Abstract
Background
Treatment options for women presenting with triple negative breast cancer (TNBC) are limited due to the lack of a therapeutic target and as a result, are managed with standard chemotherapy such as paclitaxel (Taxol®).
Following chemotherapy, the ideal tumour response is apoptotic cell death. Post-chemotherapy, cells can maintain viability by undergoing viable cellular responses such as cellular senescence, generating secretomes which can directly enhance the malignant phenotype.
Scope of Review
How tumour cells retain viability in response to chemotherapeutic engagement is discussed. In addition we discuss the implications of this retained tumour cell viability in the context of the development of recurrent and metastatic TNBC disease.
Current adjuvant and neo-adjuvant treatments available and the novel potential therapies that are being researched are also reviewed.
Major conclusions
Cellular senescence and cytoprotective autophagy are potential mechanisms of chemoresistance in TNBC. These two non-apoptotic outcomes in response to chemotherapy are inextricably linked and are neglected outcomes of investigation in the chemotherapeutic arena. Cellular fate assessments may therefore have the potential to predict TNBC patient outcome.
General Significance
Focusing on the fact that cancer cells can bypass the desired cellular apoptotic response to chemotherapy through cellular senescence and cytoprotective autophagy will highlight the importance of targeting non-apoptotic survival pathways to enhance chemotherapeutic efficacy.
Keywords: Triple negative breast cancer (TNBC), Cellular fates, Cellular senescence, Chemoresistance, Hypoxia
Highlights
-
•
How tumour cells retain viability after chemotherapeutic engagement is discussed.
-
•
The implications of tumour cell viability in TNBC are discussed.
-
•
Chemotherapy and novel therapies that are currently being researched are reviewed.
-
•
Chemoresistance mechanisms and hypoxia’s role in TNBC are outlined.
-
•
Senescence and autophagy are potential mechanisms of chemoresistance in TNBC.
1. Introduction
Triple negative breast cancers (TNBCs) are a specific subtype of epithelial breast tumours that are immunohistochemically negative for the protein expression of the oestrogen receptor (ER), the progesterone receptor (PR) and lack overexpression/gene amplification of HER2 [1].
Approximately, 10–14% of breast cancers are triple negative and the methods used to perform a diagnosis generally dictate the frequency [2]. Pre-analytic and analytic issues such as delay from tissue collection to fixation (risk of cold ischemia) and assay reproducibility (assay validation and standardisation) can impact on whether a tumour is assigned as Triple Negative or not [3].
1.1. Immunohistochemical testing
The characterisation and measurement of the oestrogen receptor (ER) protein was first described by Jensen et al., in 1967 and in 1973, McGuire et al., described the dextran-coated charcoal (DCC) biochemical assay to quantify the ER protein [4], [5]. It was also then postulated that detection of the oestrogen receptor may predict response to endocrine therapy. Immunohistochemical (IHC) methodologies have been used since the early 1990s and remain by far the most common type of assay worldwide. However, there is an inadequate standardisation of the IHC methodology. A recent “ring study” showed an initial ER testing concordance between two central laboratories at just 85%, which was fully resolved once both laboratories adopted the same ER assay methods [6].
Recent misclassification (false negative) reports have raised awareness concerning the limitations of immunohistochemistry (IHC) in the assessment of the oestrogen receptor (ER) in breast cancer. Hede et al. (2008) describe the incident in Canada where there was a 40% misclassification rate between local and central laboratories highlighting the current limitations associated with ER protein testing [7].
Rimm et al., 2011 describe a potential method for standardising ER measurement “on a slide” using a quantitative immunofluorescence (QIF) assay [8]. Moreover, Rimm’s data also show that an assessment of the intensity threshold by using this assay on two independent cohorts resulted in discordance in the 10% to 20% range with current IHC methods. Overall, the findings of the study were that (a) the threshold of immunoreactivity appears to be more important than the percentage positivity in the generation of discordant or false-negative assays and that (b) the standardisation method by using the QIF assay appeared to be more sensitive than the traditional IHC assay. This was despite the fact that the same antibody was used for the detection of ER (1D5). Rimm et al., concluded that (a) intensity threshold, in other words, what actually constitutes a “positive” nucleus is the key determinant of variable scoring that needs to be controlled to correctly define ER characterisation and that (b) the QIF assay is more sensitive than routine IHC [8].
The American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) issued guidelines to address this issue of discordance in ER assessment. An IHC cutoff of 1% positive nuclei is now used to define a tumour as ER positive compared to the previous cut off of 10% positive nuclei [9]. Few ER-positive tumours have true IHC staining between 1% and 10% [10]. However, false-negative ER results can still occur even when using a validated and reproducible IHC assay and an imaging analysis system is unlikely to improve detection of tumours with low levels of ER. This justifies the use of a low IHC cutoff (1%) for clinical use to minimise this risk of false-negative ER. In the absence of a linear correlation between ER expression by IHC and clinical outcome, available data suggest that a qualitative assessment (ER positive v ER negative) is more critical than a quantitative IHC assay [3].
Accurate testing for the HER2/neu gene overexpression is also an issue where false-positives using IHC can occur. In 1999, Jacobs et al., highlighted this with the HercepTest, where tumours identified as HER2 positive by this test were subsequently described as negative using FISH and other IHC methods using the same antibody [11]. The "gold standard" used to identify these false-positive results has been tested for HER2/neu gene amplification by FISH. This is due to the greater specificity and sensitivity of FISH when either test is compared with HER2/neu overexpression as determined by Northern and Western blot analyses [12], [13].
Studies have also shown that the problem of false-positive HER2 results, involve IHC techniques most significantly when the result is 2 + [14]. Specifically, as few as 17% of 2 + HercepTest carcinomas demonstrate gene amplification by FISH [15]. The overwhelming majority of 3 + positives demonstrate HER2 gene amplification. However, in a study by Mass et al., 2000, 11% of 3 + positive tumours did not demonstrate gene amplification by FISH. This indicates that false-positive results are a potential problem in this group as well [16]. False-negative results, in other words, IHC-negative/FISH-positive carcinomas, are rare in the 0–1 + IHC groupings.
The ASCO and the College of American Pathologists (CAP) recently updated HER2 testing guidelines to improve the accuracy of HER2 testing and its utility as a predictive marker in invasive breast cancer [3]. A systematic review was conducted to identify areas requiring clarification and to improve the accuracy of HER2 testing by immunohistochemistry (IHC) or in situ hybridisation (ISH). The guidelines have been published jointly in the Journal of Clinical Oncology and the Archives of Pathology & Laboratory Medicine. These recommendations will improve the analytical validity of HER2 testing, its clinical utility and the communication among health-care providers [17].
Recommendations include:
-
•
That HER2 status (HER2 negative or positive) be determined in all patients with invasive (early stage or recurrence) breast cancer on the basis of one or more HER2 test results (negative, equivocal, or positive).
-
•
That testing criteria define HER2-positive status when there is evidence of protein overexpression (IHC) or gene amplification (HER2 copy number or HER2/CEP17 ratio by ISH based on counting at least 20 cells within the area).
-
•
That if results are equivocal (revised criteria) reflex testing should be performed using an alternative assay (IHC or ISH).
-
•
That repeat testing should be considered if results seem discordant with other histopathological findings.
-
•
Those laboratories should demonstrate high concordance with a validated HER2 test on a sufficiently large and representative set of specimens.
-
•
That HER2 testing must be performed in a laboratory accredited by CAP or another accrediting entity.
1.2. Epidemiology
TNBC’s are typically of the histological ductal type with a high mitotic rate, increased lymphocytic infiltrate, high grade and large tumour size. In addition, TNBC patients commonly present with early visceral metastases and have lymph node involvement at the time of diagnosis [18], [19]. Approximately 15% of TNBC patients will develop brain metastases with autopsy studies reporting a 30% rate [20]. This aggressive metastatic cancer contributes to the overall shortened survival of patients with TNBC [21].
In relation to age, TNBC’s affect younger women more frequently and are more prevalent in African-Americans who are three times more likely to present with TNBC than Caucasian women [22]. In fact 39% of all African American premenopausal women diagnosed with breast cancer are diagnosed with TNBC [23]. The prevalence of TNBC in this same age group in non–African American women is demonstrably less approximating at 15%. These ethnic and/or menopausal differences are not seen in either, the ER +/HER2 + breast cancer subgroup or the ER +/HER2– subgroup [23]. Women presenting with TNBC also have a higher rate of distant recurrence and a poorer prognosis than women with other subtypes of breast cancer. Paradoxically, this higher rate of distant recurrence occurs despite these women having initial higher response rates to neoadjuvant chemotherapy [24].
1.3. TNBC molecular and gene expression subtypes; a heterogenous cancer
As a result of gene expression profiling, breast cancer has been classified into five distinct molecular subtypes, namely; normal-like, basal, luminal A & B and HER-2 enriched [25]. A Claudin-low sub-type is another described molecular subtype referring to tumours showing features of mesenchymal and mammary stem cells [26]. Interestingly, tumour cells that survive chemotherapy show features similar to mesenchymal claudin-low tumour cells [27].
In relation to TNBC’s, 75% fall into the basal-subtype and express genes normally associated with normal basal like myoepithelial cells of the breast ductal and lobular system, such as the epidermal growth factor receptor (EGFR) [28]. Basal-like breast cancers tend to express cytokeratins associated with basal types of cancers, as they arise from the outer basal layer. It has also been reported that almost 82% of basal-like breast cancers express p53 compared with 13% in the luminal A subgroup [29]. Useful immunohistochemical markers for characterising basal like carcinomas include CK5, CK6, CK14, CK8/CK18, p63, P-cadherin, vimentin, epidermal growth factor receptor 1 (EGFR1), c-kit, and other growth factors such as vascular-endothelial growth factor (VEGF) and the insulin-like growth factor receptor (IGFR) [28].
Lehmann et al., 2011 have analysed the gene-expression profiles from 21 breast cancer data sets of which 587 were TNBC [30]. Six TNBC subtypes were shown to have unique gene expression profiles, namely basal-like (BL1 & BL20), an immunomodulatory (IM), a mesenchymal (M), a mesenchymal stem-like (MSL) and a luminal androgen receptor (LAR) subtype. Identification of TNBC cell line models representative of these subtypes was performed using further gene expression (GE) analysis. Predicted “driver” signalling pathways were pharmacologically targeted in these cell line models as proof of concept that the analysis of distinct GE signatures had the potential to inform patient therapy decision.
In general, the BL1 and BL2 TNBC subtypes had higher expression of cell cycle and DNA damage response genes. Moreover, representative cell lines preferentially responded to cisplatin. In the M and MSL subtypes, these were enriched in GE profiles representing the epithelial-mesenchymal transition (EMT), and growth factor pathways with cell models responding to NVP-BEZ235 (a PI3K/mTOR inhibitor) and dasatinib (an abl/src inhibitor). The LAR subtype included patients with a decreased relapse-free survival and was characterised by androgen receptor (AR) signalling. LAR cell lines were uniquely sensitive to the AR antagonist bicalutamide. To the future, the identification and characterisation of these diverse TNBC subtypes and the molecular drivers in corresponding cell line models has the potential to provide great insight into the heterogeneity of this disease and to provide preclinical platforms for the development of effective treatment.
2. Adjuvant & neoadjuvant chemotherapy used in TNBC
It is well established that TNBC is an aggressive group of breast cancer subtypes despite having a good initial response to chemotherapy. It is also well established that patients with residual TNBC disease post neo-adjuvant chemotherapy have a worse prognosis than those presenting with non-TNBC [2]. Importantly, there is no preferred standard chemotherapy for these patients and typically tumour size, lymph node status, grade, overall performance status and the presence or absence of medical co-morbidities will determine the regimens used. For women presenting with triple negative breast cancer (TNBC), it is established that in the absence of ER, PR and HER-2, endocrine therapies such as Tamoxifen and aromatase inhibitors and HER-2 directed therapies such as Trastazumab and Lapatinib are not efficacious. Triple negative breast cancers seem to be particularly chemo-sensitive to anthracyclines and taxanes which are part of the standard therapy used for high risk patients, for example those patients with node positive disease. In spite of this, however, there is an overall poorer survival [31].
Although TNBCs are generally very susceptible to chemotherapy initially, early complete response (CR) does not correlate with overall survival. Specifically, the risk of relapse for TNBC patients in the first 3–5 years is significantly higher than for women presenting with hormone positive breast cancer [32], [33]. Clinically, this makes it particularly challenging to find the optimal chemotherapy which will result in a longer metastasis free and overall survival for these women.
Neoadjuvant chemotherapy is clinically where most of the knowledge about chemosensitivity has arisen. Specifically, there is a higher pathologic complete response, (PCR) related to neoadjuvant anthracycline based chemotherapy in TNBC compared to luminal non-TNBC subtypes. However, despite initial chemosensitivity, metastatic relapse paradoxically appears to occur at higher rates for TNBC tumours [2].
In the adjuvant therapy space, the principles for nonTNBC apply equally to TNBC. These therefore can include:-
-
1)
Doxorubicin or Epirubicin (Anthracyclines)
-
2)
AC (Doxorubicin and Cyclophosphamide)
-
3)
Paclitaxel and Docetaxel (often weekly and often every third week respectively), frequently used in combination with Cyclophosphosphamide or 5-Fluorouracil. For example, ACT (doxorubicin, cyclophosphamide, paclitaxel), TC (docetaxel and cyclophosphamide), TFEC (docetaxel, 5-Fluorouracil, epirubicin, cyclophosphamide)
-
4)
CMF (cyclophosphamide, methotrexate, 5-Fluorouracil)
-
5)
Antimetabolites such as Gemcitabine or Capecitabine and other microtubule inhibitors or stabilisers such as Vinorelbine.
-
6)
Non-taxane anti-tubulin agents such as Eribulin and Ixabepilone, which are associated with limited clinical efficacy in TNBC as opposed to non-TNBC presentations.
3. Novel potential therapies for TNBC
TNBCs can arise in BRCA1 mutation carriers and have gene expression profiles similar to those of BRCA1-deficient tumours [19]. Moreover, BRCA1-associated breast cancer appears to cluster in the basal-like subtype [34]. BRCA1 mutation carriers often display basal like gene expression profiles, and there is increasing evidence that a TNBC basal like subtype develops mainly through a BRCA1-related pathway, resulting in increased genomic instability [35]. For BRCA1 mutation carriers, the incidence of TNBC approximates to 70% [36], [37], although the incidence of BRCA mutations in TNBC can vary from 16% to 42% [38].
Even though a patient may lack the BRCA-1 somatic mutation, sporadically arising basal-like cancers often display a dysfunctional BRCA-1 pathway [39]. TNBC’s and BRCA1-deficient tumours share certain histological features including genomic instability, DNA repair defects [39] and mutations in p53, which disrupt apoptosis and which are associated with a poor prognosis [40]. Importantly, BRCA1 plays an important role in DNA double-strand break repair, contributing to the maintenance of DNA stability [41].
3.1. Poly ADP-ribose polymerase (PARP) inhibition
Poly ADP-ribose polymerase (PARP) enzymes are critical for the appropriate processing and repair of DNA breaks [42]. Tumour cell lines lacking functional BRCA1 or BRCA2 are sensitive to PARP inhibitors in preclinical studies [43]. Clinical trials using both PARP inhibitors and DNA-damaging agents in TNBC have shown some promising results in BRCA1/2-mutant tumours [44]. Specifically, the PARP inhibitor olaparib has been used in the treatment of patients with BRCA 1 and 2 deficient tumours: − cancers which share similar features to sporadic TNBC, where some efficacy has been shown in a Phase II trial in BRCA-deficient recurrent advanced breast cancers, with enrolled patients having previously been given three previous chemotherapy regimens prior to the trial. Clinically, the objective response rates were 41% (n = 11) and 22% (n = 6) for patients receiving Olaparib 400 mg twice daily and Olaparib 100 mg twice daily, respectively. Moreover, the median progression-free survival in the 400 mg BD cohort was 5.7 months, compared with 3.8 months in the 100 mg twice daily cohort [45], [46].However, clinical data from another similarly sized cohort of tumours (n = 15) failed to demonstrate any response in BRCA mutation-negative TNBC patients [47].
The PARP inhibitor Iniparib versus placebo with Gemcitabine and Carboplatin has demonstrated significant improvement in patients with pre-treated TNBC with an overall response rate (ORR) of 52% versus 33%, progression free survival (PFS) of 5.9 versus 3.6 months and overall survival (OS) of 12.3 versus 7.7 months [48]. However, results of a multicentre, phase III trial assessing the same iniparib combination in advanced TNBC failed to meet the primary study end points with a PFS of 4.1 versus 5.1 months and overall survival of 11.1 versus 11.8 months [49].
The combination of Veliparib with the oral alkylating agent temozolamide, resulted in an improved response, bearing in mind that 20% of the patients in this trial had BRCA germ line mutations. However, this Phase II trial did not include a TNBC subgroup analysis [50].
Overall, PARP inhibitors have shown little benefit in unselected TNBC populations, either as a single regimen [46] or used in combination with other standard chemotherapeutics [51], [52], although BRCA-selected patient groups may potentially benefit from ongoing PARP development [53].
3.2. Platinum salts
Three randomised trials have investigated the clinical efficacy of using platinum based regimens for the treatment of TNBC, [54], [55], [56] with further randomised trials evaluating the addition of platinum derivatives to standard adjuvant therapy (NCT01150513 & NCT01216111). NCR01378533 is an adjuvant phase III trial comparing a dose dense anthracycline–taxane regimen to dose dense paclitaxel and carboplatin. The combination of a platinum based agent with the PARP inhibitor PF-01367338 is also currently being evaluated following preoperative chemotherapy in patients with TNBC or BRCA1/2-associated BC (NCT01074970).
In the metastatic and second line setting, the addition of cisplatin to metronomic methotrexate and cyclophosphamide has resulted in an improvement in the median time-to-progression (TTP) of 6 months (from 7 to 13 months) and OS of 4 months (from 12 to 16 months) [54].
Carey et al., 2008 have also shown that when carboplatin is added to the single-agent cetuximab in pretreated advanced TNBC patients, the overall response rate (ORR) is 17% versus 6% [55]. Cetuximab plus carboplatin produced partial responses in 71% (13/18) patients compared to cetuximab monotherapy; 2 (6) of 31% patients and stable disease lasting ≥ 6 months in 6 additional patients (9%) [57].
However, the addition of cisplatin to the anthracycline-taxane based regimen showed no improvement in pathological complete response (pCR) rates or rates of breast conserving surgery in the basal like TNBC cohort of patients enrolled in The Spanish Breast Cancer Research Group (GEICAM) 2006 phase II RCT [56].
In phase II trials [56], [58], the combination of weekly cisplatin added to weekly epirubicin and paclitaxel, produced a pCR rate of 62% and 5-year disease-free survival (DFS) and OS values of 76% and 89%, respectively. In addition, Liu et al., 2013 have undertaken a meta analysis of platinum-based chemotherapy in TNBC, which included 7 studies and 717 patients of which 31% were TNBC [59]. These results showed that in the neo-adjuvant setting, the clinical complete response (cCR) rate and the pathological complete response (pCR) rates were significantly higher for the TNBC group compared with the non-TNBC group (OR, 2.68; 95% CI, 1.69-6.57; p = 0.03 and OR, 2.89; 95% CI, 1.28, 6.53; p = 0.01, respectively). However, in advanced/metastatic breast cancers, the cCR, partial response (PR) and the disease control rates for the TNBC group were not significantly different compared with the non-TNBC group. In the same study [59], the 6-month progression-free survival (PFS) rate for the TNBC group was higher than that of the non-TNBC group in all patients (OR, 1.81; 95% CI, 1.11-2.96; P = 0.02). Nonetheless, the 1- and 2-year PFS rates were not significantly different between the TNBC and non-TNBC group (OR, 1.42; 95% CI, 0.69-2.92; P = 0.35 and OR, 1.11; 95% CI, 0.35-3.52; P = 0.85, respectively). Also, there was no significant difference in the PFS rates between the groups in patients with advanced/metastatic breast cancer.
