Abstract
Cerebral collaterals are dynamically recruited after arterial occlusion and highly affect tissue outcome in acute ischemic stroke. We investigated the efficacy and safety of four pathophysiologically distinct strategies for acute modulation of collateral flow (collateral therapeutics) in the rat stroke model of transient middle cerebral artery (MCA) occlusion. A composed randomization design was used to assign rats (n = 118) to receive phenylephrine (induced hypertension), polygeline (intravascular volume load), acetazolamide (cerebral arteriolar vasodilation), head down tilt (HDT) 15° (cerebral blood flow diversion), or no treatment, starting 30 min after MCA occlusion. Compared to untreated animals, treatment with collateral therapeutics was associated with lower infarct volumes (62% relative mean difference; 51.57 mm3 absolute mean difference; p < 0.001) and higher chance of good functional outcome (OR 4.58, p < 0.001). Collateral therapeutics acutely increased cerebral perfusion in the medial (+40.8%; p < 0.001) and lateral (+19.2%; p = 0.016) MCA territory compared to pretreatment during MCA occlusion. Safety indicators were treatment-related mortality and cardiorespiratory effects. The highest efficacy and safety profile was observed for HDT. Our findings suggest that acute modulation of cerebral collaterals is feasible and provides a tissue-saving effect in the hyperacute phase of ischemic stroke prior to recanalization therapy.
Keywords: Acute ischemic stroke, cerebral collaterals, collateral therapeutics, experimental stroke, ischemic penumbra
Introduction
Cerebral collateral circulation is a subsidiary vascular network, which is dynamically recruited after arterial occlusion to provide a source of residual blood flow to ischemic areas.1 Early cerebral collateral status is emerging as a powerful predictor of functional outcome in unselected ischemic stroke patients2,3 and in stroke patients treated with intravenous thrombolysis4 or endovascular thrombectomy.5 Evidence from stroke patients and animal models supports the concept that collateral status in the acute phase of ischemic stroke is a major determinant of the “physiological” therapeutic time window, i.e. the subject-specific timing of penumbra evolution, which defines successful versus futile recanalization.6–8 Therapeutic modulation of cerebral collateral flow by “collateral therapeutics” has been advocated in the acute phase of ischemic stroke,9 but randomized preclinical or clinical studies are scarce in this field and mostly focused on induced hypertension10,11 and partial aortic occlusion.12,13
If proved safe and effective, enhancement of collateral flow may be included in the measures of hyperacute prehospital stroke care, to be applied in the “golden hour” from symptom onset, to increase the time-dependent effect of thrombolysis and thrombectomy and the delivery of neuroprotective drugs, when available.14
Notably, the physiology of cerebral collateral recruitment and maintenance during acute ischemic stroke is not entirely known and is likely dependent on more than one factor, including anatomical variants, blood pressure (arterial and venous), blood volume, and intracranial pressure.15 No data are available to compare the efficacy and safety of collateral therapeutics with different mechanisms of action under the same conditions.
In the present study, we investigated the efficacy, safety, and hemodynamic effect of four “collateral therapeutics” in a randomized preclinical trial in rats subjected to transient proximal middle cerebral artery (MCA) occlusion. The choice of the four modulation strategies (induced hypertension, intravascular volume loading, cerebroselective vasodilation, cerebral blood flow diversion) was based on their different pathophysiological mechanisms as well as their translational feasibility as emergency therapy in human ischemic stroke.
Material and methods
Experimental design, sample size determination, and composed randomization
The experimental protocol was approved by the Committee on Animal Care of the University of Milano Bicocca, in accordance with the national guidelines on the use of laboratory animals (D.L. 26/2014) and the European Union Directive for animal experiments (2010/63/EU), under project license from the Italian Ministry of Health (81/2015-PR) and reported according to the ARRIVE (Animals in Research: Reporting In Vivo Experiments) guidelines.
A group of consecutive animals undergoing successful transient proximal MCA occlusion was used to explore the effect of cerebral collateral therapeutics on ischemic stroke outcome. Primary outcomes were infarct volume and neuroscore. Secondary outcomes were cerebral perfusion, cardiorespiratory parameters, and treatment-related mortality. Outcome assessment (infarct volume, neuroscore, cerebral hemodynamics, and cardiorespiratory parameters) was performed by third party blinded to treatment.
Given that our previous data indicated that the infarct volume of untreated MCA occluded rats (90 min occlusion, 24 h reperfusion) was normally distributed with a standard deviation (SD) of 0.45 and that the minimal clinically significant difference between the means of “untreated” and “collateral therapeutics” was estimated 0.3, we planned a main study (two groups) with a sample size of 60 animals for each treatment group, with an expected power of 0.95 and type I error of 0.05 to refuse the null hypothesis of no difference between the two groups (two-sided significance level). We expected a 5–10% treatment-related mortality.
A simple unrestricted randomization was performed using an online random number generator (www.random.org) in two steps (composed randomization). A first 1:1 randomization assigned rats to “no active treatment” or “collateral therapeutics.” In case of assignment to “collateral therapeutics,” a second 1:4 randomization was immediately performed to one of the four collateral therapeutics, chosen for this study, and rats were treated accordingly.
We planned a subgroup study (five groups) in which the “collateral therapeutics” group was stratified in four groups of 14 animals, each receiving one of the four collateral therapeutics chosen for this study, and compared to an “untreated” group of 56 animals (ratio 4:1 of control to experimental subjects). Considering the within-group SD 0.45 mentioned above and a minimal clinically significant difference of 0.4 between the means of “untreated” and single collateral therapeutic treatment, we were able to reject the null hypothesis with power of 0.8 and a type I error of 0.05 (two-sided significance level).
In order to reach the calculated sample size, consecutive rats were randomized until each group reached the minimum number of 14 animals. Randomization was performed after successful MCA occlusion to guarantee allocation concealment to the experimental surgeon.
