Introduction
Atopic dermatitis (also known as atopic eczema or eczema) is by definition a chronic condition. The original diagnostic criteria proposed by Hanifin and Rajka and the most recent guidelines issued by the American Academy of Dermatology both include a “chronic or relapsing” history as an essential feature.1,2 Little is written, however about the clinical course of AD.3 This could be because many patients with AD present with symptoms early in life, and hence most research has focused on pediatric disease. Though there are relatively few publications about adult disease, recent population-based estimates of AD prevalence among US children and adults were similar.4 These data suggest either AD begins in childhood and persists through adulthood, childhood AD remits for some and begins in adulthood for others, or some combination thereof. A number of practical and methodological challenges to studying a chronic episodic condition have limited the description of the long-term course of AD. In the following sections, we review the available data and discuss the implications for clinical care and future research.
Why is the long-term course of AD important to study?
Data about the long-term course of AD are necessary for informing clinicians and patients about prognosis and guiding treatment decisions at an individual level and for planning at the health systems level. Traditionally, AD was considered a pediatric condition and families were told most children “outgrow” AD by adolescence. Such imprecise information is insufficient for patients who desire detailed prognostic data. Moreover, it is insufficient for understanding the impact of potentially disease-modifying interventions, a topic of particular salience in the current era of systemic drug development.
When does AD begin?
The symptoms of AD may begin at any age, though many sources suggest that most incident AD occurs in early childhood. It is important to consider estimates may be affected by study designs that focus only on pediatric or clinic populations or by the diagnostic criteria used. For example, a recent review in the Lancet states “in roughly 60% of cases, the disease manifests during the first year of life,” citing a prospective study that follows patients until age 7 and a retrospective cohort of clinic patients.6,7,8 Studies including only children may underestimate the average age of disease onset because they would not capture adult-onset cases. Similarly, clinic-based samples are likely biased towards patients with more persistent or severe disease that begins earlier in life, and may not be not be representative of the general AD population. Additionally, some commonly used diagnostic criteria, including the Hanifin and Rajka criteria and the UK Working Party Criteria, include onset in childhood as a minor criterion.2,9 Studies using these criteria may estimate lower rates of adult disease compared to studies that do not select patients based on age of onset. Data that are less likely to be susceptible to selection bias suggest that AD may commonly begin in adulthood. For example, a population-based survey in the US found that 54% of those with AD reported disease onset after age 18.10 More data are needed to understand disease incidence over the lifespan and whether adult-onset disease is different from disease that begins in childhood.
Does AD permanently resolve, and if so, when?
Existing data are unable to answer this question. An older review found that 50-70% of individuals with AD improved over 10 years of follow-up, though the definition of clearance varied by study and ranged from 11-92%.3 Population-based birth cohort studies with multiple assessments of individuals over 2 decades found that rates of ‘short-term’ or ‘apparent’ clearance decreased when accounting for subsequent recurrences using estimates of annual period prevalence repeated every 3-7 years.11-14 More frequent measurement of disease activity every 6 months in a US cohort of children and young adults with AD who had prior treatment with a topical calcineurin inhibitor (and therefore may be more likely to have persistent disease) suggests that while some patients seem to improve with age, most continue to have active disease at multiple time points.15 Longitudinal studies that follow individuals throughout adulthood are needed to better understand the periodicity of disease activity and patterns over the life course.
Is AD a lifelong condition?
Genetic and physiologic data support the idea of AD as a lifelong condition. It is well established that AD runs in families, and in the past decade genetic discoveries have implicated multiple genes involved in the development and maintenance of the skin barrier and in immune function in AD heritability.16,17 Patients with AD often have xerosis that predates their diagnosis. In fact, “a history of generally dry skin from birth” was found to be one of the most predictive characteristics and hence was included among the minimum set of discriminators in the UK Working Party diagnostic criteria.9 Although the evidence is mixed,18 some studies have shown that physiologic differences such as transepidermal water loss (TEWL) may precede the clinical manifestations of AD and are detectable as early as day 2 after birth.19,20 Moreover, experimental evidence from two randomized trials suggest that maintenance of the skin barrier via application of moisturizer during the neonatal period may delay or prevent the development of clinical signs of AD.21,22
These data suggest that individuals with AD have an elevated probability of developing clinical symptoms throughout life, as illustrated in Figure 1. Even normal-appearing skin in patients with AD has evidence of differences in skin barrier function, dendritic cell population, and cytokine profiles, supporting the concept of subclinical disease23 The factors influencing the transition to clinically evident disease (both disease onset and disease flares) are an area of active research: investigators have explored the modulation of immune priming by hygiene, environmental factors, psychosocial factors, and diet.24-26 Early disease onset, severe early disease, family history of atopic disease, female sex, low income, Black race, and history of filaggrin mutations, have all been associated with more active or prolonged disease. 3,27-29 Recent data also suggest the skin microbiome appears to be different in both lesional and non-lesional skin between young children and teenagers/adults with AD.33 There is little consensus, however about the causes of disease onset or progression, and there are few studies examining factors that result in long-term or permanent disease remission.
