Abstract
Background
The mechanisms of the electrocardiographic changes and arrhythmias in Brugada syndrome (BrS) remain controversial. Mutations in the sodium channel gene, SCN5A and regulatory proteins that reduce or eliminate sodium current (INa) have been linked to BrS. We studied the properties of a BS-associated SCN5A mutation in a protein kinase A (PKA) consensus phosphorylation site, R526H.
Methods and Results
In-vitro PKA phosphorylation was detected in the I-II linker peptide of wild type (WT) channels but not R526H or S528A (phosphorylation site) mutants. Cell surface expression of R526H and S528A channels were reduced compared with WT. Whole-cell INa through all channel variants revealed no significant differences in the steady-state activation, inactivation and recovery from inactivation. Peak current densities of the mutants were significantly reduced compared to WT. Infection of 2D cultures of neonatal rat ventricular myocytes with WT and mutant channels increased conduction velocity (CV) compared with non-infected cells. PKA stimulation significantly increased peak INa and CV of WT but not mutant channels. Oxidant stress inhibits cardiac Na currents; WT and mutant INa decreases with the intracellular application of NADH, an effect that is reversed by PKA stimulation in WT but not in R526H or S528A channels.
Conclusions
We identified a family with BrS and an SCN5A mutation in a PKA consensus phosphorylation site. The BrS mutation R526H is associated with a reduction in the basal level of INa and a failure of PKA stimulation to augment the current which may contribute to the predisposition to arrhythmias in patients with BrS, independent of the precipitants.
Keywords: ion channel, reactive oxygen species, SCN5A, mutation, sudden cardiac death, adrenergic stimulation, protein kinase A phosphorylation
Introduction
Mutations in the cardiac sodium channel gene, SCN5A, encoding NaV1.5 currents can produce of number of heritable diseases of cardiac rhythm and contractile function. Loss-of-function mutations have been associated with sudden cardiac death (SCD) and ST segment abnormalities in the right precordial leads of the electrocardiogram (ECG) referred to as Brugada syndrome (BrS)1. This may be part of a constellation of functional and structural abnormalities of the heart associated with perturbations of the ST segment.2-4
BrS is characterized by ECG alterations in the right precordial leads in the absence of structural cardiac abnormalities, preponderance in men particularly of Asian descent and a high rate of SCD. The prevalence of type 1 ECG changes is estimated to be ~5/10,000 5, 6 and more than double that in Japan and other parts of Asia.7, 8 The updated expert consensus statement on heritable arrhythmias recommends that a diagnosis of BrS be made in the presence of a spontaneous or drug-induced type 1 ECG pattern in at least one right precordial lead (V1-V3). 9
The genetic mechanisms of BrS have provided insights into the links between metabolism, ion channel function and cardiac arrhythmias. Approximately 20% of BrS are associated with mutations in SCN5A.10, 11 In SCN5A linked BrS, there is a reduction in Na current (INa) density that may result from mutations in the channel subunits or in modifiers of Na channel function. Mutations in glycerol phosphate dehydrogenase 1-like (GPD1-L) which is >80% homologous with glycerol phosphate dehydrogenase 1 (GPD1), 12 reduce INa through NAD/NADH dependent mechanisms 13, 14 via activation of protein kinase C (PKC). 15 There is evidence to support a direct effect on NaV 1.5 by PKC phosphorylation 15 and an NADH/PKC-mediated overproduction of reactive oxygen species (ROS) leading to a reduction in expressed INa.14 These data suggest inhibition of INa by pyridine nucleotides is mediated by mitochondrial produced ROS and that protein kinase A (PKA) activation may block ROS-induced reduction of the current and constitutes a treatment strategy for patients with BrS.13, 16
We studied a mutation in a patient with BrS presenting with a spontaneous type 1 ECG and history of syncope to elucidate the mechanism(s) of INa reduction and its reversal by adrenergic activation. Our data suggests a mechanism for the ECG changes and arrhythmias in BrS initiated by altered metabolism and an increase in oxidant burden that is not mitigated by beta adrenergic stimulation.
Methods and Materials
Detailed methods are provided in the on-line supplement. Molecular biological reagents including lentiviral vectors17-19 and ion channel plasmids were generated as previously described.20 All protocols followed US Department of Agriculture and National Institutes of Health guidelines and were approved by the Animal Care and Use Committee of the Johns Hopkins Medical Institutions.
Neonatal rat ventricular myocytes (NRVMs) were enzymatically isolated 21 and transduced as previously described.18, 22 Optical mapping was performed on plated monolayers of NRVMs and data were analyzed using custom-written scripts in MATLAB (Mathworks, Natick, MA). 18, 22
Whole cell Na currents (INa) were measured under voltage-clamp at room temperature (22°C) as previously described.23 The bath and pipette solutions are described in the on-line supplement. Recombinant human NaV1.5 peptides were expressed in E. coli, purified and incubated with PKA in the presence of γP32-labeled ATP. HEK 293 cells were transformed with WT NaV1.5, R526H and S528A NaV1.5 plasmids. Cell membrane proteins were labeled with biotin and captured on streptavidin beads. Total and membrane bound NaV1.5 expression was determined by Western blotting using an anti-Na channel antibody (Sigma S8809). The results are presented as mean ± SD or SEM. Statistical comparisons were made using a oneway ANOVA followed by Bonferroni/Dunn tests for multiple comparisons, and serial studies were assessed by repeated measures ANOVA. Statistical significance was assumed at P<0.05.
