Summary
We reviewed 37 studies reporting long‐term outcomes after a status epilepticus (SE) episode in pediatric and adult populations. Study design, length of follow‐up, outcome measures, domains investigated (mortality, SE recurrence, subsequent epilepsy, cognitive outcome, functional outcome, or quality of life), and predictors of long‐term outcomes are summarized. Despite heterogeneity in the design of prior studies, overall risk of poor long‐term outcome after SE is high in both children and adults. Etiology is the main determinant of outcome, and the effect of age or SE duration is often difficult to distinguish from the underlying cause. The effect of the treatment on long‐term outcome after SE is still unknown.
Keywords: cognitive outcome, epilepsy, functional impairment, mortality, neurological sequelae, quality of life
Key Points.
Long‐term outcomes in patients with status epilepticus are predicted by underlying etiology
Long‐term mortality after status epilepticus is seen in up to 20% of children and 55% of adults
Status epilepticus is associated with increased rates of status epilepticus recurrence, subsequent epilepsy, and worsening of previous epilepsy
Further studies using standardized tools are in progress to assess quality of life and functional and cognitive outcome
Promising research suggests that functional outcome may improve over time
1. INTRODUCTION
Status epilepticus (SE), especially refractory SE (RSE), is a life‐threatening condition often requiring intensive care.1, 2, 3, 4 Long‐term sequelae may include neurological, cognitive, and behavioral impairments and decline in quality of life (QoL),4, 5 and impose heavy burdens on the patient, the caregivers, and the healthcare system. Outcomes are influenced by type of epilepsy, type of SE, etiology, SE duration, and patient's age.4 This review aims to provide an overview of long‐term outcomes in patients with SE.
2. LITERATURE SEARCH
We performed a search of the medical literature using the following strategy in PubMed: (“status epilepticus”) AND (“long‐term outcome” OR “long‐term mortality” OR “long‐term morbidity” OR “quality of life”), gathering 570 articles. Our search was restricted to full‐length clinical studies in humans written in the English language until December 1, 2017. We excluded studies focusing on the neonatal period (1‐28 days of postnatal age at the beginning of SE) and studies evaluating solely short‐term outcome or very specific subgroups of patients. In addition, we added relevant articles from the reference lists of articles from the primary search. We included the results of 37 relevant studies, 16 on children (Table 1), 14 on adults and seven on a mixed population of adults and children (Table 2), reporting long‐term outcomes after SE (ie, after hospital discharge or >1 month from SE onset), and reviewed studies for predictors of outcome (Figure 1).
Table 1.
Pediatric studies on long‐term outcome after SE
| Authors, study design | Year | Population | Follow‐up testing/scores | Mortality rate | Outcome: epilepsy | Outcome: cognitive abilities | Outcome: QoL | Outcome: functional outcome/other | Predictors |
|---|---|---|---|---|---|---|---|---|---|
| Chevrie & Aicardi,38 prospective | 1978 | Children 28 d‐1 y, n = 313, SE: n = 40 | >1 y, median 3 y, no formal testing | 22%, patients with SE | N/A | Severe intellectual disability in SE: 60% | N/A | Neurological sequelae in SE: 43% | Cognitive abilities; poor outcome: symptomatic cases, age < 6 mo; good outcome: positive family history; functional outcome: pre‐ or perinatal abnormality |
| Maytal et al,26 retrospective and prospective | 1989 | Children in SE, n = 193 | 13.2 mo, no formal testing | 3.6%, at 3 mo | New epilepsy: 30% | N/A | N/A | Neurologic deficits, motor or cognitive: 9.1% | Functional outcome: duration of SE (in the acute symptomatic group), etiology (acute or progressive insults) |
| Shinnar et al,35 prospective | 1992 | Children with first SE, n = 95 | 29 mo, no formal testing | 3 died within 10 d | Recurrence: 17% | N/A | N/A | No deterioration in neurological function | Recurrence: underlying neurological abnormality, etiology (remote symptomatic and progressive causes) |
| Eriksson & Koivikko,27 retrospective | 1997 | Children in SE, n = 65 | 3.6 y | 0% | New epilepsy: 23% | N/A | N/A | Neurological sequelae: 15% | Functional outcome: SE duration > 2 h |
| Barnard & Wirrell,28 retrospective | 1999 | Children in SE, n = 47 | 53 mo, developmental quotient | 15.4% | New epilepsy: 36%; recurrent seizures: 85%; refractory epilepsy: 25%; recurrent SE: 50% | Neurodevelopmental deterioration: 34% (26%: severe) | School: normal (6%), resource assistance (14%), teaching aides (37%), special class (43%); behavior problems: 41% | Neurological sequelae: 79% (mild: 21%, moderate: 28%, severe: 34%) | Functional outcome: etiology (nonfebrile‐nonidiopathic), perinatal difficulties, developmental delay, abnormal neurologic examination; abnormal neuroimaging; developmental deterioration: etiology, neuroimaging, young age; epilepsy: etiology |
| Kim et al,24 case series | 2001 | Children in RSE treated with pentobarbital, n = 23 | 4 y | 43.5%, in hospital | N/A | N/A | N/A | Neurological sequelae: 61.5% | Mortality: failure of seizure control after PB coma, acute symptomatic etiology; functional outcome: treatment delayed |
| Tabarki et al,36 retrospective | 2001 | Children in SE, n = 139 | 3.5 y, no formal testing | 15.8%, within 2 mo | Epilepsy without cognitive deterioration: 5% |
Moderate intellectual disability: 11.5%; severe: 19.5%; remission: 48% |
N/A | N/A | Functional and cognitive outcome: etiology (remote symptomatic and progressive encephalopathy > acute symptomatic > febrile and idiopathic); age < 1 y |
| Sillanpää & Shinnar,34 prospective | 2002 | Children with childhood onset epilepsy, n = 150, 27% with SE, n = 41 | 30 y | 16% | Recurrence of SE: 56%; remission: 55% (compared to 80% in children without SE) | N/A | Comparing SE or not: similar social and educational outcome | N/A | Mortality and social outcome: occurrence of SE did not alter mortality rate and social and educational outcome; remission rates: slightly affected by SE |
| Metsäranta et al,6 retrospective | 2004 | Children in SE, n = 186 | 2 y and 1 mo, no formal testing | 0% | New onset epilepsy: 22% | N/A | N/A | Permanent neurological sequelae: 2.2%, temporary sequelae: 3.2% | Minor neurological sequelae: duration of seizure; major neurological sequelae: acute symptomatic cause; temporary sequelae: febrile SE |
