Abstract
Objective
To examine the mortality and cardiovascular disease (CVD) burden among a population-based cohort of patients with systemic lupus erythematosus (SLE) with previously described late mean onset and low rates of organ-threatening disease.
Methods
This retrospective population-based cohort study investigated incident cases of SLE diagnosed from 1991–2008 and followed through March 2009 to examine rates of death and CVD events: myocardial infarction, stroke, or congestive heart failure hospitalization. Cases were identified using the 1997 update of 1982 American College of Rheumatology SLE criteria. Searches included electronic records, chart audits, and state death matches, with physician review. Age and sex-matched population comparisons facilitated relative event rate calculations.
Results
70 incident SLE cases had late mean onset (52 years), with an incidence of 5 cases per 100,000/year. Matched comparisons showed similar baseline rates of hypertension, hyperlipidemia, and diabetes. However, SLE patients experienced more CVD in the 2 years preceding SLE diagnosis, odds ratio 3.8 (95% CI 1.8, 8.0). The estimated 10-year mortality rates were 26% for SLE subjects versus 19% for comparisons, hazard ratio (HR) 2.1, p<0.01. Adjusted for prior CVD, SLE cases still demonstrated increased hazards of mortality (HR 1.9, p=0.01) and CVD event or death (HR 1.8, p=0.01).
Conclusion
This incident SLE cohort demonstrated approximately doubled mortality and CVD event hazards compared to age and sex-matched comparisons, even after accounting for higher CVD events in the 2 years preceding SLE diagnosis. This raises future research questions regarding delayed lupus diagnosis versus accelerated CVD prior to SLE, particularly in older-onset SLE.
Key Indexing Terms: Systemic Lupus Erytematosus, Cardiovascular System, Mortality, Epidemiology, Incidence, Cohort Study
INTRODUCTION
While early survival among systemic lupus erythematosus (SLE) patients has improved, epidemiological studies still show an increased mortality rate and cardiovascular disease (CVD) events [1–5]. Reports regarding long-term outcomes of SLE frequently stem from patients of urban clinical referral centers [1–3], and many argue that these reports do not resemble lupus patients commonly encountered elsewhere [6,7], which include older adults in whom mortality and CVD event excesses are less clear. Few studies have examined survival in US population-based lupus inception cohorts [8,9], with the most recent concluding follow-up in 1997. To our knowledge, no prior studies have systematically examined the CVD burden among a US population-based cohort of lupus patients.
Using the 1997 update of the 1982 American College of Rheumatology SLE classification criteria [10], we assembled a population-based cohort of SLE patients newly diagnosed between January 1, 1991 and December 31, 2008. Specifically, we combined a previously identified incident lupus cohort diagnosed from 1991–2001 [11] with new incident cases observed in the same population through 2008. Our objective was to examine the mortality burden and rates of cardiovascular events within this population-based cohort of lupus patients, which had demonstrated later onset and milder SLE disease than many other lupus cohorts (9).
PATIENTS AND METHODS
Population sample
This retrospective cohort study investigated all incident cases of SLE diagnosed from 1991 to 2008 in a geographically-defined population to examine rates of death and fatal and non-fatal CVD events including myocardial infarction (MI), stroke, and hospitalization for congestive heart failure. The lupus study cohort was established within the Marshfield Epidemiologic Study Area (MESA), a defined region of 24 ZIP codes within the service area of a single health care system in north-central Wisconsin. The MESA resource was established in 1991 to facilitate population-based epidemiologic and clinical research. Nearly all MESA residents receive health care from Marshfield Clinic, its 42 regional centers, and affiliated hospitals. Patient information dating back to 1991 or earlier is archived in electronic medical records, and detailed administrative files include cohort enrollment, health care encounters, and links to state death files. Recent validation efforts indicated capture of >90% of health care encounters and 99% of deaths among the residents of central MESA [11]. The 2000 US Census data indicated that roughly 97% of the 77,280 residents of this area were white non-Hispanic. The study area population is remarkably stable geographically, with <5% annual out-migration of the original cohort. The Marshfield Clinic Institutional Review Board approved this study.
SLE and comparison cohort definitions and exclusions
A prior study in this population described 44 incident SLE cases diagnosed between 1991 and 2001, from among 239 potential cases reviewed [11]. In this new study, inpatient and outpatient encounters were again searched for International Classification of Diseases, Ninth Revision (ICD-9) SLE diagnosis codes (710.0). In total, 128 individuals with at least one SLE ICD-9 code were reviewed including 28 who had insufficient criteria for definite SLE in the 2001 review but had subsequent SLE-coded encounters, plus 100 new potential cases from 2002–2008. Chart reviews were completed by trained medical record abstractors with adjudication by study physicians, including two board certified rheumatologists. “Definite lupus” was defined by the 1997 update of the 1982 ACR revised SLE classification criteria [10]. ANA was considered positive at ≥1:80. Non-specific urinary casts were omitted to improve specificity. Rash or mucosal symptoms and arthritis required documented physician verification by history or exam. Possible antiphospholipid antibody syndrome status was assessed by presence of antiphospholipid lab abnormalities and thrombosis history confirmed by study physician review of clinical clotting event descriptions[12]. Questions were resolved by physician reviewer consensus.