Results
The patient is a 33-year-old man who presented with two syncopal spells in rapid succession during micturition and without premonitory symptoms. In the field he had a normal heart rate and blood pressure. He had experienced palpitations after vigorous exercise in the past but had never had syncope. There was a family history of a paternal aunt who died suddenly around age 40 of unknown causes and a maternal uncle with Down Syndrome who died suddenly at age 11 in the setting of a febrile illness. The mutation was transmitted maternally, and the proband's mother had multiple episodes of syncope and near syncope associated with migraine headaches which were felt to be vasodepressor. Her baseline ECG exhibited 0.5-1 mm J-point elevation in leads V1 and V2 and a saddleback segment in V2, after procainamide (PCA) infusion both V1 and V2 ST segments were changed into a coved-type. (Supplemental Figure S1) The patient's younger sister carries the mutation and has a baseline incomplete RBBB, which did not change with PCA infusion.
His 12-lead electrocardiogram (ECG) revealed coved ST segments in leads V1 and V2 consistent with a Brugada type 1 ECG pattern (Figure 1A). A transthoracic echocardiogram was normal. A single chamber defibrillator was implanted.
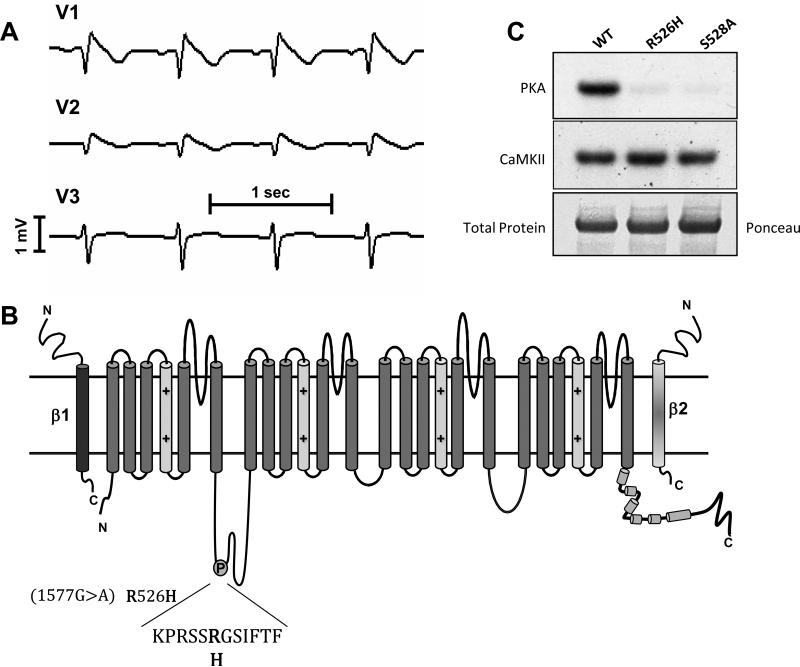
Figure 1.
(A) Leads V1-V3 of the patient's ECG exhibit typical domed J-point elevation. (B) A schematic of the NaV1.5 pore-forming α subunit and the β1 and β2 subunits. The disease causing mutation (R526H) is in the I-II interdomain linker of NaV1.5 in a canonical PKA phosphorylation recognition sequence. The residue two amino acids C-terminal, S528 was mutated to alanine in this study. (C) In vitro phosphorylation of I-II linker peptides. Purified fragments were incubated with PKA and calcium-calmodulin kinase II δ (CaMKII) in the presence of γP32 labeled ATP. The labeled peptides were separated by PAGE. Mutant peptides R526H and S528A are not labeled by PKA but are phosphorylated as efficiently as wild type in the presence of CaMKII.
BrS mutation disables PKA phosphorylation
DNA sequencing of SCN5A revealed a nucleotide transition at position 1577 encoding a missense mutation at codon 526 changing an arginine to histidine (R526H, Figure 1B). The minor allele frequency from the exome variant server is 0.016% 24 and is a chemically and structurally conservative change. 25, 26 However, the mutation resides in a PKA recognition sequence in the I-II interdomain linker of the channel, with serine 528 (S528) as the phosphorylated residue.
The I-II linker of NaV1.5 contains consensus phosphorylation sites for several kinases including PKA and calcium calmodulin kinase II (CaMKII). We created mutant I-II linker peptides with the clinical mutation, R526H and at the putative phosphorylation site, S528A. In vitro phosphorylation by PKA revealed complete elimination of 32P incorporation into the R526H and S528A peptides. In contrast, CaMKII phosphorylated all channel peptides equally (Figure 1C).
