Abstract
Adhesion of parasitized erythrocytes to post-capillary venular endothelium1 or uninfected red cells2-4 is strongly implicated in the pathogenesis of severe Plasmodium falciparum malaria. Neoantigens at the infected red-cell surface adhere to a variety of host receptors5-9, demonstrate serological diversity in field isolates10,11 and may also be a target of the host-protective immune response12. Here we use sequential cloning of P. falciparum by micromanipulation to investigate the ability of a parasite to switch antigenic and cytoadherence phenotypes. Our data show that antigens at the parasitized cell surface undergo clonal variation in vitro in the absence of immune pressure at the rate of 2% per generation with concomitant modulations of the adhesive phenotype. A clone has the potential to switch at high frequency to a variety of antigenic and adhesive phenotypes, including a new type of cytoadherence behaviour, ‘auto-agglutination’ of infected erythrocytes. This rapid appearance of antigenic and functional heterogeneity has important implications for pathogenesis and acquired immunity.
During studies on cytoadherence of infected erythrocytes we selected various phenotypes from a P.falciparum line IT 4/25/5. Parasites were selected for increased binding to C32 amelanotic melanoma cells, endothelial cells or rosetting of uninfected erythrocytes (Fig. 1). We noticed that selections for endothelial binding and rosetting were associated with changes in antigenic type. To investigate the relationship between antigenic and adhesive phenotypes and to study antigenic variation in the absence of selection pressure, we carried out a series of clonings by micromanipulation.
FIG. 1.
Tree showing derivation of the P. falciparum clones from IT 4/25/5. Clones derived by micromanipulation are shown in the bold boxes. Antigenically indistinguishable clones are boxed together. R29 was cloned from ITOR and A4R derived from A4 by 8 rounds of selection for rosetting. METHODS. Parasites were grown as described25,26 in 95% N2, 1% O2, 4% CO2 using RPMI 1640 culture medium (Gibco) supplemented with 20 mM HEPES, 20 mM glucose, 5 mM glutamine and gentamycin, and 10% pooled serum from blood donors (Blood Transfusion Service, Oxford). During experiments comparing the antigenic phenotype, the cytoadherence characteristics and surface radiolabelling, clones were grown in red blood cells from the same donor and in the same pool of serum. Rosetting parasites were selected for by sedimentation through ‘plasma gel’ (Laboratoire Roger Bellon, Neuilly, France)27. Parasites were selected for binding to C32 melanoma cells or HUVEC as previously described 9,24. Cloning was performed using a micromanipulator (Micro Instruments, Oxford), sterile micropipettes of 5 μm internal diameter drawn from capillary tubes and a binocular microscope. Parasitized cells containing single trophozoites were picked from a settled monolayer by applying an aspiration pressure of 1 cm of water, transferred into a Petri dish containing supplemented RPMI 1640 and grown as above. To exclude cross-contamination, a Southern blot of Hinfl-digested DNA from the clones and other laboratory lines was hybridized with the probe pC4. H32 labelled with 32P (Amersham) using a Quickprime labelling kit (Pharmacia). The autoradiograph showed that no cross-contamination had occurred (data not shown).
Four clones (A1-4) were derived from the endothelial binding line IT04, three of which (A2-4) were antigenically indistinguishable from the parent. A4 was subcloned, yielding 21 ‘C’ clones (Fig. 1). Cross-contamination was excluded by hybridization with the probe pC4.H32 (ref. 24). All clones gave an identical fingerprinting pattern which was different from that of other parasite lines (data not shown).
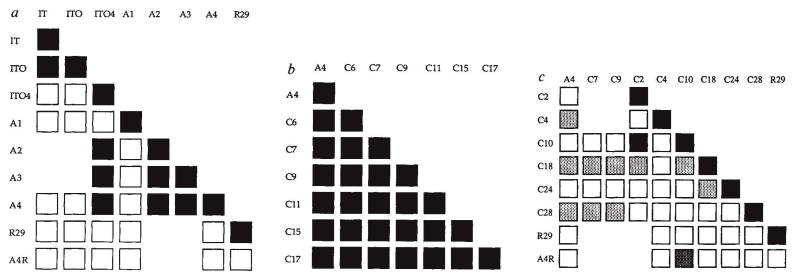
We analysed the antigenic phenotype of these clones (Fig. 2). Ten of the 21 C clones were of the same variant antigenic type as A4, but the remainder were antigenically distinct. Of six variant C clones analysed for antigenic similarity with each other, only two (C2 and C10) were antigenically indistinguishable, indicating that in P.falciparum antigenic variation is highly diverse (Fig. 2); it is also rapid. The proportion of variant C clones (11/21 or 0.52) reflects the antigenic heterogeneity within the A4 clone at the time of subcloning, provided cloning itself does not influence switching. Since 30 cycles elapsed from cloning A4 to the C subcloning, new antigenic types had appeared, on average, at 2.4% per generation, assuming equal growth rates for all variants. ((1 − r)n = p, where r is the switching rate per generation, n is the number of generations and p the proportion of the original antigenic type remaining after n generations.) This antigenic switching rate is of a similar order of magnitude to that recently observed in Trypanosoma brucei14.
