Abstract
Disease overview
Multiple myeloma accounts for approximately 10% of hematologic malignancies.
Diagnosis
The diagnosis requires ≥10% clonal bone marrow plasma cells or a biopsy proven plasmacytoma plus evidence of one or more multiple myeloma defining events (MDE): CRAB (hypercalcemia, renal failure, anemia, or lytic bone lesions) features felt related to the plasma cell disorder, bone marrow clonal plasmacytosis ≥60%, serum involved/uninvolved free light chain (FLC) ratio ≥100 (provided involved FLC is ≥100 mg/L), or >1 focal lesion on magnetic resonance imaging.
Risk stratification
Patients with del(17p), t(14;16), and t(14;20) have high-risk multiple myeloma. Patients with t(4;14) translocation and gain(1q) have intermediate-risk. All others are considered standard-risk.
Risk-adapted initial therapy
Initial treatment consists of bortezomib, lenalidomide, dexamethasone (VRd). In high-risk patients, carfilzomib, lenalidomide, dexamethasone (KRd) is an alternative to VRd. In eligible patients, initial therapy is given for approximately 3–4 cycles followed by autologous stem cell transplantation (ASCT). Standard risk patients can opt for delayed ASCT at first relapse. Patients not candidates for transplant are treated with VRd for approximately 8–12 cycles followed by lenalidomide or lenalidomide plus dexamethasone.
Maintenance therapy
After ASCT, lenalidomide maintenance is recommended for standard risk patients, while maintenance with a bortezomib-based regimen is needed for patients with intermediate or high-risk disease.
Management of refractory disease
Most patients require a triplet regimen at relapse, with the choice of regimen varying with each successive relapse. Aggressive relapse with extramedullary plasmacytomas or plasma cell leukemia may require anthracycline containing combination chemotherapy regimens.
DISEASE OVERVIEW
Multiple myeloma accounts for 1% of all cancers and approximately 10% of all hematologic malignancies.1 Each year over 30,000 new cases are diagnosed in the United States, and over 12,000 patients die of the disease.2 The annual age-adjusted incidence in the United States has remained stable for decades at approximately 4 per 100,000.3 Multiple myeloma is slightly more common in men than in women, and is twice as common in African-Americans compared with Caucasians.4 The median age of patients at the time of diagnosis is about 65 years.5
Unlike other malignancies that metastasize to bone, the osteolytic bone lesions in multiple myeloma exhibit no new bone formation.6 Bone disease is the main cause of morbidity and can be detected on routine skeletal radiographs, low-dose whole body computed tomography (WB-CT), magnetic resonance imaging (MRI), or fluoro-deoxyglucose (FDG) positron emission tomography/computed tomographic scans (PET/CT).7,8 Other major clinical manifestations are anemia, hypercalcemia, renal failure, and an increased risk of infections. Approximately 1 to 2% of patients have extramedullary disease (EMD) at the time of initial diagnosis, while 8% develop EMD later on in the disease course.9
Almost all patients with multiple myeloma evolve from an asymptomatic pre-malignant stage termed monoclonal gammopathy of undetermined significance (MGUS).10,11 MGUS is present in over 3% of the population above the age of 50,12,13 and the prevalence is approximately two-fold higher in blacks compared with whites.14,15 MGUS progresses to multiple myeloma or related malignancy a rate of 1% per year.16,17 Since MGUS is asymptomatic, over 50% of individuals who are diagnosed with MGUS have had the condition for over 10 years prior to the clinical diagnosis.18 In some patients, an intermediate asymptomatic but more advanced pre-malignant stage referred to as smoldering multiple myeloma (SMM) can be recognized clinically.19 SMM progresses to multiple myeloma at a rate of approximately 10% per year over the first 5 years following diagnosis, 3% per year over the next 5 years, and 1.5% per year thereafter. This rate of progression is influenced by the underlying cytogenetic type of disease; patients with t(4;14) translocation, del(17p), and gain(1q) are at a higher risk of progression from MGUS or SMM to multiple myeloma.20–22
DIAGNOSIS
The revised International Myeloma Working Group criteria for the diagnosis of multiple myeloma and related disorders are shown on Table 1.1 The diagnosis of multiple myeloma requires the presence of one or more myeloma defining events (MDE) in addition to evidence of either 10% or more clonal plasma cells on bone marrow examination or a biopsy-proven plasmacytoma. MDE consists of established CRAB (hypercalcemia, renal failure, anemia, or lytic bone lesions) features as well as 3 specific biomarkers: clonal bone marrow plasma cells ≥60%, serum free light chain (FLC) ratio ≥100 (provided involved FLC level is ≥100 mg/L), and more than one focal lesion on MRI. Each of the new biomarkers is associated with an approximately 80% risk of progression to symptomatic end-organ damage in two or more independent studies. The updated criteria represent a paradigm shift since they allow early diagnosis and initiation of therapy before end-organ damage.
Table 1.
International Myeloma Working Group Diagnostic Criteria for Multiple Myeloma and Related Plasma Cell Disorders
| Disorder | Disease Definition |
|---|---|
| Non-IgM monoclonal gammopathy of undetermined significance (MGUS) | All 3 criteria must be met:
|
| Smoldering multiple myeloma | Both criteria must be met:
|
| Multiple Myeloma | Both criteria must be met:
|
| IgM Monoclonal gammopathy of undetermined significance (IgM MGUS) | All 3 criteria must be met:
|
| Light Chain MGUS | All criteria must be met:
|
| Solitary Plasmacytoma | All 4 criteria must be met
|
| Solitary Plasmacytoma with minimal marrow involvement** | All 4 criteria must be met
|
Reproduced from Rajkumar SV, Dimopoulos MA, Palumbo A, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol 2014;15:e538–e548.
A bone marrow can be deferred in patients with low risk MGUS (IgG type, M protein <15 gm/L, normal free light chain ratio) in whom there are no clinical features concerning for myeloma
Solitary plasmacytoma with 10% or more clonal plasma cells is considered as multiple myeloma
When multiple myeloma is suspected clinically, patients should be tested for the presence of M proteins using a combination of tests that should include a serum protein electrophoresis (SPEP), serum immunofixation (SIFE), and the serum FLC assay.23 Approximately 2% of patients with multiple myeloma have true non-secretory disease and have no evidence of an M protein on any of the above studies.5,24 Bone marrow studies at the time of initial diagnosis should include fluorescent in situ hybridization (FISH) probes designed to detect t(11;14), t(4;14), t(14;16), t(6;14), t(14;20), trisomies, and del(17p) (see Risk-Stratification below).25 Conventional karyotyping to detect hypodiploidy and deletion 13 has value, but if FISH studies are done, additional value in initial risk-stratification is limited. Gene expression profiling (GEP) if available can provide additional prognostic value.26 Serum CrossLaps to measure carboxy-terminal collagen crosslinks (CTX) may be useful in assessing bone turnover and to determine adequacy of bisphosphonate therapy.27,28 The extent of bone disease is best assessed by low-dose WB-CT or PET/CT imaging.8 MRI scans are useful in patients with suspected SMM to rule out focal bone marrow lesions that can be seen before true osteolytic disease occurs. MRI imaging is also useful in assessing extramedullary disease, suspected cord compression, or when detailed imaging of a specific symptomatic area is needed. Conventional skeletal survey is less sensitive than low-dose WB-CT and PET/CT and recommended only if resources for more advanced imaging are not available.
The M protein is considered to be measurable if it is ≥1gm/dL in the serum and or ≥200 mg/day in the urine. The M protein level is monitored by SPEP and serum FLC assay to assess treatment response every month while on therapy, and every 3–4 months when off-therapy. The serum FLC assay is particularly useful in patients who lack a measurable M protein, provided the FLC ratio is abnormal and the involved FLC level is ≥100 mg/L.29 Urine protein electrophoresis is recommended at least once every 3–6 months, to follow the urine M protein level as well as to detect other renal complications that may result in albuminuria. Response to therapy assessment and minimal residual disease (MRD) evaluation is based on the revised International Myeloma Working Group uniform response criteria.30
MOLECULAR CLASSIFICATION
Although multiple myeloma is still considered a single disease, it is in reality a collection of several different cytogenetically distinct plasma cell malignancies (Table 2).31,32 On fluorescent in situ hybridization (FISH) studies of the bone marrow, approximately 40% of multiple myeloma is characterized by the presence of trisomies in the neoplastic plasma cells (trisomic multiple myeloma), while most of the rest have a translocation involving the immunoglobulin heavy chain (IgH) locus on chromosome 14q32 (IgH translocated multiple myeloma).33–36 A small proportion of patients have both trisomies and IgH translocations. Trisomies and IgH translocations are considered primary cytogenetic abnormalities and occur at the time of establishment of MGUS. In addition, other cytogenetic changes termed secondary cytogenetic abnormalities arise along the disease course of multiple myeloma, including gain(1q), del(1p), del(17p), del(13), RAS mutations, and secondary translocations involving MYC. Both primary and secondary cytogenetic abnormalities can influence disease course, response to therapy, and prognosis. Importantly, the interpretation and impact of cytogenetic abnormalities in multiple myeloma vary depending on the disease phase in which they are detected(Table 3).37
Table 2.
Primary Molecular Cytogenetic Classification of Multiple Myeloma
| Subtype | Gene(s)/chromosomes affected* | Percentage of myeloma patients |
|---|---|---|
| Trisomic multiple myeloma | Recurrent trisomies involving odd-numbered chromosomes with the exception of chromosomes 1, 13, and 21 | 42 |
| IgH translocated multiple myeloma | 30 | |
| t(11;14) (q13;q32) | CCND1 (cyclin D1) | 15 |
| t(4;14) (p16;q32) | FGFR-3 and MMSET | 6 |
| t(14;16) (q32;q23) | C-MAF | 4 |
| t(14;20) (q32;q11) | MAFB | <1 |
| Other IgH translocations* | CCND3 (cyclin D3) in t(6;14) multiple myeloma | 5 |
| Combined IgH translocated/trisomic multiple myeloma | Presence of trisomies and any one of the recurrent IgH translocations in the same patient | 15 |
| Isolated Monosomy 14 | Few cases may represent 14q32 translocations involving unknown partner chromosomes | 4.5 |
| Other cytogenetic abnormalities in absence of IgH translocations or trisomy or monosomy 14 | 5.5 | |
| Normal | 3 |
Modified from Kumar S et al. Trisomies in multiple myeloma: impact on survival in patients with high-risk cytogenetics. Blood 2012; 119:2100. © American Society of Hematology.
Includes the t(6;14)(p21;q32) translocation, and rarely, other IgH translocations involving uncommon partner chromosome
Table 3.
Cytogenetic Abnormalities on Clinical Course and Prognosis in Multiple Myeloma
| Cytogenetic Abnormality | Clinical Setting in which Abnormality is Detected | |
|---|---|---|
| Smoldering Multiple Myeloma | Multiple Myeloma | |
| Trisomies | Intermediate-risk of progression, median TTP of 3 years | Good prognosis, standard-risk MM, median OS 7–10 years Most have myeloma bone disease at diagnosis Excellent response to lenalidomide-based therapy |
| t(11;14) (q13;q32) | Standard-risk of progression, median TTP of 5 years | Good prognosis, standard-risk MM, median OS 7–10 years |
| t(6;14) (p21;q32) | Standard-risk of progression, median TTP of 5 years | Good prognosis, standard-risk MM, median OS 7–10 years |
| t(4;14) (p16;q32) | High-risk of progression, median TTP of 2 years | Intermediate-risk MM, median OS 5 years Needs bortezomib-based initial therapy, early ASCT (if eligible), followed by bortezomib-based consolidation/maintenance |
| t(14;16) (q32;q23) | Standard-risk of progression, median TTP of 5 years | High-risk MM, median OS 3 years Associated with high levels of FLC and 25% present with acute renal failure as initial MDE |
| t(14;20) (q32;q11) | Standard-risk of progression, median TTP of 5 years | High-risk MM, median OS 3 years |
| Gain(1q21) | High-risk of progression, median TTP of 2 years | Intermediate-risk MM, median OS 5 years |
| Del(17p) | High-risk of progression, median TTP of 2 years | High-risk MM, median OS 3 years |
| Trisomies plus any one of the IgH translocations | Standard-risk of progression, median TTP of 5 years | May ameliorate adverse prognosis conferred by high risk IgH translocations, and del 17p |
| Isolated Monosomy 13, or Isolated Monosomy 14 | Standard-risk of progression, median TTP of 5 years | Effect on prognosis is not clear |
| Normal | Low-risk of progression, median TTP of 7–10 years | Good prognosis, probably reflecting low tumor burden, median OS >7–10 years |
FISH, fluorescent in situ hybridization; TTP, time to progression; OS, overall survival; SMM, Smoldering multiple myeloma, MM, multiple myeloma; ASCT, autologous stem cell transplantation
Reproduced from Rajan AM, Rajkumar SV. Blood Cancer J. 2015;5: e365
PROGNOSIS AND RISK STRATIFICATION
Survival estimates in multiple myeloma vary based on the source of the data. Data from randomized controlled trials using modern therapy show that the median survival in multiple myeloma is approximately 6 years.38 In the subset of patients eligible for ASCT, 4-year survival rates are more than 80%;39 the median overall survival (OS) among these patients is approximately 8 years.40 Among elderly patients (age >75 years), median OS is lower, and is approximately 5 years.38 These numbers likely underestimate current survival probabilities since they predate the arrival of monoclonal antibodies and several other new agents that have been introduced in the last 3 to 5 years. On the other hand, they may be overestimates of the true population-based survival since they are derived from randomized controlled trials where patients with poor performance status and comorbidities are typically excluded. Nevertheless, these estimates are valuable benchmarks, and appear generalizable to newly diagnosed myeloma patients in good performance status.41
More precise estimation of prognosis requires an assessment of multiple factors. As in other cancers, OS in multiple myeloma is affected by host characteristics, tumor burden (stage), biology (cytogenetic abnormalities), and response to therapy.42,43 Tumor burden in multiple myeloma has traditionally been assessed using the Durie-Salmon Staging (DSS)44 and the International Staging System (ISS).45,46 Disease biology best reflected based on the molecular subtype of multiple myeloma (Table 2), the presence or absence of secondary cytogenetic abnormalities such as del(17p), gain(1q), or del(1p).25,32 In addition to cytogenetic risk factors, two other markers that are associated with aggressive disease biology are elevated serum lactate dehydrogenase and evidence of circulating plasma cells on routine peripheral smear examination (plasma cell leukemia). The Revised International Staging System (RISS) combines elements of tumor burden (ISS) and disease biology (presence of high risk cytogenetic abnormalities or elevated lactate dehydrogenase level) to create a unified prognostic index that and helps in clinical care as well as in comparison of clinical trial data (Table 4).47 In order to ensure uniform availability, only 3 widely available cytogenetic markers are used in the RISS; the Mayo Clinic mSMART risk stratification (www.msmart.org) (Table 5) has additional detail that is valuable in formulating a therapeutic strategy.48
Table 4.
