Abstract
Purpose
To assess the prevalence and severity of neurocognitive impairment in adult survivors of pediatric CNS tumors and to examine associated treatment exposures.
Patients and Methods
Participants included 224 survivors of CNS tumors who were treated at St Jude Children's Research Hospital (current median age [range], 26 years [19 to 53 years]; time from diagnosis, 18 years [11 to 42 years]) and completed neurocognitive testing. Information on cranial radiation therapy (CRT) doses and parameters of delivery were abstracted from medical records. The prevalence of severe impairment (ie, at least two standard deviations below normative mean) was compared across radiation treatment groups (no CRT, focal irradiation, craniospinal irradiation) using the χ2 test. Log-binomial models were used to estimate risk ratios (RRs) and corresponding 95% CIs for severe impairment.
Results
In multivariable models, craniospinal irradiation was associated with a 1.5- to threefold increased risk of severe impairment compared with no CRT (eg, intelligence: RR = 2.70; 95% CI, 1.37 to 5.34; memory: RR = 2.93; 95% CI, 1.69 to 5.08; executive function: RR = 1.74; 95% CI, 1.24 to 2.45). Seizures were associated with impaired academic performance (RR = 1.48; 95% CI, 1.02 to 2.14), attention (RR = 1.54; 95% CI, 1.12 to 2.13), and memory (RR = 1.44; 95% CI, 1.04 to 1.99). Hydrocephalus with shunt placement was associated with impaired intelligence (RR = 1.78; 95% CI, 1.12 to 2.82) and memory (RR = 1.42; 95% CI, 1.03 to 1.95). Differential follow-up time contributed to variability in prevalence estimates between survivors treated with older nonconformal and those treated with more contemporary conformal radiation therapy methods. Neurocognitive impairment was significantly associated with lower educational attainment, unemployment, and nonindependent living.
Conclusion
Survivors of pediatric CNS tumors are at risk of severe neurocognitive impairment in adulthood. The prevalence of severe impairment is greater than expected in the general population, even in the absence of CRT, and is associated with disrupted attainment of adult social milestones.
INTRODUCTION
The CNS is the site for nearly 20% of tumors diagnosed in children younger than 20 years of age, with an age-adjusted incidence rate of 3.1 per 100,000.1 Advances in treatment and supportive care have increased the 5-year survival from less than 60% in 1980 to more than 75% today,1 but late effects of therapy have emerged as a result of long-term survival.
Cranial radiation therapy (CRT) is an established risk factor for neurocognitive impairment in survivors of pediatric CNS tumors.2-4 Contemporary treatment protocols aim to maintain tumor control or improve overall and progression-free survival while reducing cognitive morbidities.5 One approach toward this goal, conformal or intensity modulated radiation therapy, uses three-dimensional (3D) imaging to precisely target tumor volume, thereby reducing the amount of radiation exposure to healthy brain tissue.6 Dose and target volume reduction has been shown to reduce the risk of cognitive morbidities in child and adolescent survivors.7-9 To our knowledge, no studies have reported on long-term neurocognitive outcomes in adult survivors after conformal radiation treatment.
Because brain development continues into adulthood, the degree and consequences of neurocognitive impairment may change during the decades after exposure to CRT. Despite clinical suggestion of the potential chronicity of neurocognitive morbidities in survivors of childhood CNS tumors, few studies have directly assessed long-term neurocognitive outcomes in adult survivors. In the few reports available,10-13 data are limited by exclusive reliance on survivor self-report of function,11 small sample sizes (ie, n ≤ 20),10,12 and restriction to homogeneous survivor groups (eg, medulloblastoma survivors).10,12 Therefore, the aims of this study were as follows: (1) to assess the prevalence of neurocognitive impairment in a large cohort of adult survivors of childhood CNS tumors using direct assessment and patient-reported outcomes; (2) to examine treatment exposures associated with neurocognitive impairment, including examination of outcomes by radiation delivery parameters; and (3) to report associations between neurocognitive impairment and adult social attainment.
PATIENTS AND METHODS
Study Population
The study design and characteristics of the St Jude Lifetime Cohort (SJLIFE) have been described previously.14,15 Briefly, SJLIFE is a dynamic cohort with longitudinal follow-up. Survivors are recruited randomly in blocks of 50. This Institutional Review Board–approved study is designed to examine the multifactorial cause and severity of long-term effects of childhood cancer and cancer-directed therapies. Individuals eligible for the current analysis were treated for childhood CNS tumors at St Jude Children’s Research Hospital, had a current age ≥ 18 years, and were ≥ 10 years from their original childhood cancer diagnosis. We identified 368 potentially eligible CNS tumor survivors as of June 30, 2014. Because eligibility for a neurocognitive evaluation in SJLIFE is based on the Children’s Oncology Group Long-term Follow up Guidelines,16,17 survivors who were not treated with neurosurgery and/or cranial irradiation were ineligible for the current analysis. In addition, survivors with preexisting neurologic conditions or injuries unrelated to cancer diagnosis or treatment were excluded. Of the 343 eligible survivors, 72.9% agreed to participate, and at the time of the analysis, 224 (65.3%) had completed neurocognitive evaluations (Fig 1).
Fig 1.
CONSORT diagram of study participation.
Procedures
Participants received comprehensive risk-based clinical and laboratory assessments consistent with the Children’s Oncology Group Long-term Follow up Guidelines. Medical record abstraction was performed to determine treatment exposure data, including the need for surgical resection, hydrocephalus with shunt placement, and seizures. Abstracted radiation therapy data were reviewed and coded centrally by the St Jude Children’s Research Hospital Radiation Dosimetry Core blind to functional status. Information on craniospinal irradiation (CSI) dose and focal or boost dose to the primary tumor site were abstracted through review of the treatment records and radiation films, and data included the dates of treatment, prescribed treatment doses, and technical parameters of delivery. This review yielded five primary treatment groups: (1) neurosurgery with or without chemotherapy (no CRT); (2) focal irradiation using two-dimensional treatment planning (2DRT); (3) focal irradiation using three-dimensional conformal radiation therapy (3DCRT); (4) CSI plus focal primary site boost using 2DRT (CSI + 2DRT); and (5) CSI plus focal primary site boost using 3DCRT (CSI + 3DCRT).
Neurocognitive testing was conducted during a 2-hour session in dedicated evaluation rooms. The order of testing was standardized to minimize the effects of interference and fatigue. Assessed domains (and instruments) included intelligence (Wechsler Abbreviated Scale of Intelligence18), academics (Woodcock Johnson Tests of Achievement-III: Basic Reading and Math Calculation19), memory (California Verbal Learning Test-II: Total Learning, Short-Delay Free Recall and Long-Delay Free Recall20), attention (Trail Making Test Part A21; Conners’ Continuous Performance Test-II Omissions and Variability22; Digit Span Forward subtest of the Wechsler Adult Intelligence Scale-III [WAIS-III]18), processing speed (Coding and Symbol Search subtests of the WAIS-III18; Continuous Performance Test-II Hit Rate22), fine motor dexterity (Grooved Pegboard21), and executive function (Trail Making Test Part B21; Controlled Oral Word Association Test21; Digit Span Backward subtest of the WAIS-III18). Survivors also self-reported symptoms of executive dysfunction (Behavior Rating Inventory of Executive Function23). Each test yields standard scores (age-adjusted means and standard deviations) using test-specific standardization norms. Adult social attainment was measured on the basis of educational attainment, current employment status, and independent living. These questions and response formats parallel those used in the Childhood Cancer Survivor Study.24,25
Data Analysis
We estimated overall and stratified (by radiation exposure: no CRT, focal irradiation, and CSI) means with standard deviations or medians with ranges, as appropriate. Frequency distributions of categorical variables were compared among treatment groups using the χ2 test, and continuous variables were compared using the Kruskal-Wallis one-way analysis of variance test. P < .05 suggested statistical differences in variable distributions between groups. On the basis of age-adjusted norms, we estimated the percent of survivors with severe neurocognitive or behavioral impairments in each subdomain (ie, age-adjusted z score ≤ −2.0). Age-standardized morbidity ratios (MRs) and corresponding 95% CIs were used to compare the observed prevalence of severe neurocognitive and behavioral impairments among survivors with the expected prevalence of severe impairments in the general population (ie, on the basis of age-specific population norms). In addition, we used log-binomial models to estimate risk ratios (RRs) and corresponding 95% CIs among survivors for associations between exposures and severe neurocognitive or behavioral impairments. Covariate selection was informed using the Akaike information criterion (AIC), where the AIC did not differ significantly from the optimal AIC, and a priori selected exposures of interest (eg, age, sex, cranial irradiation exposure, shunt placement, and seizures). We used Poisson regression to estimate rate ratios and corresponding 95% CIs for the associations between two-dimensional (2D) and 3D treatment eras, separately for those treated with or without CSI, and for severe neurocognitive impairment after adjustment for age at neurocognitive assessment, sex, and race. Finally, given the short follow-up duration for the 3D treatment groups compared with that of the 2D groups, we explored the sensitivity of rate ratio estimates to additional follow-up for the effect of 3D treatment by adding 5-year increments of additional follow-up to the 3D groups to more closely simulate the follow-up distribution of the 2D groups and reestimated the rate ratios. Given that all analyses were based on a priori hypotheses, the results were not adjusted for multiple comparisons.26,27 Analyses were conducted using SAS (SAS Institute, Cary, NC), R (R Foundation, Vienna, Austria), and STATA (STATA, College Station, TX).
RESULTS
Survivor Characteristics
A modestly larger proportion of participants than nonparticipants received chemotherapy and cranial irradiation, underwent surgical resection, had seizures, and had infratentorial disease, although these differences were not statistically significant. The distribution of primary cancer diagnosis differed between participants and nonparticipants (P = .02; Appendix Table A1, online only). The median age of survivors in our sample was 26 years (range, 19 to 53 years) and they were 18 years from primary diagnosis (range, 11 to 42 years). Thirty-three percent of survivors were treated with focal irradiation, 38% with CSI, and 29% did not receive CRT. The distribution of age at diagnosis (P = .02), sex (P = .04), tumor location (P < .001), surgical resection (P < .001), shunt placement (P = .02), and seizures (P = .006) varied significantly by CRT exposure (Table 1).
Table 1.
