Abstract
Background
Increased esophagogastric junction (EGJ) compliance is a key abnormality in GERD leading to increased volumes of reflux. To date, EGJ distensibility has been measured only with investigational barostatbased prototype devices.
Objectives
The aim of the study was to test the endoscopic functional luminal imaging probe (EndoFLIP), a new commercially available technology designed to measure intraluminal distensibility, by assessing the EGJ of GERD patients and controls.
Design
Prospective case-control series.
Setting
Tertiary referral center.
Subjects
Twenty GERD patients and 20 controls studied during a routine esophagogastroduodenoscopy.
Methods
The EndoFLIP was passed through the endoscopic instrumentation channel and positioned across the EGJ. The EndoFLIP uses impedance planimetry to measure 16 cross-sectional areas (CSA) along with the corresponding intrabag pressure within a 4.6-cm cylindrical segment of a fluid-filled bag.
Main Outcome Measurement
EGJ distensibility was assessed with 10- to 40-mL volume-controlled distentions.
Results
In both groups, the least distensible locus at the EGJ was usually at the hiatus. As a group, GERD patients exhibited two- to threefold increased EGJ distensibility compared with controls, particularly at 20- to 30-mL distention volumes, values quantitatively similar to previous measurements with barostat-based devices. The endoscopic estimation of EGJ distensibility, the flap valve grade, correlated poorly with EndoFLIP measurements.
Limitations
Heterogeneity of GERD patients.
Conclusions
Measurement of EGJ distensibility with EndoFLIP is feasible during clinical endoscopy. Stratifying GERD patients according to this physiological parameter may facilitate the identification of patient subgroups responsive or unresponsive to medical or surgical treatments.
Excessive esophagogastric junction (EGJ) compliance is a primary pathophysiological abnormality in many cases of GERD.1-4 Increased EGJ compliance allows greater volumes of gastric content to reflux into the esophagus,5 increases the frequency with which transient lower esophageal sphincter (LES) relaxations are elicited by proximal gastric distention,6 and allows gastric juice to track within the contracted sphincter.7-9 These physiological aberrations result in an increased number of reflux events, increased spatial distribution of refluxate within the esophagus, and increased esophageal acid exposure, all of which increase the likelihood of esophageal mucosal injury and reflux-related symptoms. Hence, quantifying EGJ compliance may discern clinically relevant subsets of GERD patients. The current clinical assessment of EGJ function is limited to endoscopic or radiographic imaging, manometric measures, or reflux testing with pH-metry. Although these studies can confirm excessive reflux, contractile defects in the LES pressure or crural diaphragm, and misalignment between the 2 manifest as a sliding hiatal hernia, which are all common in GERD,3,10,11 they do not quantify EGJ compliance. Inherent in the definition (change in volume when subject to an applied force), measuring EGJ compliance requires it being challenged with intraluminal distention.12
To date, the EGJ distensibility has been measured only with purpose-built equipment prototypes within the domain of research laboratories, which found the narrowest locus to be greater in GERD patients compared with control subjects.3,4 Although a transorally placed esophageal hydrostat or barostat can apply distending pressure at the EGJ and thus restrict dimensional measurements to the area of interest, both techniques are cumbersome and require concurrent fluoroscopic imaging.2-4 A more robust method for measuring EGJ distensibility, providing cross-sectional area (CSA) measurements at multiple adjacent segments without the need for fluoroscopy, adapts the principle of impedance planimetry13,14 into a functional luminal imaging probe (FLIP). FLIP recordings allow dynamic imaging of EGJ distention as a cylinder of varying diameter based on instantaneous CSA measurements with concurrent pressure measurements. Quantifying intraluminal pressure along with the CSA permits calculation of EGJ distensibility.15-17 The original FLIP, designed for transnasal passage, was of relatively large diameter and too poorly tolerated for general clinical use. Consequently, a new smaller probe was designed to fit through the instrumentation channel of an endoscope. The distention bag can be localized at the EGJ for compliance measurements during routine clinical endoscopy, opening the possibility of measuring EGJ compliance as part of the diagnostic evaluation of GERD. Hence, the aim of the current study was to test the performance of the new endoscopic FLIP system (EndoFLIP; Crospon Ltd, Galway, Ireland) and to compare the EGJ distensibility in GERD patients with that of asymptomatic control subjects.
