Abstract
Over the past decade, the ability to culture and differentiate human embryonic stem cells (ESCs) has offered researchers a novel therapeutic that may, for the first time, repair regions of the damaged heart. Studies of cardiac development in lower organisms have led to WKHidentification of the transforming growth factor-β superfamily (eg, activin A and bone morphogenic protein 4) and the Wnt/β-catenin pathwayV as key inducers of mesoderm and cardiovascular differentiation. These factors act in a context-specific manner (eg, Wnt/β-catenin is required initially to form mesoderm but must be antagonized thereafter to make cardiac muscle). Different lines of ESCs produce different levels of agonists and antagonists for these pathways, but with careful optimization, highly enriched populations of immature cardiomyocytes can be generated. These cardiomyocytes survive transplantation to infarcted hearts of experimental animals, where they create new human myocardial tissue and improve heart function. The grafts generated by cell transplantation have been small, however, leading to an exploration of tissue engineering as an alternate strategy. Engineered tissue generated from preparations of human cardiomyocytes survives poorly after transplantation, most likely because of ischemia. Creation of pre-organized vascular networks in the tissue markedly enhances survival, with human capillaries anastomosed to the host coronary circulation. Thus, pathways controlling formation of the human cardiovascular system are emerging, yielding the building blocks for tissue regeneration that may address the root causes of heart failure.
Keywords: Cardiomyocytes, Human embryonic stem cells, Myocardial infarction, Tissue engineering, Wnt signaling
The course of events after myocardial ischemia is: initiation of high-energy phosphate depletion within seconds, loss of contractility within 1 min, the beginning of irreversible cardiomyocyte injury within 20 min and the onset of microvascular injury within 1 h.1 Infarcted myocardium manifests necrosis by light microscopy within 24 h, followed by waves of acute and chronic inflammation that remove the necrotic tissue. Granulation tissue, the provisional tissue of wound repair, forms concomitantly with clearance of necrotic myocardium and by 6–8 weeks, the human infarct transitions into dense collagenous scar.2 This process causes an irreversible loss of contractile function in the ischemic region, decreased heart function and, depending on the location and degree of ischemia, heart failure.3 Pharmaceuticals and cardiac rehabilitation can improve heart function, but if discontinued, the heart’s function will typically deteriorate. The persistent long-term decreased heart function is related to the adult human heart’s lack of a significant regenerative capacity.4 Currently, the only long-term method for replacing dead cardiomyocytes is heart transplantation, a procedure severely limited by cost and the shortage of donor hearts.
Over the past 15 years, cell-based therapy has been actively pursued as a means to repair the damaged heart. Success in cell therapy requires the identification of a cell that is capable of differentiating into a cardiomyocyte, can electromechanically integrate into a damaged region and, ultimately, restore normal heart function. Our group, as well as others, originally focused on remuscularization using skeletal myoblasts.5 The benefit of this cell population, however, is limited, because skeletal muscle is unable to form the appropriate gap junctions for electrical coupling, which can lead to arrhythmogenesis.6,7 A diverse number of noncardiac cell types have also been assessed for their ability to regenerate myocardium, including bone marrow-derived stem cells,8,9 adipose-derived stem cells,10 resident cardiac stem cells,11 endothelial cells,12 and umbilical cord blood.13 Although many studies have demonstrated a functional benefit from these transplanted stem cells, they have demonstrated only a very limited number of de novo cardiomyocytes, which suggests that these different cell types may work through as-yet undiscovered paracrine factors rather than via true regenerative mechanisms.
Based on the limited ability of adult stem cells to remuscularize the heart, we turned our focus to pluripotent embryonic stem cells (ESCs). In 1998, Thompson et al demonstrated a method of culturing human ESCs (hESCs),14 which have an unquestioned ability to differentiate into any cell type. Furthermore, hESCs are expandable, allowing scalable expansion to replace the billion or so myocytes lost in a typical infarct.15 Understanding the biology of hESCs will help identify the factors required to determine a cardiovascular fate and may serve as a platform for future therapy. In this invited paper, we review our group’s experiences deriving cardiomyocytes from hESCs, describing the molecular basis for ESC differentiation and our efforts to use these cells to repair the infarcted heart. Although our charge was to focus on work from our own laboratory, we briefly highlight other groups as space permits. Readers interested in a broader overview of the field are referred to several recent comprehensive reviews.16–19
Embryonic Stem Cell Pluripotency: A Double-Edged Sword
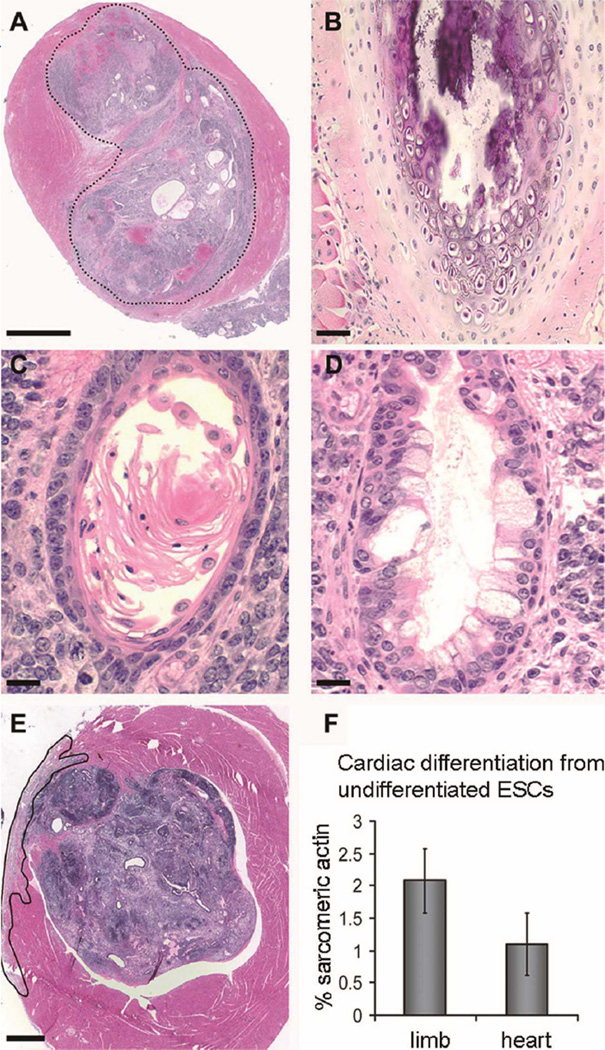
The ability of ESCs to become any cell type in the body makes them extremely useful for tissue regeneration, but it also makes them challenging to control. Initial studies attempted to introduce undifferentiated mouse ESCs directly into the myocardium, with the hope that local cues, such as signaling molecules, extracellular matrix or biomechanical stresses, would, independently, be sufficient to signal a cardiac fate and improve heart function post infarction.20,21 Although having an instructive cardiac environment would greatly simplify the therapeutic potential of ESCs, both our group22 and others23 found no preferential cardiomyocyte differentiation after transplantation into the heart. Instead, we consistently found teratomas within 3 weeks of transplanting undifferentiated mouse ESCs into the uninjured or infarcted mouse heart (Figures 1A–E). Furthermore, there was no enrichment for cardiomyocyte differentiation of ESCs transplanted into the left ventricle vs those transplanted into hindlimb skeletal muscle (Figure 1F). Finally, we found that teratomas from allogeneic ESCs were rejected unless the hosts were immunocompromised, indicating no immune privilege for ESC progeny. These studies demonstrate that more differentiated cells will be required to specify a cardiomyocyte fate and to minimize the risk of tumor formation; furthermore, immunosuppression may be necessary for allogeneic transplantation to succeed clinically.
