed] [Google Scholar]
- 140.Eliceiri BP, Cheresh DA. The role of alphav integrins during angiogenesis: insights into potential mechanisms of action and clinical development. J.Clin.Invest. 1999 May;103(9):1227. doi: 10.1172/JCI6869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Senger DR, Claffey KP, Benes JE, Perruzzi CA, Sergiou AP, Detmar M. Angiogenesis promoted by vascular endothelial growth factor: regulation through alpha1beta1 and alpha2beta1 integrins. Proc Natl Acad Sci U S A. 1997 Dec 9;94(25):13612–13617. doi: 10.1073/pnas.94.25.13612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Rupp PA, Little CD. Integrins in vascular development. Circ.Res. 2001 Sep 28;89(7):566. doi: 10.1161/hh1901.097747. [DOI] [PubMed] [Google Scholar]
- 143.Perruzzi CA, de Fougerolles AR, Koteliansky VE, Whelan MC, Westlin WF, Senger DR. Functional overlap and cooperativity among alphav and beta1 integrin subfamilies during skin angiogenesis. J.Invest.Dermatol. 2003 Jun;120(6):1100. doi: 10.1046/j.1523-1747.2003.12236.x. [DOI] [PubMed] [Google Scholar]
- 144.Takahashi S, Leiss M, Moser M, et al. The RGD motif in fibronectin is essential for development but dispensable for fibril assembly. J.Cell Biol. 2007 Jul 2;178(1):167. doi: 10.1083/jcb.200703021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Goh KL, Yang JT, Hynes RO. Mesodermal defects and cranial neural crest apoptosis in alpha5 integrin-null embryos. Development. 1997 Nov;124(21):4309. doi: 10.1242/dev.124.21.4309. [DOI] [PubMed] [Google Scholar]
- 146.Ilic D, Kovacic B, McDonagh S, et al. Focal adhesion kinase is required for blood vessel morphogenesis. Circ.Res. 2003 Feb 21;92(3):300. doi: 10.1161/01.res.0000055016.36679.23. [DOI] [PubMed] [Google Scholar]
- 147.Jalali S, del Pozo MA, Chen K, et al. Integrin-mediated mechanotransduction requires its dynamic interaction with specific extracellular matrix (ECM) ligands. Proc Natl Acad Sci U S A. 2001 Jan 30;98(3):1042–1046. doi: 10.1073/pnas.031562998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Gamble JR, Matthias LJ, Meyer G, et al. Regulation of in vitro capillary tube formation by anti-integrin antibodies. J.Cell Biol. 1993 May;121(4):931. doi: 10.1083/jcb.121.4.931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Brooks PC, Stromblad S, Sanders LC, et al. Localization of matrix metalloproteinase MMP-2 to the surface of invasive cells by interaction with integrin alpha v beta 3. Cell. 1996 May 31;85(5):683–693. doi: 10.1016/s0092-8674(00)81235-0. [DOI] [PubMed] [Google Scholar]
- 150.Seltzer JL, Lee AY, Akers KT, et al. Activation of 72-kDa type IV collagenase/gelatinase by normal fibroblasts in collagen lattices is mediated by integrin receptors but is not related to lattice contraction. Exp.Cell Res. 1994 Aug;213(2):365. doi: 10.1006/excr.1994.1211. [DOI] [PubMed] [Google Scholar]
- 151.Giancotti FG, Ruoslahti E. Integrin signaling. Science. 1999 Aug 13;285(5430):1028. doi: 10.1126/science.285.5430.1028. [DOI] [PubMed] [Google Scholar]
- 152.Senger DR, Ledbetter SR, Claffey KP, Papadopoulos-Sergiou A, Peruzzi CA, Detmar M. Stimulation of endothelial cell migration by vascular permeability factor/vascular endothelial growth factor through cooperative mechanisms involving the alphavbeta3 integrin, osteopontin, and thrombin. Am J Pathol. 1996 Jul;149(1):293–305. [PMC free article] [PubMed] [Google Scholar]
- 153.Ross RS. Molecular and mechanical synergy: cross-talk between integrins and growth factor receptors. Cardiovasc.Res. 2004 Aug 15;63(3):381. doi: 10.1016/j.cardiores.2004.04.027. [DOI] [PubMed] [Google Scholar]
- 154.Sepp NT, Li LJ, Lee KH, et al. Basic fibroblast growth factor increases expression of the alpha v beta 3 integrin complex on human microvascular endothelial cells. J Invest Dermatol. 1994 Sep;103(3):295–299. doi: 10.1111/1523-1747.ep12394617. [DOI] [PubMed] [Google Scholar]