MATERIALS AND METHODS
Subjects
Twenty control subjects (4 men; age, 18-42; median age, 27 years) and 20 patients with GERD (10 men; age, 22-79 years; median age, 31 years) were studied. The control subjects were recruited from a pool of asymptomatic volunteers with no GERD-associated symptoms. GERD patients were recruited from a pool of volunteers and patients reporting heartburn at least 2 to 3 days per week while not taking antisecretory medicines. None of the subjects had a history of GI surgery or significant medical disease. All subjects gave written informed consent. The study protocol was approved by the Northwestern University Institutional Review Board.
Take-home Message.
The EndoFLIP device yielded values for EGJ distensibility quantitatively similar to previous measurements with barostat-based devices. The least distensible locus within the EGJ was usually at the hiatus and was 2 to 3 times more distensible in GERD patients than controls. Stratifying GERD patients by EGJ distensibility can potentially match GERD patient subgroups to specific mechanistic treatments.
EndoFLIP system
EGJ distensibility was measured by using a commercially developed FLIP, the EndoFLIP, based on the concepts described previously.16 In brief, the EndoFLIP (Fig. 1) uses impedance planimetry to determine multiple adjacent CSAs within a cylindrical bag placed in a tubular organ during volumetric distention. The additional measure of the corresponding intrabag pressure allows assessment of the CSA pressure response (distensibility) of the distended area.
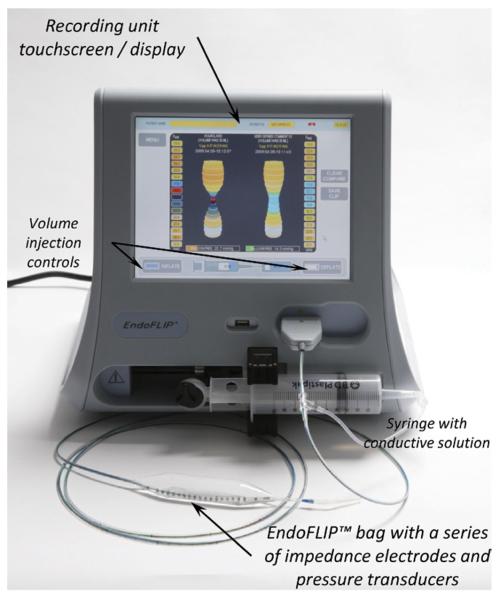
Figure 1.
The Endoscopic Functional Luminal Imaging Probe (EndoFLIP) System. Once the bag is filled with a conductive solution, 16 intraluminal cross-sectional areas (CSA) are measured within the central part of the bag by the impedance measuring electrodes, whereas the pressure transducers provide the corresponding intrabag pressure. The volume of conductive solution injected from the syringe to the bag is controlled via the touch screen on the recording unit. The screen displays the calculated CSAs as a cylinder of varying diameter in real time along with the corresponding intrabag pressure. There is also the option of a split-screen display, as demonstrated here, which can display a snapshot from previous distention simultaneously with the current acquisition. (Photograph courtesy of Crospon Ltd., Galway, Ireland.)
The EndoFLIP assembly was 240 cm long with a 3-mm outer diameter. A noncompliant bag (up to a fill-in volume of 40 mL) mounted on the distal 14 cm of the probe was designed to assume a cylindrical shape 7.0 cm long with maximal diameter of 2.5 cm, when fully distended, between the tapering cone-shaped ends sealed at the assembly. The minimal-to-maximal detectable CSA range was 10 to 450 mm2. Within the bag was a 6.4-cm segment comprising 17 ring electrodes spaced 4 mm apart for impedance planimetry measurements. Excitation electrodes at either end emitted a constant low current making the voltage measured across each of the 16 adjacent pairs of ring electrodes proportional to the impedance between them. As the bag was filled with a specially formulated conductive solution, the impedance across each segment was inversely proportional to the CSA of the bag at that locus. The probe also contained 2 low-compliance perfusion channels connected to external pressure transducers housed within the recording unit that measured intrabag pressure.