Figure 1. Hypothetical example illustrating the long-term course of AD.
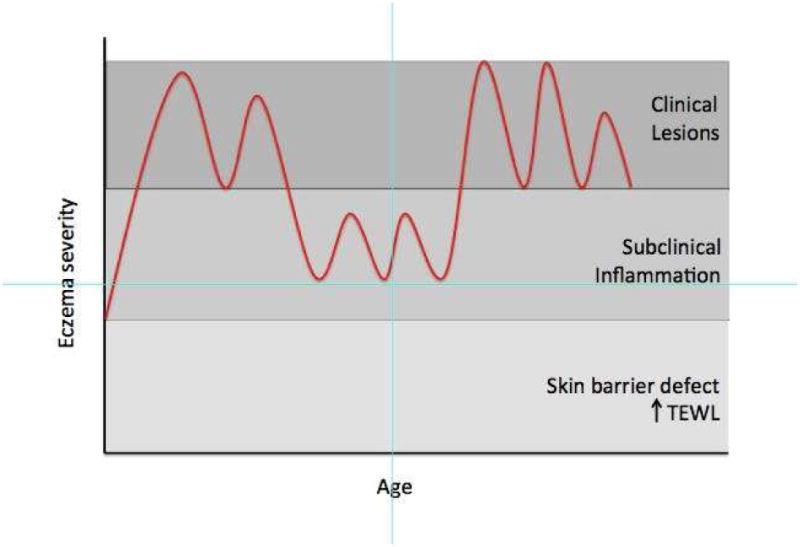
Notes: TEWL Transepidermal water loss
Some authors have suggested that there may be phases though which individuals are likely to pass including the development of “auto-sensitization” and eventually, chronic “auto-immune” disease.30 Whether eczema predisposes to asthma and allergies, a phenomenon termed the ‘atopic march,’ is also a matter of active debate.31,32 Furthermore, it is unclear whether treatment of AD might affect the long term clinical course or prevent the development of comorbid conditions, though there are some limited evidence to suggest that good control of eczema may have beneficial effects on respiratory disease as well.30 More data are needed to understand whether the onset and persistence of AD symptoms is part of a causal pathway to progressive disease, or whether barrier and immune dysfunction are always present and symptoms simply emerge when an individual encounters triggers throughout life.
Challenges to studying the long-term course of AD
The chronic and episodic nature of AD renders it particularly challenging to study its long-term course. We discuss a number of epidemiologic concepts and their application to AD below and in Table 1.
Table 1. Methodological considerations for studying the long-term course of AD.
| Concept | Considerations | Recommendations |
|---|---|---|
| Incidence | Must be chronic to meet most diagnostic criteria | Specify minimum duration of symptoms required for diagnosis (4 weeks has been proposed) |
| Prevalence | Dependent on time period and definition | Specify method of ascertainment (i.e. physical exam vs self-report) and definition used |
| Remission/ Persistence | Dependent on length of follow-up and method of detection | Avoid using these terms; specify age of patient and duration of follow up without treatment and/or symptoms |
| Flare | Dependent on patient's baseline; difficult to ascertain exact duration | Clearly define measure(s) used and delineate disease activity from disease severity. Consider combination of patient-reported outcome and severity score or a behavioral measure such as escalation of treatment |
| Long-term control | No consensus on measurement | Clearly define measure used. Consider use of well-controlled weeks |
Incidence
Incidence refers to the rate of new cases of a disease over a specified time period. However, AD may be difficult to diagnose very early in its course because its presentation is clinically heterogeneous. Diagnostic criteria for AD often include a measure of chronicity, complicating the decision of when to define an incident case. A systematic review of prevention studies found that in practice, there is a large degree of variability in the methods used to define an incident case of AD, and 27% of studies did not report any definition at all.34 The authors propose a modification to the UK Working Party criteria for AD that requires a history of an itchy skin condition that is either continuous or intermittent for at least four weeks. Longitudinal cohort studies that examine AD should be careful to explicitly state how the date of onset is defined and to ensure that their methods do not introduce immortal time bias.
