Abstract
Objective
To review the literature and conduct a meta-analysis to determine the effectiveness and safety of the combined endoscopic-transfacial approach for parotid sialolith management.
Data Sources
PubMed 1946-, Embase 1947-, CINAHL, the Cochrane Database of Systematic Reviews, Cochrane Database of Abstracts of Review Effects (DARE), Cochrane Central Register of Controlled Trials (CENTRAL), clinicaltrials.gov, Proquest Dissertations and Theses, and FirstSearch Proceedings to March, 2015.
Review Methods
Published prospective or retrospective English-language studies with reported outcomes of more than one patient undergoing the combined endoscopic-transfacial procedure for parotid sialolithiasis were included. Two independent authors screened all eligible studies and reviewed and extracted data from relevant publications. Weighted pooled proportions for stone removal, symptom improvement, gland preservation, and complications were calculated.
Results
Ten studies, primarily retrospective single-institution studies, were included in the final analysis with a total of 184 patients. Overall, the procedure was noted to be successful with low risk; the weighted pooled proportions were 0.99 (95%CI 0.97 to 1.00) for stone removal, 0.97 (95%CI 0.93 to 0.99) for symptom improvement, 1 (95%CI 0.99 to 1.00) for gland preservation, and 0.06 (95%CI 0.01 to 0.15) for complications.
Conclusion
While our analysis is primarily based on retrospective data, the evidence shown here suggests that the combined endoscopic-transfacial technique is an effective treatment for parotid gland sialolithiasis not amenable to intraoral or purely endoscopic removal. This approach results in high rates of symptom improvement and gland preservation. The complication rates are low, further supporting the use of this technique.
Keywords: parotid sialolithiasis, sialendoscopy, combined endoscopic-transfacial approach, meta-analysis
Introduction
Salivary gland obstruction is characterized by swelling and pain, often worse with eating. Frequent causes of salivary duct obstruction include sialoliths, stenosis and mucous plugs.1 Sialolithiasis is responsible for 60–70% of all salivary duct obstructions.2 While sialolithiasis is more commonly seen in the submandibular gland than the parotid gland, approximately 20% of symptomatic stones arise in Stenson’s duct or its branches.3
Over the past two decades, minimally invasive techniques for the management of symptomatic parotid sialolithiasis have been developed and accepted. Sialendoscopy utilizes specially-designed miniature semi-rigid or semi-flexible endoscopes to access the salivary duct system intraorally and visualize the lumen. Multiple reports demonstrate the feasibility of the purely endoscopic procedure in removing submandibular and parotid sialoliths with high success rates ranging from 76% to 86%.4,5 However, the intraoral approach alone fails in a small percentage of cases, due to the large size of the stone or location in the posterior or proximal aspects of the duct with a distal stricture or stenosis.6 In 2002, Nahlieli et al. reported a case series of 12 patients in which 75% of patients had successful extraction of parotid sialoliths using a combined transfacial and endoscopic or ultrasound technique. In this series, the stone was identified by endoscopy or ultrasound, and a 1 cm transfacial skin incision was used to extract the stone. McGurk et al. reported a modified version of this technique in 2006 using endoscopy to locate parotid sialoliths and a preauricular skin flap to expose the parotid gland and allow for stone removal.7 More recent publications have described the use of facial nerve monitoring and facelift type incisions.8,9
Prior to the availability of sialendoscopy, the treatment options for parotid sialoliths were limited to attempting transfacial or intraoral stone localization and removal, partial parotidectomy, or conservative management relying on eventual gland atrophy. The combined transfacial-endoscopic approach described above expands the application of the minimally invasive, gland-preserving approach to large or impacted parotid sialoliths. However, while expected to be less morbid than a parotidectomy, the combined endoscopic-transfacial technique is potentially associated with increased risks relative to a purely endoscopic approach. Major complications could include avulsion of the duct and facial nerve paralysis.10 Minor complications include postoperative sialocele, sensory deficits, salivary fistula, scar and contour deformity, or duct stricture.11 Additionally, clinical failure of the procedure is a possibility with symptomatic severe duct stenosis or stricture and/or retained stone fragments ultimately requiring repeat sialendoscopy or superficial parotidectomy. In addition, this procedure is often more technically challenging than endoscopic stone retrieval. Many small series, often with pooled data analysis of submandibular and parotid sialoliths, have demonstrated success with this combined endoscopic-transfacial approach; however, there has not been a comprehensive evaluation of this procedure for parotid sialolithiasis addressing patient outcomes and safety.
