Abstract
Objectives
The objective of this work was to investigate the polymerization behavior, neutralization capability, and mechanical properties of dentin adhesive formulations with the addition of the tertiary amine co-monomer, 2-N-morpholinoethyl methacrylate (MEMA).
Methods
A co-monomer mixture based on HEMA/BisGMA (45/55, w/w) was used as a control adhesive. Compared with the control formulation, the MEMA-containing adhesive formulations were characterized comprehensively with regard to water miscibility of liquid resin, water sorption and solubility of cured polymer, real-time photopolymerization kinetics, dynamic mechanical analysis (DMA), and modulated differential scanning calorimetry (MDSC). The neutralization capacity was characterized by monitoring the pH shift of 1 mM lactic acid (LA) solution, in which the adhesive polymers were soaked.
Results
With increasing MEMA concentrations, experimental copolymers showed higher water sorption, lower glass transition temperature and lower crosslinking density compared to the control. The pH values of LA solution gradually increased from 3.5 to about 6.0–6.5 after 90 days. With the increase in crosslinking density of the copolymers, the neutralization rate was depressed. The optimal MEMA concentration was between 20 and 40 wt%.
Conclusions
As compared to the control, the results indicated that the MEMA-functionalized copolymer showed neutralization capability. The crosslinking density of the copolymer networks influenced the neutralization rate.
Keywords: Neutralization capacity, Tertiary amine, Photopolymerization, Crosslinking density, Dentin adhesive
1. Introduction
Resin-based composite is rapidly becoming the most popular material for direct restorative dentistry. In 2006, nearly 121 million resin-based composite restorations were placed [1]. Clinical results suggest that these restorations fail at 5.7 years (NIDCR Strategic Plan 2009–13) and the patients at highest risk for decay, including children, are particularly vulnerable to composite failure [2]. The primary reason for failure is recurrent decay [3] and nearly 80–90% of recurrent decay is located at the gingival margin of Class II and V restorations [4]. At the gingival margin, the dentin adhesive is the primary barrier between the oral environment and the repaired tooth.
The structure of polymethacrylate-based dentin adhesives suggests a general mechanism for their chemical and enzymatic degradation in oral fluids. In the oral environment, water penetrates the resin; water infiltration promotes the chemical hydrolysis of ester bonds in methacrylate materials. This reaction is expected to be relatively slow at the neutral pH typical in saliva, but excursions in pH caused by foods or cariogenic bacteria may lead to transient acid or base catalysis. Over years of exposure to salivary fluids, local domains of the polymethacrylate networks are degraded. Esterases infiltrate these degraded domains and accelerate ester bond hydrolysis [5,6]. In general, the ester bonds within the polymethacrylate-based network are vulnerable to two forms of hydrolytic attack: (1) chemical hydrolysis catalyzed by acids or bases and (2) enzymatic hydrolysis catalyzed by salivary enzymes, particularly esterases [7]. Establishing and maintaining the integrity of the adhesive and the adhesive/dentin (a/d) bond has been a critical roadblock to durable composite restorations [8].
Streptococcus mutans is a microorganism and a major causative agent of dental decay [9]. Adhesion of S. mutans to the a/d interface creates a biofilm and produces lactic acid (LA). The LA damages the adjacent tooth surface by demineralization. Although numerous monomers have been investigated [10–16], the lack of dentin adhesives that are both effective and durable continues to be a major problem in restorative dentistry. Different strategies have been developed to enhance the hydrolytic stability of dentin adhesive resins. These strategies include changing the monomer structure with an emphasis on increasing the hydrophobicity of the monomers by introducing urethane groups [17–19], branched methacrylate linkages [20], or ethoxylated BisGMA (BisEMA) [21]. The extent and rate of water uptake are depressed temporarily, but most of the materials still reach saturation within 7–60 day [22]. A second strategy involves enhancing the conversion of the monomer in the hybrid layer and is done by improving the compatibility between photoinitiator and hydrophilic phase. However, due to the gel effect and vitrification phenomena of polymer matrices occurring early in the initial light-irradiation, the degree of conversion (DC) of C=C double bonds cannot reach 100% [23,24]. A third strategy involves adding effective inhibitors (such as zinc or zinc-chelators) of dentine matrix metalloproteinases (MMPs) to enhance the stability of collagen and resin-infiltrated dentine [25]. The limitations associated with these strategies have fueled the ongoing efforts to identify new approaches for achieving a durable, integrated bond at the a/d interface.
Buffers are used to control the pH of a solution in biological and chemical applications. Monomers that have basic functional groups have the potential to mitigate acidic excursions in pH. 2-(dimethylamino) ethyl methacrylate (DMAEMA) is a basic monomer (pKa 8.2) [26] and its polymer (poly(2-(dimethylamino)ethyl methacrylate, PDMAEMA) is a weak polybase, which is water-soluble both at neutral pH and in acidic media due to protonation of the tertiary amine groups. PDMAEMA is a polybase that has a critical pH point around 7, which is close to the physiological pH [27]. However, DMAEMA has been shown to be unstable in basic solution by nuclear magnetic resonance (NMR) and can be completely hydrolyzed into methacrylic acid and dimethylaminoethanol, as shown in our previous work [28]. 2-N-morpholinoethyl methacrylate (MEMA) is another basic monomer with a pKa 6.2 and is able to neutralize LA more effectively than DMAEMA [26]. Because the pKa of MEMA is lower than DMAEMA, it begins buffering and raising the pH of the solution under more acidic conditions. PDMAEMA and poly(2-N-morpholinoethyl methacrylate)-based copolymers showed low toxicity when used as a controlled drug-delivery system [29–31].