FIG. 2.
Antigenic analysis of the clones. The antigenic differences between a, IT (clone IT 4/25/5), IT0, IT04, A1-4, R29 and A4R; b, A4 and C clones with the same variant antigenic type as A4; c, A4, C7, C9, variant C clones, R29 and A4. Key: ■, 100 to 80%; square with dark shading, 79 to 15%; lightly shaded squares, 15 to 5%; and □, 5 to 0% similarity between test clones using the mixed agglutination assay. A further four C clones had the same variant type as A4, and the remaining five C clones had a different antigenic type from A4 (data not shown).
METHODS. The antigenic phenotype of the derived clones was determined by the mixed agglutination assay (C.I.N. et al., submitted). In brief, the two clones to be tested were labelled independently with either ethidium bromide (200 μg ml−1) or DAPI (4, 6-diamidino-2-methylphenylindole) (20 μg ml−1) for 10 min at 37°C, and washed three times in RPMI 1640. Labelled trophozoites (2.5 × 106) were taken from each of the two cultures and resuspended in 70 μl pooled, heat-inactivated, hyperimmune serum and 140 μl serum-free RPMI 1640. The tube was rotated at 10 r.p.m. at room temperature for 1 h. A drop of the suspension was placed on a microscope slide and sealed with a greased coverslip. Agglutinates were examined using a Leitz epifluorescence microscope with an G365 excitation filter, an FT 395 beam splitter and an LP 420 barrier filter. Agglutinates containing 5 to 10 cells were scored for whether the agglutinate contained all red cells (stained with ethidium bromide), all blue cells (stained with DAPI) or a mixture of red and blue cells. 100 agglutinates were counted in two or more separate experiments for each pair of clones tested. The proportion of mixed-colour agglutinates is a function of the similarity of the antigenic phenotype expressed at the red blood cell surface of the two clones. The distribution of the types of agglutinate can be predicted by the binomial distribution (D.J.R., unpublished observations). If the chance of homologous agglutination is p, then for an agglutinate of size n the chance of the agglutinate being of a single colour is pn−1, and the proportion of mixed-colour agglutinates 1 − pn−1. As equal numbers of parasites from the two clones are used in the test, the ratio of chance of heterologous agglutination/chance of homologous agglutination or 1−p/p is a measure of the degree of similarity between the clones. For example, assuming all agglutinates were of size 5, and the clones were antigenically indistinguishable (p =0.5), the proportion of mixed agglutinates would be 0.94 and the degree of similarity would be 0.5/0.5=1. Similarly, mixed-colour agglutinate proportions of 0.40 and 0.20 in this test represent a degree of similarity between the two clones under test of 0.15 (15%) and 0.05 (5%), respectively.
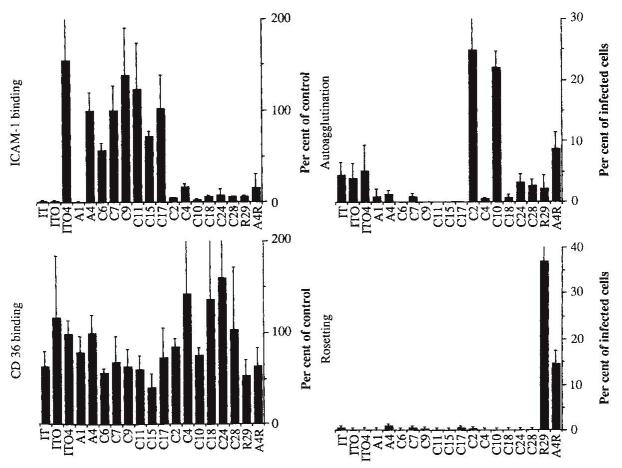
Different isolates of P.falciparum adhere to CD36 and thrombospondin5-8, intercellular adhesion molecule-1 (ICAM-1) (ref. 9) or rosette uninfected cells2,3. Clone A4 binds to purified CD36 and ICAM-1 but does not rosette. Six C clones with the same antigenic type as A4 had the same cytoadherent phenotype as A4. But six C clones with a different antigenic type from A4 had a different adhesive phenotype. They showed similar binding to CD36, but greatly reduced binding to ICAM-1 compared with A4 (Fig. 3).