Figure 1.
Formation of teratomas after transplanting undifferentiated mouse embryonic stem cells (mESCs) into a mouse heart. (A) Uninjured heart 3 weeks after mESC injection. A complex tumor (outlined by dotted lines) has replaced much of the ventricular wall and obliterated the lumen at this apical level (scale bar 200µ m). Tissue representative of all 3 germ layers is present (see B–F). (B) Mesoderm-derived cartilage and bone. (C) Ectoderm-derived keratinizing squamous epithelium. (D) Endodermderived gut epithelium with goblet cells. (Scale bar B–D, 10µ m.) (E) Infarcted heart 3 weeks after mESCs were injected at the time of coronary occlusion. A teratoma is present within the wall and growing endoluminally (scale bar 200µ m). (F) Histomorphometry demonstrates no increase in cardiomyocyte differentiation within heart teratomas when compared with teratomas from the mouse hind limb that were derived from embryoid bodies, indicating the lack of a cardiogenic environment in either the normal or injured heart.
Controlling Cardiac Differentiation With Transforming Growth Factor (TGF)-β and Wnt Family Signaling
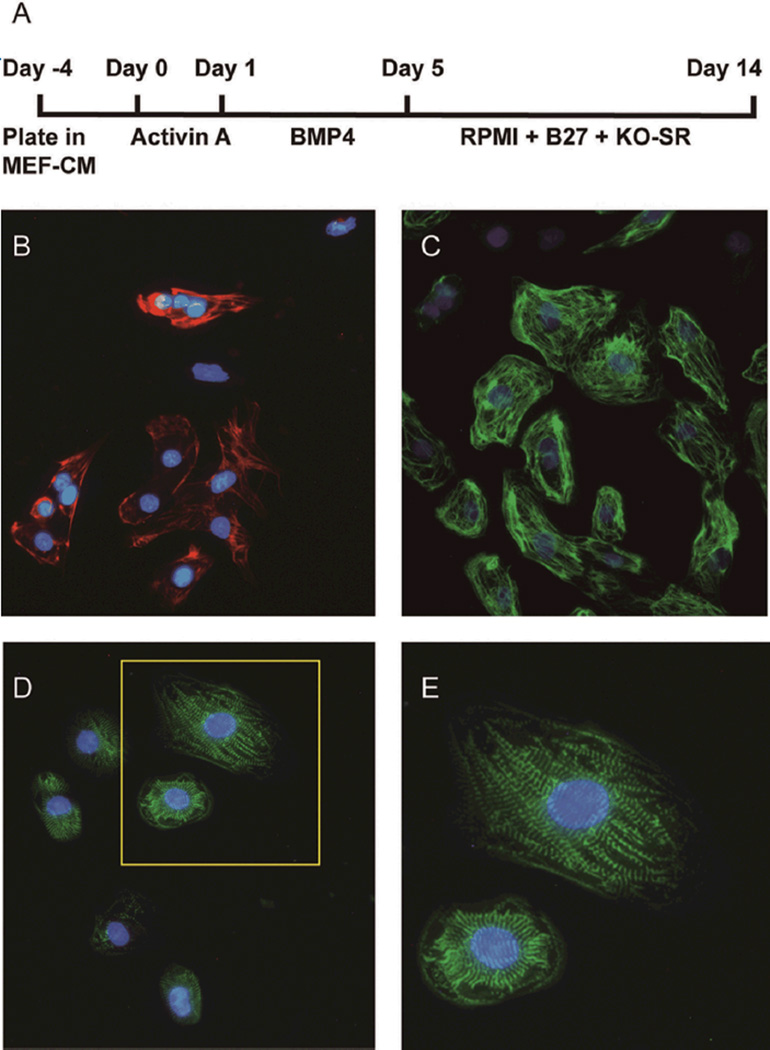
The inability of the cardiac milieu to direct ESC specification demonstrated the need to further understand the signals that induce cardiomyocyte differentiation. The original method for generating cardiomyocytes from human ESCs involved forming 3-dimensional aggregates called embryoid bodies (EBs), followed by induction of differentiation with serum. This system would create immature cardiomyocytes at a frequency of <1%,24 also giving rise to cells from all 3 germ layers. In the 1990s, a series of reports demonstrated the role of TGF-β superfamily proteins in the induction of mesoderm specification and subsequent cardiac differentiation in Xenopus and chick embryo development. Prior to gastrulation the hypoblast induces mesoderm from the primitive streak with activin or nodal signaling.25 Further induction with bone morphogenic proteins (BMPs) is able to induce cardiac differentiation.26 Drawing from this information, our group, in collaboration with scientists from the Geron Corporation, assessed these developmental cues in a hESC culture system. The H7 line of hESCs was grown as a high-density monolayer and treated with activin A (100 ng/ml) for 24 h followed by BMP4 (10 ng/ml) for 4 days. Spontaneous beating was observed at day 12, with consistent yields of >30% cardiomyocytes. If these cells were subsequently separated in a Percoll density gradient, a cardiomyocyte population of >80% could be reproducibly achieved (Figures 2A–E).27 A similar protocol developed by the Keller Laboratory again used activin A and BMP4 to induce mesoderm. However, their protocol induced differentiation in EBs grown under hypoxic conditions. The Keller Protocol also included treating the cells at the mesodermal stage with a canonical WNT inhibitor, dikkopf homolog 1 (Dkk1, see later) and the addition of vascular endothelial growth factor.28 Though there are differences between the 2 protocols, they both demonstrate that TGF-β family signaling is key to controlling mesoderm induction and cardiac differentiation from hESCs.
Figure 2.
Protocol for directed differentiation of cardiomyocytes from human embryonic stem cells (hESCs) in high-density monolayers. (A) Mesodermal fate is induced by activin A signaling. Once enriched for mesoderm, BMP4 treatment increases the percentage of spontaneously contracting cardiomyocytes. (B–E) Monolayer embryonic stem cells treated with activin A/BMP4. Immunocytochemical analyses demonstrate cardiomyocyte structural proteins: cardiac troponin T (B, red); β-MHC (C, green); α actinin (D and E, green). Panel E is a higher magnification of the insert in panel D, demonstrating sarcomeric structures. Nuclei were stained with DAPI (blue) in all panels. MHC, myosin heavy chain; BMP, bone morphogenic protein.
