Abstract
Estrogen is a key regulator of vascular responses and angioadaptation in multiple organs and tissues, including brain. However, the consequences of a loss of ovarian steroid hormone secretion on the status of microvascular networks in brain and meninges are largely unknown. Here, using the perfused dura mater model coupled with high-resolution digital epifluorescence and laser scanning confocal microscopy and computer-assisted morphometric analysis, we demonstrate that cessation of ovarian hormone production causes dramatic vascular remodeling in meningeal microvascular networks characterized by a threefold decrease in microvessel density and capillary rarefaction and an almost fourfold increase in vascular permeability. These changes were accompanied by a significant decrease in angiopoietin-1 (Ang-1) expression and Ang-1/Tie-2 ratio (1.4-fold, P < 0.01, and 1.5-fold, P < 0.05, respectively) in ovariectomized animals compared with intact females, but no changes were detected in the expression of estrogen receptors (ER)-α and -β. We conclude that estrogen-dependent control of Ang-1 expression plays an important role in stabilizing meningeal microvessel and maintaining healthy microvascular networks.
Keywords: hormones, microcirculation, imaging
Throughout Life, Vascular Networks in all organs adjust continuously to ever-changing functional needs inflicted by multiple physiological and pathological processes (5, 11, 22). Such angioadaptation, destined to maintain functional and structural integrity of the body's vascular system, is achieved by means of remodeling blood vessel walls, elimination of redundant microvessel segments (pruning), and formation of new microvessels (angiogenesis). All of these processes are tightly regulated and kept in balance by preexisting vascular architecture, hemodynamic forces exerted by the flowing blood, metabolic status of the tissue, and numerous cytokines, growth factors, and hormones. Among the latter, estrogens, particularly estradiol (E2), play a fundamental role in governing angioadaptation in an array of organs and tissues (5, 11). In reproductive organs, physiological fluctuations in estrogen levels control vascular remodeling related to the normal menstrual cycle and pregnancy (1, 2). In nonreproductive organs, E2 regulates, via genomic and nongenomic mechanisms, immediate vascular responses, vessel wall remodeling (7, 11, 12), and tumor-associated angiogenesis (19). Epidemiological evidence suggests strongly that development of multiple peri- and postmenopausal symptoms (6), as well as increased risk of stroke (13) in postmenopausal women, could be associated with the loss of E2-related angioprotection brought about by a termination of ovarian steroid hormone secretion. However, the consequences of the cessation of a gonadal hormone production upon the status of microvascular networks in brain and meninges are largely unknown. Because E2 regulates multiple proteins associated with angioadaptation and vascular maintenance, including angiopoietin-1 (Ang-1), involved directly in the control of vascular stability (3), we hypothesized that the loss of ovarian E2 production could result in destabilization and subsequent remodeling of meningeal microvasculature. In this study, we investigated changes in meningeal (dura mater) microvascular networks following bilateral ovariectomy (OVX) in Yucatan miniature pigs, determined the extent of post-OVX vascular remodeling, and identified major estrogen-dependent pathways controlling the stability and post-OVX adaptation of dura microvasculature.
Methods
Animals
Mature 9- to 12-mo-old female Yucatan miniature swine were used in this study in accordance with the University of Missouri Animal Care and Use Committee-approved protocol. Bilateral OVX was performed under general anesthesia by a team of surgeons at the National Center for Gender Physiology Swine Hormone Core. Animals were killed at indicated time points, and dura mater was acquired and processed as described previously (9, 10). The body mass at death was 38.9 ± 2.2 kg (n = 9) and 35.5 ± 3.9 kg (n = 5) for intact female (IF) and OVX animals, respectively.
Imaging
Alexa Fluor 488-conjugated soybean agglutinin (SBA) at 30μg/ml (final concentration) was used for visualizing microvasculature, as described previously (9, 10). Briefly, the dura mater corresponding to one hemisphere was collected within 15–30 min after the animal's death and placed immediately on ice in a porcine Krebs solution (PKS; Krebs physiological salt supplemented with 1.0 mg/ml porcine albumin). Special attention was paid to minimize dura mater damage during its separation from the skull. The collected dura was rinsed twice with ice-cold PKS and transferred to a refrigerated tissue dissection dish filled with PKS and connected to a circulating bath to maintain the sample temperature from 0 to 4°C during the dissection procedure. Next, the dura was dissected and trimmed under control of a low-power SMZ-2B stereomicroscope (Nikon) to remove arachnoid granulations and release the dura's folds to flatten the tissue onto a Sylgard-coated 100-mm tissue culture dish. During the trimming, particular care was taken not to damage the major blood vessels detectable under stereomicroscope. Next, an ∼5-mm-long segment of the median meningeal artery was separated carefully from surrounding tissues, the sample was transferred onto the fluorescent microscope stage, and the artery was cannulated using a pipette manufactured from a 1-mm outer diameter borosilicate glass capillary with a 90-μm opening at the tip.
