Abstract
Acute lung injury (ALI) and acute respiratory distress syndrome are characterized by protein rich alveolar edema, reduced lung compliance, and acute severe hypoxemia. A degree of pulmonary hypertension (PH) is also characteristic, higher levels of which are associated with increased morbidity and mortality. The increase in right ventricular (RV) afterload causes RV dysfunction and failure in some patients, with associated adverse effects on oxygen delivery. Although the introduction of lung protective ventilation strategies has probably reduced the severity of PH in ALI, a recent invasive hemodynamic analysis suggests that even in the modern era, its presence remains clinically important. We therefore sought to summarize current knowledge of the pathophysiology of PH in ALI.
Keywords: pulmonary vascular dysfunction, right ventricular failure, microvascular occlusion, hypoxic vasoconstriction, pulmonary vascular remodeling
acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) are characterized by acute onset hypoxemia associated with increased pulmonary vascular permeability and the development of noncardiogenic pulmonary edema (5). These syndromes are defined by the severity of hypoxemia (according to the ratio of arterial to inspired oxygen: the P-to-F ratio), with the presence of bilateral infiltrates on chest radiography and the exclusion of cardiogenic pulmonary edema (143). Despite some evidence of improvements in mortality in selected centers over recent decades (1), ALI remains a major public health problem, with 28-day mortality in the region of 25–35% (84). ALI may develop after a diverse spectrum of causes. These associated conditions may be categorized according to the nature of the insult, with for example pneumonia causing a direct lung injury and pancreatitis and nonpulmonary sepsis causing indirect lung injury, often as part of a multi-organ dysfunction syndrome, the former being associated with a higher mortality (126).
These direct or indirect insults result in neutrophil- and platelet-dependent dysfunction of the alveolar-epithelial barrier. The resultant protein-rich pulmonary edema fluid floods alveoli and causes surfactant dysfunction, which results in collapse and consolidation of lung units. Severe refractory hypoxemia results from ventilation-perfusion mismatch due to impairment of hypoxic pulmonary vasoconstriction. In addition to hypoxemia, hypercapnia is also a feature of ALI. This reflects involvement of the pulmonary microcirculation, due both to the disease process itself, as well as relating to the effects of positive pressure ventilation. Involvement of the pulmonary microcirculation is important early in ALI. For example, the inability to excrete carbon dioxide in those areas of lung being ventilated but not perfused, which equates to ventilatory dead space, can be measured at the bedside and is associated with mortality (97).
CLINICAL PERSPECTIVE
Pulmonary hypertension (PH) is defined as a mean pulmonary artery pressure of greater than 25 mmHg. According to this definition, PH is commonly found in patients who have developed ALI (149). However, this definition is largely used for patients with chronic PH. The World Health Organization further subclassified chronic PH into five groups, based on similar pathophysiology and anticipated responses to treatment (118). However, as yet there is no consensus definition of acute PH and it is difficult to assess how much information can be translated from the chronic setting to acute conditions like ALI/ARDS. As such, what may be of more clinical importance is the presence of right ventricular (RV) dysfunction or failure. Other factors may also contribute to reduced RV function in ALI including acute myocardial dysfunction, related to sepsis or a systemic inflammatory response. In the setting of ALI, RV failure with its nonspecific symptoms and signs is probably underdiagnosed. There are few studies documenting the progression of PH in patients with ALI, but a progressive increase in pulmonary pressures has been associated with a worse outcome (149). The incidence of acute RV failure appears to have fallen (from 22% to 50% to less than 10% in most studies) since the introduction of protective lung ventilation strategies (Table 1) (23, 26, 61, 62, 91, 100, 121, 135, 149).
Table 1.
Outcome studies of pulmonary hypertension and right ventricular dysfunction in patients with acute lung injury
| Author | Study Period | No. | ALI Population | How Diagnosed | Pulmonary Hemodynamics | RVF (PH) Prevalence | Outcome Associations |
|---|---|---|---|---|---|---|---|
| Zapol et al., 1977 | 1977 | 30 | Mixed | PAC | PAP, PAOP, PVR | All had PH | Not with initial but with persistent PH |
| Jardin et al., 1985 | 1985 | 23 | Mixed | TTE | RV size and systolic function | 22% had RVF | |
| Squara et al., 1998 | 1998 | 586 | Primary and secondary ALI | PAC | PAP, RVSWI | Higher PAP & RVSWI did worse | |
| Monchi et al., 1995 | 1992–1995 | 177 | Direct and indirect ALI | PAC | RAP, PAOP | Those with RAP>PAOP did worse | |
| Jardin et al., 2007 | 1980–1992 | 156 | Mixed ALI; no limitation to ventilatory plateau pressure (Pplat) | TTE | RV size and function | 56% had RVF when Pplat >35 cmH2O | At higher Pplat, RVF increases mortality |
| Vieillard-Baron et al., 2001 | 2001 | 75 | Mixed ALI (with pressure-limited ventilation) | TEE | sPAP, RV size, and function | 25% had RVF | sPAP & RVF not predictive; RVF reversible in survivors |
| Cepkova et al., 2007 | 2007 | 42 | Mixed ALI | TTE | sPAP, RV size, and function | 7% had RV dysfunction | RV and SPAP dysfunction not predictive of increased mortality |
| Jardin et al., 2007* | 1993-2006 | 196 | Mixed ALI when ventilatory pressure limited (Pplat <26 cmH2O) | TTE | RV size and function | 13% had RVF when Pplat limited | RVF did not contribute to increased mortality when Pplat low |
| Osman et al., 2009 | 2008 | 145 | Mixed ALI | PAC | PAP, PAOP, RVSVI | 9.6% had RVF | mPAP and CVP>PAOP predictive; Early RVF not predictive of mortality |
| Bull et al., 2010 | 2000–2005 | 501 | ARDSNET group | PAC | mPAP, PAOP, TPG, PVRI | 12% had RVF; (73% had PH) | High TPG predicted mortality |
| Lessons learned | Overall reduction in PH/RV dysfunction over time | Persistent PH associated with worse outcomes |
PAC, pulmonary artery catheterization; Pplat, plateau pressure; TTE, transthoracic echocardiography; TEE, transesophageal echocardiography; SPAP, systolic pulmonary arterial pressure; PH, pulmonary hypertension; ALI, acute lung injury; RVSWI, right ventricular stroke work index; RVF, right ventricular failure; TPG, transpulmonary gradient; PAP, pulmonary artery pressure; PAOP, pulmonary artery occlusion pressure; PVR, pulmonary vascular resistance; CVP, central venous pressure.
The long-term effects of ALI on pulmonary vascular function are less well studied. The most common finding is that of a reduced diffusion capacity for carbon monoxide (DLCO), seen in 70% of patients in the first year of recovery (78). This reduction in DLCO appears to persist even when other lung function changes have improved and may indicate reduced vascular reserve (36). In patients three years after ALI, in whom DLCO was decreased in only 13%, cardiopulmonary exercise testing revealed exercise-induced impairment in gas exchange in 50% (95), providing an even more sensitive indicator of reduced gas transfer. To our knowledge, there are no long-term hemodynamic studies available.
PATHOPHYSIOLOGY OF PH IN ACUTE LUNG INJURY
The pathophysiological changes occurring within the pulmonary vasculature in ALI include: 1) endothelial dysfunction, 2) pulmonary vascular occlusion, 3) increased vascular tone, 4) extrinsic vessel occlusion, and 5) vascular remodeling, as summarized in Fig. 1.
Fig. 1.
Mechanistic diagram illustrating the pathway through the pulmonary circulation and factors that may contribute to pulmonary hypertension (PH). This figure represents the factors in acute lung injury (ALI) contributing to the raised pulmonary vascular resistance (PVR) measured proximally in the pulmonary artery, as illustrated distally throughout the pulmonary vascular tree. Activation of pulmonary microvascular endothelium leads to initiation of inflammation and intravascular coagulation (also see Figs. 2–4): intravascular microthrombi, fibrin, and intravascular sequestration of cells contribute to pulmonary capillary occlusion, and clots also form in larger vessels. Vascular tone is increased in muscular arterioles and veins due to hypoxic pulmonary vasoconstriction (HPV), in an attempt to maintain ventilation/perfusion matching. Additionally, an imbalance of vasoactive mediators (e.g., endothelin-1 is raised) contributes to an increase in vasomotor tone. An overall reduction in lung volume, edema, positive end-expiratory pressure (PEEP), and atelectasis causes extrinsic vessel compression (top white circles). Later, structural changes of pulmonary vascular remodeling occur, with increased thickness of the existing pulmonary arterial smooth muscle layer, and neomuscularization of previously nonmuscularized vessels; fibrous intimal proliferative lesions are also seen in small pulmonary arteries, veins, and lymphatics. Finally, postcapillary PH may result from any cause of raised left atrial pressure such as left ventricular (LV) diastolic dysfunction due to myocardial ischemia or sepsis. Some or all of these pathological factors may contribute to the increase in PVR that is characteristic of ALI. Pplat, alveolar plateau pressure; EC, endothelial cell.
Endothelial Dysfunction
Functions of healthy pulmonary endothelium include the control of appropriate local blood flow, coagulation, vascular tone, angiogenesis, and cell proliferation (7). Pulmonary endothelial cells (EC) synthesize and release a wide variety of products (Table 2), as well as metabolizing others [e.g., angiotensin I by pulmonary endothelial angiotensin converting enzyme (ACE)]. In health, ECs provide a smooth thromboresistant surface protecting the subendothelium from procoagulant factors and platelets (80). They form a tight monolayer that acts as a barrier to control water and solute transport, maintained through complex cytoskeletal tethering forces between and within each cell (85). The large surface area and proximity to the air space, however, make the pulmonary microvascular endothelium vulnerable to injury from both direct alveolar insult and circulating mediators (144). The resultant activation of the endothelium is followed by dysfunction. Endothelial dysfunction involves an imbalance between vasodilating and vasoconstricting mediators and a shift to a prothrombotic phenotype.
Table 2.