Revised International Staging System for Myeloma47
| Stage |
|---|
| Stage 1 All of the following:
|
Stage II
|
| Stage III Both of the following:
|
Derived from: Palumbo A, et al. J Clin Oncol;2015;33:2863–2869.
Table 5.
Mayo Clinic Risk Stratification for Multiple Myeloma (mSMART)
| Risk Group | Percentage of newly diagnosed patients with the abnormality |
|---|---|
|
| |
| Standard Risk | 75% |
| Trisomies | |
| t(11;14) | |
| t(6;14) | |
| Intermediate Risk | 10% |
| t(4;14) | |
| Gain(1q) | |
| High Risk | 15% |
| t(14:16) | |
| t(14;20) | |
| del(17p) | |
Treated appropriately, the survival of patients with certain high risk categories can approach that of patients with standard risk disease. In a large trial using bortezomib-based induction, early ASCT, and bortezomib maintenance, the median OS of patients with del(17p) was approximately 8 years (8-year survival rate of 52%), and was identical to patients with standard risk multiple myeloma. In contrast, survival was lower for patients with t(4;14) translocation (8-year survival rate, 33%) and for patients with gain(1q) abnormality (8-year survival rate, 36%). These findings underscore the limitations of current risk stratification models in the context of modern therapy and highlight the need to stratify multiple myeloma based on individual cytogenetic groups rather than arbitrary heterogeneous risk categories.31
INDICATIONS FOR THERAPY
In order to initiate therapy, patients must meet criteria for multiple myeloma as outlined in Table 1. In earlier trials, treatment of asymptomatic patients with SMM was associated with a benefit in progression free survival (PFS) but not OS.49 However, a recent randomized trial found that early therapy with lenalidomide and dexamethasone in patients with high risk SMM can prolong PFS and OS.50 Although these results need further confirmation, they indicate the potential benefit of early intervention in selected asymptomatic patients.
TREATMENT OF NEWLY DIAGNOSED MYELOMA
Survival in multiple myeloma has improved significantly in the last 15 years.51 The initial impact came from the introduction of thalidomide,52 bortezomib,53 and lenalidomide.54,55 In the last 5 years, carfilzomib, pomalidomide, panobinostat, ixazomib, elotuzumab, and daratumumab have been approved by the Food and Drug Administration (FDA) for the treatment of relapsed multiple myeloma, and promise to improve outcomes further. Numerous combinations have been developed using drugs that have shown activity in multiple myeloma, and the most commonly used regimens are listed in Table 6.56–75 These drugs work through a variety of mechanisms, some of which are not fully understood. Thalidomide, lenalidomide, and pomalidomide are termed immunomodulatory agents (IMiDs). IMiDs bind to cereblon and activate cereblon E3 ligase activity, resulting in the rapid ubiquitination and degradation of two specific B cell transcription factors, Ikaros family zinc finger proteins Ikaros (IKZF 1) and Aiolos (IKZF3).76–78 They may cause direct cytotoxicity by inducing free radical mediated DNA damage.79 They also have anti-angiogenic, immunomodulatory, and tumor necrosis factor alpha inhibitory properties. Bortezomib, carfilzomib, and ixazomib are proteasome inhibitors.80–82 Elotuzumab and daratumumab are monoclonal antibodies targeting SLAMF7 and CD38 respectively.73,83,84 Panobinostat is a deacetylase inhibitor.75,85
Table 6.
Major Treatment Regimens in Multiple Myeloma
| Regimen | Usual Dosing Schedule* |
|---|---|
| Thalidomide-Dexamethasone (Td)**56,57 | Thalidomide 200 mg oral days 1–28 Dexamethasone 40 mg oral days 1, 8, 15, 22 Repeated every 4 weeks |
| Lenalidomide-Dexamethasone (Rd)58 | Lenalidomide 25 mg oral days 1–21 every 28 days Dexamethasone 40 mg oral days 1, 8, 15, 22 every 28 days Repeated every 4 weeks |
| Pomalidomide-Dexamethasone (Pom/Dex)59 | Pomalidomide 4 mg days 1–21 Dexamethasone 40 mg oral on days on days 1, 8, 15, 22 Repeated every 4 weeks |
| Bortezomib-Melphalan-Prednisone (VMP)**60–62 | Bortezomib 1.3 mg/m2 subcutaneous days 1, 8, 15, 22 Melphalan 9 mg/m2 oral days 1–4 Prednisone 60 mg/m2 oral days 1 to 4 Repeated every 35 days |
| Bortezomib-Thalidomide-Dexamethasone (VTd)**63 | Bortezomib 1.3 mg/m2 subcutaneous days 1, 8, 15, 22 Thalidomide 100–200 mg oral days 1–21 Dexamethasone 20 mg oral on day of and day after bortezomib (or 40 mg days 1, 8, 15, 22) Repeated every 4 weeks × 4 cycles as pre-transplant induction therapy |
| Bortezomib- Cyclophosphamide-Dexamethasone** (VCd or CyBord)64,65 | Cyclophosphamide 300 mg/m2 orally on days 1, 8, 15 and 22 Bortezomib 1.3 mg/m2 subcutaneous on days 1, 8, 15, 22 Dexamethasone 40 mg oral on days on days 1, 8, 15, 22 Repeated every 4 weeks† |
| Bortezomib-Lenalidomide-Dexamethasone (VRd)**65,66 | Bortezomib 1.3 mg/m2 subcutaneous days 1, 8, 15 Lenalidomide 25 mg oral days 1–14 Dexamethasone 20 mg oral on day of and day after bortezomib (or 40 mg days 1, 8, 15, 22) Repeated every 3 weeks‡ |
| Carfilzomib- Cyclophosphamide-Dexamethasone (KCd) ‡ ‡ 67 | Carfilzomib 20 mg/m2 (days 1 and 2 of Cycle 1) and 27 mg/m2 (subsequent doses) intravenously on days 1, 2, 8, 9, 15, 16 Cyclophosphamide 300 mg/m2 orally on days 1, 8, 15 Dexamethasone 40 mg oral on days on days 1, 8, 15, 22 Repeated every 4 weeks |
| Carfilzomib-Lenalidomide-Dexamethasone (KRd) ‡ ‡ 68 | Carfilzomib 20 mg/m2 (days 1 and 2 of Cycle 1) and 27 mg/m2 (subsequent doses) intravenously on days 1, 2, 8, 9, 15, 16 Lenalidomide 25 mg oral days 1–21 Dexamethasone 40 mg oral days 1, 8, 15, 22 Repeated every 4 weeks |
| Carfilzomib-Pomalidomide-Dexamethasone (KPd) ‡ ‡ 69 | Carfilzomib 20 mg/m2 (days 1 and 2 of Cycle 1) and 27 mg/m2 (subsequent cycles) intravenously on days 1, 2, 8, 9, 15, 16 Pomalidomide 4mg oral on days 1–21 Dexamethasone 40 mg oral on days on days 1, 8, 15, 22 Repeated every 4 weeks |
| Daratumumab-Lenalidomide-Dexamethasone (DRd)70 | Daratumumab 16 mg/kg intravenously weekly × 8 weeks, and then every 2 weeks for 4 months, and then once monthly Lenalidomide 25 mg oral days 1–21 Dexamethasone 40 mg intravenous days 1, 8, 15, 22 (given oral on days when no daratumumab is being administered) Lenalidomide-Dexamethasone repeated in usual schedule every 4weeks |
| Daratumumab-Bortezomib-Dexamethasone (DVd) **71 | Daratumumab 16 mg/kg intravenously weekly × 8 weeks, and then every 2 weeks for 4 months, and then once monthly Bortezomib 1.3 mg/m2 subcutaneous on days 1, 8, 15, 22 Dexamethasone 40 mg intravenous days 1, 8, 15, 22 (given oral on days when no daratumumab is being administered) Bortezomib-Dexamethasone repeated in usual schedule every 4 weeks |
| Daratumumab-Pomalidomide-Dexamethasone (DPd)72 | Daratumumab 16 mg/kg intravenously weekly × 8 weeks, and then every 2 weeks for 4 months, and then once monthly Pomalidomide 4 mg oral on days 1–21 Dexamethasone 40 mg intravenous days 1, 8, 15, 22 (given oral on days when no daratumumab is being administered) Repeated every 4 weeks |
| Elotuzumab-Lenalidomide-Dexamethasone (ERd)73 | 10 mg/kg intravenously weekly × 8 weeks, and then every 2 weeks Lenalidomide 25 mg oral days 1–21 Dexamethasone per prescribing information Lenalidomide-Dexamethasone repeated in usual schedule every 4weeks |
| Ixazomib-Lenalidomide-Dexamethasone (IRd)74 | Ixazomib 4 mg oral days 1, 8, 15 Lenalidomide 25 mg oral days 1–21 Dexamethasone 40 mg oral days 1, 8, 15, 22 Repeated every 4weeks |
| Panobinostat-Bortezomib**75 | Panobinostat 20 mg oral three times a week × 2 weeks Bortezomib 1.3 mg/m2 subcutaneous days 1, 8, 15 Repeated every 3 weeks |
All doses need to be adjusted for performance status, renal function, blood counts, and other toxicities
Doses of dexamethasone and/or bortezomib reduced based on other data showing lower toxicity and similar efficacy with reduced doses; subcutaneous route of administration of bortezomib preferred based on data showing lower toxicity and similar efficacy compared to intravenous administration
The day 22 dose of all 3 drugs is omitted if counts are low, or after initial response to improve tolerability, or when the regimen is used as maintenance therapy; When used as maintenance therapy for high risk patients, further delays can be instituted between cycles.
Omit day 15 dose if counts are low or when the regimen is used as maintenance therapy; When used as maintenance therapy for high risk patients, lenalidomide dose may be decreased to 10–15 mg per day, and delays can be instituted between cycles as done in total therapy protocols.
Carfilzomib can also considered in a once a week schedule of 70 mg/m2 on days 1, 8 and 15 every 28 days (cycle 1, day 1 should be 20 mg/m2); Day 8, 9 doses of carfilzomib can be omitted in maintenance phase of therapy after a good response to improve tolerability; KCd dosing lowered from that used in the initial trial which was conducted in newly diagnosed patients
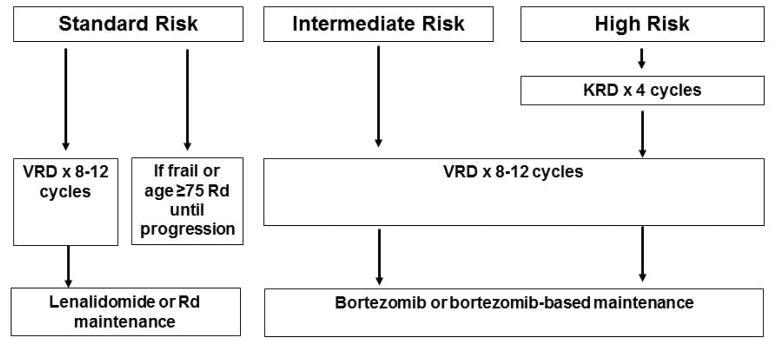
The approach to treatment of symptomatic newly diagnosed multiple myeloma is outlined in Figure 1 and is dictated by eligibility for ASCT and risk-stratification. The data to support their use from recent randomized trials using new active agents for multiple myeloma are provided in Table 7.38,39,60,86–88 There is an ongoing “cure versus control” debate on whether we should treat multiple myeloma with an aggressive multi-drug strategy targeting complete response (CR) or a sequential disease control approach that emphasizes quality of life as well as OS.89,90
Figure 1.
Approach to the treatment of newly diagnosed multiple myeloma in transplant eligible (A) and transplant ineligible (B) patients
Abbreviations: VRD, bortezomib, lenalidomide, dexamethasone; KRD, carfilzomib, lenalidomide, dexamethasone; Rd, lenalidomide plus dexamethasone; ASCT, autologous stem cell transplantation; CR, complete response; VGPR, very good partial response
Table 7.
