Abstract
The skin interfaces with the external environment and is home to a myriad of immune cells that patrol the barrier to ward off harmful agents and aid in tissue repair. The formation of the cutaneous immune arsenal begins before birth and evolves throughout our lifetime, incorporating exogenous cues from microbes and inflammatory encounters, to achieve optimal fitness and function. Here, we discuss the context-specific signals that drive productive immune responses in the skin epithelia, highlighting key modulators of these reactions, including hair follicles, neurons, and commensal microbes. We thus also discuss the causal and mechanistic underpinning of inflammatory skin diseases that have been revealed in recent years. Finally, we discuss the non-canonical functions of cutaneous immune cells including their burgeoning role in epithelial regeneration and repair. The rapidly growing field of cutaneous immunity is revealing immune mechanisms and functions that can be harnessed to boost skin health and treat disease.
Introduction
Our body’s largest organ, the skin, is a vital barrier against environmental pressures including extreme temperatures, UV, allergens, toxins, and microbes. This complex organ is composed of an array of cell types including endothelial cells, fibroblasts, neurons, adipocytes and epithelial cells that act in unison to ensure its function. Cells of the immune system have surfaced as orchestrators of many facets of skin biology that collectively serve to reinforce the barrier against external threats. The composition, function, and activation status of immune cells is constantly tuned by epidermal structures, and by microbial and inflammatory stimuli. In turn, resident and recruited immune cells engage in an active dialogue with the tissue parenchyma to limit microbial invasion and direct repair of the barrier.
Here we discuss the dynamic interactions between the myriad of innate and adaptive immune cells housed in the skin and their surrounding stroma in health and disease. In particular, we home in on the context specific cross-regulation of immunity and epithelial keratinocytes, which can either promote antimicrobial responses or, when left unchecked, lead to devastating inflammatory diseases. In this regard, emerging evidence points to hair follicles, innervating neurons and surface microbiota as key instructors of the skin’s immune repertoire and function that dictate optimal barrier function and fitness
Skin, a multi-layered barrier
The skin originates from multiple embryonic layers including the ectoderm, mesoderm, and neural crest. This multi-layered organ is comprised of the epidermis, dermis and hypodermis each of which contain unique immune cell populations that interact with and direct the function of their adjacent stroma.
The epidermis is primarily composed of keratinocytes of ectodermal origin, Merkel cells that differentiate from keratinocyte precursors, as well as melanocytes, that originate from the neural crest. Multiple hematopoietic lineages also reside in the epidermis. Langerhans cells (LC), a unique antigen presenting cell population, derived from the prenatal yolk sac take residence in the epidermis even prior to birth. LCs constantly survey the epidermal microenvironment, relaying this information to regional lymph nodes to actively generate host-protective and regulatory immune responses (Doebel et al., 2017; Merad et al., 2008). Dendritic epidermal T cells (DETC), a subset of γδ T cells, also resides alongside LCs in mouse epidermis and play crucial roles in wound healing (Nielsen et al., 2017). As the skin experiences pathogenic assaults, CD4+ and CD8+ T cells that are cellular remnants of these reactions persist long-term in the form of resident memory T cells (TRM) in the epidermis and patrol the tissue awaiting secondary stimuli (Mueller and Mackay, 2016; Schenkel and Masopust, 2014). Recent studies have also revealed that the epidermis also contains innate lymphoid cells (ILCs) of many different flavors and functions (Ricardo-Gonzalez et al., 2018; Roediger et al., 2013), and uncovered their central role in regulating the tissue hyperplasia (Kobayashi et al., 2019).
The underlying dermis houses its own functionally specialized immune populations and unique stroma. Dermal immune constituents include several distinct dendritic cell (DC) and T cell subsets that are localized in close apposition to the epithelium and, in particular, hair follicles. Macrophages, mast cells, basophils and eosinophils are also crucial components of the dermal immune milieu. Heterogeneous populations of dermal fibroblasts with distinct functions have substantial impacts on tissue repair (Driskell et al., 2013; Guerrero-Juarez et al., 2019). Adipocytes send signals to epithelial cells to regulate hair growth and mediate antibacterial host defense by producing the antimicrobial peptide, cathelicidin (Guerrero-Juarez and Plikus, 2018; Zhang et al., 2015; Zwick et al., 2018). This constellation of dermal immune cells is vital for mounting cutaneous immune responses and in aiding repair (Kabashima et al., 2019; Pasparakis et al., 2014); however, this review will focus on their role primarily in the context of their interaction with the epithelium. The hematopoietic constituents of the hypodermis and their functions remain relatively unexplored.
