Abstract
Introduction
SCA38 (MIM 611805) caused by mutations within the ELOVL5 gene, which encodes an enzyme involved in the synthesis of long-chain fatty acids with a high and specific expression in Purkinje cells, has recently been identified.
Objective
The present study was aimed at describing the clinical and neuroimaging features, and the natural history of SCA38.
Methods
We extended our clinical and brain neuroimaging data on SCA38 including 21 cases from three Italian families. All had the ELOVL5 c.689G > T (p.Gly230Val) missense mutation.
Results
Age at disease onset was in the fourth decade of life. The presenting features were nystagmus (100% of cases) and slowly progressive gait ataxia (95%). Frequent signs and symptoms included pes cavus (82%) and hyposmia (76%); rarer symptoms were hearing loss (33%) and anxiety disorder (33%). The disease progressed with cerebellar symptoms such as limb ataxia, dysarthria, dysphagia, and ophtalmoparesis followed in the later stages by ophtalmoplegia. Peripheral nervous system involvement was present in the last phase of disease with sensory loss. Dementia or extrapyramidal signs were not detected. Significant loss of abilities of daily living was reported only after 20 years of the disease. Brain imaging documented cerebellar atrophy with sparing of cerebral cortex and no white matter disease.
Conclusions
SCA38 is a rare form of inherited ataxia with characteristic clinical features, including pes cavus and hyposmia, that may guide genetic screening and prompt diagnosis in light of possible future therapeutic interventions.
Keywords: SCA38, ELOVL5, Gene, Mutation, Ataxia
Highlights
-
•
SCA38, due to ELOVL5 gene mutation, is a rare form of inherited ataxia.
-
•
Characteristic clinical features include pes cavus and hyposmia.
-
•
Limb ataxia, dysarthria, dysphagia, and ophtalmoparesis were observed during disease course.
-
•
SCA38 is slowly progressive; loss of activities of daily living were observed only after 20 years.
-
•
Imaging documented cerebellar atrophy with sparing of cerebral cortex.
1. Introduction
Autosomal dominant spinocerebellar ataxias (SCAs) are a highly heterogeneous group of movement disorders characterized by gait ataxia, eye movement deficits, dysarthria, and limb ataxia, usually associated with cerebellar atrophy [1]. To date, thirty-seven loci have been associated with SCA phenotypes with 29 defective genes identified (neuromuscular.wustl.edu/ataxia/domatax/html). However, many SCA families are still gene-orphans [1]. The majority of SCAs are caused by an expansion or insertion of a trinucleotide or polynucleotide repeat, including SCA1–3, SCA6–8, SCA10, SCA12, SCA17, SCA31, SCA36 and DRPLA. The remaining forms are caused by conventional mutations or re-arrangements in the corresponding genes [2].
Gait disorders are the initial symptom in two-thirds of all patients with SCA [3], and the clinical picture becomes more complex as the disease progresses [4], [5].
Phenotypic differences seem to allow a distinction between autosomal dominant cerebellar ataxias caused by polyglutamine expansions and conventional mutations. Genetic anticipation is present in polyglutamine expansion SCAs, which generally have a disease onset in the third or fourth decade of life, inversely correlated to the number of CAG repeats in the corresponding gene [2], [6]. Polyglutamine SCAs are multisystem diseases with rapid progression, while conventional mutations are generally associated with a pure form of ataxia and slow disease progression [1].
Polyglutamine expansions are more frequently associated with more severe forms of SCA and thus more extensive atrophy affecting the brainstem [7].
Since the phenotypes may be very heterogeneous and sometimes confusing between the different SCAs, characteristic signs are warranted for a rapid clinical diagnosis.
We recently identified SCA38 (MIM 611805) caused by mutations within the ELOVL5 gene [8], which encodes an enzyme involved in the synthesis of long-chain fatty acids with a high and specific expression in Purkinje cells [9].
In the present study, we extended the analysis of the SCA38 phenotype expanding the patients' cohort and reporting the clinical features, course of the disease and neuroimaging hallmarks of the disease.
