Abstract
Probiotics are increasingly used as a supplement to prevent adverse health outcomes in preterm infants. We conducted a systematic review, meta-analysis, and subgroup analysis of findings from randomized controlled trials (RCTs) to assess the magnitude of the effect of the probiotics on health outcomes among very-low–birth-weight (VLBW) infants. Relevant articles from January 2003 to June 2017 were selected from a broad range of databases, including Medline, PubMed, Scopus, and Embase. Studies were included if they used an RCT design, involved a VLBW infant (birthweight <1500 g or gestational age <32 wk) population, included a probiotic intervention group, measured necrotizing enterocolitis (NEC) as a primary outcome, and measured sepsis, mortality, length of hospital stay, weight gain, and intraventricular hemorrhage (IVH) as additional outcomes. The initial database search yielded 132 potentially relevant articles and 32 (n = 8998 infants) RCTs were included in the final meta-analysis. Subgroup analysis was used to evaluate the effects of the moderators on the outcome variables. In the probiotics group, it was found that NEC was reduced by 37% (95% CI: 0.51%, 0.78%), sepsis by 37% (95% CI: 0.72%, 0.97%), mortality by 20% (95% CI: 0.67%, 0.95%), and length of hospital stay by 3.77 d (95% CI: −5.94, −1.60 d). These findings were all significant when compared with the control group. There was inconsistent use of strain types among some of the studies. The results indicate that probiotic consumption can significantly reduce the risk of developing medical complications associated with NEC and sepsis, reduce mortality and length of hospital stay, and promote weight gain in VLBW infants. Probiotics are more effective when taken in breast milk and formula form, consumed for <6 wk, administered with a dosage of <109 CFU/d, and include multiple strains. Probiotics are not effective in reducing the incidence of IVH in VLBW infants.
Keywords: very preterm infants, probiotics, necrotizing enterocolitis, sepsis, mortality, hospital admission
Introduction
Survival rates of very preterm infants [very-low–birth-weight (VLBW) infants born <32 wk gestation or with a birth weight of <1500 g)] are now >90% thanks to advances in medical technology. However, with decreasing gestational age and birthweight, there is a concomitant increasing risk of short-term and long-term developmental complications and problems (1), which may be associated with mortality and poor neurodevelopmental outcomes (2).
Among the complications, necrotizing enterocolitis (NEC) is one of the most serious conditions in VLBW infants (3) and is a leading cause of death in this population (4). Infants diagnosed with NEC have a higher chance of developing poor brain growth and associated neurodevelopmental delays (2). Although the pathogenesis of NEC is multifactorial and remains unclear, it is thought that because the gut microbiota of VLBW infants may be very immature, exposure to pathogenic organisms would cause mucosal injury related–inflammatory response, leading to NEC (5).
Late-onset sepsis is another common cause of death in VLBW infants, with ∼20% of all deaths attributable to this condition (6). VLBW infants are diagnosed with sepsis when ≥1 blood culture obtained after 72 h of life are identified as positive for pathogenic bacteria (6). Sepsis is associated with increased mortality, morbidity, and prolonged hospitalization (6).
Intraventricular hemorrhage (IVH) is a condition found predominantly in preterm infants in whom fluctuation of blood pressure and impaired cerebrovascular regulation leads to bleeding into the ventricular system of the brain (7). As a result of IVH, VLBW infants have been found to have longer stays in the hospital than full-term infants (8), making them more vulnerable to nosocomial infections.
Newborns, especially VLBW infants, have not yet established a microecological balance of the intact intestinal barrier and therefore have immature immune function. Consequently, they have low resistance to infection. Pathogenic bacteria can pass through the intestinal mucosa more easily and enter the tissues of the mesenteric lymph nodes, organs, and blood flow, causing intestinal infections, such as diarrhea, neonatal necrotizing enterocolitis, and sepsis, all of which are life threatening. VLBW infants (birth weight <1500 g) develop a very different, “sparse” microbiome than that described for the full-term infant (9). The immature and fragile gut in VLBW infants, the in utero environment (e.g., prenatal maternal illness, infections, smoking, and physiological stress), and postnatal factors, such as formula feeding, invasive procedures, antibiotics, and medications, can all alter the gastroenteric pH level. In addition, VLBW infants often require prolonged hospitalization, which may affect their resistance and gastrointestinal microbiota, thereby increasing the risk of infection and inflammation (10).
In recent years, there has been increased interest in the use of probiotics in VLBW infants for the promotion of growth and development and prevention of various complications. Probiotics are defined as “live microorganisms [that], when consumed in adequate amounts, confer a health effect on the host” (11). The supplementation of probiotics may improve gastrointestinal tolerance (12). The possible mechanism through which probiotics reduce the risk of VLBW infants developing NEC is by colonizing their gastrointestinal tract with normal flora. This prevents overgrowth of pathogenic bacteria and reduces the risk of NEC, sepsis, and IVH (13), leading to a reduction in mortality in VLBW infants (14).
RCTs examining the effectiveness of probiotics on growth in VLBW infants have demonstrated mixed results. A few trials have shown improved growth (5, 15), some trials have shown significant differences between treatment and control groups in NEC (16), sepsis (17), subsequent mortality (16), and shortened length of hospital stay (18), whereas others have found no differences (19, 20). Although there are published meta-analyses that include a small number of articles (5, 15, 19, 20) or focus on infants <37 wk of gestation (16), there are no meta-analyses that examine the most recent evidence in VLBW infants born with a birth weight <1500 g or who are born at 32 wk of gestation. The present study aimed to conduct a meta-analysis of randomized control trials published in the last 14 y that used a large sample size to evaluate if probiotics have beneficial effects in VLBW infants, particularly in relation to reduction of NEC. In addition, this study assessed important health outcomes in VLBW infants, including medical complications, such as sepsis and IVH, along with mortality, prolonged length of hospital stay, and weight gain.
Methods
Search methods.
The review protocol was registered with the Prospero International Prospective Register of Systematic Reviews (http://www.crd.york.ac.uk/PROSPERO/display_record.asp?ID = CRD42016036557; registration number CRD42016036557 (21). The Population, Intervention, Control, Outcome principle was used to search the literature (21). The population was VLBW infants born <32 wk of gestation and weighing <1500 g, the intervention was the probiotic supplementation to the intervention group, the control was a control or placebo group that did not receive probiotic supplementation, and the outcomes included NEC, sepsis, IVH, neurodevelopmental growth involving weight gain, length of hospital stay, and mortality.
The keywords used to identify relevant studies were as follows: very preterm infants and probiotics and randomized controlled trial; very preterm infants and probiotics and growth and randomized controlled trial; and very preterm infants and probiotics and infections and randomized controlled trial. For cases where data were not adequately reported, authors were contacted in an attempt to retrieve the necessary information.
Inclusion criteria.
The criteria used to determine inclusion of studies in the meta-analysis were as follows: 1) publication in peer-reviewed literature in the last 14 y (January 2003 until June 2017); 2) study design involved a RCT; 3) population included VLBW infants with gestational age <32 wk or birth weight <1500 g; 4) probiotics were used as the intervention approach; 5) outcome variables included NEC as the primary outcome and sepsis, mortality, IVH, length of hospital stay, and weight gain as additional outcomes; and 6) if multiple studies were published on the same population, the most recent one was included in the analysis.
Data collection.
For articles with insufficient data reported, the corresponding authors were contacted to seek further information and acquire the necessary data. Thirteen authors were contacted for weight at the beginning and end of the study (12, 14, 20, 22–31). Some authors replied with the weight gain (24, 27, 31), and their values were included in the analysis.
Data extraction.
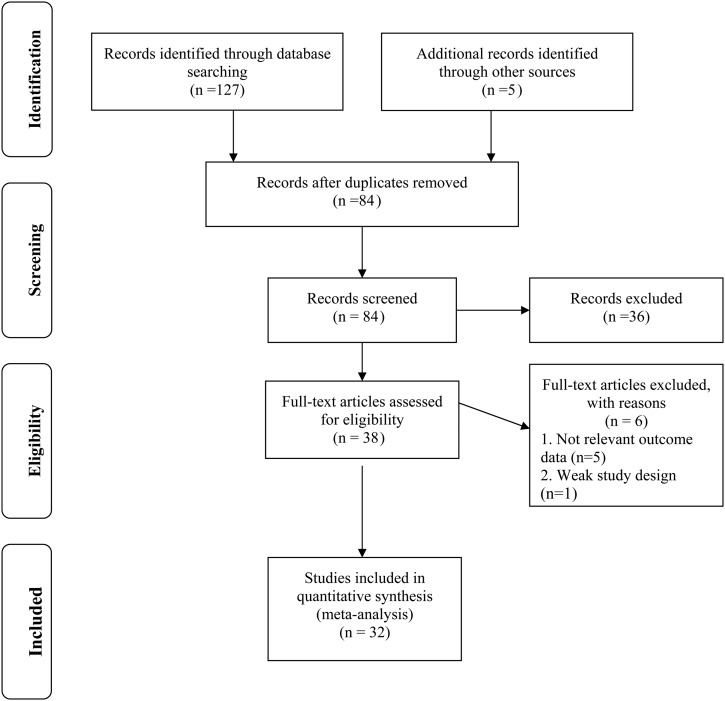
The process of identifying relevant articles for inclusion in the meta-analysis is described in Figure 1. Relevant articles were selected from a broad range of databases, including PubMed, Scopus, and Embase. The following data were extracted from each of the included articles: study setting, participant demographics, study population, type of probiotics used in the intervention, type of control used, the details of primary and secondary end points, study methodology, blinding, study completion rates, times of measurement, indicators of acceptability to users, suggested mechanisms of intervention action, information for assessment of the risk of bias and reported major findings.
FIGURE 1.
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow chart representing the reviewing process.
Quality assessment.
