This article discusses the mechanisms underlying the prothrombotic phenotype in patients with malignancy, as well as the risk factors, risk assessment, primary prevention, and treatment of cancer‐associated venous thromboembolism.
Keywords: Cancer, venous thromboembolism, diagnosis, treatment
Abstract
Venous thromboembolism (VTE), including deep‐vein thrombosis and pulmonary embolism, represents a major cause of morbidity and mortality in cancer patients. Patients with cancer are six times more likely to develop VTE than their noncancer counterparts, and VTE is the second leading cause of death in cancer patients. Despite the publication of major consensus guidelines setting out recommendations for thromboprophylaxis in cancer patients, there remains a gulf between these guidelines and clinical practice. In general, thromboprophylaxis is recommended for most patients hospitalized with active cancer. Furthermore, outpatient thromboprophylaxis may be used in carefully selected high‐risk ambulatory patients. Certain areas of controversy still remain. Although low‐molecular‐weight heparin has been shown to be superior to vitamin K antagonists in cancer patients, the role of direct oral anticoagulants is still uncertain. Moreover, recurrent thromboembolism, bleeding, and thrombocytopenia are frequently seen in cancer patients. Optimal anticoagulation in such instances presents a major challenge to clinicians. Modern computed tomography techniques have resulted in an increase in the detection of “incidental” VTE. Despite a growing body of evidence promulgating standard anticoagulant treatment in such cases, these cases present further challenges for members of the multidisciplinary team.
Implications for Practice.
This article discusses venous thromboembolism (VTE) in patients with malignancy. Practical guidance is offered on how to prevent, diagnose, and treat VTE in cancer patients. The management of “challenging” cases of VTE is also discussed.
Introduction
Since Armand Trousseau first described thrombophlebitis as a sign of visceral malignancy more than 150 years ago, the effect of cancer on blood coagulation has remained a major challenge for health care providers. Derangements in the coagulation cascade, which can manifest as thrombosis, bleeding, and disseminated intravascular coagulation, are common in patients with cancer and have significant ramifications on treatment, prognosis, and quality of life [1].
Thromboembolic events frequently complicate the course of malignancy and can involve both the venous and arterial systems. Venous thromboembolism (VTE), including deep‐vein thrombosis (DVT) and pulmonary embolism (PE), is the more common of these complications and may precede or coincide with a diagnosis of cancer. VTE can complicate surgery, hospitalizations, and systemic chemotherapy and significantly increases the cost associated with cancer treatments and hospitalizations. Patients with cancer are six times more likely to develop VTE than their noncancer counterparts and account for more than 20% of all newly diagnosed cases of VTE [2]. Indeed, VTE is the second‐leading cause of death in cancer patients after cancer itself [3]. Incidentally diagnosed VTE, commonly involving the splanchnic veins, further contributes to the thrombotic burden in patients with cancer. Arterial events, such as stroke and myocardial infarction, are also more commonly seen in patients with cancer than in the general population.
Thrombosis in cancer is associated with increased morbidity and mortality and a high economic burden. Diagnosis and management of thrombotic events interrupt essential cancer therapies and carry a risk for serious bleeding complications. Moreover, approximately 25% of patients with malignancy require readmission due to bleeding or VTE recurrence [4]. In this article, we discuss the mechanisms underlying the prothrombotic phenotype in patients with malignancy, as well as the risk factors, risk assessment, primary prevention, and treatment of cancer‐associated VTE.
Pathophysiology
Cancer cells exert a prothrombotic effect on their microenvironment through direct and indirect mechanisms, which can manifest systemically and present clinically as thrombosis. The prothrombotic state characteristic of the malignant phenotype is the result of the interplay between the factors pertaining to Virchow's triad: hemostasis results from bed rest and vascular compression by the tumor mass; vessel injury is caused by intravasation of cancer cells, intravascular devices, and systemic therapies; and hypercoagulability results from the intertwined relationship among clinical risk factors, the tumor cell, and the host response [5].
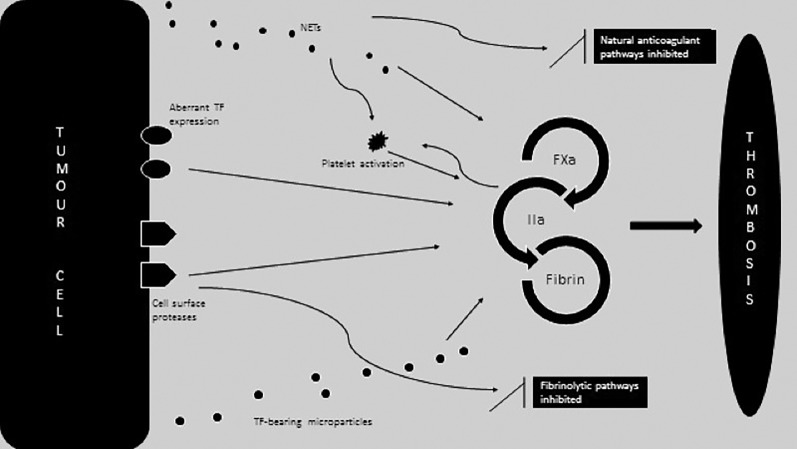
Cancer is associated with thrombin and fibrin formation directly through the release of procoagulants by neoplastic cells (e.g., tissue factor, cancer procoagulant, and, to a lesser extent, tumor mucins) and indirectly through the release of cytokines and the production of factor X‐activating cysteine proteases, mucinous glycoproteins, and circulating tissue factor‐bearing microparticles, which lead to the activation of platelets, leukocytes, and endothelial cells (Fig. 1) [6]. Furthermore, a growing body of evidence supports the idea that the same genetic mechanisms responsible for malignant transformation (including activation of oncogenes, such as RAS and PTEN, and inactivation of tumor suppressor genes, such as p53 and PTEN) drive the expression of genes controlling hemostasis [7], [8].
Figure 1.
Mechanisms underlying the cancer‐associated procoagulant state. Cancer‐mediated hypercoagulability occurs as a consequence of direct activation of procoagulant pathways by cancer cells (mediated by aberrant tumor cell TF expression, release of tumor cell‐derived, TF‐expressing microparticles, cancer procoagulant, and other cell surface proteases) or from indirect systemic effects of cancer on a variety of cell types, including leucocyte, endothelial cells, and platelets. In various malignancies, neutrophils are “primed” to release their contents in the form of NETs, resulting in direct activation of procoagulant pathways, platelet activation, and inhibition of naturally occurring anticoagulant pathways, including tissue factor pathway inhibitor. As a consequence of these various direct and indirect mechanisms, patients with cancer have an elevated risk for venous thromboembolism. Abbreviations: FXa, factor Xa; NET, neutrophil extracellular trap; TF, tissue factor.
The fibrin scaffold subsequently formed potentially confers cancer cells with a selective advantage, allowing for tumor cell anchorage and invasion [9].
A bidirectional relationship exists between cancer and thrombosis. An increased tumor burden results in a higher risk for VTE, and VTE, in turn, is a marker of tumor aggressiveness and poor prognosis. In a study from the Dutch Cancer Registry, cancer patients with VTE had a 2.2‐fold increase in mortality compared with matched cancer patients without VTE [10].