| Maegaki et al,17 retrospective | 2005 | Children in SE, n = 234 | 64 mo, no formal testing | 4% | N/A | N/A | N/A | Neurological sequelae: 15% | Mortality/functional outcome: etiology (acute neurological insult and progressive neurological disease), seizure duration (>2 h), asthmatic attack |
| Hussain et al,29 retrospective | 2007 | Children in SE, n = 137 | 1.5 y, no formal testing | 0% | New epilepsy: 5%; recurrence of SE: 10% | N/A | N/A | Neurological sequelae among previously normal children: 1.4% | New epilepsy: remote symptomatic group, no correlation with duration of SE |
| Wagenman et al,45 prospective | 2014 | Children with acute neurologic disorders neurodevelopmentally normal before PICU admission with ES or ESE, n = 137 | 2.7 y, GOS‐E Peds and PedsQoL | 0% | Subsequent seizures: 35% | N/A | PedsQoL: median = 94 for SE and 62 for ESE | Unfavorable outcome GOS‐E Peds: 35% | Functional impairment: ESE; subsequent seizures: ESE |
| Pujar et al,53 prospective | 2011 | Children with convulsive SE, n = 206 | 8 y | 3% within 1 mo, 11% within 8 y | N/A | N/A | N/A | N/A |
Mortality: preexisting clinically significant neurological impairments; better outcome: prolonged febrile convulsions and idiopathic convulsive SE |
| Camfield & Camfield,33 retrospective | 2012 | Children with focal epilepsy and normal intelligence (acute symptomatic causes not included), n = 188 (20% SE) | Mean 27 y, wide variety of testing | 0% | Seizure‐free: 61.5% with SE vs 66.4% without SE; intractability: 15% vs 11.4% | Learning disorders: 28 with SE vs 33% without SE | N/A | N/A | Cognitive outcome and epilepsy: no difference between the SE and non‐SE groups |
| Martinos et al,37 prospective | 2013 | Children from 1 to 42 mo, in SE (febrile or not), n = 54 |
At 6 wk and 1 y, Bayley Scales of Infant Development III |
1 patient died during follow‐up | N/A | Cognitive composite: FSE, 93; non‐FSE, 74; control, 107; language composite: FSE, 91; non‐FSE, 75; control, 114 | N/A | Motor composite: FSE, 96; non‐FSE, 77; control, 103 | Developmental outcome: nonfebrile SE < FSE < controls |
| Ferro et al,5 retrospective | 2014 | Children with newly diagnosed epilepsy, n = 374 (SE: 6.1%) | 24 mo, Quality of Life in Childhood Epilepsy Score | 0% | N/A | No cognitive deterioration | Poorer QoL in children with SE | N/A | N/A |
ES, electrographic seizure; ESE, electrographic SE; FSE, febrile status epilepticus; GOS‐E, Glasgow Outcome Scale–extended; GOS‐E Peds, pediatric Glasgow Outcome Scale–extended; N/A, nonapplicable; PB, pentobarbital; PedsQoL, Pediatric Quality of Life Inventory; PICU, pediatric intensive care unit; QoL, quality of life; RSE, refractory SE; SE, status epilepticus.
Table 2.
Adult or adult and pediatric studies on long‐term outcome after SE
| Authors, study design | Year | Population | Follow‐up testing/scores | Mortality rate | Outcome: epilepsy | Outcome: functional outcome/other | Predictors |
|---|---|---|---|---|---|---|---|
| DeLorenzo et al,13 retrospective and prospective | 1992 | Adults and children in SE, n = 546 | 7 y |
Children: 2.3%, adults: 25% |
N/A | N/A | Mortality: etiology (tumor, hematological disease, anoxia, metabolic and congenital malformation), age, SE duration (for short‐term mortality) |
| DeLorenzo et al,8 prospective | 1996 | Adults and children with a first SE, n = 166 | 2 y |
Children: 3%, adults: 26% (at 30 d) |
Recurrence of SE at 2 y: 13.3% | N/A | Mortality: SE duration, etiology (hypoxia); recurrence: age |
| Hesdorffer et al,30 retrospective | 1998 | Adults and children; acute symptomatic SE vs seizures, n = 416 | 10 y | N/A | New unprovoked epilepsy: 41% in the SE group vs 13% in non‐SE group | N/A | Epilepsy: SE, etiology (anoxia, structural or metabolic cause) |
| Logroscino et al,55 retrospective | 2002 | Adults and children, n = 145 | 30 d to 12 y | 43% (at 10 y) | N/A | N/A | Mortality: SE duration, etiology (acute symptomatic cause), myoclonic SE |
| Holtkamp et al,14 retrospective | 2005 | Adults and children (11‐94 y) in SE, n = 79 | Unclear, telephone calls (only for “de novo” SE) | RSE: 16.7%; SE, 8.6% (in hospital) | New symptomatic epilepsy: 22% in SE and 87.5% in RSE | N/A | Epilepsy: RSE predicted by encephalitis |
| Adachi et al,10 prospective | 2005 | Adults in SE (n = 15) or with epilepsy (n = 40) | 3.2 y, IQ and Weschler Adult Intelligence Scale | 0% | N/A | No cognitive decline | Cognitive abilities: no influence of age, CPSE, GCSE, SE duration, etiology (AED withdrawal or unknown) |
| Hesdorffer et al,9 retrospective | 2007 | Adults and children with a first afebrile SE, n = 183 | 10 y | Unclear | Recurrence: 37% (30% among 30‐d survivors) | N/A | Recurrence: female gender, failure to respond the first drug administrated, progressive symptomatic cause |
| Logroscino et al,11 retrospective | 2008 | Adults, unprovoked epilepsy (n = 291) or SE (n = 16) | 10 y | 9% without SE vs 31% with SE | N/A | N/A | Mortality: age, development of subsequent epilepsy |
| Cooper et al,15 retrospective | 2009 | Adults with PRSE, n = 14 | Median of 313 d, mRS | 43% (in hospital), 57% (last follow‐up) | N/A | No change for 2 patients and improvement for 4 patients (mRS change: −1) | None |
| Legriel et al,20 prospective | 2010 | Adults in SE, n = 248 | 90 d, GOS | 18.8% (4 patients died during the follow‐up) | New epilepsy or worsening of previous epilepsy: 33.5% | Neurological sequelae (GOS = 2‐4): 38.8% | Functional impairment (GOS < 5): older age, focal neurological signs, RSE, cerebral insults, SE duration |
| Ristić et al,12 prospective | 2010 | Adults in SE, n = 750 | 12 y | 16% (short term), 22% (long term) | N/A | N/A | Mortality: etiology (progressive and acute symptomatic), age, epilepsy, initial SE |
| Kilbride et al,21 retrospective | 2013 | Adults in PRSE, n = 63 | At least 6 mo, mRS | 34% (in hospital) and 5 died during follow‐up | Seizure‐free: 20%; no recurrence | Good outcome: mRS ≤ 3: 22%; mRS = 1: 10% | Functional outcome: normal neuroimaging and a reactive EEG at SE onset |
| Li et al,23 case series | 2014 | Adults in SRSE, n = 13 | Median 17 mo, GOS | 36.3% (at 3 mo); 15.4% (in hospital) | 50% |
At 3 mo: mean GOS = 4.1; at 17 mo: mean GOS = 4.6 |
Functional outcome at 3 mo: etiology (encephalitis), longer period of anesthesia |
| Jayalakshmi et al,16 retrospective | 2014 | Adults and children, n = 177 | 6 mo, GOS | 19% | N/A | Good outcome (GOS = 4‐5): SRSE, 33%; non‐RSE, 79%; RSE, 57% | Functional outcome: SRSE predicted by presumed encephalitis |
| Lai et al,54 retrospective | 2015 |
Adults in PRSE, n = 78 |