In summary, this meta-analysis demonstrated that in the neoadjuvant setting, platinum-based chemotherapy in the TNBC group shows short-term efficacy compared with the non-TNBC group. Unfortunately, no improvement in advanced TNBC was demonstrated [59].
The addition of platinum agents to anthracycline and/or taxane regimens in the neoadjuvant setting has shown promising outcomes, with pathological pCRs ranging from 30% to 62%. The results of the Phase II CALGB/Alliance 40603 clinical trial which looked at the impact of the addition of carboplatin and/or bevacizumab to neoadjuvant weekly paclitaxel followed by dose-dense doxorubicin and cyclophosphamide on pCR rates in TNBC were presented at the 2013 San Antonio Breast Cancer symposium [60]. The addition of carboplatin to standard neoadjuvant chemotherapy increased pCR. Despite increases in pCR with bevacizumab, side effects were more concerning with this drug.
Pathologic complete response rates were 60% for patients receiving carboplatin and 46% in patients who did not receive carboplatin; an increase of 76% (P = .0018). Defined by no disease in the breast or axilla, pCR rates increased to 54% and 41%, respectively; a 71% increase (P = .0029)
The addition of bevacizumab was also associated with an improvement in pCR in the breast only, producing pCRs in 59% of patients receiving the drug and 48% of patients not treated with bevacizumab; a 58% increase (P = 0.0089). Pathologic complete response rates for both breast and axilla were 52% and 44%, respectively; a 36% nonsignificant increase (P = .0570). When carboplatin and bevacizumab were used in combination, the highest pCR rate of 67% was achieved. However, the P value for the carboplatin/bevacizumab interaction was .52, indicating a lack of a synergistic effect.
3.3. Microtubule stabilising agents
A review in 2012, focused on potential new treatments for TNBC and included the microtubule-stabilising agent, ixabepilone [61]. Clinical trials have shown that when used with capecitabine (a second-line therapy widely used in anthracycline and taxane-resistant disease), ixabepilone has an acceptable safety profile and clinical activity, when compared with capecitabine alone. The women in this trial had locally advanced or metastatic cancer, pre-treated with an anthracycline and a taxane [62], [63]. Analysis of pooled data from these trials found that the overall response rate (ORR) (31 vs. 15%) and progression free survival (PFS) (4.2 vs. 1.7 months), were improved for women with TNBC who received combination therapy of ixabepilone and capecitabine as opposed to those women who received single-agent capecitabine alone [64].
3.4. Angiogenesis inhibitors
Tumour VEGF expression is significantly higher in TNBC compared with non TNBC presentations [65]. Studies identifying potential molecular markers of TNBC, such as VEGF [66], EGFR, Tyrosine Kinases [67], Src [68], and mTOR [69], have impacted on the design of clinical trials, investigating targeted treatments in the space of angiogenesis inhibitors.
3.4.1. Anti-VEGF monoclonal antibody; bevacizumab
Bevacizumab, the anti-VEGF agent (Trade name Avastin® Genetech/Roche), has received controversial attention despite various promising results from a variety of clinical trials. Specifically, there are multiple Phase III trials looking at the efficacy of bevacizumab in breast cancer, with three trials assessing the drug as a first line agent along with chemotherapy in the TNBC metastatic setting. These three trials are (1) the E2100 study, (2) the Avastin & Docetaxel (AVADO) trial and (3) the Regimens in Bevacizumab for Breast Oncology (RiBBOn)-1 trial [70].
In the E2100 trial, the risk of progression in first-line TNBC patients was reduced by 51% whilst the median PFS doubled (5.3 versus 10.6 months), when bevacizumab was added to paclitaxel. The AVADO trial demonstrated a 47% reduction in disease progression when bevacizumab was added to docetaxel in the TNBC subgroup. However, there did not appear to be any clear benefit in the RiBBOn-1 trial when bevacizumab was added to chemotherapy regimens.
Similarly, on TNBC subgroup analysis (N = 159), patient improvements were observed in the second line setting only in the phase III RiBBOn-2 trial, demonstrating a 51% reduced risk of disease progression and a doubling of median PFS among women treated with the bevacizumab combination, compared with women treated withchemotherapy alone [2.7 versus 6.0 months; HR = 0.49 (95% CI 0.33–0.74), P = 0.0006]. There was also a trend towards improved survival [median, 17.9 versus 12.6 months; HR = 0.624 (95% CI 0.39–1.007), P = 0.0534].
In the neo-adjuvant setting, two studies addressing the role of bevacizumab are the GeparQuinto [71] and the National Surgical Adjuvant Breast and Bowel Project 40 (NSABP)-B40 [72] trials, where this anti-VEGF agent was combined with neoadjuvant anthracycline–taxane chemotherapy. In the TNBC subgroup, the two studies showed the opposing results. Specifically, GeparQuinto demonstrated a statistically significant improvement in pCR rates (N = 663; 39.3% versus 27.9%, P = 0.003) for patients receiving bevacizumab compared with chemotherapy while, The National Surgical Adjuvant Breast and Bowel Project (NSABP)-B40 showed no improvement of pCR rates. (N = 479; 51.3% versus 47.3%, P = 0.44). The differing clinical outputs in the results from these two trials might possibly reflect variations in how HER2-negativity was defined in the patient groups treated in the trials and in the therapy regimens they received [73]. Longterm DFS and OS results are not yet available for these two studies. However, the potential use of these anti-VEGF drugs in the treatment of early TNBC will become clearer as further results become available from the GeparQuinto and (NSABP)-B40 in addition to the ongoing Bevacizumab Adjuvant Therapy in TRIple negative Breast Cancer (BEATRICE) Phase III trial.
In 2010, the FDA re-addressed the efficacy of the drug, Avastin® (bevacizumab) for the treatment of women with breast cancer, due to its reported side effects/safety concerns such as thromboembolic events, gastrointestinal perforation, wound healing complications, haemorrhage, hypertensive crisis, nephrotic syndrome, congestive heart failure and neutropenic sepsis. The FDA gradually removed breast cancer for the use of Avastin® due to these concerns and on November 18, 2011, the Food and Drug Administration (FDA) Commissioner, Margaret Hamburg revoked the agency’s accelerated approval of the breast cancer indication for bevacizumab (Avastin® (bevacizumab) made by Genentech). The Oncology Drugs Advisory Committee rejected Genentech’s appeal based in part on the modest risk-benefit ratio of this agent in the overall population [74].
In 2011, Ranpura et al., published a meta-analysis in JAMA, highlighting the adverse side effects of bevacizumab, showing that when added to chemotherapy regimens, particular taxane and platinum drugs. Specifcally bevacizumab was associated with an increased risk of fatal adverse events (FAEs), compared to using chemotherapy alone [75]. Moreover, a total of 10,217 patients with a variety of advanced solid tumours from 16 randomised controlled trials (RCTs) were included in the analyses. The overall incidence of fatal adverse events with bevacizumab was 2.5% (95% CI, 1.7%-3.9%), with haemorrhage (23.5%), neutropenia (12.2%), and gastrointestinal tract perforation (7.1%), being the most common. Bevacizumab used for metastatic breast cancer has also not been shown to provide a patient benefit in terms of delay in tumour growth that would justify its serious and potentially life-threatening risks. Therefore, there is currently no evidence that the use of bevacizumab will either help women with breast cancer live longer or improve their quality of life. This decision involves bevacizumab’s use in combination with paclitaxel for patients who have not been treated with chemotherapy for HER2 negative metastatic breast cancer. This indication has now been removed from bevacizumab's product labelling.
3.5. Epithelial growth factor receptor (EGFR) inhibitors
The epidermal growth factor receptor (EGFR) is a member of the ErbB family of receptors, a subfamily of four closely related receptor tyrosine kinases: EGFR (ErbB-1), HER2/c-neu (ErbB-2), Her 3 (ErbB-3) and Her 4 (ErbB-4).
3.5.1. Anti-EGFR monoclonal antibody; cetuximab
In patients with metastatic TNBC, a cetuximab plus cisplatin combination (BALI-I Trial) has demonstrated an overall better response rate of 20% when compared to a 10% overall better response rate with cisplatin alone [76]. Cisplatin plus cetuximab also resulted in longer PFS compared with cisplatin alone (median, 3.7 v 1.5 months) with a corresponding median OS of 12.9 versus 9.4 months. Common grade 3/4 adverse events included acne-like rash, neutropenia, and fatigue. However, the BALI-I trial failed to reach its primary endpoint, namely the overall response rate, despite apparently doubling it, compared to the single regimen. This combination also appears to lengthen PFS and OS. There was also a concern of toxicity such as diarrhoea when cetuximab was added to chemotherapy such as carboplatin and irinothecan despite an increased overall response rate in the TNBC subset of O’Shaughnessy’s phase II trial [77]. Analysis of two randomised trials has shown that basal like breast cancer patients lacking PTEN and alpha basic crystalline, preferentially respond to cetuximab treatment [78]. Therefore, the potential efficacy of anti-EGFR strategies needs further investigation with further trials such as those investigating combinations of cetuximab with the non-taxane anti-tubulin agent, ixabepilone in both the early (NCT01097642) and advanced (NCT00633464) settings.
3.5.2. EGF/Src tyrosine kinase inhibitors
Of the multiple-tyrosine kinase inhibitors, dasatinib, sunitinib and neratinib have been studied in a setting where patients have already previously received heavy doses of standard chemotherapy [79]. Dasatinib, which has activity in haematology malignancies and prostate cancer, has been shown to inhibit the growth of TNBC cell lines in vitro when used in combination with standard chemotherapy such as cisplatin and when used as a single agent [68]. However, a Phase II clinical trial of women with locally advanced or metastatic TNBC showed a clinical benefit rate of only 9.3% when dasatinib was used as a single agent [80].
In relation to sunitinib, this tyrosine kinase inhibitor was not recommended to be used in combination with doxetaxel in patients with newly diagnosed HER2 negative advanced or in patients with metastatic breast cancer following a Phase III trial [81]. Like bevacizumab, sunitinib is increasingly regarded as being ineffective in breast cancer [82].
For neratinib, the NCT01111825 Phase I/II trial is currently investigating its efficacy in combination with the mTOR Inhibitor temsirolimus, both in the metastatic TNBC and metastatic HER2 amplified setting. The primary outcome is to estimate the maximum tolerated dose with safety and efficacy as secondary outcomes. Recruitment is through the Memorial Sloan-Kettering Cancer Centre group and December 2015 is the estimated completion for this trial.
The tyrosine kinase phosphatase, PTPN12, acts by inhibiting multiple oncogenic tyrosine kinases, including HER2 and EGFR and appears to be mutated in 5% of TNBCs and absent from as many as 60% [83]. PTPN12 exhibits tumour suppressor actions when reintroduced into breast cancer cells devoid of the enzyme [83]. Sunitinib and lapatinib might therefore have beneficial activity against these tumours. Phase 1/2 clinical trials should commence to determine whether FDA approved tyrosine kinase inhibitors are effective in patients with PTPN12-deficient TNBC tumours. The possibility of a clinical trial with sunitinib and crizotinib, two drugs that are FDA-approved and marketed by Pfizer is being explored by the Stand up to Cancer Breast Cancer Dream Team.
3.6. mTOR inhibitors
The serine-threonine kinase mammalian target of rapamycin (mTOR) promotes protein translation, angiogenesis, proliferation and migration [84]. Moreover, inhibiting mTOR’s mediated PI3K/Akt signalling pathway abolishes cellular proliferative responses and causes cell cycle arrest. As PI3K/Akt overactivity has been identified in a number of breast cancers [69], rapamycin and its analogs temsirolimus, everolimus, and deforolimus, are undergoing clinical evaluation in TNBC treatment [85]. The mesenchymal subgroup of TNBC may benefit from a treatment targeting and inhibiting mTOR as the aggressive metaplastic and claudin-low type is enriched in epithelial-mesenchymal transition and stem-cell like features which make them more chemoresistant than the luminal & basal subtypes [86].
In relation to the efficacy of everolimus, the addition of this mTOR inhibitor to standard chemotherapy did not significantly improve pathological pCR rates in a phase II neoadjuvant trial of 50 TNBC patients [87] with another neoadjuvant trial in progress (NCT00930930). Ongoing studies investigating the use of everolimus in the treatment of advanced TNBC are being investigated in the NCT01272141, NCT01111825 and NCT00827567 trials. Specifically, NCT01272141 is looking at the combination of lapatinib, a dual role HER2/ EGFR inhibitor and everolimus in locally advanced or metastatic TNBC. NCT01111825 is addressing the role of temsirolimus plus neratinib in patients with either metastatic HER2-amplified or TNBC, while NCT00827567 is examining the use of everolimus as a single agent in metastatic TNBC.
The PTEN phosphatase, which inhibits the pro-growth PI3K/Akt pathway, is lost in the majority of TNBC tumours, particularly African-American and Hispanic patients [88]. PTEN loss is associated with reduced disease-free survival, but might also render these tumours susceptible to PI3K/Akt inhibitors.
The TNBC cell line MDA-MB 435 with PTEN deficiency has shown increased sensitivity to mTOR inhibition [89], while Steelman et al., 2008 also demonstrated that suppression of PTEN function in the MCF 7 non-TNBC breast cancer cell line, also increased sensitivity to the mTOR inhibitor rapamycin [90]. Overexpression of S6K1 and expression of phosphorylated Akt could be predictors of rapamycin sensitivity in breast cancer patients with changes in cyclin D1 levels providing a potential pharmacodynamic marker of response to rapamycin [91].
3.7. Androgen inhibition
Gucalp et al., 2010 have shown that androgens, through stimulating the androgen receptor can induce proliferative changes in breast cancer cell lines and promote tumourigenesis in animal models [92]. Androgen-enhanced growth of the cell line MDA-MB-453 which has the same biomarker phenotype as TNBC, is known to be ER-independent and AR-dependent [93]. Intriguingly, 10%–35% of TNBC express androgen receptors [94]. Moreover, it has been suggested that a subset of TNBC cases may benefit from the addition of androgen blockade to their therapy [95]. Bicalutamide, a nonsteroidal competitive androgen inhibitor, has been used in the treatment of advanced prostate cancer, but until recently [96], its anticancer effects had not been clinically tested in women. Specifically, the NCT00468715 is an ongoing clinical trial evaluating the use of bicalutamide in the treatment of 65 women with ER/PR-negative, AR-positive metastatic breast cancer with results presented at the 2009 ASCO Breast Cancer Symposium. Clinically, bicalutamide was well-tolerated and preliminary analyses have demonstrated disease stabilisation in ER/PR negative, AR positive tumours with AR inhibition [96].
3.8. HDAC inhibitors
Other possible novel targeted therapies for TNBC include HDAC (histone deacetylase inhibitors) inhibitors, such as vorinostat which suppress cancer-cell proliferation by inducing cell-cycle arrest and/or apoptosis [97]. A paper published in Cancer Letters discusses vorinostat-induced apoptosis in the TNBC cell line MD-MBA-231 and the role of the p38 MAP kinase in the suppression of cancer growth. Interestingly, knockdown of p38 MAP kinase was associated with decreased caspase-3 cleavage. This is of note as cleaved caspase 3 plays a role in autophagy by regulating the extracellular export of autophagic vacuoles; an alternative cellular fate [98], [99].
3.9. Immunotherapies and vaccines
TNBC patient outcome has been shown to correlate with the presence of a tumour-immune infiltrate which suggests an area of great potential for effective TNBC treatment [30]. Tumour-infiltrating lymphocytes (TILs) play a role in controlling the clinical progression of various epithelial cancers [100] with a recent review discussing the potential role that the immune microenvironment plays in breast cancer development [101].
The presence of an intense lymphocytic infiltrate may predict response to neoadjuvant chemotherapy in breast cancer [102]. This was proposed following an analysis of core biopsies from patients enrolled in the GeparDuo and GeparTrio trial. In particular, the presence of intratumoural lymphocytes and lymphocyte-predominant breast cancers were associated with a 31% and 41% pathological complete response (pCR) rates, respectively. In comparison, the pCR rates were only 2% in patients without any lymphocytic infiltration [102]. At the San Antonio Breast Cancer symposium in December 2013, Denkert et al., presented findings from the GeparSixto trial (GBG 66), where increased TILs predicted benefit from addition of carboplatin to neoadjuvant therapy for TNBC and HER2 positive breast cancer [103]. At the same session, Adams et al., demonstrated the prognostic value of TILs in two phase II randomised adjuvant breast cancer trials: Eastern Cooperative Oncology Group (ECOG) 2197 and ECOG 1199 [104].
Given that TNBC has been recently categorised based on 6 molecular subtypes [30], the immunomodulatory (IM) subtype, characterised by elevated expression of genes involved in T-cell function, immune transcription, interferon (IFN) response and antigen processing, could benefit from immunotherapies. This would be a novel treatment strategy that may be efficacious to this subtype of TNBC. Interestingly, the immunomodulatory subtype has been shown to overlap with medullary breast cancers, with gene expression profiling showing that medullary breast cancer is a subgroup of basal breast cancer displaying a prominent lymphocytic reaction that is associated with a favourable prognosis [105], [106].
Immune-checkpoint blocking antibodies and immune-stimulating therapies might act synergistically when combined with chemotherapeutic drugs. The adoptive transfer of chimeric antigen receptor (CAR) -engineered T cells and tumour vaccines against cancer-testis (CT) antigens, which appear highly expressed in TNBC as a result of epigenetic changes, are also novel therapeutic strategies for the TNBC immunomodulatory subtype [105].
The more aggressive claudin-low subtype often presents with an intense immune cell infiltrate and stem cell features with epithelial–mesenchymal transition features [107]. This subtype could therefore also potentially be targeted by immunotherapy treatment [108].
3.10. Other novel signalling pathways
New sequencing technologies have also identified promising therapeutic targets that alter apoptotic pathways. In one case, the developmental signalling pathways Wnt/β-catenin, NOTCH and Hedgehog have recently been shown to play an important role in the pathogenesis and progression of TNBC with new potential therapeutic approaches inhibiting these pathways published [109].
JAK2 amplifications in TNBCs has also been shown to be associated with a worse patient outcome and appear to be enriched in TNBCs. Balko et al., at the 2013 San Antonio Breast Cancer Symposium presented these findings and demonstrated that after neoadjuvant chemotherapy, TNBCs enriched with this amplification were associated with a poor prognosis, potentially identifying JAK2 inhibitors as novel and potential treatments for TNBC [110]. Table 1 summarizes the current novel therapies for TNBC.
Table 1.
Summary of novel therapies for TNBC.
PARP Inhibitors – Olaparib, Iniparib, Veliparib Platinum Salts – Carboplatin, Cisplatin
Ixabepilone Eribulin
Anti-VEGF Monoclonal Antibody – Bevacizumab (Avastin®)
Anti-EGFR Monoclonal Antibody – Cetuximab EGFR Tyrosine Kinase Inhibitors – Dasatinib, Neratinib, Sunitinib mTOR Inhibitor – Temsirolimus, Everolimus, Deforolimus
Bicalutamide
Vorinostat
Hedgehog – monoclonal antibodies, small molecular inhibitors NOTCH – monoclonal antibodies WNT/β-catenin signalling – monoclonal antibodies, ligand receptor inhibitors |
3.11. N-acetyl cysteine (NAC)
It has been advocated that new cancer therapies should be developed to target the metabolic inter-dependencies between epithelial cancer cells and their surrounding stromal microenvironment. Moreover, it has been suggested that cancer chemo-prevention and treatment should focus on the development of new powerful anti-oxidants to minimise oxidative stress, L-Lactate production and tumour stromal co-evolution.