Participant Demographic and Treatment Characteristics Overall and by Cranial Radiation Exposure Group
| Characteristic | All Participants (N = 224) | No CRT (n = 63) | Focal Irradiation (n = 71) | CSI (n = 83) | P |
|---|---|---|---|---|---|
| Current age, years | 26.0 (19.1-53.1) | 27.1 (19.1-37.2) | 24.5 (19.1-53.1) | 26.0 (19.3-48.9) | .25 |
| Age at diagnosis, years | 8.7 (0.1-21.0) | 11.1 (0.1-18.0) | 7.1 (0.4-21.0) | 8.7 (0.3-18.3) | .02 |
| Time since diagnosis, years | 17.7 (10.7-41.8) | 16.6 (10.7-25.0) | 18.0 (11.4-41.8) | 18.4 (11.1-40.8) | .15 |
| Radiation, Gy | |||||
| Whole brain | 23.4 (0.0-55) | — | 0 (0-0) | 35.2 (23.4-55) | |
| Tumor bed cumulative | 54 (22-72) | — | 54 (22-72) | 54.4 (36-66) | |
| Tumor bed boost dose | 18 (1.8-45) | — | 8.1 (3.6-45) | 18.6 (1.0-32.4) | |
| Tumor location | < .001 | ||||
| Supratentorial | 102 (46.8) | 39 (61.9) | 44 (62.0) | 19 (22.9) | |
| Infratentorial | 116 (53.2) | 24 (38.1) | 27 (38.0) | 64 (77.1) | |
| Sex | .04 | ||||
| Female | 91 (40.6) | 32 (50.8) | 30 (42.3) | 25 (30.1) | |
| Male | 133 (59.4) | 31 (49.2) | 41 (57.7) | 58 (69.9) | |
| Race/ethnicity | .27 | ||||
| White, non-Hispanic | 183 (81.7) | 48 (76.2) | 60 (84.5) | 70 (84.3) | |
| Black | 38 (17.0) | 15 (23.8) | 9 (12.7) | 12 (14.5) | |
| Other | 3 (1.3) | 0(0.0) | 2 (2.8) | 1 (1.2) | |
| Diagnosis | < .001 | ||||
| Medulloblastoma | 65 (29.0) | 0 (0.0) | 0 (0.0) | 63 (75.9) | |
| Ependymoma | 26 (11.6) | 0 (0.0) | 17 (23.9) | 4 (4.8) | |
| Astrocytoma | 74 (33.0) | 36 (57.1) | 36 (50.7) | 2 (2.4) | |
| Craniopharyngioma | 13 (5.8) | 7 (11.1) | 6 (8.5) | 0 (0.0) | |
| Germ cell tumor | 11 (4.9) | 1 (1.6) | 0 (0.0) | 10 (12.0) | |
| Optic pathway glioma | 8 (3.6) | 3 (4.8) | 5 (7.0) | 0 (0.0) | |
| Glioma | 17 (7.6) | 11 (17.5) | 6 (8.5) | 0 (0.0) | |
| Other | 10 (4.5) | 5 (7.9) | 1 (91.4) | 4 (4.8) | |
| Chemotherapy | |||||
| Platinum | < .001 | ||||
| Yes | 79 (35.3) | 4 (6.3) | 18 (25.4) | 55 (66.3) | |
| No | 145 (64.7) | 59 (93.7) | 53 (74.6) | 28 (33.7) | |
| Alkaloids | < .001 | ||||
| Yes | 59 (26.3) | 2 (3.2) | 12 (16.9) | 42 (50.6) | |
| No | 165 (73.7) | 61 (96.8) | 59 (83.1) | 41 (49.4) | |
| Alkylating agents | < .001 | ||||
| Yes | 67 (29.9) | 2 (3.2) | 16 (22.5) | 46 (55.4) | |
| No | 157 (70.1) | 61 (96.8) | 55 (77.5) | 37 (44.6) | |
| Topoisomerase inhibitors | < .001 | ||||
| Yes | 52 (23.2) | 2 (3.2) | 10 (14.1) | 38 (45.8) | |
| No | 172 (76.8) | 61 (96.8) | 61 (85.9) | 45 (54.2) | |
| Other | .15 | ||||
| Yes | 10 (4.5) | 0 (0) | 4 (5.6) | 5 (6.0) | |
| No | 214 (95.5) | 65 (100) | 67 (94.4) | 78 (94.0) | |
| Surgical resection | < .001 | ||||
| None | 8 (3.6) | 0 (0.0) | 7 (9.9) | 1 (1.2) | |
| Biopsy only | 35 (15.6) | 6 (9.5) | 19 (26.8) | 10 (12.0) | |
| Partial | 65 (29.0) | 14 (22.2) | 25 (35.2) | 23 (27.7) | |
| Gross total | 116 (51.8) | 43 (68.3) | 20 (28.2) | 49 (59.0) | |
| Shunt placement | .02 | ||||
| Yes* | 81 (36.2) | 15 (23.8) | 24 (33.8) | 38 (45.8) | |
| No | 143 (63.8) | 48 (76.2) | 47 (66.2) | 45 (54.2) | |
| Seizures | .006 | ||||
| Yes, resolved | 40 (17.9) | 19 (30.2) | 12 (16.9) | 7 (8.4) | |
| Yes, unresolved | 33 (14.7) | 11 (17.5) | 11 (15.5) | 10 (12) | |
| No | 151 (67.4) | 33 (52.4) | 48 (67.6) | 66 (79.5) | |
| NF-1 or tuberous sclerosis | .15 | ||||
| Yes | 8 (3.6) | 3 (5.0) | 4 (6.0) | 1 (1.0) | |
| No | 216 (96.4) | 60 (95.0) | 67 (94.0) | 1 (99.0) | |
| Education attainment† | .005 | ||||
| High school/GED or less | 102 (48.8) | 21 (35.6) | 29 (42.0) | 48 (63.2) | |
| Some college | 58 (27.8) | 18 (30.5) | 26 (37.7) | 14 (18.4) | |
| College graduate or more | 49 (23.4) | 20 (33.9) | 14 (20.3) | 14 (18.4) | |
| Employment status‡ | .26 | ||||
| Unemployed | 106 (48.0) | 25 (40.3) | 31 (43.7) | 45 (55.6) | |
| Part time | 30 (13.6) | 7 (11.3) | 11 (15.5) | 11 (13.6) | |
| Full time | 85 (38.5) | 30 (48.4) | 29 (40.8) | 25 (30.9) | |
| Independent living§ | .001 | ||||
| No | 125 (62.8) | 27 (47.7) | 42 (63.6) | 55 (73.3) | |
| Yes | 74 (37.2) | 30 (52.6) | 24 (36.4) | 20 (26.7) |
NOTE. Data are presented as median (range) or No. (%). Two hundred twenty-four survivors participated in the study. Seven survivors were treated with brachytherapy or received reirradiation and were not included in subgroup analyses using cranial radiation data. Therefore, 217 participants were included in the three treatment groups: no CRT, focal irradiation, and CSI. Cumulative dose to the tumor bed is composed of the craniospinal dose or focal irradiation dose plus the boost dose delivered specifically to the tumor bed. Tumor bed boost dose includes only the additional boost dose of radiotherapy targeting the primary tumor bed. P values were calculated using Kruskal-Wallis one way analysis of variance test comparing medians for three groups or χ2 test comparing proportions across three groups.
Abbreviations: CRT, cranial radiation therapy; CSI, craniospinal irradiation; GED, general education diploma; NF-1, neurofibromatosis type 1.
Thirty-four patients with a history of hydrocephalus with shunt placement had more than one shunt revision.
n = 209.
n = 221.
n = 199.
Neurocognitive Functioning
In the overall sample, the prevalence of severe neurocognitive impairment ranged from 8% (95% CI, 4.4 to 11.8) on direct assessment of working memory to 57% (95% CI, 50.0 to 63.6) on a task of fine motor dexterity (Fig 2; Appendix Table A2). On the basis of the general population, the expected prevalence of severe impairment was 2%, but 20% to 30% of survivors demonstrated such impairment on performance-based measures of intellect, short- and long-term memory, mathematics, and cognitive fluency. In contrast, on self-report measures, less than 10% of survivors reported a significant adverse impact of cognitive skills on daily functioning, with the exception of working memory (21%; 95% CI, 16.0 to 26.8). The prevalence of severe impairment across cognitive domains and specific abilities varied by radiation exposure (Table 2; Fig 2). For example, 11% (MR, 5.6; 95% CI, 2.5 to 11.2) of survivors without CRT exposure were impaired on long-term memory compared with 25% (MR, 12.7; 95% CI, 7.7 to 16.5) exposed to focal irradiation and 36% (MR, 18.1; 95% CI, 12.4 to 25.7) exposed to CSI (P < .001).
Fig 2.
Degree of neurocognitive impairment in adult survivors of childhood CNS tumors by cranial radiation exposure. (A) No cranial radiation therapy (CRT; n = 63). (B) Focal irradiation (n = 71). (C) Craniospinal irradiation (CSI; n = 83).
Table 2.