Risk Factors
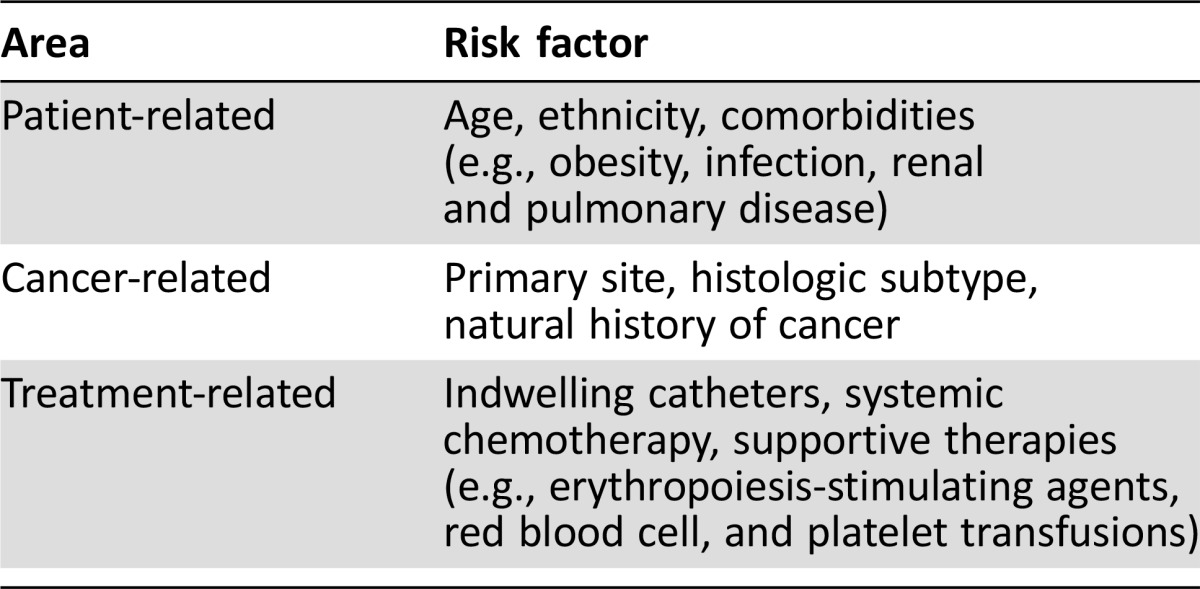
The risk for cancer‐associated VTE is highest in patients receiving systemic chemotherapy and in those hospitalized on surgical and medical floors. Clinical risk factors for VTE in patients with malignancy can be divided into patient‐related, cancer‐related, and treatment‐related risk factors (Table 1). Patient‐related risk factors include advanced age; obesity; black race; and comorbidities such as infection, anemia, and renal and pulmonary disease [11]. Conversely, Asian‐American ethnicity appears to confer some protection against VTE [12].
Table 1. Risk factors for venous thromboembolism in patients with cancer.
Furthermore, the risk for VTE varies considerably according to the primary site and histologic subtype of the cancer. The highest rates of VTE have been demonstrated in patients with primary brain tumors (47%) and pancreatic (19.2%), stomach (15.8%), and lung (13.9%) cancer [13], [14]. Those with hematologic malignancies, particularly lymphoma, are also at increased risk. The natural history of cancer also plays an important role, with studies demonstrating that patients are at greatest risk for VTE within 3 months of the initial diagnosis [15], [16], [17].
The use of systemic chemotherapy increases the risk for VTE two‐ to six‐fold [18]. The risk for VTE is also increased by the use of central catheters [19]. Furthermore, antiangiogenic agents, such as bevacizumab, a monoclonal antibody against vascular endothelial growth factor, increases the risk for both arterial and venous events, with one study demonstrating a relative risk for arterial thromboembolic events of 1.44 in patients receiving bevacizumab compared with controls (95% confidence interval [CI], 1.08–1.91; p = .013) [20], [21]. Thalidomide and lenalidomide also increase the risk for VTE when combined with dexamethasone [22], [23]. The tyrosine kinase inhibitors sunitinib and sorafenib also increase the risk for arterial thromboembolic events, suggesting a class effect for antiangiogenic therapy (relative risk [RR], 3.03; 95% CI, 1.25–7.37; p = .015) [24]. Supportive therapies, including the use of erythropoiesis‐stimulating agents, red blood cell, and platelet transfusions, further contribute to the risk for VTE [25].
Candidate biomarkers, such as P‐selectin, d‐dimers, and tissue factor, are under investigation as predictive tools in identifying patients at greater risk for events. To date, results of studies assessing the predictive utility of these biomarkers are conflicting [26], [27]. Elevated prechemotherapy leukocyte and platelet counts have also been shown to be associated with a higher incidence of VTE. One study found that VTE occurred in 4% of patients with prechemotherapy platelet counts ≥350,000 per mm3 versus 1.25% in those with prechemotherapy counts <200,000 per mm3 [28].
Risk Assessment
Current consensus guidelines discourage the use of individual risk factors in dictating clinical decision‐making. Instead, the American Society of Clinical Oncology (ASCO) guidelines, as well as the National Comprehensive Cancer Network and European Society for Medical Oncology guidelines promulgate the use of a validated risk assessment tool to identify patients at highest risk for VTE (Table 2). This risk score (the Khorana score) was initially derived from a development cohort of 2,701 patients and subsequently internally validated in an independent cohort of 1,365 patients [29]. The score has since been externally validated prospectively by the Vienna Cancer and Thrombosis Study consortium as well as numerous retrospective and prospective cohort studies [30], [31]. Risk assessment can be used for education, screening, and prophylaxis.
Table 2. Khorana score.
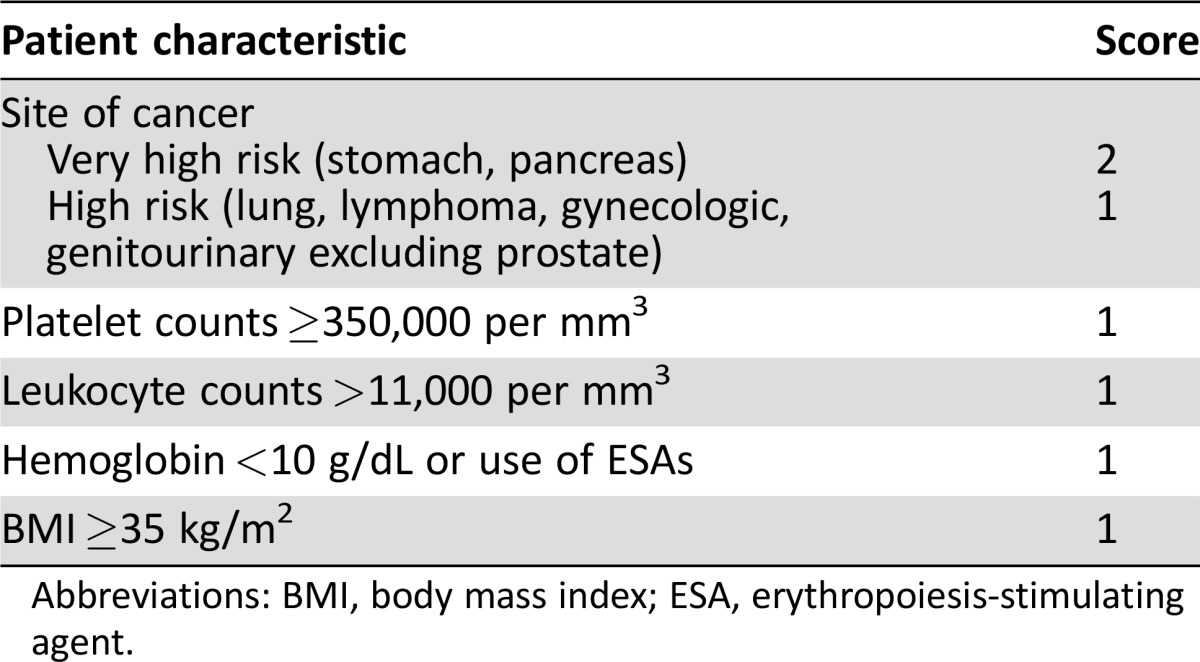
Abbreviations: BMI, body mass index; ESA, erythropoiesis‐stimulating agent.