2. Methods
2.1. Subjects
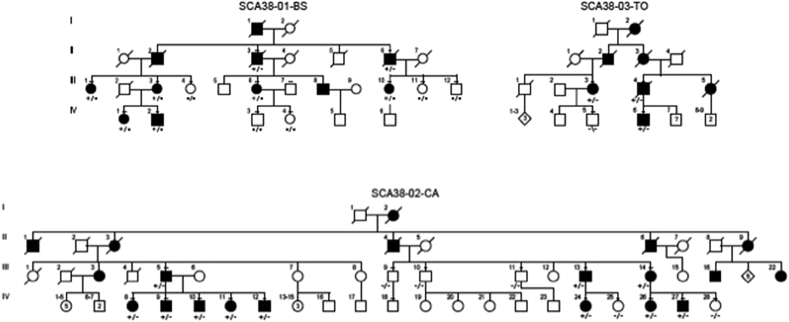
We collected clinical and neuroradiological information from twenty-one SCA38 cases carrying the c.689G > T (p.Gly230Val) mutation in ELOVL5. All patients belonged to the three Italian pedigrees described in the paper which originally reported the gene identification [8], where we also summarized the phenotype of three cases, further characterized herein (SCA38-01-BS, SCA38-02-CA and SCA38-03-TO, Fig. 1). An experienced clinician recorded clinical data of all studied cases.
Fig. 1.
Pedigrees of the three Italian families with SCA38. Unfilled symbols indicate unaffected family members, and solid black symbols affected members. A line above the symbol indicates individuals for whom DNA was available. The genotype of the ELOVL5 mutations is indicated below each tested subject. Individuals carrying ELOVL5 mutation are indicated by a “±” below the symbol.
Patients had been evaluated over the course of the disease, and signs and symptoms were periodically recorded each year. Available clinical evaluations were divided into three groups according to the onset of the disease: first decade (0–10 years from the onset of symptoms), second decade (11–20 years from the onset of symptoms), and third decade and over (over 20 years from the onset of symptoms). For each decade, we measured the incidence of symptoms by using a person-years approach. Cumulative incidence was expressed as the number of cases with the considered symptom out of the total number of person-years individuals at risk [10].
Clinical assessment was further accomplished by structural and functional imaging studies, and electrophysiological examinations, according to standard procedures. Patients underwent brain MRI (1.5 T), and atrophy was examined by visual evaluation of T1 sequences by expert neurologists and scored as absent (−) or present (+: mild, ++: moderate, +++: severe). Electromyography/electroneurography examinations considered the presence of peripheral neuropathy, according to standard procedures in the different participating centres.
2.2. Mutation screening of ELOVL5
We screened the c.689G > T (p.Gly230Val) mutation in exon 7 of ELOVL5 by Sanger sequencing. PCR conditions were: 10 μMol primers (5′-gtatgtgttgtgttcatgaagtgactg; 5′-gctccacatgcccattaagtaaata), 200 μMol dNTPs, 60 ng of genomic DNA, and 0.5 units of KAPA-fast 2G kit (Kapa Biosystems, Inc., MA, USA) with a 25 μl final volume under standard amplification conditions (56 °C annealing temperature). PCR products were purified using Agencourt AMPure XP-PCR Purification (Beckman Coulter, Miami, FL, USA) and sequenced with the Big Dye v3.1 kit (Applied Biosystems, Foster City, CA, USA). Products were purified using Agencourt CleanSEQ-Dye Terminator Removal (Beckman Coulter) and run on a ABI-3730 platform, using POP7 polymer (Applied Biosystems). Electropherograms were analysed with the SeqScape software version 2.6 (Applied Biosystems), as previously described [8].
2.3. Standard protocol approvals
The study conformed to the Declaration of Helsinki principles. The study protocol was approved by the Ethics Committee of Brescia Hospital, Brescia, Italy.
All participants signed a written informed consent form.
3. Results
3.1. Clinical features and disease course
We collected clinical data from 21 patients with the c.689G > T (p.Gly230Val) mutation in the ELOVL5 gene from three Italian families. The disease showed a pattern of fully penetrant autosomal dominant disease by 50 years of age.
All affected members showed a relatively homogeneous phenotype with reference to neurological symptoms and disease progression. As reported in Table 1, the mean age at onset was 39.1 years (range 26–50 years) with no evidence of genetic anticipation, and no significant gender bias (10 females, 11 males).
Table 1.
Clinical features of SCA38 cases.
