Abstract
Normal hypothalamic-pituitary-adrenal (HPA) axis activity leading to rhythmic and episodic release of adrenal glucocorticoids is essential for body homeostasis and survival during stress. Acting through specific intracellular receptors in the brain and periphery, glucocorticoids regulate behavior, metabolic, cardiovascular, immune, and neuroendocrine activities. In contrast to chronic elevated levels, circadian and acute stress-induced increases in glucocorticoids are necessary for hippocampal neuronal survival and memory acquisition and consolidation, through inhibiting apoptosis, facilitating glutamate transmission and inducing immediate early genes and spine formation. In addition to its metabolic actions leading to increasing energy availability, glucocorticoids have profound effects on feeding behavior, mainly through modulation of orexigenic and anorixegenic neuropeptides. Evidence is also emerging that in addition to the recognized immune suppressive actions of glucocorticoids by counteracting adrenergic proinflammatory actions, circadian elevations have priming effects in the immune system, potentiating acute defensive responses. In addition, negative feedback by glucocorticoids involves multiple mechanisms leading to limiting HPA axis activation and preventing deleterious effects of excessive glucocorticoid production. Adequate glucocorticoid secretion to meet body demands is tightly regulated by a complex neural circuitry controlling hypothalamic corticotrophin releasing hormone (CRH) and vasopressin secretion, the main regulators of pituitary adrenocorticotrophic hormone (ACTH). Rapid feedback mechanisms, likely involving non-genomic actions of glucocorticoids, mediate immediate inhibition of hypothalamic CRH and ACTH secretion, while intermediate and delayed mechanisms mediated by genomic actions involve modulation of limbic circuitry and peripheral metabolic messengers. Consistent with their key adaptive roles, HPA axis components are evolutionarily conserved, being present in the earliest vertebrates. Understanding these basic mechanisms may lead to novel approaches for the development of diagnostic and therapeutic tools for disorders related to stress and alterations of glucocorticoid secretion.
Keywords: CRH, ACTH, Glucocorticoids, cortisol/corticosterone
Introduction
Living organisms are constantly challenged by external or internal challenges or stressors threatening the internal environment equilibrium or homeostasis (1). Adaptation to this changing environment requires coordinated activation of multiple neuroendocrine responses, prominently including activation of the hypothalamic pituitary adrenal (HPA) axis (2). Control of HPA axis activity is complex involving sensors conveying signals to corticotrophin releasing hormone (CRH) neurones in the hypothalamic paraventricular nucleus (PVN), either through direct neural connections, or indirectly through pathways relaying on limbic structures such as the amygdala, frontal cortex, bed nucleus of the stria terminalis (BST) and hippocampus (2). CRH and vasopressin, released from parvocellular neurones of the hypothalamic paraventricular nucleus into the pituitary portal circulation, increases the secretion of adrenocorticotrophic hormone (ACTH) from pituitary corticotrophs, which in turn stimulates glucocorticoid secretion from the adrenal. The HPA axis is highly conserved through evolution, with its components being present in early vertebrates. The secretion of glucocorticoids (GCs) (cortisol in humans, corticosterone in rats and mice) is episodic, following circadian (daily) and ultradian (hourly) rhythms, and shows marked but transient increases after exposure to stressors. This episodic nature of glucocorticoid secretion is critical for the transcriptional activity of the steroid (3).
The action of GCs involves binding, also evolutionary conserved, to intracellular receptors belonging to the nuclear receptor family, glucocorticoid receptors type 1 or mineralocorticoid receptor (MR), and type 2 or glucocorticoid receptor (GR) (4). Although MR have equally high affinity to GCs and mineralocorticoids, the presence of 11-beta-hydroxysteroid dehydrogenase in mineralocorticoid target tissues protects MR from the much higher circulating levels of GCs, by inactivating GCs. Following binding to the steroid, GR translocates to the nucleus, where it interacts with glucocorticoid response elements in the promoter of glucocorticoid target genes inducing transcriptional activation or repression. Some biological actions of GCs are too fast to be mediated by genomic actions (which require protein synthesis) and they are believed to depend on membrane receptors (5).
GCs are essential for stress adaptation by controlling energy supply through stimulation of glycolysis, gluconeogenesis and lipolysis, as well as inducing proteolysis to supply amino acids as substrates for gluconeogenesis. In addition, GCs modulate the immune system and the synthesis and action of a number of hormones. By acting directly in neurones or by modulating the synthesis, secretion or/and activity of neuropeptides and neurotransmitters, GCs affect memory acquisition and consolidation, and impact the function of other neuroendocrine systems, such as central control of metabolic activity, feeding behaviour and reproductive activity. Given these wide ranging effects of GCs, either glucocorticoid deficiency or failure to limit HPA axis activation will have profound consequences on the organism well-being. An important mechanism for maintaining episodic HPA axis activation and for limiting HPA axis activity is the negative feedback by GCs, resulting in inhibition of expression and secretion of hypothalamic CRH and pituitary ACTH.
This review covers novel aspects about regulation and function of GCs, including the critical importance of adequate GC regulation on synaptic remodeling, interactions between the HPA axis and inflammatory processes and other neuroendocrine systems such as control of appetite, as well as the molecular mechanisms, brain and peripheral circuitries involved in HPA axis regulation and glucocorticoid feedback.
Molecular mechanisms of glucocorticoid feedback
Inhibitory feedback by GCs at the central and pituitary levels plays a major role in reducing HPA axis responses to stress. As discussed below, an important mechanism for glucocorticoid feedback involves modulation of direct and indirect circuitry controlling CRH neuron activity. In addition, GCs inhibit CRH expression (6) and secretion (7) in the hypothalamic PVN, and inhibit both ACTH output (8, 9) and the transcription of the precursor protein proopiomelanocortin (POMC) (10, 11) in the anterior pituitary corticotroph. Concerning transcriptional regulation, it is clear that inhibition of POMC transcription by GCs is a genomic effect dependent on interaction of the GR with the POMC promoter (10, 11). However, the mechanism by which GCs inhibit CRH transcription is less obvious (12). In addition, there is evidence that glucocorticoids may influence translational activity and mRNA stability of both POMC and CRH mRNA (13, 14).
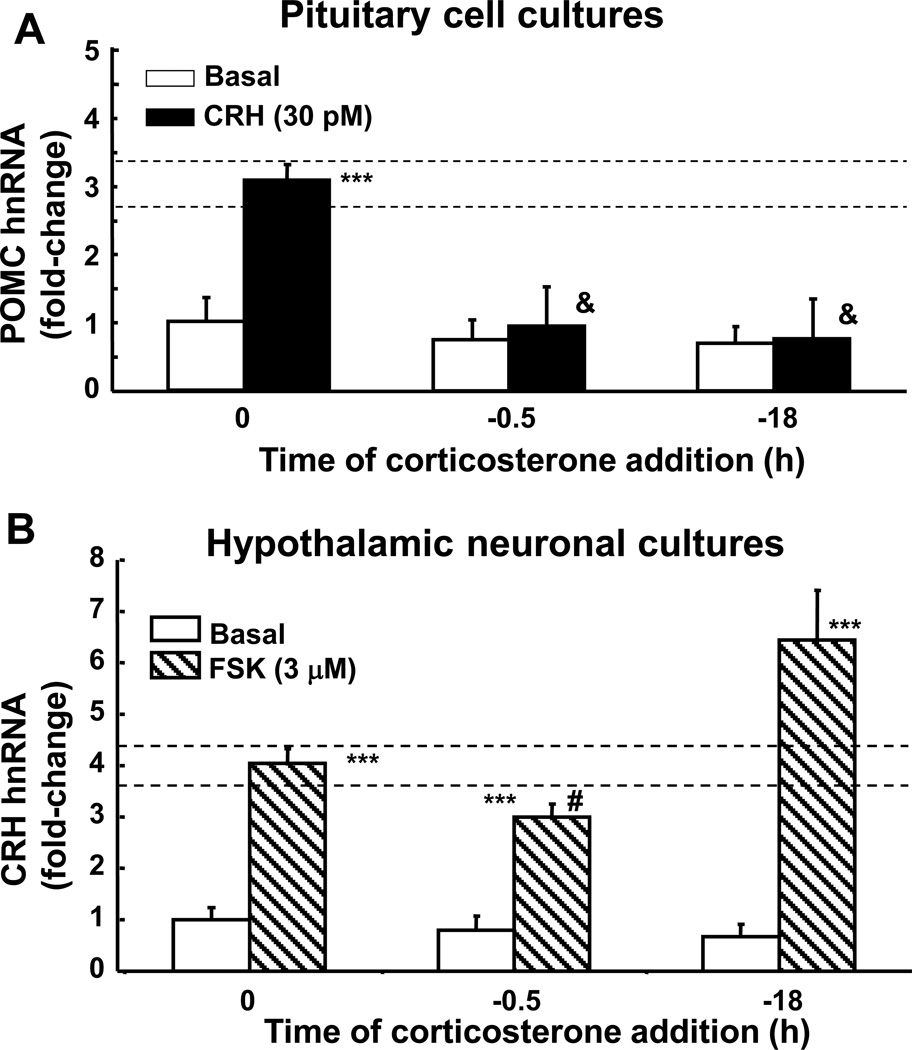
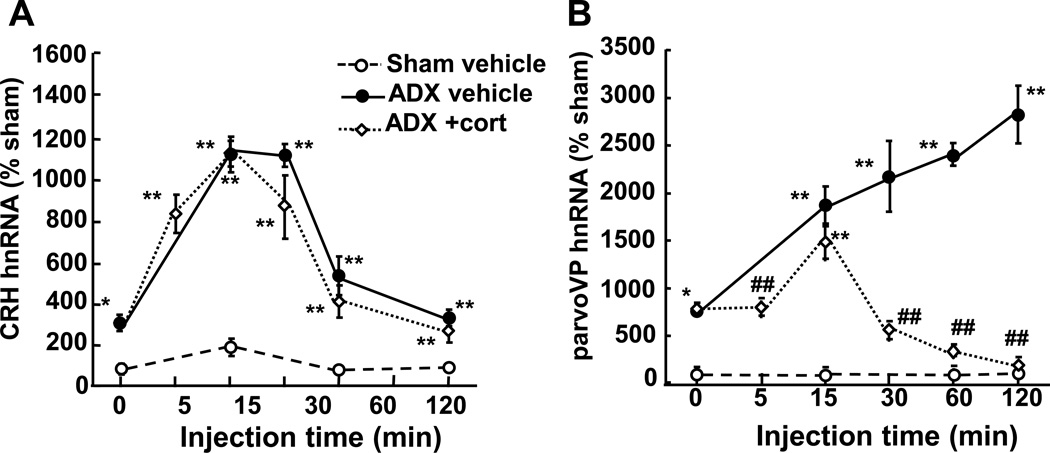
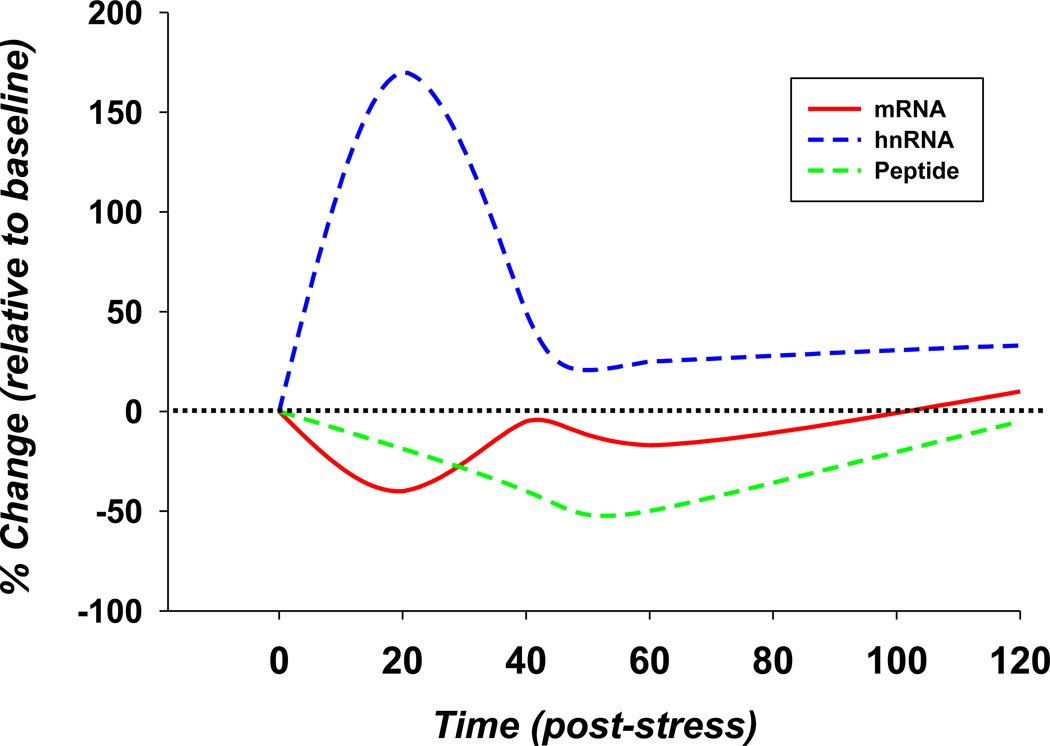
In vitro studies in primary cultures of rat anterior pituitary or hypothalamic neurones have shown marked differences between the effects of GCs on CRH and POMC transcription. In these experiments transcription rates were assessed by measuring levels of primary transcript or hnRNA. As shown in Fig. 1-A, preincubation of primary cultures of rat anterior pituitary cells with 100 nM corticosterone for 30 min before addition of 1nM CRH completely prevented the stimulatory effect of CRH on POMC hnRNA. Full inhibition of CRH-stimulated POMC transcription persisted 18h after addition of corticosterone. In contrast, exposure of primary cultures of fetal rat hypothalamic neurones to corticosterone had only minor effects of cyclic AMP-stimulated CRH hnRNA production. In these experiments, 7-day neuronal cultures maintained for 48h in steroid-free culture medium were exposed to 100 nM corticosterone before incubation with the adenylate cyclase stimulator, forskolin for an additional 45 min. As seen in Fig. 1-B, corticosterone tended to inhibit forskolin-stimulated CRH hnRNA in cells preincubated with corticosterone for 30 min, an inhibition which was statistically significant only after log transformation of the data (15). Similarly, administration of corticosterone doses increasing plasma concentration to 100-fold stress levels in adrenalectomised rats did not affect the magnitude or duration of the increase in CRH hnRNA in the PVN in response to a mild stress (15, 16) (Fig. 2-A). In the same rats, the injection of corticosterone markedly attenuated stressinduced vasopressin hnRNA levels in parvocellular neurones (Fig. 2-B) (Ma and Aguilera 1999). Similarly, Kovacs and Sawchenko (17) showed the injection of corticosterone 10 min before ether stress in rats failed to inhibit stress induced increases in CRH hnRNA.
Figure 1.