The Physiotherapy Evidence Database tool was used to assess the quality of the articles (32). This tool categorizes the quality of studies into 3 levels: high quality (≥8 points), moderate quality (4–7 points), and low quality (≤3 points). These levels are based on criteria as follows: random allocation of subjects, concealed randomization, similarity of baseline information between groups, blinding to subjects, assessors, and researchers, attrition rate, maintenance of group allocations, use of intention-to-treat analysis, use of variability measures, and use of between-group comparison methods.
Statistical analysis.
The effect of probiotics on health outcome variables was described with the use of a pooled effect size method. Random-effect meta-analyses were performed to describe the overall effect size. Risk ratio was used to present the effect size for NEC, sepsis, mortality, and IVH, and pooled mean differences for length of hospital stay and weight gain were calculated. CIs and P values were calculated for each of the outcome variables. Several parameters were used to assess the inconsistency between the pooled studies. These included visual inspection of CI overlap and I2 statistics. Subgroup analyses were performed, including quality of studies (high compared with moderate), dosage of probiotics (≥109 CFU compared with <109 CFU), strains of probiotics (single compared with multiple strains), and the forms of probiotics used (milk compared with formula or a combination of breast milk and formula). The duration of probiotics intervention (≥6 wk compared with <6 wk), the effect of probiotics on gestational age (≥29 wk compared with <29 gestation wk), and on infant birth weight (≤1000 g compared with >1000 g) were also analyzed. Publication bias was assessed with the use of the Egger test, and funnel plots were presented. Sensitivity analyses were conducted to estimate the effect of probiotics owing to a particular study or studies.
Results
The initial database search yielded 127 potentially relevant articles, and 5 additional articles were found through the reference lists of published articles. Duplicate articles (84) were removed. A total of 43 studies remained, and their titles and abstracts were screened to find any relevant articles for the topic. Following application of the Physiotherapy Evidence Database process, 38 articles remained. Of these, 5 articles were excluded due to lack of relevant outcome data (n = 4) (33–36) and use of a nonrandomized controlled trial design (n = 1) (37). This resulted in 32 studies being included in the final meta-analysis.
Participants.
Full details of the included studies are provided in Table 1. The 32 included studies reported outcomes on 4,496 infants treated with probiotics and 4,452 infants in control groups. The third inclusion criterion for all the studies was a population consisting of infants with either a birth weight <1500 g or gestational age <32 wk. Six studies met this criterion based on gestational age (14, 26, 29, 38, 39, 45, 51), 8 based on birth weight (3, 5, 20, 40–42, 44, 47), and the remaining studies included both gestational age and birth weight (12, 14, 19, 22–25, 27, 28, 30, 31, 46, 48–50, 52–54).
TABLE 1.
Characteristics of 29 included randomized controlled trials1
| Study (year) (ref.) | Participants at beginning, n (P/C) | Design; location | Gestational age, P/C, wk | Sex, n P/C, M/F | Birth weight, P/C, g | Probiotic route of administration; duration, P/C | Probiotics strain (s) | Dose, CFU/g | Form of probiotics | Placebo group | Measured outcomes and results | Attrition, n (%) | Key results |
| Al-Hosni et al. (2012) (12) | 101 (50/51) | DB; United States | 25.7 (1.4)/25.7 (1.4) | 22/28; 28/22 | 778 (138)/779 (126) | Enteral; 28 d | Lactobacillus rhamnosus GG,Bifidobacterium infantis | 5 × 108 | Breast milk | Breast milk | Weight gain, CLD, ROP, NEC, sepsis, mortality, mean volume of feeding | 0 (0) | 501–750-g infants had significant weight gain |
| Bin-Nun et al. (2005) (5) | 145 (72/73) | SB; Israel | 29.8 (2.6)/29.3 (4.3) | 44/28; 37/36 | 1152 (262)/1111 (278) | Oral; 36 wk PMA | B. infantis, Streptococcus thermophilus, B. bifidus. | 1.05 × 109 | Breast milk and preterm formula | Breast milk or formula | Weight gain, NEC, TPN | 0 (0) | NEC rate was reduced in probiotics group (4% vs. 16.4% in the control group; P = 0.03). NEC was less severe in the probiotics group |
| Braga et al. (2011) (3) | 231 (119/112) | DB; Brazil | 29.5 (2.5)/29.2 (2.6) | 58/61; 55/57 | 1194.7 (206.3)/1151.4 (224.9) | Oral; 28 d | B. breve, L. casei | 3.5 × 107 3.5 × 109 | Breast milk | Breast milk | NEC, sepsis, death | 12 (4.93) | 4 confirmed NEC cases; ≥2 occurred only in control group |
| Chou et al. (2010) (14) | 301 (153/148) | SB; China | 28.5 (2.3)/28.5 (2.3) | 83/70; 85/63 | 1103.6 (232.4)/1097 (231.4) | Oral; until discharge | L. acidophilus, B. infantis | 109 CFU for each strain | Breast milk | Breast milk | Growth: height, weight, HC, neurodevelopmental and sensory; outcomes: NEC, death, CLD, sepsis, hospitalization, visual impairment, deafness, cerebral palsy | 0 (0) | Mortality was less in probiotics group compared with control group (8 cases, 5.2% vs. 24 cases, 16.2%; P = 0.002) |
| Costalos et al. (2003) (29) | 87 (51/36) | SB; Greece | Not mentioned | 24/27; 23/13 | Not mentioned | Enteral; 30 d | Saccharomyces boulardii | 109 | Preterm formula | Maltodextrins with preterm formula | Weight gain and loss, daily milk intake, NEC, sepsis, bacterial counts | 0 (0) | No difference in weight gain, NEC, and sepsis |
| Costeloe et al. (2016) (38) | 1310 (650/660) | DB; England | 27.88 (1.65)/27.9 (1.75) | 374/276; 370/290 | 1039 (312)/1043 (317) | Oral; 36 wk PMA or discharge | B. breve BBG-001 | 109 | Breast milk | Corn starch with breast milk | ROP, death, NEC, sepsis | 5 (0.38) | No effects on death, NEC and sepsis |
| Demirel et al. (2013) (22) | 271 (135/136) | DB; Turkey | 29.4 (2.3)/29.2 (2.5) | 69/66; 66/70 | 1164 (261)/1131 (284) | Oral; until discharge | S. boulardii | 5 × 10 9 | Breast milk and preterm formula | Breast milk or formula | Feeding amount, full feeding day, weight, NEC, sepsis | 7 (2.5) | Feeding intolerance and sepsis are lower in the probiotics group |
| Dilli et al. (2015) (23) | 200 (100/100) | DB; Turkey | 28.8 (1.9)/28.2 (2.2) | 53/47; 58/42 | 1236 (212)/1147 (271) | Oral; NICU stay 34.5 (11)/37 (18) | B. lactis | 5 × 10 9 | Breast milk and preterm formula | Breast milk or formula + maltodextrin | Height, weight, HC, NEC, sepsis, feeding intolerance, RDS, stay at NICU, mortality | 0 (0) | NEC rate was lower in probiotics group (2% vs. 18% in placebo group). Sepsis and mortality rate were lower, and hospital stay was shorter in the probiotics group |
| Fernandez et al. (2013) (39) | 150 (75/75) | DB; Mexico | 30.95 (4.7)/31.25 (4.5) | Not mentioned | Not mentioned | Oral or enteral; 51.75 (57.5)/47.25 (55.5) | L acidophilus,L. rhamnosus, L. casei, L. plantarum, B. infantis, S. thermophillus | 1.0 × 1094.4 × 108 1.0 × 109 1.76 × 108 2.76 × 107 6.6 × 105 | Breast milk and preterm formula | Breast milk or preterm formula | Weight, death, NEC | 0 (0) | NEC was reduced |
| Hays et al. (2016) (24) | 102 (50/52) | DB; France | Not mentioned | Not mentioned | Not mentioned | Oral; 4–6 wk | B. lactis | 109 | Maltodextrin + water | Maltodextrin with sterile water | Weight, length, HC, NEC, sepsis, body composition, bacterial count | 22 (21.6) | No differences were found in all outcome variables |
| Hikaru et al. (2010) (40) | 208 (108/100) | SB; Japan | 28.5 (2.6)/28.1 (2.8) | 58/50; 44/56 | 1066 (272)/1009 (267) | Oral; until discharge | B. breve | 109 | Breast milk and preterm formula | Breast milk or preterm formula | Hospitalization, sepsis, death | 0 (0) | Sepsis was lower in the probiotics group. Weight gain was greater and mortality rate was lower in the probiotics group |
| Jacobs et al. (2013) (25) | 1099 (548/551) | DB; Australia and New Zealand | 27.9 (2.0)/27.8 (2.0) | 272/276; 300/248 | 1063 (259)/1048 (260) | Oral; until discharge or term-corrected age | B. infantis,S. thermophilus, B. lactis | 3 × 108, 3.5 × 108, 3.5 × 108 | Breast milk and preterm formula with maltodextrin | Maltodextrin | Sepsis, death, hospitalization, duration of parenteral nutrition, days to full enteral feeds, weight at 28 d, ROP, CLD, IVH | 0 (0) | Reduced NEC rate |
| Kanic et al. (2015) (41) | 80 (40/40) | SB; Slovenia | 28.25 (0.75)/28.55 (0.95) | 22/18; 27/13 | 1104.1 (233.2)/1024.3 (249.9) | Enteral; until discharge | L. acidophilus, Enterococcus faecium,B. infantum | 1.2 × 107 | Breast milk | Breast milk or formula | Hospitalization, late-onset sepsis, pneumonia, NEC, death, meningitis, urinary tract infection, and omphalitis | 0 (0) | Lower incidence of sepsis in probiotics group than in control group (P = 0.002) |
