Abstract
The purpose of this report is to describe the international growth, outcomes, complications and technology used in pediatric extracorporeal life support (ECLS) from 2009 to 2015 as reported by participating centers in the Extracorporeal Life Support Organization (ELSO). To date, there are 59,969 children who have received ECLS in the ELSO Registry; among those, 21,907 received ECLS since 2009 with an overall survival to hospital discharge rate of 61%. In 2009, 2,409 ECLS cases were performed at 157 centers. By 2015, that number grew to 2,992 cases in 227 centers, reflecting a 24% increase in patients and 55% growth in centers. ECLS delivered to neonates (0–28 days) for respiratory support was the largest subcategory of ECLS among children <18 years old. Overall, 48% of ECLS was delivered for respiratory support and 52% was for cardiac support or extracorporeal life support to support cardiopulmonary resuscitation (ECPR). During the study period, over half of children were supported on ECLS with centrifugal pumps (51%) and polymethylpentene oxygenators (52%). Adverse events including neurologic events were common during ECLS, a fact that underscores the opportunity and need to promote quality improvement work.
Keywords: extracorporeal membrane oxygenation (ECMO), extracorporeal life support (ECLS), outcomes, complications, pediatric, neonate, extracorporeal life support organization (ELSO), pediatric
Introduction
The Extracorporeal Life Support Organization (ELSO) and its Registry were created to improve care and outcomes for patients receiving extracorporeal life support (ECLS) or extracorporeal membrane oxygenation (ECMO). The first ECLS Registry report was published in 1988 in ASAIO.1 The report described the patient population, techniques, complications and outcomes of 715 newborns cared for at 18 U.S. institutions. Since then there have been regular ELSO Registry published reports.2–9
The intent of publishing periodic ELSO Registry reports remains the same, but over time the Registry has evolved as described by Paden et al.8 The ELSO Registry has grown to over 300 active centers divided among 60 countries with over 80,000 patients.10 Due to its broad international footprint and rapidly increasing numbers, ELSO has been organized into five chapters: European, Asia-Pacific, South West Asian and African, Latin-American and North American. In addition to ELSO’s international expansion, the annual composition of patients in the Registry has transformed from being predominated by neonatal respiratory ECLS to a majority adult ECLS. Furthermore, the ECLS technology applied to patients has also evolved with a predominance of centrifugal pumps over roller pumps, a minimization of silicone oxygenators, and the broad availability of dual lumen cannulas for patients of nearly all sizes.11,12
This report, like previous reports, will describe ECLS growth, techniques, complications and outcomes of patients supported with ECLS, but differs from other reports by focusing on pediatric ECLS in the current era. Given the rapid expansion of ECLS worldwide, as an organization we have elected to change data reporting. In addition to yearly summaries of the entire >30 year ELSO Registry, we will also provide regional and population-specific reports that focus on more recent outcomes. This manuscript represents the first of this effort and will focus on the post 2009 era of ECLS in children.
Methods
For purposes of this report, we analyzed data from children aged 0–17 years who were reported to the Registry between January 1, 2009 and December 31, 2015. Data were summarized from the ELSO Registry and the January 2017 Registry Reports: the International Summary, Outcome Trends Report and Complications Trends Reports. Beginning in September 2016, the ELSO Registry changed from using International Classification of Diseases 9th version (ICD-9), to the 10th version ICD-10. For this reason, and since not all centers have completed reporting 2016 data, we limited analyses to 2009–2015. Throughout this report we will use the term neonate to refer to children aged 0–28 days and pediatrics to refer to non-neonatal children aged 29 days −17 years.
The 2012 Registry report described the collected demographics, pre-ECLS blood gas, pre-ECLS support, diagnoses, procedures, ECLS indication, ECLS equipment, ECLS course, complications and outcomes.8 In 2016, the Registry also began adding time stamps to complications, started recording severity of illness and organ dysfunction scores on admission13–23 and added multiple logical checks to ensure accurate and complete data entry. As examples, range checks were instituted for dates and times, and mandatory fields were established that must be entered in order to complete a record and add it to the Registry.
Results
Overall Trends
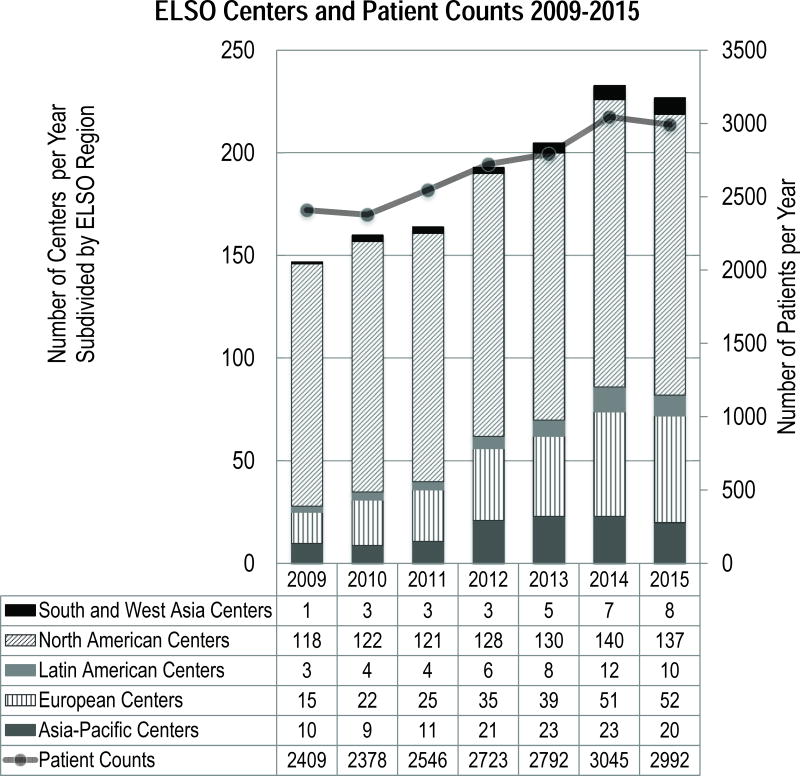
The entire ELSO Registry currently contains 59,969 ECLS cases with 61% survival (Table 1a) and 21,907 of those have been performed since 2009 (Table 1b). From 2009–2015 the number of centers reporting annual pediatric ECLS cases increased by more than 50%, from 147 centers in 2009 to 227 centers in 2015 (Figure 1). While North American centers still predominated, the proportion of non-North American centers doubled from 20% in 2009 (29/147) to 40% (90/227) in 2015. During the same time period, the annual number of children receiving ECLS increased by 24%.