- 155.Mahabeleshwar GH, Feng W, Reddy K, Plow EF, Byzova TV. Mechanisms of integrin-vascular endothelial growth factor receptor cross-activation in angiogenesis. Circ.Res. 2007 Sep 14;101(6):570. doi: 10.1161/CIRCRESAHA.107.155655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Munoz-Chapuli R, Quesada AR, Medina M Angel. Angiogenesis and signal transduction in endothelial cells. Cell Mol Life Sci. 2004 Sep;61(17):2224–2243. doi: 10.1007/s00018-004-4070-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Moon SY, Zheng Y. Rho GTPase-activating proteins in cell regulation. Trends Cell Biol. 2003 Jan;13(1):13–22. doi: 10.1016/s0962-8924(02)00004-1. [DOI] [PubMed] [Google Scholar]
- 158.Ridley AJ. Rho family proteins and regulation of the actin cytoskeleton. Prog.Mol.Subcell.Biol. 1999;22:1. doi: 10.1007/978-3-642-58591-3_1. [DOI] [PubMed] [Google Scholar]
- 159.Soga N, Namba N, McAllister S, et al. Rho family GTPases regulate VEGF-stimulated endothelial cell motility. Exp.Cell Res. 2001 Sep 10;269(1):73. doi: 10.1006/excr.2001.5295. [DOI] [PubMed] [Google Scholar]
- 160.Engelse MA, Laurens N, Verloop RE, Koolwijk P, van Hinsbergh VW. Differential gene expression analysis of tubule forming and non-tubule forming endothelial cells: CDC42GAP as a counter-regulator in tubule formation. Angiogenesis. 2008;11(2):153. doi: 10.1007/s10456-007-9086-9. [DOI] [PubMed] [Google Scholar]
- 161.Koh W, Mahan RD, Davis GE. Cdc42- and Rac1-mediated endothelial lumen formation requires Pak2, Pak4 and Par3, and PKC-dependent signaling. J.Cell.Sci. 2008 Apr 1;121(Pt 7):989. doi: 10.1242/jcs.020693. [DOI] [PubMed] [Google Scholar]
- 162.Yamamura S, Nelson PR, Kent KC. Role of protein kinase C in attachment, spreading, and migration of human endothelial cells. J.Surg.Res. 1996 Jun;63(1):349. doi: 10.1006/jsre.1996.0274. [DOI] [PubMed] [Google Scholar]
- 163.Wang A, Nomura M, Patan S, Ware JA. Inhibition of protein kinase Calpha prevents endothelial cell migration and vascular tube formation in vitro and myocardial neovascularization in vivo. Circ.Res. 2002 Mar 22;90(5):609. doi: 10.1161/01.res.0000012503.30315.e8. [DOI] [PubMed] [Google Scholar]
- 164.Schweisguth F. Regulation of notch signaling activity. Curr.Biol. 2004 Feb 3;14(3):R129. [PubMed] [Google Scholar]
- 165.Leslie JD, Ariza-McNaughton L, Bermange AL, McAdow R, Johnson SL, Lewis J. Endothelial signalling by the Notch ligand Delta-like 4 restricts angiogenesis. Development. 2007 Mar;134(5):839. doi: 10.1242/dev.003244. [DOI] [PubMed] [Google Scholar]
- 166.Siekmann AF, Lawson ND. Notch signalling limits angiogenic cell behaviour in developing zebrafish arteries. Nature. 2007 Feb 15;445(7129):781. doi: 10.1038/nature05577. [DOI] [PubMed] [Google Scholar]
- 167.Hellstrom M, Phng LK, Hofmann JJ, et al. Dll4 signalling through Notch1 regulates formation of tip cells during angiogenesis. Nature. 2007 Feb 15;445(7129):776. doi: 10.1038/nature05571. [DOI] [PubMed] [Google Scholar]
- 168.Sainson RC, Aoto J, Nakatsu MN, et al. Cell-autonomous notch signaling regulates endothelial cell branching and proliferation during vascular tubulogenesis. Faseb J. 2005 Jun;19(8):1027. doi: 10.1096/fj.04-3172fje. [DOI] [PubMed] [Google Scholar]
- 169.Ridgway J, Zhang G, Wu Y, et al. Inhibition of Dll4 signalling inhibits tumour growth by deregulating angiogenesis. Nature. 2006 Dec 21;444(7122):1083. doi: 10.1038/nature05313. [DOI] [PubMed] [Google Scholar]
- 170.Noguera-Troise I, Daly C, Papadopoulos NJ, et al. Blockade of Dll4 inhibits tumour growth by promoting non-productive angiogenesis. Nature. 2006 Dec 21;444(7122):1032. doi: 10.1038/nature05355. [DOI] [PubMed] [Google Scholar]
- 171.Kunz-Schughart LA, Schroeder JA, Wondrak M, et al. Potential of fibroblasts to regulate the formation of three-dimensional vessel-like structures from endothelial cells in vitro. Am.J.Physiol.Cell.Physiol. 2006 May;290(5):C1385. doi: 10.1152/ajpcell.00248.2005. [DOI] [PubMed] [Google Scholar]