Due to the paucity of knowledge within the otolaryngology literature regarding this relatively new combined endoscopic-transfacial approach, we conducted a systematic review to investigate the effectiveness and safety of this procedure for the management of parotid sialolithiasis. Specifically, the objective of this study was to evaluate the rates of successful stone removal, gland preservation, symptom improvement, and complications for the combined endoscopic-transfacial approach for parotid sialolithiasis. We hypothesized that this procedure would be highly effective with low rates of major adverse events.
Methods
The published literature was searched using strategies created by a medical librarian for the concepts of sialoliths, endoscopy, and open surgery. These strategies were established using a combination of standardized terms and key words, and were implemented in PubMed 1946-, Embase 1947-, CINAHL, the Cochrane Database of Systematic Reviews, Cochrane Database of Abstracts of Review Effects (DARE), Cochrane Central Register of Controlled Trials (CENTRAL), clinicaltrials.gov, Proquest Dissertations and Theses, and FirstSearch Proceedings. Searches were limited to English using database supplied filters. The search was completed in March, 2015, exported to EndNote, and duplicates were removed. Our comprehensive search strategy and list of search terms are available in online Supporting Document S1.
Candidate articles were independently reviewed by two authors familiar with the subject matter. Articles were considered eligible if they included greater than one patient who underwent a combined endoscopic-transfacial procedure for parotid stone removal and reported the outcome of the procedure. Case reports were excluded as they constitute lower level evidence than case series. In instances where multiple articles reported data from the same institution, the most recent publication was used in our analysis. We attempted to collect the following data from every eligible study: title, year of publication, timeframe of data collection, number of patients undergoing the combined approach for parotid sialolithiasis, average age of patients, follow-up time, average stone size, complication rates, rate of stone removal, rate of gland preservation, and rate of symptom improvement. Reported complications were assumed to be mutually exclusive, occurring in separate patients for the purpose of analysis.
Each included study was critically appraised and scored using a case series quality assessment checklist developed and validated by Guo et al.12 This checklist includes 20 criteria that attempt to evaluate the quality of a case series in two domains – the risk of bias and the quality of reporting. Two criteria from the checklist that were deemed to be less relevant to our analysis and thus not assessed were whether included studies reported cointerventions and whether outcome measures were applied before and after the intervention. Therefore, our modified checklist included a total of 18 criteria. For each criterion met by a study, 1 point was awarded. Instances where it was unclear or where a study partially met a criterion, half a point was awarded. No points were awarded when a study did not meet a criterion. Therefore, each study received a score ranging from 0 to 18. A single author evaluated each of the included studies according to this modified checklist and results of the quality assessment were discussed with a second author. The 2 authors were in agreement with quality assessment scores.
Analysis combining data from multiple studies was performed using STATA statistical software version 13.1 (STATA Corporation, College Station, TX). Stone removal, symptom improvement, gland preservation, and complication rates were calculated using the META module for proportions. Results were presented as weighted pooled proportions with associated 95% confidence intervals (95% CI). Following proportions meta-analysis, heterogeneity was explored where present, and a random effects model was used when appropriate. For every analysis, the Freeman-Tukey double arcsine transformation was used to stabilize the variances, accounting for cells with proportions of 100%.
Results
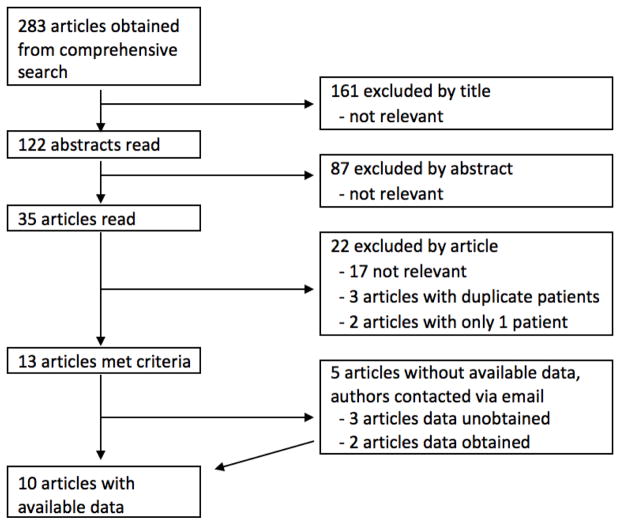
Following our comprehensive search and exclusion of duplicate studies, 283 articles were identified. A total of 122 articles remained after screening by title. After screening by abstract content, 35 articles were read in full. Two pertinent articles were excluded because they reported only one relevant case in their series. An additional 3 publications were excluded because they analyzed redundant data from the same institution. Ultimately, 13 articles met criteria for inclusion in the analysis (Figure 1). Of these 13 articles, 8 provided data for analysis. The remaining 5 articles contained relevant information; however, the success and complication rates were not specific to parotid combined endoscopic-transfacial cases. These 5 studies included pooled data on both submandibular and parotid cases, as well as purely endoscopic and combined cases. The corresponding authors for these 5 studies were contacted by email to inquire about specific results; however, data was not available for inclusion in the final analysis for 3 of these 5 studies.
