Abstract
Aim: To investigate the relationships of polymorphisms in genes whose protein products are related in the metabolic pathway of folic acid, particularly MTRR A66G, RFC1 G80A, and MTHFR C677T and A1298C, and disease activity in Mexican patients with rheumatoid arthritis (RA) treated with methotrexate (MTX).
Materials and Methods: Sixty-eight patients with RA were included in the study who were being treated with MTX, either with or without other drugs. In addition to general data, disease activity was measured by the disease activity score 28 (DAS28). Single nucleotide polymorphisms (SNPs) genotyping was performed by allelic discrimination using real-time polymerase chain reaction.
Results: Differences in genotype (homozygotic or heterozygotic for each allele), allele distributions, and phenotype were not statistically different between the RA group and control populations. We did not find any association between the studied polymorphisms and disease activity nor with the intragroup variables (e.g., clinical activity, body mass index, and single- or combined-drug treatment) or between genetic markers; we also did not find any association within the RA group or between the RA group and control populations.
Conclusion: Additional studies of more polymorphisms related to this or other metabolic pathways are required to determine the influence of genetics on disease activity in RA.
Keywords: : MTRR, RFC1, MTHFR, rheumatoid arthritis, DAS28, methotrexate
Introduction
Rheumatoid arthritis (RA) is a chronic and progressive inflammatory disease that is characterized by cell proliferation and inflammation of the joint synovial membranes (McInnes and Schett, 2011). RA is an autoimmune disease and, thus, its etiology is multifactorial with genetic and environmental components, including diet (Oliver and Silman, 2009).
Disease-modifying anti-rheumatic drugs (DMARDs) are used to decrease inflammation and pain, prevent joint damage, and preserve patient functional capacity. The DMARD methotrexate (MTX) is an antagonist of the essential nutrient folic acid, and it is the most commonly used drug to treat RA, either alone or in combination with other drugs (Calabrese et al., 2001). Although the mechanism of action of MTX in patients with autoimmune diseases is not well understood, it seems to have both antiproliferative and anti-inflammatory effects (Cutolo et al., 2001).
Differences in one or more etiological factors may predispose patients to RA onset or varying degrees of disease severity (Oliver and Silman, 2009; McInnes and Schett, 2011). Consequently, RA onset, prognosis, and response to MTX could be affected by environmental factors or genetic variations in folic acid metabolism (Inoue and Yuasa, 2014).
Previous studies have investigated associations between RA clinical activity and treatment response and the deoxyribonucleic acid (DNA) variants of genes associated with folic acid metabolism such as MTRR, RFC1, and MTHFR (Berkun et al., 2004; Hughes et al., 2006; Wessels et al., 2006; Rubini et al., 2008; Inanir et al., 2013; Salazar et al., 2014; Saad et al., 2015a, 2015b, 2016; Muralidharan et al., 2016; Remuzgo-Martínez et al., 2016). MTRR gene is located on chromosome 5p15.3 and encodes for the enzyme methionine synthase reductase, involved in the reductive regeneration of cob(I)alamin (vitamin B12) cofactor required for the maintenance of methionine synthase in a functional state (Jacques, 2003). MTRR A66G polymorphism (rs1801394) has been identified with a global minor allele frequency (MAF) G = 36% (https://www.ncbi.nlm.nih.gov/snp).
In the Caucasian population, GG genotype has been associated with an increase in plasma homocysteine (Hcy) levels, having a greater effect than the AG genotype (Gaughan et al., 2001).
RFC1 gene is located on chromosome 21q22.3 and encodes for the reduced folate transporter, which plays an important role in folate metabolism and also works as a transporter of the MTX into the cell (Matherly et al., 2007). RFC1 G80A polymorphism (rs1051266) has an overall MAF of A = 49% (https://www.ncbi.nlm.nih.gov/snp) and this genetic variant might cause an alteration in the folate transporter, affecting the availability of folate (Dervieux et al., 2004).
MTHFR gene encodes the methylene-tetrahydrofolate reductase enzyme and is located on chromosome 1p36.3. This enzyme catalyzes the conversion of 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate, a co-substrate for Hcy remethylation to methionine (Goyette et al., 1998). MTHFR C677T polymorphism (rs1801133) has a global MAF of T = 24% (https://www.ncbi.nlm.nih.gov/snp), which causes a thermolabile variant of the protein, altering the enzymatic function (Van Der Put et al., 1998).