Table 1.
| a. ECLS cases and survival to discharge, 1989–2017
| |||
|---|---|---|---|
| Number of Cases | Survived ECLS N (%) |
Survived to Hospital Discharge N (%) |
|
| Neonatal | |||
| Respiratory | 30,062 | 25,297 (84) | 22,040 (73) |
| Cardiac | 7,243 | 4,697 (65) | 2,988 (41) |
| ECPR | 1,554 | 1,048 (67) | 641 (41) |
| Pediatric | |||
| Respiratory | 8,162 | 5,487(67) | 4,699 (58) |
| Cardiac | 9,479 | 6,482 (68) | 4,844 (51) |
| ECPR | 3,469 | 1,995 (58) | 1,444 (42) |
| Total | 59,969 | 45,006 (75%) | 36,656 (61%) |
| b. ECLS cases and survival to discharge, 2009–2017
| |||
|---|---|---|---|
| Number of Cases | Survived ECLS N (%) |
Survived to Hospital Discharge N (%) |
|
| Neonatal | |||
| Respiratory | 6,586 | 5,330 (81) | 4,444 (67) |
| Cardiac | 3,285 | 2,258 (69) | 1,487 (45) |
| ECPR | 1,045 | 716 (69) | 445 (43) |
| Pediatric | |||
| Respiratory | 3,903 | 2,732 (70) | 2,353 (60) |
| Cardiac | 4,581 | 3,389 (74) | 2,600 (57) |
| ECPR | 2,507 | 1,471 (59) | 1,066 (43) |
| Total | 21,907 | 15,896 (73%) | 12,394 (59%) |
ECSL, extracorporeal life support; ECPR, ECLS to support cardiopulmonary resuscitation
Figure 1.
Number of Reporting ELSO Center and Patient Counts, 2009–2015
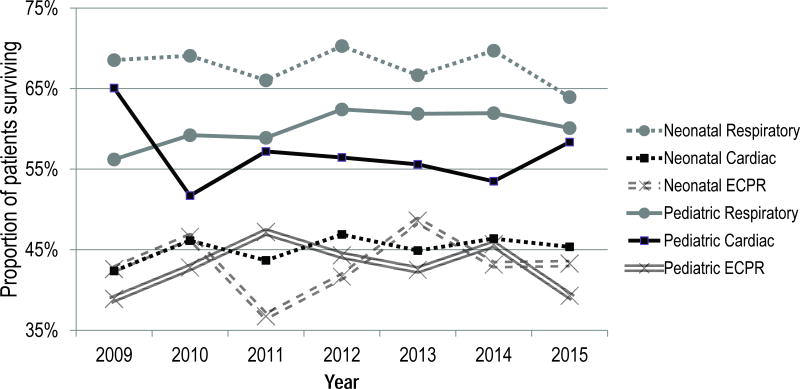
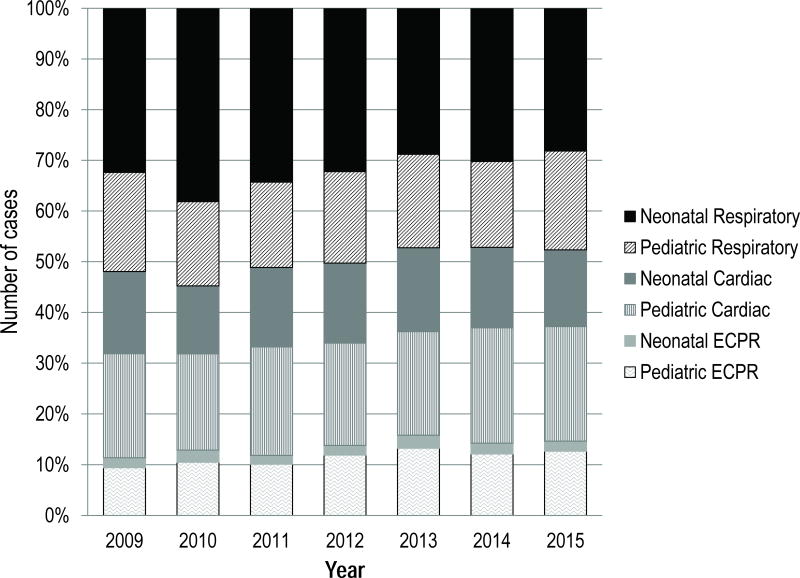
The general trends in survival and distribution of ECLS by support type remained relatively static from 2009 to 2015 (Figure 2). In 2015, ECLS survival rates clustered into two groups. Neonatal respiratory, pediatric respiratory and pediatric cardiac ECLS survival rates were all within three percentage points of 61% survival, while neonatal cardiac, neonatal ECLS to support cardiopulmonary resuscitation (ECPR) and pediatric ECPR were within three percentage points of 42% survival. In terms of distribution of support type, there was a small but persistent increase in cardiac and ECPR relative to respiratory support (Figure 3). Respiratory support constituted 52% of non-adult ECLS in 2009 and 47% in 2015.
Figure 2.
ECLS Survival by Age Group and Support Type, 2009–2015
Figure 3.