As reported, the neutralization and physicochemical properties were measured based on tertiary amine monomers, linear polymers or low crosslinking density polymers (hydrogel) [26–28,30,31]. The neutralization behavior of amine-containing dentin adhesive copolymer has not been investigated systematically and the composition-structure-properties relationship of this kind of functionalized adhesive has not been studied before. In this work, MEMA is used as one of the co-monomers in dentin adhesives and the neutralization capacity was studied with the goal of determining its potential to reduce LA-induced demineralization without compromising the other properties required for dentin adhesives. MEMA was chosen as a neutralizing co-monomer because of its nearly neutral pKa and good biocompatibility. The present study tests the hypothesis that: (i) the tertiary amine group built-in to the dentin adhesive copolymer network can neutralize LA in wet conditions, and (ii) the high crosslinking density of the network structure does not retard the neutralization capacity of the copolymers.
2. Materials and methods
2.1. Materials
2,2-Bis[4-(2-hydroxy-3-methacryloxypropoxy) phenyl]propane (BisGMA), 2-hydroxyethyl methacrylate (HEMA), and 2-N-morpholinoethyl methacrylate (MEMA) were obtained from Sigma–Aldrich (St. Louis, MO) and used as received without further purification as monomers in dentin adhesives. Camphoroquinone (CQ), ethyl-4-(dimethylamino) benzoate (EDMAB), diphenyliodonium hexafluorophosphate (DPIHP), and L(+)-lactic acid (LA) were obtained from Sigma–Aldrich (St. Louis, MO). All other chemicals were reagent grade and used without further purification.
2.2. Preparation of adhesive formulations
HEMA/BisGMA (45/55, w/w) was used as the control (C0) [10]. The experimental adhesive formulations consisting of HEMA, BisGMA, and MEMA are listed in Table 1. CQ (0.5 wt%), EDMAB (0.5 wt%), and DPIHP (0.5 wt%) were used as a three-component photoinitiator system, [32,33] with respect to the total amount of monomers. Mixtures of monomers/photoinitiators are prepared in a brown glass vial under amber light. The preparation of adhesive formulations has been reported Previously [16].
Table 1.
Control and experimental formulations and polymerization kinetic data.
| Run | HEMA/% | BisGMA/% | MEMA/% | DC (%) | ||
|---|---|---|---|---|---|---|
| C0 | 45 | 55 | / | 64.8 (0.2) | 21.2 (0.6) | |
| HBM-40-40-20 | 40 | 40 | 20 | 71.8a (0.8) | 17.8a (0.1) | |
| HBM-40-30-30 | 40 | 30 | 30 | 74.1a (0.5) | 14.6a (1.3) | |
| HBM-40-25-35 | 40 | 25 | 35 | 72.7a (1.0) | 9.3a (1.2) | |
| HBM-40-20-40 | 40 | 20 | 40 | 66.1 (0.9) | 4.6a (0.2) | |
| HBM-40-15-45 | 40 | 15 | 45 | 54.5a (0.4) | 3.3a (0.4) | |
| HBM-40-10-50 | 40 | 10 | 50 | 34.5a (0.8) | 2.8a (0.1) | |
| HBM-40-5-55 | 40 | 5 | 55 | 20.9a (0.5) | 2.7a (0.1) |
Significantly (p < 0.05) different from the control (C0). The value in the () is the standard deviation.
2.3. Water miscibility of adhesive formulations
The water miscibility of adhesive formulations has been reported [34]. About 0.5 g of each neat resin was weighed into a brown vial, and water was added in increments of ~0.005 g until the mixture was visually observed to be turbid. The percentage of water in the mixture was noted (w1). The mixture was then back-titrated using the neat resin until the turbidity disappeared and the percentage of water in the mixture was noted (w2). Then the water miscibility (Wwm, %) of the liquid formulation was calculated as the average of w1 and w2. Three specimens of each formulation were measured.
2.4. Determination of log P
The log P values (ratio of solubility in octanol to solubility in water) for each of the monomers and the model adhesive formulations were predicted using ChemBioDraw Ultra 12.0 (Cambridge Soft from PerkinElmer) [34]. The log P value for each adhesive formulation was determined using the mole fraction-average of individual monomer values as seen in the following equation:
| (1) |
where xHEMA,xBisGMA, and xMEMA are the mole fractions of HEMA, BisGMA, and MEMA, respectively.
2.5. Real-time conversion and maximum polymerization rate
The degree of conversion (DC) and polymerization behavior were determined by FTIR [33]. Real-time, in-situ monitoring of the photopolymerization behavior of the adhesive formulations was performed using an infrared spectrometer (Spectrum 400 Fourier transform infrared spectrophotometer, PerkinElmer, Waltham, MA) at a resolution of 4 cm−1. One drop of adhesive solution was placed on the diamond crystal top plate of an attenuated total reflectance (ATR) accessory (PIKE Technologies Gladi-ATR, Madison, WI) and covered with a mylar film to reduce oxygen exposure. Exposure to the commercial visible-light-polymerization unit (Spectrum® 800, Dentsply, Milford, DE) at an intensity of 550 mW/cm2 was initiated after 50 infrared spectra had been recorded. The light exposure time was 40 s. Real-time IR spectra were continuously recorded for 600 s after light activation began. A time-based spectrum collector (Spectrum TimeBase, PerkinElmer) was used for continuous and automatic collection of spectra during polymerization. A minimum of three measurements (n = 3) were carried out for each adhesive formulation. Methacrylic double bond conversion was monitored by the band ratio profile-1637 cm−1 (C=C)/1608 cm−1 (phenyl). The average of the last 50 values of the time-based spectra is reported as the DC value. The maximum polymerization rate was determined using the maximum slope of the linear region of the DC vs. time plots [32].
