Abstract
Background
Enthusiasm for laparoscopic surgical approaches to prostate cancer treatment has grown, despite limited evidence of improved outcomes compared with open radical prostatectomy. We compared laparoscopic (with or without robotic assistance) versus open radical prostatectomy in terms of postoperative outcomes and subsequent cancer-directed therapy.
Materials and Methods
Using a population-based cancer registry linked with Medicare claims, we identified men age 66 or older with localized prostate cancer who received a radical prostatectomy from 2003-2005. Outcome measures were general medical/surgical complications and mortality within 90 days following surgery; genitourinary/bowel complications within 365 days; receipt of radiation therapy, androgen deprivation therapy or both within 365 days; length of hospital stay.
Results
Of the 5,923 men,18% received a laparoscopic radical prostatectomy. Adjusting for patient and tumor characteristics, there were no differences in rates of general medical/surgical complications (OR 0.93; 95% CI: 0.77-1.14) or genitourinary/bowel complications (OR 0.96; 95% CI: 0.76-1.22) or in the use of postoperative radiation, androgen deprivation or both (OR 0.80; 95% CI: 0.60-1.08). Laparoscopic prostatectomy was associated with a 35% shorter hospital stay (p<0.0001) and a lower rate of bladder neck/urethral obstruction (OR 0.74; 95% CI 0.58-0.94). In laparoscopic patients, surgeon volume was inversely associated with length of hospital stay and the odds of any genitourinary/bowel complication.
Conclusions
Laparoscopic and open radical prostatectomy have similar rates of postoperative morbidity and use of additional treatment. Men considering prostate cancer surgery should understand the expected benefits and risks of each technique to facilitate decision-making and to set realistic expectations.
Keywords: prostate, prostatic neoplasms, prostatectomy, minimally invasive surgical procedures, laparoscopy
Introduction
Approximately 200,000 men in the US will receive a new diagnosis of prostate cancer yearly, and about one-third of these men will receive surgical treatment.1 Although open retropubic radical prostatectomy (ORP) is the gold standard for definitive resection, laparoscopic radical prostatectomy (LRP) has emerged as an alternative.2-4 Recently, robotic technology has been introduced to reduce the technical complexity associated with conventional LRP and facilitate adoption of the laparoscopic approach.5-7
Enthusiasm for LRP — and specifically for LRP with robotic assistance — has grown rapidly, despite limited evidence of its superiority to ORP. Only one randomized trial has compared ORP and LRP without robotic assistance, in a sample of 120 procedures performed by a single surgeon.8 Most observational studies comparing ORP and LRP have included a small number of patients and surgeons from a single institution.9 Two large population-based analyses were limited by a lack of information regarding tumor characteristics.10-11 Our objective was to compare the outcomes of ORP and LRP in a population-based cohort of men with clinically localized prostate cancer, controlling for patient and tumor characteristics, and to examine the impact of surgeon volume in men treated with LRP.
Materials and Methods
Data
We used Surveillance Epidemiology and End Results (SEER) cancer registry data linked with Medicare claims. SEER, sponsored by the National Cancer Institute (NCI), is a consortium of population-based cancer registries in selected geographic areas, covering approximately 25% of the US population.12 The SEER registries collect data regarding site and extent of disease, first course of therapy, and socio-demographic characteristics, with active follow-up for date and cause of death. Medicare is the primary health insurer for 97% of Americans 65 years and older, covering inpatient hospital care (Part A), and outpatient care and physician services (Part B). The SEER-Medicare files were used in accordance with a data-use agreement from NCI.
Cohort
We identified prostate cancer patients age 66 years or older who received LRP or ORP in 2003-2005 for a clinically staged T1 or T2 tumor. Men diagnosed only at death, with a prior malignancy, who were enrolled in a managed care plan, or who lacked Part A or B of Medicare were excluded. Surgical procedures were identified by Healthcare Common Procedural Coding System (HCPCS) codes (55866 - LRP; 55840, 55842, 55845 - ORP) in physician claims.
Outcomes
General medical or surgical complications were potentially life-threatening events within 90 days following surgery, including cardiac, vascular, respiratory, wound, and miscellaneous medical/surgical. Genitourinary or bowel complications included bladder neck or urethral obstruction, ureteral complications, incontinence requiring a diagnostic or corrective procedure, fistula or bowel injury, and other genitourinary complications within 365 days following surgery. Receipt of subsequent cancer therapy was defined by a claim for androgen deprivation therapy, radiation therapy or both within 365 days following surgery. Ninety-day mortality was determined from the date of death. Length of hospital stay was identified in the inpatient claim for radical prostatectomy. (Appendix with outcome codes available upon request.)
Covariates
Demographic covariates included patient age, race, geographic location, and marital status. Median income in the census tract of residence was used as a marker of socioeconomic status. Clinical covariates included clinical tumor stage, pathologic stage, number of lymph nodes examined, lymph node involvement, biopsy Gleason score, and diagnosis year. Because SEER did not record exact PSA values until 2004, we used preoperative PSA categorized as elevated, borderline, normal, or unknown. Comorbidity was estimated using the Charlson comorbidity score, based on inpatient claims in the 365 days prior to prostate cancer diagnosis.13-14
For each patient, we estimated procedure-specific annual surgeon volume as the number of procedures of the same type (ORP or LRP) performed by the patient's surgeon in the 365 days prior to and including that patient's procedure. For patients treated in 2003, annual surgeon volume was based on the surgeon's procedures during that year.
Statistical Analysis
The impact of procedure type on each outcome was estimated using multivariable regression, controlling for patient and tumor characteristics and surgeon volume. Length of hospital stay was modeled using a log transformation due to its skewed distribution. Generalized estimating equations (GEE) were used to adjust for correlation of outcomes within surgeon. To address potential overfitting, we also built more parsimonious multivariable models, but results were similar and therefore not presented. In LRP patients only, we explored the impact of surgeon volume on outcomes that were associated with surgeon volume in the full cohort. All statistical analyses were performed in SAS version 9.2 (SAS Institute, Cary, NC).
Results
Characteristics of the Cohort
We identified 5,923 men with clinical stage T1 or T2 prostate cancer in the SEER-Medicare dataset who received either LRP or ORP in 2003-2005. Overall, 4,858 men (82%) received ORP and 1,065 men (18%) received LRP (Table 1). In both groups almost 60% of patients were 66 to 69 years of age, 85% were white and about 80% were married. Men who received LRP were more likely to live in a metropolitan area, the West or Northeast and census tracts in the highest quartile of median income. LRP, as a proportion of all procedures, nearly doubled in each successive year.
