Abstract
Common Variable Immune Deficiency (CVID) and Chronic Granulomatous Disease (CGD) are two of the well-characterized primary immune defects with distinct pathologic defects. While CVID is predominantly a disorder of the adaptive immune system, in CGD, innate immunity is impaired. In both syndromes, the clinical manifestations include an increased susceptibility to infections and a number of non-infectious, inflammatory conditions including systemic autoimmunity, as well as organ-specific pathology. Among the organ-associated disorders, gastrointestinal (GI) manifestations are one of the most intractable. As such, non-infectious inflammatory disorders of the GI tract are clinically challenging as they have protean manifestations, often resembling inflammatory bowel disease (IBD) or celiac disease, are notoriously difficult to treat, and hence are associated with significant morbidity and mortality. Therefore, assessing the pathogenesis, and defining appropriate therapeutic approaches for GI disease in patients with CVID and CGD is imperative.
Keywords: common variable immune deficiency (CVID), chronic granulomatous disease (CGD), primary immune deficiency, inflammatory bowel diseases, villous atrophy, chronic enteropathy, gastrointestinal tract
INTRODUCTION
CVID is a heterogeneous primary immune deficiency disorder, characterized by a loss of B cell function [1,2]. The prevalence of CVID is estimated at 1/125,000 to 1/50,000 in Caucasians [1,2]. Most patients are diagnosed between the ages of 20 and 40, but about 20% are diagnosed under the age of 21. CVID should be considered in the differential diagnosis of patients with recurrent bacterial sino-pulmonary infections. In 20–50% of cases, interstitial lung disease, lymphoproliferative syndromes and auto-immunity are the presenting medical conditions [3]. The diagnosis of CVID is confirmed by laboratory investigations showing a significantly low serum IgG associated with low IgM and/or IgA, and poor or absent serological response to vaccines. Characteristic B cell defects include a decrease in isotype switched memory B cells (CD27+IgD−IgM−), increased numbers of “anergic” CD21lowB cells, and a loss of plasma cells in the bone marrow and peripheral tissues In making the diagnosis of CVID, other known primary or secondary causes of hypogammaglobulinemia must be excluded [1]. Additionally, a number of T cell defects, including an excess of memory T cells at the expense of naive T cells, loss of T cell proliferation, and numerous T cell-associated cytokine defects have been described in CVID and appear likely to contribute to the overall features. As a genetic defect, CVID appears mostly sporadic in inheritance, but in perhaps 10% of patients, other family members are affected. In these cases, mutations in genes that impair B cell activation (CD19, CD20, CD81 etc.), survival and function (BAFF and TACI), co-stimulation (ICOS) or other controls (LRBA, NFKB1 etc.) may be identified [4].
INFECTIOUS COMPLICATIONS OF CVID
Acute or chronic infectious diarrhea remains the most common GI symptom in CVID [5]. Patients with selective IgA deficiency [5] may have similar infections including Giardia lamblia (in up to half of cases), followed by Campylobacter jejuni and Salmonella species [5–7]. G. lamblia can be diagnosed from stool samples (PCR, ELISA, microscopy) or from duodenal biopsies (Figure 1A). In the latter, the trophozoite is generally associated with intraepithelial lymphocytosis. Other infections have been reported such as cytomegalovirus and Clostridium difficile [5,7,8]. Interestingly, despite their greater exposure to antibiotics, CVID patients do not appear to have a higher incidence of C. difficile infections, which perhaps may be due to the presence of anti-C. difficile antibodies in replacement immunoglobulin products [9]. Recently, Norovirus has been noted as an increasingly important enteric infection in patients with CVID, leading in some cases to severe chronic enteropathy. Notably, Noroviral clearance may be associated with improved histopathological features and resolution of villous atrophy [10] [11].
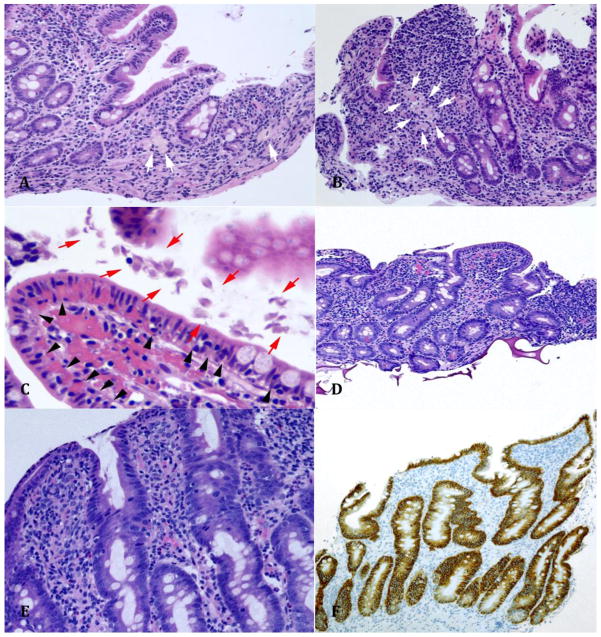
Figure 1. Pathological cases of CGD and CVID duodenal biopsies.
This duodenal mucosa exhibits small aggregates of pale, lightly pigmented macrophages (white arrows) present in the lamina propria, consistent with GI lesions of CGD (x200 magnification, hematoxylin and eosin stain) (A and B). Giardia lamblia organisms are seen between red arrows on this duodenal biopsy of a CVID patient (x600, hematoxylin and eosin stain). Intraepithelial lymphocytosis of the duodenal epithelium is also present (black arrows) (C). This biopsy shows duodenal mucosa with active inflammation characterized by a prominent neutrophil infiltrate in the lamina propria. There is blunting of the villi with mild intraepithelial lymphocytosis (D, x100 and E, x200; hematoxylin and eosin stains). Immunohistochemical stain for CD138 (plasma cell marker) shows that the lamina propria is entirely devoid of plasma cells (duodenal epithelium is a positive control) (F, x100). D, E and F biopsies are from patient 2.
The finding of other pathogens, such as Microsporidia, Cryptosporidia or unusual fungi or parasites, may raise the suspicion of a more complex immunodeficiency state including greater degrees of T cell dysfunction.
Because of increased susceptibility to bacterial infection, one might assume that patients with CVID would have a higher prevalence of Helicobacter pylori (Hp) infection. However, perhaps due to frequent exposure to antibiotics, Hp infection rates in CVID patients are equivalent to those seen in the general population. Combining data from two studies, a total of 11 patients out of 60 patients with CVID (18.3%) who had gastric biopsies were found to be Hp+ [6,12]. In contrast, in the general population, the prevalence of Hp infection ranges from 17 to 79% in Europe and around 30% in the USA, with variations according to age or ethnicity [13,14]. However Hp infection in patient with CVID may promote gastritis, gastric dysplasia or gastric cancer [15,16]. Furthermore, in the setting of CVID, gastric pathology may not resolve with Hp eradication as shown by Malamut et al. [6].
NON-INFECTIOUS COMPLICATIONS OF CVID (Table 1)
Table 1.
