Abstract
Objective
To assess incidence and mortality impact of extra-articular rheumatoid arthritis (ExRA) in patients with incident rheumatoid arthritis (RA) in 1995–2007 vs 1985–1994.
Methods
Data on incident ExRA were abstracted from medical records of RA patients, Olmsted County, Minnesota residents who first met the 1987 ACR criteria for RA between 1/1/1995 and 12/31/2007. Patients were followed until death, migration from Olmsted County, or 12/31/2008. ExRA were classified using the predefined criteria and compared to the corresponding 1985–1994 inception RA cohort (n=147).
Results
The 1995–2007 cohort included 463 RA patients followed for a mean of 6.3 years. Mean age was 55.6 years (69% female; 67% rheumatoid factor [RF] positive). The 10-year cumulative incidence of any ExRA (50.1%) and severe ExRA (6.7%) in 1995–2007 cohort was similar to the 1985–1994 cohort (46.2% and 9.7%, respectively). The 10-year cumulative incidence of vasculitis, but not other ExRA, was significantly lower in 1995–2007 cohort (0.6%) compared to the 1985–1994 cohort (3.6%). RF positivity, erosions/destructive changes, use of methotrexate, other disease-modifying antirheumatic drugs and systemic corticosteroids were significantly associated with ExRA in the 1995–2007 cohort. ExRA were associated with mortality risk (HR 2.1, 95%CI 1.2, 3.7) in the 1995–2007 cohort. The decrease in mortality following ExRA in the 1995–2007 cohort vs 1985–1994 cohort did not reach statistical significance (HR 0.6, 95%CI 0.3, 1.2, p=0.16).
Conclusions
ExRA remain a common complication associated with increased mortality in RA. The occurrence of vasculitis appears to be decreasing in the recent years.
Keywords: rheumatoid arthritis, extra-articular manifestations, incidence, mortality, risk factors
Introduction
Extra-articular manifestations of rheumatoid arthritis (ExRA) occur in a substantial proportion of patients with rheumatoid arthritis (RA) (~40%) and are associated with particularly severe and disabling disease, frequent life-threatening comorbidities and poor outcomes (1–7). Some reports suggest that in recent years, RA may be becoming milder with lower disease activity, less radiological progression and lower rates of functional disability, which to a substantial degree is likely related to improved therapeutics (8–11). Whether the epidemiological pattern of ExRA occurrence has changed in recent years has not been examined in population-based patient cohorts. To better understand the recent trends in ExRA incidence and mortality and to determine the risk factors associated with ExRA occurrence, we performed a longitudinal study on a population-based inception cohort of Olmsted County, Minnesota residents first diagnosed with RA between 1/1/1995 and 12/31/2007. This study extended our previous observations of ExRA epidemiology from earlier decades (1955–1994) (7, 12, 13).
Methods
Study setting
The population of Olmsted County, Minnesota is geographically defined with the majority of the population (65–70%, according to the recent estimates) living in the city of Rochester. The availability of comprehensive medical records for all residents seeking medical care makes the population of Olmsted County an optimal resource for investigation of longitudinal trends in RA epidemiology and associated ExRA. The resources of Rochester Epidemiology Project (REP) provide ready access to the medical records of all Olmsted County, Minnesota residents from all in-patient and out-patient health care providers including the Mayo Clinic and its affiliated hospitals for the past 65 years. This system ensures virtually complete ascertainment of all clinically recognized cases of RA in patients residing in Olmsted County, Minnesota. The unique features and potential of the REP for population-based studies have been described previously (14, 15).
Study population and data collection
The study population comprised a retrospectively identified population-based incidence cohort of RA patients who were Olmsted County residents ≥18 years of age and first met the 1987 American College of Rheumatology (ACR) criteria for RA between 1/1/1995 and 12/31/2007 (16). RA incidence date was defined as the earliest date at which the patient fulfilled ≥4 1987 ACR criteria for RA. The complete (in-patient and out-patient) medical records of this incident 1995–2007 cohort of RA patients were reviewed by one of the authors (EM) using a structured protocol. ExRA were classified according to the criteria used in our previous studies ((13), Table 1). Severe ExRA were defined according to Malmö criteria (5) and included pericarditis, pleuritis, Felty’s syndrome, glomerulonephritis, vasculitis, peripheral neuropathy, scleritis and episcleritis. All questionable cases were additionally reviewed by co-investigators supervised by the principal investigator (ELM) and confirmation or rejection of the case-status was accomplished based on the predefined criteria (5, 13). The accuracy and completeness of the data collection was confirmed by the principal investigator through the independent review of a subsample of 80 medical records with consistent results.
Table 1.