Table 1.
Characteristics of cohort by procedure type
| Characteristic | All No. Pts (%) | ORP No. (Col %) | LRP No. (Col %) | pa |
|---|---|---|---|---|
| N | 5,923 | 4,858 | 1,065 | |
| Age Category | ||||
| 66-69 | 3,461 (58) | 2,848 (59) | 613 (58) | 0.94 |
| 70-74 | 2,040 (34) | 1,666 (34) | 374 (35) | |
| 75-59 | 390 (7) | 318 (7) | (<8) | |
| 80+ | 32 (<1) | 26 (<1) | (<1) | |
| Race | ||||
| White | 5,038 (85) | 4,132 (85) | 906 (85) | <0.01 |
| Black | 430 (7) | 371 (8) | 59 (6) | |
| Other | 453 (8) | 354 (7) | 99 (9) | |
| Married | ||||
| Yes | 4,779 (81) | 3,929 (81) | 850 (80) | 0.19 |
| No | 860 (15) | 723 (15) | 137 (13) | |
| Unknown | 284 (5) | 206 (4) | 78 (7) | |
| Census tract median income | ||||
| Q1 | 1,475 (25) | 1,318 (27) | 157 (15) | <0.0001 |
| Q2 | 1,477 (25) | 1,241 (26) | 236 (22) | |
| Q3 | 1,478 (25) | 1,203 (25) | 275 (26) | |
| Q4 | 1,476 (25) | 1,083 (22) | 393 (37) | |
| Urban residence | ||||
| Metro | 5,136 (87) | 4,143 (85) | 993 (93) | <0.0001 |
| Non-Metro | 786 (13) | 714 (15) | 72 (7) | |
| Geographic area | ||||
| Northeast | 640 (11) | 504 (10) | 136 (13) | <0.0001 |
| South | 810 (14) | 719 (15) | 91 (9) | |
| Midwest | 685 (12) | 570 (12) | 115 (11) | |
| West | 3,788 (64) | 3,065 (63) | 723 (68) | |
| Year of surgery | ||||
| 2003 | 1,979 (33) | 1,830 (38) | 149 (14) | <0.0001 |
| 2004 | 2,054 (35) | 1,688 (35) | 366 (34) | |
| 2005 | 1,890 (32) | 1,340 (28) | 550 (52) | |
| Charlson comorbidity score | ||||
| 0 | 4,689 (79) | 3,826 (79) | 863 (81) | 0.22 |
| 1 | 958 (16) | 798 (16) | 160 (15) | |
| 2+ | 276 (5) | 234 (5) | 42 (4) | |
| Clinical stage | ||||
| T1 | 2,692 (45) | 2,112 (43) | 580 (54) | <0.0001 |
| T2 | 3,231 (55) | 2,746 (57) | 485 (46) | |
| Pathologic stage | ||||
| T0/Tis | 13 (<1) | (<1) | (<1) | 0.01 |
| T2 | 4,266 (72) | 3,496 (72) | 770 (72) | |
| T3 | 1,142 (19) | 977 (20) | 165 (15) | |
| T4 | 72 (1) | (<2) | (<2) | |
| Unknown | 430 (7) | 313 (6) | 117 (11) | |
| PSA | ||||
| Elevated | 4,586 (77) | 3,760 (77) | 826 (78) | 0.38 |
| Borderline | 381 (6) | 312 (6) | 69 (6) | |
| Normal | 409 (7) | 324 (7) | 85 (8) | |
| Unknown | 547 (9) | 462 (10) | 85 (8) | |
| Gleason Score | ||||
| 2-4 | 62 (1) | 52 (1) | 10 (1) | 0.81 |
| 5-6 | 3,018 (51) | 2,483 (51) | 535 (50) | |
| 7+ | 2,805 (47) | 2,293 (47) | 512 (48) | |
| Lymph nodes examined | ||||
| 0 | 1,580 (27) | 969 (20) | 611 (57) | <0.0001 |
| >0 | 4,328 (73) | 3,878 (80) | 450 (42) | |
| Lymph node involvement | ||||
| Noc | 5,735 (97) | 4,708 (97) | 1,027 (96) | <0.01 |
| Yes | 106 (2) | 99 (2) | (<1) | |
| Unknown | 82 (1) | 51 (1) | (<4) | |
| PSA laboratory value (n=3,073) | ||||
| < 4 | 362 (12) | 263 (11) | 99 (14) | <0.01 |
| 4 - 10 | 2,121 (69) | 1,602 (68) | 519 (71) | |
| >10 - ≤ 20 | 429 (14) | 342 (15) | 87 (12) | |
| > 20 | 161 (5) | 137 (6) | 24 (3) | |
| Gleason score (n=3,480) | ||||
| 2 - 5 | (<3) | (<3) | (<1) | <0.01 |
| 6 | 1,281 (37) | 960 (36) | 321 (40) | |
| 7 | 1,681 (48) | 1,279 (48) | 402 (50) | |
| 8 | 260 (7) | 208 (8) | 52 (6) | |
| 9 | 163 (5) | 138 (5) | 25 (3) | |
| 10 | (<1) | (<1) | (<1) | |
Abbreviations: ORP, open radical prostatectomy; LPR, laparoscopic radical prostatectomy (with or without robotic assistance); Q1, lowest quartile; Q4, highest quartile; PSA, prostate-specific antigen.
Unknown values and other cell counts where N<11 not shown as per SEER-Medicare Data Use Agreement.
Chi-square test; Unknowns excluded.
PSA laboratory values and exact Gleason score in SEER for cases diagnosed during 2004-2005.
Includes men who did not undergo a pelvic lymph node dissection.
LRP patients generally had a lower clinical T-stage than ORP patients and, of those with known pathology, a lower pathologic T-stage. The two groups had similar distributions of categorical PSA and Gleason score. In men diagnosed in 2004-2005, ORP patients had a higher preoperative PSA compared with LRP patients and a higher Gleason score. LRP patients were considerably less likely to have any regional lymph nodes examined (57% vs. 80%).
Outcomes
Length of Hospital Stay
Median length of stay was 2.0 days in the LRP group and 3.0 days in the ORP group (Table 2). Controlling for patient and tumor characteristics, length of stay for LRP patients was 35% shorter, on average (Table 3). Length of stay was greater in men who were older, non-white, unmarried, resided in census tracts in the lowest median income quartile, had greater comorbidity, had surgery earlier in the study period, or were operated on by lower-volume surgeons.
Table 2.