Examples of endothelial cell products
| Thrombomodulatory |
| Thrombomodulin |
| Tissue plasminogen activator |
| von Willebrand factor |
| Ecto ADPases |
| TF |
| Vasoactive |
| Prostacyclin, thromboxane, and other prostanoids |
| Endothelins |
| Nitric oxide |
| Adhesion molecules |
| E-selectin |
| ICAM 1 and 2 |
| VCAM |
| Inflammatory molecules |
| Platelet activating factor |
| Cytokines: IL-6, IL-8, MCP-1 |
| Class II MHC molecules |
MCP, monocyte chemoattractant protein; MHC, major histocompatibility class; TF, tissue factor.
Evidence for endothelial dysfunction in acute lung injury.
Endothelial dysfunction is important early and late in the course of ALI (128, 129). Histological characteristics of injury include EC swelling, the presence of enlarged mitochondria, dilated endoplasmic reticulum adjacent to the capillary lumens (129), pinocytotic vesicle formation, and inter-endothelial cell separation (114). Additionally, there is evidence for altered production of endothelial-derived molecules including Von Willebrand's factor (vWF), ACE, and angiopoietin-2. vWF is a plasma glycoprotein produced by the endothelium considered to be a biomarker of EC activation and injury: plasma vWF levels were elevated in a primate model of lung inflammation induced by intravenous bacterial lipopolysaccharide even before significant EC damage (111). Furthermore, elevated plasma levels predict the onset and increased mortality of patients with ALI (110, 139). Pulmonary endothelial ACE is responsible for regulation of vascular tone, hydrolysis of angiotensin I, and deactivation of bradykinin. Circulating levels fell early in experimental ALI before deterioration in respiratory parameters (25). Clinical indicator dilution studies suggest that pulmonary capillary endothelium-bound ACE is a good biomarker of endothelial dysfunction and ALI severity (98). Angiopoietin-2 may represent another biomarker of endothelial activation: it is produced by epithelial cells to activate EC, and levels are elevated in patients with ALI (14). Markers of endothelial injury have been associated with pulmonary dead space fraction in experimental lung injury (39) as well as with raised pulmonary vascular pressures (24).
Initiation of endothelial dysfunction in acute lung injury.
Factors responsible for activating EC in ALI include microbes, immune complexes, drugs, toxins, reactive oxygen species, cytokines, microemboli, and activated leukocytes (99), as well as mechanical forces (for example strain associated with mechanical ventilation or increased shear). These stimuli may initiate innate and adaptive immune responses. In the former, recognition of conserved components of pathogens or of endogenous ligands released by injured tissue occurs through Toll-like receptors present on vascular cells. For example, following activation of Toll-like receptor-4 on EC by gram-negative bacterial endotoxin, transcription factors such as NF-κB lead to the expression of inflammatory mediators (4). The process of endothelial activation and dysfunction is illustrated in Fig. 2 and discussed below.
Fig. 2.
Components of endothelial activation/dysfunction in acute lung injury (ALI). This schematic cartoon illustrates the components of endothelial activation including 1) endothelial cell-leukocyte interactions, 2) endothelial cell-platelet-neutrophil interactions, and 3) structural changes between endothelial cells. These are described sequentially in detail within the text. RBC, red blood cell; MLCK, myosin light chain kinases; PASMC, pulmonary artery smooth muscle cell.
Components of endothelial dysfunction in acute lung injury.
ENDOTHELIAL CELL–LEUKOCYTE INTERACTIONS (FIG. 2, POINT 1).
Activation of pulmonary microvascular endothelium in ALI generates expression of the EC-derived adhesion molecules (E- and P-selectins and ICAM-1) and the expression of corresponding neutrophil ligands (for example, L-selectin and β2-integrins such as CD11/CD18) (125, 130). These interactions result in the rolling and adhesion of neutrophils on the endothelium, as well described in the systemic endothelium (73). In addition to the upregulation of these adhesion molecules, slowing of neutrophil traffic also reflects important cytoskeletal alterations to neutrophils and a difference in their anatomical exit from the pulmonary compared with the systemic circulation (see Microvascular cell sequestration). Subsequent neutrophil extravasation through pulmonary capillaries and alveolar walls into lung tissue depends upon several factors including maintenance of chemotactic gradients and continued expression of integrins (125): these activated neutrophils are central to the pathogenesis of parenchymal lung injury (30).
ENDOTHELIAL CELL-PLATELET-NEUTROPHIL INTERACTIONS (FIG. 2, POINT 2).
It is considered that a second wave of signals follows initial EC and neutrophil activation that captures circulating erythrocytes or platelets (52). Interaction of EC with platelets was demonstrated using fluorescence microscopy hours after endotoxin infusion in rabbits, associated with an upregulation of P- and E-selectins (64). Activated platelets release prestored mediators that further activate neutrophils and via thromboxane A2 release cause pulmonary vasoconstriction and increased vascular permeability (150). The aggregation of leukocyte and platelets is likely to contribute to occlusion of small vessels with other blood cells and microemboli (see Microvascular cell sequestration).
STRUCTURAL CHANGES BETWEEN ENDOTHELIAL CELLS (FIG. 2, POINT 3).
Activated EC undergo structural changes that increase vascular permeability: ECs retract to create paracellular gaps, as well as forming pinocytotic vesicles (114, 129). The retraction process involves an increase in intracellular calcium (for example, following the binding of thrombin to its EC receptor), which through interaction with calmodulin activates myosin light chain kinases (MLCK) to phosphorylate myosin light chains (MLCs). The resulting increased actin and myosin interaction shifts intracellular actin filaments away from a cortical orientation, changing the EC morphology (35). Clinically, increased endothelial permeability is associated with both ALI severity and bronchoalveolar lavage neutrophil content (119).
Pulmonary Microvascular Occlusion
Studies demonstrate a loss of capillary density, as illustrated in Fig. 3, as well as microvascular infarcts (120, 148). Thromboemboli and fibrinous obliteration are seen in larger pulmonary arteries, veins, and lymphatics, especially beyond 10 days (129). Larger vessel pulmonary arterial thromboses were evident in patients with ALI associated with PH: administration of intravenous streptokinase to these patients led to angiographic clearance of the obstructions as well as increased microvascular filling, improved cardiac output, and oxygenation (48).
Fig. 3.
Obliteration of the pulmonary microcirculation. Silicone casts from postmortem specimens illustrating obliteration of the pulmonary microcirculation in a patient 3 wk following onset of acute lung injury (A), compared with that from a patient without pulmonary disease (B). With thanks to Professor Warren Zapol.
Sickle cell chest crises are a rare cause of ALI, often associated with severe PH. These are characterized by pulmonary microvascular occlusion due to adherent sickled erythrocytes, leukocytes, and infarcted bone marrow-derived emboli (134), as well as larger vessel thrombi in up to 20% of patients (32a). These patients have endothelial dysfunction with increased adhesiveness of erythrocytes to EC (117) and increased circulating levels of endothelin-1 (104).
The overlapping components of microvascular occlusion in ALI include sequestration of cells within the pulmonary microcirculation and the onset of intravascular coagulation. These are discussed in turn.
Microvascular cell sequestration.
Neutrophils and platelets are likely to contribute to the occlusion of small vessels in ALI. Studies illustrate intrapulmonary trapping of neutrophils upon first-pass through the lung (77, 105) preceding their migration into the lung parenchyma. This is probably due to both to a mechanical obstruction [since neutrophil migration occurs at the level of the pulmonary capillaries, rather than at the level of the postcapillary venules as in the systemic circulation (33)] and to a morphological change in the neutrophils; in the latter, their cytoskeletal stiffness increases, reducing their deformability, thereby trapping them and arresting them at sites of inflammation. Perhaps surprisingly this does not appear to depend on classic adhesion molecules including L-selectin and CD11b-CD18 (146).
Despite the obvious importance of the neutrophil, ALI also occurs in neutropenic patients (115). It is interesting that neutropenic sheep had a blunted acute pulmonary hypertensive response following infused endotoxin compared with normal sheep, associated with lower plasma thromboxane levels (59). Leukocyte-dependent platelet microvascular sequestration was also demonstrated in a model of transfusion-related lung injury forming leukocyte-platelet aggregates. In this model, ALI severity was attenuated by platelet depletion or inhibition with aspirin (76).
Intravascular coagulation.
An important shift to a procoagulant antifibrinolytic state occurs in ALI. This was suggested over 30 years ago when a thrombin infusion led to respiratory insufficiency in dogs (112) and fibrin microthrombi were found postmortem (17). Changes in measureable procoagulant and antifibrinolytic factors are summarized (Table 3). For example, increased fibrin deposition is reflected by increased bronchoalveolar lavage levels of procoagulant mediators (fibrinopeptide A, factor VII, and d-dimer) and reduced fibrinolytics [e.g., reduced urokinase and increased plasminogen activator inhibitor (PAI)] (13, 60). A reduction in the natural anticoagulant protein C occurs, suggestive of intravascular coagulation, associated with worse clinical outcomes (83). In lung injury models, administration of recombinant activated protein C (aPC) improved both lung edema and attenuated the increase in pulmonary artery pressure (136). In a large clinical trial where aPC was given to selected patients with ALI, it is noteworthy that despite no apparent benefits in terms of ventilator free days or 60-day mortality, there was an improvement in dead space fraction (74). However, given the recent negative outcome from the PROWESS-shock trial (37a) and subsequent withdrawal of Xigris, it is unlikely that any potential beneficial effects of aPC on the pulmonary circulation will be investigated further.
Table 3.
