Abstract
Importance
Prophylaxis and treatment guidelines for infective endocarditis have changed substantially over the past decade. In the United States, few population-based studies have explored the contemporary epidemiology and outcomes of endocarditis.
Objective
To quantify trends in the incidence and etiologies of infective endocarditis in the United States.
Design, Setting, and Participants
Retrospective population epidemiology study of patients hospitalized with a first episode of endocarditis identified from mandatory state databases in California and New York State between January 1, 1998, and December 31, 2013.
Exposure
Infective endocarditis.
Main Outcomes and Measures
Outcomes were crude and standardized incidence of endocarditis and trends in patient characteristics and disease etiology. Trends in acquisition mode, organism, and mortality were analyzed.
Results
Among 75 829 patients with first episodes of endocarditis (mean [SD] age, 62.3 [18.9] years; 59.1% male), the standardized annual incidence was stable between 7.6 (95% CI, 7.4 to 7.9) and 7.8 (95% CI, 7.6 to 8.0) cases per 100 000 persons (annual percentage change [APC], −0.06%; 95% CI, −0.3% to 0.2%; P = .59). From 1998 through 2013, the proportion of patients with native-valve endocarditis decreased (from 74.5% to 68.4%; APC, −0.7%; 95% CI, −0.9% to −0.5%; P < .001). Prosthetic-valve endocarditis increased (from 12.0% to 13.8%; APC, 1.3%; 95% CI, 0.8% to 1.7%; P < .001), and cardiac device–related endocarditis increased (from 1.3% to 4.1%; APC, 8.8%; 95% CI, 7.8% to 9.9%; P < .001). The proportion of patients with health care–associated nosocomial endocarditis decreased (from 17.7% to 15.3%; APC, −1.0%; 95% CI, −1.4% to −0.7%; P < .001). The proportion of patients with health care–associated nonnosocomial endocarditis increased (from 32.1% to 35.9%; APC, 0.8%; 95% CI, 0.5% to 1.1%; P < .001). The incidence of oral streptococcal endocarditis did not increase (unadjusted: APC, −0.1%; 95% CI, −0.8% to 0.6%; P = .77; adjusted: APC, −1.3%; 95% CI, −1.8% to −0.7%; P < .001). Crude 90-day mortality was unchanged (from 23.9% to 24.2%; APC, −0.3%; 95% CI, −1.0% to 0.4%; P = .44); adjusted risk of 90-day mortality decreased (adjusted hazard ratio per year, 0.982; 95% CI, 0.978 to 0.986; P < .001).
Conclusions and Relevance
In California and New York State, the overall standardized incidence of infective endocarditis was stable from 1998 through 2013, with changes in patient characteristics and etiology over this time.
Key Points
Question
What have been the trends in incidence, etiology, and mortality of infective endocarditis since 1998?
Findings
In a population epidemiology study of 75 829 inpatients with a first episode of infective endocarditis in California and New York State from 1998 through 2013, standardized incidence remained stable between 7.6 and 7.8 cases per 100 000 persons annually, with a decrease in adjusted mortality risk of 2% per year. There were shifts in patterns of patient characteristics and etiology.
Meaning
The pattern of infective endocarditis has evolved since 1998.
This population epidemiology study describes trends in incidence and etiologies of infective endocarditis in California and New York State from 1998 through 2013.
Introduction
Infective endocarditis is an uncommon illness associated with substantial morbidity and mortality. The annual incidence in developed countries was between 3 and 9 cases per 100 000 persons between 1970 and 2013, early mortality is approximately 30%, and systemic emboli, stroke, organ failure, and need for valve surgery are common. The approach to prophylaxis and therapy has changed substantially during the last decade. Most notably, consensus guidelines for antibiotic prophylaxis have become progressively more restrictive, with recommendations for infective endocarditis prophylaxis increasingly reserved for the patients with highest risk of adverse outcomes.
In the United States, recommendations for infective endocarditis prophylaxis underwent a major revision in 2007, incorporating a shift from routine antibiotic prophylaxis for patients with structural valve disease undergoing invasive procedures to prophylaxis restricted to patients undergoing specific dental procedures and with a history of valve replacement or repair, prior infective endocarditis, uncorrected congenital cyanotic heart lesions, or cardiac transplantation with valvulopathy. The effect of these changes on the incidence and outcomes of endocarditis is unknown. The increased incidence and mortality of infective endocarditis reported in several analyses after the guideline changes have not been confirmed by others. To better define trends in the epidemiology and outcomes of infective endocarditis in the United States, this population-based study used mandatory databases tracking information on all patients across all hospitalizations in California and New York State from 1998 through 2013.
Methods
Study Design
Patients with infective endocarditis were identified using International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnosis codes in the Statewide Planning and Research Cooperative System database in New York and the Office of Statewide Health Planning and Development database in California. Both are all-payer, administrative databases that prospectively collect data on every hospital discharge, ambulatory surgery, and emergency department visit in their respective states. In these databases, each patient is allocated a unique identifier linking all encounters, permitting longitudinal analysis. Patients were included in the study if they had a first episode of infective endocarditis identified by either a primary or secondary diagnostic ICD-9-CM code 421.0, 421.1, 421.9, 036.42, 098.84, 112.81, 115.04, 115.14, or 115.94 between January 1, 1998, and December 31, 2013. Hospital admissions without a unique identifier as well as readmissions were excluded from the analysis (eFigure 1 in the Supplement).