Effect of corticosterone on CRH-stimulated POMC hnRNA in primary cultures of rat anterior pituitary cells (A), and forskolin-stimulated CRH hnRNA in primary cultures of hypothalamic neurones (B). Three-day cultured trypsin-dispersed anterior pituitary cells, maintained in stripped serum for 36h were exposed to 100 nM corticosterone for 18 or 30min before addition of CRH 30 pM for an additional 30 min. Bars represent the mean and SE of POMC hnRNA levels determined by qRT-PCR in 3 cell preparations. In panel B, 10-day cultured fetal rat hypothalamic neuronal cultures were exposed to 100 nM corticosterone for 18h or 30 min before addition of forskolin (Fsk) for an additional 45 min before RNA preparation. Data points are the mean and SE of CRH hnRNA levels, normalized to GAPDH mRNA in 4 experiments. ***, p<0.001 compared with basal; # p< 0.05 lower than Fsk at 0min after log transformation of the data. &, p<0.001 vs CRH at time 0. The horizontal dashed lined represent the SE of maximal stimulated values in the absence of corticosterone.
Figure 2.
Time course of the changes in CRH hnRNA (A) and vasopressin (VP) hnRNA (B) after injection of corticosterone (2.8 mg/100 g BW, ip) or vehicle in 48-h adrenalectomised (ADX) or sham operated rats. Note that vehicle injection caused marked increases in CRH hnRNA in ADX but not in intact rats. Data points are the mean and SE of the optical density values obtained from in situ hybridization film autoradiograms in six rats per experimental group. *p<0.01 vs sham; **, p<0.001 vs time 0 and sham; ##, p< 0.01 lower than ADX vehicle. (From Ma and Aguilera, 1999).
Although the above studies suggest little effect of GCs on CRH transcription, GR are present in the CRH neuron (18, 19) and there is clear evidence that GCs negatively regulate CRH mRNA levels. For example, removal of endogenous GCs by adrenalectomy markedly increases CRH mRNA and peptide content in the PVN (20, 21), and potentiates the stimulatory effect of stress on CRH transcription (22). Glucocorticoid administration, systemic or directly in the PVN region has the converse effect (6, 23). Also, it is clear from in situ hybridization studies that chronic glucocorticoid administration inhibits basal and stress-stimulated CRH transcription (23). However, from the above evidence it is not clear whether GCs repress CRH transcription directly through interaction of the GR with the CRH promoter.
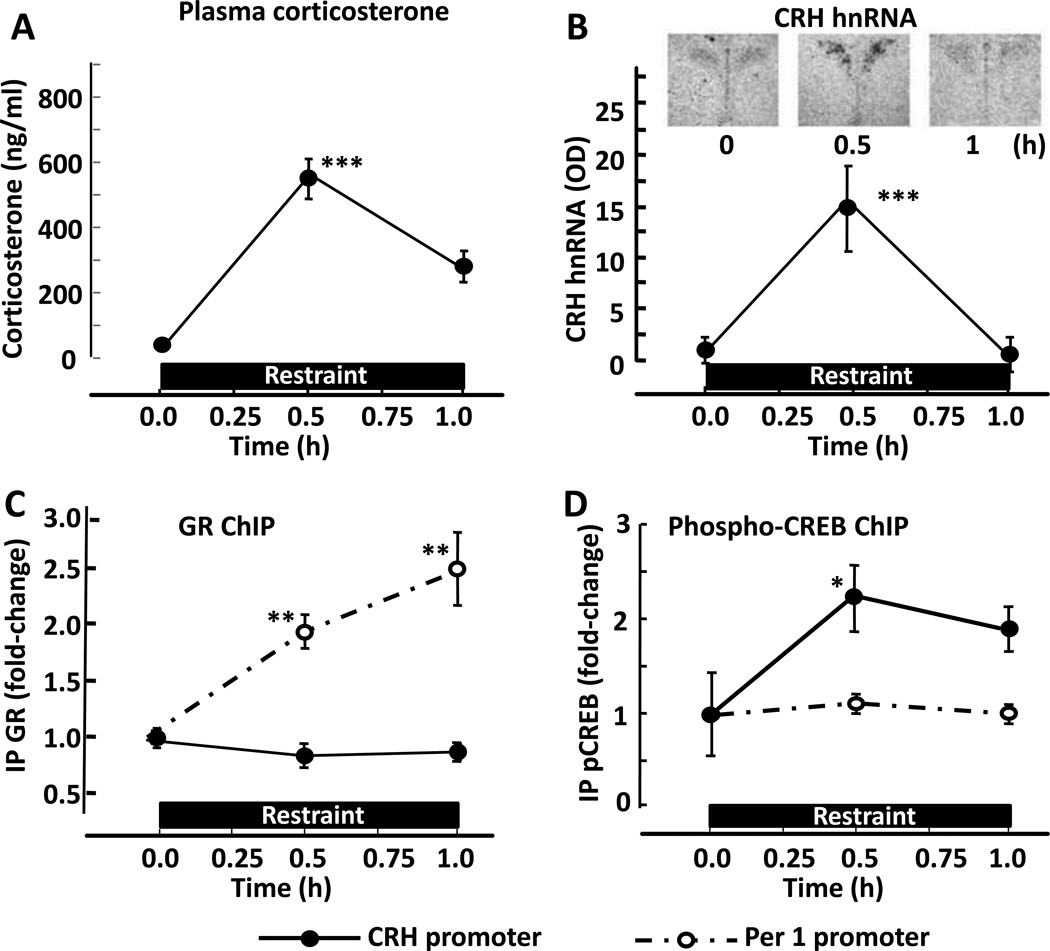
There is no classical glucocorticoid response element (GRE) in the CRH promoter, but using in vitro systems (e.g., reporter gene assays, gel shift assays), Malkoski and Dorin (24, 25) have characterized a conserved sequence located closely upstream of the essential cyclic AMP response element (CRE) of the CRH promoter, capable to mediate repression of promoter activity by GCs. However, others have not confirmed the functional activity of this site and show that the repressor effect of GCs requires the CRH promoter CRE (26), suggesting that the effect is mediated by protein–protein interactions. Since interpretation of data based on reporter gene assays or gel shift analyses can be questionable due to the lack of context of construct DNA with the chromatin landscape, studies were conducted to examine the interaction of the GR with the proximal CRH promoter during physiological changes in circulating GCs (15). Restraint stress in rats causes marked increases in circulating corticosterone (Fig. 3-A), associated with transient increases in CRH hnRNA in the PVN (22, 27, 28, 29) (Fig. 3-B), suggesting that the declining phase of transcription is due to repression by the elevated GCs. However, chromatin immunoprecipitation assays failed to identify changes in CRH promoter in chromatin pulled down by a GR antibody cocktail, while detecting marked increases in period 1 (Per 1) promoter, a recognized glucocorticoid dependent gene (15), Fig. 3-C). In contrast, immunoprecipitation with phospho-CREB antibody yielded the expected increases in CRH promoter at 30 min during restraint stress (15) (Fig. 3-D). Similar lack of change in CRH promoter immunoprecipitation was observed using qRT-PCR primers targeting regions up to 2Kb upstream from the transcription start site (15). In the same study, immunoprecipitation of hypothalamic chromatin from intact or adrenalectomised rats subjected to corticosterone injections showed minor association of the proximal CRH promoter with GR compared to phospho-CREB (pCREB) (15). The above data suggests that repression of CRH transcription by GCs is not mediated by interaction of GR with the proximal CHR promoter. However, a possible interaction of the GR with another protein in the transcriptional complex cannot be ruled out, since a short-arm crosslinker, formaldehyde, was used to stabilize GR-DNA interactions prior to sonication and immunoprecipitation. Also, interaction of GR with sites upstream of the 2000 bp scanned, could lead to changes in chromatin configuration and interaction with the CRE region in the proximal promoter, as it has been described for a number of genes (30, 31, 32).
Figure 3.
Effect of restraint stress on plasma corticosterone, CRH hnRNA levels in the PVN (B), and glucocorticoid receptor recruitment by the CRH and Per 1 promoters in intact male rats. Restraint stress caused marked increases in plasma corticosterone (A), associated with transient increases in CRH hnRNA in the PVN (B). CRH hnRNA was measured by in situ hybridization expressed as optical density (OD) of film autoradiographs (representative images are displayed at the top of data points showing the pooled values in 6 rats). Chromatin immunoprecipitation (ChIP) of microdissected hypothalamic PVN region using anti-GR antibody (IP GR) shows no association of GR with the CRH promoter (solid line and circles), but marked immunoprecipitation of Per 1 promoter (dashed lines open circles) by GR antibodies (C). Immunoprecipitation with phopho-CREB antibody (IP pCREB) yielded high CRH but not Per1 promoter (D). Measurements were performed in basal conditions (time 0), 0.5 and 1h during stress. Data points are the mean and SE of the results of 3 experiments (using pooled hypothalamic tissue from 3 rats per experimental group). The dashed lines correspond to the Per1 promoter, and solid lines show different regions of the CRH promoter. The restraint stress period is shown by the horizontal boxes above the x axis. ***, p< 0.001 vs respective basal; **, p<0.01 vs respective basal; *, p< 0.05 vs. respective basal.
CRH transcription depends on activation and binding to the CRH promoter CRE of phospho-CREB and its co-activator, transducer of regulated CREB activity (TORC) (33, 34), and it is also possible that GCs interfere with the activation and binding of these proteins to the CRH promoter. In this regards, glucocorticoid administration to rats or mice abolishes stress-induced increases in phospho-CREB immunoreactivity in the PVN and prevents nuclear translocation of TORC (35). However, this effect could reflect inhibition of afferent pathways to the PVN, rather than direct effects on the CRH neuron, as several reports show that GCs do not inhibit nuclear accumulation of phospho-CREB in vitro (15, 36, 37). The recent study showing that corticosterone has no effect on the activation and nuclear translocation of TORC in the hypothalamic cell line 4B (15), suggests that the inhibition of TORC translocation by GCs reported in mice depends on modulation of afferent inputs to the PVN neuron.
Although, it is generally believed that GCs repress CRH transcription by interacting with the putative negative GRE in the proximal promoter, accumulating evidence suggest that such an interaction does not occur during physiological elevations of the natural glucocorticoid, corticosterone. The minor effects of GCs on CRH transcription compared with the marked transcriptional repressor activity on vasopressin in parvocellular neurones, and pituitary POMC, strongly suggest that the major mechanism by which GCs repress CRH transcription is indirect by modulating neural pathways controlling CRH neuron function.
Novel mechanisms of glucocorticoid feedback signalling
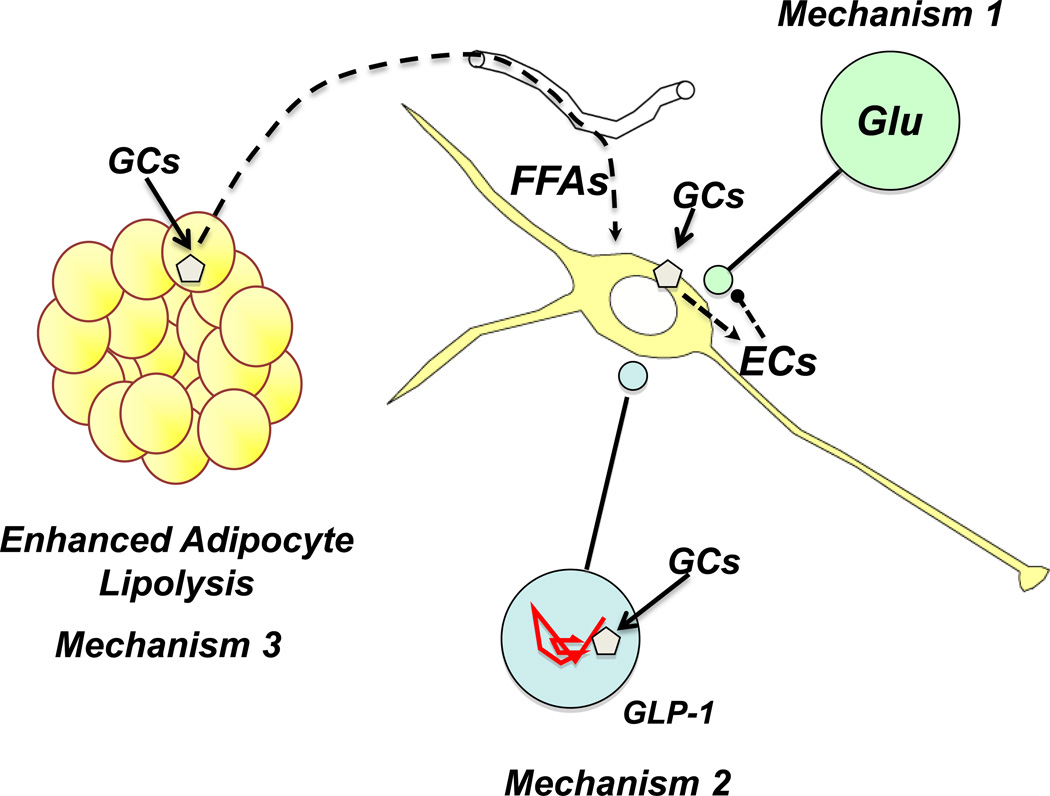
As already mentioned, the HPA axis is reliably activated by psychogenic or systemic stressors, and the largely catabolic actions of high glucocorticoid levels mandate mechanisms to limit overexposure. This is accomplished by negative feedback inhibition of hormone release, which is regulated in large part by the GR (due to its binding capacity). Whereas genomic effects of GCs contribute to long-term inhibition of ACTH release (so-called ‘delayed feedback’), the minute-to-minute control needed to constrain HPA axis function is likely to require non-genomic mechanisms. Consequently, glucocorticoid feedback appears to be modulated by multiple processes that converge to limit activation of the HPA axis by inhibition of PVN neurones driving ACTH release. In the current section, we discuss three recently-delineated feedback processes that highlight the richness of the biology of glucocorticoid signalling, including rapid PVN feedback mediated by retrograde messengers, a brainstem feedback pathway that appears to involve modulation of RNA stability, and peripheral mechanisms that capitalize on glucocorticoid interaction with metabolic effector pathways (summarized in Fig. 4).
Figure 4.
Proposed ‘non-traditional’ glucocorticoid feedback mechanisms. Mechanism 1: Glucocorticoids (GCs) act via non-genomic mechanisms to inhibit PVN CRH neurones, acting via membrane glucocorticoid receptors (pentagon) to mobilize endocannabinoids (ECs), which bind to CB1 receptors and inhibit presaynaptic glutamate release. Mechanism 2: Glucocorticoids act to destabilize preproglucagon mRNA, reducing the magnitude of glucacon-like peptide 1 (GLP-1) excitation of CRH neurones. Mechanism 3: Glucocorticoids act in the periphery (possibly at adipocytes) to generate inhibitory messengers, such as free fatty acids, that can inhibit HPA axis activation secondarily.
Glucocorticoid Fast Feedback
Rapid effects of GCs were observed as early as the 1960’s, primarily in the form of rapid feedback inhibition of glucocorticoid release following stress (38). Rapid glucocorticoid feedback inhibition is non-genomic in nature, and occurs at time delays consistent with possible membrane actions. Work from Tasker’s group has demonstrated that GCs rapidly inhibit PVN by way of a membrane-associated receptor (39, 40). The mechanism of rapid inhibitory action is mediated by postsynaptic G-protein-mediated release of endocannabinoids (39). Anandamide and 2-arachidonoylglycerol are rapidly synthesized following glucocorticoid exposure or acute stress in the PVN, and act as retrograde messengers by binding to presynaptic CB1 receptors (39). A role for endocannabinoids in glucocorticoid inhibition has been demonstrated in vitro and in vivo, as CB1 antagonism or knockout leads to elevated CRH expression in the PVN and increased plasma ACTH and CORT (41, 42, 43, 44). Furthermore, bilateral PVN injections of dexamethasone and a CB1 inverse agonist block the suppression of HPA axis responses to acute stress seen following dexamethasone administration alone (45).