- 172.Brey EM, McIntire LV, Johnston CM, Reece GP, Patrick CW., Jr. Three-dimensional, quantitative analysis of desmin and smooth muscle alpha actin expression during angiogenesis. Ann.Biomed.Eng. 2004 Aug;32(8):1100. doi: 10.1114/b:abme.0000036646.17362.c4. [DOI] [PubMed] [Google Scholar]
- 173.Ozerdem U, Grako KA, Dahlin-Huppe K, Monosov E, Stallcup WB. NG2 proteoglycan is expressed exclusively by mural cells during vascular morphogenesis. Dev Dyn. 2001 Oct;222(2):218–227. doi: 10.1002/dvdy.1200. [DOI] [PubMed] [Google Scholar]
- 174.Ozerdem U, Monosov E, Stallcup WB. NG2 proteoglycan expression by pericytes in pathological microvasculature. Microvasc.Res. 2002 Jan;63(1):129. doi: 10.1006/mvre.2001.2376. [DOI] [PubMed] [Google Scholar]
- 175.Kuzuya M, Satake S, Esaki T, et al. Induction of angiogenesis by smooth muscle cell-derived factor: possible role in neovascularization in atherosclerotic plaque. J.Cell.Physiol. 1995 Sep;164(3):658. doi: 10.1002/jcp.1041640324. [DOI] [PubMed] [Google Scholar]
- 176.Davie NJ, Gerasimovskaya EV, Hofmeister SE, et al. Pulmonary artery adventitial fibroblasts cooperate with vasa vasorum endothelial cells to regulate vasa vasorum neovascularization: a process mediated by hypoxia and endothelin-1. Am.J.Pathol. 2006 Jun;168(6):1793. doi: 10.2353/ajpath.2006.050754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Montesano R, Pepper MS, Orci L. Paracrine induction of angiogenesis in vitro by Swiss 3T3 fibroblasts. J.Cell.Sci. 1993 Aug;105(Pt 4):1013. doi: 10.1242/jcs.105.4.1013. (Pt 4) [DOI] [PubMed] [Google Scholar]
- 178.Kale S, Hanai J, Chan B, et al. Microarray analysis of in vitro pericyte differentiation reveals an angiogenic program of gene expression. Faseb J. 2005 Feb;19(2):270. doi: 10.1096/fj.04-1604fje. [DOI] [PubMed] [Google Scholar]
- 179.Chon JH, Wang HS, Chaikof EL. Role of fibronectin and sulfated proteoglycans in endothelial cell migration on a cultured smooth muscle layer. J.Surg.Res. 1997 Sep;72(1):53. doi: 10.1006/jsre.1997.5168. [DOI] [PubMed] [Google Scholar]
- 180.Dietrich F, Lelkes PI. Fine-tuning of a three-dimensional microcarrier-based angiogenesis assay for the analysis of endothelial-mesenchymal cell co-cultures in fibrin and collagen gels. Angiogenesis. 2006;9(3):111. doi: 10.1007/s10456-006-9037-x. [DOI] [PubMed] [Google Scholar]
- 181.Ozerdem U, Stallcup WB. Early contribution of pericytes to angiogenic sprouting and tube formation. Angiogenesis. 2003;6(3):241. doi: 10.1023/B:AGEN.0000021401.58039.a9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Nehls V, Denzer K, Drenckhahn D. Pericyte involvement in capillary sprouting during angiogenesis in situ. Cell Tissue Res. 1992 Dec;270(3):469–474. doi: 10.1007/BF00645048. [DOI] [PubMed] [Google Scholar]
- 183.Reynolds LP, Grazul-Bilska AT, Redmer DA. Angiogenesis in the corpus luteum. Endocrine. 2000 Feb;12(1):1–9. doi: 10.1385/ENDO:12:1:1. [DOI] [PubMed] [Google Scholar]
- 184.Nehls V, Drenckhahn D. A microcarrier-based cocultivation system for the investigation of factors and cells involved in angiogenesis in three-dimensional fibrin matrices in vitro. Histochem.Cell Biol. 1995 Dec;104(6):459. doi: 10.1007/BF01464336. [DOI] [PubMed] [Google Scholar]
- 185.St Croix B, Rago C, Velculescu V, et al. Genes expressed in human tumor endothelium. Science. 2000 Aug 18;289(5482):1197. doi: 10.1126/science.289.5482.1197. [DOI] [PubMed] [Google Scholar]
- 186.Bussolati B, Deambrosis I, Russo S, Deregibus MC, Camussi G. Altered angiogenesis and survival in human tumor-derived endothelial cells. Faseb J. 2003 Jun;17(9):1159. doi: 10.1096/fj.02-0557fje. [DOI] [PubMed] [Google Scholar]
- 187.Vallon M, Essler M. Proteolytically processed soluble tumor endothelial marker (TEM) 5 mediates endothelial cell survival during angiogenesis by linking integrin alpha(v)beta3 to glycosaminoglycans. J.Biol.Chem. 2006 Nov 10;281(45):34179. doi: 10.1074/jbc.M605291200. [DOI] [PubMed] [Google Scholar]