Trends in ECLS Support Type, 2009–2015
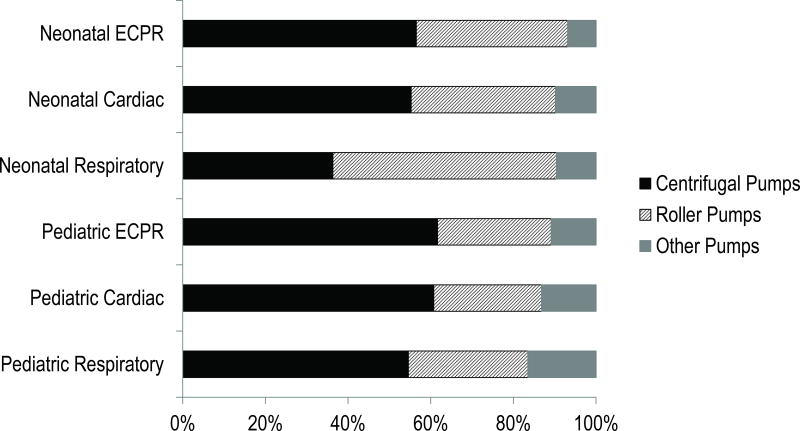
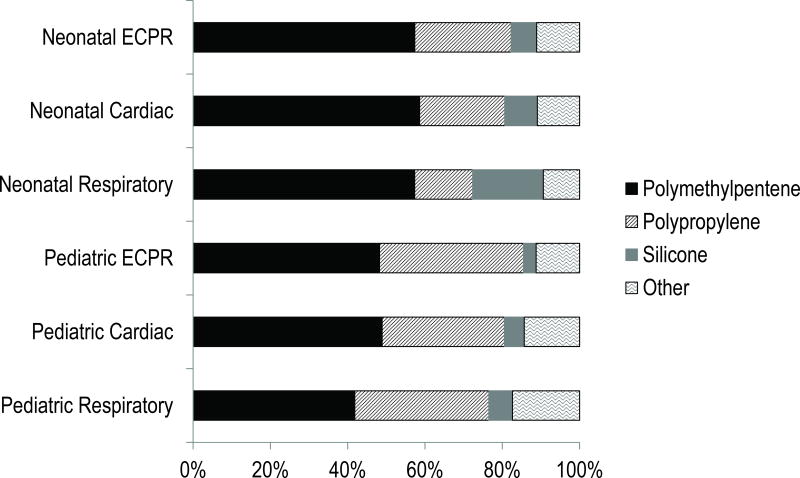
From 2009–2015, most patients received ECLS support via centrifugal pumps and polymethylpentene oxygenators (Figures 4 & 5). All types of pediatric ECLS as well as neonatal cardiac and ECPR used centrifugal pumps 55–60% of the time, but in neonatal respiratory ECLS, roller pumps predominated with a 54% utilization rate. Membrane oxygenator use differed by age group and support type. Polymethylpentene oxygenators were slightly more common (52–59%) in neonatal ECLS relative to pediatric ECLS. In pediatric cardiac and ECPR support, 49% of cases used polymethylpentene, and pediatric respiratory ECLS only utilized polymethylpentene oxygenators in 42% of cases.
Figure 4.
Trends in ECLS Mechanical Pump Type, 2009–2015
Figure 5.
Trends in ECLS Membrane Oxygenator, 2009–2015
In terms of complications, mechanical complications predominated in respiratory patients whereas they were less common in cardiac and ECPR patients. Brain death was rare, but it was much more commonly reported in non-neonates compared to neonates. Intracranial hemorrhage was more common among neonates, but intracranial hemorrhage carried a higher association with mortality in non-neonates.
Neonatal Respiratory
Despite the decrease in the use of ECLS in neonates (Figure 2), neonatal respiratory failure is still the most common indication for ECLS (Table 1a & 1b). To date, ECLS centers have submitted data on a total of 30,062 neonates with respiratory failure needing ECLS, of which 821 cases were in 2015. Neonates receiving ECLS for respiratory support have the highest survival rate, but their survival rate has declined from 75% in the 2012 Registry Report to 62% in 2015 (Table 2).8
Table 2.
Neonatal respiratory ECLS by primary diagnosis, survival and average run length, 2009–2015
| Proportion of Cases N (%) |
Survival to Hospital Discharge (%) |
Average Run Duration, days |
|
|---|---|---|---|
| Congenital diaphragmatic hernia | 1,851 (32) | 50 | 12 |
| Meconium aspiration syndrome | 1,393 (24) | 93 | 6 |
| PPHN | 1,233 (21) | 74 | 7 |
| Respiratory distress syndrome | 71 (1) | 79 | 6 |
| Sepsis | 256 (4) | 55 | 7 |
| Other | 1,035 (18) | 59 | 9 |
|
| |||
| Total | 5,839 | 68 | 9 |
ECLS, extracorporeal life support; PPHN, persistent pulmonary hypertension of the newborn
The most common neonatal diagnosis requiring respiratory ECLS was congenital diaphragmatic hernia (CDH) (32%), followed by meconium aspiration syndrome (24%) and persistent pulmonary hypertension (21%). Respiratory distress syndrome and sepsis were the least common conditions for which neonates were treated with respiratory ECLS (Table 2). CDH continued to have the lowest survival rate (50%) compared to other conditions requiring neonatal respiratory ECLS, while meconium aspiration syndrome had the highest rate of survival (93%) (Table 2). Of note, in 2015, the proportion of neonates surviving to discharge after ECLS was 64% (Figure 2), lower when compared to the earlier years of this study.