Mean Performance, Prevalence of Severe Neurocognitive Impairment, and Standardized MRs by Cranial Radiation Exposure Compared With Nationally Representative Normative Data
| No CRT (n = 63) | Focal Irradiation (n = 71) | CSI (n = 83) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Domain | Mean (SD) | % Imp | MR (95% CI) | Mean (SD) | % Imp | MR (95% CI) | Mean (SD) | % Imp | MR (95% CI) | P* |
| Intelligence | — | 9.8 | — | — | 14.3 | — | — | 32.5 | — | .001 |
| Full scale | −0.38 (1.1) | 9.8 | 4.9 (2.0 to 10.2) | −0.50 (1.1) | 14.3 | 7.1 (3.6 to 12.7) | −1.31 (1.2) | 32.5 | 16.2 (10.8 to 23.5) | < .001 |
| Verbal | −0.48 (1.2) | 14.5 | 7.3 (3.5 to 13.3) | −0.64 (1.3) | 22.5 | 11.3 (6.7 to 17.9) | −1.53 (1.2) | 45.1 | 22.6 (16.1 to 30.8) | < .001 |
| Perceptual | −0.31 (1.1) | 14.8 | 7.4 (3.6 to 13.5) | −0.28 (1.2) | 11.4 | 5.7 (2.7 to 10.9) | −1.17 (1.3) | 33.8 | 16.9 (11.4 to 24.2) | < .001 |
| Academics | — | 9.8 | — | — | 23.5 | — | — | 44.7 | — | < .001 |
| Word reading | −0.36 (0.9) | 4.9 | 2.5 (0.6 to 6.7) | −0.82 (1.3) | 16.2 | 8.1 (4.3 to 14.1) | −1.29 (1.2) | 25.0 | 12.5 (7.7 to 19.2) | < .001 |
| Mathematics | −0.66 (1.2) | 9.8 | 4.9 (2.0 to 10.2) | −0.96 (1.4) | 19.4 | 9.7 (5.4 to 16.2) | −1.84 (1.3) | 42.1 | 21.1 (14.7 to 29.4) | < .001 |
| Attention | — | 27.9 | — | — | 41.2 | — | — | 47.5 | — | .06 |
| Focus | −0.80 (1.8) | 23.3 | 11.7 (6.6 to 19.1) | −1.50 (1.8) | 32.3 | 16.2 (10.3 to 24.3) | −2.10 (2.1) | 42.5 | 21.3 (15.0 to 29.4) | < .001 |
| Sustained | −0.61 (2.9) | 8.5 | 4.2 (1.6 to 9.4) | −1.01 (3.5) | 16.7 | 8.3 (4.4 to 14.5) | −1.22 (3.5) | 20.5 | 10.3 (6.1 to 16.3) | .15 |
| Variability | −0.29 (2.5) | 6.8 | 3.4 (1.1 to 8.2) | −0.55 (2.4) | 15.2 | 7.6 (3.8 to 13.5) | −0.68 (2.2) | 14.1 | 7.1 (3.7 to 12.3) | .03 |
| Memory | — | 17.7 | — | — | 36.6 | — | — | 54.9 | — | < .001 |
| New learning | −0.27 (1.0) | 6.5 | 3.2 (1.0 to 7.8) | −0.81 (1.4) | 19.7 | 9.8 (5.6 to 16.2) | −1.56 (1.2) | 40.7 | 20.4 (14.3 to 28.3) | < .001 |
| Short-term recall | −0.41 (1.3) | 9.7 | 4.8 (2.0 to 10.1) | −1.00 (1.4) | 26.8 | 13.4 (8.3 to 20.5) | −1.22 (1.2) | 30.9 | 15.4 (10.2 to 22.5) | < .001 |
| Long-term recall | −0.61 (1.3) | 11.3 | 5.6 (2.5 to 11.2) | −1.02 (1.4) | 25.4 | 12.7 (7.7 to 16.5) | −1.48 (1.3) | 36.3 | 18.1 (12.4 to 25.7) | < .001 |
| Span | 0.10 (1.0) | 3.2 | 1.6 (0.3 to 5.3) | −0.46 (1.3) | 11.3 | 5.6 (2.6 to 10.7) | −0.95 (0.9) | 19.5 | 9.8 (5.8 to 15.5) | < .001 |
| Processing speed | — | 40.0 | — | — | 64.6 | — | — | 66.3 | — | .003 |
| Motor | −1.91 (1.9) | 39.0 | 19.5 (12.7 to 28.8) | −2.81 (1.9) | 61.5 | 30.8 (22.3 to 41.5) | −3.20 (1.9) | 64.6 | 32.3 (24.3 to 42.1) | < .001 |
| Visual | −0.22 (1.1) | 6.7 | 3.3 (1.1 to 8.0) | −0.77 (1.1) | 16.9 | 8.5 (4.5 to 14.7) | −1.23 (1.1) | 31.3 | 15.6 (10.3 to 22.7) | < .001 |
| Visual-motor | −0.42 (1.1) | 11.7 | 5.8 (2.6 to 11.5) | −1.14 (1.0) | 26.2 | 13.1 (7.9 to 20.5) | −1.55 (0.8) | 37.5 | 18.8 (12.9 to 26.4) | < .001 |
| Executive function | — | 37.1 | — | — | 52.1 | — | — | 68.3 | — | < .001 |
| Flexibility | −1.23 (2.0) | 33.3 | 16.7 (10.5 to 25.3) | −1.92 (2.2) | 44.6 | 22.3 (15.2 to 31.6) | −3.19 (2.2) | 64.9 | 32.5 (24.4 to 42.5) | < .001 |
| Fluency | −0.28 (1.4) | 11.3 | 5.6 (2.5 to 11.2) | −0.49 (1.3) | 16.9 | 8.4 (4.6 to 14.4) | −1.11 (1.2) | 26.8 | 13.4 (8.6 to 20.0) | < .001 |
| Working memory | −0.24 (1.0) | 3.2 | 1.6 (0.3 to 5.3) | −0.47 (1.1) | 8.5 | 4.2 (1.7 to 8.8) | −1.01 (0.9) | 11.0 | 5.5 (2.7 to 10.1) | < .001 |
| Cognitive rating | — | 26.2 | — | — | 26.8 | — | — | 26.8 | — | 1.0 |
| Initiation | −0.22 (1.1) | 8.2 | 4.1 (1.5 to 9.1) | −0.21 (1.2) | 12.7 | 6.3 (3.1 to 11.6) | −0.22 (1.1) | 7.3 | 3.7 (1.5 to 7.6) | .93 |
| Working memory | −0.70 (1.5) | 21.3 | 10.7 (5.9 to 17.8) | −0.68 (1.4) | 19.7 | 9.9 (5.6 to 16.2) | −1.09 (1.2) | 20.7 | 10.4 (6.2 to 16.3) | .04 |
| Planning | −0.22 (1.3) | 8.2 | 4.1 (1.5 to 9.1) | −0.28 (1.3) | 12.9 | 6.4 (3.1 to 11.8) | −0.32 (1.1) | 8.5 | 4.3 (1.9 to 8.4) | .60 |
| Task completion | −0.24 (1.3) | 8.2 | 4.1 (1.5 to 9.1) | −0.14 (1.2) | 7.1 | 3.6 (1.3 to 7.9) | −0.44 (1.1) | 7.3 | 3.7 (1.5 to 7.6) | .19 |
| Organization | −0.14 (1.1) | 6.6 | 3.3 (1.0 to 7.9) | −0.16 (1.2) | 9.9 | 4.9 (2.2 to 9.8) | −0.22 (1.1) | 3.7 | 1.8 (0.5 to 5.0) | .85 |
| Behavior rating | — | 24.6 | — | — | 14.1 | — | — | 22.0 | — | .28 |
| Inhibition | −0.19 (1.1) | 8.2 | 4.1 (1.5 to 9.1) | 0.21 (0.9) | 1.4 | 0.7 (0.03 to 3.5) | 0.17 (1.0) | 3.7 | 1.8 (0.5 to 5.0) | .05 |
| Shift | −0.29 (1.3) | 11.5 | 5.7 (2.5 to 11.4) | −0.12 (1.1) | 4.3 | 2.1 (0.5 to 5.8) | −0.45 (1.2) | 8.5 | 4.3 (1.9 to 8.4) | .20 |
| Emotional Control | −0.21 (1.4) | 11.5 | 5.7 (2.5 to 11.4) | 0.09 (1.2) | 5.6 | 2.8 (0.9 to 6.8) | −0.19 (1.1) | 8.5 | 4.3 (1.9 to 8.4) | .17 |
| Self-monitor | −0.07 (1.4) | 14.8 | 7.4 (3.6 to 13.5) | 0.02 (1.1) | 5.6 | 2.8 (0.9 to 6.8) | −0.24 (1.2) | 8.5 | 4.3 (1.9 to 8.4) | .30 |
NOTE. Expected mean = 0 (SD, 1) using nationally representative norms. Impairment defined as age-adjusted z score ≤ −2.0.
Abbreviations: CRT, cranial radiation therapy; CSI, craniospinal irradiation; Imp, impairment; MR, standardized morbidity ratio; SD, standard deviation.
P values from χ2 test comparing proportion of impairment for domains; P values from Kruskal-Wallis one-way analysis of variance test comparing mean scores for specific abilities.
In multivariable models, older age at diagnosis was inversely associated with severe neurocognitive impairment across all domains (Table 3). Treatment with CSI conferred the greatest risk of severe neurocognitive impairment (eg, intelligence: RR = 2.70; 95% CI, 1.37 to 5.34; academics: RR = 3.96; 95% CI, 1.94 to 8.08; memory: RR = 2.93; 95% CI, 1.69 to 5.08; executive function: RR = 1.74; 95% CI, 1.24 to 2.45) compared with survivors who were not treated with CRT. Seizures were associated with impaired academics (RR = 1.48; 95% CI, 1.02 to 2.14), attention (RR = 1.54; 95% CI, 1.12 to 2.13), and memory (RR = 1.44; 95% CI, 1.04 to 1.99). History of hydrocephalus with shunt placement was associated with increased risk of impaired intelligence (RR = 1.78; 95% CI, 1.12 to 2.82) and memory (RR = 1.42; 95% CI, 1.03 to 1.95). Multivariable models including tumor location are shown in Appendix Table A3. The inclusion of product terms in our multivariable regression models did not suggest significant interaction with the identified risk factors.
Table 3.
