Abstract
Background
Because of advancements in care, there has been a decline in mortality from congenital heart defects (CHD) over the last several decades. However, there are no current empirical data documenting the number of people living with CHD in the United States (US). Our aim was to estimate the CHD prevalence across all age groups in the US in the year 2010.
Methods
The age-, sex-, and severity-specific observed prevalence of CHD in Québec, Canada in the year 2010 was assumed to equal the CHD prevalence in the non-Hispanic white population in the US in 2010. A race-ethnicity adjustment factor, reflecting differential survival between racial-ethnic groups through age 5 for persons with a CHD and that in the general US population, was applied to the estimated non-Hispanic white rates to derive CHD prevalence estimates among US non-Hispanic blacks and Hispanics. Confidence intervals for the estimated CHD prevalence rates and case counts were derived using a combination of Taylor series approximations and Monte Carlo simulation.
Results
We estimated that approximately 2.4 million people (1.4 million adults, 1 million children) were living with CHD in the US in 2010. Nearly 300,000 of these individuals had severe CHD.
Conclusions
Our estimates highlight the need for two important efforts: (1) planning for health services delivery to meet the needs of the growing population of adults with CHD and; (2) the development of surveillance data across the lifespan to provide empirical estimates of the prevalence of CHD across all age groups in the US.
Keywords: heart defects, congenital, epidemiology, population, prevalence
Congenital heart defects (CHD) are diagnosed in approximately 8–10 per 1,000 live births in the United States (US).1,2 CHD are associated with lifelong comorbidity and health services utilization and the need for repeated surgical interventions3 has resulted in a significant increase in health services utilization during infancy and childhood,4 transition years,5 and adulthood.6 In the US in 2004, CHD accounted for more than 46,000 hospitalizations among individuals of all ages and nearly $1.4 billion in hospital costs.7
Advances in medical and surgical treatment have led to decreased mortality attributable to CHD across all age groups in the US.8–11 With lower mortality, a demographic shift in the population living with CHD is likely. The most compelling evidence of this demographic shift comes from recent analyses of administrative data in Québec. Analyses of these data were the first to show time trends in the lifetime CHD prevalence in the general population, documenting a 22% increase in the number of children and 85% increase in the number of adults living with severe CHD between the years 1985 and 2000.12,13 The median age at death among those with severe CHD has reportedly increased from two years of age in 1987–1993 to 23 years of age in 1999–2005.14 Similar demographic trends are expected to exist in other high-income countries, including the US, but observation of such patterns in the US is challenging in the absence of population-based surveillance systems with long-term follow-up, or extensively linked administrative datasets.
Two previous publications estimated the size of the adult population living with CHD in the US in the year 2000 based on estimates of birth prevalence and survival.15,16 In 2001, a task force of the American College of Cardiology estimated approximately 788,000 adults with CHD were living in the US in the year 2000.16 In a 2004 analysis, this estimate was refined based on survival probabilities considering either treatment or no treatment, resulting in a range – from 650,000 without treatment to 1,330,000 with treatment.15 In 2009, Marelli and colleagues estimated that 860,000 adults with CHD were living in the US in the year 2000, based on an extrapolation of the prevalence estimates from the year 2000 in Québec.13 Evidence-based planning for health services delivery to meet the needs of the growing population of adults with CHD in the US will depend on the availability of contemporary data on prevalence of CHD across the lifespan. The goal of the current study was to provide estimates of the prevalence of CHD across all age groups in the US in the year 2010.
METHODS
Based on the recently published estimates of the age-, sex- and severity-specific prevalence of CHD in Québec, Canada,17 we assumed that the Québec CHD prevalence within age and sex strata was equal to that in similar strata in the non-Hispanic white population in the United States in 2010. We then applied a race-ethnicity adjustment factor to derive CHD prevalence estimates for the U.S. non-Hispanic Black and Hispanic populations based on the non-Hispanic white estimates. A detailed description of the methods, assumptions and derivation of the estimators used in developing the U.S. CHD prevalence estimates is given in the Supplemental Material. A complete list of assumptions can be found in Supplemental Table 1.