Non-infectious and non-malignant GI disorders associated with CVID
| Organ | Manifestation | Clinical manifestations | Potential histopathological features | Differential diagnosis |
|---|---|---|---|---|
| Stomach | Chronic gastritis | Abdominal pain Vomiting Nausea |
Lymphocytic infiltration Antral and corpus atrophy Antral and corpus dysplasia Antral and corpus metaplasia Absence of plasma cells |
Pernicious anemia (Autoimmune gastritis) Hp-associated gastritis |
| Small intestine | Villous atrophy | Malnutrition Chronic diarrhea |
Villous aplasia Intraepithelial lymphocytosis Follicular lymphoid hyperplasia Lymphocytic duodenitis Neutrophilic infiltrate GVH-like lesions Absence of plasma cells |
Celiac disease Refractory sprue |
| Chronic Bacterial Overgrowth | Chronic diarrhea Abdominal discomfort Malnutrition |
Non-specific | Mechanical obstruction History of abdominal surgery |
|
| Chronic exudative enteropathy | Chronic diarrhea Malnutrition Anasarca |
Lymhangiectasia Enteritis |
Whipple’s disease Intestinal lymphoma Porto-mesenteric venous thrombosis |
|
| Chronic enteritis | Abdominal pain Diarrhea |
Ulcers Neutrophilic infiltrate GVH-like lesions Lymphocytic infiltrate Follicular lymphoid hyperplasia Granuloma Absence of plasma cells |
Celiac disease Crohn’s disease |
|
| Colon | Chronic colitis | Diarrhea Bloody stools Abdominal pain |
Ulcers Crypt abcesses Cryptitis Neutrophilic infiltrate GVH-like lesions Lymphocytic infiltrate Follicular lymphoid hyperplasia Granuloma Absence of plasma cells |
Crohn’s disease Ulcerative colitis Microscopic colitis |
Among the inflammatory complications, the most common are autoimmune cytopenias (immune thrombocytopenia and hemolytic anemia), interstitial and/or granulomatous lung disease and gastrointestinal complications including chronic enteropathy [3,7,17–19]. GI involvement is frequent in patients with CVID, reported in 9 to 20% of patients [3,20]. Up to 50% of patients have intermittent or chronic diarrhea, leading to malabsorption [5,21]. Here we discuss the non-infections GI complications in CVID in greater detail.
Non-Infectious Gastrointestinal Disease
Non-infectious enteropathy occurs in 10 to 12% of CVID patients and may resemble other GI conditions such as Crohn’s disease, ulcerative colitis or celiac disease. In a large case series, Malamut et al. examined 50 patients with CVID 40% of whom had chronic GI symptoms [6]. In this cohort, the mean age at CVID diagnosis was 36.8 years and mean age at the onset of GI symptoms was 34.5 years. Diarrhea was present in two thirds of patients and abdominal pain in more than half of cases. The mean body mass index of patients was below 20 kg.m−2, and half of patients had evidence of malnutrition [6]. Abnormalities were seen in 34 of 41 duodenal biopsies (83%) and 28 of 35 colonic biopsies (80%). Additionally, macroscopic inflammation was seen during colonoscopy in 26% of cases [6].
Similarly, in a study conducted by Maarschalk-Ellerbroek et al., 30 patients with CVID underwent upper and lower GI endoscopy. Abnormalities were observed in 25 of the 30 patients (83%), although only 18 (64%) patients had experienced GI related symptoms. Colitis and gastritis (in 20%) were the most GI common manifestations [12]. In half of these cases, gastritis was Hp (−). Five of these patients were found to have both severe gastric corpus atrophy and vitamin B12 deficiency and 3 had detectable serum auto-antibodies against gastric parietal cells. Notably, Zullo et al. previously reported that about 26% of patients with CVID have atrophic gastritis [15].
Tissue histology in CVID is characteristic for the paucity or absence of plasma cells and a high frequency of CD8+ T cell infiltrates in the intestinal lamina propria [6,7,18]. Additionally, lamina propria mononuclear cells (LPMC) of CVID patients may produce significantly more IL-12 and IFNγ (but not IL-23 and IL-17) compared to controls [22]. In Crohn’s disease, the role of IL-12 and IFNγ to promote gut inflammation is well documented [23] and therapeutic strategies targeting IL12 with briakinumab or ustekinumab have shown promising results [24,25]. Similarly, Innate lymphoid cells (ILC) have also been identified to play a crucial role in IBD pathogenesis [26]. The number of inflammatory ILC have remarkably been found increased in blood, respiratory and gastrointestinal mucosa of CVID patients with inflammatory conditions compared to patients without CVID inflammatory disorders [27] although, to date ILC have not been studied in detail in the GI mucosa of CVID patients with intestinal manifestations.
Chronic enteropathy in CVID
CVID associated CE is a complex disorder that may include features similar to ulcerative colitis, proctitis, or severe small bowel disease that leads to weight loss and malnutrition which may require long-term parenteral feeding [3]. Small bowel disease in patients with CVID is associated with exudative enteropathy, chronic bacterial overgrowth, villous atrophy and inflammatory lymphocytic infiltrates.
Exudative enteropathy is associated with a loss of serum IgG, and can be confused with CVID; however, in these cases, serum albumin may also be reduced. More importantly, IgA and IgM levels are likely to be normal in pure exudative enteropathy, and antibody function is generally preserved. Patients with CVID may develop a form of exudative enteropathy, further depleting immunoglobulins and leading to increased requirements for parenteral Ig supplementation. In such patients, protein loss is caused by epithelial exudation in the setting of mucosal inflammation and damage, due to a loss of intestinal barrier function. In certain cases this is associated with hepatopathy with or without increased portal pressures. In these cases, subcutaneous administration of Ig may be required to maintain therapeutic serum IgG levels [28].
Small intestine bacterial overgrowth (SIBO) is common in patients with CVID due to the lack of IgA and possibly IgG and IgM within the intestinal lumen [29]. In clinical practice, the diagnosis of SIBO may be difficult to establish as patients may be exposed to antibiotics on an intermittent or chronic basis. To confirm the diagnosis of SIBO, hydrogen breath test is required. Treatment with a course of non-absorbed broad-spectrum antibiotics may also be used as a diagnostic and therapeutic tool.
Small bowel villous atrophy is a frequent and challenging finding in patients with CVID who present with malabsorption. After excluding infectious causes, the first proposed diagnosis, is generally celiac disease and on this basis, gluten free diet is often initiated. As serologic studies are not helpful, genetic studies may be needed to determine the likelihood of this diagnosis. Interestingly, gluten-free diet improves approximately 20% of CVID patients with villous atrophy and the efficacy of gluten avoidance seem slightly higher in subjects with the celiac-associated genotype HLA DQ2 and DQ8 [6,30]. In these cases, a gluten free diet could be tried for 6 to 12 months, and then the histological response ascertained. However, villous atrophy is common in most or all CVID subjects with significant chronic enteropathy and wheat avoidance is more likely to lead to further loss of weight. In these cases, some histological features, such as GVH-like lesions, polymorphonuclear infiltrates and an absence of intestinal plasma cells are more suggestive of CVID-associated enteropathy than celiac disease [6,30].