Validation of ICD-9-CM coding for infective endocarditis, which preceded the final step of analytic data set creation, was performed using patients’ electronic medical records identified from the Mount Sinai Data Warehouse. Hospitalizations with any possibility of infective endocarditis were identified either by keyword search (“endocarditis” or “vegetation”) in any note from patients’ medical records or by broader ICD-9-CM codes (421.0, 421.1, 421.9, 112.81, 036.42, 098.84, 115.04, 115.14, 115.94, 424.90, 424.91, and 424.99) in hospital inpatient discharge data at Mount Sinai Medical Center, New York, New York, from January 1, 2013, through December 31, 2014. We reviewed those hospitalizations (n = 1673), identified 515 hospitalizations and 283 patients with any type of infective endocarditis, and evaluated reliability of the ICD-9-CM codes for identification of infective endocarditis. The sensitivity, specificity, and positive predictive value of the ICD-9-CM codes for acute infective endocarditis defined by the modified Duke criteria were 94% (95% CI, 92%-97%), 99% (95% CI, 99%-99%), and 94% (95% CI, 91%-97%), respectively (eTable 1A in the Supplement). To confirm generalizability of the positive predictive value in different hospital settings and different time frames, further code validation was conducted at 5 other facilities (4 urban settings and 1 rural setting; 2 academic medical centers [including 1 tertiary referral center] and 3 community hospitals) from January 1, 2013, through December 31, 2014, as well as hospitalizations at Mount Sinai Medical Center from January 1, 2003, through December 31, 2005 (eTable 1B in the Supplement). Baseline comorbidities were identified using diagnosis and procedure codes from the index hospitalization and previous hospitalizations (eTable 2 in the Supplement).
This study was approved by the Data Protection Review Board of the New York State Department of Health, the Committee for the Protection of Human Subjects of the State of California, and the Program for Protection of Human Subjects at the Icahn School of Medicine, Mount Sinai Medical Center. The approval included a waiver of informed consent.
Definitions
Infective endocarditis was categorized based on disease types as native-valve endocarditis, prosthetic-valve endocarditis, cardiac device–related endocarditis, or drug abuse–related endocarditis. The ICD-9-CM codes and definitions used are listed in eTable 3 in the Supplement, and the hierarchical allocation algorithm is shown in eFigure 2A in the Supplement.
The mode of acquisition was categorized into non–health care–associated infective endocarditis (namely community-acquired infective endocarditis) or health care–associated infective endocarditis. Health care–associated infective endocarditis was further categorized into nosocomial or nonnosocomial infective endocarditis. The ICD-9-CM codes used are listed in eTable 4 in the Supplement, and the hierarchical allocation algorithm is shown in eFigure 2B in the Supplement. Nonnosocomial infective endocarditis was defined as infective endocarditis that was present on admission in patients with health care contact before the index hospitalization. Health care contact was identified as a history of receiving intravenous therapy (including chemotherapy), transfer from a specialized nursing care facility, hemodialysis, or hospitalization for 2 days or longer in the 90 days before the index admission. Community-acquired infective endocarditis was defined as infective endocarditis that was present on admission in patients without prior health care contact. When patients were transferred from another hospital, they were classified as having nosocomial infective endocarditis if the length of stay in the previous admission was 2 days or longer; they were classified as having nonnosocomial infective endocarditis if their length of stay was shorter than 2 days and there was evidence of heath care contact before the first admission. Nosocomial infective endocarditis was defined as infective endocarditis that was not present on admission if there was no transfer from another hospital. The study definitions of disease type and mode of acquisition were validated against medical records, with positive predictive values of 87% (95% CI, 81%-92%) and 83% (95% CI, 77%-89%), respectively.
Causal microorganisms were identified using primary and secondary diagnostic codes within the limitation of ICD-9-CM, as follows: Staphylococcus aureus (including methicillin-resistant species), other Staphylococcus species, Streptococcus species, gram-negative bacilli, fungus, and unknown (which included culture-negative and uncoded cases). Among the entire cohort, 75% of patients had a causative microorganism coded. Specific ICD-9-CM codes do not exist for oral streptococci; therefore, ICD-9-CM codes primarily identifying nonpneumococcal, non-β-hemolytic, and nonenterococcal streptococci were used (eTable 5 in the Supplement). The causal microorganisms identified in the state database were validated against medical records, with a positive predictive value of 88% (95% CI, 83%-94%). For oral streptococci, the positive predictive value was 84% (95% CI, 68%-100%).
Statistical Analysis
The crude incidence of infective endocarditis was calculated by dividing the number of patients with the first episodes of infective endocarditis in each year by the California and New York census populations in the same year, and it is reported per 100 000 persons. Direct standardization was used to account for changes in age, sex, and race in the California and New York population during the study period, using the population in 1998 as the reference. Multivariable Poisson regression analysis was performed to evaluate temporal trends in the incidence of infective endocarditis, adjusting for age, sex, and race. Trends in incidence were assessed by the annual percentage change (APC) with 95% confidence interval. Interrupted time series analysis was also performed using segmented regression to assess the association with prevention guideline changes released in April 2007 and the incidence of infective endocarditis. Analyses were repeated with change points set at April 2007, October 2007, and April 2008, accounting for the possible time lag of guideline change to be effective in practice and also excluding data during 6-month or 1-year lag periods. Crude incidence trend, direct standardization, multivariable Poisson regression, and interrupted time series analysis with segmented regression analyses were all performed for both all infective endocarditis cases and oral streptococcal infective endocarditis cases.
For descriptive analysis, continuous variables are reported as means with standard deviations, and categorical variables are expressed as proportions per the total number of infective endocarditis cases. Trends in patient age were evaluated with the ordinary least squares method, and trends in proportion are presented as APC with 95% confidence interval. Log-linear Poisson regression was used to estimate the APC where the offset was a logarithm of the denominator. To evaluate the trend in 90-day mortality during the study period and predictors of 5-year mortality, multivariable Cox regression was performed, adjusting for age, sex, race, baseline comorbidities (hypertension, diabetes, chronic kidney disease, coronary artery disease, peripheral vascular disease, chronic obstructive pulmonary disease, liver disease, history of malignancy, and history of congenital heart disease), disease types, acquisition mode, organism, and admission year. For 90-day and 5-year mortality, the study cohort from January 1, 1998, through December 31, 2011, was used. Survival curves were drawn using the Kaplan-Meier method and compared using the log-rank test with Sidak adjustment for multiple comparisons. Deaths in New York were ascertained using a full Social Security Death Master File (last follow-up was May 29, 2015) as well as deceased discharge disposition at the subsequent inpatient and emergency department visits. Deaths in California were ascertained from linked vital statistics data (last available follow-up was December 31, 2011).