In homozygous individuals, this variant is correlated with a decrease in enzyme activity (35%), elevated Hcy, low levels of folic acid, and reduced formation of 5-methyltetrahydrofolate, a predominant form of folate (Frosst et al., 1995). Otherwise, a global MAF of C = 25% (https://www.ncbi.nlm.nih.gov/snp) has been reported in the MTHFR A1298C polymorphism (rs1801131) and causes loss of 40% enzyme activity in individuals homozygous for the mutant allele and only in combination with the C677T polymorphism causes hyperhomocysteinemia (Van Der Put et al., 1998).
In this study, we investigated potential associations between RA severity and the single nucleotide polymorphisms (SNPs) MTRR A66G, RFC1 G80A and MTHFR C677T and A1298C.
Materials and Methods
Study population
We analyzed data for 68 Mexican Mestizo patients (67 women and 1 man) with RA diagnosed according to the American College of Rheumatology criteria (ACR, 1987) (Arnett et al., 1988). Patients were seen in the rheumatology services clinic at Regional Hospital 110, Instituto Mexicano del Seguro Social (IMSS) or at the Hospital Civil de Guadalajara “Fray Antonio Alcalde.” and they signed a written informed before the sampling of peripheral blood when they accepted their voluntary participation in the study. All of the patients had been previously diagnosed with RA for at least 1 year and had been receiving MTX for at least 3 months. DAS criteria were used to evaluate their RA activity (low activity score, <3.2; moderate, 3.2–5.1; and high, >5.1) (Prevoo et al., 1995; Fransen et al., 2003; Makinen et al., 2005).
Control population
Each polymorphism had a different control population, for MTRR A66G n = 50 (Shi et al., 2003), RFC1 G80A n = 121 (Rodarte, unpublished data), MTHFR C677T n = 82 (González-Mercado et al., 2014), and MTHFR A1298C n = 94 (González-Mercado et al., 2014).
DNA analysis
We extracted DNA from blood samples in accordance with the methods of Miller and Gustincich (Miller et al., 1989; Gustincich et al., 1991). Polymorphisms MTRR A66G, RFC1 G80A and MTHFR C677T, and A1298C were typed in an allelic discrimination assay by using TaqMan 5′ exonuclease probes (Applied Biosystems, Foster City, CA) with the ABI 7300 real-time PCR system (Applied Biosystems).
Statistical analysis
Genotype and allele frequencies were obtained by direct counting. Data analysis included comparisons of allele frequencies and genotypes between the RA patient group and control normal Mexican population distributions (Shi et al., 2003; González-Mercado et al., 2014; Rodarte, unpublished data). All control genotypes were in agreement with Hardy–Weinberg equilibrium (HWE). We performed distribution comparisons by the exact, chi square, and likelihood ratio tests by using SPSS statistical package, v. 22 (IBM®). Quantitative variables were subjected to mean comparisons by Student's t-test or one-way analysis of variance.
Results
The general characteristics of the study participants were as follows: The mean age was 53.7 years with a SD ± 10.7 (range 72–76). Most previous studies, including some studies in Mexican populations, have estimated that the female-to-male ratio of patients with RA is ∼3:1 (Rodríguez-Acosta et al., 2001; Spindler et al., 2002). In our study, we identified 67 women and only 1 man who had been diagnosed with RA.
The disease activity score 28 (DAS28) values found in the patients in this study had a mean of 4.7 ± 1.4 SD (range 2.3–7.8). According to the DAS28 (Fransen et al., 2003), disease activity levels were as follows: low, 14 patients; moderate, 28 patients; and high, 27 patients. Forty-one patients were receiving MTX monotherapy, 26 patients were receiving MTX plus an additional DMARD, and 2 patients were receiving MTX plus two additional DMARDs. DMARDs other than MTX were leflunomide (n = 15), sulfasalazine (n = 8), chloroquine (n = 4), and penicillamine (n = 3). Since MTX monotherapy is widely recommended in most RA patients as the first line of treatment (Rodríguez-Valverde et al., 2004; Van der Heijde et al., 2005; GUIPCAR, 2011), we compared the DAS28 between patients undergoing mono- and combined therapy. The average DAS28 was 4.64 for monotherapy and 4.76 for combined therapy (p = 0.74).