2.6. Specimens preparation
The rectangular beam specimens were prepared by injecting the prepared resin into a glass-tubing mold (Fiber Optic Center, Inc., part no.: ST8100, New Bedford, MA) and light-curing them for 40 s at 23 ± 2 °C with an LED light curing unit (LED Curebox, 100 mW/cm2 irradiance, Proto-tech, Portland, OR). Disc specimens were prepared by injecting the resin into a standard aluminum hermetic lid (Tzero®, P/N:901600.901) and covering them with a mylar film to reduce oxygen exposure. The disc specimens were light-cured for 40 s at 23 ± 2 °C with a commercial visible-light-polymerization unit (Spectrum® 800, Dentsply, Milford, DE) at an intensity of 550 mW/cm2. The polymerized rectangular and disc specimens were stored in the dark at 23 ± 2 °C for at least 48 h before being used. The resultant rectangular beam specimens of cross section 1 mm × 1 mm and length 15 mm were used to determine water sorption and dynamic mechanical analysis, and the disc specimens were used to determine neutralization capacity and thermal analysis.
2.7. Water sorption and solubility of copolymer
The experimental protocol for the water sorption analyses has been reported [16,34]. In brief, water sorption and solubility were measured using rectangular beam specimens (1 mm × 1 mm × 15 mm). Five specimens were prepared for each adhesive formulation. Samples were weighed (m1) with a calibrated electronic balance (resolution of 0.01 mg, Mettler Toledo, XS 205 Dual range, Columbus, OH) and were immersed in distilled water to prewash for 7 days at 37 °C. Next, the specimens were dried in a vacuum oven in the presence of freshly dried silica gel at 37 °C; the specimens were removed every 24 h to determine the weight. This process was continued until a constant mass (m2) was recorded for each beam specimen. After prewashing, the dried specimens were immersed in distilled water and at fixed time intervals, they were removed, blotted to remove excess water, weighed (m3) and returned to the water until a constant weight was obtained. The values (%) for water sorption (Wsp) and solubility (Wsu) were calculated by the following equations:
| (2) |
| (3) |
2.8. pH-Neutralization measurements
The pH-neutralization measurements were performed with a Mettler Toledo (Columbus, OH) Accument® AP110 pH meter equipped with a micro-probe. Calibration was done using commercial buffers (Fisher Scientific, pH 4.01, 7.00, and 10.01/ 25 °C). Before the neutralization experiment, the disc specimens (~20 mg) were first prewashed in water at 37 °C for 7 days. The hydrated specimens were soaked into 2 mL LA solution (1 mM) and the pH of this solution was measured at fixed time intervals. Average and standard deviations were calculated from these pH measurements. Five specimens of each formulation were measured.
2.9. Dynamic mechanical analysis (DMA)
The dynamic mechanical properties of polymethacrylate-based dentin adhesives have been described [16,35]. In dynamic mechanical tests, a sinusoidal stress is applied, and the resultant strain is measured to obtain the storage, loss moduli, and tan δ. In the current study, DMA tests were performed using a TA instruments Q800 DMA (TA Instruments, New Castle, USA) with a three-point bending clamp. Rectangular beam specimens were used for DMA measurements and a minimum of three specimens were tested for each formulation. The following testing parameters were used: displacement amplitude of 15 µm, frequency of 1 Hz, and preload force of 0.01 N. In addition to this, temperature was ramped at the rate of 3 °C/min from 20 to 200 °C. The crosslinking density of copolymers was calculated based on the inverse ratio (ζ) of the modulus in the rubbery region to the temperature at which the modulus was measured [14].
2.10. Modulated DSC test (MDSC)
The thermal behavior in the Tg region was measured with a TA instruments model Q200 MDSC (New Castle, DE). The specimens were weighed (~20 mg) in aluminum DSC pans. The DSC cell was purged with nitrogen gas at 50 mL/min and the specimens were heated under nitrogen purge from −20 to 200 °C at 3 °C/min, with a modulation period of 60 s and amplitude of ±2 °C. The second scans were consistent with the first scan. Only the secondary cycle of heating was taken into account, and the results are shown as differential reversible heat flow vs. temperature graphs. The glass transition temperature (Tg) was determined as the position of the maximum on the derivative reversible heat flow versus temperature plots.
2.11. Statistical analysis
The results were analyzed statistically using one-way analysis of variance (ANOVA), together with Tukey’s test at α = 0.05 (Microcal Origin Version 8.0, Microcal Software Inc., Northampton, MA) to identify significant differences in the means.
3. Results
The water miscibility of the control and the experimental adhesives versus mole averaged log P is shown in Fig. 1. With increasing MEMA concentration from 0 to 55 wt%, the log P values decreased from 1.56 to 0.60 and the Wwm increased from 10.5 ± 0.1 to 54.2 ± 0.1 wt%.
Fig. 1.
Relation between the mole averaged log P value and water miscibility of adhesive formulations. Wwm stands for water miscibility.