To be considered incident cases, subjects with definite lupus were required to have resided in the MESA region at the time of their earliest diagnosis of SLE. The diagnosis “baseline” date was interpreted as the first date the patient met at least four ACR diagnostic criteria as per physician encounter notes. Cases with prior SLE diagnosis while residing outside this geographic region, miscoded diagnoses, or incomplete documentation were excluded. Discoid lupus in isolation or drug-induced lupus cases, defined by either formal diagnosis or documented use of culprit medications, were excluded.
The comparison group was defined to include all age (same birth month/year) and sex-matched MESA cohort members with health encounters on the date of the matched SLE case’s diagnosis date (n=2,565). The baseline date for comparisons was defined as the date of corresponding SLE case diagnosis. Individual comparisons were linked with a single SLE case in a weighted fashion to account for variable numbers of matched comparisons (n=3–65) for each lupus case. SLE cases and comparisons were evaluated to confirm MESA residence and assess other comorbid diagnoses 24 months preceding baseline date to verify the robustness of the population based cohort and to facilitate baseline CVD risk and comorbidity assessment.
Data sources
Data sources included clinical and administrative electronic records, matches to clinic and state death files, and lupus patient paper and electronic chart audits by trained abstractors using a pre-tested tool with study physician adjudication. A 20% random sample of cases underwent reabstraction including chart review by a second abstractor to assure quality. Electronic clinical data warehouse files were reviewed to ascertain encounter diagnosis codes and laboratory data. Administrative data files were searched to verify residency information, and death dates were verified between state vital statistics records and local mortality files.
Variables and Outcomes
Baseline CVD risk characteristics were ascertained by electronic search for both cases and comparisons. Two or more ICD-9 coded encounters in 24 months indicated presence of diabetes, hypertension, or hyperlipidemia respectively [13,14]. Administrative search strategies developed by the Center for Medicare Services for the Chronic Condition Data Warehouse were used to evaluate the presence or absence of ischemic heart disease, acute myocardial infarction (AMI), stroke or heart failure in the two years prior to lupus diagnosis or index comparison date [15]. Prior “non-fatal CVD” was defined as at least one hospitalization code for AMI, stroke, congestive heart failure, or ischemic heart disease with or without revascularization, or 2 or more outpatient codes within 24 months prior to baseline date. The same criteria were applied to screen “new” CVD events when searching from baseline diagnosis date through the earliest censoring date of date of death, migration, or the end of the follow-up period (March 21, 2009). New cardiovascular events and deaths in SLE cases were then confirmed based on queries of electronic medical record data, medical chart review, and state vital statistics data, with MD verification. New myocardial infarction events in SLE cases were confirmed using the American Heart Association universal definition including presence of (a) positive troponin or CK-MB plus chest pain, acute Q-waves or ST elevation, or imaging wall motion abnormalities; (b) angioplasty or bypass; (c) postmortem confirmation of myocardial injury; or (d) sudden death in the presence of chest pain [16]. New heart failure and stroke events in SLE cases were reviewed using Framingham CHF criteria and registry definitions of stroke including new focal or global neurologic impairment confirmed with imaging or death with only a vascular cause [17,18]. Chart audits by 2 MD investigators using these standard clinical/registry criteria confirmed 90% of CVD events that occurred in SLE subjects during the observed period, validating the automated algorithm that had been applied to compare CVD events between SLE cases and comparisons. Smoking status (current, former, or never) was ascertained by manual review for the SLE cases.
Statistical analysis
Population-based SLE incidence density was calculated as number of new diagnoses per total person time observed. Baseline demographics and cardiovascular risk factors were summarized for cases and comparisons using descriptive statistics. For comparisons, “weighted” statistics were calculated, corresponding to a hypothetical population of matched comparisons having the same age and sex distribution as the case population. Baseline cardiovascular risk factors adjusted for age and sex were compared using stratified odds ratios and Cochran-Mantel-Haenszel p-values.
All-cause mortality and a composite endpoint of death or CVD event were examined by means of weighted Kaplan-Meier (K-M) estimates. The weighted estimates may be considered the estimated cumulative incidence of CVD events for a hypothetical population drawn from the comparison group but having the same age and sex distribution as the case population. Event rates were compared using hazard ratios (HRs) from a stratified Cox proportional hazards model and stratified log-rank p-values. Individuals were censored at the earliest of: the date they left regional membership, died, or data cut-off on March 21, 2009. In a post-hoc analysis, the analysis was repeated with comparisons matched to cases on baseline prior CVD status, in addition to age and sex. K-M curves for SLE cases and weighted comparisons with and without prior CVD were presented with HRs to compare CVD risk in two groups after accounting for age, sex, and baseline status of prior CVD.
RESULTS
Patient characteristics
From 1991–2008, there were a total of 70 incident SLE cases with a mean onset age of 52 years and females outnumbering males 4:1 (Table 1). Population-based SLE incidence was 5.03 cases per 100,000 persons per year. The SLE cases had mean follow-up of 7.7 years for a total of 540 lupus patient-years observed. Similar to earlier published results [11], SLE disease was mild including only two cases (3%) of CNS disease and 23% with renal disease (Figure 1). Most were ANA positive (99%), and hematologic abnormalities (80%) and arthritis (61%) were among the most common disease manifestations. Additionally, 75% had received hydroxychloroquine and 25% received steroid sparing disease modifying drugs beyond hydroxychloroquine. 73% had received steroids and at least 56% had ever received steroids in excess of 10 mg daily for more than 2 months. Antiphospholipid Syndrome was found in 20%, and, 38% of these SLE patients had received 2 or more months of anticoagulation therapy for various reasons.
Table 1.