Results of Recent Randomized Studies in Newly Diagnosed Myeloma
| Trial | Regimen | No. of patients | Overall response rate (%) | CR plus VGPR (%) | Progression-free survival (Median in months) | P value for progression free survival | Overall survival (Median in months)* | P value for overall survival |
|---|---|---|---|---|---|---|---|---|
| San Miguel et al; Mateos et al**60,86 | MP | 331 | 35 | 8 | 17 | 43 | ||
| VMP | 337 | 71 | 41 | 24 | <0.001 | NR | <0.001 | |
| Benboubker et al87 | MPT | 547 | 62 | 28 | 21 | <0.001 | 48 | 0.016† |
| Rd × 18 months | 541 | 73 | 43 | 21 | 53 | |||
| Rd till progression | 535 | 75 | 44 | 26 | 56 | |||
| Durie et al38 | Rd | 229 | 72 | 32 | 31 | 0.002 | 64 | 0.025 |
| VRd | 242 | 82 | 43 | 43 | 75 | |||
| Moreau et al88 | VCd | 169 | 83 | 56 | N/A | N/A | N/A | |
| VTd | 169 | 92 | 66 | N/A | N/A | N/A | ||
| Attal et al39 | VRd | 350 | 97 | 77 | 36 | NR; 82% at 4 years | 0.87 | |
| VRd-ASCT | 350 | 98 | 88 | 50 | <0.001 | NR; 81% at 4 years |
Estimated from survival curves when not reported
Progression free survival not reported, numbers indicate time to progression
Rd until progression versus MPT
Abbreviations: MP, melphalan plus prednisone; VMP, bortezomib plus melphalan plus prednisone; MPT, melphalan plus prednisone plus thalidomide; Rd, lenalidomide plus dexamethasone; VTd, bortezomib, thalidomide, dexamethasone; VRd, bortezomib, lenalidomide plus dexamethasone; VCd, bortezomib, cyclophosphamide, dexamethasone; N/A, not available; NS, not significant; CR, complete response; VGPR, very good partial response.
Recent data show that MRD negative status (as estimated by next generation molecular methods or flow cytometry) has favorable prognostic value.30 However, additional trials are needed to determine if changes in treatment need to be made based on MRD status. At present, MRD results are recommended mainly as a prognostic metric and not for used in making treatment decisions. We also need additional data to determine if MRD negativity can be used as a surrogate endpoint for regulatory approval, and if sustained MRD negativity may be a marker of cure in at least a subset of patients.91
Initial Treatment in Patients Eligible for ASCT
Typically, patients are treated with approximately 3–4 cycles of induction therapy prior to stem cell harvest. After harvest, patients can either undergo frontline ASCT or resume induction therapy delaying ASCT until first relapse. There are many options for initial therapy, and the most common treatment regimens are discussed below. These regimens can also be used at the time of relapse. In general, the low-dose dexamethasone regimen (40 mg once a week) is preferred in all regimens to minimize toxicity. In a randomized trial conducted by the Eastern Cooperative Oncology Group (ECOG), the low-dose dexamethasone approach was associated with superior OS and significantly lower toxicity.58
Bortezomib-containing regimens
Bortezomib, lenalidomide, dexamethasone (VRd) is the current standard of care for newly diagnosed multiple myeloma. In a recent randomized trial conducted by the Southwest Oncology Group (SWOG), response rates, PFS, and OS were significantly superior with VRd compared with Rd (Table 7).38 If lenalidomide is not available for use as initial therapy or in the presence of acute renal failure, other bortezomib-containing regimens such as bortezomib-thalidomide-dexamethasone (VTd) or bortezomib-cyclophosphamide-dexamethasone (VCd) can be used instead of VRd. A recent randomized trial found that VTd results in superior response rates compared with VCd, but impact on long-term outcomes is not known.88 Therefore both are reasonable alternatives to VRd.
In initial studies, peripheral neuropathy was a major concern with bortezomib therapy. Neuropathy with bortezomib can occur abruptly, and can be significantly painful and debilitating. However, the neurotoxicity of bortezomib can be greatly diminished by administering bortezomib once a week instead of twice-weekly,61,62 and by administering the drug subcutaneously instead of the intravenous route.92 The once-weekly subcutaneous bortezomib schedule (Table 6) has made serious neuropathy an uncommon problem, and has made regimens such as VRd, VCd, and VTd much more tolerable. Bortezomib does not appear to have any adverse effect on stem cell mobilization.93
Lenalidomide-low dose dexamethasone (Rd)
Rd which combines lenalidomide with a lower dose of dexamethasone (40 mg once weekly) is an active regimen in newly diagnosed multiple myeloma, and has less toxicity and better OS than lenalidomide plus high dose dexamethasone or MPT.58,87 Currently Rd is recommended mainly for patients who are unable to tolerate a triplet regimen due to advanced age, poor performance status, or comorbidities. Stem cell collection with granulocyte stimulating factor (G-CSF) alone may be impaired when Rd is used as induction therapy.94 Thus patients over the age of 65 and those who have received more than 4 cycles of Rd stem cells must be mobilized with either cyclophosphamide plus G-CSF or with plerixafor.95,96 All patients treated with Rd require anti-thrombosis prophylaxis. Aspirin is adequate for most patients, but in patients who are at higher risk of thrombosis, either low-molecular weight heparin or warfarin is needed.97–99
Carfilzomib-Lenalidomide-Dexamethasone (KRd)
Two phase II trials have reported excellent results with the newly approved proteasome inhibitor carfilzomib when used in combination with lenalidomide and dexamethasone for newly diagnosed multiple myeloma.100,101 However, more data on safety and efficacy of KRd are needed before this regimen can be recommended in newly diagnosed multiple myeloma, except in young patients with high risk cytogenetics. A randomized trial in the United States (referred to as the Endurance trial) is currently ongoing comparing VRd versus KRd as initial therapy.
Multi-drug combinations
Besides the regimens discussed above, other options include anthracycline-containing regimens such as bortezomib, doxorubicin, dexamethasone (PAD)40 or multi-agent combination chemotherapy regimens such as VDT-PACE (bortezomib, dexamethasone, thalidomide, cisplatin, doxorubicin, cyclophosphamide, and etoposide).102,103 These regimens are particularly useful in patients with aggressive disease such as plasma cell leukemia or multiple extramedullary plasmacytomas. Several other regimens have been tested in newly diagnosed multiple myeloma, but there are no clear data from randomized controlled trials that they have an effect on long-term endpoints compared with the regimens discussed earlier.
Recommendations
In standard-risk and intermediate-risk patients eligible for ASCT, I favor VRd as initial therapy for 3–4 cycles, followed by ASCT and lenalidomide maintenance therapy. In patients who are tolerating therapy and responding well, an alternative is VRd for 8 to 12 cycles followed by lenalidomide maintenance therapy; in such patients stem cells must be collected for cryopreservation after the first 3–4 cycles of VRd, and ASCT must be considered at first relapse.
In high-risk patients, I favor KRd as initial therapy for 3–4 cycles followed by ASCT and then maintenance with a proteasome inhibitor-based regimen.
In patients presenting with acute renal failure suspected to be secondary to light-chain cast nephropathy, I prefer VCd or VTd as initial therapy in conjunction with plasma exchange (or dialysis with high-cut-off filter). Plasma exchange is continued daily until the serum free light chain levels are less than 50 mg/dL and then repeated as needed till chemotherapy is fully effective.
In patients presenting with plasma cell leukemia or multiple extramedullary plasmacytomas, I prefer VDT-PACE as initial therapy followed by ASCT and then maintenance with a bortezomib-based regimen.
Once weekly subcutaneous bortezomib is preferred in most patients for initial therapy, unless there is felt to be an urgent need for rapid disease control.
Dexamethasone 40 mg once a week (low-dose dexamethasone) is preferred in most patients for initial therapy, unless there is felt to be an urgent need for rapid disease control.
Initial Treatment in Patients Not Eligible for ASCT
In patients with newly diagnosed multiple myeloma who are not candidates for ASCT due to age or other comorbidities, the major options for initial therapy are the same as those discussed earlier for patients eligible for ASCT. Typically treatment is given with a bortezomib-based regimen for approximately 8–12 cycles followed by maintenance. Although melphalan-based regimens have been extensively tested in these patients, they are not recommended due to concerns about stem cell damage and secondary myelodysplastic syndrome and leukemia. In the United States transplant eligibility is not determined by a strict age cut-off, and many patients enrolled in the melphalan-based clinical trials would be considered candidates for ASCT.
Bortezomib-based regimens
VRd has shown a survival benefit compared with Rd, and is the preferred choice for initial therapy in patients who are not candidates for ASCT (Table 7).38 VRd is administered for approximately 8–12 cycles, followed by maintenance therapy. Alternatives to VRd include VCd and VTd as discussed earlier. In patients in whom initial therapy with VRd is not possible mainly for logistical reasons (such as problems with compliance due to need for parenteral administration), ixazomib can be considered in place of bortezomib.
Lenalidomide plus dexamethasone (Rd)
Rd is an option for the treatment of elderly patients with newly diagnosed multiple myeloma who are unable to tolerate triplet therapy due to advanced age, poor performance status, or co-morbidities. An international phase III trial compared MPT versus Rd for 18 cycles versus Rd until progression in 1623 patients.87 PFS was superior with Rd until progression compared with the other two arms; OS was superior with Rd until progression compared with MPT. This trial provided the first evidence that OS can be improved in patients who are not eligible for transplant using a regimen that does not contain melphalan.
Melphalan-based regimens
VMP is a bortezomib-based regimen that has shown better OS compared with MP.60,86 Substituting melphalan with thalidomide in the VMP regimen has not shown a benefit.61 Melphalan-based regimens are considered only if there is problems with access to lenalidomide. Even in these situations, the risks of melphalan can be reduced by using cyclophosphamide instead, and studies show this substitution does not alter efficacy.104 Thus, the VCd regimen can be considered as a minor modification of the VMP regimen, in which cyclophosphamide is used as the alkylating agent in place of melphalan. This variation has the advantage of not affecting stem cell mobilization, and dosing is more predictable. A randomized trial found superior PFS and OS with a 4-drug regimen of VMPT compared with VMP in a randomized phase III trial, but the contribution of the fourth drug to the induction component cannot be ascertained from this trial.62
Recommendations
In standard-risk patients, I prefer VRd as initial therapy administered for approximately 8–12 cycles, followed by lenalidomide maintenance
In frail elderly patients, I prefer Rd as initial therapy, administered until progression. Dexamethasone may be started at 20 mg once a week, then reduced as much as possible after the first 4–6 cycles, and possibly discontinued after the first year.
In intermediate- and high-risk patients, I favor VRd as initial therapy for approximately 8–12 cycles followed if possible by a lower intensity (one dose every two weeks) maintenance schedule of bortezomib.
Hematopoietic Stem Cell Transplantation
Autologous stem cell transplantation (ASCT)
ASCT improves median OS in multiple myeloma by approximately 12 months.105–108 However, randomized trials found similar OS with early ASCT (immediately following 4 cycles of induction therapy) versus delayed ASCT (at the time of relapse as salvage therapy).109–111 A recent trial by the Intergroupe Francophone du Myelome (IFM) compared early versus delayed ASCT in patients treated with VRd followed by lenalidomide maintenance.39 Patients were randomized to receive either VRd (3 cycles) followed by ASCT and then VRd consolidation (2 cycles) versus VRd × 8 cycles with ASCT reserved for relapse. Both arms received lenalidomide maintenance for one year. A significant improvement in PFS was seen as expected with early ASCT, but this has so far not translated into a difference in OS (Table 7). Based on these results, it is reasonable to consider a delayed ASCT in patients with standard-risk multiple myeloma who prefer such an approach for personal and logistic reasons.
The role of tandem (double) ASCT is unclear. In earlier randomized trials, an improvement in OS was seen in two studies,112,113 but other studies failed to show such an improvement.114,115 More recent data are available from two other randomized trials are also inconclusive. In a trial conducted in Europe, an improvement in PFS and OS was seen with tandem ASCT in both standard and high risk patients.116 However, no survival benefit was seen in a randomized trial conducted in the United States by the Bone Marrow Transplantation Clinical Trials Network (BMT-CTN) in standard or high risk multiple myeloma (BMT-CTN 0702 trial).117 The US trial more likely reflects the impact of tandem ASCT in the context of modern therapy when most new options for salvage are available. Thus routine tandem ASCT is not recommended outside of a clinical trial setting except in selected young patients with high risk multiple myeloma.
Post-transplant Consolidation
Consolidation therapy is a term used for the administration of a short course of therapy, usually with 2 or more drugs, prior to the start of long-term maintenance. The BMT-CTN 0702 trial had an arm that investigated the benefit of post-transplant consolidation therapy followed by lenalidomide maintenance versus lenalidomide maintenance alone. In this trial, additional cycles of VRd chemotherapy administered as consolidation after ASCT did not result in significant benefit. Unlike earlier trials, the BMT-CTN 0702 trial specifically isolated the effect of consolidation and is therefore more compelling than trials where one could not ascertain the precise added value of consolidation therapy on PFS and OS. Consolidation therapy after ASCT is not recommended and patients should proceed to standard low-intensity maintenance therapy.
Allogeneic Transplantation
The role of allogeneic and non-myeloablative-allogeneic transplantation in multiple myeloma is controversial with studies showing conflicting results.118,119 The treatment related mortality (TRM) rate (10–20%) and GVHD rates are fairly high.120 Although allogenic transplantation should still be considered as investigational, it may be a consideration for young patients with high-risk disease who are willing to accept a high TRM and the unproven nature of this therapy for a chance at better long-term survival.