Criteria for inclusion as extra-articular manifestations of RA
| 1. Pericarditis | (A) Clinical judgment and exudation verified by echocardiography |
| If ultrasound not available: criteria according to Hara et al* | |
| (B) Clinical criteria: (1 required) | |
| Typical pericardial pain, peripheral oedema, dyspnoea/orthopnoea, ascites, dysrythmia (heart rate > 140/min, atrial flutter/fibrillation, 2–3 degree atrioventricular block, ventricular tachycardia) | |
| Objective criteria compatible with pericarditis: (1 required) | |
| Physical examination | |
| Cardiac catheterisation findings | |
| Histological examination | |
| Other causes improbable, such as tuberculosis or other infection, metastases, primary tumor, postoperative status or other trauma | |
| 2. Pleuritis | Clinical judgment and exudation verified by x ray examination |
| Other causes improbable, such as tuberculosis or other infection, metastases, primary tumour, postoperative status or other trauma | |
| 3. Felty’s syndrome | Splenomegaly (clinically evident or measured by ultrasound) and neutropenia (<1.8×109/I) on two occasions |
| Other causes improbable, such as drug side effect or infection | |
| 4. Major cutaneous vasculitis | Diagnostic biopsy or clinical judgment by dermatologist |
| 5. Neuropathy | Clinical judgment by doctor and signs of polyneuropathy/mononeuropathy at electromyography/electroneurography |
| Other causes, such as diabetes or alcohol abuse, improbable | |
| 6. Scleritis, episcleritis or retinal vasculitis | Clinical judgment by ophthalmologist |
| 7. Glomerulonephritis | Clinical judgment by nephrologist and positive biopsy |
| 8. Vasculitis affecting other organs | Clinical judgment by organ specialist and biopsy compatible with vasculitis |
| 9. Amyloidosis | Clinical judgment and positive biopsy from affected organ |
| 10. Keratoconjunctivitis sicca | Clinical judgment |
| Positive rose-bengal staining or result of Schirmer’s test <5 mm/min | |
| 11. Xerostomia | Clinical judgment |
| Abnormal sialometry, sialography, salivary scintigraphy or salivary gland biopsy with lymphocytic infiltrate | |
| 12. Secondary sjögren’s syndrome | Two of three criteria: |
| Keratoconjunctivitis sicca (see above) | |
| Xerostomia (see above) | |
| Serological evidence: rheumatoid factor, ANA, anti-Ro (SSA), anti-La (SSB) positive, or hypergammaglobulinaemia | |
| 13. Pulmonary fibrosis | Clinical judgment and |
| Decreased vital capacity or carbon dioxide transfer factor by 15% from normal | |
| 14. Bronchiolitis obliterans organising pneumonia | Clinical judgment by pulmonologist |
| 15. Cervical myelopathy | Clinical judgment |
| Increased atlantoaxial movement—verified by x ray examination | |
| 16. Subcutaneous rheumatoid nodules | Clinical judgment |
| 17. Rheumatoid nodules in other locations | Positive biopsy |
Reproduced from [Extra-articular disease manifestations in rheumatoid arthritis: incidence trends and risk factors over 46 years, Turesson C, O'Fallon WM, Crowson CS, Gabriel SE, Matteson EL, Ann Rheum Dis. 62(8):722–7;2003] with permission from BMJ Publishing Group Ltd.
The patients were followed from RA incidence date until death, migration from Olmsted County, or 12/31/2008. The date of ExRA incidence was recorded. The date of death was noted according to the death certificate. Data on potential risk factors for ExRA were abstracted from the medical records. The risk factors included age, sex, smoking status (current and ever) at RA incidence, body mass index (BMI) at RA incidence, positivity for rheumatoid factor (RF) and/or antinuclear antibody (ANA) at any time during the follow-up, erythrocyte sedimentation rate (ESR) (highest ESR during the first year of RA and repeatedly high ESR values [≥3 ESR values of ≥60 mm/hr with a minimum interval of 30 days between 2 measurements]), joint erosions/destructive changes at any time during the follow-up, and use of antirheumatic medications (methotrexate, hydroxychloroquine, other disease-modifying anti-rheumatic drugs [DMARDs], biologic response modifiers and corticosteroids).
Statistical Methods
The cumulative incidence of ExRA, adjusted for competing risk of death, was estimated at 10 years follow-up (17, 18). These methods are similar to Kaplan-Meier method with censoring of patients who are still alive at last follow-up. However, patients who die before experiencing ExRA are appropriately accounted for to avoid the overestimation of the rate of occurrence of ExRA, which can occur if such subjects are simply censored. Incidence and mortality rates, as well as RA disease characteristics, were compared to those reported previously for the incident RA cases between 1/1/1985 and 12/31/1994 from the smaller source population (i.e. Rochester, Minnesota), followed through 12/31/2000 (13). RA disease characteristics were compared between the cohorts using chi-square and rank sum tests. Cumulative incidence rates for the two cohorts (1995–2007 and 1985–1994) were compared using methods by Gray (19). Kaplan-Meier methods were used to estimate mortality following ExRA occurrence and trends in occurrence of a second ExRA. Cox proportional hazards models were used to assess the association of ExRA with mortality, as well as to assess the association between the risk factors and development of ExRA adjusting for age, sex and calendar year of RA diagnosis. Time-dependent covariates were used to model risk factors that developed over time. Medication usage was coded as present on the day it was started and returned to absent following the stopping date. In the analysis of risk factors of second ExRA we included patients with a first ExRA and the date of the first ExRA was the index time point for the analyses.
Results
Patients’ characteristics
The study included 463 patients with incident RA in Olmsted County, Minnesota, in 1995–2007 compared to 147 patients with incident RA in Rochester, Minnesota, in 1985–1994. As expected, the mean [standard deviation] follow-up time for the 1995–2007 cohort was shorter than for the 1985–1994 cohort (6.3 [3.5] years; range 0–16 years vs 8.4 [3.8] years; range 0–14 years). Table 2 compares the characteristics of the 1995–2007 and the 1985–1994 incident RA cohorts. Age and sex were similar in both cohorts. The proportion of RF positive patients was somewhat lower in the 1995–2007 cohort vs 1985–1994 cohort, but this difference did not reach statistical significance (p=0.12). The proportion of ANA positive patients in the 1995–2007 cohort was significantly lower than in the 1985–1994 cohort (p<0.001). This is likely an artifact due to the changes in reference ranges and testing methods for the ANA testing over time. The highest ESR estimates in the 1st year after RA and the proportion of patients with repeatedly high ESR values (≥3 values ≥60 mm/hr) were significantly lower in the 1995–2007 cohort vs 1985–1994 cohort (p=0.007 and p<0.001, respectively, Table 2).