There were no missing values for age, sex, race, admission dates, or discharge dates. The 2% of the study cohort for whom race was categorized as unknown were included in the category of other. To evaluate generalizability of the results to the entire US population, the demographic characteristics and baseline comorbidities of patients with infective endocarditis identified in the Nationwide Inpatient Sample were compared between the California and New York population and the entire US population (eAppendix in the Supplement). Additionally, a sensitivity analysis was conducted to assess consistency of the findings across different geographical locations in the United States. Analyses for trends in incidence and 90-day mortality were repeated stratified by state (sensitivity analysis for 90-day mortality had a different last follow-up in each state). All tests were 2-tailed, and an α level of .05 was considered statistically significant. All statistical analyses were performed using SAS version 9.4 statistical software (SAS Institute Inc).
Results
Incidence
Between January 1, 1998, and December 31, 2013, 75 829 patients with a first episode of infective endocarditis were identified (43 114 [56.9%] in California and 32 715 [43.1%] in New York; mean [SD] age, 62.3 [18.9] years; 59.1% male). A total of 7748 patients (10.2%) were transferred from another hospital. The crude incidence increased from 7.6 to 9.3 cases per 100 000 persons annually (APC, 1.1%; 95% CI, 0.8% to 1.4%; unadjusted Poisson regression P < .001) (eFigure 3A in the Supplement; crude incidence stratified by age, sex, and race is shown in eFigure 3B-D in the Supplement). The incidence was significantly higher in male, nonwhite, and elderly patients (eFigure 4 in the Supplement). After adjusting for age, sex, and race, there was no significant increase in the incidence of endocarditis over time (range, 7.6 [95% CI, 7.4 to 7.9] to 7.8 [95% CI, 7.6 to 8.0] cases per 100 000 persons annually; APC, −0.06%; 95% CI, −0.3% to 0.2%; multivariable Poisson regression P = .59) (Figure 1). Segmented regression did not show an increase in slope of incidence after the guideline changes (eFigure 5A and eTable 6 in the Supplement). A sensitivity analysis was conducted in each state and confirmed no significant increase in the standardized incidence or in the slope of incidence after the guideline changes (eFigure 5B and C and eFigure 6 in the Supplement).
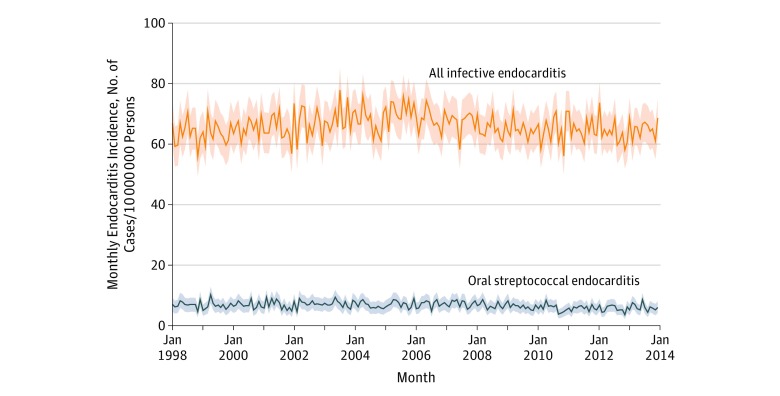
Figure 1. Standardized Incidence of Infective Endocarditis in California and New York State From 1998 Through 2013.
Direct standardization was performed to account for changes in age, sex, and race in the California and New York State census population during the study period, using the population in 1998 as the reference. For all infective endocarditis cases, the annual percentage change was −0.06% (95% CI, −0.3% to 0.2%; P = .59); for oral streptococcal endocarditis cases, the annual percentage change was −1.3% (95% CI, −1.8% to −0.7%; P < .001). P values were calculated using multivariable Poisson regression and were 2-tailed. The shaded regions indicate 95% confidence intervals.
Patient Characteristics
Patient characteristics in California and New York were similar to those of the entire US population (eTable 7 in the Supplement). Patients diagnosed with infective endocarditis in the latter part of the study period were older, more likely to be male, and more likely to have chronic obstructive pulmonary disease, cancer, and liver disease compared with patients in the earlier part of the study period (Table). From 1998 through 2013 among the patients with endocarditis, the proportion of patients with a history of valve surgery increased from 12.8% to 15.2% (APC, 1.6%; 95% CI, 1.2% to 2.0%; P < .001), and the proportion of patients with implanted pacemakers or defibrillators increased from 8.8% to 15.6% (APC, 4.8%; 95% CI, 4.3% to 5.2%; P < .001). As a result, the proportion of patients with native-valve endocarditis decreased from 74.5% to 68.4% (APC, −0.7%; 95% CI, −0.9% to −0.5%; P < .001), whereas prosthetic-valve endocarditis increased from 12.0% to 13.8% (APC, 1.3%; 95% CI, 0.8% to 1.7%; P < .001) and cardiac device–related endocarditis increased from 1.3% to 4.1% (APC, 8.8%; 95% CI, 7.8% to 9.9%; P < .001) (Table).