1 y, telephone calls (mRS) | 52% | N/A | Poor outcome (mRS = 4‐6): 67% | Functional outcome (mRS = 4‐6): vasopressor use |
| Madzar et al,44 retrospective | 2016 | Adults in RSE, n = 65 | 12 wk, mRS | 18% (in hospital) | N/A | mRS > 2: 61% | Poor functional outcome: STESS ≥ 3, longer RSE duration (10 d), sepsis |
| Legriel et al,43 retrospective | 2016 | Adults in SE, n = 268 | 90 d, GOS | 13.8% (in hospital); 14.1% (at 90 d) | N/A | Good outcome (GOS = 5): 46% | No association with hypothermia |
| Kortland et al,18 retrospective | 2017 | Adults in SE, n = 81 (RSE: n = 33) | 3 mo, mRS, QOLIE‐31, NDDI‐E | 0% | Hospitalization due to epilepsy: 22.2% |
mRS = 0‐3: 60.8% (RSE: 70%); major depression: 32.8% (QOLIE‐31: SE: 43.5%, RSE: 42.5%) |
N/A |
| Atmaca et al,19 prospective | 2017 | Adults in SE, n = 59 (RSE: n = 15) | 13.6 ± 4.6 mo | 31% | N/A | Poor outcome (death or neurological sequelae): 46% |
Mortality and functional outcome: potentially fatal etiology, EMSE score > STESS, mSTESS |
| Kantanen et al,58 retrospective | 2017 | Adults in RSE, n = 395 | 1 y | 25% | N/A | N/A | Mortality: older age, SOFA score, dependence of activities in daily living, SRSE |
| Kantanen et al,59 retrospective | 2017 | Adults in RSE, n = 75 (SRSE: 21%) | 1 y, mRS | 23 (at 1 y); 7% (in hospital) | N/A |
Neurological deficits: 29%; mRS = 4‐6: 52% |
Mortality: older age |
AED, antiepileptic drug; CPSE, complex partial SE; EEG, electroencephalogram; EMSE, epidemiology‐based mortality in SE; GCSE, generalized convulsive SE; GOS, Glasgow Outcome Scale; IQ, intelligence quotient; mRS, modified Rankin Score; mSTESS, modified STESS; N/A, nonapplicable; NDDI‐E, Neurological Disorders Depression Inventory for Epilepsy; PRSE, prolonged refractory SE; QOLIE, Quality of Life in Epilepsy; RSE, refractory SE; SE, status epilepticus; SOFA, Sequential Organ Failure Assessment; SRSE, super‐refractory SE; STESS, Status Epilepticus Severity Score.
Figure 1.
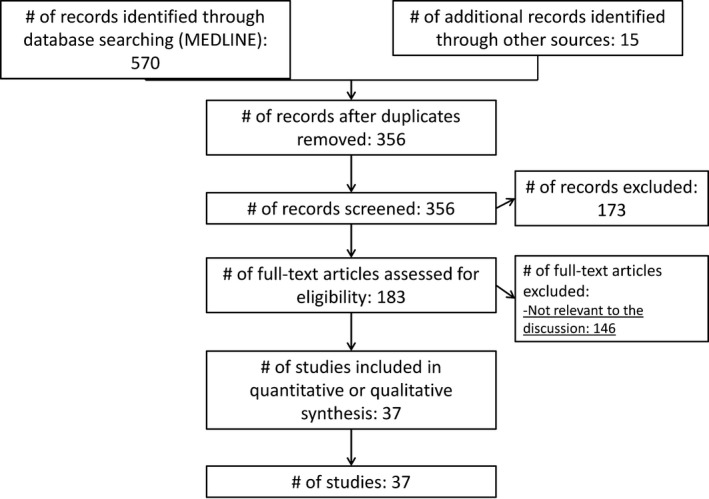
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta‐Analyses) diagram of the literature search
3. DEFINITIONS
In 15 pediatric studies, SE was defined as any seizure lasting >30 minutes or recurrent seizures lasting a total of >30 minutes without the subject fully regaining consciousness by most studies, except for one paper that used a 5‐minute limit.6 In 21 studies including adults, definitions were more variable, including 30‐minute seizure duration limits,7, 8, 9, 10, 11, 12, 13 and 5‐minute clinical seizure duration or more than two seizures without return to baseline between seizures.14, 15, 16, 17, 18, 19 One study also chose a 10‐minute clinical seizure duration cutoff limit.20 Two studies reported patients with prolonged refractory SE, including patients with SE that persists or recurs over a period of ≥7 days after the initiation of continuous general anesthesia.21, 22 Super‐refractory SE (SRSE) was defined as SE that continues or recurs ≥24 hours after the onset of anesthesic therapy, including those cases that recur on the reduction or withdrawal of anesthesia.23 Of note, the definition of RSE was also variable, but usually referred to SE that continues after administration of a benzodiazepine and a second antiseizure medication. One study defined RSE as SE lasting >60 minutes.24
4. APPROACH TO INTENSIVE CARE UNIT TREATMENT OF SE
Treatment of SE mostly follows general treatment guidelines and algorithms.1, 2 The first line usually consists of benzodiazepines, often followed by intravenous nonbenzodiazepine antiseizure medications (ASMs). If SE continues and becomes refractory, guidelines recommend transfer to the intensive care unit and additional anesthetic treatment, ideally within 30 or 40 minutes after SE onset.1, 2, 25 The most common drugs at this treatment stage are continuous infusions of midazolam, infusions of pentobarbital/thiopental, or intermittent phenobarbital doses.1 Main adverse events may include respiratory depression and hypotension, and thus mechanical ventilation and blood pressure management are often needed.25 Currently, class I evidence supports the use of benzodiazepines as first‐line treatments, and a major randomized controlled trial is in progress in an attempt to obtain data supporting choices of second‐line treatment (Established Status Epilepticus Treatment Trial). Limited data support third‐line treatment choices.2
5. LONG‐TERM OUTCOMES OF SE
5.1. Subsequent epilepsy
The risk of subsequent epilepsy after SE is high in both children and adults, with highest onset risk during the first year of follow‐up.4 In pediatric patients, this risk ranges from 5% to 36%.6, 26, 27, 28, 29 In mixed populations of adults and children, subsequent epilepsy after SE may occur in 22%‐41%,14, 30 which is less than in patients with history of RSE, who have a 87.5% risk of subsequent epilepsy.14 Only one adult study showed that 31% of patients have subsequent epilepsy or worsening of previous epilepsy after SE.20 The main predictor of subsequent epilepsy is the underlying SE etiology, and nonfebrile‐nonidiopathic,28 remote symptomatic,29 structural, and acute symptomatic (eg, anoxic brain injury)30 etiologies are usually associated with higher risk. In comparison, the risk of subsequent epilepsy after a single seizure is 40%‐50%.31, 32 In terms of epilepsy severity after SE, epilepsy becomes refractory in 15%‐25% of children after SE, which is not markedly different from the proportion of refractory cases in the general epileptic population.6, 33 In a population‐based series of 115 children with epilepsy, those who had SE, when compared with those who did not have an SE event, had a lower probability of epilepsy remission (55% vs 80%; risk ratio = 0.58, 95% confidence interval = 0.34‐0.99, P = 0.044). The patient who had SE also had a lower probability of epilepsy remission off ASMs (39% vs 65%; risk ratio = 0.50, 95% confidence interval = 0.27‐0.95, P = 0.029),34 suggesting a more refractory epilepsy in these patients.33, 34 In contrast, a study of 188 children with focal epilepsy found no statistically significant difference in the probability of epilepsy remission off ASM in patients who have SE events (61% in patients with SE vs 66% in patients without SE, P = 0.5).5
5.2. Recurrence of SE
In patients with SE, the occurrence of recurrent SE ranges from 10% to 56% in children28, 29, 34, 35 and from 13% to 37% in mixed population of adults and children.8, 9 Predictors of recurrent SE include age < 4 years,8 female gender, nonresponse to first ASM for SE,9 and remote symptomatic and progressive etiologies.9, 35