In case of severe malabsorption caused by CVID associated CE, parenteral nutrition is required which leads to risks of central venous catheter-related infections.
Gastrointestinal Pathology in CVID associated chronic enteropathy (Figure 1B–D)
Increased intraepithelial lymphocytes (IEL) are a common histological finding in gastrointestinal biopsies of subjects with CVID, especially those with chronic malabsorption [6,7,30]. In the study by Malamut et al., it was the most commonly found histological feature within the entire cohort (75.6% of cases, > 30 IELs/100 epithelial cells (ECs)). Such cases are distinguished from Celiac Disease even when associated with villous atrophy by the following criteria: In CVID, IEL count is usually lower than in Celiac Disease and is often associated with a strong - neutrophilic infiltrate, GVH-like lesions, follicular lymphoid hyperplasia and an absence of plasma cells [6]. Immunohistochemistry and flow cytometry usually reveal the IEL infiltrate to be CD8+ T cell enriched. In the study by Malamut et al. only one patient with concomitant G. lamblia infection had IELs enriched for CD4+ T cells. Additionally, and in contrast to the findings in celiac disease, only one patient with CVID had increased numbers of TCRγδ T cells, and none exhibited a clonal TCRγ rearrangement [6]. CVID patients with increased IELs are also more likely to have increased CD8+ T cells in peripheral blood (52%) and some patients may exhibit CD8+ T cell infiltrates within the hepatic sinusoids [6]. Follicular hyperplasia is yet another pathological finding in the GI tract of CVID patients [6]. The hyperplastic follicles contain B cells and sometimes CD8+ T cells but again lack plasma cells.
Between 8 and 22% of subjects with CVID develop granulomatous disease. In most cases, the non-caseating granulomas are found in lung, lymph node or liver. But granulomas may also be found in skin, brain, bone marrow, or the GI tract [31,32]. In the French national cohort of CVID patients, among 436 subjects with CVID, 59 patients (13.5 %) were diagnosed with granulomatous disease in one or more organs. Among them, 9 patients had GI involvement, associated with clinical symptoms such as diarrhea and weight loss [31]. Granulomatous disease in CVID is notoriously difficult to treat [31] (Boursiquot, Gerard et al. 2013)(2); infliximab has been used in a few cases. More recently, rituximab and azathioprine combo-therapy has shown some benefit for those with pulmonary involvement [33].
For colonic lesions in CVID, the severity ranges from microscopic colitis (mostly lymphocytic but collagenous colitis is also found) to severe colitis with ulcerations [6]. The histopathological pattern may mimic ulcerative colitis or Crohn’s disease including the presence of ulcers, crypt abscesses and crypt destruction. Alternatively, colonic lesions may show a mild inflammatory infiltrate. Follicular hyperplasia and GVH-like disease may be associated with acute or chronic colitis. Similarly, inflammation and ulcers may be seen, albeit at a lower frequency than in the small intestine [6].
GI tract cancers and CVID
Two to 10% of CVID patients may develop lymphoid malignancies [3,20,34]. These are generally non-Hodgkin’s B cell lymphomas and often involve extra nodal sites [34] which in some cases include the GI tract. In the great majority of cases, these are EBV negative. As subjects with CVID are likely to have thoracic or abdominal lymphoid hyperplasia and/or splenomegaly, the diagnosis of lymphoma can be challenging. Subjects with CVID may also develop gastrointestinal nodular lymphoid hyperplasia, leading to erroneous diagnoses of indolent GI lymphoid malignancies (such as mucosa-associated lymphoid tissue (MALT) lymphoma) [35]. Immunohistochemical and gene rearrangement studies are often helpful in the assessment of cases of atypical lymphoid hyperplasia but identifying a clonal expansion of B cells may not be pathognomonic [35]. Treatment of CVID associated lymphoma is usually similar to the treatment of lymphomas in other settings and usually incorporates rituximab as a therapeutic agent.
CVID patients have been said to have a 10-fold increased risk of gastric cancer compared to the general population [36] but recent data suggests that the risk has declined since the original reports [3,20], potentially due to the elimination of Hp by antibiotics. Chronic atrophic gastritis and extensive intestinal metaplasia within the gastric corpus are the strongest risk factors for the development of CVID-associated gastric cancer [37]. De Petris et al. focused on the characteristics of 6 CVID-associated gastric cancers and reported that these adenocarcinomas were diagnosed at a younger age, were of intestinal type and were associated with increased numbers of intra-tumoral lymphocytes. Severe atrophic metaplastic pangastritis with extensive dysplasia was present in the background in 4 on 6 cases, with features of lymphocytic gastritis in 2 cases. Histopathological features of CVID (paucity of plasma cells and nodular lymphoid hyperplasia) were also detected in these patients [38]. Notably, the risk of gastric cancer persists in CVID patients even after Hp eradication. Patients with CVID do not appear to be at a higher risk for colon and anal cancers than the general population.
Treatment of GI disease in CVID
Immunoglobulin supplementation generally has little effect on the course of non-infectious GI disease in CVID [6]. After infections have been excluded and the outside possibility that the enteropathy may be related to gluten intolerance, the first line treatment is oral steroids (budesonide or temporarily, systemic steroids). Elimination of bacterial overgrowth with oral antibiotics may be useful, and needed repeatedly. Other immunosuppressive therapies such as thiopurines have been used with variable and inconsistent results. There is anecdotal evidence regarding the efficacy of TNF-antagonists, infliximab as well as adalimumab, although a more wide spread adoption of such therapies has not occurred [39,40]. The overall efficacy of anti-IL12/IL23 therapy (ustekinumab) and anti α4β7 integrin (vedolizumab) has not yet been reported. Especially with the use of Vedolizumab, there is the potential for blocking the ingress of regulatory T cells into the mucosa, potentially worsening the clinical condition. Oral immunoglobulin therapy has been considered, as well as human breast milk. Studies are actively focused on the role of the microbiota in patients with CVID. However, to date, no data are available on fecal transplantation, which also raise the concern of the risk of introducing infections in such patients.
GASTROINTESTINAL MANIFESTATIONS OF CGD
In contrast to CVID (overall differences between CGD and CVID are exhibited in Table 2), which is a defect of adaptive immunity, CGD is an inherited immunodeficiency disorder involving the innate immune system that impairs the neutrophil oxidative burst. The basis of this defect is a loss of nicotinamide adenine dinucleotide phosphate-oxidase (NADPH Oxidase or NOX) function in phagocytic cells, which diminishes their ability to produce reactive oxygen species (ROS) [41]. This defect impairs anti-microbial killing. The CGD syndrome is a rare condition occurring in approximately 1 in 200,000 live births [42,43]. It is generally diagnosed in childhood, even though milder forms may lead to the diagnosis being made in adulthood. However, median age at diagnosis is around 3 to 5 years with earlier age at diagnosis for X-linked CGD [44,45]. Mutant genes that lead to CGD together encode the subunits of NOX. Two third of patients have an X-linked form with various mutations of CYBB, the gene encoding gp91phox. The other one third of patients have autosomal recessive mutations of CYBA, NCF1, NCF2, NCF4 which code for p22phox, p47phox, p67phox and p40phox proteins, respectively [46]. Phenotypes differ according to the genetic status. Patients with X-linked CGD generally have a more severe course and appear to be prone to developing inflammatory disorders [47].