Education
Cancer patients are woefully unaware that they are at risk for cancer‐associated thrombosis, and updated ASCO guidelines specifically call for greater education regarding this complication. An excellent demonstration of using the risk score for education has been conducted by a Canadian institution that incorporated use of the risk score into an electronic medical record [32]. Patients found to be at intermediate or high risk had a message sent to their providers to incorporate a discussion of warning signs and symptoms of VTE. Furthermore, 11% of patients in the intermediate‐ to high‐risk group developed VTE during the follow‐up period, further validating the risk score [32].
Screening
A second use of the risk score has been to identify patients with occult VTE. In a single‐institution prospective study, 35 high‐risk patients (Khorana score ≥3) were screened for VTE with monthly ultrasonography and computed tomography (CT) for restaging for 16 weeks. More than 9% of patients had occult DVT at baseline, suggesting that the Khorana score may be of value in identifying appropriate patients to screen for VTE [33]. This rate was confirmed in a multi‐institution randomized trial of prophylaxis in the same population recently reported at the American Society of Hematology meeting [34]. Current guidelines have not yet addressed these new data, but this appears to be an important future use of this or subsequent risk assessment tools.
Prophylaxis
The role of the Khorana score in identifying patients suitable for thromboprophylaxis has been demonstrated in subgroup analyses of two large randomized controlled trials (RCTs). VTE in high‐risk (Khorana score ≥3) patients enrolled in the PROTECHT trial occurred at a rate of 11.1% in the placebo arm and 4.5% in the nadroparin arm (number needed to treat [NNT], 15 vs. 77 for low‐ and intermediate‐risk patients) [35]. Similarly, in a per‐protocol subgroup analysis of SAVE‐ONCO, the NNT was 23 for high‐risk patients compared with 333 for low‐risk patients. There was no significant difference in bleeding risk between the low‐ and high‐risk patients [36]. On the basis of these findings, thromboprophylaxis may be considered in high‐risk patients. Ongoing trials are addressing the role of prophylaxis with direct oral anticoagulants in patients with Khorana score ≥2, a lower cutoff. These include CASSINI, which is using rivaroxaban, and Apixiban for the Prevention of Venous Thromboembolism in Cancer Patients (AVERT), which is using apixaban. Results are expected in 2018.
Current Guidelines
Prevention
Thromboprophylaxis in Hospitalized Patients with Cancer.
Despite the introduction of consensus guidelines encouraging thromboprophylaxis in hospitalized patients with malignancy, it remains underused in this patient population [37], [38], [39]. Indeed, one study from the United States DVT Registry found that hospitalized patients with malignancy are actually less likely to receive VTE prophylaxis than their noncancer counterparts (28% vs. 35%) [38]. However, practitioners are frequently limited by relative contraindications to pharmacologic thromboprophylaxis, including thrombocytopenia, active hemorrhage, and high risk for hemorrhage. In a recent study assessing thromboprophylaxis in hospitalized patients with cancer, 31.9% had relative contraindications to anticoagulation. Of these patients, the most common contraindication was thrombocytopenia (65.2%), followed by active hemorrhage (17.4%) [40].
There is a paucity of studies looking exclusively at thromboprophylaxis in inpatients with cancer, and most of the data on this topic has been extrapolated from randomized trials that included only a minority of cancer patients [41]. A recent subgroup analysis of three placebo‐controlled randomized trials comparing the rates of VTE events in hospitalized patients with cancer demonstrated no significant benefit of thromboprophylaxis (RR, 0.91, 95% confidence interval, 0.21–4.0; I2 = 68%), likely due to inadequate power and small sample size. Furthermore, none of these trials reported the rates of symptomatic VTE or major bleeding [41]. Given the known high incidence of VTE in this population, however, current consensus guidelines recommend the following:
Hospitalized patients with active malignancy and acute medical illness or reduced mobility should receive pharmacologic thromboprophylaxis in the absence of contraindications.
In the absence of additional risk factors, hospitalized patients with cancer may be considered for VTE prophylaxis.
There is currently insufficient evidence to support VTE prophylaxis in patients admitted for minor procedures or a short infusion of chemotherapy or those undergoing stem cell or bone marrow transplant [42].
Thromboprophylaxis in Ambulatory Patients with Cancer.
“Ambulatory” refers to the period during which a patient is not hospitalized for surgery or receiving end‐of‐life care but is in the community receiving anticancer therapy as an outpatient. One older study from the early 1990s suggested that low‐dose warfarin in women with metastatic breast cancer receiving chemotherapy was associated with an 85% relative risk reduction in the rate of VTE without a significant increase in the risk for bleeding compared with the placebo arm [43]. More recently, multiple studies have focused on prophylaxis in the ambulatory cancer population. The larger trials have enrolled broad populations, with multiple malignancies; in contrast, other investigators have taken a more focused approach on specific high‐risk malignancies, such as pancreatic cancer or myeloma. The PROTECHT (Prophylaxis Thromboembolic Events Chemotherapy) trial randomly assigned patients with metastatic or locally advanced lung, breast, gastrointestinal, ovarian, or head and neck cancer to receive daily nadroparin (3,800 U) or placebo and demonstrated a significant reduction in the composite endpoint of arterial and venous thrombosis. Rates of VTE in high‐risk patients were 11.1% in the placebo arm compared with 4.5% in the nadroparin group (NNT, 15 vs. 77 in low‐ and intermediate‐risk patients) [35].
Similarly, the SAVE‐ONCO trial randomly assigned patients with metastatic or locally advanced solid tumors commencing chemotherapy to receive either the ultralow‐molecular‐weight heparin semuloparin or placebo. Despite the relatively low incidence of VTE in the control arm (3.4%), the study demonstrated a significant reduction in the incidence of VTE in patients receiving semuloparin (1.2%), with no apparent increase in the incidence of major bleeding [44]. A per‐protocol subgroup analysis of the SAVE‐ONCO trial demonstrated an NNT of 25 for high‐risk patients compared with 333 for low‐risk patients, with no significant difference in the rates of bleeding between the two groups [36]. These studies highlight the necessity to risk‐stratify before initiating thromboprophylaxis in ambulatory cancer patients.