Data Inputs
CHD Prevalence in Québec in 2010
Age-, sex- and severity-specific prevalence of CHD in Québec in 2010 were calculated from the Québec Congenital Heart Disease Database, based on 28 years of longitudinally-linked administrative data. Detailed methods for the development of these estimates are published.17 Access to health care is universal in Québec and every individual is assigned a unique medical record number in the first year of life that is used to track all diagnoses and health services delivered. These interactions with the health care system are recorded across several administrative databases including physicians’ services and drug claims, hospital discharges, health insurance, and the death registry. The Québec Congenital Heart Disease Database was created by linking the relevant databases for all Québec patients with at least one diagnostic or procedural code for a CHD who came into contact with the healthcare system in Québec from January 1, 1983, to December 31, 2010 (inclusive).17 Diagnostic codes adhered to the International Classification of Disease-9th Revision (ICD-9) and 10th Revision (ICD-10; as of 2006). Severe CHD were defined based on these code systems as definitive diagnoses or surgical procedures to treat the following CHD types: tetralogy of Fallot, truncus arteriosus, complete and congenitally corrected transposition of the great arteries, atrioventricular septal defect, or univentricular hearts.12,17 Severe CHD and all other CHD diagnoses were included in estimates for all CHD. CHD prevalence in Québec in 2010 was calculated as the total number of persons living with CHD as of midyear per 1,000 population. Children were defined as individuals aged less than 18 years; adults were 18 years and older. (Supplemental Table 2)
US Population in the Year 2010 and Racial-Ethnic-Specific CHD Survival and All-Cause Mortality
Age-, sex- and racial-ethnic-specific estimates for the size of the US population were derived from the July 1, 2010 US Census Bureau intercensal estimates (http://www.census.gov/popest/data/intercensal/national/files/US-EST00INT-ALLDATA.pdf; Accessed November 11, 2011)). Racial-ethnic differences in CHD mortality were estimated for children 0–5 years of age based on published estimates of 5-year survival among non-Hispanic white, non-Hispanic black and Hispanic children with specific CHD using data from the Texas Birth Defects Registry.18 The published survival data were aggregated by the authors (WNN, PX) into groupings of all CHD and severe CHD to correspond to the prevalence data available from Québec. Racial-ethnic differences in all-cause mortality for children 0–5 years of age were derived from 1999–2014 detailed mortality data available from CDC WONDER (Wide-ranging Online Data for Epidemiologic Research).19 (Supplemental Tables 3 and 4).
Statistical Analysis
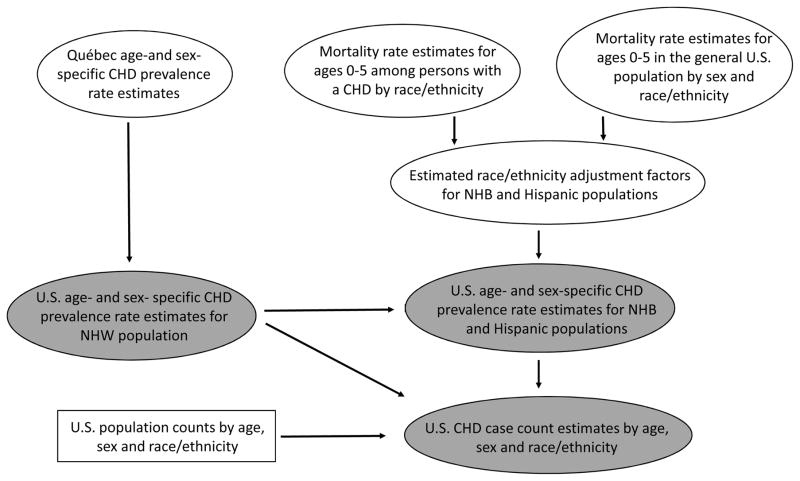
The Figure is a flowchart illustrating the input data and steps in the estimation process. As a first step in the estimation process, we used the CHD age-, sex-, and severity-specific prevalence estimates in Québec in 2010 as estimates for the corresponding age-, sex- and severity CHD prevalence in the 2010 among the US non-Hispanic white population, We then estimated race-ethnicity adjustment factors that, under some simplifying assumptions (Supplemental Table 1), correspond to the ratio of CHD prevalence among either non-Hispanic blacks to non-Hispanic whites or Hispanics to non-Hispanic whites. Therefore, multiplying the estimated non-Hispanic white CHD prevalence by the appropriate adjustment factor results in estimate of CHD prevalence within the other racial-ethnic populations. A detailed rationale for the estimator for the race-ethnicity adjustment factor is provided in the Supplemental Material, however, the key assumptions underlying this approach are that U.S. birth prevalence of CHD does not differ among the racial-ethnic groups considered in this analysis and that survival probability within racial-ethnic groups among those born with a CHD and the U.S. general population differs only though age 5.
Figure.
Flow chart illustrating input data, age and race-ethnicity specific mortality adjustments, and data outputs for the estimation of congenital heart defects (CHD) prevalence in the United States in 2010
The oval-shaped model inputs and estimates are considered to be subject to sampling variability. The rectangular-shaped population count input node is considered known.
The shaded nodes represent estimates produced by the modeling process while non-shaded nodes represent either model inputs or intermediate steps.