Prevalence
Prevalence is typically defined as the proportion of a population with the condition of interest. Many individuals with AD have periods during which they may have no visible skin lesions and/or bothersome symptoms. For conditions that are intermittent, a point prevalence based on a single examination may underestimate the condition's frequency. Therefore, a period prevalence, defined as the proportion of cases within a specified time frame, is often preferred. The ISAAC studies found self-reported annual period prevalence was 2-3× higher than prevalence based on physical exam in many settings.35
The duration of symptom-free periods among patients with AD is poorly understood and therefore it is unclear what frequency of assessment is necessary to capture those with intermittent disease. Some studies report lifetime prevalence, though such estimates should be interpreted with caution based on the age of the sample and method of reporting. Studies of older patients may be prone to recall bias (i.e. they may not remember disease which was active only in early life), while studies of younger patients will miss those with adult-onset disease. Moreover, given the increase in awareness of allergic diseases over the past few decades, there is the possibility for increased reporting over time, especially of more mild cases.
Remission and persistence
Conditions such as cancer are considered to be ‘in remission’ when they become undetectable. This concept has been applied to AD, but we warrant caution in the use of this term given the episodic nature of the condition and complexities of defining inactive disease. In AD, there are many ways inactive disease might be defined including normal skin appearance, cessation of symptoms such as itch, discontinuation of treatment, and biomarkers or tests of barrier function. It is unclear at present what the optimal approach to defining AD remission is, and whether there is a minimum time period during which AD must be undetectable for remission to be defined. Some individuals may have recurrence of skin lesions after decades without disease.
Long periods of latency add confusion around the distinction between atopic dermatitis and other types of dermatitis in adults. Patients with AD have both disrupted barrier and immune mechanisms that may potentiate them toward increased rates of allergic and irritant contact dermatitis, and it may be difficult to differentiate these conditions from AD disease activity.36,37
Conversely, many studies refer to disease ‘persistence’; yet few define what is meant by this term.38 Often, persistence is used to refer to whether patients who are followed longitudinally report symptoms at subsequent follow-up, a definition that is highly dependent on the selection of the cohort, timing of follow up, and measure of disease activity. Ideally, a readily measurable biomarker would enable prediction of lesion and symptom recurrence, though attempts to date have not elucidated useful biomarkers for either diagnosis or prognosis.39 Until better prospective data with clear markers of disease activity that can be used to define ‘remission’ and ‘persistence’ are available, we recommend that investigators avoid using these terms and instead clearly define what is being measured.
Flare
Although the term flare is commonly used to refer to disease worsening or exacerbation, many studies define flares differently.40,41 A recent systematic review highlighted that amongst 26 studies incorporating AD flares as outcome measures, 21 different definitions were used and only 4 studies incorporated a patient reported outcome. Three broad approaches have been used by investigators to define AD flares: arbitrary cut offs on severity scales, composite measures (including symptoms and signs of AD) and behavioral approaches. The challenges with these approaches are varied. Firstly, using an arbitrary cut off of an “objective” severity score may be attractive as it potentially reduces information bias, however it does so at the expense of not having a patient reported outcome, and it also adds expense and logistic challenges as it requires physician review at short notice. Secondly, using a composite measure is appealing as it includes multiple dimensions, however, interpreting the meaning of the flare definition may be difficult. For the behavioral approach, the advantages include these usually being patient reported and hence associated with face validity, however, there is considerable discussion in the eczema literature about avoidance of topical corticosteroid use, hence behavior may not totally capture disease activity.42 A validation study found that ‘use of topical anti-inflammatory medications’ and ‘escalation of treatment’ were reasonable approaches to capturing flares, and that the former outcome was feasible to collect in longer term studies.41
Given the variability of flare definitions, it is difficult to summarize patterns of disease activity. One estimate comes from the International Study of Life with Atopic Eczema (ISOLTE) trial that included 2000 patients with moderate-to-severe disease found that patients spend, on average 1 of 3 days in flare (average 9 flares/ year lasting 15 days each time), with flare defined as a “sudden worsening of symptoms requiring a physician consultation or application of prescription medication.”43 This study included patients from 8 countries, but recruited individuals from physicians offices and therefore may not be representative of the average individual with AD.