Frequency of postoperative outcomes by procedure type
| Outcome | ORP N=4,858 | LRP N=1,065 |
|---|---|---|
| Length of hospital stay, days | ||
| N | 4,697 | 1,006 |
| Median (Interquartile range) | 3.0 (2.0-4.0) | 2.0 (1.0-2.0) |
| Mortality from any cause, within 90 days (%) | <0.5 | <0.5 |
| General medical or surgical complications, within 90 days (%) | ||
| Any | 1172 (24.1) | 228 (21.4) |
| Cardiac | 180 (3.7) | 32 (3.0) |
| Respiratory | 423 (8.7) | 71 (6.7) |
| Vascular | 198 (4.1) | 35 (3.3) |
| Wound | 133 (2.7) | 26 (2.4) |
| Miscellaneous medical/surgical | 635 (13.1) | 127 (11.9) |
| Genitourinary or Bowel complications, within 365 days (%) | ||
| Any | 1722 (35.4) | 429 (40.3) |
| Bladder neck/urethral obstruction | 1422 (29.3) | 306 (28.7) |
| Ureteral stricture | 164 (3.4) | 36 (3.4) |
| Incontinence requiring procedure | 245 (5.0) | 66 (6.2) |
| Fistula/bowel injury | 115 (2.4) | 41 (3.8) |
| Other genitourinary | 205 (4.2) | 107 (10.0) |
| Subsequent cancer therapy, within 365 days (%) | ||
| Any | 563 (11.6) | 98 (9.2) |
| Radiation only | 394 (8.1) | 68 (6.4) |
| Androgen deprivation only | 330 (6.8) | 58 (5.4) |
| Radiation and androgen deprivation | 161 (3.3) | 28 (2.6) |
Abbreviations: ORP, open radical prostatectomy; LPR, laparoscopic radical prostatectomy (with or without robotic assistance).
Table 3.
Impact of procedure type, patient and tumor characteristics on general perioperative complications, length of hospital stay, and subsequent cancer therapy
| General Medical or Surgical Complicationa | Length of Hospital Stayb | Subsequent Cancer Therapyc | ||||
|---|---|---|---|---|---|---|
| OR (95% CI) | p | Estimated (95% CI) | p | OR (95% CI) | p | |
| Procedure type | ||||||
| ORP | REF | 0.49 | REF | <0.0001 | REF | 0.14 |
| LRP | 0.93 (0.77, 1.14) | 0.65 (0.61, 0.70) | 0.80 (0.60, 1.08) | |||
| Surgeon volumee | 0.991 (0.982, 0.999) | <0.05 | 0.992 (0.990, 0.994) | <0.0001 | 1.01 (0.99, 1.02) | 0.43 |
| Age | ||||||
| 66-69 | REF | <0.0001 | REF | <0.0001 | REF | 0.25 |
| 70-74 | 1.20 (1.05, 1.38) | 1.04 (1.01, 1.06) | 1.14 (0.95, 1.37) | |||
| 75+ | 1.95 (1.55, 2.44) | 1.10 (1.05, 1.15) | 1.21 (0.89, 1.65) | |||
| Race | ||||||
| White | REF | 0.19 | REF | 0.001 | REF | 0.20 |
| Black | 1.19 (0.93, 1.52) | 1.06 (1.01, 1.11) | 1.09 (0.76, 1.57) | |||
| Other | 1.15 (0.93, 1.42) | 1.06 (1.02, 1.11) | 1.33 (0.97, 1.83) | |||
| Median income | ||||||
| Q1 | REF | 0.44 | REF | <0.001 | REF | 0.58 |
| Q2 | 0.90 (0.75, 1.08) | 0.98 (0.94, 1.01) | 0.92 (0.71, 1.20) | |||
| Q3 | 0.85 (0.70, 1.04) | 0.95 (0.92, 0.98) | 1.03 (0.79, 1.34) | |||
| Q4 | 0.90 (0.74, 1.09) | 0.94 (0.91, 0.97) | 1.10 (0.85, 1.43) | |||
| Urban residence | ||||||
| Metro | REF | 0.26 | REF | 0.32 | REF | 0.41 |
| Non-Metro | 1.12 (0.92, 1.37) | 1.02 (0.98, 1.06) | 0.88 (0.65, 1.20) | |||
| Region | ||||||
| Northeast | REF | 0.07 | REF | 0.73 | REF | 0.19 |
| South | 1.02 (0.77, 1.35) | 1.00 (0.93, 1.07) | 1.21 (0.77, 1.91) | |||
| Midwest | 0.75 (0.57, 0.99) | 1.03 (0.95, 1.11) | 1.50 (0.96, 2.33) | |||
| West | 0.93 (0.74, 1.17) | 1.02 (0.96, 1.09) | 1.12 (0.76, 1.65) | |||
| Married | ||||||
| Yes | REF | 0.39 | REF | <0.0001 | REF | <0.05 |
| No | 0.92 (0.77, 1.09) | 1.06 (1.03, 1.10) | 0.88 (0.68, 1.13) | |||
| Unknown | 1.13 (0.87, 1.48) | 0.95 (0.91, 1.00) | 0.40 (0.21, 0.76) | |||
| Clinical stage | ||||||
| T1 | REF | 0.26 | REF | 0.88 | REF | <0.001 |
| T2 | 1.07 (0.95, 1.21) | 1.00 (0.98, 1.02) | 1.40 (1.18, 1.67) | |||
| PSA | ||||||
| Normal | REF | 0.63 | REF | 0.64 | REF | <0.05 |
| Borderline | 0.81 (0.57, 1.15) | 1.00 (0.94, 1.07) | 2.03 (1.17, 3.51) | |||
| Elevated | 0.93 (0.72, 1.21) | 0.99 (0.95, 1.04) | 2.02 (1.28, 3.19) | |||
| Unknown | 0.98 (0.71, 1.35) | 1.02 (0.97, 1.08) | 1.58 (0.91, 2.72) | |||
| Lymph nodes involvedf | ||||||
| No | REF | <0.0001 | ||||
| Yes | 11.73 (7.77, 17.69) | |||||
| Lymph nodes examined | ||||||
| None | REF | 0.31 | REF | 0.15 | REF | |
| Any | 1.08 (0.93, 1.26) | 1.02 (0.99, 1.05) | 1.01 (0.79, 1.28) | 0.97 | ||
| Gleason | ||||||
| 2-4 | 1.65 (0.94, 2.91) | 0.18 | 1.05 (0.95, 1.15) | 0.38 | 1.23 (0.36, 4.20) | <0.0001 |
| 5-6 | REF | REF | REF | |||
| 7+ | 0.98 (0.86, 1.11) | 0.99 (0.97, 1.01) | 3.13 (2.56, 3.83) | |||
| Comorbidity | ||||||
| 0 | REF | <0.0001 | REF | <.0001 | REF | <0.05 |
| 1 | 1.34 (1.13, 1.58) | 1.03 (1.00, 1.06) | 1.04 (0.82, 1.31) | |||
| 2+ | 3.35 (2.62, 4.27) | 1.13 (1.07, 1.19) | 0.52 (0.32, 0.86) | |||
| Year of surgery | ||||||
| 2003 | REF | 0.29 | REF | <0.01 | REF | 0.10 |
| 2004 | 1.10 (0.95, 1.27) | 0.98 (0.96, 1.00) | 0.79 (0.63, 0.98) | |||
| 2005 | 1.13 (0.96, 1.34) | 0.95 (0.92, 0.98) | 0.85 (0.68, 1.06) | |||
Abbreviations: ORP, open radical prostatectomy; LPR, laparoscopic radical prostatectomy (with or without robotic assistance); OR, odds ratio; CI, confidence interval; REF, reference; Q1, lowest quartile; Q4, highest quartile; PSA, prostate-specific antigen.