While the receptor mechanism mediating glucocorticoid signals at the membrane has not been completely worked out, most evidence suggests that effects are associated with the ‘classical’ GR molecule. Pre-administration of the GR antagonist mifepristone is not sufficient to completely block inhibitory effects of dexamethasone in vitro (39). However, rapid glucocorticoid feedback is blocked in slices from mice with targeted deletion of the GR in PVN neurones (using the simple-minded-1 (Sim1) promoter to drive cre recombinase in GR flox mice) (46). Moreover, GR can be localized to neuronal postsynaptic membranes, supporting membrane localization of the so-called ‘nuclear’ receptor (47, 48, 49, 50). Finally, PVN administration of dexamethasone-bovine serum albumin conjugate, which should not cross the cell membrane, inhibits stress-induced HPA axis activation in vivo (45).
Intermediate Time-Frame: Rapid Glucocorticoid Inhibition of Ascending Stress Effector Pathways
The medial parvocellular PVN receives synaptic innervation from ascending stressregulatory neurones in the caudal medulla and locus coeruleus (51, 52). Most of the innervation of the CRH-containing subregion of the PVN comes from the nucleus of the solitary tract (NTS) (51, 52). There is a strong body of evidence suggesting that NTS norepinephrine-epinephrine neurones are involved in excitation of PVN neurones controlling HPA axis responses to stress. Stress causes NE release in the PVN (53), and local NE administration is sufficient to cause ACTH release and enhance expression of crh gene transcription (54, 55). Moreover, local alpha adrenergic receptor blockade reduces stress-induced HPA axis activation (54). Recent data suggest that HPA axis activation is also mediated by non-catecholaminergic projections from the NTS, emanating from neurones expressing glucagon-like peptide 1 (GLP-1). These neurones send GLP-1ergic projections to CRH neurones (56, 57). Blockade of GLP-1 receptors reduces ACTH and corticosterone responses to acute physiological or psychogenic stressors, and local infusion of GLP-1 into the PVN causes release of corticosterone (58), underscoring the necessity of GLP-1 in acute stress reactivity. Moreover, central infusion of a GLP-1 receptor antagonist reduces the impact of chronic stress exposure on HPA axis end points (59). Together, the data suggest a prominent role for GLP-1 in stress excitation at the level of the PVN.
Recent data suggests that the GLP-1 system is also a target for glucocorticoid feedback. Experiments were performed to assess regulation of NTS stress-excitatory pathways following exposure to acute stress regimens. Surprisingly, a very rapid depletion of preproglucagon (PPG; the GLP-1 precursor protein) mRNA is observed following acute stress, with mRNA levels falling to less than 50% of basal values within 20–30 min of stress onset (60). This rapid downregulation is mimicked by exogenous corticosterone administration. Moreover, stress-induced down-regulation is blocked in adrenalectomised animals clamped at basal circulating corticosterone levels (60), indicating that PPG down-regulation was mediated by stress-related glucocorticoid release. Importantly, PPG hnRNA was substantially increased by acute stress exposure, suggesting that changes in PPG mRNA were not due to deceased transcription. Reductions in PPG mRNA are relevant at the level of the synapse, as stress causes a rapid decrement in PVN GLP-1 immunoreactivity (60), perhaps as a consequence of synaptic release associated with the stressor. Given the rapid timing of the stress-induced PPG decrement, we hypothesize that the effects of stress are mediated by rapid, presumably non-genomic effects of GCs on PPG mRNA stability. GCs are known to function in this capacity in cell-based systems (61, 62), and in combination with the data above suggest that glucocorticoid-mediated modulation of mRNA stability may participate in feedback regulation of the HPA axis function at the level of the NTS.
The observed time-frame of gene turn off and loss of GLP-1 immunoreactivity implies a translational ‘pause’ that will not affect immediate release of neuropeptide, but would reduce PVN excitability later in time. Consequently, the excitatory impact of GLP-1 on HPA axis activation will be reduced as a consequence of down-regulation during the period of depletion of peptide in terminals, approximately 30–60 minutes after stress (Fig. 5), predicting that the HPA axis may be less ‘excitable’ during the immediate post-stress period. This is indeed the case, as numerous studies report that imposition of a second stress 5–60 minutes after an initial stressor causes marked reduction in HPA axis responsiveness. The stress-refractory period can also be observed if the initial stressor is replaced with a bolus injection of GCs, suggesting that the inhibition is glucocorticoid-driven (referred to as ‘intermediate feedback’ by Dallman and colleagues) (63). Our data suggest that at least part of the intermediate feedback effect may be mediated by temporary destabilization of PPG mRNA by stress-induced glucocorticoid release.
Figure 5.
Schematic comparing the time course of PPG mRNA degradation (PPG mRNA) with PPG transcription (PPG mRNA) and loss of PVN GLP-1 immunoreactive terminals (peptide). Note that transcriptional effects do not correspond with loss of mRNA, suggesting the mRNA and peptide loss are likely linked to mRNA degradation or turnover.
Peripheral Glucocorticoid Signalling and Stress Regulation
In addition to actions of GCs in brain, it is important to consider that glucocorticoid receptors are abundantly expressed in the periphery, and may exert indirect effects on the HPA axis via ascending vagal afferents or hormonal messengers. Work from Dallman’s group has demonstrated that the pronounced HPA axis activation seen following adrenalectomy (e.g., increased PVN CRH synthesis) could be reversed by allowing the animals to drink sucrose, suggesting that GCs may inhibit the HPA axis by modulating peripheral metabolic signals (64). These data are supported by studies showing that central sucrose administration is not able to reduce adrenalectomy-induced activation of the HPA axis, implying a peripheral mechanism of action (65). Conversely, increasing energy stores (in the form of fat depots) by voluntary intake of a high-fat diet reduces HPA axis responses to stress, accompanied by elevated insulin secretion (66). The data suggest that glucocorticoids act in the periphery to promote glucose and insulin release, which is relayed to the hypothalamus to inhibit PVN activation.
Recent studies suggest involvement of adipose-derived signals. Specific deletion of the GR in adipocytes enhances glucocorticoid responses to stress and attenuates glucocorticoid feedback inhibition of the HPA axis (de Kloet et al, unpublished observations). GCs increase adipocyte lipolysis by activation of hormone sensitive lipase, which increases circulating free fatty acids (67). Depletion of free fatty acids, in particular palmitic acid (68, 69), causes elevated plasma ACTH and corticosterone, suggesting a role in constraining the HPA axis. One of the major sensors of free fatty acids and lipid messengers, peroxisome-proliferator activated receptor gamma (PPARγ), is localized to PVN neurones (70). Treatment with a PPARγ agonist rosiglitazone inhibits stress-induced PVN Fos induction and corticosterone release (71), whereas direct infusion of the PPARγ antagonist GW9662 into the PVN increases ACTH release. These data suggest that FFAs may provide a blood-borne inhibitory signal at the level of the PVN, and is consistent with glucocorticoid-mediated increases in lipolysis at the adipocyte.
Glucocorticoid Feedback: A Distributed Process
The data presented above highlight the richness of the glucocorticoid feedback process, identifying a number of check-points whereby secretion can be limited. These studies complement a rich literature citing trans-synaptic feedback inhibition from neural sources, such as the prefrontal cortex and hippocampus (72); genomic and non-genomic actions at the level of the pituitary (73); and rich interactions with the autonomic nervous system that can alter secretory profile at the level of the adrenal (74).
Glucocorticoids, stress and neuroplasticity
GCs in conjunction with a myriad of chemical mediators released during stress response modify several aspects of brain function, including learning and memory formation (75). Major brain areas targeted by GCs and other stress hormones include the hippocampal formation (mediating declarative memory), amygdala (mediating fear response) and prefrontal cortex (mediating working memory) (76). Morphological and biochemical studies have found that normal circadian or stress-induced variations in circulating GCs, promote neuroplasticity in these structures, especially in the hippocampus, with modifications of neuron morphology and changes in neuronal excitability and synaptic efficacy (76).
In the hippocampus, GCs regulate neuronal turnover through effects on both cell death as well as proliferation of neural progenitors. Adrenalectomy induces apoptosis of mature granule cells of the hippocampal dentate gyrus which prevented by corticosterone or MR agonist administration (77, 78,79, 80, 81). The trophic influence of adrenal steroids on granular neuron survival appears to be related to the regulation of intrinsic apoptotic signalling pathways (82). Evidence indicates that adrenalectomy increases mRNA expression of the proapoptotic gene bax (82) and promotes the processing of caspase-9 (83). Also, adrenalectomy induces an increase in the rate of proliferation of progenitor cells in the subgranular zone of the dentate gyrus, an effect which is prevented by corticosterone administration (84). On the other hand, the increase in glucocorticoid secretion induced by acute stress can act as positive or negative modulators of learning, memory and retrieval (reviewed in 85). Several studies have suggested that acute stress is associated with increased excitatory glutamatergic neurotransmission in areas of the forebrain (86, 87, 88). Some studies have demonstrated that adrenalectomy attenuates the stress-induced release of glutamate in the hippocampus and prefrontal cortex, suggesting a direct relationship between GCs and glutamate release (89). Moreover, intrahippocampal perfusion of corticosterone by retrodialysis produces a fast and reversible increase in the release of glutamate in vivo (90). In addition, electrophysiological studies have shown that application of corticosterone to hippocampal slices increases the frequency of excitatory postsynaptic potentials in the CA1 area of the hippocampus, suggesting a fast action of corticosteroids on glutamate release (91). This fast action of corticosterone is likely to be non-genomic and appears to involve the mineralocorticoid receptor (92).
Recently it has been shown that circadian glucocorticoid peaks induce the formation and development of dendritic spines (excitatory post-synaptic structures) in mouse cortex after motor skill learning and the circadian glucocorticoid troughs are required for stabilization of new spines (93). Furthermore, the improved associative learning promoted by acute stress is accompanied by a rise in the spine density of hippocampal neurones (94). In line with these observations, brief exposure of hippocampal slices to dexamethasone, a GR agonist (95) or corticosterone (100–1000 nM) (96) promotes an increase in spine density in pyramidal neurones of the CA1 hippocampal area. Furthermore, the co-administration of RU486, an antagonist of GR, abolished the effect of corticosterone (96). It has been proposed that spinogenesis is modulated by synaptic GRs and kinases including PKA, PKC (96) and ERK1/2 (97). Although GRs have been localized to neuronal cell bodies and dendrites, a recent study has shown that GRs are localized to dendritic spine heads and to spine necks of CA1 pyramidal cells (97). Additionally, a recent study in KO mice for Fragile X Mental Retardation Protein (FMRP) demonstrated a reduction in GR levels in CA1 dendrites (98). FMRP acts as a protein that transports a subset of neuronal mRNAs from the nucleus into dendrites and spines (98, 99). Furthermore, the bulk of FMRP is associated with polyribosomes and represses the translation of various mRNAs (98, 99). In accordance with these findings, it has been shown that spine GR levels increase rapidly by metabotropic glutamate receptor (mGluR) activation, an effect not observed in KO mice for FMRP (97). Thus, it has been proposed that synaptic levels of GR in CA1 spines are regulated by local GR mRNA translation involving mGluR activity and a FMRP-dependent mechanism (97).
It appears that GR binding also affects glutamatergic receptor levels. It was shown that GR activation increases surface expression of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid glutamate receptor subunit 2 (AMPAR GluA2) in primary hippocampal cultures (100). This GC-induced increase in GluA2 subunit of AMPA receptors at the hippocampal synaptic sites is involved in the facilitation of learning during acute stress (101). The variation in GluA2 levels are probably related to receptor trafficking mechanisms rather than changes in GluA2 mRNAs levels and/or translation (101).
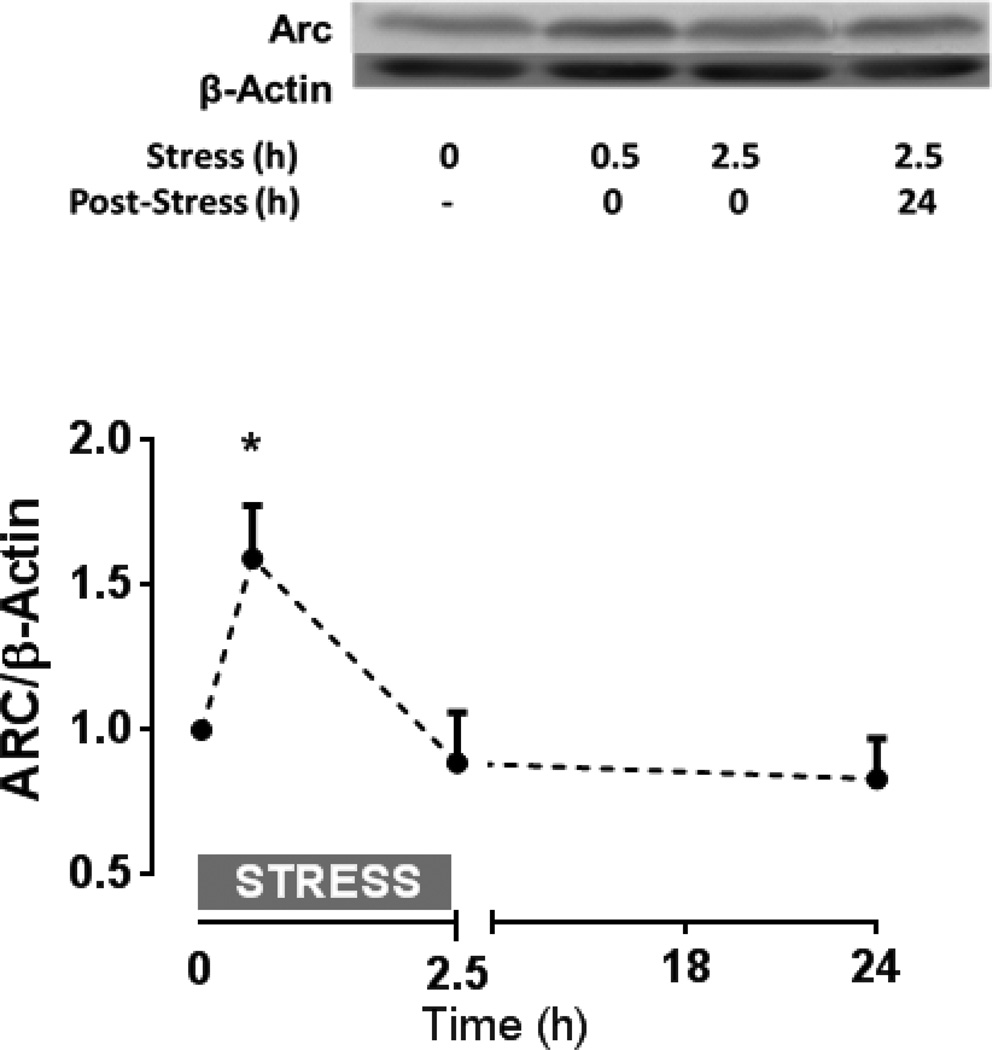
The mRNA encoding the immediate early gene, activity regulated cytoskeletal-associated protein (Arc), is increased by activation of N-methyl-D-aspartate receptor. Arc mRNA traffics to dendrites and its translation is controlled by FMRP and the activation of mGluR (reviewed in 102). Reduction of Arc protein levels in the hippocampus blocks both late-phase long-term potentiation and the consolidation of some forms of memory (103). Furthermore, it has been described that Arc reduces surface expression of GluA2/A3 receptors promoting a decrease in AMPA receptor-mediated synaptic currents (104). These findings suggest that Arc protein stabilizes the internal pool of AMPA receptors which would presumably increase or decrease the levels of these receptors in synaptic sites depending on the initial stimulus (105). Since activation of GRs produce variation in the synaptic levels of GluA2 (101), the effect of acute restraint stress on Arc protein levels was evaluated. Figure 6 shows that 0.5 h of restraint stress promotes an increase of Arc protein levels in the hippocampus, suggesting a fast Arc mRNA translation. Thus, it is plausible that stress-induced corticosterone secretion mediates mGluR activation, increasing Arc mRNA translation, which in turn may regulate surface expression of AMPA receptors. Overall, these findings suggest that stress and GCs may modulate excitatory neurotransmission by increasing glutamate release, modifying spine density, and AMPA receptor trafficking.