To remove blood, the dura mater vasculature was perfused for 20 min with PKS using a precision syringe infusion/withdrawal pump KDS210 (KD Scientific) at a physiological rate (15 μl/min). To visualize the perfused vascular tree, microvessels were perfused for an additional 30 min with PKS containing SBA lectin (30μg/ml) conjugated with Alexa Fluor 488, followed by 30-min perfusion with PKS to remove unbound dye before imaging. Terminal dura mater microvascular networks were imaged first using a fluorescence video microscopy system [Laborlux 8 microscope (Leitz Wetzlar) equipped with 75-watt xenon lamp and QICAM high-performance digital CCD camera (Quantitative Imaging, Burnaby, BC, Canada)] with controlled gain, offset, and exposure time and then a confocal system [IX-70 Olympus Microscope equipped with Solamere confocal unit with dual laser line capabilities (50 mW, used for imaging at 488 and 514 nm; and 10 mW, used for imaging at 457 nm) and acoustic coupler].
Computer-assisted morphometric analysis
The collected images were analyzed using the following software: ImageJ version 1.33u (National Institutes of Health, Bethesda, MD), Adobe Photoshop 7 (Adobe Systems, San Jose, CA), and Fovea Pro Photoshop-compatible plug-ins (Reindeer Graphics, Asheville, NC). Fovea Pro stereology tools were used to analyze microvessel volume and surface-to-volume ratio.
Analysis of changes in microvascular permeability
The changes in microvessel permeability were analyzed from fluorescent dye accumulation >10 μm in diameter circular regions of interest (ROI) located in a perivascular space immediately adjacent to the microvessels (≤10 μm in diameter). A total of 90 measurements were performed for each experimental group (10 ROI/frame, 3 frames/animal, 3 animals/group). For these purposes, fluorescent photomicrographs, acquired using the same camera settings (gain, offset, and exposure time) after 30-min perfusion of the microvasculature with 30 μg/ml solution of Alexa Fluor 488-conjugated SBA lectin in PKS followed by a 30-min wash with PKS alone, were analyzed off-line. Finally, the dye accumulation index was derived for each frame analyzed from the mean fluorescence intensity of each ROI divided by the background (the mean fluorescent intensity of the same size ROI located over the darkest unstained area of the frame).
Western blot analysis
The whole tissue lysates prepared from dura mater of IF and OVX animals (30 μg of total protein/lane) were resolved on NuPAGE 4–12% Bis-Tris gels (Invitrogen, Carlsbad, CA), transferred to nitrocellulose membranes, and analyzed using rabbit anti-estrogen receptor (ER)-α (PA1–309; Affinity BioReagents, Golden, CO), rabbit anti-human ER-β (H-150), goat anti-Ang-1 (sc-6320), goat anti-Ang-2 (sc-7015), and rabbit anti-Tie-2 (sc-324) (all Santa Cruz Biotechnology, Santa Cruz, CA) in conjunction with corresponding HRP-labeled secondary antibodies and enhanced chemiluminescence (ECL Plus) detection system (Amersham, UK). Equal loading and transfer was controlled by Ponceau S staining. Subsequently, each membrane was striped and reprobed with an anti-β-actin loading control (Abcam, Cambridge, MA), which was used for normalizing relative expression of target proteins. The whole tissue lysate prepared from the ovaries of IF animals was used as a positive control as necessary. In all Western blot experiments, the samples from six IF and five OVX pigs were analyzed.
Immunohistochemistry and capillary count
Rabbit polyclonal anti-human ER-α (Affinity BioReagents), anti-human ER-β, (Santa Cruz Biotechnology), and anti-human von Willebrand factor (Dako, Denmark) antibodies were used for immunohistochemical analysis of formaldehyde-fixed, paraffin embedded serial sections of porcine dura mater in conjunction with corresponding HRP-labeled secondary antibodies. For a capillary count, circular structures ≤10 μm in diameter stained positively for von Willebrand factor were scored in five to 10 random fields (∼11 × 104 μm2 area) for each sample. The results were calculated and presented as means ± SE.
Statistical analysis
The statistical analysis of data was performed using GraphPad Prism version 4 software (GraphPad Software, San Diego, CA). Two-tailed t-test was used to assess statistical significance of data. Box diagrams showing statistical distribution of data in Fig. 1, E–G, represent minimum value, 25th percentile, median, 75th percentile, and maximum value for each set of data. Bar graphs represent means ± SE.