Baseline Descriptives SLE Cases and Comparisons
| Characteristics | SLE (n=70) |
Comparisons (n=2565) |
Weighted Controls |
OR [95% CI] |
|---|---|---|---|---|
| Mean onset Age [mean (sd)] | 51.6 (21.2) | 45.3 (19.2) | 51.6 (21.0) | Matched |
| Age Categories: 18–25 yo | 8 (11%) | Matched | ||
| 25–<35 yo | 12 (17%) | Matched | ||
| 35–<50 yo | 15 (21%) | Matched | ||
| 50–<65 yo | 15 (21%) | Matched | ||
| 65+ yo | 20 (29%) | Matched | ||
| Female [N (%)] | 57 (81%) | 2144 (84%) | 81% | Matched |
| Hypertension [N (%)] | 17 (24%) | 354 (14%) | 19% | 1.5 [0.8,2.8] |
| Hyperlipidemia [N (%)] | 9 (13%) | 369 (14%) | 18% | 0.6 [0.3,1.3] |
| Diabetes [N (%)] | 5 (7%) | 136 (5%) | 7% | 1.0 [0.4,2.6] |
| Prior CVD [N (%)] | 16 (23%) | 166 (7%) | 10% | 3.7 [1.8,7.9]* |
| Prior Acute MI [N (%)] | 2 (3%) | 16 (0.6%) | 1% | 2.8 [0.6,12] |
| Prior Ischemia [N (%)] | 11 (16%) | 127 (5%) | 8% | 2.8 [1.3,6.1]* |
| Prior CHF [N (%)] | 7 (10%) | 60 (2%) | 4% | 2.9 [1.2,7]* |
| Prior Stroke [N (%)] | 3 (4%) | 17 (0.7%) | 1% | 5.1 [1.4,19]* |
| Mean Follow-up (years) | 7.7 | 8.3 | 8.1 |
Indicates p < 0.05
SD-Standard deviation; YO-Years old; CVD-Cardiovascular disease; MI-Myocardial Infarction; CHF-Congestive heart failure
Figure 1.
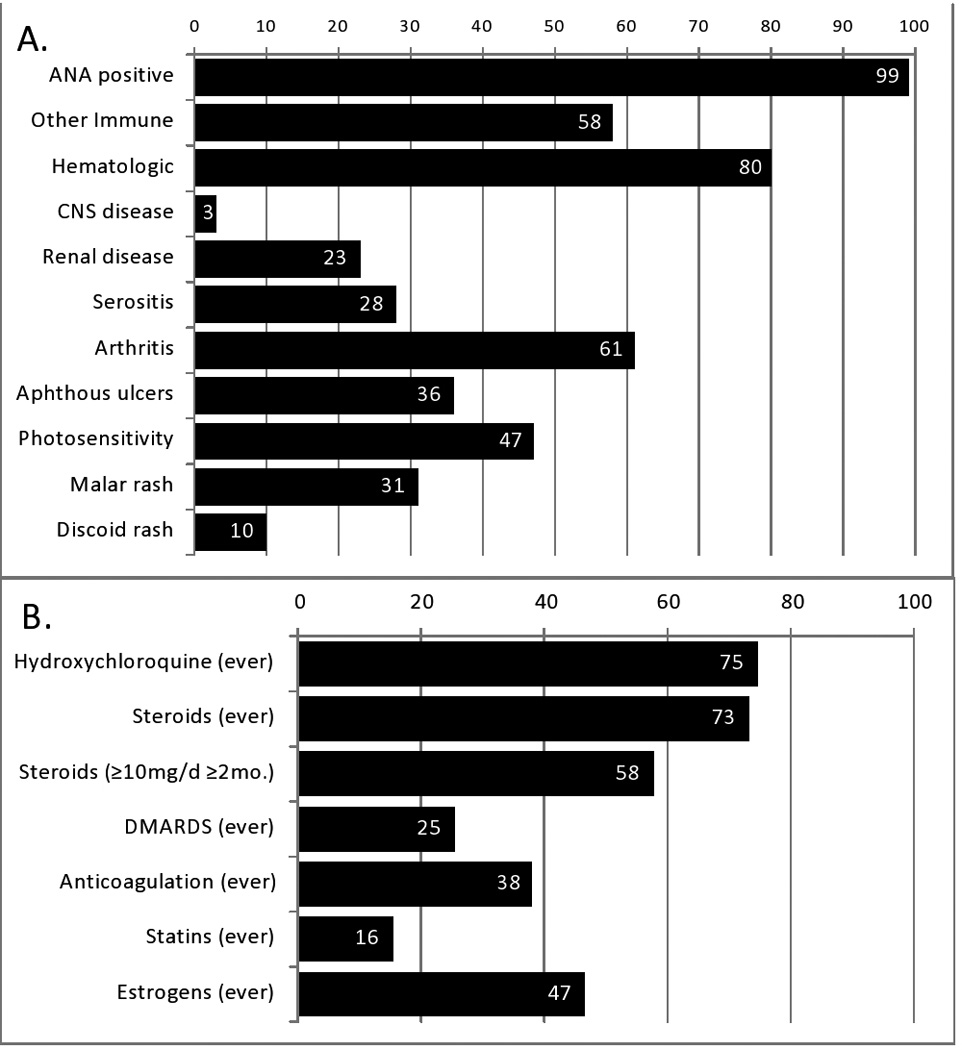
Clinical SLE features at baseline (Panel A) and medication exposure history (Panel B). Values reflect percent of SLE patients (n=70) with each feature.