Figure 1.
Flow chart of screened studies
Ultimately, 10 studies8,9,13–20 had available data for analysis, resulting in inclusion of 184 patients. Study location included the following countries: Finland, Germany, Greece, Israel, Italy, Poland, Switzerland, the UK and the US. Table 1 includes the mean age and stone size of the patient populations as well as success rate, symptom improvement rate, gland preservation rate, and complications reported by each study. The combined endoscopic-transfacial procedure was clearly described in each of the included studies. Stone localization was achieved with the endoscope and supplemented with ultrasound in some studies.17–19 In these studies, the use of intraoperative ultrasound afforded the authors quicker and easier access to the stone, more thorough evaluation for retained stone fragments, and localization of stones when the endoscope could not access the stone. Except for one study,16 all studies described the use of stents for reconstruction and stabilization of Stensen’s duct. Reported indications for stenting included concurrent duct stenosis or polyp, severely macerated duct, or an extended sialodochotomy. The techniques and materials utilized in stenting varied among studies, and stents remained in place for variable or unreported lengths of time. Many of the included studies reported the administration of prophylactic antibiotics, and 2 studies reported the additional administration of perioperative corticosteroids to prevent swelling at the surgical site and decrease the risk of postoperative ductal stenosis.16, 17
Table 1.
Description of included studies
| Author | Year | Country | Timeframe of data collection | Sample Size | Mean age | Follow-up mean/median months (range) | Mean stone size, mm | Stone removal | Symptom improvement | Gland preservation | Complications | Quality Assessment Score |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Capaccio | 2014 | Italy | 2009–2013 | 8 | 65 | 19 (3–45) | 12.6 | 88% | 100% | 100% | none | 13.5 |
| Carroll | 2013 | US | 2010–2012 | 14 | 52 | 10 (9–19) | 8.7 | 93% | 71% | 92.9% | 2 mild periauricular anesthesia 1 sialocele 1 fistula |
14 |
| Karavidas | 2010 | UK, Israel, US | 1999–2007 | 67 | 53 | 25 (2–81) | 7.2 | 99% | 97% | 100% | 1 duct perforated 2 ducts macerated 2 acute parotitis |
12 |
| Klein | 2013 | Israel | 2011–2012 | 6 | 49 | (4–23) | 8 | 100% | 100% | 100% | none | 9.5 |
| Koch | 2013 | Germany | 2006–2012 | 17 | 53 | 41 (3–68) | 9.2 | 94% | 94% | 94% | 1 hematoma | 11 |
| Konstantinidis | 2014 | Greece | 2011–2014 | 12 | 60 | 15.5 (13–20) | 8.1 | 100% | 100% | 100% | 1 hematoma 1 microbial parotitis 1 extruded stent |
13 |
| Kopec | 2013 | Poland | 2008–2011 | 5 | 62 | 20 (2–29) | 100% | 100% | 100% | none | 12 | |
| Marchal | 2007 | Switzerland | 37 | 42 | 19 | 100% | 92% | 100% | none | 9 | ||
| Mikolajczak | 2015 | Germany | 2008–2013 | 10 | 50 | 15 (6–39) | 8.7 | 100% | 100% | 100% | 2 sialoceles | 10.5 |
| Numminen | 2014 | Finland | 2009–2012 | 8 | 63 | 21.5 (2–43) | 7.6 | 75% | 75% | 87.5% | 1 suppurative parotitis | 10 |
The detailed results of our quality assessment are available in online Supporting Document S2. Studies consistently clearly described their study objectives, subject characteristics, interventions, losses to follow-up, and adverse events. Additionally, studies routinely used appropriate statistics and their conclusions were well-supported by their results. However, only 2 studies were clearly identified as prospective, and no studies utilized blinded outcome assessment or included measures of variability in reporting their results. Other shortcomings included inconsistently defined a priori outcomes, lack of validated subjective/objective outcome measures, inconsistently reported follow-up, and failure to disclose potential conflicts of interest or sources of support.