Measures of long-term control
Given the challenges inherent in studying a chronic episodic disease, how might future studies measure long-term disease control? A recent systematic review aiming to identify how long-term control was captured in published randomized controlled trials found little consistency and highlighted the need for standardized measures.44 Methods used included repeated measures of patient- or clinician-reported outcomes (such as severity, pruritus, sleep quality of life, affected body surface area, or global assessment scores), changes in medication use, and flares. Time to next disease exacerbation or number of disease flares have been proposed but depend on clear delineation of the start and end of a flare and may require frequent measurement. The number of totally and well-controlled weeks has also been recommended as a possible approach to assessing long-term control in AD.45 Defining long-term control in AD is recommended as a core outcome domain for clinical studies of AD, but there is currently no widely accepted or validated approach to defining this outcome.3 The Harmonizing Outcome Measures in Eczema initiative has prioritized this topic as a focus for their 2017 consensus meeting.
Long-term treatment
Management approaches should address the intermittent and chronic nature of AD. Much research has focused on “reactive” approaches that treat disease flares or periods of acute worsening. Newer guidelines emphasize the role of “proactive” approaches with continued use of either topical corticosteroids (1-2 times/week) or topical calcineurin inhibitors (2-3 times/ week) after disease stabilization to previously involved skin to reduce subsequent flares or relapses.1 Structured education programs and avoidance if there is evidence of a true IgE-mediated allergy are also recommended based on the highest level of evidence. Gaps in research include comparative studies to decide on the best agents for long-term maintenance therapy, long-term safety data for intermittent use of topical steroids and calcineurin inhibitors, and high-quality evidence for the long-term use of systemic therapies in AD.1,46 Additionally, a high priority for future research is to determine whether treatments may modify the clinical course and/or prevent the development of other allergic outcomes.
Conclusions
The long-term course of AD is an important area for additional research. We have highlighted a number of methodological considerations that warrant careful attention in study design and reporting. Defining the clinical course of AD provides the foundation for understanding the results of interventional studies and establishing whether new treatments might modify the course of disease. Moreover, an understanding of the long-term course of disease can help contextualize trial findings. If some patients are more likely to have persistent disease, trials should report the relevant baseline characteristics and consider pre-specified subgroup analyses (such as initial age of disease onset, duration of active disease prior to baseline, gender, and race/ethnicity). Finally, understanding associations with the long-term course of disease would help clinicians and patients better understand their prognosis and design comprehensive treatment programs.
Key Points.
AD may be a lifelong condition marked by intermittent disease activity; little is known about the variability in disease activity over the long-term.
AD incidence, prevalence, persistence, remission, flare, and long-term control require careful definition because AD is an episodic condition disease.
Synopsis.
Atopic dermatitis (AD) is a chronic, relapsing condition meaning that the intensity of symptoms usually fluctuates over time. AD is often diagnosed in childhood, though the onset of symptoms may occur at any time. Changes in skin physiology may be evident from birth even in the absence of visual changes in the skin suggesting that AD may in fact be a lifelong condition marked by intermittent symptoms/disease activity. Data from longitudinal studies suggest that some individuals with symptom onset in childhood improve with age, though many subsequently experience recurrence of their symptoms if followed for long enough. The frequency and duration of active disease periods remains poorly understood and is an area ripe for additional research. Methodological challenges to studying a chronic episodic condition are reviewed in detail. Efforts are underway to improve measurement of long-term disease control and should help to elucidate more about the clinical course of disease and the role of treatment in long-term outcomes.
Footnotes
Disclosure Statement: The Authors have nothing to disclose.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Contributor Information
Katrina Abuabara, Department of Dermatology, University of California San Francisco (UCSF), San Francisco, CA.
David J Margolis, Faculty of Epidemiology & Population Health, London School of Hygiene and Tropical Medicine, London, UK.