Most traditional CVD risk factors at baseline were similar between lupus cases and comparisons, including rates of diagnosed hypertension, hyperlipidemia, and diabetes (Table 1). However, SLE patients were more likely than comparisons to have received CVD diagnosis codes in the 2 years prior to their SLE diagnosis, OR=3.8 (95% CI 1.8, 8.0). Significant excesses were seen in prevalent ischemic heart disease, heart failure hospitalizations, and stroke, which carried the highest estimated OR=5.1 (1.4, 19) for greater baseline prevalence in SLE patients compared to age and sex matched comparisons.
New CVD events and deaths
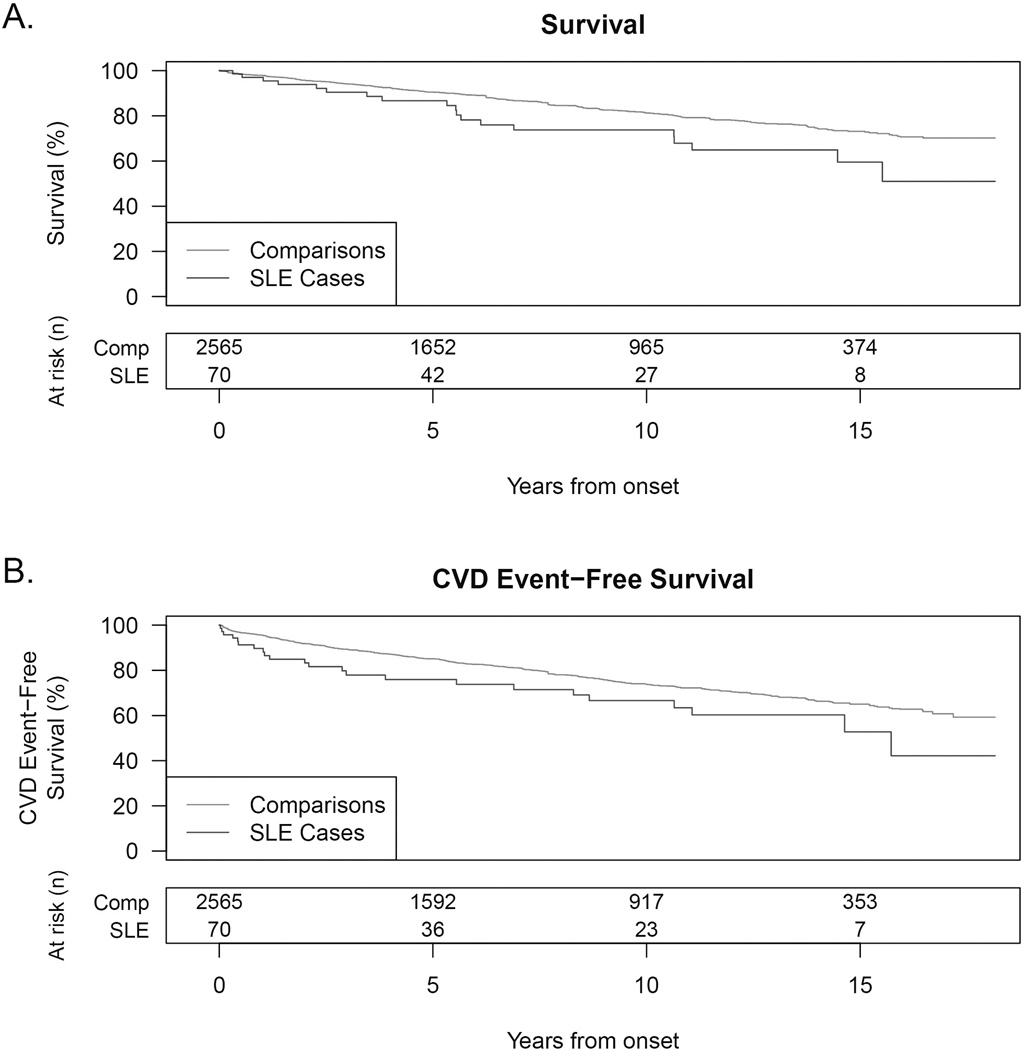
From 1991–March 2009, in the SLE cohort, there were 17 subjects who experienced 23 non-fatal CVD events, and 19 deaths were observed among the 70 patients during 540 lupus patient-years of follow-up. Compared to age and sex-matched comparisons observed over 21,181 person years, SLE subjects had approximately twice the hazards of all-cause mortality and of composite death and new CVD events with HR=2.09, p=0.002 and HR=1.83, p=0.006 respectively (Figure 2). Estimated mortality rates for the SLE cohort at 5, 10, and 15 years after diagnosis were 13%, 26%, and 41%, versus 10%, 19%, and 27% for matched comparisons (data not shown). The estimated rate for the composite endpoint of death or CVD event at 10 years after diagnosis was 33% for the SLE cohort compared to 26% for matched comparisons. Leading causes of death in SLE subjects included 32% (n=6) cardiovascular, 16% (n= 3) renal failure, 16% (n=3) infection, and (n=2) 11% malignancy, with remaining deaths attributed to GI bleed (n=1), respiratory failure/interstitial lung disease (n=1), hepatic failure (n=1), and 11% (n=2) recorded “failure to thrive” without a further death etiology documented in these two cases. The most common non-fatal CVD events in SLE cases were CHF hospitalizations (n=11), followed by stroke (n=7), and nonfatal MI (n=4).
