Abstract
Pediatric cardiomyopathies are rare diseases with an annual incidence of 1.1–1.5 per 100,000. Dilated and hypertrophic cardiomyopathies are the most common; restrictive, noncompaction, and mixed cardiomyopathies occur infrequently; and arrhythmogenic right ventricular cardiomyopathy is rare. Pediatric cardiomyopathies can result from coronary artery abnormalities, tachyarrhythmias, exposure to infection or toxins, or secondary to other underlying disorders. Increasingly, the importance of genetic mutations in the pathogenesis of isolated or syndromic pediatric cardiomyopathies is becoming apparent. Pediatric cardiomyopathies often occur in the absence of co-morbidities such as atherosclerosis, hypertension, renal dysfunction, and diabetes; as a result, they offer insights into the primary pathogenesis of myocardial dysfunction. Large international registries have characterized the epidemiology, etiology, and outcomes of pediatric cardiomyopathies. Although adult and pediatric cardiomyopathies have similar morphologic and clinical manifestations, their outcomes differ significantly. Within two years of presentation, normalization of function occurs in 20% of children with dilated cardiomyopathy, and 40% die or undergo transplantation. Infants with hypertrophic cardiomyopathy have a two-year mortality of 30%, whereas death is rare in older children. Sudden death is rare. Molecular evidence indicates that gene expression differs between adult and pediatric cardiomyopathies, suggesting treatment response may differ as well. Clinical trials to support evidence-based treatments and the development of disease-specific therapies for pediatric cardiomyopathies are in their infancy. This compendium summarizes current knowledge of the genetic and molecular origins, clinical course, and outcomes of the most common phenotypic presentations of pediatric cardiomyopathies, and highlights key areas where additional research is required.
Keywords: pediatrics, cardiomyopathy, genetics, epidemiology, etiology
Subject Terms: Genetics, Cardiomyopathy, Pediatrics
OVERVIEW
Pediatric cardiomyopathies are rare diseases with an annual incidence of 1.1–1.5 per 100,000 in children less than 18 years old.1, 2 Pediatric cardiomyopathies can result from coronary artery abnormalities, tachyarrhythmias, exposure to infection or toxins, or secondary to other underlying disorders. As the accuracy and availability of genetic testing has increased, the importance of genetic mutations in the development of pediatric cardiomyopathies has become apparent. The study of pediatric cardiomyopathies offers important insights into the pathogenesis of myocardial dysfunction in the absence of confounding co-morbidities common in adults, such as atherosclerosis, hypertension, renal dysfunction, and diabetes. The published literature in the field of pediatric cardiomyopathies ranges from large-registry epidemiologic outcome and risk factor analyses, 1–11 to individual case reports. This compendium summarizes current knowledge of the genetic and molecular origins, clinical course, management guidelines, and outcomes of the most common phenotypic presentations of pediatric cardiomyopathies, and highlights key areas where additional research is required.
Classification of Pediatric Cardiomyopathies
Cardiomyopathies are defined as abnormalities of the ventricular myocardium unexplained by abnormal loading conditions or congenital heart disease. The 1995 World Health Organization classifications were based on a combination of morphologic (“dilated” and “hypertrophic”), physiologic (“restrictive”), and etiologic (causes extrinsic to the myocardium, such as infection were excluded) characteristics.12 The identification of genetic mutations has led to controversies regarding the classification criteria. The European Society of Cardiology (ESC)13 and the American College of Cardiology/American Heart Association (ACC/AHA)14 have proposed different definitions of hypertrophic cardiomyopathy (HCM) – with the ESC definition based on morphology and including both genetic mutations and secondary forms, and the ACC/AHA definition limiting HCM to only those found with sarcomeric mutations. The ACC/AHA classification cannot be universally applied to the pediatric cardiomyopathy population because genetic testing is not widely accepted and the yield of testing in pediatric dilated (DCM), restrictive (RCM), and noncompaction cardiomyopathy (NCM) is low.15 In large population registries, pediatric cardiomyopathies have largely been defined by phenotypic characteristics, with etiology, when known, added as a modifier2, 8.
In addition to the standard phenotypes of DCM, HCM, RCM, and NCM (Figures 1–4), a “mixed” category occurs in children, and is an explicit recognition of phenotypic overlap that confounds attempts at a more specific categorization. For instance, HCM can be associated with severe hypokinesis in newborns with mitochondrial disorders, and sarcomeric HCM can transition to DCM. NCM can be seen in isolation or in combination with other phenotypes (NCM/HCM or NCM/DCM). Arrhythmogenic right ventricular cardiomyopathy is rarely diagnosed in the pediatric age group and thus will not be considered in this compedium.16
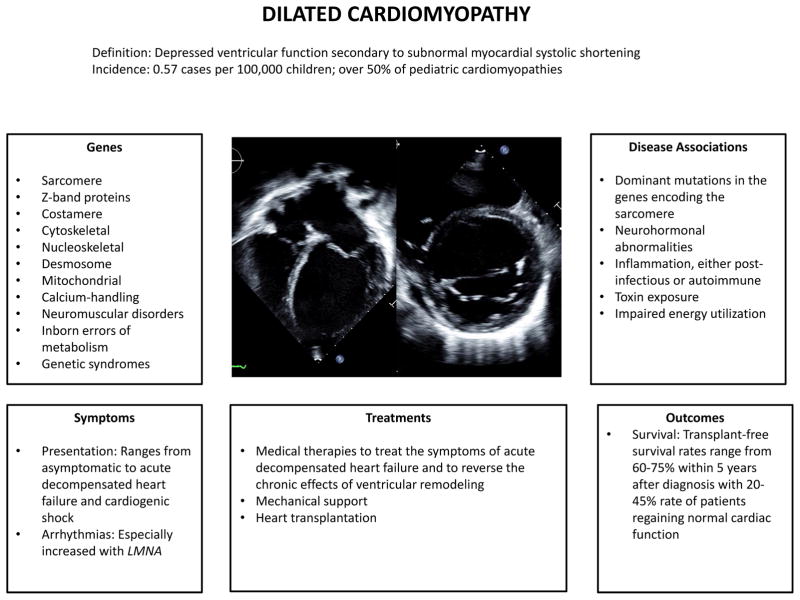
Figure 1. Dilated Cardiomyopathy.
End-diastolic apical four-chamber (left panel) and parasternal short axis end-diastolic (right panel) views of the left ventricle in a patient with severe dilated cardiomyopathy. The left ventricle is dilated and thin-walled. The apical view also demonstrates the decreased mass-to-volume ratio with sphericalization (increased short-to-long axis ratio) of the left ventricle.
Figure 4. Noncompaction Cardiomyopathy.
End-diastolic apical four-chamber view of the left ventricular in a patient with noncompaction, demonstrating multiple finger-like protrusions of myocardial trabeculations into the apex, resulting in deep inter-trabecular interstices.
Epidemiology
The Pediatric Cardiomyopathy Registry (PCMR) was first funded by the National Institutes of Health in 1994, and is the only national registry of its kind in North America. In the PCMR, DCM and HCM are the most common phenotypes with an annual incidence of 0.57 and 0.47 per 100,000 children, respectively.1, 17 Restrictive cardiomyopathy has an incidence of 0.03–0.04 per 100,000 children, and accounts for only 4.5% of pediatric cardiomyopathies1, 18 with about 30% of patients having a mixed RCM/HCM phenotype.19 The incidence of NCM is estimated to be 0.12 per 100,000 in children from birth to10 years old, and up to 0.81 per 100,000 in infants from birth to 12 months old.20 In the PCMR, NCM accounted for 4.8% of pediatric cardiomyopathies, although more than twice as many children were diagnosed in the most recent era suggesting that as the definition of NCM becomes standardized, the true incidence may be higher than previously appreciated.21 NCM may occur as an isolated phenotype (23%), a mixed NCM/DCM phenotype (59%), a mixed NCM/HCM phenotype (11%), or an indeterminate phenotype (8%).21
Genetics
Pediatric cardiomyopathies are genetically heterogeneous with many different causative genes and multiple mutations in each gene. Variants in the same gene can cause different phenotypes (e.g., variants in MYH7 can cause HCM and DCM), and variants in different genes can cause the same cardiomyopathy phenotype (e.g., variants in MYH7 and MYBPC3 both cause HCM).22, 23 Mutations in genes encoding components of the sarcomere or costamere and related binding proteins, Z-band, nuclear membrane, desmosome, mitochondrial, and calcium-handling proteins have all been found in children with cardiomyopathy.24–27 Genetic variants causing cardiomyopathy in children can also have systemic features affecting noncardiac organs.28, 29 The RASopathies, including Noonan syndrome, are the most well-known syndromic causes of pediatric cardiomyopathy.29 Inborn errors of metabolism (CPT2 deficiency in DCM) and storage disorders (Pompe disease with HCM) are associated with childhood-onset cardiomyopathy. Congenital myopathies can present during childhood with both skeletal and heart muscle involvement; Duchenne muscular dystrophy is a classic example. Table 1 summarizes the common genes found in pediatric cardiomyopathies. The NIH Genetic Testing Registry is centralized location for comprehensive genetic test information that https://www.ncbi.nlm.nih.gov/gtr/)
Table 1.
Common Genes Associated with the Pediatric Cardiomyopathies
| Gene Symbol | Inheritance | Associated Cardiac Phenotype(s) | Additional Phenotype(s) | ||||
|---|---|---|---|---|---|---|---|
| HCM | DCM | RCM | NCM | ARVC | |||
| Sarcomere | |||||||
| Thin Filament | |||||||
| ACTC1 | AD | X | X | X | X | Atrial septal defect | |
| TNNC1 | AD | X | X | ||||
| TNNI3 | AD, AR | X | X | X | |||
| TNNT2 | AD | X | X | X | X | ||
| TPM1 | AD | X | X | X | |||
| Thick Filament | |||||||
| MYBPC3 | AD | X | X | X | X | ||
| MYH7 | AD | X | X | X | X | Myopathies | |
| MYL2 | AD | X | |||||
| MYL3 | AD, AR | X | X | ||||
| Z-Disc | |||||||
| ACTN2 | AD | X | X | X | |||
| CSRP3 | AD | X | X | ||||
| LDB3 | AD | X | X | Myofibrillar myopathy | |||
| MYOZ2 | AD | X | |||||
| TCAP | AD, AR | X | X | Limb-girdle muscular dystrophy (AR) | |||
| TTN | AD | X | X | X | Hereditary myopathy with early respiratory failure | ||
| Desmosome | |||||||
| DSC2 | AD, AR | X | Palmoplantar keratoderma and woolly hair (AR) | ||||
| DSG2 | AD | X | X | ||||
| DSP | AD, AR | X | X | Carvajal syndrome (AR) | |||
| JUP | AD, AR | X | Naxos disease (AR) | ||||
| PKP2 | AD | X | |||||
| Cytoskeletal | |||||||
| VCL | AD | X | X | ||||
| Intermediate Filament | |||||||
| DES | AD, AR | X | Limb-girdle muscular dystrophy (AR), myofibrillar myopathy (AD, AR) | ||||
| Nuclear Membrane | |||||||
| EMD | X-linked | X | Emery-Dreifuss muscular dystrophy | ||||
| LMNA | AD, AR | X | X | X | Congenital muscular dystrophy (AD), Emery-Dreifuss muscular dystrophy (AD, AR) | ||
| SYNE1 | AD | X | Emery-Dreifuss muscular dystrophy | ||||
| SYNE2 | AD | X | Emery-Dreifuss muscular dystrophy | ||||
| Plasma Membrane | |||||||
| CAV3 | AD, AR | X | Limb-girdle muscular dystrophy (AD, AR), long QT (AD) | ||||
| SGCD | AD, AR | X | Limb-girdle muscular dystrophy (AR) | ||||
| Other | |||||||
| CRYAB | AD, AR | X | Myofibrillar myopathy (AD, AR) | ||||
| MIB1 | AD | X | |||||
| RMB20 | AD | X | |||||
| Syndromic Cardiomyopathies | |||||||
| BRAF | AD | X | Noonan/Costello/CFC syndrome | ||||
| HRAS | AD | X | Noonan/Costello/CFC syndrome | ||||
| KRAS | AD | X | Noonan/Costello/CFC syndrome | ||||
| PTPN11 | AD | X | Noonan/Costello/CFC syndrome | ||||
| SOS1 | AD | X | Noonan/Costello/CFC syndrome | ||||
| SPRED1 | AD | X | Noonan/Costello/CFC syndrome | ||||
| Metabolic Disorders | |||||||
| CPT2 | AR | X | Carnitine palmitoyltransferase II deficiency | ||||
| GAA | AR | X | Pompe disease (glycogen storage disease Type II) | ||||
| HADHA | AR | X | X | Long-chain 3-hydroxyacyl-CoA dehydrogenase | |||
| LAMP2 | X-linked | X | X | Danon disease (glycogen storage disease Type IIb) | |||
| MT-TL1 | Mitochondrial | X | MELAS (mitochondrial encephalopathy, lactic acidosis, and stroke-like episode) | ||||
| PRKAG2 | AD | X | Cardiac glycogen storage disease, Wolff-Parkinson-White | ||||
| SLC22A5 | AR | X | X | Primary carnitine deficiency | |||
| TAZ | X-linked | X | X | Barth syndrome | |||
| Neuromuscular/Neurodegenerative Disorders | |||||||
| DMD | X-linked | X | Duchenne/Becker muscular dystrophy | ||||
| FRDA1 | AR | X | Friedreich ataxia | ||||
AD, autosomal dominant; AR, autosomal recessive; ARVC arrhythmogenic right ventricular cardiomyopathy; CFC, cardiofaciocutaneous
There are multiple modes of inheritance for cardiomyopathies including autosomal dominant, autosomal recessive, X-linked, and mitochondrial. Isolated, autosomal-dominant cardiomyopathy is the most common genetic form of cardiomyopathy among individuals of all ages. There are shared genetic causes in children and adults, 25,27 especially in families with autosomal-dominant cardiomyopathy.30, 31 Variants can be inherited, or occur de novo. The latter are particularly common in genes within the RAS pathway and among individuals presenting with severe, early-onset cardiomyopathies that are either fatal or require transplantation. Many autosomal dominantly inherited conditions have variable age of onset and/or penetrance both within and between families. Digenic and autosomal recessive inheritance for typical, adult, autosomal-dominant causes of cardiomyopathy have been described in children with early and severe presentations.32, 33 The identification of multiple variants in one or more genes, however, explains only a small fraction of the observed clinical variability among affected individuals. Several studies have investigated the role of non-sarcomeric polymorphisms as potential disease modifiers, yet, additional studies are needed to replicate and further explore potential impact on disease.34, 35 In general, there is limited understanding of genetic, environmental, and other, as of yet undiscovered, modifying factors in pediatric cardiomyopathy.