Table 2.
Characteristics of incident RA patients in the 1985–1994 cohort (Rochester, Minnesota residents) and 1995–2007 cohort (Olmsted County, containing Rochester, Minnesota residents)
| Variable | Time period of RA incidence | ||
|---|---|---|---|
| 1985–1994 (n=147) |
1995–2007 (n=463) |
p-value | |
| Age (mean ± SD), years | 57.8 ± 16.4 | 55.6 ± 15.6 | 0.14 |
| Female, n (%) | 101 (69) | 320 (69) | 0.93 |
| RF positive, ever, n (%) | 108 (73) | 306 (67) | 0.12 |
| % tested for RF, ever | 100 | 99 | |
| ANA positive, ever, n (%) | 77 (60) | 105 (26) | <0.001 |
| % tested for ANA, ever | 88 | 86 | |
| Highest ESR in the 1st year after RA incidence (mean ± SD), mm/hr | 35.3 ± 25.1 | 30.0 ± 24.7 | 0.007 |
| ESR ≥3 values ≥60 mm/hr, n (%) | 23 (16) | 28 (6) | <0.001 |
| Joint erosions/destructive changes, n (%) | |||
| - Ever | 66 (45) | 229 (49) | 0.33 |
| - In the 1st year after RA incidence | 31 (21) | 134 (29) | 0.06 |
| % radiographed, ever | 89 | 97 | |
| Cigarette smoking, n (%) | |||
| - Ever | 88 (60) | 233 (50) | 0.04 |
| - Current | 27 (18) | 80 (17) | 0.76 |
| Obesity (BMI≥30 kg/m2), n (%) | |||
| - Ever | 37 (25) | 237 (51) | <0.001 |
| - In the 1st year after RA incidence | 32 (22) | 218 (47) | <0.001 |
| Drug exposure, ever, n (%) | |||
| - Methotrexate | 75 (51) | 299 (65) | 0.003 |
| - Hydroxychloroquine | 72 (49) | 295 (65) | 0.002 |
| - Other DMARDs | 67 (46) | 104 (23) | <0.001 |
| - Biologic Response Modifiers | 3 (2) | 96 (21) | <0.001 |
| - Corticosteroids | 109 (74) | 376 (81) | 0.06 |
Abbreviations: RA = rheumatoid arthritis; SD = standard deviation; RF = rheumatoid factor; ANA = antinuclear antibody; ESR = erythrocyte sedimentation rate; BMI = body mass index DMARDs = disease modifying antirheumatic drugs
The proportions of patients with joint erosions/destructive changes detected at some time during the follow-up period were similar in both cohorts. In the 1995–2007 cohort, 29% of patients had joint erosions/destructive changes during the 1st year after RA compared to 21% of patients who were radiographed in the 1985–1994 cohort (p=0.06), which is likely due to the improved use of diagnostic imaging in recent years. The proportion of ever smokers was lower in the 1995–2007 cohort (p=0.04), but there were no differences in the proportions of current smokers between the cohorts. Obesity (BMI≥30 kg/m2) was significantly more common in 1995–2007 vs 1985–1994 cohort (p<0.001, Table 2). Finally, the proportion of patients ever treated with methotrexate, hydroxychloroquine and biologic response modifiers was significantly higher, and the proportion of patients on other DMARDs was lower in the 1995–2007 cohort vs 1985–1994 cohort (Table 2). The use of corticosteroids was 81% in 1995–2007 compared to 74% in the 1985–1994 cohort (p=0.06).
Incidence and mortality of ExRA
Among the 463 incident RA patients identified in the 1995–2007 period, 184 patients developed an incident ExRA (25 with severe ExRA) during the follow-up (Table 3). In the comparison cohort of 147 patients with incident RA in the 1985–1994 period, there were 64 patients with incident ExRA (13 with severe ExRA). The cumulative incidence for any ExRA in the 1995–2007 cohort was similar to the 1985–1994 cohort (p=0.88) with the 10-year cumulative incidence of 50.1% vs 46.2%, respectively. The incidence of severe ExRA was also similar in 1995–2007 cohort compared to the 1985–1994 cohort (p=0.51) with the 10-year cumulative incidence of 6.7% and 9.7%, respectively. When analyzing individual ExRA, the cumulative incidence of rheumatoid vasculitis (including major cutaneous vasculitis and internal organ vasculitis) was significantly lower in the 1995–2007 cohort vs 1985–1994 cohort (p=0.011). In particular, the 10-year cumulative incidence of rheumatoid vasculitis decreased from 3.6% in the 1985–1994 cohort to 0.6% in 1995–2007 cohort, although the numbers of incident cases of vasculitis were small in both cohorts. The 10-year cumulative incidence of keratoconjunctivitis sicca in the 1995–2007 cohort was 20.4% compared to 12.2% in the 1985–1994 cohort (p=0.12). There were no substantial changes in the occurrence of other individual ExRA in the 1995–2007 cohort vs 1985–1994 cohort.