| Lin et al. (2005) (42) | 367 (180/187) | SB; Taiwan | 28.5 (2.5)/28.2 (2.5) | 84/96; 100/87 | 1104 (242)/1071 (243) | Oral or enteral; until discharge | L. acidophilus, B. infantis | 106 of each | Breast milk | Breast milk | Hospitalization, death, NEC, sepsis | 0 (0) | Death and NEC were lower in probiotics group than in control group |
| Lin et al. (2008) (43) | 434 (217/217) | SB; Taiwan | Not mentioned | 122/95; 115/102 | 1028.9 (246)/1077.3 (214.4) | Oral; 6 wk | L. acidophilus, B. bifidum | 109 | Breast milk and preterm formula | Breast milk or formula | NEC, sepsis, death, IVH, CLD, NICU stay, weight gain | 9 (2) | Death (P = 0.002) and NEC (P = 0.02) were lower in probiotics group than in control group |
| Manzoni et al. (2006) (44) | 80 (39/41) | DB; Italy | 29.5 (5)/29.3 (4) | 20/19; 21/20 | 1212 (290)/1174 (340) | Oral; 6 wk or until discharge | L. rhamnosus | 6 × 109 | Breast milk | Breast milk | Hospitalization, time of achievement of full feedings, death, NEC | 0 (0) | NEC was slightly lower in the probiotics group (P = 0.53) |
| Mihatsch et al. (2010) (45) | 180 (91/89) | SB; Germany | 26.6 (1.8)/26.7 (1.7) | 55/36; 47/42 | 856 (251)/871 (287) | Oral; 6 wk | B. lactis | 6 × 109 | Breast milk and preterm formula | Breast milk or preterm formula | NEC, death, nosocomial infections | 17 (9.3) | All variables were not statistically significant |
| Mohan et al. (2008) (26) | 69 (37/32) | DB; Germany | 31.05(2.31)/31.27 (2.56) | 14/23; 11/21 | 1449 (343)/1398 (331) | Oral; 21 d | B. lactis Bb12 | 2 × 10 9 | Sterile water | Human milk fortifier without probiotics: Nestlé FM 2000B | Body weight, alteration of gut fermentation patterns, and immunologic parameters | 0 (0) | Higher body weight in the probiotics group than in placebo group (P = 0.002) |
| Oncel et al. (2014) (46) | 400 (200/200) | DB; Turkey | 28.2 (2.4)/27.9 (2.5) | 108/92; 98/102 | 1071 (274)/1048 (298) | Oral; until discharge | L. reuteri | 1 × 108 | Oil base | Oil base | NEC, sepsis, death, hospitalization, feeding intolerance | 24 (6) | Sepsis and duration of hospital stay were lower in probiotics group than in placebo group |
| Patole et al. (2014) (27) | 153 (77/76) | DB; Australia | 28.33 (2.96)/27.67 (2.22) | 45/320; 41/35 | 1041.67 (388.89); 1031.67 (333.33) | Oral; until 37 wk PMA | B. breve M-16V | 3 × 109 | Breast milk or water | Dextrin with breast milk or sterile water | Discharge weight, death, hospitalization | 6 (3.8) | No difference between probiotics and control group in all variables |
| Rojas et al. (2012) (47) | 750 (372/378) | DB; Colombia | 31.75 (1.5)/31.5 (2) | 186/186; 185/193 | 1515.75 (124.25)/1477.75 (155.25) | Oral or enteral; until discharge | L. reuteri | 108 | Oil base | Oil-based suspension | Death, duration of hospital, NEC, sepsis | 0 (0) | Duration of hospitalization decreased in premature infants ≤1500 g (P = 0.03) |
| Rougé et al. (2009) (48) | 94 (45/49) | DB; France | 28.1 (1.9)/28.1 (1.8) | 28/17; 26/23 | 1115 (251)/1057 (260) | Oral; until discharge | L. rhamnosus,B. longum | 108 | Maltodextrin + water | Maltodextrin with sterile water | Nutrition–total calories delivered enterally, duration of hospital stay, death, oxygen therapy duration | 0 (0) | No difference between probiotics and control group in all variables |
| Roy et al. (2014) (30) | 112 (56/56) | DB; India | 32 (2)/32.2 (2) | 14/42; 16/40 | 1192 (341)/1069 (365) | Oral; 6 wk or until discharge | L. acidophilis,B. longum, B. bifidum, B. lactis | 1.25 × 109, 1.25 × 108, 1.25 × 108, 1 × 109 | Not mentioned | Breast milk | Full feed establishment, candida, death, NEC, hospitalization | 0 (0) | Lower incidence in sepsis (P = 0.02) and hospitalization (0.002) in the probiotics group |
| Samanta et al. (2009) (49) | 186 (91/95) | DB; India | 30.12 (1.63)/30.14 (1.59) | Not mentioned | 1172 (143)/1210 (143) | Oral; until discharge | B. infantis,B. bifidum,B. longum,L. acidophilus | 2.5 × 109 | Breast milk | Breast milk | NEC, death, hospitalization | 0 (0) | Duration of hospital stay was lower (13.76 vs. 19.2, P < 0.001), lower NEC incidence (5.5% vs. 15.8%, P = 0.042) and lower incidence of sepsis (14.3% vs. 29.5%, P = 0.02) in the probiotics group |
| Sari et al. (2012) (28) | 221 (110/111) | DB; Turkey | 29.7 (2.5)/29.8 (2.3) | 46/40; 49/62 | 1241 (264)/1278 (273) | Oral; 36 (22.5)/35.2 (24.9) | L. sporogenes | 3.5 × 108 | Breast milk and preterm formula | Breast milk or formula | Weight gain, length, HC, CLD, hospitalization, NEC, sepsis, feeding intolerance, oxygen days, full feeding days | 47 (21.3) | No difference between probiotics and control group in all variables |
| Serce et al. (2013) (50) | 208 (104/104) | DB; Turkey | 28.7 (2.1)/28.8 (2.2) | 50 | 1162 (216)/1126 (232) | Oral; until discharge | S. boulardii | Not mentioned | Breast milk and preterm formula | Breast milk with distilled water | NEC, weight gain, death, hospitalization, sepsis, time to reach 100 mL · kg−1 · d−1 of oral feeding | 36 (14.8) | No difference between probiotics and control group in all variables |
| Stratiki et al. (2007) (51) | 75 (41/34) | SB; Greece | 31.5 (2.5); 31(2.75) | 23/18; 17/17 | 1420 (220)/1400 (50) | Oral; 30 d | B. lactis | 2 × 107 | Preterm formula | Formula | Weight, length, NEC, time to full enteral feed | 4 (4.9) | No difference between probiotics and control group in all variables |
| Totsu et al. (2014) (20) | 283 (153/130) | DB; Japan | 28.6 (2.9)/28.5 (3.3) | 87/66; 71/59 | 1016 (289)/998 (281) | Enteral; until body weight reached 2000 g | B. bifidum | 2.5 × 109 | Water, breast milk, and formula | 500 mg dextrin | Body weight, HC, hospitalization, sepsis, accelerated establishment of enteral feeding, death, NEC, CLD | 50 (17.7) | Sepsis rate is lower in probiotics group than control group (3.9% vs. 10.0%, P < 0.05) |
| Van Niekerk et al. (2014) (31) | 184 (91/93) | DB; South Africa | Overall mean 28.7 | 43/48; 43/50 | Not mentioned | Enteral; 28 d | B. infantis | 3.5 6 × 109 | Breast milk | Breast milk with MCT oil | Daily weight gain, length, HC, feeding amount (day 14, 28) | 28 (15.2) | Higher length score in probiotics group (P < 0.01) |
| Chowdhury et al. (2016) (52) | 102 (52/50) | DB; Bangladesh | 31.38 (0.93)/31.68 (0.84) | 33/19; 36/14 | 1310.6 (110.41)/1338.0 (97.71) | Oral/oral | Capsule TS6 probiotic containingBifidobacterium species, Lactobacillus | 6 × 109 | Breast milk | Breast milk | NEC, hospital stay | 0 | Lower NEC rate and shorter hospital stay |
| Manzoni et al. (2009) (53) | 319 (151/168) | DB; Italy | 29.8 (2.8)/29.5(3.2) | 72/79; 86/82 | 1138 (253)/1109 (269) | Oral/oral | Bovine lactoferrin + L. rhamnosus GG | 6 × 109 | Breast milk | 5% glucose solution | Sepsis, NED, mortality, IVH | 0 | Lower NEC, sepsis, mortality, and IVH rates |
| Manzoni et al. (2014) (54) | 496 (238/258) | DB; Italy and New Zealand | 29.6 (2.8)/29.4 (3.1) | 116/122; 134/124 | 1129 (242)/1118 (259) | Oral/oral | Lactoferrin + L. rhamnosus GG | 6 × 109 | Breast milk | Breast milk | NEC, mortality | 0 | Lower NEC and mortality rates |
Values are means (SDs) unless otherwise indicated. CLD, chronic lung disease; DB, double blinded; HC, head circumference; IVH, intraventricular hemorrhage; MCT, medium-chain TG; NEC, necrotizing enterocolitis; NICU, neonatal intensive care unit; P/C, probiotic group/control group; PMA, postmenstrual age; RDS, respiratory distress syndrome; ref., reference; ROP, retinopathy of prematurity; SB, single blinded; TPN, total parenteral nutrition.
Probiotics were initiated at different times, ranging from the first feed to 7 d after birth. Fifteen studies introduced probiotics or placebo with the first feeding (5, 12, 19, 22, 27, 28, 31, 39–42, 46, 48–50, 52), 5 studies initiated probiotics on the second day of birth (3, 20, 38, 47, 51) and 2 trials initiated probiotics within 3 d of life (30, 44), within a week (24, 29), and after the first week (14, 24, 53, 54). Only 1 study introduced probiotics after the initiation of breast milk (45), from day 4 of life (26), or when a VLBW infant was receiving 1 mL of breast milk every 4 h (25). The studies were conducted in 19 different locations, including Turkey (22, 23, 28, 46, 50), France (24, 48), Germany (26, 45), Greece (29, 51), India (30, 49), Bangladesh (52), Taiwan (19, 42), Japan (20, 40), Brazil (3), China (14), Colombia (47), England (38), Italy (44, 53, 54), Israel (5), Mexico (39), Slovenia (41), South Africa (31), Australia (27), or in multiple centers (i.e., Australia and New Zealand) (25), and the United States (12).