Within 90 days following surgery
For initial surgical hospitalization
Radiation or androgen deprivation therapy or both administered within 365 days following surgery
Estimate denotes change in the ratio of expected mean length of stay for covariate vs. reference value
OR interpretable as the decrease in odds of the outcome for every additional procedure performed by a patient's surgeon in the 365 days prior to his own surgery; length of stay estimate interpretable as the percentage change in average length of hospital stay for every additional procedure performed by a patient's surgeon in the year prior to their own surgery
Only considered clinically relevant for subsequent cancer therapy outcome, therefore not included in other models
Perioperative Mortality, General Medical or Surgical Complications
In both groups, the 90-day mortality rate was < 0.5% (Table 2). A general medical or surgical complication within 90 days postoperatively occurred in 21% of LRP patients and 24% of ORP patients. Adjusted for covariates, the odds of a general medical or surgical complication did not differ significantly between groups (Table 3). These complications were more likely in men who were older, had greater comorbidity or whose surgeons had lower procedure-specific volume.
Genitourinary or Bowel Complications
In the first year following surgery, almost 40% of LRP patients had a genitourinary or bowel complication, compared with 35% of ORP patients (Table 2). Controlling for patient and tumor characteristics, procedure type was not significantly associated with the odds of a genitourinary or bowel complication (Table 4). Only surgeon volume, marital status, and clinical stage were significant predictors of this outcome.
Table 4.
Impact of procedure type, patient and tumor characteristics on postoperative genitourinary or bowel complications within 1 year following surgery
| Any Genitourinary or Bowel Complication | Bladder Neck or Urethral Obstruction | |||
|---|---|---|---|---|
| OR (95% CI) | p | OR (95% CI) | P | |
| Procedure type | ||||
| ORP | REF | 0.74 | REF | <0.05 |
| LRP | 0.96 (0.76, 1.22) | 0.74 (0.58, 0.94) | ||
| Surgeon volumea | 0.98 (0.97, 0.99) | <0.001 | 0.98 (0.97, 0.99) | <0.01 |
| Age | ||||
| 66-69 | REF | 0.80 | REF | 0.97 |
| 70-74 | 1.03 (0.91, 1.16) | 1.01 (0.90, 1.15) | ||
| 75+ | 1.07 (0.86, 1.32) | 1.01 (0.80, 1.28) | ||
| Race | ||||
| White | REF | 0.90 | REF | 0.50 |
| Black | 0.95 (0.77, 1.18) | 1.02 (0.81, 1.28) | ||
| Other | 0.99 (0.78, 1.24) | 0.87 (0.69, 1.10) | ||
| Median income | ||||
| Q1 | REF | 0.23 | REF | 0.37 |
| Q2 | 0.97 (0.83, 1.13) | 1.05 (0.89, 1.23) | ||
| Q3 | 0.85 (0.71, 1.00) | 0.91 (0.76, 1.10) | ||
| Q4 | 0.89 (0.74, 1.08) | 0.92 (0.76, 1.12) | ||
| Urban residence | ||||
| Metro | REF | 0.54 | REF | 0.55 |
| Non-Metro | 1.07 (0.87, 1.30) | 1.06 (0.87, 1.31) | ||
| Region | ||||
| Northeast | REF | 0.07 | REF | <0.01 |
| South | 0.68 (0.51, 0.91) | 0.61 (0.44, 0.82) | ||
| Midwest | 0.80 (0.59, 1.09) | 0.81 (0.59, 1.12) | ||
| West | 0.83 (0.66, 1.04) | 0.87 (0.68, 1.12) | ||
| Married | ||||
| Yes | REF | <0.01 | REF | <0.01 |
| No | 1.29 (1.11, 1.49) | 1.28 (1.09, 1.50) | ||
| Unknown | 0.91 (0.69, 1.20) | 0.89 (0.66, 1.19) | ||
| Clinical stage | ||||
| T1 | REF | <0.01 | REF | <0.05 |
| T2 | 1.18 (1.06, 1.33) | 1.14 (1.01, 1.28) | ||
| PSA | ||||
| Normal | REF | 0.21 | REF | 0.29 |
| Borderline | 0.81 (0.59, 1.11) | 0.77 (0.57, 1.04) | ||
| Elevated | 0.97 (0.77, 1.23) | 0.95 (0.78, 1.17) | ||
| Unknown | 1.10 (0.83, 1.45) | 1.00 (0.75, 1.32) | ||
| Lymph node examination | ||||
| None | REF | 0.09 | REF | <0.05 |
| Any | 0.89 (0.77, 1.02) | 0.86 (0.74, 1.00) | ||
| Gleason | ||||
| 2-4 | 1.63 (0.98, 2.69) | 0.17 | 1.45 (0.88, 2.39) | 0.30 |
| 5-6 | REF | REF | ||
| 7+ | 1.02 (0.91, 1.14) | 0.98 (0.87, 1.10) | ||
| Comorbidity | ||||
| 0 | REF | 0.20 | REF | 0.26 |
| 1 | 1.08 (0.94, 1.25) | 1.12 (0.96, 1.31) | ||
| 2+ | 1.21 (0.95, 1.55) | 1.14 (0.87, 1.49) | ||
| Year of surgery | ||||
| 2003 | REF | 0.68 | REF | 0.82 |
| 2004 | 1.05 (0.91, 1.21) | 1.02 (0.88, 1.19) | ||
| 2005 | 1.06 (0.92, 1.23) | 1.05 (0.90, 1.22) | ||
Abbreviations: ORP, open radical prostatectomy; LPR, laparoscopic radical prostatectomy (with or without robotic assistance); OR, odds ratio; CI, confidence interval; REF, reference; Q1, lowest quartile; Q4, highest quartile; PSA, prostate-specific antigen.