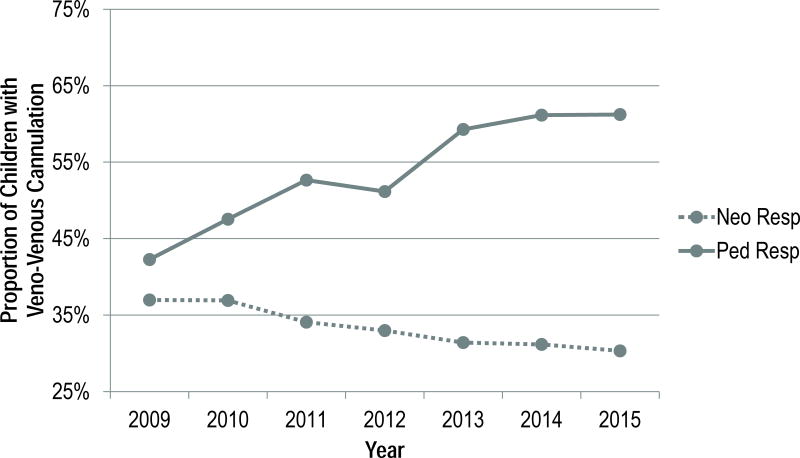
Neonatal respiratory ECLS was the only subtype where roller pumps predominated (Figure 4), and there was a decreasing trend in venovenous relative to venoarterial cannulation in neonatal respiratory ECLS (Figure 6).
Figure 6.
Trends in use of venovenous cannulation for respiratory support ECLS, 2009–2015
The most commonly reported type of mechanical complication for neonatal respiratory ECLS was cannula related (12%). The most common patient-level complication was intra-cerebral hemorrhage (11%) where its potential contribution to mortality was 28% (Table 3). Surgical site bleeding and GI hemorrhage were less common, but two of every three children suffering these complications died compared to an average mortality of one in three.
Table 3.
Mechanical and patient related complications with respiratory ECLS, 2009–2015
| Neonatal Respiratory | Pediatric Respiratory | |||||
|---|---|---|---|---|---|---|
| Complications N (%) |
After complication survival N (%) |
Difference between average and after complication survival (%) |
Complications N (%) |
After complication survival N (%) |
Difference between average and after complication survival (%) |
|
| Mechanical | ||||||
| Oxygenator Failure | 280 (5) | 147 (53) | 16 | 251 (8) | 106 (42) | 18 |
| Pump Malfunction | 84 (1) | 46 (55) | 13 | 47 (1) | 24 (51) | 9 |
| Cannula Problem | 696 (12) | 400 (57) | 11 | 515 (15) | 305 (59) | 1 |
| Air in Circuit | 209 (4) | 119 (57) | 11 | 181 (5) | 90 (50) | 10 |
| Patient | ||||||
| Seizure by EEG | 158 (3) | 77 (49) | 19 | 111 (3) | 39 (35) | 25 |
| Cerebral Infarct | 180 (3) | 79 (44) | 24 | 158 (7) | 54 (34) | 26 |
| ICH | 643 (11) | 255 (40) | 28 | 243 (5) | 52 (21) | 39 |
| Brain Death | 23 (0.4) | 0 | 68 | 117 (4) | 0 | 60 |
| Cardiac Tamponade | 13 (0.2) | 5 (38) | 30 | 84 (3) | 38 (45) | 15 |
| Surgical Site Bleeding | 386 (7) | 134 (35) | 33 | 332 (10) | 168 (51) | 9 |
| GI Hemorrhage | 89 (2) | 29 (33) | 35 | 135 (4) | 53 (39) | 21 |
| Amputation | 0 (0) | - | - | 5 (0.1) | 4 (80) | −20 |
ECLS, extracorporeal life support; ICH, intracranial hemorrhage
Pediatric Respiratory
Pediatric respiratory ECLS refers to children aged 29 days through 17 years who were placed on ECLS for respiratory support. In 2009, during the H1N1 influenza pandemic, the 459 annual cases exceeded the 385 cases in 2010, but every year since then the number of cases has grown. In 2015 there were 569 pediatric respiratory ECLS runs, a 48% increase over 6 years.
The most common etiology for pediatric respiratory failure was infectious lung disease (bronchiolitis, pneumonia, and pertussis) representing over 30% of cases. Survival remained stable between 56% – 62%, with asthma having the best survival to hospital discharge (88%) and pertussis having the lowest survival (32%). Average length of pediatric runs varied between 7 – 17 days, depending on the indication (Table 4).
Table 4.
Pediatric respiratory ECLS by primary diagnosis, survival and average run length, 2009–2015
| Proportion of Cases N (%) |
Survival to Hospital Discharge (%) |
Average Run Duration, days |
|
|---|---|---|---|
| Asthma | 91 (3) | 88 | 6 |
| Bronchiolitis | 329 (10) | 78 | 13 |
| Viral pneumonia | 338 (10) | 63 | 14 |
| Aspiration pneumonia | 28 (1) | 82 | 12 |
| Bacterial pneumonia | 257 (8) | 67 | 13 |
| Other pneumonia | 29 (1) | 45 | 17 |
| Pertussis | 69 (2) | 32 | 17 |
| Acute Respiratory Failure | 730 (22) | 60 | 13 |
| Chronic Lung Disease | 98 (3) | 56 | 15 |
| Pulmonary Hemorrhage | 35 (1) | 60 | 9 |
| Drowning, Inhalation, Foreign Body | 75 (2) | 69 | 7 |
| Postoperative or Trauma | 119 (4) | 57 | 11 |
| Sepsis | 281 (8) | 52 | 10 |
| Congenital Heart Disease | 152 (5) | 53 | 9 |
| Other | 681 (21) | 52 | 9 |
|
| |||
| Total | 3,312 | 60 | 12 |
ECLS, extracorporeal life support
Coincident with the growth in pediatric respiratory ECLS support there has also been a growth in the proportion of respiratory support utilizing venovenous ECLS. Venovenous cannulations increased from 41% of pediatric respiratory ECLS cases in 2009 to 59% in 2015 (Figure 6).