Altered channel regulation in response to stress
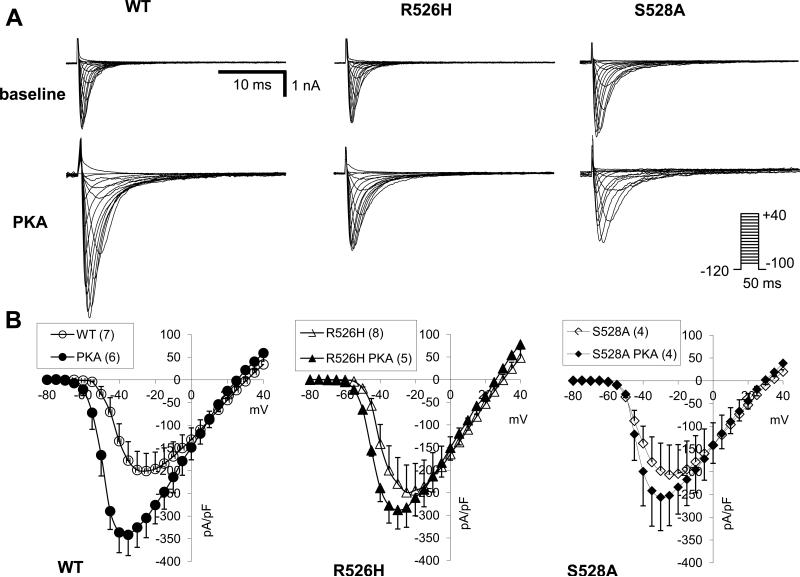
We speculate that stressors that reduce Na current density (oxidants, fever, Na channel blocking drugs) may in part be offset by PKA stimulation of the channel. In order to study the functional effects of these mutations in response to PKA phosphorylation, we expressed WT and mutant NaV1.5 currents in HEK293 cells. Selected families of currents in standard recording solutions revealed current densities through the mutant channels that were only modestly smaller than WT but not different from each other in the absence of PKA stimulation (Figure 2A and 2B). The baseline whole-cell properties including the current-voltage (I-V) relationships, voltage dependence and kinetics of gating, and kinetics of current decay were not different between WT and the mutants (Figure 2C, Table S1). In the presence of PKA,WT NaV1.5 currents were significantly up regulated with a hyperpolarizing shift in the peak I-V and activation curves (Figure 2), and hastened recovery from inactivation (Table S1). In contrast, neither the peak current, voltage dependence of gating or recovery kinetics (Figure 2, Table S1) of R526H or S528A were significantly affected by the addition of PKA.
Figure 2.
PKA regulation of Na current variants. (A) Representative families of current through WT, R526H and S528A channels expressed in HEK 293 cells in the presence and absence of PKA activation. (B) I-V relationships in the presence (filled symbols) and absence (open symbols) of PKA stimulation. There is no increase in the current through the mutant channels. (C) Activation (circles) and steady state inactivation (triangles) curves in the presence (filled symbols) and absence (open symbols) of PKA stimulation. The data are fit to a Boltzman function as described in the methods. There are no significant differences between the WT and mutant channels in the basal voltage dependence and kinetics of gating.
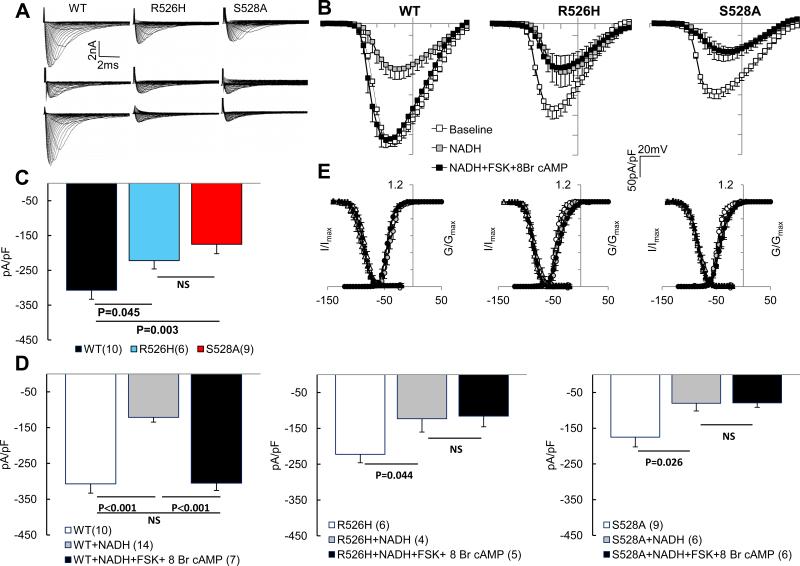
Oxidative stress and an increase in glycolysis will lead to increased levels of cytosolic NADH which rapidly decreases INa. We recorded currents in a metabolic stabilizing solution (Supplemental Methods, inhibiting kinases and phosphatases) to examine the effect of intracellular NADH on the WT and mutant currents. The baseline peak density of the WT current was significantly larger than either R526H or S528A mutant currents (Figure 3 A-C). The addition of 100 μM NADH to the pipette solution reduced the average INa by >50% in all channel variants (Figure 3A and 3B). The addition of NADH had no significant effect on the decay kinetics or voltage dependence of gating of WT NaV1.5 or the mutant channels (Figure 3E, Table S2). We speculated that the current density might be restored by PKA in the setting of an increase in NADH and that this restoration might be compromised in the mutant channels. In order to study the effect of activation PKA signaling on NADH induced suppression of NaV1.5 current, cells were incubated with forskolin (FSK) and 8-Br cAMP for 20 minutes prior to patch clamp recording. Incubation with FSK and 8-Br cAMP completely reversed the down regulation of WT NaV1.5 current by intracellular NADH (Figure 3A, 3B, 3D) and produced a modest but significant hyperpolarizing shift in the voltage dependence of activation gating (Table S2). The reductions in R526H and S528A currents by NADH were not affected by the addition of FSK and 8-Br cAMP (Figure 3D, Table S2).