Although event rates in broad populations have been low, studies addressing individual tumors have demonstrated a profound benefit to anticoagulation. For instance, FRAGEM (A Phase II Randomized Study of Chemo‐Anticoagulation [Gemcitabine‐Dalteparin] Versus Chemotherapy Alone [Gemcitabine] for Locally Advanced and Metastatic Pancreatic Adenocarcinoma) focused on patients with pancreatic cancer and demonstrated a reduction in the rate of VTE from 23% in the placebo arm to 3.4% in the thromboprophylaxis arm (p = .002; NNT, 6) [45]. The PROSPECT‐CONKO 004 trial also addressed patients with pancreatic cancer and found a reduction in the rate of VTE from 9.87% to 1.25% at 3 months and from 15.13% to 5% at 12 months [46]. In patients with newly diagnosed multiple myeloma treated with lenalidomide, aspirin and low‐molecular‐weight heparin (LMWH) have demonstrated similar benefit in reducing the incidence of VTE [47]. Multiple myeloma is the only malignancy in which aspirin thromboprophylaxis is recommended. ASCO recommendations include the following:
Routine pharmacologic thromboprophylaxis is not recommended in unselected ambulatory cancer patients. However, LMWH may be considered in high‐risk outpatients with solid tumors receiving systemic chemotherapy.
Thalidomide and lenalidomide are associated with an increased risk for VTE when used in combination with dexamethasone. As a result, patients with multiple myeloma receiving either regimen should receive thromboprophylaxis with aspirin or LMWH in low‐risk patients and LMWH in high‐risk patients [42].
Thromboprophylaxis in Surgical Patients with Cancer.
Compared with non‐cancer patients, those with malignancy have a 2‐ to 3‐fold higher risk for VTE during the perioperative period [48], [49], [50]. The ideal duration of thromboprophylaxis in patients with malignancy during the perioperative period has been a source of debate. The ENOXACAN II (Enoxaparin and Cancer II) trial randomly assigned patients with cancer undergoing laparotomy to receive short‐duration (7–10 days) or extended‐duration (28 days) VTE prophylaxis with enoxaparin (40 mg, once daily). Patients underwent screening venography at the end of the study period; the frequency of thromboembolic events was decreased from 12% in the short‐duration arm to 4.8% in the extended duration group [51]. A further study compared short‐duration (7 days) and extended‐duration (27 days) prophylaxis with dalteparin in patients undergoing laparotomy and found a 55% reduction in the rates of VTE, from 16.3% in the short‐duration group to 7.3% in the extended‐duration group [52].
A recent study randomly assigned patients with colorectal cancer undergoing surgery to receive nadroparin or enoxaparin preoperatively. Despite a reduction in the rates of major bleeding in the nadroparin group, there was no significant difference in symptomatic or asymptomatic VTE occurrence [53].
A variety of mechanical thromboprophylactic methods have also been evaluated in surgical patients with cancer, but in general, they are less effective than pharmacologic prophylaxis [54], [55]. Current recommendations for thromboprophylaxis during the perioperative period include the following:
All patients with malignancy undergoing major surgical intervention should be considered for pharmacologic thromboprophylaxis with unfractionated heparin (UFH) or LMWH unless contraindications exist. VTE prophylaxis should commence preoperatively.
Moreover, it is recommended that pharmacologic thromboprophylaxis be continued for 7–10 days in all patients, with the exception of “patients undergoing major abdominal or pelvic surgery for cancer who have high‐risk features such as restricted mobility, obesity, history of VTE, or with additional risk factors,” in whom VTE prophylaxis should be continued for up to 4 weeks [42].
Treatment and Secondary Prevention
Choice and Duration of Treatment.
VTE in patients with malignancy is associated with high rates of complications, including a 12% annual risk for bleeding complications and up to a 21% annual risk of recurrence [56]. Cancer‐associated VTE has traditionally been treated with vitamin K antagonists (VKAs) despite a body of evidence suggesting that cancer‐associated VTE may be resistant to warfarin. Indeed, data from the SWIVTER (Swiss Venous Thromboembolism) and MASTER registries demonstrated that a majority of patients with cancer‐associated VTE undergo treatment with warfarin (51% and 62%, respectively) [57], [58]. LMWH has been shown to be superior to VKAs as a mainstay of treatment for VTE in cancer patients. The CLOT (Randomized Comparison of Low‐Molecular‐Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer) study randomly assigned cancer patients with VTE to receive dalteparin initially, followed by 6 months of therapeutic dalteparin or warfarin with a target international normalized ratio (INR) of 2.5. This study demonstrated an 8‐percentage point absolute risk reduction (9% vs. 17%) and a 55% relative risk reduction of recurrent VTE in patients treated with dalteparin [59]. A Cochrane review of treatment of cancer‐associated VTE further highlighted the superiority of LMWH over warfarin [60].
More recently, the CATCH (Comparison of Acute Treatments in Cancer Haemostasis) study randomly assigned 900 patients with active cancer to receive the LMWH tinzaparin at a dose of 175 IU/kg once daily for 6 months or initial tinzaparin for 5–10 days, overlapped and followed by 6 months of warfarin therapy with a target INR of 2–3. The incidence of VTE decreased from 10% in the warfarin group to 6.9% in the tinzaparin group (hazard ratio, 0.65; 95% CI, 0.41–1.03; p = .07), although it did not reach statistical significance; symptomatic DVT rates were significantly lower with tinzaparin. No significant difference in major bleeding was observed, while the rates of clinically relevant nonmajor bleeding events were significantly lower in the tinzaparin group (11% and 16%, respectively; p = .03) [61].
The ideal duration of anticoagulation has not been assessed, but there is a consensus that anticoagulation should be continued for a minimum of 6 months. Thereafter, the need for ongoing anticoagulation should be re‐evaluated by assessing risk factors, including metastatic or progressive disease and ongoing systemic chemotherapy.
The ideal duration of anticoagulation has not been assessed, but there is a consensus that anticoagulation should be continued for a minimum of 6 months. Thereafter, the need for ongoing anticoagulation should be re‐evaluated by assessing risk factors, including metastatic or progressive disease and ongoing systemic chemotherapy. In such patients, the risk for recurrent VTE is sufficiently high to justify extending treatment beyond 6 months.
Managing “Challenging” Cases of Cancer‐Associated Thrombosis.
As mentioned previously, use of thromboprophylaxis in hospitalized patients with cancer is frequently limited by contraindications to anticoagulation. The International Society of Thrombosis and Hemostasis (ISTH) recently published guidelines on the management of “challenging” cases of cancer‐associated thrombosis, including those with thrombocytopenia, those with recurrent VTE despite anticoagulation, and patients who are actively bleeding. Recurrent thrombosis is a frequently encountered problem in patients receiving anticoagulation with 10%–17% of patients treated with warfarin and 6%–9% of patients treated with LMWH experiencing recurrent VTE during follow‐up [59], [62], [63]. In patients treated with warfarin, the guidelines recommend switching to LMWH for those who develop recurrent VTE. In those treated with LMWH, it is recommended to increase the dose by 25% (or therapeutic, weight‐adjusted doses in those receiving lower doses) should they experience recurrence. Patients should be reassessed at 5–7 days. Those with symptomatic improvement should continue at the same dose, whereas those without symptomatic improvement should have their peak anti‐factor Xa level checked to dictate the dose of the next escalation [64].