Principle coagulation and fibrinolytic factors and their function in ALI
| Factor | Location | Biological Function | Levels in ALI |
|---|---|---|---|
| Procoagulants | |||
| TF | Constitutively expressed subendothelium (away from plasma coagulation factors) | Complexes with FVIIa to initiate extrinsic pathway to form thrombin (from prothrombin) and fibrin | Increased BAL levels in ALI (60) |
| Upregulated on platelets, monocytes, macrophages, and EC in response to cytokines | In ALI, generation of alveolar thrombin is mediated by TF (72) | Increased staining in lung tissue in ALI (in hyaline membranes, alveolar epithelial cells and macrophages) (9) | |
| BAL contains elevated thrombin and fibrin (TF-induced) (60). | |||
| von Willebrand factor | Procoagulant amino acid/protein on EC | Binds to other proteins, especially factor VIII; important in platelet adhesion | Increased in ALI (111) |
| Anticoagulants | |||
| TF pathway inhibitor | Anticoagulant protein | Natural anticoagulant for extrinsic pathway: inactivates TF-FVIIa complexes after binding to Xa | Increased in ALI after endothelial injury (but not to match TF increase) (42) |
| Made by EC | Attenuates LPS-induced inflammatory responses in rats by inhibiting TNF-α production by monocytes | ||
| 15% Secreted into blood | |||
| Protein C | Serine protease inactive zymogen on EC surface | 1) Natural anticoagulant | Low plasma levels in ALI (140), associated with worse outcomes (83) |
| Activated (to aPC) by thrombin /thrombomodulin complex | aPC initiates FVa and FVIIIa so reducing downstream thrombin generation | ||
| 2) Anti-inflammatory—e.g., aPC inhibits TNF production via NF-κB and AP-1 signaling | |||
| Thrombomodulin | EC surface protein that binds thrombin | Anticoagulant cofactor in the thrombin-induced activation of protein C | Increased in edema fluid in ALI, associated with worse outcomes (140) |
| Plasminogen activator inhibitor type-1 | Produced by EC | Main inhibitor of tPA and urokinase: inhibits fibrinolysis | BAL levels reduced (13) |
| Antithrombin | Plasma glycoprotein inhibitor of thrombin | Anticoagulant; shown to prevent LPS-induced vascular injury and promotes endothelial release of prostacyclin (131) | Reduced in ALI (101) |
EC, endothelial cell; LPS, lipopolysaccharide; tPA, tissue plasminogen activator; aPC, activated protein C.
Coagulation and inflammation.
Activation of coagulation overlaps with inflammatory processes at the EC surface as illustrated in Fig. 4. For example, IL-6 upregulates tissue factor (TF) expression on EC (113), and tumor necrosis factor (TNF)-α attenuates fibrinolysis by stimulating the release of inhibitors of plasminogen activators (72). Exposure of TF activates the extrinsic pathway, resulting in thrombin release, fibrinogen cleavage to produce fibrin, and activation of platelets by binding protease-activated receptors (28). Release of vWF is also increased from EC in response to fibrin (107). In an attempt to balance increased intravascular coagulation, anticoagulant mechanisms are in place: thrombin binds to the surface anticoagulant thrombomodulin and the surface endothelial protein C receptor (EPCR) releases aPC that degrades thrombin. Furthermore, there is a concurrent increase in tissue factor pathway inhibitor, although this is deemed insufficient to match the increase in TF in patients with ALI (42). Finally, inhibition of fibrinolysis is characteristic in ALI, which at least in part leads to a reduction in PAI-1 (13). The linkage of inflammation and intravascular coagulation is further supported by studies where prevention of vascular injury by aPC is dependent on inhibition of leukocyte activation (132).
Fig. 4.
Overview of processes leading to intravascular coagulation and inflammation at microvascular endothelial surface in ALI. A: activation of intravascular coagulation. Vascular injury initiates the tissue factor-driven extrinsic pathway to generate thrombin. Tissue factor (TF), usually hidden on the subendothelium, is upregulated on platelets, leukocytes, and on EC during inflammation. TF interacts with factor (F) Va to activate FIX and X, leading to prothrombin (P) activation, involving the formation of complexes between FVa and factor Xa, with subsequent thrombin (T) formation. Thrombin binds to protease-activated receptors (PARs) expressed on platelets and EC and has several functions, most importantly to enable formation of fibrin from fibrinogen and the subsequent stabilization of stable clot by activating FVIII. Thrombin also activates FV and FVIII from the intrinsic pathway, the function of which is to amplify coagulation. B: reduction in anticoagulants. Protein C dysfunction Thrombin usually binds thrombomodulin (TM) on the luminal side of the EC and the thrombin-TM complex converts protein C to activated protein C (APC). APC interacts with the endothelial cell protein C receptor (EPCR), and protein S, and acts as a natural anticoagulant by inactivating FVa and FVIIIa. APC is also thought to inhibit plasminogen activator inhibitor-type 1 (PAI-1) thus indirectly promoting fibrinolysis (see section C). In addition to its anticoagulant properties, aPC also has anti-inflammatory effects. These include inhibition of TNF-α production and blocking adhesion of leukocytes to selectins. In ALI, protein C levels are low, thereby resulting in both (intravascular and extravascular) coagulation and inflammation. C: reduction in fibrinolysis. Once a stable clot has formed, fibrinolysis occurs to break down stable clot through the actions of plasmin. Plasminogen is activated to form plasmin by tissue plasminogen activator (tPA), usually released slowly into blood by damaged EC to enable clot breakdown following vessel injury. Other plasminogen activators include urokinase, and FXII, XIIa and kallikrein. To balance this, the principle inhibitor of tPA and urokinase is plasminogen activator inhibitor-type 1 (PAI-1). During inflammation, there is evidence that cytokines such as TNF-α can stimulate the release of inhibitors of plasminogen activators, thus attenuating fibrinolysis.
Alterations in Pulmonary Vasomotor Tone
Rather than overt pulmonary vasoconstriction per se, increased pulmonary vascular resistance seen in ALI may reflect a loss of control of vascular tone, with an excess of pulmonary vasoconstrictor over vasodilator substances (Table 4). In animal models, direct injection of endotoxin leads to acute rises in pulmonary vascular resistance, probably through inhibition of nitric oxide (NO) (94), a phenomenon that may be mediated by endothelin B receptors (109). Derangements in acid-base balance also influence pulmonary vascular resistance (PVR) in patients with ALI (41, 86). The role of hypoxic pulmonary vasoconstriction in ALI is discussed, followed by a brief description of potential roles of relevant vasoactive mediators.
Table 4.
Local factors that may increase pulmonary vascular tone in acute lung injury
| Low mixed venous Po2: hypoxic vasoconstriction |
| Acidosis (hypercapnia) |
| Increased sympathetic tone |
| High airway pressure |
| Vasoconstrictor: vasodilator imbalance |
| Excess ET-1, TXA-1, 5-HT |
| Reduced nitric oxide, prostanoids |
| Effects of endotoxin |
5-HT, serotonin; ET-1, endothelin-1; Po2, partial pressure of oxygen; TXA-1, thromboxane A1.
Hypoxic pulmonary vasoconstriction.
Hypoxic pulmonary vasoconstriction (HPV) is a dynamic process predominantly mediated by precapillary arterioles (21) that promotes ventilation-perfusion matching by diverting blood flow away from poorly ventilated (diseased) and therefore hypoxic lung units. Although the mechanism of action of HPV is not fully understood, it is likely to involve inhibition of oxygen-sensitive K channels on vascular smooth muscle, leading to activation of voltage-gated calcium channels, Ca2+ influx and vasoconstriction (147). Basal NO release is likely to be important in limiting HPV (75), whereas reduced NO levels maintain it (93).
In ALI, HPV has a protective effect on gas exchange (21) while also contributing to mild levels of PH (20, 81). HPV may be inhibited by many factors (Table 5), which reverse this protective effect, resulting in a right-to-left intrapulmonary shunt and contributing to the profound characteristic hypoxemia (6, 31, 51).
Table 5.
Conditions that may inhibit hypoxic pulmonary vasoconstriction in acute lung injury
| Pathological | Treatment-Related |
|---|---|
| Endotoxin (59) | β-Agonists, α-adrenoceptor antagonists, nitro-glycerine, prostacyclin (82) |
| Alkalosis (22) | Volatile anesthetic agents (88) |
| Hypothermia (10) | Calcium channel blockers (65) Nitroprusside (63) |
| Postural effects (138) | Increased positive end-expiratory pressure (71) |
| Raised left atrial pressure (70) |
Disordered, inhomogeneous HPV is a potential contributor to high altitude pulmonary edema, a condition also characterized by pulmonary hypertension, which may have parallels to ALI (8). The resulting uneven distribution of pulmonary blood flow with exaggerated hypoxic pulmonary venoconstriction in turn exposes some pulmonary capillaries to high pressure (79) thought to induce endothelial stress failure (142).
Pulmonary vasoactive mediators.
In the setting of pulmonary arterial hypertension (PAH), of many potential mediators, reduced NO and cyclooxygenase (COX) pathway signaling, and increased endothelin (ET)-1 signaling occur. The resultant excessive pulmonary vasoconstriction and promotion of vascular cell proliferation are well recognized to contribute to the pathophysiology of PAH (58). Whether the same or similar mediators are responsible for the increase in pulmonary vascular tone and/or the pulmonary vascular remodeling in ALI is uncertain. The possible contribution of the COX, ET-1, and NO pathways to increased vasomotor tone in ALI is discussed.
COX PATHWAY.
Arachidonic acid is metabolized by isoforms of the COX enzyme to prostaglandins, prostacyclin, and thromboxane. Some of these products are vasoactive and may be implicated in dysregulated vascular tone in ALI. For example, raised levels of prostacyclin and thromboxane-A2 were seen in models of sepsis and ALI (12), and reduced removal of prostaglandin E1 was demonstrated in dilution studies in patients with ALI (47). Thromboxane-A2 release from leukocytes following endotoxin challenge is likely to contribute to the acute elevation in pulmonary artery pressure, as demonstrated in a sheep model of ALI (59).
ET-1.
ET-1 is a potent naturally occurring vasoconstrictor peptide and smooth muscle mitogen. It is produced predominantly by EC, and also by the more numerous PASMC (and fibroblasts). Stimuli for ET-1 production include hypoxia, endotoxin, and cytokines, via NF-κB–dependent signaling (145). There is evidence for upregulation of ET-1 in models of direct lung injury (68, 116) and in patients with ALI (34), where circulating levels are elevated and fall in association with clinical improvement and a fall in PVR (69). Treatment with an endothelin receptor antagonist in an oleic acid model suggested that ET-1 contributes to the early increase in PVR in ALI (56).
NO.
NO is an endogenous pulmonary vasodilator. It is produced by EC and other cells and acts via soluble guanylate cyclase to produce cGMP in vascular smooth muscle. In addition to basal vasodilatation, NO inhibits platelet and leukocyte adhesion, platelet aggregation, and smooth muscle cell proliferation (102, 122). In PAH, expression of pulmonary endothelial NO synthase is decreased (46). It is not known whether this is also the case in ALI, where NO levels may even be increased. In a rodent lipopolysaccharide-induced model of severe sepsis, NO synthase was induced in both systemic and pulmonary vessel walls leading to the production of large quantities of vasoactive NO (50). Rather than increasing vasodilatation, however, these high NO levels may combine with reactive oxygen species to form the highly damaging reactive nitrogen species peroxynitrite (40, 124). Finally, there is evidence for decreased exhaled NO in ALI (18), although the relevance of this to the vasculature is uncertain.
