ABSTRACT
Rectoceles are common and involve a herniation of the rectum into the posterior vaginal wall that results in a vaginal bulge. Women with rectoceles generally complain of perineal and vaginal pressure, obstructive defecation, constipation, or the need to splint or digitally reduce the vagina to effectuate a bowel movement. Rectoceles are associated with age and parturition and arise from either a tear or stretching of the rectovaginal fascia, and can be repaired via a vaginal, anal, or perineal approach. Although the rate of successful anatomic repair is high, reports of functional outcome are more variable.
Keywords: Rectocele, posterior colporrhaphy, defecation disorders
The rectovaginal septum is weakened by age and parturition, allowing the rectum to protrude into the vagina during evacuation efforts.1 This outpouching of the rectum into the vagina is termed a rectocele. Although rectoceles are common, associated symptoms vary widely. This manuscript covers rectocele anatomy, symptoms, physical exam, functional evaluations, indications for repair, surgical techniques, and treatment outcomes.
ANATOMY
Rectoceles result from defects in the integrity of the rectovaginal septum and herniation of the rectal wall into the vaginal lumen.2 The normal posterior vagina is lined by squamous epithelium that overlies the lamina propria, a layer of loose connective tissue. A fibromuscular layer of tissue composed of smooth muscle, collagen, and elastin underlies this lamina propria, and is referred to as the rectovaginal fascia. This is an extension of the endopelvic fascia that surrounds the pelvic organs and allows for their support, and contains blood vessels, lymphatics, and nerves supplying the pelvic organs.
Denonvilliers originally described a dense tissue layer in men between the bladder and the rectum and named it the rectovesical septum.3 Many clinicians refer to this layer as Denonvilliers' fascia. The layer of tissue between the vagina and the rectum was felt to be analogous to the rectovesical septum and became known as Denonvilliers' fascia in the female or the rectovaginal septum.3 Uhlenhuth,4 Milley, and Nichols,5 and Richardson3 described the rectovaginal septum, or rectovaginal fascia, as a support mechanism of the pelvic organs and were successful in identifying this layer during surgical and autopsy dissections.
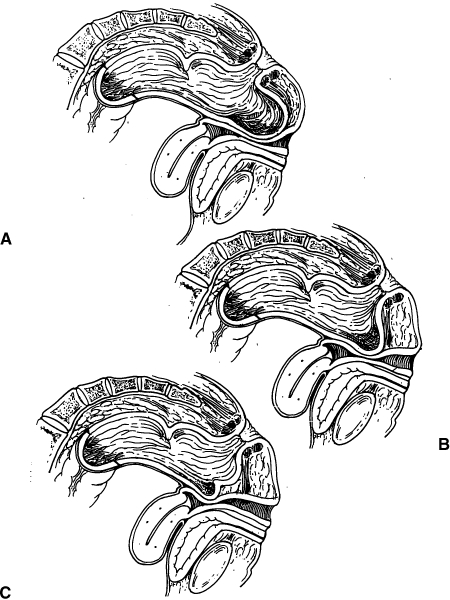
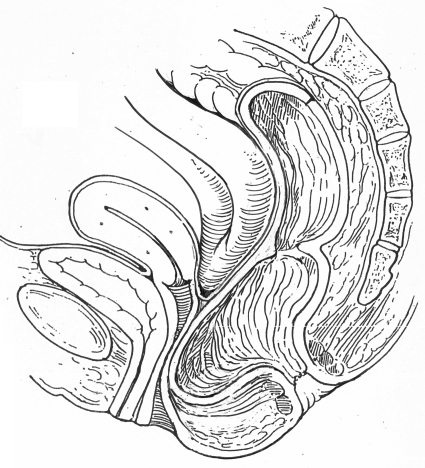
The normal vagina is stabilized and supported on three levels. Superiorly, the vaginal apical endopelvic fascia is attached to the cardinal-uterosacral ligament complex. Laterally, the endopelvic fascia is connected to the arcus tendineus fasciae pelvis, with the lateral posterior vagina attaching to the fascia overlying the levator ani muscles. Inferiorly, the lower, posterior vagina connects to the perineal body.6 The rectovaginal septum is described as “a continuous layer of support extending from the sacrum above to the perineal body below.”5 Many consider the cervix (or vaginal cuff in the hysterectomized woman) to be the “superior central tendon” and the perineal body the “inferior central tendon” between which the endopelvic fascia extends in the rectovaginal septum. A rectocele results from a stretching or actual separation or tear of the rectovaginal fascia, thus leading to a bulging of the posterior vaginal wall noted on examination during a Valsalva maneuver. Rectoceles may be located proximal (high), medial (mid), or distal (low) in the septum (Fig. 1). Defects in the rectovaginal fascia or separation from its attachments often result from childbirth or improperly healed episiotomies, and commonly lead to transverse defects above the usual location of the connection to the perineal body.3,7 In addition, patients may present with lateral, midline, or high transverse fascial defects. Separation of the rectovaginal septum fascia from the vaginal cuff results in the development of an enterocele (Fig. 2).