Figure 3.
Regulation of Na channel variants by NADH and PKA. The currents are recorded with fluoride in the pipette. (A) Representative families of current through WT, R526H and S528A channels expressed in HEK 293 cells (top row), in the presence of 100 μM NADH (middle) and NADH plus PKA activation with forskolin and 8-Br-cAMP (bottom). (B) I-V relationships in the absence (open symbols), presence of NADH (gray symbols) and NADH + PKA stimulation (filled symbols). NADH reduces the current through all channel variants, while PKA activation does not increase the current density through the mutant channels. (C) Bar plots of peak current densities of WT (black), R526H (blue) and S528A (red) under basal conditions. (D) Summary bar plots for WT, R526H and S528A channel peak current density at baseline (open bars) with NADH (gray bars) and NADH + PKA activation (black bars). (E) Activation (circles) and steady state inactivation (triangles) curves in the absence (open symbols), presence of NADH (gray symbols) and NADH + PKA stimulation (filled symbols) for each of the channel variants, there are no significant differences in any of the variants under any of the conditions.
Trafficking of mutant channels
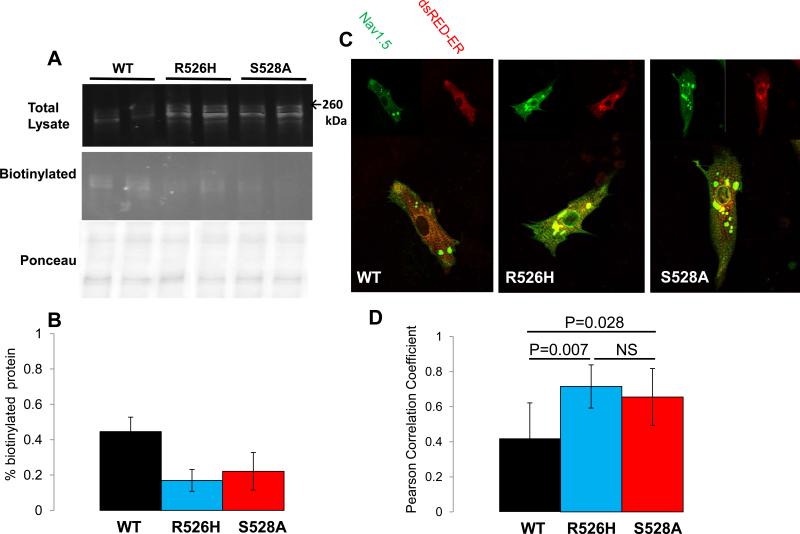
The reduction in basal current density of R526H and S528A channels without a change in biophysical characteristics compared to WT suggested the possibility of an alteration in trafficking to the surface membrane. Surface membrane expression of WT and mutant NaV1.5 channels in HEK 293 was quantified by labeling intact cells with biotin and purifying the protein on immobilized streptavadin. Overall expression of the channel proteins did not differ; however, Figure 4A illustrates that even with less WT NaV1.5 protein in the total lysate, a greater proportion of the WT channel was biotinylated compared with both the R526H and S528A mutants (Figure 4B).
Figure 4.
Mutations and channel trafficking. (A) Surface membrane expression of the channel measured by biotinylation in intact cells. The top panel shows total lysate, the middle panel the protein captured on streptavadin beads stained with an anti-NaV1.5 antibody and the bottom panel is a Ponceau stained blot demonstrating equivalent loading. (B) The percentage of total lysate modified by biotin is significantly decreased in the R526H (blue bar) and S528A (red bars) mutant channels compared to WT (black bars). (C) Immuncytochemical localization of the channel proteins in transduced NRVMs. The NaV1.5 channel variant is co-transfected with the endoplasmic reticulum marker dsRed-ER. There is significantly increased co-localization of both mutant channel proteins with the ER marker compared to the wild type. (D) Bar plots of the percent co-localization of the channel protein and dsRed-ER.
In order to study trafficking in a cardiac cell background, neonatal rat ventricular myocytes (NRVMs) were infected with lentiviruses containing the WT or mutant channels fused to eGFP at the carboxyl terminus. The channel-infected NRVMs were co-transfected with an endoplasmic reticulum marker dsRED-ER. Consistent with the biotinylation experiments, NRVMs infected with the viruses encoding the R526H and S528A mutants exhibited more channel protein in subcellular membranes, co-localized with dsRED-ER compared with WT channels (Figures 4C and 4D).