At the time of those studies, the role of Wnt/β-catenin signaling in cardiomyocyte differentiation was less clear. Various studies suggested this pathway both induces and represses cardiomyocyte differentiation. Data from Xenopus and chick development showed that overexpression of Wnts in the pre-cardiac mesoderm suppresses heart induction and instead forms primitive erythrocytes.29 Furthermore, inhibition of Wnt induces ectopic heart formation in the noncardiac mesoderm in chick embryos.30 Although this would suggest Wnt directs stem cells away from a cardiac specification, a separate study demonstrated Wnt/β-catenin signaling enhances rather than represses cardiac differentiation by inducing lateral mesoderm formation, a tissue that gives rise to the heart.31
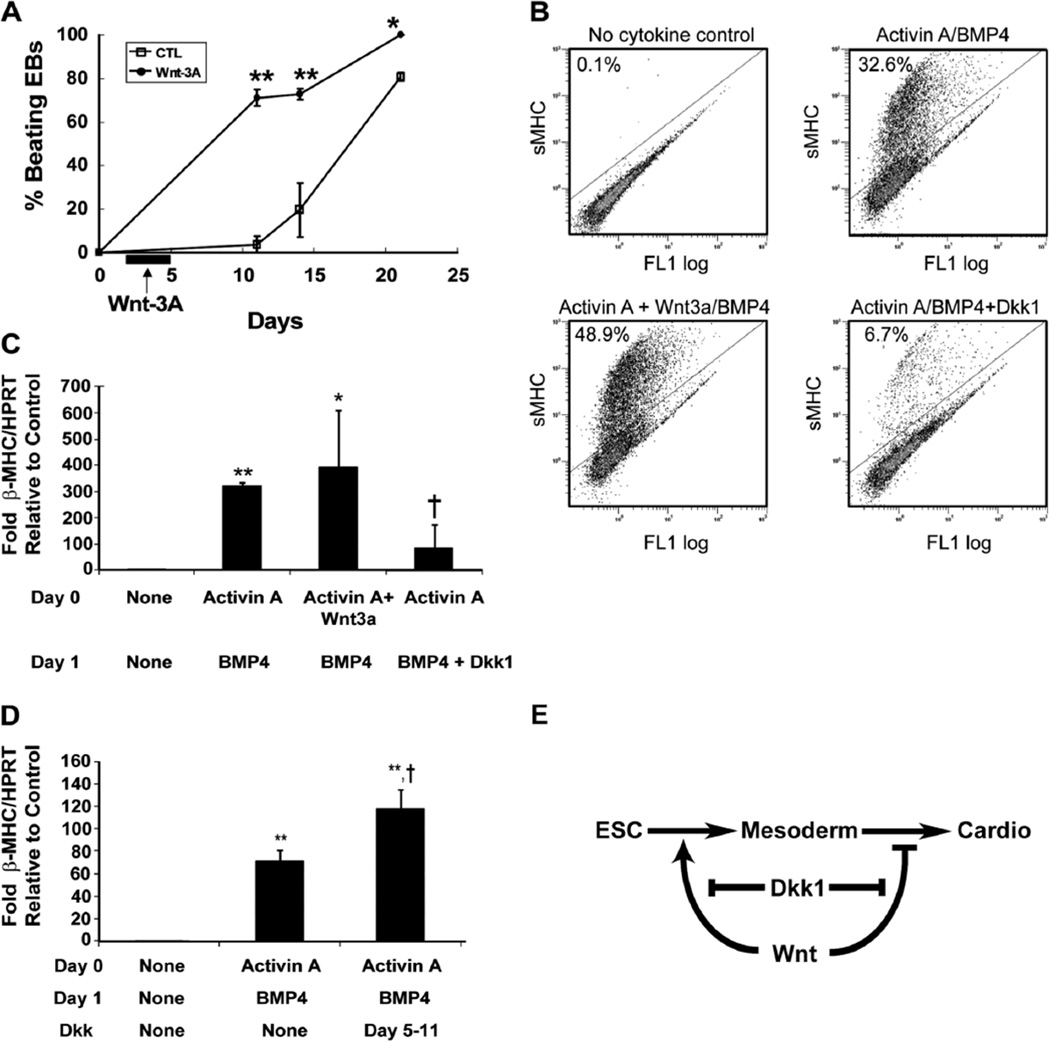
Confounded by these results, our group assessed this divergent role for Wnt signaling. Zebrafish lines were engineered to contain heat shock inducible Wnt activators (Wnt8) and Wnt repressors (Dkk1). Using these transgenic animals, we demonstrated that if Wnt activation occurs prior to gastrulation, the amount of lateral mesoderm tissue is increased, which subsequently increases the number of cardiac progenitors (determined by Nkx2.5 expression). If, however, Wnt overexpression is induced after gastrulation, cardiac progenitor numbers are reduced.32 Conversely, repressing Wnt signaling by overexpressing Dkk1 prior to gastrulation reduces the pool of lateral mesoderm cells and diminishes cardiac differentiation, whereas treating the cells after mesoderm induction with Dkk1 increases the resulting percentage of cardiomyocytes. Interestingly, the transition from activator to repressor occurs over a 1-h period, demonstrating a striking temporal dependence. This biphasic role for canonical Wnt in mesoderm and cardiac specification was also tested in mouse ESCs. Mouse EBs were subjected to Wnt3a from days 2–5 of differentiation, which resulted in a 20-fold increase of EB spontaneous contraction (Figure 3A) and a likewise 22-fold increase in cardiac specific α myosin heavy chain RNA (data not shown).32
Figure 3.
The Wnt/b-catenin signaling pathway has a biphasic effect on cardiac differentiation in mouse and human embryonic stem cells (ESCs). (A) The addition of Wnt-3A to mouse ESCs from days 2–5 hastens the onset of spontaneous contraction in embryoid bodies (EBs) when compared with a control. (B) Flow cytometry for sarcomeric myosin heavy chain (MF20 antibody, vertical axis) demonstrates that early treatment with Wnt3a enhances the induction of cardiac differentiation by activin A and BMP4. Conversely, blocking endogenous Wnt signaling with Dkk1 markedly inhibits the ability of activin A and BMP4 to induce cardiac differentiation. (C) Quantitative RT-PCR demonstrates enhanced expression of β-MHC mRNA when treated with Activin A/BMP4. This is further increased with the addition of Wnt3a to activin A. If a Wnt inhibitor, Dkk1, is added with BMP4, β-MHC transcription is markedly decreased. (D) Addition of Dkk1 at later time points (days 5–11), post mesoderm induction, enhances β-MHC transcript levels, indicating that endogenous Wnt signals have a biphasic effect, inducing mesoderm early and later inhibiting differentiation of cardiomyocytes from mesoderm. (E) Schematic of Wnt signaling during cardiac differentiation, initially inducing a population of precardiac mesodermal cells. Once mesodermal, further treatment of Wnt will induce other mesodermal fates but will inhibit further cardiac differentiation. The canonical Wnt antagonist, Dkk1, can block Wnt signaling. Dkk1-mediated Wnt inhibition prior to mesoderm formation decreases the population of precardiac mesoderm that will give rise to cardiomyoyctes. Dkk1-mediated Wnt inhibition after mesoderm formation promotes a cardiac phenotype. MHC, myosin heavy chain; BMP, bone morphogenic protein. Dkk1, dikkopf homolog 1; RT-PCR, reverse transcriptase polymerase chain reaction.