NHW, Non-Hispanic White; NHB, Non-Hispanic Black
We used a combination of Taylor series approximations and Monte Carlo simulation, in which the model inputs were repeatedly drawn from their assumed sampling distributions, to estimate the sampling variability for the U.S. CHD prevalence and case count estimates. While the U.S. Census-based population counts were considered fixed, all other inputs to the model, observed prevalence from Québec, mortality among persons with a CHD observed in the Texas Birth Defect Registry and death counts from the detailed mortality file, were considered to be subject to sampling variability. Standard errors for these inputs were estimated under a Poisson assumption on the number of CHD cases or number of deaths as appropriate. The sampling distributions for these inputs were assumed to be normal with mean equal to the observed value and variance equal to the square of the estimated standard error. The Monte Carlo simulation process was repeated 10,000 times with CHD prevalence and case count estimates for each repetition calculated based on values for the model inputs drawn from their assumed sampling distributions in each iteration. The resulting 10,000 calculated values for CHD prevalence and case counts were assumed to reflect the sampling variability of the estimators. These generated sampling distributions for U.S. CHD prevalence and cases counts are summarized using the mean value and 95% confidence intervals defined by the 2.5th percentile and 97.5th percentile of the generated values. To reflect the lack of certainty in these modeled estimates, all CHD case count estimates are rounded to the nearest 1,000. A detailed description of the methods used to estimate the sampling error in the CHD estimates is provided in the Supplemental Material. This study did not involve human subjects and is exempt from Institutional Review Board review.
RESULTS
Approximately 2.4 million people were estimated to be alive with CHD in the US in 2010 (Table 1). Approximately 290,000 (12%) of these individuals had severe CHD. The prevalence of CHD ranged from approximately 6 per 1,000 in adults to 13 per 1,000 in children although there are more adults estimated to be living with CHD (approximately 1.4 million) compared to children (approximately 1 million). Considering differences by race-ethnicity, the prevalence was slightly higher in the non-Hispanic white and Hispanic population compared with the non-Hispanic black population. Given the relative sizes of the US population, however, the vast majority of individuals estimated to be living with CHD in the US are non-Hispanic white (approximately 1.7 million) compared with non-Hispanic black or Hispanic (approximately 700,000).
TABLE 1.
Estimated prevalence of congenital heart defects (CHD) for individuals of all ages, among children (0–17 years), and among adults (18 years and older) by severity, and by race-ethnicity in the United States, 2010
| Age Group | CHD Severity | Estimated US Prevalence per 1,000 (95% Confidence Interval) | Estimated Number of Individuals (95% Confidence Interval)* |
|---|---|---|---|
| All ages | Overall | 7.85 (7.79 – 7.92) | 2,425,000 (2,405,000 – 2,444,000) |
| Severe | 0.92 (0.90 – 0.94) | 283,000 (277,000 – 290,000) | |
| Children | Overall | 13.21 (13.03 – 13.39) | 980,000 (966,000 – 993,000) |
| Severe | 1.66 (1.60 – 1.73) | 123,000 (119,000 – 128,000) | |
| Adults | Overall | 6.16 (6.10 – 6.22) | 1,444,500 (1,431,000 – 1,459,000) |
| Severe | 0.68 (0.66 – 0.70) | 160,000 (155,000 – 165,000) |
| Age Group | Race-Ethnicity | Estimated US Prevalence per 1,000 (95% Confidence Interval) | Estimated Number of Individuals (95% Confidence Interval)* |
|---|---|---|---|
| Children | Non-Hispanic White | 13.31 (13.12 – 13.49) | 620,000 (612,000 – 629,000) |
| Non-Hispanic Black | 12.69 (12.50 – 12.88) | 133,000 (131,000 – 135,000) | |
| Hispanic | 13.26 (13.08 – 13.45) | 227,000 (224,000 – 230,000) | |
| Adults | Non-Hispanic White | 6.36 (6.29 – 6.42) | 1,104,000 (1,094,000 – 1,115,000) |
| Non-Hispanic Black | 5.63 (5.56 – 5.69) | 155,000 (153,000 – 156,000) | |
| Hispanic | 5.58 (5.52 – 5.65) | 186,000 (184,000 – 188,000) |
Overall, there was a slight predominance of females compared to males (Table 2). The most striking difference between the sexes was among individuals 25–44 years of age; women in this age group had a prevalence of 6 per 1,000 while men had a prevalence of 3.8 per 1,000 – which translated to approximately 247,000 affected women and 157,000 affected men.
TABLE 2.