OR interpretable as the decrease in odds of the outcome for every additional procedure performed by a patient's surgeon in the 365 days prior to his own surgery
In both groups, about 29% of men had a bladder neck or urethral obstruction (Table 2). Controlling for patient and tumor characteristics, LRP was associated with lower odds of this complication (OR 0.74, 95% CI 0.58–0.94) (Table 4), which was more common in men who were unmarried, lived in the Northeast, had a clinically staged T2 tumor, or did not have a pelvic lymphadenectomy. Surgeon volume was inversely associated with bladder neck or urethral obstruction.
Subsequent Cancer Therapy
About 9% of LRP patients and 12% of ORP patients received radiation therapy, androgen deprivation therapy or both in the year following surgery (Table 2). Adjusted for patient and tumor characteristics, procedure type was not significantly associated with receipt of subsequent cancer therapy (Table 3). When we limited the endpoint to subsequent cancer therapy in postoperative days 120 through 365, results were similar.
Surgeon Volume in LRP
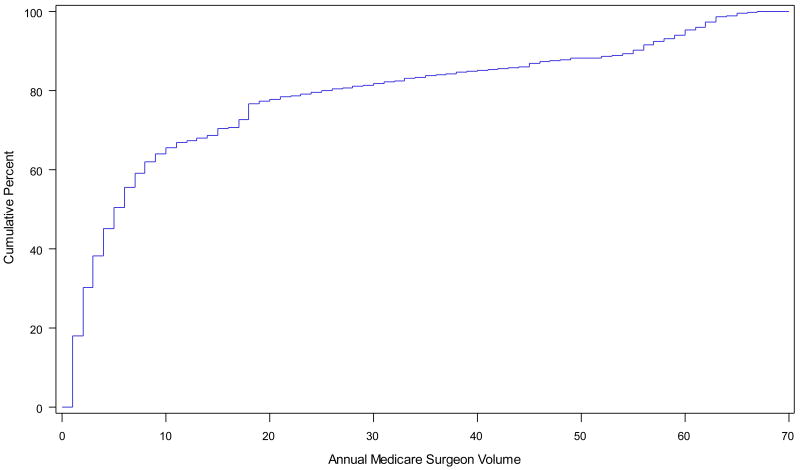
Among LRP patients, annual procedure-specific surgeon volume varied from 1 to 68. More than half of these men had a LRP performed by a surgeon whose annual volume was less than 5 (Figure 1). Fewer than 20% of LRP patients had a surgeon whose annual volume was 30 or more.
Figure 1.
Cumulative Distribution of Annual Procedure-Specific Surgeon Volume in Laparoscopic Radical Prostatectomy (With or Without Robotic Assistance) Patients
For each patient, we estimated procedure-specific annual Medicare surgeon volume as the number of procedures of the same type (ORP or LRP) performed by the patient's surgeon in the 365 days prior to that patient's procedure, including the patient's surgery.
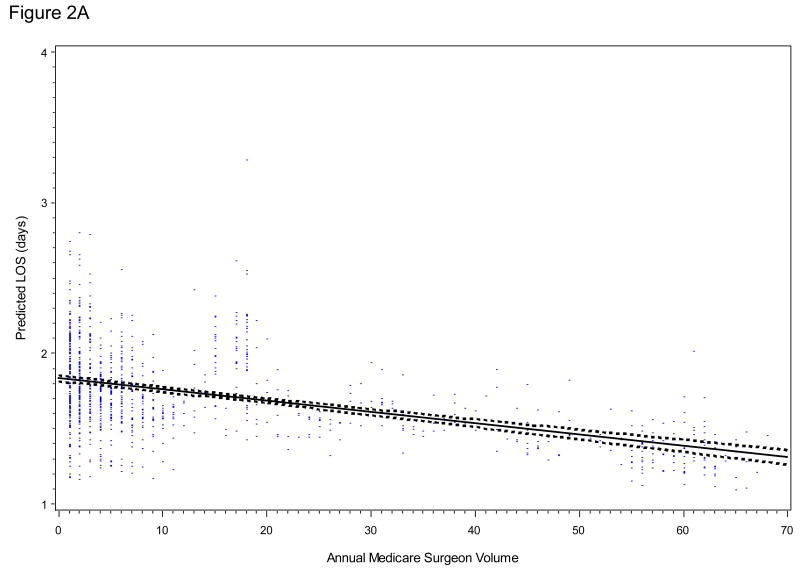
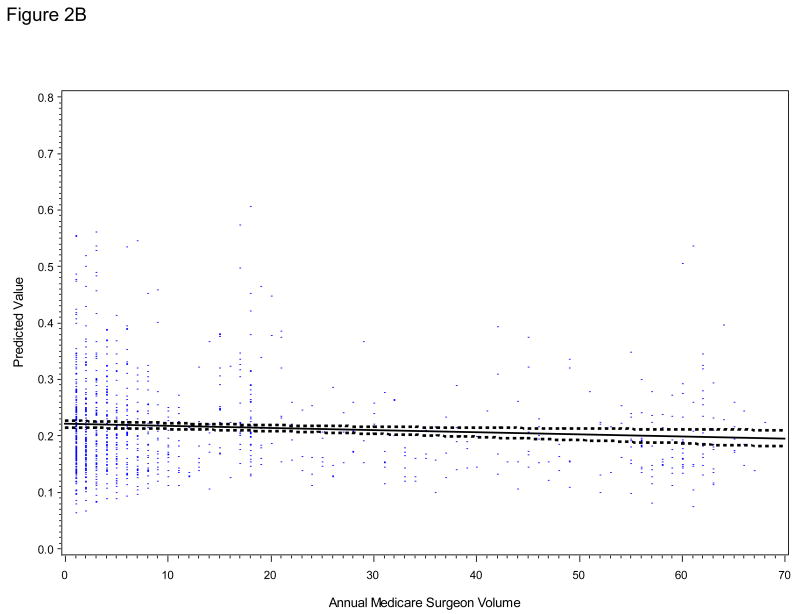
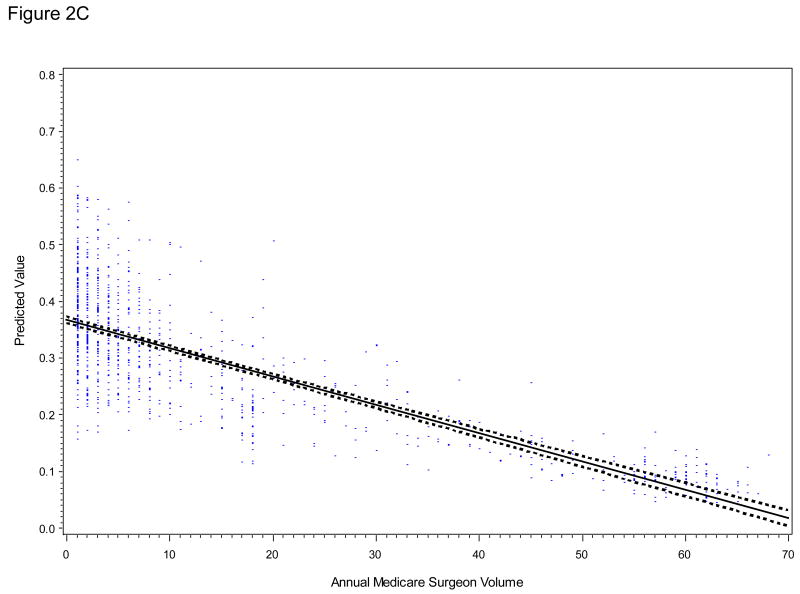
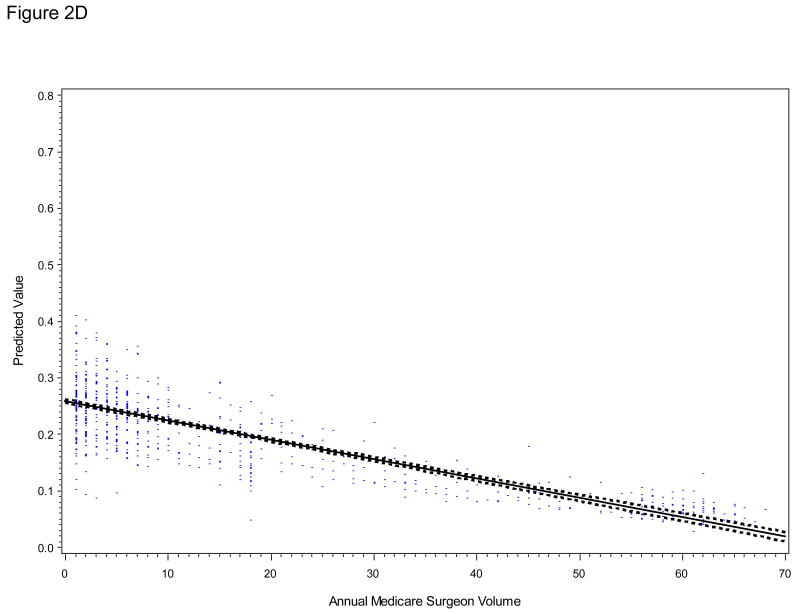
Controlling for patient and tumor characteristics, surgeon volume was associated with a shorter length of stay (p<0.001, Figure 2A), but not with general medical or surgical complications (p=0.33, Figure 2B) in the LRP group. The inverse relationship between surgeon volume and both genitourinary or bowel complications (p<0.01, Figure 2C) and bladder neck or urethral obstruction (p<0.05, Figure 2D) persisted in the LRP group. The predicted probability of a bladder neck or urethral obstruction after LRP was 31% for the average patient whose surgeon performed 5 LRPs in the preceding year and decreased to 16% for the average patient whose surgeon performed 30 LRPs. Results were similar when we relaxed the assumption of linearity between surgeon volume and each outcome.
Figure 2 (A - D).
Relationship of Annual Procedure-Specific Surgeon Volume in SEER-Medicare with Postoperative Outcomes