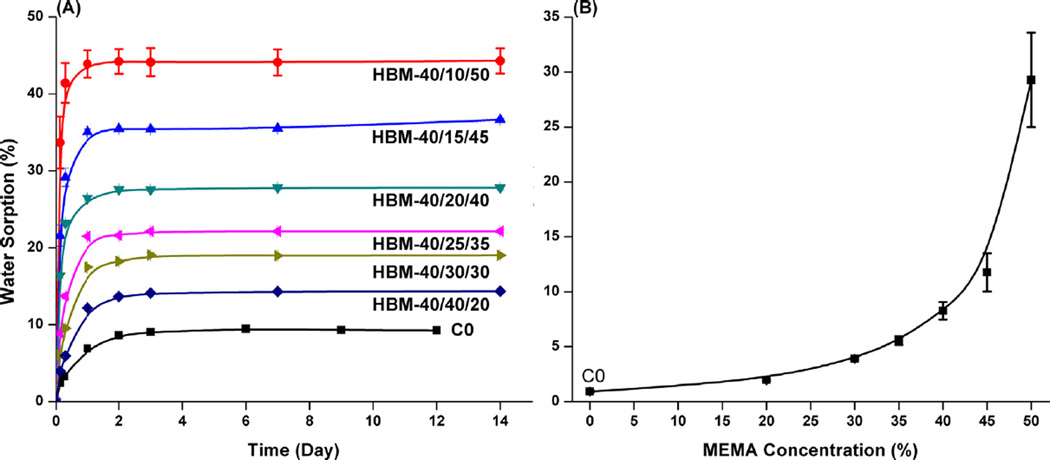
As can be seen in Fig. 2, the Wsp and Wsu of the adhesive formulations are enhanced with the increase in MEMA concentration. With the increase of MEMA from 0 to 50 wt%, the Wsp increased from about 9.5 to 45 wt% and the time required to reach balance is shortened from ~3 days to 1 day. At the same time, the Wsu increased from about 0.9 to 30 wt%.
Fig. 2.
Water sorption (A) and solubility (B) of dentin adhesive copolymers as a function of storage time (25 °C). (C0:HEMA/BisGMA 45/55, w/w).
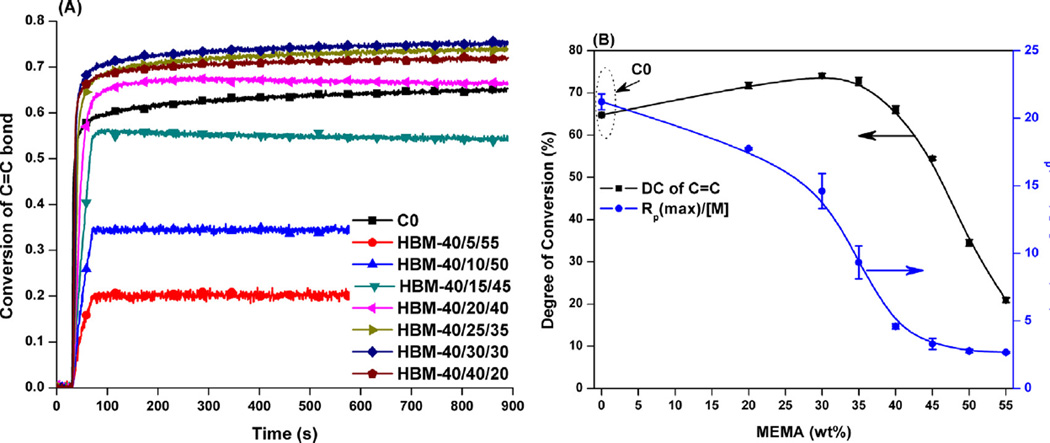
Real-time photopolymerization kinetic behavior of the control and experimental formulations are shown in Fig. 3 and the data are summarized in Table 1. When MEMA concentration was about 30 wt%, the DC (10 min) showed a maximum value and was about 74%. Further increase in MEMA concentration led to a decrease in DC. With an increase in MEMA concentration, the maximum polymerization rate decreased from 0.21 to 0.027 s−1.
Fig. 3.
Real-time conversion (A), degree of conversion and maximum polymerization rate vs. MEMA concentration (B). The adhesives were light-cured for 40 s at 23 ± 2 °C using a commercial visible light lamp (Spectrum® 800, Intensity is 550 mW/cm2).
Fig. 4A shows the pH values of 1 mM LA solution containing the control or experimental copolymer specimens versus storage time. When the BisGMA concentration was 55 wt% (C0), the pH value increased from 3.5 to about 3.75 and finally equilibrated at 3.7–3.8 after 90 days; the control copolymer specimens did not show neutralization capability. When MEMA concentrations were 50 or 45 wt%, the pH values increased to about 4.3 after 10 days and gradually reached 5.0 after 90 days. Decreasing MEMA concentrations from 40 to 35, or 30 wt% led to an increase in the pH values slowly in the initial 10 days and also led to a rapid increase during 15 to 60 days. Overall the pH values after 90 days were similar at about 6.0–6.5 when the MEMA concentrations were 40, 35 or 30 wt%. When MEMA concentration was 20 wt%, the pH value increased slightly in the initial 20 days, it accelerated to 6.0 after 60 days, and reached around 6.3 after 90 days. Fig. 4B shows the Δ pH values at 90 days. After 90 days, the pH value of LA solution containing the control copolymer specimens slightly increased about 0.3; however, the difference in the pH values of the LA solution containing the experimental copolymer specimens showed an obvious increase and the Δ pH values varied from about 1.5 to 3.0.
Fig. 4.
Plots of the pH values of 1 mM LA solution containing control and experimental copolymer specimens versus storage time (A), and the difference pH value after 90 days (B). (Volume of LA is 2 mL, initial pH is 3.50, and temperature is 23 ± 2 °C).
The dynamic mechanical properties of the amine-containing copolymers in dry conditions at various temperatures are shown in Fig. 5A and B. With the decrease in the BisGMA concentration, the rubbery moduli for the experimental copolymers decreased obviously and were significantly lower than that of the control (p < 0.05). The values of the tan Δ peak of experimental was significantly higher than that of the control (p < 0.05). Fig. 5C shows the inverse ratio (ζ) of the modulus in the rubbery region to the temperature at which the modulus was measured and the final pH values of LA solution over 90 days, which were plotted as a function of MEMA content (%). The value of ζ is considered to be inversely correlated to the crosslinking density of the copolymer networks, with higher ζ values corresponding to lower crosslinking density [14,36]. The experimental results showed higher ζ values than that of the control. It also showed that the pH values of LA solution had little dependence on the MEMA concentration in the experimental formulations, except for the HBM-40-15-45 samples with relatively low DC.