Effect of mutants on conduction velocity (CV)
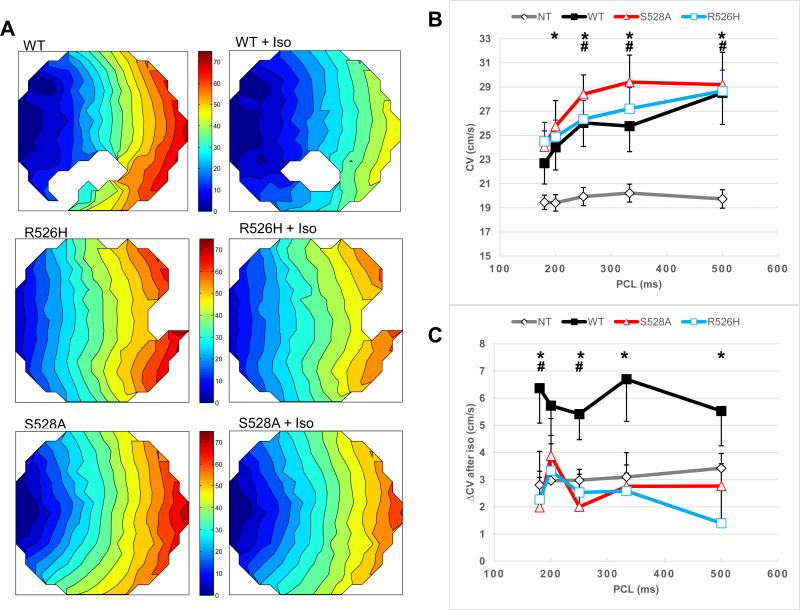
The functional consequences of the reduced surface expression and altered regulation of the mutant channels were studied using optical mapping of 2D cultures of NRVMs. In order to control for variability in baseline CVs cultures that were transduced with Na channel variants were compared with naïve non-transfected (NT) cultures. The cultures were stimulated over a range of pacing cycle lengths (175 – 500 msec) to determine the CVs; isochronal maps at baseline and in the presence of isoproterenol are shown for WT, R526H and S528A transfected cultures (Figure 5A). The baseline CVs of NRVM cultures infected with WT NaV1.5 and the mutant channels were similar and approximately 20-50% (5-10 cm/s) faster than NT cultures over a range of pacing cycle lengths (Figure 5B).
Figure 5.
Optical mapping of Na channel variants. (A) Representative isochronal maps from cultures of NRVMs infected with the Na channel variants and stimulated at the left side of the culture. (B) Plots of the average conduction velocity (CV) ± SEM of WT, R526H and S528A transduced cultures and non-infected (NT) control cultures over a range of pacing cycle lengths (PCL). CV is consistently faster in WT, R526H and S528A NaV1.5 infected cultures compared with the non-transduced (NT) controls over the entire range of PCLs. *p<0.05, all vs. NT; #p<0.01, all vs. NT. (C) Plots of the change in average CV ± SEM of WT, R526H and S528A transduced cultures and NT control cultures over a range of PCL after application of 1 μM isoproterenol. Change in CV of WT transduced cultures is significantly larger than CV changes of NT cultures and mutant transduced cultures after isoproterenol application. *p<0.05, R526H vs. WT; #p<0.05, S528A vs. WT. The number cultures studied at all PCLs in both the absence and presence of isoproterenol were NT (14), WT (13), S528A (5) and R526H (11).
At more rapid pacing rates accumulation of ROS is anticipated which would decrease INa. The CVs of the WT and mutant channel transduced cultures are greater than that of NT control cultures at slower pacing rates, at faster rates the difference is diminished and no longer significant at a PCL of 180 msec (Figure 5B).. The CVs of the WT infected cultures are significantly increased by exposure to isoproterenol while that of the mutant infected cultures is no more responsive than NT control cultures (Figure 5C).
Discussion
Mutations in the Na channel, SCN5A 10or regulatory proteins such as GPD1L 12 that reduce current have been associated with BrS and sudden infant death syndrome (SIDS). In both conditions arrhythmias are more prevalent under conditions of various types of stress (oxidant stress, fever, ischemia, Na channel blocking drugs). We describe an SCN5A mutation in a patient with BrS that produces both a chronic reduction in INa and absence of augmentation of the current by adrenergic stimulation. Interestingly, the reduction in the basal current produced by the R526H and S528A mutations compared to WT NaV1.5 is more pronounced when fluoride is included in the patch pipette to inhibit protein phosphatase, consistent with increased basal phosphorylation of the WT channel. The elimination of a PKA site appears to be central to both the signaling and trafficking deficits. The disease causing mutation, R526H, which is chemically and structurally conservative,25, 26 eliminates the basic priming residue in a consensus PKA phosphorylation recognition sequence. Replacement of the phosphorylation target residue, S528A, alters channel trafficking and regulation by PKA in a manner that is comparable to the disease-causing mutation and similar to previously described defects in PKA-mediated INa potentiation.27 The mutations reduce channel expression at the membrane surface as assessed by biotinylation and immunocytochemistry studies (Figure 4) and are reflected in a lower expressed current density in HEK cells (Figure 3).