Extrinsic Vessel Compression
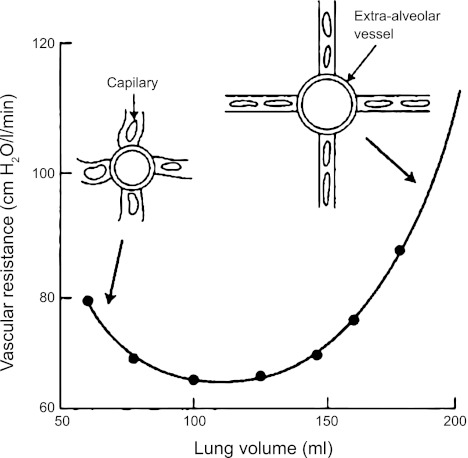
Several factors causing mechanical compression of pulmonary capillaries and veins may increase intravascular pressure in ALI (Fig. 1). In areas of lung with high alveolar volumes, alveolar pressure exceeds intra-capillary pressure (West zone 1), which through compression of capillaries may increase PVR (Fig. 5). This is demonstrated in patients following a Fontan procedure, where venous blood is shunted from the right atrium to the pulmonary arteries, and pulmonary blood flow is directly dependent on venous return to the right heart: pulmonary blood flow is reduced by positive pressure ventilation and augmented by negative pressure ventilation (103). In some patients with ALI elevated PVR, the resulting RV dysfunction may be rapidly reversed by reducing the airway pressure (135). At low lung volumes, such as in areas of atelectasis, PVR may also be increased through increased tone in extra-alveolar muscular vessels (Fig. 5) (141). Pulmonary veins may also be compressed by edema or areas of atelectasis, which in theory will also increase capillary pressure and pulmonary arterial pressure, as well as increasing vascular inflammation (66). Finally, the overall reduction in total lung volume will increase PVR, which may at least in part be due to compressive effects.
Fig. 5.
Increased pulmonary vascular resistance at extremes of lung volumes. This figure represents measurements made in an animal lobe preparation in which the transmural pressure of the capillaries is held constant. It illustrates, at least in noninjured lungs, that at low lung volumes (as may occur with atelectasis), extra-alveolar vessels become narrow, and smooth muscle and elastic fibers in these collapsed vessels increase PVR. At high lung volumes, as alveolar volumes are increased and walls are thinned, capillaries are stretched, reducing their caliber and also increasing PVR (adapted from John West's Essential Physiology, 8th edition, Philadelphia: Lippincott & Williams, with permission) (141).
Pulmonary Vascular Remodeling and Neomuscularization
Evidence for structural changes to small pulmonary vessels including fibrous intimal proliferative lesions was derived from postmortem studies in the era when higher airway pressures and inspired oxygen fraction were deemed to be acceptable (129). In intermediate and late cases (beyond 18 days′ duration) smooth muscle hypertrophy and neomuscularization of previously nonmuscular vessels were evident, with morphometric analysis revealing thrombosis, medial thickening, and decreased vascular density of pre- and intra-acinar vessels (120, 129). Proposed mechanisms may be similar to other causes of PH associated with chronic respiratory disease, where hypoxia is an important precipitant, and also likely to reflect the smooth muscle mitogenic effects of ET-1 as well as inflammation itself. For example, cytokines such as IL-6 are implicated in vascular remodeling in PAH (123). It has indeed been shown that repeated lung injury induced by 10–14 wk of endotoxin injection can lead to PH associated with remodeling (90).
Diagnosis of PH in ALI
The diagnosis of PH and RV dysfunction in ALI may be difficult since clinical signs may be nonspecific. Potential aides to clinical diagnosis include biomarkers such as brain natriuretic peptide (BNP), a marker of ventricular stretch and biomarkers reflecting endothelial dysfunction (see Table 3). These are, however, not validated in patients with pulmonary vascular dysfunction in ALI and likely to be very difficult to interpret. One study investigated BNP as a potential biomarker of RV dilatation early in ALI: serum BNP levels were found to be increased in the cohort overall, and RV dilatation on echocardiography was seen in 11 (26%) of patients. However, no correlation was found between elevated BNP and RV dilatation, although there was a moderate correlation between BNP and pulmonary dead space (27). It would be of interest to assess potential correlation of BNP with RV dysfunction, but this would require a much larger number of patients.The use of pulmonary artery catheters for invasive hemodynamic monitoring was previously commonplace in the management of ALI. We are not suggesting this practice is resumed, but given the adverse prognostic association of pulmonary vascular dysfunction with survival, consideration of the vascular component of ALI is indicated. Echocardiography is a very useful first-line diagnostic tool and is frequently used; however, it does have some limitations. These might include difficult visualization of the RV during trans-thoracic echocardiographic studies, and the nonsymmetrical shape of the RV, making it hard to reproducibly assess RV contractility or volume (53). Pulmonary arterial catheterization remains the gold standard for assessment of PH in other settings, and it may be useful both as a diagnostic and monitoring tool in ALI in the setting of RV dysfunction secondary to pulmonary hypertension. Cardiac output can be measured by several indicator dilution techniques and by oesophageal Doppler, but none of these provide a measure of PH and RV afterload, which therapies may manipulate directly or indirectly.
Potential Therapeutic Strategies
RV dysfunction in ALI may prevent the cardiac output rising appropriately or only doing so at the expense of systemic venous hypertension. This may limit oxygen delivery and contribute to other organ dysfunction and worse outcomes. Therapies can be directed at reducing PH directly. The systemic administration of vasodilators to patients with ALI may, however, worsen oxygenation due to blunting of HPV (29), as seen in the setting of PH in chronic hypoxic lung disease (44). Mechanical ventilation may also have profound effects on RV afterload and function with reductions in airway pressures improving RV function and hemodynamic stability (62, 135).
Inhaled NO.
Inhaled NO is a potent selective pulmonary vasodilator, which reduced PVR and improved cardiac output in patients with acute RV failure in the setting of ALI (11, 15, 16, 38, 108). Beneficial effects on the RV probably require higher doses of NO than those required to improve oxygenation (54). Continuous administration of NO is needed to reduce PVR, and oxygenation may worsen at high doses (43). A reduction in RV afterload, however, does not appear to correlate with any clinical outcome benefits in ALI (19, 32, 127), and its use is largely confined. Potential limitations of NO include its expense, an association with acute kidney injury (2), and accumulation of toxic metabolites, although this is not usually a clinically significant problem (49). Rebound PH with RV dysfunction on weaning from NO may be reduced with phosphodiesterase type 5 inhibitors (45).
Other pulmonary vasodilators.
Intravenous prostacyclin also reduced PVR and improved RV function in studies of ALI, although at the expense of increased intrapulmonary shunt (106). Inhaled administration of prostacyclin (133, 137, 151) and inhaled prostaglandin E1 (89) improved oxygenation and reduced PVR in ALI with minimal effects on SVR: further studies of prostacyclin and iloprost in patients with PH and ALI (3, 117a, 133a) are ongoing. NO and intravenous prostacyclin have been combined in ALI with effective reduction of PVR without adverse effects (67). Levosimendan is a calcium-sensitizing agent that also vasodilates the pulmonary, systemic, and coronary vessels by activating adenosine triphosphate-sensitive potassium channels. In a preliminary study of 35 patients, a 24-h infusion of levosimendan increased RV ejection fraction and cardiac output compared with placebo (92). Agents such as sildenafil and bosentan have not been investigated formally in ALI (96).
Potential future therapies.
Potential ALI therapies such as those targeting intravascular coagulation, neomuscularization, or endothelial/epithelial function may limit PH. There are no data about their concomitant effects on PH and RV function. For example, prior treatment with antiplatelet therapy may protect patients from ALI (37) but the effects on PH are unknown. The effects of other potential agents such as interferon-β, anti-tissue factor, anti-VEGF, HMG-CoA reductase inhibitors, and neuromuscular blockade are also unknown.
CONCLUSION
In ALI, the presence of significant PH is associated with increased mortality, although the incidence of acute RV failure does appear to have fallen over the last few decades probably in relation to use of lung protective ventilation strategies. Although PH may simply be a surrogate for the extent of pulmonary damage, it is possible also that the associated right ventricular dysfunction and systemic venous hypertension contribute significantly to further organ dysfunction. Moreover, the reduced gas transfer factor in patients who survive ALI suggests that pulmonary vascular dysfunction persists, at least in a selection of patients.
Several mechanisms may contribute to PH in ALI, including endothelial dysfunction, microvascular occlusion, an imbalance of vasoactive mediators, extrinsic compression of small vessels, pulmonary vascular neomuscularization, and remodeling. Whether these processes could be targeted to reduce PH and thereby modify acute outcome or even longer term cardiorespiratory performance is not known, but represent strategies that are largely untested.
GRANTS
The work was supported by the National Institute for Health Research Respiratory Disease Biomedical Research Unit at the Royal Brompton and Harefield National Health Service (NHS) Foundation Trust and Imperial College London. L. C. Price is supported by the British Heart Foundation. D. F. McAuley is supported by the Northern Ireland Public Health Agency, Research and Development Division Translational Research Group for Critical Care. The views expressed in this publication are those of the authors and not necessarily those of the NHS, the National Institute for Health Research, or the Department of Health.
DISCLOSURES
No conflicts of interest, financial or otherwise are declared by the author(s).
AUTHOR CONTRIBUTIONS
Author contributions: L.C.P., D.F.M., M.J.G., and S.J.W. conception and design of research; L.C.P. analyzed data; L.C.P. prepared figures; L.C.P. drafted manuscript; L.C.P., D.F.M., P.S.M., S.J.F., M.J.G., and S.J.W. edited and revised manuscript; L.C.P., D.F.M., P.S.M., S.J.F., M.J.G., and S.J.W. approved final version of manuscript.