Multivariable Models With Radiation Exposure Predicting Risk of Severe Impairment by Neurocognitive Domain
| Parameter | RR | 95% CI | P |
|---|---|---|---|
| Intelligence | |||
| Age at diagnosis (per 1 year) | 0.83 | 0.77 to 0.88 | < .001 |
| Time since diagnosis (per 1 year) | 1.03 | 0.97 to 1.09 | .33 |
| Sex | |||
| Female (referent) | 1.0 | ||
| Male | 0.91 | 0.56 to 1.47 | .69 |
| CRT | |||
| None (referent) | 1.0 | ||
| Focal irradiation | 1.09 | 0.48 to 2.51 | .83 |
| CSI | 2.70 | 1.37 to 5.34 | .004 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.78 | 1.12 to 2.82 | .015 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.31 | 0.84 to 2.07 | .24 |
| Academics | |||
| Age at diagnosis (per 1 year) | 0.86 | 0.81 to 0.92 | < .001 |
| Time since diagnosis (per 1 year) | 1.01 | 0.97 to 1.05 | .65 |
| Sex | |||
| Female (referent) | 1.0 | ||
| Male | 1.04 | 0.72 to 1.52 | .82 |
| CRT | |||
| None (referent) | 1.0 | ||
| Focal irradiation | 1.93 | 0.89 to 4.18 | .096 |
| CSI | 3.96 | 1.94 to 8.08 | .002 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.43 | 0.99 to 2.04 | .05 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.48 | 1.02 to 2.14 | .039 |
| Attention | |||
| Age at diagnosis (per 1 year) | 0.95 | 0.92 to 0.99 | .009 |
| Time since diagnosis (per 1 year) | — | ||
| Sex | |||
| Female (referent) | — | ||
| Male | — | ||
| CRT | |||
| None (referent) | 1.0 | ||
| Focal irradiation | 1.41 | 0.87 to 2.30 | .17 |
| CSI | 1.72 | 1.09 to 2.70 | .02 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.22 | 0.89 to 1.67 | .21 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.54 | 1.12 to 2.13 | .008 |
| Memory | |||
| Age at diagnosis (per 1 year) | 0.97 | 0.94 to 1.01 | .14 |
| Time since diagnosis (per 1 year) | 1.02 | 0.99 to 1.05 | .21 |
| Sex | |||
| Female (referent) | 1.0 | ||
| Male | 1.08 | 0.77 to 1.50 | .67 |
| CRT | |||
| None (referent) | 1.0 | ||
| Focal irradiation | 1.90 | 1.06 to 3.41 | .03 |
| CSI | 2.93 | 1.69 to 5.08 | < .001 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.42 | 1.03 to 1.95 | .03 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.44 | 1.04 to 1.99 | .03 |
| Processing speed | |||
| Age at diagnosis (per 1 year) | 0.96 | 0.93 to 0.98 | .001 |
| Time since diagnosis (per 1 year) | 1.02 | 1.00 to 1.04 | .03 |
| Sex | |||
| Female (referent) | 1.0 | ||
| Male | 1.22 | 0.97 to 1.53 | .09 |
| CRT | |||
| None (referent) | 1.0 | ||
| Focal irradiation | 1.42 | 1.00 to 2.01 | .05 |
| CSI | 1.45 | 1.03 to 2.04 | .03 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.24 | 1.00 to 1.54 | .05 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.24 | 1.00 to 1.56 | .05 |
| Executive function | |||
| Age at diagnosis (per 1 year) | 0.95 | 0.92 to 0.97 | < .001 |
| Time since diagnosis (per 1 year) | — | ||
| Sex | |||
| Female (referent) | 1.0 | ||
| Male | 1.12 | 0.88 to 1.42 | .38 |
| CRT | |||
| None (referent) | 1.0 | ||
| Focal irradiation | 1.27 | 0.86 to 1.86 | .23 |
| CSI | 1.74 | 1.24 to 2.45 | .001 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.08 | 0.86 to 1.35 | .52 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.22 | 0.96 to 1.56 | .11 |
NOTE. Dash indicates variables that were not retained in best-fitting models on the basis of Akaike information criterion. No interactions terms were selected in any best-fitting models (age at diagnosis × CRT; sex × CRT; time since diagnosis × CRT).
Abbreviations: CRT, cranial radiation therapy; CSI, craniospinal irradiation; RR, risk ratio.
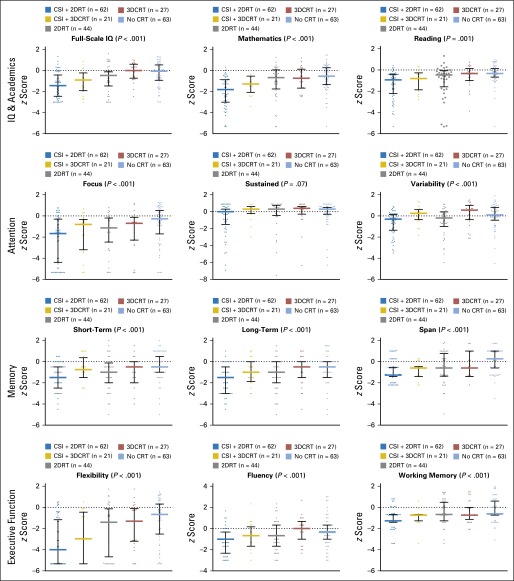
Given the small number of observations, the type of radiation therapy (ie, 2DRT v 3DCRT) could not be considered in multivariable models. Nevertheless, we examined performance on direct assessment measures by 2DRT and 3DCRT within CSI and focal irradiation groups (Appendix Table A4). A lower prevalence of impairment was observed across nearly all domains for survivors treated with CSI plus 3DCRT compared with those treated with CSI plus 2DRT, as well as for those treated with focal 3DCRT compared with those treated with focal 2DRT (Fig 3; Appendix Table A5). However, comparisons are limited given the difference in follow-up time between survivors treated with 2D and 3D techniques. Survivors treated with 3DCRT had, on average, 14 years of follow-up, whereas survivors treated with 2DRT were followed for 21 years. Rate ratios accounting for differences in follow-up time with adjustment for age at assessment, sex, and race suggest elevated rates of severe impairment in survivors treated with 3D compared with 2D radiation therapy, but were not statistically significant (Appendix Tables A6 and A7).
Fig 3.
Neurocognitive performance by cranial radiation exposure. Medians, interquartile ranges, and individual z scores plotted for each exposure by outcome. The y-axis intervals are extended for sustained attention and verbal fluency to account for extreme values. 2DRT, two-dimensional treatment planning; 3DCRT, three-dimensional conformal radiation therapy; CRT, cranial radiation therapy; CSI, craniospinal irradiation; IQ, intelligence quotient.
Sensitivity analyses were conducted to explore the potential impact of shorter follow-up times for the 3D groups compared with the 2D groups. For example, assuming no additional individuals with executive function impairment were identified in the CSI + 2DRT group, an additional 5 years of follow-up time for the CSI + 3DCRT group would result in a similar rate of executive function impairment for the 3D and 2D groups (rate ratio = 0.99). With the addition of 10 years of follow-up time, survivors treated with CSI + 3DCRT had a 29% reduction in the rate of severe impairment compared with those treated with CSI + 2DRT (rate ratio = 0.71). Similar patterns were observed with the addition of 5 and 10 years of follow-up time for 3DCRT compared with 2DRT.
Social Attainment
In multivariable models adjusted for current age and sex, neurocognitive impairment was associated with increased risk of reduced educational attainment, unemployment, and nonindependent living. For example, survivors with impaired intellect (RR = 1.37; 95% CI, 1.23 to 1.52), academics (RR = 1.41; 95% CI, 1.26 to 1.57), or executive function (RR=1.41; 95% CI, 1.20 to 1.66), had a 40% increased risk of not graduating from college. The risk of unemployment was two times greater in survivors with impaired attention (RR = 2.37; 95% CI, 1.75 to 3.21), memory (RR = 1.87; 95% CI, 1.42 to 2.47), or executive function (RR = 2.17; 95% CI, 1.54 to 3.07). Survivors with severe impairment in intelligence (RR = 1.47; 95% CI, 1.22 to 1.76), academics (RR = 1.55; 95% CI, 1.29 to 1.86), memory (RR = 1.49; 95% CI, 1.23 to 1.80) or executive function (RR = 1.47; 95% CI, 1.18 to 1.83) had an approximately 50% increased risk of nonindependent living compared with survivors without such impairment.
DISCUSSION
To our knowledge, this report presents the most comprehensive analysis of outcome data on a large cohort of adult survivors of pediatric CNS tumors, with direct assessment of neurocognitive function and the resulting impact on social attainment. Nearly 20 years after diagnosis, survivors demonstrated severe impairment of up to 15 times the frequency of that observed in the general population. Our results contribute several novel and important findings to the literature: (1) the prevalence of severe impairment is significantly greater than that observed in the general population, even among survivors who were not treated with CRT; (2) directly assessed neurocognitive skills are more severely impaired compared with self-reported cognition and behavior; (3) the risk of severe neurocognitive impairment in adulthood is increased significantly by a history of seizures and hydrocephalus with shunt placement; (4) the potential long-term benefit of conformal radiation therapy is yet to be established in adult survivors, although our results suggest that such benefit may be observed with additional follow-up time; and, (5) neurocognitive impairment is associated with reduced attainment of adult developmental milestones.
The frequency of severe neurocognitive impairment was greater than expected across all domains of neurocognitive function. Even among survivors who were not treated with CRT, we observed a fivefold increased prevalence of impairment on measures of short- and long-term memory and a 17-fold increased prevalence of impaired cognitive flexibility. Moreover, 37% of survivors without CRT exposure were severely impaired on at least one measure of processing speed, a cognitive domain often presumed to be affected by radiation-induced changes to cerebral white matter.
Although a larger than expected proportion of survivors demonstrated severe neurocognitive impairment, few perceived a similar degree of impairment in daily life. With the exception of working memory, less than 10% of survivors reported severe cognitive or behavioral impairment, whereas up to 50% of survivors were severely impaired on performance-based measures. This contrast may be a result of survivor adaptation to deficits over time. Survivors may learn to compensate for their deficits or to avoid situations that require specific skills, thereby reducing the potential impact on daily activities. Alternatively, survivors may lack the insight or awareness to appreciate the impact of their deficits. Our results highlight the need to consider clinical measurement and patient-reported outcomes when assessing neurocognitive function in this population.