Thrombocytopenia is a common reason for withholding anticoagulation in patients with cancer. The guidelines recommend that patients with a platelet count of ≥50 × 109 per L should receive full‐dose anticoagulation without a concurrent platelet transfusion. In patients with a platelet count <50 × 109 per L, platelet transfusions should be given with full therapeutic anticoagulation to achieve a platelet count >50 × 109 per L. If platelet transfusion is not possible, consideration should be given to insertion of a retrievable inferior vena cava (IVC) filter with removal of the filter and commencement of anticoagulation when the platelet count recovers [64].
Furthermore, the management of central venous port (CVP) related VTE frequently presents a conundrum for clinicians. Hohl Moinat et al. recently conducted a prospective observational cohort study of 1,097 patients undergoing CVP insertion. They defined catheter‐related VTE as DVT in the arm with or without PE, or isolated PE after CVP insertion. In this study, the incidence of CVP‐associated VTE was 5.9% at 3 months (95% CI, 4.4%–7.3%) and 11.3% (95% CI, 9.4%–13.2%) at 12 months. A high Khorana score and lung cancer were significant predictors of VTE at 3 months [65]. Recent randomized trials have not shown benefit for thromboprophylaxis in patients with central catheters and so anticoagulation is not routinely recommended.
A high Khorana score and lung cancer were significant predictors of VTE at 3 months. Recent randomized trials have not shown benefit for thromboprophylaxis in patients with central catheters and so anticoagulation is not routinely recommended.
Anticoagulation in patients with advanced disease and near the end of life represents a further challenging area. Studies informing the management of cancer‐associated thrombosis have largely excluded patients with advanced disease and a short life expectancy. Clinicians may find themselves in situations in the palliative care setting in which anticoagulation seems inappropriate. There is currently no evidence guiding anticoagulation in these patients. In a review article, Noble and Johnson advocate a pragmatic approach, suggesting that patients near the end of life with anticipated deterioration attributed to VTE may be managed with appropriate end‐of‐life medications. Conversely, they suggest that patients who are not imminently dying may benefit from anticoagulation for relief of symptoms attributable to VTE [66].
Direct Oral Anticoagulants.
The direct oral anticoagulants (DOACs), including the direct factor IIa inhibitor dabigatran and the factor Xa inhibitors apixaban, rivaroxaban, and edoxaban, are being investigated for use in cancer patients [67]. These agents offer many benefits over traditional anticoagulants, including no requirement for laboratory monitoring, feasibility of oral administration, and a reduced risk for food‐drug interactions. All three agents have received regulatory approval for the treatment of acute VTE in the general population, but there remains a paucity of data on the efficacy and safety of these agents in patients with cancer and European regulatory authorities have advised against the use of apixaban for the treatment of cancer‐associated VTE.
The EINSTEIN‐DVT study randomly assigned patients with acute DVT to receive rivaroxaban (15 mg twice daily for 3 weeks, followed by 20 mg once daily) or conventional treatment with LMWH followed by warfarin and showed similar rates of VTE recurrence and nonmajor bleeding events in the two groups. A subgroup analysis of the EINSTEIN‐DVT study exploring the safety and efficacy of rivaroxaban in patients with active malignancy demonstrated no significant difference in VTE recurrence or bleeding complications between the two groups [68]. Similarly, the RE‐COVER trial highlighted the noninferiority of dabigatran compared with standard therapy with LMWH and warfarin in patients with acute symptomatic DVT. A subgroup analysis of the RE‐COVER study looking at acute symptomatic DVT in patients with active cancer showed a nonsignificant difference in the rates of recurrence between the dabigatran and conventional therapy groups (3.1% in the dabigatran group vs. 5.3% in the standard treatment group) [69].
A major limitation of the aforementioned studies is that they used warfarin in the control arm rather than LMWH, the standard of therapy for cancer‐associated VTE. A recent meta‐analysis evaluated 9 RCTs and included 2,310 patients treated with DOACs. This analysis demonstrated a reduction in recurrent VTE in patients treated with LMWH compared with those receiving warfarin (RR, 0.52; 95% CI, 0.36–0.74). Conversely, compared with warfarin, DOACs were not associated with a significant reduction in recurrent VTE (RR, 0.66; 95% CI, 0.39–1.11). In this study, LMWH was associated with a nonsignificant increase in major bleeding (RR, 1.06; 95% CI, 0.5–2.23), whereas DOACs showed a nonsignificant decrease (RR, 0.78; 95% CI, 0.42–1.44) [70]. Overall, in light of the paucity of data demonstrating the safety and efficacy of these agents in cancer patients and lack of appropriate control arms, current published consensus guidelines do not recommend their use in patients with cancer.
Current guidelines recommend judicious use of IVC filters in patients with absolute contraindications to anticoagulation or in whom proximal clot extension is observed despite maximal LMWH therapy. One prospective randomized trial of 200 patients (including 56 with malignancy) who received IVC filters found that they conferred some short‐term protection from PE but were associated higher rates of DVT and filter‐site thrombosis compared with no filters (20.8% vs. 11.6%) [71]. IVC filters have not been shown to offer any short‐ or long‐term survival benefit with some nonrandomized cohort studies demonstrating that IVC filters are associated with increased tumor metastases and decreased survival [72]. Recommendations include the following:
LMWH rather than UFH should be used for the initial 5–10 days of anticoagulation in cancer patients with confirmed VTE.
LMWH should be continued for 6 months thereafter for secondary prevention, and anticoagulant therapy should be extended beyond the initial 6‐month period in patients with active malignancy or metastatic disease and in those still receiving systemic chemotherapy.
IVC filter placement is advisable only when absolute contraindications to anticoagulant therapy exist and may be considered where there is proximal extension of thrombosis despite maximal LMWH therapy.
Incidental VTE.
Modern CT techniques with higher resolution and sensitivity have resulted in an increase in the detection of incidental VTE. “Incidental” VTE refers to DVTs and PEs that are not clinically suspected at the time of diagnosis. As mentioned previously, incidental VTE contributes to the overall thrombotic burden in patients with cancer and accounts for up to half of all diagnosed cases of VTE in major cancer centers [73], [74]. Management of incidental VTE remains controversial. An array of retrospective studies have demonstrated similar rates of mortality and recurrent VTE between patients with incidental and symptomatic VTE [75], [76], [77]. With this in mind, recent guidelines from the ISTH advocate standard therapy for incidental VTE in patients with cancer (i.e. at least 6 months of LMWH monotherapy) [78].
Incidental subsegmental PE (SSPE) represents an area of particular controversy. A recent meta‐analysis identified that the risk for symptomatic recurrence in patients with SSPE is similar to that in patients with more proximal PE [79]. Further case‐control and cohort studies found similar rates of recurrence and survival in patients with incidental SSPE not treated with anticoagulation compared with those without PE. Rates of major bleeding were as high as 5% in those treated with anticoagulation, suggesting that routine anticoagulation might unjustifiably expose patients with SSPE to the risk for bleeding complications [80].
Conclusion
At present, there remains a gulf between consensus guidelines and clinical practice. Although implementation of a risk‐assessment tool allows for identification of high‐risk patients who will benefit most from thromboprophylaxis in the outpatient setting, many contentious issues remain. Additional studies are required to explore the safety and efficacy of the DOACs in patients with cancer. Furthermore, there remains a wide discrepancy with respect to individual clinician philosophies and treatment modalities used for incidental VTE, most prominently with respect to SSPE. In addition, the exact mechanisms underlying the prothrombotic phenotype in patients with cancer continue to be elucidated. As these mechanisms become clearer, future research should focus on manipulating these mechanisms to help predict ambulatory patients at highest risk for VTE.