Measurements from the 16 electrode pairs and pressure transducers were sampled at 10 Hz with the data acquisition system and transmitted to the recording unit, which displayed them in real time as a 6.4-cm cylinder of varying diameters along its length reflecting the 16 measured CSAs. The probe and the pressure transducers were precalibrated during the manufacturing process and required no additional calibration before use (CSA resolution 0.8 mm2, accuracy ±0.8 mm2; intrabag pressure resolution 0.1 mm Hg, accuracy ±0.8 mm Hg). Before use, the air was removed from the EndoFLIP assembly by using an automated purge sequence. Subsequent establishment of pressure baseline and infusion of the conductive solution were controlled by using the touch screen on the recording unit positioned at the level of the subject's torso.
Endoscopy
Subjects underwent esophagogastroduodenoscopy in the left lateral decubitus position to evaluate for esophagitis and EGJ anatomy and to place the EndoFLIP probe across the EGJ. A therapeutic gastroscope with an 11.3-mm diameter with a 3.7-mm diameter instrument channel was used (Olympus GIF type 1TQ160; Olympus Corp, Tokyo, Japan). Moderate sedation was administered during the procedure (5-15 mg midazolam, 20-200 μg fentanyl with similar doses in each subject group). Esophagitis was graded according to the Los Angeles classification where mucosal breaks were less (A) than 5 mm long, but not extending between the tops of 2 mucosal folds; (B) more than 5 mm long, but not extending between the tops of 2 mucosal folds; (C) continuous between the tops of 2 or more mucosal folds, but involving less than 75% of the circumference; and (D) involving 75% or more of the esophageal circumference.18 EGJ morphology was graded according to the “flap valve” concept: (I) a normal ridge of tissue closely approximated to the gastroscope, (II) the ridge slightly less well defined and opening with respiration, (III) the ridge effaced and the hiatus patulous, and (IV) the hiatus open at all times and the squamocolumnar junction displaced axially.19
EndoFLIP protocol
At the end of the endoscopy, the EndoFLIP probe was threaded through the instrumentation channel until the entire bag was seen within the stomach. The air insufflated during endoscopy was removed and the intrabag pressure was zeroed within the stomach; therefore, subsequent pressure measurements were relative to the baseline gastric pressure. The endoscope and probe were withdrawn such that its bag straddled the EGJ and the endoscope tip remained in the distal esophagus. CSA and the corresponding distention pressure measurements were made with the bag filled to 10, 20, 30, and 40 mL. EndoFLIP measurements were monitored in real-time to ensure proper bag placement by using both the direct visualization through the endoscope and the display of CSAs on the recording unit. If the bag migrated, it was repositioned, and the measurement was repeated. Measurements interrupted by esophageal peristalsis were also repeated.
Data analysis
The 16 CSAs and intrabag pressure were assessed at each distention volume by quantifying the 50th percentile of each measure during each 30-second test recording.
EGJ distensibility (CSA vs pressure) was based on the narrowest CSA (invariably the diaphragmatic hiatus) and the corresponding intrabag pressure and expressed as the EGJ distensibility index at each distention volume. To account for the data with 0-mm Hg intrabag pressure, the index was calculated as [narrowest CSA/(intrabag pressure + intragastric pressure offset)], where intragastric pressure was given as 4.0 mm Hg based on previous reports of typical normal intragastric pressure values across healthy control and GERD patient groups.3
Statistical analysis
Data were expressed as median (5th-95th percentiles). Statistical comparisons used Wilcoxon matched pairs and Kruskal-Wallis tests. The bivariate relationship between gastroesophageal flap valve grade (categorical variable) and hiatal CSA or EGJ distensibility index (continuous variable) was assessed by using the Kruskal-Wallis test. Significance was set at P < .05.
RESULTS
Demographic data
Demographic data are summarized in Table 1. Eight of the 20 GERD patients had esophagitis: 5 Los Angeles grade A, 2 grade B, and 1 grade C. Surprisingly, gastroesophageal flap valve grade III was the most common grade reported in both the controls (10/20) and the GERD patients (11/20). None of the controls had a grade IV flap valve indicative of hiatal hernia. Esophageal acid exposure had been assessed in 4 of the patients with 2 having abnormal values (total percentage time pH <4>, 5%); neither had esophagitis.