Pediatric respiratory ECLS patients who experienced complications had a higher mortality rate than the average pediatric respiratory ECLS mortality rate. While mechanical complications remained more common than patient complications, fortunately, they also seemed to have a lower associated mortality risk (Table 3). Among all non-adult ECLS cases, oxygenator and cannula malfunctions were most common in the pediatric respiratory ECLS at 8% and 15% of cases respectively. Patient related complications were reported in 10% of cases or less, but patients who suffered these complications had lower survival rates. Intracranial hemorrhage was reported in 5% of patients, but it was the most consequential of patient-related complications with 79% of those patients dying before hospital discharge (Table 3).
Neonatal and Pediatric Cardiac
From 1989 to present the ELSO Registry contained 7,243 neonatal cardiac ECLS cases and 9,479 pediatric cardiac ECLS cases (Table 1a). Between 2009 and 2015, neonatal and pediatric cardiac ECLS cases included 2,849 neonates and 3,850 pediatric patients (Table 5 & 6). Neonates had a mean ECLS run duration of 6 days and 45% survival to hospital discharge (Table 5). Pediatric patients had longer runs (mean run length 7 days) and better survival, 57% (Table 6). Annual ECLS survival to hospital discharge for the neonatal cardiac cohort was relatively stable from 2009 to 2015, ranging between 42%-47%. Pediatric cardiac ECLS survival was more variable ranging from 52% to 65% (Figure 2). Annual volumes varied for cardiac ECLS with a general trend towards increasing annual volumes. Yearly volumes were between 310 and 470 runs for neonates and between 439 and 673 runs for pediatric cardiac ECLS (Figure 3).
Table 5.
Neonatal cardiac ECLS by primary diagnosis, survival and average run length, 2009–2015
| Proportion of Cases N (%) |
Survival to Hospital Discharge (%) |
Average Run Duration, days |
|
|---|---|---|---|
| Congenital Heart Disease | 2,301 (81) | 44 | 6 |
| HLHS | 644 (23) | 40 | 6 |
| LVOTO | 178 (6) | 41 | 6 |
| RVOTO | 95 (3) | 39 | 6 |
| Septal Defects | 172 6) | 44 | 6 |
| Cyanotic with decreased pulmonary flow | 348 (12) | 48 | 7 |
| Cardiac Arrest | 41 (1) | 41 | 7 |
| Cardiogenic Shock | 57 (2) | 39 | 5 |
| Cardiomyopathy | 44 (2) | 59 | 9 |
| Myocarditis | 38 (1) | 50 | 11 |
| Other | 368 (13) | 50 | 7 |
|
| |||
| Total | 2,849 | 45 | 6 |
ECLS, extracorporeal life support; HLHS, hypoplastic left heart syndrome; LVOTO, left ventricular outflow tract obstruction; RVOTO, right ventricular outflow tract obstruction
Table 6.
Pediatric Cardiac ECLS by primary diagnosis, survival and average run length, 2009–2015
| Proportion of Cases N (%) |
Survival to Hospital Discharge (%) |
Average Run Duration, days |
|
|---|---|---|---|
| Congenital Heart Disease | 2,010 (52) | 54 | 6 |
| HLHS | 283 (7) | 46 | 7 |
| LVOTO | 212 (5) | 57 | 6 |
| RVOTO | 108 (3) | 62 | 6 |
| Septal Defects | 323 (8) | 49 | 6 |
| Cyanotic with decreased pulmonary flow | 271 (7) | 52 | 6 |
| Cardiac Arrest | 128 (3) | 45 | 6 |
| Cardiogenic Shock | 175 (5) | 61 | 6 |
| Cardiomyopathy | 317 (8) | 65 | 8 |
| Myocarditis | 204 (5) | 76 | 8 |
| Other | 1,016 (26) | 57 | 8 |
|
| |||
| Total | 3,850 | 57 | 7 |
ECLS, extracorporeal life support; HLHS, hypoplastic left heart syndrome; LVOTO, left ventricular outflow tract obstruction; RVOTO, right ventricular outflow tract obstruction
Congenital heart disease was the most common neonatal cardiac indication for ECLS, comprising more than 80% of cases (Table 5). Neonates with cardiomyopathy and myocarditis had longer support duration but higher survival compared to other indications. Among neonates with congenital heart disease, those with hypoplastic left heart syndrome (HLHS), left ventricular outflow obstruction and cyanosis with increased pulmonary blood flow (transposition of the great arteries and truncus arteriosus) had lower survival relative to those with decreased pulmonary blood flow (including tetralogy of Fallot, double outlet right ventricle with restricted pulmonary blood flow, Ebstein’s anomaly).
Pediatric cardiac ECLS was also most often delivered to support children with congenital heart disease (52%). Among pediatric cardiac ECLS patients, those with myocarditis had the highest survival rate (76%), while those requiring ECLS for a cardiac arrest had the lowest survival rate (45%) (Table 6). Among non-neonatal children with congenital heart disease, patients with HLHS had the longest average duration of support and lowest survival (46%), while right-sided obstructive lesions were associated with shorter runs and highest survival (62%).
Mechanical malfunctions during ECLS were uncommon in this cohort, but patient survival to discharge after mechanical complications was reduced by 10–16% compared to average survival (Table 7). Bleeding at the surgical site was common in both neonatal and pediatric patients (26% and 25% respectively). Intracranial pathology, cerebral infarction or intracranial hemorrhage was associated with reduced survival compared to average in both neonates (12%–17%) and children (21%-31%).
Table 7.