Acknowledgments
Dr. Khorana is supported by the Sondra and Stephen Hardis Chair in Oncology Research and the Scott Hamilton CARES Foundation.
Glossary
Abbreviations
- FXa
factor Xa
- NET
neutrophil extracellular trap
- TF
tissue factor.
Footnotes
For Further Reading: Nicole M. Kuderer, Eva Culakova, Gary H. Lyman et al. A Validated Risk Score for Venous Thromboembolism Is Predictive of Cancer Progression and Mortality. The Oncologist 2016;21:861‐867.
Implications for Practice: The risk of venous thromboembolism (VTE) is increased in patients receiving cancer chemotherapy. In this article, the authors demonstrate that a popular risk score for VTE in patients with cancer is also associated with the risk of early mortality in this setting. It is important that clinicians evaluate the risk of VTE in patients receiving cancer treatment and discuss the risk and associated symptoms of VTE with patients. Individuals at increased risk should be advised that VTE is a medical emergency and should be urgently diagnosed and appropriately treated to reduce the risk of serious and life‐threatening complications.
Author Contributions
Conception/Design: Eoin Donnellan, Alok A. Khorana
Manuscript writing: Eoin Donnellan, Alok A. Khorana
Final approval of manuscript: Eoin Donnellan, Alok A. Khorana
Disclosures
Alok Khorana: Leo Pharma, Sanofi, Janssen, Roche, Pfizer, Bayer (C/A); Janssen, Leo, Amgen (RF). The other author indicated no financial relationships.
(C/A) Consulting/advisory relationship; (RF) Research funding; (E) Employment; (ET) Expert testimony; (H) Honoraria received; (OI) Ownership interests; (IP) Intellectual property rights/inventor/patent holder; (SAB) Scientific advisory board
References
- 1. Khorana AA. Malignancy, thrombosis and Trousseau: The case for an eponym. J Thromb Haemost 2003;1:2463–2465. [DOI] [PubMed] [Google Scholar]
- 2. Geerts WH, Bergqvist D, Pineo GF et al. Prevention of venous thromboembolism: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th edition). Chest 2008;133(suppl 6):381S–453S. [DOI] [PubMed] [Google Scholar]
- 3. Khorana AA, Francis CW, Culakova E et al. Thromboembolism is a leading cause of death in cancer patients receiving outpatient chemotherapy. J Thromb Haemost 2007;5:632–634. [DOI] [PubMed] [Google Scholar]
- 4. Bullano MF, Willey V, Hauch O et al. Longitudinal evaluation of health plan cost per venous thromboembolism or bleed event in patients with a prior venous thromboembolism event during hospitalization. J Manag Care Pharm 2005;11:663–673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Zwicker JI, Furie BC, Furie B. Cancer‐associated thrombosis. Crit Rev Oncol Hematol 2007;62:126–136. [DOI] [PubMed] [Google Scholar]
- 6. Donnellan E, Kevane B, Bird BRH et al. Cancer and venous thromboembolic disease: From molecular mechanisms to clinical management. Curr Oncol 2014;21:134–143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Rong Y, Post DE, Pieper RO et al. PTEN and hypoxia regulate tissue factor expression and plasma coagulation by glioblastoma. Cancer Res 2005;65:1406–1413. [DOI] [PubMed] [Google Scholar]
- 8. Regina S, Valentin J‐B, Lachot S et al. Increased tissue factor expression is associated with reduced survival in non–small cell lung cancer and with mutations of TP53 and PTEN. Clin Chem 2009;55:1834–1842. [DOI] [PubMed] [Google Scholar]
- 9. Boccaccio C, Comoglio PM. Genetic link between cancer and thrombosis. J Clin Oncol 2009;27:4827–4833. [DOI] [PubMed] [Google Scholar]
- 10. Sørensen HT, Mellemkjaer L, Olsen JH et al. Prognosis of cancers associated with venous thromboembolism. N Engl J Med 2000;343:1846–1850. [DOI] [PubMed] [Google Scholar]
- 11. Khorana AA, Francis CW, Culakova E et al. Frequency, risk factors, and trends for venous thromboembolism among hospitalized cancer patients. Cancer 2007;110:2339–2346. [DOI] [PubMed] [Google Scholar]
- 12. White RH, Dager WE, Zhou H et al. Racial and gender differences in the incidence of recurrent venous thromboembolism. Thromb Haemost 2006;96:267–273. [DOI] [PubMed] [Google Scholar]
- 13. Khorana AA, Dalal M, Lin J et al. Incidence and predictors of venous thromboembolism (VTE) among ambulatory high‐risk cancer patients undergoing chemotherapy in the United States. Cancer 2013;119:648–655. [DOI] [PubMed] [Google Scholar]
- 14. Petterson TM, Marks RS, Ashrani AA et al. Risk of site‐specific cancer in incident venous thromboembolism: A population‐based study. Thromb Res 2015;135:472–478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Blom JW, Doggen CJM, Osanto S et al. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA 2005;293:715–722. [DOI] [PubMed] [Google Scholar]
- 16. Sallah S, Wan JY, Nguyen NP. Venous thrombosis in patients with solid tumors: determination of frequency and characteristics. Thromb Haemost 2002;87:575–579. [PubMed] [Google Scholar]
- 17. Park LC, Woo S‐Y, Kim S et al. Incidence, risk factors and clinical features of venous thromboembolism in newly diagnosed lymphoma patients: Results from a prospective cohort study with Asian population. Thromb Res 2012;130:e6–e12. [DOI] [PubMed] [Google Scholar]
- 18. Heit JA, Silverstein MD, Mohr DN et al. Risk factors for deep vein thrombosis and pulmonary embolism: A population‐based case‐control study. Arch Intern Med 2000;160:809–815. [DOI] [PubMed] [Google Scholar]
- 19. Verso M, Agnelli G. Venous thromboembolism associated with long‐term use of central venous catheters in cancer patients. J Clin Oncol 2003;21:3665–3675. [DOI] [PubMed] [Google Scholar]
- 20. Nalluri SR, Chu D, Keresztes R et al. Risk of venous thromboembolism with the angiogenesis inhibitor bevacizumab in cancer patients: A meta‐analysis. JAMA 2008;300:2277–2285. [DOI] [PubMed] [Google Scholar]
- 21. Ranpura V, Hapani S, Chuang J et al. Risk of cardiac ischemia and arterial thromboembolic events with the angiogenesis inhibitor bevacizumab in cancer patients: A meta‐analysis of randomized controlled trials. Acta Oncol 2010;49:287–297. [DOI] [PubMed] [Google Scholar]
- 22. Knight R, DeLap RJ, Zeldis JB. Lenalidomide and venous thrombosis in multiple myeloma. N Engl J Med 2006;354:2079–2080. [DOI] [PubMed] [Google Scholar]
- 23. Zangari M, Anaissie E, Barlogie B et al. Increased risk of deep‐vein thrombosis in patients with multiple myeloma receiving thalidomide and chemotherapy. Blood 2001;98:1614–1615. [DOI] [PubMed] [Google Scholar]
- 24. Choueiri TK, Schutz FAB, Je Y et al. Risk of arterial thromboembolic events with sunitinib and sorafenib: A systematic review and meta‐analysis of clinical trials. J Clin Oncol 2010;28:2280–2285. [DOI] [PubMed] [Google Scholar]