TABLE 1.
Subject demographics
| Control subjects (n = 20) | GERD patients (n = 20) | |
|---|---|---|
| Male:female | 4:16 | 10:10 |
| Age, y | 18-42 | 22-79 |
| Gastroesophageal flap valve grade | I: n = 7; II: n = 3; III: n = 10; IV: n = 0 | I: n = 2; II: n = 4; III: n = 11; IV: n = 3 |
| Esophagitis (Los Angeles grading) | None | 8/20 |
| Abnormal pH study* | NA | 2/4 |
Total percentage time pH <4>, 5%.
Patients were included in the study based on the criterion of experiencing heartburn at least 2 to 3 days per week while not taking antisecretory medications. Additional symptoms potentially related to reflux included regurgitation (2/20), chest pain (1/20), cough (2/20), throat burning (2/20), and globus sensation (1/20). The majority of the patients (16/20) were on long-term proton pump inhibitor therapy: esomeprazole (9), omeprazole (4), rabeprazole (2), and lansoprazole (1), which provided adequate symptomatic relief. A minority of the patients opted to control their symptoms with over-the-counter calcium carbonate products instead (2/16) or remain off any medications despite having favorable experience with them in the past (2/16).
Hence, GERD diagnosis was based on objective evidence (esophagitis or positive pH-metry) in 10 of 20 patients and the combination of symptoms and response to antisecretory therapy in 10 of 20 patients.
EGJ distensibility
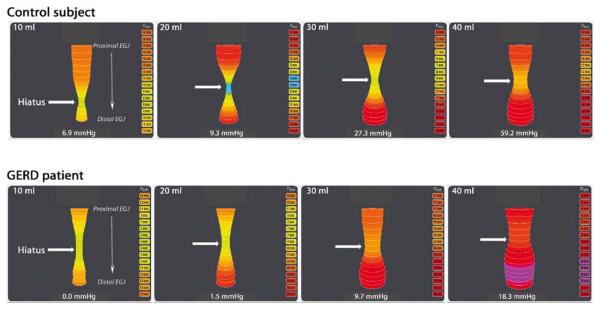
The EndoFLIP bag assumed an hourglass shape when distended straddling the EGJ with the central constriction at the diaphragmatic hiatus in both control subjects and GERD patients (Fig. 2). The hiatus was typically the least distensible locus with the majority of the conductive solution within the bag displaced proximally or distally to it. The hiatal CSA increased progressively with distending volumes (P < .0001) to a similar degree in both groups (P = .68, Table 2). The hiatal CSA was similar in patients with objectively documented and symptomatically defined GERD (P = .46). However, the hourglass shape was absent in 2 of 20 control subjects (both with gastroesophageal flap valve grade III) and 6 of 20 GERD patients (1 gastroesophageal flap valve grade II, 4 grade III, 1 grade IV). The bag instead assumed a cylindrical shape of uniform diameter. A “double” hourglass shape with 2 constriction loci was not seen.
Figure 2.
Examples of a set of volumetric EndoFLIP distentions in a control subject (top) and a GERD patient (bottom). Typically the hourglass shape of the EGJ narrowed at the hiatus (white arrow) in both groups. In each panel, EGJ distention is illustrated as a cylinder of varying diameter corresponding to the 16 CSAs measured by impedance planimetry within the EndoFLIP bag along with the corresponding intrabag pressures. These examples were selected to illustrate the greater EGJ distensibility in a GERD patient with consistently larger hiatal distention at 20- to 40-mL distention volumes coinciding with lower intrabag pressure.
TABLE 2.
CSA (mm2) of the diaphragmatic hiatus (narrowest EGJ CSA measured by the EndoFLIP) during volume distentions reported as median (5th to 95th percentiles)
| EndoFLIP bag volume (mL) |
Control subjects | GERD patients |
|---|---|---|
| 10 | 20 (12-110) | 20 (12-119) |
| 20 | 38 (13-94) | 36 (13-201) |
| 30 | 94 (27-225) | 116 (33-344) |
| 40 | 264 (99-496) | 180 (86-409) |
CSA, Cross-sectional area.