Recommendations
ASCT should be considered in all eligible patients. But in standard-risk patients responding well to therapy, ASCT can be delayed until first relapse provided stem cells are harvested early in the disease course.
Tandem ASCT is not recommended outside of clinical trials except in selected young patients with high risk multiple myeloma.
At present, allogeneic transplantation as frontline therapy should largely be considered investigational.
Maintenance Therapy
Maintenance therapy is indicated following ASCT. Maintenance therapy should also be considered in following completion of 8–12 cycles of initial therapy in patients treated without ASCT. Lenalidomide is the standard of care for maintenance therapy for most patients.87,121–125 In a meta-analysis of randomized trials, a significant improvement in PFS and OS was seen with lenalidomide maintenance compared with placebo or no therapy.126 Lenalidomide maintenance is associated with a 2–3 fold increase in the risk of second cancers and patients must be counseled in this regard and monitored.
The impact of lenalidomide maintenance in patients with intermediate- and high risk multiple myeloma is unclear. In the meta-analysis, no significant OS benefit was seen in these subsets of high risk patients.126 In contrast, bortezomib administered every other week has been shown to improve OS, particularly in patients with del(17p).124 Bortezomib-based maintenance may thus be preferable for intermediate- and high-risk patients.
The benefit of maintenance therapy in patients who did not undergo upfront ASCT is less clear. Based on the results of the SWOG trial, maintenance therapy with lenalidomide or Rd should be considered in patients who are in good performance status after completion of initial 8–12 cycles of triplet therapy. Patients who are treated with double therapy with Rd due to frailty or performance status are usually treated with this regimen until disease progression. After the first 18 cycles, the dose of dexamethasone can be lowered to minimize side effects. In some patients dexamethasone may need to be discontinued.
Although the benefit of maintenance is now established, data on optimal duration are lacking. We also need to consider the cost, toxicity, and inconvenience of long-term indefinite maintenance therapy. Many patients seek a drug-free interval. An ECOG trial is comparing lenalidomide maintenance given until progression versus a limited duration of 2 years. Trials are also examining if the duration of maintenance can be modified based on MRD results.
Recommendations
I recommend lenalidomide maintenance for standard-risk patients following ASCT. I also recommend lenalidomide or Rd maintenance following 8–12 cycles of initial therapy among patients who did not receive ASCT as part of initial therapy.
I recommend maintenance with bortezomib (or an alternative proteasome inhibitor) for patients with intermediate- and high-risk multiple myeloma
TREATMENT OF RELAPSED MULTIPLE MYELOMA
Almost all patients with multiple myeloma eventually relapse. The remission duration in relapsed multiple myeloma decreases with each regimen.127 The median PFS and OS in patients with relapsed multiple myeloma refractory to lenalidomide and bortezomib is poor, with median times of 5 months and 9 months, respectively.128 The choice of a treatment regimen at relapse is complicated and is affected by many factors including the timing of the relapse, response to prior therapy, aggressiveness of the relapse, and performance status (TRAP). Patients are eligible for an ASCT should be considered for the procedure if they have never had one before, or if they have had an excellent remission duration with the first ASCT defined as a remission of at least 36 months or longer with maintenance. In terms of drug therapy, a triplet regimen containing at least two new drugs that the patient is not refractory to should be considered. An approach to the treatment of relapsed multiple myeloma is given in Figure 2. Major regimens used in the treatment of multiple myeloma, including relapsed disease are listed in Table 6. Recent advances in the treatment of relapsed multiple myeloma, including new active agents and results of major randomized trials are discussed below (Table 8).68,70,71,73–75,129–132 One important consideration is that the lenalidomide-containing regimens listed in Table 8 were tested mainly in patient populations who were not previously exposed to lenalidomide. In contrast, current clinical practice typically consists of patients who have been treated with lenalidomide and are often relapsing while on a lenalidomide-containing regimen. In patients who are considered refractory to lenalidomide, one option is to consider pomalidomide-based regimens.
Figure 2.
Suggested options for the treatment of relapsed multiple myeloma in first relapse (A) and second or higher relapse (B)
Abbreviations: DRd, daratumumab, lenalidomide, dexamethasone; KRd, carfilzomib, lenalidomide, dexamethasone; Elo-Rd, Elotuzumab, lenalidomide, dexamethasone; IRd, ixazomib, lenalidomide, dexamethasone; DPd, daratumumab, pomalidomide, dexamethasone; DVd, daratumumab, bortezomib, dexamethasone; KPd, carfilzomib, pomalidomide, dexamethasone; VCD, bortezomib, cyclophosphamide, dexamethasone; Pd, pomalidomide, dexamethasone; ASCT, autologous stem cell transplantation
Table 8.
Results of Recent Randomized Studies in Relapsed Myeloma
| Trial | Regimen | No. of patients | Overall response rate (%) | CR plus VGPR (%) | Progression-free survival (Median in months) | P value for progression free survival | Overall survival* (Median in months) | P value for overall survival |
|---|---|---|---|---|---|---|---|---|
| Lonial et al73,154 | Rd | 325 | 66 | 28 | 15 | 40 | N/A | |
| Elo-Rd | 321 | 79 | 33 | 19 | <0.001 | 44 | 0.03 | |
| Stewart et al68,129 | Rd | 396 | 67 | 14 | 18 | 40 | 0.04 | |
| KRd | 396 | 87 | 32 | 26 | 0.0001 | 48 | ||
| Moreau et al74 | Rd | 362 | 72 | 7 | 15 | N/A | N/A | |
| IRd | 360 | 78 | 12 | 21 | 0.012 | N/A | ||
| Dimopoulos et al70 | Rd | 283 | 76 | 44 | 18.4 | <0.001 | N/A; 87% at 1 year | NS |
| DRd | 286 | 93 | 76 | NR | N/A; 92% at 1 year | |||
| Palumbo et al71 | Vd | 247 | 63 | 29 | 7.2 | <0.001 | N/A; 70% at 1 year | 0.30 |
| DVd | 251 | 83 | 59 | NR | N/A; 80% at 1 year | |||
| San Miguel et al75,155 | Vd | 381 | 55 | 6 | 8.1 | 36 | 0.54 | |
| Pano-Vd | 387 | 61 | 11 | 12 | <0.0001 | 40 | ||
| San Miguel et al130 | Dex | 153 | 10 | 0 | 1.9 | 8 | NS | |
| Pd | 302 | 31 | 1 | 4.0 | <0.0001 | 13 | ||
| Dimopoulos et al131,132 | Vd | 465 | 63 | 6 | 9 | 40 | 0.01 | |
| Kd | 464 | 77 | 13 | 19 | <0.0001 | 48 |
Estimated from updated publication when available; estimated from survival curves when not reported
Abbreviations: Elo-Rd, Elotuzumab, lenalidomide, dexamethasone; KRd, carfilzomib, lenalidomide, dexamethasone; Rd, lenalidomide plus dexamethasone; Kd, carfilzomib, dexamethasone; IRd, ixazomib, lenalidomide, dexamethasone; DRd, daratumumab, lenalidomide, dexamethasone; DVd, daratumumab, bortezomib, dexamethasone; Vd, bortezomib, dexamethasone; Pano-Vd, Panobinostat, bortezomib, dexamethasone; Pd, pomalidomide, dexamethasone; Dex, high dose dexamethasone; Kd, carfilzomib, dexamethasone; N/A, not available; NS, not significant; CR, complete response.
Bortezomib-based regimens
These regimens are appropriate for patients who received a bortezomib-based triplet for a period of time, and then stopped therapy. In these patients if relapse occurs after a reasonable period of remission off all therapy, then restarting the same (or similar) bortezomib-based triplet is reasonable and also carries lower cost and risk. As in newly diagnosed multiple myeloma, VRd, VCd, and VTd are active regimens in relapsed disease.133,134
Daratumumab
Daratumumab targeting CD38 has shown promise in relapsed, refractory multiple myeloma.83 In a phase II trial, daratumumab as a single-agent was produced a response rate of approximately 30% in heavily pre-treated patients.84 Based on these findings, daratumumab was first granted accelerated approval by the FDA in 2015 for the treatment of patients with multiple myeloma who have received at least three prior lines of therapy including a proteasome inhibitor and an immunomodulatory agent, or who are double-refractory to a proteasome inhibitor and an immunomodulatory agent. Subsequently 3 other daratumumab-based combinations have shown efficacy and have been approved by the FDA. These include daratumumab, lenalidomide, dexamethasone (DRd), daratumumab, bortezomib, dexamethasone (DVd), and daratumumab, pomalidomide, dexamethasone (DPd)(Table 8). The various triplets available for use in relapsed disease have not been compared head-to-head, daratumumab-based regimens appear to have the greatest reduction in risk of progression, and may be preferred for first relapse subject to availability and cost considerations.135
Carfilzomib
Carfilzomib is a novel keto-epoxide tetrapeptide proteasome inhibitor initially approved in 2013 for the treatment of relapsed refractory multiple myeloma in patients who have been previously treated with lenalidomide and bortezomib. In a phase 2 study (PX-171-003-A1), of 266 patients (80% of patients whom were refractory or intolerant to both bortezomib and lenalidomide), single-agent carfilzomib resulted in a response rate of 24% for a median duration of approximately 8 months.136 KRd has been subsequently shown to be effective in a randomized trial, and is a major option for the treatment of relapsed disease (Table 8).68 In another randomized trial carfilzomib plus dexamethasone was associated with an improvement in PFS and OS compared with bortezomib plus dexamethasone in relapsed multiple myeloma.131,132 However, the dose of carfilzomib used in this trial (56mg/m2) is twice the standard dose, and carries a much higher cost compared with bortezomib. Carfilzomib is typically administered twice-weekly at a dose of 27 mg/m2 (refer to Table 6), but a once-weekly schedule of 70 mg/m2 may be equally effective and safe, and more convenient. Carfilzomib carries a lower risk of neurotoxicity than bortezomib, but a small proportion (5%) of patients can experience serious cardiac side effects. Carfilzomib-based regimens are important options at relapse, and can work well even in patients who are refractory to a bortezomib-containing regimen.
Pomalidomide
Pomalidomide is an analog of lenalidomide and thalidomide initially approved in 2013 for the treatment of relapsed refractory multiple myeloma. It has significant activity in relapsed refractory multiple myeloma, even in patients failing lenalidomide.137,138 Response rate with pomalidomide plus dexamethasone (Pd) in patients refractory to lenalidomide and bortezomib is approximately 30%.59,139 In a randomized trial, Pd was found superior to high-dose dexamethasone in patients refractory to other forms of therapy for multiple myeloma (Table 8).130 Pomalidomide-containing triplet regimens such as daratumumab, pomalidomide, dexamethasone (DPd) and carfilzomib, pomalidomide, dexamethasone (KPd) are active and are important options at relapse for patients who are considered lenalidomide-refractory. In frail patients and in those with indolent relapse, the doublet regimen of Pd is a reasonable option for patients with indolent relapse.
Elotuzumab
Elotuzumab, a monoclonal antibody targeting the signaling lymphocytic activation molecule F7 (SLAMF7).73 Unlike daratumumab, elotuzumab does not have single-agent activity but shows synergistic activity when combined with Rd. In a phase III trial of 646 patients, elotuzumab, lenalidomide, dexamethasone (ERd) was superior to Rd (Table 8).73 Elotuzumab is well tolerated, and was initially approved in 2015 by the FDA to be given in combination with Rd for the treatment of patients with multiple myeloma who have received one to three prior therapies.
Ixazomib
Ixazomib is an oral proteasome inhibitor that is active in both the relapsed refractory setting and in newly diagnosed multiple myeloma.74,140 It has the advantage of once-weekly oral administration. Compared with bortezomib it has more gastrointestinal adverse events, but lower risk of neurotoxicity. In a randomized controlled trial in relapsed multiple myeloma, ixazomib, lenalidomide, dexamethasone (IRd) was found to improve PFS compared with Rd (Table 8).74 Based on these results ixazomib was initially approved by the FDA in 2015 to be given in combination with Rd for the treatment of patients with multiple myeloma who have received at least one prior therapy.
Doxorubicin and Liposomal Doxorubicin
Anthracyclines have marginal single-agent activity in multiple myeloma. A phase III randomized trial found that median time to progression (TTP) was superior with bortezomib plus pegylated liposomal doxorubicin compared with bortezomib alone, 9.3 months versus 6.5 months, respectively, P<0.001.141 OS at 15 months was also superior, 76% compared with 65%, respectively, P = 0.03. Despite this study, liposomal doxorubicin is infrequently used in the treatment of relapsed multiple myeloma given availability of other active agents. Doxorubicin-containing regimens such as PAD or VDT-PACE may be useful in the treatment of patients with aggressive multiple myeloma refractory to other standard myeloma agents.
Panobinostat
Panobinostat is a pan-deacetylase inhibitor initially approved by the FDA in 2015 for the treatment of patients with multiple myeloma who have received at least two prior standard therapies, including bortezomib and an immunomodulatory agent.75 Its putative mechanism of action is blockade of the aggresome pathway, an alternative route for cells to bypass the lethal effects of proteasome inhibition. By combining bortezomib and panobinostat, there is simultaneous blockade of both proteasome and aggresome pathways.142,143 In a randomized trial of 768 patients, bortezomib/dexamethasone plus panobinostat was associated with superior PFS compared with bortezomib/dexamethasone plus placebo.75 However, panobinostat therapy is associated with grade 3 diarrhea in approximately 25% of patients, and care should be exercised when using this drug. I recommend a lower initial dose of panobinostat than the approved starting dose, and that bortezomib be used in the once-weekly subcutaneous schedule rather than the twice weekly regimen used in the pivotal trial (Table 6)
Other Options
Despite the multiple option available, more patients eventually become refractory to all drug classes. Some additional options to consider for relapsed disease in refractory multiple myeloma include bendamustine-containing regimens such as bendamustine, lenalidomide, dexamethasone or bendamustine, bortezomib, dexamethasone. 144,145 Other options include the addition of panobinostat to a proteasome-inhibitor containing regimen, or the use of quadruplet regimen in which daratumumab is added to a standard triplet regimen. Venetoclax is not approved for use in multiple myeloma, but is commercially available, and appears to have single-agent activity in patients with t(11;14) subtype of multiple myeloma.146 For young high-risk patients with a suitable donor, allogeneic transplantation is an option as well.