The thiol anti-oxidant N-acetyl cysteine (NAC) behaves as an anti-inflammatory due to NFKappaB inhibition and inhibits L-Lactate production. Due to the fact that lactate has been shown to decrease after chemotherapy or radiotherapy in animals [225], the monitoring of this metabolite may be predictive of therapeutic response [226]. Clinically, N-acetyl cysteine (NAC) is already used in clinical practice as an antidote for acute drug intoxication, in particular as a result of Paracetamol poisoning with safe dosages and careful assessments of its side effect profile already known even at very high doses for long-term treatments [227], [228], [229]. It also has a clinical role in inflammation and acute respiratory distress [230].
However, the potential therapeutic use of NAC as an anti-cancer agent is yet to be tried out in the clinical setting. Intriguingly, its ability to negatively impact on the ability of cells to undergo senescence and or autophagy and indeed resistance to anoikis, key cellular fates induced in hypoxic environments, identifies it as an attractive candidate to be used in combination with our established adjuvant and neo-adjuvant treatments in TNBC, an established and recognised hypoxic tumour with an innately chemoresistant biology. Table 2a, Table 2b summarise the evidence to date that NAC could have a role as an antioxidant in cancer and more specifically in TNBC.
Table 2a.
Evidence that N-acetyl cysteine (NAC) can act as an anti-cancer agent.
| 1. NAC induces p53-dependent apoptosis, has anti-tumour activities and has been shown to be cancer chemo preventitive in clinical studies, with promise in preventing tumour progression [229], [231]. |
| 2. Treatment with thiol-containing antioxidants (such as NAC) has the potential to preferentially induce apoptosis in preneoplastic and neoplastic human lung fibroblasts cells [232]. |
| 3. NAC is involved in the downregulation of VEGF expression, by limiting hypoxia-induced transcription via hypoxia inducible factor-1-alpha (HIF1-α) and repression of reactive oxygen species (ROS) [233]. Clinically this has resulted in angiogenesis inhibition and thus reduction in tumour growth in Kaposi Sarcoma [234]. |
Table 2b.
Evidence that N-acetyl cysteine (NAC) has a role in targeting TNBC viability.
| 1. In comparison to the non-malignant breast cell line MCF-10A, it has been shown in the TNBC cell line MB-MDA-231 in vitro, that redox controls progression of the cell cycle suggesting that loss of redox control could drive aberrant cancer cell proliferation [235]. Indeed redox regulation of cancer cells is emerging as a potential therapeutic target in cancer [152]. 2. Treatment of the TNBC cell line MDA MB 435 with NAC results in apoptosis and reduction of microvascular density within the core of the tumour leading to significant tumour cell apoptosis/necrosis. This study demonstrated that NAC promotes anti-angiogenesis resulting in endothelial apoptosis and vascular collapse in the tumour [236]. 3. NAC appears lethal in the BRCA1-deficient TNBC cell lines HCC 1937 and MDA MB 231. Immunostaining for MCT4 (a functional marker of hypoxia, oxidative stress, aerobic glycolysis, and L-lactate efflux) [237] could be used as a cost-effective biomarker to monitor the response to antioxidant therapy [238]. |
4. Summary
Women presenting with triple negative breast cancer (TNBC) are treated in the most part similarly in the adjuvant and neoadjuvant setting to women who present with non-triple negative breast cancer. This is despite the widespread novel therapy strategies that are currently being investigated in clinical trials. Due to varying histological subtypes and molecular profiles within the heterogenous biology that is TNBC, future treatment efficacy and personalised treatment will need to consider the uniqueness of these presentations in order to progress a much needed individualised treatment strategy for these women.
5. Chemoresistance in TNBC
Chemoresistance can be attributable to the fact that although cytotoxic chemotherapy aims to kill cancer cells through apoptosis, tumour cells have the ability to maintain viability following chemotherapeutic exposure by undergoing alternative cellular fates such as cellular senescence, therapeutic induced senescence (TIS) and autophagy.
Tumour environmental stresses such as starvation, hypoxia and DNA damage have been shown to induce such fates as autophagy [111] and senescence [112]. Both cellular fates being significantly associated with cancer cell survival and chemoresistance [113], [114].
Genomic acknowledgment of at least 5 intrinsic breast cancer subtypes coupled with at least 6–7 TNBC molecularly characterised subtypes, makes it not surprising that the task of tailoring treatment for breast cancer presentations a minefield [25], [30].
However, adopting the approach of understanding the cellular fates induced in TNBC and using this information to bypass their induction or impede them from survival, has the potential to harness the efficacy of our current therapeutics more effectively. Despite the heterogeneity of TNBC, if we can demonstrate that cellular fates and the tumour microenvironment play a significant role in chemoresistance, a potential treatment target could be exploited for this tumour type by targeting cellular fates and not specific signalling pathways.
5.1. Established mechanisms of chemoresistance in TNBC
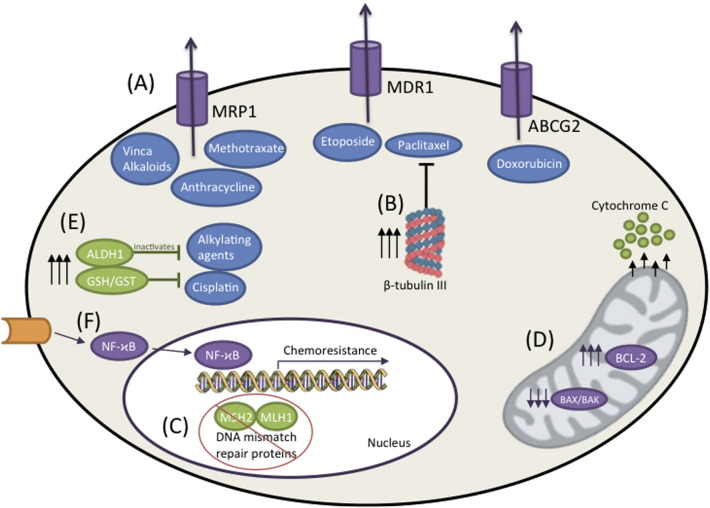
Standard chemotherapy remains the backbone of systemic triple negative breast cancer (TNBC) treatment even amidst advances in cancer treatment as neither hormonal therapy nor anti-HER2 agents are efficacious against TNBC due to the absence of a target [73]. At present, the cure for cancer continues to escape oncologists due in a large part to chemoresistance, which accounts for 90% of drug failures in metastatic cancers [115]. Fig. 1 demonstrates six mechanisms of chemoresistance in TNBC that are discussed in detail in this section namely, 1) APC transposrters, 2) β-tubulin III, 3) mutations in DNA repair enzymes such as topoisomerase II and DNA mismatch repair enzymes, 4) alterations in genes involved in apoptosis, 5) ALDH1 and glutathione (GSH)/Glutathione-S-transferase (GST) and 6) NF-ϰB signaling pathways.
Fig. 1.
Mechanisms of Chemoresistance in TNBC.
(A) ABC transporters efflux chemotherapeutics out of cancer cells (B) Overexpression of β-tubulin III subunit induces paclitaxel resistance (C) Mutations in DNA repair enzymes and enzymes altering drug sensitivity (D) Alterations in genes involved in apoptosis prevent chemotherapy-induced apoptosis (E) ALDH1 and glutathione (GSH)/Glutathione-S-transferase (GST) mediate chemotherapeutic inactivation/detoxification (F) Role of NF-ϰB signaling pathway in chemoresistance.
5.1.1. ABC transporters
One proposed method of chemoresistance involves ATP-binding cassette (ABC) transporters found in a subpopulation of cancer stem cells (CSCs), called side population (SP) cells [116]. Efflux of chemotherapeutic drugs by SP cells has been linked to the ABC transporter expressed in these cells. Three ABC transporters in particular have been extensively studied in TNBC namely (a)multidrug-resistant protein-1 (MRP1) which confers resistance to agents such as vinca alkaloids, anthracyclines, and high-dose methotrexate but not paclitaxel or mitoxantrone [117], (b) breast cancer resistance protein (ABCG2) [118], [119], [120] which is responsible for the efflux of drugs such as doxorubicin, and (c) the P-glycoprotein (MDR1) pump which pumps a wide array of chemotherapeutics out of cancer cells, including paclitaxel [115]. Specific inhibitors of ABC transporters are currently being tested as a means for overcoming this chemoresistance along with non-specific methods such as nanodiamond conjugation of chemotherapeutic drugs to impair efflux through MDR1 overexpressing cells.
5.1.2. Overexpression of β-tubulin III subunit induces paclitaxel resistance
Tomassi et al., 2007 and Paradiso et al., 2005 have found that in advanced breast cancer, βIII overexpression correlated with paclitaxel resistance and progression of disease in patients given first-line paclitaxel chemotherapy [121], [122].
5.1.3. Mutations in DNA repair enzymes and enzymes altering drug sensitivity
Reduced levels of expression or function of topoisomerase II, an enzyme critical in DNA replication and repair, have been shown to lead to chemoresistance to drugs such as the anthracyclines and epipodophyllotoxins [123], [124]. Chemoresistance may also arise from aberrant DNA mismatch repair enzymes that function to repair DNA damage from various chemotherapeutics such as alkylating agents, platinum compounds, and anthracyclines. Fedier et al., 2001 have further shown that there is a correlation between the loss of DNA-mismatch repair proteins MSH2 and MLH1, and the associated microsatellite instability genotype coupled with resistance to topoisomerase II inhibitors (epirubicin), doxorubicin, and mitoxantrone [125].
5.1.4. Alterations in genes involved in apoptosis
Alterations in genes regulating apoptosis (p53, caspase-3 s, bcl-2, bcl-x) have been linked with chemoresistance to various chemotherapeutics such as cyclophosphamide, doxorubicin, methotrexate, fluorouracil, and tubulin inhibitors [126], [127], [128], [129], [130].
5.1.5. Drug inactivation/detoxification as a mechanism of chemoresistance
Overexpression of ALDH1A1 and ALDH3A1 have been shown to result in greater inactivation of cyclophosphamide leading to chemoresistance [131]. Moreover, increased glutathione and glutathione-S-transferase activity results in increased breakdown of alkylating agents and cisplatin thereby increasing resistance to these compounds [132]. Lastly, polymorphisms in the cytochrome P450 system (namely CYP3A4 and CYP2C8) have been associated with highly basal enzymatic activity which may also contribute to chemoresistance [133].
5.1.6. Role of the NF-ϰB signaling pathway in chemoresistance
Treatment of breast cancer with disulfiram and copper sensitises breast CSCs to paclitaxel possibly by inhibiting constitutively active NF-ϰB thereby alluding to the correlation between aberrant NF-ϰB regulation and chemoresistance in TNBC [134].
5.1.7. KIF14-mediated AKT phosphorylation promotes chemoresistance in TNBC
Singel et al., 2014 published that overexpression of KIF14 increased chemoresistance to docetaxel in 34 cases of locally advanced TNBC. Conversely KIF14 knockdown and chemical inhibition resulted in lower levels of AKT phosphorylation and activity, leading to significant chemosensitisation when cells were treated with docetaxel. Importantly, phosphorylated AKT (pAKT) is an anti-apoptotic protein kinase in the well-characterised prosurvival PI3K/Akt signaling pathway [135].
5.1.8. Molecular markers that could predict resistance to chemotherapy in TNBC
Chekhun et al., 2009 have found that overexpression of metallothioneins and glutathione-S-transferase were associated with resistance to platinum salts while Zhou et al., 2011 reported that low expression of the eukaryotic translation initiation factor 4E (eIF4E) increased sensitivity to platinum salts [136], [137]. As for taxane chemotherapy, overexpression of the microtubule-associated protein 2 (MAP2), interleukin 6 (IL6), matrix metalloproteinase 9 (MMP-9), and low expression of inhibitor of tissue-plasminogen activation (t-PAI) increased sensitivity [138], [139] while overexpression of GRB-7 protein, alpha B-crystallin, and P-glycoprotein conferred a chemoresistant phenotype [140], [141]. Resistance to PARP inhibitors was increased by the overexpression of the multi-drug resistance efflux pumps (MDR 1,2) [142] and decreased by a deficiency of BRCA-1, BRCA-2, XRCC2, and XRCC3 genes [143]. Conversely, the restoration of function of the tumour suppressor gene BRCA-1 led to an increased chemoresistance against PARP inhibitors [142].
Lastly, the low expression of the zinc-finger enhancer binding protein ZEB-1 increased sensitivity of TNBC cells to bicalutamide [144].
5.2. Selective resistance of cancer stem cells (CSCs) to chemotherapy
Treatment of TNBC with chemotherapeutics such as taxanes is initially very effective in most patients. However, the majority of these tumours will recur following chemotherapy treatment [145]. These recurrent tumours are typically chemoresistant, and are associated with a poor prognosis [146]. There is an increasing amount of evidence that suggests that following treatment, a small population of cells survive, with stem-like properties that may be responsible for tumour recurrence [147]. These cells have been termed cancer stem cells (CSCs). CSCs are tumour cells with self-renewal properties that have the ability form a recurrent chemoresistant tumour. Lapidot et al. provided early evidence in 1994 of the existence of CSCs by identifying a subpopulation of cells in acute myeloid leukaemia (AML) that were capable of initiating leukemic growth when introduced into severe combined immunodeficiency (SCID) mice [148].
Recent evidence suggests that chemoresistant populations of cancer stem cells may be responsible for relapse in triple negative breast cancer (TNBC). The TGF-β family of cytokines has been implicated in CSCs. TGF-β1 and the TGF-β type 1 receptor (TGF-βR1) have been shown to be over-expressed in a subpopulation of breast cancer cells with CSC features. TGF-β induces epithelial-to-mesenchymal transition (EMT) in mammary cells, which has been associated with tumour stem-like properties in cells [149].
In 2013, Bhola et al. reported that following chemotherapy treatment, TNBC biopsies showed an increase in RNA transcripts of genes associated with CSCs and TGF-β signalling. Treatment with paclitaxel also increased TGF-β signalling and CSC properties in TNBC cell lines and mouse xenografts [150]. This suggests that initial treatment may form a subpopulation of cancerous cells with stem-like properties that can regenerate a chemoresistant tumour.
However, Bhola et al. also reported that using the TGF-β type 1 receptor kinase inhibitor LY2157299 in combination with paclitaxel, prevented re-establishment of tumours in TNBC xenografts in mice [150].
In 2014, Samanta et al. published that treatment of human TNBC cell lines with paclitaxel or gemcitabine resulted in an increase in hypoxia-inducible factors (HIFs) expression and transcriptional activity. This resulted in an enrichment of the CSCs population through interleukin-6 and interleukin-8 signalling [151]. This indicated that HIFs may be involved in acquired chemoresistance of breast cancer stem cells.
This study also indicated that the administration of HIF inhibitors in combination with paclitaxel or gemcitabine, overcame the CSCs resistance to the chemotherapeutics, which lead to tumour eradication, both in vitro and in vivo [151].
Both these studies show evidence that the treatment of TNBC with current therapeutics may result in the formation of a subpopulation of chemoresistant cancer cells with stem-like properties, that if not eliminated are free to form a tumour that is not responsive to current chemotherapeutics.
5.3. Tumour hypoxia’s role in TNBC chemoresistance
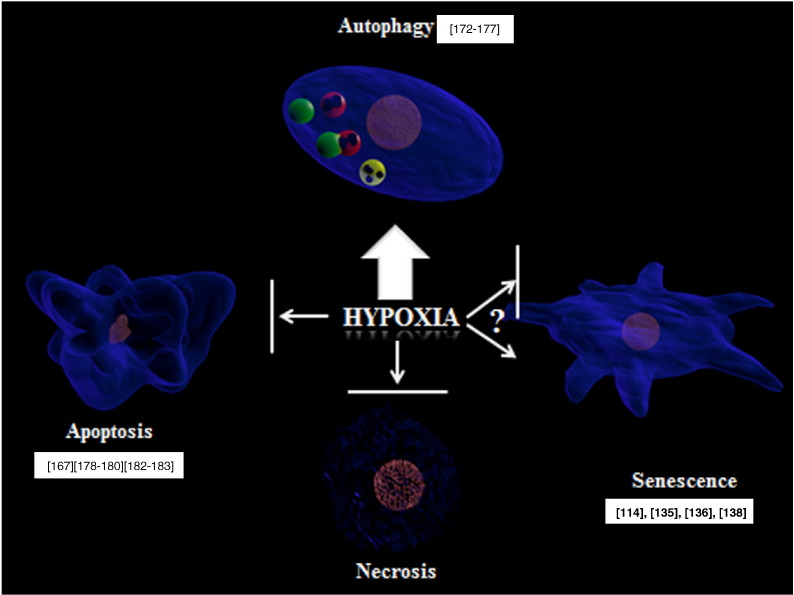
Breast cancer cells are subjected to a high level of oxidative stress, both intra- and extra cellularly [152]. Moreover, it is known that TNBC is associated with a more hypoxic phenotype possibly due to activation of the mTOR pathway and HIF-1α stabilisation [153]. Fig. 2 is a referenced illustration demonstrating the role that hypoxia plays in cellular fates. Clinically, the fact that TNBC’s are amenable to fluroeoxyglucose-positron emission tomography (FDG-PET) scanning as a result of the “Warburg effect” [154] also highlights their more hypoxic phenotype (Table 3).
Fig. 2.
The role hypoxia plays in cellular fates including references.
Table 3.
Clinical published evidence that triple negative breast cancer (TNBC) is more hypoxic than non-TNBC histologies.
| Hypoxia Appreciation | Patient Numbers | Details | Reference |
|---|---|---|---|
| FDG-PET | N = 88 TNBC = 29 ER +/PR +/HER2- = 59 |
TNBC tumours associated with enhanced FDG uptake, and detected with very high sensitivity using FDG-PET imaging. | Basu et al., 2008 [154] |
| FDG-PET | N = 41 TNBC = 22 Non-TNBC = 19 |
The degree of tumour FDG uptake correlated significantly with proliferation in women with TNBC, suggesting a potential role of FDG-PET in monitoring treatment response for this group of women. | Tchou et al., 2010 [167] |
| FDG PET/CT | N = 78 (Stage II / III Breast Cancers) |
FDG-PET/CTs were acquired before and after the first cycle of chemotherapy to evaluate early metabolic response. In TNBC, the pre-chemotherapy standardised uptake value (SUV) was higher than in non-TNBC. | Keam et al., 2011 [168] |
| 18 F-FDG PET/CT | N = 152 (Primary Breast Cancers) |
High SUV levels were significantly correlated with tumour size, poorer grade, nuclear atypia, increased mitotic index, negative hormone receptor status, high score of HER-2 expression, lymph node metastasis, and IDC in comparison with invasive lobular carcinoma. | Ueda et al., 2008 [169] |
| 18 F-FDG-PET | N = 91 (Invasive Ductal Carcinomas) |
High FDG uptake correlated with poor prognosis factors, such as tumour invasiveness > 2 cm, higher tumour grade, hormonal receptor negativity, and triple negativity. | Kim et al., 2012 [170] |
| 18 F-FDG–PET | N/A | 18 F-FDG–PET had a higher sensitivity for TNBCs than oestrogen-positive and human epidermal growth factor receptor 2-positive tumours. | Dogan et al., 2012 [171] |
| Protein expression of CAIX and BRCA1 evaluated by AQUA (Automated Quantitative Analysis) Technology (IHC) | N = 619 TNBC = 111 Unselected Cohort = 508 |
CAIX was expressed in 7% of the unselected breast cancer cohort and in 25% of the TNBCs and significantly associated with the triple negative phenotype. CAIX also associated with poor outcome. | Neumeister et al., 2012 [161] |
| FFPE Tissue Sections stained for various hypoxic markers, including; HIF-1α, PHD1-3, and CAIX |
CAIX Staining: N = 456 Basal-like = 62 |
50% of basal-like tumours expressed HIF-1α, and more than half expressed at least one of the PHD enzymes and FIH-1. Basal-like tumours were nine times more likely to be associated with CAIX expression. Patients with any CAIX-positive breast tumour phenotype or CAIX basal tumours had a significantly worse prognosis than CAIX-negative tumours, partly attributable to an enhanced hypoxic response. | Tan et al., 2009 [162] |
Note:- FDG-PET = fludeoxyglucose-positron emission tomography, SUV = standardised uptake value, SUV represents the ratio of the image derived radioactivity concentration found in a selected part of the body at a certain time point. CAIX = carbonic anhydrase, HIF-1-α = hypoxia inducible factor, IDC = invasive ductal carcinoma, PHD = prolyl hydroxylase domain, FIH = factor inhibiting hypoxia-inducible factor.