- 188.Rmali KA, Puntis MC, Jiang WG. TEM-8 and tubule formation in endothelial cells, its potential role of its vW/TM domains. Biochem.Biophys.Res.Commun. 2005 Aug 19;334(1):231. doi: 10.1016/j.bbrc.2005.06.085. [DOI] [PubMed] [Google Scholar]
- 189.Wang XQ, Sheibani N, Watson JC. Modulation of tumor endothelial cell marker 7 expression during endothelial cell capillary morphogenesis. Microvasc.Res. 2005 Nov;70(3):189. doi: 10.1016/j.mvr.2005.08.004. [DOI] [PubMed] [Google Scholar]
- 190.Rmali KA, Puntis MC, Jiang WG. Prognostic values of tumor endothelial markers in patients with colorectal cancer. World J.Gastroenterol. 2005 Mar 7;11(9):1283. doi: 10.3748/wjg.v11.i9.1283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Davis GE, Senger DR. Endothelial extracellular matrix: biosynthesis, remodeling, and functions during vascular morphogenesis and neovessel stabilization. Circ.Res. 2005 Nov 25;97(11):1093. doi: 10.1161/01.RES.0000191547.64391.e3. [DOI] [PubMed] [Google Scholar]
- 192.Bonanno E, Iurlaro M, Madri JA, Nicosia RF. Type IV collagen modulates angiogenesis and neovessel survival in the rat aorta model. In Vitro Cell.Dev.Biol.Anim. 2000 May;36(5):336. doi: 10.1290/1071-2690(2000)036<0336:TICMAA>2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 193.Colorado PC, Torre A, Kamphaus G, et al. Anti-angiogenic Cues from Vascular Basement Membrane Collagen. Cancer Res. 2000 May 1;60(9):2520–2526. 2000. [PubMed] [Google Scholar]
- 194.Kamphaus GD, Colorado PC, Panka DJ, et al. Canstatin, a Novel Matrix-derived Inhibitor of Angiogenesis and Tumor Growth. J. Biol. Chem. 2000 January 14;275(2):1209–1215. doi: 10.1074/jbc.275.2.1209. 2000. [DOI] [PubMed] [Google Scholar]
- 195.DiPietro LA, Nissen NN, Gamelli RL, Koch AE, Pyle JM, Polverini PJ. Thrombospondin 1 synthesis and function in wound repair. Am J Pathol. 1996 Jun;148(6):1851–1860. [PMC free article] [PubMed] [Google Scholar]
- 196.Ren B, Yee KO, Lawler J, Khosravi-Far R. Regulation of tumor angiogenesis by thrombospondin-1. Biochimica et Biophysica Acta (BBA) - Reviews on Cancer. 2006;1765(2):178–188. doi: 10.1016/j.bbcan.2005.11.002. [DOI] [PubMed] [Google Scholar]
- 197.Baker LH, Rowinsky EK, Mendelson D, et al. Randomized, phase II study of the thrombospondin-1-mimetic angiogenesis inhibitor ABT-510 in patients with advanced soft tissue sarcoma. J Clin Oncol. 2008 Dec 1;26(34):5583–5588. doi: 10.1200/JCO.2008.17.4706. [DOI] [PubMed] [Google Scholar]
- 198.Kulke MH, Bergsland EK, Ryan DP, et al. Phase II study of recombinant human endostatin in patients with advanced neuroendocrine tumors. J.Clin.Oncol. 2006 Aug 1;24(22):3555. doi: 10.1200/JCO.2006.05.6762. [DOI] [PubMed] [Google Scholar]
- 199.Khasigov PZ, Podobed OV, Gracheva TS, Salbiev KD, Grachev SV, Berezov TT. Role of matrix metalloproteinases and their inhibitors in tumor invasion and metastasis. Biochemistry (Mosc) 2003 Jul;68(7):711. doi: 10.1023/a:1025051214001. [DOI] [PubMed] [Google Scholar]
- 200.Qi JH, Ebrahem Q, Moore N, et al. A novel function for tissue inhibitor of metalloproteinases-3 (TIMP3): inhibition of angiogenesis by blockage of VEGF binding to VEGF receptor-2. Nat.Med. 2003 Apr;9(4):407. doi: 10.1038/nm846. [DOI] [PubMed] [Google Scholar]
- 201.Bergers G, Song S. The role of pericytes in blood-vessel formation and maintenance. Neuro Oncol. 2005 Oct;7(4):452–464. doi: 10.1215/S1152851705000232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Lindahl P, Johansson BR, Leveen P, Betsholtz C. Pericyte loss and microaneurysm formation in PDGF-B-deficient mice. Science. 1997 Jul 11;277(5323):242. doi: 10.1126/science.277.5323.242. [DOI] [PubMed] [Google Scholar]