Figure 1.
Rectocele. (A) High rectocele; (B) mid-rectocele; (C) low rectocele.
Figure 2.
Enterocele.
The levator plate extends from the pubic bone to the sacrum/coccyx and provides support for the change in vaginal axis from vertical to horizontal along the mid vagina. A rectocele typically develops at, or below, the levator plate, along the vertical vagina.
SYMPTOMS
A posterior vaginal wall herniation of the rectum into the vaginal canal results in a visible bulge, which may produce symptoms. Classically, women will report a need to digitally reduce or splint the posterior vaginal bulge or their perineum to either initiate or complete a bowel movement. Obstructive defecation due to accumulation of stool within the rectocele reservoir leads to increasing degrees of perineal pressure. In the absence of digital reduction, women will note incomplete emptying associated with increasing degrees of perineal pressure. This leads to a high degree of frustration and a vicious cycle of increasing pelvic pressure, need for stronger Valsalva efforts, enlargement of the rectocele bulge, and increasing perineal pressure. Rectal digitation is less commonly reported.
Constipation is not clearly understood by many practitioners. Its vague nature, coupled with a poor understanding of the complexity of colorectal function, results in a lack of interest to further evaluate the symptoms of constipation. Unfortunately, this may lead to surgical treatment of the rectocele when initial conservative therapy of the constipation may resolve many of the symptoms and lower recurrence rates.
An enlarging rectocele will widen the levator hiatus, increase vaginal caliber, and enlarge the genital hiatuses.8,9 This may lead to sexual difficulties including symptoms of vaginal looseness and decreased sensation during intercourse. Hemorrhoids can be associated with a rectocele. They typically occur secondary to increased Valsalva efforts by the patient to have a bowel movement.
Women with a large rectocele may present with a palpable vaginal bulge. Once a rectocele has extended beyond the hymeneal ring, it becomes exteriorized. A large, exteriorized rectocele places the patient at risk for vaginal mucosal erosion and ulceration, and accelerates progression of the rectocele due to further weakening of the posterior vaginal wall.2 There is frequently an association with other anatomic support defects such as an enterocele or a cystocele.
PHYSICAL EXAMINATION
The typical physical finding in a woman with a symptomatic rectocele is a lower posterior vaginal wall bulge. In an isolated rectocele, the bulge extends from the edge of the levator plate to the perineal body. With an enlarging rectocele, the perineal body may become distended and loose its bulk, leading to a perineocele. As such, physical examination should include not only a vaginal exam but a rectal exam as well. A perineocele may not be evident on vaginal examination and may be only identified upon digital rectal examination and confirmation of absent fibromuscular tissue in the perineal body. Although most gynecologists examine patients in the lithotomy position, colorectal surgeons favor the prone-jackknife or left lateral position.
Various classification schemes have been developed to describe rectocele severity. In the traditional Baden–Walker system using the midvaginal plane as a landmark, a rectocele can be graded from 0 to 4. In the pelvic organ prolapse quantification (POP-Q) system, two points along the posterior vaginal wall are identified (Ap: 3 cm proximal to the hymen, and Bp: the most dependent part), and their distances from the hymeneal ring are measured in centimeters with maximum Valsalva effort. In the POP-Q system, discrete points and their displacement are measured, rather than the underlying organ (i.e., rectum).
Additional factors that should be evaluated during the physical examination include vaginal mucosal thickness and estrogenization, associated pelvic support defects such as vaginal vault prolapse or cystocele, and pelvic neuromuscular function.2 Poorly estrogenized, thin vaginal mucosa should be treated with local estrogen prior to surgical therapy. Levator tone and contraction strength are important factors in enhancing the long-term success rate of pelvic reconstructive surgery. Regularly performed Kegel exercises should be recommended following pelvic reconstructive procedures.2 Biofeedback therapy may be necessary to instruct patients how to adequately isolate and contract their pelvic floor muscles.