Table. Patient Characteristics in California and New York State From 1998 Through 2013, Overall and Trends by Years.
| Characteristic | No. (%) | Annual Percentage Change, % (95% CI) | P Valuea | ||||
|---|---|---|---|---|---|---|---|
| Overall (N = 75 829) |
1998-2001 (n = 16 511) |
2002-2005 (n = 18 887) |
2006-2009 (n = 19 611) |
2010-2013 (n = 20 820) |
|||
| Demographic | |||||||
| Age, mean (SD), y | 62.3 (18.9) | 61.0 (19.4) | 61.9 (19.0) | 63.0 (18.5) | 63.3 (18.5) | <.001b | |
| Male | 44 804 (59.1) | 9550 (57.8) | 10 971 (58.1) | 11 672 (59.5) | 12 611 (60.6) | 0.5 (0.2 to 0.7) | <.001 |
| Race | |||||||
| White | 52 776 (69.6) | 11 750 (71.2) | 13 262 (70.2) | 13 534 (69.0) | 14 230 (68.3) | −0.3 (−0.5 to −0.2) | <.001 |
| Black | 10 074 (13.3) | 2189 (13.3) | 2547 (13.5) | 2714 (13.8) | 2624 (12.6) | −0.4 (−0.8 to 0.03) | .07 |
| Other | 12 979 (17.1) | 2572 (15.6) | 3078 (16.3) | 3363 (17.1) | 3966 (19.0) | 1.7 (1.3 to 2.1) | <.001 |
| Comorbidities | |||||||
| Hypertension | 47 604 (62.8) | 8176 (49.5) | 11 495 (60.9) | 13 189 (67.3) | 14 744 (70.8) | 2.8 (2.6 to 3.0) | <.001 |
| Complicated diabetes | 11 165 (14.7) | 1921 (11.6) | 2632 (13.9) | 3097 (15.8) | 3515 (16.9) | 3.0 (2.6 to 3.4) | <.001 |
| Coronary artery disease | 30 326 (40.0) | 5589 (33.9) | 7433 (39.4) | 8303 (42.3) | 9001 (43.2) | 2.0 (1.7 to 2.2) | <.001 |
| Peripheral vascular disease | 11 763 (15.5) | 2246 (13.6) | 3295 (17.4) | 3145 (16.0) | 3077 (14.8) | 0.3 (−0.1 to 0.7) | .11 |
| Chronic obstructive pulmonary disease | 19 185 (25.3) | 3327 (20.2) | 4675 (24.8) | 5450 (27.8) | 5733 (27.5) | 2.5 (2.2 to 2.8) | <.001 |
| Chronic kidney disease | 22 308 (29.4) | 3059 (18.5) | 4397 (23.3) | 6776 (34.6) | 8076 (38.8) | 6.3 (6.0 to 6.6) | <.001 |
| Dialysis dependent | 13 989 (18.4) | 2459 (14.9) | 3797 (20.1) | 4000 (20.4) | 3733 (17.9) | 1.0 (0.6 to 1.3) | <.001 |
| Liver disease | 15 477 (20.4) | 2544 (15.4) | 3625 (19.2) | 4108 (20.9) | 5200 (25.0) | 3.9 (3.5 to 4.3) | <.001 |
| Cancer | 13 121 (17.3) | 2480 (15.0) | 3157 (16.7) | 3538 (18.0) | 3946 (19.0) | 1.9 (1.5 to 2.3) | <.001 |
| Human immunodeficiency virusc | 715 (1.7) | 182 (2.0) | 203 (1.9) | 178 (1.6) | 152 (1.3) | −3.4 (−4.9 to −1.8) | <.001 |
| Predisposing factor | |||||||
| History of congenital heart disease | 3437 (4.5) | 658 (4.0) | 857 (4.5) | 1043 (5.3) | 879 (4.2) | 0.7 (−0.01 to 1.5) | .05 |
| History of valve surgery | 10 591 (14.0) | 2116 (12.8) | 2515 (13.3) | 2800 (14.3) | 3160 (15.2) | 1.6 (1.2 to 2.0) | <.001 |
| History of implanted pacemaker or defibrillator | 9765 (12.9) | 1446 (8.8) | 2207 (11.7) | 2865 (14.6) | 3247 (15.6) | 4.8 (4.3 to 5.2) | <.001 |
| Disease type | |||||||
| Native-valve endocarditis | 54 332 (71.7) | 12 299 (74.5) | 13 747 (72.8) | 14 038 (71.6) | 14 248 (68.4) | −0.7 (−0.9 to −0.5) | <.001 |
| Prosthetic-valve endocarditis | 9777 (12.9) | 1989 (12.0) | 2355 (12.5) | 2558 (13.0) | 2875 (13.8) | 1.3 (0.8 to 1.7) | <.001 |
| Cardiac device–related endocarditis | 2236 (3.0) | 217 (1.3) | 454 (2.4) | 717 (3.7) | 848 (4.1) | 8.8 (7.8 to 9.9) | <.001 |
| Drug abuse–related endocarditis | 9484 (12.5) | 2006 (12.1) | 2331 (12.3) | 2298 (11.7) | 2849 (13.7) | 0.9 (0.4 to 1.3) | <.001 |
| Mode of acquisition | |||||||
| Community-acquired | 35 701 (47.1) | 8288 (50.2) | 8571 (45.4) | 8683 (44.3) | 10 159 (48.8) | −0.2 (−0.4 to 0.03) | .09 |
| Health care–associated | 40 128 (52.9) | 8223 (49.8) | 10 316 (54.6) | 10 928 (55.7) | 10 661 (51.2) | 0.2 (−0.04 to 0.4) | .10 |
| Nosocomial | 13 304 (17.5) | 2923 (17.7) | 3475 (18.7) | 3721 (19.0) | 3185 (15.3) | −1.0 (−1.4 to −0.7) | <.001 |
| Nonnosocomial | 26 824 (35.4) | 5300 (32.1) | 6841 (36.2) | 7207 (36.7) | 7476 (35.9) | 0.8 (0.5 to 1.1) | <.001 |
| Causative organism | |||||||
| Staphylococcus | 29 172 (38.5) | 6011 (36.4) | 7424 (39.3) | 7520 (38.3) | 8217 (39.5) | 0.6 (0.3 to 0.8) | <.001 |
| Staphylococcus aureus | 24 179 (31.9) | 4786 (29.0) | 6170 (32.7) | 6272 (32.0) | 6951 (33.4) | 1.0 (0.7 to 1.3) | <.001 |
| Methicillin-resistant | 9161 (12.1) | 878 (5.3) | 2399 (12.7) | 2886 (14.7) | 2998 (14.4) | 6.3 (5.8 to 6.8) | <.001 |
| Methicillin-sensitive | 15 018 (19.8) | 3908 (23.7) | 3771 (20.0) | 3386 (17.3) | 3953 (19.0) | −2.0 (−2.3 to −1.6) | <.001 |
| Streptococcus | 20 157 (26.6) | 4321 (26.2) | 4761 (25.2) | 5257 (26.8) | 5818 (27.9) | 0.7 (0.4 to 1.0) | <.001 |