Fig. 1.
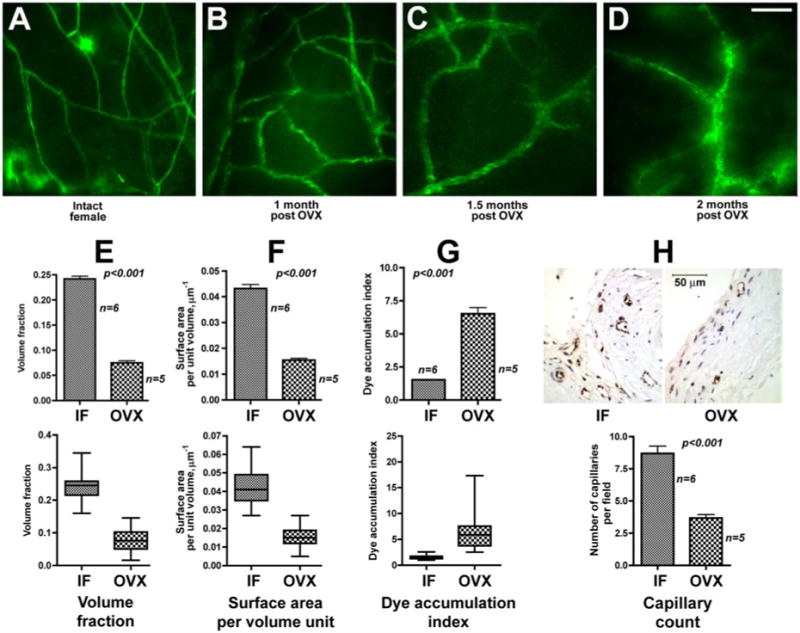
Gradual remodeling of the terminal dura mater microvascular networks in pigs following ovariectomy (OVX). A–D: microvascular networks of intact female (IF; A) and OVX animals 1 mo (B), 1.5 mo (C), and 2 mo (D) post-OVX. Note the apparent loss of capillaries, increase in average microvessel size, and enhanced dye accumulation in perivascular space with time following OVX. Scale bar shown in D, 100 μm. E–G: response to OVX on a tissue volume occupied by terminal microvascular networks (E), blood vessel surface-to-volume ratio (F), and microvessel permeability (G). Bar graphs (E–G, top) show means ± SE, and box diagrams (E–G, bottom) show statistical distribution of data as lowest value, 25th percentile, median, 75th percentile, and highest value. H: immunohistochemical analysis of capillary density using antibody directed against endothelium-specific von Willebrand factor. Scale bar, 50 μm. Bar graph, means ± SE.
Results and Discussion
In this study, we used the perfused dura mater model (9, 10) coupled with high-resolution digital epifluorescence and laser scanning confocal microscopy to investigate the status of terminal microvascular networks in dura mater of intact and ovariectomized miniature pigs. Our results show that within 2 mo post-OVX, terminal microvascular networks in meninges of ovariectomized animals undergo gradual remodeling characterized by a dramatic decrease in microvessel density, capillary rarefaction, and an increase in average microvessel diameter (Fig. 1, A–D). Moreover, the augmented fluorescent dye accumulation in the perivascular space immediately adjacent to the microvessels indicates that this process alters functional properties of terminal microvessels related to permeability and water-solute exchange (Fig. 1D).
Computer-assisted morphometric analysis (Fig. 1, E–G) revealed a greater than threefold decrease (P < 0.001) in a tissue volume occupied by terminal microvascular networks in ovariectomized animals compared with the control (Fig. 1E). A significant (almost 3-fold) decrease in the calculated blood vessel surface-to-volume ratio (Fig. 1F) in OVX swine (P < 0.001) indicates that the observed overall decrease in terminal dura mater microvessel density was due to a prevalent loss of capillaries and accompanied by an apparent increase in the average microvessel size. Furthermore, there was a greater than fourfold increase (P < 0.001) in perivascular fluorescent dye accumulation in OVX pigs compared with IFs (Fig. 1G), indicative of changes in vascular permeability. The prevalent loss of capillaries, detected by epifluorescence and laser scanning confocal microscopy in dura mater of post-OVX animals compared with IFs, was further confirmed immunohistochemically using antibody directed against endothelium-specific von Willebrand factor (Fig. 1H).