ANORECTAL PHYSIOLOGIC TESTS
Due to their increasing availability, anorectal physiologic testing is frequently used in the diagnosis and treatment of anorectal pathology. Specifically, defecography allows dynamic visualization of rectal evacuation, detects the presence of a rectocele, and evaluates degree, ease, and rapidity of emptying.1 Defecography is performed by introducing 100 to 250 cc of radiopaque paste into the rectum. An additional 20 to 50 cc of liquid barium can be added to highlight details with a double-contrast technique. Rectal contrast is inserted with the patient lying on the left lateral position on the x-ray table. Subsequently, the x-ray table is positioned upright so that the patient is seated in a lateral position over a plastic radiolucent commode filled with water to create a phantom similar to soft tissue.10,11
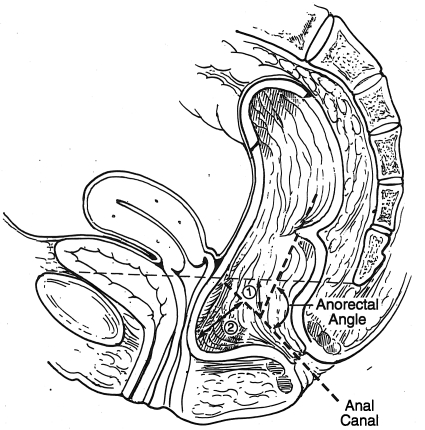
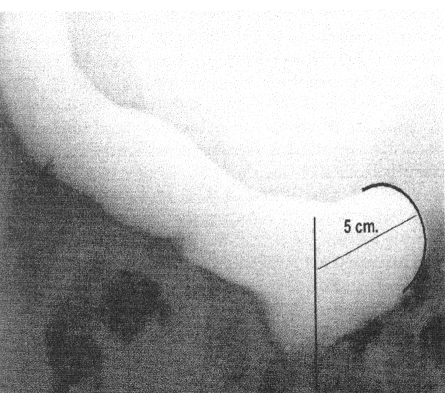
The fluoroscopic monitor is connected to a video recording machine to allow the entire radiologic procedure to be recorded. Instant radiographs are also obtained at rest (R), squeeze (S), and during push (P); an optional postevacuatory film may also be taken. Measurements of anorectal angle (ARA), perineal descent (PD), and rectocele diameter can be accomplished as shown in Fig. 3. As shown in Fig. 4, rectocele diameter is the distance between the interpolated anterior anorectal axis and the anterior-most portion of the rectocele.12,13,14
Figure 3.
Measurements of anorectal angle (ARA), perineal descent (PD), and rectocele diameter can be obtained from defecography.
Figure 4.
Rectocele diameter is the distance between the interpolated anterior anorectal axis and the anterior-most portion of the rectocele.
Another cinedefecography criterion than can indicate surgery is barium retention inside the rectocele.1 The volume of barium retention depends on a methodologic variable including the volume of barium injected and the technique used to determine the volume. A radionuclide technique can yield 19 to 33% retention in volunteers as any radioactivity adherence to the rectal wall will be recorded as retained, whereas with standard defecography, a thin layer of barium on the mucosa is considered normal.15 Halligan et al,16 in a retrospective study, tried to determine whether rectocele size and contrast retention are significant findings. Neither evacuation time, completeness, maximal and distal intrarectal pressure, nor the need to digitate significantly differed among patients with no rectocele, with rectocele, and rectoceles with more than 10% of contrast retention. The authors concluded that because no impairment of evacuation appeared to be associated with a large rectocele or barium retention, these evacuation disorders should not be directly attributed to these proctographic findings. Ting et al17 presented similar results and concluded that defecographic findings do not, in general, explain incomplete emptying or the sense of incomplete evacuation, but may determine the location of the retained barium.
Coexisting causes of constipation such as rectoanal intussusception, paradoxical puborectalis contraction, and sigmoidocele are commonly observed. These associated conditions could be predictors of a poor outcome after surgical repair of rectocele, as the defect could be a secondary condition.18 In addition, slow colonic transit time should be excluded as the cause of constipation prior to rectocele repair. Despite these controversies, surgical treatment of rectocele should be restricted to patients in whom clinical, physical, and dynamic findings confirm this defect as the primary cause of the symptoms. It has been demonstrated that at most only 10 to 20% of rectoceles are clinically significant as the major cause of symptoms.18,19 Adequate patient selection for surgical repair of rectocele yields a success rate of up to 82% after one year of follow-up.20,21
TREATMENT
Medical
Once the diagnosis is confirmed, a high fiber diet (25 to 35 g/day) plus ingestion of two to three liters of noncaffeinated nonalcoholic fluids per 24 hours is recommended as initial conservative treatment. If control of the patient's symptoms is suboptimal, the authors will consider surgical therapy.
Surgical
Surgical indications for a symptomatic rectocele repair include the presence of obstructive defecation symptoms, lower pelvic pressure and heaviness, prolapse of the posterior vaginal wall, pelvic relaxation, or enlarged vaginal hiatus. Several approaches have been described to repair rectoceles. Gynecologists and many colorectal surgeons (including the authors) prefer the transvaginal approach (posterior colporrhaphy). Other options include a transanal and perineal approach.