Emerging Options
There are several investigational approaches that are promising and patients should be considered for clinical trials investigating these approaches. Two of the most exciting options include antigen receptor T cells (CAR-T) targeting B cell maturation antigen (BCMA) such as bb2121,147 and GSK2857916 (a humanized anti-BCMA antibody that is conjugated to monomethyl auristatin-F, a microtubule disrupting agent).148 Other agents with single-agent activity that are promising include isatuximab (a CD38 monoclonal antibody), marizomib (a new proteasome inhibitor), oprozomib, (an oral proteasome inhibitor related to carfilzomib), filanesib (a kinesin spindle protein inhibitor), dinaciclib (a cyclin dependent kinase inhibitor), and LGH-447 (a pan PIM kinase inhibitor).
Recommendations
Patients who are eligible for ASCT should consider ASCT as salvage therapy at first relapse if they have never had a transplant before, or if they have had a prolonged remission with the first ASCT.
If relapse occurs more than 6 months after stopping therapy, the initial treatment regimen that successfully controlled the multiple myeloma initially can be re-instituted when possible.
At first relapse, for patients who are not refractory to lenalidomide, my preferred option is DRd. Alternatives include KRd, IRd, and ERd.
At first relapse, for patients who are refractory to lenalidomide, my preferred option is DPd. Alternatives include DVd, KPd, and VCd
Patients who have an indolent relapse or who are frail can be treated with oral regimens such as IRd or Pd.
At second or higher relapse, I switch to a triplet regimen that contains at least 2 new drugs that the patient is not refractory to.
Additional options to consider in patients with multiple relapses and disease that is refractory to conventional regimens include bendamustine-based regimens, the addition of panobinostat to a proteasome-inhibitor containing regimen, quadruplet daratumumab-containing regimens, multi-drug chemotherapy regimens, allogenic transplantation in young high risk patients with a suitable donor, and venetoclax in patients with t(11;14) multiple myeloma.
Patients with more aggressive relapse with plasma cell leukemia or extramedullary plasmacytomas often require therapy with a multi-drug anthracycline containing regimen such as VDT-PACE.
The duration of therapy has not been well addressed in relapsed multiple myeloma, and in some regimens such as those employing parenteral proteasome inhibitors it may be reasonable to stop therapy once a stable plateau has been reached in order to limit minimize risks of serious toxicity.
SMOLDERING MULTIPLE MYELOMA
SMM is a stage that is clinically positioned between MGUS and multiple myeloma.149 It comprises of a heterogeneous group of patients, some of whom have multiple myeloma which has not yet manifested with MDEs, and some who have premalignant MGUS. Patients with SMM have a risk of progression of approximately 10% per year for the first 5 years, 3% per year for the next 5 years, and 1% per year thereafter.19 Patients with the highest risk of progression (ultra-high risk) have now been reclassified as having multiple myeloma by the new IMWG criteria.1 Within the current definition of SMM (Table 1), there are two groups of patients: high risk (25% per year risk of progression in the first 2 years) and low risk (~ 5% per year risk of progression).149 Criteria for high risk SMM are given on Table 9. Presence of one or more of these factors is associated with a median TTP to multiple myeloma of approximately 2 years. Early studies in SMM failed to show an advantage to early intervention, but were limited by lack of power, safe and effective drugs, and a risk-adapted strategy.150,151 A recent randomized trial conducted in Spain found that patients with high risk SMM had significant prolongation of PFS and OS with Rd compared with observation.50,152 These are very promising results, and further confirmatory studies are ongoing. Observation is still the standard of care for SMM; however, selected high risk SMM patients with multiple risk factors can be considered for therapy. They are also candidates for clinical trials testing early intervention, some of which are testing intensive therapy with curative intent.153
Table 9.
Criteria for High Risk Smoldering Multiple Myeloma*
| Bone marrow clonal plasma cells ≥10% and any one or more of the following: |
|---|
| Serum M protein ≥30g/L |
| IgA SMM |
| Immunoparesis with reduction of two uninvolved immunoglobulin isotypes |
| Serum involved/uninvolved free light chain ratio ≥8 (but less than 100) |
| Progressive increase in M protein level (Evolving type of SMM)† |
| Bone marrow clonal plasma cells 50–60% |
| Abnormal plasma cell immunophenotype (≥95% of bone marrow plasma cells are clonal) and reduction of one or more uninvolved immunoglobulin isotypes |
| t (4;14) or del 17p or 1q gain |
| Increased circulating plasma cells |
| MRI with diffuse abnormalities or 1 focal lesion |
| PET-CT with focal lesion with increased uptake without underlying osteolytic bone destruction |
SMM, smoldering multiple myeloma; M, monoclonal; MRI, magnetic resonance imaging; PET-CT, positron emission tomography-computed tomography
Note that the term smoldering multiple myeloma excludes patients without end-organ damage who meet revised definition of multiple myeloma, namely clonal bone marrow plasma cells ≥60% or serum free light chain (FLC) ratio ≥100 (plus measurable involved FLC level ≥100 mg/L), or more than one focal lesion on magnetic resonance imaging. The risk factors listed in this Table are not meant to be indications for therapy; they are variables associated with a high risk of progression of SMM, and identify patients who need close follow up and consideration for clinical trials
Increase in serum monoclonal protein by ≥25% on two successive evaluations within a 6 month period
Reproduced from: Rajkumar SV, Landgren O, Mateos MV. Smoldering Multiple Myeloma. Blood. 2015 Apr 2. pii: blood-2014-09-568899 © American Society of Hematology.
Recommendations
I recommend observation for most patients with SMM.
Consideration of multiple myeloma therapy can be given to the small subset of patients with SMM who have multiple high risk factors especially if there is progressive rise in monoclonal protein levels.
Acknowledgments
Supported in part by grants CA 107476, CA 168762, and CA186781 from the National Cancer Institute, Rockville, MD, USA.
Footnotes
Conflict of interest: No conflicts of interest to be disclosed.
Authorship Contribution Statement
SVR conceived of the paper, researched the literature, and wrote the manuscript.
Disclosure of Conflicts of Interest
SVR declares no conflict of interest.
References
- 1.Rajkumar SV, Dimopoulos MA, Palumbo A, et al. International Myeloma Working Group Updated Criteria for the Diagnosis of Multiple Myeloma. Lancet Oncol. 2014;15:e538–48. doi: 10.1016/S1470-2045(14)70442-5. [DOI] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68:7–30. doi: 10.3322/caac.21442. [DOI] [PubMed] [Google Scholar]
- 3.Kyle RA, Therneau TM, Rajkumar SV, Larson DR, Plevak MF, Melton LJ., 3rd Incidence of multiple myeloma in Olmsted County, Minnesota: Trend over 6 decades. Cancer. 2004;101:2667–74. doi: 10.1002/cncr.20652. [DOI] [PubMed] [Google Scholar]
- 4.Landgren O, Weiss BM. Patterns of monoclonal gammopathy of undetermined significance and multiple myeloma in various ethnic/racial groups: support for genetic factors in pathogenesis. Leukemia. 2009;23:1691–7. doi: 10.1038/leu.2009.134. [DOI] [PubMed] [Google Scholar]
- 5.Kyle RA, Gertz MA, Witzig TE, et al. Review of 1,027 patients with newly diagnosed multiple myeloma. Mayo Clinic Proc. 2003;78:21–33. doi: 10.4065/78.1.21. [DOI] [PubMed] [Google Scholar]
- 6.Roodman GD. Pathogenesis of myeloma bone disease. Leukemia. 2009;23:435–41. doi: 10.1038/leu.2008.336. [DOI] [PubMed] [Google Scholar]
- 7.Regelink JC, Minnema MC, Terpos E, et al. Comparison of modern and conventional imaging techniques in establishing multiple myeloma-related bone disease: a systematic review. British journal of haematology. 2013;162:50–61. doi: 10.1111/bjh.12346. [DOI] [PubMed] [Google Scholar]
- 8.Hillengass J, Moulopoulos LA, Delorme S, et al. Whole-body computed tomography versus conventional skeletal survey in patients with multiple myeloma: a study of the International Myeloma Working Group. Blood Cancer J. 2017;7:e599. doi: 10.1038/bcj.2017.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Short KD, Rajkumar SV, Larson D, et al. Incidence of extramedullary disease in patients with multiple myeloma in the era of novel therapy, and the activity of pomalidomide on extramedullary myeloma. Leukemia. 2011;25:906–8. doi: 10.1038/leu.2011.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Landgren O, Kyle RA, Pfeiffer RM, et al. Monoclonal gammopathy of undetermined significance (MGUS) consistently precedes multiple myeloma: a prospective study. Blood. 2009;113:5412–7. doi: 10.1182/blood-2008-12-194241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Weiss BM, Abadie J, Verma P, Howard RS, Kuehl WM. A monoclonal gammopathy precedes multiple myeloma in most patients. Blood. 2009;113:5418–22. doi: 10.1182/blood-2008-12-195008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kyle RA, Therneau TM, Rajkumar SV, et al. Prevalence of Monoclonal Gammopathy of Undetermined Significance. N Engl J Med. 2006;354:1362–9. doi: 10.1056/NEJMoa054494. [DOI] [PubMed] [Google Scholar]
- 13.Dispenzieri A, Katzmann JA, Kyle RA, et al. Prevalence and risk of progression of light-chain monoclonal gammopathy of undetermined significance: a retrospective population-based cohort study. Lancet. 2010;375:1721–8. doi: 10.1016/S0140-6736(10)60482-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Landgren O, Graubard BI, Katzmann JA, et al. Racial disparities in the prevalence of monoclonal gammopathies: a population-based study of 12 482 persons from the national health and nutritional examination survey. Leukemia. 2014;28:1537–42. doi: 10.1038/leu.2014.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Landgren O, Graubard BI, Kumar S, et al. Prevalence of myeloma precursor state monoclonal gammopathy of undetermined significance (MGUS) in 12,309 individuals 10 to 49 years old: a population-based study from the National Health and Nutritional Examination Survey. Blood Cancer J. 2017 doi: 10.1038/bcj.2017.97. In Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kyle RA, Therneau TM, Rajkumar SV, et al. A long-term study of prognosis of monoclonal gammopathy of undetermined significance. N Engl J Med. 2002;346:564–9. doi: 10.1056/NEJMoa01133202. [DOI] [PubMed] [Google Scholar]
- 17.Kyle RA, Larson DR, Therneau TM, et al. Long-Term Follow-up of Monoclonal Gammopathy of Undetermined Significance. N Engl J Med. 2018;378:241–9. doi: 10.1056/NEJMoa1709974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Therneau TM, Kyle RA, Melton LJ, III, et al. Incidence of monoclonal gammopathy of undetermined significance and estimation of duration before first clinical recognition. Mayo Clin Proc. 2012;87:1071–9. doi: 10.1016/j.mayocp.2012.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kyle RA, Remstein ED, Therneau TM, et al. Clinical Course and Prognosis of Smoldering (Asymptomatic) Multiple Myeloma. N Engl J Med. 2007;356:2582–90. doi: 10.1056/NEJMoa070389. [DOI] [PubMed] [Google Scholar]
- 20.Rajkumar SV, Gupta V, Fonseca R, et al. Impact of primary molecular cytogenetic abnormalities and risk of progression in smoldering multiple myeloma. Leukemia. 2013;27:1738–44. doi: 10.1038/leu.2013.86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Neben K, Jauch A, Hielscher T, et al. Progression in smoldering myeloma is independently determined by the chromosomal abnormalities del(17p), t(4;14), gain 1q, hyperdiploidy, and tumor load. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2013;31:4325–32. doi: 10.1200/JCO.2012.48.4923. [DOI] [PubMed] [Google Scholar]