Figure 6.
Effect of acute restraint stress on the Arc (activity-regulated cytoskeleton-associated protein) protein levels in the hippocampus of rats. Acute restraint stress induces an increase of Arc protein levels in rat hippocampus. Sprague Dawley male rats were restrained during 0.5 h or 2.5 h and immediately sacrificed or restrained during 2.5 h and sacrificed 24 h after the restraint. A) Representative immunoblots of homogenates from the hippocampus of stressed rats using anti-Arc and β-actin antibodies. B) Graph shows the relative ratio of Arc levels relative to β-actin. Data represent mean ± SD of n=4 per experimental condition. *p<0.05 when comparing to non-stressed animal (i.e. time= 0).
Glucocorticoids in the control of food intake
Food intake is a basic behaviour regulated by multiple factors, including the adiposity signals leptin and insulin, and satiety signals, such as mechanical and chemical stimulation of stomach and small intestine, as well as hormones released during a meal, as the cholecystokinin (CCK) (106). The adiposity factors are involved in the long-term control of energy balance and act primarily in hypothalamic neurones expressing orexigenic or anorexigenic neuropeptides, which are key mediators in the control of energy homeostasis (106). Neuropeptide Y (NPY) and agouti related protein (AgRP) in the arcuate nucleus of the hypothalamus, and orexins and melanin-concentrating hormone (MCH) in the lateral hypothalamic area (LHA) comprise important hypothalamic orexigenic neuropeptides (107, 108, 109). On the other hand, POMC and cocaine and amphetamine-regulated transcript (CART) in the arcuate nucleus of the hypothalamus, and CRH and oxytocin (OT) in the PVN are the main hypothalamic mediators involved in the inhibition of food intake (106, 109). The satiety signals, in turn, are implicated in the short-term control of food intake and have their actions mediated by brainstem areas, mainly the nucleus of the solitary tract (NTS), controlling the size of a meal (110).
In addition to their important role regulating metabolic activity in the periphery, there are profound interactions between GCs and appetite control (111). Feeding is a major synchronizer of HPA axis rhythmicity (112), and the amount of food ingested is associated with GC secretion (113). Reciprocally, increased food intake and body weight gain in humans are associated with increases in circulating glucocorticoids associated with therapeutic administration of or Cushing disease (114). Glucocorticoid excess in these cases is associated with increased glucose production, decreased glucose transport and utilization, decreased protein synthesis, increased protein degradation in muscle and body weight gain (115, 116). In contrast, chronic glucocorticoid deficiency as occurs in Addison’s disease or primary adrenal insufficiency is characterized malaise, fatigue, weight loss, joint and back pain, darkening of the skin, hyponatremia, hypoglycemia, hyperkalemia, with anorexia being a remarkable symptom (117).
Studies in rodents have shown that GC effects on food intake depend on the dose of corticosterone replacement (118). Low doses of corticosterone in adrenalectomised rats, with corticosterone plasma levels ranging from 1 to 2 µg/dL, were demonstrated to activate MR and to have a stimulatory effect on fat intake, body weight gain and fat depots, which occur at the late phase of the feeding period (119, 120, 121). On the other hand, GRs are activated by higher doses of circulating corticosterone (2–10 µg/dL) just before or in the first hours of the active feeding period, which induce carbohydrate ingestion and metabolism (119, 120,121, 122, 123, 124). In addition, extremely high corticosterone plasma concentrations (>10 µg/dL), as in consequence of stress or food restriction, stimulate fat and protein catabolism, mainly muscle protein catabolism, and consequently body weight loss, to increase the availability of gluconeogenesis substrates and to enhance glucose plasma concentration as energy source (121, 125). Central actions of GCs on feeding behaviour has been demonstrated by different studies, which showed that dexamethasone injection into the lateral ventricle not only stimulated food intake but also enhanced body weight gain in rats, accompanied by hyperleptinemia and hyperinsulinemia, (126, 127).
The central effects of GCs on the regulation of food intake seem to be mediated by the interaction of GCs with hypothalamic neuropeptides involved in the control of energy homeostasis, as demonstrated by the presence of glucocorticoid receptors in neurones expressing neuropeptides involved in the control of energy homeostasis (128, 129). This interaction between GCs and hypothalamic neuropeptides involved in the control of food intake has been evaluated, showing that central glucocorticoid treatment increases hypothalamic NPY levels and decreases those of CRH (127). In addition, circulating GCs were shown to be required for feeding-induced decrease in the expression of orexigenic neuropeptides in the arcuate nucleus of the hypothalamus and increase in the expression of the anorexigenic neuropeptide POMC in this nucleus, being these effects consequence of a direct modulation of GCs in these neurones in the arcuate nucleus of the hypothalamus, and indirectly, in consequence of changes in leptin and insulin secretion induced by food intake (130).
As observed in humans with Addison disease, removal of endogenous GCs induced by bilateral adrenalectomy in rodents has been shown to reduce food intake and body weight gain, and these effects are reversed by glucocorticoid replacement in rats (131, 132, 133, 134). Furthermore, adrenalectomy diminishes hyperphagia and obesity in different experimental models, whereas glucocorticoid replacement reverses these effects (131, 135, 136, 137). The hypophagic effect induced by the removal of endogenous GCs by adrenalectomy is associated with increased expression of the anorexigenic neuropeptides CRH and OT in the PVN, as well as reduction in the expression of the orexigenic neuropeptides NPY and AgRP in the arcuate nucleus of the hypothalamus, indicating that changes in the expression of these hypothalamic neuropeptides could account to the reduction on appetite following adrenalectomy (130, 133, 134). Concerning the anorexigenic neuropeptides of the arcuate nucleus of the hypothalamus, adrenalectomy was shown to reduce POMC and CART, suggesting that the anorexigenic effects in response to the removal of adrenal glands are dissociated from elevations of these neuropeptides in the arcuate nucleus of the hypothalamus (130, 138).
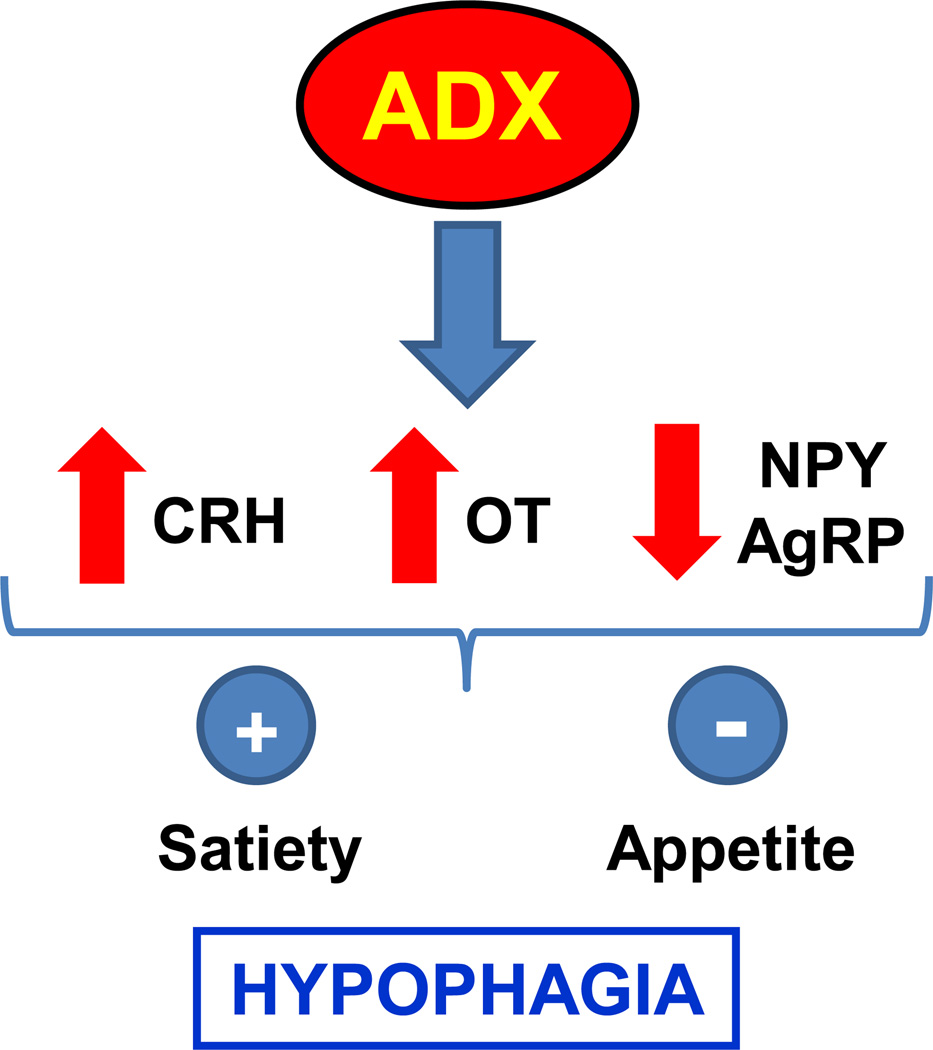
It is well established that the action of GCs on food intake involve their stimulatory drive to eat, and thus adrenalectomy-induced hypophagia involves, at least in part, a reduction on the drive to eat. However, recent studies have highlighted the role of GCs on the short-term control of food intake, indicating that the hypophagic effect in response to adrenal gland removal is associated with increased activation of satiety-related responses through brainstem and hypothalamic pathways (132, 133). Accordingly, NTS neurones as well as CRH and OT neurones in the PVN were also more activated in response to meal after adrenalectomy (132, 133). In addition, CRH and OT neurones in the PVN project to the NTS, and OT axonal projections from the PVN to the NTS are enhanced after adrenalectomy (139). Interestingly, CRH and OT were shown to be involved in the enhanced satiety-related responses after adrenalectomy since both the hypophagia and the increased activation of NTS neurones induced by feeding following adrenalectomy were reversed by CRH type 2 receptor and OT receptor antagonists (132, 134, 139). Accordingly, CRH and OT rise as pivotal mediators of the enhanced satiety-related responses in the NTS, contributing to the hypophagia in the primary adrenal insufficiency. Thus, GCs have well-established roles in the regulation of feeding behaviour and energy homeostasis, stimulating the drive to eat as well as reducing the satiety-related responses (Fig. 7).
Figure 7.
Schematic illustration showing the hypophagic effect in response to adrenalectomy (ADX) through the stimulation of appetite and the inhibition of satiety pathways, mediated by the increase on the expression of the anorexigenic neuropeptides corticotrophin releasing hormone (CRH) and oxytocin (OT) and the decrease of the orexigenic neuropeptides neuropeptide Y (NPY) and agouti related protein (AgRP).
Interactions between the HPA axis and the immune system
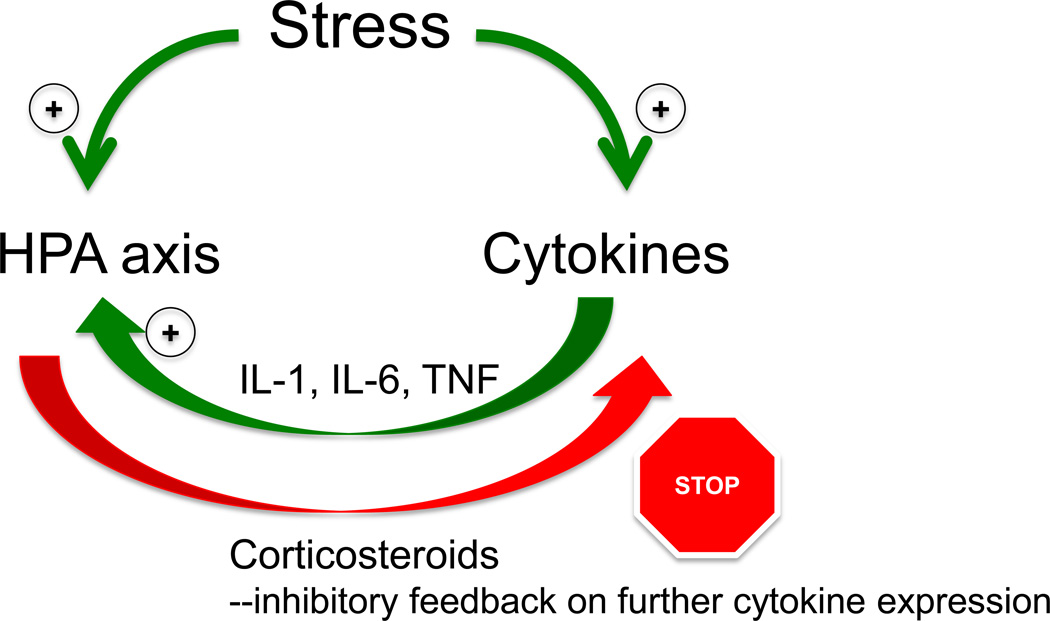
Glucocorticoids have well recognized effects as immune suppressors, and their release during immune challenge acts to dampen cytokine production and inflammatory responses. However, the HPA axis and immune system show complex bidirectional interactions not only under immune challenges but also during exposure to non-immune stressors. A growing body of literature suggests that stress significantly impacts many facets of neuroimmune function. For instance, exposure to a variety of acute stress challenges increased the expression of Interleukin-1 beta (IL-1β) in the hypothalamus (140, 141, 142), whereas increased prostaglandin activity has been observed throughout the cortex in response to acute stress (143). Converging lines of evidence suggest that these changes in neuroimmune signalling factors (cytokines, chemokines, prostaglandins, etc) are associated with other tell-tale signs of neuroinflammation such as priming (144), proliferation (145) and activation (146, 147) of microglia. Consistently, injection of the putative microglial inhibitor minocycline blocked cytokine expression evoked by stress (146, 148). Thus, a rich constellation of neuroimmune changes occur in response to stress, which raises several critical issues, including: (i) how do hormones classically associated with stress (nor/epinephrine and GCs, in particular) influence cytokine expression and other aspects of neuroimmune function?; (ii) to what extent do neuroimmune signalling pathways serve as either moderators or mediators of HPA axis output across natural diurnal rhythms and/or in response to later stress challenges? (iii) under what circumstances do neuroimmune consequences of stress either compromise or protect against the development of pathological states of the CNS?