Table 2.
Comparison of clinico-pathological characteristics between CVID and CGD
| CVID | CGD | |
|---|---|---|
| Dysfunctional immune cell subset | B cells | Phagocytic cells (neutrophils and macrophages) |
| Dysfunctional immune function | Adaptive immunity Immunoglobulin production |
Innate immunity Neutrophil oxidative burst |
| Major mutant genes and functions | - B cell activation: CD19, CD20, CD81 - B cell survival and function: TACI, BAFF - Others: LRBA, NFKB1 |
- NOX GP91phox subunit X-linked encoding: CYBB - Others NOX subunits autosomal encoding: CYBA, NCF1, NCF2, NCF4 |
| Median age of diagnosis | 20 to 40 year-old 20% of diagnosis made under 21 year-old |
X-linked form = 3 year-old Autosomal form = 11 year-old |
| Most frequent infections | - Bronchitis - Sinusitis - Pneumonia - GI infections - Meningitis |
- Pneumonia - Suppurative adenitis - Skin infections - Liver and GI tract abscesses |
| Usual involved microorganisms | - Respiratory tract: Streptococcus pneumoniae, Haemophilus influenzae - GI tract: Giardia intestinalis, Salmonella species |
- Catalase-positive microorganisms: Aspergillus sp., Staphylococcus aureus, Serratia sp., Salmonella sp., Burkholderia sp. |
| Non infectious/non GI manifestations | - Autoimmunity: autoimmune cytopenias (Immunologic thrombocytopenic purpura, Autoimmune hemolytic anemia), vitiligo, etc - Inflammatory lung disorders: bronchiectasis, interstitial disease - Lymphoproliferative features: systemic granulomatous, lymphoproliferation, B cell non Hodgkin lymphoma |
- Systemic granulomatous: brain, lungs, liver, spleen involvement may be seen - Nodular regenerative hyperplasia |
Even though the prognosis of CGD patients has improved during the past few years due to antibiotic prophylaxis, CGD is generally a severe condition that requires intermittent and life long medical care. The main clinical features include bacterial and fungal infections and inflammatory manifestations. Most patients have the early appearance of severe and recurrent bacterial and/or fungal infections. The diagnosis is established following a functional evaluation of phagocytes (neutrophil oxidative burst) which demonstrate impaired production of reactive oxygen species (ROS). Genetic assessments confirm the diagnosis and the inheritance pattern. With appropriate antibiotic prophylaxis, infectious conditions are less frequent after the age of 10, when inflammatory disorders become the major clinical phenotype. GI tract is the most frequently affected organ as more than 80% of patients have inflammatory GI disorders [47]. The most common inflammatory clinical manifestations are non-infectious diarrhea, followed by oral aphthae, anal fistulae, vomiting, anorexia, and abdominal pain [47]. Aside from infections, intestinal dysmotility, obstruction and ulceration may occur along the entire length of the GI tract even though the colon is the most frequently involved site [48]. CGD patients are highly predisposed to perianal disease and rectal abscesses in infancy are often the first clue to this disease. In other cases, the GI involvement can mimic Crohn’s disease and lead to misdiagnosis, especially in the absence of a suggestive history of infections [48,49]. A dysregulated inflammatory response to chronic exposure of unresolved microbial infections is thought to drive the disorder. Indeed, high rates of CD-associated anti-microbial antibodies are present in patients with CGD-associated colitis [50]. Moreover, Acetobacteracae have been sequenced from patient granulomas [51].
EVALUATION OF PATIENTS WITH CGD
Imaging techniques such as CT-scans or magnetic resonance imaging (MRI) may be useful to show GI ulceration, wall thickening, strictures or fistulae [48]. Endoscopic features are not specific and are generally indistinguishable from Crohn’s disease, ranging from mild erythema to deep ulcers. Inflammation is almost always discontinuous [52]. Histopathologic findings are also very similar to Crohn’s disease. Both acute and chronic inflammatory infiltrates are seen and are generally transmural [52,53]. Granulomas are found in approximately half of patients [47,52,53]. Even though the constellation of pathological findings resemble Crohn’s disease, CGD-specific features include a lack of neutrophils, increased eosinophils, eosinophilic cryptitis, and the presence of pigment-laden macrophages in the lamina propria (Figure 1E and F) [52,53].
Marks et al. have reported a case series of 25 patients with CGD. Retrospective analysis of the cohort revealed that more than half of them experienced GI symptoms in the previous 3 years. Intestinal inflammation was documented in 79% of them with a more frequent involvement of the colon and rectum (73%). Furthermore, discontinuous segments of the colon were affected and the perianal area was always involved. Additionally, granulomas were found in 55% of patients with documented intestinal inflammation [52]. Khangura et al. have reported the results of 211 endoscopies in 78 patients with CGD seen at the National Institute of Health (NIH). 74% of patients had gastric and colonic inflammation. Esophageal inflammation was less frequent (21%) but esophageal dysmotility and structural abnormalities were observed in 26% of cases. Similar to the data reported by Marks et al., there was a high prevalence of perianal disease (93%), including fistulae, in patients with colonic CGD. Finally, colonic strictures were common (24%) and mainly involved ano-rectal area (80%) [54].
TREATMENT OF INFLAMMATORY GI DISEASE ASSOCIATED WITH CGD
In terms of treatment, infectious etiologies should first be excluded, even though the presence of pathogens is not necessarily the cause of symptoms. The treatment of inflammatory GI disorders associated with CGD is empirical and not well established as it is based on scant data. Anti-infectious agents are often used empirically and should broadly target bacterial and fungal pathogens. Short courses of systemic or ileal release steroid therapy (budesonide) have been administered, but with equivocal results [52]. In case of steroid non-responsiveness or as steroid sparing agents, immunomodulatory therapy, such as 5-aminosalicylates, thiopurines, methotrexate, cyclosporine and thalidomide have been prescribed [52,55,56]. However, the scientific evidence of treatment efficacy is weak and anecdotal. Anti-TNFα therapy should be avoided as it critically increases the risk of serious bacterial and fungal infections [57]. G-CSF and GM-CSF have been used with variable success [58]. Additionally, surgical options may be considered in patients with refractory colitis, perianal fistulae and complicated GI lesions. Finally, in CGD hematopoetic bone marrow transplantation has been offered to patients with a suitable stem cell match and severe disease [59].
CASE RECORDS OF REPRESENTATIVE PATIENTS FROM THE MOUNT SINAI COHORT
The following cases are illustrative of the complexity and severity of GI manifestations in patients with CVID-associated CE (Table 3) and IBD-like CGD. As described, the management of GI disease in such cases is often symptom directed and empiric.
Table 3.