The minimal detectable CSA was approximately 10 mm2.
As the hiatal diameter increased with increasing distention volume, so did the pressure within the bag in both subject groups (P < .0001, Table 3). However, unlike the hiatal diameter, the distending pressure was consistently lower in GERD patients than in control subjects, particularly at 20-, 30-, and 40-mL bag volumes (P < .0005, Table 3). At any given distensile pressure, the extent of the EGJ opening in GERD patients was greater than in control subjects (Fig. 3). The intrabag pressure in patients with objectively documented or symptomatically defined GERD was comparable (P = .47).
TABLE 3.
Pressure (mm Hg) within the EndoFLIP bag during distention reported as median (5th to 95th percentiles)
| EndoFLIP bag volume (mL) |
Control subjects |
GERD patients |
|---|---|---|
| 10 | 3 (0-8) | 0.5 (0-11) |
| 20 | 9 (2-20) | 3 (0-9)* |
| 30 | 25 (6-47) | 10 (3-20)* |
| 40 | 39 (17-60) | 24 (13-51)* |
P < .01 versus controls.
Figure 3.
EGJ distensibility in control subjects and GERD patients. The EndoFLIP bag distensile pressure (x-axis) and the hiatal CSA (y-axis) were measured with the EndoFLIP bag filled to 10 mL (squares), 20 mL (diamonds), 30 mL (circles) and 40 mL (triangles). Both control subjects (open symbols) and GERD patients (closed symbols) exhibited measurable EGJ distention, particularly with 20- and 30-mL EndoFLIP bag volumes. However, the intrabag pressures associated with EGJ distention were consistently 6 to 15 mm Hg less in the GERD patients compared with the control subjects (see also Table 3). Data shown as medians.
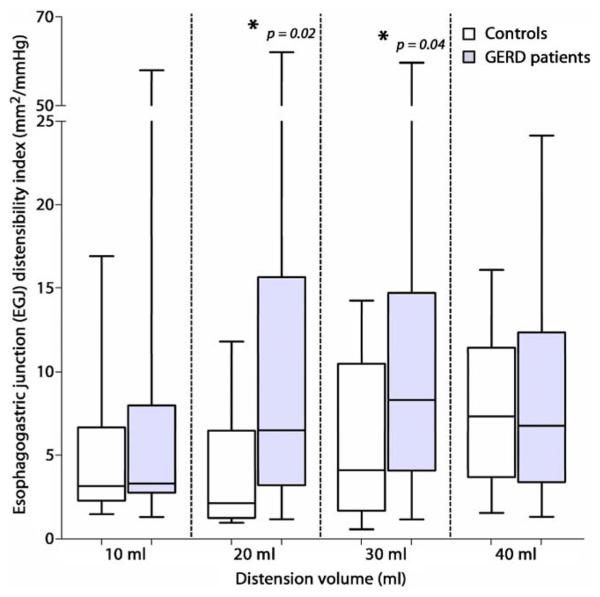
The data in Figure 3 were used to calculate an EGJ distensibility index (defined in the data analysis section). The index was significantly greater in the GERD patients (P = .01, Fig. 4). This difference was most prominent at the 20-mL (controls 2 [1-9] mm2/Hg vs GERD 7 [1-32] mm2/Hg, P = .02) and 30-mL distention volumes (controls 4 [1-14] mm2/Hg vs GERD 8 [1-46] mm2/Hg, P = .04) at which the index was three- and twofold greater, respectively. As with other measures, the EGJ distensibility index was similar for the objectively documented or symptomatically defined GERD subgroups (P = .88).
Figure 4.
Esophagogastric junction distensibility index in control subjects (white boxes) and GERD patients (gray boxes). *P = .02 versus controls.