- 203.Morikawa S, Baluk P, Kaidoh T, Haskell A, Jain RK, McDonald DM. Abnormalities in pericytes on blood vessels and endothelial sprouts in tumors. Am J Pathol. 2002 Mar;160(3):985–1000. doi: 10.1016/S0002-9440(10)64920-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Benjamin LE, Golijanin D, Itin A, Pode D, Keshet E. Selective ablation of immature blood vessels in established human tumors follows vascular endothelial growth factor withdrawal. J.Clin.Invest. 1999 Jan;103(2):159. doi: 10.1172/JCI5028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Song S, Ewald AJ, Stallcup W, Werb Z, Bergers G. PDGFRbeta+ perivascular progenitor cells in tumours regulate pericyte differentiation and vascular survival. Nat Cell Biol. 2005 Sep;7(9):870–879. doi: 10.1038/ncb1288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Sennino B, Falcon BL, McCauley D, et al. Sequential loss of tumor vessel pericytes and endothelial cells after inhibition of platelet-derived growth factor B by selective aptamer AX102. Cancer Res. 2007 Aug 1;67(15):7358–7367. doi: 10.1158/0008-5472.CAN-07-0293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Sussman MA. Showing up isn’t enough for vascularization: persistence is essential. Circ Res. 2008 Nov 21;103(11):1200–1201. doi: 10.1161/CIRCRESAHA.108.189076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Benjamin LE, Hemo I, Keshet E. A plasticity window for blood vessel remodelling is defined by pericyte coverage of the preformed endothelial network and is regulated by PDGF-B and VEGF. Development. 1998 May;125(9):1591–1598. doi: 10.1242/dev.125.9.1591. [DOI] [PubMed] [Google Scholar]
- 209.Rucker HK, Wynder HJ, Thomas WE. Cellular mechanisms of CNS pericytes. Brain Res Bull. 2000 Mar 15;51(5):363–369. doi: 10.1016/s0361-9230(99)00260-9. [DOI] [PubMed] [Google Scholar]
- 210.Gerhardt H, Wolburg H, Redies C. N-cadherin mediates pericytic-endothelial interaction during brain angiogenesis in the chicken. Dev Dyn. 2000 Jul;218(3):472–479. doi: 10.1002/1097-0177(200007)218:3<472::AID-DVDY1008>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- 211.Gerhardt H, Betsholtz C. Endothelial-pericyte interactions in angiogenesis. Cell Tissue Res. 2003 Oct;314(1):15–23. doi: 10.1007/s00441-003-0745-x. [DOI] [PubMed] [Google Scholar]
- 212.Tillet E, Vittet D, Feraud O, Moore R, Kemler R, Huber P. N-cadherin deficiency impairs pericyte recruitment, and not endothelial differentiation or sprouting, in embryonic stem cell-derived angiogenesis. Exp Cell Res. 2005 Nov 1;310(2):392–400. doi: 10.1016/j.yexcr.2005.08.021. [DOI] [PubMed] [Google Scholar]
- 213.Andrae J, Gallini R, Betsholtz C. Role of platelet-derived growth factors in physiology and medicine. Genes Dev. 2008 May 15;22(10):1276–1312. doi: 10.1101/gad.1653708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Clowes AW. Regulation of smooth muscle cell proliferation and migration. Transplant.Proc. 1999 Feb-Mar;31(12):810. doi: 10.1016/s0041-1345(98)01781-3. [DOI] [PubMed] [Google Scholar]
- 215.Fredriksson L, Li H, Eriksson U. The PDGF family: four gene products form five dimeric isoforms. Cytokine Growth Factor Rev. 2004 Aug;15(4):197. doi: 10.1016/j.cytogfr.2004.03.007. [DOI] [PubMed] [Google Scholar]
- 216.Soriano P. Abnormal kidney development and hematological disorders in PDGF beta-receptor mutant mice. Genes Dev. 1994 Aug 15;8(16):1888–1896. doi: 10.1101/gad.8.16.1888. [DOI] [PubMed] [Google Scholar]
- 217.Leveen P, Pekny M, Gebre-Medhin S, Swolin B, Larsson E, Betsholtz C. Mice deficient for PDGF B show renal, cardiovascular, and hematological abnormalities. Genes Dev. 1994 Aug 15;8(16):1875–1887. doi: 10.1101/gad.8.16.1875. [DOI] [PubMed] [Google Scholar]
- 218.Hellstrom M, Kalen M, Lindahl P, Abramsson A, Betsholtz C. Role of PDGF-B and PDGFR-beta in recruitment of vascular smooth muscle cells and pericytes during embryonic blood vessel formation in the mouse. Development. 1999 Jun;126(14):3047–3055. doi: 10.1242/dev.126.14.3047. [DOI] [PubMed] [Google Scholar]