Stone removal
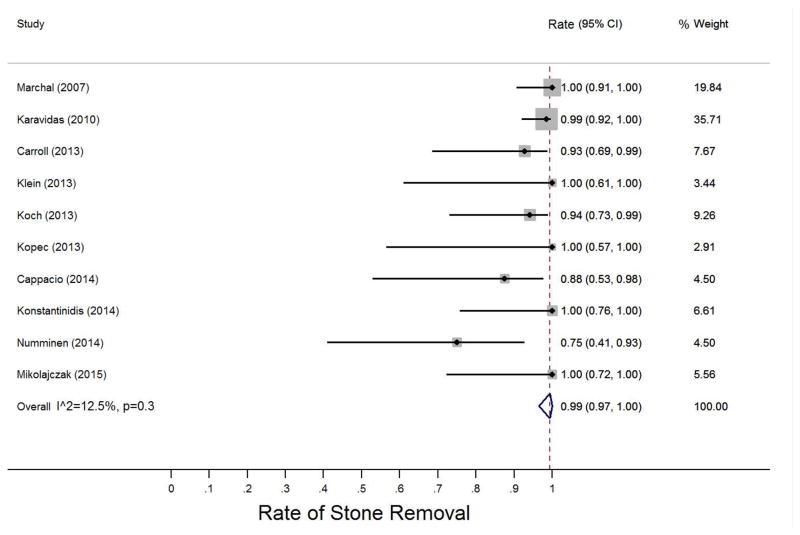
Stone removal was determined by visual inspection using the endoscope in all studies. Two studies reported use of ultrasound at 6-month follow-up, and one study reported use of sialendoscopy and scintigraphy in follow-up to evaluate for stone fragments and potential ductal stenosis. The weighted pooled proportion of stone removal was 0.99 (95%CI 0.97 to 1.00) (Figure 2). There was little heterogeneity (I2 = 12.5%, p=0.3) and a fixed-effects model was utilized.
Figure 2.
Pooled analysis for rates of stone removal
Abbreviations: CI, confidence interval
Symptom improvement
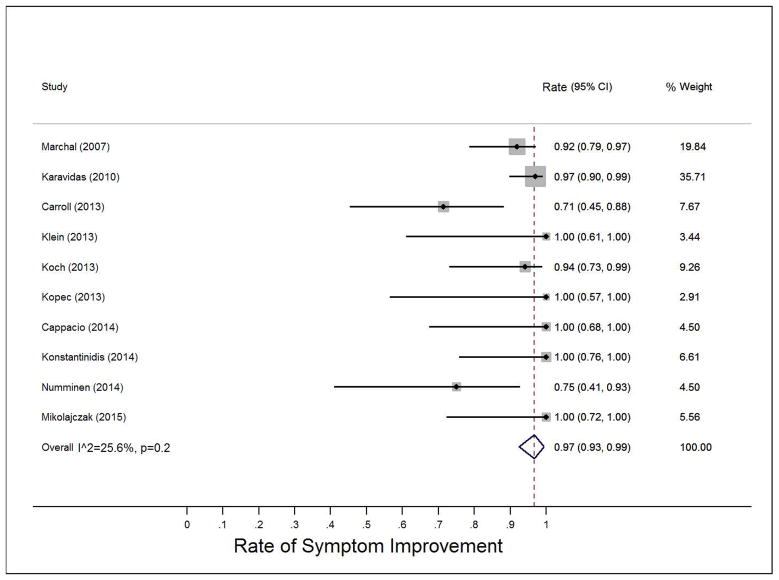
Symptom improvement was assessed at last follow-up regardless of success of stone removal. For retrospective studies, symptom improvement was self-reported and identified by chart review; but in prospective studies, symptom improvement was determined by questioning at follow-up appointments, telephone interviews, and postoperative questionnaires. The duration of follow-up and the timing of symptom assessment varied among studies. Patients requiring parotidectomy for symptom improvement were considered to be failures of symptom improvement, as they required an invasive procedure to relieve pain. The weighted pooled proportion of symptom improvement was 0.97 (95%CI 0.93 to 0.99) (Figure 3). Again, a fixed-effects model was used due to little heterogeneity (I2 = 25.6%, p=0.2).