Polymorphism analysis
Polymorphism distributions for the RA patient group and for the control populations are presented in Table 1. The genetic frequencies of MTRR A66G, RFC1 G80A, and MTHFR C677T and A1298C SNPs have been analyzed in previous studies of several Mexican populations; the genotypes were in agreement with HWE (Shi et al., 2003; González-Mercado et al., 2014; Rodarte, unpublished data).
Table 1.
Genotypes and Allele Frequencies for MTRR A66G, RFC1 G80A, and MTHFR C677T and A1298C Single Nucleotide Polymorphisms in Rheumatoid Arthritis Patients and Control Mexican Populations
| Genotype, counts (%) | Allele, counts (%)a | |||||||
|---|---|---|---|---|---|---|---|---|
| Polymorphism | Group | 1/1 | 1/2 | 2/2 | p | 1 | 2 | p |
| MTRR A66G | RA | 38 (56) | 25 (37) | 5 (7) | 0.67 | 101 (74) | 35 (26) | 0.40 |
| Control (Shi et al., 2003) (n = 50) | 32 (64) | 15 (30) | 3 (6) | 79 (79) | 21 (21) | |||
| RFC1 G80A | RA | 19 (28) | 37 (54) | 12 (18) | 0.99 | 75 (55) | 61 (45) | 0.97 |
| Control (Rodarte, unpublished results) (n = 121) | 34 (28) | 65 (54) | 22 (18) | 133 (55) | 109 (45) | |||
| MTHFR C677T | RA | 23 (34) | 32 (47) | 13 (19) | 0.75 | 78 (57) | 58 (43) | 0.52 |
| Control (González-Mercado et al., 2014) (n = 82) | 23 (28) | 42 (51) | 17 (21) | 88 (53) | 76 (47) | |||
| MTHFR A1298C | RA | 43 (63) | 21 (31) | 4 (6) | 0.70 | 107 (79) | 29 (21) | 0.38 |
| Control (González-Mercado et al., 2014) (n = 94) | 54 (57) | 32 (34) | 8 (9) | 140 (74) | 48 (26) | |||
Allele 1: A in MTRR 66, G in RFC-1 80, C in MTHFR 677, and A in MTHFR 1298. Allele 2: G in MTRR 66, A in RFC-1 80, T in MTHFR 677, and C in MTHFR 1298. Differences in genotype, phenotype (homozygous or heterozygous for each allele), and allele distributions between the RA group and control populations were not statistically significant.
RA, rheumatoid arthritis; SNPs, single nucleotide polymorphisms.
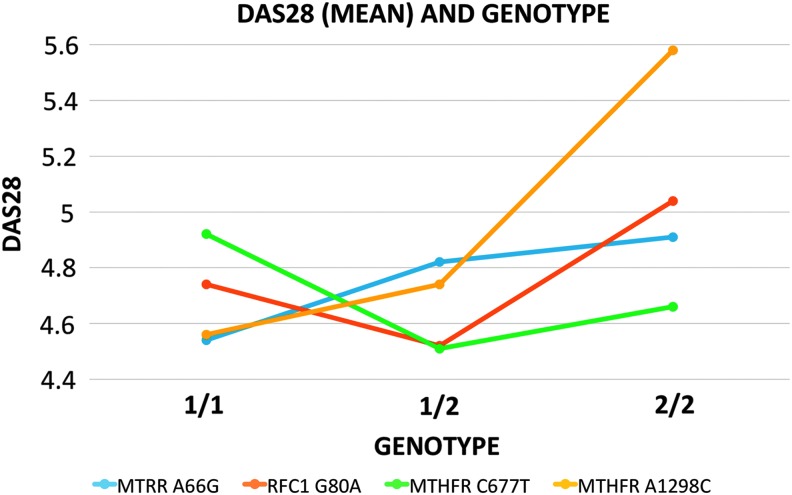
When comparing the allele and genotype frequencies of the four polymorphisms included in this study between patients with RA and various Mexican healthy populations, we found no statistically significant differences between the groups (Table 1). DAS28 values were compared with the genotypes of the polymorphisms studied and there was a trend toward increased activity in the genotypes 2/2 of the polymorphisms MTRR A66G, RFC1 G80A, and MTHFR A1298C; however, there were no significant differences within the RA patient group with respect to clinical activity (Fig. 1). There were also no significant differences with body mass index (BMI), and monotherapy versus combined therapy.
FIG. 1.