Table 3.
Cumulative incidence of ExRA at 10 years follow-up after RA incidence in the 1985–1994 cohort (Rochester, Minnesota residents) and 1995–2007 cohort (Olmsted County, containing Rochester, Minnesota residents)
| Extra-articular manifestation |
Time of RA incidence | |||
|---|---|---|---|---|
| 1985–1994 (n=147) | 1995–2007 (n=463) | |||
| Number of events* |
10-year cumulative incidence (%) |
Number of events* |
10-year cumulative incidence (%) |
|
| Any ExRA | 64 | 46.2 | 184 | 50.1 |
| Severe ExRA* | 13 | 9.7 | 25 | 6.7 |
| Pericarditis | 2 | 2.0 | 8 | 2.6 |
| Pleuritis | 3 | 2.1 | 6 | 1.9 |
| Felty’s syndrome | 0 | 0 | 2 | 0.5 |
| Vasculitis** | 5 | 3.6 | 2 | 0.6 |
| Neuropathy | 2 | 1.4 | 6 | 1.2 |
| - Mononeuropathy | 0 | 0 | 2 | 0.2 |
| - Polyneuropathy | 2 | 1.4 | 4 | 1.0 |
| Scleritis | 0 | 0 | 0 | 0 |
| Episcleritis | 1 | 0.7 | 3 | 0.7 |
| Retinal vasculitis | 0 | 0 | 0 | 0 |
| Glomerulonephritis | 0 | 0 | 0 | 0 |
| Other ExRA | 60 | 43.4 | 169 | 45.7 |
| Amyloidosis | 0 | 0 | 0 | 0 |
| Keratoconjunctivitis sicca | 16 | 12.2 | 74 | 20.4 |
| Xerostomia | 1 | 0.7 | 2 | 0.5 |
| Sjögren’s syndrome | 13 | 9.7 | 38 | 9.6 |
| Pulmonary fibrosis | 9 | 6.6 | 15 | 5.0 |
| Bronchiolitis obliterans | 0 | 0 | 2 | 0.7 |
| Organizing pneumonia | 1 | 0 | 2 | 0.4 |
| Cervical myelopathy | 3 | 2.2 | 2 | 0.7 |
| Subcutaneous nodules | 43 | 31.2 | 110 | 31.2 |
| Other nodules | 0 | 0 | 1 | 0.2 |
Abbreviations: ExRA = extra-articular rheumatoid arthritis; RA = rheumatoid arthritis
Malmö criteria (5)
Includes major cutaneous and internal organ vasculitis
(dashed line) compared to patients with incident RA in 1995–2007 period (solid line)*
Similar to the 1985–1994 cohort, the most common ExRA in the 1995–2007 cohort were subcutaneous nodules, keratoconjunctivitis sicca and Sjögren’s syndrome (Table 3). There were no cases of scleritis, retinal vasculitis, glomerulonephritis or amyloidosis in either 1995–2007 cohort or 1985–1994 cohort. Occurrence of a second ExRA appeared to be similar in the two cohorts with 10-year cumulative incidence of 37.5% in the 1995–2007 cohort and 36.6% in the 1985–1994 cohort. The trends in occurrence of a second ExRA subsequent to the first ExRA were similar in both cohorts (data not shown).
In the 1995–2007 cohort, ExRA was a significant predictor of premature mortality (HR 2.1, 95% CI 1.2, 3.7) adjusting for age, sex and calendar year. Neither severe ExRA, nor the development of a second ExRA conferred a greater mortality risk than occurrence of any one ExRA (HR 2.2, 95%CI 0.8, 6.2 and HR 0.5, 95%CI 0.2, 1.5, respectively). During the follow-up 53 deaths occurred per 2,956 person-years of observation in the 1995–2007 cohort. Figure 1 shows mortality trends after occurrence of any ExRA in 1995–2007 vs 1985–1994 cohort. Survival curves diverged during the first year after ExRA occurrence and remained separated during the following years. However, the difference in mortality following ExRA in 1995–2007 cohort vs 1985–1994 cohort did not reach statistical significance (HR 0.6, 95%CI 0.3, 1.2, p=0.16). Although the mortality risk after the occurrence of severe ExRA was nearly halved in the 1995–2007 cohort compared with 1985–1994 cohort, this difference was not statistically significant (HR 0.5, 95%CI 0.1, 1.7, p=0.25).
Figure 1.