Figure 3.
Pooled analysis for rates of symptom improvement
Abbreviations: CI, confidence interval
Gland preservation
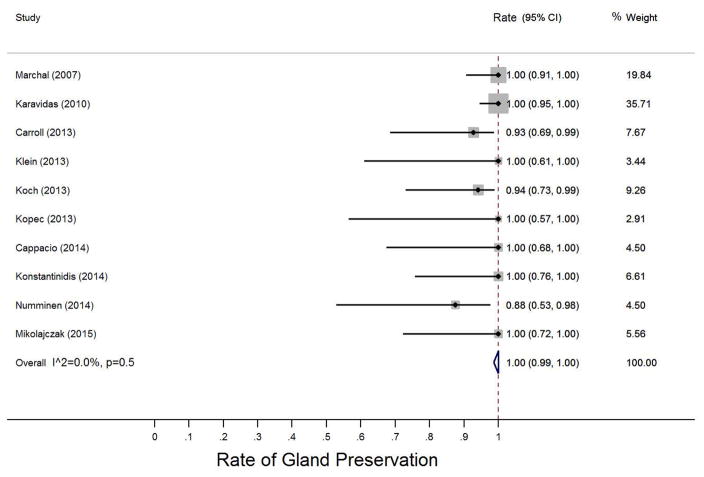
Gland preservation, or avoidance of need to remove the affected parotid gland, was determined at the last follow-up appointment. The weighted pooled proportion of gland preservation was approximated to be 1 (95%CI 0.99 to 1.00) (Figure 4) with little heterogeneity (I2 = 0.0%, p=0.5). A fixed-effects model was used in this analysis.
Figure 4.
Pooled analysis for rates of gland preservation
Abbreviations: CI, confidence interval
Complications
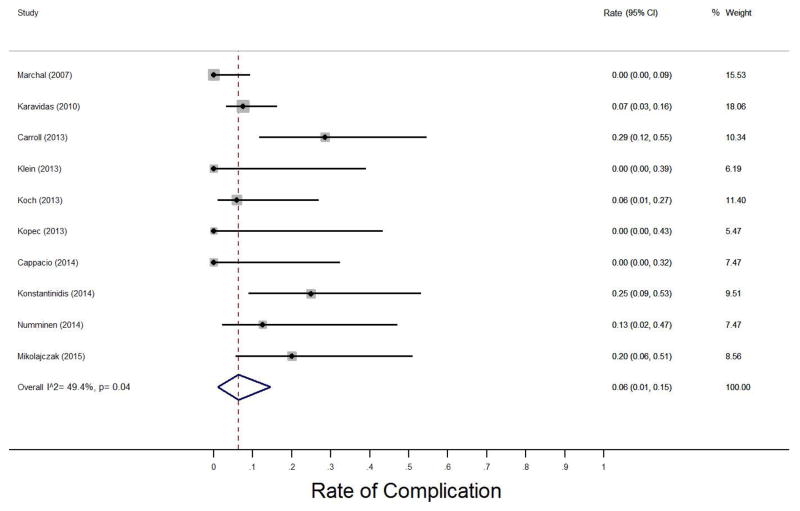
All 10 studies provided information regarding complications of their combined endoscopic-transfacial cases. Minor complications included parotitis, extruded stent, sialocele, periauricular anesthesia, fistula, and hematoma. Major complications included duct perforation and macerated duct. There were a total of 3 major complications. There were no reports of facial paralysis, which would have been considered a major complication. The weighted pooled proportion for complications was 0.06 (95%CI 0.01 to 0.15) (Figure 5). A random-effects model was used in this analysis to account for heterogeneity (I2 = 49.4%, p=0.04).
Figure 5.
Pooled analysis for rates of complication
Abbreviations: CI, confidence interval
Discussion
This is the first systematic review and meta-analysis to focus on the combined endoscopic-transfacial procedure for removal of parotid sialoliths. The pooled results suggest that this surgical approach is effective in stone removal, symptom improvement and gland preservation, with a very low risk of complications. All outcome measures evaluated – stone removal, symptom improvement and gland preservation – had success rates of 97% or higher, and the complication rate for both major and minor adverse events was only 6%.