The reduction in INa is further complicated by the absence of a response of the channel to adrenergic stimulation. Neither the direct activation of adenylyl cyclase or β-AR stimulation with isoproterenol had a significant effect on current density or CV mediated by the mutant channel variants. These findings suggest a mechanism of stress-induced arrhythmias that may be due to changes of cellular redox balance with alterations in the ratio of NAD+/NADH and consequent reduction in INa. Such metabolic stressors are typically accompanied by activation of the sympathetic nervous system which may partially offset the redox-induced reduction in INa and would dampen stress-induced ECG changes in BrS and alterations in CV. In such cases, arrhythmias would not be predicted to be induced by sympathetic activation but instead would be mitigated by an increase in sympathetic tone. This concept is consistent with the utility of isoproterenol in the treatment of arrhythmic storm in patients with BrS.28-30
There are a number of mechanisms by which metabolism has been proposed to alter the Na current. Direct oxidation of the channel can alter channel conductance,31, 32 gating33, 34 and trafficking.35 Mutations associated with BrS have informed other mechanisms of metabolic regulation of Na channels. NADH is generated from NAD+ during glycolysis, and NAD+ must be regenerated for glycolysis to continue. GPD1 reduces dihydroxyacetone phosphate to glycerol-3-phosphate, causing oxidation of NADH and regeneration of NAD+ with the electrons released from this reaction entering the electron transport chain. GPD1-L is highly homologous to GPD1 and harbors mutations associated with BrS.12 When mutant GPD1-L is co-expressed with NaV 1.5, INa is significantly reduced.12,15 The BrS mutations have been proposed to reduce the enzymatic function of GPD1-L and would be expected to increase intracellular NADH levels. An increase in NADH possibly via activation of PKC, 13 enhancing phosphorylation of complex III resulting in an increase in reactive oxygen species (ROS) release, 14 reduces channel function. Alternatively, inactivating mutants of GPD1-L have been proposed to increase PKC phosphorylation of the Na channel in the III-IV linker, reducing current density.15 Increased mitochondrial ROS release, elevated levels of NADH and PKC activation have been implicated in INa down regulation in models of non-ischemic cardiomyopathy and in CV slowing in diseased human ventricles. 36 Our work supports another mechanism of coupling of metabolism to channel function through altered PKA activation.27 Increased NADH causes a reduction in current through WT and mutant R526H and S528A expressed channels. The absence of the PKA phosphorylation site in the mutant channels precludes current augmentation in the setting of sympathetic activation.
The mechanism of arrhythmias in this SCN5A mediated BrS appears to involve a substrate that is characterized by decreased basal current expression as a result of altered mutant channel trafficking. Although it is difficult to infer general mechanisms from a single family, this disease-causing mutation produces the requisite basal reduction in Na current compounded by defective current augmentation that generates the substrate for a potentially lethal arrhythmia. A trigger that further reduces INa – through any number of mechanisms such as alterations in glycolysis, ROS levels or PKC activation cannot be mitigated by PKA activation, leading to a further reduction in current. The absence of a PKA-mediated reversal of NADH- induced current reduction (Figure 3) is consistent with the lack of effect on conduction in infected NRVMs. The reduction of INa could alter either regional dispersion of repolarization or conduction in the heart; one or both may contribute to the genesis of the potentially lethal ventricular arrhythmias in patients harboring this mutation.
Supplementary Material
Acknowledgments
We thank Deborah DiSilvestre, Victoria Halperin Kuhns and Dr. Yan Li Tian for technical support.
Funding Sources: The work was supported by R01 HL 50411 (GFT) and a Mid-Atlantic Affiliate AHA fellowship (GK).
Footnotes
Conflict of Interest Disclosures: None.