- 22.Lakshman A, Paul S, Rajkumar SV, et al. Prognostic significance of interphase FISH in monoclonal gammopathy of undetermined significance. Leukemia. 2018 doi: 10.1038/s41375-018-0030-3. [DOI] [PubMed] [Google Scholar]
- 23.Katzmann JA, Dispenzieri A, Kyle R, et al. Elimination of the Need for Urine Studies in the Screening Algorithm for Monoclonal Gammopathies by Using Serum Immunofixation and Free Light Chain Assays. Mayo Clin Proc. 2006;81:1575–8. doi: 10.4065/81.12.1575. [DOI] [PubMed] [Google Scholar]
- 24.Chawla SS, Kumar SK, Dispenzieri A, et al. Clinical course and prognosis of non-secretory multiple myeloma. European journal of haematology. 2014 doi: 10.1111/ejh.12478. [DOI] [PubMed] [Google Scholar]
- 25.Kumar SK, Mikhael JR, Buadi FK, et al. Management of Newly Diagnosed Symptomatic Multiple Myeloma: updated Mayo Stratification of Myeloma and Risk-Adapted Therapy (mSMART) Consensus Guidelines. Mayo Clinic Proceedings. 2009;84:1095–110. doi: 10.4065/mcp.2009.0603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhou Y, Barlogie B, Shaughnessy JD., Jr The molecular characterization and clinical management of multiple myeloma in the post-genome era. Leukemia. 2009;23:1941–56. doi: 10.1038/leu.2009.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dizdar O, Barista I, Kalyoncu U, et al. Biochemical markers of bone turnover in diagnosis of myeloma bone disease. Am J Hematol. 2007;82:185–91. doi: 10.1002/ajh.20794. [DOI] [PubMed] [Google Scholar]
- 28.Silvestris F, Lombardi L, De Matteo M, Bruno A, Dammacco F. Myeloma bone disease: pathogenetic mechanisms and clinical assessment. Leuk Res. 2007;31:129–38. doi: 10.1016/j.leukres.2006.04.014. [DOI] [PubMed] [Google Scholar]
- 29.Dispenzieri A, Kyle R, Merlini G, et al. International Myeloma Working Group guidelines for serum-free light chain analysis in multiple myeloma and related disorders. Leukemia. 2009;23:215–24. doi: 10.1038/leu.2008.307. [DOI] [PubMed] [Google Scholar]
- 30.Kumar S, Paiva B, Anderson KC, et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. The Lancet Oncology. 2016;17:e328–46. doi: 10.1016/S1470-2045(16)30206-6. [DOI] [PubMed] [Google Scholar]
- 31.Kumar S, Rajkumar SV. The multiple myelomas — current concepts in cytogenetic classification and therapy. Nat Rev Clin Oncol. 2018 doi: 10.1038/s41571-018-0018-y. In Press. [DOI] [PubMed] [Google Scholar]
- 32.Kumar S, Fonseca R, Ketterling RP, et al. Trisomies in multiple myeloma: impact on survival in patients with high-risk cytogenetics. Blood. 2012;119:2100–5. doi: 10.1182/blood-2011-11-390658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kuehl WM, Bergsagel PL. Multiple myeloma: evolving genetic events and host interactions. Nature Reviews Cancer. 2002;2:175–87. doi: 10.1038/nrc746. [DOI] [PubMed] [Google Scholar]
- 34.Bergsagel PL, Kuehl WM. Chromosome translocations in multiple myeloma. Oncogene. 2001;20:5611–22. doi: 10.1038/sj.onc.1204641. [DOI] [PubMed] [Google Scholar]
- 35.Fonseca R, Bailey RJ, Ahmann GJ, et al. Genomic abnormalities in monoclonal gammopathy of undetermined significance. Blood. 2002;100:1417–24. [PubMed] [Google Scholar]
- 36.Seidl S, Kaufmann H, Drach J. New insights into the pathophysiology of multiple myeloma. Lancet Oncology. 2003;4:557–64. doi: 10.1016/s1470-2045(03)01195-1. [DOI] [PubMed] [Google Scholar]
- 37.Rajan AM, Rajkumar SV. Interpretation of cytogenetic results in multiple myeloma for clinical practice. Blood Cancer J. 2015;5:e365. doi: 10.1038/bcj.2015.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Durie BGM, Hoering A, Abidi MH, et al. Bortezomib, Lenalidomide and Dexamethasone vs. Lenalidomide and Dexamethasone Induction Followed by Lenalidomide and Dexamethasone Maintenance in Patients with Newly Diagnosed Myeloma without Intent for Immediate Autologous Stem Cell Transplant: Results of the Randomised Phase III SWOG Trial S0777. Lancet. 2017;389:519–27. doi: 10.1016/S0140-6736(16)31594-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Attal M, Lauwers-Cances V, Hulin C, et al. Lenalidomide, Bortezomib, and Dexamethasone with Transplantation for Myeloma. N Engl J Med. 2017;376:1311–20. doi: 10.1056/NEJMoa1611750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Goldschmidt H, Lokhorst HM, Mai EK, et al. Bortezomib before and after high-dose therapy in myeloma: long-term results from the phase III HOVON-65/GMMG-HD4 trial. Leukemia. 2018;32:383–90. doi: 10.1038/leu.2017.211. [DOI] [PubMed] [Google Scholar]
- 41.Paquin A, Kumar SK, Buadi FK, et al. Overall Survival of Transplant Eligible Patients with Newly Diagnosed Multiple Myeloma: Comparative Effectiveness Analysis of Modern Induction Regimens on Outcome. Blood. 2017;130:3138. doi: 10.1038/s41408-018-0163-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Russell SJ, Rajkumar SV. Multiple myeloma and the road to personalised medicine. Lancet Oncol. 2011;12:617–9. doi: 10.1016/S1470-2045(11)70143-7. [DOI] [PubMed] [Google Scholar]
- 43.Vu T, Gonsalves W, Kumar S, et al. Characteristics of exceptional responders to lenalidomide-based therapy in multiple myeloma. Blood Cancer J. 2015;5:e363. doi: 10.1038/bcj.2015.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Durie BG, Salmon SE. A clinical staging system for multiple myeloma. Correlation of measured myeloma cell mass with presenting clinical features, response to treatment, and survival. Cancer. 1975;36:842–54. doi: 10.1002/1097-0142(197509)36:3<842::aid-cncr2820360303>3.0.co;2-u. [DOI] [PubMed] [Google Scholar]
- 45.Greipp PR, San Miguel JF, Durie BG, et al. International Staging System for Multiple Myeloma. J Clin Oncol. 2005;23:3412–20. doi: 10.1200/JCO.2005.04.242. [DOI] [PubMed] [Google Scholar]
- 46.Hari PN, Zhang MJ, Roy V, et al. Is the international staging system superior to the Durie-Salmon staging system? A comparison in multiple myeloma patients undergoing autologous transplant. Leukemia. 2009;23:1528–34. doi: 10.1038/leu.2009.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Palumbo A, Avet-Loiseau H, Oliva S, et al. Revised International Staging System for Multiple Myeloma: A Report From International Myeloma Working Group. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2015;33:2863–9. doi: 10.1200/JCO.2015.61.2267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mikhael JR, Dingli D, Roy V, et al. Management of Newly Diagnosed Symptomatic Multiple Myeloma: Updated Mayo Stratification of Myeloma and Risk-Adapted Therapy (mSMART) Consensus Guidelines 2013. Mayo Clin Proc. 2013;88:360–76. doi: 10.1016/j.mayocp.2013.01.019. [DOI] [PubMed] [Google Scholar]
- 49.Witzig TE, Laumann KM, Lacy MQ, et al. A phase III randomized trial of thalidomide plus zoledronic acid versus zoledronic acid alone in patients with asymptomatic multiple myeloma. Leukemia. 2013;27:220–5. doi: 10.1038/leu.2012.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mateos M-V, Hernández M-T, Giraldo P, et al. Lenalidomide plus Dexamethasone for High-Risk Smoldering Multiple Myeloma. N Engl J Med. 2013;369:438–47. doi: 10.1056/NEJMoa1300439. [DOI] [PubMed] [Google Scholar]
- 51.Kumar SK, Dispenzieri A, Lacy MQ, et al. Continued improvement in survival in multiple myeloma: changes in early mortality and outcomes in older patients. Leukemia. 2014;28:1122–8. doi: 10.1038/leu.2013.313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Singhal S, Mehta J, Desikan R, et al. Antitumor activity of thalidomide in refractory multiple myeloma [see comments] N Engl J Med. 1999;341:1565–71. doi: 10.1056/NEJM199911183412102. [DOI] [PubMed] [Google Scholar]
- 53.Richardson PG, Sonneveld P, Schuster MW, et al. Bortezomib or high-dose dexamethasone for relapsed multiple myeloma. [see comment] N Engl J Med. 2005;352:2487–98. doi: 10.1056/NEJMoa043445. [DOI] [PubMed] [Google Scholar]
- 54.Rajkumar SV, Hayman SR, Lacy MQ, et al. Combination therapy with lenalidomide plus dexamethasone (Rev/Dex) for newly diagnosed myeloma. Blood. 2005;106:4050–3. doi: 10.1182/blood-2005-07-2817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Richardson PG, Blood E, Mitsiades CS, et al. A randomized phase 2 study of lenalidomide therapy for patients with relapsed or relapsed and refractory multiple myeloma. Blood. 2006;108:3458–64. doi: 10.1182/blood-2006-04-015909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Rajkumar SV, Blood E, Vesole DH, Fonseca R, Greipp PR. Phase III Clinical Trial of Thalidomide Plus Dexamethasone Compared With Dexamethasone Alone in Newly Diagnosed Multiple Myeloma: A Clinical Trial Coordinated by the Eastern Cooperative Oncology Group. J Clin Oncol. 2006;24:431–6. doi: 10.1200/JCO.2005.03.0221. [DOI] [PubMed] [Google Scholar]
- 57.Rajkumar SV, Rosiñol L, Hussein M, et al. A Multicenter, Randomized, Double-Blind, Placebo-Controlled Study of Thalidomide plus Dexamethasone Versus Dexamethasone as Initial Therapy for Newly Diagnosed Multiple Myeloma. J Clin Oncol. 2008;26:2171–7. doi: 10.1200/JCO.2007.14.1853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Rajkumar SV, Jacobus S, Callander NS, et al. Lenalidomide plus high-dose dexamethasone versus lenalidomide plus low-dose dexamethasone as initial therapy for newly diagnosed multiple myeloma: an open-label randomised controlled trial. Lancet Oncol. 2010;11:29–37. doi: 10.1016/S1470-2045(09)70284-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Richardson PG, Siegel DS, Vij R, et al. Pomalidomide alone or in combination with low-dose dexamethasone in relapsed and refractory multiple myeloma: a randomized phase 2 study. Blood. 2014;123:1826–32. doi: 10.1182/blood-2013-11-538835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.San Miguel JF, Schlag R, Khuageva NK, et al. Bortezomib plus Melphalan and Prednisone for Initial Treatment of Multiple Myeloma. N Engl J Med. 2008;359:906–17. doi: 10.1056/NEJMoa0801479. [DOI] [PubMed] [Google Scholar]
- 61.Mateos MV, Oriol A, Martinez-Lopez J. Bortezomib/melphalan/prednisone (VMP) versus Bortezomib/Thalidomide/Prednisone (VTP) as induction therapy followed by maintenance treatment with Bortezomib/Thalidomide (VT) versus Bortezomib/Prednisone (VP): A randomised trial in elderly untreated patients with multiple myeloma older than 65 years. Lancet Oncol. 2010;11:934–41. doi: 10.1016/S1470-2045(10)70187-X. [DOI] [PubMed] [Google Scholar]
- 62.Palumbo A, Bringhen S, Rossi D, et al. Bortezomib-melphalan-prednisone-thalidomide followed by maintenance with bortezomib-thalidomide compared with bortezomib-melphalan-prednisone for initial treatment of multiple myeloma: a randomized controlled trial. J Clin Oncol. 2010;28:5101–9. doi: 10.1200/JCO.2010.29.8216. [DOI] [PubMed] [Google Scholar]
- 63.Cavo M, Tacchetti P, Patriarca F, et al. Bortezomib with thalidomide plus dexamethasone compared with thalidomide plus dexamethasone as induction therapy before, and consolidation therapy after, double autologous stem-cell transplantation in newly diagnosed multiple myeloma: a randomised phase 3 study. Lancet. 2010;376:2075–85. doi: 10.1016/S0140-6736(10)61424-9. [DOI] [PubMed] [Google Scholar]
- 64.Reeder CB, Reece DE, Kukreti V, et al. Cyclophosphamide, bortezomib and dexamethasone induction for newly diagnosed multiple myeloma: high response rates in a phase II clinical trial. Leukemia. 2009;23:1337–41. doi: 10.1038/leu.2009.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kumar S, Flinn I, Richardson PG, et al. Randomized, multicenter, phase 2 study (EVOLUTION) of combinations of bortezomib, dexamethasone, cyclophosphamide, and lenalidomide in previously untreated multiple myeloma. Blood. 2012;119:4375–82. doi: 10.1182/blood-2011-11-395749. [DOI] [PubMed] [Google Scholar]
- 66.Richardson PG, Weller E, Lonial S, et al. Lenalidomide, bortezomib, and dexamethasone combination therapy in patients with newly diagnosed multiple myeloma. Blood. 2010;116:679–86. doi: 10.1182/blood-2010-02-268862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Bringhen S, Petrucci MT, Larocca A, et al. Carfilzomib, cyclophosphamide, and dexamethasone in patients with newly diagnosed multiple myeloma: a multicenter, phase 2 study. Blood. 2014;124:63–9. doi: 10.1182/blood-2014-03-563759. [DOI] [PubMed] [Google Scholar]