5.3. Cognitive outcome and functional outcome
Cognitive, functional, and QoL sequelae are frequent, especially in RSE and SRSE.16, 20 Long‐term cognitive sequelae occur in 28%‐34% of children.28, 33, 36, 37 In one adult study on long‐term cognitive sequelae after SE, no cognitive decline was found after 3 years of follow‐up, but this series included only 15 patients.10 Cognitive outcomes are usually evaluated based on clinical judgment or measured using a wide variety of neuropsychological tests. The underlying etiology is the main factor associated with long‐term cognitive outcome in children,6, 28, 33, 36, 38 with symptomatic SE28, 36, 38 or progressive encephalopathy36 contributing to increased risk. Other factors are young age at the time of SE28, 36, 38 and neuroimaging abnormalities28 (Table 1). Also, seizure burden in uncontrolled epilepsy, rather than SE, is more frequently associated with poor cognitive outcome.39, 40
The impact of SE on cognitive outcome is debatable. Animal models show that prolonged seizures result in neuronal loss and brain connectivity changes.41, 42 A clinical study showed that children with SE had worse long‐term cognitive outcome than healthy controls, with nonfebrile SE associated with worse cognitive impairments than febrile SE.37 In contrast, large studies showed no difference in cognitive outcome when comparing children with and without SE, although controls in this study were children with epilepsy.33, 34 Most adult studies focus on functional rather than cognitive outcomes using standardized scales like the modified Rankin Score and the Glasgow Outcome Scale, with functional deficits seen in 21‐61%,16, 20, 21, 43, 44 and these could be more severe in RSE20 or SRSE (67%).16 Functional outcomes in children are mostly based on clinical impression, yielding a wide spectrum of functional impairment after SE from 0% to 79%,6, 17, 24, 26, 27, 28, 29, 35, 36, 38, 45 and this range may also be related to different definitions and assessment of impairment, and often lack of good baseline information. The evaluation of long‐term cognitive outcomes is further complicated by evolution over time in some cases, and in particular outcomes in children are often not static as development progresses. In a pediatric study, impaired performance at discharge persisted at 1 year,37 whereas in another series deficits disappeared or improved over time.15, 22, 46 Predictors of poor functional outcome include etiology (nonfebrile SE, acute symptomatic SE, progressive encephalopathy)17, 26, 28, 36, 37 and SE duration17, 26, 27, 45 (Table 1).
5.4. QoL
There is limited literature on QoL after SE. Compared to short seizures, convulsive or electrographic‐only SE45 has a negative impact on the long‐term QoL.5 However, population‐based studies comparing adults with childhood‐onset epilepsy with or without SE showed no association with educational attainment, employment status, and income.33 Patients after RSE may achieve an equivalent QoL as compared to patients after non‐RSE.18 However, patients in seizure remission present better QoL results as compared to patients with SE.18 Other prospective studies showed only small associations between SE and selected domains of QoL.34, 47
5.5. Mortality
Short‐term mortality of SE ranges from 0% to 4%26, 48, 49 in children and 2%‐40% in adults, with higher mortality in RSE.26, 48, 50, 51, 52 Long‐term mortality data after an episode of SE, including in‐hospital deaths, is 0%‐22% in children5, 6, 17, 26, 27, 28, 29, 33, 34, 35, 37, 38, 45, 53 and 0%‐57% in adults.10, 11, 12, 17, 19, 20, 43, 54 Although long‐term mortality rates are high, the underlying etiology and the period of follow‐up are major determinants of outcome. A population‐based study reported 24 deaths among 150 patients with childhood onset epilepsy, but mortality was similar in those with or without prior SE.34 A prospective study including 206 children identified preexisting neurological comorbidities as a predictor of mortality.53 Risk factors for mortality in adults include etiology (progressive, remote, or acute symptomatic causes),8, 12, 13, 24, 55 older age,11, 12 SE duration,8, 13, 56 and development of subsequent epilepsy.11 The Status Epilepticus Severity Score is a valuable tool to assess in‐hospital mortality but has not been clearly validated to estimate long‐term mortality.57, 58, 59 The Epidemiology‐Based Mortality in Status Epilepticus score considers etiology, age, electroencephalogram, and comorbidities and has been associated with poor long‐term outcome in one prospective study.19
5.6. Health care utilization and cost
There are limited data on short‐term resource utilization in SE. Studies on mean SE cost estimated up to US$18 834 in the USA and up to €14 946 in Germany per admission, significantly higher than those related to admissions of patients with epilepsy (€1998‐€3475).56, 60, 61 However, these studies reflect the in‐hospital treatment, but SE is also associated with indirect costs because of unfavorable outcomes and costs or tentative income loss for those caring for patients with epilepsy.62 Surprisingly, there is a lack of studies on long‐term resource utilization due to SE.
6. DIFFERENCES BETWEEN ADULT AND PEDIATRIC POPULATION
Age is one of the main outcome predictors after SE,53 with the youngest (<1 year)13, 28, 36, 38 and oldest11, 12, 20 patients having the poorest long‐term outcomes (>65 years11 or odds/risk ratio = 1.04‐1.05/year12, 20). The higher mortality reported in younger children may also reflect the higher proportion of acute symptomatic cases in this age group.63, 64, 65 Of note, animal models have shown that immature neurons are more resistant to neuronal damage after a prolonged seizure.66, 67 This may be reflected in the finding that children have fewer cognitive sequelae of SE and lower mortality than adults.13, 68 However, sequelae in children usually affect a longer expected lifespan than in adults and the elderly.69
7. FACTORS AFFECTING OUTCOME
7.1. SE etiology
As in short‐term studies, etiology is the main determinant of long‐term morbidity and mortality related to SE, probably more than the SE episode itself.33, 34, 55 A large majority of studies assigned the etiology of SE into broad categories (acute symptomatic, progressive symptomatic, remote symptomatic, and idiopathic/cryptogenic) based on previous work and International League Against Epilepsy recommendations,70 but the category assignments may in part contain information bias, and various classifications are used.