The term hypoxia describes the phenomenon of an inadequate supply of oxygen as a result of cells outgrowing their vasculature [155] beyond the size of several mm3. This compromises the cells biological functions with a concomitant increase in compensatory glycolysis [156]. Importantly, hypoxia is a key microenvironment associated with the tumour milieu with this acidic setting compromising chemotherapeutic efficacy due to the varying levels of oxygen present in the tumour [157]. Moreover, a hypoxic tumour environment results in an increase in cellular senescence [114], [159] and therefore potentially propagating a chemoresistance phenotype.
Clinically, evidence that TNBC tumours are more hypoxic than non-TNBC histologies is presented in Table 3 by either an increased FDG uptake and standardise uptake values (SUV) on PET/CT imaging or increased Carbonic Anhydrase IX (CAIX) immunohistochemical assessment on full face paraffin embedded (FFPE) TNBC clinical material. TNBCs are amenable to FGP-PET scanning on account of the “Warburg effect” [154] with one study addressing the use of 18F-FDG PET/CT as an early assessment in TNBC during neoadjuvant chemotherapy to identify patients who are unlikely to achieve a pathologic complete response and are at a high risk of early relapse [160]. CAIX, is an accepted marker for tissue hypoxia and has been shown to be associated with a worse prognosis for patients with TNBC [161], [162]. Recent evidence shows that basal endoplasmic reticulum stress (ERS) is typically activated in TNBC and cooperates with hypoxia signalling to promote tumour progression and relapse [163].
5.4. Cellular senescence
The term cellular senescence was first discovered and the term coined in aging human fibroblasts by Hayflick and Moorhead in 1961 who observed an irreversible exit of the cells from the cell cycle. Senescent cells, however, although initially having lost their proliferative capacity, maintain cellular viability and metabolic activity [112] generating a senescent secretome called the SASP phenotype or senescence associated secretory phenotype.
In relation to the role of cellular senescence in cancer, on the one hand it can be regarded as a tumour suppressor mechanism for cells in response to DNA damage, telomere attrition, oxidative stress, DNA damage and other tumour-promoting insults [112] and indeed there is evidence that the progression of malignant invasive tumours is believed to arise from the evasion of senescence in the pre-invasive precursor tumour [164]. Recent evidence however also implicates the senescence pathway in breast carcinogenesis [165] through promoting tumour progression by stimulating growth and transformation in adjacent cells through the senescence associated secretory phenotype (SASP) [166].
5.5. Hypoxia, senescence and stroma
Table 4 demonstrates the role hypoxia plays in determining cellular senescence in cancer with clear conflicting views evident.
Table 4.
The role hypoxia plays in determining cellular senescence in cancer.
| Cellular Fate | Determination of Cellular Fate | Details/Results |
|---|---|---|
| Senescence Watson et al., 2009 [114] | SA-β-gal | PwR-1E benign prostate epithelial cells and equivalently aged hypoxia-adapted PwR-1E cells were used to identify the phenotypic and epigenetic consequences of chronic hypoxia in prostate cells. |
| The impact of chronic hypoxia (1% O2) on cellular senescence was assessed by β-galactosidase activity for both cell lines. | ||
| Increased levels of cellular senescence were identified in the hypoxic population of PwR-1E cells compared to their normoxic counterparts. | ||
| Senescence Hammond et al., 2005 [172] | p53 Immunoblotting | RKO, 293 T, RCC4, and HCT116 cell lines used. |
| HIF-1α protein accumulated with similar kinetics at 0.2% and 2% oxygen. p53 protein accumulated in response to 0.02% oxygen only. p53 accumulated in response to hypoxia independently of HIF-1α. | ||
| Accumulation of p53 correlated with replication arrest. | ||
| Senescence Welford et al., 2011 [173] | N/A | Reports conflicting evidence on the role of hypoxia in senescence: |
| Excess O2 can lead to the production of Reactive Oxygen Species (ROS) which can promote senescence. Reduced O2 levels (hypoxia) can reduce the amount of ROS. | ||
| Hypoxia leads to the stabilisation of HIF, which impacts on many pathways that can affect senescence: | ||
| HIF binding to Myc can induce p21 expression and inhibit CDC25C, promoting cell-cycle arrest. | ||
| HIF controls several genes in the senescence-associated secretory phenotype (SASP) that can promote senescence. | ||
| However, HIF also promotes glycolysis, and can induce expression of TERT, negatively regulating p53, both of which are inhibitory to a senescent phenotype. |
Note:- TERT = Telomerase reverse transcriptase, CDC25 phosphatases are important regulators
of the cell cycle, Myc = transcription factor.
The contribution of the stromal component and the inter-dependencies between tumour epithelial cells (TECs) and their stromal environment, tumour associated fibroblasts (TAF) in an oxygen stressed /L-lactate rich environment has been identified as a key cancer research area. This is new model of cancer metabolism, identifying the role of aerobic glycolysis and L-lactate production in fuelling tumour growth and metastasis [137].
Intriguingly, senescent cells produce considerable amounts of lactate and play a key role in the tumour-stroma co-evolution. In relation to the relevance of this to breast cancer, lower levels of lactate have been observed in breast cancer patients with long-term survival (≥ 5 years, survivors) compared to patients who died of cancer recurrence [174].
This novel hypothesis led by Professor Lisanti in Thomas Jefferson University, Philadelphia is coined the “Reverse Warbung effect” as aerobic glycolysis takes place in stromal fibroblasts, rather than epithelial cancer cells. This group demonstrated that MCT4 immunostaining of human TNBC micro arrays can be used to directly assess prognosis [175]. MCT4 is a functional marker of hypoxia, oxidative stress, aerobic glycolysis, and L-lactate efflux. High stromal MCT4 levels were associated with decreased overall survival (< 18% survival at 10 years post-diagnosis). In contrast, patients with absent stromal MCT4 expression had 10 year survival rates of ~ 97%. Interestingly, epithelial MCT4 staining demonstrated no prognostic value, suggesting that the tumour stroma may represent a needed focus for targeted therapy.
Oxidative stress induced by the tumour epithelial cells (TECs), results in increased levels of fibronectin (FN) on the surface of adjacent senescent tumour associated fibroblasts (TAFs) [176]. Increased production of FN is one of the most typical features of senescent cells [177], [178] and potentially is an antigenic determinant unique to cellular senescence [179]. Intriguingly FN is differentially upregulated in TNBC compared to Her2 +, ER-PR- [180].This increased synthesis of FN is mediated by TGF-α1 [176] which can stimulate p16 (INK4), a marker of cellular senescence [181].
Mechanistically, reconstitution of p16 has been shown to increase the expression of the FN receptor α5β1 on TECs [182]. Moreover, elevated intracellular reactive oxygen species (ROS) produced by oxidatively stressed mitochondria increases α5β1 expression on tumour epithelial cells (TECs) [183]. When bound to extracellular matrix components such as FN, α5β1 co-operates with growth factors to activate anti-apoptotic signalling.
It has also been shown that with increasing age, the accumulation of senescent cells alter the tissue architecture to the extent that it provides a permissive environment of uncontrolled growth of adjacent premalignant cells [184]. Moreover, fibroblasts adjacent to malignant tissue in contrast to those neighbouring the normal epithelium have been found to be senescent [185] suggesting a possible role for their assessment in identifying tumour free margins.
The role of epithelial versus stromal features of senescent cells in predicting outcome for patients presenting with TNBC, irrespective of the chemotherapeutic used or the individual subtype of TNBC, is a potential area of research.
5.6. Autophagy
Autophagy, type II programmed cell death, is an evolutionarily conserved, self-degenerative process involved in protein and organelle recycling and cellular homeostasis [186]. There are three types of autophagy; macroautophagy [187], microautophagy [188] and chaperone mediated autophagy (CMA) [189]. Macroautophagy involves the formation of a double-membraned structure in the cytoplasm (the autophagosome), which grows and eventually fuses at either end thus capturing the majority of cytoplasm and some organelles in the process. This autophagosome then fuses with the lysosome for degradation [151]. Microautophagy, on the other hand, refers to the direct transfer of cytosolic components into the lysosome by invagination of the lysosomal membrane and subsequent budding into vesicles within the lysosome itself [152]. Chaperone mediated autophagy (CMA) uses a different approach completely with specific selection and removal of proteins with particular peptide sequence motifs that are recognised by molecular chaperones, which then fuse with the lysosome via a lysosome-associated membrane protein Lamp2a for selected removal [151].
Autophagy appears to modulate both cell viability and death [190]. However, the role of autophagy in cell death is controversial. The presence of autophagic vacuoles in dying cells may be interpreted in one of two ways: either cells activate autophagy in an attempt to survive, or autophagy is a part of the process of cell death [191].
Autophagy is often expressed at a basal level in cells representing its role in the recycling of proteins and tissue homeostasis [192]. However, in addition to the role of autophagy in protein quality control, this cellular fate mechanism also plays an integral role in cell survival during cytotoxic stresses such as hypoxia, nutrient deprivation, DNA damage and in response to chemotherapeutics and radiotherapy [193], [194].
However, the overall role of autophagy in cancer is far more controversial with its inhibition and induction both showing beneficial and negative effects of tumour cell survival [195], [196], [197].
In tumour cells the role of autophagy may depend on the type of tumour, the stage of tumourigenesis and the nature and extent of the insult [198]. Inhibiting cytoprotective autophagy or promoting type II programmed cell death are mechanisms that could enhance the effects of cytotoxic chemotherapeutics [195].
5.7. Autophagy and hypoxia
Autophagy provides the cell with an anaerobic metabolic mechanism which supplies the cell with the basic metabolic portfolio to survive during oxidative stress or nutrient deprivation. It is also responsible for the removal of ROS [199] and unfolded proteins generated during oxidative stress and cellular aging [200].
The role of autophagy in an oncological setting has received much attention, specifically coined as the “Autophagic Tumour-Stromal Model” of cancer metabolism and tumour cell survival [201].
This model explains why angiogenesis inhibitors might not work, and instead induce lethal tumour recurrence, and metastasis. This is partially attributable to the fact that angiogenesis inhibitors drive “hypoxia” in the tumour stromal micro-environment. Hypoxia, in turn, drives oxidative stress and autophagy.
It has already been shown that paclitaxel induces autophagy in the cell line A549 human alveolar adenocarcinoma. However, conversely, inhibiting autophagy potentiates paclitaxel’s-mediated apoptotic effects [202].
In vitro studies have also demonstrated that hypoxia protects the TNBC cell line, MDA-MB-231 from paclitaxel-induced apoptosis and concomitantly induced an autophagic response after 2 hours in normoxic and hypoxic condition. Specifically, the mechanism of decreased apoptosis was studied using p62siRNA transfection, a protein involved in autophagosome formation. These results demonstrated that hypoxia diminishes paclitaxel-induced apoptosis in MDA-MB-231 cells via mTOR/JNK pathway [203].
Recently, a new form of autophagy has emerged known as mitophagy, which is believed to play a very important role in cancer cell survival [204], particularly in hypoxic regions of tumours. This is supported by studies of models with monoallelic deletion of Beclin-1 showing increased cell death in these tumour regions [205]. Mitophagy involves the selective removal of damaged mitochondria, subsequently decreasing ROS and cytotoxic molecules of apoptosis and thus, promoting cell survival [206], [207]. Mitophagy is induced by BNIP3 whose expression in turn is regulated by hypoxia-inducible factor-1 (HIF-1) expression or the Ras/raf/Erk pathway [207], [208]. Ras mutated tumours can therefore become very dependent on autophagic pathways, particularly mitophagy, through overactivation of autophagic pathways such as Ras/Raf/Erk or indirectly through hypoxia-induced expression of HIF-1. Due to the hypoxic nature of tumours, HIF pathways are of great importance in tumour progression. Cells have been found to adapt energy metabolism to hypoxic conditions through 4 main HIF-mediated mechanisms: (1) COX4 subunit switching, (2) inhibition of acetyl-CoA synthesis by activation of PDK1, (3) c-Myc repression and inhibtion of mitochondrial biogenesis and the newer concept of mitochondrial autophagy, which as described above, decreases ROS release from damaged mitochondria thus promoting cell survival [206].
5.8. Autophagy inhibition & TNBC
Protein expression of the autophagy-related microtubule-associated proteins, including beclin-1, light chain (LC) 3A and LC3B have been shown to be the highest in TNBC cells compared to the other breast cancer subtypes, with the lowest expression in the stroma of TNBC [209]. High expression of LC3B is also associated with tumour progression and poor outcome in TNBC [210] demonstrating its role as a potential prognostic marker in TNBC.
Intriguingly cancer cell viability has also been shown to be reduced through induction of non-apoptotic, non-autophagic cytoplasmic vacuolation death in TNBC cells through the expression of the LC3 protein and p62/SQSTM1; a protein involved in autophagosome formation [211].
Moreover, inhibition of autophagy has been identified as a potential adjunctive strategy for enhancing the chemotherapeutic effect of paclitaxel. In addition, by inhibiting autophagy with the pharmacological autophagy inhibitor chloroquine, a synergistic effect of enhanced TNBC cell death both in vivo and in vitro was noted when used with the endoplasmic reticulum stress aggravator (ERSA) compounds such as the antiretroviral Nelfinavir, and the COX2 inhibitor Celecoxib [212].
Table 5, Table 6 summarise the relationship between hypoxia and the fates of autophagy and apoptosis respectively in TNBC specifically.
Table 5.
The role hypoxia plays in determining the cellular fate autophagy.
| Pike et al., 2013 [213] | MCF7, MDA-MB-231, HCT116, HEK293, MEF3T6, T47D, and HeLa cell lines | ULK1 | Autophagy-initiating kinase ULK1 (UNC51-like kinase 1) is a direct transcriptional target of ATF4 (activating transcription factor 4), which drives the expression of ULK1 mRNA and protein in severe hypoxia. ULK1 is required for autophagy in severe hypoxia and ablation of ULK1 causes caspase-3/7-independent cell death. ULK1 expression is associated with a poor prognosis in breast cancer. |
| Salem et al., 2012 [214] | Human hTERT-immortalized fibroblasts MDA-MB-231 cell |
HIF-1α | shBRCA1 fibroblasts displayed an elevated growth rate. Using immunofluorescence and immunoblot analysis, shBRCA1 fibroblasts demonstrated an increase in markers of autophagy and mitophagy. Most notably, shBRCA1 fibroblasts also displayed an elevation of HIF-1α expression. Using xenografts demonstrated that shBRCA1 fibroblasts induced an approximate 2.2-fold increase in tumour growth when co-injected with MDA-MB-231 cells into nude mice. |
| Sotgia et al., 2011 [215] Witkiewicz et al., 2011 [216] |
Malignant melanoma 44 cases: 24 lymph node metastasis, 20 primary tumours. IHC stained for Cav-1 |
stromal Cav-1 | Coupling of metabolism between epithelial cells and surrounding stroma between oxygenated and hypoxia compartments. Increased autophagy/mitophagy in the stroma drives metabolism in the epithelial cells. Ammonia production in the epithelial cells then drives autophagy in stroma to promote this cycle. Loss of Cav-1 in stroma leads to early recurrence, metastasis, drug resistance and poor clinical outcome. |
| Azad et al., 2008 [217] | Two glioma cell lines (U87, U373). Two breast cancer cell lines (MDA-MB-231, ZR75). One embryonic cell line (HEK293). |
BNIP3 | Under normoxic conditions, all five cell lines displayed etoposide-induced apoptosis whereas hypoxia failed to induce these apoptotic responses BNIP3 overexpression induced autophagy, while expression of BNIP3 siRNA or a dominant-negative form of BNIP3 reduced hypoxia-induced autophagy |
| Tan et al., 2007 [218] | Normal breast tissue n = 11 DCIS n = 81 IDC n = 251 |
BNIP3 and HIF1a | Stained for BNIP3 and hypoxia-inducible factor-1alpha. BNIP3 expression was significantly up-regulated in the cytoplasm of DCIS and invasive carcinoma compared with normal breast Nuclear BNIP3 expression was associated with smaller tumour size, low tumour grade (P = 0.005), and oestrogen receptor positivity in invasive tumours Nuclear BNIP3 expression in DCIS was associated with a 3-fold increase in recurrence and a shorter disease-free survival No association between HIF-1α and BNIP3 expression |
Note: ULK = autophagy-initiating kinase, a homologue of yeast ATG1,CAV1 = caveolin 1 leads to oxidative stress, BNIP3 = Bcl-2 and nineteen-kilodalton interacting protein-37 and is a member of the Bcl-2 protein family.
Table 6.
The role hypoxia plays in determining the cellular fate, apoptosis.
| Cellular model | Details | Reference |
|---|---|---|
| MDA-MB-231 Breast cell line |
Hypoxia protected MDA-MB-231 cells form paclitaxel-induced apoptosis and concomitantly induced autophagy response after 2 hours in normoxic and hypoxic condition. The mechanism of decreased apoptosis was studied using siRNA and showed that hypoxia decreases the apoptotic response via mTOR and JNK activation. | Notte et al., 2013 [203] |
| MCF7 Breast cell line |
Hypoxia decreased paclitaxel-induced apoptosis and G2/M arrest was visualised by MTT assay and flow cytometry. Also, hypoxia diminished paclitaxel-dependent polymerisation of tubulin. Interestingly it was found that under hypoxic conditions, Cyclin B1 was down-regulated which lead to decreased apoptotic death. Overexpression of Cyclin B1 in MCF7 cells partially restored sensitivity to paclitaxel in hypoxic cells. | Dong et al., 2012 [219] |
| A2780 Ovarian cell line |
Human ovarian xenograft tumours (A2780) in mice were severely hypoxic. Phosphorylation of protein STAT3 was observed under hypoxia and knock-down experiments revealed that STAT3 might be an important protein in mediating resistance to cisplatin and paclitaxel under hypoxic conditions. | Selvendiran et al., 2009 [220] |
| MDA-MB-231 Breast cell line Du-145 Prostate cell line |
Hypoxia induces resistance to etoposide in breast and prostate cancer due to the prevention of DNA damage and strand breaks after exposure. Cell treatment with small interfering RNA targeting hypoxia-inducible factor 1 prevented the hypoxia-induced decreases in topoisomerase IIalpha levels, abolished the protective effect of hypoxia against etoposide-induced DNA damage, and inhibited hypoxia-induced etoposide resistance. | Sullivan et al., 2009 [221] |
| C13K Ovarian cell line |
Ovarian cancer C13K cells under hypoxic conditions are resistant to cisplatin treatment. Inhibition of HIF1α transcriptional activity by the small molecule – noscapine improves sensitivity to cisplatin. | Su et al., 2011 [222] |
| MDA-MB-231 Breast cell line |
Hypoxia protected MDA-MB-231 breast cancer cells against paclitaxel-induced apoptosis. The implication of HIF-1 and AP-1 in the hypoxia-induced anti-apoptotic pathway was investigated by the use of specific siRNA. Results showed that both transcription factors are important in mediating resistance. | Flamant et al., 2010 [223] |
| MDA-MB-468 MCF-7 HCC1937 Breast cell lines |
Hypoxia induced change in BRCA1 subcellular localisation to the nucleus without a change in the overall protein levels. Hypoxia also induced TRAIL expression on the surface of the cell which increased apoptotic response to apoptotic stimuli. BRCA1 mutant cells were defective in apoptotic activation under hypoxic condition compared to BRCA1 wt cells. | Fitzgerald et al., 2007 [224] |
Note: JNK = c-Jun N-terminal kinase, STAT3 = Signal transducer and activator of transcription 3, TRAIL = TNF-related apoptosis-inducing ligand, TNF = tumour necrosis factor, wt = wild type.