For LRP (with or without robotic assistance) patients, plots are derived from multivariable linear regression models adjusted for patient and tumor characteristics and corrected for within-surgeon correlation. A, predicted length of stay (LOS, days); B, predicted risk of general medical or surgical complication within 90 days of surgery; C, predicted risk of any genitourinary or bowel complication within 365 days of surgery; D, predicted risk of bladder neck or urethral obstruction within 365 days of surgery. Blue dots represent individual predicted outcomes. The linear relationship between each outcome (solid line) and 95% confidence intervals around the mean predicted values (dashed lines) are shown.
Discussion
Since its introduction, the use of robotic radical prostatectomy has increased exponentially, despite a lack of high-quality evidence demonstrating superiority to ORP.5-6 In the large population-based dataset analyzed here, we found no difference between procedures in 90-day mortality, 90-day general medical or surgical complications, or use of additional cancer therapy in the year following surgery. Although we observed some differences in unadjusted rates of complications and subsequent cancer therapy, these differences were not statistically significant when we controlled for patient and tumor characteristics and surgeon volume. Differences in length of stay and bladder neck or urethral obstruction persisted in multivariable analysis, both favoring LRP.
Our findings of similar 90-day general complication rates and rates of any genitourinary or bowel complication at one year are consistent with several single-institution reports, and suggest that procedure type has little, if any, impact on morbidity.9, 15-16 Patient age, comorbidity, and surgeon volume were the only significant predictors of general medical or surgical complications, while surgeon volume, marital status, and clinical stage were the only significant predictors of any genitourinary or bowel complication.
Some of our results conflict with other recent population-based studies. In a nationwide sample of Medicare beneficiaries, Hu et al. found a lower risk of postoperative complications, but a greater likelihood of additional cancer treatment in LRP patients.11 In the absence of information about disease characteristics, the investigators were able to control only for age, race, comorbidity, and geographic region. We found that controlling for both patient and tumor characteristics, including clinical or pathologic stage, lymph node examination and involvement, preoperative PSA, and preoperative Gleason score, had a substantial impact on our conclusions. This likely reflects confounding, particularly by higher-risk tumor characteristics which were more common in the ORP group.