Animals and surgery
Animals were housed in single cages, exposed to 12/12 h light/dark cycle, at controlled room temperature, with free access to food and water, in a specific pathogen-free facility. One hundred and twenty-six adult male Wistar rats (291 ± 7% g; Charles River, Calco, Italy) were anesthetized with 3% isoflurane in O2/N2O (1:3) and maintained with 1.5% isoflurane. Occlusion of the origin of the right MCA was induced transiently for 90 min with a reperfusion period of 24 h. Briefly, a silicone-coated filament (diameter 0.39 ± 0.02 mm, Doccol Corporation, Redlands, CA, USA) was introduced in the right external carotid artery and pushed through the right internal carotid artery to occlude the origin of the right MCA. Common carotid artery was transiently occluded immediately before the insertion of the filament and subsequently reopened during ischemia period and reperfusion. Pterygopalatine artery and proximal branches of the external carotid artery (occipital artery and cranial thyroid artery) were ligated during the surgical procedure, before the insertion of the filament. During surgery, the core temperature of 37℃ was controlled by a rectal thermometer connected to a feedback-controlled heating pad. After reperfusion, rats were allowed to recover and had free access to food and water. After 24 h from the onset of ischemia, animals were assessed for neurobehavioral score (see below), then euthanized using CO2 inhalation and the brains were collected for fixation. Eight rats were excluded from the experimental series for early death due to procedural subarachnoid hemorrhage (SAH) occurred before successful MCA occlusion. SAH was suspected by cerebral perfusion monitoring (> 90% perfusion deficit in both laser Doppler (LD) probes +absence of reperfusion after filament withdrawal, see below) and verified by necropsy (macroscopic bleeding around the circle of Willis). All successfully occluded rats (n = 118) were randomized and used for analysis.
Administration of collateral therapeutics
Collateral therapeutics were administered 30 min after successful MCA occlusion, at the doses specified below. Phenylephrine (PHE) 0.25 mg/ml solution in 0.9% saline was administered by continuous intravenous infusion at 45 ± 5 μl/min for 60 min in order to increase mean arterial pressure (MAP) by 30%. Polygeline (PLG) 0.35% solution was administered as a single intravenous bolus of 4.0 ± 0.5 ml over 5 min in order to increase intravascular volume by 25%. Acetazolamide (ACZ) 100 mg/kg solution in 0.9% saline was administered in a single intravenous bolus of 0.5 ml over 1 min to promote carbon dioxide-mediated cerebral vasodilation. Head down tilt (HDT) was administered using a 15° tilted platform for 60 min in order to promote gravitational cerebral flow diversion from the lower body to the head.
Multisite LD cerebral perfusion monitoring
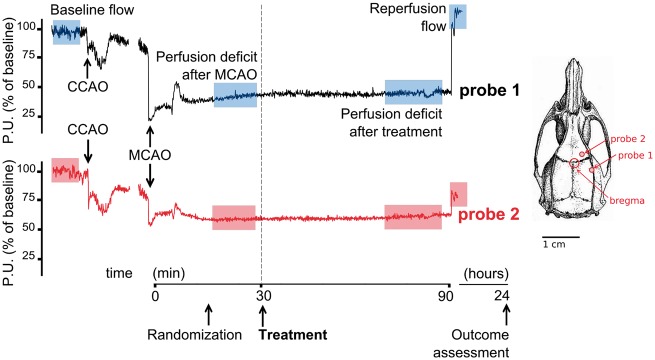
The induction of focal cerebral ischemia was assessed using LD perfusion monitoring (dual channel moorVMS-LDF™, Moor, Axminster, UK) using two blunt needle probes (VP12).16 A first probe (Probe 1) was attached to the skull 1 mm posterior to the Bregma and 5 mm lateral to the midline (corresponding to the lateral MCA territory, within the “ischemic core”). A second probe (Probe 2) was attached to the skull 2 mm anterior to the Bregma and 2 mm lateral to the midline (corresponding to the medial MCA territory, to provide an indication of “collateral flow”). Three hemodynamic parameters (expressed as % of baseline) were considered for each probe: (i) stabilized perfusion deficit during MCA occlusion (0–30 min after MCA occlusion), (ii) maximal change of cerebral perfusion after treatment (30–90 min after MCA occlusion), and (iii) reperfusion after filament withdrawal.
Monitoring of cardiorespiratory parameters
Cardiorespiratory parameters (arterial pressure, heart rate, respiratory rate) were continuously monitored using Samba Preclin 420 transducer (Samba Sensors, Harvard Apparatus, UK) inserted in the right femoral artery before ischemia induction. MAP, heart rate, and respiratory rate of each animal were recorded at 40 Hz sampling rate during the ischemic period (0–90 min after MCA occlusion) and synchronized to cerebral perfusion tracings using a Matlab dedicated software.
Neurobehavioral assessment
Rats were assessed 24 h after transient MCA occlusion with a functional neuroscore for neurological outcome on a scale from 3 (most severe) to 18 (no deficit), which required scoring spontaneous movement, sensory function, and motor function.17 Functional outcome was dichotomized and expressed as “good functional outcome” (scores 14–18) or “poor functional outcome” (scores 3–13).
Histology and infarct volume determination
Brains were fixed in ice cold 10% neutral buffer formalin and coronal sections (50 µm) were stained using Cresyl Violet 0.1% (Bioptica, Milano, Italy). Infarct areas were measured in 19 consecutive sections with 250 µm interval (bregma +2.5 mm to −3.0 mm). Infarct volume was calculated using ImageJ image processing software (National Institute of Health, Bethesda, MD, USA), corrected for interhemispheric asymmetries due to cerebral edema and expressed in mm3.