6. Conclusion
Finding efficacious treatments for TNBC is critical due to the fact that these women are treated, in the most part with standard chemotherapy. This approach does not tailor to the specific biology of TNBC and its molecular subtypes. Even though the concept of standard chemotherapy in breast cancer is becoming a thing of the past with a) oncotyping and b) other molecular markers identifying hormone sensitive tumours that gain no benefit from chemotherapy, these do not however, apply to patients with TNBC resulting inevitably in a significant number of women not responding to first line treatment.
With this in mind and in order to improve patient outcome it is crucial to establish a) what factors influence a patients’ individualised response to chemotherapy and b) which factors may explain why some patients fail to benefit from standard first-line chemotherapy, while others achieve long-term remission.
In this regard, research specifically focused on interrogating the mechanisms and ability of tumour cells to circumvent and bypass apoptotic death mechanisms through adopting viable cellular fates such as cellular senescence and cytoprotective autophagy is warranted. In particular, targeting these non-apoptotic survival pathways has the potential to enhance the chemotherapeutic efficacy of our current artillery of drugs for women presenting with TNBC.
6.1. Summary
In specifically targeting the survival mechanisms that cancer cells can use to bypass death through apoptosis, viable cellular fates such as cellular senescence and cytoprotective autophagy could potentially be a target for impedance or manipulation and thus increase the death rate of cancer cells. Importantly, both these cellular fates are induced in tumour hypoxia, a key tumour microenvironment associated with TNBC and integral to chemoresistance. In the case of senescence, anti-oxidants such as NAC in combination with standard chemotherapuetics hold potential in eradicating these metabolically active SASP secreting cells, as do the combinatorial use of mTOR inhibitors or glycolytic inhibitors such as rapamycin to promote terminal autophagy. Such approaches are worth serious consideration and may offer more potential than attempts to find specific tailored therapies for the myriad of genetic and epigenetic changes and tumour cell populations unique to each patient’s tumour and tumour microenvironment.
Transparency document
Transparency document.
Acknowledgements
We acknowledge the support of the Health Research Board (grant number HRA-POR/2011/113), the Breast Health Research Mater Grant and the Mater Surgical Oncology Appeal. We also acknowledge the UCD School of Medicine and Medical Science for supporting the Intercalated M.Sc. of Matthew Ho Zhing Guang.
Footnotes
The Transparency document associated with this article can be found, in online version.
References
- 1.Schneider B.P., Winer E.P., Foulkes W.D., Garber J., Perou C.M., Richardson A. Triple-negative breast cancer: risk factors to potential targets. Clin. Cancer Res. 2008;14:8010–8018. doi: 10.1158/1078-0432.CCR-08-1208. [DOI] [PubMed] [Google Scholar]
- 2.Carey L.A., Dees E.C., Sawyer L., Gatti L., Moore D.T., Collichio F. The triple negative paradox: primary tumor chemosensitivity of breast cancer subtypes. Clin. Cancer Res. 2007;13:2329–2334. doi: 10.1158/1078-0432.CCR-06-1109. [DOI] [PubMed] [Google Scholar]
- 3.Wolff A.C., Hammond M.E.H., Hicks D.G., Dowsett M., McShane L.M., Allison K.H. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J. Clin. Oncol. 2013;31:3997–4013. doi: 10.1200/JCO.2013.50.9984. [DOI] [PubMed] [Google Scholar]
- 4.Jensen E.V., Desombre E.R., Hurst D.J., Kawashima T., Jungblut P.W. Estrogen-receptor interactions in target tissues. Arch. Anat. Microsc. Morphol. Exp. 1967;56:547–569. ( http://www.ncbi.nlm.nih.gov/pubmed/5615176) [PubMed] [Google Scholar]
- 5.McGuire W.L. Estrogen receptors in human breast cancer. J. Clin. Invest. 1973;52:73–77. doi: 10.1172/JCI107175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.McCullough A.E., Dell’orto P., Reinholz M.M., Gelber R.D., Dueck A.C., Russo L. Central pathology laboratory review of HER2 and ER in early breast cancer: an ALTTO trial [BIG 2-06/NCCTG N063D (Alliance)] ring study. Breast Cancer Res. Treat. 2014;143:485–492. doi: 10.1007/s10549-013-2827-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hede K. Breast cancer testing scandal shines spotlight on black box of clinical laboratory testing. J. Natl. Cancer Inst. 2008;100(836–7):844. doi: 10.1093/jnci/djn200. [DOI] [PubMed] [Google Scholar]
- 8.Welsh A.W., Moeder C.B., Kumar S., Gershkovich P., Alarid E.T., Harigopal M. Standardization of estrogen receptor measurement in breast cancer suggests false-negative results are a function of threshold intensity rather than percentage of positive cells. J. Clin. Oncol. 2011;29:2978–2984. doi: 10.1200/JCO.2010.32.9706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hammond M.E.H., Hayes D.F., Dowsett M., Allred D.C., Hagerty K.L., Badve S. American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J. Clin. Oncol. 2010;28:2784–2795. doi: 10.1200/JCO.2009.25.6529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Khoshnoud M.R., Löfdahl B., Fohlin H., Fornander T., Stål O., Skoog L. Immunohistochemistry compared to cytosol assays for determination of estrogen receptor and prediction of the long-term effect of adjuvant tamoxifen. Breast Cancer Res. Treat. 2011;126:421–430. doi: 10.1007/s10549-010-1202-7. [DOI] [PubMed] [Google Scholar]
- 11.Jacobs T.W., Gown A.M., Yaziji H., Barnes M.J., Schnitt S.J. Specificity of HercepTest in determining HER-2/neu status of breast cancers using the United States Food and Drug Administration-approved scoring system. J. Clin. Oncol. 1999;17:1983–1987. doi: 10.1200/JCO.1999.17.7.1983. [DOI] [PubMed] [Google Scholar]
- 12.Pauletti G., Godolphin W., Press M.F., Slamon D.J. Detection and quantitation of HER-2/neu gene amplification in human breast cancer archival material using fluorescence in situ hybridization. Oncogene. 1996;13:63–72. [PubMed] [Google Scholar]
- 13.Press M.F., Bernstein L., Thomas P.A., Meisner L.F., Zhou J.Y., Ma Y. HER-2/neu gene amplification characterized by fluorescence in situ hybridization: poor prognosis in node-negative breast carcinomas. J. Clin. Oncol. 1997;15:2894–2904. doi: 10.1200/JCO.1997.15.8.2894. [DOI] [PubMed] [Google Scholar]
- 14.Tubbs R.R., Pettay J.D., Roche P.C., Stoler M.H., Jenkins R.B., Grogan T.M. Discrepancies in clinical laboratory testing of eligibility for trastuzumab therapy: apparent immunohistochemical false-positives do not get the message. J. Clin. Oncol. 2001;19:2714–2721. doi: 10.1200/JCO.2001.19.10.2714. [DOI] [PubMed] [Google Scholar]
- 15.Lebeau A., Deimling D., Kaltz C., Sendelhofert A., Iff A., Luthardt B. Her-2/neu analysis in archival tissue samples of human breast cancer: comparison of immunohistochemistry and fluorescence in situ hybridization. J. Clin. Oncol. 2001;19:354–363. doi: 10.1200/JCO.2001.19.2.354. ( http://www.ncbi.nlm.nih.gov/pubmed/11208826) [DOI] [PubMed] [Google Scholar]
- 16.Mass R., Sanders C., Charlene K. The Concordance Between the Clinical Trials Assay (CTA) and Fluorescence in Situ Hybridization (FISH) in the Herceptin Pivotal Trials. Am. Soc. Clin. Oncol. 2000:A291. ( http://life.nthu.edu.tw/~b821605/herceptinew/ab291.htm) [Google Scholar]
- 17.Rakha E.A., Starczynski J., Lee A.H.S., Ellis I.O. The updated ASCO/CAP guideline recommendations for HER2 testing in the management of invasive breast cancer: a critical review of their implications for routine practice. Histopathology. 2014;64:609–615. doi: 10.1111/his.12357. [DOI] [PubMed] [Google Scholar]
- 18.Viale G., Rotmensz N., Maisonneuve P., Bottiglieri L., Montagna E., Luini A. Invasive ductal carcinoma of the breast with the “triple-negative” phenotype: prognostic implications of EGFR immunoreactivity. Breast Cancer Res. Treat. 2009;116:317–328. doi: 10.1007/s10549-008-0206-z. [DOI] [PubMed] [Google Scholar]
- 19.Haffty B.G., Yang Q., Reiss M., Kearney T., Higgins S.A., Weidhaas J. Locoregional relapse and distant metastasis in conservatively managed triple negative early-stage breast cancer. J. Clin. Oncol. 2006;24:5652–5657. doi: 10.1200/JCO.2006.06.5664. [DOI] [PubMed] [Google Scholar]
- 20.Anders C., Carey L.A. Understanding and treating triple-negative breast cancer. Oncology (Williston Park) 2008;22:1233–1239. (discussion 1239–40, 1243. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2868264&tool=pmcentrez&rendertype=abstract) [PMC free article] [PubMed] [Google Scholar]
- 21.Anders C.K., Carey L.A. Biology, metastatic patterns, and treatment of patients with triple-negative breast cancer. Clin. Breast Cancer. 2009;9(Suppl. 2):S73–S81. doi: 10.3816/CBC.2009.s.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Morris G.J., Naidu S., Topham A.K., Guiles F., Xu Y., McCue P. Differences in breast carcinoma characteristics in newly diagnosed African-American and Caucasian patients: a single-institution compilation compared with the National Cancer Institute’s Surveillance, Epidemiology, and End Results database. Cancer. 2007;110:876–884. doi: 10.1002/cncr.22836. [DOI] [PubMed] [Google Scholar]
- 23.Carey L.A., Perou C.M., Livasy C.A., Dressler L.G., Cowan D., Conway K. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006;295:2492–2502. doi: 10.1001/jama.295.21.2492. [DOI] [PubMed] [Google Scholar]
- 24.Dent R., Trudeau M., Pritchard K.I., Hanna W.M., Kahn H.K., Sawka C.A. Triple-negative breast cancer: clinical features and patterns of recurrence. Clin. Cancer Res. 2007;13:4429–4434. doi: 10.1158/1078-0432.CCR-06-3045. [DOI] [PubMed] [Google Scholar]
- 25.Perou C.M., Sørlie T., Eisen M.B., van de Rijn M., Jeffrey S.S., Rees C.A. Molecular portraits of human breast tumours. Nature. 2000;406:747–752. doi: 10.1038/35021093. [DOI] [PubMed] [Google Scholar]
- 26.Prat A., Parker J.S., Karginova O., Fan C., Livasy C., Herschkowitz J.I. Phenotypic and molecular characterization of the claudin-low intrinsic subtype of breast cancer. Breast Cancer Res. 2010;12:R68. doi: 10.1186/bcr2635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Creighton C.J., Li X., Landis M., Dixon J.M., Neumeister V.M., Sjolund A. Residual breast cancers after conventional therapy display mesenchymal as well as tumor-initiating features. Proc. Natl. Acad. Sci. U. S. A. 2009;106:13820–13825. doi: 10.1073/pnas.0905718106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rakha E.A., Reis-Filho J.S., Ellis I.O. Basal-like breast cancer: a critical review. J. Clin. Oncol. 2008;26:2568–2581. doi: 10.1200/JCO.2007.13.1748. [DOI] [PubMed] [Google Scholar]
- 29.Sørlie T., Perou C.M., Tibshirani R., Aas T., Geisler S., Johnsen H. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. U. S. A. 2001;98:10869–10874. doi: 10.1073/pnas.191367098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lehmann B.D., Bauer J. a, Chen X., Sanders M.E., Chakravarthy a B., Shyr Y. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J. Clin. Invest. 2011;121:2750–2767. doi: 10.1172/JCI45014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chacón R.D., Costanzo M.V. Triple-negative breast cancer. Breast Cancer Res. 2010;12(Suppl. 2):S3. doi: 10.1186/bcr2574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hudis C. a, Gianni L. Triple-negative breast cancer: an unmet medical need. Oncologist. 2011;16(Suppl. 1):1–11. doi: 10.1634/theoncologist.2011-S1-01. [DOI] [PubMed] [Google Scholar]
- 33.Cheang M.C.U., Voduc D., Bajdik C., Leung S., McKinney S., Chia S.K. Basal-like breast cancer defined by five biomarkers has superior prognostic value than triple-negative phenotype. Clin. Cancer Res. 2008;14:1368–1376. doi: 10.1158/1078-0432.CCR-07-1658. [DOI] [PubMed] [Google Scholar]
- 34.Sorlie T., Tibshirani R., Parker J., Hastie T., Marron J.S., Nobel A. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc. Natl. Acad. Sci. U. S. A. 2003;100:8418–8423. doi: 10.1073/pnas.0932692100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.de Ruijter T.C., Veeck J., de Hoon J.P.J., van Engeland M., Tjan-Heijnen V.C. Characteristics of triple-negative breast cancer. J. Cancer Res. Clin. Oncol. 2011;137:183–192. doi: 10.1007/s00432-010-0957-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Foulkes W.D., Stefansson I.M., Chappuis P.O., Bégin L.R., Goffin J.R., Wong N. Germline BRCA1 mutations and a basal epithelial phenotype in breast cancer. J. Natl. Cancer Inst. 2003;95:1482–1485. doi: 10.1093/jnci/djg050. ( http://www.ncbi.nlm.nih.gov/pubmed/14519755) [DOI] [PubMed] [Google Scholar]
- 37.Lakhani S.R., Reis-Filho J.S., Fulford L., Penault-Llorca F., van der Vijver M., Parry S. Prediction of BRCA1 status in patients with breast cancer using estrogen receptor and basal phenotype. Clin. Cancer Res. 2005;11:5175–5180. doi: 10.1158/1078-0432.CCR-04-2424. [DOI] [PubMed] [Google Scholar]
- 38.Gonzalez-Angulo A.M., Timms K.M., Liu S., Chen H., Litton J.K., Potter J. Incidence and outcome of BRCA mutations in unselected patients with triple receptor-negative breast cancer. Clin. Cancer Res. 2011;17:1082–1089. doi: 10.1158/1078-0432.CCR-10-2560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Turner N., Tutt A., Ashworth A. Hallmarks of “BRCAness” in sporadic cancers. Nat. Rev. Cancer. 2004;4:814–819. doi: 10.1038/nrc1457. [DOI] [PubMed] [Google Scholar]
- 40.Overgaard J., Yilmaz M., Guldberg P., Hansen L.L., Alsner J. TP53 mutation is an independent prognostic marker for poor outcome in both node-negative and node-positive breast cancer. Acta Oncol. 2000;39:327–333. doi: 10.1080/028418600750013096. ( http://www.ncbi.nlm.nih.gov/pubmed/10987229) [DOI] [PubMed] [Google Scholar]
- 41.D’Andrea A.D., Grompe M. The Fanconi anaemia/BRCA pathway. Nat. Rev. Cancer. 2003;3:23–34. doi: 10.1038/nrc970. [DOI] [PubMed] [Google Scholar]
- 42.Hoeijmakers J.H. Genome maintenance mechanisms for preventing cancer. Nature. 2001;411:366–374. doi: 10.1038/35077232. [DOI] [PubMed] [Google Scholar]
- 43.Farmer H., McCabe N., Lord C.J., Tutt A.N.J., Johnson D.A., Richardson T.B. Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature. 2005;434:917–921. doi: 10.1038/nature03445. [DOI] [PubMed] [Google Scholar]
- 44.Fong P.C., Boss D.S., Yap T.A., Tutt A., Wu P., Mergui-Roelvink M. Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N. Engl. J. Med. 2009;361:123–134. doi: 10.1056/NEJMoa0900212. [DOI] [PubMed] [Google Scholar]
- 45.Tutt A., Robson M., Garber J.E., Domchek S., Audeh M.W., Weitzel J.N. Vol. 27. 2009. Phase II trial of the oral PARP inhibitor olaparib in BRCA-deficient advanced breast cancer; p. CRA501. (ASCO, Meet. Abstr.). ( http://hwmaint.meeting.ascopubs.org/cgi/content/abstract/27/18S/CRA501) [Google Scholar]
- 46.Tutt A., Robson M., Garber J.E., Domchek S.M., Audeh M.W., Weitzel J.N. Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and advanced breast cancer: a proof-of-concept trial. Lancet. 2010;376:235–244. doi: 10.1016/S0140-6736(10)60892-6. [DOI] [PubMed] [Google Scholar]
- 47.Gelmon K.A., Tischkowitz M., Mackay H., Swenerton K., Robidoux A., Tonkin K. Olaparib in patients with recurrent high-grade serous or poorly differentiated ovarian carcinoma or triple-negative breast cancer: a phase 2, multicentre, open-label, non-randomised study. Lancet Oncol. 2011;12:852–861. doi: 10.1016/S1470-2045(11)70214-5. [DOI] [PubMed] [Google Scholar]
- 48.O’Shaughnessy J., Osborne C., Pippen J.E., Yoffe M., Patt D., Rocha C. Iniparib plus chemotherapy in metastatic triple-negative breast cancer. N. Engl. J. Med. 2011;364:205–214. doi: 10.1056/NEJMoa1011418. [DOI] [PubMed] [Google Scholar]
- 49.O’Shaughnessy J., Schwartzberg L., Danso M., Rugo H., Miller K., Yardley D. 2011. A randomized phase III study of iniparib (BSI-201) in combination with gemcitabine/carboplatin (G/C) in metastatic triple-negative breast cancer (TNBC) (| 2011 ASCO Annual Meeting | Abstracts | Meeting Library, J. Clin. Oncol.). ( http://meetinglibrary.asco.org/content/78038-102) [Google Scholar]
- 50.Isakoff S.J., Overmoyer B., Tung N.M., Gelman R.S., Giranda V.L., Bernhard K.M. Vol. 28. 2010. A phase II trial of the PARP inhibitor veliparib (ABT888) and temozolomide for metastatic breast cancer; p. 1019. (ASCO, Meet. Abstr.). ( http://hwmaint.meeting.ascopubs.org/cgi/content/abstract/28/15_suppl/1019) [Google Scholar]
- 51.Dent R.A., Lindeman G.J., Clemons M., Wildiers H., Chan A., McCarthy N.J. Phase I trial of the oral PARP inhibitor olaparib in combination with paclitaxel for first- or second-line treatment of patients with metastatic triple-negative breast cancer. Breast Cancer Res. 2013;15:R88. doi: 10.1186/bcr3484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tan A., Gibbon D., Stein M., Moss R., Karantza V., Lin H. Preliminary results of a phase I trial of ABT-888, a poly(ADP-ribose) polymerase (PARP) inhibitor, in combination with cyclophosphamide. J. Clin. Oncol. 2010;28(15):3000. ( http://meetinglibrary.asco.org/content/43189-74) [Google Scholar]