In adults, etiologies associated with poor outcome include hypoxia,7, 8, 13 acute symptomatic,12, 20, 44, 45, 55 and progressive symptomatic causes.9, 12 In children, many studies have pointed out remote29, 35, 36 and acute6, 17, 24, 26, 71 symptomatic causes, progressive encephalopathies,17, 26, 35, 36 or more extensively “nonfebrile‐nonidiopathic SE”28 as predictors of poor outcome. In contrast, the risks of mortality53 and neurological deficits are low with febrile SE and cryptogenic/idiopathic SE.26, 27, 28, 72 However, the specific subcategory of presumed encephalitis or new onset refractory SE may be associated with worse long‐term outcome16, 73, 74 and prolonged duration of SE.21
7.2. Treatment
There is not sufficient evidence for the efficacy of treatment of RSE with anesthetic medications.2
Notably, some studies suggest that the use of intravenous anesthetic drugs (IVADs) is associated with negative outcomes.75, 76 Especially pentobarbital has been linked to the development of hypotension requiring prolonged duration of mechanical ventilation and vasopressor therapies,76 which have in turn been associated with poor long‐term outcome.54 Continuous infusion of thiopental was also associated with more frequent adverse events and worse outcome at 6 months compared to continuous infusions of midazolam.77 However, it is discussed whether the association with negative outcomes is effectively due to the use of IVADs, or due to confounding by indication, as patients who require continuous infusions are probably more critically ill.78 A recent prospective two‐site cohort study matched 406 patients (139 with IVADs) and found worse outcome in the group receiving IVADs, after adjusting for known outcome predictors.79 In a review of long‐term mortality in relationship to IVADs, the death rate was higher with thiopental/pentobarbital (46%) compared with propofol (36%), midazolam (34%), and ketamine (44%).80 However, there may be unknown predictors affecting outcome, and therefore additional comparative effectiveness data and randomized controlled trials are needed in this area.
Patients with SRSE are more likely to require multiple medication combinations and prolonged hospitalizations and to present severe deconditioning and often systemic complications. Systemic complications of SE can determine long‐term morbidity and mortality, including cardiomyopathy, pulmonary edema, and renal failure.11, 81 It remains unclear how medical treatments affect long‐term outcome of SE.
7.3. Short‐ and long‐term effect of acute treatment of SE
Most literature on the treatment of SE considers short‐term endpoints like seizure control or in‐hospital mortality. Delays in time to treatment are independently associated with worse outcomes in the short term (higher mortality, higher need for continuous infusions, longer convulsive duration, and more frequent hypotension).1, 2, 3, 33, 82, 83 Families and caregivers play a crucial role, as timely treatment is often possible if families and caregivers administer a rescue medication at home and quickly call emergency services.84, 85, 86 However, a survey of 100 families of patients with epilepsy showed that 87% had a rescue medication prescription, but only 61% of them reported receiving training on how to use it.86, 87 Furthermore, a study showed that only 37.5% of patients received prehospital treatment.85 Improving these factors could impact short‐term outcomes. However, the influence of acute treatment of SE on long‐term outcomes appears unclear, and the main predictor of long‐term outcome appears to be SE etiology.53
8. LIMITATIONS
Current data need to be interpreted in the setting of often retrospective or unstructured outcome assessment. Additionally, study populations are heterogeneous, different definitions of SE are often applied, and data are acquired in different geographical, socioeconomic, and health care system settings. In addition, follow‐up duration and the outcome measures are often variable, reflecting lack of standardized data collection or related guidelines in this field. The current literature does not permit a comparison between results from SE and RSE studies, as they differ in many aspects, including heterogeneity of study populations and study design. Thus far, SE patients are by and large not systematically followed with validated tests repeated over time, or through standardized clinical outcome tools over time. Lastly, we also acknowledge publication bias of specific results, and many publications may suffer from selection and information biases.80
9. OUTLOOK
Long‐term outcome after SE encompasses multiple domains including development of subsequent epilepsy, functional and cognitive deficits, and QoL. Mortality remains high, exacerbated by underlying neurological comorbidities, and about one‐third of the children develop cognitive sequelae after RSE. Etiology is the main determinant of long‐term outcome, but age, treatment timing, and status duration may also play a role, with potential opportunity for care improvements in the latter two. Future studies may either include larger numbers to adjust for confounders or focus on specific etiologies. There is an urgent need for large prospective and multicenter studies, adjusted for confounders and stratified by seizure type, etiology, treatment timing, and age to account for SE heterogeneity, using validated outcome measures, responsive to the intervention. For example, safety studies comparing midazolam, propofol, and barbiturates could be considered, also taking into the account the impact of adverse events caused by prolonged deep sedation88 and considering electroencephalographic endpoint and duration of anesthetic treatments, such as seizure suppression or burst suppression, and related long‐term outcomes. Neuroprotective approaches are likely to improve outcome of patients with acute symptomatic causes, which seems to be one of the most important risk factors.20 Some studies have demonstrated that functional outcome and likely QoL may improve over time.15, 18, 22, 23 Promising research in animal models is in the process of identifying biomarkers that can be modulated to minimize long‐term functional impairment.89, 90 Additional comparative effectiveness and interventional trials are underway to provide additional data. Translational and clinical research may move these findings to clinical practice in the near future.
DISCLOSURE OF CONFLICTS OF INTEREST
T.L. serves on the Laboratory Accreditation Board for Long Term (Epilepsy and Intensive Care Unit) Monitoring, on the Council (and as President) of the American Clinical Neurophysiology Society, on the American Board of Clinical Neurophysiology, as an Associate Editor for Seizure, and as an Associate Editor for Wyllie's Treatment of Epilepsy, 6th and 7th editions. He is part of pending patent applications to detect and predict seizures and to diagnose epilepsy. He receives research support from the National Institutes of Health, Epilepsy Research Fund, American Epilepsy Society, Epilepsy Foundation of America, Epilepsy Therapy Project, Patient‐Centered Outcomes Research Institute, and Pediatric Epilepsy Research Foundation, and received research grants from Lundbeck, Eisai, Upsher‐Smith, Acorda, and Pfizer. He serves as a consultant for Zogenix, Sunovion, Upsher‐Smith, Advance Medical, and Lundbeck. He performs video electroencephalogram long‐term and intensive care unit monitoring, electroencephalograms, and other electrophysiological studies at Boston Children's Hospital and affiliated hospitals and bills for these procedures and he evaluates pediatric neurology patients and bills for clinical care. He has received speaker honorariums from national societies including the American Academy of Neurology, American Epilepsy Society, and American Clinical Neurophysiology Society, and for grand rounds at various academic centers. His wife, Dr Karen Stannard, is a pediatric neurologist; she performs video‐electroencephalographic long‐term and intensive care unit monitoring, electroencephalograms, and other electrophysiological studies and bills for these procedures, and she evaluates pediatric neurology patients and bills for clinical care. The authors have no conflicts of interest to report. We confirm that we have read the Journal's position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
ACKNOWLEDGMENTS
I.S.F., M.G.L. and T.L. are supported by the Pediatric Epilepsy Research Foundation and the Epilepsy Research Fund. C.S. is supported by a scholarship by the IFCN. I.S.F. is funded by a grant for the study of Epileptic Encephalopathies from “Fundación Alfonso Martín Escudero” and by the HHV6 Foundation.