Consistent with prior studies among child and adolescent survivors, CSI was significantly associated with severe impairment across all neurocognitive domains. However, observed associations were not restricted to radiation therapy exposure. We also observed that survivors with a history of seizures were at risk of severely impaired academics, attention, and memory compared with survivors without a history of seizures. Seizures conferred a nearly 50% increased risk of impairment even after adjustment for exposure to CRT. In adults with epilepsy, deficits in memory and attention are well documented.28,29 The impact of seizures on neurocognitive function may be related to structural brain abnormalities and/or the cumulative neurobiologic consequences of recurrent seizures. However, because of the small number of survivors with seizures, we were unable to examine the contribution of seizure status (resolved v unresolved) or the use anticonvulsant medications, which have previously been associated with neurocognitive morbidities in adult survivors of childhood cancer.30 A history of hydrocephalus with shunt placement conferred a 40% increased risk of memory impairment, consistent with the literature documenting hippocampal vulnerability to hydrocephalus.31
Our results also provide important information regarding the functional implications of neurocognitive impairment. Specifically, severe neurocognitive impairment was associated with a reduced attainment of expected adult developmental milestones, including education, employment, and independent living. These findings are consistent with reports from other childhood cancer survivors32 and reports linking self-report of cognitive impairment with lower socioeconomic attainment among CNS tumor survivors.11
A unique contribution of our study is the assessment of potential differences in neurocognitive outcomes by parameters of CRT delivery, particularly 2D compared with 3D techniques. Data from survivors within 5 years of diagnosis and treatment suggest that radiation dose and volume reduction may confer benefit with respect to the established cognitive decline observed in the first decade after treatment completion. Our results are not compatible with the hypothesized benefit of 3DCRT therapy on long-term cognitive outcomes; however, the studied cohort was treated in the early stages of conformal treatment planning when the chosen clinical target volume margins were conservative and large, resulting in a modest level of normal tissue sparing. In addition, the duration of follow-up is markedly different between survivors treated with older 2DRT and those treated with more contemporary 3DCRT techniques. Neurocognitive assessments were completed later among survivors treated with 2DRT; the consequence is that the time to impairment may have been overestimated for survivors treated with 2DRT. We used sensitivity analysis to explore the potential impact of follow-up duration on our effect estimates comparing 2DRT and 3DCRT groups. The results suggested that the risk of severe cognitive impairment may be lower after 3DCRT than after 2DRT techniques with 5 to 10 years of additional follow-up for survivors treated with 3D techniques. These results emphasize the need for systematic follow-up of survivors beyond 5 years after therapy, with appropriate assessment and comparison of long-term outcomes as treatment protocols change.
Other limitations should be considered when interpreting our findings. Survivors who returned to campus and participated in a neurocognitive evaluation may have differed with respect to health or neurocognitive status compared with survivors who did not participate. Although comparisons of participants and nonparticipants in our study and the SJLIFE cohort as a whole do not suggest selective participation, the missing outcomes for nonparticipants precludes certainty about a lack of bias from selective participation. Because concern regarding selection bias cannot be eliminated, generalization of our results to other samples may be limited. In addition, although our exposure and outcome measurements are compatible with a temporal order between CRT exposure and neurocognitive morbidities, the lack of serial neurocognitive assessments precludes inferences about potential cognitive decline associated with aging in survivors.
Our results suggest that adult survivors of pediatric brain tumors are at risk of pervasive neurocognitive impairment several decades after diagnosis and treatment. Survivors may adapt to or lack insight related to their deficits, but observed cognitive difficulties confer a risk of reduced socioeconomic attainment in adulthood. Systematic monitoring of cognitive functioning of survivors is recommended. Prophylactic interventions during therapy,33,34 as well as remedial approaches,35,36 may reduce the severity and functional impact of neurocognitive impairment in survivors of pediatric brain tumors.
Appendix
Table A1.
Characteristics of Participants and Nonparticipants
| Characteristic | Participants (n = 224) | Nonparticipants (n = 119) | P |
|---|---|---|---|
| Age at diagnosis, years, mean (SD) | 8.78 (4.6) | 8.56 (4.9) | .70 |
| Sex | .61 | ||
| Female | 91 (40.6) | 45 (37.8) | |
| Male | 133 (59.4) | 74 (62.2) | |
| Race | .43 | ||
| White, non-Hispanic | 183 (81.7) | 93 (78.3) | |
| Other | 41 (18.3) | 26 (21.8) | |
| Cranial radiation therapy | .25 | ||
| Yes | 155 (69.2) | 75 (63) | |
| No | 69 (30.8) | 44 (37.0) | |
| Chemotherapy | .38 | ||
| Yes | 90 (40.2) | 42 (35.3) | |
| No | 134 (59.8) | 77 (64.7) | |
| Surgical resection | .21 | ||
| Yes | 181 (80.8) | 88 (73.9) | |
| No/biopsy only | 43 (19.2) | 31 (26.1) | |
| Shunt placement | .23 | ||
| Yes | 81 (36.2) | 51 (42.9) | |
| No | 143 (63.8) | 68 (57.1) | |
| Seizures | .21 | ||
| Yes | 73 (32.6) | 31 (26.1) | |
| No | 151 (67.4) | 88 (73.9) | |
| Tumor location | .08 | ||
| Infratentorial | 120 (53.6) | 52 (43.7) | |
| Supratentorial | 104 (46.4) | 67 (56.3) | |
| Diagnosis | .02 | ||
| Medulloblastoma | 65 (29.0) | 22 (18.5) | |
| Ependymoma | 26 (11.6) | 7 (5.9) | |
| Glioma | 17 (7.6) | 15 (12.6) | |
| Astrocytoma | 74 (33.0) | 46 (38.7) | |
| Craniopharyngioma | 13 (5.8) | 2 (1.7) | |
| Germ cell tumor | 11 (4.9) | 10 (8.4) | |
| Optic pathway glioma | 8 (3.6) | 5 (4.2) | |
| Other | 10 (4.5) | 12 (10.1) |
NOTE. Data are presented as No. (%) unless indicated otherwise.
Table A2.
Degree of Neurocognitive Impairment in Adult Survivors of Pediatric CNS Tumors
| Domain | No. | Mean | SD | None | Mild | Moderate | Severe | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. | % | 95% CI | No. | % | 95% CI | No. | % | 95% CI | No. | % | 95% CI | ||||
| Intelligence | |||||||||||||||
| Full scale | 216 | −0.79 | 1.21 | 126 | 58.3 | 51.7 to 64.8 | 29 | 13.4 | 9.4 to 18.5 | 17 | 7.9 | 4.8 to 12.1 | 44 | 20.4 | 15.4 to 26.1 |
| Verbal | 220 | −0.95 | 1.33 | 114 | 51.8 | 45.2 to 58.4 | 28 | 12.7 | 8.8 to 17.6 | 13 | 5.9 | 3.3 to 9.6 | 65 | 29.5 | 23.8 to 35.8 |
| Perceptual | 216 | −0.65 | 1.29 | 146 | 67.6 | 61.1 to 73.6 | 15 | 6.9 | 4.1 to 11.0 | 9 | 4.2 | 2.1 to 7.5 | 46 | 21.3 | 16.2 to 27.1 |
| Academics | |||||||||||||||
| Word reading | 210 | −0.86 | 1.24 | 145 | 69.0 | 62.6 to 75.0 | 19 | 9.0 | 5.7 to 13.5 | 12 | 5.7 | 3.3 to 9.5 | 34 | 16.2 | 11.7 to 21.6 |
| Mathematics | 209 | −1.2 | 1.4 | 100 | 47.8 | 41.1 to 54.6 | 38 | 18.2 | 13.4 to 23.9 | 19 | 9.1 | 5.7 to 13.6 | 52 | 24.9 | 19.4 to 31.1 |
| Attention | |||||||||||||||
| Focus | 209 | −1.52 | 1.96 | 108 | 51.7 | 44.9 to 58.4 | 15 | 7.2 | 4.2 to 11.3 | 16 | 7.7 | 4.6 to 11.9 | 70 | 33.5 | 27.3 to 40.1 |
| Sustained | 208 | −0.97 | 3.3 | 163 | 78.4 | 72.4 to 83.6 | 10 | 4.8 | 2.5 to 8.4 | 3 | 1.4 | 0.4 to 3.9 | 32 | 15.4 | 11.0 to 20.8 |
| Variability | 208 | −0.52 | 2.31 | 161 | 77.4 | 71.4 to 82.7 | 15 | 7.2 | 4.2 to 11.4 | 7 | 3.4 | 1.5 to 6.5 | 25 | 12.0 | 8.1 to 17.0 |
| Memory | |||||||||||||||
| New learning | 219 | −0.94 | 1.29 | 120 | 54.8 | 48.2 to 61.3 | 28 | 12.8 | 8.8 to 17.7 | 20 | 9.1 | 5.8 to 13.5 | 51 | 23.3 | 18.1 to 29.2 |
| Short-term recall | 219 | −0.9 | 1.33 | 110 | 50.2 | 43.6 to 56.8 | 59 | 26.9 | 21.4 to 33.1 | 0 | 0.0 | 0.0 to 1.4 | 50 | 22.8 | 17.6 to 28.7 |
| Long-term recall | 218 | −1.08 | 1.35 | 101 | 46.3 | 39.8 to 53.0 | 62 | 28.4 | 22.8 to 34.7 | 0 | 0.0 | 0.0 to 1.4 | 55 | 25.2 | 19.8 to 31.3 |
| Span | 220 | −0.5 | 1.17 | 127 | 57.7 | 51.1 to 64.1 | 64 | 29.1 | 23.4 to 35.4 | 1 | 0.5 | 0.0 to 2.2 | 28 | 12.7 | 8.8 to 17.6 |
| Processing speed | |||||||||||||||
| Motor | 207 | −2.7 | 1.95 | 43 | 20.8 | 15.7 to 26.7 | 28 | 13.5 | 9.4 to 18.7 | 18 | 8.7 | 5.4 to 13.1 | 118 | 57.0 | 50.2 to 63.6 |
| Visual | 209 | −0.69 | 1.16 | 118 | 56.5 | 49.7 to 63.1 | 36 | 17.2 | 12.6 to 22.8 | 14 | 6.7 | 3.9 to 10.7 | 41 | 19.6 | 14.7 to 25.4 |
| Visual–motor | 209 | −0.6 | 1.06 | 72 | 34.4 | 28.2 to 41.1 | 57 | 27.3 | 21.6 to 33.6 | 26 | 12.4 | 8.5 to 17.5 | 54 | 25.8 | 20.3 to 32.1 |
| Executive function | |||||||||||||||
| Flexibility | 206 | −2.2 | 2.27 | 84 | 40.8 | 34.2 to 47.6 | 14 | 6.8 | 3.9 to 10.9 | 6 | 2.9 | 1.2 to 6.0 | 102 | 49.5 | 42.7 to 56.3 |
| Fluency | 220 | −0.69 | 1.32 | 113 | 51.4 | 44.8 to 57.9 | 46 | 20.9 | 15.9 to 26.7 | 17 | 7.7 | 4.7 to 11.8 | 44 | 20.2 | 15.1 to 25.7 |
| Working memory | 220 | −0.6 | 1.02 | 148 | 67.3 | 60.9 to 73.2 | 51 | 23.2 | 18.0 to 29.1 | 4 | 1.8 | 0.6 to 4.3 | 17 | 7.7 | 4.7 to 11.8 |
| Cognitive rating | |||||||||||||||
| Initiation | 219 | −0.21 | 1.14 | 156 | 71.2 | 65.0 to 77.0 | 31 | 14.2 | 10.0 to 19.3 | 12 | 5.5 | 3.0 to 9.1 | 20 | 9.1 | 5.8 to 13.5 |
| Working memory | 219 | −0.84 | 1.37 | 123 | 56.2 | 49.5 to 62.6 | 21 | 9.6 | 6.2 to 14.1 | 29 | 13.2 | 9.2 to 18.2 | 46 | 21.0 | 16.0 to 26.8 |
| Planning | 218 | −0.26 | 1.19 | 151 | 69.3 | 62.9 to 75.1 | 39 | 17.9 | 13.2 to 23.4 | 7 | 3.2 | 1.4 to 6.2 | 21 | 9.6 | 6.2 to 14.1 |
| Task completion | 219 | −0.28 | 1.17 | 159 | 72.9 | 66.4 to 78.2 | 34 | 15.6 | 11.2 to 20.8 | 9 | 4.1 | 2.0 to 7.4 | 16 | 7.3 | 4.4 to 11.4 |
| Organization | 219 | −0.17 | 1.14 | 166 | 75.8 | 69.8 to 81.1 | 21 | 9.6 | 6.2 to 14.1 | 18 | 8.2 | 5.1 to 12.4 | 14 | 6.4 | 3.7 to 10.3 |
| Behavior rating | |||||||||||||||
| Inhibition | 219 | 0.09 | 1 | 180 | 82.2 | 76.7 to 86.8 | 22 | 10.0 | 6.6 to 14.6 | 8 | 3.7 | 1.7 to 6.8 | 9 | 4.1 | 2.0 to 7.4 |
| Shift | 218 | −0.28 | 1.17 | 151 | 69.3 | 62.9 to 75.1 | 38 | 17.4 | 12.8 to 22.9 | 12 | 5.5 | 3.0 to 9.2 | 17 | 7.8 | 4.8 to 12.0 |
| Emotional control | 219 | −0.09 | 1.2 | 161 | 73.5 | 67.4 to 79.0 | 30 | 13.7 | 9.6 to 18.7 | 10 | 4.6 | 2.3 to 8.0 | 18 | 8.2 | 5.1 to 12.4 |
| Self-monitor | 219 | −0.11 | 1.22 | 172 | 78.5 | 72.7 to 83.6 | 12 | 5.5 | 3.0 to 9.1 | 15 | 6.8 | 4.0 to 10.8 | 20 | 9.1 | 5.8 to 13.5 |
NOTE. Expected mean = 0 (SD, 1) using nationally representative norms. Age-adjusted z scores for impairment: none, > −1.0; mild, > −1.5 to −1.0; moderate, > −2.0 to −1.5; severe, ≤ −2.0.