Sex-, age-group, and severity-specific estimates of prevalence of congenital heart defects (CHD) in the United States, 2010
| Age Group (y) | CHD Severity | Estimated US Prevalence per 1,000 (95% Confidence Interval) | Estimated Number of Individuals (95% Confidence Interval)* |
|---|---|---|---|
|
Females
| |||
| 0 to 5 | Overall | 11.54 (11.12 – 11.96) | 137,000 (132,000 – 142,000) |
| Severe | 1.07 (0.95 – 1.20) | 13,000 (11,000 – 14,000) | |
| 6 to 12 | Overall | 15.57 (15.11 – 16.05) | 218,000 (212,000 – 225,000) |
| Severe | 1.69 (1.53 – 1.84) | 24,000 (21,000 – 26,000) | |
| 13 to 17 | Overall | 12.62 (12.15 – 13.08) | 130,000 (126,000 – 135,000) |
| Severe | 1.87 (1.70 – 2.05) | 19,000 (18,000 – 21,000) | |
| 18 to 24 | Overall | 6.35 (6.08 – 6.62) | 95,000 (91,000 – 99,000) |
| Severe | 1.33 (1.20 – 1.45) | 20,000 (18,000 – 22,000) | |
| 25 to 44 | Overall | 6.01 (5.86 – 6.16) | 247,000 (241,000 – 253,000) |
| Severe | 1.13 (1.06 – 1.19) | 46,000 (43,000 – 49,000) | |
| 45 and older | Overall | 6.70 (6.58 – 6.81) | 433,000 (426,000 – 441,000) |
| Severe | 0.41 (0.38 – 0.44) | 26,000 (25,000 – 28,000) | |
| All ages | Overall | 8.03 (7.94 – 8.13) | 1,261,000 (1,246,000 – 1,275,000) |
| Severe | 0.95 (0.91 – 0.98) | 148,000 (143,000 – 153,000) | |
|
| |||
|
Males
| |||
| 0 to 5 | Overall | 13.96 (13.52 – 14.42) | 173,000 (167,000 – 179,000) |
| Severe | 1.45 (1.30 – 1.59) | 18,000 (16,000 – 20,000) | |
| 6 to 12 | Overall | 14.33 (13.89 – 14.78) | 210,000 (204,000 – 217,000) |
| Severe | 2.00 (1.84 – 2.17) | 29,000 (27,000 – 32,000) | |
| 13 to 17 | Overall | 10.18 (9.78 – 10.59) | 111,000 (107,000 – 115,000) |
| Severe | 1.85 (1.68 – 2.03) | 20,000 (18,000 – 22,000) | |
| 18 to 24 | Overall | 5.05 (4.82 – 5.29) | 79,000 (75,000 – 83,000) |
| Severe | 1.11 (1.00 – 1.22) | 17,000 (16,000 – 19,000) | |
| 25 to 44 | Overall | 3.83 (3.71 – 3.95) | 157,000 (153,000 – 162,000) |
| Severe | 0.76 (0.71 – 0.81) | 31,000 (29,000 – 33,000) | |
| 45 and older | Overall | 7.59 (7.46 – 7.72) | 434,000 (426,000 – 441,000) |
| Severe | 0.33 (0.30 – 0.36) | 19,000 (17,000 – 20,000) | |
|
| |||
| All ages | Overall | 7.67 (7.58 – 7.76) | 1,164,000 (1,150,000 – 1,177,000) |
| Severe | 0.89 (0.86 – 0.92) | 135,000 (130,000 – 140,000) | |
DISCUSSION
There are published estimates of the size of the CHD-affected population in the US in the year 2000.13,15,16 A task force that convened as part of the American College of Cardiology’s 32nd Bethesda Conference estimated that in the year 2000, the total number of adults living with CHD in the United States was approximately 788,000 – a combination of adults with simple (368,800), moderate (302,500) and complex CHD (117,000) – based on estimates of the birth prevalence and survival from the New England Regional Infant Cardiac Program.16,20 Subsequently, Hoffman and colleagues15 refined these estimates under two extreme scenarios. If all individuals born with CHD were treated, they estimated approximately 1.3 million adult survivors - 750,000 with simple CHD and 580,000 with moderate or complex CHD; without treatment, the number of survivors in each group would be 400,000 and 250,000 respectively.15 Marelli and colleagues, in 2007, published estimates of the lifetime prevalence of CHD in Québec in 200012 categorized by those with “severe” CHD and all other CHD. The proportion of the population with severe CHD was smaller (9%) than the 15% with complex CHD estimated by Hoffman and colleagues in 200415 primarily due to differences in the specific CHD included in the respective categories. In 2009, Marelli and colleagues extrapolated that lifetime prevalence estimate to the US population in 2000 and estimated approximately 1.7 million affected individuals, evenly divided among adults and children.13
Importantly, the current study is an extension of Marelli’s 2009 analysis, using updated 2010 Québec estimates as input data,17 and attempts to account for racial-ethnic differences in survival in the US. Analyses of cross-sectional US death certificate data have shown that age-adjusted mortality due to CHD is greater for non-Hispanic Blacks than non-Hispanic whites and that decreases in mortality due to CHD have been greatest for non-Hispanic whites compared to other racial-ethnic groups in the US.9 In addition, survival analyses of population-based birth defects surveillance data have shown that mortality in ages 0 to 5 among non-Hispanic black and Hispanic infants born with CHD is generally higher than among non-Hispanic whites, although there are differences among CHD subtypes.11,18 The reasons for higher mortality among those age 0 to 5 born with a CHD in minority populations are not well understood; although differential access to healthcare among minority populations has frequently been a proposed explanation, it is likely not the only explanation. For example, there are persistent racial-ethnic differences in post-operative mortality after CHD surgery that do not seem to be explained by differences in access to care.21 Nembhard and colleagues18 have hypothesized that other factors, such as the severity of the defect, use of home care both before and after surgery, and differences in the frequency and severity of comorbid conditions such as asthma, could be responsible for racial-ethnic differences in mortality among those age 0 to 5 with CHD. Previous reports of the size of the population living with CHD have not attempted to account for these recognized, but largely unexplained, differences in child survival.