| Patient | Pedigree | Gender | Age at last evaluation | Age at onset | Symptom at onset | Additional clinical features |
|||
|---|---|---|---|---|---|---|---|---|---|
| Pes cavus | Hearing loss | Hyposmia | Anxiety disorder | ||||||
| III-6 | SCA38-01-BS | F | 59 | 45 | gait ataxia | – | - | + | - |
| III-10 | SCA38-01-BS | F | 60 | 38 | gait ataxia | + | - | + | - |
| III-1 | SCA38-01-BS | F | 76 | 40 | gait ataxia | + | - | + | - |
| III-2 | SCA38-01-BS | F | 74 | 33 | gait ataxia | n.a. | + | n.a. | - |
| II-6 | SCA38-01-BS | M | 80 (deceased) | 50 | gait ataxia | n.a. | - | n.a. | - |
| II-3 | SCA38-01-BS | M | 91 (deceased) | 45 | gait ataxia | n.a. | - | n.a. | - |
| IV-2 | SCA38-01-BS | M | 43 | 38 | gait ataxia | - | - | - | - |
| IV-1 | SCA38-01-BS | F | 45 | 38 | gait ataxia | - | - | - | - |
| IV-8 | SCA38-02-CA | F | 48 | 35 | gait ataxia | + | + | + | + |
| III-5 | SCA38-02-CA | M | 80 | 38 | gait ataxia | + | + | + | + |
| IV-11 | SCA38-02-CA | F | 43 | 38 | gait ataxia | + | + | + | + |
| IV-9 | SCA38-02-CA | M | 47 | 37 | gait ataxia | + | + | + | + |
| IV-12 | SCA38-02-CA | M | 38 | 34 | gait ataxia | + | + | + | + |
| IV-10 | SCA38-02-CA | M | 46 | 46 | gait ataxia | + | + | - | - |
| III-14 | SCA38-02-CA | F | 55 | 37 | gait ataxia | + | - | + | + |
| III-13 | SCA38-02-CA | M | 66 | 30 | gait ataxia | + | - | n.a. | + |
| IV-27 | SCA38-02-CA | M | 32 | 30 | gait ataxia | n.a. | - | + | - |
| IV-26 | SCA38-02-CA | F | 33 | 26 | gait ataxia | + | - | + | - |
| III-4 | SCA38-03-TO | M | 70 (deceased) | 50 | gait ataxia, dys. | + | - | + | - |
| III-3 | SCA38-03-TO | F | 72 | 50 | gait ataxia | + | - | - | - |
| IV-6 | SCA38-03-TO | M | 47 | 44 | dysarthria | + | - | + | - |
| 47% F (10/21) | - | mean 39.1 | - | 82.3% (14/17) | 33.3% (7/21) | 76.5% (13/17) | 33.3% (7/21) | ||
F: female; M: male; dys.: dysarthria; n.a.: not available; +: presence of sign/symptom; -: absence of sign/symptom.
The first sign of the disease was unbalanced gait, and in some cases slurred speech. At neurological examination, patients presented nystagmus, in the lateral and vertical gaze (100%), and gait ataxia (95%). In one case, dysarthria was the first presenting symptom (5%), and in a second dysarthria was the first presenting symptom along with gait ataxia (5%).
At the first neurological examination, further features were observed in the majority of patients such as pes cavus (14/17, 82.3%) and hyposmia (13/17, 76.5%). Less frequently we observed hearing loss (7/21, 33.3%) and anxiety disorder (7/21, 33.3%), the two latter symptoms were present only in patients from SCA38-02-CA families. No other comorbidities were significantly associated with the disease, including epilepsy and significant metabolic diseases.
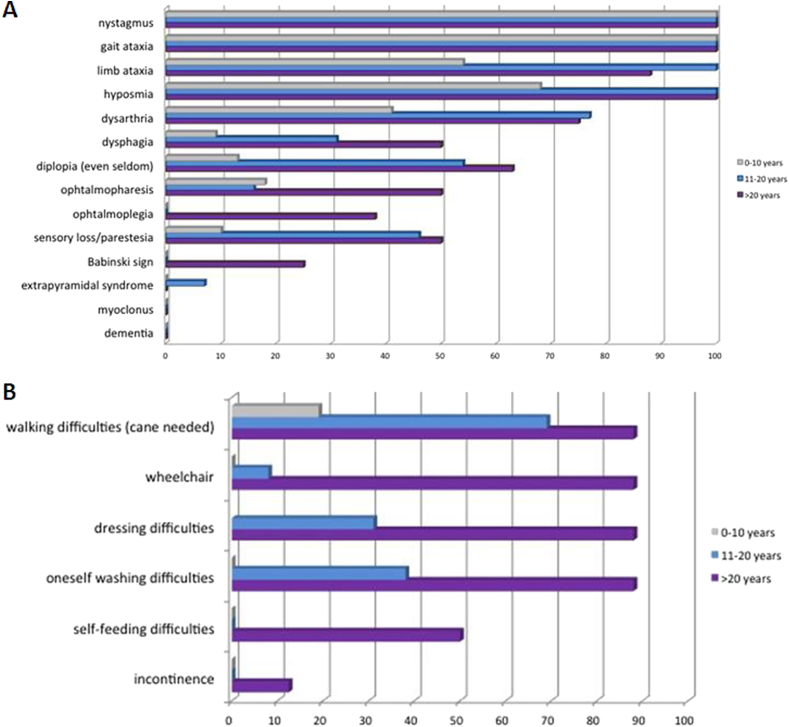
Forty-three cumulative clinical evaluations were available: 22 in the first decade from onset, 13 in the second decade, and 8 in the third decade (Fig. 2, panel A). Gait ataxia and nystagmus were the most frequent features in the early disease stage, whereas limb ataxia, and dysarthria were present in almost half of the cases in the first decade, and increased in frequency in the more advanced stages. Dysphagia was a rare symptom in the first two decades from onset. Half of the patients developed dysphagia in the third decade.