Our findings with respect to bladder neck or urethral obstruction are consistent with those reported by Hu et al. in a separate claims-based analysis.10 In that cohort, which included men of all ages, LRP patients had lower odds of bladder neck or urethral obstruction. However, in their analysis of Medicare beneficiaries, Hu et al. reached the opposite conclusion, finding a greater risk of bladder neck or urethral obstruction in men who received LRP.11 Although bladder neck and urethral obstructions requiring operative intervention were uncommon even in our ORP group, these complications can lead to chronic urinary problems, impairing function and quality of life and requiring multiple corrective procedures.17
Comparisons of LRP and ORP have consistently found reduced blood loss and shorter length of hospital stay with LRP.9 We were not able to assess the former, but our results corroborate the latter. While reduced length of stay is commonly stated as an advantage of LRP, it is not clear that it yields the economic advantages its advocates promote. Cost comparisons of the two procedures suggest that savings in hospital room and board costs with LRP do not necessarily offset the increased operative costs associated with use of the robot, particularly in low-volume settings.18-19 Standardization of care pathways may reduce differences between the two procedures in length of stay.20-21
The rapid uptake of LRP has important implications for providers and patients.22 Hospital marketing of robotic prostatectomy is widespread, and is not regulated by the Food and Drug Administration rules that govern direct-to-consumer advertising by device manufacturers. In a simple Internet search of hospital websites, we found that many promoted the robotic procedure with claims that overstated its potential benefits or implied a level of certainty that is not currently supported by existing evidence. It is therefore not surprising that in a survey of 400 men who had ORP or robot-assisted LRP, those who had the robotic procedure were more than 3 times as likely to express regret with their treatment choice, while men who had ORP were more than 4 times as likely to express satisfaction.23
Increased use of the robotic approach also means that the procedure is being adopted by less experienced surgeons. We found that annual surgeon volume was associated with a shorter hospital stay, lower risk of general medical or surgical complications, genitourinary or bowel complications, and specifically bladder neck or urethral obstruction. In LRP patients only, surgeon volume was predictive of most of these endpoints, but not of general medical or surgical complications. While our measure of annual surgeon volume included only procedures performed on men age 66 or older in the SEER-Medicare dataset, it should be highly correlated with the volume of procedures performed on men of all ages.24 Efforts to regionalize robotic prostatectomy may yield both clinical and economic benefits.
Based on the large, population-based linked SEER-Medicare dataset, our findings are generalizable to surgical prostate cancer patients age 66 and older, and we were able to control for important patient and tumor. However, there may have been residual confounding by unmeasured factors. Misclassification of outcomes is another possible limitation; our methods favored inclusion of all potentially relevant billing codes to identify complications. Similarly, because all LRPs shared a single HCPCS code (55866), it was impossible to distinguish procedures performed with and without robotic assistance. We estimate that most LRPs were performed with robotic assistance, based on information provided by the sole manufacturer of the robotic system. Finally, we could not distinguish with certainty between adjuvant and salvage therapies. Therefore, receipt of subsequent cancer therapy is not an ideal measure of short-term cancer control.
While we evaluated important outcomes of prostate cancer surgery, there are numerous other endpoints we could not analyze. Long-term oncologic control, urinary and erectile function, quality of life and costs are pertinent to both patients and providers. We hope that recent enthusiasm and funding for comparative effectiveness studies prompts further comparisons of the different approaches to radical prostatectomy, and address these important endpoints in prospective clinical studies.25
Conclusion
Our results suggest that ORP and LRP have similar rates of postoperative mortality and morbidity. Controlling for important patient and tumor characteristics, the only differences favoring LRP were a shorter length of stay and a lower risk of bladder neck or urethral obstruction. All men considering radical prostatectomy should be clearly informed about the differences between the two techniques as well as the similarities in their expected outcomes and make treatment decisions in collaboration with an experienced surgeon.
Acknowledgments
The authors gratefully acknowledge the Applied Research Program, NCI; the Office of Information Services and Office of Strategic Planning, Centers for Medicare & Medicaid Services (CMS); Information Management Services, Inc; and the SEER Program tumor registries for creation of the SEER-Medicare dataset. Substantive editing was performed by Janet Novak, PhD, of Helix Editing. This work was paid for by Memorial Sloan-Kettering Cancer Center.
Funding: This work was supported in part by funds from the National Institutes of Health [T32-CA82088 to P.S. and W.L.]; the National Cancer Institute [P50-CA92629 SPORE to P.S., CA118189-01A2 to E.E]; Sidney Kimmel Center for Prostate and Urologic Cancers; and David H. Koch provided through the Prostate Cancer Foundation.
Abbreviations
- AJCC
American Joint Committee on Cancer Staging Manual, 6th Edition
- CMS
Centers for Medicare and Medicaid Services
- GEE
Generalized Estimating Equations
- HCPCS
Healthcare Common Procedural Coding System
- ICD-9
International Classification of Diseases codes, 9th Edition
- LRP
Laparoscopic Radical Prostatectomy
- NCI
National Cancer Institute
- PSA
Prostate-Specific Antigen
- ORP
Open Radical Prostatectomy
- SEER
Surveillance Epidemiology and End Results
Contributor Information
William T. Lowrance, Department of Surgery (Urology Service), Sidney Kimmel Center for Prostate and Urologic Cancers, Memorial Sloan-Kettering Cancer Center, New York, NY.
Elena B. Elkin, Department of Epidemiology and Biostatistics (Health Outcomes Research Group), Memorial Sloan-Kettering Cancer Center, New York, NY.
Lindsay M. Jacks, Department of Epidemiology and Biostatistics (Health Outcomes Research Group), Memorial Sloan-Kettering Cancer Center, New York, NY.
David S. Yee, Department of Surgery (Urology Service), Sidney Kimmel Center for Prostate and Urologic Cancers, Memorial Sloan-Kettering Cancer Center, New York, NY.
Thomas L. Jang, Department of Surgery (Urology Service), Sidney Kimmel Center for Prostate and Urologic Cancers, Memorial Sloan-Kettering Cancer Center, New York, NY.
Vincent P. Laudone, Department of Surgery (Urology Service), Sidney Kimmel Center for Prostate and Urologic Cancers, Memorial Sloan-Kettering Cancer Center, New York, NY.
Bertrand D. Guillonneau, Department of Surgery (Urology Service), Sidney Kimmel Center for Prostate and Urologic Cancers, Memorial Sloan-Kettering Cancer Center, New York, NY.
Peter T. Scardino, Department of Surgery (Urology Service), Sidney Kimmel Center for Prostate and Urologic Cancers, Memorial Sloan-Kettering Cancer Center, New York, NY.
James A. Eastham, Department of Surgery (Urology Service), Sidney Kimmel Center for Prostate and Urologic Cancers, Memorial Sloan-Kettering Cancer Center, New York, NY.