- 53.Gelmon K., Dent R., Mackey J.R., Laing K., McLeod D., Verma S. Targeting triple-negative breast cancer: optimising therapeutic outcomes. Ann. Oncol. 2012;23:2223–2234. doi: 10.1093/annonc/mds067. [DOI] [PubMed] [Google Scholar]
- 54.Bhattacharyya G.S., Basu S., Agarwal V., Malhotra H., Pareekh P.M., Babu K.G. 41LBA Single institute phase II study of weekly cisplatinum and metronomic dosing of endoxan and methotrexate in second line metastatic breast cancer triple-negative. Eur. J. Cancer. 2009;(Suppl. 7):18–19. [Google Scholar]
- 55.Carey L.A., Rugo H.S., Marcom P.K., Mayer E.L., Esteva F.J., Ma C.X. TBCRC 001: randomized phase II study of cetuximab in combination with carboplatin in stage IV triple-negative breast cancer. J. Clin. Oncol. 2012;30:2615–2623. doi: 10.1200/JCO.2010.34.5579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Alba E., Chacon J.I., Lluch A., Anton A., Estevez L., Cirauqui B. A randomized phase II trial of platinum salts in basal-like breast cancer patients in the neoadjuvant setting. Results from the GEICAM/2006-03, multicenter study. Breast Cancer Res. Treat. 2012;136:487–493. doi: 10.1007/s10549-012-2100-y. [DOI] [PubMed] [Google Scholar]
- 57.Pal S.K., Childs B.H., Pegram M. Triple negative breast cancer: unmet medical needs. Breast Cancer Res. Treat. 2011;125:627–636. doi: 10.1007/s10549-010-1293-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Frasci G., Comella P., Rinaldo M., Iodice G., Di Bonito M., D’Aiuto M. Preoperative weekly cisplatin-epirubicin-paclitaxel with G-CSF support in triple-negative large operable breast cancer. Ann. Oncol. 2009;20:1185–1192. doi: 10.1093/annonc/mdn748. [DOI] [PubMed] [Google Scholar]
- 59.Liu M., Mo Q.-G., Wei C.-Y., Qin Q.-H., Huang Z., He J. Platinum-based chemotherapy in triple-negative breast cancer: A meta-analysis. Oncol. Lett. 2013;5:983–991. doi: 10.3892/ol.2012.1093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sikov W.M., Berry D.A., Perou C.M., Singh B., Cirrincione C.T., Tolaney S.M. Impact of the Addition of Carboplatin and/or Bevacizumab to Neoadjuvant Once-per-Week Paclitaxel Followed by Dose-Dense Doxorubicin and Cyclophosphamide on Pathologic Complete Response Rates in Stage II to III Triple-Negative Breast Cancer: CALGB 40603. Am. J. Clin. Oncol. 2014 doi: 10.1200/JCO.2014.57.0572. (JCO.2014.57.0572–) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Fornier M., Fumoleau P. The paradox of triple negative breast cancer: novel approaches to treatment. Breast J. 2012;18:41–51. doi: 10.1111/j.1524-4741.2011.01175.x. [DOI] [PubMed] [Google Scholar]
- 62.Thomas E.S., Gomez H.L., Li R.K., Chung H.-C., Fein L.E., Chan V.F. Ixabepilone plus capecitabine for metastatic breast cancer progressing after anthracycline and taxane treatment. J. Clin. Oncol. 2007;25:5210–5217. doi: 10.1200/JCO.2007.12.6557. [DOI] [PubMed] [Google Scholar]
- 63.Bunnell C., Vahdat L., Schwartzberg L., Gralow J., Klimovsky J., Poulart V. Phase I/II study of ixabepilone plus capecitabine in anthracycline-pretreated/resistant and taxane-resistant metastatic breast cancer. Clin. Breast Cancer. 2008;8:234–241. doi: 10.3816/CBC.2008.n.026. [DOI] [PubMed] [Google Scholar]
- 64.Rugo H., Roche H., Thomas E., Blackwell K., Chung H., Lerzo G. Ixabepilone plus capecitabine vs. capecitabine in patients with triple negative tumors: a pooled analysis of patients from two large phase III clinical studies [abstract 3057] Cancer Res. 2009;69:225. [Google Scholar]
- 65.Linderholm B.K., Hellborg H., Johansson U., Elmberger G., Skoog L., Lehtiö J. Significantly higher levels of vascular endothelial growth factor (VEGF) and shorter survival times for patients with primary operable triple-negative breast cancer. Ann. Oncol. 2009;20:1639–1646. doi: 10.1093/annonc/mdp062. [DOI] [PubMed] [Google Scholar]
- 66.Burstein H.J., Elias A.D., Rugo H.S., Cobleigh M.A., Wolff A.C., Eisenberg P.D. Phase II study of sunitinib malate, an oral multitargeted tyrosine kinase inhibitor, in patients with metastatic breast cancer previously treated with an anthracycline and a taxane. J. Clin. Oncol. 2008;26:1810–1816. doi: 10.1200/JCO.2007.14.5375. [DOI] [PubMed] [Google Scholar]
- 67.Nielsen T.O., Hsu F.D., Jensen K., Cheang M., Karaca G., Hu Z. Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin. Cancer Res. 2004;10:5367–5374. doi: 10.1158/1078-0432.CCR-04-0220. [DOI] [PubMed] [Google Scholar]
- 68.Finn R.S., Dering J., Ginther C., Wilson C.A., Glaspy P., Tchekmedyian N. Dasatinib, an orally active small molecule inhibitor of both the src and abl kinases, selectively inhibits growth of basal-type/“triple-negative” breast cancer cell lines growing in vitro. Breast Cancer Res. Treat. 2007;105:319–326. doi: 10.1007/s10549-006-9463-x. [DOI] [PubMed] [Google Scholar]
- 69.Ellard S.L., Clemons M., Gelmon K.A., Norris B., Kennecke H., Chia S. Randomized phase II study comparing two schedules of everolimus in patients with recurrent/metastatic breast cancer: NCIC Clinical Trials Group IND.163. J. Clin. Oncol. 2009;27:4536–4541. doi: 10.1200/JCO.2008.21.3033. [DOI] [PubMed] [Google Scholar]
- 70.O’Shaughnessy J., Dieras V., Glaspy J., Brufsky A., Miller K., Miles D. Comparison of subgroup analyses of PFS from three phase III studies of bevacizumab in combination with chemotherapy in patients with HER2-negative metastatic breast cancer (MBC) [Astract 207] Cancer Res. 2009;69 [Google Scholar]
- 71.von Minckwitz G., Eidtmann H., Rezai M., Fasching P.A., Tesch H., Eggemann H. Neoadjuvant chemotherapy and bevacizumab for HER2-negative breast cancer. N. Engl. J. Med. 2012;366:299–309. doi: 10.1056/NEJMoa1111065. [DOI] [PubMed] [Google Scholar]
- 72.Bear H.D., Tang G., Rastogi P., Geyer C.E., Robidoux A., Atkins J.N. Bevacizumab added to neoadjuvant chemotherapy for breast cancer. N. Engl. J. Med. 2012;366:310–320. doi: 10.1056/NEJMoa1111097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Crown J., O’Shaughnessy J., Gullo G. Emerging targeted therapies in triple-negative breast cancer. Ann. Oncol. 2012;23(Suppl. 6):vi56–vi65. doi: 10.1093/annonc/mds196. [DOI] [PubMed] [Google Scholar]
- 74.Goozner M. Avastin hearing leads to more uncertainty over drug’s future. J. Natl. Cancer Inst. 2011;103:1148–1150. doi: 10.1093/jnci/djr293. [DOI] [PubMed] [Google Scholar]
- 75.Ranpura V., Hapani S., Wu S. Treatment-related mortality with bevacizumab in cancer patients: a meta-analysis. JAMA. 2011;305:487–494. doi: 10.1001/jama.2011.51. [DOI] [PubMed] [Google Scholar]
- 76.Baselga J., Gomez P., Awada A., Greil R., Braga S., Climent M. The addition of cetuximab to cisplatin increases overall response rate (ORR) and progression free survival (PFS) in metastatic triple-negative breast cancer (TNBC): results of a randomized phase II study (BALI-1) Ann. Oncol. 2010;21:viii96–viii121. [Google Scholar]
- 77.O’Shaughnessy J., Weckstein D., Vukelja S., McIntyre K., Krekow L., Holmes F. Randomized phase II study of weekly irinotecan/carboplatin with or without cetuximab in patients with metastatic breast cancer [Abstract 308] Breast Cancer Res. Treat. 2007;106 [Google Scholar]
- 78.Carey L., O’Shaughnessy J., Hoadley K., Khambata-Ford S., Horak C., Xu L. Potential predictive markers of benefit from cetuximab in metastatic breast cancer: an analysis of two randomized phase 2 trials [Abstract 2014] Cancer Res. 2009;69 [Google Scholar]
- 79.Finn R.S., Bengala C., Ibrahim N., Roché H., Sparano J., Strauss L.C. Dasatinib as a single agent in triple-negative breast cancer: results of an open-label phase 2 study. Clin. Cancer Res. 2011;17:6905–6913. doi: 10.1158/1078-0432.CCR-11-0288. [DOI] [PubMed] [Google Scholar]
- 80.Brower V. Search for new treatments intensifies for triple-negative breast cancer. J. Natl. Cancer Inst. 2009;101:1536–1537. doi: 10.1093/jnci/djp422. [DOI] [PubMed] [Google Scholar]
- 81.Bergh J., Bondarenko I.M., Lichinitser M.R., Liljegren A., Greil R., Voytko N.L. First-line treatment of advanced breast cancer with sunitinib in combination with docetaxel versus docetaxel alone: results of a prospective, randomized phase III study. J. Clin. Oncol. 2012;30:921–929. doi: 10.1200/JCO.2011.35.7376. [DOI] [PubMed] [Google Scholar]
- 82.Crown J., Dieras V., Staroslawska E., Yardley D.A., Davidson N., Bachelot T.D. Vol. 28. 2010. Phase III trial of sunitinib (SU) in combination with capecitabine (C) versus C in previously treated advanced breast cancer (ABC) p. LBA1011. (ASCO Meet. Abstr.). ( http://hwmaint.meeting.ascopubs.org/cgi/content/abstract/28/18_suppl/LBA1011) [Google Scholar]
- 83.Sun T., Aceto N., Meerbrey K.L., Kessler J.D., Zhou C., Migliaccio I. Activation of multiple proto-oncogenic tyrosine kinases in breast cancer via loss of the PTPN12 phosphatase. Cell. 2011;144:703–718. doi: 10.1016/j.cell.2011.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Meric-Bernstam F., Gonzalez-Angulo A.M. Targeting the mTOR signaling network for cancer therapy. J. Clin. Oncol. 2009;27:2278–2287. doi: 10.1200/JCO.2008.20.0766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Minami C.A., Chung D.U., Chang H.R. Management options in triple-negative breast cancer. Breast Cancer (Auckl.) 2011;5:175–199. doi: 10.4137/BCBCR.S6562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hennessy B.T., Gonzalez-Angulo A.-M., Stemke-Hale K., Gilcrease M.Z., Krishnamurthy S., Lee J.-S. Characterization of a naturally occurring breast cancer subset enriched in epithelial-to-mesenchymal transition and stem cell characteristics. Cancer Res. 2009;69:4116–4124. doi: 10.1158/0008-5472.CAN-08-3441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Gonzalez-Angulo A., Green M., Murray J., Palla S., Koenig K., Brewster A. Open label, randomized clinical trial of standard neoadjuvant chemotherapy with paclitaxel followed by FEC (T-FEC) versus the combination of paclitaxel and RAD001 followed by FEC (TR-FEC) in women with triple receptor-negative breast cancer (TNBC) J. Clin. Oncol. 2011;29 doi: 10.1093/annonc/mdu124. (Abstr 1016, presented data—ASCO Annual Meeting 2011. http://meetinglibrary.asco.org/content/77698-102) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Wu Y., Sarkissyan M., Elshimali Y., Vadgama J.V. Triple negative breast tumors in African-American and Hispanic/Latina women are high in CD44 +, low in CD24 +, and have loss of PTEN. PLoS One. 2013;8:e78259. doi: 10.1371/journal.pone.0078259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Neshat M.S., Mellinghoff I.K., Tran C., Stiles B., Thomas G., Petersen R. Enhanced sensitivity of PTEN-deficient tumors to inhibition of FRAP/mTOR. Proc. Natl. Acad. Sci. U. S. A. 2001;98:10314–10319. doi: 10.1073/pnas.171076798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Steelman L.S., Navolanic P.M., Sokolosky M.L., Taylor J.R., Lehmann B.D., Chappell W.H. Suppression of PTEN function increases breast cancer chemotherapeutic drug resistance while conferring sensitivity to mTOR inhibitors. Oncogene. 2008;27:4086–4095. doi: 10.1038/onc.2008.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Noh W.-C., Mondesire W.H., Peng J., Jian W., Zhang H., Dong J. Determinants of rapamycin sensitivity in breast cancer cells. Clin. Cancer Res. 2004;10:1013–1023. doi: 10.1158/1078-0432.ccr-03-0043. ( http://www.ncbi.nlm.nih.gov/pubmed/14871980) [DOI] [PubMed] [Google Scholar]
- 92.Gucalp A., Traina T.A. Triple-negative breast cancer: role of the androgen receptor. Cancer J. 2010;16 doi: 10.1097/PPO.0b013e3181ce4ae1. 62–5. http://dx.doi.org/10.1097/PPO.0b013e3181ce4ae1. [DOI] [PubMed] [Google Scholar]
- 93.Doane A.S., Danso M., Lal P., Donaton M., Zhang L., Hudis C. An estrogen receptor-negative breast cancer subset characterized by a hormonally regulated transcriptional program and response to androgen. Oncogene. 2006;25:3994–4008. doi: 10.1038/sj.onc.1209415. [DOI] [PubMed] [Google Scholar]
- 94.Park S., Koo J., Park H.S., Kim J.-H., Choi S.-Y., Lee J.H. Expression of androgen receptors in primary breast cancer. Ann. Oncol. 2010;21:488–492. doi: 10.1093/annonc/mdp510. [DOI] [PubMed] [Google Scholar]
- 95.Gonzalez-Angulo A.M., Stemke-Hale K., Palla S.L., Carey M., Agarwal R., Meric-Berstam F. Androgen receptor levels and association with PIK3CA mutations and prognosis in breast cancer. Clin. Cancer Res. 2009;15:2472–2478. doi: 10.1158/1078-0432.CCR-08-1763. [DOI] [PubMed] [Google Scholar]
- 96.Traina T., Wolff A., Giri D., Feigin K., Patil S., Yuan J. 2009. Androgen receptor inhibition for the treatment of AR +/ER-/PR- metastatic breast cancer [Abstract 251] (ASCO Breast Cancer Symp.). [Google Scholar]
- 97.Brehm A., Miska E.A., McCance D.J., Reid J.L., Bannister A.J., Kouzarides T. Retinoblastoma protein recruits histone deacetylase to repress transcription. Nature. 1998;391:597–601. doi: 10.1038/35404. [DOI] [PubMed] [Google Scholar]
- 98.Uehara N., Kanematsu S., Miki H., Yoshizawa K., Tsubura A. Requirement of p38 MAPK for a cell-death pathway triggered by vorinostat in MDA-MB-231 human breast cancer cells. Cancer Lett. 2012;315:112–121. doi: 10.1016/j.canlet.2011.07.032. [DOI] [PubMed] [Google Scholar]
- 99.Sirois I., Groleau J., Pallet N., Brassard N., Hamelin K., Londono I. Caspase activation regulates the extracellular export of autophagic vacuoles. Autophagy. 2012;8:927–937. doi: 10.4161/auto.19768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Fridman W.H., Pagès F., Sautès-Fridman C., Galon J. The immune contexture in human tumours: impact on clinical outcome. Nat. Rev. Cancer. 2012;12:298–306. doi: 10.1038/nrc3245. [DOI] [PubMed] [Google Scholar]
- 101.de la Cruz-Merino L., Barco-Sánchez A., Henao Carrasco F., Nogales Fernández E., Vallejo Benítez A., Brugal Molina J. New insights into the role of the immune microenvironment in breast carcinoma. Clin. Dev. Immunol. 2013;2013:785317. doi: 10.1155/2013/785317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Denkert C., Loibl S., Noske A., Roller M., Müller B.M., Komor M. Tumor-associated lymphocytes as an independent predictor of response to neoadjuvant chemotherapy in breast cancer. J. Clin. Oncol. 2010;28:105–113. doi: 10.1200/JCO.2009.23.7370. [DOI] [PubMed] [Google Scholar]
- 103.Denkert C., Loibl S., Salat C., Sinn B., Schem C., Endris V. Abstract S1-06: Increased tumor-associated lymphocytes predict benefit from addition of carboplatin to neoadjuvant therapy for triple-negative and HER2-positive early breast cancer in the GeparSixto trial (GBG 66) Cancer Res. 2014;73 (S1–06–S1–06) [Google Scholar]
- 104.Adams S., Gray R.J., Demaria S., Goldstein L., Perez E.A., Shulman L.N. Prognostic Value of Tumor-Infiltrating Lymphocytes in Triple-Negative Breast Cancers From Two Phase III Randomized Adjuvant Breast Cancer Trials: ECOG 2197 and ECOG 1199. J. Clin. Oncol. 2014 doi: 10.1200/JCO.2013.55.0491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Stagg J., Allard B. Immunotherapeutic approaches in triple-negative breast cancer: latest research and clinical prospects. Ther. Adv. Med. Oncol. 2013;5:169–181. doi: 10.1177/1758834012475152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Bertucci F., Finetti P., Cervera N., Charafe-Jauffret E., Mamessier E., Adélaïde J. Gene expression profiling shows medullary breast cancer is a subgroup of basal breast cancers. Cancer Res. 2006;66:4636–4644. doi: 10.1158/0008-5472.CAN-06-0031. [DOI] [PubMed] [Google Scholar]
- 107.Haakensen V.D., Lingjaerde O.C., Lüders T., Riis M., Prat A., Troester M.A. Gene expression profiles of breast biopsies from healthy women identify a group with claudin-low features. BMC Med. Genomics. 2011;4:77. doi: 10.1186/1755-8794-4-77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Arnedos M., Bihan C., Delaloge S., Andre F. Triple-negative breast cancer: are we making headway at least? Ther. Adv. Med. Oncol. 2012;4:195–210. doi: 10.1177/1758834012444711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.O’Toole S.A., Beith J.M., Millar E.K.A., West R., McLean A., Cazet A. Therapeutic targets in triple negative breast cancer. J. Clin. Pathol. 2013;66:530–542. doi: 10.1136/jclinpath-2012-201361. [DOI] [PubMed] [Google Scholar]
- 110.Balko J.M., Giltnane J.M., Wang K., Schwarz L.J., Young C.D., Cook R.S. Molecular profiling of the residual disease of triple-negative breast cancers after neoadjuvant chemotherapy identifies actionable therapeutic targets. Cancer Discov. 2014;4:232–245. doi: 10.1158/2159-8290.CD-13-0286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Janku F., McConkey D.J., Hong D.S., Kurzrock R. Autophagy as a target for anticancer therapy. Nat. Rev. Clin. Oncol. 2011;8:528–539. doi: 10.1038/nrclinonc.2011.71. [DOI] [PubMed] [Google Scholar]