This biphasic role for Wnt occurs in the hESC differentiation pathway as well.33 When assessing the role of Wnt expression in the activin A/BMP4 directed differentiation protocol, endogenous Wnts 1, 3a and 8a are induced after activin A treatment and mesoderm specification. If hESCs are treated with exogenous Wnt3a prior to mesoderm specification, the ultimate extent of the induction of cardiomyocytes (Figure 3B) and β-myosin heavy chain RNA (MHC; Figure 3C) is further increased. Conversely, when Wnt signaling is inhibited by Dkk1 application at day 1 of activin A/BMP4 treatment, the induction of cardiomyocyte numbers and β-MHC RNA is markedly reduced (Figures 3B,C). If, however, Wnt is inhibited after mesoderm specification, by the addition of Dkk1 (days 5–11), the ultimate amount of βMHC RNA is increased by 70% compared with control hESCs treated only with activin A/BMP4 (Figure 3D).33 These findings indicate that endogenously produced Wnt ligands control mesoderm and cardiac differentiation from hESCs. Data from our group further suggests that line-toline variation in the activity of key signaling pathways likely explains the variance in cardiogenesis among different hESC lines.33 Studies such as these also demonstrate that cardiac promoting signals learned from Xenopus and zebrafish embryology have a similar function in controlling cardiac specific differentiation in mouse and human ESCs. These advancements greatly increase our ability to prepare and isolate larger populations of hESC-derived cardiomyocytes for transplantation experiments.
Generation of Endothelium From Human ESCs
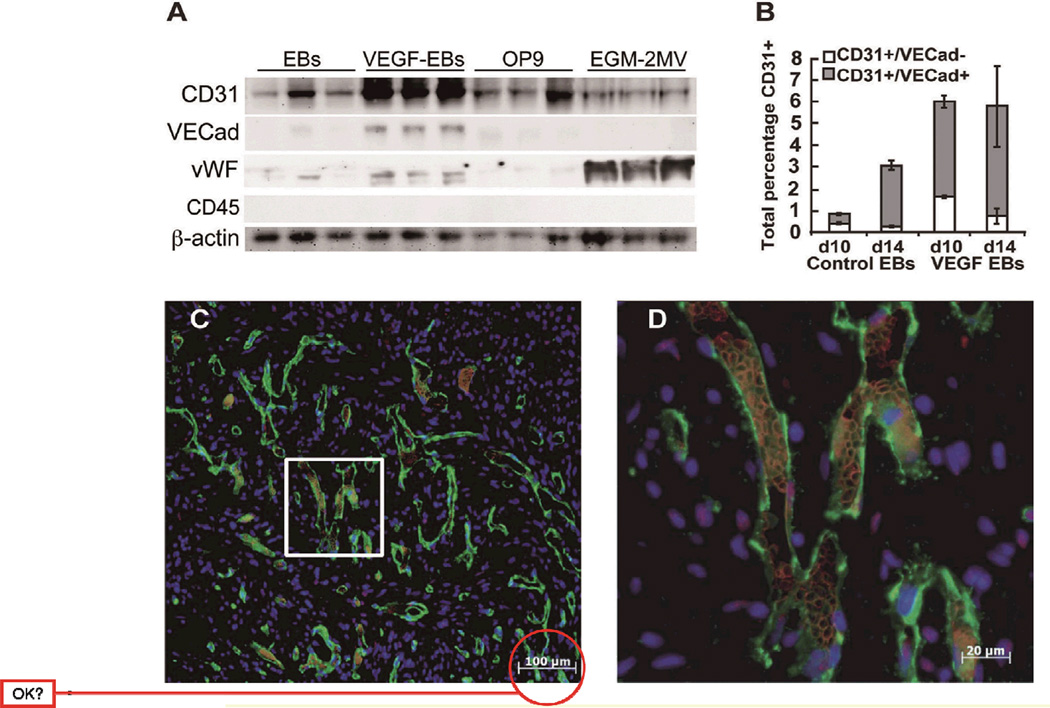
Although the principal goal of cell-based cardiac repair has been the replacement of lost cardiomyocytes, the transplanted cardiomyocytes will require the establishment of a significant vascular supply, either from the host tissue or from a therapeutic cell source. In early development, blood vessels are derived from angioblasts initially generated by the yolk sac. Vascular endothelial growth factor (VEGF) is a major effector for the proliferation and differentiation of the angioblasts,34 ultimately leading to the coalescing of blood islands and the formation of a primary capillary bed.35 If hESCs are grown to form EBs and allowed to differentiate in 20% fetal bovine serum (FBS), typically only 1–2% of cells will obtain an endothelial fate. We therefore tested a variety of protocols to enhance endothelial differentiation. Neither differentiation in commercial endothelial growth medium (EGM-2MV, effective for monkey ESCs)36 nor coculture with OP9 stromal cells (shown to enhance hematopoiesis from human ESCs)37 enhanced expression of the endothelial surface marker CD31. If the same protocol were repeated, however, with 20% FBS and vascular endothelial growth factor (VEGF: 50 ng/ml every 2 days), after 14 days of treatment, the VEGF-treated EBs had significant increases in the level of endothelial markers CD31, VE-cadherin, and von Willebrand factor (Figures 4A,B). Furthermore, CD31 antibody staining of VEGF-treated EBs demonstrated vascular structures with cords, branches and lumens, whereas control EBs had only rudimentary structures.
Figure 4.
Generation of endothelial cells from human embryonic stem cells (hESCs). (A) Embryoid bodies (EBs) treated with multiple factors for the induction of a vascular phenotype demonstrate that VEGF leads to increased levels of CD31, VE-cadherin, and vWF proteins at 14 days. Conversely, coculture with OP9 stromal cells or growth in endothelial growth medium, 2MV, did not consistently enhance endothelial markers. (B) Flow cytormetry analysis demonstrating that VEGF treatment of EBs leads to increased numbers of total VE-cadherin+/CD31+ cells at days 10 and 14 when compared with untreated EBs. (C,D) hESC-derived endothelial cells purified by CD31 expression, seeded into collagen gel constructs and implanted onto the surface of hearts previously injured by ischemia–reperfusion. The human endothelial cells (human-specific CD31 staining, green) demonstrate vascular networks that are perfused with rat red blood cells (red, autofluorescence). (Scale bars: C, 100 µm; D, 20 µm.) VEGF, vascular endothelial growth factor; vWF, von Willebrand factor.
We assessed whether hESC-derived endothelium was able to differentiate into functional vascular beds by treating EBs with VEGF. These treated cells were then sorted and a CD31-positive population was isolated and mixed in a 2:1 ratio with human mesenchymal stem cells, to provide a stromal support, and seeded into a collagen gel. We found that these constructs could be implanted onto the surfaces of 7week-old rat myocardial infarcts. When analyzed after 1 week, human vascular networks were identified and, even more encouraging, host red blood cells were readily detected within the lumens of the engineered vasculature (Figures 4C,D). This finding demonstrated that hESC-derived endothelial cells could survive transplantation and, within a relatively short period, incorporate into the existing vasculature, which encourages further study of hESC-derived endothelial cells in cell therapy and tissue engineering.