Fig. 5.
Representative storage modulus (A), tan δ (B) vs. temperature curves of the control and experimental adhesive copolymers, and plots of the inverse ratio of the modulus in the rubbery region to temperature and the pH value of LA solution measured after 90 days plotted as a function of MEMA concentration (%) in adhesive copolymers (C).
Fig. 6 shows the derivative reversible heat flow signals and the Tg of the control and experimental copolymers. With a decrease in BisGMA concentration from 40 to 15 wt%, the Tg decreased from about 110 to 65 °C. Compared with the control (135 °C), Tg of the experimental was significantly lower (p < 0.05). Fig. 6B shows the reverse correlation between the Tg and the ζ value of copolymers, which indicated that higher crosslinking density leads to a higher Tg.
Fig. 6.
Derivative reversible heat flow vs. temperature (A) and Tg vs. BisGMA concentration of control and experimental copolymers (B) HB-45/55 was used as the control.
4. Discussion
MEMA is a rather weaker base than DMAEMA and more stable in solution [37]. Its copolymer shows good biocompatibility, low toxicity, and has been used in drug delivery systems [30,31]. However, the neutralization behavior of its polymers has not been investigated when basic monomers are used as co-monomers in dentin adhesive formulations. In this study, we sought to characterize and optimize the neutralization capability of dentin adhesive copolymers using MEMA as co-monomer.
The oral environment is aqueous, so water miscibility of the basic monomer-containing liquid resin is an important aspect of compatibility with the formulation. MEMA (log P = 0.47) is more hydrophilic than BisGMA (log P = 5.09), so with the increase of MEMA concentration from 0 to 55 wt%, the water miscibility values increase. Log P values of the mixed co-monomers were calculated using Eq. (1) and found to vary from 1.56 to 0.60. The log P values decreased with the concentration of hydrophilic co-monomer MEMA, as expected.
The monomer-to-polymer conversion and polymerization behavior play important roles in determining the quality of the adhesive bond at the interface with dentin. When MEMA concentration was less than 35 wt%, the DC (10 min) was significantly higher than that of the control. With further increases in MEMA concentration, the DC showed a decreasing trend and was even as low as 20%. With the increasing MEMA concentration, the viscosities of experimental formulations also decreased considerably (unpublished data), which weakened the auto-acceleration effect. The decreasing maximum polymerization rates also supported the results. Many studies have shown that free radical polymerization of multifunctional methacrylates does not result in the complete conversion of C=C bonds and that the DCs are between 40 and 85% [10,16,38]. It has also been shown that the extent of cure (DC) influences the bulk physicochemical properties [35,39]. In the present study, with an increase in MEMA concentration from 20 to 40 wt%, the DCs were comparable or higher than that of the control. From the plot of polymer's solubility versus MEMA concentration (Fig. 2B), it can be observed that the higher the MEMA concentration, the higher the solubility. The experimental adhesive (30 wt% MEMA) with the highest DC (~74%, Wsu = 3.90 ± 0.11%) still possessed four times the solubility of the control (DC at ~64%, Wsu = 0.93 ± 0.13%). This could be due to the higher level of unreacted monomers or soluble oligomers when monomethacrylate MEMA was used in the adhesive formulation. From the water sorption experiment, the balanced Wsp showed a monotonic increasing trend with the increase of MEMA concentration. These results indicated that the hydrophilicity or hydrophobicity of monomers was the primary factor in determining the swelling behavior of copolymers. From Fig. 2A, it can be observed that the control copolymer was completely saturated within 3 days and, with the increase of MEMA concentration, the time to reach saturation was shortened to two or even one day.
Fig. 4 shows the pH trend of 1 mM LA containing copolymer specimens over storage time. In the control formulation, HEMA and BisGMA lacked basic moiety and did not neutralize the acid. After 90 days, the pH values increased to about 3.8, which may be related to the presence of the amine co-initiator, EDMAB. The initial burst increase in pH values can be observed in all of the experiments, which could be due to the adsorption of protons onto the surface of hydrated specimens. With the addition of MEMA in the formulations, the pH values of LA solution showed a gradually increasing trend, which shows that the copolymers are capable of neutralization. Despite differences in the pH values after 90 days, all of the pH values of LA solution containing experimental specimens exhibited an increasing trend. Therefore, the hypothesis (i) that the tertiary amine group built-in to the copolymer network can neutralize acid, was accepted.
When the copolymer specimens were soaked in LA solution, the neutralization ionized the tertiary amine groups on the copolymer chains and generated positive charges along the surface of copolymer sample quickly. It was observed that the pH value increased quickly from 3.5 to about 3.7, after the addition of copolymer specimens, in 3 h. After the tertiary amine-functionalized copolymers met LA, repulsions of the like charges could cause the reconfiguration of copolymer chains from a coiled status to an extended structure. However, due to the highly cross-linked network structure, even with a decrease in BisGMA concentration to 25 wt%, mobility of the backbone structure may still be very limited. When the functional groups exposed on the surface were neutralized by LA, a positively charged layer formed which severely restricted the diffusion of protons. In addition, the thickness of the polymer samples was in millimeters [34]. Therefore, it takes several weeks to reach plateau for the pH. The results indicated that the highly cross-linked network limited the mobility of amine groups and retarded the penetration of protons from the solution into the polymer network. The results of longer time to reach plateau indicated that the neutralization rate was depressed in highly cross-linked networks. When the MEMA concentration was 45 or 50 wt%, the pH value after 90 days reached about 4.8, which was due to the lower DC and higher solubility. During prewash with water, a large portion of the unpolymerized monomers and oligomers were removed, including the MEMA monomer. After prewash, the polymer could be porous, which could facilitate the penetration of LA molecules and protons.