The observed changes in microvascular architecture and function apparently occurred as a consequence of the cessation of a gonadal steroid hormone production. Thus, we investigated next how OVX affected plasma levels of E2 as well as the expression of ER-α and -β in porcine dura mater. In our experiments, estrous and diestrous E2 plasma levels in IFs were 43.79 ± 6.24 and 8.05 ± 1.04 pg/ml, respectively (Fig. 2A). Of note, E2 plasma levels in post-OVX animals decreased significantly (2.7- and 6.4-fold, respectively) compared with overall IF animals and estrous E2 levels in normal cycling swine (Fig. 2A). However, compared with diestrous E2 levels in intact animals (Fig. 2A), post-OVX E2 levels showed only a marginal (1.2-fold) decrease from 8.05 ± 0.93 pg/ml to 6.89 ± 0.84 pg/ml, suggesting that diestrous E2 levels could be maintained largely by extragonadal E2 production.
Fig. 2.
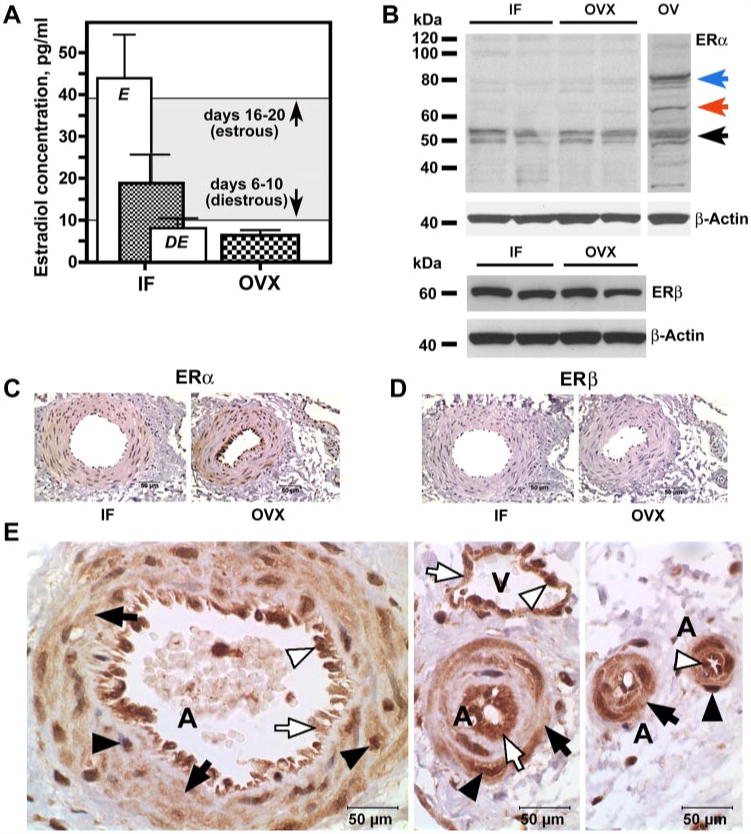
Changes in estradiol (E2) plasma levels and estrogen receptor (ER) expression following OVX. A: plasma levels of E2 in IF (n = 9) and OVX (n = 5) pigs. In IF animals, in addition to the overall E2 level (hatched bar), estrous (E; n = 3) and diestrous (DE; n = 6) E2 levels are shown (open bars). The gray area indicates the magnitude of physiological fluctuations of plasma E2 levels as per the literature. B: Western blot analysis of ER-α and ER-β expression in dura mater of IF and OVX Yucatan miniature pigs 2 mo post-OVX. In porcine ovary extract (OV), anti-ER-α antibody recognizes a full-length (∼66 kDa) receptor (red arrow) as well as higher (∼80 kDa) molecular weight (blue arrow) and truncated (∼50–52 kDa; black arrow) species. In dura mater samples (IF and OVX), however, truncated (∼50–52 kDa) ER-α-immunoreactive bands are clearly present, whereas ∼66 and ∼80 kDa are barely detectable. In contrast, the anti-ER-β antibody recognizes a single ∼59-kDa immu-noreactive band corresponding to a full-length receptor. C–E: immunohistochemical analysis of ER-α (C and E) and ER-β (D) expression in dura mater of IF and OVX Yucatan miniature pigs 2 mo post-OVX. The brown color represents ER-α (C and E) and ER-β (D) immunoreactivity. In E, note cytoplasmic (arrows) and nuclear (arrowheads) expression of ER-α in both vascular smooth muscle (black arrows and black arrowheads) and endothelial (white arrows and white arrowheads) cells in arteries (A) and veins (V) of various diameters. Scale bars, 50 μm.