Figure 2.
Kaplan-Meier estimates for SLE cases and their age and sex-matched comparisons for (A) all-cause mortality and (B) the composite of death or CVD event. SLE subjects had 2-fold increased hazards for each endpoint (A and B).
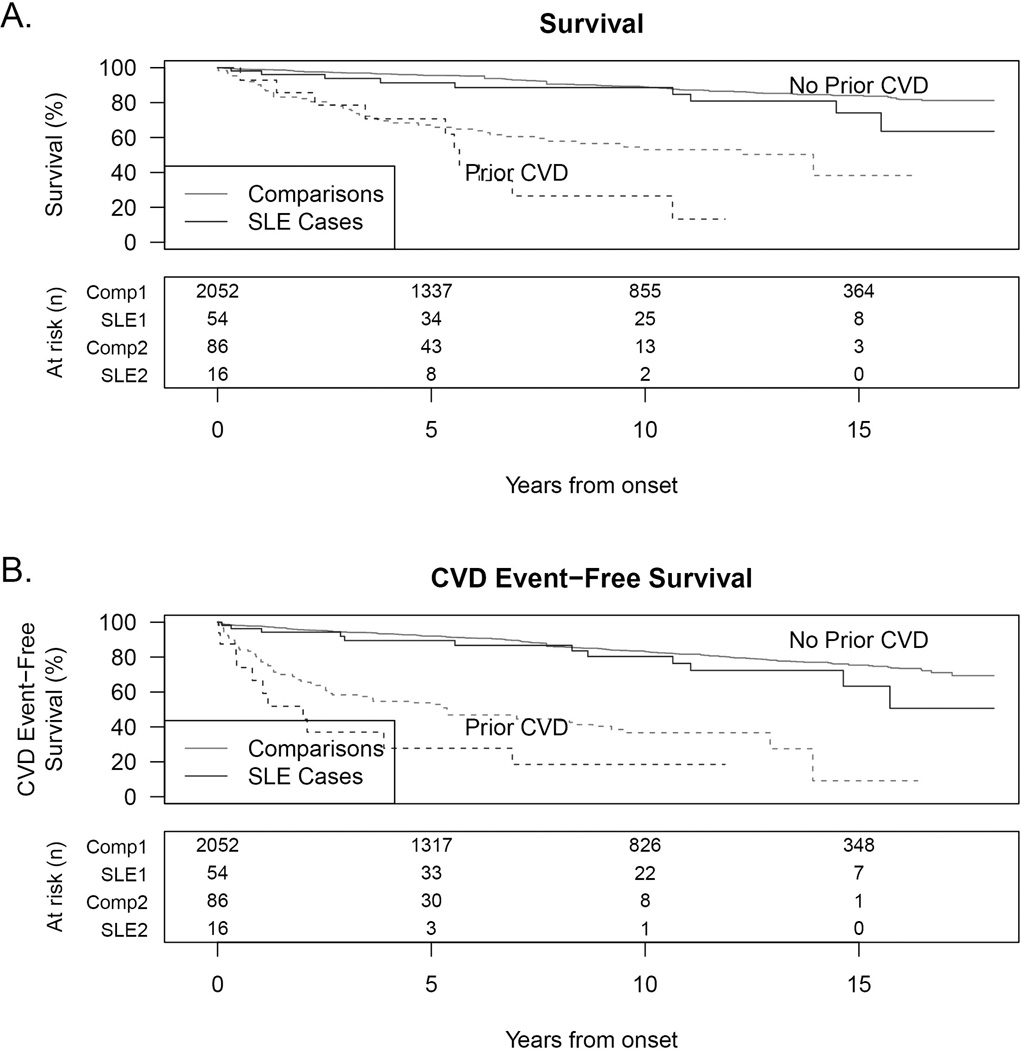
Post-hoc, the survival and composite death or CVD event analyses were repeated with stratification by baseline CVD status. K-M estimates were computed for SLE subjects and comparisons with and without baseline CVD (Figure 3) to estimate the additional hazard observed in the SLE cohort not accounted for by age, sex, and baseline CVD status. As shown in Figure 2, all-cause mortality was highest in SLE cases with prior CVD (dashed black line) which exceeded comparisons with prior CVD (dashed grey line), and also higher in SLE cases without prior CVD (solid black) than in comparisons without prior CVD (solid grey). The HRs for SLE cases compared to population comparisons, after matching by age, sex, and baseline CVD status, were 1.91, p=0.01 for mortality and HR=1.76, p=0.01 for the composite of death or new CVD event, similar to the unstratified analysis that yielded HRs of 2.09 and 1.83 respectively. Although sample size was limited to n=13 men with SLE, interaction of lupus and gender was not detected and mortality hazard ratios were not significantly different from women.
Figure 3.
Kaplan-Meier estimates for SLE cases and their age and sex-matched comparisons for (A) all-cause mortality and (B) the composite of death or CVD event endpoints stratified by baseline CVD status. Increased hazard ratios of SLE for event risk remained even after stratifying for baseline CVD status.