The major alternative to the combined endoscopic-transfacial procedure for parotid sialolithiasis not amenable to intra-oral and/or endoscopic-only removal is superficial parotidectomy. There are obvious disadvantages for this invasive option including scar and contour deformity, increased risk of facial nerve paralysis, and removal of a potentially functional gland. A case series from Sydney, Australia, reported on 47 patients who underwent superficial or near-total parotidectomy for parotid sialadenitis. Of a total of 49 surgeries (2 patients had bilateral surgery), there were 18 complications, accounting for a 37% complication rate. These complications included 12 temporary facial nerve weaknesses and 1 permanent injury21. A second study that evaluated 75 patients after parotidectomy for sialadenitis reported a rate of 33% for temporary facial paralysis and 1% for permanent paralysis22. In contrast, none of the case series in our analysis of the combined endoscopic-transfacial approach reported temporary or permanent facial nerve paralysis. Seven of the included studies specifically mentioned that no patients experienced any degree of facial nerve injury, damage, or weakness. However, 3 studies reported no facial paralysis without further elaborating.13,14,19 The intraoperative localization of the sialolith, through endoscopy with or without ultrasound, provides for limited dissection of the parotid gland and duct, and thus reduces the risk to the facial nerve. Additionally, the buccal branch of the facial nerve, which typically is found in the vicinity of the parotid duct, often has redundancy of its muscular innervation, reducing the risk of a clinical facial nerve weakness.
Multiple studies have reported the successful use of sialendoscopy4,5 as well as combined endoscopic-assisted procedures 23 for both submandibular and parotid sialoliths. However, there has not been a pooled report including only combined parotid cases. Management of parotid sialoliths is inherently different than treatment of submandibular sialoliths due to limited intra-oral access to the mid and proximal parotid salivary duct, risk of injury to the facial nerve (buccal branch), and risk of increased morbidity with removal of the parotid gland as compared to the submandibular gland. A meta-analysis from 2014 included 5 studies using the combined endoscopic-transfacial technique for parotid sialoliths, however the final analysis included endoscopically-assisted transoral submandibular cases in addition to parotid cases.23 Large or impacted parotid sialoliths require a more complicated surgical technique with greater risk of complications as compared to submandibular sialoliths; thus, these procedures should be considered differently when considering safety, effectiveness and gland preservation.
While this analysis and review focuses on the endoscopic-assisted transfacial surgery, similar approaches have been described utilizing intra-operative ultrasound without sialendoscopy or other minimally invasive techniques for sialolith localization. The findings in this analysis supporting the safety, clinical improvement, and excellent gland preservation rates would be expected to apply to ultrasound or other methods for stone localization.
While sialendoscopy alone has been shown to be effective in many cases of sialolithiasis, procedure failure can result from distal stricture, multiple stones, or large sialoliths. Certain pre-operative evaluations, such as diagnostic sialendoscopy or sialogram, may provide insight into the feasibility of an endoscopic-only approach. Further study is required, however, to correlate such evaluations with operative success. We feel the possibility of a transfacial approach should be discussed with all sialolithiasis patients pre-operatively, even for a “favorable” sialolith. For parotid sialolithiasis, we recommend starting with endoscopic visualization and attempted removal. If the stone is not able to be removed endoscopically nor moved to the distal duct, and, thus, accessible intra-orally, then transition to a transfacial-endoscopic combined procedure would be undertaken. Parotidectomy would be a last resort and may be postponed to a later date depending on the clinical scenario. It is, therefore, important to have additional options in the surgeon’s skill-set for the management of large or impacted sialoliths, whether identified intra-operatively or pre-operatively. Combined endoscopic-assisted surgery for the parotid gland is a relatively new surgical approach; however, it serves as an excellent option for management of difficult cases. This procedure is not performed frequently, even in large academic medical centers, and the reported literature is sparse. Increasing the knowledge of endoscopic-assisted transfacial parotid surgery allows for more widespread access to this procedure, expanding the spectrum of salivary gland-preserving surgical approaches.
There are several limitations to this study. One is the heterogeneity of studies which were combined. While little heterogeneity was seen for stone removal, symptom improvement, and gland preservation rates with overall very high rates of success, heterogeneity was identified for complication rates. This heterogeneity may be due to small sample sizes, variable surgical techniques and instruments, variable levels of surgeon experience, and inconsistent reporting. We are unable to account for these specific differences in the analysis. Our conclusions are also limited by the inclusion of primarily retrospective studies. There is an inherent risk of recall bias when retrospectively assessing both symptom improvement and complications, and utilizing chart review is not ideal for identifying either symptom improvement or complications. Additionally, the included studies had highly variable and inconsistently reported length of follow-up. The short follow-up of several studies precludes evaluation of long-term obstructive symptoms and xerostomia. A final limitation of this study is the lack of validated subjective and objective outcome measures utilized by the included studies. Symptom improvement is perhaps the most important outcome following the combined endoscopic-transfacial technique, but without utilization of a validated outcome measure, symptom improvement is often poorly defined, evaluated, and reported.