REFERENCES
- 1. Abel SJ, Finney SJ, Brett SJ, Keogh BF, Morgan CJ, Evans TW. Reduced mortality in association with the acute respiratory distress syndrome (ARDS). Thorax 53: 292–294, 1998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Adhikari NK, Burns KE, Friedrich JO, Granton JT, Cook DJ, Meade MO. Effect of nitric oxide on oxygenation and mortality in acute lung injury: systematic review and meta-analysis. BMJ 334: 779–786, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Afshari A, Brok J, Moller AM, Wetterslev J. Aerosolized prostacyclin for acute lung injury (ALI) and acute respiratory distress syndrome (ARDS). Cochrane Database Syst Rev 8: CD007733, 2010 [DOI] [PubMed] [Google Scholar]
- 4. Akira S, Takeda K. Toll-like receptor signalling. Nat Rev Immunol 4: 499–511, 2004 [DOI] [PubMed] [Google Scholar]
- 5. Ashbaugh DG, Bigelow DB, Petty TL, Levine BE. Acute respiratory distress in adults. Lancet 2: 319–323, 1967. 4143721 [Google Scholar]
- 6. Baraka AS, Taha SK, Yaacoub CI. Alarming hypoxemia during one-lung ventilation in a patient with respiratory bronchiolitis-associated interstitial lung disease. Can J Anaesth 50: 411–414, 2003 [DOI] [PubMed] [Google Scholar]
- 7. Barnes PJ, Liu SF. Regulation of pulmonary vascular tone. Pharmacol Rev 47: 87–131, 1995 [PubMed] [Google Scholar]
- 8. Bartsch P, Mairbaurl H, Maggiorini M, Swenson ER. Physiological aspects of high-altitude pulmonary edema. J Appl Physiol 98: 1101–1110, 2005 [DOI] [PubMed] [Google Scholar]
- 9. Bastarache JA, Wang L, Geiser T, Wang Z, Albertine KH, Matthay MA, Ware LB. The alveolar epithelium can initiate the extrinsic coagulation cascade through expression of tissue factor. Thorax 62: 608–616, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Benumof JL, Wahrenbrock EA. Dependency of hypoxic pulmonary vasoconstriction on temperature. J Appl Physiol 42: 56–58, 1977 [DOI] [PubMed] [Google Scholar]
- 11. Benzing A, Mols G, Beyer U, Geiger K. Large increase in cardiac output in a patient with ARDS and acute right heart failure during inhalation of nitric oxide. Acta Anaesthesiol Scand 41: 643–646, 1997 [DOI] [PubMed] [Google Scholar]
- 12. Bernard GR, Reines HD, Halushka PV, Higgins SB, Metz CA, Swindell BB, Wright PE, Watts FL, Vrbanac JJ. Prostacyclin and thromboxane A2 formation is increased in human sepsis syndrome. Effects of cyclooxygenase inhibition. Am Rev Respir Dis 144: 1095–1101, 1991 [DOI] [PubMed] [Google Scholar]
- 13. Bertozzi P, Astedt B, Zenzius L, Lynch K, LeMaire F, Zapol W, Chapman HA., Jr Depressed bronchoalveolar urokinase activity in patients with adult respiratory distress syndrome. N Engl J Med 322: 890–897, 1990 [DOI] [PubMed] [Google Scholar]
- 14. Bhandari V, Choo-Wing R, Lee CG, Zhu Z, Nedrelow JH, Chupp GL, Zhang X, Matthay MA, Ware LB, Homer RJ, Lee PJ, Geick A, de Fougerolles AR, Elias JA. Hyperoxia causes angiopoietin 2-mediated acute lung injury and necrotic cell death. Nat Med 12: 1286–1293, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Bhorade S, Christenson J, O'Connor M, Lavoie A, Pohlman A, Hall JB. Response to inhaled nitric oxide in patients with acute right heart syndrome. Am J Respir Crit Care Med 159: 571–579, 1999 [DOI] [PubMed] [Google Scholar]
- 16. Bigatello LM, Hurford WE, Kacmarek RM, Roberts JD, Jr, Zapol WM. Prolonged inhalation of low concentrations of nitric oxide in patients with severe adult respiratory distress syndrome. Effects on pulmonary hemodynamics and oxygenation. Anesthesiology 80: 761–770, 1994 [DOI] [PubMed] [Google Scholar]
- 17. Bone RC, Francis PB, Pierce AK. Intravascular coagulation associated with the adult respiratory distress syndrome. Am J Med 61: 585–589, 1976 [DOI] [PubMed] [Google Scholar]
- 18. Brett SJ, Evans TW. Measurement of endogenous nitric oxide in the lungs of patients with the acute respiratory distress syndrome. Am J Respir Crit Care Med 157: 993–997, 1998 [DOI] [PubMed] [Google Scholar]
- 19. Brett SJ, Hansell DM, Evans TW. Clinical correlates in acute lung injury: response to inhaled nitric oxide. Chest 114: 1397–1404, 1998 [DOI] [PubMed] [Google Scholar]
- 20. Brimioulle S, Julien V, Gust R, Kozlowski JK, Naeije R, Schuster DP. Importance of hypoxic vasoconstriction in maintaining oxygenation during acute lung injury. Crit Care Med 30: 874–880, 2002 [DOI] [PubMed] [Google Scholar]
- 21. Brimioulle S, LeJeune P, Naeije R. Effects of hypoxic pulmonary vasoconstriction on pulmonary gas exchange. J Appl Physiol 81: 1535–1543, 1996 [DOI] [PubMed] [Google Scholar]
- 22. Brimioulle S, Lejeune P, Vachiery JL, Leeman M, Melot C, Naeije R. Effects of acidosis and alkalosis on hypoxic pulmonary vasoconstriction in dogs. Am J Physiol 258: H347–H353, 1990 [DOI] [PubMed] [Google Scholar]
- 23. Bull TM, Clark B, McFann K, Moss M. Pulmonary vascular dysfunction is associated with poor outcomes in patients with acute lung injury. Am J Respir Crit Care Med 182: 1123–1128, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Calfee C, Gallagher D, Abbott J, Aird WC, Thompson BT, Matthay MA, the NHLBI ARDS Network Plasma angiopoietin-2 in clinical acute lung injury: pathogenetic and prognostic significance. Crit Care Med. In Press [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Casey L, Krieger B, Kohler J, Rice C, Oparil S, Szidon P. Decreased serum angiotensin converting enzyme in adult respiratory distress syndrome associated with sepsis: a preliminary report. Crit Care Med 9: 651–654, 1981 [DOI] [PubMed] [Google Scholar]
- 26. Cepkova M, Kapur V, Ren X, Quinn T, Zhuo H, Foster E, Liu KD, Matthay MA. Pulmonary dead space fraction and pulmonary artery systolic pressure as early predictors of clinical outcome in acute lung injury. Chest 132: 836–842, 2007 [DOI] [PubMed] [Google Scholar]
- 27. Cepkova M, Kapur V, Ren X, Quinn T, Zhuo H, Foster E, Matthay MA, Liu KD. Clinical significance of elevated B-type natriuretic peptide in patients with acute lung injury with or without right ventricular dilatation: an observational cohort study. Ann Intensive Care 1: 18, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Chambers RC. Procoagulant signalling mechanisms in lung inflammation and fibrosis: novel opportunities for pharmacological intervention? Br J Pharmacol 153, Suppl 1: S367–S378, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Cornet AD, Hofstra JJ, Swart EL, Girbes AR, Juffermans NP. Sildenafil attenuates pulmonary arterial pressure but does not improve oxygenation during ARDS. Intensive Care Med 36: 758–764, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Cowburn AS, Condliffe AM, Farahi N, Summers C, Chilvers ER. Advances in neutrophil biology: clinical implications. Chest 134: 606–612, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Daoud FS, Reeves JT, Schaefer JW. Failure of hypoxic pulmonary vasoconstriction in patients with liver cirrhosis. J Clin Invest 51: 1076–1080, 1972 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Dellinger RP, Zimmerman JL, Taylor RW, Straube RC, Hauser DL, Criner GJ, Davis K, Jr, Hyers TM, Papadakos P. Effects of inhaled nitric oxide in patients with acute respiratory distress syndrome: results of a randomized phase II trial. Inhaled nitric oxide in ARDS Study Group. Crit Care Med 26: 15–23, 1998 [DOI] [PubMed] [Google Scholar]
- 32a. Dessap AM, Deux JF, Abidi N, Lavenu-Bombled C, Melica G, Renaud B, Godeau B, Adnot S, Brochard L, Brun-Buisson C, Galacteros F, Rahmouni A, Habibi A, Maitre B. Pulmonary artery thrombosis during acute chest syndrome in sickle cell disease. Am J Respir Crit Care Med 184: 1022–1029, 2011 [DOI] [PubMed] [Google Scholar]
- 33. Doerschuk CM, Beyers N, Coxson HO, Wiggs B, Hogg JC. Comparison of neutrophil and capillary diameters and their relation to neutrophil sequestration in the lung. J Appl Physiol 74: 3040–3045, 1993 [DOI] [PubMed] [Google Scholar]
- 34. Druml W, Steltzer H, Waldhausl W, Lenz K, Hammerle A, Vierhapper H, Gasic S, Wagner OF. Endothelin-1 in adult respiratory distress syndrome. Am Rev Respir Dis 148: 1169–1173, 1993 [DOI] [PubMed] [Google Scholar]
- 35. Dudek SM, Garcia JG. Cytoskeletal regulation of pulmonary vascular permeability. J Appl Physiol 91: 1487–1500, 2001 [DOI] [PubMed] [Google Scholar]
- 36. Elliott CG, Morris AH, Cengiz M. Pulmonary function and exercise gas exchange in survivors of adult respiratory distress syndrome. Am Rev Respir Dis 123: 492–495, 1981 [DOI] [PubMed] [Google Scholar]
- 37. Erlich JM, Talmor DS, Cartin-Ceba R, Gajic O, Kor DJ. Prehospitalization antiplatelet therapy is associated with a reduced incidence of acute lung injury: a population-based cohort study. Chest 139: 289–295, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37a. FDA Drug Safety Podcast for Healthcare Professionals: Voluntary market withdrawal of Xigris [drotrecogin alfa (activated)] due to failure to show a survival benefit (Online). http://www.fda.gov/Drugs/DrugSafety/DrugSafetyPodcasts/ucm277212 [4 November 2011].