Western blot analysis of dura mater whole tissue lysates revealed no difference in total levels of expression of either ER-α or ER-β between IF and ovariectomized animals (Fig. 2B). This observation is somewhat in contrast with the results published previously by Stirone et al. (21). In a related study, they reported an ∼1.5-fold decrease in ER-α expression in rat brain vessels following OVX. At this point, it is not clear whether this contradiction is due to species-related (pigs versus rats) or tissue-specific (dura mater versus pia mater and brain) differences in ovarian-dependent regulation of ER-α expression. Of note, Stirone et al. (21) detected multiple ER-α immunoreactive bands of ∼50, 82, 93, and 110kDa in the lysates of rat cerebral blood vessels, whereas a detectability of the ∼66-kDa band, corresponding to a full-length receptor, was characterized as “little to none.” Similarly, in porcine dura mater lysates, we were unable to detect a 66-kDa ER-α band. In our experiments, we detected mostly two ER-α immunoreactive bands of ∼50 and 52kDa, consistent with the existence of alternatively spliced ER-α isoforms (8,16,21). In porcine ovary extracts, however, the anti-ER-α antibody recognized a full-length ∼66kDa receptor (Fig. 2B) as well as higher-molecular-weight (∼80kDa) species, which have been identified previously as phosphorylated ER-α, and lower-molecular-weight (∼50–52 kDa) isoforms described previously in rat brain microvasculature (21). The latter (∼50–52kDa) bands were the only ER-a-immunoreactive species expressed strongly in dura mater extracts (Fig. 2B), whereas ∼66- and ∼80kDa bands were barely detectable.
Immunohistochemically, ER-α (but not ER-β) expression in porcine dura appeared to be restricted mostly to the vascular tissue (Fig. 2, C and D). Strong ER-α immunoreactivity was observed in the cytoplasm and the nuclei of both vascular smooth muscle cells (VSMC) and endothelial cells (EC) of arteries and veins of all sizes (Fig. 2E). Such a prominent expression of ER-α in the dura mater microvasculature undoubtedly suggests that E2 plays an important role in meningeal vascularity.
Estrogen acts upon vascular tissue through multiple genomic and nongenomic mechanisms (20). One aspect of E2 vascular function, however, could be of particular importance in regulating vascular stability and overall microvessel maintenance. Recently, using a rat model of stroke, Ardelt et al. (3) demonstrated that E2, but not hypoxia, positively regulates Ang-1 expression in vascular tissue via ER-α. Ang-1 binding to its receptor Tie-2, resulting in a subsequent Tie-2 autophosphorylation, promotes EC interactions with ECM and stabilizes quiescent microvessels. In contrast, Ang-2 competitive binding to Tie-2 cancels the stabilizing effects of Ang-1 and causes overall microvessel destabilization. Such destabilized vessels may undergo regression in the absence of angiogenic stimuli or angiogenesis when angiogenic signals are present (22).
Therefore, we analyzed next the expression of Ang-1, Ang-2, and Tie-2 in dura mater of IF and ovariectomized animals. Of note, we were unable to detect Ang-2 protein by Western blot analysis in dura mater lysates of either intact or ovariectomized swine. However, there was a marked (1.4-fold, P < 0.01) decrease in Ang-1 expression in dura mater of ovariectomized animals compared with IFs (Fig. 3, A and C), whereas Tie-2 expression remained unchanged (Fig. 3, B and D). Because of that, there was a significant (1.5-fold, P < 0.05) reduction in the Ang-1-to-Tie-2 ratio in dura mater of post-OVX pigs compared with control animals (Fig. 3E). The immunohistochemical analysis confirmed further our Western blot results showing an apparent reduction in Ang-1 expression in the vasculature of post-OVX animals compared with IF (Fig. 3F). The Ang-1 immunoreactivity was confined to VSMC in both IF and post-OVX pigs (Fig. 3F). However, it was markedly reduced in post-OVX swine (Fig. 3F). The expression of the Ang-1 receptor Tie-2 was also restricted to the vascular tissue (Fig. 3G). However, unlike Ang-1, Tie-2 was expressed in both VSMC and EC, and there was no difference in the levels of Tie-2 immunoreactivity between the IF and OVX groups (Fig. 3G). Because Ang-1 binding to Tie-2 is essential for Tie-2 vessel-stabilizing signaling, such a reduction in Ang-1 to Tie-2 ratio would inevitably result in destabilization of blood vessels in post-OVX animals. Apparently, even 2 mo post-OVX, meningeal microvessels remain destabilized and therefore susceptible to remodeling stimuli. Furthermore, because Ang-1 reduces vascular leakage by restricting the number and size of gaps that form at EC junctions (4), a decrease in Ang-1 expression may account at least in part for the marked increase in microvessel permeability following OVX (Fig. 1, D and G).
Fig. 3.