Comparison between DAS28 mean values and the genotypes of each polymorphism in patients with RA treated with MTX. A slight upward trend in disease activity was observed in genotypes 2/2 of polymorphisms (MTRR A66G p = 0.73, RFC1 G80A p = 0.55, MTHFR A1298C p = 0.48) with the exception of MTHFR C677T (p = 0.58); however, it was not statistically significant. DAS28, disease activity score 28; RA, rheumatoid arthritis.
Discussion
General characteristics
Although disease onset can occur at any age (Rodríguez-Acosta et al., 2001; Firestein, 2005), the Diagnóstico y Tratamiento de Artritis Reumatoide del Adulto, (Diagnosis and Treatment of Adult RA) reports that the mean age at diagnosis is 40 ± 10 SD years in Mexico (Barrera-Cruz et al., 2010). In this series, the mean age at disease onset was 44.7 ± 12.0 SD years, which is similar to that reported in other populations.
In our patients, the mean BMI was 26.9 ± 4.8 SD (range 15–41), which is above the threshold for overweight. Other studies have reported similar findings with respect to increased BMI in Mexican patients with RA (Puente-Torres et al., 2009). However, the proportion of overweight patients that we observed is comparable to that of the general Mexican population (INEGI, 2016).
Polymorphism analysis
Studies of the genotype and allele frequencies of the polymorphisms that we analyzed have revealed that they vary widely worldwide (Dávalos et al., 2001; Boughrara et al., 2015; Saad et al., 2015a; Li et al., 2016; Remuzgo-Martínez et al., 2016). Several studies have investigated the associations of these polymorphisms with autoimmune diseases, including systemic lupus erythematosus (Summers et al., 2008), multiple sclerosis (Naghibalhossaini et al., 2015), and thyroiditis (Arakawa et al., 2012). Previous studies of RA patients have focused on disease evolution (Brambila-Tapia et al., 2012; Remuzgo-Martínez et al., 2016) and severity or the therapeutic response to MTX. Associations of these polymorphisms with MTX treatment response seem to be consistently significant, but studies of their association with disease onset and clinical severity have yielded conflicting results (Table 2).
Table 2.
Associations Between Polymorphisms and Rheumatoid Arthritis Disease Course and Clinical Response to Methotrexate Treatment in Previous Studies
| Polymorphism | |||||
|---|---|---|---|---|---|
| Disease | Population | n | Allele frequency | Association | References |
| RA and MTX response | Spain | 61 RA patients responders | MTHFR C677T | N.A. | Salazar et al. (2014) |
| RA responders | |||||
| 16 RA patients no responders | C 0.68 | ||||
| T 0.32 | |||||
| RA no responders | |||||
| C 0.75 | |||||
| T 0.25 | |||||
| MTHFR A1298C | N.A. | ||||
| RA responders | |||||
| A 0.67 | |||||
| C 0.33 | |||||
| RA no responders | |||||
| A 0.56 | |||||
| T 0.46 | |||||
| RA and MTX response | Netherlands | 205 RA patients | MTRR A66G | N.A. | Wessels et al. (2006) |
| Not available | |||||
| RA and MTX response | India | 327 RA patients | RFC1 G80A | N.A. but polymorphism confers protection for RA response | Muralidharan et al. (2016) |
| 322 controls | RA patients | ||||
| G 0.57 | |||||
| A 0.43 | |||||
| Controls | |||||
| G 0.57 | |||||
| A 0.43 | |||||
| RA and MTX toxicity | Mexico | 57 RA patients without toxicity | MTHFR C677T | N.A. | Mena et al. (2011) |
| 13 RA patients with toxicity | RA without toxicity | ||||
| C 0.55 | |||||
| T 0.45 | |||||
| RA with toxicity | |||||
| C 0.58 | |||||
| T 0.42 | |||||
| MTHFR A1298C | Associated with elevation of transaminases | ||||
| RA without toxicity | |||||
| A 0.79 | |||||
| C 0.21 | |||||
| RA with toxicity | |||||
| A 0.58 | |||||
| C 0.42 | |||||
| RA and MTX toxicity | Tunisia | 141 RA | MTRR A66G | N.A. | Chaabane et al. (2016) |
| MTX tolerant/MTX intolerant | MTX tolerant | ||||
| A 0.51 | |||||
| G 0.49 | |||||
| MTX intolerant | |||||
| A 0.52 | |||||
| G 0.48 | |||||
| MTHFR C677T | Associated with MTX toxicity | ||||
| MTX tolerant | |||||
| C 0.75 | |||||
| T 0.25 | |||||
| MTX intolerant | |||||
| C 0.65 | |||||
| T 0.35 | |||||