FIG. 3.
The cytoadherence phenotype of the clones. Adherence of infected red cells to a, purified CD36; b, an ICAM-1–Fc chimaera; c, rosetting of uninfected by infected red cells, and d, auto-agglutination of infected red cells. Binding to CD36 and to the ICAM-1–Fc chimaera is given as the average ± s.d. of two experiments using 4 determinations on each occasion. The percentage of infected erythrocytes rosetting is the mean ± s.d. for 500 infected cells on four occasions, and for auto-agglutination is the mean ± s.d. counting 1,000 infected cells in four determinations.
METHODS. CD36 was isolated from human platelets28 and an ICAM-1–Fc chimaera prepared by transient expression in COS cells and purification on protein G-Sepharose29. A solution of CD36 (3 μl; 1 μg ml−1) or ICAM-1–Fc (3 μl; 10 μg ml−1) were adsorbed on to a plastic dish (Falcon no. 1005) in a moist atmosphere at 4 °C overnight. The spots were aspirated and the dish washed five times with binding medium (RPMI, HEPES 20 mM, pH 6.9) before blocking with 0.5% BSA/RPMI 1640 for 4 h at 37 °C and washing 5 times in binding medium. A suspension of trophozoites (5 ml) in binding medium at 1% haematocrit and between 1 and 2% parasitaemia was placed in the dish, incubated at 37 °C, resuspending the red cells every 10 min. This was repeated five times. Unbound cells were removed from the dish by careful washing. The adherent cells were fixed with 1% glutaraldehyde, stained with Giemsa and the number of cells per high power field counted. The counts were normalized to the binding of A4 clone for both CD36 and ICAM-1 binding in each experiment. Rosetting was measured by counting the number of trophozites rosetting two or more uninfected red cells immediately after removing a sample from the culture flask. The percentage of trophozites agglutinating with other infected cells (that is, auto-agglutinating) was counted after 200 μl culture at 10% haematocrit was mixed at 10 r.p.m. for 1 h.
Neither A4 nor any C clone showed significant rosetting of uninfected erythrocytes. A rosetting line, A4R, could however be derived from A4 by selection. A4R was antigenically distinct from both A4 and all the other C clones. The variety of adhesive phenotypes that can be generated within a clone was underlined by finding a novel adhesive phenotype in two of the variant C clones. These clones, C2 and C10, showed auto-agglutination of mature infected, but not uninfected, erythrocytes in serum from donors never exposed to malaria. This phenotype has also been observed in field isolates (our unpublished obervations). Rosetting and auto-agglutination are distinct phenotypes; the auto-agglutinating clones do not rosette and a freshly cloned rosetting parasite (R29) does not auto-agglutinate (Fig. 3). This distinction may be important as the relative pathological importance of the various adhesive phenotypes has not been established. Auto-agglutination in vivo could lead to the focal accumulation of infected erythrocytes and so contribute to the pathogenesis of severe malaria.
Clonal antigenic variation is an established mechanism of immune evasion for a variety of pathogens15-20. It is distinguished from antigenic diversity generated by somatic mutation by the retention of a copy of each gene. This permits the subsequent appearance of previously expressed antigenic phenotypes15. It has recently been shown that a single clonal antigenic switch occurred in an antigen expressed at the surface of P. falciparurn-infected erythrocytes21. To determine whether the capacity to express a previous phenotype was retained after switching, we selected a variant clone, C28, for ICAM-1 binding. After a single round of selection, the new line, C28-I, had regained ICAM-1 binding and showed increased antigenic similarity to A4, but was now antigenically distinct from C28 (Fig. 4).
FIG. 4.