Statistical analysis
The data were analyzed using the SPSS Statistics (IBM Corporation, Armonk, New York, USA) package for PC (version 23). Values were expressed as mean and SD. For the two-group analyses on infarct volume, an unpaired two-sided Student’s t-test was used, while the effect on neuroscore was evaluated by means of a logistic regression model; a subanalysis, adjusting both effects for Probe 2 perfusion deficit after MCA occlusion, was done using ANCOVA for infarct volume and logistic regression model for neuroscore. For the subgroup analyses, infarct volume was analyzed by means of a one-way ANOVA with Bonferroni’s correction for multiple comparisons and neuroscore was analyzed by means of a logistic regression model (in both cases, untreated group was the reference). Comparisons between control and treated group on secondary outcomes (cerebral perfusion and cardiorespiratory parameters) were done by means of unpaired two-sided Student’s t-test for two-group analyses and one-way ANOVA with Bonferroni’s correction for multiple comparisons for subgroup analyses, while a paired two-sided Student’s t-test was used when comparing pre- versus posttreatment (cerebral perfusion, by probe) in untreated rats and in each collateral therapeutic. A p-value less than 0.05 was considered significant. Effect size was estimated using Cohen’ d for infarct volume and odds ratio (OR) for neuroscore.
Results
Study population
A total of 118 consecutive rats, subjected to successful MCA occlusion, were randomized according to the study design (Figure 1 and Supplementary Figure 1), 58 rats received “no active treatment” and 60 rats received “collateral therapeutics.” A second randomization was immediately performed within the “collateral therapeutics” group to allocate animals to one of the four modulation strategies, resulting in 14 rats treated with PHE, 16 rats with PLG, 16 rats with ACZ, and 14 rats with HDT. The baseline (before treatment) characteristics of the main study groups are summarized in Table 1. No difference was observed between the two groups with the exception of the residual perfusion in the medial MCA territory during MCA occlusion, which was approximately 10% higher for collateral therapeutics compared to controls (p = 0.004) and was included as a covariate (named: Probe 2) in the primary outcome analysis.
Figure 1.
Graphical representation of the study design. Timing of the experiment (MCA occlusion, randomization, treatment, reperfusion, outcome assessment) is shown in relation to cerebral hemodynamic monitoring. Representative multisite LD tracings from an untreated rat are shown. The right panel represents the position of the two LD probes with reference to the underlying MCA territory: Colored boxes indicate cerebral perfusion sampling. CCAO: common carotid artery occlusion; LD: laser Doppler; MCA: middle cerebral artery.
Table 1.
Baseline characteristics of the study population.
| Unit | Collateral therapeutics (60) mean (SD) | No active treatment (58) mean (SD) | p-value | |
|---|---|---|---|---|
| Weight | (g) | 293.8 (20.5) | 289.2 (26.5) | 0.374 |
| Lateral MCA territory perfusion deficit (Probe 1)a | PU (% of baseline) | 43.8 (2.1) | 39.6 (2.1) | 0.180 |
| Medial MCA territory perfusion deficit (Probe 2)a | PU (% of baseline) | 71.2 (21.4) | 60.0 (19.1) | 0.004 |
| Arterial blood pressurea | mmHg | 115 (33) | 113 (23) | 0.895 |
| Heart ratea | bpm | 433 (38) | 434 (30) | 0.901 |
| Respiratory ratea | brpm | 58 (11) | 57 (11) | 0.854 |
bpm: beats per minute; brpm: breaths per minute; MCA: middle cerebral artery; PU: perfusion unit; SD: standard deviation.
Comparisons were made by means of unpaired Student’s t-tests.
0–30 min after MCA occlusion, before treatment. Bold values are statistically significant with p < 0.05.
Efficacy of collateral therapeutics on infarct volume and functional outcome
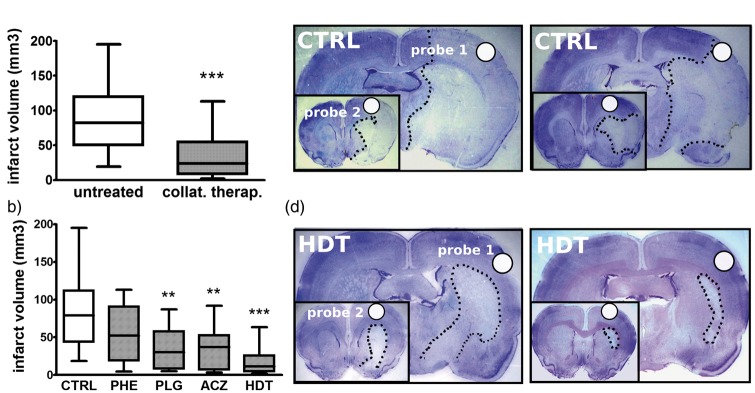
Infarct volume at 24 h was lower in rats treated with collateral therapeutics compared to untreated rats (relative mean difference 62%, absolute mean difference 51.57 mm3, 95% confidence interval (CI) 35.69–67.46, Cohen’s d 1.32, p < 0.001) (Figure 2(a)). After adjusting for the covariate (Probe 2), this finding was confirmed (absolute difference 44.33 mm3, 95% CI 27.49–61.19, Cohen’s d 1.18, p < 0.001). Subgroup analysis showed that the largest effect size in infarct volume reduction was observed with HDT (64.93 mm3, Cohen’s d 1.96, p < 0.001), followed by ACZ (46.64 mm3, Cohen’s d 1.32, p < 0.01) and PLG (40.45 mm3, Cohen’s d 1.31, p < 0.01), while PHE (33.51 mm3, Cohen’s d 0.79, p = 0.264) did not reach a significant effect (Figure 2(b)). The difference in infarct volume was mostly driven by a reduction of ischemic areas in the cortex rather than in the basal ganglia (Figure 2(c) and (d)).
Figure 2.
Effect of collateral therapeutics on infarct volume in the main study (a) and in the subgroups (b). Representative brain histological sections obtained 24 h after MCA occlusion from two untreated (CTRL) rats (c) and two rats treated with HDT (d). Sections correspond to LD positioning (Probe 1 larger pictures; Probe 2 smaller pictures). A dotted line indicates the borders between ischemic lesions and healthy brain tissue. CTRL n = 58. PHE n = 13. PLG n = 15. ACZ n = 14. HDT n = 14. **p < 0.01 compared to untreated. ***p < 0.001 compared to untreated. Comparisons were made by means of unpaired Student’s t-tests (main study) and ANOVA with Bonferroni’s correction (subgroups analysis). ACZ: acetazolamide; HDT: head down tilt; LD: laser Doppler; MCA: middle cerebral artery; PHE: phenylephrine; PLG: polygeline.