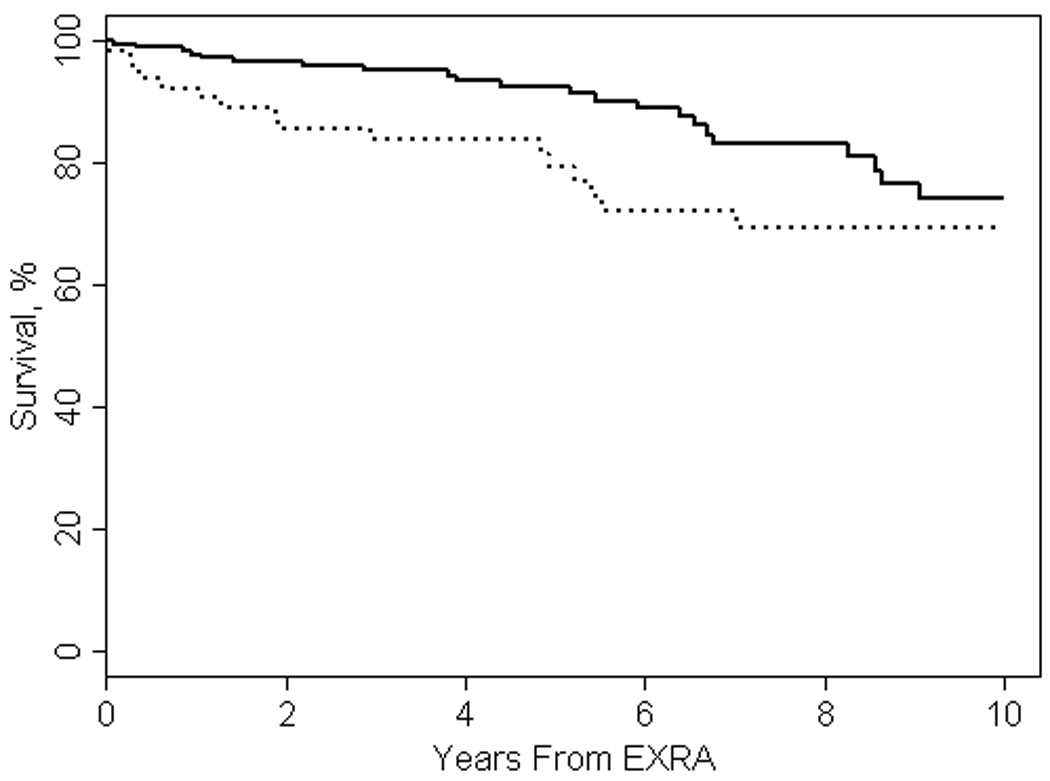
Mortality after any ExRA in patients with incident RA in 1985–1994 decade (dashed line) compared to patients with incident RA in 1995–2007 period (solid line)*
Abbreviations: ExRA = extra-articular manifestations of rheumatoid arthritis
* p=0.16 adjusted for age and sex
Risk factors associated with ExRA occurrence
To better understand the impact of ExRA occurrence in the recent years, we analyzed the associations of the potential risk factors with ExRA incidence in the 1995–2007 cohort (Table 4). RF positivity, joint erosions/destructive changes, use of methotrexate, other DMARDs and systemic corticosteroids were found to be significantly associated with the occurrence of any ExRA. Use of biologic response modifiers and systemic corticosteroids were significantly associated with severe ExRA. Male sex, current smoking, RF positivity, joint erosions/destructive changes, use of methotrexate, other DMARDs and biologic response modifiers were significantly associated with the development of rheumatoid nodules. Use of biologic response modifiers was significantly associated with the occurrence of a second ExRA (HR 2.3, 95%CI 1.1, 4.9). None of the other risk factors, nor subcutaneous nodules as the first ExRA was significantly associated with the occurrence of a second ExRA. Following the additional adjustment for characteristics of RA activity and severity (i.e. ≥3 ESR values of ≥60 mm/hr, RF positivity and presence of erosions/destructive changes on radiographs), the strength of some of these associations has slightly weakened (Table 4).
Table 4.
Risk factors associated with the occurrence of ExRA in patients with incident RA in 1995–2007 period *
| Variable | Any ExRA HR (95% CI) |
Severe ExRA HR (95% CI) |
Rheumatoid nodules HR (95% CI) |
|---|---|---|---|
| Age (per 10 year increase) | 1.0 (0.9, 1.1) | 0.9 (0.7, 1.2) | 1.0 (0.9, 1.1) |
| Male sex | 1.2 (0.8, 1.6) | 1.3 (0.5, 3.0) | 1.5 (1.01, 2.2) |
| Cigarette smoking | |||
| Ever | 0.9 (0.7, 1.3) | 1.0 (0.4, 2.3) | 1.2 (0.8, 1.8) |
| Current | 1.2 (0.8, 1.8) | 1.6 (0.6, 4.1) | 1.6 (1.01, 2.4) |
| BMI (per 1 kg/m2 increase) | 1.0 (0.98, 1.03) | 0.98 (0.9, 1.05) | 1.02 (0.99, 1.05) |
| RF positivity | 2.2 (1.5, 3.3) | 0.8 (0.3, 2.0) | 4.5 (2.4, 8.2) |
| ANA positivity | 1.3 (0.9, 1.8) | 1.7 (0.7, 4.2) | 1.0 (0.6, 1.7) |
| ESR ≥3 values ≥60 mm/hr | 1.5 (0.6, 3.7) | 3.2 (0.7, 14.9) | 1.6 (0.6, 4.5) |
| Highest ESR in the 1st year of RA (per 10 mm/hr increase) | 1.0 (0.96, 1.1) | 1.1 (0.9, 1.2) | 1.0 (0.97, 1.1) |
| Erosions/destructive changes on radiographs | 2.2 (1.6, 3.0) | 1.1 (0.5, 2.7) | 2.8 (1.9, 4.2) |
| Medication usage | |||
| Methotrexate | 1.5 (1.1, 2.2) | 0.7 (0.3, 1.9) | 1.9 (1.3, 2.9) |
| Hydroxychloroquine | 1.3 (0.93, 1.8) | 0.6 (0.2, 1.7) | 1.2 (0.8, 1.9) |
| Other DMARDs | 2.6 (1.6, 4.3) | 1.9 (0.5, 6.8) | 2.5 (1.4, 4.6) |
| Biologic Response Modifiers | 1.9 (0.99, 3.5) | 4.0 (1.4, 11.5) | 2.2 (1.1, 4.4) |
| Corticosteroids (systemic) | 1.4 (1.03, 2.0) | 3.8 (1.5, 10.0) | 1.1 (0.8, 1.7) |
| Medication usage, adjusted# | |||
| Methotrexate | 1.2 (0.9, 1.8) | 0.6 (0.2, 1.7) | 1.5 (0.98, 2.3) |
| Hydroxychloroquine | 1.3 (0.94, 1.9) | 0.8 (0.3, 2.3) | 1.3 (0.8, 2.0) |
| Other DMARDs | 2.4 (1.4, 4.0) | 1.5 (0.4, 5.5) | 2.4 (1.3, 4.5) |
| Biologic Response Modifiers | 1.6 (0.8, 3.1) | 3.1 (1.04, 9.5) | 1.8 (0.9, 3.7) |
| Corticosteroids (systemic) | 1.3 (0.96, 1.9) | 3.5 (1.3, 9.1) | 1.1 (0.7, 1.6) |
Abbreviations: ExRA = extra-articular rheumatoid arthritis; HR = hazard ratio; BMI = body mass index; RF = rheumatoid factor; ANA = antinuclear antibody; ESR = erythrocyte sedimentation rate; DMARDs = disease modifying antirheumatic drugs
Statistically significant associations (p<0.05) are shown in bold
Adjusted for ESR ≥3 values ≥60 mm/hr, RF positivity, erosions/destructive changes on radiographs
Discussion