| Oral streptococci | 7640 (10.1) | 1759 (10.7) | 1970 (10.4) | 2019 (10.3) | 1892 (9.1) | −1.2 (−1.7 to −0.8) | <.001 |
| Gram-negative bacilli | 4235 (5.6) | 897 (5.4) | 1022 (5.4) | 1186 (6.0) | 1130 (5.4) | 0.1 (−0.5 to 0.8) | .68 |
| Fungus | 1316 (1.7) | 285 (1.7) | 356 (1.9) | 365 (1.9) | 310 (1.5) | −1.0 (−2.2 to 0.2) | .10 |
| Other | 1745 (2.3) | 386 (2.3) | 389 (2.1) | 456 (2.3) | 514 (2.5) | 0.8 (−0.2 to 1.9) | .12 |
| Unspecified | 19 204 (25.3) | 4611 (27.9) | 4935 (26.1) | 4827 (24.6) | 4831 (23.2) | −1.5 (−1.9 to −1.2) | <.001 |
P values were calculated using 2-tailed log-linear Poisson regression.
P values was calculated using the ordinary least squares method.
These data were available for California admissions only (sample sizes were n = 43 114 overall; n = 9353 for 1998-2001; n = 10 818 for 2002-2005; n = 11 248 for 2006-2009; and n = 11 695 for 2010-2013).
Mode of Acquisition and Pathogens
The proportion of health care–associated infective endocarditis accounted for nearly half of all infective endocarditis cases during the study period, with 49.8% in 1998 through 2001 and 51.2% in 2010 through 2013 (APC, 0.2%; 95% CI, −0.04% to 0.4%; P = .10). The proportion of patients with health care–associated nonnosocomial infective endocarditis among all patients with endocarditis increased from 32.1% to 35.9% (APC, 0.8%; 95% CI, 0.5% to 1.1%; P < .001), and health care–associated nosocomial infective endocarditis decreased from 17.7% to 15.3% (APC, −1.0%; 95% CI, −1.4% to −0.7%; P < .001). The proportion of patients who were dependent on dialysis increased by 38.3% (95% CI, 23.7% to 54.6%) from 14.9% to 17.9% (APC, 1.0%; 95% CI, 0.6% to 1.3%; P < .001), accounting for 35.0% of health care–associated infective endocarditis cases in 2010 through 2013 (Table).
The standardized incidence of infective endocarditis caused by S aureus increased from 2.1 (95% CI, 2.0 to 2.2) to 2.7 (95% CI, 2.6 to 2.9) cases per 100 000 persons annually during the study period (APC, 1.0%; 95% CI, 0.6% to 1.4%; P < .001), and the standardized incidence of infective endocarditis among patients with methicillin-resistant S aureus infections increased from 0.23 (95% CI, 0.19 to 0.27) to 1.13 (95% CI, 1.05 to 1.22) cases per 100 000 persons annually (APC, 6.1%; 95% CI, 5.5% to 6.7%; P < .001) (eFigure 7 in the Supplement). The crude incidence of patients diagnosed with oral streptococcal infective endocarditis did not change (from 0.84 to 0.88 cases per 100 000 persons annually; APC, −0.1%; 95% CI, −0.8% to 0.6%; unadjusted Poisson regression P = .77) (eFigure 3A in the Supplement). After adjusting for age, sex, and race, the incidence decreased over time (from 0.84 [95% CI, 0.76 to 0.92] to 0.73 [95% CI, 0.67 to 0.80] cases per 100 000 persons annually; APC, −1.3%; 95% CI, −1.8% to −0.7%; multivariable Poisson regression P < .001) (Figure 1). Segmented regression did not show an increase in the slope of incidence of oral streptococcal infective endocarditis after the guideline changes (eFigure 5A and eTable 6 in the Supplement). Sensitivity analysis was conducted in each state and confirmed no significant increase in the standardized incidence of oral streptococcal infective endocarditis or in the slope of its incidence after the guideline change (eFigure 5B and C and eFigure 6 in the Supplement).
Mortality
Crude 90-day mortality was unchanged from 1998 through 2011 (from 23.9% to 24.2%, respectively; APC, −0.3%; 95% CI, −1.0% to 0.4%; P = .44). After risk adjustment for patient demographic characteristics and comorbidities, the risk of mortality decreased 2% per year over time (adjusted hazard ratio per year, 0.982; 95% CI, 0.978 to 0.986; P < .001). The proportion of patients who underwent cardiac surgery during or within 30 days of their index admission increased during the study period from 10.6% to 13.3% (APC, 2.2%; 95% CI, 0.8% to 3.6%; P < .001). Sensitivity analysis confirmed a decreased adjusted risk of 90-day mortality during the study period in each state. In California, crude 90-day mortality was 23.4% in 1998 and 23.6% in 2011 (APC, 0.3%; 95% CI, −0.5% to 1.1%; adjusted hazard ratio per year, 0.985; 95% CI, 0.980 to 0.990; P < .001). In New York, crude 90-day mortality was 24.4% in 1998 and 24.1% in 2013 (APC, −0.3%; 95% CI, −1.0% to 0.4%; adjusted hazard ratio per year, 0.978; 95% CI, 0.973 to 0.983; P < .001).