Abbreviation: SD, standard deviation.
Table A3.
Multivariable Models With Tumor Location Predicting Risk of Severe Impairment by Neurocognitive Domain
| Parameter | RR | 95% CI | P |
|---|---|---|---|
| Intelligence | |||
| Age at diagnosis (per 1 year) | 0.83 | 0.77 to 0.89 | < .001 |
| Time since diagnosis (per 1 year) | 1.02 | 0.96 to 1.08 | .58 |
| Sex | |||
| Female (referent) | 1.0 | ||
| Male | 0.98 | 0.59 to 1.64 | .94 |
| Tumor location | |||
| Supratentorial (referent) | 1.0 | ||
| Infratentorial | 1.81 | 1.06 to 3.09 | .03 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.71 | 1.08 to 2.72 | .02 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.37 | 0.85 to 2.21 | .20 |
| Academics | |||
| Age at diagnosis (per 1 year) | 0.86 | 0.81 to 0.91 | < .001 |
| Time since diagnosis (per 1 year) | — | ||
| Sex | |||
| Female (referent) | 1.0 | ||
| Male | 1.12 | 0.74 to 1.68 | .60 |
| Tumor location | |||
| Supratentorial (referent) | 1.0 | ||
| Infratentorial | 1.27 | 0.84 to 1.92 | .25 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.58 | 1.08 to 2.31 | .02 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.32 | 0.87 to 2.00 | .19 |
| Attention | |||
| Age at diagnosis (per 1 year) | 0.97 | 0.93 to 1.00 | .06 |
| Time since diagnosis (per 1 year) | 1.02 | 0.99 to 1.05 | .20 |
| Sex | |||
| Female (referent) | — | ||
| Male | — | ||
| Tumor location | |||
| Supratentorial (referent) | 1.0 | ||
| Infratentorial | 1.16 | 0.83 to 1.62 | .38 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.37 | 1.00 to 1.88 | .05 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.38 | 1.00 to 1.91 | .05 |
| Memory | |||
| Age at diagnosis (per 1 year) | 0.97 | 0.93 to 1.01 | .09 |
| Time since diagnosis (per 1 year) | 1.03 | 1.00 to 1.06 | .05 |
| Sex | |||
| Female (referent) | — | ||
| Male | — | ||
| Tumor location | |||
| Supratentorial (referent) | 1.0 | ||
| Infratentorial | 1.01 | 0.72 to 1.41 | .97 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.65 | 1.20 to 2.27 | .002 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.17 | 0.84 to 1.63 | .36 |
| Processing speed | |||
| Age at diagnosis (per 1 year) | 0.96 | 0.93 to 0.98 | .001 |
| Time since diagnosis (per 1 year) | 1.02 | 1.01 to 1.04 | .01 |
| Sex | |||
| Female (referent) | 1.0 | ||
| Male | 1.27 | 1.02 to 1.58 | .04 |
| Tumor location | |||
| Supratentorial (referent) | 1.0 | ||
| Infratentorial | 1.21 | 0.96 to 1.52 | .11 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.3 | 1.06 to 1.60 | .01 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.22 | 0.98 to 1.51 | .08 |
| Executive function | |||
| Age at diagnosis (per 1 year) | 0.95 | 0.92 to 0.97 | < .001 |
| Time since diagnosis (per 1 year) | |||
| Sex | |||
| Female (referent) | — | ||
| Male | — | ||
| Tumor location | |||
| Supratentorial (referent) | 1.0 | ||
| Infratentorial | 1.04 | 0.82 to 1.32 | .74 |
| Shunt | |||
| No (referent) | 1.0 | ||
| Yes | 1.19 | 0.95 to 1.49 | .13 |
| Seizure | |||
| No (referent) | 1.0 | ||
| Yes | 1.13 | 0.89 to 1.44 | .32 |
NOTE. Dash indicates variables that were not retained in best-fitting models on the basis of Akaike information criterion. No interactions terms were selected in any best-fitting models (age at diagnosis × CRT; sex × CRT; time since diagnosis × CRT).
Abbreviations: CRT, cranial radiation therapy; RR, risk ratio.
Table A4.
Participant Demographic and Treatment Characteristics by Cranial Radiation Therapy
| Characteristic | CSI + 2DRT (n = 62) | CSI + 3DCRT (n = 21) | 2DRT (n = 44) | 3DCRT (n = 27) |
|---|---|---|---|---|
| Current age, years | 27.7 (21.7-48.9) | 24.4 (19.3-30.9) | 26.7 (20.9-53.1) | 22.6 (19.1-32.3) |
| Age at diagnosis, years | 7.7 (0.3-16.3) | 11.8 (5.3-18.3) | 7.0 (0.4-21.0) | 7.7 (1.9-17.7) |
| Time since diagnosis, years | 19.9 (14.2-40.8) | 13.2 (11.1-16.1) | 20.6 (13.1-41.8) | 14.1 (11.4-21.5) |
| Radiation, Gy | ||||
| Whole brain | 35.2 (23.4-55) | 25.5 (23.4-39.6) | 0 (0-0) | 0 (0-0) |
| Tumor bed cumulative | 54 (0-66) | 55.8 (41.4-59.4) | 54 (22-72) | 54 (50.4-59.4) |
| Tumor bed boost dose | 18 (9-32.4) | 19.8 (1.8-32.4) | 72 (7.2-7.2) | 3.6 (0-45) |
| Tumor location | ||||
| Supratentorial | 13 (21.0) | 6 (28.6) | 28 (63.6) | 16 (59.3) |
| Infratentorial | 49 (79.0) | 15 (71.4) | 16 (36.4) | 11 (40.7) |
| Sex | ||||
| Female | 20 (32.3) | 5 (23.8) | 18 (40.9) | 12 (44.4) |
| Male | 42 (67.7) | 16 (76.2) | 26 (59.1) | 15 (55.6) |
| Race/ethnicity | ||||
| White, non-Hispanic | 55 (88.7) | 15 (71.4) | 34 (77.3) | 26 (96.3) |
| Black | 6 (9.7) | 6 (28.6) | 8 (18.2) | 1 (3.7) |
| Other | 1 (1.6) | 0 (0.0) | 2 (4.5) | 0 (0.0) |
| Diagnosis | ||||
| Medulloblastoma | 48 (77.4) | 15 (71.4) | 0 (0.0) | 0 (0.0) |
| Ependymoma | 4 (6.5) | 0 (0.0) | 9 (20.5) | 8 (29.6) |
| Astrocytoma | 2 (3.2) | 0 (0.0) | 23 (52.3) | 13 (48.1) |
| Craniopharyngioma | 0 (0.0) | 0 (0.0) | 3 (6.8) | 3 (11.1) |
| Germ cell tumor | 4 (6.5) | 6 (28.6) | 0 (0.0) | 0 (0.0) |
| Glioma | 0 (0.0) | 0 (0.0) | 5 (11.4) | 1 (3.7) |
| Optic pathway glioma | 0 (0.0) | 0 (0.0) | 4 (9.1) | 1 (3.7) |
| Other | 4 (6.5) | 0 (0.0) | 0 (0.0) | 1 (3.7) |
| Surgical resection | ||||
| None | 1 (1.6) | 0 (0.0) | 6 (13.6) | 1 (3.7) |
| Biopsy only | 5 (8.1) | 5 (23.8) | 13 (29.5) | 6 (22.2) |
| Partial | 19 (30.6) | 4 (19.0) | 12 (27.3) | 13 (48.1) |
| Gross total | 37 (59.7) | 12 (57.1) | 13 (29.5) | 7 (25.9) |
NOTE. Data are presented as median (range) or No. (%).