| MTHFR A1298C | N.A. | ||||
| MTX tolerant | |||||
| A 0.70 | |||||
| C 0.30 | |||||
| MTX intolerant | |||||
| A 0.74 | |||||
| C 0.26 | |||||
| RA and serum MTX levels | Japan | 100 RA patients | RFC1 G80A | N.A. | Fukino et al. (2007) |
| G 0.37 | |||||
| A 0.63 | |||||
| MTHFR C677T | N.A. | ||||
| C 0.57 | |||||
| T 0.43 | |||||
| MTHFR A1298C | N.A. | ||||
| A 0.80 | |||||
| C 0.20 | |||||
| RA and MTX | Japan | 170 RA patients | RFC1 G80A | G allele may be associated with lower intracellular MTX uptake and poor efficacy | Hayashi et al. (2013) |
| G 0.48 | |||||
| A 0.52 | |||||
| RA and DAS28 | Japan | 55 RA patients | MTRR A66G | N.A. | Kato et al. (2012) |
| A 0.75 | |||||
| G 0.25 | |||||
| RFC1 G80A | N.A. | ||||
| G 0.47 | |||||
| A 0.53 | |||||
| MTHFR C677T | N.A. | ||||
| C 0.6 | |||||
| T 0.4 | |||||
| MTHFR A1298C | AA genotype had lower mean DAS28 than 1298AC/CC genotypes | ||||
| A 0.86 | |||||
| C 0.14 | |||||
DAS28, disease activity score 28; MTX, methotrexate; N.A., no association.
The role of polymorphisms MTRR A66G, RFC1 G80A, and MTHFR C677T and A1298C in RA and other autoimmune diseases remains unclear.
Our failure to find an association between RA and these SNPs could be due to the small sample size included in this study or to the weak contribution of these genes to RA onset and evolution, either in Mexico or worldwide.
Polymorphisms and disease activity
The patients of this study showed the three levels of disease activity, with the most frequent being moderate activity. Although there are clinical or laboratory measures used in clinical practice, a good individual performance in the assessment of disease activity has not been demonstrated, so it has been decided to use DAS28 for a better classification of the disease (Prevoo et al., 1995; Fransen et al., 2003), which is used as the basis for the evaluation of the response to treatment established by the European League Against Rheumatism. Although we found no statistical differences between polymorphisms and disease activity, there was a trend in three of the polymorphisms studied (MTRR 66GG, RFC1 80AA, and MTHFR 1298CC) and we concluded that perhaps a larger sample size might have yielded data with statistical significance.
This study has some limitations to be discussed: Because this study was exploratory, there was no previous information related with our main objective of investigating potential associations between RA severity and the SNPs MTRR A66G, RFC1 G80A, and MTHFR C677T and A1298C in the Mexican population from Western Mexico. In the results of the comparisons (described in Table 1) of allele and genotype frequencies observed in RA cases versus controls, we were not able to exclude the probability of an insufficient statistical power to identify differences between these two groups (type II error). Nevertheless, we consider that this study might help future investigations based in our data on the computation of the sample size required to perform these comparisons.
Otherwise, we have observed that although the n of controls increases, the allelic frequencies of the polymorphisms are maintained, since a minimum of 100 alleles is required. Our results represent relevant information demonstrating that these polymorphisms might not be related with disease severity in RA. These findings support the importance of seeking other genetic factors that might predispose to the observed phenotype differences in the severity of this disease among these patients.
In conclusion, in this study, we did not find any significant associations between RA or RA characteristics such as activity disease and polymorphisms MTRR A66G, RFC1 G80A, and MTHFR C677T and A1298C. Additional studies that include greater numbers of patients and more polymorphisms related to this or other metabolic pathways are required to determine the influence of genetics on disease activity in RA in Mexican populations, and thus provide greater knowledge about individualized pharmacological therapies for a better response to treatment.
Author Disclosure Statement
No competing financial interests exist.