Mechanical and patient related complications with cardiac ECLS, 2009–2015
| Neonatal Cardiac | Pediatric Cardiac | |||||
|---|---|---|---|---|---|---|
| Complications N (%) |
After complication survival N (%) |
Difference between average and after complication survival (%) |
Complications N (%) |
After complication survival N (%) |
Difference between average and after complication survival (%) |
|
| Mechanical | ||||||
| Oxygenator Failure | 123 (4) | 36 (29) | 16 | 205 (5) | 94 (46) | 11 |
| Pump Malfunction | 37 (1) | 12 (32) | 13 | 49 (1) | 22 (45) | 12 |
| Cannula Problem | 156 (5) | 52 (33) | 12 | 194 (5) | 92 (47) | 10 |
| Air in Circuit | 101 (3) | 33 (33) | 12 | 105 (3) | 49 (47) | 10 |
| Patient | ||||||
| Seizure by EEG | 100 (4) | 41 (41) | 4 | 101 (3) | 42 (42) | 15 |
| Cerebral Infarct | 93 (3) | 31 (33) | 12 | 231 (6) | 83 (36) | 21 |
| ICH | 326 (11) | 91 (28) | 17 | 251 (6) | 65 (26) | 31 |
| Brain Death | 21 (1) | 0 | 45 | 107 (3) | 0 | 57 |
| Cardiac Tamponade | 148 (5) | 62 (42) | 3 | 171 (4) | 66 (39) | 18 |
| Surgical Site Bleeding | 739 (26) | 257 (35) | 10 | 974 (25) | 496 (51) | 6 |
| GI Hemorrhage | 35 (1) | 7 (20) | 25 | 79 (2) | 18 (23) | 34 |
| Amputation | 3 (0.1) | 2 (67) | −22 | 4 (0.1) | 3 (75) | −18 |
ECLS, extracorporeal life support; ICH, intracranial hemorrhage; EEG, Electroencephalogram
Neonatal and Pediatric ECPR
The use of ECPR is increasing in children. During the study period, 3,005 ECPR runs were reported to the Registry with overall survival to hospital discharge of 43%. The ECPR cohort included 887 neonates and 2,118 pediatric patients with similar survival to discharge rate of 43% and mean ECLS run duration of five days for both age groups. From 2009 to 2015, neonatal ECPR increased by 35% from 108 to 146 annual cases and pediatric ECPR increased by 67% from 221 to 369 cases per year.
The ELSO Registry created a data collection addendum on cardiopulmonary resuscitation techniques and management for ECPR patients in 2011. Selected data from the addendum are presented in Table 8. Nearly all cardiac arrests were witnessed and over 80% of cardiac arrests leading to ECPR cannulation occurred in highly monitored environments such as the intensive care unit, operating room and emergency department. The majority of ECLS circuits used in ECPR patients were blood primed and therapeutic hypothermia was used in more than half of cases. Median duration of CPR prior to ECLS was 40 minutes [interquartile range (IQR) 25–61 minutes]; the majority (73%) received CPR < 60 minutes prior to ECLS support. Therapeutic hypothermia was commonly used following ECPR.
Table 8.
Characteristics of ECPR, 2011–2015
| Variable, N (%) | ECPR Cases N=1,828 |
|---|---|
| Arrest Location | |
| Operating Room | 116 (6) |
| Intensive care unit | 1310 (72) |
| Emergency Department | 56 (3) |
| Ward | 87 (5) |
| During Transport | 24 (1) |
| Outside Hospital | 37 (2) |
| Other Location | 127 (7) |
| Missing | 71 (4) |
| Witnessed Arrest | 1723 (94) |
| Cannulation Location | |
| Operating Room | 150 (8) |
| Intensive care unit | 1473 (81) |
| Emergency Department | 33 (2) |
| Cardiac Catheterization Laboratory | 97 (5) |
| Other Location | 29 (2) |
| Missing | 46 (3) |
| Circuit Prime | |
| Blood | 1185 (65) |
| Clear | 423 (23) |
| Other | 25 (1) |
| Missing | 195 (11) |
| Therapeutic hypothermia use | 1051 (57) |
| Duration of CPR in minutes, median [IQR] | 40 [25, 61] |
ECPR, extracorporeal life support to support cardiopulmonary resuscitation; IQR, interquartile range
ECLS complications occurred frequently in those requiring ECPR; selected complications are shown in Table 9. As might be expected, neurological injury was common in this cohort. Approximately 10% of pediatric ECPR patients met brain death criteria, but brain death was uncommonly reported in the neonatal ECPR population (2%). These rates are considerably higher than in respiratory and cardiac ECLS support.
Table 9.
Mechanical and patient related complications after ECPR support, 2009–2015
| Neonatal ECPR | Pediatric ECPR | |||||
|---|---|---|---|---|---|---|
| Complications N (%) |
After complication survival N (%) |
Difference between average and after complication survival (%) |
Complications N (%) |
After complication survival N (%) |
Difference between average and after complication survival (%) |
|
| Mechanical | ||||||
| Oxygenator Failure | 26 (3) | 10 (38) | 5 | 82 (4) | 29 (35) | 8 |
| Pump Malfunction | 12 (1) | 5 (42) | 1 | 20 (1) | 7 (35) | 8 |
| Cannula Problem | 61 (7) | 23 (38) | 5 | 175 (8) | 59 (34) | 9 |
| Air in Circuit | 58 (6) | 19 (33) | 10 | 63 (3) | 27 (43) | 0 |
| Patient | ||||||
| Seizure by EEG | 54 (6) | 12 (22) | 21 | 137 (6) | 44 (32) | 11 |
| Cerebral Infarct | 46 (5) | 12 (26) | 17 | 238 (11) | 67 (28) | 15 |
| ICH | 128 (14) | 41 (32) | 11 | 182 (9) | 43 (24) | 19 |
| Brain Death | 14 (2) | 0 | 43 | 205 (10) | 0 | 43 |
| Cardiac Tamponade | 20 (2) | 6 (30) | 13 | 50 (2) | 19 (38) | 5 |
| Surgical Site Bleeding | 150 (17) | 47 (31) | 12 | 298 (14) | 124 (42) | 1 |
| GI Hemorrhage | 12 (1) | 2 (17) | 26 | 64 (3) | 7 (11) | 32 |
| Amputation | 0 (0) | - | - | 5 (0.2) | 2 (40) | 3 |
ECPR, extracorporeal life support to support cardiopulmonary resuscitation; ICH, intracranial hemorrhage; EEG, Electroencephalogram; ECPR, extracorporeal life support to support cardiopulmonary resuscitation
Discussion
Since 2009 we have continued to see growth in the utilization of ECLS both in the number of centers and in the number of patients reported to the ELSO Registry. ECLS survival and the distribution of ECLS support types have been stable. From 2009 to 2015, approximately 48% of ECLS delivered to children was respiratory and 52% was cardiac or ECPR. Respiratory ECLS continued to use venoarterial cannulations in 70% of neonatal cases and 40% of pediatric cases. Roller pumps continued to predominate in neonatal respiratory disease, but centrifugal pumps were most common in all other ECLS subgroups. Among oxygenators, polymethylpentene was the most common in all age groups and support types.