Clinical features of representative CVID-associated CE cases from the Mount Sinai cohort
| Age at CVID diagnosis | Infections history (except GI) | IgG | IgA | IgM | Age at GI first symptoms | GI infections history | GI clinical features | GI endoscopic and pathologic features | Treatments received for GI purpose | |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 18y | - Recurrent childhood ear infections |

|

|

|
18 y | - Giardia intestinalis - Acute Norovirus |
- Chronic intestinal failure/malnutrition - Chronic diarrhea - Vomiting/nausea - Exudative enteropathy |
- Normal macroscopic assessment - Chronic Hp(−) gastritis - Chronic duodenitis with increased intraepithelial lymphocytes (IEL) - Microscopic lymphocytic colitis - Absence of plasma cells |
- TPN/multivitamin supplementation - Budesonide - Ribavirin |
| 2 | 11y | - Recurrent bronchiolitis and pneumoniae since 6 months-old - Bronchectiasis with multiple surinfections |

|

|

|
8y | - Giardia intestinalis - C. difficile |
- Chronic diarrhea - Vomiting/nausea - Chronic intestinal failure/malnutrition |
- Normal macroscopic assessment - Chronic Hp(−) gastritis - Chronic duodenitis with increased intraepithelial lymphocytes (IEL) - Chronic ileitis with increased IELs - Microscopic lymphocytic colitis - Absence of plasma cells |
- Enteral feeding - Budesonide - 6-MP - Azathioprine |
| 3 | 40y | - Sino-pulmonary |

|

|

|
44y | - Non remarkable | - Chronic diarrhea - Chronic intestinal failure/malnutrition - Exuudative enteropathy - Bloating - SIBO |
- Normal macroscopic assessment - Chronic Hp(−) gastritis - Chronic duodenitis with increased IELs - Villous atrophy - Normal colonic mucosa - Absence of plasma cell |
- TPN/multivitamin supplementation - Octreotide - Loperamide - Budesonide - Vedolizumab - Pancreatic enzymes - Sequential antibiotics |
IELs: intraepithelial lymphocytes; SIBO: small bowel bacterial overgrowth; TPN: total parenteral nutrition
Patient 1 is a male who was diagnosed CVID in 2010 at the age of 18 with GI symptoms. The patient developed a rapidly progressive enteropathy syndrome characterized by nausea, vomiting and diarrhea with evidence of protein loss (low plasma protein level and pedal edema) and wasting. Pertinent labs included a profound reduction in IgG, IgA and IgM levels, decreased peripheral B cells (150/mm3) and T cells (comprised of CD4 cytopenia ~300/mm3) as well as multiple micronutrient deficiencies. Additionally and characteristically, the patient had the absence of plasma cells in GI biopsies. Notably, the patient’s prior medical history was only significant for recurrent ear infections since childhood.
Due to progressive and severe malnutrition, total parenteral nutrition (TPN) with micro and macronutrient supplementation was started. Additionally, budesonide and intravenous immunoglobulin therapies were initiated. Following clinical response, the patient was subsequently weaned off of TPN and budesonide.
From 2011 the patient has exhibited chronic osteoarticular pain suggestive of osteomalacia and has presented with multiple relapses of GI disease due to recurrent Giardia infections and as well as CVID-associated non-infectious chronic enteropathy.
In summer of 2015, the patient again developed rapidly progressive GI symptoms because of an acute Norovirus infection with a relapse in October. The patient was treated with Ribavirin and TPN was reinitiated. The patient’s disease course was complicated by a PICC-line associated septic shock. At the present time, the patient is on oral budesonide and has been discharged from the hospital after successful treatment of septic shock and resolution of the Norovirus infection with oral ribavirin.
Patient 2 is a female who presented with recurrent pneumonia and bronchiolitis requiring multiple hospitalizations since the age of 6 months.
At the age of 8y, the patient developed colitis and was diagnosed with Giardia intestinalis. However, even after the treatment of Giardia her colitic symptoms persisted. She was started on enteral feeding due to failure to thrive. Additionally, immunomodulator therapy with 6-mercaptopurine was started due to ongoing GI disease. The patient continued to develop recurrent respiratory tract infections which resulted in bronchiectasis. She was finally diagnosed with CVID at the age of 11. At the time, the patient had a severe deficiency of IgG, IgA and IgM antibodies and the absence of B cells in the peripheral blood and intestinal biopsies. In contrast, her T cells were in the low-normal range. Soon after diagnosis, the patient was started on IVIG therapy.
In addition to chronic lung disease, the patient has chronic diarrhea, failure to thrive and deficiency of multiple, micro- and macronutrients. Endoscopic examination of the upper and lower GI tracts were grossly unremarkable. However, pathologic evaluation of biopsies revealed chronic gastritis (Hp−), pan-enteritis and colitis. Additionally, the plasma cells were absent and intra-epithelial lymphocytes (IEL) were increased in the small bowel and the colon. The patient showed mild improvement in intestinal symptoms with budesonide and azathioprine but continues to have recurrent hospitalizations. She was recently diagnosed with an autosomal dominant, frame shift mutation in the NFkB1 gene. Currently, infliximab is being considered for the refractory GI disease in this patient.
Patient 3 is a female born with recurrent mild sino-pulmonary infections since mild childhood, which resulted in the diagnosis of CVID at the age of 30. She had an IgG of 81, an IgA <1 and IgM of 17mg/dl and was placed on IVIG. Four years after diagnosis, the patient developed GI symptoms comprising of chronic diarrhea and wasting. Upper GI endoscopy revealed villous atrophy initially. Although the patient continued to have recurrent GI symptoms, in 2014, 7 years after the initial diagnosis, her GI symptoms worsened dramatically and were associated with marked weight loss and malnutrition. Multiple tests could not diagnose an infectious etiology. Endoscopic examination, though grossly unremarkable revealed H. pylori negative chronic gastritis and duodenitis with mild intraepithelial lymphocytosis. There was a complete absence of plasma cells in the upper and lower intestinal tracts. The patient was empirically started on TPN, Octreotide, Loperamide and pancreatic enzymes. Due to ongoing GI symptoms, she was started on vedolizumab, a novel inhibitor of integrin α4β7. Remarkably and perhaps instructively, her GI symptoms worsened with vedolizumab and this therapy was abruptly discontinued. The patient continues to be treated with sequential courses of antibiotics, budesonide and is on home TPN at the present time.
Patient 4 was diagnosed with autosomal recessive p47phox CGD at the age of 5 in the context of recurrent lymphadenitis with abscesses. His medical history was notable for consanguineous parents and G6PD deficiency. The patient has since developed chronic diarrhea, vomiting, malnutrition and failure to thrive. Endoscopic evaluation of his GI tract, grossly normal, has revealed chronic, H. pylori negative gastritis, enteritis and colitis. The patient continues to have chronic, systemic inflammation. Subsequent disease course is complicated by a rectal abscesses and an intractable rectal fistula. Repeat endoscopic evaluation has revealed erythematous and nodular lesions in esophagus and stomach, scattered aphthae in the colon as well as a left colonic stricture. Pathologic assessments do not show granulomas, typical of patients with CGD but pigmented macrophages are seen. The patient was placed on 6-mercaptopurine therapy but did not show a good clinical response, likely due to non-compliance with medications. Additional immunomodulator therapies are being considered for this patient.