Sinéad M Langan, Departments of Dermatology and of Biostatistics and Epidemiology, University of Pennsylvania Perelman School of Medicine, Philadelphia, PA.
References
- 1.Eichenfield LF, Tom WL, Chamlin SL, et al. Guidelines of care for the management of atopic dermatitis: section 1. Diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70(2):338–351. doi: 10.1016/j.jaad.2013.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hanifin JM, R G. Diagnostic features of atopic dermatitis. Acta Derm Venereol. 1980;92(suppl):44–47. [Google Scholar]
- 3.Williams HC, W B. Atopic Dermaittis: The epidemiology, causes and prevention of atopic eczema. United Kingdom: Cambridge University Press; 2000. The natural history of atopic dermatitis. [Google Scholar]
- 4.Abuabara K, Margolis DJ. Do children really outgrow their eczema, or is there more than one eczema? J Allergy Clin Immunol. 2013;132(5):1139–1140. doi: 10.1016/j.jaci.2013.08.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hay RJ, Johns NE, Williams HC, et al. The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J Invest Dermatol. 2014;134(6):1527–1534. doi: 10.1038/jid.2013.446. [DOI] [PubMed] [Google Scholar]
- 6.Weidinger S, Novak N. Atopic dermatitis. Lancet. 2016;387(10023):1109–1122. doi: 10.1016/S0140-6736(15)00149-X. [DOI] [PubMed] [Google Scholar]
- 7.Illi S, von Mutius E, Lau S, et al. The natural course of atopic dermatitis from birth to age 7 years and the association with asthma. J Allergy Clin Immunol. 2004;113(5):925–931. doi: 10.1016/j.jaci.2004.01.778. [DOI] [PubMed] [Google Scholar]
- 8.Garmhausen D, Hagemann T, Bieber T, et al. Characterization of different courses of atopic dermatitis in adolescent and adult patients. Allergy. 2013;68(4):498–506. doi: 10.1111/all.12252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Williams HC, Burney PG, Hay RJ, et al. The U.K. Working Party's Diagnostic Criteria for Atopic Dermatitis. I. Derivation of a minimum set of discriminators for atopic dermatitis. Br J Dermatol. 1994;131(3):383–396. doi: 10.1111/j.1365-2133.1994.tb08530.x. [DOI] [PubMed] [Google Scholar]
- 10.Hanifin JM, Reed ML, Eczema P, Impact Working G A population-based survey of eczema prevalence in the United States. Dermatitis. 2007;18(2):82–91. doi: 10.2310/6620.2007.06034. [DOI] [PubMed] [Google Scholar]
- 11.Ballardini N, Kull I, Lind T, et al. Development and comorbidity of eczema, asthma and rhinitis to age 12: data from the BAMSE birth cohort. Allergy. 2012;67(4):537–544. doi: 10.1111/j.1398-9995.2012.02786.x. [DOI] [PubMed] [Google Scholar]
- 12.Burr ML, Dunstan FD, Hand S, Ingram JR, Jones KP. The natural history of eczema from birth to adult life: a cohort study. Br J Dermatol. 2013;168(6):1339–1342. doi: 10.1111/bjd.12216. [DOI] [PubMed] [Google Scholar]
- 13.Williams HC, Strachan DP. The natural history of childhood eczema: observations from the British 1958 birth cohort study. Br J Dermatol. 1998;139(5):834–839. doi: 10.1046/j.1365-2133.1998.02509.x. [DOI] [PubMed] [Google Scholar]
- 14.Ziyab AH, Raza A, Karmaus W, et al. Trends in eczema in the first 18 years of life: results from the Isle of Wight 1989 birth cohort study. Clin Exp Allergy. 2010;40(12):1776–1784. doi: 10.1111/j.1365-2222.2010.03633.x. [DOI] [PubMed] [Google Scholar]