The increased availability of genetic testing has led to increasing detection of genetic causes in pediatric patients. In PCMR data published in 2003, approximately one third of children with cardiomyopathy had a confirmed etiology. Diagnostic categories included myocarditis (DCM 16%), metabolic (4% DCM, 9% HCM), syndromic (1% DCM, 9%, HCM), neuromuscular (9% DCM, 9% HCM), familial (5% DCM), and idiopathic.7, 8 A more recent single-center study, incorporating clinical genetic evaluation and testing, identified an underlying etiology in approximately 75% of affected children (excluding those with neuromuscular disease); 42% familial, 20.5% metabolic, and 14.5% syndromic with the remainder being idiopathic.27 While genetic testing and evaluation has improved the ability to identify underlying etiology, diagnostic rates in clinical practice remain uncertain, as genetic testing is not routinely and universally incorporated into the clinical care of children with cardiomyopathy.
Published guidelines have recommended approaches for genetic testing and family screening in patients with isolated, autosomal-dominant cardiomyopathy.36, 37 These guidelines do not specifically address many circumstances commonly encountered in children, and both the timing and type of genetic testing in children varies by the clinical context. Phenotype-specific gene panels are the most appropriate baseline test for isolated cardiomyopathy (e.g., HCM gene panel). The size of these panels varies by clinical genetic testing laboratory and phenotype. While the number of genes has increased, the overall test sensitivity has not, suggesting that expanding from phenotype-specific gene panel testing to a larger panel or whole exome sequencing has limited utility in most cases.38 In the context of a complex phenotype, whole exome sequencing may be considered.
Genetic testing should be guided by the medical and family histories, ideally by teams experienced in caring for children with cardiomyopathy (Figure 5). Not surprisingly, individuals with a positive family history of cardiomyopathy have a higher diagnostic yield with testing.27, 39 Currently, clinical gene panel tests detect disease-causing variants in up to 60% of children with HCM but are informative in < 25% of children with DCM, NCM, or RCM.40 If a causal genetic variant is identified, cascade genetic testing can be recommended for at-risk relatives. Genetic screening of family members requires expertise and resources that may not be available in all practice settings. In centers with personnel and resources dedicated to family-based screening, 39% to 66% of at risk relatives completed recommended genetic testing, and 57% complete cardiac screening.41, 42
Figure 5. Genetic testing algorithm for pediatric cardiomyopathy.
The pediatric patient being evaluated may have a clinical diagnosis of cardiomyopathy or may be seen because of a family history of cardiomyopathy. Genetic testing should be initiated in the most clearly affected individual in the family whenever possible. Positive testing is defined as genetic testing for cardiomyopathy that identifies a pathogenic mutation. Likely pathogenic variants should be handled on an individual basis. Within this algorithm, variants of uncertain significance are treated as a negative test result. In clinical practice, co-segregation studies should be performed if possible to improve interpretation. Genetic testing result interpretation is probabilistic and may change over time as new information is identified. Testing results should therefore be reviewed and updated every two to three years. When a pathogenic variant is identified, testing should be offered to affected relatives to confirm co-segregation with disease. Cardiac screening applies to individuals at-risk for developing cardiomyopathy based on their family history and/or genotype. All affected individuals should receive medical management for their specific diagnosis and symptoms.
*CM, cardiomyopathy
Even in families with multiple affected relatives, genetic testing does not always identify a causal variant, suggesting the presence of missed variants, genes not yet to be identified, or complex gene-to-gene and gene-to-environment interactions. Environmental and/or infectious factors either causing or contributing to cardiomyopathy may be present, particularly in children with DCM. Copy number variation analysis been performed for genes known to cause cardiomyopathy and data suggest that deletions and duplications account for only a minority of cases.43 It may be that smaller base pair deletions or intronic sequence variants are missed due to limitations at the technological or bioinformatic level. With increasing access to genomic analysis of pediatric cardiomyopathy cases, including exome/genome sequencing, additional single gene causes are likely to be identified.
Medical Evaluation
In DCM, 75–80% of children present with signs and symptoms of heart failure, while only 20% of infants and 4% of older children with HCM present with overt heart failure.1, 15, 44 Signs and symptoms of biventricular failure are more common in children compared to adults. Infants and young children present with poor feeding, growth failure, tachypnea, and hepatomegaly while older children present with abdominal symptoms due to hepatomegaly and low cardiac output.45 Grading heart failure symptoms in infants and children can be challenging, as the New York Heart Association class is not applicable. Thus, the Ross Heart Failure Class, which defines symptoms based on respiratory distress, feeding intolerance and failure to thrive, has been adopted but has not been validated against outcomes.46 Heart failure with preserved ejection fraction can be found in patients with HCM, RCM, and NCM. These patients often have symptoms related to diastolic dysfunction and poor cardiac output, including dyspnea, orthopnea and growth failure.
In addition to clinically assessing for heart failure, evaluating the child with cardiomyopathy includes searching for an underlying metabolic, congenital, or acquired etiology. This is particularly important in the infant population, where the incidence of metabolic disease is greater and includes potentially reversible conditions, such as primary carnitine deficiency. A thorough medical history, including growth and development, developmental milestone assessment, chronic medical problem review, and a three-generation pedigree should be performed, as well as comprehensive physical examination attending to features suggesting a genetic syndrome. (Table 2) This evaluation can be extensive, and many centers have gone to a staged approach based on the age at presentation and clinical history and with the involvement of a multidisciplinary team, including geneticists and neurologists.39, 47, 48
Table 2.
Clinical Considerations for Evaluation of Etiology
| History | Developmental delay or regression |
| Poor growth or failure to thrive | |
| Feeding difficulties or intolerance | |
| Seizures | |
| Metabolic decompensation with illness | |
|
| |
| Physical | Dysmorphic features |
| Short stature | |
| Vision or hearing loss | |
| Hypotonia or hypertonia | |
| Gait abnormalities or muscle weakness | |
| Congenital anomaly | |
|
| |
| Laboratory | Urine organic acids, serum amino acids, acylcarnitine profile, lactate, pyruvate, creatine phosphokinase, enzyme testing for concern of Pompe |
| Sequence based genetic testing (e.g., cardiomyopathy panel testing, Noonan syndrome testing, mitochondrial panel) | |
| Directed biochemical (e.g., urine glycosaminoglycans, transferrin isoelectric focusing), genetic (e.g., muscular dystrophy panel), or invasive testing (e.g., muscle biopsy) based on initial laboratory results | |
Echocardiography establishes the cardiac phenotype, and is performed serially to aid in prognosis and treatment. In children with DCM, the degree of ventricular dysfunction and dilation are strong predictors of death or transplantation and sudden death.49, 50 In patients with HCM, septal wall thickness has been associated with sudden death, and is also the primary method of assessing the severity and progression of hypertrophy, left ventricular outflow tract obstruction, and of evaluating systolic and diastolic performance.50 Cardiac magnetic resonance imaging (MRI) has been useful to evaluate for inflammatory processes as well as late gadolinium enhancement, which can further define etiology as well as assist in management and risk stratification.51–53 The use of cardiac biomarkers, including B-type natriuretic peptide in DCM, have been helpful in defining children with DCM at high-risk.54–56 However, extrapolation of adult data has been difficult given pediatric patients have different normative ranges and there is more heterogeneity in this population.57
DILATED CARDIOMYOPATHY
Molecular Genetics of DCM
Between 35% and 40% of genetic DCM cases are thought to be caused by sarcomere gene mutations, with mutations in the giant protein titin estimated to be responsible for about 25%.61, 62 Gene mutations can also affect multiple Z-band proteins, which connect the thin filaments and titin, thereby serving as an important nodal point of mechanosignalling.63, 64 Mutations in members of the LINC complex, that links the nucleus to the cytoplasm, have been described in pediatric DCM including the Lamin A and C proteins, emerin, and nesprins-1 and -2.66, 67 Both LMNA and emerin-null fibroblasts have altered expression of mechanosensitive genes in response to mechanical stress.67 While genotype-phenotype correlations are lacking for most cases of DCM, mutations in genes such as LMNA (and DES) are known to be highly associated with conduction system disease (sinoatrial node disease, atrial arrhythmias, atrioventricular heart block, and ventricular tachyarrhythmias). Thus, the presence of these genes is a risk factor for sudden death.26, 68–71 Mutations in dystrophin and the sarcoglycans produce skeletal muscle disease and cardiomyopathy; as such, heart failure in these patients may be further compromised by hypoventilation from respiratory muscle weakness.65
Inflammatory Causes of DCM
Evidence of viral myocarditis is common in children with DCM. From registry data, between 35–48% of children with DCM who undergo endomyocardial biopsy have evidence of myocarditis.72–74 Parvovirus B19, influenza, Epstein-Barr, HIV, coxsackie virus, herpes, and adenovirus have all been identified. 73–77 Murine models of myocarditis (coxsackie or adenoviral) demonstrate evidence of disruption of several important pathways in the innate and activated immune systems.78, 79 The enteroviral protease 2A has been demonstrated to cleave the cytoskeletal protein- dystrophin which results in severe DCM. 80, 81 Genetic mutations in the dystrophin gene have been demonstrated in Duchenne muscular dystrophy and other X-linked forms of DCM, creating a link between the acquired and genetic forms of DCM.82
Toxic Causes of DCM
Pediatric DCM can occur following exposure to toxins, such as anthracycline exposure during chemotherapy. The mechanism of anthracycline-induced injury is incompletely understood, but oxidative stress and reactive oxygen species activation are felt to play a key role in cell damage.83 Radiation exposure and genetic polymorphisms have been associated with a higher frequency of anthracycline toxicity.84, 85 Survivors of childhood cancer are six times more likely than their siblings to develop congestive heart failure.86 The cumulative incidence of heart failure in childhood cancer survivors has been difficult to accurately determine but recent estimates are 4–5% over a 20- to 30-year period.86–88 Risk factors for heart failure include female gender, young age at diagnosis, treatment during the 1980’s, and total anthracycline or radiation dose.86–90 The relative hazard of congestive heart failure with anthracycline treatment was 2.4-fold for doses < 250 mg/m2 and 5.2-fold for doses > 250 mg/m2.87
Neurohormonal Activation in DCM
The role of neurohormonal activation in the pathophysiology of chronic heart failure in adults is well-described. The neurohormonal derangements in pediatric DCM are described in small series and include elevations in circulating norepinephrine and epinephrine, 91 and decreases in plasma renin activity, aldosterone, and sympathetic nervous system activation with carvedilol treatment.92 There is some evidence that the cardiac molecular response to stress in pediatric DCM is distinct from adult DCM. Tissue analysis of explanted hearts found downregulation of beta-2 adrenergic receptors and upregulation of connexin-43 in pediatric DCM, while upregulation of the beta-2 adrenergic receptor and down regulation of connexin-43 was found in adult DCM.93 In addition, phosphatase expression and phosphorylation of phospholamban were unchanged in pediatric DCM, while adult DCM hearts had upregulation in phosphatase expression and decreased phosphorylation of phospholamban.93 In another study, expression of adenylyl cyclase and phosphodiesterase isoforms after treatment with a phosphodiesterase-3 inhibitor also differed between pediatric and adult explanted DCM hearts.94 Treatment of human induced pluripotent stem cell-derived cardiomyocytes and neonatal rat ventricular myocytes with serum from pediatric DCM patients showed pathogenic changes in gene expression independent of the renin-angiotensin-aldosterone and adrenergic systems.95 These data suggest that the response to adult heart failure therapies targeting the neurohormonal system may be different in pediatric DCM, and also suggest the possibility of identifying therapeutic targets specific to pediatric DCM.
Clinical Concepts in DCM
Presentation
The clinical presentation of children with DCM ranges from asymptomatic to acute decompensated heart failure and cardiogenic shock.96 Many children require hospitalization at the time of diagnosis because of advanced heart failure.97, 98 In a large population-based study of admissions for new-onset heart failure in children with cardiomyopathy, 54% received intravenous inotropic support, 41% were placed on mechanical ventilation, 13% were treated with extracorporeal membrane oxygenation, and 11% underwent urgent transplantation.98 Compared to adults, children with cardiomyopathy hospitalized with heart failure had greater morbidity and mortality and utilized advanced heart failure therapies more frequently.99 In patients with DCM as a component of a multisystem disease (e.g., neuromuscular, metabolic, and mitochondrial disorders) the underlying disease is often an important determinant of patient outcome.1, 3, 15
Comorbid conditions are also key contributors to morbidity and mortality in pediatric DCM.100 Hyponatremia occurs in nearly half of children presenting with acute heart failure, and anemia may occur in up to 40% of patients; both factors are associated with death, transplant, and mechanical circulatory support.101, 102 Additional serious comorbidities, such as sepsis, acute renal failure, and respiratory failure, are less common but are strongly associated with mortality.100 It is not clear if comorbidities are a consequence of worse heart failure or causative. The effectiveness of treatment interventions targeting modifiable comorbidities has not been studies and is an area of research need.
Outcomes and Risk Prediction
The outcome after presentation with DCM in children is by no means certain, with some patients requiring urgent mechanical assist support followed by transplantation and others regaining normal function, despite presenting with fulminant heart failure.103, 104 Registries from the U.S., Australia, and Europe report transplant-free survival rates ranging from 60% to 75% within five years after diagnosis, with most events occurring within two years of presentation.4, 10, 15, 105–107 These same registries also report that 20% to 45% of patients regain normal cardiac function during the same time period.4, 10, 103, 105, 106
Risk factor analyses have confirmed several obvious predictors of worse outcome, such as the worse heart failure, ejection fraction, and ventricular dilation, along with other factors, such as older age (greater than six) and higher B-type natriuretic peptide concentrations.49, 54, 104 B-type natriuretic peptide levels at hospital admission may be less important than the level when clinically stable or the change in levels with therapy.55, 56 More specific biomarkers are needed to distinguish patients at highest risk death from those likely to recover. Potential targets for investigation include genetic variants, circulating or imaging markers of inflammation or stress, neurohormonal abnormalities, microRNA, viral genome on endomyocardial biopsy, and markers of extracardiac impairment.51, 73, 74, 94, 101, 102, 108, 109 Identifying biomarkers that are important for pediatric patients is the focus of a large ongoing study by the PCMR and is an area of research need.