TRANSVAGINAL REPAIR (POSTERIOR COLPORRHAPHY)
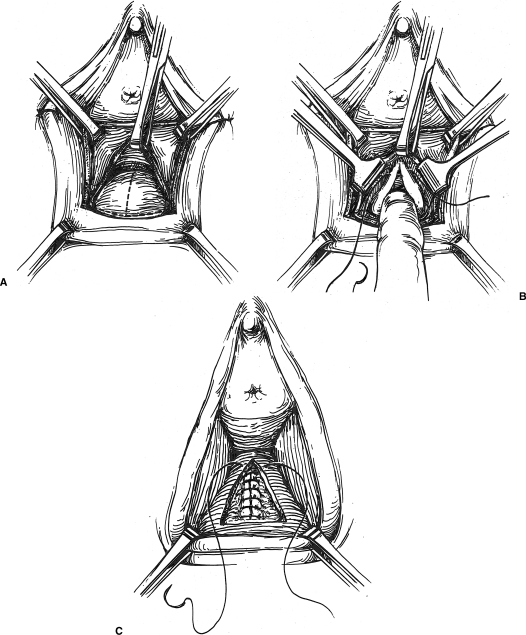
Posterior colporrhaphy is commonly performed in conjunction with a perineoplasty to address a relaxed perineum and widened genital hiatus. Preoperatively, the severity of the rectocele is assessed, as well as the desired final vaginal caliber. Allis clamps are placed on the inner labia minora/hymen remnants bilaterally and then approximated in the midline. The resultant vagina should loosely admit two to three fingers. A triangular (or transverse) incision over the perineal body is made between the Allis clamps (Fig. 5), and sharp and blunt dissection is then performed to separate the posterior vagina from the underlying rectovaginal fascia. A midline incision along the length of the vagina to a site above the superior edge of the rectocele may enhance exposure and allow excision of redundant vaginal mucosa.
Figure 5.
Transvaginal repair.
The dissection is carried laterally to the lateral vaginal sulcus and medial margins of the puborectalis muscles. The rectovaginal fascia with or without the underlying the levator ani muscles is then plicated with interrupted sutures of 2–0 polyglycolic acid (VicrylTM, Ethicon, Somerville, NJ) while depressing the anterior rectal wall. Typically, numerous slowly dissolving stitches are placed along the length of the rectocele. Excess vaginal mucosa is carefully trimmed and then reapproximated. A concomitant perineoplasty may be performed by plicating the bulbocavernosus and transverse perineal muscles. This reinforces the perineal body (or inferior central tendon) and provides enhanced support to the corrected rectocele.
Discrete tears or breaks in the rectovaginal fascia or rectovaginal septum have been described and may contribute to the formation of rectoceles. The intent of the discrete facial defect repair of rectoceles is to identify the fascial tears and reapproximate the edges. The surgical dissection is similar to the traditional posterior colporrhaphy whereby the vaginal mucosa is dissected off the underlying rectovaginal fascia to the lateral border of the levator muscles. However, instead of plicating the fascia and levator muscles in the midline, the fascial tears are identified and repaired with interrupted sutures. Richardson3 describes using a finger in the rectum to push anteriorly to identify areas of rectal muscularis that are not covered by the rectovaginal septum, thereby locating fascial defects. The operator can then identify fascial margins and reapproximate them. A perineoplasty may be necessary if a widened vaginal hiatus is present.
The posterior colporrhaphy has been the traditional approach to rectocele repair by gynecologists.2 Although commonly performed, it has been described as “among the most misunderstood and poorly performed” gynecologic surgeries.22 Although many authors have reported satisfactory anatomic results, conflicting effects on postoperative bowel and sexual function have been noted.23,24,25,26 These have included symptoms of a lump or pressure, symptoms of incomplete bowel emptying, constipation, fecal incontinence, and sexual dysfunction. A major component of sexual dysfunction is dyspareunia, which has been reported in up to 50% of patients and associated with plication of the levator ani muscles.24 This led some authors to recommend a modified rectocele repair, the discrete fascial defect repair.25 Quality of life measures have generally improved when studied postoperatively.26 Although published studies show promising anatomic and functional results; however, long-term prospective studies are warranted. In addition, unlike the traditional posterior colporrhaphy, the discrete fascial defect repair has thus far resulted in lessened incidence of postoperative dyspareunia.