- 15.Abuabara K, Hoffstad O, Troxel A, Gelfand JM, Margolis DJ. Atopic dermatitis disease control and age: A cohort study. J Allergy Clin Immunol. 2015;136(1):190–192 e193. doi: 10.1016/j.jaci.2015.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Paternoster L, Standl M, Waage J, et al. Multi-ancestry genome-wide association study of 21,000 cases and 95,000 controls identifies new risk loci for atopic dermatitis. Nat Genet. 2015;47(12):1449–1456. doi: 10.1038/ng.3424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Elmose C, Thomsen SF. Twin Studies of Atopic Dermatitis: Interpretations and Applications in the Filaggrin Era. J Allergy (Cairo) 2015;2015:902359. doi: 10.1155/2015/902359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kikuchi K, Kobayashi H, O'Goshi K, Tagami H. Impairment of skin barrier function is not inherent in atopic dermatitis patients: a prospective study conducted in newborns. Pediatr Dermatol. 2006;23(2):109–113. doi: 10.1111/j.1525-1470.2006.00191.x. [DOI] [PubMed] [Google Scholar]
- 19.Kelleher M, Dunn-Galvin A, Hourihane JO, et al. Skin barrier dysfunction measured by transepidermal water loss at 2 days and 2 months predates and predicts atopic dermatitis at 1 year. J Allergy Clin Immunol. 2015;135(4):930–935 e931. doi: 10.1016/j.jaci.2014.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 20.Flohr C, England K, Radulovic S, et al. Filaggrin loss-of-function mutations are associated with early-onset eczema, eczema severity and transepidermal water loss at 3 months of age. Br J Dermatol. 2010;163(6):1333–1336. doi: 10.1111/j.1365-2133.2010.10068.x. [DOI] [PubMed] [Google Scholar]
- 21.Simpson EL, Chalmers JR, Hanifin JM, et al. Emollient enhancement of the skin barrier from birth offers effective atopic dermatitis prevention. J Allergy Clin Immunol. 2014;134(4):818–823. doi: 10.1016/j.jaci.2014.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Horimukai K, Morita K, Narita M, et al. Application of moisturizer to neonates prevents development of atopic dermatitis. J Allergy Clin Immunol. 2014;134(4):824–830 e826. doi: 10.1016/j.jaci.2014.07.060. [DOI] [PubMed] [Google Scholar]
- 23.Tang TS, Bieber T, Williams HC. Are the concepts of induction of remission and treatment of subclinical inflammation in atopic dermatitis clinically useful? J Allergy Clin Immunol. 2014;133(6):1615–1625 e1611. doi: 10.1016/j.jaci.2013.12.1079. [DOI] [PubMed] [Google Scholar]
- 24.Flohr C, Mann J. New insights into the epidemiology of childhood atopic dermatitis. Allergy. 2014;69(1):3–16. doi: 10.1111/all.12270. [DOI] [PubMed] [Google Scholar]
- 25.Langan SM, Bourke JF, Silcocks P, Williams HC. An exploratory prospective observational study of environmental factors exacerbating atopic eczema in children. Br J Dermatol. 2006;154(5):979–980. doi: 10.1111/j.1365-2133.2006.07153.x. [DOI] [PubMed] [Google Scholar]
- 26.Langan SM, Silcocks P, Williams HC. What causes flares of eczema in children? Br J Dermatol. 2009;161(3):640–646. doi: 10.1111/j.1365-2133.2009.09320.x. [DOI] [PubMed] [Google Scholar]
- 27.Margolis DJ, Gupta J, Apter AJ, et al. Filaggrin-2 variation is associated with more persistent atopic dermatitis in African American subjects. J Allergy Clin Immunol. 2014;133(3):784–789. doi: 10.1016/j.jaci.2013.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Margolis DJ, Apter AJ, Gupta J, et al. The persistence of atopic dermatitis and filaggrin (FLG) mutations in a US longitudinal cohort. J Allergy Clin Immunol. 2012;130(4):912–917. doi: 10.1016/j.jaci.2012.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Margolis JS, Abuabara K, Bilker W, Hoffstad O, Margolis DJ. Persistence of mild to moderate atopic dermatitis. JAMA Dermatol. 2014;150(6):593–600. doi: 10.1001/jamadermatol.2013.10271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bieber T, Cork M, Reitamo S. Atopic dermatitis: a candidate for disease-modifying strategy. Allergy. 2012;67(8):969–975. doi: 10.1111/j.1398-9995.2012.02845.x. [DOI] [PubMed] [Google Scholar]