Fig. 2.
Signs and symptoms (panel A) and loss of activities of daily living (panel B) of SCA38 patients according to decades from disease onset. In grey, symptoms reported in the first 10 years of the disease, in blue those reported from 10 to 20 years of the disease, in violet those reported over 20 years from the onset of symptoms. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
At the beginning of the disease ocular anomalies included diplopia on the horizontal line and were present in half of the cases in the second decade. As the disease progressed, patients developed ophtalmoparesis, which became ophtalmoplegia in almost 40% of cases, after 20 years of the disease.
Sensory loss increased in frequency over time, being present in 40–50% of patients after 10 years from onset. Babinski reflex was rarely reported, and only in the later stages of the disease.
Neither cognitive impairment, nor bradykinesia, tremors, hypertonia, myoclonus nor dystonia was associated with SCA38.
Overall, SCA38 was a slowly progressive disorder (Fig. 2, panel B). In the first decade of the disease, all patients were independent in basic activities of daily living. In the second decade, 70% of patients had walking difficulties and a cane was needed, and almost 30% had dressing and washing difficulties. In the third decade of the disease, almost 80% of patients were wheelchair-bound and not independent in basic activities of daily living; half of them had to be helped with feeding, whereas incontinence was a very rare finding.
The mean disease duration from onset to death (available in 3 patients) was 41 years (range 20–52). The cause of death was hearth disease (2 cases) and pulmonary infection (1 case) at the age of 70, 80 and 91 years old, respectively.
3.2. Neuroimaging features
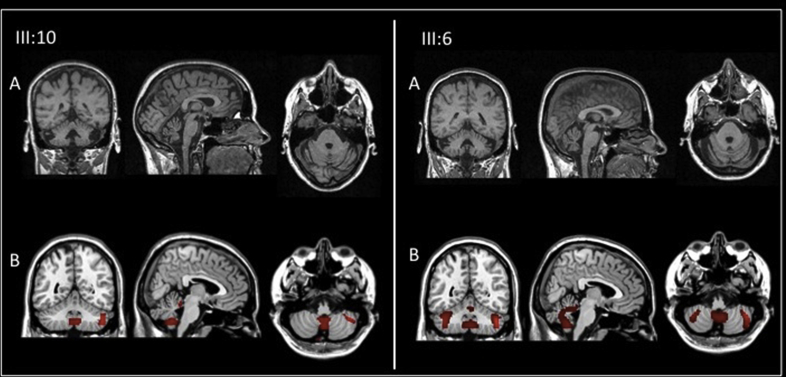
Brain MRI showed mild cerebellar atrophy mainly affecting the vermis, with normal brainstem and no cerebral cortex atrophy. These findings were supported by functional brain FDG-Positron Emission Tomography (FDG-PET) studies, which confirmed selective hypometabolism of the cerebellar vermis and of the cerebellar hemisphere to a lesser extent (in Fig. 3, structural and functional brain studies in two SCA38 patients, namely cases III:10 and III:6, family SCA38-01-BS).
Fig. 3.
Structural and functional neuroimaging findings in two patients with SCA38 (SCA38-01-BS III:10 and SCA38-01-BS III:6). Panel A. Coronal, sagittal and axial magnetic resonance imaging sections (MRI). Panel B. FDG-PET images were processed by Statistical Parametric Mapping (SPM8), and single-subject analysis using a group of 19 healthy controls (mean age: 53 years; female: 59%) was performed (patient < controls) and results were superimposed to T1 MRI template. Statistical threshold was set at P < 0.001, uncorrected for multiple comparisons, with voxel threshold = 100 voxels.
As reported in Supplementary Table, visual rating of the available MRI scans over the course of the disease (14 patients evaluated over time with 25 MRI scans available) confirmed selective cerebellar atrophy with spared brainstem and cerebral cortex in almost all cases. No significant white matter disease was observed. Only one patient had significant cortical atrophy and white matter abnormalities, but along with significant cerebrovascular comorbidities and previous strokes (III:4, family SCA38-03-TO).