Compared with our previous study, the DMAEMA monomer, which was evaluated by NMR experiments using LA as a probe, showed fast neutralization and buffering capacity [28]. In the present study, when the basic monomer MEMA was polymerized, the copolymers showed a relatively slow neutralization rate. Increasing MEMA concentration in formulation from 20 to 50 wt% led to the molar ratio of MEMA to LA in the solution increasing from 10:1 to 25:1. Several weeks were still required for the pH values to reach a plateau. Armes et al. have reported that the MEMA homopolymer’s pKa value was 4.9 by titration methodology [40]. In our group, the titration method was used to measure the pKa value of MEMA monomer (6.2) [26]. The relatively low pKa of MEMA homopolymer is due to the polymer chains resisting the local build-up of cationic charge density so that it becomes progressively harder to protonate the remaining neutral amine groups. In this work, the excess in MEMA concentration was enough to neutralize the LA in the solution. Therefore, the final pH values of LA solution were very close to the monomer’s pKa. As a result, the hypothesis (ii) that the highly cross-linked network structure does not retard the neutralization capability, was accepted.
Dynamic mechanical analysis (DMA) is suitable to characterize viscoelastic behavior of materials and has been suggested as a valuable tool for obtaining information such as crosslinking density and heterogeneity of polymer networks [41,42]. In the present study, the widths of the tan δ curves indicated that the glass transition occurred over a wide temperature range (Fig. 5B). With the decrease in BisGMA concentration, the intensity of tan δ peak increased, which indicated that the mobility of copolymer chains was improved. The ratio of rubbery modulus to the absolute temperature at which that modulus was measured, ζ, is inversely related to the crosslinking density of polymer network [14,36]. By this measure, the experimental formulations showed lower crosslink densities than that of the control. When BisGMA concentration was between 20 and 40 wt%, the equilibrium pH values of LA solution were similar at about 6.0–6.5. Combined with the neutralization results, it can be concluded that the neutralization capacity was mainly determined by the tertiary amine comonomer and not directly dependent on the crosslinking density of copolymers.
The modulated temperature DSC (MDSC) method has been used to obtain the thermal properties and to provide related information on the degree of crosslinking in polymers [43]. Fig. 6 shows the results obtained in the MDSC analysis for the control and experimental copolymers. From the DC obtained by the FTIR method, the specimens were only partly cured with the DC at about 60–70%. As the sample was heated, it could reach the glass transition region and the thermal energy provided sufficient molecular mobility to facilitate continuation of the curing process, causing a shift in the transition region. The non-reversible components showed an exothermic peak arising from the simultaneous thermal curing (not shown). As the temperature was increased still further, the reaction finally ceased as the system reached full cure. MEMA is monomethacrylate (only one C=C double bond in the monomeric structure), so a loosely cross-linked network formed after the light-irradiation with the increasing of MEMA concentration. The Tg showed an inverse correlation with the ζ values, which indicated the higher crosslinking density and the higher transition temperature (Fig. 6B). This result was consistent with that determined by DMA.
5. Conclusion
Copolymers with neutralization capacity are a promising strategy for reducing LA-induced demineralization of the tooth at the interface between the adhesive and dentin. In the present work, tertiary amine-functionalized dentin adhesive copolymers have been prepared by using MEMA as a functional co-monomer. The MEMA-functionalized copolymers showed a neutralization capability in 1 mM LA solution, and the final pH values were about 6.0–6.5, except for the formulations with relatively low DC (<50%). Considering the neutralization capacity, polymerization behavior and mechanical properties, the MEMA concentration should be between 20 and 40 wt%. The crosslinking density of the copolymer networks did not show a significant effect on the neutralization capacity. Crosslinking density was related to neutralization rate, i.e., the lower crosslinking density, the faster neutralization rate. The decreased crosslink density and increased water sorption of MEMA-containing copolymers would potentially limit the durability of these materials under clinical conditions. Further investigation is required to balance the functionality and stability of amine-containing copolymers.
Acknowledgments
This investigation was supported by research grants R01DE022054 and 3R01DE022054-04S1 from the National Institute of Dental and Craniofacial Research, National Institutes of Health, Bethesda, Maryland, United States of America.
Footnotes
Conflict of interest
The authors declare no potential conflicts of interest with respect to the authorship and/or publication of this article.