Effects on NEC.
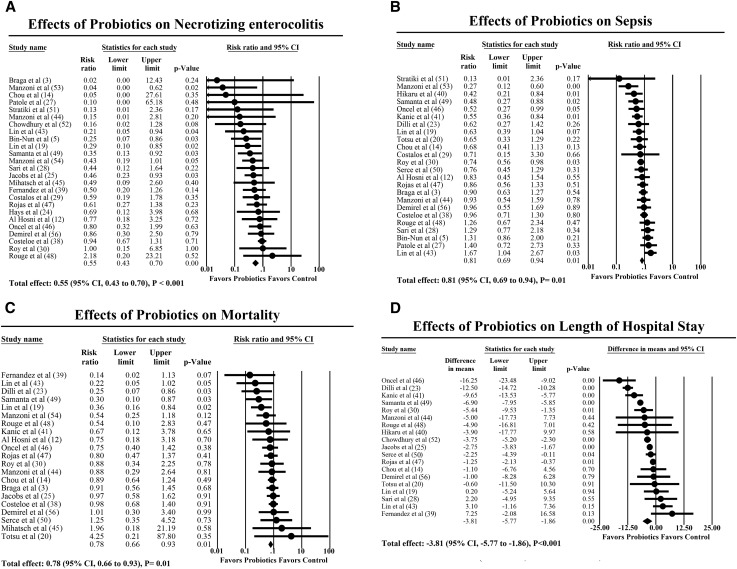
Twenty-five studies with a total sample of 8492 infants measured rates of infant NEC. The mean NEC rate in the probiotics group was 3.6% compared with 6.9% in the control group. The meta-analysis showed there was a significantly lower risk ratio in NEC by 45.0% (95% CI: 0.43%, 0.70%; P < 0.001) in the probiotics group compared with the control groups. The heterogeneity was not statistically significant (I2 = 10.7%, P = 0.31). The forest plot of the effect is presented in Figure 2A (see also Table 2).
FIGURE 2.
Forest plots of the effects of probiotics on necrotizing enterocolitis (A), sepsis (B), mortality (C), and length of hospital stay (D).
TABLE 2.
Total effects of probiotics on necrotizing enterocolitis, sepsis, mortality, hospital stay, intraventricular hemorrhage, and weight gain
| Variable | Studies, n | Risk ratio or mean difference | 95% CI | P | I2 | 2 P |
| Necrotizing enterocolitis, risk ratio | 25 | 0.55 | 0.43, 0.70 | <0.001 | 10.68 | 0.31 |
| Sepsis, risk ratio | 24 | 0.83 | 0.71, 0.96 | 0.02 | 48 | 0.01 |
| Mortality, risk ratio | 21 | 0.78 | 0.66, 0.93 | 0.01 | 6.92 | 0.37 |
| Hospital stay, mean difference, d | 19 | −3.81 | −5.77, −1.86 | <0.001 | 89.62 | <0.001 |
| Intraventricular hemorrhage, risk ratio | 10 | 0.91 | 0.73, 1.14 | 0.42 | 0 | 0.63 |
| Weight gain, mean difference, g | 15 | −0.29 | −1.16, 0.58 | 0.51 | 0 | 0.97 |
A subgroup analysis (Table 3) found that the effect of the probiotics on NEC was statistically significant when the study quality was high (P < 0.001) compared with studies of moderate quality (P > 0.05). It was also found that the effect of probiotics on NEC was statistically significant when it was in a dosage <109 CFU (P < 0.001) compared with those trials that used a larger dosage of ≥109 CFU (P > 0.05). Probiotics were more effective when they consisted of multiple strains (P < 0.001) compared with a single strain (P < 0.05). The trials that used a combination of breast milk and preterm formula (P < 0.001) as a medium to administer probiotics had better results compared with trials that used other forms (P > 0.05). A significant effect was also found in trials with infants <29 wk of gestation (P < 0.001) compared with trials with infants >29 wk of gestation. The effect was not different between trials that used probiotics for <6 wk compared with trials of >6 wk.
TABLE 3.
Subgroup analysis on the effects of probiotics on necrotizing enterocolitis and sepsis1
| Necrotizing enterocolitis |
Sepsis |
|||||||||||||
| Subgroups | Studies, n | Participants, n | I2 | Q-test | Risk ratio | 95% CI | P | Studies, n | Participants, n | I2 | Q-test | Risk ratio | 95% CI | P |
| Quality of study | 0.01 | <0.02 | ||||||||||||
| High | 15 | 3374 | 0.00 | 10.94 | 0.44*** | 0.33, 0.60 | 12 | 2357 | 68.53** | 34.95 | 0.90 | 0.70, 1.16 | ||
| Moderate | 10 | 981 | 0.00 | 8.44 | 0.79 | 0.60, 1.03 | 11 | 1101 | 14.36 | 12.85 | 0.78*** | 0.54, 1.12 | ||
| Dosage | 0.42 | 0.57 | ||||||||||||
| <109 | 13 | 2410 | 0.00 | 7.49 | 0.40*** | 0.28, 0.58 | 12 | 2508 | 68.40*** | 34.81 | 0.68* | 0.46, 0.99 | ||
| ≥109 | 11 | 1945 | 23.37 | 13.05 | 0.57 | 0.37, 0.90 | 10 | 1846 | 11.92 | 10.22 | 0.86 | 0.68, 1.08 | ||
| Strains | <0.01 | 0.77 | ||||||||||||
| Single | 11 | 1792 | 0.00 | 8.10 | 0.74* | 0.56, 0.97 | 11 | 2116 | 37.09 | 15.89 | 0.79 | 0.60, 1.03 | ||
| Multiple | 14 | 2563 | 0.00 | 10.13 | 0.37*** | 0.26, 0.54 | 12 | 1342 | 63.56*** | 30.19 | 0.74 | 0.52, 1.04 | ||
| Form of probiotics | 0.14 | 0.84 | ||||||||||||
| Breast milk and formula | 16 | 5791 | 0.00 | 13.06 | 0.44*** | 0.33, 0.60 | 13 | 3082 | 63.49*** | 32.88 | 0.75 | 0.54, 1.04 | ||
| Other | 7 | 2431 | 17.33 | 7.26 | 0.67 | 0.42, 1.07 | 9 | 3281 | 37.78 | 12.86 | 0.79 | 0.58, 1.07 | ||
| Gestational age, wk | 0.66 | 0.48 | ||||||||||||
| <29 | 9 | 2262 | 0.00 | 6.15 | 0.42*** | 0.28, 0.64 | 13 | 2179 | 28.51 | 16.79 | 0.70** | 0.56, 0.88 | ||
| ≥29 | 10 | 1684 | 0.00 | 8.81 | 0.47*** | 0.33, 0.69 | 9 | 1011 | 73.40*** | 22.55 | 0.77 | 0.43, 1.40 | ||
| Duration of probiotics, wk | 0.15 | 0.61 | ||||||||||||
| ≤6 | 14 | 2094 | 0.00 | 9.23 | 0.41*** | 0.28, 0.60 | 9 | 1559 | 66.81** | 24.10 | 0.63 | 0.39, 1.01 | ||
| >6 | 11 | 2261 | 20.72 | 12.61 | 0.60*** | 0.42, 0.87 | 7 | 1746 | 62.56** | 16.03 | 0.81 | 0.54, 1.22 | ||
If there were ≤2 studies in each subgroup, they were excluded. *P < 0.05, **P < 0.01, ***P < 0.001.
Effects on sepsis.
Twenty-four studies, with 7894 infant participants, measured rates of infant sepsis. The mean sepsis rate in the probiotics group was 15.1% compared with 18.5% in the control group following probiotics administration. The meta-analysis showed that there was a significantly lower risk ratio of 17% (95% CI: 0.71%, 0.96%; P = 0.02) in the probiotics group compared with the control group. The heterogeneity was significant (I2 = 48%, P = 0.01). The forest plot of the effect is presented in Figure 2B (see also Table 2).
Subgroup analysis (Table 3) indicated that the effect of probiotics on sepsis was statistically significant in reducing the incidence of sepsis when the study quality was moderate (P < 0.05) compared with high-quality studies (P > 0.05). It was also found that the effect of probiotics on sepsis was statistically significant in reducing its incidence when probiotics consumption was <109 CFU (P < 0.05) compared with >109 CFU (P > 0.05). A significant effect was also found in trials of infants with gestational age <29 wk (P < 0.01) compared with trials of infants with a gestational age of ≥29 wk (P > 0.05).
Effects on mortality.
Twenty-one studies with 7332 participants measured infant mortality. The mean mortality rate in the probiotics group was 6.8% compared with 8.8% in the control group. The meta-analysis showed that there was a significantly lower risk ratio in mortality of 22% (95% CI: 0.66%, 0.93%; P = 0.01) in the probiotics group compared with the control group. The heterogeneity was not significant (I2 = 7%, P = 0.37). The forest plot of the effect is presented in Figure 2C (see also Table 2).
Subgroup analysis (Table 4) found that the effect of the probiotics on mortality was statistically significant when trials were of high quality (P < 0.001) compared with those trials of moderate quality (P > 0.05), when the dosage was <109 CFU (P < 0.05) compared with a larger dosage of ≥109 CFU, when the probiotics consisted of multiple strains (P < 0.001) rather than a single strain, and when the trials used breast milk and formula (P < 0.01) compared with trials that used other forms (P > 0.05). A significant effect was also found in trials with infants of <29 wk gestation (P < 0.05) compared with trials with infants of >29 wk gestation and when the duration of probiotics administration was ≤6 wk (P < 0.05) compared with trials ≥6 wk (P > 0.05).