Systemic disease has a major impact on morbidity and mortality in RA. This retrospective population-based cohort study reveals that the occurrence of ExRA overall (including any ExRA and severe ExRA) in the 1995–2007 RA incidence cohort was similar to that in the 1985–1994 RA incidence cohort from the same population base. While not significantly affecting the major trends in the ExRA incidence, the pattern of occurrence of some individual ExRA (in particular, rheumatoid vasculitis and perhaps keratoconjunctivitis sicca) appears to be changing. Concordantly, the decline in the occurrence of rheumatoid vasculitis after 1995 has been described in some clinic-based studies (20, 21). The decline in prevalence of serious ExRA including vasculitis from 1985 to 2006 among US veterans also suggests evolving changes in epidemiology of some ExRA (22).
The use of more aggressive treatment strategies including increased usage of DMARDs and biologic response modifiers in the 1995–2007 cohort vs 1985–1994 cohort may be one of the possible factors affecting the pattern of ExRA occurrence in the recent years. While the use of advanced therapeutic regimens could have an impact on decrease in the occurrence of some ExRA (e. g. rheumatoid vasculitis) (23, 24), these benefits could be counteracted by increased risk of other ExRA events (e.g. rheumatoid lung disease and rheumatoid nodules) associated with antirheumatic medication use (26–28). In fact, the use of some antirheumatic medications was associated with increased risk of ExRA occurrence in our study and these associations were only slightly attenuated after the adjustment for available characteristics of RA activity and severity (i.e. repeatedly high ESR, RF positivity and presence of erosions/destructive changes). These potential risks of antirheumatic therapies with respect to ExRA occurrence may therefore, to some extent, explain the lack of significant decrease in the occurrence of certain ExRA in the era of improved therapeutics.
Non-treatment related changes in RA disease pattern towards milder disease in recent decades could also influence the occurrence of ExRA, particularly, the decrease in vasculitis (5, 8–11). Concordantly, we found significantly lower ESR and marginally lower proportion of RF positive patients in 1995–2007 vs 1985–1994 cohort. However, despite a suggested decrease in RA activity over time, the presence of active and severe RA in our study (i.e. RF positivity and joint erosions/destructive changes) continued to confer a significantly increased risk for ExRA similar to the earlier years (25, 26). This suggests that patients with advanced RF positive RA remain a high risk group for the development of ExRA, thus contributing to the sustained ExRA burden.
Increased awareness of ExRA and improved diagnostic techniques can be another potential explanation for the lack of decline in the occurrence of some ExRA and suggested marginal increase in keratoconjunctivitis sicca in 1995–2007 vs 1985–1994 cohort. The impact of some other factors, including environmental factors (e.g. smoking, obesity) cannot be excluded. There was a significant decrease in the obesity rates in the 1995–2007 vs 1985–1994 cohort, but BMI did not appear to have substantial impact on the occurrence of ExRA in our study. The impact of smoking may be greater on the occurrence of some manifestations (e.g. rheumatoid nodules) than on other subsets of ExRA. Similarly to previous decades, during the 1995–2007 period ExRA conferred a significant mortality risk (4). However, in contrast to our previous findings (7), the mortality risk associated with severe ExRA and second ExRA was not greater than that of any ExRA. The decrease in the occurrence of vasculitis which has been previously shown to have a major impact on mortality and better control of RA activity in the recent years may be a possible explanation (7, 27). While not reaching statistical significance, an estimated 40% decrease in mortality risk after any ExRA and 50% decrease in mortality risk after severe ExRA in 1995–2007 cohort vs 1985–1994 may be promising for the improvement in survival following ExRA in the future. However, this requires further study.