Overall mortality at 1 year and 5 years was 37.1% (95% CI, 36.8%-37.5%) and 52.9% (95% CI, 52.5%-53.3%), respectively. Health care–associated infective endocarditis was associated with significantly higher mortality than community-acquired endocarditis (adjusted hazard ratio, 1.52; 95% CI, 1.48-1.56; P < .001) (Figure 2). Figure 3 depicts survival stratified by pathogen. Compared with streptococcal infections, mortality was higher for gram-negative infections (adjusted hazard ratio, 1.22; 95% CI, 1.16-1.28; P < .001), staphylococcal infections (adjusted hazard ratio, 1.38; 95% CI, 1.34-1.42; P < .001), and fungal infections (adjusted hazard ratio, 1.84; 95% CI, 1.72-1.99; P < .001).
Figure 2. Five-Year Survival of Patients With Infective Endocarditis, Stratified by Mode of Acquisition, in California and New York State.
Patients admitted from 1998 through 2011 were included in the analysis for long-term outcomes.
Figure 3. Five-Year Survival of Patients With Infective Endocarditis, Stratified by Pathogen, in California and New York State.
Patients admitted from 1998 through 2011 were included in the analysis for long-term outcomes. Staphylococcus and Streptococcus each include the entire genus.
Discussion
In this retrospective cohort study of 75 829 inpatients with a first episode of infective endocarditis in California and New York from 1998 through 2013, the standardized incidence remained stable between 7.6 and 7.8 cases per 100 000 persons, and the adjusted hazard ratio of 90-day mortality decreased approximately 2% per year. There were shifts in patterns of patient characteristics and etiology, with increases in the proportion of prosthetic- and device-related endocarditis and in health care–associated nonnosocomial endocarditis, decreases in the proportion of native-valve endocarditis and in health care–associated nosocomial endocarditis, and no increase in the incidence of oral streptococcal endocarditis. To our knowledge, this study is the first to report population trends in standardized incidence of de novo endocarditis in a US population; to report trends in mortality, surgical treatment, and long-term outcomes; and to report shifts in modes of acquisition and pathogens, including nosocomial infections and high-risk groups, such as patients receiving dialysis.
The study period spanned changes in prevention of infective endocarditis. The American Heart Association revised the consensus guidelines for antibiotic prophylaxis of infective endocarditis in 2007, recommending much more restrictive use of antibiotic prophylaxis. The main changes were removal of the recommendations for routine antibiotic prophylaxis for invasive dental procedures in low- and moderate-risk patients and for most other invasive procedures in all patients. A substantial decrease in prescribing antibiotic prophylaxis following revised consensus guidelines occurred in the United Kingdom, and regional survey data suggest that prescribing patterns were similarly affected in the United States. In Canada, a 45% decrease in antibiotic prophylaxis recommendations for low-risk patients and a 10% decrease for high-risk patients were reported. The overall standardized incidence of infective endocarditis appeared to remain stable, which was contrary to previous studies. This may be because previous studies reported the crude incidence of endocarditis without adjusting for changing population mix, because they analyzed first episodes and recurrent infective endocarditis together (resulting in overestimates of the incidence of infective endocarditis), or because of the trend analysis methods used. These data supported findings of a much smaller county-based study and of a Medicare database in a large sample more representative of the US population and suggested that the recommendations restricting antibiotic prophylaxis for dental procedures had not contributed to increased oral streptococcal endocarditis. This study highlights the emergence of infective endocarditis unlikely to be associated with dental procedures, such as staphylococcal or health care–associated endocarditis.
Health care–associated endocarditis accounted for more than 50% of native-valve endocarditis and was associated with 50% mortality at 1 year. Nonnosocomial health care–associated endocarditis (ie, infections likely acquired through outpatient health care) increased steadily across the study period, whereas nosocomial endocarditis cases declined during this period. This decrease coincided with coordinated efforts to reduce hospital-acquired infections through evidence-based consensus guidelines, bundled interventions, and public reporting. Prospective studies are needed to assess whether these approaches could and should be applied to outpatient care.
The mortality rate associated with de novo endocarditis remained high along with the increase in resistant pathogens and patient morbidity, with marginal improvement in adjusted mortality risk. The excess mortality observed with health care–associated endocarditis compared with community-acquired endocarditis underscores the importance of efforts focused on reducing the occurrence of preventable health care–acquired infection. Given the limits of available therapeutic interventions, it is likely that any further improvements in the outcomes of endocarditis will take place in small increments. Consequently, more effective prevention of health care–associated endocarditis may achieve the greatest reductions in the burden of this disease.
The main strengths of this study were the population data drawn from mandatory, all-comer state data sets allowing comprehensive identification and follow-up of all patients diagnosed with infective endocarditis in California and New York, representing 18.6% of the US population during the study period. This study demonstrated that this large patient population was representative of the overall population of patients hospitalized with endocarditis in the United States (eTable 7 in the Supplement). Used in conjunction with state census data, these data sets permitted accurate estimates of standardized infective endocarditis incidence and changes in the risk profile of the population, providing a more informed analysis of trends than crude incidence in isolation. The ability to follow patients up longitudinally provided new insights into the evolving etiology of infective endocarditis, particularly nonnosocomial infection. The lower proportion of patients who underwent valve surgery in the present cohort compared with those reported in previous prospective cohort studies is likely explained by the more broadly representative nature of the sample of the present study in contrast with the highly selected populations seen by cardiac surgery programs. Additionally, previous studies either were performed using data sets that did not permit accurate longitudinal follow-up and consequently conflated de novo endocarditis cases with recurrences, relapses, and transferred cases, or consisted of US patient populations that were too small or not representative of the general population. The use of single trend testing and less specific ICD-9-CM codes may undermine the validity of observed trends in endocarditis and their association with guideline changes.