Lastly, SLE cases were also assessed for tobacco use and lipid testing as additional CVD risk factors. In the SLE patients at the time of diagnosis, 16.2% were current smokers, 36.8% were past smokers, and 47.1% indicated they had never smoked. There were no deaths among current smokers, and there was no indication of increased risk of mortality or death/CVD composite among current or past smokers compared to lupus cases who had never smoked (p=1.0). Lipid tests were ever performed in only 66% of SLE patients over a mean observation time of >7 years. Overall 84% of SLE men and only 61% of women were ever tested. Gaps in hyperlipidemia diagnosis and statin treatment (<20%) were also observed, suggesting suboptimal control of traditional cardiovascular risk particularly in SLE women, although this was not compared to population comparison subjects.
DISCUSSION
Among 70 incident SLE cases with mean age of 52 years at diagnosis, SLE cases experienced increased mortality and CVD event rates. Cases had approximately twice the hazard of death or CVD events compared to age and sex-matched comparisons. Though baseline rates for other traditional cardiovascular risk factors were similar between cases and comparisons, SLE cases had nearly four times the odds of baseline CVD 2 years prior to SLE diagnosis. Even after accounting for prior CVD status, in addition to matching age and sex, we observed HRs of SLE cases versus comparisons that remained significantly elevated. This shows that the differences in observed rates of mortality and new CVD events in SLE patients are not fully accounted for by differences in prior CVD, although that observation also deserves more study.
Our findings fit with published literature including a summary by Lim et al describing reported US population-based SLE incidence rates ranging from 3.4 to7.6 per 100,000 [19]. One US population-based study from the Mayo Rochester Epidemiology Project defined an SLE incidence cohort similar to ours including a mean age of 49 years old, and incidence estimates of 3.1–5.6/100,000 in another predominantly white Midwestern cohort [9]. They too noted increased mortality, though that study concluded follow-up in 1997 and did not specifically measure CVD events [9].
Reports on “late onset lupus,” defined as onset after age 50, also support our findings by describing lower female to male ratios 3–4:1, white race predilection, more arthritis, less renal disease, and high mortality [5,20,21]. In our incident SLE cohort, 50% of new cases were >50 years old at first SLE diagnosis and 61% had arthritis and renal and CNS disease were rare. Accordingly, use of medications beyond hydroxychloroquine or steroids was infrequent, yet SLE was associated with increased mortality and CVD event hazards. Our 10 year SLE survival rate of 74% was nearly identical to a recent late-onset SLE cohort from Spain [5], although most lupus studies with younger mean age cite >90% 10 year survival [3] likely reflecting age related hazards more than disease severity. While our cohort size did not allow multivariate modeling, patients more often received steroids than other immunosuppression, and a recent Hopkins Lupus Cohort correlated steroid use to SLE CVD events more strongly than SLE activity [22]. Likewise surveillance studies regarding atherosclerosis progression in SLE have reported that risk factors such as age, hypertension, and steroid exposure may interact to compound risk [23,24].
Cardiovascular disease remains a major cause of excess SLE deaths [2,25,26]. A frequently cited study demonstrated that women with lupus between the ages 35–44 years old had a hazard for cardiac events that was elevated 50-fold compared to age-matched peers [27]. However prior studies have drawn disparate conclusions regarding relationships between age at onset, sex, and lupus mortality [2–4], driving our interest in examining the CVD burden and mortality among this older, more gender-balanced cohort. Moreover, SLE patients with baseline CVD have often been excluded from studies examining the link between SLE and CVD [27]. To our knowledge, no prior studies have systematically captured CVD diagnosis and events preceding the SLE diagnosis.
In light of higher observed CVD event rates in the two years preceding SLE diagnosis, we must also consider possible explanations such as rapid atherosclerotic acceleration upon, or even prior to, disease onset in subclinical disease states, or perhaps delayed SLE diagnoses. Rural patients often have fewer health care encounters [28]. Moreover, with older-onset and male sex, a lupus diagnosis may be less frequently considered [29]. Conversely, early accelerated CVD may be an inherent feature of this predominantly older lupus cohort.
As with any study, some limitations are inherent in this analysis, and findings such as increased CVD events prior to SLE diagnosis should be re-examined in larger SLE populations beyond our well-described population-based cohort. We may have missed lupus patients who did not receive care or whose encounters used alternate ICD-9 codes including unspecified connective tissue disease or misclassified/attributed disease to drug-induced lupus. Next, although ascertainment of CVD risk factors and events used established algorithms, capture of CVD risk factors and outcomes may have been incomplete. Manual audits for the >21,000 comparison patient years were not feasible, but could have offered improved CVD event ascertainment beyond the validated CVD event ICD/procedure code algorithms used to compare SLE cases and comparison subjects. Moreover the 2 SLE cases with reported “failure to thrive” as a source of out of hospital death, and perhaps also in some comparison cases, may have been misclassified as non-cardiac. As cardiac risks, obesity, activity levels and comparison smoking rates were not systematically examined. However, smoking rates in SLE subjects did not differ significantly from regional smoking rates [30]. Sensitivity analysis showed that all SLE cases with current tobacco use were alive at last observation, with no evidence of increased risk of mortality or CV events in smokers. This suggests that smoking cannot explain observed differences. Also, although subjects were censored at the date they were known to have left regional MESA population, CVD events may have been missed due to subject migration or acute management outside of the region. Lastly, the region is 97% non-Hispanic white race, and our cases had a mean age of onset of 51 years; as such, care should be taken in applying the conclusions to other lupus patient populations. Important strengths of our approach include systematic ascertainment of nearly all SLE cases in a geographically defined population and strong clinical data systems to capture residence, medical encounters, cardiovascular events and mortality.