References
- Arakawa Y, Watanabe M, Inoue N, et al. (2012) Association of polymorphisms in DNMT1, DNMT3A, DNMT3B, MTHFR and MTRR genes with global DNA methylation levels and prognosis of autoimmune thyroid disease. Clin Exp Immunol 170:194–201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arnett FC, Edworthy SM, Bloch DA, et al. (1988) The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31:315–324 [DOI] [PubMed] [Google Scholar]
- Barrera Cruz A, Beltran Castillo J, Blanco Favela F, et al. (2010) Diagnóstico y Tratamiento de Artritis Reumatoide del Adulto. México: Secretaria de Salud [Google Scholar]
- Berkun Y, Levartovsky D, Rubinow A, et al. (2004) Methotrexate related adverse effects in patients with rheumatoid arthritis are associated with the A1298C polymorphism of the MTHFR gene. Ann Rheum Dis 63:1227–1231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boughrara W, Aberkane M, Fodil M, et al. (2015) Impact of MTHFR rs1801133, MTHFR rs1801131 and ABCB1 rs1045642 polymorphisms with increased susceptibility of rheumatoid arthritis in the West Algerian population: a case-control study. Acta Reumatol Port 40:363–371 [PubMed] [Google Scholar]
- Brambila-Tapia AJ, Durán-González J, Sandoval-Ramírez L, et al. (2012) MTHFR C677T, MTHFR A1298C, and OPG A163G polymorphisms in Mexican patients with rheumatoid arthritis and osteoporosis. Dis Markers 32:109–114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calabrese DC, Konop R, Cannon E, et al. (2001) Discussion. Treatment algorithm: managing rheumatoid arthritis. Manag Care 10:19–24 [PubMed] [Google Scholar]
- Chaabane S, Marzouk S, Akrout R, et al. (2016) Genetic determinants of methotrexate toxicity in tunisian patients with rheumatoid arthritis: a study of polymorphisms involved in the MTX metabolic pathway. Eur J Drug Metab Pharmacokinet 41:385–393 [DOI] [PubMed] [Google Scholar]
- Cutolo M, Sulli A, Pizzorni C, et al. (2001) Anti-inflammatory mechanisms of methotrexate in rheumatoid arthritis. Ann Rheum Dis 60:729–735 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dávalos IP, Olivares N, Castillo MT, et al. (2001) The C677T polymorphism of the methylenetetrahydrofolate reductase gene in Mexican mestizo, neural tube defects parents, control Mestizo and native populations. Ann Genet 43:89–92 [DOI] [PubMed] [Google Scholar]
- Dervieux T, Furst D, Lein DO, et al. (2004) Polyglutamation of methotrexate with common polymorphisms in reduced folate carrier, aminoimidazole carboxamide ribonucleotide transformylase, and thymidylate synthase are associated with methotrexate effects in rheumatoid arthritis. Arthritis Rheum 50:2766–2774 [DOI] [PubMed] [Google Scholar]
- Firestein G. (2005) Pathogenesis of rheumatoid arthritis: how early is easy? Arthritis Res Ther 7:157–159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fransen J, Stucki G, van Riel PLCM. (2003) Rheumatoid arthritis measures. Disease activity score (DAS), disease activity score 28 (DAS28), rapid assessment of disease activity in rheumatology (RADAR), and rheumatoid arthritis disease activity index (RADAI). Arthritis Rheum 49:214–224 [Google Scholar]
- Frosst P, Blom Hj, Milos R, et al. (1995) A candidate genetic risk factor for vascular disease: a common mutation in methylenetetrahydrofolate reductase. Nature Genet 10:111–113 [DOI] [PubMed] [Google Scholar]
- Fukino K, Kawashima T, Suzuki M, et al. (2007) Methylenetetrahydrofolate reductase and reduced folate carrier-1 genotypes and methotrexate serum concentrations in patients with rheumatoid arthritis. J Toxicol Sci 32:449–452 [DOI] [PubMed] [Google Scholar]
- Gaughan DJ, Kluijtmans LAJ, Barbaux S, et al. (2001) The methionine synthase reductase (MTRR) A66G polymorphism is a novel genetic determinant of plasma homocysteine concentrations. Atherosclerosis 157:451–456 [DOI] [PubMed] [Google Scholar]
- González-Mercado MG, Rivas-Solis F, Salazar-Páramo M, et al. (2014) Homocisteína, polimorfismos MTHFR C677T y A1298C y variables clínico/bioquímicas en población mexicana. Acta Bioq Clin Lat 48:23–31 [Google Scholar]