The propensity to reach a good functional outcome (Garcia neuroscore 14–18 at 24 h) was significantly higher in rats treated with collateral therapeutics compared to untreated rats (OR 4.58; p < 0.001) (Table 2). After adjusting for the covariate (Probe 2), this finding was confirmed (Adj OR 4.50; p < 0.001). Subgroup analysis showed that HDT was significantly associated with a good neuroscore compared to controls (OR 28.88; p = 0.002), PLG showed a significant effect (OR 3.33; p = 0.044) which was not confirmed after adjusting for Probe 2, while ACZ and PHE did not reach a significant effect (Table 2).
Table 2.
Functional outcome 24 h after reperfusion.
| Study | Dichotomized neuroscore | n success/ total (%) | p-value | OR (95% CI) | Adj OR (95% CI) |
|---|---|---|---|---|---|
| Main | No active treatment | 18/58 (31.0) | ref. | 1.00 | 1.00 |
| Collateral therapeutics | 36/56 (64.2) | < 0.001 | 4.58 (2.07–10.12) | 4.50 (1.93–10.48) | |
| Subgroups | No treatment | 18/58 (31.0) | ref. | 1.00 | 1.00 |
| PHE | 6/13 (46.1) | 0.127 | 2.59 (0.76–8.81) | 2.80 (0.78–9.14) | |
| PLG | 9/15 (60.0) | 0.044 | 3.33 (1.03–10.77) | 2.68 (0.78–9.14) | |
| ACZ | 8/14 (57.1) | 0.075 | 2.96 (0.89–9.79) | 2.88 (0.85–9.69) | |
| HDT | 13/14 (92.8) | 0.002 | 28.88 (3.50–237.96) | 26.71 (3.20–222.64) |
ACZ: acetazolamide; Adj OR: adjusted odds ratio; CI: confidence intervals; HDT: head down tilt; number; OR: odds ratio; PHE: phenylephrine; PLG: polygeline.
Success” identifies animals reaching a good functional outcome (Garcia neuroscore 14–18). Comparisons were made by means of OR and 95% CI. Bold values are statistically significant with p < 0.05.
Effect of collateral therapeutics on cerebral perfusion during MCA occlusion
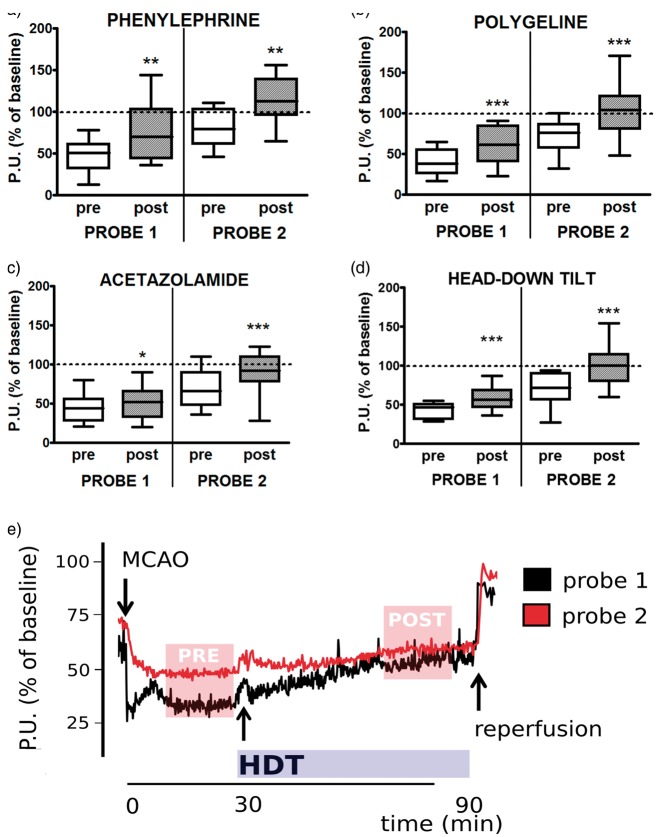
Treatment with collateral therapeutics was associated with a significant increase in cerebral perfusion in both lateral (Probe 1 + 19.2%; p = 0.016) and medial (Probe 2 +40.8%; p < 0.001) MCA territory, compared to pretreatment during MCA occlusion (Figure 3(a)). Reperfusion values did not differ between untreated rats and rats treated with collateral therapeutics for both LD probes (Figure 3(b)). In untreated animals, a small but significant increase in cerebral perfusion was observed during the 90 min of MCA occlusion for both LD probe (Probe 1 +8.5%, p = 0.002; Probe 2 +8.6%, p = 0.002; Figure 3(c)). Subgroup analysis showed that all collateral therapeutics significantly increased cerebral perfusion compared to pretreatment values for both LD probes (Figure 4). When compared to untreated animals, all collateral therapeutics increased cerebral perfusion in Probe 2, while only PHE and HDT increased cerebral perfusion in Probe 1 (Table 3).
Figure 3.