Sculier C, Gaínza‐Lein M, Sánchez Fernández I, Loddenkemper T. Long‐term outcomes of status epilepticus: A critical assessment. Epilepsia. 2018;59(S2):155–169. 10.1111/epi.14515
REFERENCES
- 1. Brophy GM, Bell R, Claassen J, et al. Guidelines for the evaluation and management of status epilepticus. Neurocrit Care. 2012;17:3–23. [DOI] [PubMed] [Google Scholar]
- 2. Glauser T, Shinnar S, Gloss D, et al. Evidence‐based guideline: treatment of convulsive status epilepticus in children and adults: report of the Guideline Committee of the American Epilepsy Society. Epilepsy Curr. 2016;16:48–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Abend NS, Loddenkemper T. Pediatric status epilepticus management. Curr Opin Pediatr. 2014;26:668–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Raspall‐Chaure M, Chin RFM, Neville BG, Scott RC. Outcome of paediatric convulsive status epilepticus: a systematic review. Lancet Neurol. 2006;5:769–79. [DOI] [PubMed] [Google Scholar]
- 5. Ferro MA, Chin RFM, Camfield CS, Wiebe S, Levin SD, Speechley KN. Convulsive status epilepticus and health‐related quality of life in children with epilepsy. Neurology. 2014;83:752–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Metsäranta P, Koivikko M, Peltola J, Eriksson K. Outcome after prolonged convulsive seizures in 186 children: low morbidity, no mortality. Dev Med Child Neurol. 2004;46:4–8. [DOI] [PubMed] [Google Scholar]
- 7. Hesdorffer DC, Logroscino G, Cascino G, Annegers JF, Hauser WA. Incidence of status epilepticus in Rochester, Minnesota, 1965‐1984. Neurology. 1998;50:735–41. [DOI] [PubMed] [Google Scholar]
- 8. DeLorenzo RJ, Hauser WA, Towne AR, et al. A prospective, population‐based epidemiologic study of status epilepticus in Richmond, Virginia. Neurology. 1996;46:1029–35. [DOI] [PubMed] [Google Scholar]
- 9. Hesdorffer DC, Logroscino G, Cascino GD, Hauser WA. Recurrence of afebrile status epilepticus in a population‐based study in Rochester, Minnesota. Neurology. 2007;69:73–8. [DOI] [PubMed] [Google Scholar]
- 10. Adachi N, Kanemoto K, Muramatsu R, et al. Intellectual prognosis of status epilepticus in adult epilepsy patients: analysis with Wechsler Adult Intelligence Scale‐revised. Epilepsia. 2005;46:1502–9. [DOI] [PubMed] [Google Scholar]
- 11. Logroscino G, Hesdorffer DC, Cascino G, Hauser WA. Status epilepticus without an underlying cause and risk of death: a population‐based study. Arch Neurol. 2008;65:221–4. [DOI] [PubMed] [Google Scholar]
- 12. Ristić AJ, Sokić DV, Trajković G, et al. Long‐term survival in patients with status epilepticus: a tertiary referral center study. Epilepsia. 2010;51:57–61. [DOI] [PubMed] [Google Scholar]
- 13. DeLorenzo RJ, Towne AR, Pellock JM, Ko D. Status epilepticus in children, adults, and the elderly. Epilepsia. 1992;33(suppl 4):S15–25. [DOI] [PubMed] [Google Scholar]
- 14. Holtkamp M, Othman J, Buchheim K, Meierkord H. Predictors and prognosis of refractory status epilepticus treated in a neurological intensive care unit. J Neurol Neurosurg Psychiatry. 2005;76:534–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Cooper AD, Britton JW, Rabinstein AA. Functional and cognitive outcome in prolonged refractory status epilepticus. Arch Neurol. 2009;66:1505–9. [DOI] [PubMed] [Google Scholar]
- 16. Jayalakshmi S, Ruikar D, Vooturi S, et al. Determinants and predictors of outcome in super refractory status epilepticus—a developing country perspective. Epilepsy Res. 2014;108:1609–17. [DOI] [PubMed] [Google Scholar]
- 17. Maegaki Y, Kurozawa Y, Hanaki K, Ohno K. Risk factors for fatality and neurological sequelae after status epilepticus in children. Neuropediatrics. 2005;36:186–92. [DOI] [PubMed] [Google Scholar]
- 18. Kortland L‐M, Knake S, von Podewils F, Rosenow F, Strzelczyk A. Socioeconomic outcome and quality of life in adults after status epilepticus: a multicenter, longitudinal, matched case‐control analysis from Germany. Front Neurol. 2017;8:507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Atmaca MM, Bebek N, Baykan B, Gökyiğit A, Gürses C. Predictors of outcomes and refractoriness in status epilepticus: a prospective study. Epilepsy Behav. 2017;75:158–64. [DOI] [PubMed] [Google Scholar]
- 20. Legriel S, Azoulay E, Resche‐Rigon M, et al. Functional outcome after convulsive status epilepticus. Crit Care Med. 2010;38:2295–303. [DOI] [PubMed] [Google Scholar]
- 21. Kilbride RD, Reynolds AS, Szaflarski JP, Hirsch LJ. Clinical outcomes following prolonged refractory status epilepticus (PRSE). Neurocrit Care. 2013;18:374–85. [DOI] [PubMed] [Google Scholar]
- 22. Erklauer JO, Wilfong A, Jeanine G. Outcome in refractory and super‐refractory status epilepticus in children. Neurocrit Care. 2016;25:S1–310. [Google Scholar]
- 23. Li Y, Tian L, Zeng T, Chen J, Chen L, Zhou D. Clinical features and outcome of super‐refractory status epilepticus: a retrospective analysis in west China. Seizure. 2014;23:722–7. [DOI] [PubMed] [Google Scholar]
- 24. Kim SJ, Lee DY, Kim JS. Neurologic outcomes of pediatric epileptic patients with pentobarbital coma. Pediatr Neurol. 2001;25:217–20. [DOI] [PubMed] [Google Scholar]
- 25. Loddenkemper T, Goodkin HP. Treatment of pediatric status epilepticus. Curr Treat Options Neurol. 2011;13:560–73. [DOI] [PubMed] [Google Scholar]
- 26. Maytal J, Shinnar S, Moshé SL, Alvarez LA. Low morbidity and mortality of status epilepticus in children. Pediatrics. 1989;83:323–31. [PubMed] [Google Scholar]
- 27. Eriksson KJ, Koivikko MJ. Status epilepticus in children: aetiology, treatment, and outcome. Dev Med Child Neurol. 1997;39:652–8. [DOI] [PubMed] [Google Scholar]
- 28. Barnard C, Wirrell E. Does status epilepticus in children cause developmental deterioration and exacerbation of epilepsy? J Child Neurol. 1999;14:787–94. [DOI] [PubMed] [Google Scholar]
- 29. Hussain N, Appleton R, Thorburn K. Aetiology, course and outcome of children admitted to paediatric intensive care with convulsive status epilepticus: a retrospective 5‐year review. Seizure. 2007;16:305–12. [DOI] [PubMed] [Google Scholar]