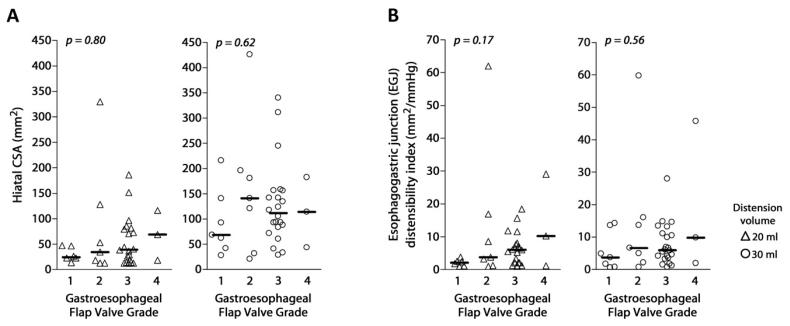
The agreement between endoscopically assessed EGJ morphology and EndoFLIP measures was tested with pooled data for control and GERD subjects. There was no relationship between the flap valve grade and either the hiatal CSA (P ≥ .62) or the EGJ distensibility index (P ≥ .17) at 20- and 30-mL distention volumes (Fig. 5).
Figure 5.
Endoscopic assessment of EGJ morphology versus functional EGJ assessment with EndoFLIP. The qualitative grading of EGJ patency (gastroesophageal flap valve grade19) was not related to either hiatal CSA measurement (A) or EGJ distensibility index (B) in the pooled data from control subjects and GERD patients at the distention volumes of 20 and 30 mL. Bold lines denote medians.
DISCUSSION
This investigation tested the ability of the EndoFLIP, a novel device based on impedance planimetry technology, to quantify EGJ distensibility in the course of routine endoscopy. Studies were done on 20 control subjects and 20 GERD patients. The major findings were that (1) EndoFLIP provided technically successful measurements in all cases; (2) in both subject groups, the least distensible locus within the EGJ (usually the hiatus) was most successfully isolated with 20- to 30-mL distentions; (3) consistent with previous investigations that used prototype barostat-based technology, EGJ distensibility was significantly greater in GERD patients than in control subjects; and (4) the nearest endoscopic gauge of EGJ distensibility, the flap valve grade, correlated poorly with EndoFLIP measurements.
The poor correlation between the endoscopic and EndoFLIP estimates of EGJ distensibility (Fig. 5) was somewhat unexpected. The endoscopic assessment is highly qualitative, and distending pressure is uncontrolled during endoscopy; still, one would have expected a correlation. This raises the issue of which measure is correct. Addressing that concern, it is useful to compare the EndoFLIP measurements with previous findings made with hydrostat device pressure-controlled distentions in similar groups of GERD and control subjects.3,4 The similarities are particularly apparent in Figure 3. In the hydrostat study, at the maximal distention pressure used (6 mm Hg relative to gastric pressure), the hiatal CSA was 38 ± 5 mm2 in controls, and 79 ± 11 mm2 in GERD patients. Extrapolating from Figure 3, the corresponding EndoFLIP measurements referenced to intragastric pressure would be 40 mm2 for control and 80 mm2 for GERD subjects, remarkably similar numbers. These opening diameters are less than the diameter of the endoscope used in the current study and emphasize the nonphysiologic circumstances implicit in endoscopic assessment.
Increased EGJ distensibility has major repercussions on the volume of reflux. Whether reflux events are attributable to transient LES relaxation or otherwise, there is a very robust relationship in which the volume of flow through the EGJ is proportional to the opening diameter raised to the fourth power.3,4 Clinical and scientific opinion20-24 suggests this to be an important distinction that potentially identifies a GERD subgroup with syndromes driven by large reflux volumes (regurgitation and chest pain) that may benefit from anatomic correction, by either surgical or evolving endoluminal techniques. It may also isolate a GERD subgroup with normal EGJ distensibility and little to gain from such interventions. Similarly, EndoFLIP measurements may be potentially useful in the course of surgical or endoluminal procedures to calibrate the magnitude of the intervention. That is not to say that EndoFLIP measurements, or any other putative measure, will represent a single unifying physiological feature defining GERD. GERD is recognized as a heterogeneous condition, and Figure 4 confirms this once again with respect to the metric of EGJ distensibility. When we used pooled data, the distensibility index in GERD patients was distinctly different from that of controls, but it also varied substantially among GERD patients.