Conceptually, the lifetime prevalence of CHD in a closed population (i.e., no migration) or in a population with zero net migration is the product of the birth prevalence and survival. The estimated birth prevalence of CHD in the US is approximately 8–10 per 1,000 live births1,2 and has increased over the last 30–40 years, driven largely by increasing prevalence and improved detection of less severe phenotypes such as ventricular septal defects and atrial septal defects.22,23 Survival, however, has also increased because of advancements in medical and surgical care. According to US death certificate data, age adjusted annual infant mortality due to CHD decreased 40% between 1979 and 19938 and continued to decrease through the mid-2000s9 although death from CHD still remains the most common cause of infant mortality from birth defects in the US.24 In addition, according to death certificates, overall mortality decreased 40% in people with tetralogy of Fallot and 60% in those with ventricular septal defect between 1980 and 2005.25 Similarly, in the Québec CHD database, among over 8,000 patients with severe CHD followed for over 1 million life-years, the median age of death increased from two years of age in 1987–1993 to 23 years of age in 1999–2005.14 The authors concluded that mortality from CHD has changed from a bimodal distribution of death to a distribution skewed towards the elderly, matching that of the population unaffected by CHD.14 Finally, with respect to sex, it has been observed that among adults aged 18–45 years with CHD, women had lower in-hospital mortality than men, independent of pregnancy history.26
Despite the fact that most CHD patients require lifelong cardiology care,27 data from a recent study indicated that over 40% of individuals with CHD reported a gap of at least three years in such care28 with the late teens being the most common age of the first gap. This corresponds to the typical time of transition from pediatric to adult oriented medical care during which pediatric patients can be lost to follow-up and fail to undergo a successful transition.28,29 A long lapse in medical care may result in adverse outcomes, since adults with CHD (ACHD) must be monitored frequently for the development of complications from their structural heart defects, such as arrhythmias and pulmonary hypertension.29,30 As of 2010, the consensus among the ACHD community was that the ACHD clinics in the US at that time were inadequate to meet the needs of this population.29 The treatment recommendations from the 32nd Bethesda Conference were that adults with moderate or complex CHD should be followed in a regional ACHD center at least once or twice each year.16 Based on the 2009 estimate of nearly 860,000 adults with CHD in the US in the year 2000, and assuming that 50% of them were seen at least once each year in an ACHD regional center, it was estimated that 150 large regional centers would be required to care for the ACHD population in the US.13 If the current estimate of 1.4 million adults with CHD in 2010 is considered, it represents a 63% increase in the estimated size of the adult population with CHD in the US since the year 2000. It is conceivable that over 200 ACHD regional centers will be required to meet the needs of this rapidly growing population. According to the Adult Congenital Heart Association, currently there are 114 ACHD regional centers in the United States (http://www.achaheart.org/home/clinic-directory.aspx - Accessed 12 February 2015).
The study findings presented here must be interpreted in light of several potential methodological limitations. First, the Canadian input prevalence data were derived from administrative data in which CHD were identified via ICD-9 coding; cases identified in this fashion are more subject to misclassification than those actively ascertained through medical records review.10 However, to minimize the possibility of misclassification resulting from the use of administrative data, Marelli and colleagues developed extensive algorithms to exclude individuals who seemed to have miscoded CHD and manually reviewed approximately 28% of their original dataset (17,474/61,386) to validate the algorithm’s categorization.12 In addition, the input data for the current analysis17 reassuringly had a CHD birth prevalence comparable those reported from active birth defects surveillance systems.2 An advantage of using input data derived from inpatient hospitalization and outpatient records across the lifespan, is that individuals with CHD who were clinically diagnosed at any age were included in the dataset, and diagnosis at birth (which can be missed depending on the CHD subtype) was not required for inclusion. Second, given that individuals who neither represent a visible racial-ethnic minority nor consider themselves Aboriginal represent 89% of the Québec population according to the 2011 Canadian National Household Survey,31 the calculated age-, sex- and severity-specific prevalences for 2010 in Québec were assumed to be equivalent to that of the US non-Hispanic white population for the same time period. We assumed there were no differences in either birth prevalence or survival between the Québec population and the US non-Hispanic white population with CHD. Any differences in environmental or genetic risk factors or health care provision between Canada and the US were assumed to have no impact on prevalence or survival. Third, for individuals six years of age and older, we assumed the impact of mortality due to CHD was the same across all age groups. For children 0–5 years, we approximated CHD racial-ethnic prevalence ratios using their corresponding ratios of cumulative survival with CHD thereby assuming no racial-ethnic differences in CHD prevalence at birth. Given that racial-ethnic differences in CHD prevalence in the US are inconsistent across CHD subtypes and eras,22,23,32 accounting for differences in birth prevalence across racial-ethnic groups was not feasible in this study. Other nuances of differential survival by race-ethnicity could not be modeled. For example, recently published data from California pediatric cardiology specialty care centers indicated substantial differences in hospital discharges among Hispanics and non-Hispanic whites over a 28-year period.33 From 1983–2011, the percent of discharges identified as non-Hispanic white decreased from 58% to 30%; in the same time period, the percent identified as Hispanic rose from 24% to 48%. This demographic shift, specific to California, might be attributable to underlying population growth, immigration, or changing access to health care.