- 30. Hesdorffer DC, Logroscino G, Cascino G, Annegers JF, Hauser WA. Risk of unprovoked seizure after acute symptomatic seizure: effect of status epilepticus. Ann Neurol. 1998;44:908–12. [DOI] [PubMed] [Google Scholar]
- 31. Shinnar S, Berg AT, Moshe SL, et al. The risk of seizure recurrence after a first unprovoked afebrile seizure in childhood: an extended follow‐up. Pediatrics. 1996;98:216–25. [PubMed] [Google Scholar]
- 32. Berg AT. Risk of recurrence after a first unprovoked seizure. Epilepsia. 2008;49(suppl 1):13–8. [DOI] [PubMed] [Google Scholar]
- 33. Camfield P, Camfield C. Unprovoked status epilepticus: the prognosis for otherwise normal children with focal epilepsy. Pediatrics. 2012;130:e501–6. [DOI] [PubMed] [Google Scholar]
- 34. Sillanpää M, Shinnar S. Status epilepticus in a population‐based cohort with childhood‐onset epilepsy in Finland. Ann Neurol. 2002;52:303–10. [DOI] [PubMed] [Google Scholar]
- 35. Shinnar S, Maytal J, Krasnoff L, Moshe SL. Recurrent status epilepticus in children. Ann Neurol. 1992;31:598–604. [DOI] [PubMed] [Google Scholar]
- 36. Tabarki B, Yacoub M, Selmi H, Oubich F, Barsaoui S, Essoussi AS. Infantile status epilepticus in Tunisia. Clinical, etiological and prognostic aspects. Seizure. 2001;10:365–9. [DOI] [PubMed] [Google Scholar]
- 37. Martinos MM, Yoong M, Patil S, et al. Early developmental outcomes in children following convulsive status epilepticus: a longitudinal study. Epilepsia. 2013;54:1012–9. [DOI] [PubMed] [Google Scholar]
- 38. Chevrie JJ, Aicardi J. Convulsive disorders in the first year of life: neurological and mental outcome and mortality. Epilepsia. 1978;19:67–74. [DOI] [PubMed] [Google Scholar]
- 39. Hermann BP, Seidenberg M, Dow C, et al. Cognitive prognosis in chronic temporal lobe epilepsy. Ann Neurol. 2006;60:80–7. [DOI] [PubMed] [Google Scholar]
- 40. De Marchis GM, Pugin D, Meyers E, et al. Seizure burden in subarachnoid hemorrhage associated with functional and cognitive outcome. Neurology. 2016;86:253–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Holmes GL. Effects of early seizures on later behavior and epileptogenicity. Ment Retard Dev Disabil Res Rev. 2004;10:101–5. [DOI] [PubMed] [Google Scholar]
- 42. Deshpande LS, Lou JK, Mian A, Blair RE, Sombati S, DeLorenzo RJ. In vitro status epilepticus but not spontaneous recurrent seizures cause cell death in cultured hippocampal neurons. Epilepsy Res. 2007;75:171–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Legriel S, Lemiale V, Schenck M, et al. Hypothermia for neuroprotection in convulsive status epilepticus. N Engl J Med. 2016;375:2457–67. [DOI] [PubMed] [Google Scholar]
- 44. Madžar D, Geyer A, Knappe RU, et al. Association of seizure duration and outcome in refractory status epilepticus. J Neurol. 2016;263:485–91. [DOI] [PubMed] [Google Scholar]
- 45. Wagenman KL, Blake TP, Sanchez SM, et al. Electrographic status epilepticus and long‐term outcome in critically ill children. Neurology. 2014;82:396–404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Krumholz A, Sung GY, Fisher RS, Barry E, Bergey GK, Grattan LM. Complex partial status epilepticus accompanied by serious morbidity and mortality. Neurology. 1995;45:1499–504. [DOI] [PubMed] [Google Scholar]
- 47. Berg AT, Shinnar S, Testa FM, et al. Status epilepticus after the initial diagnosis of epilepsy in children. Neurology. 2004;63:1027–34. [DOI] [PubMed] [Google Scholar]
- 48. Chin RFM, Neville BGR, Peckham C, et al. Incidence, cause, and short‐term outcome of convulsive status epilepticus in childhood: prospective population‐based study. Lancet. 2006;368:222–9. [DOI] [PubMed] [Google Scholar]
- 49. Singh RK, Stephens S, Berl MM, et al. Prospective study of new‐onset seizures presenting as status epilepticus in childhood. Neurology. 2010;74:636–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Vignatelli L, Tonon C, D'Alessandro R, et al. Incidence and short‐term prognosis of status epilepticus in adults in Bologna, Italy. Epilepsia. 2003;44:964–8. [DOI] [PubMed] [Google Scholar]
- 51. Loddenkemper T, Syed TU, Ramgopal S, et al. Risk factors associated with death in in‐hospital pediatric convulsive status epilepticus. PLoS One. 2012;7:e47474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Lv RJ, Wang Q, Cui T, et al. Status epilepticus‐related etiology, incidence and mortality: a meta‐analysis. Epilepsy Res. 2017;136:12–7. [DOI] [PubMed] [Google Scholar]
- 53. Pujar S, Neville B, Scott R, Chin R. Death within 8 years after childhood convulsive status epilepticus: a population‐based study. Brain. 2011;134:2819–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Lai A, Outin HD, Jabot J, et al. Functional outcome of prolonged refractory status epilepticus. Crit Care. 2015;19:199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Logroscino G, Hesdorffer DC, Cascino GD, Annegers JF, Bagiella E, Hauser WA. Long‐term mortality after a first episode of status epilepticus. Neurology. 2002;58:537–41. [DOI] [PubMed] [Google Scholar]
- 56. Strzelczyk A, Knake S, Oertel WH, Rosenow F, Hamer HM. Inpatient treatment costs of status epilepticus in adults in Germany. Seizure. 2013;22:882–5. [DOI] [PubMed] [Google Scholar]
- 57. Aukland P, Lando M, Vilholm O, Christiansen EB, Beier CP. Predictive value of the Status Epilepticus Severity Score (STESS) and its components for long‐term survival. BMC Neurol. 2016;16:213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Kantanen A‐M, Reinikainen M, Parviainen I, Kälviäinen R. Long‐term outcome of refractory status epilepticus in adults: a retrospective population‐based study. Epilepsy Res. 2017;133:13–21. [DOI] [PubMed] [Google Scholar]
- 59. Kantanen A‐M, Kälviäinen R, Parviainen I, et al. Predictors of hospital and one‐year mortality in intensive care patients with refractory status epilepticus: a population‐based study. Crit Care. 2017;21:71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Penberthy LT, Towne A, Garnett LK, Perlin JB, DeLorenzo RJ. Estimating the economic burden of status epilepticus to the health care system. Seizure. 2005;14:46–51. [DOI] [PubMed] [Google Scholar]