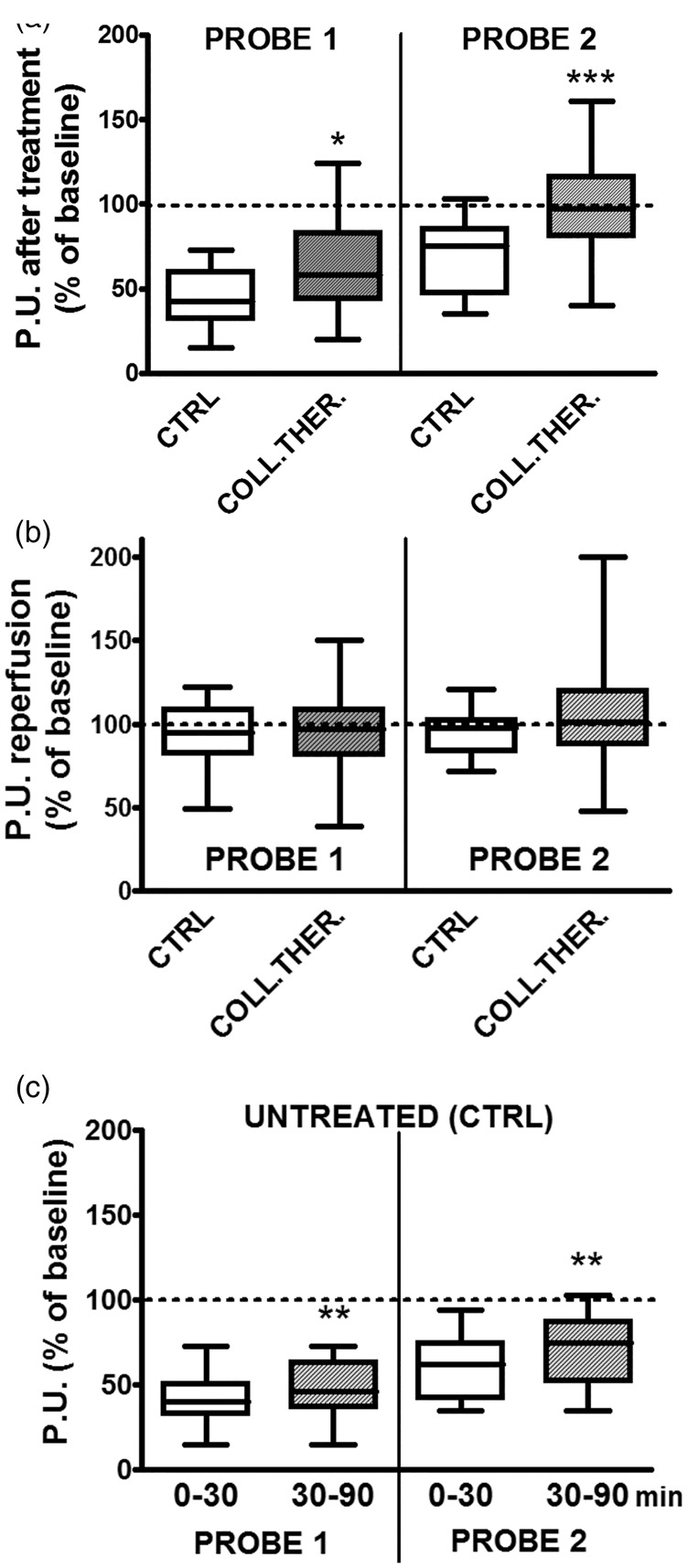
Effect of collateral therapeutics on cerebral perfusion during MCA occlusion and reperfusion. Maximal cerebral perfusion after treatment during MCA occlusion (a) and reperfusion (b) are shown for Probe 1 and Probe 2, comparing untreated rats (CTRL) and rats treated with collateral therapeutics. Cerebral perfusion of untreated rats during MCA occlusion, comparing the first 30 min with the last 60 min, is shown in (c). CTRL n = 58. PHE n = 13. PLG n = 15. ACZ n = 14. HDT n = 14. *p < 0.05 compared to untreated. **p < 0.01 compared to the first 30 min of MCA occlusion. ***p < 0.001 compared to untreated. Comparisons were made by means of unpaired Student’s t-tests (a and b) and paired Student’s t-test (c). ACZ: acetazolamide; HDT: head down tilt; MCA: middle cerebral artery; PHE: phenylephrine; PLG: polygeline.
Figure 4.
Effect of single collateral therapeutics on cerebral perfusion during MCA occlusion. The first 30 min (before treatment; pre) were compared with the last 60 min (after treatment; post). Cerebral perfusion is indicated as % of baseline flow in Probe 1 and Probe 2 before and after treatment with PHE (a), PLG (b), ACZ (c), and HDT (d). A representative multisite LD tracing of a rat treated with HDT is shown in (e). CTRL n = 58. PHE n = 13. PLG n = 15. ACZ n = 14. HDT n = 14. *p < 0.05 compared to pretreatment. **p < 0.01 compared to pretreatment. ***p < 0.001 compared to pretreatment. Comparisons were made by means of paired Student’s t-test. ACZ: acetazolamide; HDT: head down tilt; LD: laser Doppler; MCA: middle cerebral artery; PHE: phenylephrine; PLG: polygeline.
Table 3.
Changes in cerebral perfusion in the lateral and medial MCA territory after treatment.
| Study | Lateral MCA (Probe 1) | Peak flow (mean) | 95% CI | p-value |
|---|---|---|---|---|
| Main | No active treatment | 46.60 | 38.76–54.43 | ref. |
| Collateral therapeutics | 61.59 | 54.90–68.28 | 0.006 | |
| Subgroups | No active treatment | 46.60 | 38.76–54.43 | ref. |
| PHE | 75.00 | 60.34–89.65 | 0.001 | |
| PLG | 61.64 | 49.25–74.03 | 0.044 | |
| ACZ | 47.07 | 34.68–59.65 | 0.949 | |
| HDT | 66.50 | 54.1–78.88 | 0.008 | |
| Medial MCA (Probe 2) |
|
|
|
|
| Main | No active treatment | 66.03 | 57.77–74.88 | ref. |
| Collateral therapeutics | 98.54 | 90.65–106.64 | <0.001 | |
| Subgroups | No active treatment | 66.03 | 57.77–74.88 | ref. |
| PHE | 114.30 | 98.21–130.38 | <0.001 | |
| PLG | 105.57 | 91.97–119.16 | <0.001 | |
| ACZ | 94.83 | 80.14–109.51 | 0.008 | |
| HDT | 92.23 | 78.12–106.34 | 0.002 |
ACZ: acetazolamide; CI: confidence intervals; HDT: head down tilt; MCA: middle cerebral artery; PHE: phenylephrine; PLG: polygeline.