Limitations
This study has several limitations related to the use of administrative data sets, which may be subject to inaccurate coding of patient clinical diagnoses and procedures, with clinical information limited to conditions and treatments defined by ICD-9-CM codes. Consequently, the changes we observed and the long-term outcomes may have been affected by unmeasured confounders. However, validation of ICD-9-CM codes was performed using clinical records in a subset of patients with high positive predictive values. There were no ICD-9-CM codes to identify skilled home nursing care and wound care, which are normally included in the definition of health care–associated infection; consequently, the number of health care–associated infections could have been underestimated. It was not possible to identify the date when blood cultures were performed during hospitalization, and it was therefore assumed that patients who had a diagnosis of infective endocarditis on admission had either nonnosocomial or community-acquired endocarditis based on health care contact prior to admission. The organisms identified by ICD-9-CM codes were assumed to be causative if they were coded during the hospitalizations with infective endocarditis, but this could not be validated for individual patients. It was also not possible to differentiate between culture-negative infective endocarditis and infective endocarditis for which the causative organism was simply not recorded. Oral streptococcal endocarditis was defined by exclusion, so despite high positive predictive value, this may still be subject to misclassification; without additional information on dental procedures and concurrent antibiotic prophylaxis, our data did not confirm that the organisms were indeed of oral origin and did not confirm the role of dental prophylaxis in incidence. Microbiological etiology was unknown or not specified in 23% to 27% of cases, allowing a margin for error. Patients who recently moved from different states or different countries may not have had previous admissions in the database, which would lead to an underestimate of baseline comorbidities and the incidence of nonnosocomial infections. Additionally, the most recent data were 4 years old, and the study findings may not reflect the current characteristics and outcomes associated with infective endocarditis.
Conclusions
In California and New York State, the overall standardized incidence of infective endocarditis did not increase from 1998 through 2013, with changes in patient characteristics and etiology over this time.
eFigure 1. Exclusion Criteria
eFigure 2. Hierarchical Allocation Algorithm of Disease Type and Mode of Acquisition
eFigure 3. Trends in Crude Incidence of Infective Endocarditis, 1998-2013
eFigure 4. Odds Ratio of Incidence of Infective Endocarditis Between 1998 and 2013
eFigure 5. Interrupted Time Series Analysis, 1998-2013
eFigure 6. Trends in Standardized Incidence of Infective Endocarditis, 1998-2013
eFigure 7. Trends in Incidence of Infective Endocarditis, 1998-2013, Stratified by Causative Organism
eTable 1. Code Validation of ICD-9 Codes for Infective Endocarditis
eTable 2. ICD-9 Codes for Comorbidities
eTable 3. ICD-9 Codes for Disease Types
eTable 4. ICD-9 Codes for Mode of Acquisition
eTable 5. ICD-9 Codes for Causative Organisms
eTable 6. Segmented Regression Analysis on Incidence Using April 2007, October 2007, and April 2008 as Change Points and Model Excluding 6-Month and 12-Month Lag Time After April 2007 in California and New York State
eTable 7. Patients Baseline Characteristics Hospitalized With Infective Endocarditis in California and New York State vs All States in the Nationwide Inpatient Sample, 2005-2011
eAppendix. Supplemental Methods
eReferences
References
- 1.Tleyjeh IM, Steckelberg JM, Murad HS, et al. Temporal trends in infective endocarditis: a population-based study in Olmsted County, Minnesota. JAMA. 2005;293(24):3022-3028. [DOI] [PubMed] [Google Scholar]
- 2.DeSimone DC, Tleyjeh IM, Correa de Sa DD, et al. Temporal trends in infective endocarditis epidemiology from 2007 to 2013 in Olmsted County, MN. Am Heart J. 2015;170(4):830-836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Duval X, Delahaye F, Alla F, et al. ; AEPEI Study Group . Temporal trends in infective endocarditis in the context of prophylaxis guideline modifications: three successive population-based surveys. J Am Coll Cardiol. 2012;59(22):1968-1976. [DOI] [PubMed] [Google Scholar]
- 4.Sy RW, Kritharides L. Health care exposure and age in infective endocarditis: results of a contemporary population-based profile of 1536 patients in Australia. Eur Heart J. 2010;31(15):1890-1897. [DOI] [PubMed] [Google Scholar]
- 5.Murdoch DR, Corey GR, Hoen B, et al. ; International Collaboration on Endocarditis-Prospective Cohort Study (ICE-PCS) Investigators . Clinical presentation, etiology, and outcome of infective endocarditis in the 21st century: the International Collaboration on Endocarditis–Prospective Cohort Study. Arch Intern Med. 2009;169(5):463-473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bor DH, Woolhandler S, Nardin R, Brusch J, Himmelstein DU. Infective endocarditis in the U.S., 1998-2009: a nationwide study. PLoS One. 2013;8(3):e60033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bikdeli B, Wang Y, Kim N, Desai MM, Quagliarello V, Krumholz HM. Trends in hospitalization rates and outcomes of endocarditis among Medicare beneficiaries. J Am Coll Cardiol. 2013;62(23):2217-2226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fedeli U, Schievano E, Buonfrate D, Pellizzer G, Spolaore P. Increasing incidence and mortality of infective endocarditis: a population-based study through a record-linkage system. BMC Infect Dis. 2011;11:48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pant S, Patel NJ, Deshmukh A, et al. Trends in infective endocarditis incidence, microbiology, and valve replacement in the United States from 2000 to 2011. J Am Coll Cardiol. 2015;65(19):2070-2076. [DOI] [PubMed] [Google Scholar]