CONCLUSIONS
GI involvement, common in CVID and CGD, could be the dominant disease manifestation in a subset of patients. GI symptoms, often protean can mimic other GI diseases such as celiac disease and IBD. Acute infections remain the most frequent manifestation and should always be ruled out. Current therapies are empiric and based on self-experience. To improve the quality of available therapies, well-designed prospective studies are urgently needed.
Acknowledgments
Funding Acknowledgement: This work was supported by the National Institutes of Health AI 101093, AI-086037, AI-48693, T32-GM007280, The Jeffrey Modell Foundation, and the David S Gottesman Immunology Chair (all for CCR), and 5R01DA033777 (SM).
Footnotes
Conflicts of Interest
MU, HMK, SM, and CCR declare that they have no conflicts of interest.
Compliance with Ethics Guidelines
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
- 1.Bonilla FA, Barlan I, Chapel H, Costa-Carvalho BT, Cunningham-Rundles C, de la Morena MT, et al. ICON: Common Variable Immunodeficiency Disorders. J allergy Clin Immunol Pract. 2015 doi: 10.1016/j.jaip.2015.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Picard C, Al-Herz W, Bousfiha A, Casanova J-L, Chatila T, Conley ME, et al. Primary Immunodeficiency Diseases: an Update on the Classification from the International Union of Immunological Societies Expert Committee for Primary Immunodeficiency 2015. J Clin Immunol. 2015 doi: 10.1007/s10875-015-0201-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Resnick ES, Moshier EL, Godbold JH, Cunningham-Rundles C. Morbidity and mortality in common variable immune deficiency over 4 decades. Blood. 2012;119:1650–7. doi: 10.1182/blood-2011-09-377945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Salzer U, Warnatz K, Peter HH. Common variable immunodeficiency: an update. Arthritis Res Ther. 2012;14:223. doi: 10.1186/ar4032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Oksenhendler E, Gérard L, Fieschi C, Malphettes M, Mouillot G, Jaussaud R, et al. Infections in 252 Patients with Common Variable Immunodeficiency. Clin Infect Dis. 2008;46:1547–54. doi: 10.1086/587669. [DOI] [PubMed] [Google Scholar]
- **6.Malamut G, Verkarre V, Suarez F, Viallard J-F, Lascaux A-S, Cosnes J, et al. The enteropathy associated with common variable immunodeficiency: the delineated frontiers with celiac disease. Am J Gastroenterol. 2010;105:2262–75. doi: 10.1038/ajg.2010.214. 50 patients with CVID-associated GI symptoms were assessed with chronic diarrhea as the most frequent clinical feature (92%). Pathological small bowel assessment was remarkable for the high frequency of increased IEL (75%) and villous atrophy (50%) whereas plasma cells were almost always absent. IVIg desmonstrated no effect on GI symptoms. [DOI] [PubMed] [Google Scholar]
- 7.Daniels Ja, Lederman HM, Maitra A, Montgomery Ea. Gastrointestinal tract pathology in patients with common variable immunodeficiency (CVID): a clinicopathologic study and review. Am J Surg Pathol. 2007;31:1800–12. doi: 10.1097/PAS.0b013e3180cab60c. [DOI] [PubMed] [Google Scholar]
- 8.Kralickova P, Mala E, Vokurkova D, Krcmova I, Pliskova L, Stepanova V, et al. Cytomegalovirus disease in patients with common variable immunodeficiency: three case reports. Int Arch Allergy Immunol. 2014;163:69–74. doi: 10.1159/000355957. [DOI] [PubMed] [Google Scholar]
- 9.Salcedo J, Keates S, Pothoulakis C, Warny M, Castagliuolo I, LaMont JT, et al. Intravenous immunoglobulin therapy for severe Clostridium difficile colitis. Gut. 1997;41:366–70. doi: 10.1136/gut.41.3.366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- **10.Woodward JM, Gkrania-Klotsas E, Cordero-Ng AY, Aravinthan A, Bandoh BN, Liu H, et al. The Role Of Chronic Norovirus Infection In The Enteropathy Associated With Common Variable Immunodeficiency. Am J Gastroenterol Nature Publishing Group. 2015;110:320–7. doi: 10.1038/ajg.2014.432. Stools and small bowel biopsies from 8 CVID patients with CE were analysed by PCR for the presence of Norovirus RNA and compared to 10 CVID patients without GI disorder. All 8 patients with CVID-associated CE showed evidence of chronic Norovirus carriage (none in the control group). Remarkably, 3 patients with Norovirus clearance (one spontaneously, 2 with Ribavirin therapy) exhibited complete symptomatic and histological recovery. [DOI] [PubMed] [Google Scholar]
- 11.van de Ven AAJM, Janssen WJM, Schulz LS, van Loon AM, Voorkamp K, Sanders EAM, et al. Increased prevalence of gastrointestinal viruses and diminished secretory immunoglobulin a levels in antibody deficiencies. J Clin Immunol. 2014;34:962–70. doi: 10.1007/s10875-014-0087-3. [DOI] [PubMed] [Google Scholar]
- 12.Maarschalk-Ellerbroek L, Oldenburg B, Mombers I, Hoepelman A, Brosens L, Offerhaus G, et al. Outcome of screening endoscopy in common variable immunodeficiency disorder and X-linked agammaglobulinemia. Endoscopy. 2013;45:320–3. doi: 10.1055/s-0032-1326078. [DOI] [PubMed] [Google Scholar]
- 13.Roberts SE, Morrison-Rees S, Samuel DG, Thorne K, Akbari A, Williams JG. Review article: the prevalence of Helicobacter pylori and the incidence of gastric cancer across Europe. Aliment Pharmacol Ther. 2015 doi: 10.1111/apt.13474. [DOI] [PubMed] [Google Scholar]
- 14.Everhart JE, Kruszon-Moran D, Perez-Perez GI, Tralka TS, McQuillan G. Seroprevalence and ethnic differences in Helicobacter pylori infection among adults in the United States. J Infect Dis. 2000;181:1359–63. doi: 10.1086/315384. [DOI] [PubMed] [Google Scholar]
- 15.Zullo A, Romiti A, Rinaldi V, Vecchione A, Tomao S, Aiuti F, et al. Gastric pathology in patients with common variable immunodeficiency. Gut. 1999;45:77–81. doi: 10.1136/gut.45.1.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Desar IME, van Deuren M, Sprong T, Jansen JBMJ, Namavar F, Vandenbroucke-Grauls CM, et al. Serum bactericidal activity against Helicobacter pylori in patients with hypogammaglobulinaemia. Clin Exp Immunol. 2009;156:434–9. doi: 10.1111/j.1365-2249.2009.03909.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Washington K, Stenzel TT, Buckley RH, Gottfried MR. Gastrointestinal pathology in patients with common variable immunodeficiency and X-linked agammaglobulinemia. Am J Surg Pathol. 1996;20:1240–52. doi: 10.1097/00000478-199610000-00010. [DOI] [PubMed] [Google Scholar]