Medical Management
The follow-up and management of pediatric DCM is challenging because invasive strategies, such as mechanical support or transplantation, must be weighed against the possibility of full recovery. Although acute and chronic adult heart failure therapies are routinely applied to children with DCM (Table 3), extrapolating the evidence for treatment efficacy from adults to children is fraught with difficulties given the significant differences in age, etiology, comorbidities, and outcomes between the two populations.110 Studies of heart failure medications in pediatric DCM are limited, with few supporting a treatment strategy and some studies suggesting no benefit.107, 111 Drug trials in pediatric cardiomyopathy are challenging because of limitations in power due to small sample size, lack of validated endpoints, and incomplete pharmacokinetic/pharmacodynamic data.110, 112, 113 A current study of sacubitril/valsartan in children is using a novel global rank endpoint as the primary outcome.114 If successful, this may prove to be a useful endpoint the design of future studies. Identifying appropriate surrogate outcomes and developing accurate pharmacokinetic and pharmacodynamic models are the key research areas needed to advance the development of new heart failure agents in pediatric DCM.
Table 3.
Heart Failure Therapies Routinely Used in Children
| Chronic Heart Failure Medication |
|---|
| Loop Diuretics |
| Bumetanide (oral, IV) |
| Furosemide (oral, IV) |
| Thiazide Diuretics |
| Chlorothiazide (oral, IV) |
| Hydrochlorothiazide (HCTZ) (oral) |
| Metolazone (oral) |
| ACE Inhibitors |
| Captopril (oral) |
| Enalapril (oral) |
| ARBs |
| Candesartan (oral) |
| Losartan (oral) |
| Valsartan (oral) |
| Aldosterone Antagonists |
| Spironolactone (oral) |
| Eplerenone (oral) |
| Beta Blockers |
| Carvedilol (oral) |
| Metoprolol (oral) |
| Digoxin (oral, IV) |
ACE, angiotensin converting enzyme; ARBs, angiotensin II receptor blockers; IV, intravenous
Mechanical Support and Transplantation
Contemporary outcomes after heart transplantation in children with DCM are excellent, with a one-year survival of 94%.115 The use of mechanical support to bridge patients to heart transplantation is increasing, due to an increasing number of children being listed and an increasing wait time.116 Neurologic injury and bleeding are common complications of ventricular assist devices in children including a 20% to 30% incidence of stroke, with smaller children at highest risk.117 The indications for implanted mechanical assist devices are not well defined in children, and are an important area of research need in order to reserve this therapy for those at highest risk.
Disease-Specific Therapies in DCM
If an inborn error of metabolism is diagnosed, treatment strategies can be directed toward the underlying metabolic abnormality and may include dietary management and supplementation to decrease the accumulation of toxic metabolic by-products. Specific management is also directed towards avoiding metabolic or energetic crises.25, 120, 121 Supplementation with carnitine in primary carnitine deficiency cures DCM, emphasizing the critical need to diagnose this disease promptly.122
Duchenne muscular dystrophy is one of the rare genetic DCM diseases with a substantial focus on gene-directed therapies, including viral vector delivery of minigenes, exon-skipping approaches, and nonsense suppression therapy.118 CRISPR/Cas9 technology has been used to remove the mutation in the dystrophin gene and thereby modulate protein expression in mouse models.119 These approaches may have broader application to other genetic causes of DCM.
There have been several novel approaches to the treatment of DCM that are relevant to the pediatric population. A small study of pulmonary artery banding in children with DCM reported clinical improvement occurred in eight of ten patients (delisted for transplant or improved left ventricular function).123 The rationale for the improvement includes an increase right ventricular pressure that alters right and left ventricular interaction and decreases mitral regurgitation. Human mesenchymal stem cells have anti-fibrotic, pro-regenerative, and anti-inflammatory effects – all features that argue for a therapeutic effect in pediatric DCM. A small adult trial in nonischemic DCM has demonstrated a favorable safety profile with better quality of life and lower adverse event scores after transendocardial injection of allogenic stem cells.124 Another small trial has demonstrated increased left ventricular ejection fraction with better functional and clinical outcome following intramyocardial stem cell injection.125
HYPERTROPHIC CARDIOMYOPATHY
Molecular Genetics of HCM
Hypertrophic cardiomyopathy was the first cardiac disease to be described at the molecular level when a disease-causing mutation in the β-myosin heavy chain was discovered.126 Since then, more than 1400 mutations in different sarcomeric genes have been identified.127 Mutations, primarily missense, in the MYBPC3 and MYH7 genes are found in about 70% of HCM cases of all ages.127 Overall penetrance of the disease is unpredictable but may be age-dependent with highly variable expression.128 In infants and children with HCM, non-sarcomeric HCM phenotypes have more diverse genetic etiologies and include the RASopathies, metabolic storage disorders, neurodegenerative disorders (Friedreich ataxia) and mitochondrial disorders (Table 1).1, 3, 25, 129
Clinical Concepts in HCM
Presentation
HCM due to inborn errors of metabolism and malformation syndromes generally present in infancy, and are often associated with neurologic and musculoskeletal abnormalities.44 HCM from sarcomeric mutations is commonly diagnosed during adolescence or early adulthood, although onset can occur from fetal life onward. HCM can also be diagnosed in asymptomatic individuals from testing performed for other reasons or at the time of screening in first-degree relatives. Symptoms of heart failure are rare in the older child. Progressive left atrial enlargement can occur as a result of diastolic dysfunction and predisposes the HCM patient to atrial arrhythmias. Cardiac arrest or sudden death may be the presenting event in a previously healthy child. Older children may experience progressive left ventricular dysfunction and dilation with a transition to DCM and chronic heart failure. Recently the HCMNet Study has shown that HCM mutation carriers have abnormal echocardiographic findings despite a negative phenotype (no hypertrophy).130 Pre-clinical identification of HCM patients raises the possibility of treatments aimed at preventing disease progression prior to the development of hypertrophy.
Outcomes and Risk Prediction
Overall survival for pediatric HCM is 97% at five years and 94% at10 years after presentation. The age at death in children with HCM peaks before one year of age and again at eight to 17 years of age.44, 131 Heart failure is the leading cause of death, although sudden death can occur. Risk factors for poor outcomes in pediatric HCM vary by subgroup (pure HCM vs mixed with DCM or RCM) but importantly children with worse prognosis were diagnosed less than one year of age, had mixed phenotypes, low weight, heart failure, lower left ventricular fractional shortening, or higher left ventricular end-diastolic posterior wall thickness or end-diastolic ventricular septal thickness at the time of diagnosis. In a PCMR analysis, the risk of death or heart transplantation was significantly increased when two or more risk factors were present.85 Identifying patients at risk for ventricular arrhythmias is important in risk stratification of adult HCM patients, however, the association of specific arrhythmia patterns with the risk of sudden death has not been described in children.85 In adults, cardiac MRI may be more accurate than echocardiography at measuring wall thickness, and late gadolinium enhancement can delineate the amount of scar tissue which may predict a poor clinical outcome, but this has not been studied in children with HCM.132, 133
Better risk stratification to identify patients at risk for worse outcome is needed in pediatric HCM. In adult HCM patients, a blunted blood pressure response on exercise testing and elevations in cardiac troponins 134 have been identified as risk factors for worse outcome, but these have not been evaluated in children. Biomarkers such as B-type natriuretic peptide or markers of myocardial scarring have not been studied rigorously in children. Population-based data combining imaging, arrhythmia monitoring, biomarker testing, and exercise testing would help refine risk groups and if validated could serve as surrogate endpoints for treatment trials.132, 135
Medical Management
The treatment goals for children with HCM include relieving symptoms and preventing sudden death. General measures include maintaining adequate hydration and avoiding situations and medications that cause marked peripheral vasodilation. Medical therapy for treating symptoms in pediatric HCM has not been rigorously studied; thus treatment strategies are extrapolated from adult studies. Beta blockade is used to treat symptomatic children with HCM and outflow tract obstruction, and few small studies suggest that beta blockade may reduce the risk of sudden unexpected death in asymptomatic children prompting the routine use in many centers.136, 137 If patients cannot tolerate beta blockade, or if beta blockade does not alleviate symptoms, a calcium channel blocker, specifically verapamil, is often added or substituted. Disopyramide may be used in symptom-resistant patients, but children may not tolerate the side effects. Diuretics are used cautiously when reactive pulmonary edema limits mobility. Studies of the efficacy and optimal doses of the various medical therapies used in pediatric HCM are area of great research need.
Patients with HCM are at increased risk of sudden death during exercise, and are therefore advised to stop participating in competitive sports.14 Restricting sports participation during adolescence puts patients at risk for social isolation, depression, suicide, obesity, and loss of post-high school educational opportunities. Restricting sports participation in HCM has not been shown to improve survival, in part because randomized testing of this hypothesis is at best impractical and is considered by many to be unethical. Regular exercise is likely beneficial in HCM, but the level of exercise at which this benefit is gained and the level at which risk is significant is unknown. The LIVE-HCM study (ClinicTrials.gov identifier NCT02549664) is an observational study evaluating the risk and benefits of exercise and sports participation on cardiac events and quality of life in patients with HCM as young as eight years old. The follow-up period is three years but longer follow-up is likely needed to more accurately assess risk.
Surgical myectomy to relieve left ventricular outflow obstruction is considered when symptoms persist despite medications, the left ventricular outflow gradient exceeds 60–70mmHg at rest, or mitral regurgitation increases. In children and young adults, gradient relief is effective and symptom improvement is excellent.138 The benefits of myectomy also include improving mitral regurgitation by decreasing systolic anterior motion of the mitral valve, although there may be an increased risk of aortic or mitral valve injury in children.138
Automatic implantable cardioverter defibrillator (AICD) implantation in adults with HCM has substantially decreased mortality. Children with HCM have much lower incidence of sudden cardiac death as compared to adults, and sudden death in pediatric HCM before adolescence is rare.3, 44, 132 Risk factors for sudden death in adults have not been validated in children, thus there is a pressing need for data to support pediatric HCM-specific guidelines for AICD use in order to maximize the benefit of AICD use and to minimize risk. Given the low event rate, a large, multicenter, prospective observational registry that includes long-term, comprehensive data collection is needed.
Disease-Specific Therapies in HCM
The infantile form of Pompe disease presents in the first few months with severe HCM, failure to thrive, hypotonia, and respiratory failure. Enzyme replacement therapy is most effective when started early and is associated with decreased cardiac hypertrophy. Certain lysosomal storage diseases, specifically mucopolysaccharidosis I, II, IV, and VI, are also currently treated with enzyme replacement therapy or bone marrow transplantation. Results are variable and early treatment seems to be associated with better outcomes.
Pharmacologic interventions in rodent models of HCM have demonstrated prevention or attenuation of hypertrophy prior to full expression of the phenotype.139–141 Screening of first-degree relatives for HCM can identify children in whom pre-emptive therapy may favorably modify disease expression.142 A study of diltiazem was carried out in genotype positive-phenotype-negative children and demonstrated slower progression of echocardiographic markers of disease.143 Another multicenter clinical trial using valsartan in a similar population is currently in progress. (VANISH trial; ClinicTrials.gov identifier NCT01912534).
HCM in patients with RASopathies presents earlier and can be more rapidly progressive than HCM caused by sarcomeric mutations, and heart failure symptoms and left ventricular outflow tract obstruction are common.129 A PCMR study showed that children with Noonan syndrome (compared to HCM from other causes) were far more likely to present prior to the age of six months (51% vs 28%), more likely to present with heart failure (24% vs 9%), and had much higher mortality with 22% surviving less than one year.144 Additional cardiac (pulmonary stenosis or atrial septal defect) or extracardiac manifestations (coagulation defects and lymphatic dysplasia) can complicate management and worsen prognosis. Translational studies in the RASopathies that blocked dysregulation of the RAS signaling pathway have eliminated cardiac hypertrophy in rodents.145
RESTRICTIVE CARDIOMYOPATHY
Molecular Genetics of RCM
The most common genes implicated in RCM are sarcomeric, including the troponin I (TNNI3)148, β-myosin heavy chain (MYH7), alpha cardiac actin (ACTC1), titin (TTN) 149, and myosin light chain genes.150 Several non-sarcomeric gene abnormalities also cause RCM. The desmin gene (DES), which encodes the chief intermediate filament of skeletal and cardiac muscle, has been associated with RCM.151 Desmin mutations are also associated with conduction abnormalities, including high-grade atrioventricular block.151 Mutations affecting calcium homeostasis within the sarcomere have also been described.152 In the end, the phenotype relies on sarcomere functional abnormalities, similar to those in HCM. Cardiomyocyte alterations and their persistent responses at the cellular level also cause changes that are correlated with sudden cardiac death and other cardiac problems.153
Clinical Concepts in RCM
Presentation
The clinical presentation of pediatric RCM varies, ranging from no symptoms to overt heart failure, syncope, or sudden death. Age at diagnosis ranges from early infancy through late adulthood. Nearly a quarter of RCM patients have a family history of cardiomyopathy.19 A third of patients with RCM have a mixed phenotype with characteristics of RCM and HCM.19 Many of clinical manifestations of RCM are the result of elevated filling pressures that cause pulmonary edema, pulmonary hypertension, hepatomegaly, and peripheral edema. Children with RCM may have a history of reactive airway disease or frequent respiratory infections that prompt referral to cardiology after a chest radiograph shows cardiomegaly. Syncope is an ominous presenting manifestation, presumably caused by ischemia, arrhythmia, or thromboembolism, and increases the risk of sudden death. Enlarged atria increase the risk of clot formation and stroke in addition to atrial arrhythmias. Patients are also at risk of sudden cardiac death from ventricular arrhythmia or heart block.156 In the later stages of disease, systolic function may fail. Although cardiac MRI may help distinguish RCM from constrictive pericarditis and infiltrative disease (e.g., amyloidosis, sarcoidosis, and hemochromatosis) in adults, its diagnostic usefulness in children is minimal given that these other diagnoses are rarely seen in the pediatric age group.157 Cardiac catheterization is an excellent method to distinguish RCM from constrictive pericarditis. In addition, it can assess the degree of abnormal left- and right-heart filling pressures and pulmonary hypertension.