3.3. Electrophysiological studies
To further characterize SCA38, peripheral nerve electrophysiological studies and evoked potentials were performed. The conduction velocities study and electromyography (EMG) denoted sensory-motor axonal polyneuropathy at four limbs, and was pathological in all 9 available cases but one, who performed EMG one year from disease onset.
Auditory evoked potentials, markers of brainstem damage, were available in 12 of the 20 patients, and they were found to be pathological in 92% of cases (11/12). Somatosensory and motor evoked potentials led to variable findings, showing alterations in 33% (3/9 and 1/3) of patients, respectively, and in later disease stages. Visual evoked potentials were found to be unremarkable overall in all 8 cases available.
4. Discussion
In this study, we provide clinical and neuroradiological details on SCA38, a recently described form of autosomal dominant ataxia [8]. The availability of cases for phenotype characterization is presently limited to three Italian families, which were however enough to allow us to gather 21 cases. All have the same ELOVL5 c.689G > T (p.Gly230Val) mutation: two of the families were related to one another, whereas for the third a common ancestor was not demonstrated [8].
Cases with different mutations may in future reveal a broader phenotypic spectrum, but on the other hand the study of many patients with the same mutation is a unique opportunity to define the disease characteristics associated with the shared variant.
Conventional mutations in spinocerebellar ataxia genes are suggested to lead to a milder phenotype and a slower progressive disorder if compared to polyglutamine expansions [1]. SCA38 seems to confirm this rule, with an onset in the fourth-fifth decade of life, and complete penetrance by age 50 is a common finding, shared by many forms of SCAs [2].
The neurological picture is that of a pure cerebellar ataxia with symptoms including gait ataxia, nystagmus and dysarthria as the foremost manifestations. However, two features seem peculiar in SCA38 and may help the diagnosis: pes cavus without paraparesis (82%) and early hyposmia/anosmia (73%). Pes cavus has never been described in SCAs, while olfactory deficit has recently been reported in SCA3 [11], [12], SCA7 [13] and in SCA2 [14].
Other features may help in the differential diagnosis of SCA: 1) eye movement anomalies, frequently associated with polyglutamine expansion SCAs [15], [16], and with SCA28 in which ophtalmoplegia and ptosis have been described at later stages [18]; 2) peripheral sensory loss, although quite a common symptom in hereditary ataxias [19], [20]; 3) the absence of cognitive decline and extrapyramidal movement disorders suggests the lack of involvement of cortical and subcortical brain regions, and excludes a subset of autosomal dominant ataxias (e.g., SCA8, 13, 17, 21 and DRPLA).
Hearing loss detected at diagnosis, and supported by auditory evoked potential deficit, was not very common (33.3%), previously reported only in SCA36 [21], and debated in SCA31 [22]. Anxiety disorder might be considered an additional symptom for SCA38, present in 33.3%, of the reported cases; however, this was present in only one family, and further cases may elucidate the specific association between SCA38 and anxiety disorder.
The disease is slowly progressive with significant functional impairment occurring only after the third decade from the onset of the disease.
Brain MRI showed selective cerebellar atrophy without significant involvement of the cerebral hemispheres. No significant white matter disease was associated with SCA38.
SCA38 and SCA34 are two spinocerebellar ataxias associated with the synthesis of Poly Unsaturated Fatty Acids (PUFAs) with more than 20 carbon atoms [23]. Pathogenic mechanisms are still to be elucidated in both these diseases, although our previous work detected decreased level of ELOVL5 products in patients' serum, indicating that Gly230Val mutation might affect the elongase activity of this enzyme causing a loss of function mechanism. In these new series of patients, we could not collect further serum samples for measuring PUFA levels.
On the other hand, we had initial evidence that the mutant ELOVL5 protein is not degraded, but upregulated and delocalized suggesting that it gains a toxic function. Both loss and gain of function might therefore be effective in causing SCA38.
Interestingly, ELOVL4, another elongase involved in the synthesis of fatty acids, has recently been described in a single Canadian family with an autosomal dominant form of cerebellar ataxia with or without erythrokeratodermia (SCA34, OMIM133190) [24]. The clinical features of SCA34 resembled to some extent those of SCA38, being slowly progressive with onset in the fourth/fifth decade of life, with no cognitive impairment and axonal peripheral neuropathy [25], [26].