Overall, our study offers an updated look at survival among a US population-based lupus inception cohort, and serves as a comparison to many tertiary clinical cohort survival reports. It offers a novel examination of CVD diagnoses and event codes preceding SLE diagnosis and suggests new hypotheses regarding the timing of SLE diagnoses and atherosclerosis.
We observed increased mortality and CVD events despite mild, late onset SLE disease. This SLE-effect was independent of observing higher prior CVD at baseline, supporting the need for further CVD prevention efforts in lupus patients. Higher rates of CVD events in the 2 years preceding SLE diagnosis in lupus patients versus comparisons draw attention to questions of delayed SLE diagnosis versus early rapidly accelerated CVD in this predominantly older lupus cohort, and future work should explore these possibilities.
Acknowledgements
Grant support: Marshfield Clinic Research Foundation provided grant support for this project. Additional support provided by University of Wisconsin Institute for Clinical and Translational Research (UW-ICTR CTSA) 9U54TR000021 NIH/NCATS and the UW Health Innovation Program. Bartels is currently supported by NIH/NIAMS K23 AR062381-02.
Authors thank Lorelle Benetti for meticulous data preparation, and Joanna Wong and Courtney Maxcy for formatting and manuscript assistance.
Footnotes
Authors have no financial interests or potential conflict of interest or the appearance of a conflict of interest with regard to the work.
REFERENCES
- 1.Alarcon GS, McGwin G, Jr, Bastian HM, Roseman J, Lisse J, Fessler BJ, et al. Systemic lupus erythematosus in three ethnic groups. VII [correction of VIII]. Predictors of early mortality in the LUMINA cohort. LUMINA Study Group. Arthritis Rheum. 2001;45:191–202. doi: 10.1002/1529-0131(200104)45:2<191::AID-ANR173>3.0.CO;2-2. [DOI] [PubMed] [Google Scholar]
- 2.Cervera R, Khamashta MA, Font J, Sebastiani GD, Gil A, Lavilla P, et al. Morbidity and mortality in systemic lupus erythematosus during a 10-year period: a comparison of early and late manifestations in a cohort of 1,000 patients. Medicine (Baltimore) 2003;82:299–308. doi: 10.1097/01.md.0000091181.93122.55. [DOI] [PubMed] [Google Scholar]
- 3.Kasitanon N, Magder LS, Petri M. Predictors of survival in systemic lupus erythematosus. Medicine (Baltimore) 2006;85:147–156. doi: 10.1097/01.md.0000224709.70133.f7. [DOI] [PubMed] [Google Scholar]
- 4.Bernatsky S, Boivin JF, Joseph L, Manzi S, Ginzler E, Gladman DD, et al. Mortality in systemic lupus erythematosus. Arthritis Rheum. 2006;54:2550–2557. doi: 10.1002/art.21955. [DOI] [PubMed] [Google Scholar]
- 5.Alonso MD, Martinez-Vazquez F, de Teran TD, Miranda-Filloy JA, Dierssen T, Blanco R, et al. Late-onset systemic lupus erythematosus in Northwestern Spain: differences with early-onset systemic lupus erythematosus and literature review. Lupus. 2012;21:1135–1148. doi: 10.1177/0961203312450087. [DOI] [PubMed] [Google Scholar]
- 6.Ward MM, Pyun E, Studenski S. Long-term survival in systemic lupus erythematosus. Patient characteristics associated with poorer outcomes. Arthritis Rheum. 1995;38:274–283. doi: 10.1002/art.1780380218. [DOI] [PubMed] [Google Scholar]
- 7.Duran S, Apte M, Alarcon GS. Poverty, not ethnicity, accounts for the differential mortality rates among lupus patients of various ethnic groups. J Natl Med Assoc. 2007;99:1196–1198. [PMC free article] [PubMed] [Google Scholar]
- 8.Michet CJ, Jr, McKenna CH, Elveback LR, Kaslow RA, Kurland LT. Epidemiology of systemic lupus erythematosus and other connective tissue diseases in Rochester, Minnesota, 1950 through 1979. Mayo Clinic proceedings Mayo Clinic [Research Support, U.S. Gov't, P.H.S.] 1985;60:105–113. doi: 10.1016/s0025-6196(12)60294-8. [DOI] [PubMed] [Google Scholar]
- 9.Uramoto KM, Michet CJ, Jr, Thumboo J, Sunku J, O'Fallon WM, Gabriel SE. Trends in the incidence and mortality of systemic lupus erythematosus, 1950–1992. Arthritis Rheum. 1999;42:46–50. doi: 10.1002/1529-0131(199901)42:1<46::AID-ANR6>3.0.CO;2-2. [DOI] [PubMed] [Google Scholar]
- 10.Hochberg MC. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997;40:1725. doi: 10.1002/art.1780400928. [DOI] [PubMed] [Google Scholar]
- 11.Naleway AL, Davis ME, Greenlee RT, Wilson DA, McCarty DJ. Epidemiology of systemic lupus erythematosus in rural Wisconsin. Lupus. 2005;14:862–866. doi: 10.1191/0961203305lu2182xx. [DOI] [PubMed] [Google Scholar]
- 12.Miyakis S, Lockshin MD, Atsumi T, Branch DW, Brey RL, Cervera R, et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS) J Thromb Haemost [Consensus Development Conference Research Support, Non-U.S. Gov't] 2006;4:295–306. doi: 10.1111/j.1538-7836.2006.01753.x. [DOI] [PubMed] [Google Scholar]