- 18.Agarwal S, Smereka P, Harpaz N, Cunningham-Rundles C, Mayer L. Characterization of immunologic defects in patients with common variable immunodeficiency (CVID) with intestinal disease. Inflamm Bowel Dis. 2011;17:251–9. doi: 10.1002/ibd.21376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chapel H, Lucas M, Lee M, Bjorkander J, Webster D, Grimbacher B, et al. Common variable immunodeficiency disorders: division into distinct clinical phenotypes. Blood. 2008;112:277–86. doi: 10.1182/blood-2007-11-124545. [DOI] [PubMed] [Google Scholar]
- 20.Gathmann B, Mahlaoui N, Gérard L, Oksenhendler E, Warnatz K, Schulze I, et al. Clinical picture and treatment of 2212 patients with common variable immunodeficiency. J Allergy Clin Immunol. 2014;134:116–26. e11. doi: 10.1016/j.jaci.2013.12.1077. [DOI] [PubMed] [Google Scholar]
- 21.Cunningham-Rundles C, Bodian C. Common variable immunodeficiency: clinical and immunological features of 248 patients. Clin Immunol. 1999;92:34–48. doi: 10.1006/clim.1999.4725. [DOI] [PubMed] [Google Scholar]
- 22.Mannon PJ, Fuss IJ, Dill S, Friend J, Groden C, Hornung R, et al. Excess IL-12 but not IL-23 accompanies the inflammatory bowel disease associated with common variable immunodeficiency. Gastroenterology. 2006;131:748–56. doi: 10.1053/j.gastro.2006.06.022. [DOI] [PubMed] [Google Scholar]
- 23.Baumgart DC, Sandborn WJ. Crohn’s disease. Lancet Elsevier Ltd. 2012;380:1590–605. doi: 10.1016/S0140-6736(12)60026-9. [DOI] [PubMed] [Google Scholar]
- 24.Mannon PJ, Fuss IJ, Mayer L, Elson CO, Sandborn WJ, Present D, et al. Anti-interleukin-12 antibody for active Crohn’s disease. N Engl J Med. 2004;351:2069–79. doi: 10.1056/NEJMoa033402. [DOI] [PubMed] [Google Scholar]
- 25.Sandborn WJ, Gasink C, Gao L-L, Blank Ma, Johanns J, Guzzo C, et al. Ustekinumab induction and maintenance therapy in refractory Crohn’s disease. N Engl J Med. 2012;367:1519–28. doi: 10.1056/NEJMoa1203572. [DOI] [PubMed] [Google Scholar]
- 26.Peters CP, Mjösberg JM, Bernink JH, Spits H. Innate lymphoid cells in inflammatory bowel diseases. Immunol Lett. 2015 doi: 10.1016/j.imlet.2015.10.004. [DOI] [PubMed] [Google Scholar]
- **27.Cols M, Rahman A, Maglione PJ, Garcia-Carmona Y, Simchoni N, Ko H-BM, et al. Expansion of inflammatory innate lymphoid cells in patients with common variable immune deficiency. J Allergy Clin Immunol. 2015 doi: 10.1016/j.jaci.2015.09.013. Lymphoid cells were identified and analyzed from peripheral blood and GI tissue of CVID patients (31 with and 24 without inflammatory/autoimmune complications). In the blood of CVID patients with inflammatory associated disorders, type 3 ILC and -associated cytokines were expanded. Moreover, numerous IFNγ+RORγt+CD3− were found in GI biopsies of patients with IBD-like suggesting a role in this mucosal inflammation. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sanges M, Spadaro G, Miniero M, Mattera D, Sollazzo R, D’Armiento FP, et al. Efficacy of subcutaneous immunoglobulins in primary immunodeficiency with Crohn’s-like phenotype: report of a case. Eur Rev Med Pharmacol Sci. 2015;19:2641–5. [PubMed] [Google Scholar]
- 29.Pignata C, Budillon G, Monaco G, Nani E, Cuomo R, Parrilli G, et al. Jejunal bacterial overgrowth and intestinal permeability in children with immunodeficiency syndromes. Gut. 1990;31:879–82. doi: 10.1136/gut.31.8.879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Biagi F, Bianchi PI, Zilli A, Marchese A, Luinetti O, Lougaris V, et al. The significance of duodenal mucosal atrophy in patients with common variable immunodeficiency: A clinical and histopathologic study. Am J Clin Pathol. 2012;138:185–9. doi: 10.1309/AJCPEIILH2C0WFYE. [DOI] [PubMed] [Google Scholar]
- 31.Boursiquot J-N, Gérard L, Malphettes M, Fieschi C, Galicier L, Boutboul D, et al. Granulomatous disease in CVID: retrospective analysis of clinical characteristics and treatment efficacy in a cohort of 59 patients. J Clin Immunol. 2013;33:84–95. doi: 10.1007/s10875-012-9778-9. [DOI] [PubMed] [Google Scholar]
- 32.Ardeniz O, Cunningham-Rundles C. Granulomatous disease in common variable immunodeficiency. Clin Immunol. 2009;133:198–207. doi: 10.1016/j.clim.2009.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chase NM, Verbsky JW, Hintermeyer MK, Waukau JK, Tomita-Mitchell A, Casper JT, et al. Use of Combination Chemotherapy for Treatment of Granulomatous and Lymphocytic Interstitial Lung Disease (GLILD) in Patients with Common Variable Immunodeficiency (CVID) J Clin Immunol. 2012;33:30–9. doi: 10.1007/s10875-012-9755-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cunningham-Rundles C. The many faces of common variable immunodeficiency. Hematology Am Soc Hematol Educ Program. 2012;2012:301–5. doi: 10.1182/asheducation-2012.1.301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sander CA, Medeiros LJ, Weiss LM, Yano T, Sneller MC, Jaffe ES. Lymphoproliferative lesions in patients with common variable immunodeficiency syndrome. Am J Surg Pathol. 1992;16:1170–82. doi: 10.1097/00000478-199212000-00004. [DOI] [PubMed] [Google Scholar]
- 36.Dhalla F, da Silva SP, Lucas M, Travis S, Chapel H. Review of gastric cancer risk factors in patients with common variable immunodeficiency disorders, resulting in a proposal for a surveillance programme. Clin Exp Immunol. 2011;165:1–7. doi: 10.1111/j.1365-2249.2011.04384.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cassaro M, Rugge M, Gutierrez O, Leandro G, Graham DY, Genta RM. Topographic patterns of intestinal metaplasia and gastric cancer. Am J Gastroenterol. 2000;95:1431–8. doi: 10.1111/j.1572-0241.2000.02074.x. [DOI] [PubMed] [Google Scholar]
- 38.De Petris G, Dhungel BM, Chen L, Chang Y-HH. Gastric adenocarcinoma in common variable immunodeficiency: features of cancer and associated gastritis may be characteristic of the condition. Int J Surg Pathol. 2014;22:600–6. doi: 10.1177/1066896914532540. [DOI] [PubMed] [Google Scholar]