Mutations in ELOVL4 have been associated with autosomal dominant Startgard macular dystrophy disease type 3 (MIM 600110) and an autosomal recessive form of spastic quadriplegia with ichthyosis and mental retardation (MIM 614457). The heterogeneous clinical picture associated with ELOVL4 mutations is intriguing and mirrors the role of ELOVL4 products in the skin, eye and brain. We may speculate that mutations affecting specific ELOVL5 domains, or both alleles, might also therefore be associated with clinical features that are phenotypically different from SCA38.
5. Conclusion
In conclusion, we extended the survey and detailed the phenotypic spectrum associated with the Gly230Val mutation in ELOVL5. Our study suggests that the presence of pes cavus without paraparesis and early hyposmia/anosmia, in cases with a pure form of cerebellar ataxia would be hallmarks for testing SCA38 as the first tier genetic analysis. Ophtalmopharesis and ophthalmoplegia may be considered in advanced cases.
Further assessment of disease heterogeneity and further characterization of the clinical picture of SCA38 by evaluation of other causative mutations will be warranted.
The identification of further cases may be also instrumental in clarifying the pathogenic mechanism of SCA38 in light of future therapeutic approaches.
Author's contributions
All authors made substantial contributions to the conception and design, and/or acquisition of data, and/or analysis and interpretation of data.
In detail:
Barbara Borroni: first draft of the manuscript, patient cohort, analysis and interpretation of data, study conceptualization and design.
Eleonora Di Gregorio: genetic analysis, critical revision of the manuscript for content.
Giovanna Vaula: patient cohort, critical revision of the manuscript for content.
Laura Orsi: patient cohort, critical revision of the manuscript for content.
Chiara Costanzi: patient cohort, critical revision of the manuscript for content.
Filippo Tempia: critical revision of the manuscript for content.
Nico Mitro: critical revision of the manuscript for content.
Donatella Caruso: critical revision of the manuscript for content.
Lorenzo Pinessi: critical revision of the manuscript for content.
Marta Manes: patient cohort, critical revision of the manuscript for content.
Alessandro Padovani: critical revision of the manuscript for content.
Alfredo Brusco: genetic analysis, analysis and interpretation of data, critical revision of the manuscript for content.
Loredana Boccone: patient cohort, analysis and interpretation of data, critical revision of the manuscript for content.
Disclosures
Barbara Borroni: no disclosures.
Eleonora Di Gregorio: no disclosures.
Giovanna Vaula: no disclosures.
Laura Orsi: no disclosures.
Chiara Costanzi: no disclosures.
Filippo Tempia: no disclosures.
Nico Mitro: no disclosures.
Donatella Caruso: no disclosures.
Lorenzo Pinessi: no disclosures.
Marta Manes: no disclosures.
Alessandro Padovani: no disclosures.
Alfredo Brusco: no disclosures.
Loredana Boccone: no disclosures.
Acknowledgements
The authors are indebted to the patients and their families for taking part into the study.
This work was supported by Fondazione Telethon grant number GGP14225.
Footnotes
Supplementary data related to this article can be found at http://dx.doi.org/10.1016/j.parkreldis.2016.04.030.
Appendix A. Supplementary data
The following is the supplementary data related to this article:
Magnetic Resonance Imaging (MRI) features of SCA38 patients according to disease duration.
References
- 1.Durr A. Autosomal dominant cerebellar ataxias: polyglutamine expansions and beyond. Lancet Neurol. 2010;9:885–894. doi: 10.1016/S1474-4422(10)70183-6. [DOI] [PubMed] [Google Scholar]
- 2.Schöls L., Bauer P., Schmidt T., Schulte T., Riess O. Autosomal dominant cerebellar ataxias: clinical features, genetics, and pathogenesis. Lancet Neurol. 2004;3:291–304. doi: 10.1016/S1474-4422(04)00737-9. [DOI] [PubMed] [Google Scholar]