Outcomes and Risk Prediction
Children with RCM have notably poor outcomes. In the largest cohort study to date, five-year survival from the diagnosis of RCM was 68%.19 Transplant-free survival in children with pure RCM is worse than in children with a mixed RCM/HCM phenotype, with one- and five-year survivals of 48% and 22% vs 77% and 68%, respectively. Congestive heart failure and lower shortening fraction z-score at presentation in all RCM patients, and higher posterior wall thickness z-score in the mixed RCM/HCM phenotype predicted worse outcomes.19 Arrhythmia monitoring is necessary to identify patients with conduction disturbances or tachyarrhythmias. Atrial or ventricular arrhythmias; ischemic changes, or pulmonary hypertension predict worse outcomes.156 Pulmonary hypertension can develop rapidly in patients with RCM in the absence of significant heart failure.158, 159
Medical Management
No medical therapies have been described to treat diastolic dysfunction in RCM. Diuretics can improve pulmonary and systemic venous congestion but must be balanced with the need to maintain adequate preload. Anticoagulation should be considered to avoid clot formation in the atria and prevent thromboembolic events. Arrhythmias can be treated with anti-arrhythmia medications and/or AICD placement, but the effectiveness of these therapies is not well described.156
Transplantation
Given the limited medical therapies, poor mechanical support options, and the absence of risk factors that consistently predict rapid disease progression or sudden death, early consideration for heart transplantation has been promoted for RCM patients. Overall wait list mortality for children with RCM is low, at 10%.160 Children requiring mechanical support and infants have a significantly higher risk of death while on the waitlist. After transplantation, one-and five-year survival is 89% and 77%, respectively.160 Overall survival ten years after transplantation is similar to that of children receiving transplants for other forms of cardiomyopathy.161 Better methods of risk stratification to identify patients who would benefit from early heart transplantation are needed.
Disease-Specific Therapies in RCM
Although medical therapies that treat diastolic dysfunction are lacking, the genetic characterization of RCM has improved understanding of the pathways involved in the development of RCM, and may offer insights into potential disease-specific therapeutic targets. Mutations in the desmin gene have been found in areas of the gene that are critically relevant for filament assembly and interaction with other cytoskeletal proteins.162 Multiple mutation in the sarcomeric genes have been identified in RCM that potentially disrupt the actin-binding domain of troponin I and areas of the tropomyosin-binding domain through mechanisms that may increase calcium sensitivity of contraction and the cooperatively of thin filament interactions.30, 152 A knock-in murine model of mutant myopalladin that results in RCM demonstrated changes in the mechanosensory proteins without contractile impairment, which may explain why medical therapies that target beta-receptors or the renin–angiotensin–aldosterone system pathway are not effective in RCM.163
NONCOMPACTION CARDIOMYOPATHY
Molecular Genetics of NCM
Noncompaction of the left ventricle is a common finding in Barth Syndrome, an X-linked recessive disorder caused by a mutation in the tafazzin (TAZ) gene on chromosome Xq28. Presentation is commonly mixed in association with DCM.164 In addition, other inborn errors of metabolism including glycogen storage disease type 1b, malonyl coenzyme A decarboxylase deficiency, and cobalamin C deficiency have been reported with NCM. Although specific genetic mutations have not been identified, NCM can occur with any form of congenital heart disease, but is most commonly associated with pulmonary stenosis or septal defects.165 NCM has also been associated with aneuploidies (Turner syndrome or trisomy 21, 18, and 13), copy number variations (22q11 deletion, 1p36 deletion), neuromuscular disease (Duchenne, Becker, limb-girdle, multiminicore) and other genetic syndromes (Soto, Marfan, and the RASopathies).
Clinical Concepts in NCM
Despite its designation as a separate cardiomyopathy by the AHA in 2006,146 the definition of NCM remains controversial. The debate focuses on whether it is a distinct cardiomyopathy or a description of a morphologic feature of other cardiomyopathies,166 as well as whether different etiologies represent unique diagnoses with different characteristics and outcomes. Current evidence suggests that NCM can be a manifestation of a developmental abnormality or secondary to other diseases.
Data support NCM as a manifestation of premature arrest of myocardial development during embryogenesis. Before the coronary arteries develop, the myocardium is trabeculated, which regresses once the coronary arteries form between weeks five and eight of embryogenesis. The premature arrest of this final stage of cardiac morphogenesis leads to incomplete myocardial compaction with persistence of ventricular trabeculations and deep intertrabecular recesses, which may adversely affect subendocardial perfusion.167 Subendocardial ischemia increases fibrous and elastic tissue on the endocardial surface, contributing to the clinical phenotype of NCM.168
Presentation
The clinical phenotype of NCM in children ranges from a benign to a severe course with progressive systolic or diastolic dysfunction, life-threatening arrhythmias, or thromboembolism. In the largest pediatric NCM study, nearly 40% of patients were infants and 25% had a family history of cardiomyopathy. The signs and symptoms at presentation were heart failure (25%), suspected arrhythmia (17%), or murmur (18%).169 Electrocardiograms were abnormal in nearly all patients, with 8% displaying pre-excitation. A third of patients had a documented tachyarrhythmia, including ventricular tachycardia (17%), atrial tachycardia (6%), and re-entrant supraventricular tachycardia (8%). Patients with NCM and Barth syndrome have associated skeletal myopathy, neutropenia, pre-pubertal growth delay, and cardiomyopathy.170, 171
NCM is associated with an undulating phenotype characterized by transient improvements and declines in systolic function. NCM is commonly a component of a mixed phenotype: HCM-DCM (28%), HCM (27%) and DCM (19%), with more than half of patients presenting with systolic dysfunction and the eventual development of systolic dysfunction in an additional subset.169
Outcomes and Risk Prediction
Children with NCM that have normal cardiac dimensions and normal systolic function are at very low risk for an adverse outcome. These findings are in contrast outcomes in adult with NCM in whom heart failure and ventricular arrhythmia are frequent and outcomes are poor.172 Children with a NCM and a mixed or dilated phenotype have a worse prognosis with an incidence of death or transplantation (18% to 25%) that is higher in infants or those with congenital heart disease. Thus, differentiating the NCM subtype has important prognostic implications.21, 169 Systolic dysfunction has been associated with an increased risk of arrhythmia; and arrhythmia independent of systolic dysfunction is a separate risk factor for sudden death. The mechanisms for arrhythmia remain unclear, with studies suggesting that progressive subendocardial ischemia and arrest of fetal myocardial development contribute to arrhythmogenisis.169 The majority of NCM patients that have undergone heart transplantation are pediatric with an average age of three years.173
Medical Management
Approaches to the diagnosis and management of pediatric NCM are variable. The lack of consensus on diagnostic criteria leads to under- and over-diagnosis of the phenotype and limits understanding of the natural history of the disease. Single-center and multicenter data provide a basis for evaluating children with NCM, but often focus on isolated NCM. Given these data, many centers evaluate children annually with non-invasive imaging (echocardiography or cardiac MRI), electrocardiography, and ambulatory Holter monitoring. For older children who can comply, stress testing may also screen for exercise-induced arrhythmias. For those children with concomitant phenotypes, such as NCM/DCM or NCM/HCM, evaluation is typically more frequent, with many centers evaluating patients every six months or more often if symptoms change.
Thromboembolic disease in association with NCM occurs in up to 24% of adults.172 Many centers treat adults with antiplatelet therapy or systemic anticoagulation, especially those with a history of systolic dysfunction. No data have been reported regarding the risk or occurrence of thromboembolic disease in pediatric NCM. Whether the risk profile for stroke is different in children is unclear, and is a needed area of study given the potential impact of under- or over-treatment with anticoagulants on morbidity and mortality. The ability to identify patients at risk of sudden cardiac death remains limited, and there are no specific risk scores for pediatric NCM. The risks and benefits of AICD use must be weighed against the potential risk of sudden arrest. A better understanding of appropriate medical therapies is needed to assess efficacy. Preliminary data suggest that conventional remodeling therapies may be effective in NCM with a concomitant DCM phenotype, but larger studies are needed in the setting of mixed NCM disease.174
Disease-Specific Therapies in NCM
Tafazzin is an acyltransferase responsible for acylation of cardiolipin, the major phospholipid of the mitochondrial membrane. TAZ deficiency impairs mature cardiolipin production and mitochondrial function and has substantial downstream effects on sarcomere assembly and myocardial contraction. TAZ deficiency destabilizes mitochondrial respiratory chain complexes and affects supercomplex assembly. Changes in cardiac proteome have been identified and measured in TAZ knockdown mouse models of human Barth syndrome.175 The ability to generate induced pluripotent stem cell-cardiomyocyte models from individuals with Barth syndrome is an important development that may speed the assessment of potential therapies to correct the metabolic phenotype of the disease.176
FUTURE RESEARCH DIRECTIONS
Several large longitudinal registries have provided invaluable data characterizing the epidemiology and outcomes of pediatric cardiomyopathies over the past two decades. Pediatric cardiomyopathies have a significant burden of disease in the affected population and more precise biomarkers are needed to distinguish patients at risk who would benefit from invasive therapies such as AICD, mechanical assist, or transplantation from those who will remain clinically well or even improve over time. The increasing application of genomic analysis to the pediatric cardiomyopathy population is creating in a wealth of information that requires expanded registry participation in order to further understanding of the pathogenic mechanisms underlying pediatric cardiomyopathies and the genetic, environmental, and other, as of yet undiscovered, modifying factors that impact the severity of disease. Clinical trials evaluating adult heart failure therapies in children are desperately needed to establish their safety and efficacy, as there is evidence that the pathophysiology of heart failure and arrhythmias in the pediatric cardiomyopathy population differs from adults. Disease-specific therapies based the underlying pathophysiology and genetics hold promise for the future.
Supplementary Material
Figure 2. Hypertrophic Cardiomyopathy.
End-diastolic (left panel) and end-systolic (right panel) apical four-chamber views of the left ventricle in a patient with severe hypertrophic cardiomyopathy. Regional left ventricular hypertrophy is most notable in the mid-septum, lateral free wall, and lateral apex. The end-diastolic frame shows extension of the left ventricular cavity to the apex and the end-systolic frame shows systolic apical obliteration.
Figure 3. Restrictive Cardiomyopathy.
End-systolic apical four-chamber view of the left and right ventricles in a patient with restrictive cardiomyopathy demonstrating mildly small right and left ventricular cavities with massive bi-atrial dilation.
Acknowledgments
We thank the Children’s Cardiomyopathy Foundation for its ongoing and consistent support for the work of the PCMR.
SOURCES OF FUNDING
Children’s Cardiomyopathy Foundation
NON-STANDARD ABBREVIATIONS AND ACRONYMS
- ESC
European Society of Cardiology
- ACC
American College of Cardiology
- AHA
American Heart Association
- HCM
Hypertrophic Cardiomyopathy
- DCM
Dilated Cardiomyopathy
- RCM
Restrictive Cardiomyopathy
- NCM
Noncompaction Cardiomyopathy
- PCMR
Pediatric Cardiomyopathy Registry
- MRI
Magnetic Resonance Imaging
- AICD
Automatic Implantable Cardioverter Defibrillator
Footnotes
DISCLOSURES
The authors have reported that they have no relationships to disclose.