- Goyette P, Pai A, Milos R. (1998) Gene structure of human and mouse methylenetetrahydrofolate reductase (MTHFR). Mamm Genome 9:652–656 [DOI] [PubMed] [Google Scholar]
- GUIPCAR (2011) Guía de práctica clínica para el manejo de la Artritis Reumatoidea (Sociedad Española de Reumatologia 2011). http://portal.guiasalud.es/web/guest/home (last accessed September3, 2016)
- Gustincich S, Manfioletti G, Del Sal G, et al. (1991) A fast method for high quality genomic-DNA extraction from whole human blood. Biotechniques 11:300–302 [PubMed] [Google Scholar]
- Hayashi H, Tazoe Y, Tsuboi S, et al. (2013) A single nucleotide polymorphism of reduced folate carrier 1 predicts methotrexate efficacy in Japanese patients with rheumatoid arthritis. Drug Metab Pharmacokinet 28:164–168 [DOI] [PubMed] [Google Scholar]
- Hughes LB, Beasley TM, Patel H, et al. (2006) Racial or ethnic differences in allele frequencies of single-nucleotide polymorphisms in the methylenetetrahydrofolate reductase gene and their influence on response to methotrexate in rheumatoid arthritis. Ann Rheum Dis 65:1213–1218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inanir A, Yigit S, Tekcan A, et al. (2013) IL-4 and MTHFR gene polymorphism in rheumatoid arthritis and their effects. Immunol Lett 152:104–108 [DOI] [PubMed] [Google Scholar]
- INEGI (2016) Instituto Nacional de Estadística y Geografía. Available at www.beta.inegi.org.mx (last accessed September3, 2016)
- Inoue K, Yuasa H. (2014) Molecular basis for pharmacokinetics and pharmacodynamics of methotrexate in rheumatoid arthritis therapy. Drug Metab Pharmacokinet 29:12–19 [DOI] [PubMed] [Google Scholar]
- Jacques P. (2003) Effects of polymorphisms of methionine synthase and methionine synthase reductase on total plasma homocysteine in the NHLBI Family Heart Study. Atherosclerosis 166:49–55 [DOI] [PubMed] [Google Scholar]
- Kato T, Hamada A, Mori S, et al. (2012) Genetic polymorphisms in metabolic and cellular transport pathway of methotrexate impact clinical outcome of methotrexate monotherapy in Japanese patients with rheumatoid arthritis. Drug Metab Pharmacokinet 27:192–199 [DOI] [PubMed] [Google Scholar]
- Li X, Hu M, Li W, et al. (2016) The association between reduced folate carrier-1 gene 80G/A polymorphism and methotrexate efficacy or methotrexate related-toxicity in rheumatoid arthritis: a meta-analysis. Int Immunopharmacol 38:8–15 [DOI] [PubMed] [Google Scholar]
- Makinen H, Kautiainen H, Hannonen P, et al. (2005) Is DAS28 an appropiate tool to assess remission in rheumatoid arthritis? Ann Rheum Dis 64:1410–1413 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matherly LH, Hou Z, Deng Y. (2007) Human reduced folate carrier: translation of basic biology to cancer etiology and therapy. Cancer Metastasis Rev 26:111–128 [DOI] [PubMed] [Google Scholar]
- McInnes IB, Schett G. (2011) The pathogenesis of rheumatoid arthritis. N Engl J Med 365:2205–2219 [DOI] [PubMed] [Google Scholar]
- Mena JP, Salazar-Páramo M, González-López L, et al. (2011) Polymorphisms C677T and A1298C in the MTHFR gene in Mexican patients with rheumatoid arthritis treated with methotrexate: implication with elevation of transaminases. Pharmacogenomics J 11:287–291 [DOI] [PubMed] [Google Scholar]
- Miller SA, Dikes DD, Polesky HP. (1989) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acid Res 16:1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muralidharan N, Mariaselvam CM, Mithun CB, et al. (2016) Reduced folate carrier-1 80G>A gene polymorphism is not associated with methotrexate treatment response in South Indian Tamils with rheumatoid arthritis. Clin Rheumatol 35:879–885 [DOI] [PubMed] [Google Scholar]
- Naghibalhossaini F, Ehyakonandeh H, Nikseresht A, et al. (2015) Association between MTHFR genetic variants and multiple sclerosis in a Southern Iranian population. Int J Mol Cell Med 4:87–93 [PMC free article] [PubMed] [Google Scholar]