The relative a, antigenic and b, adhesive phenotypes of the parental clone A4, progeny clone C28 and line C28-1 derived from C28 by selection once on ICAM-1. Key: ■, 100 to 80%; shaded square, 79 to 25%; and □, 15 to 0% similarity between clones using the mixed agglutination assay. Binding to CD36 and to the ICAM-1–Fc chimaera are given as the average ± s.d. of four determinations. Results of a representative experiment are shown.
METHODS. ICAM-1–Fc chimaera (50 μg ml−1 in PBS/gentamycin) was adsorbed on to a plastic dish (Falcon no. 1005) in a moist atmosphere at 37 °C for 2 h. The solution was removed and the dish blocked with 2% BSA/PBS/gentamycin for 2 h at 37 °C and finally wahed three times in binding medium. A suspension of late trophozoites (1.25 ml) purified by gelatin flotation27 was allowed to settle on the dish for 1 h before the unbound parasites were washed off. Erythrocytes (50 μl) were added to the bound cells in culture medium, and the dish placed in a gas jar at 37 °C overnight. Red cells were collected the next day and cultured as described (Fig. 1). The mixed agglutination and cytoadherence assays were done as described.
Clinical immunity to malaria has been linked to antibody responses directed against the infected red-cell surface12. Although antigenic variation in vivo would permit chronic infection, exposure to multiple variant antigenic types generated by the diverse pattern of switching we have observed may prime the immune system for the rapid control of future heterologous challenges. This would explain how protective immunity can be achieved after relatively few clinical attacks of malaria12,22, despite the large repertoire of variant antigenic types10,11.
Changes in antigenic23 or cytoadherence phenotypes24 have been associated with a family of high-molecular-weight proteins expressed at the red-cell surface. We have found that antigenic variation is also accompanied by size changes in these proteins (data not shown). Thus the biochemical, antigenic and adhesive phenotypes are all co-modulated by clonal antigenic variation and so these properties will not be stable characteristics of an isolate, but will be determined by the interaction between the parasite genotype and the host environment. The same parasite infecting different individuals may therefore give rise to a variety of phenotypes depending on the selective pressures exerted by the host. These pressures could include constitutive or induced levels of endothelial or red-cell surface receptor expression and the speed and effectiveness of the variant specific antibody response, Furthermore, in view of the apparent linkage between antigenic and adhesive phenotypes (Figs 2 and 3) and the large antigenic repertoire10,11, additional host receptors may exist. If severe disease is associated with specific adhesive phenotypes, such as rosetting, auto-agglutination or ICAM-1 binding, characterizing the antigenic determinants of these pathogenic subsets of the total repertoire of variant antigenic types may form the basis of a realistic strategy to control severe disease.
Note added in proof: Data linking antigenic and cytoadherence phenotypes of p. falciparum IT derived clones has also been obtained by Biggs et al.30.
ACKNOWLEDGEMENTS
We thank K. Robson for DNA samples from the FCB, 3D7 and D4 laboratory lines; R. Howard for IT 4/25/5; T. Wellems for the probe pC4.H32; D. Ferguson for blood donations; B. Elford. C. Mulholland and R. Allan for help with growing clones; G. Brown, D. Weatherall, C. Bangham and S. Kyes for discussion and E. Piza for support. We thank the MRC, the Wellcome Trust. the Lister Institute for Preventive Medicine and the UNDP/World Bank/WHO special programme for Research and Training in Tropical Diseases for financial support. D.J.R. is a Medical Research Council Training Fellow and A.RB. is a Lister Institute Research Fellow.