Flow is expressed as perfusion units (% of baseline) and represents the maximum flow obtained after treatment.
CTRL n = 58. PHE n = 13. PLG n = 15. ACZ n = 14. HDT n = 14. Comparisons were made by means of unpaired Student’s t-tests (main study) and ANOVA with Bonferroni’s correction (subgroups analysis). Bold values are statistically significant with p < 0.05.
Effect of collateral therapeutics on cardiorespiratory parameters during MCA occlusion
Arterial pressure, heart rate, and respiratory rate were monitored before and after treatment during MCA occlusion (Supplementary Figure 2). No significant treatment-related differences in cardiorespiratory parameters were observed for PLG, ACZ, or HDT, compared to untreated animals. Treatment with PHE was associated with a 29.5% increase in MAP (+34 mmHg; 95% CI 23–45) and a 6% decrease in heart rate (−25 bpm; 95% CI −52 to +1).
Safety of collateral therapeutics
Treatment-related mortality occurred in four rats (6.6% of treated animals) within 20–60 min after treatment administration (Supplementary Table 1). Treatment-related SAH accounted for two deaths (50%) and was associated with PHE (one rat) and ACZ (one rat). ACZ determined the death of one further rat, unrelated to SAH and with no identified cause. Respiratory failure occurred as cause of death in one rat treated with PLG. No rats died after treatment with HDT. Animals who died due to treatment were excluded from the analysis of the primary and secondary outcomes.
Discussion
The functional status of cerebral collaterals is crucially important in the pathophysiology of acute ischemic stroke,18 being associated with the timing of penumbra loss19 and response to reperfusion therapies.20 Although several pharmacological and nonpharmacological strategies may theoretically enhance cerebral collateral perfusion in acute ischemic stroke,21 the efficacy and safety of “collateral therapeutics” have not been rigorously investigated and compared in experimental studies. Recent studies demonstrated the feasibility of prehospital drug administration in acute stroke,22,23 in order to provide ultra-early neuroprotection and maximize efficacy. In this context, the ideal collateral therapeutic should be easy to administer, fast acting, and safe in case of misdiagnosis (hemorrhagic stroke or stroke mimics).
Our randomized preclinical study investigated the efficacy and safety of four different collateral therapeutics in the rat stroke model of intraluminal proximal MCA occlusion (90 min) followed by 24 h reperfusion. A composed randomization design was chosen in order to obtain two parallel studies. The aim of the high-powered (actual power 0.94) study was to compare collateral therapeutics as a “class” with “no active treatment” and provide evidence for the concept of collateral flow modulation in acute ischemic stroke. The aim of the standard-powered (actual power 0.84) study was to compare each therapeutic strategy with “no active treatment” and provide preliminary evidence about the collateral therapeutic with the best efficacy and safety profile. Given the use of two-sided tests, this study needs to be considered as exploratory.
The four therapeutic strategies were selected according to two criteria: pathophysiology and feasibility in a translational paradigm. The dose of each collateral therapeutic was determined in order to obtain the largest effect without incurring excessive risks of organ damage.
PHE was used to increase MAP by 30%, guided by invasive blood pressure monitoring. A target of 30% increase in MAP was used in a previous study of distal MCA occlusion in mice,11 while a lower target of 10–20% was used in pilot clinical studies of induced hypertension in ischemic stroke patients.10,24
PLG was used to acutely expand intravascular volume by 25%. Large volumes of PLG are commonly used in shock treatment25 and display a neutral effect on the coagulation system.26
ACZ was used to promote carbon dioxide-mediated cerebral arteriolar vasodilation.27 The dose chosen for ACZ (100 mg/kg) represents a high (but not harmful) dose in healthy rats and has been shown to produce a maximal cerebral vasodilation.28
HDT was applied by carefully tilting the operating table by 15° to promote gravitational diversion of cerebral blood flow from the lower body to the head. Previous studies indicated that head positioning affects cerebral perfusion in ischemic stroke patients.29 Moreover, HDT from 6° to 12° has been studied in the field of aerospace medicine as a model of microgravity and it has been applied for time periods ranging from minutes to several days in healthy subjects.30,31
Our findings strongly support the feasibility and efficacy of collateral therapeutics as a hyperacute therapy in experimental ischemic stroke prior to reperfusion. Postischemic administration of collateral therapeutics, with a therapeutic time window of 30 min, largely reduced infarct volume and improved functional outcome at 24 h with a number needed to treat (NNT) of 3. Although all four therapeutic strategies acutely modified cerebral perfusion in the affected MCA territory, subgroups analysis showed significant differences as far as stroke outcome. PLG and ACZ favorably affected structural outcome, while HDT favorably affected both structural and functional outcome. PHE showed a smaller and more variable effect on either structural or functional outcome.
Cerebral perfusion during MCA occlusion was monitored in the lateral and medial MCA territory, as previously described.16 Perfusion deficit in the lateral MCA territory (Probe 1) was considered an indicator of the “ischemic core” and corresponds to the LD position commonly used in this stroke model.32 Perfusion deficit in the medial MCA territory (Probe 2) was considered an indicator of “collateral flow,” corresponding to the border zone territory between anterior cerebral artery and MCA.8,33 Collateral therapeutics acutely improved cerebral perfusion in both lateral and medial MCA territory. Notably, perfusion deficit in Probe 2 was almost completely reversed by collateral therapeutics, while perfusion deficit in Probe 1 was favorably modified only by PHE and HDT.
Interestingly, no significant variation of blood pressure, heart rate, and respiratory rate was detected after treatment with PLG, ACZ, or HDT. Conversely, PHE induced the expected increase in blood pressure and a mild reflex bradycardia. Safety assessment showed 6% treatment-related mortality. Treatment-related SAH was the most frequent (50%) cause of death and was observed in animals treated with PHE and ACZ. Notably, no animal died after treatment with HDT.
A major limitation of our study is that acute MRI imaging (including perfusion or penumbral imaging) was not performed. However, dynamics of cerebral collateral flow was the main focus of the study and this can be reliably monitored with multisite LD monitoring, which has been validated with acute MRI for outcome prediction in this stroke model.34 For logistic reasons, multisite LD monitoring and MRI are mutually exclusive.