References
- 1.Lipshultz SE, Sleeper LA, Towbin JA, Lowe AM, Orav EJ, Cox GF, Lurie PR, McCoy KL, McDonald MA, Messere JE, Colan SD. The Incidence of Pediatric Cardiomyopathy in Two Regions of the United States. N Engl J Med. 2003;348:1647–1655. doi: 10.1056/NEJMoa021715. [DOI] [PubMed] [Google Scholar]
- 2.Nugent AW, Daubeney PEF, Chondros P, Carlin JB, Cheung M, Wilkinson LC, Davis AM, Kahler SG, Chow CW, Wilkinson JL, Weintraub RG. The Epidemiology of Childhood Cardiomyopathy in Australia. N Engl J Med. 2003;348:1639–1646. doi: 10.1056/NEJMoa021737. [DOI] [PubMed] [Google Scholar]
- 3.Nugent AW, Daubeney PE, Chondros P, Carlin JB, Colan SD, Cheung M, Davis AM, Chow CW, Weintraub RG National Australian Childhood Cardiomyopathy S. Clinical features and outcomes of childhood hypertrophic cardiomyopathy: results from a national population-based study. Circulation. 2005;112:1332–8. doi: 10.1161/CIRCULATIONAHA.104.530303. [DOI] [PubMed] [Google Scholar]
- 4.Daubeney PE, Nugent AW, Chondros P, Carlin JB, Colan SD, Cheung M, Davis AM, Chow CW, Weintraub RG National Australian Childhood Cardiomyopathy S. Clinical features and outcomes of childhood dilated cardiomyopathy: results from a national population-based study. Circulation. 2006;114:2671–8. doi: 10.1161/CIRCULATIONAHA.106.635128. [DOI] [PubMed] [Google Scholar]
- 5.Alexander PM, Daubeney PE, Nugent AW, Lee KJ, Turner C, Colan SD, Robertson T, Davis AM, Ramsay J, Justo R, Sholler GF, King I, Weintraub RG National Australian Childhood Cardiomyopathy S. Long-term outcomes of dilated cardiomyopathy diagnosed during childhood: results from a national population-based study of childhood cardiomyopathy. Circulation. 2013;128:2039–46. doi: 10.1161/CIRCULATIONAHA.113.002767. [DOI] [PubMed] [Google Scholar]
- 6.Wilkinson JD, Sleeper LA, Alvarez JA, Bublik N, Lipshultz SE. The Pediatric Cardiomyopathy Registry: 1995–2007. Prog Pediatr Cardiol. 2008;25:31–36. doi: 10.1016/j.ppedcard.2007.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wilkinson JD, Landy DC, Colan SD, Towbin JA, Sleeper LA, Orav EJ, Cox GF, Canter CE, Hsu DT, Webber SA, Lipshultz SE. The pediatric cardiomyopathy registry and heart failure: key results from the first 15 years. Heart Fail Clin. 2010;6:401–13. vii. doi: 10.1016/j.hfc.2010.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Grenier MA, Osganian SK, Cox GF, Towbin JA, Colan SD, Lurie PR, Sleeper LA, Orav EJ, Lipshultz SE. Design and implementation of the North American Pediatric Cardiomyopathy Registry. Am Heart J. 2000;139:S86–95. doi: 10.1067/mhj.2000.103933. [DOI] [PubMed] [Google Scholar]
- 9.Puggia I, Merlo M, Barbati G, Rowland TJ, Stolfo D, Gigli M, Ramani F, Di Lenarda A, Mestroni L, Sinagra G. Natural History of Dilated Cardiomyopathy in Children. J Am Heart Assoc. 2016:5. doi: 10.1161/JAHA.116.003450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.den Boer SL, Lennie van Osch-Gevers M, van Ingen G, du Marchie Sarvaas GJ, van Iperen GG, Tanke RB, Backx AP, Ten Harkel AD, Helbing WA, Delhaas T, Bogers AJ, Rammeloo LA, Dalinghaus M. Management of children with dilated cardiomyopathy in The Netherlands: Implications of a low early transplantation rate. J Heart Lung Transplant. 2015;34:963–9. doi: 10.1016/j.healun.2015.01.980. [DOI] [PubMed] [Google Scholar]
- 11.Arola A, Jokinen E, Ruuskanen O, Saraste M, Pesonen E, Kuusela AL, Tikanoja T, Paavilainen T, Simell O. Epidemiology of idiopathic cardiomyopathies in children and adolescents. A nationwide study in Finland. Am J Epidemiol. 1997;146:385–93. doi: 10.1093/oxfordjournals.aje.a009291. [DOI] [PubMed] [Google Scholar]
- 12.Report of the WHO/ISFC task force on the definition and classification of cardiomyopathies. Br Heart J. 1980;44:672–3. doi: 10.1136/hrt.44.6.672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Elliott PM, Anastasakis A, Borger MA, et al. 2014 ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy: the Task Force for the Diagnosis and Management of Hypertrophic Cardiomyopathy of the European Society of Cardiology (ESC) Eur Heart J. 2014;35:2733–79. doi: 10.1093/eurheartj/ehu284. [DOI] [PubMed] [Google Scholar]
- 14.Gersh BJ, Maron BJ, Bonow RO, et al. 2011 ACCF/AHA guideline for the diagnosis and treatment of hypertrophic cardiomyopathy: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2011;124:e783–831. doi: 10.1161/CIR.0b013e318223e2bd. [DOI] [PubMed] [Google Scholar]
- 15.Towbin JA, Lowe AM, Colan SD, Sleeper LA, Orav EJ, Clunie S, Messere J, Cox GF, Lurie PR, Hsu D, Canter C, Wilkinson JD, Lipshultz SE. Incidence, causes, and outcomes of dilated cardiomyopathy in children. JAMA. 2006;296:1867–76. doi: 10.1001/jama.296.15.1867. [DOI] [PubMed] [Google Scholar]
- 16.Yokogoshi H, Ashida K. Comparison between the metabolic effects of tryptophan and histidine deficiencies in the rat. J Nutr. 1975;105:550–6. doi: 10.1093/jn/105.5.550. [DOI] [PubMed] [Google Scholar]
- 17.Wilkinson JD, Westphal JA, Bansal N, Czachor JD, Razoky H, Lipshultz SE. Lessons learned from the Pediatric Cardiomyopathy Registry (PCMR) Study Group. Cardiol Young. 2015;25(Suppl 2):140–53. doi: 10.1017/S1047951115000943. [DOI] [PubMed] [Google Scholar]
- 18.Nugent AW, Daubeney PE, Chondros P, Carlin JB, Cheung M, Wilkinson LC, Davis AM, Kahler SG, Chow CW, Wilkinson JL, Weintraub RG. The epidemiology of childhood cardiomyopathy in Australia. N Engl J Med. 2003;348:1639–46. doi: 10.1056/NEJMoa021737. [DOI] [PubMed] [Google Scholar]
- 19.Webber SA, Lipshultz SE, Sleeper LA, et al. Outcomes of restrictive cardiomyopathy in childhood and the influence of phenotype: a report from the Pediatric Cardiomyopathy Registry. Circulation. 2012;126:1237–44. doi: 10.1161/CIRCULATIONAHA.112.104638. [DOI] [PubMed] [Google Scholar]
- 20.Botto LD. Orphanet encyclopedia. 2004. Left ventricular noncompaction. [Google Scholar]
- 21.Jefferies JL, Wilkinson JD, Sleeper LA, et al. Cardiomyopathy Phenotypes and Outcomes for Children With Left Ventricular Myocardial Noncompaction: Results From the Pediatric Cardiomyopathy Registry. J Card Fail. 2015;21:877–84. doi: 10.1016/j.cardfail.2015.06.381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Morimoto S. Sarcomeric proteins and inherited cardiomyopathies. Cardiovasc Res. 2008;77:659–66. doi: 10.1093/cvr/cvm084. [DOI] [PubMed] [Google Scholar]
- 23.Watkins H, Ashrafian H, Redwood C. Inherited cardiomyopathies. N Engl J Med. 2011;364:1643–56. doi: 10.1056/NEJMra0902923. [DOI] [PubMed] [Google Scholar]
- 24.Towbin JA. Inherited cardiomyopathies. Circ J. 2014;78:2347–56. doi: 10.1253/circj.cj-14-0893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bates MG, Bourke JP, Giordano C, d’Amati G, Turnbull DM, Taylor RW. Cardiac involvement in mitochondrial DNA disease: clinical spectrum, diagnosis, and management. Eur Heart J. 2012;33:3023–33. doi: 10.1093/eurheartj/ehs275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Taylor MR, Slavov D, Ku L, et al. Prevalence of desmin mutations in dilated cardiomyopathy. Circulation. 2007;115:1244–51. doi: 10.1161/CIRCULATIONAHA.106.646778. [DOI] [PubMed] [Google Scholar]
- 27.Kindel SJ, Miller EM, Gupta R, Cripe LH, Hinton RB, Spicer RL, Towbin JA, Ware SM. Pediatric cardiomyopathy: importance of genetic and metabolic evaluation. J Card Fail. 2012;18:396–403. doi: 10.1016/j.cardfail.2012.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fiorillo C, Astrea G, Savarese M, et al. MYH7-related myopathies: clinical, histopathological and imaging findings in a cohort of Italian patients. Orphanet J Rare Dis. 2016;11:91. doi: 10.1186/s13023-016-0476-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jhang WK, Choi JH, Lee BH, Kim GH, Yoo HW. Cardiac Manifestations and Associations with Gene Mutations in Patients Diagnosed with RASopathies. Pediatr Cardiol. 2016;37:1539–1547. doi: 10.1007/s00246-016-1468-6. [DOI] [PubMed] [Google Scholar]
- 30.Kaski JP, Syrris P, Burch M, Tome-Esteban MT, Fenton M, Christiansen M, Andersen PS, Sebire N, Ashworth M, Deanfield JE, McKenna WJ, Elliott PM. Idiopathic restrictive cardiomyopathy in children is caused by mutations in cardiac sarcomere protein genes. Heart. 2008;94:1478–84. doi: 10.1136/hrt.2007.134684. [DOI] [PubMed] [Google Scholar]
- 31.Morita H, Rehm HL, Menesses A, McDonough B, Roberts AE, Kucherlapati R, Towbin JA, Seidman JG, Seidman CE. Shared genetic causes of cardiac hypertrophy in children and adults. N Engl J Med. 2008;358:1899–908. doi: 10.1056/NEJMoa075463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zahka K, Kalidas K, Simpson MA, Cross H, Keller BB, Galambos C, Gurtz K, Patton MA, Crosby AH. Homozygous mutation of MYBPC3 associated with severe infantile hypertrophic cardiomyopathy at high frequency among the Amish. Heart. 2008;94:1326–30. doi: 10.1136/hrt.2007.127241. [DOI] [PubMed] [Google Scholar]
- 33.Rasmussen TB, Palmfeldt J, Nissen PH, Magnoni R, Dalager S, Jensen UB, Kim WY, Heickendorff L, Molgaard H, Jensen HK, Baandrup UT, Bross P, Mogensen J. Mutated desmoglein-2 proteins are incorporated into desmosomes and exhibit dominant-negative effects in arrhythmogenic right ventricular cardiomyopathy. Hum Mutat. 2013;34:697–705. doi: 10.1002/humu.22289. [DOI] [PubMed] [Google Scholar]
- 34.Ding Y, Long PA, Bos JM, et al. A modifier screen identifies DNAJB6 as a cardiomyopathy susceptibility gene. JCI Insight. 2016:1. doi: 10.1172/jci.insight.88797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rani B, Kumar A, Bahl A, Sharma R, Prasad R, Khullar M. Renin-angiotensin system gene polymorphisms as potential modifiers of hypertrophic and dilated cardiomyopathy phenotypes. Mol Cell Biochem. 2017;427:1–11. doi: 10.1007/s11010-016-2891-y. [DOI] [PubMed] [Google Scholar]
- 36.Ackerman MJ, Priori SG, Willems S, et al. HRS/EHRA expert consensus statement on the state of genetic testing for the channelopathies and cardiomyopathies. Heart Rhythm. 2011;8:1308–39. doi: 10.1016/j.hrthm.2011.05.020. [DOI] [PubMed] [Google Scholar]
- 37.Hershberger RE, Cowan J, Morales A, Siegfried JD. Progress with genetic cardiomyopathies: screening, counseling, and testing in dilated, hypertrophic, and arrhythmogenic right ventricular dysplasia/cardiomyopathy. Circ Heart Fail. 2009;2:253–61. doi: 10.1161/CIRCHEARTFAILURE.108.817346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Alfares AA, Kelly MA, McDermott G, et al. Results of clinical genetic testing of 2,912 probands with hypertrophic cardiomyopathy: expanded panels offer limited additional sensitivity. Genet Med. 2015;17:880–8. doi: 10.1038/gim.2014.205. [DOI] [PubMed] [Google Scholar]
- 39.Cox GF, Sleeper LA, Lowe AM, Towbin JA, Colan SD, Orav EJ, Lurie PR, Messere JE, Wilkinson JD, Lipshultz SE. Factors associated with establishing a causal diagnosis for children with cardiomyopathy. Pediatrics. 2006;118:1519–31. doi: 10.1542/peds.2006-0163. [DOI] [PubMed] [Google Scholar]
- 40.Murphy SL, Anderson JH, Kapplinger JD, Kruisselbrink TM, Gersh BJ, Ommen SR, Ackerman MJ, Bos JM. Evaluation of the Mayo Clinic Phenotype-Based Genotype Predictor Score in Patients with Clinically Diagnosed Hypertrophic Cardiomyopathy. J Cardiovasc Transl Res. 2016;9:153–61. doi: 10.1007/s12265-016-9681-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Christiaans I, Birnie E, Bonsel GJ, Wilde AA, van Langen IM. Uptake of genetic counselling and predictive DNA testing in hypertrophic cardiomyopathy. Eur J Hum Genet. 2008;16:1201–7. doi: 10.1038/ejhg.2008.92. [DOI] [PubMed] [Google Scholar]
- 42.Miller EM, Wang Y, Ware SM. Uptake of cardiac screening and genetic testing among hypertrophic and dilated cardiomyopathy families. J Genet Couns. 2013;22:258–67. doi: 10.1007/s10897-012-9544-4. [DOI] [PubMed] [Google Scholar]
- 43.Lopes LR, Murphy C, Syrris P, Dalageorgou C, McKenna WJ, Elliott PM, Plagnol V. Use of high-throughput targeted exome-sequencing to screen for copy number variation in hypertrophic cardiomyopathy. Eur J Med Genet. 2015;58:611–6. doi: 10.1016/j.ejmg.2015.10.001. [DOI] [PubMed] [Google Scholar]
- 44.Colan SD, Lipshultz SE, Lowe AM, Sleeper LA, Messere J, Cox GF, Lurie PR, Orav EJ, Towbin JA. Epidemiology and cause-specific outcome of hypertrophic cardiomyopathy in children: findings from the Pediatric Cardiomyopathy Registry. Circulation. 2007;115:773–81. doi: 10.1161/CIRCULATIONAHA.106.621185. [DOI] [PubMed] [Google Scholar]
- 45.Hollander SA, Addonizio LJ, Chin C, Lamour JM, Hsu DT, Bernstein D, Rosenthal DN. Abdominal complaints as a common first presentation of heart failure in adolescents with dilated cardiomyopathy. Am J Emerg Med. 2013;31:684–6. doi: 10.1016/j.ajem.2012.12.009. [DOI] [PubMed] [Google Scholar]