References
- 1.Antzelevitch C, Brugada P, Borggrefe M, Brugada J, Brugada R, Corrado D, et al. Brugada syndrome: Report of the second consensus conference: Endorsed by the Heart Rhythm Society and the European Heart Rhythm Association. Circulation. 2005;111:659–670. doi: 10.1161/01.CIR.0000152479.54298.51. [DOI] [PubMed] [Google Scholar]
- 2.Martini B, Nava A, Thiene G, Buja GF, Canciani B, Scognamiglio R, et al. Ventricular fibrillation without apparent heart disease: Description of six cases. Am Heart J. 1989;118:1203–1209. doi: 10.1016/0002-8703(89)90011-2. [DOI] [PubMed] [Google Scholar]
- 3.Haissaguerre M, Derval N, Sacher F, Jesel L, Deisenhofer I, de Roy L, et al. Sudden cardiac arrest associated with early repolarization. N Engl J Med. 2008;358:2016–2023. doi: 10.1056/NEJMoa071968. [DOI] [PubMed] [Google Scholar]
- 4.Hoogendijk MG, Opthof T, Postema PG, Wilde AA, de Bakker JM, Coronel R. The Brugada ECG pattern: A marker of channelopathy, structural heart disease, or neither? Toward a unifying mechanism of the Brugada syndrome. Circ Arrhythm Electrophysiol. 2010;3:283–290. doi: 10.1161/CIRCEP.110.937029. [DOI] [PubMed] [Google Scholar]
- 5.Patel SS, Anees S, Ferrick KJ. Prevalence of a Brugada pattern electrocardiogram in an urban population in the United States. Pacing Clin Electrophysiol. 2009;32:704–708. doi: 10.1111/j.1540-8159.2009.02354.x. [DOI] [PubMed] [Google Scholar]
- 6.Sinner MF, Pfeufer A, Perz S, Schulze-Bahr E, Monnig G, Eckardt L, et al. Spontaneous Brugada electrocardiogram patterns are rare in the German general population: Results from the KORA study. Europace. 2009;11:1338–1344. doi: 10.1093/europace/eup205. [DOI] [PubMed] [Google Scholar]
- 7.Sidik NP, Quay CN, Loh FC, Chen LY. Prevalence of Brugada sign and syndrome in patients presenting with arrhythmic symptoms at a heart rhythm clinic in singapore. Europace. 2009;11:650–656. doi: 10.1093/europace/eup079. [DOI] [PubMed] [Google Scholar]
- 8.Nademanee K, Veerakul G, Nimmannit S, Chaowakul V, Bhuripanyo K, Likittanasombat K, et al. Arrhythmogenic marker for the sudden unexplained death syndrome in thai men. Circulation. 1997;96:2595–2600. doi: 10.1161/01.cir.96.8.2595. [DOI] [PubMed] [Google Scholar]
- 9.Priori SG, Wilde AA, Horie M, Cho Y, Behr ER, Berul C, et al. Executive summary: HRS/EHRA/APHRS expert consensus statement on the diagnosis and management of patients with inherited primary arrhythmia syndromes. Europace. 2013;15:1389–1406. doi: 10.1093/europace/eut272. [DOI] [PubMed] [Google Scholar]
- 10.Chen Q, Kirsch GE, Zhang D, Brugada R, Brugada J, Brugada P, et al. Genetic basis and molecular mechanism for idiopathic ventricular fibrillation. Nature. 1998;392:293–296. doi: 10.1038/32675. [DOI] [PubMed] [Google Scholar]
- 11.Miura D, Nakamura K, Ohe T. Update on genetic analysis in brugada syndrome. Heart Rhythm. 2008;5:1495–1496. doi: 10.1016/j.hrthm.2008.07.026. [DOI] [PubMed] [Google Scholar]
- 12.London B, Michalec M, Mehdi H, Zhu X, Kerchner L, Sanyal S, et al. Mutation in glycerol-3-phosphate dehydrogenase 1 like gene (GPD1-L) decreases cardiac Na+ current and causes inherited arrhythmias. Circulation. 2007;116:2260–2268. doi: 10.1161/CIRCULATIONAHA.107.703330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Liu M, Sanyal S, Gao G, Gurung IS, Zhu X, Gaconnet G, et al. Cardiac Na+ current regulation by pyridine nucleotides. Circ Res. 2009;105:737–745. doi: 10.1161/CIRCRESAHA.109.197277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Liu M, Liu H, Dudley SC., Jr. Reactive oxygen species originating from mitochondria regulate the cardiac sodium channel. Circ Res. 2010;107:967–974. doi: 10.1161/CIRCRESAHA.110.220673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Valdivia CR, Ueda K, Ackerman MJ, Makielski JC. GPD1-L links redox state to cardiac excitability by PKC-dependent phosphorylation of the sodium channel SCN5A. Am J Physiol Heart Circ Physiol. 2009;297:H1446–1452. doi: 10.1152/ajpheart.00513.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chahine M. Cardiac metabolic state and Brugada syndrome: A link revealed. Circ Res. 2009;105:721–723. doi: 10.1161/CIRCRESAHA.109.208405. [DOI] [PubMed] [Google Scholar]
- 17.Fleming J, Ginn SL, Weinberger RP, Trahair TN, Smythe JA, Alexander IE. Adeno-associated virus and lentivirus vectors mediate efficient and sustained transduction of cultured mouse and human dorsal root ganglia sensory neurons. Hum Gene Ther. 2001;12:77–86. doi: 10.1089/104303401450997. [DOI] [PubMed] [Google Scholar]
- 18.Kizana E, Chang CY, Cingolani E, Ramirez-Correa GA, Sekar RB, Abraham MR, et al. Gene transfer of connexin43 mutants attenuates coupling in cardiomyocytes: Novel basis for modulation of cardiac conduction by gene therapy. Circ Res. 2007;100:1597–1604. doi: 10.1161/CIRCRESAHA.106.144956. [DOI] [PubMed] [Google Scholar]