In conclusion, this experiment appraised the ability of EndoFLIP, a new commercially available technology, to evaluate EGJ distensibility in the course of clinical endoscopy. The device performed well, both confirming previously described differences in EGJ distensibility between GERD patients and controls and producing results that were quantitatively very similar to measurements made with cumbersome purpose-built barostat-based prototypes requiring concurrent fluoroscopy. As anticipated, GERD patients were heterogeneous with respect to EGJ distensibility, but stratifying them according to this physiological parameter may identify those uniquely responsive to endoluminal or surgical therapies. Further studies, especially management trials based on EndoFLIP measures, will be needed to ascertain the degree to which that is possible.
Acknowledgments
Supported and sponsored by Crospon Ltd, Galway, Ireland; grant R01 DC00646 (P.J. Kahrilas and J.E. Pandolfino) from the Public Health Service; and the 2008 AGA June and Donald O. Castell Esophageal Clinical Research Award (J.E. Pandolfino).
Abbreviations
- CSA
cross-sectional area
- EGJ
esophagogastric junction
- FLIP
functional luminal imaging probe
- LES
lower esophageal sphincter
Footnotes
DISCLOSURE: The following author disclosed a financial relationship relevant to this publication: J.E. Pandolfino Scientific Advisory Board member, Crospon Ltd. The other authors disclosed no financial relationships relevant to this publication.
REFERENCES
- 1.Jenkinson AD, Scott SM, Yazaki E, et al. Compliance measurement of lower esophageal sphincter and esophageal body in achalasia and gastroesophageal reflux disease. Dig Dis Sci. 2001;46:1937–42. doi: 10.1023/a:1010639232137. [DOI] [PubMed] [Google Scholar]
- 2.Pandolfino JE, Curry J, Shi G, et al. Restoration of normal distensive characteristics of the esophagogastric junction after fundoplication. Ann Surg. 2005;242:43–8. doi: 10.1097/01.sla.0000167868.44211.f0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pandolfino JE, Shi G, Trueworthy B, et al. Esophagogastric junction opening during relaxation distinguishes nonhernia reflux patients, hernia patients, and normal subjects. Gastroenterology. 2003;125:1018–24. doi: 10.1016/s0016-5085(03)01210-1. [DOI] [PubMed] [Google Scholar]
- 4.Pandolfino JE, Shi G, Curry J, et al. Esophagogastric junction distensibility: a factor contributing to sphincter incompetence. Am J Physiol Gastrointest Liver Physiol. 2002;282:G1052–8. doi: 10.1152/ajpgi.00279.2001. [DOI] [PubMed] [Google Scholar]
- 5.Ghosh SK, Kahrilas PJ, Brasseur JG. Liquid in the gastroesophageal segment promotes reflux, but compliance does not: a mathematical modeling study. Am J Physiol Gastrointest Liver Physiol. 2008;295:G920–33. doi: 10.1152/ajpgi.90310.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kahrilas PJ, Shi G, Manka M, et al. Increased frequency of transient lower esophageal sphincter relaxation induced by gastric distention in reflux patients with hiatal hernia. Gastroenterology. 2000;118:688–95. doi: 10.1016/s0016-5085(00)70138-7. [DOI] [PubMed] [Google Scholar]
- 7.Pandolfino JE, Zhang Q, Ghosh SK, et al. Acidity surrounding the squamocolumnar junction in GERD patients: “acid pocket” versus “acid film.”. Am J Gastroenterol. 2007;102:2633–41. doi: 10.1111/j.1572-0241.2007.01488.x. [DOI] [PubMed] [Google Scholar]
- 8.Fletcher J, Wirz A, Young J, et al. Unbuffered highly acidic gastric juice exists at the gastroesophageal junction after a meal. Gastroenterology. 2001;121:775–83. doi: 10.1053/gast.2001.27997. [DOI] [PubMed] [Google Scholar]