- 46.Kantor PF, Lougheed J, Dancea A, et al. Presentation, diagnosis, and medical management of heart failure in children: Canadian Cardiovascular Society guidelines. Can J Cardiol. 2013;29:1535–52. doi: 10.1016/j.cjca.2013.08.008. [DOI] [PubMed] [Google Scholar]
- 47.Ware SM. Evaluation of genetic causes of cardiomyopathy in childhood. Cardiol Young. 2015;25(Suppl 2):43–50. doi: 10.1017/S1047951115000827. [DOI] [PubMed] [Google Scholar]
- 48.Byers SL, Ficicioglu C. Infant with cardiomyopathy: When to suspect inborn errors of metabolism? World J Cardiol. 2014;6:1149–55. doi: 10.4330/wjc.v6.i11.1149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Alvarez JA, Orav EJ, Wilkinson JD, et al. Competing risks for death and cardiac transplantation in children with dilated cardiomyopathy: results from the pediatric cardiomyopathy registry. Circulation. 2011;124:814–23. doi: 10.1161/CIRCULATIONAHA.110.973826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bharucha T, Lee KJ, Daubeney PE, et al. Sudden death in childhood cardiomyopathy: results from a long-term national population-based study. J Am Coll Cardiol. 2015;65:2302–10. doi: 10.1016/j.jacc.2015.03.552. [DOI] [PubMed] [Google Scholar]
- 51.Gopalakrishnan V, Menon PG, Madan S. cMRI-BED: A novel informatics framework for cardiac MRI biomarker extraction and discovery applied to pediatric cardiomyopathy classification. Biomed Eng Online. 2015;14(Suppl 2):S7. doi: 10.1186/1475-925X-14-S2-S7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sachdeva S, Song X, Dham N, Heath DM, DeBiasi RL. Analysis of clinical parameters and cardiac magnetic resonance imaging as predictors of outcome in pediatric myocarditis. Am J Cardiol. 2015;115:499–504. doi: 10.1016/j.amjcard.2014.11.029. [DOI] [PubMed] [Google Scholar]
- 53.Mavrogeni S, Bratis K, Georgakopoulos D, Karanasios E, Kolovou G, Pavlides G, Papadopoulos G. Evaluation of myocarditis in a pediatric population using cardiovascular magnetic resonance and endomyocardial biopsy. Int J Cardiol. 2012;160:192–5. doi: 10.1016/j.ijcard.2011.04.019. [DOI] [PubMed] [Google Scholar]
- 54.Auerbach SR, Richmond ME, Lamour JM, Blume ED, Addonizio LJ, Shaddy RE, Mahony L, Pahl E, Hsu DT. BNP levels predict outcome in pediatric heart failure patients: post hoc analysis of the Pediatric Carvedilol Trial. Circ Heart Fail. 2010;3:606–11. doi: 10.1161/CIRCHEARTFAILURE.109.906875. [DOI] [PubMed] [Google Scholar]
- 55.den Boer SL, Rizopoulos D, du Marchie Sarvaas GJ, Backx AP, Ten Harkel AD, van Iperen GG, Rammeloo LA, Tanke RB, Boersma E, Helbing WA, Dalinghaus M. Usefulness of Serial N-terminal Pro-B-type Natriuretic Peptide Measurements to Predict Cardiac Death in Acute and Chronic Dilated Cardiomyopathy in Children. Am J Cardiol. 2016;118:1723–1729. doi: 10.1016/j.amjcard.2016.08.053. [DOI] [PubMed] [Google Scholar]
- 56.Medar SS, Hsu DT, Ushay HM, Lamour JM, Cohen HW, Killinger JS. Serial Measurement of Amino-Terminal Pro-B-Type Natriuretic Peptide Predicts Adverse Cardiovascular Outcome in Children With Primary Myocardial Dysfunction and Acute Decompensated Heart Failure. Pediatr Crit Care Med. 2015;16:529–34. doi: 10.1097/PCC.0000000000000408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kantor PF, Rusconi P, Lipshultz S, Mital S, Wilkinson JD, Burch M. Current applications and Future Needs for Biomarkers in Pediatric Cardiomyopathy and Heart Failure: Summary From The Second International Conference On Pediatric Cardiomyopathy. Prog Pediatr Cardiol. 2011;32:11–14. doi: 10.1016/j.ppedcard.2011.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Hershberger RE, Hanson EL, Jakobs PM, Keegan H, Coates K, Bousman S, Litt M. A novel lamin A/C mutation in a family with dilated cardiomyopathy, prominent conduction system disease, and need for permanent pacemaker implantation. Am Heart J. 2002;144:1081–6. doi: 10.1067/mhj.2002.126737. [DOI] [PubMed] [Google Scholar]
- 59.Khan IA, Biddle WP, Najeed SA, Abdul-Aziz S, Mehta NJ, Salaria V, Murcek AL, Harris DM. Isolated noncompaction cardiomyopathy presenting with paroxysmal supraventricular tachycardia--case report and literature review. Angiology. 2003;54:243–50. doi: 10.1177/000331970305400216. [DOI] [PubMed] [Google Scholar]
- 60.Towbin JA, Lorts A. Arrhythmias and dilated cardiomyopathy common pathogenetic pathways? J Am Coll Cardiol. 2011;57:2169–71. doi: 10.1016/j.jacc.2010.11.061. [DOI] [PubMed] [Google Scholar]
- 61.Herman DS, Lam L, Taylor MR, et al. Truncations of titin causing dilated cardiomyopathy. N Engl J Med. 2012;366:619–28. doi: 10.1056/NEJMoa1110186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Gautel M. Cytoskeletal protein kinases: titin and its relations in mechanosensing. Pflugers Arch. 2011;462:119–34. doi: 10.1007/s00424-011-0946-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Knoll R, Hoshijima M, Hoffman HM, et al. The cardiac mechanical stretch sensor machinery involves a Z disc complex that is defective in a subset of human dilated cardiomyopathy. Cell. 2002;111:943–55. doi: 10.1016/s0092-8674(02)01226-6. [DOI] [PubMed] [Google Scholar]
- 64.Hassel D, Dahme T, Erdmann J, et al. Nexilin mutations destabilize cardiac Z-disks and lead to dilated cardiomyopathy. Nat Med. 2009;15:1281–8. doi: 10.1038/nm.2037. [DOI] [PubMed] [Google Scholar]
- 65.Yasuda S, Townsend D, Michele DE, Favre EG, Day SM, Metzger JM. Dystrophic heart failure blocked by membrane sealant poloxamer. Nature. 2005;436:1025–9. doi: 10.1038/nature03844. [DOI] [PubMed] [Google Scholar]
- 66.Puckelwartz MJ, Kessler EJ, Kim G, Dewitt MM, Zhang Y, Earley JU, Depreux FF, Holaska J, Mewborn SK, Pytel P, McNally EM. Nesprin-1 mutations in human and murine cardiomyopathy. J Mol Cell Cardiol. 2010;48:600–8. doi: 10.1016/j.yjmcc.2009.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lammerding J, Schulze PC, Takahashi T, Kozlov S, Sullivan T, Kamm RD, Stewart CL, Lee RT. Lamin A/C deficiency causes defective nuclear mechanics and mechanotransduction. J Clin Invest. 2004;113:370–8. doi: 10.1172/JCI19670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Arbustini E, Pilotto A, Repetto A, Grasso M, Negri A, Diegoli M, Campana C, Scelsi L, Baldini E, Gavazzi A, Tavazzi L. Autosomal dominant dilated cardiomyopathy with atrioventricular block: a lamin A/C defect-related disease. J Am Coll Cardiol. 2002;39:981–90. doi: 10.1016/s0735-1097(02)01724-2. [DOI] [PubMed] [Google Scholar]
- 69.Fatkin D, MacRae C, Sasaki T, et al. Missense mutations in the rod domain of the lamin A/C gene as causes of dilated cardiomyopathy and conduction-system disease. N Engl J Med. 1999;341:1715–24. doi: 10.1056/NEJM199912023412302. [DOI] [PubMed] [Google Scholar]
- 70.Taylor MR, Fain PR, Sinagra G, et al. Natural history of dilated cardiomyopathy due to lamin A/C gene mutations. J Am Coll Cardiol. 2003;41:771–80. doi: 10.1016/s0735-1097(02)02954-6. [DOI] [PubMed] [Google Scholar]
- 71.Li D, Tapscoft T, Gonzalez O, Burch PE, Quinones MA, Zoghbi WA, Hill R, Bachinski LL, Mann DL, Roberts R. Desmin mutation responsible for idiopathic dilated cardiomyopathy. Circulation. 1999;100:461–4. doi: 10.1161/01.cir.100.5.461. [DOI] [PubMed] [Google Scholar]
- 72.Nugent AW, Davis AM, Kleinert S, Wilkinson JL, Weintraub RG. Clinical, electrocardiographic, and histologic correlations in children with dilated cardiomyopathy. J Heart Lung Transplant. 2001;20:1152–7. doi: 10.1016/s1053-2498(01)00334-5. [DOI] [PubMed] [Google Scholar]
- 73.Gagliardi MG, Fierabracci A, Pilati M, Chinali M, Bassano C, Saura F, Giovannoni I, Francalanci P. The Impact of Specific Viruses on Clinical Outcome in Children Presenting with Acute Heart Failure. Int J Mol Sci. 2016;17:486. doi: 10.3390/ijms17040486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Bowles NE, Ni J, Kearney DL, Pauschinger M, Schultheiss HP, McCarthy R, Hare J, Bricker JT, Bowles KR, Towbin JA. Detection of viruses in myocardial tissues by polymerase chain reaction. evidence of adenovirus as a common cause of myocarditis in children and adults. J Am Coll Cardiol. 2003;42:466–72. doi: 10.1016/s0735-1097(03)00648-x. [DOI] [PubMed] [Google Scholar]
- 75.Vigneswaran TV, Brown JR, Breuer J, Burch M. Parvovirus B19 myocarditis in children: an observational study. Arch Dis Child. 2016;101:177–80. doi: 10.1136/archdischild-2014-308080. [DOI] [PubMed] [Google Scholar]
- 76.Klugman D, Berger JT, Sable CA, He J, Khandelwal SG, Slonim AD. Pediatric patients hospitalized with myocarditis: a multi-institutional analysis. Pediatr Cardiol. 2010;31:222–8. doi: 10.1007/s00246-009-9589-9. [DOI] [PubMed] [Google Scholar]
- 77.Matsuura H, Ichida F, Saji T, Ogawa S, Waki K, Kaneko M, Tahara M, Soga T, Ono Y, Yasukochi S. Clinical Features of Acute and Fulminant Myocarditis in Children- 2nd Nationwide Survey by Japanese Society of Pediatric Cardiology and Cardiac Surgery. Circ J. 2016;80:2362–2368. doi: 10.1253/circj.CJ-16-0234. [DOI] [PubMed] [Google Scholar]
- 78.Yao HL, Song J, Sun P, Song QQ, Sheng LJ, Chi MM, Han J. Gene expression analysis during recovery process indicates the mechanism for innate immune injury and repair from Coxsackievirus B3-induced myocarditis. Virus Res. 2016;213:314–21. doi: 10.1016/j.virusres.2016.01.003. [DOI] [PubMed] [Google Scholar]
- 79.Stephenson E, Savvatis K, Mohiddin SA, Marelli-Berg FM. T-cell immunity in myocardial inflammation: pathogenic role and therapeutic manipulation. Br J Pharmacol. 2016 doi: 10.1111/bph.13613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Badorff C, Lee GH, Lamphear BJ, Martone ME, Campbell KP, Rhoads RE, Knowlton KU. Enteroviral protease 2A cleaves dystrophin: evidence of cytoskeletal disruption in an acquired cardiomyopathy. Nat Med. 1999;5:320–6. doi: 10.1038/6543. [DOI] [PubMed] [Google Scholar]
- 81.Xiong D, Yajima T, Lim BK, Stenbit A, Dublin A, Dalton ND, Summers-Torres D, Molkentin JD, Duplain H, Wessely R, Chen J, Knowlton KU. Inducible cardiac-restricted expression of enteroviral protease 2A is sufficient to induce dilated cardiomyopathy. Circulation. 2007;115:94–102. doi: 10.1161/CIRCULATIONAHA.106.631093. [DOI] [PubMed] [Google Scholar]
- 82.Maekawa Y, Ouzounian M, Opavsky MA, Liu PP. Connecting the missing link between dilated cardiomyopathy and viral myocarditis: virus, cytoskeleton, and innate immunity. Circulation. 2007;115:5–8. doi: 10.1161/CIRCULATIONAHA.106.670554. [DOI] [PubMed] [Google Scholar]
- 83.Trachtenberg BH, Landy DC, Franco VI, Henkel JM, Pearson EJ, Miller TL, Lipshultz SE. Anthracycline-associated cardiotoxicity in survivors of childhood cancer. Pediatr Cardiol. 2011;32:342–53. doi: 10.1007/s00246-010-9878-3. [DOI] [PubMed] [Google Scholar]
- 84.Blanco JG, Sun CL, Landier W, et al. Anthracycline-related cardiomyopathy after childhood cancer: role of polymorphisms in carbonyl reductase genes--a report from the Children’s Oncology Group. J Clin Oncol. 2012;30:1415–21. doi: 10.1200/JCO.2011.34.8987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Lipshultz SE, Orav EJ, Wilkinson JD, Towbin JA, Messere JE, Lowe AM, Sleeper LA, Cox GF, Hsu DT, Canter CE, Hunter JA, Colan SD Pediatric Cardiomyopathy Registry Study G. Risk stratification at diagnosis for children with hypertrophic cardiomyopathy: an analysis of data from the Pediatric Cardiomyopathy Registry. Lancet. 2013;382:1889–97. doi: 10.1016/S0140-6736(13)61685-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.van Dalen EC, van der Pal HJ, Kok WE, Caron HN, Kremer LC. Clinical heart failure in a cohort of children treated with anthracyclines: a long-term follow-up study. Eur J Cancer. 2006;42:3191–8. doi: 10.1016/j.ejca.2006.08.005. [DOI] [PubMed] [Google Scholar]
- 87.Mulrooney DA, Yeazel MW, Kawashima T, Mertens AC, Mitby P, Stovall M, Donaldson SS, Green DM, Sklar CA, Robison LL, Leisenring WM. Cardiac outcomes in a cohort of adult survivors of childhood and adolescent cancer: retrospective analysis of the Childhood Cancer Survivor Study cohort. BMJ. 2009;339:b4606. doi: 10.1136/bmj.b4606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kremer LC, van der Pal HJ, Offringa M, van Dalen EC, Voute PA. Frequency and risk factors of subclinical cardiotoxicity after anthracycline therapy in children: a systematic review. Ann Oncol. 2002;13:819–29. doi: 10.1093/annonc/mdf167. [DOI] [PubMed] [Google Scholar]