This is the first ELSO Registry report to summarize data collected as part of the ECPR addendum. The report offers new insights into pediatric ECPR. It describes the duration of chest compressions, the location of arrest and cannulation and the proportion of patients receiving therapeutic hypothermia. Thirty percent of patients are cannulated less than 30 minutes after compressions are started and 81% of arrests leading to ECPR occur in resource intense areas such as the intensive care unit, operating room and emergency department. Half of ECPR patients received therapeutic hypothermia in the ELSO Registry. The therapeutic hypothermia after in-hospital cardiac arrest in children (THAPCA) trial was conducted from 2009–2015 and found no benefit of therapeutic hypothermia.24 It will be interesting to observe if the proportion of patients receiving therapeutic hypothermia declines in the future.
Among patient complications, brain death was much less common in neonates than in pediatric patients for each support type. We suspect that this difference is at least partially due to difficulty in diagnosis of brain death in neonates as compared to non-neonatal children.
As of September 2016, the ELSO Registry began collecting ICD-10 diagnostic codes, severity of illness data, date and times for procedures and complications and implemented logical limits to data entry. In addition, researchers have published pre-ECLS specific measures of severity of illness for adult respiratory, adult cardiac, pediatric respiratory and neonatal respiratory ECLS using the ELSO Registry.25–30 Also, there are ongoing initiatives to update a formal database dictionary for all data fields, to validate data entry both through external validation and measures of inter-rater reliability of data abstraction. Finally, there is ongoing work in the ELSO Registry to update dynamic quality reporting of outcomes, processes and structures in ECLS care.
The ELSO Registry has been and continues to be used by regulatory agencies and industry to evaluate technology. The Registry is regularly used by clinicians to inform and support the care of ECLS patients. The Registry has been used in research trials such as the prospective trial of a pediatric ventricular assist device31 as well as over 225 retrospective studies listed in PubMed (April 1, 2017). The ELSO Registry is also used to promote quality improvement through real-time benchmarking reports that compare an individual center’s survival and complication rates to those of peer institutions. These initiatives will continue to improve the world’s largest ECLS Registry so that future patients requiring ECLS will benefit from improved care and technology.
Acknowledgments
Source of Funding: This research did not have funding support.
Footnotes
Conflict of Interest: Drs. Barbaro and Thiagarajan are on the steering committee for the Extracorporeal Life Support Organization. Dr. Barbaro is the ELSO Registry Chair and Dr. Thiagarajan is the immediate past Registry Chair. The authors have no other conflicts of interest relevant to this article to disclose.
References
- 1.Toomasian JM, Snedecor SM, Cornell RG, Cilley RE, Bartlett RH. National experience with extracorporeal membrane oxygenation for newborn respiratory failure. Data from 715 cases. ASAIO Trans. 1988;34(2):140–7. doi: 10.1097/00002480-198804000-00011. [DOI] [PubMed] [Google Scholar]
- 2.Stolar CJ, Snedecor SM, Bartlett RH. Extracorporeal membrane oxygenation and neonatal respiratory failure: experience from the extracorporeal life support organization. J Pediatr Surg. 1991;26(5):563–71. doi: 10.1016/0022-3468(91)90708-2. [DOI] [PubMed] [Google Scholar]
- 3.Stolar CJ, Delosh T, Bartlett RH. Extracorporeal Life Support Organization 1993. ASAIO J. 1993;39(4):976–9. [PubMed] [Google Scholar]
- 4.Tracy TF, Jr, DeLosh T, Bartlett RH. Extracorporeal Life Support Organization 1994. ASAIO J. 1994;40(4):1017–9. [PubMed] [Google Scholar]
- 5.Bartlett RH. Extracorporeal Life Support Registry Report 1995. ASAIO J. 1997;43(1):104–7. [PubMed] [Google Scholar]
- 6.Conrad SA, Rycus PT, Dalton H. Extracorporeal Life Support Registry Report 2004. ASAIO J. 2005;51(1):4–10. doi: 10.1097/01.mat.0000151922.67540.e9. [DOI] [PubMed] [Google Scholar]
- 7.Haines NM, Rycus PT, Zwischenberger JB, Bartlett RH, Undar A. Extracorporeal Life Support Registry Report 2008: neonatal and pediatric cardiac cases. ASAIO J. 2009;55(1):111–6. doi: 10.1097/MAT.0b013e318190b6f7. [DOI] [PubMed] [Google Scholar]
- 8.Paden ML, Conrad SA, Rycus PT, Thiagarajan RR. Extracorporeal Life Support Organization Registry Report 2012. ASAIO J. 2013;59(3):202–10. doi: 10.1097/MAT.0b013e3182904a52. [DOI] [PubMed] [Google Scholar]
- 9.Thiagarajan RR, Barbaro RP, Rycus PT, et al. Extracorporeal Life Support Organization Registry International Report 2016. Asaio j. 2017;63(1):60–67. doi: 10.1097/MAT.0000000000000475. [DOI] [PubMed] [Google Scholar]
- 10.Organization ELS. ECLS Registry Report. Organization ELS; 2017. Available at: http://www.elso.org. [Google Scholar]