- 31.Bantz SK, Zhu Z, Zheng T. The Atopic March: Progression from Atopic Dermatitis to Allergic Rhinitis and Asthma. J Clin Cell Immunol. 2014;5(2) doi: 10.4172/2155-9899.1000202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kapoor R, Menon C, Hoffstad O, Bilker W, Leclerc P, Margolis DJ. The prevalence of atopic triad in children with physician-confirmed atopic dermatitis. J Am Acad Dermatol. 2008;58(1):68–73. doi: 10.1016/j.jaad.2007.06.041. [DOI] [PubMed] [Google Scholar]
- 33.Shi B, Bangayan NJ, Curd E, et al. The skin microbiome is different in pediatric versus adult atopic dermatitis. J Allergy Clin Immunol. 2016 doi: 10.1016/j.jaci.2016.04.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Simpson EL, Keck LE, Chalmers JR, Williams HC. How should an incident case of atopic dermatitis be defined? A systematic review of primary prevention studies. J Allergy Clin Immunol. 2012;130(1):137–144. doi: 10.1016/j.jaci.2012.01.075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Flohr C, Weinmayr G, Weiland SK, et al. How well do questionnaires perform compared with physical examination in detecting flexural eczema? Findings from the International Study of Asthma and Allergies in Childhood (ISAAC) Phase Two. Br J Dermatol. 2009;161(4):846–853. doi: 10.1111/j.1365-2133.2009.09261.x. [DOI] [PubMed] [Google Scholar]
- 36.Gittler JK, Krueger JG, Guttman-Yassky E. Atopic dermatitis results in intrinsic barrier and immune abnormalities: implications for contact dermatitis. J Allergy Clin Immunol. 2013;131(2):300–313. doi: 10.1016/j.jaci.2012.06.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mowad CM, Anderson B, Scheinman P, Pootongkam S, Nedorost S, Brod B. Allergic contact dermatitis: Patient diagnosis and evaluation. J Am Acad Dermatol. 2016;74(6):1029–1040. doi: 10.1016/j.jaad.2015.02.1139. [DOI] [PubMed] [Google Scholar]
- 38.Kim JP, Chao LX, Simpson EL, Silverberg JI. Persistence of atopic dermatitis (AD): A systematic review and meta-analysis. J Am Acad Dermatol. 2016;75(4):681–687 e611. doi: 10.1016/j.jaad.2016.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Thijs J, Krastev T, Weidinger S, et al. Biomarkers for atopic dermatitis: a systematic review and meta-analysis. Curr Opin Allergy Clin Immunol. 2015;15(5):453–460. doi: 10.1097/ACI.0000000000000198. [DOI] [PubMed] [Google Scholar]
- 40.Langan SM, Thomas KS, Williams HC. What is meant by a “flare” in atopic dermatitis? A systematic review and proposal. Arch Dermatol. 2006;142(9):1190–1196. doi: 10.1001/archderm.142.9.1190. [DOI] [PubMed] [Google Scholar]
- 41.Thomas KS, Stuart B, O'Leary CJ, et al. Validation of treatment escalation as a definition of atopic eczema flares. PLoS One. 2015;10(4):e0124770. doi: 10.1371/journal.pone.0124770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Charman CR, Morris AD, Williams HC. Topical corticosteroid phobia in patients with atopic eczema. Br J Dermatol. 2000;142(5):931–936. doi: 10.1046/j.1365-2133.2000.03473.x. [DOI] [PubMed] [Google Scholar]
- 43.Zuberbier T, Orlow SJ, Paller AS, et al. Patient perspectives on the management of atopic dermatitis. J Allergy Clin Immunol. 2006;118(1):226–232. doi: 10.1016/j.jaci.2006.02.031. [DOI] [PubMed] [Google Scholar]
- 44.Barbarot S, Rogers NK, Abuabara K, et al. Strategies used for measuring long-term control in atopic dermatitis trials: A systematic review. J Am Acad Dermatol. 2016 doi: 10.1016/j.jaad.2016.05.043. [DOI] [PubMed] [Google Scholar]
- 45. [Accessed October 1, 2016];(HOME) HOMfE. http://www.nottingham.ac.uk/research/groups/cebd/resources/flare-atopic-dermatitis.aspx.
- 46.Ring J, Alomar A, Bieber T, et al. Guidelines for treatment of atopic eczema (atopic dermatitis) Part II. J Eur Acad Dermatol Venereol. 2012;26(9):1176–1193. doi: 10.1111/j.1468-3083.2012.04636.x. [DOI] [PubMed] [Google Scholar]