A second limitation of our study is early outcome assessment, which was limited to the first 24 h after MCA occlusion and reperfusion. Further studies using longer time points are warranted to consolidate our findings for both structural and behavioral outcome.
Our results indicate that HDT has favorable efficacy and safety profile as a collateral therapeutic in experimental ischemic stroke and exhibits a high translational potential. For its simplicity, low cost, and translational appeal, HDT may be an attractive candidate for a rigorous, international, multicenter preclinical trial in experimental ischemic stroke, in order to consolidate the evidence before moving to studies in large animals and humans.35
In the present study, we did not address the effect of single collateral therapeutics on specific molecular or biochemical pathways in neural or glial cells, which are known to affect tissue outcome.36 We believe that this issue is highly important, may help to explain the differences in the protective effect among the four investigated strategies, and needs further research.
Nonetheless, we believe that the major mechanism of action of collateral therapeutics relies on acute enhancement of cerebral blood flow in ischemic areas, with slightly different dynamics among the four strategies, as shown by multisite LD results. We also believe that any acute collateral-enhancing therapy would be of little benefit if not followed by reperfusion, since collateral failure frequently develops with persistent large artery occlusion.37
Although primarily aimed at enhancing perfusion prior to recanalization therapies, HDT could also precede or be combined with neuroprotective therapies, which are being studied for the hyperacute or acute phase of ischemic stroke, such as hypothermia, antiexcitotoxic, or immunomodulating agents.38
Further studies are needed to assess whether low HDT angles (below −15℃) will provide the same benefit in terms of increased collateral perfusion. More importantly, further safety assessments of HDT in acute cerebral ischemia are required, particularly in term of intracranial pressure changes and risk of hemorrhagic transformation. Although HDT appears as an extremely simple, low cost, and feasible measure to be applied in the prehospital setting when an acute ischemic stroke is suspected, its effect on hemorrhagic stroke (intracerebral hemorrhage and SAH) or stroke mimics needs to be carefully determined. A preliminary, but important step to address this issue would be to assess safety of HDT in rodent models of hemorrhagic stroke, particularly if applied during the time window of hematoma expansion.
Conclusions
Acute therapeutic modulation of cerebral collateral flow is feasible and improves structural and functional outcome after transient proximal MCA occlusion in rats. The highest efficacy and safety profile was observed for 15° HDT. Further experimental and clinical research is urgently needed to establish collateral therapeutics as a novel tissue-saving strategy in the hyperacute phase of ischemic stroke prior to recanalization therapy.
Supplementary Material
Acknowledgements
We thank Dr Virginia Rodriguez Menendez and Ms Elena Pirovano for technical assistance.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the University of Milano Bicocca Fondo di Ateneo 2012–2015.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
SB, AV, and ER designed the study; SB, AV, DC, MR, VDE, EC, RC, LM, SP, GP, FS, and GP performed the experiments; SB, DC, LP, CG, EPS, and CF analyzed data; SB wrote the article, which was critically revised by the other authors.
Data availability
Data are available upon request to the corresponding author.
Supplementary Material
Supplementary material for this paper can be found at http://journals.sagepub.com/doi/suppl/10.1177/0271678X16688705.
References
- 1.Liebeskind DS. Collateral circulation. Stroke 2003; 34: 2279–2284. [DOI] [PubMed] [Google Scholar]
- 2.van den Wijngaard IR, Boiten J, Holswilder G, et al. Impact of collateral status evaluated by dynamic computed tomographic angiography on clinical outcome in patients with ischemic stroke. Stroke 2015; 46: 3398–3404. [DOI] [PubMed] [Google Scholar]
- 3.Fanou E, Knight J, Aviv R, et al. Effect of collaterals on clinical presentation, baseline imaging, complications, and outcome in acute stroke. Am J Neuroradiol 2015; 36: 2285–2291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brunner F, Tomandl B, Hanken K, et al. Impact of collateral circulation on early outcome and risk of hemorrhagic complications after systemic thrombolysis. Int J Stroke 2014; 9: 992–998. [DOI] [PubMed] [Google Scholar]
- 5.Leng X, Fang H, Leung TW, et al. Impact of collateral status on successful revascularization in endovascular treatment: a systematic review and meta-analysis. Cerebrovasc Dis 2015; 41: 27–34. [DOI] [PubMed] [Google Scholar]
- 6.Bivard A, Levi C, Krishnamurthy V, et al. Perfusion computed tomography to assist decision making for stroke thrombolysis. Brain 2015; 138: 1919–1931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sheth SA, Liebeskind DS. Collaterals in endovascular therapy for stroke. Curr Opin Neurol 2015; 28: 10–15. [DOI] [PubMed] [Google Scholar]
- 8.Beretta S, Cuccione E, Versace A, et al. Cerebral collateral flow defines topography and evolution of molecular penumbra in experimental ischemic stroke. Neurobiol Dis 2015; 74: 305–313. [DOI] [PubMed] [Google Scholar]
- 9.Liebeskind DS. Reperfusion for acute ischemic stroke: arterial revascularization and collateral therapeutics. Curr Opin Neurol 2010; 23: 36–45. [DOI] [PubMed] [Google Scholar]
- 10.Marzan AS, Hungerbühler H-J, Studer A, et al. Feasibility and safety of norepinephrine-induced arterial hypertension in acute ischemic stroke. Neurology 2004; 62: 1193–1195. [DOI] [PubMed] [Google Scholar]