The major strengths of the study include its community-based longitudinal design, use of comprehensive medical records linkage system with complete medical information for each patient, and the use of the same classification for ExRA as in our previous studies. The comparative analysis of characteristics of successive incidence RA cohorts from the same underlying population further strengthens the results.
This study has several potential limitations. The retrospective design of the study necessarily means that only information available from medical records was considered and that only those events that came to the medical attention were included in the study. However, availability of complete medical records from all health care providers in the area is likely to minimize this weakness, as these REP records include the vast majority of clinically important ExRA events. Inherent to retrospective data is the possibility of misclassification of ExRA manifestations. However, independent review of the cases by several investigators-rheumatologists based on the uniform case definition and structured protocol consistently used in the number of our studies likely reduced misinterpretation of the data.
Mean follow-up was 6.3 years in the 1995–2007 cohort compared to 8.4 years in the 1985–1994 cohort. This difference is unlikely to have an impact on the analysis because our methods appropriately account for the length of follow-up. There is a possibility of limited power due to the small number of some specific ExRA events and limited number of deaths. The number of RA patients in the 1995–2007 cohort was significantly higher than in 1985–1994 cohort. This is because, in contrast to the previous studies enrolling the residents of the city of Rochester, this study involved RA patients from a larger geographical area (i.e. Olmsted County) which includes the city of Rochester. In fact, there have been no dramatic changes in RA incidence in Olmsted County during the study period. Rather, there was a modest increase in RA incidence among women in the recent years (28), which is unlikely to have a major impact on the results of this study. In an observational study of this kind it is difficult to draw firm conclusions about any causal relationships between potential risk factors (particularly, antirheumatic treatment) and ExRA occurrence. Thus, further efforts including prospective controlled studies are needed to explore the nature of the associations between drug exposure and ExRA. Finally, we cannot exclude the possibility that more attention has been paid in the clinic to recording ExRA in recent years, but have no data to support this.
In conclusion, in the 1995–2007 incidence RA cohort, ExRA were common and the incidence of ExRA remained similar to that of the 1985–1994 RA incidence RA cohort. However, there was a significant decrease in the incidence of vasculitis suggesting that the pattern of occurrence of some individual ExRA may be changing in the recent years. Markers of RA severity and drug exposure were significantly associated with ExRA development during the recent years. ExRA remain a significant predictor of mortality. Improved control of RA and milder disease in recent years may contribute to the decrease in the incidence of vasculitis, while some other factors, including some disease and treatment-related effects, may counteract these beneficial changes contributing to the lack of improvement in the overall occurrence and mortality of ExRA. Given the possibility of confounding by indication in this observational study, the assessment of the full impact of treatment on disease expression is a matter of ongoing research.
Acknowledgments
Funding Source: This work was funded by a grant from the National Institutes of Health, NIAMS (R01 AR46849) and made possible the Rochester Epidemiology Project (R01 AG034676 from the National Institute on Aging).
Footnotes
Financial Disclosures: None
References
- 1.Calguneri M, Ureten K, Akif Ozturk M, Onat AM, Ertenli I, Kiraz S, et al. Extra-articular manifestations of rheumatoid arthritis: results of a university hospital of 526 patients in Turkey. Clin Exp Rheumatol. 2006;24:305–308. [PubMed] [Google Scholar]
- 2.Carmona L, Gonzalez-Alvaro I, Balsa A, Angel Belmonte M, Tena X, Sanmarti R. Rheumatoid arthritis in Spain: occurrence of extra-articular manifestations and estimates of disease severity. Ann Rheum Dis. 2003;62:897–900. doi: 10.1136/ard.62.9.897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cimmino MA, Salvarani C, Macchioni P, Montecucco C, Fossaluzza V, Mascia MT, et al. Extra-articular manifestations in 587 Italian patients with rheumatoid arthritis. Rheumatol Int. 2000;19:213–217. doi: 10.1007/pl00006853. [DOI] [PubMed] [Google Scholar]
- 4.Gabriel SE, Crowson CS, Kremers HM, Doran MF, Turesson C, O'Fallon WM, et al. Survival in rheumatoid arthritis: a population-based analysis of trends over 40 years. Arthritis Rheum. 2003;48:54–58. doi: 10.1002/art.10705. [DOI] [PubMed] [Google Scholar]
- 5.Turesson C, Jacobsson L, Bergstrom U. Extra-articular rheumatoid arthritis: prevalence and mortality. Rheumatology (Oxford) 1999;38:668–674. doi: 10.1093/rheumatology/38.7.668. [DOI] [PubMed] [Google Scholar]