- 3.Globas C., du Montcel S.T., Baliko L., Boesch S., Depondt C., DiDonato S., Durr A., Filla A., Klockgether T., Mariotti C., Melegh B., Rakowicz M., Ribai P., Rola R., Schmitz-Hubsch T., Szymanski S., Timmann D., Van de Warrenburg B.P., Bauer P., Schols L. Early symptoms in spinocerebellar ataxia type 1, 2, 3, and 6. Mov. Disord. 2008;23:2232–2238. doi: 10.1002/mds.22288. [DOI] [PubMed] [Google Scholar]
- 4.Yamada M., Sato T., Tsuji S., Takahashi H. CAG repeat disorder models and human neuropathology: similarities and differences. Acta Neuropathol. 2008;115:71–86. doi: 10.1007/s00401-007-0287-5. [DOI] [PubMed] [Google Scholar]
- 5.Rossi M., Perez-Lloret S., Doldan L., Cerquetti D., Balej J., Millar Vernetti P., Hawkes H., Cammarota A., Merello M. Autosomal dominant cerebellar ataxias: a systematic review of clinical features. Eur. J. Neurol. 2014;21:607–615. doi: 10.1111/ene.12350. [DOI] [PubMed] [Google Scholar]
- 6.Tezenas du Montcel S., Durr A., Bauer P., Figueroa K.P., Ichikawa Y., Brussino A., Forlani S., Rakowicz M., Schöls L., Mariotti C., van de Warrenburg B.P., Orsi L., Giunti P., Filla A., Szymanski S., Klockgether T., Berciano J., Pandolfo M., Boesch S., Melegh B., Timmann D., Mandich P., Camuzat A., Clinical Research Consortium for Spinocerebellar Ataxia (CRC-SCA); EUROSCA network, Goto J., Ashizawa T., Cazeneuve C., Tsuji S., Pulst S.M., Brusco A., Riess O., Brice A., Stevanin G. Modulation of the age at onset in spinocerebellar ataxia by CAG tracts in various genes. Brain. 2014;137:2444–2455. doi: 10.1093/brain/awu174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Guerrini L., Lolli F., Ginestroni A., Belli G., Della Nave R., Tessa C., Foresti S., Cosottini M., Piacentini S., Salvi F., Plasmati R., De Grandis D., Siciliano G., Filla A., Mascalchi M. Brainstem neurodegeneration correlates with clinical dysfunction in SCA1 but not in SCA2. A quantitative volumetric, diffusion and proton spectroscopy MR study. Brain. 2004;127:1785–1795. doi: 10.1093/brain/awh201. [DOI] [PubMed] [Google Scholar]
- 8.Di Gregorio E., Borroni B., Giorgio E., Lacerenza D., Ferrero M., Lo Buono N., Ragusa N., Mancini C., Gaussen M., Calcia A., Mitro N., Hoxha E., Mura I., Coviello D.A., Moon Y.A., Tesson C., Vaula G., Couarch P., Orsi L., Duregon E., Papotti M.G., Deleuze J.F., Imbert J., Costanzi C., Padovani A., Giunti P., Maillet-Vioud M., Durr A., Brice A., Tempia F., Funaro A., Boccone L., Caruso D., Stevanin G., Brusco A. ELOVL5 mutations cause spinocerebellar ataxia 38. Am. J. Hum. Genet. 2014;95:209–217. doi: 10.1016/j.ajhg.2014.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Moon Y.A., Hammer R.E., Horton J.D. Deletion of ELOVL5 leads to fatty liver through activation of SREBP-1c in mice. J. Lipid Res. 2009;50:412–423. doi: 10.1194/jlr.M800383-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fletcher R., Fletcher S.W. Lippincott Williams & Wilkins; 2013. Clinical Epidemiology: the Essentials. [Google Scholar]
- 11.Moscovich M., Munhoz R.P., Teive H.A., Raskin S., Carvalho Mde J., Barbosa E.R., Ranvaud R., Liu J., McFarland K., Ashizawa T., Lees A.J., Silveira-Moriyama L. Olfactory impairment in familial ataxias. J. Neurol. Neurosurg. Psychiatry. 2012;83:970–974. doi: 10.1136/jnnp-2012-302770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Braga-Neto P., Felicio A.C., Hoexter M.Q., Pedroso J.L., Dutra L.A., Alessi H., Minett T., Santos-Galduroz R.F., da Rocha A.J., Garcia L.A., Bertolucci P.H., Bressan R.A., Barsottini O.G. Cognitive and olfactory deficits in Machado-Joseph disease: a dopamine transporter study. Parkinsonism Relat. Disord. 2012;18:854–858. doi: 10.1016/j.parkreldis.2012.04.015. [DOI] [PubMed] [Google Scholar]