Despite these limitations, this is the first report to provide a contemporary estimate of the number of people living with CHD in the US stratified by age, sex, race-ethnicity and CHD severity. Compared with population estimates generated for the year 2000,13 the current estimates for the year 2010 represent a 40% increase in the total number of individuals living with CHD in the US and over a 60% increase in the size of the adult population alone. Results from this analysis can help inform two efforts critical to this emergent public health issue: (1) planning for health services delivery to meet the needs of the growing population of adults with CHD and; (2) the development of surveillance data across the lifespan to provide more accurate, empirical estimates of the prevalence of CHD across all age groups in the US.
Supplementary Material
Clinical Perspective.
What is new?
There are no current empirical data documenting the number of people living with congenital heart defects (CHD) in the United States (US).
The aim of this study was to estimate the CHD prevalence across all age groups in the US in the year 2010. Using prevalence data from Québec, Canada in the year 2010 as a foundation for a mathematical model, we estimated that approximately 2.4 million people (1.4 million adults, 1 million children) were living with CHD in the US in the year 2010. Nearly 300,000 people had severe CHD.
Overall, there was a slight predominance of females compared to males.
What are the clinical implications?
This current estimate of 1.4 million adults with CHD in 2010 corresponds to a 63% increase in the estimated size of the adult population with CHD since the year 2000.
Resource allocation for health services delivery will need to account for this growing population of adults with CHD.
Acknowledgments
The authors would like to acknowledge Dr. Raluca Ionescu-Ittu for her assistance with acquisition and analysis of the Québec data.
Sources of Funding
Dr. Ariane Marelli received peer-reviewed public funding from the Canadian Institute of Health Research, the Heart and Stroke Foundation of Canada, and the Fonds de la recherche en santé du Québec. Dr. Adolfo Correa was supported by contracts HHSN268201300046C (PI: Correa) and HHSN268201300047C (PI: Correa); subaward from HHSN268201300049C (PI: Payton) from the National Heart, Lung, and Blood Institute and the National Institute on Minority Health and Health Disparities; grant 1U01AT006239-01 (PI: Marshall) from the National Center for Complementary and Alternative Medicine; and subaward from P60MD002249-01 (PI: Diez-Roux) from the National Institute on Minority Health and Health Disparities. Dr. Wendy Nembhard was supported by funds from the Arkansas Biosciences Institute.
Footnotes
Disclosures
None.
Disclaimer
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
References
- 1.Hoffman J, Kaplan S. The incidence of congenital heart disease. J Am Coll Cardiol. 2002;39:1890–1900. doi: 10.1016/s0735-1097(02)01886-7. [DOI] [PubMed] [Google Scholar]
- 2.Reller MD, Strickland MJ, Riehle-Colarusso T, Mahle WT, Correa A. Prevalence of congenital heart defects in metropolitan Atlanta, 1998–2005. J Pediatr. 2008;153:807–813. doi: 10.1016/j.jpeds.2008.05.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ionescu-Ittu R, Mackie AS, Abrahamowicz M, Pilote L, Tchervenkov C, Martucci G, Marelli AJ. Valvular operations in patients with congenital heart disease: increasing rates from 1988 to 2005. Ann Thorac Surg. 2010;90:1563–1569. doi: 10.1016/j.athoracsur.2010.07.017. [DOI] [PubMed] [Google Scholar]
- 4.Mackie AS, Ionescu-Ittu R, Pilote L, Rahme E, Marelli AJ. Hospital readmissions in children with congenital heart disease: a population-based study. Am Heart J. 2008;155:577–584. doi: 10.1016/j.ahj.2007.11.003. [DOI] [PubMed] [Google Scholar]