- 10.Wilson W, Taubert KA, Gewitz M, et al. ; American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee; American Heart Association Council on Cardiovascular Disease in the Young; American Heart Association Council on Clinical Cardiology; American Heart Association Council on Cardiovascular Surgery and Anesthesia; Quality of Care and Outcomes Research Interdisciplinary Working Group . Prevention of infective endocarditis: guidelines from the American Heart Association: a guideline from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group. Circulation. 2007;116(15):1736-1754. [DOI] [PubMed] [Google Scholar]
- 11.National Institute for Health and Care Excellence Prophylaxis against infective endocarditis: antimicrobial prophylaxis against infective endocarditis in adults and children undergoing interventional procedures. https://www.nice.org.uk/guidance/CG64. Published March 2008. Updated July 2016. Accessed August 1, 2016. [PubMed]
- 12.Habib G, Lancellotti P, Antunes MJ, et al. ; Document Reviewers . 2015 ESC guidelines for the management of infective endocarditis: the Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC), endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J. 2015;36(44):3075-3128. [DOI] [PubMed] [Google Scholar]
- 13.Baddour LM, Wilson WR, Bayer AS, et al. ; American Heart Association Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease of the Council on Cardiovascular Disease in the Young, Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and Stroke Council . Infective endocarditis in adults: diagnosis, antimicrobial therapy, and management of complications: a scientific statement for healthcare professionals from the American Heart Association. Circulation. 2015;132(15):1435-1486. [DOI] [PubMed] [Google Scholar]
- 14.Dayer MJ, Jones S, Prendergast B, Baddour LM, Lockhart PB, Thornhill MH. Incidence of infective endocarditis in England, 2000-13: a secular trend, interrupted time-series analysis. Lancet. 2015;385(9974):1219-1228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Desimone DC, Tleyjeh IM, Correa de Sa DD, et al. ; Mayo Cardiovascular Infections Study Group . Incidence of infective endocarditis caused by viridans group streptococci before and after publication of the 2007 American Heart Association’s endocarditis prevention guidelines. Circulation. 2012;126(1):60-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li JS, Sexton DJ, Mick N, et al. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis. 2000;30(4):633-638. [DOI] [PubMed] [Google Scholar]
- 17.Friedman ND, Kaye KS, Stout JE, et al. Health care–associated bloodstream infections in adults: a reason to change the accepted definition of community-acquired infections. Ann Intern Med. 2002;137(10):791-797. [DOI] [PubMed] [Google Scholar]
- 18.Wagner AK, Soumerai SB, Zhang F, Ross-Degnan D. Segmented regression analysis of interrupted time series studies in medication use research. J Clin Pharm Ther. 2002;27(4):299-309. [DOI] [PubMed] [Google Scholar]
- 19.Lockhart PB, Hanson NB, Ristic H, Menezes AR, Baddour L. Acceptance among and impact on dental practitioners and patients of American Heart Association recommendations for antibiotic prophylaxis. J Am Dent Assoc. 2013;144(9):1030-1035. [DOI] [PubMed] [Google Scholar]
- 20.Grattan MJ, Power A, Fruitman DS, Islam S, Mackie AS. The impact of infective endocarditis prophylaxis recommendations on the practices of pediatric and adult congenital cardiologists. Can J Cardiol. 2015;31(12):1497.e23-1497.e28. [DOI] [PubMed] [Google Scholar]
- 21.DeSimone DC, Wilson WR, Baddour LM. Trends in infective endocarditis, incidence, microbiology and valve replacement in the United States from 2000 to 2011: the devil is in the details. J Am Coll Cardiol. 2015;66(10):1201-1202. [DOI] [PubMed] [Google Scholar]
- 22.Duval X, Hoen B. Prophylaxis for infective endocarditis: let’s end the debate. Lancet. 2015;385(9974):1164-1165. [DOI] [PubMed] [Google Scholar]
- 23.Iung B, Tubiana S, Alla F, Lavielle M. Infective endocarditis and antibiotic prophylaxis. Lancet. 2015;386(9993):529-530. [DOI] [PubMed] [Google Scholar]
- 24.Berwick DM, Calkins DR, McCannon CJ, Hackbarth AD. The 100,000 lives campaign: setting a goal and a deadline for improving health care quality. JAMA. 2006;295(3):324-327. [DOI] [PubMed] [Google Scholar]
- 25.Marsteller JA, Hsu YJ, Weeks K. Evaluating the impact of mandatory public reporting on participation and performance in a program to reduce central line-associated bloodstream infections: evidence from a national patient safety collaborative. Am J Infect Control. 2014;42(10)(suppl):S209-S215. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eFigure 1. Exclusion Criteria
eFigure 2. Hierarchical Allocation Algorithm of Disease Type and Mode of Acquisition
eFigure 3. Trends in Crude Incidence of Infective Endocarditis, 1998-2013
eFigure 4. Odds Ratio of Incidence of Infective Endocarditis Between 1998 and 2013
eFigure 5. Interrupted Time Series Analysis, 1998-2013
eFigure 6. Trends in Standardized Incidence of Infective Endocarditis, 1998-2013
eFigure 7. Trends in Incidence of Infective Endocarditis, 1998-2013, Stratified by Causative Organism
eTable 1. Code Validation of ICD-9 Codes for Infective Endocarditis
eTable 2. ICD-9 Codes for Comorbidities
eTable 3. ICD-9 Codes for Disease Types
eTable 4. ICD-9 Codes for Mode of Acquisition
eTable 5. ICD-9 Codes for Causative Organisms
eTable 6. Segmented Regression Analysis on Incidence Using April 2007, October 2007, and April 2008 as Change Points and Model Excluding 6-Month and 12-Month Lag Time After April 2007 in California and New York State
eTable 7. Patients Baseline Characteristics Hospitalized With Infective Endocarditis in California and New York State vs All States in the Nationwide Inpatient Sample, 2005-2011
eAppendix. Supplemental Methods
eReferences