TABLE 4.
Subgroup analyses on the effects of probiotics on mortality and hospital stay1
| Mortality |
Hospital stay |
|||||||||||||
| Subgroups | Studies, n | Participants, n | I2 | Q-test | Risk ratio | 95% CI | P | Studies, n | Participants, n | I2 | Q-test | Mean difference, d | 95% CI | P |
| Quality of study | 0.03 | 0.13 | ||||||||||||
| High | 11 | 3080 | 4.99 | 10.53 | 0.58*** | 0.42, 0.80 | 11 | 1769 | 90.56*** | 95.61 | −2.32 | −4.62, 0.01 | ||
| Moderate | 10 | 834 | 0 | 5.81 | 0.89 | 0.74, 1.06 | 8 | 903 | 90.34*** | 72.06 | −6.32** | −10.97, −1.68 | ||
| Dosage | 0.96 | 0.10 | ||||||||||||
| <109 | 10 | 1685 | 16.84 | 10.82 | 0.75* | 0.57, 0.97 | 8 | 1402 | 85.03*** | 53.45 | −3.66** | −6.25, −1.08 | ||
| ≥109 | 10 | 2125 | 14.89 | 10.57 | 0.78 | 0.57, 1.08 | 8 | 1064 | 89.28*** | 74.63 | −4.15* | −8.18, −0.13 | ||
| Strains | 0.02 | 0.49 | ||||||||||||
| Single | 11 | 1844 | 0.0 | 2.54 | 0.94 | 0.75, 1.18 | 11 | 1063 | 58.42** | 24.05 | −3.00*** | −4.55, −1.46 | ||
| Multiple | 10 | 2340 | 41.75 | 15.45 | 0.58*** | 0.41, 0.81 | 6 | 1507 | 91.84*** | 85.81 | −6.14 | −6.90, −5.39 | ||
| Form of probiotics | 0.38 | 0.80 | ||||||||||||
| Breast milk and formula | 14 | 4542 | 13.06 | 14.95 | 0.74** | 0.59, 0.93 | 12 | 3692 | 89.09*** | 100.78 | −4.12** | −7.13, −1.12 | ||
| Other | 6 | 2660 | 0 | 4.94 | 0.86 | 0.66, 1.12 | 6 | 1525 | 92.89*** | 70.29 | −3.29* | −6.48, −0.09 | ||
| Gestational age, wk | 0.01 | 0.93 | ||||||||||||
| <29 | 11 | 2223 | 19.71 | 12.45 | 0.74* | 0.57, 0.97 | 9 | 1397 | 93.85*** | 130.18 | −3.82* | −7.09, −0.56 | ||
| ≥29 | 9 | 1383 | 0.00 | 7.39 | 0.73 | 0.54, 0.99 | 10 | 1173 | 78.01*** | 40.93 | −3.66 | −6.16, −1.15 | ||
| Duration of probiotics, wk | 0.32 | 0.07 | ||||||||||||
| ≤6 | 10 | 1735 | 8.52 | 10.93 | 0.69* | 0.53, 0.93 | 8 | 1081 | 94.90*** | 137.13 | −4.32* | −7.23, −1.41*** | ||
| >6 | 9 | 2026 | 3.69 | 8.31 | 0.84 | 0.69, 1.03 | 10 | 1370 | 72.88*** | 33.19 | −3.25 | −6.59, −0.08 | ||
If ≤2 studies in each subgroup, they were excluded. *P < 0.05, **P < 0.01, ***P < 0.001.
Effects on length of hospital stay.
Nineteen studies with 5443 infant participants measured length of hospital stay. The mean hospital stay time in the probiotics group was 48.07 d (SD: 20.82 d) compared with 50.40 d (SD: 21.65 d) in the control group. The meta-analysis demonstrated a significantly reduced hospital stay by 3.81 d (95% CI: −5.77; −1.86 d; P < 0.001) in the probiotics group compared with the control group. The heterogeneity was significant (I2 = 89%, P < 0.001). The forest plot of the effect is presented in Figure 2D (see also Table 2).
Subgroup analysis (Table 4) found that moderate-quality studies had a significantly shorter length of hospital stay of ≤6.32 d (95% CI: −10.97, −1.68 d; P < 0.01) compared with high-quality studies (−2.32 d; 95% CI: −4.62, 0.01 d; P > 0.05). Probiotics with dosages of <109 CFU had a significantly greater effect in reducing the hospital length of stay (−3.66 d; 95% CI: −6.25, −1.08 d; P < 0.01) compared with dosages ≤109 CFU (−2.2 d; 95% CI: −8.0, 3.7 d). Probiotics consisting of multiple strains had a significantly greater effect in reducing the length of hospital stay (−6.14 d; 95% CI: −6.90, −5.30 d; P < 0.001) compared with a single strain (−3.00 d; 95% CI: −4.55, −1.46 d; P > 0.05). Infants with a gestational age of <29 wk had a shorter length of hospital stay (−3.82 d; 95% CI: −7.09, −0.56 d; P < 0.05) compared with infants with a gestational age of ≥29 wk (−3.66 d; 95% CI: −6.16, −1.15 d). Infants who had <6 wk of probiotics consumption had a significantly shorter length of hospital stay (−4.32 d; 95% CI: −7.23, −1.41 d; P < 0.05) compared with infants who had ≥6 wk of probiotics consumption (−3.25 d; 95% CI: −6.59, −0.8 d; P > 0.05).
Effects on weight gain.
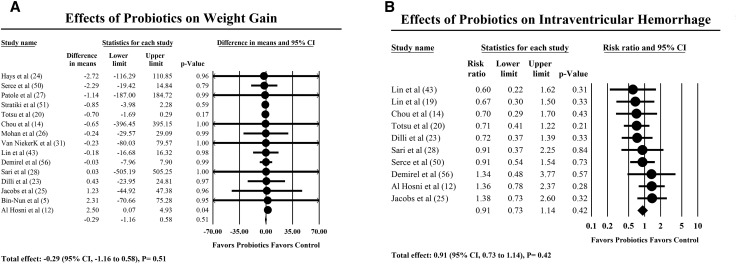
Fifteen studies with 3751 infant participants measured weight gain as an outcome variable. The mean weight gain in the probiotics group was 16.41 g/d compared with 16.57 g/d in the control group. The meta-analysis showed that there was a 0.29-g lower weight gain in the probiotics group (95% CI: −1.16, 0.58 g, P = 0.51) compared with the control group, but this difference was not statistically significant. The forest plot of the effect is presented in Figure 3A (see also Table 2). The heterogeneity was unmeasurable (I2 = 0%, P = 0.97).
FIGURE 3.
Effects of probiotics on weight gain (A) and intraventricular hemorrhage (B).
Subgroup analysis (Table 5) found that VLBW infants in the probiotics group who took breast milk or preterm formula had a significantly greater weight gain (mean weight gain = 2.20 g; 95% CI: 0.08, 4.48 g; P < 0.05) compared with those infants who received probiotics in other forms (weight gain = −0.89 g; 95% CI: −3.97, 2.18 g; P = 0.57). Other variables, including study quality, dosage amounts, number of probiotic strains, form of probiotics, and gestational age, had no effect on the weight gain.
TABLE 5.
Subgroup analyses on the effects of probiotics on everyday weight gain and intraventricular hemorrhage1
| Everyday weight gain |
Intraventricular hemorrhage |
|||||||||||||
| Subgroups | Studies, n | Participants, n | I2 | Q-test | Mean difference, g | 95% CI | P | Studies, n | Participants, n | I2 | Q-test | Risk ratio | 95% CI | P |
| Quality of study | 0.51 | 0.41 | ||||||||||||
| High | 7 | 1051 | 0.0 | 0.68 | 2.17 | −0.11, 4.45 | 5 | 934 | 0.00 | 1.66 | 0.86 | 0.60, 1.21 | ||
| Moderate | 8 | 829 | 0.0 | 0.02 | −0.71 | −1.65, 0.23 | 5 | 789 | 22.15 | 5.14 | 0.95 | 0.69, 1.31 | ||
| Dosage | 0.51 | 0.35 | ||||||||||||
| <109 | 5 | 816 | 0.0 | 2.75 | 1.24 | −0.68, 3.15 | 5 | 965 | 15.02 | 4.71 | 1.00 | 0.70, 1.43 | ||
| ≥109 | 9 | 960 | 0.0 | 0.05 | −0.69 | −1.66, 0.29 | 5 | 758 | 0.00 | 1.50 | 0.83 | 0.62, 1.12 | ||
| Strains | 0.51 | 0.42 | ||||||||||||
| Single | 9 | 749 | 0.0 | 0.08 | −0.71 | −1.64, 0.23 | 5 | 578 | 0.00 | 2.93 | 0.96 | 0.71, 1.28 | ||
| Multiple | 6 | 1131 | 0.0 | 0.11 | 2.44* | 0.04, 4.83 | 5 | 1145 | 0.00 | 3.90 | 0.86 | 0.62, 1.20 | ||
| Form of probiotics | 0.51 | 0.42 | ||||||||||||
| Breast milk and formula | 10 | 3025 | 0.0 | 0.50 | 2.20* | −0.08, 4.48 | 8 | 2766 | 0.41 | 7.03 | 0.92 | 0.71, 1.17 | ||
| Other | 3 | 459 | 0.0 | 0.03 | −0.89 | −3.97, 2.18 | 2 | 382 | 0 | 0 | 0.91 | 0.58, 1.44 | ||
| Birth weight, g | 0.51 | 0.37 | ||||||||||||
| ≥1000 | 12 | 1689 | 0.0 | 0.10 | −0.70 | −1.64, 0.23 | 9 | 1676 | 0.00 | 4.90 | 0.84 | 0.64, 1.10 | ||
| <1000 | ||||||||||||||
| Gestational age, wk | 0.51 | 0.42 | ||||||||||||
| <29 | 8 | 1242 | 0.0 | 5.79 | −0.25 | −1.16, 0.66 | 7 | 1285 | 0.00 | 4.62 | 0.88 | 0.69, 1.13 | ||
| ≥29 | 5 | 371 | 0.0 | 0.04 | −0.73 | −3.62, 2.16 | ||||||||
| Duration of probiotics, wk | 0.51 | 0.37 | ||||||||||||
| ≤6 | 8 | 690 | 0.0 | 2.96 | 1.17 | −0.72, 3.05 | 4 | 468 | 0.00 | 2.89 | 0.89 | 0.59, 1.32 | ||
| >6 | 6 | 1071 | 0.0 | 0.01 | 0.03 | −7.73, 7.79 | 5 | 1102 | 0.00 | 2.93 | 0.99 | 0.67, 1.46 | ||
If ≤2 studies in each subgroup, they were excluded. *P < 0.05.