In the future, a prospective multi-institutional or large database study evaluating procedural success, clinical outcome, and complication rates, with consideration for stone size and location and duct stenosis/stricture, would advance knowledge and understanding of this technique. Additionally, we advocate for the incorporation of a validated symptom questionnaire specific to salivary symptoms in clinical follow-up of sialolithiasis patients. While long-term follow-up with imaging or sialendoscopy to evaluate stenosis or stone recurrence is helpful, ultimately symptom scores will determine treatment success. While more research is warranted, the data published thus far supports the use of the combined endoscopic-transfacial technique for parotid sialoliths not manageable by sialendoscopy alone.
Conclusion
This analysis supports the combined endoscopic-transfacial technique as an effective treatment for stone removal, symptom improvement, and gland preservation. The complication rates are low, further supporting the use of this technique in clinical practice.
Supplementary Material
Supporting Document S1. Comprehensive search strategy and list of search terms
Supporting Document S2. Quality assessment of included studies
Acknowledgments
We would like to acknowledge Angela Hardi, medical librarian, for her expertise, guidance and assistance with the search process for this study.
This work was supported by the T32DC00022 grant from the National Institutes of Deafness and Other Communications Disorders. This publication was also supported by the Washington University Institute of Clinical and Translational Sciences grants UL1 TR000448 and TL1 TR000449 from the National Center for Advancing Translational Sciences. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Footnotes
Conflicts of interest: none
References
- 1.Ngu RK, Brown JE, Whaites EJ, Drage NA, Ng SK, Makdissi J. Salivary duct strictures: nature and incidence in benign salivary obstruction. Dentomaxillofac Radiol. 2007;36:63–67. doi: 10.1259/dmfr/24118767. [DOI] [PubMed] [Google Scholar]
- 2.Singh PP, Gupta N, Goyal A, Tomar S. Interventional Sialendoscopy for Parotid Ductal Calculi: Our Preliminary Experience. Indian J Otolaryngol Head Neck Surg. 2011 doi: 10.1007/s12070-011-0361-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zenk J, Koch M, Klintworth N, et al. Sialendoscopy in the diagnosis and treatment of sialolithiasis: a study on more than 1000 patients. Otolaryngol Head Neck Surg. 2012;147(5):858–863. doi: 10.1177/0194599812452837. [DOI] [PubMed] [Google Scholar]
- 4.Atienza G, Lopez-Cedrun JL. Management of obstructive salivary disorders by sialendoscopy: a systematic review. Br J Oral Maxillofac Surg. 2015;53(6):507–519. doi: 10.1016/j.bjoms.2015.02.024. [DOI] [PubMed] [Google Scholar]
- 5.Strychowsky JE, Sommer DO, Gupta MK, Cohen N, Nahlieli O. Sialendoscopy for the Management of Obstructive Salivary Gland Disease: A Systematic Review and Meta-Analysis. Arch Otolaryngol Head Neck Surg. 2012;138(6) doi: 10.1001/archoto.2012.856. [DOI] [PubMed] [Google Scholar]
- 6.Nahlieli O, London D, Zagury A, Eliav E. Combined approach to impacted parotid stones. J Oral Maxillofac Surg. 2002;60(12):1418–1423. doi: 10.1053/joms.2002.36097. [DOI] [PubMed] [Google Scholar]
- 7.McGurk M, MacBean AD, Fan KF, Sproat C, Darwish C. Endoscopically assisted operative retrieval of parotid stones. Br J Oral Maxillofac Surg. 2006;44(2):157–160. doi: 10.1016/j.bjoms.2005.03.026. [DOI] [PubMed] [Google Scholar]
- 8.Marchal F. A combined endoscopic and external approach for extraction of large stones with preservation of parotid and submandibular glands. Laryngoscope. 2007;117(2):373–377. doi: 10.1097/mlg.0b013e31802c06e9. [DOI] [PubMed] [Google Scholar]