References
- 1.Kingman A, Hyman J, Masten SA, Jayaram B, Smith C, Eichmiller F, et al. Bisphenol A and other compounds in human saliva and urine associated with the placement of composite restorations. J. Am. Dent. Assoc. 2012;143:1292–1302. doi: 10.14219/jada.archive.2012.0090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.DeRouen TA, Martin MD, Leroux BG, Townes BD, Woods JS, Leitao J, et al. Neurobehavioral effects of dental amalgam in children—a randomized clinical trial. Jama-J. Am. Med. Assoc. 2006;295:1784–1792. doi: 10.1001/jama.295.15.1784. [DOI] [PubMed] [Google Scholar]
- 3.Opdam NJM, Bronkhorst EM, Loomans BAC, Huysmans MCDNJM. 12-year survival of composite vs. amalgam restorations. J. Dent. Res. 2010;89:1063–1067. doi: 10.1177/0022034510376071. [DOI] [PubMed] [Google Scholar]
- 4.Li Y, Carrera C, Chen R, Li J, Lenton P, Rudney JD, et al. Degradation in the dentin-composite interface subjected to multi-species biofilm challenges. Acta Biomater. 2014;10:375–383. doi: 10.1016/j.actbio.2013.08.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ferracane JL. Hygroscopic and hydrolytic effects in dental polymer networks. Dent. Mater. 2006;22:211–222. doi: 10.1016/j.dental.2005.05.005. [DOI] [PubMed] [Google Scholar]
- 6.Spencer P, Ye Q, Park J, Topp EM, Misra A, Marangos O, et al. Adhesive/dentin interface: the weak link in the composite restoration. Ann. Biomed. Eng. 2010;38:1989–2003. doi: 10.1007/s10439-010-9969-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kostoryz EL, Dharmala K, Ye Q, Wang Y, Huber J, Park JG, et al. Enzymatic biodegradation of HEMA/BisGMA adhesives formulated with different water content. J. Biomed. Mater. Res. Part B-Appl. Biomater. 2009;88B:394–401. doi: 10.1002/jbm.b.31095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Spencer P, Ye Q, Misra A, Goncalves SE, Laurence JS. Proteins, pathogens, and failure at the composite-tooth interface. J. Dent. Res. 2014 doi: 10.1177/0022034514550039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Takahashi N, Nyvad B. The role of bacteria in the caries process: ecological perspectives. J. Dent. Res. 2011;90:294–303. doi: 10.1177/0022034510379602. [DOI] [PubMed] [Google Scholar]
- 10.Park J, Ye Q, Singh V, Kieweg SL, Misra A, Spencer P. Synthesis and evaluation of novel dental monomer with branched aromatic carboxylic acid group. J. Biomed. Mater. Res. Part B-Appl. Biomater. 2012;100B:569–576. doi: 10.1002/jbm.b.31987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Park J, Eslick J, Ye Q, Misra A, Spencer P. The influence of chemical structure on the properties in methacrylate-based dentin adhesives. Dent. Mater. 2011;27:1086–1093. doi: 10.1016/j.dental.2011.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Moszner N. New monomers for dental application. Macromol. Symp. 2004;217:63–75. [Google Scholar]
- 13.Moszner N, Hirt T. New polymer-chemical developments in clinical dental polymer materials: enamel-dentin adhesives and restorative composites. J. Polym. Sci. Part A-Polym. Chem. 2012;50:4369–4402. [Google Scholar]
- 14.Park JG, Ye Q, Topp EM, Misra A, Spencer P. Water sorption and dynamic mechanical properties of dentin adhesives with a urethane-based multifunctional methacrylate monomer. Dent. Mater. 2009;25:1569–1575. doi: 10.1016/j.dental.2009.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ge XP, Ye Q, Song LY, Misra A, Spencer P. Synthesis and evaluation of novel siloxane-methacrylate monomers used as dentin adhesives. Dent. Mater. 2014;30:1073–1087. doi: 10.1016/j.dental.2014.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Song L, Ye Q, Ge X, Misra A, Laurence JS, Berrie CL, et al. Synthesis and evaluation of novel dental monomer with branched carboxyl acid group. J. Biomed. Mater. Res. B Appl. Biomater. 2014;102:1473–1484. doi: 10.1002/jbm.b.33126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Park JG, Ye Q, Topp EM, Spencer P. Enzyme-catalyzed hydrolysis of dentin adhesives containing a new urethane-based trimethacrylate monomer. J. Biomed. Mater. Res. Part B-Appl. Biomater. 2009;91B:562–571. doi: 10.1002/jbm.b.31430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Finer Y, Santerre JP. Salivary esterase activity and its association with the biodegradation of dental composites. J. Dent. Res. 2004;83:22–26. doi: 10.1177/154405910408300105. [DOI] [PubMed] [Google Scholar]
- 19.Hagio M, Kawaguchi M, Motokawa W, Miyazaki K. Degradation of methacrylate monomers in human saliva. Dent. Mater. J. 2006;25:241–246. doi: 10.4012/dmj.25.241. [DOI] [PubMed] [Google Scholar]
- 20.Yourtee DM, Smith RE, Russo KA, Burmaster S, Cannon JM, Eick JD, et al. The stability of methacrylate biomaterials when enzyme challenged: kinetic and systematic evaluations. J. Biomed. Mater. Res. 2001;57:522–531. doi: 10.1002/1097-4636(20011215)57:4<522::aid-jbm1198>3.0.co;2-9. [DOI] [PubMed] [Google Scholar]
- 21.Sideridou I, Tserki V, Papanastasiou G. Study of water sorption, solubility and modulus of elasticity of light-cured dimethacrylate-based dental resins. Biomaterials. 2003;24:655–665. doi: 10.1016/s0142-9612(02)00380-0. [DOI] [PubMed] [Google Scholar]
- 22.Ferracane JL. Water sorption and solubility of experimental dental composites. Abstr. Pap. Am. Chem. Soc. 1997;214 142–POLY. [Google Scholar]