Although the precise mechanisms controlling cytokine expression during times of stress have not been fully delineated, several key mechanisms have emerged. Prior work has shown that administration of β-adrenergic blockers inhibit or severely blunt the expression of Interleukin-1 evoked by stress, and administration of the β-adrenergic agonist isoproterenol both recapitulates and potentiates the expression of IL-1 induced by stress (146, 148, 149). In contrast to the dependence of central cytokine responses on β-adrenergic receptor activation, plasma cytokine responses to stress appear to be selectively mediated by α1-adrenergic receptors (150). These findings are supported by lesion studies, where complete lesions of central adrenergic systems incurred by injection of the neurotoxin DSP4 completely blocked the IL-1 response produced by stress in several brain structures (149), whereas more targeted lesions of the ventral noradrenergic bundle (VNAB) only partially attenuated the IL-1 response to stress in the PVN (150). These data fit well with strain differences in central cytokine responses to stress showing that hyper-adrenergic Fisher 344 rats show much greater increases in central cytokine expression relative to their less reactive Sprague Dawley comparators (151). In fact, the potentiation of cytokine responses to stress in hyperadrenergic, Fisher 344 rats is particularly noteworthy given that this strain also evinces a potentiated HPA axis response relative to other strains (152). Thus, norepinephrine appears to be a key driver of stress-induced cytokine expression (see Fig. 8). However, it should be noted that other transmitter systems such as glutamate (153) and other intermediaries such as Danger Associated Molecular Patterns (DAMPs; 154) may participate in various features of the neuroimmune response to stressor exposure as well.
Figure 8.
Schematic illustration describing the interaction between corticosteroids and cytokine expression. Stress exposure directly induces the release of HPA axis hormones and increases expression of cytokines. Although there is emerging evidence to suggest that corticosteroids may enhance cytokine expression under certain circumstances, a primary effect of corticosteroids is suppression of cytokine expression. Cytokines, on the other hand, directly stimulates activation of the HPA axis via actions both intrinsic and extrinsic to the axis, and appear to augment HPA axis sensitivity to later stress challenges.
GCs, on the other hand, powerfully constrain the expression of cytokines during times of stress. Evidence to support this comes from the findings that removal of endogenous GCs via adrenalectomy (155), administration of glucocorticoid receptor antagonists (156) or the glucocorticoid synthesis inhibitor metyrapone (146) massively potentiates the expression of cytokines evoked by stress. These findings fit well with a range of molecular interactions through which corticosteroid receptors squelch inflammatory signalling pathways (157, 158, 159). Very recently, however, a few studies have begun to reveal effects that indicate priminglike actions of GCs as well (160, 161). However, it should be noted that these non-canonical actions may be (i) unique to specific tissue types within the body; (ii) occur at only low-dose or low-physiological concentrations of circulating GCs; or (iii) reflect a short-term, early response to glucocorticoid signalling. These intriguing findings suggest that the relationship between corticosteroid signalling and neuroimmune consequences of stress will require detailed consideration of numerous key parameters (tissue, dose, timing, etc). Nevertheless, it is clear that in the absence of effective glucocorticoid signalling, neuroimmune consequences of stress are quite severe.
Moving beyond mechanisms, neuroimmune consequences of stress (particularly increased cytokine expression) may play an important role as moderators of HPA axis activation. For instance, IL-1 receptor 1 knockout mice show blunted HPA axis response to mild but not intense stress challenges (162). Previously stressed rats showed a faster HPA axis response to later LPS injection, an effect that was reversed by IL-1 receptor antagonist (163). Furthermore, it has been noted that dissociations between ACTH and CORT are often observed under inflammatory conditions (164, 165), including after immune activation by LPS (166, 167), as well as in inflammatory disease states (168, 169). Thus, cytokines and other immune-related signalling factors have the capacity to induce (through actions extrinsic to the axis) or augment (through intrinsic actions) HPA axis responses to later stress challenges.
The impact of neuroimmune consequences of stress is not restricted to the HPA axis. For instance, neuroimmune consequences of stress seem to control certain behavioural consequences of stress exposure (170), impairments in cognitive function precipitated by stress (171, 172), and may be involved in certain depressive-like consequences of stress such as learned helplessness (173). Indeed, stress-related cytokines have been argued as a potentially critical mechanistic bridge between stressful experiences and the development of stress-related pathology (174, 175, 176). Thus, with such far-reaching implications of stress-related changes in neuroimmune function, the interaction between classic stress-responsive systems (HPA axis, SNS) and neuroimmune function provides an important starting block for better understanding long term, cumulative impacts of stress on animals and humans (175).
Concluding remarks
Important progress has been made during the last few years in understanding the physiological actions of glucocorticoids and the mechanisms of glucocorticoid feedback regulation of the HPA axis. Glucocorticoids have pleotropic actions regulating metabolism, brain function and modulating the function of most cells in the body. Growing evidence indicates that normal glucocorticoid-dependent regulation depends upon the prevailing levels of the steroid, secretion pattern, as well as glucocorticoids interaction with neurotransmitters and neuropeptides. This is partly illustrated by the concentration-dependent effects of glucocorticoids in hippocampal neuroplasticity and immune function discussed above. It is also clear that not all the effects of glucocorticoids are direct, with a number of actions depending of changes in other regulatory factors. For example, the profound modulatory effects of glucocorticoids on food intake largely depend on the opposing effects of glucocorticoids on orexigenic and anorexigenic neuropeptides.
Negative glucocorticoid feedback is essential for the fine control of HPA axis activity to avoid deleterious consequences of excessive CRH and glucocorticoid production. New evidence indicates that glucocorticoid feedback inhibits HPA axis activity at a number of anatomical and molecular targets, rapidly shutting-off hypothalamic and pituitary responses at the cell membrane level, controlling the intensity and duration of stress responses at limbic sites, and inducing long-term inhibition by modulating transcription and mRNA stability at the central and pituitary levels.
In spite of the recent advances in the field of HPA axis regulation and glucocorticoid actions, a number of challenges still remain. This includes identification of peripheral metabolic signals and central sensors impacting on the sensitivity of glucocorticoid feedback and HPA axis activity, and the exact identity of the membrane glucocorticoid receptor and its signalling mechanisms. Understanding the functions of glucocorticoids and mechanisms of feedback regulation of HPA axis activity will contribute to developing new diagnostic and therapeutic tools for disorders related to stress and alterations of glucocorticoid secretion.
Supplementary Material
Acknowledgements
This study was supported by FAPESP and CNPq to E.T.U., Intramural Research Program, National Institute of Child Health and Human Development to G.A., NIH grants MH049698 and MH069860 to J.P.H., FONDECYT 1120528 to J.L.F., National Science Foundation (NSF) grant 0822129 to T.D. and CNPq (Proc. No.302592/2009-1) to M.B.C.S.
References
- 1.Johnson EO, Kamilaris TC, Chrousos GP, Gold PW. Mechanisms of stress: a dynamic overview of hormonal and behavioral homeostasis. Neurosci Biobehav Rev. 1992;16(2):115–130. doi: 10.1016/s0149-7634(05)80175-7. [DOI] [PubMed] [Google Scholar]
- 2.Smith SM, Vale WW. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin Neurosci. 2006;8(4):383–395. doi: 10.31887/DCNS.2006.8.4/ssmith. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lightman SL, Wiles CC, Atkinson HC, Henley DE, Russell GM, Leendertz JA, McKenna MA, Spiga F, Wood SA, Conway-Campbell BL. The significance of glucocorticoid pulsatility. Eur J Pharmacol. 2008;583(2–3):255–262. doi: 10.1016/j.ejphar.2007.11.073. [DOI] [PubMed] [Google Scholar]
- 4.Joëls M, de Kloet ER. Mineralocorticoid and glucocorticoid receptors in the brain. Implications for ion permeability and transmitter systems. Prog Neurobiol. 1994;43(1):1–36. doi: 10.1016/0301-0082(94)90014-0. [DOI] [PubMed] [Google Scholar]
- 5.Hill MN, Tasker JG. Endocannabinoid signaling, glucocorticoid-mediated negative feedback, and regulation of the hypothalamic-pituitary-adrenal axis. Neuroscience. 2012;204:5–16. doi: 10.1016/j.neuroscience.2011.12.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sawchenko PE. Evidence for a local site of action for glucocorticoids in inhibiting CRF and vasopressin expression in the paraventricular nucleus. Brain Research. 1987;403(2):213–224. doi: 10.1016/0006-8993(87)90058-8. [DOI] [PubMed] [Google Scholar]
- 7.Plotsky PM, Otto S, Sapolsky RM. Inhibition of immunoreactive corticotropin-releasing factor secretion into the hypophysial-portal circulation by delayed glucocorticoid feedback. Endocrinology. 1986;119(3):1126–1130. doi: 10.1210/endo-119-3-1126. [DOI] [PubMed] [Google Scholar]
- 8.Plotsky PM, Sawchenko PE. Hypophysial-Portal Plasma Levels, Median Eminence Content, and Immunohistochemical Staining of Corticotropin-Releasing Factor, Arginine Vasopressin, and Oxytocin after Pharmacological Adrenalectomy. Endocrinology. 1987;120(4):1361–1369. doi: 10.1210/endo-120-4-1361. [DOI] [PubMed] [Google Scholar]
- 9.Dallman MF, Akana SF, Levin N, Walker CD, Bradbury MJ, Suemaru S, Scribner KS. Corticosteroids and the control of function in the hypothalamo- pituitary-adrenal (HPA) axis. Ann NY Acad Sci. 1994;746:22–28. doi: 10.1111/j.1749-6632.1994.tb39206.x. [DOI] [PubMed] [Google Scholar]
- 10.Gagner JP, Drouin J. Opposite regulation of pro-opiomelanocortin gene transcription by glucocorticoids and CRH. Mol Cell Endocrinol. 1985;40(1):25–32. doi: 10.1016/0303-7207(85)90154-6. [DOI] [PubMed] [Google Scholar]
- 11.Eberwine JH, Jonassen JA, Evinger MJ, Roberts JL. Complex transcriptional regulation by glucocorticoids and corticotropin-releasing hormone of proopiomelanocortin gene expression in rat pituitary cultures. DNA. 1987;6(5):483–492. doi: 10.1089/dna.1987.6.483. [DOI] [PubMed] [Google Scholar]
- 12.Aguilera G, Liu Y. The molecular physiology of CRH neurons. Front Neuroendocrinol. 2012;33(1):17. doi: 10.1016/j.yfrne.2011.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ma XM, Camacho C, Aguilera G. Regulation of corticotropin-releasing hormone (CRH) transcription and CRH mRNA stability by glucocorticoids. Cell Mol Neurobiol. 2001;21(5):465–475. doi: 10.1023/A:1013863205647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Spencer CM, Eberwine J. Cytoplasmic proteins interact with a translational control element in the protein-coding region of proopiomelanocortin mRNA. DNA Cell Biol. 1999;18(1):39–49. doi: 10.1089/104454999315600. [DOI] [PubMed] [Google Scholar]
- 15.Evans AN, Liu Y, Macgregor R, Huang V, Aguilera G. Regulation of hypothalamic corticotropin releasing hormone transcription by elevated glucocorticoids. Mol Endocrinol. 2013;27(11):1796–1807. doi: 10.1210/me.2013-1095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ma XM, Aguilera G. Differential regulation of corticotropin-releasing hormone and vasopressin transcription by glucocorticoids. Endocrinology. 1999;140(12):5642–5650. doi: 10.1210/endo.140.12.7214. [DOI] [PubMed] [Google Scholar]
- 17.Kovács KJ, Földes A, Sawchenko PE. Glucocorticoid negative feedback selectively targets vasopressin transcription in parvocellular neurosecretory neurons. J Neurosci. 2000;20(10):3843–3852. doi: 10.1523/JNEUROSCI.20-10-03843.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liposits Z, Uht RM, Harrison RW, Gibbs FP, Paull WK, Bohn MC. Ultrastructural localization of glucocorticoid receptor (GR) in hypothalamic paraventricular neurons synthesizing corticotropin releasing factor (CRF) Histochemistry. 1987;87(5):407–412. doi: 10.1007/BF00496811. [DOI] [PubMed] [Google Scholar]
- 19.Fenoglio KA, Brunson KL, Avishai-Eliner S, Chen Y, Baram TZ. Region-Specific Onset of Handling-Induced Changes in Corticotropin-Releasing Factor and Glucocorticoid Receptor Expression. Endocrinology. 2004;145(6):2702–2706. doi: 10.1210/en.2004-0111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Harbuz MS, Lightman SL. Glucocorticoid inhibition of stress-induced changes in hypothalamic corticotrophin-releasing factor messenger RNA and proenkephalin A messenger RNA. Neuropeptides. 1989;14(1):17–20. doi: 10.1016/0143-4179(89)90029-2. [DOI] [PubMed] [Google Scholar]
- 21.Makino S, Smith MA, Gold PW. Increased expression of corticotropin-releasing hormone and vasopressin messenger ribonucleic acid (mRNA) in the hypothalamic paraventricular nucleus during repeated stress: association with reduction in glucocorticoid receptor mRNA levels. Endocrinology. 1995;136(8):3299–3309. doi: 10.1210/endo.136.8.7628364. [DOI] [PubMed] [Google Scholar]