Human ESC-Derived Cardiomyocytes Improve Heart Function Post-Infarct
The activin A/BMP4 protocol increased our population of cardiomyocytes for transplantation and minimized the risk of teratoma formation by decreasing the pool of undifferentiated ESCs. The next hurdle to overcome was achieving engraftment. Infarcts present a hostile environment: they are hypoxic, acidic, replete with reactive oxygen species, and contain leukocytes that participate in cytokine and protease production. Could immature cardiomyocytes survive, integrate and function? Prior studies demonstrated that hESCderived cardiomyocytes could survive in a normal rodent heart,38 and Kehat et al39 showed that these cells electrically integrated when grafted in the normal pig heart. We therefore tested whether hESC-derived cardiomyocytes could engraft and improve function in a rodent model of myocardial infarction. Our initial results were disappointing. Human cardiomyocyte preparations did not survive in the infarct, and cardiac function was not affected. After extensive testing, we developed a pro-survival cocktail that blocks multiple pathways involved in cell death through membrane damage or apoptosis.27 The pro-survival interventions improve survival markedly, with >90% of animals now showing human cardiac grafts.
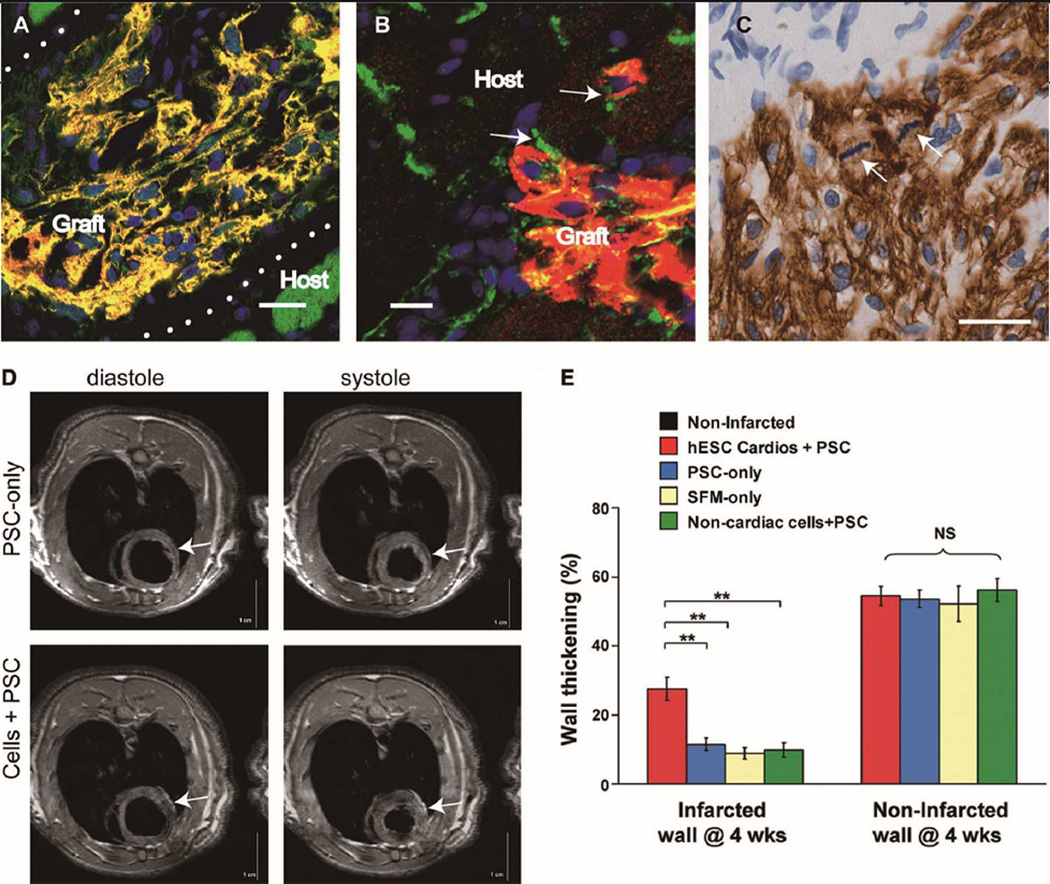
Next, we transplanted 10 million hESC-derived cardiomyocytes into the hearts of athymic rats, injured 4 days prior by 60 min of ischemia of the left anterior descending artery followed by reperfusion. At 1 month after transplantation, >85% of the transplanted cells were correctly targeted to the infarct, repopulating approximately 4% of the infarcted area (Figure 5A). The human cardiomyocytes occasionally established junctions with host cardiomyocytes (Figure 5B), but most were separated from the host by scar tissue. The cardiomyocytes expressed the expected cardiac marker proteins, such as Nkx2.5, MLC2V and β-MHC (in contrast to host rat α-MHC), and had immature sarcomeres (Figure 5A). Interestingly, the human cardiomyocytes remained proliferative until 4 weeks post transplantation. When given a 1-h BrdU pulse to detect DNA synthesis, 1.4% of cardiomyocytes were labeled, and mitotic figures were readily detected (Figure 5C). Correspondingly, graft size grew substantially between 1 and 4 weeks. Heart function was likewise improved. Cardiac magnetic resonance imaging demonstrated a 2.5-fold increase in systolic wall thickening in the infarcted region of rats receiving human cardiomyocytes (Figures 5D,E), and echocardiography demonstrated a 40% improvement in fractional shortening among the 3 different control groups.
Figure 5.
Human cardiomyocytes engraft in rat infarcts and enhance ventricular function. Human embryonic stem cells (hESCs) were differentiated to a cardiomyocyte cell fate and transplanted to 4-day old infarcts in athymic rats. (A) When assessed at 4 weeks post transplantation, grafts expressed cardiac troponin I (green) and β-MHC (red; yellow in merged image), whereas rat cardiomyocytes were typically β-MHC-negative (scale bar 20µ m). (B) Junctions between host and graft cardiomyocytDense graftses (*) could of transplantedbe demonstrated cell could by be staining determined for cadherin by β-MHC (green (brown);) and the β- nucleiMHC (ofred some) (nuclei β-MHC-positive blue, scale barcells 10 haveµ m). con- (C) densed DNA (arrows), demonstrating mitotic activity post transplantation (scale bar 20µ m). (C) Ongoing proliferation of 4week-old grafts identified by mitotic figures (arrows) in β-MHC-positive graft cells (brown). (D–G) Cardiac MRI used to assess recipient animals for improvement in heart function. Control (pro-survival cocktail (PSC)) only, animals demonstrated a thinned anterior wall infarct (arrow) and dilated chamber in both diastole (D) and systole (E), whereas animals receiving differentiated cardiomyocytes had reduced dilation and increased anterior wall thickening (arrow) in both diastole (F) and systole (G) (scale bar 1 cm). (H) Rats receiving human cardiomyocytes with PSC had a 2.5-fold increase in systolic wall thickening, by MRI, compared with hearts treated with PSC alone, serum-free medium or hESCs differentiated in FBS + PSC, which have low rates of cardiomyocyte production (~0.8%). MHC, myosin heavy chain; FBS, fetal bovine serum; MRI, magnetic resonance imaging.