- 11.MacLaren G, Combes A, Bartlett RH. Contemporary extracorporeal membrane oxygenation for adult respiratory failure: life support in the new era. Intensive Care Med. 2012;38(2):210–20. doi: 10.1007/s00134-011-2439-2. [DOI] [PubMed] [Google Scholar]
- 12.Gadepalli SK, Hirschl RB. Extracorporeal life support: updates and controversies. Semin Pediatr Surg. 2015;24(1):8–11. doi: 10.1053/j.sempedsurg.2014.11.002. [DOI] [PubMed] [Google Scholar]
- 13.Leteurtre S, Martinot A, Duhamel A, et al. Validation of the paediatric logistic organ dysfunction (PELOD) score: prospective, observational, multicentre study. Lancet. 2003;362(9379):192–7. doi: 10.1016/S0140-6736(03)13908-6. [DOI] [PubMed] [Google Scholar]
- 14.Leteurtre S, Duhamel A, Salleron J, Grandbastien B, Lacroix J, Leclerc F. PELOD-2: an update of the PEdiatric logistic organ dysfunction score. Crit Care Med. 2013;41(7):1761–73. doi: 10.1097/CCM.0b013e31828a2bbd. [DOI] [PubMed] [Google Scholar]
- 15.Shann F, Pearson G, Slater A, Wilkinson K. Paediatric index of mortality (PIM): a mortality prediction model for children in intensive care. Intensive Care Med. 1997;23(2):201–7. doi: 10.1007/s001340050317. [DOI] [PubMed] [Google Scholar]
- 16.Slater A, Shann F, Pearson G. PIM2: a revised version of the Paediatric Index of Mortality. Intensive Care Med. 2003;29(2):278–85. doi: 10.1007/s00134-002-1601-2. [DOI] [PubMed] [Google Scholar]
- 17.Straney L, Clements A, Parslow RC, et al. Paediatric index of mortality 3: an updated model for predicting mortality in pediatric intensive care*. Pediatr Crit Care Med. 2013;14(7):673–81. doi: 10.1097/PCC.0b013e31829760cf. [DOI] [PubMed] [Google Scholar]
- 18.Pollack MM, Patel KM, Ruttimann UE. PRISM III: an updated Pediatric Risk of Mortality score. Crit Care Med. 1996;24(5):743–52. doi: 10.1097/00003246-199605000-00004. [DOI] [PubMed] [Google Scholar]
- 19.Pollack MM, Holubkov R, Funai T, et al. The Pediatric Risk of Mortality Score: Update 2015. Pediatr Crit Care Med. 2015 doi: 10.1097/PCC.0000000000000558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med. 1985;13(10):818–29. [PubMed] [Google Scholar]
- 21.Zimmerman JE, Kramer AA, McNair DS, Malila FM. Acute Physiology and Chronic Health Evaluation (APACHE) IV: hospital mortality assessment for today’s critically ill patients. Crit Care Med. 2006;34(5):1297–310. doi: 10.1097/01.CCM.0000215112.84523.F0. [DOI] [PubMed] [Google Scholar]
- 22.Le Gall JR, Lemeshow S, Saulnier F. A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. Jama. 1993;270(24):2957–63. doi: 10.1001/jama.270.24.2957. [DOI] [PubMed] [Google Scholar]
- 23.Ferreira FL, Bota DP, Bross A, Melot C, Vincent JL. Serial evaluation of the SOFA score to predict outcome in critically ill patients. Jama. 2001;286(14):1754–8. doi: 10.1001/jama.286.14.1754. [DOI] [PubMed] [Google Scholar]
- 24.Moler FW, Silverstein FS, Holubkov R, et al. Therapeutic Hypothermia after In-Hospital Cardiac Arrest in Children. N Engl J Med. 2017;376(4):318–329. doi: 10.1056/NEJMoa1610493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schmidt M, Bailey M, Sheldrake J, et al. Predicting survival after extracorporeal membrane oxygenation for severe acute respiratory failure. The Respiratory Extracorporeal Membrane Oxygenation Survival Prediction (RESP) score. Am J Respir Crit Care Med. 2014;189(11):1374–82. doi: 10.1164/rccm.201311-2023OC. [DOI] [PubMed] [Google Scholar]
- 26.Schmidt M, Burrell A, Roberts L, et al. Predicting survival after ECMO for refractory cardiogenic shock: the survival after veno-arterial-ECMO (SAVE)-score. Eur Heart J. 2015;36(33):2246–56. doi: 10.1093/eurheartj/ehv194. [DOI] [PubMed] [Google Scholar]
- 27.Barbaro RP, Boonstra PS, Paden ML, et al. Development and validation of the pediatric risk estimate score for children using extracorporeal respiratory support (Ped-RESCUERS) Intensive Care Med. 2016;42(5):879–88. doi: 10.1007/s00134-016-4285-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bailly DK, Reeder RW, Zabrocki LA, et al. Development and Validation of a Score to Predict Mortality in Children Undergoing Extracorporeal Membrane Oxygenation for Respiratory Failure: Pediatric Pulmonary Rescue With Extracorporeal Membrane Oxygenation Prediction Score. Crit Care Med. 2016 doi: 10.1097/CCM.0000000000002019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Barbaro RP, Bartlett RH, Chapman RL, et al. Development and Validation of the Neonatal Risk Estimate Score for Children Using Extracorporeal Respiratory Support. J Pediatr. 2016;173:56–61.e3. doi: 10.1016/j.jpeds.2016.02.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Maul TM, Kuch BA, Wearden PD. Development of Risk Indices for Neonatal Respiratory Extracorporeal Membrane Oxygenation. Asaio j. 2016;62(5):584–90. doi: 10.1097/MAT.0000000000000402. [DOI] [PubMed] [Google Scholar]
- 31.Fraser CD, Jr, Jaquiss RD, Rosenthal DN, et al. Prospective trial of a pediatric ventricular assist device. N Engl J Med. 2012;367(6):532–41. doi: 10.1056/NEJMoa1014164. [DOI] [PubMed] [Google Scholar]