TRANSPERINEAL REPAIRS
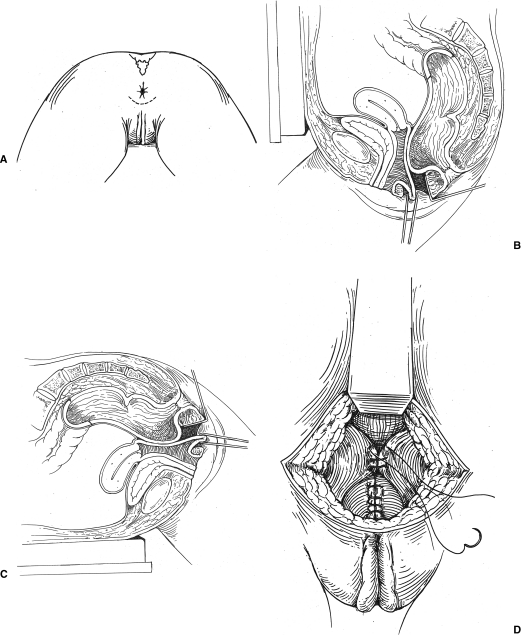
For a perineal approach, the patient is placed in the prone jackknife position and a U-shape incision is created (Fig. 6). A high dissection is undertaken to reach the vaginal cupola after which a trapezoid, L-shaped strip of posterior redundant vaginal wall is resected. Reconstruction is effected by a running suture of 3–0 polyglactin, and the space between the rectal and vaginal walls is closed and a levator plication is then executed by placing two to three single sutures. The skin is then completely closed; combined procedures for hemorrhoidectomies and/or fissures can be undertaken.
Figure 6.
Transperineal rectocele repair. (A) Surgical repair is performed using the perineal approach, through a U-shaped incision. (B, C) Redundant vaginal mucosa is retracted and resected. (D) Resected vaginal wall is sutured closed (inferiorly) and lateral rectovaginal fascia is plicated with sutures. The skin is closed without drainage.
Synthetic and biologic mesh has been used to repair or augment tissue repairs of rectoceles. One study showed no significant difference in recurrence or function between posterior colporrhaphy, site-specific repairs, or site-specific repairs with porcine small intestine submucosa mesh.27 Another study sutured Vicryl mesh to the posterior wall of the vagina in one arm and no mesh in the other arm. Recurrence was equal in both.28 There were no reports of mesh erosion in the two studies.27,28 All the studies employed small numbers of participants; therefore, any definitive conclusion as to the efficacy of mesh cannot be made.
TRANSANAL REPAIR
In 1967, Marks29 described a transanal repair of rectoceles. He felt that the prolapsed anterior rectal mucosa was a source of defecation difficulties that aggravated hemorrhoidal disease despite correction of the posterior vaginal wall and rectovaginal musculofascial layer. His procedure was performed in the lithotomy position. The redundant rectal mucosa was grasped and pulled outward until taut. A two-layer suture closure was performed underlying the rectal mucosa, including the hemorrhoid. The anterior rectal mucosa was then removed. The formation of scar at the suture line added to support.
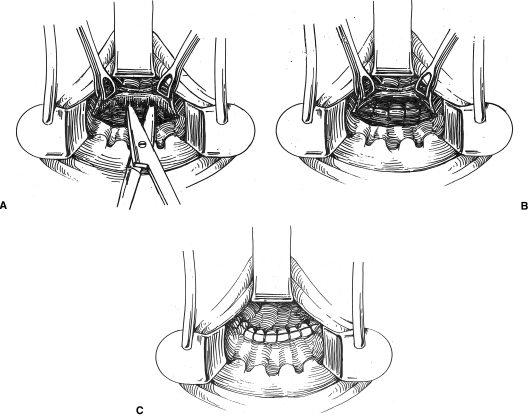
The transanal approach is preferred by many colorectal surgeons; many of their patients have other anorectal pathology such as hemorrhoids, and the postoperative pain may be less than with a transvaginal approach.30,31,32,33,34 Patients receive a mechanical and antibiotic bowel preparation. After institution of general or regional anesthesia, the patient is positioned in the prone jackknife position with the buttocks taped apart. The rectum is cleaned with povidone-iodine and exposure maintained with a retractor (e.g., Pratt bivalve). The size and location of the rectovaginal defect is confirmed by manual palpation. The submucosal plane is infiltrated with a 1:200,000 epinephrine solution. Depending on the type of repair that is to be performed, a vertical, horizontal, or elliptical incision in the anorectal mucosal is created using the electrocautery. Mucosal flaps are created with sharp and blunt dissection. Care is taken to maintain this dissection in the submucosal plane. Interrupted sutures of 2–0 polyglycolic acid are placed in a vertical or horizontal manner to plicate the rectal musculature and deeper supporting tissue (rectovaginal fascia). Redundant mucosa is then excised and the mucosa is closed with absorbable sutures (Fig. 7).
Figure 7.
Transanal repair. (A) Anal mucosa is excised; (B) rectal wall is plicated; (C) mucosa is closed.