- 5.Gurvitz MZ, Inkelas M, Lee M, Stout K, Escarce J, Chang RK. Changes in hospitalization patterns among patients with congenital heart disease during the transition from adolescence to adulthood. J Am Coll Cardiol. 2007;49:875–882. doi: 10.1016/j.jacc.2006.09.051. [DOI] [PubMed] [Google Scholar]
- 6.Mackie AS, Pilote L, Ionescu-Ittu R, Rahme E, Marelli AJ. Health care resource utilization in adults with congenital heart disease. Am J Cardiol. 2007;99:839–843. doi: 10.1016/j.amjcard.2006.10.054. [DOI] [PubMed] [Google Scholar]
- 7.Russo CA, Elixhauser A. Healthcare Cost and Utilization Project (HCUP) Statistical Briefs. Rockville (MD): 2006. Hospitalizations for Birth Defects, 2004: Statistical Brief #24. [Google Scholar]
- 8.Boneva RS, Botto LD, Moore CA, Yang Q, Correa A, Erickson JD. Mortality associated with congenital heart defects in the United States: trends and racial disparities, 1979–1997. Circulation. 2001;103:2376–2381. doi: 10.1161/01.cir.103.19.2376. [DOI] [PubMed] [Google Scholar]
- 9.Gilboa SM, Salemi JL, Nembhard WN, Fixler DE, Correa A. Mortality resulting from congenital heart disease among children and adults in the United States, 1999 to 2006. Circulation. 2010;122:2254–2263. doi: 10.1161/CIRCULATIONAHA.110.947002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Oster ME, Lee KA, Honein MA, Riehle-Colarusso T, Shin M, Correa A. Temporal trends in survival among infants with critical congenital heart defects. Pediatrics. 2013;131:e1502–1508. doi: 10.1542/peds.2012-3435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang Y, Liu G, Druschel CM, Kirby RS. Maternal race/ethnicity and survival experience of children with congenital heart disease. J Pediatr. 2013;163:1437–1442. doi: 10.1016/j.jpeds.2013.06.084. [DOI] [PubMed] [Google Scholar]
- 12.Marelli AJ, Mackie AS, Ionescu-Ittu R, Rahme E, Pilote L. Congenital heart disease in the general population: changing prevalence and age distribution. Circulation. 2007;115:163–172. doi: 10.1161/CIRCULATIONAHA.106.627224. [DOI] [PubMed] [Google Scholar]
- 13.Marelli AJ, Therrien J, Mackie AS, Ionescu-Ittu R, Pilote L. Planning the specialized care of adult congenital heart disease patients: from numbers to guidelines; an epidemiologic approach. Am Heart J. 2009;157:1–8. doi: 10.1016/j.ahj.2008.08.029. [DOI] [PubMed] [Google Scholar]
- 14.Khairy P, Ionescu-Ittu R, Mackie AS, Abrahamowicz M, Pilote L, Marelli AJ. Changing mortality in congenital heart disease. J Am Coll Cardiol. 2010;56:1149–1157. doi: 10.1016/j.jacc.2010.03.085. [DOI] [PubMed] [Google Scholar]
- 15.Hoffman JI, Kaplan S, Liberthson RR. Prevalence of congenital heart disease. Am Heart J. 2004;147:425–439. doi: 10.1016/j.ahj.2003.05.003. [DOI] [PubMed] [Google Scholar]
- 16.Warnes CA, Liberthson R, Danielson GK, Dore A, Harris L, Hoffman JI, Somverville J, Williams RG, Webb GD. Task force 1: the changing profile of congenital heart disease in adult life. J Am Coll Cardiol. 2001;37:1170–1175. doi: 10.1016/s0735-1097(01)01272-4. [DOI] [PubMed] [Google Scholar]
- 17.Marelli AJ, Ionescu-Ittu R, Mackie AS, Guo L, Dendukuri N, Kaouache M. Lifetime prevalence of congenital heart disease in the general population from 2000 to 2010. Circulation. 2014;130:749–756. doi: 10.1161/CIRCULATIONAHA.113.008396. [DOI] [PubMed] [Google Scholar]
- 18.Nembhard WN, Xu P, Ethen MK, Fixler DE, Salemi JL, Canfield MA. Racial/ethnic disparities in timing of death during childhood among children with congenital heart defects. Birth Defects Res A Clin Mol Teratol. 2013;97:628–640. doi: 10.1002/bdra.23169. [DOI] [PubMed] [Google Scholar]
- 19.Centers for Disease Control and Prevention, National Center for Health Statistics. Compressed Mortality File 1999–2007. [Nov 7, 2011];CDC WONDER On-line Database, compiled from Compressed Mortality File 1999–2007. 2010 Series 20 No. 2M. Accessed at http://wonder.cdc.gov/cmf-icd10.html on.