- 22.Ma XM, Lightman SL, Aguilera G. Vasopressin and Corticotropin-Releasing Hormone Gene Responses to Novel Stress in Rats Adapted to Repeated Restraint. Endocrinology. 1999;140(8):3623–3632. doi: 10.1210/endo.140.8.6943. [DOI] [PubMed] [Google Scholar]
- 23.Kóvacs KJ, Makara GB. Corticosterone and dexamethasone act at different brain sites to inhibit adrenalectomy-induced adrenocorticotropin hypersecretion. Brain Research. 1988;474:205–210. doi: 10.1016/0006-8993(88)90435-0. [DOI] [PubMed] [Google Scholar]
- 24.Malkoski SP, Handanos CM, Dorin RI. Localization of a negative glucocorticoid response element of the human corticotropin releasing hormone gene. Mol Cell Endocrinol. 1997;127(2):189–199. doi: 10.1016/s0303-7207(96)04004-x. [DOI] [PubMed] [Google Scholar]
- 25.Malkoski SP, Dorin RI. Composite glucocorticoid regulation at a functionally defined negative glucocorticoid response element of the human corticotropin-releasing hormone gene. Mol Endocrinol. 1999;13(10):1629–1644. doi: 10.1210/mend.13.10.0351. [DOI] [PubMed] [Google Scholar]
- 26.Guardiola-Diaz HM, Kolinske JS, Gates LH, Seasholtz AF. Negative glucorticoid regulation of cyclic adenosine 3', 5'-monophosphate-stimulated corticotropin-releasing hormone-reporter expression in AtT-20 cells. Mol Endocrinol. 1996;10(3):317–329. doi: 10.1210/mend.10.3.8833660. [DOI] [PubMed] [Google Scholar]
- 27.Ma XM, Levy A, Lightman SL. Rapid changes of heteronuclear RNA for arginine vasopressin but not for corticotropin releasing hormone in response to acute corticosterone administration. J Neuroendocrinol. 1997;9(10):723–728. doi: 10.1046/j.1365-2826.1997.00646.x. [DOI] [PubMed] [Google Scholar]
- 28.Shepard JD, Liu Y, Sassone-Corsi P, Aguilera G. Role of Glucocorticoids and cAMP-Mediated Repression in Limiting Corticotropin-Releasing Hormone Transcription during Stress. J Neurosci. 2005;25(16):4073–4081. doi: 10.1523/JNEUROSCI.0122-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liu Y, Poon V, Sanchez-Watts G, Watts AG, Takemori H, Aguilera G. Salt-Inducible Kinase Is Involved in the Regulation of Corticotropin-Releasing Hormone Transcription in Hypothalamic Neurons in Rats. Endocrinology. 2012;153(1):223–233. doi: 10.1210/en.2011-1404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.So AY, Chaivorapol C, Bolton EC, Li H, Yamamoto KR. Determinants of cell- and genespecific transcriptional regulation by the glucocorticoid receptor. PLOS Genet. 2007;3(6):e94–e94. doi: 10.1371/journal.pgen.0030094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hakim O, John S, Ling JQ, Biddie SC, Hoffman AR, Hager GL. Glucocorticoid Receptor Activation of the Ciz1-Lcn2 Locus by Long Range Interactions. J Biol Chem. 2009;284(10):6048–6052. doi: 10.1074/jbc.C800212200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Paakinaho V, Makkonen H, Jaaskelainen T, Palvimo JJ. Glucocorticoid Receptor Activates Poised FKBP51 Locus through Long-Distance Interactions. Mol Endocrinol. 2010;24(3):511–525. doi: 10.1210/me.2009-0443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Liu Y, Coello AG, Grinevich V, Aguilera G. Involvement of Transducer of Regulated cAMP Response Element-Binding Protein Activity on Corticotropin Releasing Hormone Transcription. Endocrinology. 2010;151(3):1109–1118. doi: 10.1210/en.2009-0963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liu Y, Knobloch HS, Grinevich V, Aguilera G. Stress induces nuclear translocation of the CREB co-activator, transducer of regulated CREB activity (TORC) in corticotropin releasing hormone neurons. J Neuroendocrinol. 2011;23:216–223. doi: 10.1111/j.1365-2826.2010.02101.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jeanneteau FD, Lambert WM, Ismaili N, Bath KG, Lee FS, Garabedian MJ, Chao MV. BDNF and glucocorticoids regulate corticotrophin-releasing hormone (CRH) homeostasis in the hypothalamus. Proc Natl Acad Sci. 2012;109(4):1305–1310. doi: 10.1073/pnas.1114122109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Adler GK, Smas CM, Majzoub JA. Expression and dexamethasone regulation of the human corticotropin-releasing hormone gene in a mouse anterior pituitary cell line. J Biol Chem. 1988;263(12):5846–5852. [PubMed] [Google Scholar]
- 37.Kageyama K, Akimoto K, Suda T. Corticotrophin-releasing factor gene transcription is directly activated after deprivation of glucocorticoids in hypothalamic cells. J Neuroendocrinol. 2010;22(9):971–978. doi: 10.1111/j.1365-2826.2010.02048.x. [DOI] [PubMed] [Google Scholar]
- 38.Dallman MF, Yates FE. Dynamic asymmetries in the corticosteroid feedback path and distribution-metabolism-binding elements of the adrenocortical system. Ann N Y Acad Sci. 1969;156:696–721. doi: 10.1111/j.1749-6632.1969.tb14008.x. [DOI] [PubMed] [Google Scholar]
- 39.Di S, Malcher-Lopes R, Halmos KC, Tasker JG. Nongenomic glucocorticoid inhibition via endocannabinoid release in the hypothalamus: a fast feedback mechanism. J Neurosci. 2003;23:4850–4857. doi: 10.1523/JNEUROSCI.23-12-04850.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Malcher-Lopes R, Di S, Marcheselli VS, Weng FJ, Stuart CT, Bazan NG, Tasker JG. Opposing crosstalk between leptin and glucocorticoids rapidly modulates synaptic excitation via endocannabinoid release. J Neurosci. 2006;26:6643–6650. doi: 10.1523/JNEUROSCI.5126-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Patel S, Roelke CT, Rademacher DJ, Cullinan WE, Hillard CJ. Endocannabinoid signaling negatively modulates stress-induced activation of the hypothalamic-pituitary-adrenal axis. Endocrinology. 2004;145:5431–5438. doi: 10.1210/en.2004-0638. [DOI] [PubMed] [Google Scholar]
- 42.Cota D, Steiner MA, Marsicano G, Cervino C, Herman JP, Grubler Y, Stalla J, Pasquali R, Lutz B, Stalla GK, Pagotto U. Requirement of cannabinoid receptor type 1 for the basal modulation of hypothalamic-pituitary-adrenal axis function. Endocrinology. 2007;148:1574–1581. doi: 10.1210/en.2005-1649. [DOI] [PubMed] [Google Scholar]
- 43.Ginsberg AB, Pecoraro NC, Warne JP, Horneman HF, Dallman MF. Rapid alteration of stress-induced hypothalamic-pituitary-adrenal hormone secretion in the rat: a comparison of glucocorticoids and cannabinoids. Stress. 2010;13:248–257. doi: 10.3109/10253890903336839. [DOI] [PubMed] [Google Scholar]
- 44.Hill MN, McEwen BS. Involvement of the endocannabinoid system in the neurobehavioural effects of stress and glucocorticoids. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34:791–797. doi: 10.1016/j.pnpbp.2009.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Evanson NK, Tasker JG, Hill MN, Hillard CJ, Herman JP. Fast feedback inhibition of the HPA axis by glucocorticoids is mediated by endocannabinoid signaling. Endocrinology. 2010;151:4811–4819. doi: 10.1210/en.2010-0285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hamm J, Halmos KC, Muglia LJ, Tasker JG. 2010 Neuroscience Meeting Planner. San Diego, CA: Society for Neuroscience; 2010. Rapid synaptic modulation of hypothalamic neurons by glucocorticoids requires the glucocorticoid receptor. Program No. 389.19. [Google Scholar]
- 47.Johnson LR, Farb C, Morrison JH, McEwen BS, LeDoux JE. Localization of glucocorticoid receptors at postsynaptic membranes in the lateral amygdala. Neuroscience. 2005;136:289–299. doi: 10.1016/j.neuroscience.2005.06.050. [DOI] [PubMed] [Google Scholar]
- 48.Komatsuzaki Y, Murakami G, Tsurugizawa T, Mukai H, Tanabe N, Mitsuhashi K, Kawata M, Kimoto T, Ooishi Y, Kawato S. Rapid spinogenesis of pyramidal neurons induced by activation of glucocorticoid receptors in adult male rat hippocampus. Biochem Biophys Res Commun. 2005;335:1002–1007. doi: 10.1016/j.bbrc.2005.07.173. [DOI] [PubMed] [Google Scholar]
- 49.Wang CC, Wang SJ. Modulation of presynaptic glucocorticoid receptors on glutamate release from rat hippocampal nerve terminals. Synapse. 2009;63:745–751. doi: 10.1002/syn.20654. [DOI] [PubMed] [Google Scholar]
- 50.Prager EM, Brielmaier J, Bergstrom HC, McGuire J, Johnson LR. Localization of mineralocorticoid receptors at mammalian synapses. PLoS One. 2010;5:e14344. doi: 10.1371/journal.pone.0014344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cunningham ET, Jr, Sawchenko PE. Anatomical specificity of noradrenergic inputs to the paraventricular and supraoptic nuclei of the rat hypothalamus. J Comp Neurol. 1988;274:60–76. doi: 10.1002/cne.902740107. [DOI] [PubMed] [Google Scholar]
- 52.Cunningham ET, Jr, Bohn MC, Sawchenko PE. Organization of adrenergic inputs to the paraventricular and supraoptic nuclei of the hypothalamus in the rat. J Comp Neurol. 1990;292:651–667. doi: 10.1002/cne.902920413. [DOI] [PubMed] [Google Scholar]
- 53.Pacak K, Palkovits M, Kopin IJ, Goldstein DS. Stress-induced norepinephrine release in the hypothalamic paraventricular nucleus and pituitary-adrenocortical and sympathoadrenal activity: in vivo microdialysis studies. Front Neuroendocrinol. 1995;16:89–150. doi: 10.1006/frne.1995.1004. [DOI] [PubMed] [Google Scholar]
- 54.Szafarczyk A, Malaval F, Laurent A, Gibaud R, Assenmacher I. Further evidence for a central stimulatory action of catecholamines on adrenocorticotropin release in the rat. Endocrinology. 1987;121:883–892. doi: 10.1210/endo-121-3-883. [DOI] [PubMed] [Google Scholar]
- 55.Itoi K, Suda T, Tozawa F, Dobashi I, Ohmori N, Sakai Y, Abe K, Demura H. Microinjection of norepinephrine into the paraventricular nucleus of the hypothalamus stimulates corticotropin-releasing factor gene expression in conscious rats. Endocrinology. 1994;135:2177–2182. doi: 10.1210/endo.135.5.7956940. [DOI] [PubMed] [Google Scholar]
- 56.Sarkar S, Fekete C, Legradi G, Lechan RM. Glucagon like peptide-1 (7–36) amide (GLP-1) nerve terminals densely innervate corticotropin-releasing hormone neurons in the hypothalamic paraventricular nucleus. Brain Res. 2003;985:163–168. doi: 10.1016/s0006-8993(03)03117-2. [DOI] [PubMed] [Google Scholar]
- 57.Tauchi M, Zhang R, D'Alessio DA, Stern JE, Herman JP. Distribution of glucagon-like peptide-1 immunoreactivity in the hypothalamic paraventricular and supraoptic nuclei. J Chem Neuroanat. 2008a;36:144–149. doi: 10.1016/j.jchemneu.2008.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kinzig KP, D'Alessio DA, Herman JP, Sakai RR, Vahl TP, Figueiredo HF, Murphy EK, Seeley RJ. CNS glucagon-like peptide-1 receptors mediate endocrine and anxiety responses to interoceptive and psychogenic stressors. J Neurosci. 2003;23:6163–6170. doi: 10.1523/JNEUROSCI.23-15-06163.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Tauchi M, Zhang R, D'Alessio DA, Seeley RJ, Herman JP. Role of central glucagon-like peptide-1 in hypothalamo-pituitary-adrenocortical facilitation following chronic stress. Exp Neurol. 2008b;210:458–466. doi: 10.1016/j.expneurol.2007.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zhang R, Packard BA, Tauchi M, D'Alessio DA, Herman JP. Glucocorticoid regulation of preproglucagon transcription and RNA stability during stress. Proc Natl Acad Sci USA. 2009;106:5913–5918. doi: 10.1073/pnas.0808716106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Fossom LH, Sterling CR, Tank AW. Regulation of tyrosine hydroxylase gene transcription rate and tyrosine hydroxylase mRNA stability by cyclic AMP and glucocorticoid. Mol Pharmacol. 1992;42:898–908. [PubMed] [Google Scholar]
- 62.Dhawan L, Liu B, Blaxall BC, Taubman MB. A novel role for the glucocorticoid receptor in the regulation of monocyte chemoattractant protein-1 mRNA stability. J Biol Chem. 2007;282:10146–10152. doi: 10.1074/jbc.M605925200. [DOI] [PubMed] [Google Scholar]
- 63.Keller-Wood M, Dallman MF. Corticosteroid inhibition of ACTH secretion. Endocrine Rev. 1984;5:1–24. doi: 10.1210/edrv-5-1-1. [DOI] [PubMed] [Google Scholar]
- 64.Laugero KD, Bell ME, Bhatnagar S, Soriano L, Dallman MF. Sucrose ingestion normalizes central expression of corticotropin-releasing-factor messenger ribonucleic acid and energy balance in adrenalectomized rats: a glucocorticoid-metabolic-brain axis? Endocrinology. 2001;142:2796–2804. doi: 10.1210/endo.142.7.8250. [DOI] [PubMed] [Google Scholar]
- 65.Dallman MF, Akana SF, Laugero KD, Gomez F, Manalo S, Bell ME, Bhatnagar S. A spoonful of sugar: feedback signals of energy stores and corticosterone regulate responses to chronic stress. Physiol Behav. 2003;79:3–12. doi: 10.1016/s0031-9384(03)00100-8. [DOI] [PubMed] [Google Scholar]
- 66.la Fleur SE, Houshyar H, Roy M, Dallman MF. Choice of lard, but not total lard calories, damps adrenocorticotropin responses to restraint. Endocrinology. 2005;146:2193–2199. doi: 10.1210/en.2004-1603. [DOI] [PubMed] [Google Scholar]
- 67.Campbell JE, Peckett AJ, D'Souza AM, Hawke TJ, Riddell MC. Adipogenic and lipolytic effects of chronic glucocorticoid exposure. Am J Physiol Cell Physiol. 2011;300:C198–C209. doi: 10.1152/ajpcell.00045.2010. [DOI] [PubMed] [Google Scholar]