- Oliver JE, Silman AJ. (2009) What epidemiology has told us about risk factors and aetiopathogenesis in rheumatic diseases. Arthritis Res Ther 11:223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prevoo MLL, van't Hof MA, Kuper HH, et al. (1995) Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum 38:44–48 [DOI] [PubMed] [Google Scholar]
- Puente-Torres L, Hurtado-Torres GF, Abud-Mendoza C, et al. (2009) Evaluación del estado nutricio en una población mexicana de pacientes adultos con artritis reumatoide. Nutr Hosp 24:233–238 [PubMed] [Google Scholar]
- Remuzgo-Martínez S, Genre F, López-Mejías R, et al. (2016) Decreased expression of methylene tetrahydrofolate reductase (MTHFR) gene in patients with rheumatoid arthritis. Clin Exp Rheumatol 34:106–110 [PubMed] [Google Scholar]
- Rodríguez-Acosta R, Arévalo-Tan CE, Pérez-García D, et al. (2001) Factores sociodemográficos y severidad radiológica de la artritis reumatoide. Rev Cubana Med Gen Integr 17:423–428 [Google Scholar]
- Rodríguez-Valverde V, García-Alvaro JM, Tornero-Molina J. (2004) En representación de los médicos del panel. Segunda actualizacion del consenso de la Sociedad Española de Reumatologia sobre la terapia biologica en la Artritis Reumatoidea. Rev Esp Reumatol 31:394–401 [Google Scholar]
- Rubini M, Padovan M, Baricordi O, et al. (2008) The c.1298A>C polymorphism in the methylenetetrahydrofolate reductase gene is associated with rheumatoid arthritis susceptibility in Italian patients. Clin Exp Rheumatol 26:163. [PubMed] [Google Scholar]
- Saad MN, Mabrouk MS, Eldeib AM, et al. (2015a) Genetic case-control study for eight polymorphisms associated with rheumatoid arthritis. PLoS One 10:e0131960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saad MN, Mabrouk MS, Eldeib AM, et al. (2015b) Effect of MTHFR, TGFbeta1, and TNFB polymorphisms on osteoporosis in rheumatoid arthritis patients. Gene 568:124–128 [DOI] [PubMed] [Google Scholar]
- Saad MN, Mabrouk MS, Eldeib AM, et al. (2016) Identification of rheumatoid arthritis biomarkers based on single nucleotide polymorphisms and haplotype blocks: a systematic review and meta-analysis. J Adv Res 7:1–16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salazar J, Moya P, Altés A, et al. (2014) Polymorphisms in genes involved in the mechanism of action of methotrexate: are they associated with outcome in rheumatoid arthritis patients? Pharmacogenomics 15:1079–1090 [DOI] [PubMed] [Google Scholar]
- Shi M, Caprau D, Romitti P, et al. (2003) Genotype frequencies and linkage disequilibrium in the CEPH human diversity panel for variants in folate pathway genes MTHFR, MTHFD, MTRR, RFC1, and GCP2. Birth Defects Res A Clin Mol Teratol 67:545–549 [DOI] [PubMed] [Google Scholar]
- SNP [Internet]. Bethesda, MD: National Library of Medicine (US), National Center for Biotechnology Information; 2004. Available at https://www.ncbi.nlm.nih.gov/snp (last accessed date July27, 2017) [Google Scholar]
- Spindler A, Bellomio V, Berman A. (2002) Prevalence of rheumatoid arthritis in Tucuman, Argentina. J Rheumatol 29:1166–1170 [PubMed] [Google Scholar]
- Summers CM, Cucchiara AJ, Nackos E, et al. (2008) Functional polymorphisms of folate-metabolizing enzymes in relation to homocysteine concentrations in systemic lupus erythematosus. J Rheumatol 35:2179–2186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van der Heijde D, Landewé R, Klareskog L, et al. (2005) Presentation and analysis of data on radiographic outcome in clinical trials: experience from the TEMPO study. Arthritis Rheum 52:49–60 [DOI] [PubMed] [Google Scholar]
- Van Der Put NM, Gabreels F, Stevens EM, et al. (1998) A second common mutation in the methylenetetrahydrofolate reductase gene: an additional risk factor for neural-tube defects? Am J Hum Genet 62:1044–1051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wessels JA, Kooloos WM, De Jonge R, et al. (2006) Relationship between genetic variants in the adenosine pathway and outcome of methotrexate treatment in patients with recent-onset rheumatoid arthritis. Arthritis Rheum 54:2830–2839 [DOI] [PubMed] [Google Scholar]