- 13.Borzecki AM, Wong AT, Hickey EC, Ash AS, Berlowitz DR. Identifying hypertension-related comorbidities from administrative data: what's the optimal approach? Am J Med Qual. 2004;19:201–206. doi: 10.1177/106286060401900504. [DOI] [PubMed] [Google Scholar]
- 14.Rector TS, Wickstrom SL, Shah M, Thomas Greeenlee N, Rheault P, Rogowski J, et al. Specificity and sensitivity of claims-based algorithms for identifying members of Medicare+Choice health plans that have chronic medical conditions. Health Serv Res. 2004;39:1839–1857. doi: 10.1111/j.1475-6773.2004.00321.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Buccaneer Computer Systems & Service, Inc. Chronic Condition Data Warehouse User Manual. Warrenton, VA: Buccaneer Computer Systems & Service, Inc.; 2009. [cited 2010 April 13]. [updated 2009;]. Available from: http://ccwdata.org/downloads/CCW%20User%20Manual.pdf. [Google Scholar]
- 16.Thygesen K, Alpert JS, White HD. Universal definition of myocardial infarction. J Am Coll Cardiol. 2007;50:2173–2195. doi: 10.1016/j.jacc.2007.09.011. [DOI] [PubMed] [Google Scholar]
- 17.Ho KK, Anderson KM, Kannel WB, Grossman W, Levy D. Survival after the onset of congestive heart failure in Framingham Heart Study subjects. Circulation. 1993;88:107–115. doi: 10.1161/01.cir.88.1.107. [DOI] [PubMed] [Google Scholar]
- 18.Minnesota Department of Health. [cited August 12, 2010];Minnesota Stroke Registry: Case Ascertainment Protocol and Case Definition. 2010 [updated 2010 Version 1/19/10;]. Available from: http://www.mnstrokeregistry.org/documents/MSRCaseAscertainmentProtocol100119_000.pdf.
- 19.Lim SS, Drenkard C, McCune WJ, Helmick CG, Gordon C, Deguire P, et al. Population-based lupus registries: advancing our epidemiologic understanding. Arthritis Rheum. 2009;61:1462–1466. doi: 10.1002/art.24835. [DOI] [PubMed] [Google Scholar]
- 20.Boddaert J, Huong DL, Amoura Z, Wechsler B, Godeau P, Piette JC. Late-onset systemic lupus erythematosus: a personal series of 47 patients and pooled analysis of 714 cases in the literature. Medicine (Baltimore) 2004;83:348–359. doi: 10.1097/01.md.0000147737.57861.7c. [DOI] [PubMed] [Google Scholar]
- 21.Lalani S, Pope J, de Leon F, Peschken C. Clinical features and prognosis of late-onset systemic lupus erythematosus: results from the 1000 faces of lupus study. The Journal of rheumatology. 2010;37:38–44. doi: 10.3899/jrheum.080957. [DOI] [PubMed] [Google Scholar]
- 22.Magder LS, Petri M. Incidence of and risk factors for adverse cardiovascular events among patients with systemic lupus erythematosus. American Journal of Epidemiology. 2012;176:708–719. doi: 10.1093/aje/kws130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Doria A, Iaccarino L, Ghirardello A, Zampieri S, Arienti S, Sarzi-Puttini P, et al. Long-term prognosis and causes of death in systemic lupus erythematosus. Am J Med. 2006;119:700–706. doi: 10.1016/j.amjmed.2005.11.034. [DOI] [PubMed] [Google Scholar]
- 24.Kiani AN, Magder L, Petri M. Coronary calcium in systemic lupus erythematosus is associated with traditional cardiovascular risk factors, but not with disease activity. J Rheumatol. 2008;35:1300–1306. [PubMed] [Google Scholar]
- 25.Bjornadal L, Yin L, Granath F, Klareskog L, Ekbom A. Cardiovascular disease a hazard despite improved prognosis in patients with systemic lupus erythematosus: results from a Swedish population based study 1964–95. J Rheumatol. 2004;31:713–719. [PubMed] [Google Scholar]
- 26.Trager J, Ward MM. Mortality and causes of death in systemic lupus erythematosus. Curr Opin Rheumatol. 2001;13:345–351. doi: 10.1097/00002281-200109000-00002. [DOI] [PubMed] [Google Scholar]
- 27.Manzi S, Meilahn EN, Rairie JE, Conte CG, Medsger TA, Jr, Jansen-McWilliams L, et al. Age-specific incidence rates of myocardial infarction and angina in women with systemic lupus erythematosus: comparison with the Framingham Study. Am J Epidemiol. 1997;145:408–415. doi: 10.1093/oxfordjournals.aje.a009122. [DOI] [PubMed] [Google Scholar]
- 28.Larson SL, Fleishman JA. Rural-urban differences in usual source of care and ambulatory service use: analyses of national data using Urban Influence Codes. Med Care. 2003;41:III65–III74. doi: 10.1097/01.MLR.0000076053.28108.F2. [DOI] [PubMed] [Google Scholar]
- 29.Font J, Pallares L, Cervera R, Lopez-Soto A, Navarro M, Bosch X, et al. Systemic lupus erythematosus in the elderly: clinical and immunological characteristics. Ann Rheum Dis. 1991;50:702–705. doi: 10.1136/ard.50.10.702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. [updated 2012 2012; cited 01/30/2013];2011–2012 Wisconsin Collaborative for Healthcare Quality (WCHQ) Reports. 2012 Available from: http://www.wchq.org/reporting/measures.php.