- 68.Oh YT, Oh KS, Kang I, Youn JH. A Fall in plasma free fatty acid (FFA) level activates the hypothalamic-pituitary-adrenal axis independent of plasma glucose: evidence for brain sensing of circulating FFA. Endocrinology. 2012;153:3587–3592. doi: 10.1210/en.2012-1330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Oh YT, Kim J, Kang I, Youn JH. Regulation of Hypothalamic-pituitary-adrenal Axis by Circulating Free Fatty Acids in Male Wistar Rats: Role of Individual Free Fatty Acids. Endocrinology. 2014 doi: 10.1210/en.2013-1700. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Sarruf DA, Yu F, Nguyen HT, Williams DL, Printz RL, Niswender KD, Schwartz MW. Expression of peroxisome proliferator-activated receptor-gamma in key neuronal subsets regulating glucose metabolism and energy homeostasis. Endocrinology. 2009;150:707–712. doi: 10.1210/en.2008-0899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ryan KK, Grayson BE, Jones KR, Schneider AL, Woods SC, Seeley RJ, Herman JP, Ulrich-Lai YM. Physiological responses to acute psychological stress are reduced by the PPARgamma agonist rosiglitazone. Endocrinology. 2012;153:1279–1287. doi: 10.1210/en.2011-1689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ulrich-Lai YM, Herman JP. Neural regulation of endocrine and autonomic stress responses. Nat Rev Neurosci. 2009;10:397–409. doi: 10.1038/nrn2647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Buckingham JC, John CD, Solito E, Tierney T, Flower RJ, Christian H, Morris J. Annexin 1, glucocorticoids, and the neuroendocrine-immune interface. Ann N Y Acad Sci. 2006;1088:396–409. doi: 10.1196/annals.1366.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Jasper MS, Engeland WC. Splanchnicotomy increases adrenal sensitivity to ACTH in nonstressed rats. Am J Physiol. 1997;273:E363–E368. doi: 10.1152/ajpendo.1997.273.2.E363. [DOI] [PubMed] [Google Scholar]
- 75.Joels M, Baram TZ. The neuro-symphony of stress. Nat Rev Neurosci. 2009;10(6):459–466. doi: 10.1038/nrn2632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.McEwen BS. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev. 2007;87(3):873–904. doi: 10.1152/physrev.00041.2006. [DOI] [PubMed] [Google Scholar]
- 77.Bravo JA, Parra CS, Arancibia S, Andres S, Morales P, Herrera-Marschitz M, Herrera L, Lara HE, Fiedler JL. Adrenalectomy promotes a permanent decrease of plasma corticoid levels and a transient increase of apoptosis and the expression of Transforming Growth Factor beta1 (TGF-beta1) in hippocampus: effect of a TGF-beta1 oligo-antisense. BMC Neurosci. 2006:740. doi: 10.1186/1471-2202-7-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Greiner M, Cardenas S, Parra C, Bravo J, Avalos AM, Paredes A, Lara HE, Fiedler JL. Adrenalectomy regulates apoptotic-associated genes in rat hippocampus. Endocrine. 2001;15(3):323–33. doi: 10.1385/ENDO:15:3:323. [DOI] [PubMed] [Google Scholar]
- 79.Sloviter RS, Sollas AL, Dean E, Neubort S. Adrenalectomy-induced granule cell degeneration in the rat hippocampal dentate gyrus: characterization of an in vivo model of controlled neuronal death. J Comp Neurol. 1993;330(3):324–36. doi: 10.1002/cne.903300304. [DOI] [PubMed] [Google Scholar]
- 80.Sloviter RS, Valiquette G, Abrams GM, Ronk EC, Sollas AL, Paul LA, Neubort S. Selective loss of hippocampal granule cells in the mature rat brain after adrenalectomy. Science. 1989;243(4890):535–538. doi: 10.1126/science.2911756. [DOI] [PubMed] [Google Scholar]
- 81.Woolley CS, Gould E, Sakai RR, Spencer RL, McEwen BS. Effects of aldosterone or RU28362 treatment on adrenalectomy-induced cell death in the dentate gyrus of the adult rat. Brain Res. 1991;554(1–2):312–315. doi: 10.1016/0006-8993(91)90207-c. [DOI] [PubMed] [Google Scholar]
- 82.Cardenas SP, Parra C, Bravo J, Morales P, Lara HE, Herrera-Marschitz M, Fiedler JL. Corticosterone differentially regulates bax, bcl-2 and bcl-x mRNA levels in the rat hippocampus. Neurosci Lett. 2002;331(1):9–12. doi: 10.1016/s0304-3940(02)00744-9. [DOI] [PubMed] [Google Scholar]
- 83.Andres S, Cardenas S, Parra C, Bravo J, Greiner M, Rojas P, Morales P, Lara H, Fiedler J. Effects of long-term adrenalectomy on apoptosis and neuroprotection in the rat hippocampus. Endocrine. 2006;29(2):299–308. doi: 10.1385/ENDO:29:2:299. [DOI] [PubMed] [Google Scholar]
- 84.Cameron HA, Gould E. Adult neurogenesis is regulated by adrenal steroids in the dentate gyrus. Neuroscience. 1994;61(2):203–209. doi: 10.1016/0306-4522(94)90224-0. [DOI] [PubMed] [Google Scholar]
- 85.Joels M, Pu Z, Wiegert O, Oitzl MS, Krugers HJ. Learning under stress: how does it work? Trends Cogn Sci. 2006;10(4):152–158. doi: 10.1016/j.tics.2006.02.002. [DOI] [PubMed] [Google Scholar]
- 86.Bagley J, Moghaddam B. Temporal dynamics of glutamate efflux in the prefrontal cortex and in the hippocampus following repeated stress: effects of pretreatment with saline or diazepam. Neuroscience. 1997;77(1):65–73. doi: 10.1016/s0306-4522(96)00435-6. [DOI] [PubMed] [Google Scholar]
- 87.Musazzi L, Milanese M, Farisello P, Zappettini S, Tardito D, Barbiero VS, Bonifacino T, Mallei A, Baldelli P, Racagni G, Raiteri M, Benfenati F, Bonanno G, Popoli M. Acute stress increases depolarization-evoked glutamate release in the rat prefrontal/frontal cortex: the dampening action of antidepressants. PloS One. 2010;5(1):e8566. doi: 10.1371/journal.pone.0008566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Reznikov LR, Grillo CA, Piroli GG, Pasumarthi RK, Reagan LP, Fadel J. Acute stress-mediated increases in extracellular glutamate levels in the rat amygdala: differential effects of antidepressant treatment. Eur J Neurosci. 2007;25(10):3109–3114. doi: 10.1111/j.1460-9568.2007.05560.x. [DOI] [PubMed] [Google Scholar]
- 89.Moghaddam B, Bolinao ML, Stein-Behrens B, Sapolsky R. Glucocorticoids mediate the stress-induced extracellular accumulation of glutamate. Brain Res. 1994;655(1–2):251–254. doi: 10.1016/0006-8993(94)91622-5. [DOI] [PubMed] [Google Scholar]
- 90.Venero C, Borrell J. Rapid glucocorticoid effects on excitatory amino acid levels in the hippocampus: a microdialysis study in freely moving rats. Eur J Neurosci. 1999;11(7):2465–2473. doi: 10.1046/j.1460-9568.1999.00668.x. [DOI] [PubMed] [Google Scholar]
- 91.Karst H, Joels M. Corticosterone slowly enhances miniature excitatory postsynaptic current amplitude in mice CA1 hippocampal cells. J Neurophysiol. 2005;94(5):3479–3486. doi: 10.1152/jn.00143.2005. [DOI] [PubMed] [Google Scholar]
- 92.Karst H, Berger S, Turiault M, Tronche F, Schutz G, Joels M. Mineralocorticoid receptors are indispensable for nongenomic modulation of hippocampal glutamate transmission by corticosterone. Proc Nal Acad Sci USA. 2005;102(52):19204–19207. doi: 10.1073/pnas.0507572102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Liston C, Cichon JM, Jeanneteau F, Jia Z, Chao MV, Gan WB. Circadian glucocorticoid oscillations promote learning-dependent synapse formation and maintenance. Nature Neurosci. 2013;16(6):698–705. doi: 10.1038/nn.3387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Shors TJ, Chua C, Falduto J. Sex differences and opposite effects of stress on dendritic spine density in the male versus female hippocampus. J Neurosci. 2001;21(16):6292–6297. doi: 10.1523/JNEUROSCI.21-16-06292.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Komatsuzaki Y, Murakami G, Tsurugizawa T, Mukai H, Tanabe N, Mitsuhashi K, Kawata M, Kimoto T, Ooishi Y, Kawato S. Rapid spinogenesis of pyramidal neurons induced by activation of glucocorticoid receptors in adult male rat hippocampus. Biochem Biophys Res Commun. 2005;335(4):1002–1007. doi: 10.1016/j.bbrc.2005.07.173. [DOI] [PubMed] [Google Scholar]
- 96.Komatsuzaki Y, Hatanaka Y, Murakami G, Mukai H, Hojo Y, Saito M, Kimoto T, Kawato S. Corticosterone induces rapid spinogenesis via synaptic glucocorticoid receptors and kinase networks in hippocampus. PloS One. 2012;7(4):e34124. doi: 10.1371/journal.pone.0034124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Jafari M, Seese RR, Babayan AH, Gall CM, Lauterborn JC. Glucocorticoid receptors are localized to dendritic spines and influence local actin signaling. Mol Neurobiol. 2012;46(2):304–315. doi: 10.1007/s12035-012-8288-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Miyashiro KY, Beckel-Mitchener A, Purk TP, Becker KG, Barret T, Liu L, Carbonetto S, Weiler IJ, Greenough WT, Eberwine J. RNA cargoes associating with FMRP reveal deficits in cellular functioning in Fmr1 null mice. Neuron. 2003;37(3):417–431. doi: 10.1016/s0896-6273(03)00034-5. [DOI] [PubMed] [Google Scholar]
- 99.Sidorov MS, Auerbach BD, Bear MF. Fragile X mental retardation protein and synaptic plasticity. Mol Brain. 2013;6:15. doi: 10.1186/1756-6606-6-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Martin S, Henley JM, Holman D, Zhou M, Wiegert O, van Spronsen M, Joels M, Hoogenraad CC, Krugers HJ. Corticosterone alters AMPAR mobility and facilitates bidirectional synaptic plasticity. PloS One. 2009;4(3):e4714. doi: 10.1371/journal.pone.0004714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Conboy L, Sandi C. Stress at learning facilitates memory formation by regulating AMPA receptor trafficking through a glucocorticoid action. Neuropsychopharmacology. 2010;35(3):674–685. doi: 10.1038/npp.2009.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Bramham CR, Worley PF, Moore MJ, Guzowski JF. The immediate early gene arc/arg3.1: regulation, mechanisms, and function. J Neurosci. 2008;28(46):11760–11767. doi: 10.1523/JNEUROSCI.3864-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Guzowski JF, Lyford GL, Stevenson GD, Houston FP, McGaugh JL, Worley PF, Barnes CA. Inhibition of activity-dependent arc protein expression in the rat hippocampus impairs the maintenance of long-term potentiation and the consolidation of long-term memory. J Neurosci. 2000;20(11):3993–4001. doi: 10.1523/JNEUROSCI.20-11-03993.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Rial Verde EM, Lee-Osbourne J, Worley PF, Malinow R, Cline HT. Increased expression of the immediate-early gene arc/arg3.1 reduces AMPA receptor-mediated synaptic transmission. Neuron. 2006;52(3):461–474. doi: 10.1016/j.neuron.2006.09.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Shepherd JD, Bear MF. New views of Arc, a master regulator of synaptic plasticity. Nat Neurosci. 2011;14(3):279–284. doi: 10.1038/nn.2708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Schwartz MW, Woods SC, Porte JRD, Seeley RJ, Baskin DG. Central nervous system control of food intake. Nature. 2000;404(6778):661–671. doi: 10.1038/35007534. [DOI] [PubMed] [Google Scholar]
- 107.Gehlert DR. Role of hypothalamic neuropeptide Y in feeding and obesity. Neuropeptides. 1999;33(5):329–338. doi: 10.1054/npep.1999.0057. [DOI] [PubMed] [Google Scholar]
- 108.Smith PM, Ferguson AV. Neurophysiology of hunger and satiety. Dev Disabil Res Rev. 2008;14(2):96–104. doi: 10.1002/ddrr.13. [DOI] [PubMed] [Google Scholar]
- 109.Valassi E, Scacchi M, Cavagnini F. Neuroendocrine control of food intake. Nutr Metab Cardiovasc Dis. 2008;18(2):158–168. doi: 10.1016/j.numecd.2007.06.004. [DOI] [PubMed] [Google Scholar]
- 110.Havel PJ. Peripheral signals conveying metabolic information to the brain: short-term and long-term regulation of food intake and energy homeostasis. Exp Biol Med. 2001;226(11):963–977. doi: 10.1177/153537020122601102. [DOI] [PubMed] [Google Scholar]
- 111.La Fleur SE. The effects of glucocorticoids on feeding behavior in rats. Physiol Behav. 2006;89(1):110–114. doi: 10.1016/j.physbeh.2006.01.028. [DOI] [PubMed] [Google Scholar]
- 112.Leal AM, Moreira AC. Food and the circadian activity of the hypothalamic-pituitaryadrenal axis. Braz J Med Biol Res. 1997;30(12):1391–1405. doi: 10.1590/s0100-879x1997001200003. [DOI] [PubMed] [Google Scholar]
- 113.Honma KI, Honma S, Hiroshige T. Critical role of food amount for prefeeding corticosterone peak in rats. Am J Physiol. 1983;245(3):R339–R344. doi: 10.1152/ajpregu.1983.245.3.R339. [DOI] [PubMed] [Google Scholar]
- 114.Tataranni PA, Larson DE, Snitker S, Young JB, Flatt JP, Ravussin E. Effects of glucocorticoids on energy metabolism and food intake in humans. Am J Physiol. 1996;271(2):E317–E325. doi: 10.1152/ajpendo.1996.271.2.E317. [DOI] [PubMed] [Google Scholar]
- 115.Nieuwenhuizen AG, Rutters F. The hypothalamic-pituitary-adrenal-axis in the regulation of energy balance. Physiol Behav. 2008;94(2):169–177. doi: 10.1016/j.physbeh.2007.12.011. [DOI] [PubMed] [Google Scholar]
- 116.Shibli-Rahhal A, Van Beek M, Schlechte JA. Cushing's syndrome. Clin Dermatol. 2006;24(4):260–265. doi: 10.1016/j.clindermatol.2006.04.012. [DOI] [PubMed] [Google Scholar]
- 117.Nieman LK, Chanco Turner ML. Addison's disease. Clin Dermatol. 2006;24(4):276–280. doi: 10.1016/j.clindermatol.2006.04.006. [DOI] [PubMed] [Google Scholar]
- 118.Devenport L, Knehans A, Sundstrom A, Thomas T. Corticosterone's dual metabolic actions. Life Sci. 1989;45(15):1389–1396. doi: 10.1016/0024-3205(89)90026-x. [DOI] [PubMed] [Google Scholar]
- 119.Tempel DL, Leibowitz SF. PVN steroid implants: effect on feeding patterns and macronutrient selection. Brain Res Bull. 1989;23(6):553–560. doi: 10.1016/0361-9230(89)90200-1. [DOI] [PubMed] [Google Scholar]
- 120.Tempel DL, McEwen BS, Leibowitz SF. Adrenal steroid receptors in the PVN: studies with steroid antagonists in relation to macronutrient intake. Neuroendocrinology. 1993;57(6):1106–1113. doi: 10.1159/000126477. [DOI] [PubMed] [Google Scholar]
- 121.Tempel DL, Leibowitz SF. Adrenal steroid receptors: interactions with brain neuropeptide systems in relation to nutrient intake and metabolism. J Neuroendocrinol. 1994;6(5):479–501. doi: 10.1111/j.1365-2826.1994.tb00611.x. [DOI] [PubMed] [Google Scholar]
- 122.Kumar BA, Leibowitz SF. Impact of acute corticosterone administration on feeding and macronutrient self-selection patterns. Am J Physiol. 1988;254(2):R222–R228. doi: 10.1152/ajpregu.1988.254.2.R222. [DOI] [PubMed] [Google Scholar]
- 123.Kumar BA, Papamichael M, Leibowitz SF. Feeding and macronutrient selection patterns in rats: adrenalectomy and chronic corticosterone replacement. Physiol Behav. 1988;42(6):581–589. doi: 10.1016/0031-9384(88)90161-8. [DOI] [PubMed] [Google Scholar]
- 124.Goldstein RE, Wasserman DH, McGuinness OP, Lacy DB, Cherrington AD, Abumrad NN. Effects of chronic elevation in plasma cortisol on hepatic carbohydrate metabolism. Am J Physiol. 1993;264(1):E119–E127. doi: 10.1152/ajpendo.1993.264.1.E119. [DOI] [PubMed] [Google Scholar]