Abbreviations: 2DRT, two-dimensional treatment planning; 3DCRT, three-dimensional conformal radiation therapy; CSI, craniospinal irradiation; IQ, intelligence quotient.
Table A5.
Mean Performance and Severe Neurocognitive Impairment by Cranial Radiation Therapy
| CSI + 2DRT (n = 62) | CSI + 3DCRT (n = 21) | 2DRT (n = 44) | 3DCRT (n = 27) | |||||
|---|---|---|---|---|---|---|---|---|
| Domain | Mean (SD) | % Imp | Mean (SD) | % Imp | Mean (SD) | % Imp | Mean (SD) | % Imp |
| Intelligence | — | 35.0 | — | 25.0 | — | 20.9 | — | 3.7 |
| Full scale | −1.38 (1.2) | 35.0 | −1.08 (1.1) | 25.0 | −0.74 (1.3) | 20.9 | −0.14 (0.9) | 3.7 |
| Verbal | −1.57 (1.2) | 44.3 | −1.40 (1.3) | 47.6 | −0.94 (1.4) | 34.1 | −0.15 (1.0) | 3.7 |
| Perceptual | −1.28 (1.3) | 38.3 | −0.85 (1.2) | 20.0 | −0.55 (1.2) | 16.3 | 0.16 (0.9) | 3.7 |
| Academics | — | 49.1 | — | 31.6 | — | 26.8 | — | 18.5 |
| Word reading | −1.39 (1.3) | 26.3 | −1.00 (0.09) | 21.1 | −1.00 (1.5) | 19.5 | −0.54 (1.0) | 11.1 |
| Mathematics | −2.01 (1.4) | 47.4 | −1.31 (0.9) | 26.3 | −1.09 (1.6) | 22.5 | −0.78 (1.0) | 14.8 |
| Attention | — | 51.7 | — | 35.0 | — | 43.9 | — | 37.0 |
| Focus | −2.25 (2.1) | 45.0 | −1.65 (1.0) | 35.0 | −1.62 (1.8) | 33.3 | −1.32 (1.8) | 30.8 |
| Sustained | −1.64 (4.0) | 25.9 | −0.01 (1.0) | 5.0 | −1.61 (4.2) | 22.5 | −0.09 (1.5) | 7.7 |
| Variability | −0.94 (2.4) | 19 | −0.09 (0.8) | 0.0 | −1.01 (2.9) | 20.0 | 0.16 (1.1) | 7.7 |
| Memory | — | 62.3 | — | 33.3 | — | 38.6 | — | 33.3 |
| New learning | −1.67 (1.1) | 45.9 | −1.20 (1.3) | 25.0 | −1.03 (1.4) | 25.0 | −0.46 (1.3) | 11.1 |
| Short-term recall | −1.39 (1.2) | 36.1 | −0.70 (1.2) | 15.0 | −1.07 (1.5) | 27.3 | −0.89 (1.3) | 25.9 |
| Long-term recall | −1.65 (1.3) | 40.0 | −0.95 (1.2) | 25.0 | −1.16 (1.4) | 27.3 | −0.80 (1.4) | 22.2 |
| Span | −0.97 (1.0) | 21.3 | −0.89 (0.8) | 14.3 | −0.58 (1.4) | 15.9 | −0.26 (1.1) | 3.7 |
| Processing speed | — | 71.6 | — | 50.0 | — | 71.8 | — | 53.9 |
| Motor | −3.42 (1.9) | 71.2 | −2.55 (1.7) | 45.0 | −2.97 (1.9) | 66.7 | −2.57 (2.0) | 53.8 |
| Visual | −1.39 (1.0) | 36.7 | −0.73 (1.2) | 15.0 | −0.92 (1.1) | 20.5 | −0.54 (1.2) | 11.5 |
| Visual-motor | −1.66 (0.8) | 43.3 | −1.23 (0.8) | 20.0 | −1.28 (0.9) | 30.8 | −0.94 (1.3) | 19.2 |
| Executive function | — | 72.1 | — | 57.1 | — | 52.3 | — | 51.9 |
| Flexibility | −3.36 (2.1) | 68.4 | −2.70 (2.3) | 55.0 | −2.03 (2.3) | 43.6 | −1.77 (2.1) | 46.2 |
| Fluency | −1.23 (1.1) | 31.1 | −0.76 (1.2) | 14.3 | −0.65 (1.4) | 20.5 | −0.22 (1.2) | 11.1 |
| Working memory | −1.04 (0.9) | 13.1 | −0.91 (0.7) | 4.8 | −0.50 (1.1) | 9.1 | −0.40 (1.0) | 7.4 |
NOTE. Expected mean = 0, SD = 1 using nationally representative norms. Impairment defined as age-adjusted z score ≤ −2.0.
Abbreviations: 2DRT, two-dimensional treatment planning; 3DCRT, three-dimensional conformal radiation therapy; CSI, craniospinal irradiation; Imp, impairment; SD, standard deviation.
Table A6.
Effect of CSI + 3DCRT Compared With CSI + 2DRT on Neurocognitive Impairment
| Domain/Radiation Modality | Total (No.) | Severely Impaired (No.) | Person-Years | Rate Ratio | 95% CI |
|---|---|---|---|---|---|
| Intelligence | |||||
| CSI + 3DCRT | 20 | 5 | 61.75 | 1.5 | 0.59 to 3.7 |
| CSI + 2DRT | 58 | 19 | 581.9 | 1.0 | |
| Academic | |||||
| CSI + 3DCRT | 19 | 6 | 58.42 | 1.4 | 0.69 to 2.9 |
| CSI + 2DRT | 55 | 26 | 546.2 | 1.0 | |
| Attention | |||||
| CSI + 3DCRT | 20 | 7 | 61.75 | 1.8 | 0.87 to 3.7 |
| CSI + 2DRT | 58 | 29 | 581.9 | 1.0 | |
| Memory | |||||
| CSI + 3DCRT | 21 | 7 | 66.62 | 1.4 | 0.70 to 2.7 |
| CSI + 2DRT | 59 | 36 | 595.4 | 1.0 | |
| Processing speed | |||||
| CSI + 3DCRT | 20 | 10 | 61.75 | 1.8 | 1.2 to 2.9 |
| CSI + 2DRT | 58 | 41 | 581.9 | 1.0 | |
| Executive function | |||||
| CSI + 3DCRT | 21 | 12 | 66.62 | 1.7 | 1.0 to 2.6 |
| CSI + 2DRT | 59 | 42 | 595.4 | 1.0 |
NOTE. Rate ratios and 95% CIs were adjusted for age at neurocognitive assessment, sex, and race. Severely impaired was defined as age-adjusted z score ≤ −2.0.
Abbreviations: 2DRT, two-dimensional treatment planning; 3DCRT, three-dimensional conformal radiation therapy; CSI, craniospinal irradiation.
Table A7.
Effect of 3DCRT Compared With 2DRT on Neurocognitive Impairment
| Domain/Radiation Modality | Total (No.) | Severely Impaired (No.) | Person-Years | Rate Ratio | 95% CI |
|---|---|---|---|---|---|
| Intelligence | |||||
| 3DCRT | 27 | 1 | 130.8 | 0.19 | 0.02 to 1.6 |
| 2DRT | 45 | 11 | 510.9 | 1.0 | |
| Academic | |||||
| 3DCRT | 27 | 5 | 130.8 | 0.89 | 0.36 to 2.2 |
| 2DRT | 43 | 13 | 491.2 | 1.0 | |
| Attention | |||||
| 3DCRT | 27 | 10 | 130.8 | 1.3 | 0.68 to 2.6 |
| 2DRT | 43 | 20 | 491.8 | 1.0 | |
| Memory | |||||
| 3DCRT | 27 | 9 | 130.8 | 1.6 | 0.79 to 3.4 |
| 2DRT | 46 | 19 | 521.2 | 1.0 | |
| Processing speed | |||||
| 3DCRT | 26 | 14 | 122.0 | 1.4 | 0.87 to 2.1 |
| 2DRT | 41 | 30 | 465.8 | 1.0 | |
| Executive function | |||||
| 3DCRT | 27 | 14 | 130.8 | 1.6 | 0.99 to 2.7 |
| 2DRT | 46 | 25 | 521.2 | 1.0 |
NOTE. Rate ratios and 95% CIs were adjusted for age at neurocognitive assessment, sex, and race. Severely impaired was defined as age-adjusted z score ≤ −2.0.
Abbreviations: 2DRT, two-dimensional treatment planning; 3DCRT, three-dimensional conformal radiation therapy.
Footnotes
Supported by the National Cancer Institute (CA195547, M. Hudson, Principal Investigator). Support to St Jude Children's Research Hospital also provided by the Cancer Center Support (CORE) grant (CA21765, C. Roberts, Principal Investigator) and the American Lebanese-Syrian Associated Charities.
Authors' disclosures of potential conflicts of interest are found in the article online at www.jco.org. Author contributions are found at the end of this article.
AUTHOR CONTRIBUTIONS
Conception and design: Tara M Brinkman, Matthew J. Krasin, Gregory T. Armstrong, Rohit P. Ojha, Zsila S. Sadighi, Thomas E. Merchant, Amar Gajjar, Leslie L. Robison, Melissa M. Hudson, Kevin R. Krull
Administrative support: Leslie L. Robison
Provision of study materials: Thomas E. Merchant
Collection and assembly of data: Tara M Brinkman, Matthew J. Krasin, Zsila S. Sadighi, Cara Kimberg, Thomas E. Merchant, Melissa M. Hudson, Kevin R. Krull
Data analysis and interpretation: Tara M Brinkman, Matthew J. Krasin, Wei Liu, Gregory T. Armstrong, Rohit P. Ojha, Pankaj Gupta, Cara Kimberg, Deokumar Srivastava, Amar Gajjar, Leslie L. Robison, Melissa M. Hudson, Kevin R. Krull
Manuscript writing: All authors
Final approval of manuscript: All authors
AUTHORS' DISCLOSURES OF POTENTIAL CONFLICTS OF INTEREST
Long-Term Neurocognitive Functioning and Social Attainment in Adult Survivors of Pediatric CNS Tumors: Results From the St Jude Lifetime Cohort Study
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to www.asco.org/rwc or jco.ascopubs.org/site/ifc.