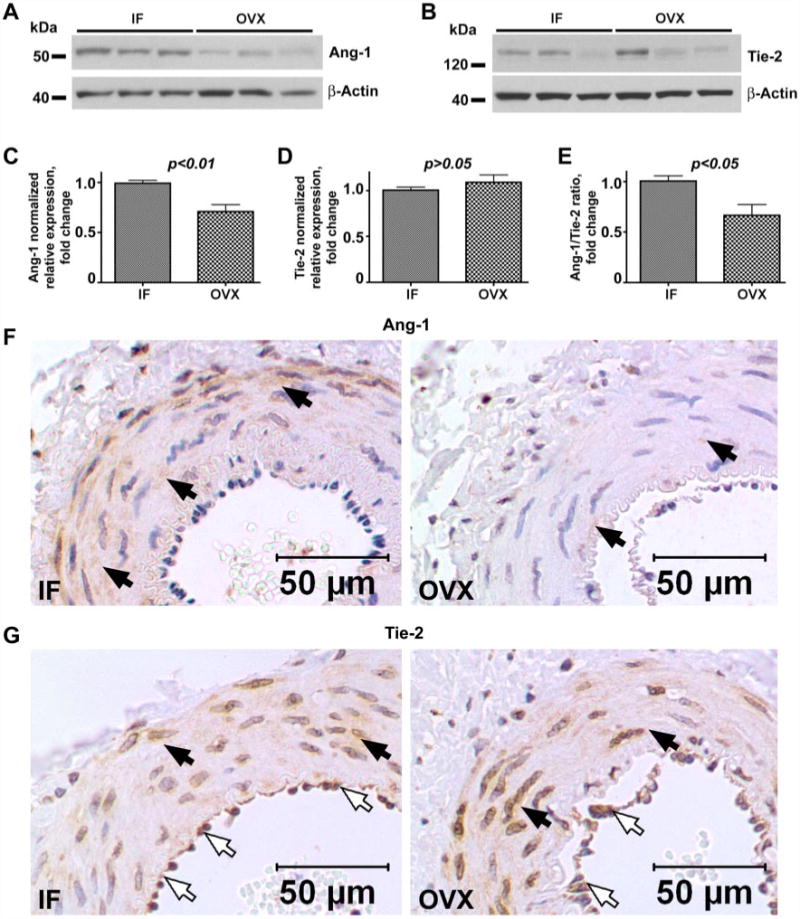
Western blot and immunohistochemical analysis of angiopoietin-1 (Ang-1; A, C, and F) and Tie-2 (B, D, and G) expression in dura mater of IF and OVX Yucatan miniature pigs 2 mo post-OVX. Note the 1.4-fold decrease in Ang-1-normalized relative expression (C) and 1.5-fold decrease in Ang-1-to-Tie-2 ratio (E) in OVX animals compared with IF, whereas Tie-2 expression (D) remains unchanged. C–E: bar graphs show means ± SE. F and G: note vascular expression of Ang-1 (F) and Tie-2 (G). The Ang-1 immunoreactivity (F) is confined to the vascular smooth muscle cells (black arrows) in both IF (left) and OVX (right) animals and is markedly reduced in OVX pigs compared with IF. In contrast, there is no difference in the immunoreactivity of the Ang-1 receptor Tie-2 between the groups (G), and Tie-2 expression is evident in both smooth muscle cells (black arrows) and endothelial cells (white arrows). Scale bars, 50 μm.
Undoubtedly, described herein, ovarian-dependent vascular remodeling is an extremely complex process that could not be governed by a single molecular or cellular mechanism and likely involves multiple factors regulated directly or indirectly by E2. It appears, though, that the interplay of E2 with the angiopoietin system is important in controlling overall blood vessel maintenance and stability. We hereby propose a mechanism by which a cessation of ovarian E2 production results in a reduced Ang-1 synthesis, microvessel destabilization, and post-OVX vascular remodeling (Fig. 4).
Fig. 4.
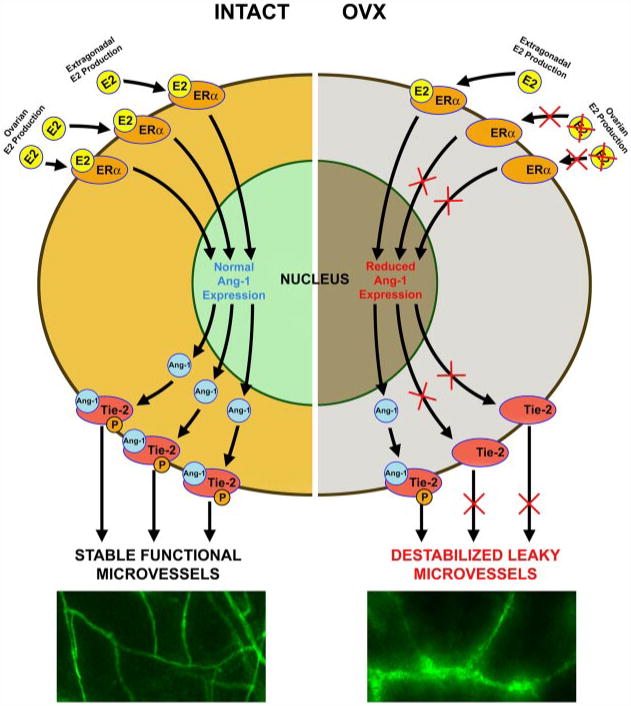
Schematic representation of a proposed mechanism of the ovarian-dependent control of vascular stability. Loss of ovarian E2 production results in a reduced ER-α-dependent Ang-1 synthesis, leading to a decrease in vessel-stabilizing Tie-2 signaling, microvessel destabilization, and post-OVX vascular remodeling.