- 112.Saab R. Cellular senescence: many roads, one final destination. ScientificWorldJournal. 2010;10:727–741. doi: 10.1100/tsw.2010.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Degenhardt K., Mathew R., Beaudoin B., Bray K., Anderson D., Chen G. Autophagy promotes tumor cell survival and restricts necrosis, inflammation, and tumorigenesis. Cancer Cell. 2006;10:51–64. doi: 10.1016/j.ccr.2006.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Watson J. a, Watson C.J., McCrohan A.-M., Woodfine K., Tosetto M., McDaid J. Generation of an epigenetic signature by chronic hypoxia in prostate cells. Hum. Mol. Genet. 2009;18:3594–3604. doi: 10.1093/hmg/ddp307. [DOI] [PubMed] [Google Scholar]
- 115.Longley D.B., Johnston P.G. Molecular mechanisms of drug resistance. J. Pathol. 2005;205:275–292. doi: 10.1002/path.1706. [DOI] [PubMed] [Google Scholar]
- 116.Abdullah L.N., Chow E.K.-H. Mechanisms of chemoresistance in cancer stem cells. Clin. Transl. Med. 2013;2:3. doi: 10.1186/2001-1326-2-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Leonessa F., Clarke R. ATP binding cassette transporters and drug resistance in breast cancer. Endocr. Relat. Cancer. 2003;10:43–73. doi: 10.1677/erc.0.0100043. ( http://www.ncbi.nlm.nih.gov/pubmed/12653670) [DOI] [PubMed] [Google Scholar]
- 118.Chow E.K.-H., Fan L., Chen X., Bishop J.M. Oncogene-specific formation of chemoresistant murine hepatic cancer stem cells. Hepatology. 2012;56:1331–1341. doi: 10.1002/hep.25776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Shapiro A.B., Corder A.B., Ling V. P-glycoprotein-mediated Hoechst 33342 transport out of the lipid bilayer. Eur. J. Biochem. 1997;250:115–121. doi: 10.1111/j.1432-1033.1997.00115.x. ( http://www.ncbi.nlm.nih.gov/pubmed/9431998) [DOI] [PubMed] [Google Scholar]
- 120.Scharenberg C.W., Harkey M.A., Torok-Storb B. The ABCG2 transporter is an efficient Hoechst 33342 efflux pump and is preferentially expressed by immature human hematopoietic progenitors. Blood. 2002;99:507–512. doi: 10.1182/blood.v99.2.507. ( http://www.ncbi.nlm.nih.gov/pubmed/11781231) [DOI] [PubMed] [Google Scholar]
- 121.Tommasi S., Mangia A., Lacalamita R., Bellizzi A., Fedele V., Chiriatti A. Cytoskeleton and paclitaxel sensitivity in breast cancer: the role of beta-tubulins. Int. J. Cancer. 2007;120:2078–2085. doi: 10.1002/ijc.22557. [DOI] [PubMed] [Google Scholar]
- 122.Paradiso A., Mangia A., Chiriatti A., Tommasi S., Zito A., Latorre A. Biomarkers predictive for clinical efficacy of taxol-based chemotherapy in advanced breast cancer. Ann. Oncol. 2005;16(Suppl. 4):iv14–iv19. doi: 10.1093/annonc/mdi902. [DOI] [PubMed] [Google Scholar]
- 123.Giaccone G., Pinedo H. Drug Resistance. Oncologist. 1996;1:82–87. ( http://www.ncbi.nlm.nih.gov/pubmed/10387972) [PubMed] [Google Scholar]
- 124.Valkov N.I., Sullivan D.M. Drug resistance to DNA topoisomerase I and II inhibitors in human leukemia, lymphoma, and multiple myeloma. Semin. Hematol. 1997;34:48–62. ( http://www.ncbi.nlm.nih.gov/pubmed/940896) [PubMed] [Google Scholar]
- 125.Fedier A., Schwarz V.A., Walt H., Carpini R.D., Haller U., Fink D. Resistance to topoisomerase poisons due to loss of DNA mismatch repair. Int. J. Cancer. 2001;93:571–576. doi: 10.1002/ijc.1356. ( http://www.ncbi.nlm.nih.gov/pubmed/11477562) [DOI] [PubMed] [Google Scholar]
- 126.Végran F., Boidot R., Oudin C., Riedinger J.-M., Bonnetain F., Lizard-Nacol S. Overexpression of caspase-3 s splice variant in locally advanced breast carcinoma is associated with poor response to neoadjuvant chemotherapy. Clin. Cancer Res. 2006;12:5794–5800. doi: 10.1158/1078-0432.CCR-06-0725. [DOI] [PubMed] [Google Scholar]
- 127.O’Connor P.M., Jackman J., Bae I., Myers T.G., Fan S., Mutoh M. Characterization of the p53 tumor suppressor pathway in cell lines of the National Cancer Institute anticancer drug screen and correlations with the growth-inhibitory potency of 123 anticancer agents. Cancer Res. 1997;57:4285–4300. ( http://www.ncbi.nlm.nih.gov/pubmed/9331090) [PubMed] [Google Scholar]
- 128.Aas T., Børresen A.L., Geisler S., Smith-Sørensen B., Johnsen H., Varhaug J.E. Specific P53 mutations are associated with de novo resistance to doxorubicin in breast cancer patients. Nat. Med. 1996;2:811–814. doi: 10.1038/nm0796-811. ( http://www.ncbi.nlm.nih.gov/pubmed/8673929) [DOI] [PubMed] [Google Scholar]
- 129.Andersson J., Larsson L., Klaar S., Holmberg L., Nilsson J., Inganäs M. Worse survival for TP53 (p53)-mutated breast cancer patients receiving adjuvant CMF. Ann. Oncol. 2005;16:743–748. doi: 10.1093/annonc/mdi150. [DOI] [PubMed] [Google Scholar]
- 130.Dumontet C., Sikic B.I. Mechanisms of action of and resistance to antitubulin agents: microtubule dynamics, drug transport, and cell death. J. Clin. Oncol. 1999;17:1061–1070. doi: 10.1200/JCO.1999.17.3.1061. ( http://www.ncbi.nlm.nih.gov/pubmed/10071301) [DOI] [PubMed] [Google Scholar]
- 131.Sládek N.E., Kollander R., Sreerama L., Kiang D.T. Cellular levels of aldehyde dehydrogenases (ALDH1A1 and ALDH3A1) as predictors of therapeutic responses to cyclophosphamide-based chemotherapy of breast cancer: a retrospective study. Rational individualization of oxazaphosphorine-based cancer chemotherap. Cancer Chemother. Pharmacol. 2002;49:309–321. doi: 10.1007/s00280-001-0412-4. [DOI] [PubMed] [Google Scholar]
- 132.DeGregorio M., Perez E. Molecular mechanisms of drug resistance. Cecil Textb. Med. 1996:1056–1060. [Google Scholar]
- 133.Bedard P.L., Di Leo A., Piccart-Gebhart M.J. Taxanes: optimizing adjuvant chemotherapy for early-stage breast cancer. Nat. Rev. Clin. Oncol. 2010;7:22–36. doi: 10.1038/nrclinonc.2009.186. [DOI] [PubMed] [Google Scholar]
- 134.Yip N.C., Fombon I.S., Liu P., Brown S., Kannappan V., Armesilla A.L. Disulfiram modulated ROS-MAPK and NFκB pathways and targeted breast cancer cells with cancer stem cell-like properties. Br. J. Cancer. 2011;104:1564–1574. doi: 10.1038/bjc.2011.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Singel S.M., Cornelius C., Zaganjor E., Batten K., Sarode V.R., Buckley D.L. KIF14 promotes AKT phosphorylation and contributes to chemoresistance in triple-negative breast cancer. Neoplasia. 2014;16(247–56):256.e2. doi: 10.1016/j.neo.2014.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Chekhun V.F., Zhylchuk V.E., Lukyanova N.Y., Vorontsova A.L., Kudryavets Y.I. Expression of drug resistance proteins in triple-receptor-negative tumors as the basis of individualized therapy of the breast cancer patients. Exp. Oncol. 2009;31:123–124. ( http://www.ncbi.nlm.nih.gov/pubmed/19550405) [PubMed] [Google Scholar]
- 137.Zhou F., Yan M., Guo G., Wang F., Qiu H., Zheng F. Knockdown of eIF4E suppresses cell growth and migration, enhances chemosensitivity and correlates with increase in Bax/Bcl-2 ratio in triple-negative breast cancer cells. Med. Oncol. 2011;28:1302–1307. doi: 10.1007/s12032-010-9630-0. [DOI] [PubMed] [Google Scholar]
- 138.Nolen B.M., Marks J.R., Ta’san S., Rand A., Luong T.M., Wang Y. Serum biomarker profiles and response to neoadjuvant chemotherapy for locally advanced breast cancer. Breast Cancer Res. 2008;10:R45. doi: 10.1186/bcr2096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Bauer J.A., Chakravarthy A.B., Rosenbluth J.M., Mi D., Seeley E.H., De Matos Granja-Ingram N. Identification of markers of taxane sensitivity using proteomic and genomic analyses of breast tumors from patients receiving neoadjuvant paclitaxel and radiation. Clin. Cancer Res. 2010;16:681–690. doi: 10.1158/1078-0432.CCR-09-1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Ivanov O., Chen F., Wiley E.L., Keswani A., Diaz L.K., Memmel H.C. alphaB-crystallin is a novel predictor of resistance to neoadjuvant chemotherapy in breast cancer. Breast Cancer Res. Treat. 2008;111:411–417. doi: 10.1007/s10549-007-9796-0. [DOI] [PubMed] [Google Scholar]
- 141.Giricz O., Calvo V., Pero S.C., Krag D.N., Sparano J.A., Kenny P.A. GRB7 is required for triple-negative breast cancer cell invasion and survival. Breast Cancer Res. Treat. 2012;133:607–615. doi: 10.1007/s10549-011-1822-6. [DOI] [PubMed] [Google Scholar]
- 142.Anders C.K., Winer E.P., Ford J.M., Dent R., Silver D.P., Sledge G.W. Poly(ADP-Ribose) polymerase inhibition: “targeted” therapy for triple-negative breast cancer. Clin. Cancer Res. 2010;16:4702–4710. doi: 10.1158/1078-0432.CCR-10-0939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Alli E., Sharma V.B., Sunderesakumar P., Ford J.M. Defective repair of oxidative dna damage in triple-negative breast cancer confers sensitivity to inhibition of poly(ADP-ribose) polymerase. Cancer Res. 2009;69:3589–3596. doi: 10.1158/0008-5472.CAN-08-4016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Graham T.R., Yacoub R., Taliaferro-Smith L., Osunkoya A.O., Odero-Marah V.A., Liu T. Reciprocal regulation of ZEB1 and AR in triple negative breast cancer cells. Breast Cancer Res. Treat. 2010;123:139–147. doi: 10.1007/s10549-009-0623-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Liedtke C., Mazouni C., Hess K.R., André F., Tordai A., Mejia J.A. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J. Clin. Oncol. 2008;26:1275–1281. doi: 10.1200/JCO.2007.14.4147. [DOI] [PubMed] [Google Scholar]
- 146.André F., Zielinski C.C. Optimal strategies for the treatment of metastatic triple-negative breast cancer with currently approved agents. Ann. Oncol. 2012;23(Suppl. 6):vi46–vi51. doi: 10.1093/annonc/mds195. [DOI] [PubMed] [Google Scholar]
- 147.Dalerba P., Cho R.W., Clarke M.F. Cancer stem cells: models and concepts. Annu. Rev. Med. 2007;58:267–284. doi: 10.1146/annurev.med.58.062105.204854. [DOI] [PubMed] [Google Scholar]
- 148.Lapidot T., Sirard C., Vormoor J., Murdoch B., Hoang T., Caceres-Cortes J. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature. 1994;367:645–648. doi: 10.1038/367645a0. [DOI] [PubMed] [Google Scholar]
- 149.Mani S.A., Guo W., Liao M.-J., Eaton E.N., Ayyanan A., Zhou A.Y. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell. 2008;133:704–715. doi: 10.1016/j.cell.2008.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Bhola N., Balko J. TGF-β inhibition enhances chemotherapy action against triple-negative breast cancer. J. Clin. Investig. 2013;123 doi: 10.1172/JCI65416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Samanta D., Gilkes D.M., Chaturvedi P., Xiang L., Semenza G.L. Hypoxia-inducible factors are required for chemotherapy resistance of breast cancer stem cells. Proc. Natl. Acad. Sci. U. S. A. 2014:5429–5438. doi: 10.1073/pnas.1421438111. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 152.Vera-Ramirez L., Sanchez-Rovira P., Ramirez-Tortosa M.C., Ramirez-Tortosa C.L., Granados-Principal S., Lorente J.A. Free radicals in breast carcinogenesis, breast cancer progression and cancer stem cells. Biological bases to develop oxidative-based therapies. Crit. Rev. Oncol. Hematol. 2011;80:347–368. doi: 10.1016/j.critrevonc.2011.01.004. [DOI] [PubMed] [Google Scholar]
- 153.Koo J.S., Jung W. Alteration of REDD1-mediated mammalian target of rapamycin pathway and hypoxia-inducible factor-1α regulation in human breast cancer. Pathobiology. 2010;77:289–300. doi: 10.1159/000320936. [DOI] [PubMed] [Google Scholar]
- 154.Basu S., Chen W., Tchou J., Mavi A., Cermik T., Czerniecki B. Comparison of triple-negative and estrogen receptor-positive/progesterone receptor-positive/HER2-negative breast carcinoma using quantitative fluorine-18 fluorodeoxyglucose/positron emission tomography imaging parameters: a potentially useful method for d. Cancer. 2008;112:995–1000. doi: 10.1002/cncr.23226. [DOI] [PubMed] [Google Scholar]
- 155.Lee S.-J., Kim H.-P., Jin Y., Choi A.M.K., Ryter S.W. Beclin 1 deficiency is associated with increased hypoxia-induced angiogenesis. Autophagy. 2011;7:829–839. doi: 10.4161/auto.7.8.15598. ( http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3149693&tool=pmcentrez&rendertype=abstract) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Cosse J.-P., Michiels C. Tumour hypoxia affects the responsiveness of cancer cells to chemotherapy and promotes cancer progression. Anti Cancer Agents Med. Chem. 2008;8:790–797. doi: 10.2174/187152008785914798. ( http://www.ncbi.nlm.nih.gov/pubmed/18855580) [DOI] [PubMed] [Google Scholar]
- 157.Milane L., Duan Z., Amiji M. Role of hypoxia and glycolysis in the development of multi-drug resistance in human tumor cells and the establishment of an orthotopic multi-drug resistant tumor model in nude mice using hypoxic pre-conditioning. Cancer Cell Int. 2011;11:3. doi: 10.1186/1475-2867-11-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Prencipe M., McGoldrick A., Perry A.S., O’Grady A., Phelan S., McGrogan B. MAD2 downregulation in hypoxia is independent of promoter hypermethylation. Cell Cycle. 2010;9:2856–2865. ( http://www.ncbi.nlm.nih.gov/pubmed/20676051) [PubMed] [Google Scholar]
- 160.Groheux D., Hindié E., Giacchetti S., Delord M., Hamy A.-S., de Roquancourt A. Triple-negative breast cancer: early assessment with 18 F-FDG PET/CT during neoadjuvant chemotherapy identifies patients who are unlikely to achieve a pathologic complete response and are at a high risk of early relapse. J. Nucl. Med. 2012;53:249–254. doi: 10.2967/jnumed.111.094045. [DOI] [PubMed] [Google Scholar]
- 161.Neumeister V.M., Sullivan C. a, Lindner R., Lezon-Geyda K., Li J., Zavada J. Hypoxia-induced protein CAIX is associated with somatic loss of BRCA1 protein and pathway activity in triple negative breast cancer. Breast Cancer Res. Treat. 2012;136:67–75. doi: 10.1007/s10549-012-2232-0. [DOI] [PubMed] [Google Scholar]
- 162.Tan E.Y., Yan M., Campo L., Han C., Takano E., Turley H. The key hypoxia regulated gene CAIX is upregulated in basal-like breast tumours and is associated with resistance to chemotherapy. Br. J. Cancer. 2009;100:405–411. doi: 10.1038/sj.bjc.6604844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Bernardi R., Gianni L. Hallmarks of triple negative breast cancer emerging at last? Cell Res. 2014;24:904–905. doi: 10.1038/cr.2014.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Collado M., Gil J., Efeyan A., Guerra C., Schuhmacher A.J., Barradas M. Tumour biology: senescence in premalignant tumours. Nature. 2005;436:642. doi: 10.1038/436642a. [DOI] [PubMed] [Google Scholar]
- 165.Chakrabarti J., Turley H., Campo L., Han C., Harris A.L., Gatter K.C. The transcription factor DEC1 (stra13, SHARP2) is associated with the hypoxic response and high tumour grade in human breast cancers. Br. J. Cancer. 2004;91:954–958. doi: 10.1038/sj.bjc.6602059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Pare R., Yang T., Shin J.-S., Lee C.S. The significance of the senescence pathway in breast cancer progression. J. Clin. Pathol. 2013;66:491–495. doi: 10.1136/jclinpath-2012-201081. [DOI] [PubMed] [Google Scholar]
- 167.Tchou J., Sonnad S.S., Bergey M.R., Basu S., Tomaszewski J., Alavi A. Degree of tumor FDG uptake correlates with proliferation index in triple negative breast cancer. Mol. Imaging Biol. 2010;12:657–662. doi: 10.1007/s11307-009-0294-0. [DOI] [PubMed] [Google Scholar]
- 168.Keam B., Im S.-A., Koh Y., Han S.-W., Oh D.-Y., Cho N. Early metabolic response using FDG PET/CT and molecular phenotypes of breast cancer treated with neoadjuvant chemotherapy. BMC Cancer. 2011;11:452. doi: 10.1186/1471-2407-11-452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Ueda S., Tsuda H., Asakawa H., Shigekawa T., Fukatsu K., Kondo N. Clinicopathological and prognostic relevance of uptake level using 18 F-fluorodeoxyglucose positron emission tomography/computed tomography fusion imaging (18 F-FDG PET/CT) in primary breast cancer. Jpn. J. Clin. Oncol. 2008;38:250–258. doi: 10.1093/jjco/hyn019. [DOI] [PubMed] [Google Scholar]
- 170.Kim B.S., Sung S.H. Usefulness of 18 F-FDG uptake with clinicopathologic and immunohistochemical prognostic factors in breast cancer. Ann. Nucl. Med. 2012;26:175–183. doi: 10.1007/s12149-011-0556-1. [DOI] [PubMed] [Google Scholar]
- 171.Dogan B.E., Turnbull L.W. Imaging of triple-negative breast cancer. Ann. Oncol. 2012;23(Suppl. 6):vi23–vi29. doi: 10.1093/annonc/mds191. [DOI] [PubMed] [Google Scholar]
- 172.Hammond E.M., Giaccia A.J. The role of p53 in hypoxia-induced apoptosis. Biochem. Biophys. Res. Commun. 2005;331:718–725. doi: 10.1016/j.bbrc.2005.03.154. [DOI] [PubMed] [Google Scholar]
- 173.Welford S.M., Giaccia A.J. Hypoxia and senescence: the impact of oxygenation on tumor suppression. Mol. Cancer Res. 2011;9:538–544. doi: 10.1158/1541-7786.MCR-11-0065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Cao M.D., Sitter B., Bathen T.F., Bofin A., Lønning P.E., Lundgren S. Predicting long-term survival and treatment response in breast cancer patients receiving neoadjuvant chemotherapy by MR metabolic profiling. NMR Biomed. 2012;25:369–378. doi: 10.1002/nbm.1762. [DOI] [PubMed] [Google Scholar]