- 13.Galvez V., Diaz R., Hernandez-Castillo C.R., Campos-Romo A., Fernandez-Ruiz J. Olfactory performance in spinocerebellar ataxia type 7 patients. Park. Relat. Disord. 2014;20:499–502. doi: 10.1016/j.parkreldis.2014.01.024. [DOI] [PubMed] [Google Scholar]
- 14.Velazquez-Perez L., Fernandez-Ruiz J., Diaz R., González R.P., Ochoa N.C., Cruz G.S., Mederos L.E., Góngora E.M., Hudson R., Drucker-Colin R. Spinocerebellar ataxia type 2 olfactory impairment shows a pattern similar to other major neurodegenerative diseases. J. Neurol. 2006;253:1165–1169. doi: 10.1007/s00415-006-0183-2. [DOI] [PubMed] [Google Scholar]
- 15.Rivaud-Pechoux S., Dürr A., Gaymard B., Cancel G., Ploner C.J., Agid Y., Brice A., Pierrot-Deseilligny C. Eye movement abnormalities correlate with genotype in autosomal dominant cerebellar ataxia type I. Ann. Neurol. 1998;43:297–302. doi: 10.1002/ana.410430306. [DOI] [PubMed] [Google Scholar]
- 16.Stevanin G., Dürr A., David G., Didierjean O., Cancel G., Rivaud S., Tourbah A., Warter J.M., Agid Y., Brice A. Clinical and molecular features of spinocerebellar ataxia type 6. Neurology. 1997;49:1243–1246. doi: 10.1212/wnl.49.5.1243. [DOI] [PubMed] [Google Scholar]
- 18.Cagnoli C., Mariotti C., Taroni F., Seri M., Brussino A., Michielotto C., Grisoli M., Di Bella D., Migone N., Gellera C., Di Donato S., Brusco A. SCA28, a novel form of autosomal dominant cerebellar ataxia on chromosome 18p11.22-q11.2. Brain. 2006;129:235–242. doi: 10.1093/brain/awh651. [DOI] [PubMed] [Google Scholar]
- 19.Whaley N.R., Fujioka S., Wszolek Z.K. Autosomal dominant cerebellar ataxia type I: a review of the phenotypic and genotypic characteristics. Orphanet J. Rare Dis. 2011;6:33. doi: 10.1186/1750-1172-6-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Berciano J., Garcia A., Infante J. Peripheral nerve involvement in hereditary cerebellar and multisystem degenerative disorders. Handb. Clin. Neurol. 2013;115:907–932. doi: 10.1016/B978-0-444-52902-2.00051-5. [DOI] [PubMed] [Google Scholar]
- 21.Ikeda Y., Ohta Y., Kurata T., Shiro Y., Takao Y., Abe K. Acoustic impairment is distinguishable clinical features of Asidan/SCA36. J. Neurol. Sci. 2013;324:109–112. doi: 10.1016/j.jns.2012.10.013. [DOI] [PubMed] [Google Scholar]
- 22.Ikeda Y., Nagai M., Kurata T., Yamashita T., Ohta Y., Nagotani S., Deguchi K., Takehisa Y., Shiro Y., Matsuura T., Abe K. Comparisons of acoustic function in SCA31 and other forms of ataxia. Neurol. Res. 2011;33:427–432. doi: 10.1179/1743132810Y.0000000011. [DOI] [PubMed] [Google Scholar]
- 23.Kihara A. Very long-chain fatty acids: elongation, physiology and related disorders. J. Biochem. 2012;152:387–395. doi: 10.1093/jb/mvs105. [DOI] [PubMed] [Google Scholar]
- 24.Cadieux-Dion M., Turcotte-Gauthier M., Noreau A., Martin C., Meloche C., Gravel M., Drouin C.A., Rouleau G.A., Nguyen D.K., Cosette P. Expanding the clinical phenotype associated with ELOVL4 mutation: study of a large French-Canadian family with autosomal dominant spinocerebellar ataxia and erythrokeratodermia. JAMA Neurol. 2014;71:470–475. doi: 10.1001/jamaneurol.2013.6337. [DOI] [PubMed] [Google Scholar]
- 25.Bourassa C.V., Raskin S., Serafini S., Teive H.A., Dion P.A., Rouleau G.A. A New ELOVL4 mutation in a case of Spinocerebellar Ataxia with erythrokeratodermia. JAMA Neurol. 2015;72:942–943. doi: 10.1001/jamaneurol.2015.0888. [DOI] [PubMed] [Google Scholar]
- 26.Ozaki K., Doi H., Mitsui J., Sato N., Iikuni Y., Majima T., Yamane K., Irioka T., Ishiura H., Doi K., Morishita S., Higashi M., Sekiguchi T., Koyama K., Ueda N., Miura Y., Miyatake S., Matsumoto N., Yokota T., Tanaka F., Tsuji S., Mizusawa H., Ishikawa K. A novel mutation in ELOVL4 leading to Spinocerebellar Ataxia (SCA) with the hot cross bun sign but lacking erythrokeratodermia: a broadened spectrum of SCA34. JAMA Neurol. 2015;72:797–805. doi: 10.1001/jamaneurol.2015.0610. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.