Sehapayak33 described a transrectal repair of rectocele with multilayer closure, but with resection of the lateral quadrants of the mucosal prolapse rather than the anterior quadrant. In addition, he described repair of the rectovaginal septum through a transanal approach. A midline incision is made distal to the dentate line, continued 7 to 8 cm above the anorectal ring, and deepened to the rectovaginal septum. The levator ani muscle and rectovaginal fascia are then plicated separately. Ninety-eight percent of 355 patients reported improvement. In terms of bowel function, 49.5% were asymptomatic, 35% had occasional straining, and 14% used laxatives. They did not report on postoperative sexual function. Complications included a rectovaginal fistula and infection. The author warns against performing this procedure for high rectoceles or enteroceles or combining it with transvaginal surgery secondary to the risk of infection.
Arnold et al34 retrospectively compared rectocele operations performed transanally versus transvaginally. In all, fifty-four percent had postoperative constipation, and 34% had gas, liquid, or stool incontinence. Sexual dysfunction was reported in 22%. These complications occurred equally among the two groups of patients. The only significant difference was that the patients repaired transvaginally had more persistent pain. Khubchandani and colleagues31 recently reported a retrospective study of 123 consecutive cases of transanal repair of rectocele. Patient satisfaction was noted as 82% and the complication rate was only 3%, confirming the validity of this simple technique.
OTHER TECHNIQUES
Fox and Stanton35 describe mesh interposition to correct a rectocele at the time of abdominal sacrocolpopexy for vaginal vault prolapse. This simplifies the approach for patients with both vaginal vault prolapse and rectocele, as it alleviates the need for a concomitant vaginal procedure. The rectum is dissected from the posterior vaginal wall to the perineal body and a continuous piece of mesh is placed from the perineal body to the vaginal vault. The mesh is then tied to the anterior longitudinal ligament overlying the sacral promontory in a tension-free fashion. The authors treated 29 patients with this surgery and reported significant improvement in prolapse symptoms. They were dissatisfied, however, by the continued bowel symptoms including constipation and incomplete defecation. Similarly, Taylor et al36 reported a persistence or increase in bowel symptoms in 39% of their patients who underwent this type of surgery.
Laparoscopic rectocele repair involves opening the rectovaginal space and dissecting inferiorly to the perineal body. The perineal body is sutured to the rectovaginal septum and rectovaginal fascial defects are identified and closed. The levator ani muscles may be plicated. The advantages are reported to be better visualization secondary to magnification and insufflation, and more rapid recovery, with decreased pain and hospitalization. Disadvantages are many, including difficulty with laparoscopic suturing, increased operating time and expense, and an extended learning curve.37
Few reports describing outcomes of laparoscopic surgery for pelvic organ prolapse exist in the literature. Lyons and Winer38 described the use of polyglactin mesh in laparoscopic rectocele repair in 20 patients, with 80% reporting relief of both prolapse symptoms and the need for manual assistance to defecate. Further studies are needed to assess this surgical approach for rectocele repair.
DISCUSSION
Gynecologic indications for rectocele repair are numerous compared with traditional colorectal indications. It must be remembered that gynecologists primarily address vaginal symptoms when repairing a rectocele. Thus, obstructive defecation symptoms are only one of a list of accepted indications. Preoperative evaluation is typically restricted to clinical assessment including history and physical exam, and gynecologists rarely depend on defecography results in planning a reconstructive procedure for rectocele. Overall, surgical correction success rates are quite high when using a vaginal approach for rectocele correction. Vaginal dissection results in better visualization and access to the endopelvic fascia and levator musculature, which allows for a more firm anatomic correction. Further, and more comprehensive, data collection is necessary to better understand the effect of various rectocele repair techniques on defecatory, vaginal, and sexual symptoms.
REFERENCES
- 1.Rosato G O. In Beck DE, Wexner SD, editor. Fundamentals of Anorectal Surgery. London: WB Saunders; 1998. Rectocele and perineal hernias. pp. 187–197.