Although these are encouraging findings, we do not yet know whether the transplanted cells were under control of the sinoatrial node and beating synchronously with the rest of the heart. The increased systolic function does, however, suggest the transplanted population induces local force generation. Another concern is the potential for arrhythmia arising from transplanted cells, particularly given that both nodal and ventricular cells can be generated from immature cardiomyocytes. Rhythm disturbances from pacemaker activity within the transplanted cells would be difficult to detect, given that the rat heart rate is approximately 450 beats/min and would thus overdrive suppress any automatic focus created by donor human cardiomyocytes. Further study in species with slower heart rates will be needed. Despite these limitations, this work demonstrates an ability to make an enriched population of mitotically active, immature cardiomyocytes for transplantation in a scalable system, and it shows that these cells can improve cardiac function post infarction.
Creation of Vascularized Cardiac Patches for Transplantation
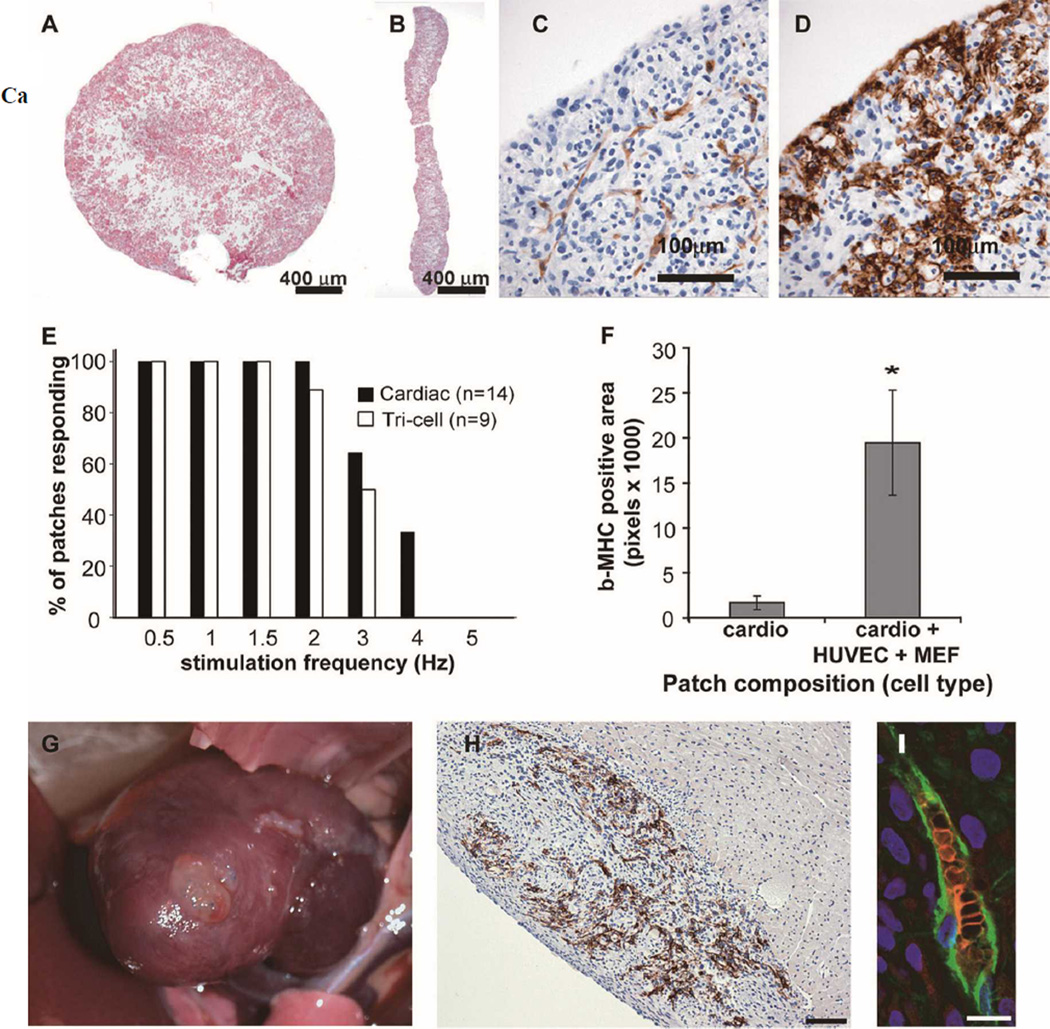
Although individual cells survived in a rodent transplant model, the graft size remained relatively small and variable. Tissue engineering presents a complementary approach to cell therapy, potentially offering larger and more predictable grafts. Many groups have approached cardiac tissue engineering by using synthetic or natural biodegradable scaffolds seeded with cardiomyocytes and other cell types.40–42 Although these methods show promise, there are several drawbacks, including possible toxicity of the scaffold (eg, degradation products), induction of an inflammatory response, poor compliance with heart function, non-uniform and lowdensity cell seeding, and electrical isolation of the graft. Concerned by these limitations, our group has generated scaffoldfree human cardiac tissue patches for transplantation.43 When hESC-derived cardiomyocytes are cultured on an orbital shaker, within 2 days they self-adhere, forming a disk-shaped patch of tissue. The diameter of the patch can be manipulated both by the initial number of cells added to the culture and the orbital speed, while the thickness remains approximately 400µ m (Figures 6A,B). Interestingly, although the initial population of cells used to create the patches was approximately 30% human cardiomyocytes, after 10 days of 3-D growth, the patches were >75% cardiomyocytes. Unfortunately, these cardiac patches had poor survival when transplanted into skeletal muscle, most likely because of ischemia. We hypothesized that a “prevascularized” patch would develop a circulation more rapidly and enhance overall survival. To increase vascularization, human umbilical vein endothelial cells or hESC-derived endothelial cells plus mouse embryonic fibroblasts, as a mesenchymal support, were added at a 1:1:0.5 ratio (Figures 6C–F). The resultant tri-cell patches developed vessel-like structures (Figure 6C) without compromise of a cardiac phenotype (Figure 6D). These patches could be field-stimulated up to approximately 3 Hz (180 beats/min) with gradual loss at faster rates (Figure 6E). When loaded with a fluorescent indicator of calcium transients, these patches clearly demonstrated a synchronized calcium transient, appearing as waves across the macroscopic tissue patch, which implies the existence of coupled depolarization between cardiomyocytes.
Figure 6.
Human cardiac tissue engineering. Human cardiac patches are created by placing hESC-derived cardiomyocytes in a suspension culture on an orbital shaker. This method reproducibly creates patches of contractile myocardium with a tunable diameter (A), dependent on the input cell number, while its thickness is typically ~400µ m (B). (C) Pretreatment of patches with human umbilical vein endothelial cells (HUVECs) and mouse embryo fibroblasts (MEFs) form vascular-like networks (red, CD31) prior to transplant without compromising a β-MHC phenotype (D, red). These patches are able to be electrically stimulated (E). Prevascularization conveyed a survival advantage over nonvascularized patches when transplanted to rat skeletal muscle, typically demonstrating 11-fold larger graft size (F). The patches also could be sutured to the rat epicardium (G) and after 1 week in vivo, had significant levels of β-MHC-positive human cardiomyocytes (brown) (scale bar 100µ m). (H) Patches also contained robust human CD31 immunofluorescence in vessel-like structures that also held Ter 119+ red blood cells (green, human CD31; scale bar 10µ m) demonstrating connection of the engineered vessels to the vasculature of the rat. MHC, myosin heavy chain; hESC, human embryonic stem cells.