- 219.Abramsson A, Lindblom P, Betsholtz C. Endothelial and nonendothelial sources of PDGF-B regulate pericyte recruitment and influence vascular pattern formation in tumors. J Clin Invest. 2003 Oct;112(8):1142–1151. doi: 10.1172/JCI18549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Guo P, Hu B, Gu W, et al. Platelet-derived growth factor-B enhances glioma angiogenesis by stimulating vascular endothelial growth factor expression in tumor endothelia and by promoting pericyte recruitment. Am J Pathol. 2003 Apr;162(4):1083–1093. doi: 10.1016/S0002-9440(10)63905-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Lindblom P, Gerhardt H, Liebner S, et al. Endothelial PDGF-B retention is required for proper investment of pericytes in the microvessel wall. Genes Dev. 2003 Aug 1;17(15):1835–1840. doi: 10.1101/gad.266803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Powell RJ, Carruth JA, Basson MD, Bloodgood R, Sumpio BE. Matrix-specific effect of endothelial control of smooth muscle cell migration. J.Vasc.Surg. 1996 Jul;24(1):51. doi: 10.1016/s0741-5214(96)70144-1. [DOI] [PubMed] [Google Scholar]
- 223.Yoshida H, Nakamura M, Makita S, Hiramori K. Paracrine effect of human vascular endothelial cells on human vascular smooth muscle cell proliferation: transmembrane co-culture method. Heart Vessels. 1996;11(5):229. doi: 10.1007/BF01746202. [DOI] [PubMed] [Google Scholar]
- 224.Newcomb PM, Herman IM. Pericyte growth and contractile phenotype: modulation by endothelial-synthesized matrix and comparison with aortic smooth muscle. J.Cell.Physiol. 1993 May;155(2):385. doi: 10.1002/jcp.1041550220. [DOI] [PubMed] [Google Scholar]
- 225.Axel DI, Riessen R, Athanasiadis A, Runge H, Koveker G, Karsch KR. Growth factor expression of human arterial smooth muscle cells and endothelial cells in a transfilter coculture system. J.Mol.Cell.Cardiol. 1997 Nov;29(11):2967. doi: 10.1006/jmcc.1997.0541. [DOI] [PubMed] [Google Scholar]
- 226.Wu XJ, Huang L, Song DL, Jin J, Zhao G. Effects of endothelial cell growth states on the proliferation and migration of vascular smooth muscle cells in vitro. Sheng Li Xue Bao. 2003 Oct 25;55(5):554. [PubMed] [Google Scholar]
- 227.Fillinger MF, O’Connor SE, Wagner RJ, Cronenwett JL. The effect of endothelial cell coculture on smooth muscle cell proliferation. J.Vasc.Surg. 1993 Jun;17(6):1058. [PubMed] [Google Scholar]
- 228.Hirschi KK, Rohovsky SA, D’Amore PA. PDGF, TGF-beta, and heterotypic cell-cell interactions mediate endothelial cell-induced recruitment of 10T1/2 cells and their differentiation to a smooth muscle fate. J.Cell Biol. 1998 May 4;141(3):805. doi: 10.1083/jcb.141.3.805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Hirschi KK, Rohovsky SA, Beck LH, Smith SR, D’Amore PA. Endothelial cells modulate the proliferation of mural cell precursors via platelet-derived growth factor-BB and heterotypic cell contact. Circ.Res. 1999 Feb 19;84(3):298. doi: 10.1161/01.res.84.3.298. [DOI] [PubMed] [Google Scholar]
- 230.Hirschi KK, Skalak TC, Peirce SM, Little CD. Vascular assembly in natural and engineered tissues. Ann.N.Y.Acad.Sci. 2002 Jun;961:223. doi: 10.1111/j.1749-6632.2002.tb03090.x. [DOI] [PubMed] [Google Scholar]
- 231.Bjarnegard M, Enge M, Norlin J, et al. Endothelium-specific ablation of PDGFB leads to pericyte loss and glomerular, cardiac and placental abnormalities. Development. 2004 April 15;131(8):1847–1857. doi: 10.1242/dev.01080. 2004. [DOI] [PubMed] [Google Scholar]
- 232.Betsholtz C. Insight into the physiological functions of PDGF through genetic studies in mice. Cytokine Growth Factor Rev. 2004 Aug;15(4):215–228. doi: 10.1016/j.cytogfr.2004.03.005. [DOI] [PubMed] [Google Scholar]
- 233.Betsholtz C, Lindblom P, Bjarnegard M, Enge M, Gerhardt H, Lindahl P. Role of platelet-derived growth factor in mesangium development and vasculopathies: lessons from platelet-derived growth factor and platelet-derived growth factor receptor mutations in mice. Curr Opin Nephrol Hypertens. 2004 Jan;13(1):45–52. doi: 10.1097/00041552-200401000-00007. [DOI] [PubMed] [Google Scholar]