- 2.Pollak J, Davila G W. Rectocele repair: the gynecologic approach. Clin Colon Rectal Surg. 2003;16:61–69. [Google Scholar]
- 3.Richardson A C. The rectovaginal septum revisited: its relationship to rectocele and its importance in rectocele repair. Clin Obstet Gynecol. 1993;36(4):976–983. doi: 10.1097/00003081-199312000-00022. [DOI] [PubMed] [Google Scholar]
- 4.Uhlenhuth E, Wolfe W M, Smith E M, Middleton E B. The rectogenital septum. Surg Gynecol Obstet. 1948;86(2):148–163. [PubMed] [Google Scholar]
- 5.Milley P S, Nichols D H. A correlative investigation of the human rectovaginal septum. Anat Rec. 1969;163(3):443–451. doi: 10.1002/ar.1091630307. [DOI] [PubMed] [Google Scholar]
- 6.DeLancey J OL. Anatomic aspects of vaginal eversion after hysterectomy. Am J Obstet Gynecol. 1992;166(6 Pt 1):1717–1724. discussion 1724–1728. doi: 10.1016/0002-9378(92)91562-o. [DOI] [PubMed] [Google Scholar]
- 7.Shull B L, Bachofen C G. In: Walters MD, Karram MM, editor. Urogynecology and reconstructive pelvic surgery. 2nd ed. St. Louis: Mosby; 1999. Enterocele and rectocele. pp. 221–234.
- 8.Kahn M A, Stanton S L. Techniques of rectocele repair and their effects of bowel function. Int Urogynecol J. 1998;9:37–47. doi: 10.1007/BF01900540. [DOI] [PubMed] [Google Scholar]
- 9.Delancey J OL, Hurd W W. Size of the urogenital hiatus in the levator ani muscles in normal women and women with pelvic organ prolapse. Obstet Gynecol. 1998;91(3):364–368. doi: 10.1016/s0029-7844(97)00682-0. [DOI] [PubMed] [Google Scholar]
- 10.Bartram C I, Mahieu P HG. In: Henry MM, Swash M, editor. Coloproctology and the Pelvic Floor. 2nd ed. London: Butterworth-Heinemann; 1992. Evacuation proctography and anal endosonography. pp. 149–561.
- 11.Jorge J MN, Wexner S D, Marchetti F, Rosato G O, Sullivan M L, Jagelman D G. How reliable are currently available methods of measuring the anorectal angle? Dis Colon Rectum. 1992;35(4):332–338. doi: 10.1007/BF02048110. [DOI] [PubMed] [Google Scholar]
- 12.Yoshioka K, Matsui Y, Yamada O, et al. Physiologic and anatomic assessment of patients with rectocele. Dis Colon Rectum. 1991;34(8):704–708. doi: 10.1007/BF02050355. [DOI] [PubMed] [Google Scholar]
- 13.Lu R H. [Rectocele: analysis of 239 cases] Ching Hua Wai Ko Tsa Chih. 1990;28(2):102–104. 127. [PubMed] [Google Scholar]
- 14.Lucas J D, Landy L B. The gynecologist's approach to anterior rectoceles. Semin Colon Rectal Surg. 1992;3(2):138–143. [Google Scholar]
- 15.Ambroze W L, Pemberton J H, Bell A M, Brown M L, Zinsmeister A R. The effect of stool consistency on rectal and neorectal emptying. Dis Colon Rectum. 1991;34(1):1–7. doi: 10.1007/BF02050199. [DOI] [PubMed] [Google Scholar]
- 16.Halligan S, Bartram C I. Is barium trapping in rectoceles significant? Dis Colon Rectum. 1995;38(7):764–768. doi: 10.1007/BF02048038. [DOI] [PubMed] [Google Scholar]
- 17.Ting K-H, Mangel E, Eibl-Eibesfeldt B, Müller-Lissner S A. Is the volume retained after defecation a valuable parameter at defecography? Dis Colon Rectum. 1992;35(8):762–767. doi: 10.1007/BF02050326. [DOI] [PubMed] [Google Scholar]
- 18.Johansson C, Nilsson B Y, Holmström B, Dolk A, Mellgren A. Association between rectocele and paradoxical sphincter response. Dis Colon Rectum. 1992;35(5):503–509. doi: 10.1007/BF02049410. [DOI] [PubMed] [Google Scholar]
- 19.Block I R. Transrectal repair of rectocele using obliterative suture. Dis Colon Rectum. 1986;29:707–711. doi: 10.1007/BF02555314. [DOI] [PubMed] [Google Scholar]
- 20.Khubchandani I T, Sheets J A, Stasik J J, Hakki A R. Endorectal repair of rectocele. Dis Colon Rectum. 1983;26(12):792–796. doi: 10.1007/BF02554752. [DOI] [PubMed] [Google Scholar]
- 21.Sehapayak S. Transrectal repair of rectocele: an extended armamentarium of colorectal surgeons. A report of 355 cases. Dis Colon Rectum. 1985;28(6):422–433. doi: 10.1007/BF02560230. [DOI] [PubMed] [Google Scholar]