- 25. Khorana AA, Francis CW, Blumberg N et al. Blood transfusions, thrombosis, and mortality in hospitalized patients with cancer. Arch Intern Med 2008;168:2377–2381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Thaler J, Ay C, Mackman N et al. Microparticle‐associated tissue factor activity, venous thromboembolism and mortality in pancreatic, gastric, colorectal and brain cancer patients. J Thromb Haemost 2012;10:1363–1370. [DOI] [PubMed] [Google Scholar]
- 27. Zwicker JI, Liebman HA, Neuberg D et al. Tumor‐derived tissue factor–bearing microparticles are associated with venous thromboembolic events in malignancy. Clin Cancer Res 2009;15:6830–6840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Khorana AA, Francis CW, Culakova E et al. Risk factors for chemotherapy‐associated venous thromboembolism in a prospective observational study. Cancer 2005;104:2822–2829. [DOI] [PubMed] [Google Scholar]
- 29. Khorana AA, Kuderer NM, Culakova E et al. Development and validation of a predictive model for chemotherapy‐associated thrombosis. Blood 2008;111:4902–4907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Khorana AA, McCrae KR. Risk stratification strategies for cancer‐associated thrombosis: An update. Thromb Res 2014;133 Suppl 2:S35–S38. [DOI] [PubMed] [Google Scholar]
- 31. Ay C, Dunkler D, Marosi C et al. Prediction of venous thromboembolism in cancer patients. Blood 2010;116:5377–5382. [DOI] [PubMed] [Google Scholar]
- 32. Lustig DB, Rodriguez R, Wells PS. Implementation and validation of a risk stratification method at The Ottawa Hospital to guide thromboprophylaxis in ambulatory cancer patients at intermediate‐high risk for venous thrombosis. Thromb Res 2015;136:1099–1102. [DOI] [PubMed] [Google Scholar]
- 33. Khorana AA, Rubens D, Francis CW. Screening high‐risk cancer patients for VTE: A prospective observational study. Thromb Res 2014;134:1205–1207. [DOI] [PubMed] [Google Scholar]
- 34. Khorana AA, Francis CW, Kuderer NM, et al. Dalteparin thromboprophylaxis in cancer patients at high risk for venous thromboembolism: A randomized trial. Oral presentation at: 57th American Society of Hematology (ASH) Annual Meeting & Exposition, December 5–8, 2015; Orlando, FL.
- 35. Verso M, Agnelli G, Barni S et al. A modified Khorana risk assessment score for venous thromboembolism in cancer patients receiving chemotherapy: The Protecht score. Intern Emerg Med 2012;7:291–292. [DOI] [PubMed] [Google Scholar]
- 36. George D, Agnelli G, Fisher W et al. Venous thromboembolism (VTE) prevention with semuloparin in cancer patients initiating chemotherapy: Benefit‐risk assessment by VTE risk in SAVE‐ONCO. Blood 2011;118:206a. [Google Scholar]
- 37. Amin A, Stemkowski S, Lin J et al. Thromboprophylaxis rates in US medical centers: Success or failure? J Thromb Haemost 2007;5:1610–1616. [DOI] [PubMed] [Google Scholar]
- 38. Burleigh E, Wang C, Foster D et al. Thromboprophylaxis in medically ill patients at risk for venous thromboembolism. Am J Health Syst Pharm 2006;63(suppl 6):S23–S29. [DOI] [PubMed] [Google Scholar]
- 39. Kahn SR, Panju A, Geerts W et al. Multicenter evaluation of the use of venous thromboembolism prophylaxis in acutely ill medical patients in Canada. Thromb Res 2007;119:145–155. [DOI] [PubMed] [Google Scholar]
- 40. Zwicker JI, Rojan A, Campigotto F et al. Pattern of frequent but nontargeted pharmacologic thromboprophylaxis for hospitalized patients with cancer at academic medical centers: A prospective, cross‐sectional, multicenter study. J Clin Oncol 2014;32:1792–1796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Carrier M, Khorana AA, Moretto P et al. Lack of evidence to support thromboprophylaxis in hospitalized medical patients with cancer. Am J Med 2014;127:82–86.e1. [DOI] [PubMed] [Google Scholar]
- 42. Lyman GH, Khorana AA, Kuderer NM et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol 2013;31:2189–2204. [DOI] [PubMed] [Google Scholar]
- 43. Levine M, Hirsh J, Arnold A et al. Double‐blind randomised trial of very‐low‐dose warfarin for prevention of thromboembolism in stage IV breast cancer. Lancet 1994;343:886–889. [DOI] [PubMed] [Google Scholar]
- 44. Agnelli G, George DJ, Kakkar AK et al. Semuloparin for thromboprophylaxis in patients receiving chemotherapy for cancer. N Engl J Med 2012;366:601–609. [DOI] [PubMed] [Google Scholar]
- 45. Maraveyas A, Waters J, Roy R et al. Gemcitabine versus gemcitabine plus dalteparin thromboprophylaxis in pancreatic cancer. Eur J Cancer 2012;48:1283–1292. [DOI] [PubMed] [Google Scholar]
- 46. Riess H, Pelzer U, Hilbig A et al. Rationale and design of PROSPECT‐CONKO 004: A prospective, randomized trial of simultaneous pancreatic cancer treatment with enoxaparin and chemotherapy). BMC Cancer 2008;8:361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Larocca A, Cavallo F, Bringhen S et al. Aspirin or enoxaparin thromboprophylaxis for patients with newly diagnosed multiple myeloma treated with lenalidomide. Blood 2012;119:933–939; quiz 1093. [DOI] [PubMed] [Google Scholar]
- 48. White RH, Zhou H, Romano PS. Incidence of symptomatic venous thromboembolism after different elective or urgent surgical procedures. Thromb Haemost 2003;90:446–455. [DOI] [PubMed] [Google Scholar]
- 49. Huber O, Bounameaux H, Borst F, Rohner A. Postoperative pulmonary embolism after hospital discharge. An underestimated risk. Arch Surg 1992;127:310–313. [DOI] [PubMed] [Google Scholar]
- 50. Agnelli G, Bolis G, Capussotti L et al. A clinical outcome‐based prospective study on venous thromboembolism after cancer surgery: The @RISTOS project. Ann Surg 2006;243:89–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Bergqvist D, Agnelli G, Cohen AT et al. Duration of prophylaxis against venous thromboembolism with enoxaparin after surgery for cancer. N Engl J Med 2002;346:975–980. [DOI] [PubMed] [Google Scholar]
- 52. Rasmussen MS, Jorgensen LN, Wille‐Jørgensen P et al. Prolonged prophylaxis with dalteparin to prevent late thromboembolic complications in patients undergoing major abdominal surgery: A multicenter randomized open‐label study. J Thromb Haemost 2006;4:2384–2390. [DOI] [PubMed] [Google Scholar]
- 53. Simonneau G, Laporte S, Mismetti P et al. A randomized study comparing the efficacy and safety of nadroparin 2850 IU (0.3 mL) vs. enoxaparin 4000 IU (40 mg) in the prevention of venous thromboembolism after colorectal surgery for cancer. J Thromb Haemost 2006;4:1693–1700. [DOI] [PubMed] [Google Scholar]