In contrast to the cardiomyocyte-only patches, the prevascularized tri-cell cardiac patches were resilient in transplantation experiments. When transplanted into hindlimb skeletal muscle, the patches showed >10-fold enhancement of cardiomyocyte survival compared with the cardiomyocyte-only constructs (Figure 6F). Tri-cell patches were then sutured directly onto the epicardial surface of uninjured nude rat hearts (Figure 6G).43 After 7 days these grafts demonstrated persistent β-MHC expression, with immature sarcomeric structures (Figure 6H). Importantly, the human endothelial cell structures had transitioned into definitive microvessels. The human vessels were found to contain both rat erythrocytes and leukocytes, demonstrating anastomosis to the host coronary circulation (Figure 6I).
The tissue engineering experiments are subject to some of the same limitations discussed for cell therapy. It is not known whether the grafts electrically couple with the host, and possible arrhythmic complications may be masked by the rat’s high heart rate. Nevertheless, we have found our largest human cardiac grafts to date using these engineered cardiac tissue patches and this will be an important area for the field to continue advancing.
Conclusions
Since the initial report on hESC culture over a decade ago, there have been many advances in the ability to direct these stem cells to either a cardiac or an endothelial cell fate. Fascinatingly, the differentiation cues that control cardiac development in lesser vertebrates have a similar function in hESC differentiation. Work from our laboratory, as well as those of others, is focused on dissecting the molecular pathways that determine a cardiovascular fate. By understanding the basic biology of ESCs, we are furthering our ability to manipulate these cells for a therapeutic purpose. Improvements in cell culture conditions, transplantation technology and tissue engineering may lead us to a new method of treating cardiovascular disease.
Acknowledgments
This work was supported by NIH Grants R01HL084642, P01HL094374, R01HL64387, P01GM81619, U01HL100405, and U24DK076126.
References
- 1.Jennings RB, Reimer KA. The cell biology of acute myocardial ischemia. Annu Rev Med. 1991;42:225–246. doi: 10.1146/annurev.me.42.020191.001301. [DOI] [PubMed] [Google Scholar]
- 2.Mallory GK, White PD, Salcedo-Salga J. The speed of healing of myocardial infarction: A study of the pathologic anatomy in 72 cases. Am Heart J. 1939;18:647–671. [Google Scholar]
- 3.Velagaleti RS, Pencina MJ, Murabito JM, Wang TJ, Parikh NI, D’Agostino RB, et al. Long-term trends in the incidence of heart failure after myocardial infarction. Circulation. 2008;118:2057–2062. doi: 10.1161/CIRCULATIONAHA.108.784215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bergmann O, Bhardwaj RD, Bernard S, Zdunek S, Barnabé-Heider F, Walsh S, et al. Evidence for cardiomyocyte renewal in humans. Science. 2009;324:98–102. doi: 10.1126/science.1164680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Reinecke H, Poppa V, Murry CE. Skeletal muscle stem cells do not transdifferentiate into cardiomyocytes after cardiac grafting. J Mol Cell Cardiol. 2002;34:241–249. doi: 10.1006/jmcc.2001.1507. [DOI] [PubMed] [Google Scholar]
- 6.Reinecke H, MacDonald GH, Hauschka SD, Murry CE. Electromechanical coupling between skeletal and cardiac muscle: Implications for infarct repair. J Cell Biol. 2000;149:731–740. doi: 10.1083/jcb.149.3.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Leobon B, Garcin I, Menasche P, Vilquin JT, Audinat E, Charpak S. Myoblasts transplanted into rat infarcted myocardium are functionally isolated from their host. Proc Natl Acad Sci USA. 2003;100:7808–7811. doi: 10.1073/pnas.1232447100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Orlic D, Kajstura J, Chimenti S, Jakoniuk I, Anderson SM, Li B, et al. Bone marrow cells regenerate infarcted myocardium. Nature. 2001;410:701–705. doi: 10.1038/35070587. [DOI] [PubMed] [Google Scholar]
- 9.Tang XL, Rokosh DG, Guo Y, Bolli R. Cardiac progenitor cells and bone marrow-derived very small embryonic-like stem cells for cardiac repair after myocardial infarction. Circ J. 2010;74:390–404. doi: 10.1253/circj.cj-09-0923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Planat-Benard V, Menard C, Andre M, Puceat M, Perez A, GarciaVerdugo JM, et al. Spontaneous cardiomyocyte differentiation from adipose tissue stroma cells. Circ Res. 2004;94:223–229. doi: 10.1161/01.RES.0000109792.43271.47. [DOI] [PubMed] [Google Scholar]
- 11.Beltrami AP, Barlucchi L, Torella D, Baker M, Limana F, Chimenti S, et al. Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell. 2003;114:763–776. doi: 10.1016/s0092-8674(03)00687-1. [DOI] [PubMed] [Google Scholar]
- 12.Condorelli G, Borello U, De Angelis L, Latronico M, Sirabella D, Coletta M, et al. Cardiomyocytes induce endothelial cells to transdifferentiate into cardiac muscle: Implications for myocardium regeneration. Proc Natl Acad Sci USA. 2001;98:10733–10738. doi: 10.1073/pnas.191217898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schlechta B, Wiedemann D, Kittinger C, Jandrositz A, Bonaros NE, Huber JC, et al. Ex-vivo expanded umbilical cord blood stem cells retain capacity for myocardial regeneration. Circ J. 2009;74:188–194. doi: 10.1253/circj.cj-09-0409. [DOI] [PubMed] [Google Scholar]
- 14.Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145–1147. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- 15.Murry CE, Reinecke H, Pabon LM. Regeneration gaps: Observations on stem cells and cardiac repair. J Am Coll Cardiol. 2006;47:1777–1785. doi: 10.1016/j.jacc.2006.02.002. [DOI] [PubMed] [Google Scholar]
- 16.Shiba Y, Hauch KD, Laflamme MA. Cardiac applications for human pluripotent stem cells. Curr Pharm Des. 2009;15:2791–2806. doi: 10.2174/138161209788923804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Freund C, Mummery CL. Prospects for pluripotent stem cellderived cardiomyocytes in cardiac cell therapy and as disease models. J Cell Biochem. 2009;107:592–599. doi: 10.1002/jcb.22164. [DOI] [PubMed] [Google Scholar]
- 18.Irion S, Nostro MC, Kattman SJ, Keller GM. Directed differentiation of pluripotent stem cells: From developmental biology to therapeutic applications. Cold Spring Harb Symp Quant Biol. 2008;73:101–110. doi: 10.1101/sqb.2008.73.065. [DOI] [PubMed] [Google Scholar]
- 19.Murry CE, Keller G. Differentiation of embryonic stem cells to clinically relevant populations: Lessons from embryonic development. Cell. 2008;132:661–680. doi: 10.1016/j.cell.2008.02.008. [DOI] [PubMed] [Google Scholar]