Changes in the meningeal microvasculature associated with a cessation of the ovarian steroid hormone production may have a profound effect on microvessel function and the overall health in postmenopausal women. For example, a capillary rarefaction of this magnitude would inevitably result in a corresponding increase in peripheral vascular resistance (17), require microvascular network accommodation to altered hemodynamic and water-solute exchange requirements, and affect vascular function. Indeed, capillary rarefaction in dermal and skeletal muscle microvasculature, leading to the increase in peripheral resistance and altered vascular function, is characteristic of hypertensive animals (15) and humans (17). In addition, if similar changes occur in pia mater and brain, ovarian-dependent vascular remodeling could be intimately involved in age-related decrease in brain microvessel density, leading to a decline in neuronal metabolic activity and consequent decrease in cognitive ability as well (18). Furthermore, a recent study from Luchsinger et al. (14) established a potential link between the aggregation of vascular risk factors and the risk of Alzheimer's disease. Thus, described herein, microvascular changes associated with the loss of ovarian hormone secretion may constitute a morphological basis for the development of multiple perimenopausal and age-related symptoms and explain at least in part why postmenopausal women become more susceptible to cardiovascular and cerebrovascular conditions.
Acknowledgments
We thank the National Center for Gender Physiology and Environmental Adaptation Swine Hormone Core and Hormone Assay Core for technical assistance.
Grants: This research was supported by grants from the Veterans Affairs Merit Review Program (to V. V. Glinsky), National Aeronautics and Space Administration (to V. H. Huxley and L. J. Rubin), and National Institutes of Health (to J. R. Turk, L. J. Rubin, and V. H. Huxley). O. V. Glinskii was supported by the fellowship from the American Heart Association Heartland Affiliate.
References
- 1.Albrecht ED, Pepe GJ. Steroid hormone regulation of angiogenesis in the primate endometrium. Front Biosci. 2003;8:d416–d429. doi: 10.2741/1001. [DOI] [PubMed] [Google Scholar]
- 2.Albrecht ED, Robb VA, Pepe GJ. Regulation of placental vascular endothelial growth/permeability factor expression and angiogenesis by estrogen during early baboon pregnancy. J Clin Endocrinol Metab. 2004;89:5803–5809. doi: 10.1210/jc.2004-0479. [DOI] [PubMed] [Google Scholar]
- 3.Ardelt AA, McCullough LD, Korach KS, Wang MM, Munzenmaier DH, Hurn PD. Estradiol regulates angiopoietin-1 mRNA expression through estrogen receptor-α in a rodent experimental stroke model. Stroke. 2005;36:337–341. doi: 10.1161/01.STR.0000153795.38388.72. [DOI] [PubMed] [Google Scholar]
- 4.Baffert F, Le T, Thurston G, McDonald DM. Angiopoietin-1 decreases plasma leakage by reducing number and size of endothelial gaps in venules. Am J Physiol Heart Circ Physiol. 2006;290:H107–H118. doi: 10.1152/ajpheart.00542.2005. [DOI] [PubMed] [Google Scholar]
- 5.Bake S, Sohrabji F. 17beta-estradiol differentially regulates blood-brain barrier permeability in young and aging female rats. Endocrinology. 2004;145:5471–5475. doi: 10.1210/en.2004-0984. [DOI] [PubMed] [Google Scholar]
- 6.Brass LM. Estrogens and stroke: use of oral contraceptives and postmenopausal use of estrogen: current recommendations. Curr Treat Options Neurol. 2004;6:459–467. doi: 10.1007/s11940-004-0004-9. [DOI] [PubMed] [Google Scholar]
- 7.Dubey RK, Jackson EK. Estrogen-induced cardiorenal protection: potential cellular, biochemical, and molecular mechanisms. Am J Physiol Renal Physiol. 2001;280:F365–F388. doi: 10.1152/ajprenal.2001.280.3.F365. [DOI] [PubMed] [Google Scholar]