- 68.Stewart AK, Rajkumar SV, Dimopoulos MA, et al. Carfilzomib, lenalidomide, and dexamethasone for relapsed multiple myeloma. The New England journal of medicine. 2015;372:142–52. doi: 10.1056/NEJMoa1411321. [DOI] [PubMed] [Google Scholar]
- 69.Shah JJ, Stadtmauer EA, Abonour R, et al. Carfilzomib, pomalidomide, and dexamethasone for relapsed or refractory myeloma. Blood. 2015;126:2284–90. doi: 10.1182/blood-2015-05-643320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Dimopoulos MA, Oriol A, Nahi H, et al. Daratumumab, Lenalidomide, and Dexamethasone for Multiple Myeloma. The New England journal of medicine. 2016;375:1319–31. doi: 10.1056/NEJMoa1607751. [DOI] [PubMed] [Google Scholar]
- 71.Palumbo A, Chanan-Khan A, Weisel K, et al. Daratumumab, Bortezomib, and Dexamethasone for Multiple Myeloma. The New England journal of medicine. 2016;375:754–66. doi: 10.1056/NEJMoa1606038. [DOI] [PubMed] [Google Scholar]
- 72.Chari A, Suvannasankha A, Fay JW, et al. Daratumumab plus pomalidomide and dexamethasone in relapsed and/or refractory multiple myeloma. Blood. 2017;130:974–81. doi: 10.1182/blood-2017-05-785246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lonial S, Dimopoulos M, Palumbo A, et al. Elotuzumab Therapy for Relapsed or Refractory Multiple Myeloma. The New England journal of medicine. 2015;373:621–31. doi: 10.1056/NEJMoa1505654. [DOI] [PubMed] [Google Scholar]
- 74.Moreau P, Masszi T, Grzasko N, et al. Oral Ixazomib, Lenalidomide, and Dexamethasone for Multiple Myeloma. The New England journal of medicine. 2016;374:1621–34. doi: 10.1056/NEJMoa1516282. [DOI] [PubMed] [Google Scholar]
- 75.San-Miguel MDJF, Hungria VTM, Yoon MDS. Randomized phase 3 trial of the deacetylase inhibitor panobinostat plus bortezomib and dexamethasone versus placebo plus bortezomib and dexamethasone in relapsed or relapsed and refractory multiple myeloma. Lancet Oncol. 2014;15:1195–206. doi: 10.1016/S1470-2045(14)70440-1. [DOI] [PubMed] [Google Scholar]
- 76.Ito T, Ando H, Suzuki T, et al. Identification of a primary target of thalidomide teratogenicity. Science. 2010;327:1345–50. doi: 10.1126/science.1177319. [DOI] [PubMed] [Google Scholar]
- 77.Krönke J, Udeshi ND, Narla A, et al. Lenalidomide Causes Selective Degradation of IKZF1 and IKZF3 in Multiple Myeloma Cells. Science. 2014;343:301–5. doi: 10.1126/science.1244851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lu G, Middleton RE, Sun H, et al. The Myeloma Drug Lenalidomide Promotes the Cereblon-Dependent Destruction of Ikaros Proteins. Science. 2014;343:305–9. doi: 10.1126/science.1244917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Parman T, Wiley MJ, Wells PG. Free radical-mediated oxidative DNA damage in the mechanism of thalidomide teratogenicity [see comments] Nature Medicine. 1999;5:582–5. doi: 10.1038/8466. [DOI] [PubMed] [Google Scholar]
- 80.Rajkumar SV, Richardson PG, Hideshima T, Anderson KC. Proteasome Inhibition as a Novel Therapeutic Target in Human Cancer. J Clin Oncol. 2004;23:630–9. doi: 10.1200/JCO.2005.11.030. [DOI] [PubMed] [Google Scholar]
- 81.Kumar S, Rajkumar SV. Many facets of bortezomib resistance/susceptibility. Blood. 2008;112:2177–8. doi: 10.1182/blood-2008-07-167767. [DOI] [PubMed] [Google Scholar]
- 82.Gutman D, Morales AA, Boise LH. Acquisition of a multidrug-resistant phenotype with a proteasome inhibitor in multiple myeloma. Leukemia. 2009;23:2181–3. doi: 10.1038/leu.2009.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Lokhorst HM, Plesner T, Laubach JP, et al. Targeting CD38 with Daratumumab Monotherapy in Multiple Myeloma. The New England journal of medicine. 2015;373:1207–19. doi: 10.1056/NEJMoa1506348. [DOI] [PubMed] [Google Scholar]
- 84.Lonial Sagar, Weiss Brendan M, Usmani Saad Zafar, et al. Single-agent daratumumab in heavily pre-treated patients with Multiple Myeloma: An Open-Label, International, Multicentre Phase 2 trial (Sirius) Lancet. 2016 doi: 10.1016/S0140-6736(15)01120-4. [DOI]
- 85.Rajkumar SV. Panobinostat for the treatment of multiple myeloma. The Lancet Oncology. 2014;15:1178–9. doi: 10.1016/S1470-2045(14)70443-7. [DOI] [PubMed] [Google Scholar]
- 86.Mateos M-V, Richardson PG, Schlag R, et al. Bortezomib Plus Melphalan and Prednisone Compared With Melphalan and Prednisone in Previously Untreated Multiple Myeloma: Updated Follow-Up and Impact of Subsequent Therapy in the Phase III VISTA Trial. J Clin Oncol. 2010;28:2259–66. doi: 10.1200/JCO.2009.26.0638. [DOI] [PubMed] [Google Scholar]
- 87.Benboubker L, Dimopoulos MA, Dispenzieri A, et al. Lenalidomide and dexamethasone in transplant-ineligible patients with myeloma. The New England journal of medicine. 2014;371:906–17. doi: 10.1056/NEJMoa1402551. [DOI] [PubMed] [Google Scholar]
- 88.Moreau P, Hulin C, Macro M, et al. VTD is superior to VCD prior to intensive therapy in multiple myeloma: results of the prospective IFM2013-04 trial. Blood. 2016;127:2569–74. doi: 10.1182/blood-2016-01-693580. [DOI] [PubMed] [Google Scholar]
- 89.Rajkumar SV. Treatment of Myeloma: Cure vs Control. Mayo Clinic Proceedings. 2008;83:1142–5. doi: 10.4065/83.10.1142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Rajkumar SV, Gahrton G, Bergsagel PL. Approach to the treatment of multiple myeloma: a clash of philosophies. Blood. 2011;118:3205–11. doi: 10.1182/blood-2011-06-297853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Moreau P, Rajkumar SV. Multiple myeloma--translation of trial results into reality. Lancet. 2016;388:111–3. doi: 10.1016/S0140-6736(16)30954-0. [DOI] [PubMed] [Google Scholar]
- 92.Moreau P, Pylypenko H, Grosicki S, et al. Subcutaneous versus intravenous administration of bortezomib in patients with relapsed multiple myeloma: a randomised, phase 3, non-inferiority study. Lancet Oncol. 2011;12:431–40. doi: 10.1016/S1470-2045(11)70081-X. [DOI] [PubMed] [Google Scholar]
- 93.Moreau P, Hulin C, Marit G, et al. Stem cell collection in patients with de novo multiple myeloma treated with the combination of bortezomib and dexamethasone before autologous stem cell transplantation according to IFM 2005-01 trial. Leukemia. 2010;24:1233–5. doi: 10.1038/leu.2010.82. [DOI] [PubMed] [Google Scholar]
- 94.Kumar S, Dispenzieri A, Lacy MQ, et al. Impact of lenalidomide therapy on stem cell mobilization and engraftment post-peripheral blood stem cell transplantation in patients with newly diagnosed myeloma. Leukemia. 2007;21:2035–42. doi: 10.1038/sj.leu.2404801. [DOI] [PubMed] [Google Scholar]
- 95.Kumar S, Giralt S, Stadtmauer EA, et al. Mobilization in myeloma revisited: IMWG consensus perspectives on stem cell collection following initial therapy with thalidomide-, lenalidomide-, or bortezomib-containing regimens. Blood. 2009;114:1729–35. doi: 10.1182/blood-2009-04-205013. [DOI] [PubMed] [Google Scholar]
- 96.Giralt S, Stadtmauer EA, Harousseau JL, et al. International myeloma working group (IMWG) consensus statement and guidelines regarding the current status of stem cell collection and high-dose therapy for multiple myeloma and the role of plerixafor (AMD 3100) Leukemia. 2009;23:1904–12. doi: 10.1038/leu.2009.127. [DOI] [PubMed] [Google Scholar]
- 97.Palumbo A, Cavo M, Bringhen S, et al. Aspirin, warfarin, or enoxaparin thromboprophylaxis in patients with multiple myeloma treated with thalidomide: a phase III, open-label, randomized trial. J Clin Oncol. 2011;29:986–93. doi: 10.1200/JCO.2010.31.6844. [DOI] [PubMed] [Google Scholar]
- 98.Larocca A, Cavallo F, Bringhen S, et al. Aspirin or enoxaparin thromboprophylaxis for newly-diagnosed multiple myeloma patients treated with lenalidomide. Blood. 2011;119:933–9. doi: 10.1182/blood-2011-03-344333. [DOI] [PubMed] [Google Scholar]
- 99.Palumbo A, Rajkumar SV, Dimopoulos MA, et al. Prevention of thalidomide- and lenalidomide-associated thrombosis in myeloma. Leukemia. 2008;22:414–23. doi: 10.1038/sj.leu.2405062. [DOI] [PubMed] [Google Scholar]
- 100.Jakubowiak AJ, Dytfeld D, Griffith KA, et al. A phase 1/2 study of carfilzomib in combination with lenalidomide and low-dose dexamethasone as a frontline treatment for multiple myeloma. Blood. 2012;120:1801–9. doi: 10.1182/blood-2012-04-422683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Zingone A, Kwok ML, Manasanch EE, et al. Phase II Clinical and Correlative Study Of Carfilzomib, Lenalidomide, and Dexamethasone Followed By Lenalidomide Extended Dosing (CRD-R) Induces High Rates Of MRD Negativity In Newly Diagnosed Multiple Myeloma (MM) Patients. Blood. 2013;122:538. [Google Scholar]
- 102.Barlogie B, Anaissie E, van Rhee F, et al. Incorporating bortezomib into upfront treatment for multiple myeloma: early results of total therapy 3. British Journal of Haematology. 2007;138:176–85. doi: 10.1111/j.1365-2141.2007.06639.x. [DOI] [PubMed] [Google Scholar]
- 103.van Rhee F, Szymonifka J, Anaissie E, et al. Total therapy 3 for multiple myeloma: prognostic implications of cumulative dosing and premature discontinuation of VTD maintenance components, bortezomib, thalidomide and dexamethasone, relevant to all phases of therapy. Blood. 116:1220–7. doi: 10.1182/blood-2010-01-264333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Magarotto V, Bringhen S, Offidani M, et al. A Randomized Phase 3 Trial Of Melphalan-Lenalidomide-Prednisone (MPR) Or Cyclophosphamide-Prednisone-Lenalidomide (CPR) Vs Lenalidomide Plus Dexamethsone (Rd) In Elderly Newly Diagnosed Multiple Myeloma Patients. Blood. 2013;122:536. [Google Scholar]
- 105.Attal M, Harousseau JL, Stoppa AM, et al. A prospective, randomized trial of autologous bone marrow transplantation and chemotherapy in multiple myeloma. Intergroupe Francais du Myelome. N Engl J Med. 1996;335:91–7. doi: 10.1056/NEJM199607113350204. [DOI] [PubMed] [Google Scholar]
- 106.Child JA, Morgan GJ, Davies FE, et al. High-dose chemotherapy with hematopoietic stem-cell rescue for multiple myeloma. N Engl J Med. 2003;348:1875–83. doi: 10.1056/NEJMoa022340. [DOI] [PubMed] [Google Scholar]
- 107.Blade J, Vesole DH, Gertz M. Transplantation for multiple myeloma: who, when, how often? Blood. 2003;102:3469–77. [Google Scholar]
- 108.Kumar A, Loughran T, Alsina M, Durie BG, Djulbegovic B. Management of multiple myeloma: a systematic review and critical appraisal of published studies. Lancet Oncology. 2003;4:293–304. doi: 10.1016/s1470-2045(03)01077-5. [DOI] [PubMed] [Google Scholar]
- 109.Fermand JP, Ravaud P, Chevret S, et al. High-dose therapy and autologous peripheral blood stem cell transplantation in multiple myeloma: up-front or rescue treatment?. Results of a multicenter sequential randomized clinical trial. Blood. 1998;92:3131–6. [PubMed] [Google Scholar]
- 110.Facon T, Mary JY, Harousseau JL, et al. Front-line or rescue autologous bone marrow transplantation (ABMT) following a first course of high dose melphalan (HDM) in multiple myeloma (MM). Preliminary results of a prospective randomized trial (CIAM) protocol. Blood. 1996;88(Suppl1):685a. [Google Scholar]
- 111.Barlogie B, Kyle RA, Anderson KC, et al. Standard chemotherapy compared with high-dose chemoradiotherapy for multiple myeloma: final results of phase III US Intergroup Trial S9321. Journal of Clinical Oncology. 2006;24:929–36. doi: 10.1200/JCO.2005.04.5807. [DOI] [PubMed] [Google Scholar]
- 112.Attal M, Harousseau JL, Facon T, et al. Single versus double autologous stem-cell transplantation for multiple myeloma. [see comment] N Engl J Med. 2003;349:2495–502. doi: 10.1056/NEJMoa032290. [DOI] [PubMed] [Google Scholar]
- 113.Cavo M, Tosi P, Zamagni E, et al. Prospective, Randomized Study of Single Compared With Double Autologous Stem-Cell Transplantation for Multiple Myeloma: Bologna 96 Clinical Study. J Clin Oncol. 2007;25:2434–41. doi: 10.1200/JCO.2006.10.2509. [DOI] [PubMed] [Google Scholar]