- 234.Bergers G, Song S, Meyer-Morse N, Bergsland E, Hanahan D. Benefits of targeting both pericytes and endothelial cells in the tumor vasculature with kinase inhibitors. J.Clin.Invest. 2003 May;111(9):1287. doi: 10.1172/JCI17929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Potapova O, Laird AD, Nannini MA, et al. Contribution of individual targets to the antitumor efficacy of the multitargeted receptor tyrosine kinase inhibitor SU11248. Mol Cancer Ther. 2006 May;5(5):1280–1289. doi: 10.1158/1535-7163.MCT-03-0156. [DOI] [PubMed] [Google Scholar]
- 236.Hasumi Y, Klosowska-Wardega A, Furuhashi M, Ostman A, Heldin CH, Hellberg C. Identification of a subset of pericytes that respond to combination therapy targeting PDGF and VEGF signaling. Int J Cancer. 2007 Dec 15;121(12):2606–2614. doi: 10.1002/ijc.22999. [DOI] [PubMed] [Google Scholar]
- 237.Timke C, Zieher H, Roth A, et al. Combination of vascular endothelial growth factor receptor/platelet-derived growth factor receptor inhibition markedly improves radiation tumor therapy. Clin Cancer Res. 2008 Apr 1;14(7):2210–2219. doi: 10.1158/1078-0432.CCR-07-1893. [DOI] [PubMed] [Google Scholar]
- 238.Kuhnert F, Tam BY, Sennino B, et al. Soluble receptor-mediated selective inhibition of VEGFR and PDGFRbeta signaling during physiologic and tumor angiogenesis. Proc Natl Acad Sci U S A. 2008 Jul 22;105(29):10185–10190. doi: 10.1073/pnas.0803194105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Chen RR, Silva EA, Yuen WW, Mooney DJ. Spatio-temporal VEGF and PDGF delivery patterns blood vessel formation and maturation. Pharm.Res. 2007 Feb;24(2):258. doi: 10.1007/s11095-006-9173-4. [DOI] [PubMed] [Google Scholar]
- 240.Korpisalo P, Karvinen H, Rissanen TT, et al. Vascular endothelial growth factor-A and platelet-derived growth factor-B combination gene therapy prolongs angiogenic effects via recruitment of interstitial mononuclear cells and paracrine effects rather than improved pericyte coverage of angiogenic vessels. Circ Res. 2008 Nov 7;103(10):1092–1099. doi: 10.1161/CIRCRESAHA.108.182287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Betsholtz C, Karlsson L, Lindahl P. Developmental roles of platelet-derived growth factors. Bioessays. 2001 Jun;23(6):494–507. doi: 10.1002/bies.1069. [DOI] [PubMed] [Google Scholar]
- 242.Darland DC, D’Amore PA. TGF beta is required for the formation of capillary-like structures in three-dimensional cocultures of 10T1/2 and endothelial cells. Angiogenesis. 2001;4(1):11. doi: 10.1023/a:1016611824696. [DOI] [PubMed] [Google Scholar]
- 243.Lien S-C, Usami S, Chien S, Chiu J-J. Phosphatidylinositol 3-kinase/Akt pathway is involved in transforming growth factor-[beta]1-induced phenotypic modulation of 10T1/2 cells to smooth muscle cells. Cellular Signalling. 2006;18(8):1270–1278. doi: 10.1016/j.cellsig.2005.10.013. [DOI] [PubMed] [Google Scholar]
- 244.Brown DJ, Rzucidlo EM, Merenick BL, Wagner RJ, Martin KA, Powell RJ. Endothelial cell activation of the smooth muscle cell phosphoinositide 3-kinase/Akt pathway promotes differentiation. J.Vasc.Surg. 2005 Mar;41(3):509. doi: 10.1016/j.jvs.2004.12.024. [DOI] [PubMed] [Google Scholar]
- 245.Kutcher ME, Kolyada AY, Surks HK, Herman IM. Pericyte Rho GTPase mediates both pericyte contractile phenotype and capillary endothelial growth state. Am.J.Pathol. 2007 Aug;171(2):693. doi: 10.2353/ajpath.2007.070102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Orlidge A, D’Amore PA. Inhibition of capillary endothelial cell growth by pericytes and smooth muscle cells. J.Cell Biol. 1987 Sep;105(3):1455. doi: 10.1083/jcb.105.3.1455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Korff T, Kimmina S, Martiny-Baron G, Augustin HG. Blood vessel maturation in a 3-dimensional spheroidal coculture model: direct contact with smooth muscle cells regulates endothelial cell quiescence and abrogates VEGF responsiveness. Faseb J. 2001 Feb;15(2):447. doi: 10.1096/fj.00-0139com. [DOI] [PubMed] [Google Scholar]