- 8.Flouriot G, Brand H, Denger S, Metivier R, Kos M, Reid G, Sonntag-Buck V, Gannon F. Identification of a new isoform of the human estrogen receptor-α that is encoded by distinct transcripts and that is able to repress hER-α activation function 1. EMBO J. 2000;19:4688–4700. doi: 10.1093/emboj/19.17.4688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Glinskii OV, Huxley VH, Turk JR, Deutscher SL, Quinn TP, Pienta KJ, Glinsky VV. Continuous real time ex vivo epifluorescent video microscopy for studying cancer cell interactions with dura mater microvasculature. Clin Exp Metastasis. 2003;20:451–458. doi: 10.1023/a:1025449031136. [DOI] [PubMed] [Google Scholar]
- 10.Glinskii OV, Turk JR, Pienta KJ, Huxley VH, Glinsky VV. Evidence of porcine and human endothelium activation by cancer-associated carbohydrates expressed on glycoproteins and tumor cells. J Physiol. 2004;554:89–99. doi: 10.1113/jphysiol.2003.054783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Huang A, Kaley G. Gender-specific regulation of cardiovascular function: estrogen as key player. Microcirculation. 2004;11:9–38. doi: 10.1080/10739680490266162. [DOI] [PubMed] [Google Scholar]
- 12.Kang DH, Yu ES, Yoon KI, Johnson R. The impact of gender on progression of renal disease. Potential role of estrogen-mediated vascular endothelial growth factor regulation and vascular protection. Am J Pathol. 2004;164:679–688. doi: 10.1016/S0002-9440(10)63155-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lowe GD. Hormone replacement therapy and cardiovascular disease: increased risks of venous thromboembolism and stroke, and no protection from coronary heart disease. J Intern Med. 2004;256:361–374. doi: 10.1111/j.1365-2796.2004.01400.x. [DOI] [PubMed] [Google Scholar]
- 14.Luchsinger JA, Reitz C, Honig LS, Tang MX, Shea S, Mayeux R. Aggregation of vascular risk factors and risk of incident Alzheimer disease. Neurology. 2005;65:545–551. doi: 10.1212/01.wnl.0000172914.08967.dc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Papanek PE, Rieder MJ, Lombard JH, Greene AS. Gender-specific protection from microvessel rarefaction in female hypertensive rats. Am J Hypertens. 1998;11:998–1005. doi: 10.1016/s0895-7061(98)00114-9. [DOI] [PubMed] [Google Scholar]
- 16.Pasqualini C, Guivarc'h D, Barnier JV, Guibert B, Vincent JD, Vernier P. Differential subcellular distribution and transcriptional activity of ΣE3, ΣE4, ΣE3–4 isoforms of the rat estrogen receptor-α. Mol Endocrinol. 2001;15:894–908. doi: 10.1210/mend.15.6.0642. [DOI] [PubMed] [Google Scholar]
- 17.Prasad A, Dunnill GS, Mortimer PS, MacGregor GA. Capillary rarefaction in the forearm skin in essential hypertension. J Hypertens. 1995;13:265–268. [PubMed] [Google Scholar]
- 18.Riddle DR, Sonntag WE, Lichtenwalner RJ. Microvascular plasticity in aging. Ageing Res Rev. 2003;2:149–168. doi: 10.1016/s1568-1637(02)00064-8. [DOI] [PubMed] [Google Scholar]
- 19.Seo KH, Lee HS, Jung B, Ko HM, Choi JH, Park SJ, Choi IH, Lee HK, Im SY. Estrogen enhances angiogenesis through a pathway involving platelet-activating factor-mediated nuclear factor-kappaB activation. Cancer Res. 2004;64:6482–6488. doi: 10.1158/0008-5472.CAN-03-2774. [DOI] [PubMed] [Google Scholar]
- 20.Simoncini T, Mannella P, Fornari L, Caruso A, Varone G, Genazzani AR. Genomic and non-genomic effects of estrogens on endothelial cells. Steroids. 2004;69:537–542. doi: 10.1016/j.steroids.2004.05.009. [DOI] [PubMed] [Google Scholar]
- 21.Stirone C, Duckles SP, Krause DN. Multiple forms of estrogen receptor-α in cerebral blood vessels: regulation by estrogen. Am J Physiol Endocrinol Metab. 2003;284:E184–E192. doi: 10.1152/ajpendo.00165.2002. [DOI] [PubMed] [Google Scholar]
- 22.Zakrzewicz A, Secomb TW, Pries AR. Angioadaptation: keeping the vascular system in shape. News Physiol Sci. 2002;17:197–201. doi: 10.1152/nips.01395.2001. [DOI] [PubMed] [Google Scholar]