Tara M. Brinkman
No relationship to disclose
Matthew J. Krasin
No relationship to disclose
Wei Liu
No relationship to disclose
Gregory T. Armstrong
No relationship to disclose
Rohit P. Ojha
No relationship to disclose
Zsila S. Sadighi
No relationship to disclose
Pankaj Gupta
No relationship to disclose
Cara Kimberg
No relationship to disclose
Deokumar Srivastava
Consulting or Advisory Role: SRA International, Department of Defense
Thomas E. Merchant
Consulting or Advisory Role: AstraZeneca
Travel, Accommodations, Expenses: IBA
Amar Gajjar
Consulting or Advisory Role: Celgene
Research Funding: Genentech
Leslie L. Robison
No relationship to disclose
Melissa M. Hudson
No relationship to disclose
Kevin R. Krull
No relationship to disclose
REFERENCES
- 1.Howlander N, Noone A, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2011. National Cancer Institute,: Bethesda, MD; 2011. [Google Scholar]
- 2.Mulhern RK, Palmer SL, Merchant TE, et al. Neurocognitive consequences of risk-adapted therapy for childhood medulloblastoma. J Clin Oncol. 2005;23:5511–5519. doi: 10.1200/JCO.2005.00.703. [DOI] [PubMed] [Google Scholar]
- 3.Palmer SL, Gajjar A, Reddick WE, et al. Predicting intellectual outcome among children treated with 35-40 Gy craniospinal irradiation for medulloblastoma. Neuropsychology. 2003;17:548–555. doi: 10.1037/0894-4105.17.4.548. [DOI] [PubMed] [Google Scholar]
- 4.Spiegler BJ, Bouffet E, Greenberg ML, et al. Change in neurocognitive functioning after treatment with cranial radiation in childhood. J Clin Oncol. 2004;22:706–713. doi: 10.1200/JCO.2004.05.186. [DOI] [PubMed] [Google Scholar]
- 5.Merchant TE, Conklin HM, Wu S, et al. Late effects of conformal radiation therapy for pediatric patients with low-grade glioma: Prospective evaluation of cognitive, endocrine, and hearing deficits. J Clin Oncol. 2009;27:3691–3697. doi: 10.1200/JCO.2008.21.2738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Merchant TE. Three-dimensional conformal radiation therapy for ependymoma. Childs Nerv Syst. 2009;25:1261–1268. doi: 10.1007/s00381-009-0892-9. [DOI] [PubMed] [Google Scholar]
- 7.Merchant TE, Mulhern RK, Krasin MJ, et al. Preliminary results from a phase II trial of conformal radiation therapy and evaluation of radiation-related CNS effects for pediatric patients with localized ependymoma. J Clin Oncol. 2004;22:3156–3162. doi: 10.1200/JCO.2004.11.142. [DOI] [PubMed] [Google Scholar]
- 8.Merchant TE, Kiehna EN, Li C, et al. Radiation dosimetry predicts IQ after conformal radiation therapy in pediatric patients with localized ependymoma. Int J Radiat Oncol Biol Phys. 2005;63:1546–1554. doi: 10.1016/j.ijrobp.2005.05.028. [DOI] [PubMed] [Google Scholar]
- 9.Merchant TE, Sharma S, Xiong X, et al. Effect of cerebellum radiation dosimetry on cognitive outcomes in children with infratentorial ependymoma. Int J Radiat Oncol Biol Phys. 2014;90:547–553. doi: 10.1016/j.ijrobp.2014.06.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Edelstein K, Spiegler BJ, Fung S, et al. Early aging in adult survivors of childhood medulloblastoma: Long-term neurocognitive, functional, and physical outcomes. Neuro-oncol. 2011;13:536–545. doi: 10.1093/neuonc/nor015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ellenberg L, Liu Q, Gioia G, et al. Neurocognitive status in long-term survivors of childhood CNS malignancies: A report from the Childhood Cancer Survivor Study. Neuropsychology. 2009;23:705–717. doi: 10.1037/a0016674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Maddrey AM, Bergeron JA, Lombardo ER, et al. Neuropsychological performance and quality of life of 10 year survivors of childhood medulloblastoma. J Neurooncol. 2005;72:245–253. doi: 10.1007/s11060-004-3009-z. [DOI] [PubMed] [Google Scholar]
- 13.Ness KK, Leisenring WM, Huang S, et al. Predictors of inactive lifestyle among adult survivors of childhood cancer: A report from the Childhood Cancer Survivor Study. Cancer. 2009;115:1984–1994. doi: 10.1002/cncr.24209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hudson MM, Ness KK, Nolan VG, et al. Prospective medical assessment of adults surviving childhood cancer: Study design, cohort characteristics, and feasibility of the St. Jude Lifetime Cohort study. Pediatr Blood Cancer. 2011;56:825–836. doi: 10.1002/pbc.22875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ojha RP, Oancea SC, Ness KK, et al. Assessment of potential bias from non-participation in a dynamic clinical cohort of long-term childhood cancer survivors: Results from the St. Jude Lifetime Cohort Study. Pediatr Blood Cancer. 2013;60:856–864. doi: 10.1002/pbc.24348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Landier W, Wallace WH, Hudson MM. Long-term follow-up of pediatric cancer survivors: Education, surveillance, and screening. Pediatr Blood Cancer. 2006;46:149–158. doi: 10.1002/pbc.20612. [DOI] [PubMed] [Google Scholar]
- 17.Landier W, Bhatia S, Eshelman DA, et al. Development of risk-based guidelines for pediatric cancer survivors: The Children’s Oncology Group Long-Term Follow-Up Guidelines from the Children’s Oncology Group Late Effects Committee and Nursing Discipline. J Clin Oncol. 2004;22:4979–4990. doi: 10.1200/JCO.2004.11.032. [DOI] [PubMed] [Google Scholar]
- 18.Wechsler D. Wechsler Abbreviated Scale of Intelligence. San Antonio, TX: Psychological Corporation; 1999. [Google Scholar]
- 19.Woodcock R, McGrew K, Mather N. Woodcock-Johnson Tests of Achievement. Itasca, IL, : Riverside; 2001. [Google Scholar]
- 20.Delis D, Kramer J, Kaplan E, et al. California Verbal Learning Test. (ed 2). San Antonio, TX,: Psychological Coorporation; 2000. [Google Scholar]
- 21.Strauss E, Sherman E, Spreen O. A Compendium of Neuropsychological Tests: Administration, Norms, and Comentray. (ed 3). New York, NY,: Oxford University Press; 2006. [Google Scholar]
- 22.Conners C. Conners’ Continuous Performance Test II. North Tonawanda, NY,: Multihealth Systems; 2001. [Google Scholar]
- 23.Roth R, Isquith P, Gioia G. Behavior Rating Inventory of Executive Function - Adult Version. Lutz, FL,: Psychological Assessment Resources; 2005. [Google Scholar]
- 24.Gurney JG, Krull KR, Kadan-Lottick N, et al. Social outcomes in the Childhood Cancer Survivor Study cohort. J Clin Oncol. 2009;27:2390–2395. doi: 10.1200/JCO.2008.21.1458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Robison LL, Green DM, Hudson M, et al. Long-term outcomes of adult survivors of childhood cancer. Cancer. 2005;104:2557–2564. doi: 10.1002/cncr.21249. [DOI] [PubMed] [Google Scholar]
- 26.Rothman KJ. No adjustments are needed for multiple comparisons. Epidemiology. 1990;1:43–46. [PubMed] [Google Scholar]
- 27.Rothman KJ. Six persistent research misconceptions. J Gen Intern Med. 2014;29:1060–1064. doi: 10.1007/s11606-013-2755-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Elger CE, Helmstaedter C, Kurthen M. Chronic epilepsy and cognition. Lancet Neurol. 2004;3:663–672. doi: 10.1016/S1474-4422(04)00906-8. [DOI] [PubMed] [Google Scholar]
- 29.Vingerhoets G. Cognitive effects of seizures. Seizure. 2006;15:221–226. doi: 10.1016/j.seizure.2006.02.012. [DOI] [PubMed] [Google Scholar]
- 30.Brinkman TM, Zhang N, Ullrich NJ, et al. Psychoactive medication use and neurocognitive function in adult survivors of childhood cancer: A report from the Childhood Cancer Survivor study. Pediatr Blood Cancer. 2013;60:486–493. doi: 10.1002/pbc.24255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Savolainen S, Laakso MP, Paljärvi L, et al. MR imaging of the hippocampus in normal pressure hydrocephalus: Correlations with cortical Alzheimer’s disease confirmed by pathologic analysis. AJNR Am J Neuroradiol. 2000;21:409–414. [PMC free article] [PubMed] [Google Scholar]
- 32.Krull KR, Brinkman TM, Li C, et al. Neurocognitive outcomes decades after treatment for childhood acute lymphoblastic leukemia: A report from the St Jude lifetime cohort study. J Clin Oncol. 2013;31:4407–4415. doi: 10.1200/JCO.2012.48.2315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Moore IM, Hockenberry MJ, Anhalt C, et al. Mathematics intervention for prevention of neurocognitive deficits in childhood leukemia. Pediatr Blood Cancer. 2012;59:278–284. doi: 10.1002/pbc.23354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zou P, Conklin HM, Scoggins MA, et al. Functional MRI in medulloblastoma survivors supports prophylactic reading intervention during tumor treatment. Brain Imaging Behav. doi: 10.1007/s11682-015-9390-8. [epub ahead of print on May 14, 2015] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hardy KK, Willard VW, Allen TM, et al. Working memory training in survivors of pediatric cancer: A randomized pilot study. Psychooncology. 2013;22:1856–1865. doi: 10.1002/pon.3222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Butler RW, Copeland DR, Fairclough DL, et al. A multicenter, randomized clinical trial of a cognitive remediation program for childhood survivors of a pediatric malignancy. J Consult Clin Psychol. 2008;76:367–378. doi: 10.1037/0022-006X.76.3.367. [DOI] [PMC free article] [PubMed] [Google Scholar]