- 89.Lipshultz SE, Lipsitz SR, Sallan SE, Dalton VM, Mone SM, Gelber RD, Colan SD. Chronic progressive cardiac dysfunction years after doxorubicin therapy for childhood acute lymphoblastic leukemia. J Clin Oncol. 2005;23:2629–36. doi: 10.1200/JCO.2005.12.121. [DOI] [PubMed] [Google Scholar]
- 90.Lipshultz SE, Lipsitz SR, Mone SM, Goorin AM, Sallan SE, Sanders SP, Orav EJ, Gelber RD, Colan SD. Female sex and higher drug dose as risk factors for late cardiotoxic effects of doxorubicin therapy for childhood cancer. N Engl J Med. 1995;332:1738–43. doi: 10.1056/NEJM199506293322602. [DOI] [PubMed] [Google Scholar]
- 91.Ohuchi H, Takasugi H, Ohashi H, Okada Y, Yamada O, Ono Y, Yagihara T, Echigo S. Stratification of pediatric heart failure on the basis of neurohormonal and cardiac autonomic nervous activities in patients with congenital heart disease. Circulation. 2003;108:2368–76. doi: 10.1161/01.CIR.0000101681.27911.FA. [DOI] [PubMed] [Google Scholar]
- 92.Giardini A, Formigari R, Bronzetti G, Prandstraller D, Donti A, Bonvicini M, Picchio FM. Modulation of neurohormonal activity after treatment of children in heart failure with carvedilol. Cardiol Young. 2003;13:333–6. [PubMed] [Google Scholar]
- 93.Miyamoto SD, Stauffer BL, Nakano S, Sobus R, Nunley K, Nelson P, Sucharov CC. Beta-adrenergic adaptation in paediatric idiopathic dilated cardiomyopathy. Eur Heart J. 2014;35:33–41. doi: 10.1093/eurheartj/ehs229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Nakano SJ, Sucharov J, van Dusen R, Cecil M, Nunley K, Wickers S, Karimpur-Fard A, Stauffer BL, Miyamoto SD, Sucharov CC. Cardiac Adenylyl Cyclase and Phosphodiesterase Expression Profiles Vary by Age, Disease, and Chronic Phosphodiesterase Inhibitor Treatment. J Card Fail. 2016 doi: 10.1016/j.cardfail.2016.07.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Jiang X, Sucharov J, Stauffer BL, Miyamoto SD, Sucharov CC. Exosomes from Pediatric Dilated Cardiomyopathy Patients Modulate a Pathological Response in Cardiomyocytes. Am J Physiol Heart Circ Physiol. 2017 doi: 10.1152/ajpheart.00673.2016. ajpheart.00673.2016. [DOI] [PMC free article] [PubMed]
- 96.Rossano JW, Shaddy RE. Heart failure in children: etiology and treatment. J Pediatr. 2014;165:228–33. doi: 10.1016/j.jpeds.2014.04.055. [DOI] [PubMed] [Google Scholar]
- 97.Shamszad P, Hall M, Rossano JW, Denfield SW, Knudson JD, Penny DJ, Towbin JA, Cabrera AG. Characteristics and outcomes of heart failure-related intensive care unit admissions in children with cardiomyopathy. J Card Fail. 2013;19:672–7. doi: 10.1016/j.cardfail.2013.08.006. [DOI] [PubMed] [Google Scholar]
- 98.Andrews RE, Fenton MJ, Ridout DA, Burch M British Congenital Cardiac A. New-onset heart failure due to heart muscle disease in childhood: a prospective study in the United kingdom and Ireland. Circulation. 2008;117:79–84. doi: 10.1161/CIRCULATIONAHA.106.671735. [DOI] [PubMed] [Google Scholar]
- 99.Wittlieb-Weber CA, Lin KY, Zaoutis TE, O’Connor MJ, Gerald K, Paridon SM, Shaddy RE, Rossano JW. Pediatric Versus Adult Cardiomyopathy and Heart Failure–Related Hospitalizations: A Value-Based Analysis. J Card Fail. 2015;21:76–82. doi: 10.1016/j.cardfail.2014.10.011. [DOI] [PubMed] [Google Scholar]
- 100.Rossano JW, Kim JJ, Decker JA, Price JF, Zafar F, Graves DE, Morales DLS, Heinle JS, Bozkurt B, Towbin JA, Denfield SW, Dreyer WJ, Jefferies JL. Prevalence, Morbidity, and Mortality of Heart Failure–Related Hospitalizations in Children in the United States: A Population-Based Study. J Card Fail. 2012;18:459–470. doi: 10.1016/j.cardfail.2012.03.001. [DOI] [PubMed] [Google Scholar]
- 101.Goldberg JF, Shah MD, Kantor PF, Rossano JW, Shaddy RE, Chiou K, Hanna J, Hagan JL, Cabrera AG, Jeewa A, Price JF. Prevalence and Severity of Anemia in Children Hospitalized with Acute Heart Failure. Congenit Heart Dis. 2016 doi: 10.1111/chd.12355. [DOI] [PubMed] [Google Scholar]
- 102.Price JF, Kantor PF, Shaddy RE, Rossano JW, Goldberg JF, Hagan J, Humlicek TJ, Cabrera AG, Jeewa A, Denfield SW, Dreyer WJ, Akcan-Arikan A. Incidence, Severity, and Association With Adverse Outcome of Hyponatremia in Children Hospitalized With Heart Failure. Am J Cardiol. 2016;118:1006–10. doi: 10.1016/j.amjcard.2016.07.014. [DOI] [PubMed] [Google Scholar]
- 103.Everitt MD, Sleeper LA, Lu M, et al. Recovery of echocardiographic function in children with idiopathic dilated cardiomyopathy: results from the pediatric cardiomyopathy registry. J Am Coll Cardiol. 2014;63:1405–13. doi: 10.1016/j.jacc.2013.11.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Foerster SR, Canter CE, Cinar A, et al. Ventricular remodeling and survival are more favorable for myocarditis than for idiopathic dilated cardiomyopathy in childhood: an outcomes study from the Pediatric Cardiomyopathy Registry. Circ Heart Fail. 2010;3:689–97. doi: 10.1161/CIRCHEARTFAILURE.109.902833. [DOI] [PubMed] [Google Scholar]
- 105.Andrews RE, Fenton MJ, Dominguez T, Burch M British Congenital Cardiac A. Heart failure from heart muscle disease in childhood: a 5–10 year follow-up study in the UK and Ireland. ESC Heart Fail. 2016;3:107–114. doi: 10.1002/ehf2.12082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Weintraub RG, Everitt MD, Alexander PMA, Wilkinson JD, Lipshultz SE. Do selection criteria for children with dilated cardiomyopathy enrolled in national registries explain differences in outcomes? Prog Pediatr Cardiol. 2014;37:47–48. [Google Scholar]
- 107.Tsirka AE, Trinkaus K, Chen SC, Lipshultz SE, Towbin JA, Colan SD, Exil V, Strauss AW, Canter CE. Improved outcomes of pediatric dilated cardiomyopathy with utilization of heart transplantation. J Am Coll Cardiol. 2004;44:391–7. doi: 10.1016/j.jacc.2004.04.035. [DOI] [PubMed] [Google Scholar]
- 108.den Boer SL, Joosten KF, van den Berg S, Backx AP, Tanke RB, Sarvaas GJ, Helbing WA, Rammeloo LA, ten Harkel AD, van Iperen GG, Dalinghaus M. Prospective Evaluation of Sleep Apnea as Manifestation of Heart Failure in Children. Pediatr Cardiol. 2016;37:248–54. doi: 10.1007/s00246-015-1269-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Miyamoto SD, Karimpour-Fard A, Peterson V, Auerbach SR, Stenmark KR, Stauffer BL, Sucharov CC. Circulating microRNA as a biomarker for recovery in pediatric dilated cardiomyopathy. J Heart Lung Transplant. 2015;34:724–33. doi: 10.1016/j.healun.2015.01.979. [DOI] [PubMed] [Google Scholar]
- 110.Hsu DT, Shaddy R. Advantages, disadvantages and alternatives to using adult heart failure clinical trials to guide pediatric heart failure therapy. Prog Pediatr Cardiol. 2016 in press:x. [Google Scholar]
- 111.Kantor PF, Abraham JR, Dipchand AI, Benson LN, Redington AN. The Impact of Changing Medical Therapy on Transplantation-Free Survival in Pediatric Dilated Cardiomyopathy. J Am Coll Cardiol. 2010;55:1377–1384. doi: 10.1016/j.jacc.2009.11.059. [DOI] [PubMed] [Google Scholar]
- 112.Kechagia IA, Kalantzi L, Dokoumetzidis A. Extrapolation of enalapril efficacy from adults to children using pharmacokinetic/pharmacodynamic modelling. J Pharm Pharmacol. 2015;67:1537–45. doi: 10.1111/jphp.12471. [DOI] [PubMed] [Google Scholar]
- 113.Laer S, Mir TS, Behn F, Eiselt M, Scholz H, Venzke A, Meibohm B, Weil J. Carvedilol therapy in pediatric patients with congestive heart failure: a study investigating clinical and pharmacokinetic parameters. Am Heart J. 2002;143:916–22. doi: 10.1067/mhj.2002.121265. [DOI] [PubMed] [Google Scholar]
- 114.McMurray JJ, Packer M, Desai AS, Gong J, Lefkowitz MP, Rizkala AR, Rouleau JL, Shi VC, Solomon SD, Swedberg K, Zile MR. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371:993–1004. doi: 10.1056/NEJMoa1409077. [DOI] [PubMed] [Google Scholar]
- 115.Singh TP, Almond CS, Piercey G, Gauvreau K. Current outcomes in US children with cardiomyopathy listed for heart transplantation. Circ Heart Fail. 2012;5:594–601. doi: 10.1161/CIRCHEARTFAILURE.112.969980. [DOI] [PubMed] [Google Scholar]
- 116.Davies RR, Haldeman S, McCulloch MA, Pizarro C. Ventricular assist devices as a bridge-to-transplant improve early post-transplant outcomes in children. J Heart Lung Transplant. 2014;33:704–12. doi: 10.1016/j.healun.2014.02.010. [DOI] [PubMed] [Google Scholar]
- 117.Almond CS, Morales DL, Blackstone EH, et al. Berlin Heart EXCOR pediatric ventricular assist device for bridge to heart transplantation in US children. Circulation. 2013;127:1702–11. doi: 10.1161/CIRCULATIONAHA.112.000685. [DOI] [PubMed] [Google Scholar]
- 118.Robinson-Hamm JN, Gersbach CA. Gene therapies that restore dystrophin expression for the treatment of Duchenne muscular dystrophy. Hum Genet. 2016;135:1029–40. doi: 10.1007/s00439-016-1725-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Nelson CE, Hakim CH, Ousterout DG, Thakore PI, Moreb EA, Castellanos Rivera RM, Madhavan S, Pan X, Ran FA, Yan WX, Asokan A, Zhang F, Duan D, Gersbach CA. In vivo genome editing improves muscle function in a mouse model of Duchenne muscular dystrophy. Science. 2016;351:403–7. doi: 10.1126/science.aad5143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Meyers DE, Basha HI, Koenig MK. Mitochondrial cardiomyopathy: pathophysiology, diagnosis, and management. Tex Heart Inst J. 2013;40:385–94. [PMC free article] [PubMed] [Google Scholar]
- 121.Arnold GL, Van Hove J, Freedenberg D, Strauss A, Longo N, Burton B, Garganta C, Ficicioglu C, Cederbaum S, Harding C, Boles RG, Matern D, Chakraborty P, Feigenbaum A. A Delphi clinical practice protocol for the management of very long chain acyl-CoA dehydrogenase deficiency. Mol Genet Metab. 2009;96:85–90. doi: 10.1016/j.ymgme.2008.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Magoulas PL, El-Hattab AW. Systemic primary carnitine deficiency: an overview of clinical manifestations, diagnosis, and management. Orphanet J Rare Dis. 2012;7:68. doi: 10.1186/1750-1172-7-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Schranz D, Rupp S, Muller M, Schmidt D, Bauer A, Valeske K, Michel-Behnke I, Jux C, Apitz C, Thul J, Hsu D, Akinturk H. Pulmonary artery banding in infants and young children with left ventricular dilated cardiomyopathy: a novel therapeutic strategy before heart transplantation. J Heart Lung Transplant. 2013;32:475–81. doi: 10.1016/j.healun.2013.01.988. [DOI] [PubMed] [Google Scholar]
- 124.Hare JM, DiFede DL, Rieger AC, et al. Randomized Comparison of Allogeneic Versus Autologous Mesenchymal Stem Cells for Nonischemic Dilated Cardiomyopathy: POSEIDON-DCM Trial. J Am Coll Cardiol. 2017;69:526–537. doi: 10.1016/j.jacc.2016.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Vrtovec B, Poglajen G, Lezaic L, Sever M, Domanovic D, Cernelc P, Socan A, Schrepfer S, Torre-Amione G, Haddad F, Wu JC. Effects of intracoronary CD34+ stem cell transplantation in nonischemic dilated cardiomyopathy patients: 5-year follow-up. Circ Res. 2013;112:165–73. doi: 10.1161/CIRCRESAHA.112.276519. [DOI] [PubMed] [Google Scholar]
- 126.Geisterfer-Lowrance AA, Kass S, Tanigawa G, Vosberg HP, McKenna W, Seidman CE, Seidman JG. A molecular basis for familial hypertrophic cardiomyopathy: a beta cardiac myosin heavy chain gene missense mutation. Cell. 1990;62:999–1006. doi: 10.1016/0092-8674(90)90274-i. [DOI] [PubMed] [Google Scholar]
- 127.Maron BJ, Maron MS, Semsarian C. Genetics of hypertrophic cardiomyopathy after 20 years: clinical perspectives. J Am Coll Cardiol. 2012;60:705–15. doi: 10.1016/j.jacc.2012.02.068. [DOI] [PubMed] [Google Scholar]
- 128.Wang S, Wang J, Zou Y, Wang J, Wang H, Hui R. Angiotensinogen gene variations and LV outflow obstruction in hypertrophic cardiomyopathy. Herz. 2014;39:258–63. doi: 10.1007/s00059-013-3818-y. [DOI] [PubMed] [Google Scholar]
- 129.Gelb BD, Roberts AE, Tartaglia M. Cardiomyopathies in Noonan syndrome and the other RASopathies. Prog Pediatr Cardiol. 2015;39:13–19. doi: 10.1016/j.ppedcard.2015.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Ho CY, Day SM, Colan SD, et al. The Burden of Early Phenotypes and the Influence of Wall Thickness in Hypertrophic Cardiomyopathy Mutation Carriers: Findings From the HCMNet Study. JAMA Cardiol. 2017 doi: 10.1001/jamacardio.2016.5670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Ostman-Smith I, Rossano JW, Shaddy RE. Hypertrophic cardiomyopathy: do sudden death prevention strategies in children differ between Europe and North America? Curr Opin Cardiol. 2013;28:130–8. doi: 10.1097/HCO.0b013e32835dd020. [DOI] [PubMed] [Google Scholar]
- 132.Windram JD, Dragelescu A, Benson L, Forsey J, Shariat M, Yoo SJ, Mertens L, Wong D, Grosse-Wortmann L. Myocardial Dimensions in Children With Hypertrophic Cardiomyopathy: A Comparison Between Echocardiography and Cardiac Magnetic Resonance Imaging. Can J Cardiol. 2016;32:1507–1512. doi: 10.1016/j.cjca.2016.06.014. [DOI] [PubMed] [Google Scholar]