- 61. Kortland L‐M, Alfter A, Bähr O, et al. Costs and cost‐driving factors for acute treatment of adults with status epilepticus: a multicenter cohort study from Germany. Epilepsia. 2016;57:2056–66. [DOI] [PubMed] [Google Scholar]
- 62. Kortland L‐M, Knake S, Rosenow F, Strzelczyk A. Cost of status epilepticus: a systematic review. Seizure. 2015;24:17–20. [DOI] [PubMed] [Google Scholar]
- 63. Aicardi J, Chevrie JJ. Convulsive status epilepticus in infants and children. A study of 239 cases. Epilepsia. 1970;11:187–97. [DOI] [PubMed] [Google Scholar]
- 64. Logroscino G, Hesdorffer DC, Cascino G, Annegers JF, Hauser WA. Short‐term mortality after a first episode of status epilepticus. Epilepsia. 1997;38:1344–9. [DOI] [PubMed] [Google Scholar]
- 65. Shinnar S, Pellock JM, Moshé SL, et al. In whom does status epilepticus occur: age‐related differences in children. Epilepsia. 1997;38:907–14. [DOI] [PubMed] [Google Scholar]
- 66. Holmes GL, Khazipov R, Ben‐Ari Y. Seizure‐induced damage in the developing human: relevance of experimental models. Prog Brain Res. 2002;135:321–34. [DOI] [PubMed] [Google Scholar]
- 67. Lothman EW, Bertram EH. Epileptogenic effects of status epilepticus. Epilepsia. 1993;34(suppl 1):S59–70. [DOI] [PubMed] [Google Scholar]
- 68. Shorvon S, Walker M. Status epilepticus in idiopathic generalized epilepsy. Epilepsia. 2005;46(suppl 9):73–79. [DOI] [PubMed] [Google Scholar]
- 69. Helmstaedter C. Cognitive outcome of status epilepticus in adults. Epilepsia. 2007;48(suppl 8):85–90. [DOI] [PubMed] [Google Scholar]
- 70. Hauser WA, Annegers JF, Kurland LT. Prevalence of epilepsy in Rochester, Minnesota: 1940‐1980. Epilepsia. 1991;32:429–45. [DOI] [PubMed] [Google Scholar]
- 71. Sahin M, Menache CC, Holmes GL, Riviello JJ. Prolonged treatment for acute symptomatic refractory status epilepticus: outcome in children. Neurology. 2003;61:398–401. [DOI] [PubMed] [Google Scholar]
- 72. Shinnar S, Pellock JM, Berg AT, et al. Short‐term outcomes of children with febrile status epilepticus. Epilepsia. 2001;42:47–53. [DOI] [PubMed] [Google Scholar]
- 73. Howell KB, Katanyuwong K, Mackay MT, et al. Long‐term follow‐up of febrile infection‐related epilepsy syndrome. Epilepsia. 2012;53:101–10. [DOI] [PubMed] [Google Scholar]
- 74. Sahin M, Menache CC, Holmes GL, Riviello JJ. Outcome of severe refractory status epilepticus in children. Epilepsia. 2001;42:1461–7. [DOI] [PubMed] [Google Scholar]
- 75. Marchi NA, Novy J, Faouzi M, Stähli C, Burnand B, Rossetti AO. Status epilepticus: impact of therapeutic coma on outcome. Crit Care Med. 2015;43:1003–9. [DOI] [PubMed] [Google Scholar]
- 76. Kowalski RG, Ziai WC, Rees RN, et al. Third‐line antiepileptic therapy and outcome in status epilepticus: the impact of vasopressor use and prolonged mechanical ventilation. Crit Care Med. 2012;40:2677–84. [DOI] [PubMed] [Google Scholar]
- 77. Bellante F, Legros B, Depondt C, Créteur J, Taccone FS, Gaspard N. Midazolam and thiopental for the treatment of refractory status epilepticus: a retrospective comparison of efficacy and safety. J Neurol. 2016;263:799–806. [DOI] [PubMed] [Google Scholar]
- 78. Sutter R, Kaplan PW. Can anesthetic treatment worsen outcome in status epilepticus? Epilepsy Behav. 2015;49:294–297. [DOI] [PubMed] [Google Scholar]
- 79. Sutter R, De Marchis GM, Semmlack S, et al. Anesthetics and outcome in status epilepticus: a matched two‐center cohort study. CNS Drugs. 2017;31:65–74. [DOI] [PubMed] [Google Scholar]
- 80. Ferlisi M, Shorvon S. The outcome of therapies in refractory and super‐refractory convulsive status epilepticus and recommendations for therapy. Brain J Neurol. 2012;135:2314–28. [DOI] [PubMed] [Google Scholar]
- 81. Vooturi S, Jayalakshmi S, Sahu S, Mohandas S. Prognosis and predictors of outcome of refractory generalized convulsive status epilepticus in adults treated in neurointensive care unit. Clin Neurol Neurosurg. 2014;126:7–10. [DOI] [PubMed] [Google Scholar]
- 82. Gaínza‐Lein M, Sánchez Fernández I, Jackson M, et al. Association of time to treatment with short‐term outcomes for pediatric patients with refractory convulsive status epilepticus. JAMA Neurol. 2018;75:410–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Wilkes R, Tasker RC. Pediatric intensive care treatment of uncontrolled status epilepticus. Crit Care Clin. 2013;29:239–57. [DOI] [PubMed] [Google Scholar]
- 84. Pellock JM, Marmarou A, DeLorenzo R. Time to treatment in prolonged seizure episodes. Epilepsy Behav. 2004;5:192–6. [DOI] [PubMed] [Google Scholar]
- 85. Sánchez Fernández I, Abend NS, Agadi S, et al. Time from convulsive status epilepticus onset to anticonvulsant administration in children. Neurology. 2015;84:2304–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Alldredge BK, Wall DB, Ferriero DM. Effect of prehospital treatment on the outcome of status epilepticus in children. Pediatr Neurol. 1995;12:213–6. [DOI] [PubMed] [Google Scholar]
- 87. Gainza‐Lein M, Benjamin R, Stredny C, McGurl M, Kapur K, Loddenkemper T. Rescue medications in epilepsy patients: a family perspective. Seizure. 2017;52:188–94. [DOI] [PubMed] [Google Scholar]
- 88. Sutter R, Kaplan PW, Rüegg S. Outcome predictors for status epilepticus—what really counts. Nat Rev Neurol. 2013;9:525–34. [DOI] [PubMed] [Google Scholar]
- 89. Rutten A, van Albada M, Silveira DC, et al. Memory impairment following status epilepticus in immature rats: time‐course and environmental effects. Eur J Neurosci. 2002;16:501–13. [DOI] [PubMed] [Google Scholar]
- 90. Wang C‐A, Lai M‐C, Lui C‐C, et al. An enriched environment improves cognitive performance after early‐life status epilepticus accompanied by an increase in phosphorylation of extracellular signal‐regulated kinase 2. Epilepsy Behav. 2007;11:303–9. [DOI] [PubMed] [Google Scholar]