- 175.Witkiewicz A.K., Whitaker-Menezes D., Dasgupta A., Philp N.J., Lin Z., Gandara R. Using the “reverse Warburg effect” to identify high-risk breast cancer patients: stromal MCT4 predicts poor clinical outcome in triple-negative breast cancers. Cell Cycle. 2012;11:1108–1117. doi: 10.4161/cc.11.6.19530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Ksiazek K., Mikula-Pietrasik J., Korybalska K., Dworacki G., Jörres A., Witowski J. Senescent peritoneal mesothelial cells promote ovarian cancer cell adhesion: the role of oxidative stress-induced fibronectin. Am. J. Pathol. 2009;174:1230–1240. doi: 10.2353/ajpath.2009.080613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Fodil-Bourahla I., Drubaix I., Robert L., Labat-Robert J. Effect of in vitro aging on the modulation of protein and fibronectin biosynthesis by the elastin-laminin receptor in human skin fibroblasts. Gerontology. 1999;45:23–30. doi: 10.1159/000022051. ( http://www.ncbi.nlm.nih.gov/pubmed/9852377) [DOI] [PubMed] [Google Scholar]
- 178.Grillari J., Hohenwarter O., Grabherr R.M., Katinger H. Subtractive hybridization of mRNA from early passage and senescent endothelial cells. Exp. Gerontol. 2000;35:187–197. doi: 10.1016/s0531-5565(00)00080-2. ( http://www.ncbi.nlm.nih.gov/pubmed/10767578) [DOI] [PubMed] [Google Scholar]
- 179.Porter M.B., Pereira-Smith O.M., Smith J.R. Novel monoclonal antibodies identify antigenic determinants unique to cellular senescence. J. Cell. Physiol. 1990;142:425–433. doi: 10.1002/jcp.1041420228. [DOI] [PubMed] [Google Scholar]
- 180.Schulz D.M., Böllner C., Thomas G., Atkinson M., Esposito I., Höfler H. Identification of differentially expressed proteins in triple-negative breast carcinomas using DIGE and mass spectrometry. J. Proteome Res. 2009;8:3430–3438. doi: 10.1021/pr900071h. [DOI] [PubMed] [Google Scholar]
- 181.Saab R. Senescence and pre-malignancy: how do tumors progress? Semin. Cancer Biol. 2011;21:385–391. doi: 10.1016/j.semcancer.2011.09.013. [DOI] [PubMed] [Google Scholar]
- 182.Plath T., Detjen K., Welzel M., von Marschall Z., Murphy D., Schirner M. A novel function for the tumor suppressor p16(INK4a): induction of anoikis via upregulation of the alpha(5)beta(1) fibronectin receptor. J. Cell Biol. 2000;150:1467–1478. doi: 10.1083/jcb.150.6.1467. ( http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2150704&tool=pmcentrez&rendertype=abstract) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Cho S.O., Kim K.H., Yoon J.-H., Kim H. Signaling for integrin alpha5/beta1 expression in Helicobacter pylori-infected gastric epithelial AGS cells. Ann. N. Y. Acad. Sci. 2006;1090:298–304. doi: 10.1196/annals.1378.032. [DOI] [PubMed] [Google Scholar]
- 184.Krtolica A., Campisi J. Cancer and aging: a model for the cancer promoting effects of the aging stroma. Int. J. Biochem. Cell Biol. 2002;34:1401–1414. doi: 10.1016/s1357-2725(02)00053-5. ( http://www.ncbi.nlm.nih.gov/pubmed/12200035) [DOI] [PubMed] [Google Scholar]
- 185.Yang G., Rosen D.G., Zhang Z., Bast R.C., Mills G.B., Colacino J.A. The chemokine growth-regulated oncogene 1 (Gro-1) links RAS signaling to the senescence of stromal fibroblasts and ovarian tumorigenesis. Proc. Natl. Acad. Sci. U. S. A. 2006;103:16472–16477. doi: 10.1073/pnas.0605752103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Nishida K., Yamaguchi O., Otsu K. Crosstalk between autophagy and apoptosis in heart disease. Circ. Res. 2008;103:343–351. doi: 10.1161/CIRCRESAHA.108.175448. [DOI] [PubMed] [Google Scholar]
- 187.Yang Y.-P., Liang Z.-Q., Gu Z.-L., Qin Z.-H. Molecular mechanism and regulation of autophagy. Acta Pharmacol. Sin. 2005;26:1421–1434. doi: 10.1111/j.1745-7254.2005.00235.x. [DOI] [PubMed] [Google Scholar]
- 188.Kunz J.B., Schwarz H., Mayer A. Determination of four sequential stages during microautophagy in vitro. J. Biol. Chem. 2004;279:9987–9996. doi: 10.1074/jbc.M307905200. [DOI] [PubMed] [Google Scholar]
- 189.Dice J.F. Chaperone-mediated autophagy. Autophagy. 2007;3:295–299. doi: 10.4161/auto.4144. ( http://www.ncbi.nlm.nih.gov/pubmed/17404494) [DOI] [PubMed] [Google Scholar]
- 190.Levine B., Yuan J. Autophagy in cell death: an innocent convict? J. Clin. Invest. 2005;115:2679–2688. doi: 10.1172/JCI26390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Thorburn A. Apoptosis and autophagy: regulatory connections between two supposedly different processes. Apoptosis. 2008;13:1–9. doi: 10.1007/s10495-007-0154-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Hoare M., Young A.R.J., Narita M. Autophagy in cancer: having your cake and eating it. Semin. Cancer Biol. 2011;21:397–404. doi: 10.1016/j.semcancer.2011.09.004. [DOI] [PubMed] [Google Scholar]
- 193.Martinet W., Agostinis P., Vanhoecke B., Dewaele M., De Meyer G.R.Y. Autophagy in disease: a double-edged sword with therapeutic potential. Clin. Sci. (Lond.) 2009;116:697–712. doi: 10.1042/CS20080508. [DOI] [PubMed] [Google Scholar]
- 194.Swampillai A.L., Salomoni P., Short S.C. The role of autophagy in clinical practice. Clin. Oncol. (R. Coll. Radiol.) 2012;24:387–395. doi: 10.1016/j.clon.2011.09.010. [DOI] [PubMed] [Google Scholar]
- 195.Rikiishi H. Novel Insights into the Interplay between Apoptosis and Autophagy. Int. J. Cell Biol. 2012;2012:317645. doi: 10.1155/2012/317645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Notte A., Leclere L., Michiels C. Autophagy as a mediator of chemotherapy-induced cell death in cancer. Biochem. Pharmacol. 2011;82:427–434. doi: 10.1016/j.bcp.2011.06.015. [DOI] [PubMed] [Google Scholar]
- 197.Mathew R., Karantza-Wadsworth V., White E. Role of autophagy in cancer. Nat. Rev. Cancer. 2007;7:961–967. doi: 10.1038/nrc2254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Lorin S., Hamaï A., Mehrpour M., Codogno P. Autophagy regulation and its role in cancer. Semin. Cancer Biol. 2013;23:361–379. doi: 10.1016/j.semcancer.2013.06.007. [DOI] [PubMed] [Google Scholar]
- 199.Eng C.H., Abraham R.T. The autophagy conundrum in cancer: influence of tumorigenic metabolic reprogramming. Oncogene. 2011;30:4687–4696. doi: 10.1038/onc.2011.220. [DOI] [PubMed] [Google Scholar]
- 200.Essick E.E., Sam F. Oxidative stress and autophagy in cardiac disease, neurological disorders, aging and cancer. Oxidative Med. Cell. Longev. 2010;3:168–177. doi: 10.4161/oxim.3.3.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Martinez-Outschoorn U.E., Pavlides S., Howell A., Pestell R.G., Tanowitz H.B., Sotgia F. Stromal-epithelial metabolic coupling in cancer: integrating autophagy and metabolism in the tumor microenvironment. Int. J. Biochem. Cell Biol. 2011;43:1045–1051. doi: 10.1016/j.biocel.2011.01.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Xi G., Hu X., Wu B., Jiang H., Young C.Y.F., Pang Y. Autophagy inhibition promotes paclitaxel-induced apoptosis in cancer cells. Cancer Lett. 2011;307:141–148. doi: 10.1016/j.canlet.2011.03.026. [DOI] [PubMed] [Google Scholar]
- 203.Notte A., Ninane N., Arnould T., Michiels C. Hypoxia counteracts taxol-induced apoptosis in MDA-MB-231 breast cancer cells: role of autophagy and JNK activation. Cell Death Dis. 2013;4:e638. doi: 10.1038/cddis.2013.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Mathew R., White E. Autophagy, stress, and cancer metabolism: what doesn’t kill you makes you stronger. Cold Spring Harb. Symp. Quant. Biol. 2011;76:389–396. doi: 10.1101/sqb.2012.76.011015. [DOI] [PubMed] [Google Scholar]
- 205.Ávalos Y., Canales J., Bravo-Sagua R., Criollo A., Lavandero S., Quest A.F.G. Tumor suppression and promotion by autophagy. Biomed. Res. Int. 2014;2014:603980. doi: 10.1155/2014/603980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Zhang H., Bosch-Marce M., Shimoda L.A., Tan Y.S., Baek J.H., Wesley J.B. Mitochondrial autophagy is an HIF-1-dependent adaptive metabolic response to hypoxia. J. Biol. Chem. 2008;283:10892–10903. doi: 10.1074/jbc.M800102200. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 207.Bellot G., Garcia-Medina R., Gounon P., Chiche J., Roux D., Pouysségur J. Hypoxia-induced autophagy is mediated through hypoxia-inducible factor induction of BNIP3 and BNIP3L via their BH3 domains. Mol. Cell. Biol. 2009;29:2570–2581. doi: 10.1128/MCB.00166-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Semenza G.L. Hypoxia-inducible factor 1: regulator of mitochondrial metabolism and mediator of ischemic preconditioning. Biochim. Biophys. Acta. 2011;1813:1263–1268. doi: 10.1016/j.bbamcr.2010.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Choi J., Kim D.H., Jung W.H., Koo J.S. Metabolic interaction between cancer cells and stromal cells according to breast cancer molecular subtype. Breast Cancer Res. 2013;15:R78. doi: 10.1186/bcr3472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Zhao H., Yang M., Zhao J., Wang J., Zhang Y., Zhang Q. High expression of LC3B is associated with progression and poor outcome in triple-negative breast cancer. Med. Oncol. 2013;30:475. doi: 10.1007/s12032-013-0475-1. [DOI] [PubMed] [Google Scholar]
- 211.Singha P.K., Pandeswara S., Venkatachalam M.A., Saikumar P. Manumycin A inhibits triple-negative breast cancer growth through LC3-mediated cytoplasmic vacuolation death. Cell Death Dis. 2013;4:e457. doi: 10.1038/cddis.2012.192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Thomas S., Sharma N., Golden E.B., Cho H., Agarwal P., Gaffney K.J. Preferential killing of triple-negative breast cancer cells in vitro and in vivo when pharmacological aggravators of endoplasmic reticulum stress are combined with autophagy inhibitors. Cancer Lett. 2012;325:63–71. doi: 10.1016/j.canlet.2012.05.030. [DOI] [PubMed] [Google Scholar]
- 213.Pike L.R.G., Singleton D.C., Buffa F., Abramczyk O., Phadwal K., Li J.-L. Transcriptional up-regulation of ULK1 by ATF4 contributes to cancer cell survival. Biochem. J. 2013;449:389–400. doi: 10.1042/BJ20120972. [DOI] [PubMed] [Google Scholar]
- 214.Salem A.F., Howell A., Sartini M., Sotgia F., Lisanti M.P. Downregulation of stromal BRCA1 drives breast cancer tumor growth via upregulation of HIF-1α, autophagy and ketone body production. Cell Cycle. 2012;11:4167–4173. doi: 10.4161/cc.22316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Sotgia F., Martinez-Outschoorn U.E., Pavlides S., Howell A., Pestell R.G., Lisanti M.P. Understanding the Warburg effect and the prognostic value of stromal caveolin-1 as a marker of a lethal tumor microenvironment. Breast Cancer Res. 2011;13:213. doi: 10.1186/bcr2892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Witkiewicz A.K., Kline J., Queenan M., Brody J.R., Tsirigos A., Bilal E. Molecular profiling of a lethal tumor microenvironment, as defined by stromal caveolin-1 status in breast cancers. Cell Cycle. 2011;10:1794–1809. doi: 10.4161/cc.10.11.15675. ( http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3142463&tool=pmcentrez&rendertype=abstract) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Azad M.B., Chen Y., Henson E.S., Cizeau J., McMillan-Ward E., Israels S.J. Hypoxia induces autophagic cell death in apoptosis-competent cells through a mechanism involving BNIP3. Autophagy. 2008;4:195–204. doi: 10.4161/auto.5278. ( http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3164855&tool=pmcentrez&rendertype=abstract) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Tan E.Y., Campo L., Han C., Turley H., Pezzella F., Gatter K.C. BNIP3 as a progression marker in primary human breast cancer; opposing functions in in situ versus invasive cancer. Clin. Cancer Res. 2007;13:467–474. doi: 10.1158/1078-0432.CCR-06-1466. [DOI] [PubMed] [Google Scholar]
- 219.Dong X.L., Xu P.F., Miao C., Fu Z.Y., Li Q.P., Tang P.Y. Hypoxia decreased chemosensitivity of breast cancer cell line MCF-7 to paclitaxel through cyclin B1. Biomed. Pharmacother. 2012;66:70–75. doi: 10.1016/j.biopha.2011.11.016. [DOI] [PubMed] [Google Scholar]
- 220.Selvendiran K., Bratasz A., Kuppusamy M.L., Tazi M.F., Rivera B.K., Kuppusamy P. Hypoxia induces chemoresistance in ovarian cancer cells by activation of signal transducer and activator of transcription 3. Int. J. Cancer. 2009;125:2198–2204. doi: 10.1002/ijc.24601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Sullivan R., Graham C.H. Hypoxia prevents etoposide-induced DNA damage in cancer cells through a mechanism involving hypoxia-inducible factor 1. Mol. Cancer Ther. 2009;8:1702–1713. doi: 10.1158/1535-7163.MCT-08-1090. [DOI] [PubMed] [Google Scholar]
- 222.Su W., Huang L., Ao Q., Zhang Q., Tian X., Fang Y. Noscapine sensitizes chemoresistant ovarian cancer cells to cisplatin through inhibition of HIF-1α. Cancer Lett. 2011;305:94–99. doi: 10.1016/j.canlet.2011.02.031. [DOI] [PubMed] [Google Scholar]
- 223.Flamant L., Notte A., Ninane N., Raes M., Michiels C. Anti-apoptotic role of HIF-1 and AP-1 in paclitaxel exposed breast cancer cells under hypoxia. Mol. Cancer. 2010;9:191. doi: 10.1186/1476-4598-9-191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Fitzgerald L.D., Bailey C.K., Brandt S.J., Thompson M.E. BRCA1 accumulates in the nucleus in response to hypoxia and TRAIL and enhances TRAIL-induced apoptosis in breast cancer cells. FEBS J. 2007;274:5137–5146. doi: 10.1111/j.1742-4658.2007.06033.x. [DOI] [PubMed] [Google Scholar]
- 225.Plathow C., Weber W.A. Tumor cell metabolism imaging. J. Nucl. Med. 2008;49(Suppl. 2):43S–63S. doi: 10.2967/jnumed.107.045930. [DOI] [PubMed] [Google Scholar]
- 226.Hirschhaeuser F., Sattler U.G.A., Mueller-Klieser W. Lactate: a metabolic key player in cancer. Cancer Res. 2011;71:6921–6925. doi: 10.1158/0008-5472.CAN-11-1457. [DOI] [PubMed] [Google Scholar]
- 227.Woo O.F., Mueller P.D., Olson K.R., Anderson I.B., Kim S.Y. Shorter duration of oral N-acetylcysteine therapy for acute acetaminophen overdose. Ann. Emerg. Med. 2000;35:363–368. ( http://www.ncbi.nlm.nih.gov/pubmed/10736123) [PubMed] [Google Scholar]
- 228.Bird S., Mazzola J., Brush D., Boyer E., Aaron C. A prospective evaluation of abbreviated oral N-acetylcysteine (NAC) therapy for acetaminophen poisoning. Acad. Emerg. Med. 2003;10:521. [Google Scholar]
- 229.De Flora S., Izzotti A., D’Agostini F., Balansky R.M. Mechanisms of N-acetylcysteine in the prevention of DNA damage and cancer, with special reference to smoking-related end-points. Carcinogenesis. 2001;22:999–1013. doi: 10.1093/carcin/22.7.999. ( http://www.ncbi.nlm.nih.gov/pubmed/11408342) [DOI] [PubMed] [Google Scholar]
- 230.Moradi M., Mojtahedzadeh M., Mandegari A., Soltan-Sharifi M.S., Najafi A., Khajavi M.R. The role of glutathione-S-transferase polymorphisms on clinical outcome of ALI/ARDS patient treated with N-acetylcysteine. Respir. Med. 2009;103:434–441. doi: 10.1016/j.rmed.2008.09.013. [DOI] [PubMed] [Google Scholar]
- 231.D’Agostini F., Bagnasco M., Giunciuglio D., Albini A., De Flora S. Inhibition by oral N-acetylcysteine of doxorubicin-induced clastogenicity and alopecia, and prevention of primary tumors and lung micrometastases in mice. Int. J. Oncol. 1998;13:217–224. doi: 10.3892/ijo.13.2.217. ( http://www.ncbi.nlm.nih.gov/pubmed/9664114) [DOI] [PubMed] [Google Scholar]
- 232.Havre P.A., O’Reilly S., McCormick J.J., Brash D.E. Transformed and tumor-derived human cells exhibit preferential sensitivity to the thiol antioxidants, N-acetyl cysteine and penicillamine. Cancer Res. 2002;62:1443–1449. ( http://www.ncbi.nlm.nih.gov/pubmed/11888918) [PubMed] [Google Scholar]
- 233.Gao P., Zhang H., Dinavahi R., Li F., Xiang Y., Raman V. HIF-dependent antitumorigenic effect of antioxidants in vivo. Cancer Cell. 2007;12:230–238. doi: 10.1016/j.ccr.2007.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Albini A., Morini M., D’Agostini F., Ferrari N., Campelli F., Arena G. Inhibition of angiogenesis-driven Kaposi’s sarcoma tumor growth in nude mice by oral N-acetylcysteine. Cancer Res. 2001;61:8171–8178. ( http://www.ncbi.nlm.nih.gov/pubmed/11719447) [PubMed] [Google Scholar]
- 235.Menon S.G., Coleman M.C., Walsh S.A., Spitz D.R., Goswami P.C. Differential susceptibility of nonmalignant human breast epithelial cells and breast cancer cells to thiol antioxidant-induced G(1)-delay. Antioxid. Redox Signal. 2005;7:711–718. doi: 10.1089/ars.2005.7.711. [DOI] [PubMed] [Google Scholar]
- 236.Agarwal A., Muñoz-Nájar U., Klueh U., Shih S.-C., Claffey K.P. N-acetyl-cysteine promotes angiostatin production and vascular collapse in an orthotopic model of breast cancer. Am. J. Pathol. 2004;164:1683–1696. doi: 10.1016/S0002-9440(10)63727-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Whitaker-Menezes D., Martinez-Outschoorn U.E., Lin Z., Ertel A., Flomenberg N., Witkiewicz A.K. Evidence for a stromal-epithelial “lactate shuttle” in human tumors: MCT4 is a marker of oxidative stress in cancer-associated fibroblasts. Cell Cycle. 2011;10:1772–1783. doi: 10.4161/cc.10.11.15659. ( http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3142461&tool=pmcentrez&rendertype=abstract) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Martinez-Outschoorn U.E., Balliet R., Lin Z., Whitaker-Menezes D., Birbe R.C., Bombonati A. BRCA1 mutations drive oxidative stress and glycolysis in the tumor microenvironment: implications for breast cancer prevention with antioxidant therapies. Cell Cycle. 2012;11:4402–4413. doi: 10.4161/cc.22776. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Transparency document.