Effects on IVH.
Ten studies, with 3431 infant participants, reported the rate of IVH. The mean IVH rate in the probiotics group was 7.77% compared with 8.49% in the control group. The meta-analysis showed that there was a lower risk ratio of 9% (95% CI: 0.73%, 1.14%; P = 0.42) in the probiotics group compared with the control group, but this finding was not statistically significant (P = 0.42). The heterogeneity was not significant (I2 = 0%, P = 0.63). The forest plot of the effect is presented in Figure 3B (see also Table 2).
Subgroup analysis (Table 5) found that study quality, dosage amounts, number of strains of probiotics, form of probiotics, infant gestational age, and duration of probiotics administration had no effect on the rate of IVH.
Publication bias.
The results for all factors except NEC showed minimal asymmetry, suggesting minimal publication bias. A visual inspection of the funnel plots showed no clear evidence of publication bias with regard to effects on NEC, sepsis, mortality, and length of hospital stay (), weight gain, or IVH (). Findings from the Egger’s test (NEC: P = 0.001; sepsis: P = 0.19; mortality: P = 0.07; length of hospital stay: P = 0.88; weight gain: P = 0.54; and IVH: P = 0.56) supported the finding that there was no publication bias, except for NEC. Applying the trim and fill method did not change the overall effects for NEC when 9 studies were excluded from the analysis (3, 5, 12, 14, 27, 29, 41, 47, 51). These tests suggest that there was no statistically significant evidence of publication bias for any of the assessed outcomes.
Discussion
The pooled effects based on the meta-analysis found that probiotics improved VLBW infants’ health outcomes, including reducing the incidence of major medical complications, such as NEC and sepsis, and reducing mortality and length of hospital stay. However, the effects of probiotics on weight gain and IVH were not statistically significant. These findings are important. With worldwide advances in neonatology and modern neonatal intensive care units, the incidence of NEC has increased in VLBW infants (55). These risk factors need to be addressed, because reductions in the risk of NEC and sepsis can significantly reduce the high mortality rate associated with a very preterm birth.
Our results are consistent with previous published reviews regarding the effects of probiotics on reducing the risk of NEC (16) and sepsis (17), subsequent mortality (16) and shortened length of hospital stay (18). However, unlike our study, previous reviews focused either on preterm infants with a gestational age <37 wk (17, 21), rather than VLBW infants or on 1 outcome variable (17). None of the studies provided subgroup analyses on key variables. Our study included RCT trials focusing on VLBW infants with a birthweight <1500 g or gestational age <32 wk. A subgroup analysis was also conducted to demonstrate the effect of probiotics administration in VLBW infants with a gestational age of <29 wk, and therefore, the data pertaining to this high-risk group could be extracted from the included studies.
Effects of probiotics on NEC, sepsis, mortality, and length of hospital stay.
Subgroup analyses revealed that the significant effects of probiotics on reducing the risk of NEC, sepsis, and mortality and reducing the length of hospital stay may be due to the effects of probiotics dosage, probiotic forms (i.e., breast milk, preterm formula, and formula), number of strains in the products, duration of probiotics intake, and preterm infants’ gestational age. Most of the trials used breast milk and formula (3, 5, 12, 14, 23–27, 39, 40–44, 46, 51, 53, 54, 56). Probiotics in breast milk form and preterm formula had a significant effect in enhancing weight gain. Probiotics in the breast milk and formula had significant pooled effects on the reduced risk of NEC and mortality, and probiotics in the other forms had no significant impact in reducing NEC and the risk of mortality. The heterogeneity of the studies was small, indicating the consistency of the findings in the effect of milk plus formula on the reduction of risk of medical complications and mortality and the effect of breast milk on enhancing weight gain.
The mechanism involved in the reduction of NEC during hospitalization may be related to the prevention of bacterial toxins and bacteria growth through “clearing” intestinal bacteria, the inhibition of potential pathogens to enhance immune response to the bacterial products’ host, and the reduction in the incidence of intestinal infections (57). Use of human breast milk significantly reduces the risk of NEC and sepsis in the neonatal intensive care unit (58). Other potential mechanisms by which probiotics in breast milk may protect high-risk infants from developing NEC and mortality include increased barriers to migrating bacteria and their products across the mucosa (15), competitive exclusion of potential pathogens (15), modification of host response to microbial products (19), augmentation of IgA mucosal responses, and upregulation of immune responses (20). Another possible mechanism may occur because probiotics can reduce the degree of inflammation of the immature intestine of VLBW infants by entering the enteral feeding stage early and reducing hypoxia-related intestinal injury (59).
Fourteen trials that used multiple probiotic strains showed a statistically significant reduction of NEC and mortality and enhanced VLBW infant weight gain, whereas trials with a single strain did not. These results are similar to the finding that trials that used multiple probiotic strains had greater effects compared with those that used a single probiotics strain on the reduction of NEC and mortality and increase in body weight (4). In the studies we reviewed, a number of general bacteria were used in probiotics, including Bifidobacterium infantis, Bifidobacterium lactis, Lactobacillus acidophilus, Lactobacillus plantarum, Bifidobacterium, Enterococcus, Streptococcus thermophilus, Lactobacillus bulgaricus, Streptococcus, and thermophiles. We found that B. infantis had the strongest effect on the reduction of risk of NEC, sepsis, and mortality. This may be because B. infantis can reduce diversity of the gut microbiota and pathogens, thereby optimizing its activity in the large intestine (60).
Probiotics in small dosages (<109 CFU) had a significant effect on the reduction of NEC, mortality, and length of hospital stay. The finding was confirmed in 13 studies for NEC and 8 studies for length of hospital stay. Probiotic supplementation in VLBW infants should be considered carefully to avoid overwhelming their immune system (1). Research recommends that VLBW infants be administered only 1.5 × 109 CFU/d of probiotics (61). This limitation is because infants in this population have impaired immune systems, poor nutrition, and frequent exposure to harmful microorganisms (1).
No significant differences were found on 2 variables, namely weight gain and rate of IVH. The lack of a significant difference in weight gain may be explained by measurement differences. In some studies, weight gain was measured daily (12), and in others, it was measured weekly (29) or when the study concluded, resulting in an averaging effect, which may mean that significant differences that occurred at particular times during the measurement period were missed. With regards to IVH, it should be noted that the number of VLBW infants with this condition included in the studies was insufficient to identify an effect. For example, there were only 6–8 infants in each of the studies cited (14, 19, 28, 42, 56) and a total of 279 with the IVH condition.
Strength and limitations.
Our review used a thorough and comprehensive search strategy to identify and assess the quality of articles included in the meta-analysis. To minimize reviewer bias, authors conducted each step of this review independently before collaborating to decide on the included studies. A strength of our meta-analysis is that it included long-term intervention RCT studies with large sample sizes [with the exception of Mohan et al (26) and Stratiki et al (51)], so the power of the analyses was sufficient to generate firm conclusions. Any missing data were collected after contacting pertinent authors, and therefore all possible data were included in this meta-analysis at the time of publication. All studies involved RCT trials with VLBW infants, and therefore the effects of the probiotics could be assessed. A subgroup analysis to demonstrate the effect of probiotics administration in infants <29 wk of gestational age was performed, and therefore the data pertaining to this high-risk group could be extracted from the included studies. This meta-analysis builds on published findings. For example, a meta-analysis (16), which included 19 studies (5338 VLBW infants) reported no clinical effects of probiotics on sepsis, whereas our analysis had 24 RCT trials (n = 8492) with sufficient sample sizes that found probiotics reduced sepsis in VLBW infants.
A limitation of the present study is that there was inconsistent use of strain types among some of the studies, with some studies using one strain and others using multiple strains, making it difficult to assess which specific strain or strains contributed to the effect of the probiotics. Further RCTs involving long-term interventions that consistently compare specific single compared with multiple strains of probiotics supplement regimens are required to clarify these findings.
Clinical implications.
The results of this study have identified that probiotics can provide a significant benefit to VLBW infant health, including a reduction of the incidence of major medical complications, such as NEC and sepsis, a reduction of mortality incidence, and a reduction in length of hospital stay. Considering these outcomes, the use of probiotics in neonatal intensive care units would be highly beneficial for VLBW infants to reduce the risk of developing medical complications, including NEC and sepsis. Through the provision of probiotics, taking into consideration appropriate dosage amounts, probiotic forms (i.e., breast milk, preterm formula, and formula), and multiple strains, VLBW infants would have the opportunity to strengthen their immune systems and optimize the amount of microbial activity in their large intestines. Finally, the results of this study highlight that probiotics provide positive outcomes for improving VLBW infants’ health outcomes and that gestational age is also an important consideration. However, due to the multifaceted nature of probiotics, it is recommended that the process of probiotics intake become integrated into VLBW infants’ usual care, providing them with ongoing monitoring and support.