- 125.Tomas FM, Munro HN, Young VR. Effect of glucocorticoid administration on the rate of muscle protein breakdown in vivo in rats, as measured by urinary excretion of N tau-methylhistidine. Biochem J. 1979;178(1):139–146. doi: 10.1042/bj1780139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Cusin I, Rouru J, Rohner-Jeanrenaud F. Intracerebroventricular glucocorticoid infusion in normal rats: induction of parasympathetic-mediated obesity and insulin resistance. Obes Res. 2001;9(7):401–406. doi: 10.1038/oby.2001.52. [DOI] [PubMed] [Google Scholar]
- 127.Zakrzewska KE, Cusin I, Stricker-Krongrad A, Boss O, Ricquier D, Jeanrenaud B, Rohner-Jeanrenaud F. Induction of obesity and hyperleptinemia by central glucocorticoid infusion in the rat. Diabetes. 1999;48(2):365–370. doi: 10.2337/diabetes.48.2.365. [DOI] [PubMed] [Google Scholar]
- 128.Aronsson M, Fuxe K, Dong Y, Agnati LF, Okret S, Gustafsson JA. Localization of glucocorticoid receptor mRNA in the male rat brain by in situ hybridization. Proc Natl Acad Sci USA. 1988;85(23):9331–9335. doi: 10.1073/pnas.85.23.9331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Hisano S, Kagotani Y, Tsuruo Y, Daikoku S, Chihara K, Whitnall MH. Localization of glucocorticoid receptor in neuropeptide Y-containing neurons in the arcuate nucleus of the rat hypothalamus. Neurosci Lett. 1988;95(1–3):13–18. doi: 10.1016/0304-3940(88)90624-6. [DOI] [PubMed] [Google Scholar]
- 130.Uchoa ET, Silva LE, de Castro M, Antunes-Rodrigues J, Elias LL. Glucocorticoids are required for meal-induced changes in the expression of hypothalamic neuropeptides. Neuropeptides. 2012;46(3):119–124. doi: 10.1016/j.npep.2012.02.002. [DOI] [PubMed] [Google Scholar]
- 131.Freedman MR, Castonguay TW, Stern JS. Effect of adrenalectomy and corticosterone replacement on meal patterns of Zucker rats. Am J Physiol. 1985;249(5):R584–R594. doi: 10.1152/ajpregu.1985.249.5.R584. [DOI] [PubMed] [Google Scholar]
- 132.Uchoa ET, Sabino HA, Ruginsk SG, Antunes-Rodrigues J, Elias LL. Hypophagia induced by glucocorticoid deficiency is associated with an increased activation of satiety-related responses. J Appl Physiol. 2009;106(2):596–604. doi: 10.1152/japplphysiol.90865.2008. [DOI] [PubMed] [Google Scholar]
- 133.Uchoa ET, Silva LE, de Castro M, Antunes-Rodrigues J, Elias LL. Hypothalamic oxytocin neurons modulate hypophagic effect induced by adrenalectomy. Horm Behav. 2009;56(5):532–538. doi: 10.1016/j.yhbeh.2009.09.007. [DOI] [PubMed] [Google Scholar]
- 134.Uchoa ET, Silva LE, de Castro M, Antunes-Rodrigues J, Elias LL. Corticotrophin-releasing factor mediates hypophagia after adrenalectomy, increasing meal-related satiety responses. Horm Behav. 2010;58(5):714–719. doi: 10.1016/j.yhbeh.2010.07.003. [DOI] [PubMed] [Google Scholar]
- 135.Bruce BK, King BM, Phelps GR, Veitia MC. Effects of adrenalectomy and corticosterone administration on hypothalamic obesity in rats. Am J Physiol. 1982;243(2):E152–E157. doi: 10.1152/ajpendo.1982.243.2.E152. [DOI] [PubMed] [Google Scholar]
- 136.Dubuc PU, Wilden NJ. Adrenalectomy reduces but does not reverse obesity in ob/ob mice. Int J Obes. 1986;10(2):91–98. [PubMed] [Google Scholar]
- 137.Yukimura Y, Bray GA, Wolfsen AR. Some effects of adrenalectomy in the fatty rat. Endocrinology. 1978;103(5):1924–1928. doi: 10.1210/endo-103-5-1924. [DOI] [PubMed] [Google Scholar]
- 138.Germano CM, Castro M, Rorato R, Laguna MT, Antunes-Rodrigues J, Elias CF, Elias LL. Time course effects of adrenalectomy and food intake on cocaine- and amphetamine-regulated transcript expression in the hypothalamus. Brain Res. 2007;1166:55–64. doi: 10.1016/j.brainres.2007.05.077. [DOI] [PubMed] [Google Scholar]
- 139.Uchoa ET, Zahm DS, de Carvalho Borges B, Rorato R, Antunes-Rodrigues J, Elias LL. Oxytocin projections to the nucleus of the solitary tract contribute to the increased meal-related satiety responses in primary adrenal insufficiency. Exp Physiol. 2013;98(10):1495–1504. doi: 10.1113/expphysiol.2013.073726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Deak T, Bordner K, McElderry N, Barnum C, Blandino P, Jr, Deak M, Tammariello S. Stress-induced increases in hypothalamic IL-1: a systematic analysis of multiplt stressor paradigms. Brain Res Bull. 2005;64:541–556. doi: 10.1016/j.brainresbull.2004.11.003. [DOI] [PubMed] [Google Scholar]
- 141.Hueston CM, Barnum CJ, Eberle JA, Ferraioli FJ, Buck HM, Deak T. Stress-dependent changes in neuroinflammatory markers observed after common laboratory stressors are not seen following acute social defeat of the Sprague Dawley rat. Physiol Behav. 2011;104:187–198. doi: 10.1016/j.physbeh.2011.03.013. [DOI] [PubMed] [Google Scholar]
- 142.Hueston CM, Buck HM, Deak T. The inflamed axis: The interaction between cytokines and HPA hormones in the stress response. Physiol Behav. 2014;124:77–91. doi: 10.1016/j.physbeh.2013.10.035. [DOI] [PubMed] [Google Scholar]
- 143.Garcia-Bueno B, Madrigal JL, Perez-Nievas BG, Leza JC. Stress mediators regulate brain prostaglandin synthesis and peroxisome proliferator-activated receptor-gamma activation after stress in rats. Endocrinology. 2008;149:1969–1978. doi: 10.1210/en.2007-0482. [DOI] [PubMed] [Google Scholar]
- 144.Frank MG, Baratta MV, Sprunger DB, Watkins LR, Maier SF. Microglia serve as a neuroimmune substrate for stress-induced potentiation of CNS pro-inflammatory cytokine responses. Brain Behav Immun. 2007;21:47–59. doi: 10.1016/j.bbi.2006.03.005. [DOI] [PubMed] [Google Scholar]
- 145.Nair A, Bonneau RH. Stress-induced elevation of glucocorticoids increases microglia proliferation through NMDA receptor activation. J Neuroimmunol. 2006;171:72–85. doi: 10.1016/j.jneuroim.2005.09.012. [DOI] [PubMed] [Google Scholar]
- 146.Blandino P, Jr, Barnum CJ, Solomon LG, Larish Y, Lankow BS, Deak T. Gene expression changes in the hypothalamus provide evidence for regionally-selective changes in IL-1 and microglial markers after acute stress. Brain Behav Immun. 2009;23:958–968. doi: 10.1016/j.bbi.2009.04.013. [DOI] [PubMed] [Google Scholar]
- 147.Sugama S, Fujita M, Hashimoto M, Conti B. Stress induced morphological microglial activation in the rodent brain: involvement of interleukin-18. Neuroscience. 2007;146:1388–1399. doi: 10.1016/j.neuroscience.2007.02.043. [DOI] [PubMed] [Google Scholar]
- 148.Blandino P, Jr, Barnum CJ, Deak T. The involvement of norepinephrine and microglia in hypothalamic and splenic IL-1beta responses to stress. J Neuroimmunol. 2006;173:87–95. doi: 10.1016/j.jneuroim.2005.11.021. [DOI] [PubMed] [Google Scholar]
- 149.Johnson JD, Campisi J, Sharkey CM, Kennedy SL, Nickerson M, Greenwood BN, Fleshner M. Catecholamines mediate stress-induced increases in peripheral and central inflammatory cytokines. Neuroscience. 2005;135:1295–1307. doi: 10.1016/j.neuroscience.2005.06.090. [DOI] [PubMed] [Google Scholar]
- 150.Blandino P, Jr, Hueston CM, Barnum CJ, Bishop C, Deak T. The impact of ventral noradrenergic bundle lesions on increased IL-1 in the PVN and hormonal responses to stress in male sprague dawley rats. Endocrinology. 2013;154:2489–2500. doi: 10.1210/en.2013-1075. [DOI] [PubMed] [Google Scholar]
- 151.Porterfield VM, Zimomra ZR, Caldwell EA, Camp RM, Gabella KM, Johnson JD. Rat strain differences in restraint stress-induced brain cytokines. Neuroscience. 2011;188:48–54. doi: 10.1016/j.neuroscience.2011.05.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Windle RJ, Wood SA, Lightman SL, Ingram CD. The pulsatile characteristics of hypothalamo-pituitary-adrenal activity in female Lewis and Fischer 344 rats and its relationship to differential stress responses. Endocrinology. 1998;139(10):4044–4052. doi: 10.1210/endo.139.10.6238. [DOI] [PubMed] [Google Scholar]
- 153.Nair A, Bonneau RH. Stress-induced elevation of glucocorticoids increases microglia proliferation through NMDA receptor activation. J Neuroimmunol. 2006;171(1–2):72–85. doi: 10.1016/j.jneuroim.2005.09.012. [DOI] [PubMed] [Google Scholar]
- 154.Maslanik T, Mahaffey L, Tannura K, Beninson L, Greenwood BN, Fleshner M. The inflammasome and danger associated molecular patterns (DAMPs) are implicated in cytokine and chemokine responses following stressor exposure. Brain Behav Immun. 2013;28:54–62. doi: 10.1016/j.bbi.2012.10.014. [DOI] [PubMed] [Google Scholar]
- 155.Nguyen K, Deak T, Owens S, Kohno T, Fleshner M, Watkins L, Maier S. Exposure ot acute stress induces brain interleukin-1 beta protein in the rat. J Neurosci. 1998;18:2239–2246. doi: 10.1523/JNEUROSCI.18-06-02239.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Nguyen KT, Deak T, Will MJ, Hansen MK, Hunsaker BN, Fleshner M, Watkins LR, Maier SF. Timecourse and corticosterone sensitivity of the brain, pituitary, and serum interleukin-1b protein response to acute stress. Brain Res. 2000;859:193–201. doi: 10.1016/s0006-8993(99)02443-9. [DOI] [PubMed] [Google Scholar]
- 157.Busillo JM, Cidlowski JA. The five Rs of glucocorticoid action during inflammation: ready, reinforce, repress, resolve, and restore. Trends Endocrinol Metab. 2013;24:109–119. doi: 10.1016/j.tem.2012.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Necela BM, Cidlowski JA. Mechanisms of glucocorticoid receptor action in noninflammatory and inflammatory cells. Proc Am Thorac Soc. 2004;1:239–246. doi: 10.1513/pats.200402-005MS. [DOI] [PubMed] [Google Scholar]
- 159.Watkins LR, Hansen MK, Nguyen KT, Lee JE, Maier SF. Dynamic regulation of the proinflammatory cytokine interleukin-1b: molecular biology for non-molecular biologists. Life Sci. 1999;65:449–481. doi: 10.1016/s0024-3205(99)00095-8. [DOI] [PubMed] [Google Scholar]
- 160.Frank MG, Watkins LR, Maier SF. Stress-induced glucocorticoids as a neuroendocrine alarm signal of danger. Brain Behav Immun. 2013;33:1–6. doi: 10.1016/j.bbi.2013.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Sorrells SF, Sapolsky RM. An inflammatory review of glucocorticoid actions in the CNS. Brain Behav Immun. 2007;21:259–272. doi: 10.1016/j.bbi.2006.11.00. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Goshen I, Kreisel T, Ben-Menachem-Zidon O, Licht T, Weidenfeld J, Ben-Hur T, Yirmiya R. Brain interleukin-1 mediates chronic stress-induced depression in mice via adrenocortical activation and hippocampal neurogenesis suppression. Mol Psychiatry. 2008;13:717–728. doi: 10.1038/sj.mp.4002055. [DOI] [PubMed] [Google Scholar]
- 163.Johnson JD, O'Connor KA, Watkins LR, Maier SF. The role of IL-1beta in stress-induced sensitization of proinflammatory cytokine and corticosterone responses. Neuroscience. 2004;127:569–577. doi: 10.1016/j.neuroscience.2004.05.046. [DOI] [PubMed] [Google Scholar]
- 164.Bornstein SR, KZiegler CG, Krug AW, Kanczkowski W, Rettori V, McCann SM, Wirth M, Zacharowski K. The role of toll-like receptors in the immune-adrenal crosstalk. Ann NY Acad Sci. 2006;1088:307–318. doi: 10.1196/annals.1366.027. [DOI] [PubMed] [Google Scholar]
- 165.Deak T. From classic aspects of the stress response to neuroinflammation and sickness: implications for individuals and offspring of diverse species. International. J Comp Psychol. 2007;20:96–110. [Google Scholar]
- 166.Engstrom L, Rosen K, Angel A, Fyrberg A, Mackerlova L, Konsman JP, Engblom D, Blomqvist A. Systemic immune challenge activates an intrinsically regulated local inflammatory circuit in the adrenal gland. Endocrinology. 2008;19(4):1436–14350. doi: 10.1210/en.2007-1456. [DOI] [PubMed] [Google Scholar]
- 167.Mazzocchi G, Gottardo G, Nussdorfer GG. A local immuno-endocrine interaction may mediate rat adrenal glucocorticoid response to bacterial endotoxins. Life Sci. 1998;62:1783–1787. doi: 10.1016/s0024-3205(98)00140-4. [DOI] [PubMed] [Google Scholar]
- 168.Beishuizen A, Thijs LG, Haanen C, Vermes I. Macrophage migration inhibitory factor and hypothalamo-pituitary-adrenal function during critical illness. J Clin Endocrinol Metab. 2001;86:2811–2816. doi: 10.1210/jcem.86.6.7570. [DOI] [PubMed] [Google Scholar]
- 169.Vermes I, Beishuizen A, Hampsink RM, Haanen C. Dissociation of plasma adrenocorticotropin and cortisol levels in critically ill patients: possible role of endothelin and atrial natriuretic hormone. J Clin Endocrinol Metab. 1995;80:1238–1242. doi: 10.1210/jcem.80.4.7714094. [DOI] [PubMed] [Google Scholar]
- 170.Arakawa H, Blandino P, Jr, Deak T. Central infusion of interleukin-1 receptor antagonist blocks the reduction in social behavior produced by prior stressor exposure. Physiol Behav. 2009;98:139–146. doi: 10.1016/j.physbeh.2009.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Koo JW, Duman RS. Interleukin-1 receptor null mutant mice show decreased anxietylike behavior and enhanced fear memory. Neurosci Lett. 2009;456:39–43. doi: 10.1016/j.neulet.2009.03.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Pugh CR, Nguyen KT, Gonyea JL, Fleshner M, Watkins LR, Maier SF, Rudy JW. Role of interleukin-1 beta in impairment of contextual fear conditioning caused by social isolation. Behav Brain Res. 1999;106:109–118. doi: 10.1016/s0166-4328(99)00098-4. [DOI] [PubMed] [Google Scholar]
- 173.Maier SF, Watkins LR. Intracerebroventricular interleukin-1 receptor antagonist blocks the enhancement of fear conditioning and interference with escape produced by inescapable shock. Brain Res. 1995;695:279–282. doi: 10.1016/0006-8993(95)00930-o. [DOI] [PubMed] [Google Scholar]
- 174.Barnum CJ, Tansey MG. Neuroinflammation and non-motor symptoms: the dark passenger of Parkinson's disease? Curr Neurol Neurosci Rep. 2012;12:350–358. doi: 10.1007/s11910-012-0283-6. [DOI] [PubMed] [Google Scholar]
- 175.Deak T. From hippocampus to dorsal horn: the pervasive impact of IL-1 on learning and memory spans the length of the neuroaxis. Brain, Behav Immun. 2007;21:746–747. doi: 10.1016/j.bbi.2007.04.001. [DOI] [PubMed] [Google Scholar]
- 176.Wager-Smith K, Markou A. Depression: a repair response to stress-induced neuronal microdamage that can grade into a chronic neuroinflammatory condition? Neurosci Biobehav Rev. 2011;35:742–764. doi: 10.1016/j.neubiorev.2010.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.