- 38. Fierobe L, Brunet F, Dhainaut JF, Monchi M, Belghith M, Mira JP, Dall'ava-Santucci J, Dinh-Xuan AT. Effect of inhaled nitric oxide on right ventricular function in adult respiratory distress syndrome. Am J Respir Crit Care Med 151: 1414–1419, 1995 [DOI] [PubMed] [Google Scholar]
- 39. Frank JA, McAuley DF, Gutierrez JA, Daniel BM, Dobbs L, Matthay MA. Differential effects of sustained inflation recruitment maneuvers on alveolar epithelial and lung endothelial injury. Crit Care Med 33: 181–188, 2005 [DOI] [PubMed] [Google Scholar]
- 40. Freeman B. Free radical chemistry of nitric oxide. Looking at the dark side. Chest 105, Suppl 3: 79S–84S, 1994 [DOI] [PubMed] [Google Scholar]
- 41. Fullerton DA, McIntyre RC, Jr, Kirson LE, Cyr JA, Whitman GJ, Grover FL. Impact of respiratory acid-base status in patients with pulmonary hypertension. Ann Thorac Surg 61: 696–701, 1996 [DOI] [PubMed] [Google Scholar]
- 42. Gando S, Kameue T, Matsuda N, Hayakawa M, Morimoto Y, Ishitani T, Kemmotsu O. Imbalances between the levels of tissue factor and tissue factor pathway inhibitor in ARDS patients. Thromb Res 109: 119–124, 2003 [DOI] [PubMed] [Google Scholar]
- 43. Gerlach H, Keh D, Semmerow A, Busch T, Lewandowski K, Pappert DM, Rossaint R, Falke KJ. Dose-response characteristics during long-term inhalation of nitric oxide in patients with severe acute respiratory distress syndrome: a prospective, randomized, controlled study. Am J Respir Crit Care Med 167: 1008–1015, 2003 [DOI] [PubMed] [Google Scholar]
- 44. Ghofrani HA, Wiedemann R, Rose F, Schermuly RT, Olschewski H, Weissmann N, Gunther A, Walmrath D, Seeger W, Grimminger F. Sildenafil for treatment of lung fibrosis and pulmonary hypertension: a randomised controlled trial. Lancet 360: 895–900, 2002 [DOI] [PubMed] [Google Scholar]
- 45. Giacomini M, Borotto E, Bosotti L, Denkewitz T, Reali-Forster C, Carlucci P, Centanni S, Mantero A, Iapichino G. Vardenafil and weaning from inhaled nitric oxide: effect on pulmonary hypertension in ARDS. Anaesth Intensive Care 35: 91–93, 2007 [DOI] [PubMed] [Google Scholar]
- 46. Giaid A, Saleh D. Reduced expression of endothelial nitric oxide synthase in the lungs of patients with pulmonary hypertension. N Engl J Med 333: 214–221, 1995 [DOI] [PubMed] [Google Scholar]
- 47. Gillis CN, Pitt BR, Wiedemann HP, Hammond GL. Depressed prostaglandin E1 and 5-hydroxytryptamine removal in patients with adult respiratory distress syndrome. Am Rev Respir Dis 134: 739–744, 1986 [DOI] [PubMed] [Google Scholar]
- 48. Greene R, Lind S, Jantsch H, Wilson R, Lynch K, Jones R, Carvalho A, Reid L, Waltman AC, Zapol W. Pulmonary vascular obstruction in severe ARDS: angiographic alterations after i.v. fibrinolytic therapy. AJR Am J Roentgenol 148: 501–508, 1987 [DOI] [PubMed] [Google Scholar]
- 49. Griffiths MJ, Evans TW. Inhaled nitric oxide therapy in adults. N Engl J Med 353: 2683–2695, 2005 [DOI] [PubMed] [Google Scholar]
- 50. Griffiths MJ, Liu S, Curzen NP, Messent M, Evans TW. In vivo treatment with endotoxin induces nitric oxide synthase in rat main pulmonary artery. Am J Physiol 268: L509–L518, 1995 [DOI] [PubMed] [Google Scholar]
- 51. Gust R, McCarthy TJ, Kozlowski J, Stephenson AH, Schuster DP. Response to inhaled nitric oxide in acute lung injury depends on distribution of pulmonary blood flow prior to its administration. Am J Respir Crit Care Med 159: 563–570, 1999 [DOI] [PubMed] [Google Scholar]
- 52. Hidalgo A, Chang J, Jang JE, Peired AJ, Chiang EY, Frenette PS. Heterotypic interactions enabled by polarized neutrophil microdomains mediate thromboinflammatory injury. Nat Med 15: 384–391, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Hoeper MM, Granton J. ICU management of patients with severe pulmonary hypertension and right heart failure. Am J Respir Crit Care Med 184: 1114–1124, 2011 [DOI] [PubMed] [Google Scholar]
- 54. Hsu CW, Lee DL, Lin SL, Sun SF, Chang HW. The initial response to inhaled nitric oxide treatment for intensive care unit patients with acute respiratory distress syndrome. Respiration 75: 288–295, 2008 [DOI] [PubMed] [Google Scholar]
- 56. Hubloue I, Biarent D, Abdel Kafi S, Bejjani G, Mélot C, Naeije R, Leeman M. Endothelin receptor blockade in canine oleic acid-induced lung injury. Intensive Care Med 29: 1003–1006, 2003 [DOI] [PubMed] [Google Scholar]
- 58. Humbert M, Sitbon O, Simonneau G. Treatment of pulmonary arterial hypertension. N Engl J Med 351: 1425–1436, 2004 [DOI] [PubMed] [Google Scholar]
- 59. Huttemeier PC, Watkins WD, Peterson MB, Zapol WM. Acute pulmonary hypertension and lung thromboxane release after endotoxin infusion in normal and leukopenic sheep. Circ Res 50: 688–694, 1982 [DOI] [PubMed] [Google Scholar]
- 60. Idell S, Koenig KB, Fair DS, Martin TR, McLarty J, Maunder RJ. Serial abnormalities of fibrin turnover in evolving adult respiratory distress syndrome. Am J Physiol 261: L240–L248, 1991 [DOI] [PubMed] [Google Scholar]
- 61. Jardin F, Gueret P, Dubourg O, Farcot JC, Margairaz A, Bourdarias JP. Two-dimensional echocardiographic evaluation of right ventricular size and contractility in acute respiratory failure. Crit Care Med 13: 952–956, 1985 [DOI] [PubMed] [Google Scholar]
- 62. Jardin F, Vieillard-Baron A. Is there a safe plateau pressure in ARDS? The right heart only knows. Intensive Care Med 33: 444–447, 2007 [DOI] [PubMed] [Google Scholar]
- 63. Johnson D, Hurst T, Mayers I. Insufflated halothane increases venous admixture less than nitroprusside in canine atelectasis. Anesthesiology 77: 301–308, 1992 [DOI] [PubMed] [Google Scholar]
- 64. Kiefmann R, Heckel K, Schenkat S, Dorger M, Wesierska-Gadek J, Goetz AE. Platelet-endothelial cell interaction in pulmonary micro-circulation: the role of PARS. Thromb Haemost 91: 761–770, 2004 [DOI] [PubMed] [Google Scholar]
- 65. Kjaeve J, Bjertnaes LJ. Interaction of verapamil and halogenated inhalation anesthetics on hypoxic pulmonary vasoconstriction. Acta Anaesthesiol Scand 33: 193–198, 1989 [DOI] [PubMed] [Google Scholar]
- 66. Kuebler WM, Ying X, Singh B, Issekutz AC, Bhattacharya J. Pressure is proinflammatory in lung venular capillaries. J Clin Invest 104: 495–502, 1999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Kuhlen R, Walbert E, Frankel P, Thaden S, Behrendt W, Rossaint R. Combination of inhaled nitric oxide and intravenous prostacyclin for successful treatment of severe pulmonary hypertension in a patient with acute respiratory distress syndrome. Intensive Care Med 25: 752–754, 1999 [DOI] [PubMed] [Google Scholar]
- 68. Kuo C, Chen J. Effect of meconium aspiration on plasma endothelin-1 level and pulmonary hemodynamics in a piglet model. Biol Neonate 76: 228–234, 1999 [DOI] [PubMed] [Google Scholar]
- 69. Langleben D, DeMarchie M, Laporta D, Spanier AH, Schlesinger RD, Stewart DJ. Endothelin-1 in acute lung injury and the adult respiratory distress syndrome. Am Rev Respir Dis 148: 1646–1650, 1993 [DOI] [PubMed] [Google Scholar]
- 70. Lejeune P, De Smet JM, de Francquen P, Leeman M, Brimioulle S, Hallemans R, Melot C, Naeije R. Inhibition of hypoxic pulmonary vasoconstriction by increased left atrial pressure in dogs. Am J Physiol 259: H93–H100, 1990 [DOI] [PubMed] [Google Scholar]
- 71. Lejeune P, Vachiery JL, De Smet JM, Leeman M, Brimioulle S, Delcroix M, Melot C, Naeije R. PEEP inhibits hypoxic pulmonary vasoconstriction in dogs. J Appl Physiol 70: 1867–1873, 1991 [DOI] [PubMed] [Google Scholar]
- 72. Levi M, ten Cate H, van der Poll T. Endothelium: interface between coagulation and inflammation. Crit Care Med 30, Suppl 5: S220–S224, 2002 [DOI] [PubMed] [Google Scholar]
- 73. Ley K, Laudanna C, Cybulsky MI, Nourshargh S. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol 7: 678–689, 2007 [DOI] [PubMed] [Google Scholar]
- 74. Liu KD, Levitt J, Zhuo H, Kallet RH, Brady S, Steingrub J, Tidswell M, Siegel MD, Soto G, Peterson MW, Chesnutt MS, Phillips C, Weinacker A, Thompson BT, Eisner MD, Matthay MA. Randomized clinical trial of activated protein C for the treatment of acute lung injury. Am J Respir Crit Care Med 178: 618–623, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Liu SF, Crawley DE, Evans TW, Barnes PJ. Endogenous nitric oxide modulates adrenergic neural vasoconstriction in guinea-pig pulmonary artery. Br J Pharmacol 104: 565–569, 1991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Looney MR, Nguyen JX, Hu Y, Van Ziffle JA, Lowell CA, Matthay MA. Platelet depletion and aspirin treatment protect mice in a two-event model of transfusion-related acute lung injury. J Clin Invest 119: 3450–3461, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Looney MR, Su X, Van Ziffle JA, Lowell CA, Matthay MA. Neutrophils and their Fc gamma receptors are essential in a mouse model of transfusion-related acute lung injury. J Clin Invest 116: 1615–1623, 2206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Luhr O, Aardal S, Nathorst-Westfelt U, Berggren L, Johansson LA, Wahlin L, Frostell C. Pulmonary function in adult survivors of severe acute lung injury treated with inhaled nitric oxide. Acta Anaesthesiol Scand 42: 391–398, 1998 [DOI] [PubMed] [Google Scholar]