- 6.Turesson C, Jarenros A, Jacobsson L. Increased incidence of cardiovascular disease in patients with rheumatoid arthritis: results from a community based study. Ann Rheum Dis. 2004;63:952–955. doi: 10.1136/ard.2003.018101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Turesson C, O'Fallon WM, Crowson CS, Gabriel SE, Matteson EL. Occurrence of extraarticular disease manifestations is associated with excess mortality in a community based cohort of patients with rheumatoid arthritis. J Rheumatol. 2002;29:62–67. [PubMed] [Google Scholar]
- 8.Odegard S, Kvien TK, Uhlig T. Incidence of clinically important 10-year health status and disease activity levels in population-based cohorts with rheumatoid arthritis. J Rheumatol. 2008;35:54–60. [PubMed] [Google Scholar]
- 9.Pincus T, Sokka T, Kautiainen H. Patients seen for standard rheumatoid arthritis care have significantly better articular, radiographic, laboratory, and functional status in 2000 than in 1985. Arthritis Rheum. 2005;52:1009–1019. doi: 10.1002/art.20941. [DOI] [PubMed] [Google Scholar]
- 10.Tanaka E, Mannalithara A, Inoue E, Hara M, Tomatsu T, Kamatani N, et al. Efficient management of rheumatoid arthritis significantly reduces long-term functional disability. Ann Rheum Dis. 2008;67:1153–1158. doi: 10.1136/ard.2007.072751. [DOI] [PubMed] [Google Scholar]
- 11.Welsing PM, Fransen J, van Riel PL. Is the disease course of rheumatoid arthritis becoming milder? Time trends since 1985 in an inception cohort of early rheumatoid arthritis. Arthritis Rheum. 2005;52:2616–2624. doi: 10.1002/art.21259. [DOI] [PubMed] [Google Scholar]
- 12.Turesson C, McClelland RL, Christianson TJ, Matteson EL. No decrease over time in the incidence of vasculitis or other extraarticular manifestations in rheumatoid arthritis: results from a community-based study. Arthritis Rheum. 2004;50:3729–3731. doi: 10.1002/art.20590. [DOI] [PubMed] [Google Scholar]
- 13.Turesson C, O'Fallon WM, Crowson CS, Gabriel SE, Matteson EL. Extra-articular disease manifestations in rheumatoid arthritis: incidence trends and risk factors over 46 years. Ann Rheum Dis. 2003;62:722–727. doi: 10.1136/ard.62.8.722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Maradit Kremers H, Crowson CS, Gabriel SE. Rochester Epidemiology Project: a unique resource for research in the rheumatic diseases. Rheum Dis Clin North Am. 2004;30:819–834. doi: 10.1016/j.rdc.2004.07.010. [DOI] [PubMed] [Google Scholar]
- 15.Melton LJ., 3rd History of the Rochester Epidemiology Project. Mayo Clin Proc. 1996;71:266–274. doi: 10.4065/71.3.266. [DOI] [PubMed] [Google Scholar]
- 16.Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988;31:315–324. doi: 10.1002/art.1780310302. [DOI] [PubMed] [Google Scholar]
- 17.Aalen O. Nonparametric estimation of partial transition probabilities in multiple decrement models. Ann Stat. 1978;6:534–535. [Google Scholar]
- 18.Kalbfleisch JGPR. Statistical analysis of failure time data. New York: 2002. [Google Scholar]
- 19.Gray R. A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat. 1988;16:1141–1154. [Google Scholar]
- 20.Ward MM. Decreases in rates of hospitalizations for manifestations of severe rheumatoid arthritis, 1983–2001. Arthritis Rheum. 2004;50:1122–1131. doi: 10.1002/art.20158. [DOI] [PubMed] [Google Scholar]
- 21.Watts RA, Mooney J, Lane SE, Scott DG. Rheumatoid vasculitis: becoming extinct? Rheumatology (Oxford) 2004;43:920–923. doi: 10.1093/rheumatology/keh210. [DOI] [PubMed] [Google Scholar]
- 22.Bartels CM, Bell CL, Rosenthal A, Bridges AJ. Changing trends in serious extra-articular manifestations of rheumatoid arthritis among United State veterans over 20 years. Rheumatology (Oxford) 2010;49:1670–1675. doi: 10.1093/rheumatology/keq135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hellmann M, Jung N, Owczarczyk K, Hallek M, Rubbert A. Successful treatment of rheumatoid vasculitis-associated cutaneous ulcers using rituximab in two patients with rheumatoid arthritis. Rheumatology (Oxford) 2008;47:929–930. doi: 10.1093/rheumatology/ken129. [DOI] [PubMed] [Google Scholar]
- 24.Puechal X, Miceli-Richard C, Mejjad O, Lafforgue P, Marcelli C, Solau-Gervais E, et al. Anti-tumour necrosis factor treatment in patients with refractory systemic vasculitis associated with rheumatoid arthritis. Ann Rheum Dis. 2008;67:880–884. doi: 10.1136/ard.2007.081679. [DOI] [PubMed] [Google Scholar]
- 25.Nyhall-Wahlin BM, Petersson IF, Nilsson JA, Jacobsson LT, Turesson C. High disease activity disability burden and smoking predict severe extra-articular manifestations in early rheumatoid arthritis. Rheumatology (Oxford) 2009;48:416–420. doi: 10.1093/rheumatology/kep004. [DOI] [PubMed] [Google Scholar]
- 26.Turesson C, Eberhardt K, Jacobsson LT, Lindqvist E. Incidence and predictors of severe extra-articular disease manifestations in an early rheumatoid arthritis inception cohort. Ann Rheum Dis. 2007;66:1543–1544. doi: 10.1136/ard.2007.076521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Erhardt CC, Mumford PA, Venables PJ, Maini RN. Factors predicting a poor life prognosis in rheumatoid arthritis: an eight year prospective study. Ann Rheum Dis. 1989;48:7–13. doi: 10.1136/ard.48.1.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Myasoedova E, Crowson CS, Kremers HM, Therneau TM, Gabriel SE. Is the incidence of rheumatoid arthritis rising?: results from Olmsted County, Minnesota, 1955–2007. Arthritis Rheum. 2010;62:1576–1582. doi: 10.1002/art.27425. [DOI] [PMC free article] [PubMed] [Google Scholar]