- 248.Kurzen H, Manns S, Dandekar G, Schmidt T, Pratzel S, Kraling BM. Tightening of endothelial cell contacts: a physiologic response to cocultures with smooth-muscle-like 10T1/2 cells. J.Invest.Dermatol. 2002 Jul;119(1):143. doi: 10.1046/j.1523-1747.2002.01792.x. [DOI] [PubMed] [Google Scholar]
- 249.Shih SC, Ju M, Liu N, Mo JR, Ney JJ, Smith LE. Transforming growth factor beta1 induction of vascular endothelial growth factor receptor 1: mechanism of pericyte-induced vascular survival in vivo. Proc Natl Acad Sci U S A. 2003 Dec 23;100(26):15859–15864. doi: 10.1073/pnas.2136855100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Darland DC, Massingham LJ, Smith SR, Piek E, Saint-Geniez M, D’Amore PA. Pericyte production of cell-associated VEGF is differentiation-dependent and is associated with endothelial survival. Dev Biol. 2003 Dec 1;264(1):275–288. doi: 10.1016/j.ydbio.2003.08.015. [DOI] [PubMed] [Google Scholar]
- 251.Sundberg C, Kowanetz M, Brown LF, Detmar M, Dvorak HF. Stable expression of angiopoietin-1 and other markers by cultured pericytes: phenotypic similarities to a subpopulation of cells in maturing vessels during later stages of angiogenesis in vivo. Lab Invest. 2002 Apr;82(4):387–401. doi: 10.1038/labinvest.3780433. [DOI] [PubMed] [Google Scholar]
- 252.Thurston G, Suri C, Smith K, et al. Leakage-resistant blood vessels in mice transgenically overexpressing angiopoietin-1. Science. 1999 Dec 24;286(5449):2511. doi: 10.1126/science.286.5449.2511. [DOI] [PubMed] [Google Scholar]
- 253.Uemura A, Ogawa M, Hirashima M, et al. Recombinant angiopoietin-1 restores higher-order architecture of growing blood vessels in mice in the absence of mural cells. J Clin Invest. 2002 Dec;110(11):1619–1628. doi: 10.1172/JCI15621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Wilkinson-Berka JL, Babic S, De Gooyer T, et al. Inhibition of platelet-derived growth factor promotes pericyte loss and angiogenesis in ischemic retinopathy. Am J Pathol. 2004 Apr;164(4):1263–1273. doi: 10.1016/s0002-9440(10)63214-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.McIlroy M, O’Rourke M, McKeown SR, Hirst DG, Robson T. Pericytes influence endothelial cell growth characteristics: role of plasminogen activator inhibitor type 1 (PAI-1) Cardiovasc Res. 2006 Jan;69(1):207–217. doi: 10.1016/j.cardiores.2005.08.003. [DOI] [PubMed] [Google Scholar]
- 256.Baker AH, Edwards DR, Murphy G. Metalloproteinase inhibitors: biological actions and therapeutic opportunities. J.Cell.Sci. 2002 Oct 1;115(Pt 19):3719. doi: 10.1242/jcs.00063. [DOI] [PubMed] [Google Scholar]
- 257.Lafleur MA, Forsyth PA, Atkinson SJ, Murphy G, Edwards DR. Perivascular cells regulate endothelial membrane type-1 matrix metalloproteinase activity. Biochem.Biophys.Res.Commun. 2001 Mar 30;282(2):463. doi: 10.1006/bbrc.2001.4596. [DOI] [PubMed] [Google Scholar]
- 258.Grant DS, Kleinman HK. Regulation of capillary formation by laminin and other components of the extracellular matrix. Exs. 1997;79:317. doi: 10.1007/978-3-0348-9006-9_13. [DOI] [PubMed] [Google Scholar]
- 259.Davis GE, Senger DR. Extracellular matrix mediates a molecular balance between vascular morphogenesis and regression. Curr.Opin.Hematol. 2008 May;15(3):197. doi: 10.1097/MOH.0b013e3282fcc321. [DOI] [PubMed] [Google Scholar]
- 260.Zhou Z, Doi M, Wang J, et al. Deletion of laminin-8 results in increased tumor neovascularization and metastasis in mice. Cancer Res. 2004 Jun 15;64(12):4059. doi: 10.1158/0008-5472.CAN-04-0291. [DOI] [PubMed] [Google Scholar]
- 261.Riha GM, Lin PH, Lumsden AB, Yao Q, Chen C. Roles of hemodynamic forces in vascular cell differentiation. Ann.Biomed.Eng. 2005 Jun;33(6):772. doi: 10.1007/s10439-005-3310-9. [DOI] [PubMed] [Google Scholar]
- 262.Lehoux S, Castier Y, Tedgui A. Molecular mechanisms of the vascular responses to haemodynamic forces. J.Intern.Med. 2006 Apr;259(4):381. doi: 10.1111/j.1365-2796.2006.01624.x. [DOI] [PubMed] [Google Scholar]