- 23.Truffier-Boutry D, Demoustier-Champagne S, Devaux J, Biebuyck JJ, Mestdagh M, Larbanois P, et al. A physico-chemical explanation of the post-polymerization shrinkage in dental resins. Dent. Mater. 2006;22:405–412. doi: 10.1016/j.dental.2005.04.030. [DOI] [PubMed] [Google Scholar]
- 24.Sideridou I, Tserki V, Papanastasiou G. Effect of chemical structure on degree of conversion in light-cured dimethacrylate-based dental resins. Biomaterials. 2002;23:1819–1829. doi: 10.1016/s0142-9612(01)00308-8. [DOI] [PubMed] [Google Scholar]
- 25.Toledano M, Yamauti M, Osorio E, Osorio R. Zinc-inhibited MMP-mediated collagen degradation after different dentine demineralization procedures. Caries Res. 2012;46:201–207. doi: 10.1159/000337315. [DOI] [PubMed] [Google Scholar]
- 26.Laurence JS, Nelson BN, Ye Q, Park J, Spencer P. Characterization of acid-neutralizing basic monomers in co-solvent systems by NMR. Int. J. Polym. Mater. Polym. Biomater. 2014;63:361–367. doi: 10.1080/00914037.2013.845192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Liu SY, Weaver JVM, Tang YQ, Billingham NC, Armes SP, Tribe K. Synthesis of shell cross-linked micelles with pH-responsive cores using ABC triblock copolymers. Macromolecules. 2002;35:6121–6131. [Google Scholar]
- 28.Park J, Ye Q, Spencer P, Laurence JS. Determination of neutralization capacity and stability of a basic methacrylate monomer using NMR. Int. J. Polym. Mater. 2012;61:144–153. doi: 10.1080/00914037.2011.574660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Taktak FF, Butun V. Synthesis and physical gels of pH- and thermo-responsive tertiary amine methacrylate based ABA triblock copolymers and drug release studies. Polymer. 2010;51:3618–3626. [Google Scholar]
- 30.Van Overstraeten-Schlogel N, Shim YH, Tevel V, Piel G, Piette J, Dubois P, et al. Assessment of new biocompatible Poly(N-(morpholino) ethyl methacrylate)-based copolymers by transfection of immortalized keratinocytes. Drug Deliv. 2012;19:112–122. doi: 10.3109/10717544.2011.649219. [DOI] [PubMed] [Google Scholar]
- 31.Reschner A, Shim YH, Dubois P, Delvenne P, Evrard B, Marcelis L, et al. Evaluation of a new biocompatible poly(N-(morpholino ethyl methacrylate)-based copolymer for the delivery of ruthenium oligonucleotides, targeting HPV16 E6 oncogene. J. Biomed. Nanotechnol. 2013;9:1432–1440. doi: 10.1166/jbn.2013.1634. [DOI] [PubMed] [Google Scholar]
- 32.Guo X, Wang Y, Spencer P, Ye Q, Yao X. Effects of water content and initiator composition on photopolymerization of a model BisGMA/HEMA resin. Dent. Mater. 2008;24:824–831. doi: 10.1016/j.dental.2007.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ye Q, Park J, Topp E, Spencer P. Effect of photoinitiators on the in vitro performance of a dentin adhesive exposed to simulated oral environment. Dent. Mater. 2009;25:452–458. doi: 10.1016/j.dental.2008.09.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Parthasarathy R, Misra A, Park J, Ye Q, Spencer P. Diffusion coefficients of water and leachables in methacrylate-based crosslinked polymers using absorption experiments. J. Mater. Sci. Mater. Med. 2012;23:1157–1172. doi: 10.1007/s10856-012-4595-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Park J, Ye Q, Topp EM, Misra A, Kieweg SL, Spencer P. Effect of photoinitiator system and water content on dynamic mechanical properties of a light-cured bisGMA/HEMA dental resin. J. Biomed. Mater. Res. A. 2010;93A:1245–1251. doi: 10.1002/jbm.a.32617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Podgorski M, Matynia T. Network structure/mechanical property relationship in multimethacrylates-derivatives of nadic anhydride. J. Appl. Polym. Sci. 2008;109:2624–2635. [Google Scholar]
- 37.van de Wetering P, Zuidam NJ, van Steenbergen MJ, van der Houwen OAGJ, Underberg WJM, Hennink WE. A mechanistic study of the hydrolytic stability of poly(2-(dimethylamino) ethyl methacrylate) Macromolecules. 1998;31:8063–8068. [Google Scholar]
- 38.Santerre JP, Shajii L, Leung BW. Relation of dental composite formulations to their degradation and the release of hydrolyzed polymeric-resin-derived products. Crit. Rev. Oral Biol. Med. 2001;12:136–151. doi: 10.1177/10454411010120020401. [DOI] [PubMed] [Google Scholar]
- 39.Lovell LG, Lu H, Elliott JE, Stansbury JW, Bowman CN. The effect of cure rate on the mechanical properties of dental resins. Dent. Mater. 2001;17:504–511. doi: 10.1016/s0109-5641(01)00010-0. [DOI] [PubMed] [Google Scholar]
- 40.Butun V, Armes SP, Billingham NC. Synthesis and aqueous solution properties of near-monodisperse tertiary amine methacrylate homopolymers and diblock copolymers. Polymer. 2001;42:5993–6008. [Google Scholar]
- 41.Sideridou ID, Karabela MM, Vouvoudi EC. Dynamic thermomechanical properties and sorption characteristics of two commercial light cured dental resin composites. Dent. Mater. 2008;24:737–743. doi: 10.1016/j.dental.2007.08.004. [DOI] [PubMed] [Google Scholar]
- 42.Mesquita V, Geis-Gerstorfer J. Influence of temperature on the visco-elastic properties of direct and indirect dental composite resins. Dent. Mater. 2008;24:623–632. doi: 10.1016/j.dental.2007.06.025. [DOI] [PubMed] [Google Scholar]
- 43.Ye Q, Spencer P, Wang Y, Misra A. Relationship of solvent to the photopolymerization process, properties, and structure in model dentin adhesives. J. Biomed. Mater. Res. A. 2007;80A:342–350. doi: 10.1002/jbm.a.30890. [DOI] [PMC free article] [PubMed] [Google Scholar]