- 20.Min JY, Yang Y, Converso KL, Liu L, Huang Q, Morgan JP, et al. Transplantation of embryonic stemcells improves cardiac function in postinfarcted rats. J Appl Physiol. 2002;92:288–296. doi: 10.1152/jappl.2002.92.1.288. [DOI] [PubMed] [Google Scholar]
- 21.Behfar A, Zingman LV, Hodgson DM, Rauzier JM, Kane GC, Terzic A, et al. Stem cell differentiation requires a paracrine pathway in the heart. FASEB J. 2002;16:1558–1566. doi: 10.1096/fj.02-0072com. [DOI] [PubMed] [Google Scholar]
- 22.Nussbaum J, Minami E, Laflamme MA, Virag JA, Ware CB, Masino A, et al. Transplantation of undifferentiated murine embryonic stem cells in the heart: Teratoma formation and immune response. FASEB J. 2007;21:1345–1357. doi: 10.1096/fj.06-6769com. [DOI] [PubMed] [Google Scholar]
- 23.Swijnenburg RJ, Tanaka M, Vogel H, Baker J, Kofidis T, Gunawan F, et al. Embryonic stem cell immunogenicity increases upon differentiation after transplantation into ischemic myocardium. Circulation. 2005;112:I166–I172. doi: 10.1161/CIRCULATIONAHA.104.525824. [DOI] [PubMed] [Google Scholar]
- 24.Kehat I, Kenyagin-Karsenti D, Snir M, Segev H, Amit M, Gepstein A, et al. Human embryonic stem cells can differentiate into myocytes with structural and functional properties of cardiomyocytes. J Clin Invest. 2001;108:407–414. doi: 10.1172/JCI12131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ladd AN, Yatskievych TA, Antin PB. Regulation of avian cardiac myogenesis by activin/TGFbeta and bone morphogenetic proteins. Dev Biol. 1998;204:407–419. doi: 10.1006/dbio.1998.9094. [DOI] [PubMed] [Google Scholar]
- 26.Sugi Y, Sasse J, Barron M, Lough J. Developmental expression of fibroblast growth factor receptor-1 (cek-1; flg) during heart development. Dev Dyn. 1995;202:115–125. doi: 10.1002/aja.1002020203. [DOI] [PubMed] [Google Scholar]
- 27.Laflamme MA, Chen KY, Naumova AV, Muskheli V, Fugate JA, Dupras SK, et al. Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts. Nat Biotechnol. 2007;25:1015–1024. doi: 10.1038/nbt1327. [DOI] [PubMed] [Google Scholar]
- 28.Yang L, Soonpaa MH, Adler ED, Roepke TK, Kattman SJ, Kennedy M, et al. Human cardiovascular progenitor cells develop from a KDR+ embryonic-stem-cell-derived population. Nature. 2008;453:524–528. doi: 10.1038/nature06894. [DOI] [PubMed] [Google Scholar]
- 29.Marvin MJ, Di Rocco G, Gardiner A, Bush SM, Lassar AB. Inhibition of Wnt activity induces heart formation from posterior mesoderm. Genes Dev. 2001;15:316–327. doi: 10.1101/gad.855501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schneider VA, Mercola M. Wnt antagonism initiates cardiogenesis in Xenopus laevis. Genes Dev. 2001;15:304–315. doi: 10.1101/gad.855601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nakamura T, Sano M, Songyang Z, Schneider MD. A Wnt-and beta-catenin-dependent pathway for mammalian cardiac myogenesis. Proc Natl Acad Sci USA. 2003;100:5834–5839. doi: 10.1073/pnas.0935626100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ueno S, Weidinger G, Osugi T, Kohn AD, Golob JL, Pabon L, et al. Biphasic role for Wnt/beta-catenin signaling in cardiac specification in zebrafish and embryonic stem cells. Proc Natl Acad Sci USA. 2007;104:9685–9690. doi: 10.1073/pnas.0702859104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Paige SL, Osugi T, Afanasiev OK, Pabon L, Reinecke H, Murry CE. Endogenous Wnt/beta-catenin signaling is required for cardiac differentiation in human embryonic stem cells. PLoS One. 2010;5:1–8. doi: 10.1371/journal.pone.0011134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Millauer B, Wizigmann-Voos S, Schnurch H, Martinez R, Moller NP, Risau W, et al. High affinity VEGF binding and developmental expression suggest Flk-1 as a major regulator of vasculogenesis and angiogenesis. Cell. 1993;72:835–846. doi: 10.1016/0092-8674(93)90573-9. [DOI] [PubMed] [Google Scholar]
- 35.Drake CJ, Hungerford JE, Little CD. Morphogenesis of the first blood vessels. Ann NY Acad Sci. 1998;857:155–179. doi: 10.1111/j.1749-6632.1998.tb10115.x. [DOI] [PubMed] [Google Scholar]
- 36.Kaufman DS, Lewis RL, Hanson ET, Auerbach R, Plendl J, Thomson JA. Functional endothelial cells derived from rhesus monkey embryonic stem cells. Blood. 2004;103:1325–1332. doi: 10.1182/blood-2003-03-0799. [DOI] [PubMed] [Google Scholar]
- 37.Vodyanik MA, Bork JA, Thomson JA, Slukvin II. Human embryonic stem cell-derived CD34+ cells: Efficient production in the coculture with OP9 stromal cells and analysis of lymphohematopoietic potential. Blood. 2005;105:617–626. doi: 10.1182/blood-2004-04-1649. [DOI] [PubMed] [Google Scholar]
- 38.Laflamme MA, Gold J, Xu C, Hassanipour M, Rosler E, Police S, et al. Formation of human myocardium in the rat heart from human embryonic stem cells. Am J Pathol. 2005;167:663–671. doi: 10.1016/S0002-9440(10)62041-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kehat I, Khimovich L, Caspi O, Gepstein A, Shofti R, Arbel G, et al. Electromechanical integration of cardiomyocytes derived from human embryonic stem cells. Nat Biotechnol. 2004;22:1282–1289. doi: 10.1038/nbt1014. [DOI] [PubMed] [Google Scholar]
- 40.Radisic M, Park H, Martens TP, Salazar-Lazaro JE, Geng W, Wang Y, et al. Pre-treatment of synthetic elastomeric scaffolds by cardiac fibroblasts improves engineered heart tissue. J Biomed Mater Res A. 2008;86:713–724. doi: 10.1002/jbm.a.31578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bursac N, Loo Y, Leong K, Tung L. Novel anisotropic engineered cardiac tissues: Studies of electrical propagation. Biochem Biophys Res Commun. 2007;361:847–853. doi: 10.1016/j.bbrc.2007.07.138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gonen-Wadmany M, Gepstein L, Seliktar D. Controlling the cellular organization of tissue-engineered cardiac constructs. Ann NY Acad Sci. 2004;1015:299–311. doi: 10.1196/annals.1302.025. [DOI] [PubMed] [Google Scholar]
- 43.Stevens KR, Pabon L, Muskheli V, Murry CE. Scaffold-free human cardiac tissue patch created from embryonic stem cells. Tissue Eng Part A. 2009;15:1211–1222. doi: 10.1089/ten.tea.2008.0151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Stevens KR, Kreutziger KL, Dupras SK, Korte FS, Regnier M, Muskheli V, et al. Physiological function and transplantation of scaffold-free and vascularized human cardiac muscle tissue. Proc Natl Acad Sci USA. 2009;106:16568–16573. doi: 10.1073/pnas.0908381106. [DOI] [PMC free article] [PubMed] [Google Scholar]