- 114.Fermand JP, Alberti C, Marolleau JP. Single versus tandem high dose therapy (HDT) supported with autologous blood stem cell (ABSC) transplantation using unselected or CD34-enriched ABSC: results of a two by two designed randomized trial in 230 young patients with multiple myeloma (MM) Hematol J. 2003;4(Suppl 1):S59. [Google Scholar]
- 115.Goldschmidt H. Single vs. tandem autolgous transplantation in multiple myeloma: the GMMG experience. Hematol J. 2003;4(Suppl 1):S61. [Google Scholar]
- 116.Cavo M, Gay FM, Patriarca F, et al. Double Autologous Stem Cell Transplantation Significantly Prolongs Progression-Free Survival and Overall Survival in Comparison with Single Autotransplantation in Newly Diagnosed Multiple Myeloma: An Analysis of Phase 3 EMN02/HO95 Study. Blood. 2017;130:401. [Google Scholar]
- 117.Stadtmauer EA, Pasquini MC, Blackwell B, et al. Comparison of Autologous Hematopoietic Cell Transplant (autoHCT), Bortezomib, Lenalidomide (Len) and Dexamethasone (RVD) Consolidation with Len Maintenance (ACM), Tandem Autohct with Len Maintenance (TAM) and Autohct with Len Maintenance (AM) for up-Front Treatment of Patients with Multiple Myeloma (MM): Primary Results from the Randomized Phase III Trial of the Blood and Marrow Transplant Clinical Trials Network (BMT CTN 0702 - StaMINA Trial) Blood. 2016;128 LBA-1-LBA- [Google Scholar]
- 118.Krishnan A, Pasquini MC, Logan B, et al. Autologous haemopoietic stem-cell transplantation followed by allogeneic or autologous haemopoietic stem-cell transplantation in patients with multiple myeloma (BMT CTN 0102): a phase 3 biological assignment trial. Lancet Oncol. 2011;12:1195–203. doi: 10.1016/S1470-2045(11)70243-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Bruno B, Rotta M, Patriarca F, et al. A Comparison of Allografting with Autografting for Newly Diagnosed Myeloma. N Engl J Med. 2007;356:1110–20. doi: 10.1056/NEJMoa065464. [DOI] [PubMed] [Google Scholar]
- 120.Stewart AK. Reduced-intensity allogeneic transplantation for myeloma: reality bites. Blood. 2009;113:3135–6. doi: 10.1182/blood-2008-12-173526. [DOI] [PubMed] [Google Scholar]
- 121.Attal M, Lauwers-Cances V, Marit G, et al. Lenalidomide Maintenance after Stem-Cell Transplantation for Multiple Myeloma. N Engl J Med. 2012;366:1782–91. doi: 10.1056/NEJMoa1114138. [DOI] [PubMed] [Google Scholar]
- 122.McCarthy PL, Owzar K, Hofmeister CC, et al. Lenalidomide after Stem-Cell Transplantation for Multiple Myeloma. N Engl J Med. 2012;366:1770–81. doi: 10.1056/NEJMoa1114083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Palumbo A, Cavallo F, Gay F, et al. Autologous transplantation and maintenance therapy in multiple myeloma. The New England journal of medicine. 2014;371:895–905. doi: 10.1056/NEJMoa1402888. [DOI] [PubMed] [Google Scholar]
- 124.Sonneveld P, Schmidt-Wolf IGH, van der Holt B, et al. Bortezomib Induction and Maintenance Treatment in Patients With Newly Diagnosed Multiple Myeloma: Results of the Randomized Phase III HOVON-65/GMMG-HD4 Trial. Journal of Clinical Oncology. 2012;30:2946–55. doi: 10.1200/JCO.2011.39.6820. [DOI] [PubMed] [Google Scholar]
- 125.Palumbo A, Hajek R, Delforge M, et al. Continuous Lenalidomide Treatment for Newly Diagnosed Multiple Myeloma. N Engl J Med. 2012;366:1759–69. doi: 10.1056/NEJMoa1112704. [DOI] [PubMed] [Google Scholar]
- 126.Attal M, Palumbo A, Holstein SA, et al. Lenalidomide (LEN) maintenance (MNTC) after high-dose melphalan and autologous stem cell transplant (ASCT) in multiple myeloma (MM): A meta-analysis (MA) of overall survival (OS) J Clin Oncol. 2016;34(suppl):A8001. (abstract) [Google Scholar]
- 127.Kumar SK, Therneau TM, Gertz MA, et al. Clinical course of patients with relapsed multiple myeloma. Mayo Clinic Proceedings. 2004;79:867–74. doi: 10.4065/79.7.867. [DOI] [PubMed] [Google Scholar]
- 128.Kumar SK, Lee JH, Lahuerta JJ, et al. Risk of progression and survival in multiple myeloma relapsing after therapy with IMiDs and bortezomib: A multicenter international myeloma working group study. Leukemia. 2012;26:149–57. doi: 10.1038/leu.2011.196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Siegel DS, Dimopoulos MA, Ludwig H, et al. Improvement in Overall Survival With Carfilzomib, Lenalidomide, and Dexamethasone in Patients With Relapsed or Refractory Multiple Myeloma. Journal of Clinical Oncology. 2018;36:728–34. doi: 10.1200/JCO.2017.76.5032. [DOI] [PubMed] [Google Scholar]
- 130.San Miguel J, Weisel K, Moreau P, et al. Pomalidomide plus low-dose dexamethasone versus high-dose dexamethasone alone for patients with relapsed and refractory multiple myeloma (MM-003): a randomised, open-label, phase 3 trial. Lancet Oncol. 2013;14:1055–66. doi: 10.1016/S1470-2045(13)70380-2. [DOI] [PubMed] [Google Scholar]
- 131.Dimopoulos MA, Moreau P, Palumbo A, et al. Carfilzomib and dexamethasone versus bortezomib and dexamethasone for patients with relapsed or refractory multiple myeloma (ENDEAVOR): a randomised, phase 3, open-label, multicentre study. Lancet Oncol. 2016;17:27–38. doi: 10.1016/S1470-2045(15)00464-7. [DOI] [PubMed] [Google Scholar]
- 132.Dimopoulos MA, Goldschmidt H, Niesvizky R, et al. Carfilzomib or bortezomib in relapsed or refractory multiple myeloma (ENDEAVOR): an interim overall survival analysis of an open-label, randomised, phase 3 trial. Lancet Oncol. 2017;18:1327–37. doi: 10.1016/S1470-2045(17)30578-8. [DOI] [PubMed] [Google Scholar]
- 133.Pineda-Roman M, Zangari M, van Rhee F, et al. VTD combination therapy with bortezomib-thalidomide-dexamethasone is highly effective in advanced and refractory multiple myeloma. Leukemia. 2008;22:1419–27. doi: 10.1038/leu.2008.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Richardson PG, Weller E, Jagannath S, et al. Multicenter, Phase I, Dose-Escalation Trial of Lenalidomide Plus Bortezomib for Relapsed and Relapsed/Refractory Multiple Myeloma. Journal of Clinical Oncology. 2009;27:5713–9. doi: 10.1200/JCO.2009.22.2679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Rajkumar SV, Kyle RA. Progress in Myeloma - A Monoclonal Breakthrough. The New England journal of medicine. 2016;375:1390–2. doi: 10.1056/NEJMe1609835. [DOI] [PubMed] [Google Scholar]
- 136.Siegel DS, Martin T, Wang M, et al. A phase 2 study of single-agent carfilzomib (PX-171-003-A1) in patients with relapsed and refractory multiple myeloma. Blood. 2012;120:2817–25. doi: 10.1182/blood-2012-05-425934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Lacy MQ, Hayman SR, Gertz MA, et al. Pomalidomide (CC4047) Plus Low-Dose Dexamethasone As Therapy for Relapsed Multiple Myeloma. J Clin Oncol. 2009;27:5008–14. doi: 10.1200/JCO.2009.23.6802. [DOI] [PubMed] [Google Scholar]
- 138.Lacy MQ, Hayman SR, Gertz MA, et al. Pomalidomide (CC4047) plus low dose dexamethasone (Pom/dex) is active and well tolerated in lenalidomide refractory multiple myeloma (MM) Leukemia. 2010;24:1934–9. doi: 10.1038/leu.2010.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Lacy MQ, Allred JB, Gertz MA, et al. Pomalidomide plus low-dose dexamethasone in myeloma refractory to both bortezomib and lenalidomide: comparison of 2 dosing strategies in dual-refractory disease. Blood. 2011;118:2970–5. doi: 10.1182/blood-2011-04-348896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Kumar SK, Berdeja JG, Niesvizky R, et al. Safety and tolerability of ixazomib, an oral proteasome inhibitor, in combination with lenalidomide and dexamethasone in patients with previously untreated multiple myeloma: an open-label phase 1/2 study. Lancet Oncol. 2014;15:1503–12. doi: 10.1016/S1470-2045(14)71125-8. [DOI] [PubMed] [Google Scholar]
- 141.Orlowski RZ, Nagler A, Sonneveld P, et al. Randomized Phase III Study of Pegylated Liposomal Doxorubicin Plus Bortezomib Compared With Bortezomib Alone in Relapsed or Refractory Multiple Myeloma: Combination Therapy Improves Time to Progression. J Clin Oncol. 2007;25:3892–901. doi: 10.1200/JCO.2006.10.5460. [DOI] [PubMed] [Google Scholar]
- 142.Hideshima T, Bradner JE, Wong J, et al. Small-molecule inhibition of proteasome and aggresome function induces synergistic antitumor activity in multiple myeloma. Proc Natl Acad Sci U S A. 2005;102:8567–72. doi: 10.1073/pnas.0503221102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.San-Miguel JF, Richardson PG, Gunther A, et al. Phase Ib study of panobinostat and bortezomib in relapsed or relapsed and refractory multiple myeloma. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2013;31:3696–703. doi: 10.1200/JCO.2012.46.7068. [DOI] [PubMed] [Google Scholar]
- 144.Mey UJ, Brugger W, Schwarb H, et al. Bendamustine, lenalidomide and dexamethasone (BRd) has high activity as 2(nd) -line therapy for relapsed and refractory multiple myeloma - a phase II trial. Br J Haematol. 2017;176:770–82. doi: 10.1111/bjh.14481. [DOI] [PubMed] [Google Scholar]
- 145.Rodon P, Hulin C, Pegourie B, et al. Phase II study of bendamustine, bortezomib and dexamethasone as second-line treatment for elderly patients with multiple myeloma: the Intergroupe Francophone du Myelome 2009-01 trial. Haematologica. 2015;100:e56–9. doi: 10.3324/haematol.2014.110890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Kumar S, Kaufman JL, Gasparetto C, et al. Efficacy of venetoclax as targeted therapy for relapsed/refractory t(11;14) multiple myeloma. Blood. 2017;130:2401–9. doi: 10.1182/blood-2017-06-788786. [DOI] [PubMed] [Google Scholar]
- 147.Berdeja JG, Lin Y, Raje N, et al. Durable Clinical Responses in Heavily Pretreated Patients with Relapsed/Refractory Multiple Myeloma: Updated Results from a Multicenter Study of bb2121 Anti-Bcma CAR T Cell Therapy. Blood. 2017;130:740. [Google Scholar]
- 148.Trudel S, Lendvai N, Popat R, et al. Deep and Durable Responses in Patients (Pts) with Relapsed/Refractory Multiple Myeloma (MM) Treated with Monotherapy GSK2857916, an Antibody Drug Conjugate Against B-Cell Maturation Antigen (BCMA): Preliminary Results from Part 2 of Study BMA117159. Blood. 2017;130:741. [Google Scholar]
- 149.Rajkumar SV, Landgren O, Mateos MV. Smoldering multiple myeloma. Blood. 2015;125:3069–75. doi: 10.1182/blood-2014-09-568899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Hjorth M, Hellquist L, Holmberg E, Magnusson B, Rodjer S, Westin J. Initial versus deferred melphalan-prednisone therapy for asymptomatic multiple myeloma stage I--a randomized study. Myeloma Group of Western Sweden. European Journal of Haematology. 1993;50:95–102. doi: 10.1111/j.1600-0609.1993.tb00148.x. [DOI] [PubMed] [Google Scholar]
- 151.Grignani G, Gobbi PG, Formisano R, et al. A prognostic index for multiple myeloma. Br J Cancer. 1996;73:1101–7. doi: 10.1038/bjc.1996.212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Mateos MV, Hernandez MT, Giraldo P, et al. Lenalidomide plus dexamethasone versus observation in patients with high-risk smouldering multiple myeloma (QuiRedex): long-term follow-up of a randomised, controlled, phase 3 trial. Lancet Oncol. 2016;17:1127–36. doi: 10.1016/S1470-2045(16)30124-3. [DOI] [PubMed] [Google Scholar]
- 153.Mateos M-V, Martinez Lopez J, Rodriguez-Otero P, et al. Curative Strategy for High-Risk Smoldering Myeloma (GEM-CESAR): Carfilzomib, Lenalidomide and Dexamethasone (KRd) As Induction Followed By HDT-ASCT, Consolidation with Krd and Maintenance with Rd. Blood. 2017;130:402. [Google Scholar]
- 154.Dimopoulos MA, Lonial S, White D, et al. Elotuzumab plus lenalidomide/dexamethasone for relapsed or refractory multiple myeloma: ELOQUENT-2 follow-up and post-hoc analyses on progression-free survival and tumour growth. Br J Haematol. 2017;178:896–905. doi: 10.1111/bjh.14787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.San-Miguel JF, Hungria VT, Yoon SS, et al. Overall survival of patients with relapsed multiple myeloma treated with panobinostat or placebo plus bortezomib and dexamethasone (the PANORAMA 1 trial): a randomised, placebo-controlled, phase 3 trial. Lancet Haematol. 2016;3:e506–e15. doi: 10.1016/S2352-3026(16)30147-8. [DOI] [PubMed] [Google Scholar]