References
- 1.Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71. doi: 10.3322/CA.2007.0010. [DOI] [PubMed] [Google Scholar]
- 2.Guillonneau B, Vallancien G. Laparoscopic radical prostatectomy: the Montsouris experience. J Urol. 2000;163:418. doi: 10.1016/s0022-5347(05)67890-1. [DOI] [PubMed] [Google Scholar]
- 3.Schuessler WW, Kavoussi LR, Clayman RV, et al. Laparoscopic radical prostatectomy: initial case report. J Urol. 1992;147:246A. doi: 10.1016/S0090-4295(97)00543-8. [DOI] [PubMed] [Google Scholar]
- 4.Schuessler WW, Schulam PG, Clayman RV, et al. Laparoscopic radical prostatectomy: initial short-term experience. Urology. 1997;50:854. doi: 10.1016/S0090-4295(97)00543-8. [DOI] [PubMed] [Google Scholar]
- 5.Abbou CC, Hoznek A, Salomon L, et al. Remote laparoscopic radical prostatectomy carried out with a robot. Report of a case. Prog Urol. 2000;10:520. [PubMed] [Google Scholar]
- 6.Binder J, Kramer W. Robotically-assisted laparoscopic radical prostatectomy. BJU Int. 2001;87:408. doi: 10.1046/j.1464-410x.2001.00115.x. [DOI] [PubMed] [Google Scholar]
- 7.Menon M, Tewari A, Peabody JO, et al. Vattikuti Institute prostatectomy, a technique of robotic radical prostatectomy for management of localized carcinoma of the prostate: experience of over 1100 cases. Urol Clin North Am. 2004;31:701. doi: 10.1016/j.ucl.2004.06.011. [DOI] [PubMed] [Google Scholar]
- 8.Guazzoni G, Cestari A, Naspro R, et al. Intra- and peri-operative outcomes comparing radical retropubic and laparoscopic radical prostatectomy: results from a prospective, randomised, single-surgeon study. Eur Urol. 2006;50:98. doi: 10.1016/j.eururo.2006.02.051. [DOI] [PubMed] [Google Scholar]
- 9.Ficarra V, Novara G, Artibani W, et al. Retropubic, Laparoscopic, and Robot-Assisted Radical Prostatectomy: A Systematic Review and Cumulative Analysis of Comparative Studies. Eur Urol. 2009 doi: 10.1016/j.eururo.2009.01.036. [DOI] [PubMed] [Google Scholar]
- 10.Hu JC, Hevelone ND, Ferreira MD, et al. Patterns of care for radical prostatectomy in the United States from 2003 to 2005. J Urol. 2008;180:1969. doi: 10.1016/j.juro.2008.07.054. [DOI] [PubMed] [Google Scholar]
- 11.Hu JC, Wang Q, Pashos CL, et al. Utilization and outcomes of minimally invasive radical prostatectomy. J Clin Oncol. 2008;26:2278. doi: 10.1200/JCO.2007.13.4528. [DOI] [PubMed] [Google Scholar]
- 12.Warren JL, Klabunde CN, Schrag D, et al. Overview of the SEER-Medicare data: content, research applications, and generalizability to the United States elderly population. Med Care. 2002;40:IV. doi: 10.1097/01.MLR.0000020942.47004.03. [DOI] [PubMed] [Google Scholar]
- 13.Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373. doi: 10.1016/0021-9681(87)90171-8. [DOI] [PubMed] [Google Scholar]
- 14.Klabunde CN, Warren JL, Legler JM. Assessing comorbidity using claims data: an overview. Med Care. 2002;40:IV. doi: 10.1097/00005650-200208001-00004. [DOI] [PubMed] [Google Scholar]
- 15.Guillonneau B, Rozet F, Cathelineau X, et al. Perioperative complications of laparoscopic radical prostatectomy: the Montsouris 3-year experience. J Urol. 2002;167:51. [PubMed] [Google Scholar]
- 16.Smith JA, Jr, Herrell SD. Robotic-assisted laparoscopic prostatectomy: do minimally invasive approaches offer significant advantages? J Clin Oncol. 2005;23:8170. doi: 10.1200/JCO.2005.03.1963. [DOI] [PubMed] [Google Scholar]
- 17.Eastham JA, Kattan MW, Rogers E, et al. Risk factors for urinary incontinence after radical prostatectomy. J Urol. 1996;156:1707. [PubMed] [Google Scholar]
- 18.Lotan Y, Cadeddu JA, Gettman MT. The new economics of radical prostatectomy: cost comparison of open, laparoscopic and robot assisted techniques. J Urol. 2004;172:1431. doi: 10.1097/01.ju.0000139714.09832.47. [DOI] [PubMed] [Google Scholar]
- 19.Scales CD, Jr, Jones PJ, Eisenstein EL, et al. Local cost structures and the economics of robot assisted radical prostatectomy. J Urol. 2005;174:2323. doi: 10.1097/01.ju.0000181830.43340.e7. [DOI] [PubMed] [Google Scholar]
- 20.Nelson B, Kaufman M, Broughton G, et al. Comparison of length of hospital stay between radical retropubic prostatectomy and robotic assisted laparoscopic prostatectomy. J Urol. 2007;177:929. doi: 10.1016/j.juro.2006.10.070. [DOI] [PubMed] [Google Scholar]
- 21.Wood DP, Schulte R, Dunn RL, et al. Short-term health outcome differences between robotic and conventional radical prostatectomy. Urology. 2007;70:945. doi: 10.1016/j.urology.2007.06.1120. [DOI] [PubMed] [Google Scholar]
- 22.Pruthi RS, Wallen EM. Current status of robotic prostatectomy: promises fulfilled. J Urol. 2009;181:2420. doi: 10.1016/j.juro.2009.03.028. [DOI] [PubMed] [Google Scholar]
- 23.Schroeck FR, Krupski TL, Sun L, et al. Satisfaction and regret after open retropubic or robot-assisted laparoscopic radical prostatectomy. Eur Urol. 2008;54:785. doi: 10.1016/j.eururo.2008.06.063. [DOI] [PubMed] [Google Scholar]
- 24.Begg CB, Riedel ER, Bach PB, et al. Variations in morbidity after radical prostatectomy. N Engl J Med. 2002;346:1138. doi: 10.1056/NEJMsa011788. [DOI] [PubMed] [Google Scholar]
- 25.Committee on Comparative Effectiveness Research Prioritization, Institute of Medicince: Initial national priorities for comparative effectiveness research. Washington, DC: Institute of Medicine; 2009. [Google Scholar]