- 79. Maggiorini M, Melot C, Pierre S, Pfeiffer F, Greve I, Sartori C, Lepori M, Hauser M, Scherrer U, Naeije R. High-altitude pulmonary edema is initially caused by an increase in capillary pressure. Circulation 103: 2078–2083, 2001 [DOI] [PubMed] [Google Scholar]
- 80. Maniatis NA, Orfanos SE. The endothelium in acute lung injury/acute respiratory distress syndrome. Curr Opin Crit Care 14: 22–30, 2008 [DOI] [PubMed] [Google Scholar]
- 81. Marshall BE, Hanson CW, Frasch F, Marshall C. Role of hypoxic pulmonary vasoconstriction in pulmonary gas exchange and blood flow distribution. 2. Pathophysiology. Intensive Care Med 20: 379–389, 1994 [DOI] [PubMed] [Google Scholar]
- 82. Marshall BE, Marshall C, Frasch F, Hanson CW. Role of hypoxic pulmonary vasoconstriction in pulmonary gas exchange and blood flow distribution. 1. Physiologic concepts. Intensive Care Med 20: 291–297, 1994 [DOI] [PubMed] [Google Scholar]
- 83. Matthay MA, Ware LB. Plasma protein C levels in patients with acute lung injury: prognostic significance. Crit Care Med 32: S229–S232, 2004 [DOI] [PubMed] [Google Scholar]
- 84. McIntyre RC, Jr, Pulido EJ, Bensard DD, Shames BD, Abraham E. Thirty years of clinical trials in acute respiratory distress syndrome. Crit Care Med 28: 3314–3331, 2000 [DOI] [PubMed] [Google Scholar]
- 85. Mehta D, Malik AB. Signaling mechanisms regulating endothelial permeability. Physiol Rev 86: 279–367, 2006 [DOI] [PubMed] [Google Scholar]
- 86. Mekontso Dessap A, Charron C, Devaquet J, Aboab J, Jardin F, Brochard L, Vieillard-Baron A. Impact of acute hypercapnia and augmented positive end-expiratory pressure on right ventricle function in severe acute respiratory distress syndrome. Intensive Care Med 35: 1850–1858, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Mendoza CU, Suarez M, Castaneda R, Hernandez A, Sanchez R. Comparative study between the effects of total intravenous anesthesia with propofol and balanced anesthesia with halothane on the alveolar-arterial oxygen tension difference and on the pulmonary shunt. Arch Med Res 23: 139–142, 1992 [PubMed] [Google Scholar]
- 89. Meyer J, Theilmeier G, Van Aken H, Bone HG, Busse H, Waurick R, Hinder F, Booke M. Inhaled prostaglandin E1 for treatment of acute lung injury in severe multiple organ failure. Anesth Analg 86: 753–758, 1998 [DOI] [PubMed] [Google Scholar]
- 90. Meyrick B, Brigham KL. Repeated Escherichia coli endotoxin-induced pulmonary inflammation causes chronic pulmonary hypertension in sheep. Structural and functional changes. Lab Invest 55: 164–176, 1986 [PubMed] [Google Scholar]
- 91. Monchi M, Bellenfant F, Cariou A, Joly LM, Thebert D, Laurent I, Dhainaut JF, Brunet F. Early predictive factors of survival in the acute respiratory distress syndrome. A multivariate analysis. Am J Respir Crit Care Med 158: 1076–1081, 1998 [DOI] [PubMed] [Google Scholar]
- 92. Morelli A, Teboul JL, Maggiore SM, Vieillard-Baron A, Rocco M, Conti G, De Gaetano A, Picchini U, Orecchioni A, Carbone I, Tritapepe L, Pietropaoli P, Westphal M. Effects of levosimendan on right ventricular afterload in patients with acute respiratory distress syndrome: a pilot study. Crit Care Med 34: 2287–2293, 2006 [DOI] [PubMed] [Google Scholar]
- 93. Moudgil R, Michelakis ED, Archer SL. Hypoxic pulmonary vasoconstriction. J Appl Physiol 98: 390–403, 2005 [DOI] [PubMed] [Google Scholar]
- 94. Myers PR, Parker JL, Tanner MA, Adams HR. Effects of cytokines tumor necrosis factor alpha and interleukin 1 beta on endotoxin-mediated inhibition of endothelium-derived relaxing factor bioactivity and nitric oxide production in vascular endothelium. Shock 1: 73–78, 1994 [DOI] [PubMed] [Google Scholar]
- 95. Neff TA, Stocker R, Frey HR, Stein S, Russi EW. Long-term assessment of lung function in survivors of severe ARDS. Chest 123: 845–853, 2003 [DOI] [PubMed] [Google Scholar]
- 96. Ng J, Finney SJ, Shulman R, Bellingan GJ, Singer M, Glynne PA. Treatment of pulmonary hypertension in the general adult intensive care unit: a role for oral sildenafil? Br J Anaesth 94: 774–777, 2005 [DOI] [PubMed] [Google Scholar]
- 97. Nuckton TJ, Alonso JA, Kallet RH, Daniel BM, Pittet JF, Eisner MD, Matthay MA. Pulmonary dead-space fraction as a risk factor for death in the acute respiratory distress syndrome. N Engl J Med 346: 1281–1286, 2002 [DOI] [PubMed] [Google Scholar]
- 98. Orfanos SE, Armaganidis A, Glynos C, Psevdi E, Kaltsas P, Sarafidou P, Catravas JD, Dafni UG, Langleben D, Roussos C. Pulmonary capillary endothelium-bound angiotensin-converting enzyme activity in acute lung injury. Circulation 102: 2011–2018, 2000 [DOI] [PubMed] [Google Scholar]
- 99. Orfanos SE, Mavrommati I, Korovesi I, Roussos C. Pulmonary endothelium in acute lung injury: from basic science to the critically ill. Intensive Care Med 30: 1702–1714, 2004 [DOI] [PubMed] [Google Scholar]
- 100. Osman D, Monnet X, Castelain V, Anguel N, Warszawski J, Teboul JL, Richard C, French Pulmonary Artery Catheter Study Group Incidence and prognostic value of right ventricular failure in acute respiratory distress syndrome. Intensive Care Med 35: 69–76, 2009 [DOI] [PubMed] [Google Scholar]
- 101. Owings JT, Gosselin R. Acquired antithrombin deficiency following severe traumatic injury: rationale for study of antithrombin supplementation. Semin Thromb Hemost 23, Suppl 1: 17–24, 1997 [PubMed] [Google Scholar]
- 102. Palmer RM, Ashton DS, Moncada S. Vascular endothelial cells synthesize nitric oxide from L-arginine. Nature 333: 664–666, 1988 [DOI] [PubMed] [Google Scholar]
- 103. Penny DJ, Redington AN. Doppler echocardiographic evaluation of pulmonary blood flow after the Fontan operation: the role of the lungs. Br Heart J 66: 372–374, 1991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Phelan M, Perrine SP, Brauer M, Faller DV. Sickle erythrocytes, after sickling, regulate the expression of the endothelin-1 gene and protein in human endothelial cells in culture. J Clin Invest 96: 1145–1151, 1995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Powe JE, Short A, Sibbald WJ, Driedger AA. Pulmonary accumulation of polymorphonuclear leukocytes in the adult respiratory distress syndrome. Crit Care Med 10: 712–718, 1982 [DOI] [PubMed] [Google Scholar]
- 106. Radermacher P, Santak B, Wust HJ, Tarnow J, Falke KJ. Prostacyclin and right ventricular function in patients with pulmonary hypertension associated with ARDS. Intensive Care Med 16: 227–232, 1990 [DOI] [PubMed] [Google Scholar]
- 107. Ribes JA, Francis CW, Wagner DD. Fibrin induces release of von Willebrand factor from endothelial cells. J Clin Invest 79: 117–123, 1987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Romberg-Camps MJ, Korsten HH, Botman CJ, Bindels AJ, Roos AN. Right ventricular failure in acute respiratory distress syndrome. Neth J Med 57: 94–97, 2000 [DOI] [PubMed] [Google Scholar]
- 109. Rossi P, Persson B, Boels PJ, Arner A, Weitzberg E, Oldner A. Endotoxemic pulmonary hypertension is largely mediated by endothelin-induced venous constriction. Intensive Care Med 34: 873–880, 2008 [DOI] [PubMed] [Google Scholar]
- 110. Rubin DB, Wiener-Kronish JP, Murray JF, Green DR, Turner J, Luce JM, Montgomery AB, Marks JD, Matthay MA. Elevated von Willebrand factor antigen is an early plasma predictor of acute lung injury in nonpulmonary sepsis syndrome. J Clin Invest 86: 474–480, 1990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Sabharwal AK, Bajaj SP, Ameri A, Tricomi SM, Hyers TM, Dahms TE, Taylor FB, Jr, Bajaj MS. Tissue factor pathway inhibitor and von Willebrand factor antigen levels in adult respiratory distress syndrome and in a primate model of sepsis. Am J Respir Crit Care Med 151: 758–767, 1995 [DOI] [PubMed] [Google Scholar]
- 112. Saldeen T. Blood coagulation and shock. Pathol Res Pract 165: 221–252, 1979 [DOI] [PubMed] [Google Scholar]
- 113. Scarpati EM, Sadler JE. Regulation of endothelial cell coagulant properties. Modulation of tissue factor, plasminogen activator inhibitors, and thrombomodulin by phorbol 12-myristate 13-acetate and tumor necrosis factor. J Biol Chem 264: 20705–20713, 1989 [PubMed] [Google Scholar]
- 114. Schnells G, Voigt WH, Redl H, Schlag G, Glatzl A. Electron-microscopic investigation of lung biopsies in patients with post-traumatic respiratory insufficiency. Acta Chir Scand Suppl 499: 9–20, 1980 [PubMed] [Google Scholar]
- 115. Schwartz MD, Moore EE, Moore FA, Shenkar R, Moine P, Haenel JB, Abraham E. Nuclear factor-kappa B is activated in alveolar macrophages from patients with acute respiratory distress syndrome. Crit Care Med 24: 1285–1292, 1996 [DOI] [PubMed] [Google Scholar]
- 116. Shekerdemian LS, Penny DJ, Ryhammer PK, Reader JA, Ravn HB. Endothelin-A receptor blockade and inhaled nitric oxide in a porcine model of meconium aspiration syndrome. Pediatr Res 56: 353–358, 2004 [DOI] [PubMed] [Google Scholar]
- 117. Shiu YT, Udden MM, McIntire LV. Perfusion with sickle erythrocytes up-regulates ICAM-1 and VCAM-1 gene expression in cultured human endothelial cells. Blood 95: 3232–3241, 2000 [PubMed] [Google Scholar]
- 117a. Siddiqui S. Inhaled Prostacyclin for Adult Respiratory Distress Syndrome (ARDS) and Pulmonary Hypertension (Online). http://clinicaltrials.gov/ct2/show/NCT00314548 [12 March 2012].