- 9.Capaccio P, Michele G, Lorenzo P. Sialendoscopy-assisted transfacial surgical removal of parotid stones. J Craniomaxillofac Surg. 2014;42:1964–1969. doi: 10.1016/j.jcms.2014.08.009. [DOI] [PubMed] [Google Scholar]
- 10.Nahlieli O. Complications of sialendoscopy: Personal experience, literature analysis, and suggestions. J Oral Maxillofac Surg. 2014;73(1):75–80. doi: 10.1016/j.joms.2014.07.028. [DOI] [PubMed] [Google Scholar]
- 11.Walvekar RR, Razfar A, Carrau RL, Schaitkin B. Sialendoscopy and associated complications: a preliminary experience. Laryngoscope. 2008;118(5):776–779. doi: 10.1097/MLG.0b013e318165e355. [DOI] [PubMed] [Google Scholar]
- 12.Guo B, Moga C, Harstall C, Schopflocher D. A principal component analysis is conducted for a case series quality appraisal checklist. J Clin Epidemiol. 2016;69:199–207. e192. doi: 10.1016/j.jclinepi.2015.07.010. [DOI] [PubMed] [Google Scholar]
- 13.Konstantinidis I, Chatziavramidis A, Iakovou I, Constantinidis J. Long-term results of combined approach in parotid sialolithiasis. Eur Arch Otorhinolaryngol. 2014 doi: 10.1007/s00405-014-3391-x. [DOI] [PubMed] [Google Scholar]
- 14.Kopec T, Szyfter W, Wierzbicka M. Sialoendoscopy and combined approach for the management of salivary gland stones. Eur Arch Otorhinolaryngol. 2013;270(1):219–223. doi: 10.1007/s00405-012-2145-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mikolajczak S, Bremke M, Beutner D, Luers JC. Combined endoscopic and transcutaneous approach for immobile parotid stones. Acta Otolaryngol. 2015;135(1):85–89. doi: 10.3109/00016489.2014.953204. [DOI] [PubMed] [Google Scholar]
- 16.Numminen J, Sillanpaa S, Virtanen J, Sipila M, Rautiainen M. Retrospective analysis of a combined endoscopic and transcutaneous technique for the management of parotid salivary gland stones. ORL J Otorhinolaryngol Relat Spec. 2014;76(5):282–287. doi: 10.1159/000368719. [DOI] [PubMed] [Google Scholar]
- 17.Carroll WW, Walvekar RR, Gillespie MB. Transfacial ultrasound-guided gland-preserving resection of parotid sialoliths. Otolaryngol Head Neck Surg. 2013;148(2):229–234. doi: 10.1177/0194599812471514. [DOI] [PubMed] [Google Scholar]
- 18.Karavidas K, Nahlieli O, Fritsch M, McGurk M. Minimal surgery for parotid stones: a 7-year endoscopic experience. Int J Oral Maxillofac Surg. 2010;39(1):1–4. doi: 10.1016/j.ijom.2009.06.030. [DOI] [PubMed] [Google Scholar]
- 19.Koch M, Iro H, Zenk J. Combined endoscopic-transcutaneous surgery in parotid gland sialolithiasis and other ductal diseases: reporting medium- to long-term objective and patients’ subjective outcomes. Eur Arch Otorhinolaryngol. 2013;270(6):1933–1940. doi: 10.1007/s00405-012-2286-y. [DOI] [PubMed] [Google Scholar]
- 20.Klein H, Ardekian L. The treatment of large sialoliths by sialendoscopic combined approach. J Oral Maxillofac Surg. 2014;72(4):737–743. doi: 10.1016/j.joms.2013.09.003. [DOI] [PubMed] [Google Scholar]
- 21.Bates D, O’Brien CJ, Tikaram K, Painter DM. Parotid and submandibular sialadenitis treated by salivary gland excision. Aust N Z J Surg. 1998;68(2):120–124. doi: 10.1111/j.1445-2197.1998.tb04720.x. [DOI] [PubMed] [Google Scholar]
- 22.Patel R, Low T, Gao K, O’Brien C. Clinical Outcome After Surgery for 75 Patients with Parotid Sialadenitis. Laryngoscope. 2007;117(4):644–647. doi: 10.1097/mlg.0b013e318030acc0. [DOI] [PubMed] [Google Scholar]
- 23.Jadu FM, Jan AM. A meta-analysis of the efficacy and safety of managing parotid and submandibular sialoliths using sialendoscopy assisted surgery. Saudi Med J. 2014;35(10):1188–1194. [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supporting Document S1. Comprehensive search strategy and list of search terms
Supporting Document S2. Quality assessment of included studies