- 22.Nichols D H. Posterior colporrhaphy and perineorrhaphy: separate and distinct operations. Am J Obstet Gynecol. 1991;164(3):714–721. doi: 10.1016/0002-9378(91)90503-j. [DOI] [PubMed] [Google Scholar]
- 23.Francis W J, Jeffcoate T N. Dyspareunia following vaginal operations. J Obstet Gynaecol Br Commonw. 1961;68:1–10. doi: 10.1111/j.1471-0528.1961.tb02679.x. [DOI] [PubMed] [Google Scholar]
- 24.Kahn M A, Stanton S L. Posterior colporrhaphy: its effects on bowel and sexual function. Br J Obstet Gynaecol. 1997;104(1):82–86. doi: 10.1111/j.1471-0528.1997.tb10654.x. [DOI] [PubMed] [Google Scholar]
- 25.Cundiff G W, Weidner A C, Visco A G, Addison W A, Bump R C. An anatomic and functional assessment of the discrete defect rectocele repair. Am J Obstet Gynecol. 1998;179(6 Pt 1):1451–1456. discussion 1456–1457. doi: 10.1016/s0002-9378(98)70009-2. [DOI] [PubMed] [Google Scholar]
- 26.Porter W E, Steele A, Walsh P, Kohli N, Karram M M. The anatomic and functional outcomes of defect-specific rectocele repairs. Am J Obstet Gynecol. 1999;181(6):1353–1358. discussion 1358–1359. doi: 10.1016/s0002-9378(99)70376-5. [DOI] [PubMed] [Google Scholar]
- 27.Paraiso M F, Barber M D, Muir T W, Walters M D. Rectocele repair: a randomized trial of three surgical techniques including graft augmentation. Am J Obstet Gynecol. 2006;195(6):1762–1771. doi: 10.1016/j.ajog.2006.07.026. [DOI] [PubMed] [Google Scholar]
- 28.Sand P K, Koduri S, Lobel R W, et al. Prospective randomized trial of polyglactin 910 mesh to prevent recurrence of cystoceles and rectoceles. Am J Obstet Gynecol. 2001;184(7):1357–1362. discussion 1362–1364. doi: 10.1067/mob.2001.115118. [DOI] [PubMed] [Google Scholar]
- 29.Marks M M. The rectal side of the rectocele. Dis Colon Rectum. 1967;10(5):387–388. doi: 10.1007/BF02617024. [DOI] [PubMed] [Google Scholar]
- 30.Arnold M W, Stewart W RC, Aguilar P S. Rectocele repair. Four years' experience. Dis Colon Rectum. 1990;33(8):684–687. doi: 10.1007/BF02150745. [DOI] [PubMed] [Google Scholar]
- 31.Khubchandani I T, Clancy J P, III, Rosen L, Riether R D, Stasik J J., Jr Endorectal repair of rectocele revisited. Br J Surg. 1997;84(1):89–91. doi: 10.1046/j.1365-2168.1997.02463.x. [DOI] [PubMed] [Google Scholar]
- 32.Murthy V K, Orkin B A, Smith L E, Glassman L M. Excellent outcome using selective criteria for rectocele repair. Dis Colon Rectum. 1996;39:374–378. doi: 10.1007/BF02054049. [DOI] [PubMed] [Google Scholar]
- 33.Sehapayak S. Transrectal repair of rectocele: an extended armamentarium of colorectal surgeons. Dis Colon Rectum. 1985;6:422–433. doi: 10.1007/BF02560230. [DOI] [PubMed] [Google Scholar]
- 34.Arnold M W, Stewart W RC, Aguilar P S. Rectocele repair. Four years' experience. Dis Colon Rectum. 1990;33(8):684–687. doi: 10.1007/BF02150745. [DOI] [PubMed] [Google Scholar]
- 35.Fox S D, Stanton S L. Vault prolapse and rectocele: assessment of repair using sacrocolpopexy with mesh interposition. Br J Obstet Gynaecol. 2000;107:1371–1375. doi: 10.1111/j.1471-0528.2000.tb11650.x. [DOI] [PubMed] [Google Scholar]
- 36.Taylor G M, Ballard P, Jarvis G J. Vault prolapse and rectocele: assessment of repair using sacrocolpopexy with mesh interposition. Br J Obstet Gynaecol. 2001;8:775–776. doi: 10.1111/j.1471-0528.2001.00167.x. [DOI] [PubMed] [Google Scholar]
- 37.Paraiso M FR, Falcone T, Walters M D. Laparoscopic surgery for enterocele, vaginal apex prolapse and rectocele. Int Urogynecol J. 1997;8:146–152. doi: 10.1007/s001920050050. [DOI] [PubMed] [Google Scholar]
- 38.Lyons T L, Winer W K. Laparoscopic rectocele repair using polyglactin mesh. J Am Assoc Gynecol Laparosc. 1997;4(3):381–384. doi: 10.1016/s1074-3804(05)80232-4. [DOI] [PubMed] [Google Scholar]