In conclusion, the findings of this study suggest that supplementation with probiotics is effective in reducing the risk of major medical complications and the coexisting risk of mortality and shortens the length of hospital stay. Probiotics are more effective when taken in breast milk or breast milk plus formula form, consumed for <6 wk, administered at a dosage of <109 CFU/d, and include multiple strains. By managing the risks of medical complications and coexisting risk factors and through facilitating growth and enteral feeding, probiotics may provide an avenue to reduce the current high rates of mortality associated with NEC, sepsis, and delayed growth in VLBW infants.
Acknowledgments
The authors’ responsibilities were as follows—JS: conceptualized and designed the study, collected the data, conducted the statistical analysis, drafted, edited, submitted the manuscript, and critically revised the article; GM and MW: collected the data for the first version of the article; NB: conceptualized the study and critically reviewed, edited, and revised the manuscript; DE and PHG: conceptualized the study, critically reviewed and revised the manuscript, and interpreted the data; and all authors: approved the final manuscript as submitted and agreed to be accountable for all aspects of the work.
Footnotes
Abbreviations used: IVH, intraventricular hemorrhage; NEC, necrotizing enterocolitis; RCT, randomized controlled trial; VLBW, very low birth weight.
References
- 1.Behrman RE, Butler AS. Preterm birth: causes, consequences, and prevention. Washington (DC): National Academies Press; 2007. [PubMed] [Google Scholar]
- 2.Neu J, Walker WA. Necrotizing enterocolitis. N Engl J Med 2011;364:255–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Braga TD, da Silva GAP, de Lira PIC, de Carvalho Lima M. Efficacy of bifidobacterium breve and Lactobacillus casei oral supplementation on necrotizing enterocolitis in very-low-birth-weight preterm infants: a double-blind, randomized, controlled trial. Am J Clin Nutr 2011;93:81–6. [DOI] [PubMed] [Google Scholar]
- 4.Aceti A, Gori D, Barone G, Callegari ML, Di Mauro A, Fantini MP, Indrio F, Maggio L, Meneghin F, Morelli L, et al. ; Italian Society of Neonatology. Probiotics for prevention of necrotizing enterocolitis in preterm infants: systematic review and meta-analysis. Ital J Pediatr 2015;41:89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bin-Nun A, Bromiker R, Wilschanski M, Kaplan M, Rudensky B, Caplan M, Hammerman C. Oral probiotics prevent necrotizing enterocolitis in very low birth weight neonates. J Pediatr 2005;147:192–6. [DOI] [PubMed] [Google Scholar]
- 6.Stoll BJ, Hansen N, Fanaroff AA, Wright LL, Carlo WA, Ehrenkranz RA, Lemons JA, Donovan EF, Stark AR, Tyson JE, et al. Late-onset sepsis in very low birth weight neonates: the experience of the NICHD Neonatal Research Network. Pediatrics 2002;110:285–91. [DOI] [PubMed] [Google Scholar]
- 7.Al Rifai MT, Al Tawil KI. The neurological outcome of isolated PVL and severe IVH in preterm infants: is it fair to compare? Pediatr Neurol 2015;53:427–33. [DOI] [PubMed] [Google Scholar]
- 8.Blosky MA, Qian Z, Wood GC, Betoni J, Black L, Wary A, Mateer H, Stewart W. PS1–22: premature birth, initial hospital length of stay, and costs. Clin Med Res 2010;8:184. [Google Scholar]
- 9.Brooks B, Firek BA, Miller CS, Sharon I, Thomas BC, Baker R, Morowitz MJ, Banfield JF. Microbes in the neonatal intensive care unit resemble those found in the gut of premature infants. Microbiome 2014;2:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jacquot A, Neveu D, Aujoulat F, Mercier G, Marchandin H, Jumas-Bilak E, Picaud JC. Dynamics and clinical evolution of bacterial gut microflora in extremely premature patients. J Pediatr 2011;158:390–6. [DOI] [PubMed] [Google Scholar]
- 11.Gilliland SE, Morelli L, Reid G. Health and nutritional properties of probiotics in food including powder milk with live lactic acid bacteria [Internet]. Report of a joint FAO/WHO Expert consultation on evaluation of health and nutritional properties of probiotics in food including powder milk with live lactic acid bacteria. Rome; WHO; 2006. [cited 2017 May 30]. Available from: http://www.fao.org/3/a-a0512e.pdf.
- 12.Al-Hosni M, Duenas M, Hawk M, Stewart LA, Borghese RA, Cahoon M, Atwood L, Howard D, Ferrelli K, Soll R. Probiotics-supplemented feeding in extremely low-birth-weight infants. J Perinatol 2012;32:253. [DOI] [PubMed] [Google Scholar]
- 13.Soll RF. Probiotics: are we ready for routine use? Pediatrics 2010;125:1071–2. [DOI] [PubMed] [Google Scholar]
- 14.Chou IC, Kuo HT, Chang JS, Wu SF, Chiu HY, Su BH, Lin HC. Lack of effects of oral probiotics on growth and neurodevelopmental outcomes in preterm very low birth weight infants. J Pediatr 2010;156:393–6. [DOI] [PubMed] [Google Scholar]
- 15.Kitajima H, Sumida Y, Tanaka R, Yuki N, Takayama H, Fujimura M. Early administration of bifidobacterium breve to preterm infants: randomised controlled trial. Arch Dis Child Fetal Neonatal Ed 1997;76:F101–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.AlFaleh K, Anabrees J. Probiotics for prevention of necrotizing enterocolitis in preterm infants. Cochrane Database Syst Rev 2014;CD005496. [DOI] [PubMed] [Google Scholar]
- 17.Rao SC, Athalye-Jape GK, Deshpande GC, Simmer KN, Patole SK. Probiotic supplementation and late-onset sepsis in preterm infants: a meta-analysis. Pediatrics 2016;137:e20153684. [DOI] [PubMed] [Google Scholar]
- 18.Athalye-Jape G, Deshpande G, Rao S, Patole S. Benefits of probiotics on enteral nutrition in preterm neonates: a systematic review. Am J Clin Nutr 2014;100:1508–19. [DOI] [PubMed] [Google Scholar]
- 19.Lin HC, Hsu CH, Chen HL, Chung MY, Hsu JF, Lien RI, Tsao LY, Chen CH, Su BH. Oral probiotics prevent necrotizing enterocolitis in very low birth weight preterm infants: a multicenter, randomized, controlled trial. Pediatrics 2008;122:693–700. [DOI] [PubMed] [Google Scholar]
- 20.Totsu S, Yamasaki C, Terahara M, Uchiyama A, Kusuda S. bifidobacterium and enteral feeding in preterm infants: cluster‐randomized trial. Pediatr Int 2014;56:714–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Huang X, Lin J, Demner-Fushman D. Evaluation of PICO as a knowledge representation for clinical questions. AMIA Annu Symp Proc 2006;2006:359–63. [PMC free article] [PubMed] [Google Scholar]
- 22.Demirel G, Erdeve O, Celik IH, Dilmen U. Saccharomyces boulardii for prevention of necrotizing enterocolitis in preterm infants: a randomized, controlled study. Acta Paediatr 2013;102:e560–5. [DOI] [PubMed] [Google Scholar]
- 23.Dilli D, Aydin B, Fettah ND, Özyazıcı E, Beken S, Zenciroğlu A, Okumuş N, Özyurt BM, İpek MŞ, Akdağ A, et al. The propre-save study: effects of probiotics and prebiotics alone or combined on necrotizing enterocolitis in very low birth weight infants. J Pediatr 2015;166:545–51.e1. [DOI] [PubMed] [Google Scholar]
- 24.Hays S, Jacquot A, Gauthier H, Kempf C, Beissel A, Pidoux O, Jumas-Bilak E, Decullier E, Lachambre E, Beck L, et al. Probiotics and growth in preterm infants: a randomized controlled trial, PREMAPRO study. Clin Nutr 2016;35:802–11. [DOI] [PubMed] [Google Scholar]
- 25.Jacobs SE, Tobin JM, Opie GF, Donath S, Tabrizi SN, Pirotta M, Morley CJ, Garland SM. Probiotic effects on late-onset sepsis in very preterm infants: a randomized controlled trial. Pediatrics 2013;132:1055. [DOI] [PubMed] [Google Scholar]
- 26.Mohan R, Koebnick C, Schildt J, Mueller M, Radke M, Blaut M. Effects of bifidobacterium lactis Bb12 supplementation on body weight, fecal pH, acetate, lactate, calprotectin, and IgA in preterm infants. Pediatr Res 2008;64:418–22. [DOI] [PubMed] [Google Scholar]
- 27.Patole S, Keil AD, Chang A, Nathan E, Doherty D, Simmer K, Esvaran M, Conway P. Effect of bifidobacterium breve M-16V supplementation on fecal bifidobacteria in preterm neonates - a randomised double blind placebo controlled trial. PLoS One 2014;9: e89511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sari FN, Eras Z, Dizdar EA, Erdeve O, Oguz SS, Uras N, Dilmen U. Do oral probiotics affect growth and neurodevelopmental outcomes in very low birth weight preterm infants? Am J Perinatol 2012;29:579–86. [DOI] [PubMed] [Google Scholar]