References
- 1.MacPherson GG, Warrell MJ, White NJ, Looareesuran S, Warrell DA. Am. J. Path. 1985;119:385–401. [PMC free article] [PubMed] [Google Scholar]
- 2.David PH, Handunetti SM, Leech JH, Gamage P, Mendis KM. Am. J. trop. Med. Hyg. 1988;38:289–297. doi: 10.4269/ajtmh.1988.38.289. [DOI] [PubMed] [Google Scholar]
- 3.Udomsangpetch R, et al. J. exp. Med. 1989;169:1835–1840. doi: 10.1084/jem.169.5.1835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Carlson J, et al. Lancet. 1990;336:1457–1460. doi: 10.1016/0140-6736(90)93174-n. [DOI] [PubMed] [Google Scholar]
- 5.Barnwell JW, et al. J. clin. Invest. 1989;84:765–772. doi: 10.1172/JCI114234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ockenhouse CF, Tandon NN, Magowan C, Jamieson GA, Chulay JO. Science. 1989;243:1469–1471. doi: 10.1126/science.2467377. [DOI] [PubMed] [Google Scholar]
- 7.Oquendo P, Hundt E, Lawler J, Seed B. Cell. 1989;58:95–101. doi: 10.1016/0092-8674(89)90406-6. [DOI] [PubMed] [Google Scholar]
- 8.Roberts DD, et al. Nature. 1985;318:64–66. doi: 10.1038/318064a0. [DOI] [PubMed] [Google Scholar]
- 9.Berendt AR, Simmons DL, Tansey J, Newbold CI, Marsh K. Nature. 1989;341:57–59. doi: 10.1038/341057a0. [DOI] [PubMed] [Google Scholar]
- 10.Marsh K, Howard RJ. Science. 1988;231:150–153. doi: 10.1126/science.2417315. [DOI] [PubMed] [Google Scholar]
- 11.Forsyth KP, et al. Am. J. trop. Med. Hyg. 1989;41:259–267. [PubMed] [Google Scholar]
- 12.Marsh K, Otoo L, Hayes RJ, Carson DC, Greenwood BM. Trans. R. Soc. trop. Med. 1989;83:293–303. doi: 10.1016/0035-9203(89)90478-1. [DOI] [PubMed] [Google Scholar]
- 13.Sinnis P, Wellems TE. Genomics. 1988;3:287–295. doi: 10.1016/0888-7543(88)90117-6. [DOI] [PubMed] [Google Scholar]
- 14.Turner CMR, Barry JD. Parasitology. 1989;99:67–75. doi: 10.1017/s0031182000061035. [DOI] [PubMed] [Google Scholar]
- 15.Borst P, Greaves DR. Science. 1987;235:658–667. doi: 10.1126/science.3544215. [DOI] [PubMed] [Google Scholar]
- 16.Brown KN, Brown IN. Nature. 1965;208:1286–1288. doi: 10.1038/2081286a0. [DOI] [PubMed] [Google Scholar]
- 17.Barnwell JR, Howard RJ, Coon HG, Miller LH. Infect. Immun. 1983;40:985–994. doi: 10.1128/iai.40.3.985-994.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McLean SA, Pearson CD, Phillips RS. Expl Parasitol. 1986;54:296–302. doi: 10.1016/0014-4894(82)90038-8. [DOI] [PubMed] [Google Scholar]
- 19.Handunnetti SM, Mendis KN, David PD. J. exp. Med. 1987;165:1269–1283. doi: 10.1084/jem.165.5.1269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gilks CF, Walliker D, Newbold CI. Parasite Immun. 1990;12:45–64. doi: 10.1111/j.1365-3024.1990.tb00935.x. [DOI] [PubMed] [Google Scholar]
- 21.Biggs BA, et al. Proc. natn. Acad. Sci. U.S.A. 1991;88:9171–9174. doi: 10.1073/pnas.88.20.9171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Baird JK, et al. Am. J. trop. Med. Hyg. 1991;45:65–76. doi: 10.4269/ajtmh.1991.45.65. [DOI] [PubMed] [Google Scholar]
- 23.Leech JH, Barnwell JW, Miller LH, Howard RJ. J. exp. Med. 1984;159:1587–1575. doi: 10.1084/jem.159.6.1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Magowan C, Woolish W, Anderson L, Leech J. J. exp. Med. 1988;188:1307–1319. doi: 10.1084/jem.168.4.1307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Trager W, Jensen JB. Science. 1976;193:673–675. doi: 10.1126/science.781840. [DOI] [PubMed] [Google Scholar]
- 26.Haynes JD, Diggs CL, Hines FA, Desjardins RE. Nature. 1976;263:767–769. doi: 10.1038/263767a0. [DOI] [PubMed] [Google Scholar]
- 27.Jensen JB. Am. J. trop. Med Hyg. 1978;27:1274–1280. doi: 10.4269/ajtmh.1978.27.1274. [DOI] [PubMed] [Google Scholar]
- 28.Tandon NN, Lipsky RH, Burgess WH, Jamieson GA. J. biol. Chem. 1989;264:7570–7575. [PubMed] [Google Scholar]
- 29.Berendt AR, et al. Cell. 1992;68:71–81. doi: 10.1016/0092-8674(92)90207-s. [DOI] [PubMed] [Google Scholar]
- 30.Biggs BA, et al. J. Immun. in the press. [Google Scholar]