- 19.Sekar RB, Kizana E, Cho HC, Molitoris JM, Hesketh GG, Eaton BP, et al. IK1 heterogeneity affects genesis and stability of spiral waves in cardiac myocyte monolayers. Circ Res. 2009;104:355–364. doi: 10.1161/CIRCRESAHA.108.178335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Biswas S, DiSilvestre D, Tian Y, Halperin VL, Tomaselli GF. Calcium-mediated dual-mode regulation of cardiac sodium channel gating. Circ Res. 2009;104:870–878. doi: 10.1161/CIRCRESAHA.108.193565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bursac N, Parker KK, Iravanian S, Tung L. Cardiomyocyte cultures with controlled macroscopic anisotropy: A model for functional electrophysiological studies of cardiac muscle. Circ Res. 2002;91:e45–54. doi: 10.1161/01.res.0000047530.88338.eb. [DOI] [PubMed] [Google Scholar]
- 22.Sekar RB, Kizana E, Smith RR, Barth AS, Zhang Y, Marban E, et al. Lentiviral vector-mediated expression of GFP or Kir2.1 alters the electrophysiology of neonatal rat ventricular myocytes without inducing cytotoxicity. Am J Physiol Heart Circ Physiol. 2007;293:H2757–2770. doi: 10.1152/ajpheart.00477.2007. [DOI] [PubMed] [Google Scholar]
- 23.Aiba T, Hesketh GG, Liu T, Carlisle R, Villa-Abrille MC, O'Rourke B, et al. Na+ channel regulation by Ca2+/calmodulin and Ca2+/calmodulin-dependent protein kinase II in guinea-pig ventricular myocytes. Cardiovasc Res. 2010;85:454–463. doi: 10.1093/cvr/cvp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Exome variant server. http://evs.gs.washington.edu/EVS/ date last accessed November 11,2013.
- 25.Grantham R. Amino acid difference formula to help explain protein evolution. Science. 1974;185:862–864. doi: 10.1126/science.185.4154.862. [DOI] [PubMed] [Google Scholar]
- 26.Adzhubei IA, Schmidt S, Peshkin L, Ramensky VE, Gerasimova A, Bork P, et al. A method and server for predicting damaging missense mutations. Nat Methods. 2010;7:248–249. doi: 10.1038/nmeth0410-248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhou J, Shin HG, Yi J, Shen W, Williams CP, Murray KT. Phosphorylation and putative er retention signals are required for protein kinase a-mediated potentiation of cardiac sodium current. Circ Res. 2002;91:540–546. doi: 10.1161/01.res.0000033598.00903.27. [DOI] [PubMed] [Google Scholar]
- 28.Miyazaki T, Mitamura H, Miyoshi S, Soejima K, Aizawa Y, Ogawa S. Autonomic and antiarrhythmic drug modulation of st segment elevation in patients with Brugada syndrome. J Am Coll Cardiol. 1996;27:1061–1070. doi: 10.1016/0735-1097(95)00613-3. [DOI] [PubMed] [Google Scholar]
- 29.Tanaka H, Kinoshita O, Uchikawa S, Kasai H, Nakamura M, Izawa A, et al. Successful prevention of recurrent ventricular fibrillation by intravenous isoproterenol in a patient with Brugada syndrome. Pacing Clin Electrophysiol. 2001;24:1293–1294. doi: 10.1046/j.1460-9592.2001.01293.x. [DOI] [PubMed] [Google Scholar]
- 30.Maury P, Couderc P, Delay M, Boveda S, Brugada J. Electrical storm in Brugada syndrome successfully treated using isoprenaline. Europace. 2004;6:130–133. doi: 10.1016/j.eupc.2003.11.009. [DOI] [PubMed] [Google Scholar]
- 31.Spalding BC. Properties of toxin-resistant sodium channels produced by chemical modification in frog skeletal muscle. J Physiol. 1980;305:485–500. doi: 10.1113/jphysiol.1980.sp013377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sigworth FJ, Spalding BC. Chemical modification reduces the conductance of sodium channels in nerve. Nature. 1980;283:293–295. doi: 10.1038/283293a0. [DOI] [PubMed] [Google Scholar]
- 33.Ward CA, Giles WR. Ionic mechanism of the effects of hydrogen peroxide in rat ventricular myocytes. J Physiol. 1997;500:631–642. doi: 10.1113/jphysiol.1997.sp022048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Song Y, Shryock JC, Wagner S, Maier LS, Belardinelli L. Blocking late sodium current reduces hydrogen peroxide-induced arrhythmogenic activity and contractile dysfunction. J Pharmacol Exp Ther. 2006;318:214–222. doi: 10.1124/jpet.106.101832. [DOI] [PubMed] [Google Scholar]
- 35.Hallaq H, Wang DW, Kunic JD, George AL, Jr., Wells KS, Murray KT. Activation of protein kinase c alters the intracellular distribution and mobility of cardiac Na+ channels. Am J Physiol Heart Circ Physiol. 2012;302:H782–789. doi: 10.1152/ajpheart.00817.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu M, Gu L, Sulkin MS, Liu H, Jeong EM, Greener I, et al. Mitochondrial dysfunction causing cardiac sodium channel downregulation in cardiomyopathy. J Mol Cell Cardiol. 2013;54:25–34. doi: 10.1016/j.yjmcc.2012.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.