- 11.Shin HK, Nishimura M, Jones PB, et al. Mild induced hypertension improves blood flow and oxygen metabolism in transient focal cerebral ischemia. Stroke 2008; 39: 1548–1555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Winship IR, Armitage GA, Ramakrishnan G, et al. Augmenting collateral blood flow during ischemic stroke via transient aortic occlusion. J Cereb Blood Flow Metab 2014; 34: 61–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shuaib A, Bornstein NM, Diener H-C, et al. Partial aortic occlusion for cerebral perfusion augmentation safety and efficacy of neuroflo in acute ischemic stroke trial. Stroke 2011; 42: 1680–1690. [DOI] [PubMed] [Google Scholar]
- 14.Weber J, Ebinger M, Audebert HJ. Prehospital stroke care: telemedicine, thrombolysis and neuroprotection. Expert Rev Neurother 2015; 15: 753–761. [DOI] [PubMed] [Google Scholar]
- 15.Ginsberg MD. Expanding the concept of neuroprotection for acute ischemic stroke: the pivotal roles of reperfusion and the collateral circulation. Prog Neurobiol 2016; 145–146: 46–47. [DOI] [PubMed]
- 16.Beretta S, Riva M, Carone D, et al. Optimized system for cerebral perfusion monitoring in the rat stroke model of intraluminal middle cerebral artery occlusion. J Vis Exp 2013; 72: pii: 50214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Garcia JH, Wagner S, Liu K-F, et al. Neurological deficit and extent of neuronal necrosis attributable to middle cerebral artery occlusion in rats statistical validation. Stroke 1995; 26: 627–635. [DOI] [PubMed] [Google Scholar]
- 18.Liebeskind DS. Collateral perfusion: time for novel paradigms in cerebral ischemia. Int J Stroke 2012; 7: 309–310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jung S, Gilgen M, Slotboom J, et al. Factors that determine penumbral tissue loss in acute ischaemic stroke. Brain 2013; 136: 3554–3560. [DOI] [PubMed] [Google Scholar]
- 20.Hwang Y-H, Kang D-H, Kim Y-W, et al. Impact of time-to-reperfusion on outcome in patients with poor collaterals. Am J Neuroradiol 2015; 36: 495–500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shuaib A, Butcher K, Mohammad AA, et al. Collateral blood vessels in acute ischaemic stroke: a potential therapeutic target. Lancet Neurol 2011; 10: 909–921. [DOI] [PubMed] [Google Scholar]
- 22.Saver JL, Starkman S, Eckstein M, et al. Prehospital use of magnesium sulfate as neuroprotection in acute stroke. N Engl J Med 2015; 372: 528–536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kim DH, Saver JL, Starkman S, et al. Enrollment yield and reasons for screen failure in a large prehospital stroke trial. Stroke 2016; 47: 232–235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Koenig MA, Geocadin RG, de Grouchy M, et al. Safety of induced hypertension therapy in patients with acute ischemic stroke. Neurocrit Care 2006; 4: 3–7. [DOI] [PubMed] [Google Scholar]
- 25.Raghunathan K, Bonavia A, Nathanson BH, et al. Association between initial fluid choice and subsequent in-hospital mortality during the resuscitation of adults with septic shock. Anesthesiology 2015; 123: 1385–1393. [DOI] [PubMed] [Google Scholar]
- 26.Coats T, Brazil E, Heron M. The effects of commonly used resuscitation fluids on whole blood coagulation. Emerg Med J 2006; 23: 546–549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Inoue Y, Tanaka Y, Hata H, et al. Arterial spin-labeling evaluation of cerebrovascular reactivity to acetazolamide in healthy subjects. Am J Neuroradiol 2014; 35: 1111–1116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Frankel HM, Garcia E, Malik F, et al. Effect of acetazolamide on cerebral blood flow and capillary patency. J Appl Physiol 1992; 73: 1756–1761. [DOI] [PubMed] [Google Scholar]
- 29.Wojner-Alexander AW, Garami Z, Chernyshev OY, et al. Heads down: flat positioning improves blood flow velocity in acute ischemic stroke. Neurology 2005; 64: 1354–1357. [DOI] [PubMed] [Google Scholar]
- 30.Palombo C, Morizzo C, Baluci M, et al. Large artery remodeling and dynamics following simulated microgravity by prolonged head-down tilt bed rest in humans. Biomed Res Int 2015; 2015: 342565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fortrat J-O, Sigaudo D, Hughson RL, et al. Effect of prolonged head-down bed rest on complex cardiovascular dynamics. Auton Neurosci 2001; 86: 192–201. [DOI] [PubMed] [Google Scholar]
- 32.Taninishi H, Jung JY, Izutsu M, et al. A blinded randomized assessment of laser Doppler flowmetry efficacy in standardizing outcome from intraluminal filament MCAO in the rat. J Neurosci Methods 2015; 241: 111–120. [DOI] [PubMed] [Google Scholar]
- 33.Riva M, Pappadà GB, Papadakis M, et al. Hemodynamic monitoring of intracranial collateral flow predicts tissue and functional outcome in experimental ischemic stroke. Exp Neurol 2012; 233: 815–820. [DOI] [PubMed] [Google Scholar]
- 34.Cuccione E, Versace A, Cho TH, et al. Multi-site laser Doppler flowmetry for assessing collateral flow in experimental ischemic stroke: validation of outcome prediction with acute MRI. J Cereb Blood Flow Metab. Epub ahead of print 27 July 2016. DOI: 0271678X16661567. [DOI] [PMC free article] [PubMed]
- 35.Dirnagl U, Fisher M. International, multicenter randomized preclinical trials in translational stroke research: it’s time to act. J Cereb Blood Flow Metab 2012; 32: 933–935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Moskowitz MA, Lo EH, Iadecola C. The science of stroke: mechanisms in search of treatments. Neuron 2010; 67: 181–198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Beard DJ, McLeod DD, Logan CL, et al. Intracranial pressure elevation reduces flow through collateral vessels and the penetrating arterioles they supply. A possible explanation for ‘collateral failure’ and infarct expansion after ischemic stroke. J Cereb Blood Flow Metab 2015; 35: 861–872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chamorro Á, Dirnagl U, Urra X, et al. Neuroprotection in acute stroke: targeting excitotoxicity, oxidative and nitrosative stress, and inflammation. Lancet Neurol 2016; 15: 869–881. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data are available upon request to the corresponding author.