- 118. Simonneau G, Robbins IM, Beghetti M, Channick RN, Delcroix M, Denton CP, et al. Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol 54, Suppl 1: S43–S54, 2009 [DOI] [PubMed] [Google Scholar]
- 119. Sinclair DG, Braude S, Haslam PL, Evans TW. Pulmonary endothelial permeability in patients with severe lung injury. Clinical correlates and natural history. Chest 106: 535–539, 1994 [DOI] [PubMed] [Google Scholar]
- 120. Snow RL, Davies P, Pontoppidan H, Zapol WM, Reid L. Pulmonary vascular remodeling in adult respiratory distress syndrome. Am Rev Respir Dis 126: 887–892, 1982 [DOI] [PubMed] [Google Scholar]
- 121. Squara P, Dhainaut JF, Artigas A, Carlet J. Hemodynamic profile in severe ARDS: results of the European Collaborative ARDS Study. Intensive Care Med 24: 1018–1028, 1998 [DOI] [PubMed] [Google Scholar]
- 122. Stamler JS, Loh E, Roddy MA, Currie KE, Creager MA. Nitric oxide regulates basal systemic and pulmonary vascular resistance in healthy humans. Circulation 89: 2035–2040, 1994 [DOI] [PubMed] [Google Scholar]
- 123. Steiner MK, Syrkina OL, Kolliputi N, Mark EJ, Hales CA, Waxman AB. Interleukin-6 overexpression induces pulmonary hypertension. Circ Res 104: 236–244, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Steudel W, Hurford WE, Zapol WM. Inhaled nitric oxide: basic biology and clinical applications. Anesthesiology 91: 1090–1121, 1999 [DOI] [PubMed] [Google Scholar]
- 125. Strieter RM, Kunkel SL. Acute lung injury: the role of cytokines in the elicitation of neutrophils. J Investig Med 42: 640–651, 1994 [PubMed] [Google Scholar]
- 126. Suntharalingam G, Regan K, Keogh BF, Morgan CJ, Evans TW. Influence of direct and indirect etiology on acute outcome and 6-month functional recovery in acute respiratory distress syndrome. Crit Care Med 29: 562–566, 2001 [DOI] [PubMed] [Google Scholar]
- 127. Taylor RW, Zimmerman JL, Dellinger RP, Straube RC, Criner GJ, Davis K, Jr, et al. Low-dose inhaled nitric oxide in patients with acute lung injury: a randomized controlled trial. JAMA 291: 1603–1609, 2004 [DOI] [PubMed] [Google Scholar]
- 128. Tomashefski JF., Jr Pulmonary pathology of acute respiratory distress syndrome. Clin Chest Med 21: 435–466, 2000 [DOI] [PubMed] [Google Scholar]
- 129. Tomashefski JF, Jr, Davies P, Boggis C, Greene R, Zapol WM, Reid LM. The pulmonary vascular lesions of the adult respiratory distress syndrome. Am J Pathol 112: 112–126, 1983 [PMC free article] [PubMed] [Google Scholar]
- 130. Tsokos M, Fehlauer F, Puschel K. Immunohistochemical expression of E-selectin in sepsis-induced lung injury. Int J Legal Med 113: 338–342, 2000 [DOI] [PubMed] [Google Scholar]
- 131. Uchiba M, Okajima K. Antithrombin III (AT III) prevents LPS-induced pulmonary vascular injury: novel biological activity of AT III. Semin Thromb Hemost 23: 583–590, 1997 [DOI] [PubMed] [Google Scholar]
- 132. Uchiba M, Okajima K, Murakami K, Johno M, Okabe H, Takatsuki K. Recombinant thrombomodulin prevents endotoxin-induced lung injury in rats by inhibiting leukocyte activation. Am J Physiol 271: L470–L475, 1996 [DOI] [PubMed] [Google Scholar]
- 133. van Heerden PV, Barden A, Michalopoulos N, Bulsara MK, Roberts BL. Dose-response to inhaled aerosolized prostacyclin for hypoxemia due to ARDS. Chest 117: 819–827, 2000 [DOI] [PubMed] [Google Scholar]
- 133a. Veterans Affairs Medical Center, Oklahoma City, OK Iloprost Effects on Gas Exchange and Pulmonary Mechanics (Online). http://clinicaltrials.gov/ct2/show/NCT01274481 [12 March 2012].
- 134. Vichinsky EP, Neumayr LD, Earles AN, Williams R, Lennette ET, Dean D, Nickerson B, Orringer E, McKie V, Bellevue R, Daeschner C, Manci EA. Causes and outcomes of the acute chest syndrome in sickle cell disease. National Acute Chest Syndrome Study Group. N Engl J Med 342: 1855–1865, 2000 [DOI] [PubMed] [Google Scholar]
- 135. Vieillard-Baron A, Schmitt JM, Augarde R, Fellahi JL, Prin S, Page B, Beauchet A, Jardin F. Acute cor pulmonale in acute respiratory distress syndrome submitted to protective ventilation: incidence, clinical implications, and prognosis. Crit Care Med 29: 1551–1555, 2001 [DOI] [PubMed] [Google Scholar]
- 136. Waerhaug K, Kirov MY, Kuzkov VV, Kuklin VN, Bjertnaes LJ. Recombinant human activated protein C ameliorates oleic acid-induced lung injury in awake sheep. Crit Care 12: R146, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Walmrath D, Schneider T, Pilch J, Grimminger F, Seeger W. Aerosolised prostacyclin in adult respiratory distress syndrome. Lancet 342: 961–962, 1993 [DOI] [PubMed] [Google Scholar]
- 138. Walther SM, Domino KB, Hlastala MP. Effects of posture on blood flow diversion by hypoxic pulmonary vasoconstriction in dogs. Br J Anaesth 81: 425–429, 1998 [DOI] [PubMed] [Google Scholar]
- 139. Ware LB, Eisner MD, Thompson BT, Parsons PE, Matthay MA. Significance of von Willebrand factor in septic and nonseptic patients with acute lung injury. Am J Respir Crit Care Med 170: 766–772, 2004 [DOI] [PubMed] [Google Scholar]
- 140. Ware LB, Fang X, Matthay MA. Protein C and thrombomodulin in human acute lung injury. Am J Physiol Lung Cell Mol Physiol 285: L514–L521, 2003 [DOI] [PubMed] [Google Scholar]
- 141. West J. Respiratory Physiology: The Essentials (7th ed.). Philadelphia, PA: Lippincott Williams & Wilkins, 2005 [Google Scholar]
- 142. West J. Pulmonary capillary stress failure. J Appl Physiol 89: 2483–2489, 2000 [DOI] [PubMed] [Google Scholar]
- 143. Wheeler AP, Bernard GR. Acute lung injury and the acute respiratory distress syndrome: a clinical review. Lancet 369: 1553–1564, 2007 [DOI] [PubMed] [Google Scholar]
- 144. Wort SJ, Evans TW. The role of the endothelium in modulating vascular control in sepsis and related conditions. Br Med Bull 55: 30–48, 1999 [DOI] [PubMed] [Google Scholar]
- 145. Wort SJ, Ito M, Chou PC, Mc Master SK, Badiger R, Jazrawi E, de Souza P, Evans TW, Mitchell JA, Pinhu L, Ito K, Adcock IM. Synergistic induction of endothelin-1 by tumor necrosis factor alpha and interferon gamma is due to enhanced NF-kappaB binding and histone acetylation at specific kappaB sites. J Biol Chem 284: 24297–24305, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146. Yoshida K, Kondo R, Wang Q, Doerschuk CM. Neutrophil cytoskeletal rearrangements during capillary sequestration in bacterial pneumonia in rats. Am J Respir Crit Care Med 174: 689–698, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Yuan XJ, Goldman WF, Tod ML, Rubin LJ, Blaustein MP. Hypoxia reduces potassium currents in cultured rat pulmonary but not mesenteric arterial myocytes. Am J Physiol 264: L116–L123, 1993 [DOI] [PubMed] [Google Scholar]
- 148. Zapol WM, Kobayashi K, Snider MT, Greene R, Laver MB. Vascular obstruction causes pulmonary hypertension in severe acute respiratory failure. Chest 71, Suppl 2: 306–307, 1977 [DOI] [PubMed] [Google Scholar]
- 149. Zapol WM, Snider MT. Pulmonary hypertension in severe acute respiratory failure. N Engl J Med 296: 476–480, 1977 [DOI] [PubMed] [Google Scholar]
- 150. Zarbock A, Singbartl K, Ley K. Complete reversal of acid-induced acute lung injury by blocking of platelet-neutrophil aggregation. J Clin Invest 116: 3211–3219, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Zwissler B, Kemming G, Habler O, Kleen M, Merkel M, Haller M, Briegel J, Welte M, Peter K. Inhaled prostacyclin (PGI2) versus inhaled nitric oxide in adult respiratory distress syndrome. Am J Respir Crit Care Med 154: 1671–1677, 1996 [DOI] [PubMed] [Google Scholar]