- 133.Maron BJ, Maron MS. A Discussion of Contemporary Nomenclature, Diagnosis, Imaging, and Management of Patients With Hypertrophic Cardiomyopathy. Am J Cardiol. 2016;118:1897–1907. doi: 10.1016/j.amjcard.2016.08.086. [DOI] [PubMed] [Google Scholar]
- 134.Kubo T, Kitaoka H, Yamanaka S, Hirota T, Baba Y, Hayashi K, Iiyama T, Kumagai N, Tanioka K, Yamasaki N, Matsumura Y, Furuno T, Sugiura T, Doi YL. Significance of high-sensitivity cardiac troponin T in hypertrophic cardiomyopathy. J Am Coll Cardiol. 2013;62:1252–9. doi: 10.1016/j.jacc.2013.03.055. [DOI] [PubMed] [Google Scholar]
- 135.Maron BJ, Maron MS. Contemporary strategies for risk stratification and prevention of sudden death with the implantable defibrillator in hypertrophic cardiomyopathy. Heart Rhythm. 2016;13:1155–65. doi: 10.1016/j.hrthm.2015.12.048. [DOI] [PubMed] [Google Scholar]
- 136.Ostman-Smith I, Wettrell G, Riesenfeld T. A cohort study of childhood hypertrophic cardiomyopathy: improved survival following high-dose beta-adrenoceptor antagonist treatment. J Am Coll Cardiol. 1999;34:1813–22. doi: 10.1016/s0735-1097(99)00421-0. [DOI] [PubMed] [Google Scholar]
- 137.Decker JA, Rossano JW, Smith EO, Cannon B, Clunie SK, Gates C, Jefferies JL, Kim JJ, Price JF, Dreyer WJ, Towbin JA, Denfield SW. Risk factors and mode of death in isolated hypertrophic cardiomyopathy in children. J Am Coll Cardiol. 2009;54:250–4. doi: 10.1016/j.jacc.2009.03.051. [DOI] [PubMed] [Google Scholar]
- 138.Altarabsheh SE, Dearani JA, Burkhart HM, Schaff HV, Deo SV, Eidem BW, Ommen SR, Li Z, Ackerman MJ. Outcome of septal myectomy for obstructive hypertrophic cardiomyopathy in children and young adults. Ann Thorac Surg. 2013;95:663–9. doi: 10.1016/j.athoracsur.2012.08.011. discussion 669. [DOI] [PubMed] [Google Scholar]
- 139.Semsarian C, Ahmad I, Giewat M, Georgakopoulos D, Schmitt JP, McConnell BK, Reiken S, Mende U, Marks AR, Kass DA, Seidman CE, Seidman JG. The L-type calcium channel inhibitor diltiazem prevents cardiomyopathy in a mouse model. J Clin Invest. 2002;109:1013–20. doi: 10.1172/JCI14677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Cannon L, Yu ZY, Marciniec T, Waardenberg AJ, Iismaa SE, Nikolova-Krstevski V, Neist E, Ohanian M, Qiu MR, Rainer S, Harvey RP, Feneley MP, Graham RM, Fatkin D. Irreversible triggers for hypertrophic cardiomyopathy are established in the early postnatal period. J Am Coll Cardiol. 2015;65:560–9. doi: 10.1016/j.jacc.2014.10.069. [DOI] [PubMed] [Google Scholar]
- 141.Teekakirikul P, Eminaga S, Toka O, et al. Cardiac fibrosis in mice with hypertrophic cardiomyopathy is mediated by non-myocyte proliferation and requires Tgf-beta. J Clin Invest. 2010;120:3520–9. doi: 10.1172/JCI42028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Nagueh SF, Bachinski LL, Meyer D, Hill R, Zoghbi WA, Tam JW, Quinones MA, Roberts R, Marian AJ. Tissue Doppler imaging consistently detects myocardial abnormalities in patients with hypertrophic cardiomyopathy and provides a novel means for an early diagnosis before and independently of hypertrophy. Circulation. 2001;104:128–30. doi: 10.1161/01.cir.104.2.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Ho CY, Lakdawala NK, Cirino AL, et al. Diltiazem treatment for pre-clinical hypertrophic cardiomyopathy sarcomere mutation carriers: a pilot randomized trial to modify disease expression. JACC Heart Fail. 2015;3:180–8. doi: 10.1016/j.jchf.2014.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Wilkinson JD, Lowe AM, Salbert BA, Sleeper LA, Colan SD, Cox GF, Towbin JA, Connuck DM, Messere JE, Lipshultz SE. Outcomes in children with Noonan syndrome and hypertrophic cardiomyopathy: a study from the Pediatric Cardiomyopathy Registry. Am Heart J. 2012;164:442–8. doi: 10.1016/j.ahj.2012.04.018. [DOI] [PubMed] [Google Scholar]
- 145.Wu X, Simpson J, Hong JH, Kim KH, Thavarajah NK, Backx PH, Neel BG, Araki T. MEK-ERK pathway modulation ameliorates disease phenotypes in a mouse model of Noonan syndrome associated with the Raf1(L613V) mutation. J Clin Invest. 2011;121:1009–25. doi: 10.1172/JCI44929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Maron BJ, Towbin JA, Thiene G, Antzelevitch C, Corrado D, Arnett D, Moss AJ, Seidman CE, Young JB, Association AH Council on Clinical Cardiology HaFaTC, Groups QoCaORaFGaTBIW and Prevention CoEa. Contemporary definitions and classification of the cardiomyopathies. Circulation. 2006;113:1807–16. doi: 10.1161/CIRCULATIONAHA.106.174287. [DOI] [PubMed] [Google Scholar]
- 147.Gewillig M, Mertens L, Moerman P, Dumoulin M. Idiopathic restrictive cardiomyopathy in childhood. A diastolic disorder characterized by delayed relaxation. Eur Heart J. 1996;17:1413–20. doi: 10.1093/oxfordjournals.eurheartj.a015076. [DOI] [PubMed] [Google Scholar]
- 148.Mogensen J, Kubo T, Duque M, Uribe W, Shaw A, Murphy R, Gimeno JR, Elliott P, McKenna WJ. Idiopathic restrictive cardiomyopathy is part of the clinical expression of cardiac troponin I mutations. J Clin Invest. 2003;111:209–16. doi: 10.1172/JCI16336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Peled Y, Gramlich M, Yoskovitz G, et al. Titin mutation in familial restrictive cardiomyopathy. Int J Cardiol. 2014;171:24–30. doi: 10.1016/j.ijcard.2013.11.037. [DOI] [PubMed] [Google Scholar]
- 150.Olson TM, Karst ML, Whitby FG, Driscoll DJ. Myosin light chain mutation causes autosomal recessive cardiomyopathy with mid-cavitary hypertrophy and restrictive physiology. Circulation. 2002;105:2337–40. doi: 10.1161/01.cir.0000018444.47798.94. [DOI] [PubMed] [Google Scholar]
- 151.Dalakas MC, Park KY, Semino-Mora C, Lee HS, Sivakumar K, Goldfarb LG. Desmin myopathy, a skeletal myopathy with cardiomyopathy caused by mutations in the desmin gene. N Engl J Med. 2000;342:770–80. doi: 10.1056/NEJM200003163421104. [DOI] [PubMed] [Google Scholar]
- 152.Pinto JR, Parvatiyar MS, Jones MA, Liang J, Potter JD. A troponin T mutation that causes infantile restrictive cardiomyopathy increases Ca2+ sensitivity of force development and impairs the inhibitory properties of troponin. J Biol Chem. 2008;283:2156–66. doi: 10.1074/jbc.M707066200. [DOI] [PubMed] [Google Scholar]
- 153.Harvey PA, Leinwand LA. The cell biology of disease: cellular mechanisms of cardiomyopathy. J Cell Biol. 2011;194:355–65. doi: 10.1083/jcb.201101100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Chen SC, Balfour IC, Jureidini S. Clinical spectrum of restrictive cardiomyopathy in children. J Heart Lung Transplant. 2001;20:90–2. doi: 10.1016/s1053-2498(00)00162-5. [DOI] [PubMed] [Google Scholar]
- 155.Denfield SW. Sudden death in children with restrictive cardiomyopathy. Card Electrophysiol Rev. 2002;6:163–7. doi: 10.1023/a:1017980331651. [DOI] [PubMed] [Google Scholar]
- 156.Walsh MA, Grenier MA, Jefferies JL, Towbin JA, Lorts A, Czosek RJ. Conduction abnormalities in pediatric patients with restrictive cardiomyopathy. Circ Heart Fail. 2012;5:267–73. doi: 10.1161/CIRCHEARTFAILURE.111.964395. [DOI] [PubMed] [Google Scholar]
- 157.Gupta A, Singh Gulati G, Seth S, Sharma S. Cardiac MRI in restrictive cardiomyopathy. Clin Radiol. 2012;67:95–105. doi: 10.1016/j.crad.2011.05.020. [DOI] [PubMed] [Google Scholar]
- 158.Weller RJ, Weintraub R, Addonizio LJ, Chrisant MR, Gersony WM, Hsu DT. Outcome of idiopathic restrictive cardiomyopathy in children. Am J Cardiol. 2002;90:501–6. doi: 10.1016/s0002-9149(02)02522-5. [DOI] [PubMed] [Google Scholar]
- 159.Murtuza B, Fenton M, Burch M, Gupta A, Muthialu N, Elliott MJ, Hsia TY, Tsang VT, Kostolny M. Pediatric heart transplantation for congenital and restrictive cardiomyopathy. Ann Thorac Surg. 2013;95:1675–84. doi: 10.1016/j.athoracsur.2013.01.014. [DOI] [PubMed] [Google Scholar]
- 160.Zangwill SD, Naftel D, L’Ecuyer T, Rosenthal D, Robinson B, Kirklin JK, Stendahl G, Dipchand AI. Outcomes of children with restrictive cardiomyopathy listed for heart transplant: a multi-institutional study. J Heart Lung Transplant. 2009;28:1335–40. doi: 10.1016/j.healun.2009.06.028. [DOI] [PubMed] [Google Scholar]
- 161.Dipchand AI, Naftel DC, Feingold B, Spicer R, Yung D, Kaufman B, Kirklin JK, Allain-Rooney T, Hsu D. Outcomes of children with cardiomyopathy listed for transplant: a multi-institutional study. J Heart Lung Transplant. 2009;28:1312–21. doi: 10.1016/j.healun.2009.05.019. [DOI] [PubMed] [Google Scholar]
- 162.Arbustini E, Pasotti M, Pilotto A, Pellegrini C, Grasso M, Previtali S, Repetto A, Bellini O, Azan G, Scaffino M, Campana C, Piccolo G, Vigano M, Tavazzi L. Desmin accumulation restrictive cardiomyopathy and atrioventricular block associated with desmin gene defects. Eur J Heart Fail. 2006;8:477–83. doi: 10.1016/j.ejheart.2005.11.003. [DOI] [PubMed] [Google Scholar]
- 163.Huby AC, Mendsaikhan U, Takagi K, Martherus R, Wansapura J, Gong N, Osinska H, James JF, Kramer K, Saito K, Robbins J, Khuchua Z, Towbin JA, Purevjav E. Disturbance in Z-disk mechanosensitive proteins induced by a persistent mutant myopalladin causes familial restrictive cardiomyopathy. J Am Coll Cardiol. 2014;64:2765–76. doi: 10.1016/j.jacc.2014.09.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Jefferies JL. Barth syndrome. Am J Med Genet C Semin Med Genet. 2013;163C:198–205. doi: 10.1002/ajmg.c.31372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Towbin JA. Left ventricular noncompaction: a new form of heart failure. Heart Fail Clin. 2010;6:453–69. viii. doi: 10.1016/j.hfc.2010.06.005. [DOI] [PubMed] [Google Scholar]
- 166.Arbustini E, Weidemann F, Hall JL. Left Ventricular Noncompaction: A Distinct Cardiomyopathy or a Trait Shared by Different Cardiac Diseases? J Am Coll Cardiol. 2014;64:1840–1850. doi: 10.1016/j.jacc.2014.08.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Burke A, Mont E, Kutys R, Virmani R. Left ventricular noncompaction: a pathological study of 14 cases. Hum Pathol. 2005;36:403–11. doi: 10.1016/j.humpath.2005.02.004. [DOI] [PubMed] [Google Scholar]
- 168.Sen-Chowdhry S, McKenna WJ. Left ventricular noncompaction and cardiomyopathy: cause, contributor, or epiphenomenon? Curr Opin Cardiol. 2008;23:171–5. doi: 10.1097/HCO.0b013e3282fdc939. [DOI] [PubMed] [Google Scholar]
- 169.Brescia ST, Rossano JW, Pignatelli R, Jefferies JL, Price JF, Decker JA, Denfield SW, Dreyer WJ, Smith O, Towbin JA, Kim JJ. Mortality and sudden death in pediatric left ventricular noncompaction in a tertiary referral center. Circulation. 2013;127:2202–8. doi: 10.1161/CIRCULATIONAHA.113.002511. [DOI] [PubMed] [Google Scholar]
- 170.Roberts AE, Nixon C, Steward CG, Gauvreau K, Maisenbacher M, Fletcher M, Geva J, Byrne BJ, Spencer CT. The Barth Syndrome Registry: distinguishing disease characteristics and growth data from a longitudinal study. Am J Med Genet A. 2012;158A:2726–32. doi: 10.1002/ajmg.a.35609. [DOI] [PubMed] [Google Scholar]
- 171.Ting TW, Jamuar SS, Brett MS, Tan ES, Cham BW, Lim JY, Law HY, Tan EC, Choo JT, Lai AH. Left Ventricular Non-compaction: Is It Genetic? Pediatr Cardiol. 2015;36:1565–72. doi: 10.1007/s00246-015-1222-5. [DOI] [PubMed] [Google Scholar]
- 172.Oechslin EN, Attenhofer Jost CH, Rojas JR, Kaufmann PA, Jenni R. Long-term follow-up of 34 adults with isolated left ventricular noncompaction: a distinct cardiomyopathy with poor prognosis. J Am Coll Cardiol. 2000;36:493–500. doi: 10.1016/s0735-1097(00)00755-5. [DOI] [PubMed] [Google Scholar]
- 173.Al-Kindi SG, El-Amm C, Ginwalla M, Hoit BD, Park SJ, Oliveira GH. Heart transplant outcomes in patients with left ventricular non-compaction cardiomyopathy. J Heart Lung Transplant. 2015;34:761–5. doi: 10.1016/j.healun.2014.11.005. [DOI] [PubMed] [Google Scholar]
- 174.Parent JJ, Towbin JA, Jefferies JL. Medical Therapy Leads to Favorable Remodeling in Left Ventricular Non-compaction Cardiomyopathy: Dilated Phenotype. Pediatr Cardiol. 2016;37:674–7. doi: 10.1007/s00246-015-1330-2. [DOI] [PubMed] [Google Scholar]
- 175.Huang Y, Powers C, Madala SK, Greis KD, Haffey WD, Towbin JA, Purevjav E, Javadov S, Strauss AW, Khuchua Z. Cardiac metabolic pathways affected in the mouse model of barth syndrome. PLoS One. 2015;10:e0128561. doi: 10.1371/journal.pone.0128561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Wang G, McCain ML, Yang L, et al. Modeling the mitochondrial cardiomyopathy of Barth syndrome with induced pluripotent stem cell and heart-on-chip technologies. Nat Med. 2014;20:616–23. doi: 10.1038/nm.3545. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.