- 20.Report of the New England Regional Infant Cardiac Program. Pediatrics. 1980;65(2 Pt 2):375–461. [PubMed] [Google Scholar]
- 21.Oster ME, Strickland MJ, Mahle WT. Racial and ethnic disparities in post-operative mortality following congenital heart surgery. J Pediatr. 2011;159:222–226. doi: 10.1016/j.jpeds.2011.01.060. [DOI] [PubMed] [Google Scholar]
- 22.Bjornard K, Riehle-Colarusso T, Gilboa SM, Correa A. Patterns in the prevalence of congenital heart defects, metropolitan Atlanta, 1978 to 2005. Birth Defects Res A Clin Mol Teratol. 2013;97:87–94. doi: 10.1002/bdra.23111. [DOI] [PubMed] [Google Scholar]
- 23.Botto LD, Correa A, Erickson JD. Racial and temporal variations in the prevalence of heart defects. Pediatrics. 2001;107:E32. doi: 10.1542/peds.107.3.e32. [DOI] [PubMed] [Google Scholar]
- 24.Broussard CS, Gilboa SM, Lee KA, Oster M, Petrini JR, Honein MA. Racial/ethnic differences in infant mortality attributable to birth defects by gestational age. Pediatrics. 2012;130:e518–527. doi: 10.1542/peds.2011-3475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pillutla P, Shetty KD, Foster E. Mortality associated with adult congenital heart disease: Trends in the US population from 1979 to 2005. Am Heart J. 2009;158:874–879. doi: 10.1016/j.ahj.2009.08.014. [DOI] [PubMed] [Google Scholar]
- 26.Zomer AC, Ionescu-Ittu R, Vaartjes I, et al. Sex differences in hospital mortality in adults with congenital heart disease: the impact of reproductive health. J Am Coll Cardiol. 2013;62:58–67. doi: 10.1016/j.jacc.2013.03.056. [DOI] [PubMed] [Google Scholar]
- 27.Warnes CA, Williams RG, Bashore TM, Child JS, Connolly JM, Dearani JA, del Nido P, Fasules JW, Graham TP, Hijazi ZM, Hunt SA, King ME, Landzberg ML, Miner PD, Radford MJ, Walsh EP, Webb GD, Smith SC, Jacobs AK, Adams CD, Anderson JL, Antman EM, Buller CE, Creager MA, Ettinger SM, Halperin JL, Hunt SA, Krumholz HM, Kushner FG, Lytle BW, Nishimura RA, Page RL, Riegel B, Tarkington LG, Yancy CW American College of Cardiology American Heart Association Task Force on Practice Guidelines, American Society of Echocardiography, Heart Rhythm Society, International Society for Adult Congenital Heart Disease, Society for Cardiovascular Angiography and Interventions, Society of Thoracic Surgeons. ACC/AHA 2008 guidelines for the management of adults with congenital heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Develop Guidelines on the Management of Adults With Congenital Heart Disease). Developed in Collaboration With the American Society of Echocardiography, Heart Rhythm Society, International Society for Adult Congenital Heart Disease, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2008;52:e143–263. doi: 10.1016/j.jacc.2008.10.001. [DOI] [PubMed] [Google Scholar]
- 28.Gurvitz M, Valente AM, Broberg C, Cook S, Stout K, Kay J, Ting J, Kuehl K, Earing M, Webb G, Houser L, Opotowsky A, Harmon A, Graham D, Khairy P, Gianola A, Verstappen A, Landzberg M Alliance for Adult Research in Congenital Cardiology, Adult Congenital Heart Association. Prevalence and predictors of gaps in care among adult congenital heart disease patients: HEART-ACHD (The Health, Education, and Access Research Trial) J Am Coll Cardiol. 2013;61:2180–2184. doi: 10.1016/j.jacc.2013.02.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Patel MS, Kogon BE. Care of the adult congenital heart disease patient in the United States: a summary of the current system. Pediatr Cardiol. 2010;31:511–514. doi: 10.1007/s00246-009-9629-5. [DOI] [PubMed] [Google Scholar]
- 30.Williams RG. Transitioning youth with congenital heart disease from pediatric to adult health care. J Pediatr. 2015;166:15–19. doi: 10.1016/j.jpeds.2014.09.054. [DOI] [PubMed] [Google Scholar]
- 31.Statistics Canada. [March 22, 2016];National Household Survey. 2011 Accessed at http://www12.statcan.gc.ca/nhs-enm/2011/dp-pd/dt-td/Rp-eng.cfm?LANG=E&APATH=3&DETAIL=0&DIM=0&FL=A&FREE=0&GC=0&GID=0&GK=0&GRP=1&PID=107647&PRID=0&PTYPE=105277&S=0&SHOWALL=0&SUB=0&Temporal=2013&THEME=95&VID=0&VNAMEE=&VNAMEF= on.
- 32.Canfield MA, Mai CT, Wang Y, O’Halloran A, Marengo LK, Olney RS, Borger CL, Rutkowski R, Fornoff J, Irwin N, Copeland G, Flood TJ, Meyer RE, Rickard R, Alverson CJ, Sweatlock F, Kirby RS. The association between race/ethnicity and major birth defects in the United States, 1999–2007. Am J Public Health. 2014;104:e14–23. doi: 10.2105/AJPH.2014.302098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chamberlain LJ, Fernandes SM, Saynina O, Grady S, Sanders L, Staves K, Wise PH. Variation in Use of Pediatric Cardiology Subspecialty Care: A Total Population Study in California, 1983 to 2011. J Am Coll Cardiol. 2015;66:37–44. doi: 10.1016/j.jacc.2015.04.053. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.