- 54. Roderick P, Ferris G, Wilson K et al. Towards evidence‐based guidelines for the prevention of venous thromboembolism: Systematic reviews of mechanical methods, oral anticoagulation, dextran and regional anaesthesia as thromboprophylaxis. Health Technol Assess 2005;9:iii–iv, ix–x, 1–78. [DOI] [PubMed] [Google Scholar]
- 55.Cardiovascular Disease Educational and Research Trust , Cyprus Cardiovascular Disease Educational and Research Trust, European Venous Forum, International Surgical Thrombosis Forum, International Union of Angiology, Union Internationale de Phlébologie. Prevention and treatment of venous thromboembolism. International Consensus Statement (guidelines according to scientific evidence). Int Angiol 2006;25:101–161. [PubMed] [Google Scholar]
- 56. Prandoni P, Lensing AWA, Piccioli A et al. Recurrent venous thromboembolism and bleeding complications during anticoagulant treatment in patients with cancer and venous thrombosis. Blood 2002;100:3484–3488. [DOI] [PubMed] [Google Scholar]
- 57. Spirk D, Ugi J, Korte W et al. Long‐term anticoagulation treatment for acute venous thromboembolism in patients with and without cancer. The SWIss Venous ThromboEmbolism Registry (SWIVTER) II. Thromb Haemost 2011;105:962–967. [DOI] [PubMed] [Google Scholar]
- 58. Imberti D, Agnelli G, Ageno W et al. Clinical characteristics and management of cancer‐associated acute venous thromboembolism: Findings from the MASTER Registry. Haematologica 2008;93:273–278. [DOI] [PubMed] [Google Scholar]
- 59. Lee AYY, Levine MN, Baker RI et al. Low‐molecular‐weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med 2003;349:146–153. [DOI] [PubMed] [Google Scholar]
- 60. Akl EA, Barba M, Rohilla S et al. Low‐molecular‐weight heparins are superior to vitamin K antagonists for the long term treatment of venous thromboembolism in patients with cancer: A Cochrane systematic review. J Exp Clin Cancer Res 2008;27:21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Lee AYY, Bauersachs R, Janas MS et al. CATCH: A randomised clinical trial comparing long‐term tinzaparin versus warfarin for treatment of acute venous thromboembolism in cancer patients. BMC Cancer 2013;13:284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Hull RD, Pineo GF, Brant RF et al. Long‐term low‐molecular‐weight heparin versus usual care in proximal‐vein thrombosis patients with cancer. Am J Med 2006;119:1062–1072. [DOI] [PubMed] [Google Scholar]
- 63. Meyer G, Marjanovic Z, Valcke J et al. Comparison of low‐molecular‐weight heparin and warfarin for the secondary prevention of venous thromboembolism in patients with cancer: A randomized controlled study. Arch Intern Med 2002;162:1729–1735. [DOI] [PubMed] [Google Scholar]
- 64. Oo TH. Management of challenging cases of patients with cancer‐associated thrombosis including recurrent thrombosis and bleeding: guidance from the SSC of the ISTH: A rebuttal. J Thromb Haemost 2014;12:115–116. [DOI] [PubMed] [Google Scholar]
- 65. Hohl Moinat C, D Périard, A Grueber et al. Predictors of venous thromboembolic events associated with central venous port insertion in cancer patients. J Oncol, doi: 10.1155/2014/743181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Noble S, Johnson MJ. Management of cancer‐associated thrombosis in people with advanced disease. BMJ Support Palliat Care 2012;2:163–167. [DOI] [PubMed] [Google Scholar]
- 67. Levine MN. New antithrombotic drugs: potential for use in oncology. J Clin Oncol 2009;27:4912–4918. [DOI] [PubMed] [Google Scholar]
- 68. Agnelli G, Berkowitz S, Bounameaux H et al. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med 2010;363:2499–2510. [DOI] [PubMed] [Google Scholar]
- 69. Schulman S, Kearon C, Kakkar AK et al. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med 2009;361:2342–2352. [DOI] [PubMed] [Google Scholar]
- 70. Carrier M, Cameron C, Delluc A et al. Efficacy and safety of anticoagulant therapy for the treatment of acute cancer‐associated thrombosis: A systematic review and meta‐analysis. Thromb Res 2014;134:1214–1219. [DOI] [PubMed] [Google Scholar]
- 71.Eight‐year follow‐up of patients with permanent vena cava filters in the prevention of pulmonary embolism : The PREPIC (Prevention du Risque d'Embolie Pulmonaire par Interruption Cave) Randomized Study. Circulation 2005;112:416–422. [DOI] [PubMed] [Google Scholar]
- 72. Matsuo K, Carter CM, Ahn EH et al. Inferior vena cava filter placement and risk of hematogenous distant metastasis in ovarian cancer. Am J Clin Oncol 2013;36:362–367. [DOI] [PubMed] [Google Scholar]
- 73. van Es N, Bleker SM, Di Nisio M. Cancer‐associated unsuspected pulmonary embolism. Thromb Res 2014;(suppl 2):S172–S178. [DOI] [PubMed] [Google Scholar]
- 74. Sun J‐M, Kim TS, Lee J et al. Unsuspected pulmonary emboli in lung cancer patients: The impact on survival and the significance of anticoagulation therapy. Lung Cancer 2010;69:330–336. [DOI] [PubMed] [Google Scholar]
- 75. den Exter PL, Hooijer J, Dekkers OM, Huisman MV. Risk of recurrent venous thromboembolism and mortality in patients with cancer incidentally diagnosed with pulmonary embolism: A comparison with symptomatic patients. J Clin Oncol 2011;29:2405–2409. [DOI] [PubMed] [Google Scholar]
- 76. Menapace LA, Peterson DR, Berry A et al. Symptomatic and incidental thromboembolism are both associated with mortality in pancreatic cancer. Thromb Haemost 2011;106:371–378. [DOI] [PubMed] [Google Scholar]
- 77. O'Connell C, Razavi P, Ghalichi M et al. Unsuspected pulmonary emboli adversely impact survival in patients with cancer undergoing routine staging multi‐row detector computed tomography scanning. J Thromb Haemost 2011;9:305–311. [DOI] [PubMed] [Google Scholar]
- 78. Di Nisio M, Lee AYY, Carrier M et al. Diagnosis and treatment of incidental venous thromboembolism in cancer patients: Guidance from the SSC of the ISTH. J Thromb Haemost 2015;13:880–883. [DOI] [PubMed] [Google Scholar]
- 79. van der Hulle T, den Exter PL, Meyer G et al. Risk of recurrent venous thromboembolism and major bleeding in cancer‐associated incidental pulmonary embolism amongst treated and untreated patients: A pooled analysis of 926 patients. Blood 2014;124:590–590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Donato AA, Khoche S, Santora J et al. Clinical outcomes in patients with isolated subsegmental pulmonary emboli diagnosed by multidetector CT pulmonary angiography. Thromb Res 2010;126:e266–e270. [DOI] [PubMed] [Google Scholar]