- 39.Chua I, Standish R, Lear S, Harbord M, Eren E, Raeiszadeh M, et al. Anti-tumour necrosis factor-alpha therapy for severe enteropathy in patients with common variable immunodeficiency (CVID) Clin Exp Immunol. 2007;150:306–11. doi: 10.1111/j.1365-2249.2007.03481.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vázquez-Morón JM, Pallarés-Manrique H, Martín-Suárez IJ, Benítez-Rodríguez B, Ramos-Lora M. Crohn’s-like disease in a patient with common variable immunodeficiency treated with azathioprine and adalimumab. Rev espan ola enfermedades Dig organo Of la Soc Espan ola Patol Dig. 105:299–302. doi: 10.4321/s1130-01082013000500010. [DOI] [PubMed] [Google Scholar]
- 41.Holland SM. Chronic granulomatous disease. Hematol Oncol Clin North Am. 2013;27:89–99. viii. doi: 10.1016/j.hoc.2012.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Winkelstein JA, Marino MC, Johnston RB, Boyle J, Curnutte J, Gallin JI, et al. Chronic granulomatous disease. Report on a national registry of 368 patients. Medicine (Baltimore) 2000;79:155–69. doi: 10.1097/00005792-200005000-00003. [DOI] [PubMed] [Google Scholar]
- 43.van den Berg JM, van Koppen E, Ahlin A, Belohradsky BH, Bernatowska E, Corbeel L, et al. Chronic granulomatous disease: the European experience. PLoS One. 2009;4:e5234. doi: 10.1371/journal.pone.0005234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jones LBKR, McGrogan P, Flood TJ, Gennery AR, Morton L, Thrasher A, et al. Special article: chronic granulomatous disease in the United Kingdom and Ireland: a comprehensive national patient-based registry. Clin Exp Immunol. 2008;152:211–8. doi: 10.1111/j.1365-2249.2008.03644.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Martire B, Rondelli R, Soresina A, Pignata C, Broccoletti T, Finocchi A, et al. Clinical features, long-term follow-up and outcome of a large cohort of patients with Chronic Granulomatous Disease: An Italian multicenter study. Clin Immunol. 2008;126:155–64. doi: 10.1016/j.clim.2007.09.008. [DOI] [PubMed] [Google Scholar]
- 46.Raptaki M, Varela I, Spanou K, Tzanoudaki M, Tantou S, Liatsis M, et al. Chronic granulomatous disease: a 25-year patient registry based on a multistep diagnostic procedure, from the referral center for primary immunodeficiencies in Greece. J Clin Immunol. 2013;33:1302–9. doi: 10.1007/s10875-013-9940-z. [DOI] [PubMed] [Google Scholar]
- **47.Magnani A, Brosselin P, Beauté J, de Vergnes N, Mouy R, Debré M, et al. Inflammatory manifestations in a single-center cohort of patients with chronic granulomatous disease. J Allergy Clin Immunol. 2014;134:655–62. e8. doi: 10.1016/j.jaci.2014.04.014. 68 on 98 analyzed CGD patients experienced inflammatory episodes. GI tract was the most commonly affected organ (88% of episodes) and patients with X-linked CGD were at higher risk of inflammatory episodes compared to patients with autosomal-recessive CGD (RR 2.2, 95% CI=1.5–3.5) [DOI] [PubMed] [Google Scholar]
- 48.Huang a, Abbasakoor F, Vaizey CJ. Gastrointestinal manifestations of chronic granulomatous disease. Colorectal Dis. 2006;8:637–44. doi: 10.1111/j.1463-1318.2006.01030.x. [DOI] [PubMed] [Google Scholar]
- 49.Marciano BE, Rosenzweig SD, Kleiner DE, Anderson VL, Darnell DN, Anaya-O’Brien S, et al. Gastrointestinal involvement in chronic granulomatous disease. Pediatrics. 2004;114:462–8. doi: 10.1542/peds.114.2.462. [DOI] [PubMed] [Google Scholar]
- 50.Yu JE, De Ravin SS, Uzel G, Landers C, Targan S, Malech HL, et al. High levels of Crohn’s disease-associated anti-microbial antibodies are present and independent of colitis in chronic granulomatous disease. Clin Immunol Elsevier Inc. 2011;138:14–22. doi: 10.1016/j.clim.2010.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Greenberg DE, Ding L, Zelazny AM, Stock F, Wong A, Anderson VL, et al. A novel bacterium associated with lymphadenitis in a patient with chronic granulomatous disease. PLoS Pathog. 2006;2:e28. doi: 10.1371/journal.ppat.0020028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Marks DJB, Miyagi K, Rahman FZ, Novelli M, Bloom SL, Segal AW. Inflammatory bowel disease in CGD reproduces the clinicopathological features of Crohn’s disease. Am J Gastroenterol. 2009;104:117–24. doi: 10.1038/ajg.2008.72. [DOI] [PubMed] [Google Scholar]
- 53.Alimchandani M, Lai J-P, Aung PP, Khangura S, Kamal N, Gallin JI, et al. Gastrointestinal histopathology in chronic granulomatous disease: a study of 87 patients. Am J Surg Pathol. 2013;37:1365–72. doi: 10.1097/PAS.0b013e318297427d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- **54.Khangura SK, Kamal N, Ho N, Quezado M, Zhao X, Marciano B, et al. Clin Gastroenterol Hepatol. Elsevier Ltd; 2015. Gastrointestinal Features of Chronic Granulomatous Disease Found During Endoscopy. 211 GI tract endoscopies were performed at the NIH on 78 CGD patients.. Inflammatory lesions were frequent in all parts of the GI tract with a classically skip pattern. Ano-rectal area was the most often involved GI tract part (93% of patients with colonic inflammation) including sometimes fistulae. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Noel N, Mahlaoui N, Blanche S, Suarez F, Coignard-Biehler H, Durieu I, et al. Efficacy and safety of thalidomide in patients with inflammatory manifestations of chronic granulomatous disease: a retrospective case series. J Allergy Clin Immunol. 2013;132:997–1000. e1–4. doi: 10.1016/j.jaci.2013.04.059. [DOI] [PubMed] [Google Scholar]
- 56.Rosh JR, Tang HB, Mayer L, Groisman G, Abraham SK, Prince A. Treatment of intractable gastrointestinal manifestations of chronic granulomatous disease with cyclosporine. J Pediatr. 1995;126:143–5. doi: 10.1016/s0022-3476(95)70519-8. [DOI] [PubMed] [Google Scholar]
- 57.Uzel G, Orange JS, Poliak N, Marciano BE, Heller T, Holland SM. Complications of tumor necrosis factor-α blockade in chronic granulomatous disease-related colitis. Clin Infect Dis. 2010;51:1429–34. doi: 10.1086/657308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Myrup B, Valerius NH, Mortensen PB. Treatment of enteritis in chronic granulomatous disease with granulocyte colony stimulating factor. Gut. 1998;42:127–30. doi: 10.1136/gut.42.1.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Güngör T, Teira P, Slatter M, Stussi G, Stepensky P, Moshous D, et al. Reduced-intensity conditioning and HLA-matched haemopoietic stem-cell transplantation in patients with chronic granulomatous disease: a prospective multicentre study. Lancet (London, England) 2014;383:436–48. doi: 10.1016/S0140-6736(13)62069-3. [DOI] [PubMed] [Google Scholar]