- 9.Fletcher J, Wirz A, Henry E, et al. Studies of acid exposure immediately above the gastro-oesophageal squamocolumnar junction: evidence of short segment reflux. Gut. 2004;53:168–73. doi: 10.1136/gut.2003.022160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kahrilas PJ, Lin S, Manka M, et al. Esophagogastric junction pressure topography after fundoplication. Surgery. 2000;127:200–8. doi: 10.1067/msy.2000.102045. [DOI] [PubMed] [Google Scholar]
- 11.Lord RV, DeMeester SR, Peters JH, et al. Hiatal hernia, lower esophageal sphincter incompetence, and effectiveness of Nissen fundoplication in the spectrum of gastroesophageal reflux disease. J Gastrointest Surg. 2009;13:602–10. doi: 10.1007/s11605-008-0754-x. Epub 2008 Dec 3. [DOI] [PubMed] [Google Scholar]
- 12.Harris LD, Pope CE., 2nd “Squeeze” vs. resistance: an evaluation of the mechanism of sphincter competence. J Clin Invest. 1964;43:2272–8. doi: 10.1172/JCI105101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.McMahon BP, Drewes AM, Gregersen H. Functional oesophago-gastric junction imaging. World J Gastroenterol. 2006;12:2818–24. doi: 10.3748/wjg.v12.i18.2818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gregersen H. Biomechanics of the gastrointestinal tract: new perspectives in motility research and diagnostics. Springer-Verlag; Heidelberg: 2003. [Google Scholar]
- 15.McMahon BP, Frokjaer JB, Kunwald P, et al. The functional lumen imaging probe (FLIP) for evaluation of the esophagogastric junction. Am J Physiol Gastrointest Liver Physiol. 2007;292:G377–84. doi: 10.1152/ajpgi.00311.2006. [DOI] [PubMed] [Google Scholar]
- 16.McMahon BP, Frokjaer JB, Liao D, et al. A new technique for evaluating sphincter function in visceral organs: application of the functional lumen imaging probe (FLIP) for the evaluation of the oesophago-gastric junction. Physiol Meas. 2005;26:823–36. doi: 10.1088/0967-3334/26/5/019. [DOI] [PubMed] [Google Scholar]
- 17.Kwiatek MA, Kahrilas PJ, Soper NJ, et al. Esophagogastric junction distensibility after fundoplication assessed with a novel functional luminal imaging probe. J Gastrointest Surg. 2009;14:268–76. doi: 10.1007/s11605-009-1086-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lundell LR, Dent J, Bennett JR, et al. Endoscopic assessment of oesophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut. 1999;45:172–80. doi: 10.1136/gut.45.2.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hill LD, Kozarek RA, Kraemer SJ, et al. The gastroesophageal flap valve: in vitro and in vivo observations. Gastrointest Endosc. 1996;44:541–7. doi: 10.1016/s0016-5107(96)70006-8. [DOI] [PubMed] [Google Scholar]
- 20.Kahrilas PJ. Clinical practice. Gastroesophageal reflux disease. N Engl J Med. 2008;359:1700–7. doi: 10.1056/NEJMcp0804684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lundell L. Anti-reflux surgery in the laparoscopic era. Baillieres Best Pract Res Clin Gastroenterol. 2000;14:793–810. doi: 10.1053/bega.2000.0125. [DOI] [PubMed] [Google Scholar]
- 22.Kahrilas PJ, Shaheen NJ, Vaezi MF. American Gastroenterological Association Institute technical review on the management of gastroesophageal reflux disease. Gastroenterology. 2008;135:1392–413. doi: 10.1053/j.gastro.2008.08.044. 1413.e1-5. Epub 2008 Sep 16. [DOI] [PubMed] [Google Scholar]
- 23.Kahrilas PJ, Shaheen NJ, Vaezi MF, et al. American Gastroenterological Association Medical Position Statement on the management of gastroesophageal reflux disease. Gastroenterology. 2008;135:1383–91. doi: 10.1053/j.gastro.2008.08.045. 1391.e1-5. [DOI] [PubMed] [Google Scholar]
- 24.Sifrim D, Mittal R, Fass R, et al. Review article: acidity and volume of the refluxate in the genesis of gastro-oesophageal reflux disease symptoms. Aliment Pharmacol Ther. 2007;25:1003–17. doi: 10.1111/j.1365-2036.2007.03281.x. [DOI] [PubMed] [Google Scholar]