- 29.Costalos C, Skouteri V, Gounaris A, Sevastiadou S, Triandafilidou A, Ekonomidou C, Kontaxaki F, Petrochilou V. Enteral feeding of premature infants with Saccharomyces boulardii. Early Hum Dev 2003;74:89–96. [DOI] [PubMed] [Google Scholar]
- 30.Roy A, Chaudhuri J, Sarkar D, Ghosh P, Chakraborty S. Role of enteric supplementation of probiotics on late-onset sepsis by candida species in preterm low birth weight neonates: a randomized, double blind, placebo-controlled trial. N Am J Med Sci 2014;6:50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Van Niekerk E, Kirsten GF, Nel DG, Blaauw R. Probiotics, feeding tolerance, and growth: a comparison between HIV-exposed and unexposed very low birth weight infants. Nutrition 2014;30:645–53. [DOI] [PubMed] [Google Scholar]
- 32.Verhagen AP, de Vet HC, de Bie RA, Kessels AG, Boers M, Bouter LM, Knipschild PG. The delphi list: a criteria list for quality assessment of randomized clinical trials for conducting systematic reviews developed by Delphi consensus. J Clin Epidemiol 1998;51:1235–41. [DOI] [PubMed] [Google Scholar]
- 33.Chrzanowska-Liszewska D, Seliga-Siwecka J, Kornacka MK. The effect of Lactobacillus rhamnosus GG supplemented enteral feeding on the microbiotic flora of preterm infants-double blinded randomized control trial. Early Hum Dev 2012;88:57–60. [DOI] [PubMed] [Google Scholar]
- 34.Havranek T, Al-Hosni M, Armbrecht E. Probiotics supplementation increases intestinal blood flow velocity in extremely low birth weight preterm infants. J Perinatol 2013;33:40–4. [DOI] [PubMed] [Google Scholar]
- 35.Wang C, Shoji H, Sato H, Nagata S, Ohtsuka Y, Shimizu T, Yamashiro Y. Effects of oral administration of bifidobacterium breve on fecal lactic acid and short-chain fatty acids in low birth weight infants. J Pediatr Gastroenterol Nutr 2007;44:252–7. [DOI] [PubMed] [Google Scholar]
- 36.Li Y, Shimizu T, Hosaka A, Kaneko N, Ohtsuka Y, Yamashiro Y. Effects of bifidobacterium breve supplementation on intestinal flora of low birth weight infants. Pediatr Int 2004;46:509–15. [DOI] [PubMed] [Google Scholar]
- 37.Hu XY, Zhou YX, Xu SZ, Lin YY. [Effects of probiotics on feeding intolerance in low birth weight premature infants. ] Zhongguo Dang Dai Er Ke Za Zhi 2010;12:693–5 (in Chinese). [PubMed] [Google Scholar]
- 38.Costeloe K, Hardy P, Juszczak E, Wilks M, Millar MR; Probiotics in Preterm Infants Study Collaborative Group. Bifidobacterium breve BBG-001 in very preterm infants: a randomised controlled phase 3 trial. Lancet 2016;387:649–60. [DOI] [PubMed] [Google Scholar]
- 39.Fernández-Carrocera LA, Solis-Herrera A, Cabanillas-Ayón M, Gallardo-Sarmiento RB, García-Pérez CS, Montaño-Rodríguez R, Echániz-Aviles MO. Double-blind, randomised clinical assay to evaluate the efficacy of probiotics in preterm newborns weighing less than 1500 g in the prevention of necrotising enterocolitis. Arch Dis Child Fetal Neonatal Ed 2013;98:F5–9. [DOI] [PubMed] [Google Scholar]
- 40.Hikaru U, Koichi S, Yayoi S, Hiromichi S, Hiroaki S, Yoshikazu O, Seigo S, Satoru N, Toshiaki S, Yuichiro Y. Bifidobacteria prevents preterm infants from developing infection and sepsis. Int J Probiotics Prebiotics 2010;5:33–6. [Google Scholar]
- 41.Kanic Z, Micetic Turk D, Burja S, Kanic V, Dinevski D. Influence of a combination of probiotics on bacterial infections in very low birthweight newborns. Wien Klin Wochenschr 2015;127:S210–5. [DOI] [PubMed] [Google Scholar]
- 42.Lin HC, Su BH, Chen AC, Lin TW, Tsai CH, Yeh TF, Oh W. Oral probiotics reduce the incidence and severity of necrotizing enterocolitis in very low birth weight infants. Pediatrics 2005;115:1–4. [DOI] [PubMed] [Google Scholar]
- 43.Lin PW, Nasr TR, Stoll BJ. Necrotizing enterocolitis: recent scientific advances in pathophysiology and prevention. Semin Perinatol 2008;32:70–82. [DOI] [PubMed] [Google Scholar]
- 44.Manzoni P, Mostert M, Leonessa ML, Priolo C, Farina D, Monetti C, Latino MA, Gomirato G. Oral supplementation with Lactobacillus casei subspecies rhamnosus prevents enteric colonization by Candida species in preterm neonates: a randomized study. Clin Infect Dis 2006;42:1735–42. [DOI] [PubMed] [Google Scholar]
- 45.Mihatsch WA, Vossbeck S, Eikmanns B, Hoegel J, Pohlandt F. Effect of bifidobacterium lactis on the incidence of nosocomial infections in very-low-birth-weight infants: a randomized controlled trial. Neonatology 2010;98:156–63. [DOI] [PubMed] [Google Scholar]
- 46.Oncel MY, Sari FN, Arayici S, Guzoglu N, Erdeve O, Uras N, Oguz SS, Dilmen U. Lactobacillus Reuteri for the prevention of necrotising enterocolitis in very low birthweight infants: a randomised controlled trial. Arch Dis Child Fetal Neonatal Ed 2014;99:F110–5. [DOI] [PubMed] [Google Scholar]
- 47.Rojas MA, Lozano JM, Rojas MX, Rodriguez VA, Rondon MA, Bastidas JA, Perez LA, Rojas C, Ovalle O, Garcia-Harker JE, et al. Prophylactic probiotics to prevent death and nosocomial infection in preterm infants. Pediatrics 2012;130:e1113–20. [DOI] [PubMed] [Google Scholar]
- 48.Rougé C, Piloquet H, Butel M-J, Berger B, Rochat F, Ferraris L, Des Robert C, Legrand A, de la Cochetière M-F, N’Guyen J-M, et al. Oral supplementation with probiotics in very-low-birth-weight preterm infants: a randomized, double-blind, placebo-controlled trial. Am J Clin Nutr 2009;89:1828–35. [DOI] [PubMed] [Google Scholar]
- 49.Samanta M, Sarkar M, Ghosh P, Ghosh JK, Sinha MK, Chatterjee S. Prophylactic probiotics for prevention of necrotizing Enterocolitis in very low birth weight newborns. J Trop Pediatr 2009;55:128–31. [DOI] [PubMed] [Google Scholar]
- 50.Serce O, Benzer D, Gursoy T, Karatekin G, Ovali F. Efficacy of Saccharomyces boulardii on necrotizing enterocolitis or sepsis in very low birth weight infants: a randomised controlled trial. Early Hum Dev 2013;89:1033–6. [DOI] [PubMed] [Google Scholar]
- 51.Stratiki Z, Costalos C, Sevastiadou S, Kastanidou O, Skouroliakou M, Giakoumatou A, Petrohilou V. The effect of a bifidobacter supplemented bovine milk on intestinal permeability of preterm infants. Early Hum Dev 2007;83:575–9. [DOI] [PubMed] [Google Scholar]
- 52.Chowdhury T, Ali MM, Hossain MM, Singh J, Yousuf AN, Yasmin F, Chowdhury FR. Efficacy of probiotics versus placebo in the prevention of necrotizing enterocolitis in preterm very low birth weight infants: a double-blind randomized controlled trial. J Coll Physicians Surg Pak 2016;26:770–4. [PubMed] [Google Scholar]
- 53.Manzoni P, Rinaldi M, Cattani S, Pugni L, Romeo MG, Messner H, Stolfi I, Decembrino L, Laforgia N, Vagnarelli F, et al. Bovine lactoferrin supplementation for prevention of late-onset sepsis in very low-birth-weight neonates: a randomized trial. JAMA 2009;302:1421–8. [DOI] [PubMed] [Google Scholar]
- 54.Manzoni P, Meyer M, Stolfi I, Rinaldi M, Cattani S, Pugni L, Romeo MG, Messner H, Decembrino L, Laforgia N, et al. Bovine lactoferrin supplementation for prevention of necrotizing enterocolitis in very-low-birth-weight neonates: a randomized clinical trial. Early Hum Dev 2014;90:S60–5. [DOI] [PubMed] [Google Scholar]
- 55.Henry MCW, Moss R. Necrotizing enterocolitis. Annu Rev Med 2009;60:111–24. [DOI] [PubMed] [Google Scholar]
- 56.Demirel G, Celik IH, Erdeve O, Saygan S, Dilmen U, Canpolat FE. Prophylactic Saccharomyces boulardii versus nystatin for the prevention of fungal colonization and invasive fungal infection in premature infants. Eur J Pediatr 2013;172:1321–6. [DOI] [PubMed] [Google Scholar]
- 57.Ren YF, Wang LL. Effects of probiotics on intestinal bacterial colonization in premature infants. Chin Comtemp Pediatr 2010;12:192–4. [PubMed] [Google Scholar]
- 58.Meier PP, Bode L. Health, nutrition, and cost outcomes of human milk feedings for very low birthweight infants. Adv Nutr 2013;4:670–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hsueh W, Caplan MS, Qu XW, Tan XD, De Plaen IG, Gonzalez-Crussi F. Neonatal necrotizing enterocolitis: clinical considerations and pathogenetic concepts. Pediatr Dev Pathol 2003;6:6–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Underwood MA, German JB, Lebrilla CB, Mills DA. Bifidobacterium longum subspecies infantis: champion colonizer of the infant gut. Pediatr Res 2015;77:229–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Deshpande GC, Rao SC, Keil AD, Patole SK. Evidence-based guidelines for use of probiotics in preterm neonates. BMC Med 2011;9:92. [DOI] [PMC free article] [PubMed] [Google Scholar]