There has been an increasing body of literature cataloguing and functionally examining the immune compartment of human skin. Structurally, human and mouse skin are quite distinct. Human epidermis is about 6–10 cell layers thick, while mouse epidermis is only 3 layers thick (Gudjonsson et al., 2007). Unsurprisingly, mouse skin exhibits greater barrier permeability than human skin, with significant implications for differential uptake of environmental antigens and their influence over the cutaneous immune milieu (Menon, 2002). There are also key differences in the types of immune cells, in particular lymphocytes, found in the human versus mouse skin. While mouse epidermis is predominantly populated by DETCs, human epidermis harbors many more CD4+ and CD8+ TRMs (Clark, 2015). Human skin also houses a significant population of innate-like T cells called Mucosal-associated invariant T (MAIT) cells that can only be elicited in mice with the application of certain surface commensals (Constantinides et al., 2017). The development and composition of innate immune subsets also differs markedly between humans and mice. Most notably, human skin dendritic cells (DC) express a distinct repertoire of antigen presenting molecules, CD1a, b and c, that enable presentation of lipids to T cells (Collin et al., 2013). These are only a few key differences between human and mouse skin immunity and, despite these seeming differences, it is important to note that many key principles uncovered in mice have enormously aided in our understanding of human skin disease. For example, mouse models were instrumental to the discovery that psoriasis is a T cell-mediated autoimmune disease and the underlying roles of IL-17A in the pathogenesis of this disease (Dainichi et al., 2018; Nestle et al., 2009).
Another consideration when understanding the cutaneous immune system, human or mouse, is the remarkable architectural heterogeneity of this large organ. Different skin sites have varied densities of hair follicles, sebaceous glands and sweat glands, and variations in the thickness of the epidermal and dermal layers (Otberg et al., 2004; Robertson and Rees, 2010). Moreover, the composition of resident microbiota also varies drastically between skin sites (Grice et al., 2009). Along with these structural and microbial differences, the skin’s immune milieu (Tong et al., 2015) is also uniquely tailored to each skin area and influences responsiveness to pathogens (Doehl et al., 2017). Although the mechanistic underpinnings of such site-specific immune variation remain elusive, it is tempting to speculate that skin structures in collaboration with distinct microbial stimuli locally dictate the skin’s immune repertoire in topologically distinct regions.
Epithelial cells govern immune cell localization
Far from a static barrier, the epithelium dynamically reacts to environmental stimuli, recruiting and retaining its own immune arsenal. Attracting immune cells to the skin, however, is not an equivalent feature of all skin epithelial cells, as these functions appear to be regionally compartmentalized. Mammalian epidermis uniquely equipped with invaginations such as hair follicles, interspersed with flat interfollicular epidermal regions. The follicular and interfollicular epithelial layers are contiguous and contain resident DETCs, LCs, and ILCs. Hair follicles, however, have their own unique immune microcosm that also includes dermal immune populations.
Conjuring immune cells appears to be a specialized property of follicle epithelium, which is particularly adept at transmitting homeostatic and distress signals to draw cells to their vicinity. Evolutionarily, the emergence of innate and adaptive immunity precedes the occurrence of hair follicles as hairless vertebrate are known to have prototypic lymphoid cells (Vivier et al., 2016). Perhaps these epidermal invaginations evolved as bidirectional passageway for environmental signals to tune the skin’s immune surveillance system while simultaneously providing immune cells passage to the epidermis.
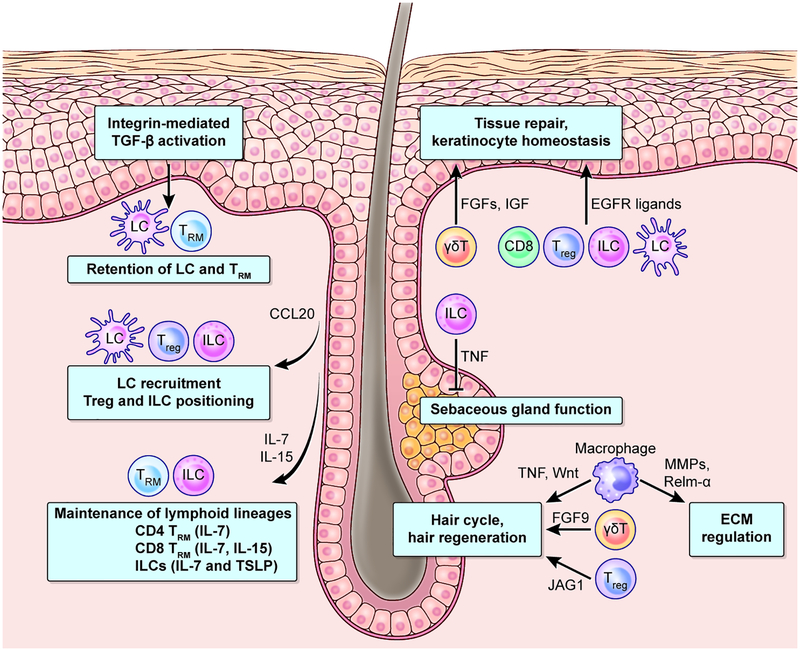
Lymphocytes and myeloid cells have long been observed in the vicinity of hair follicles (Paus et al., 1998) (Christoph et al., 2000), but the biological significance and specific mechanisms underlying this observation have only recently been appreciated (Figure 1). Tissue-derived chemokines are crucial for guiding immune cells to specific tissues within the body and even to certain regions within a given tissue. In the skin, hair follicles serve as epicenters for positioning immune cells both at steady state and under duress. We have previously reported that certain regions of the follicle are highly specialized and produce specific chemokines that either attract or repel immune cells (Nagao et al., 2012). Following mechanical stress, keratinocytes in the upper follicular regions, the isthmus and infundibulum, uniquely express the chemokines Ccl2 and Ccl20, while the bulge area of the follicle, which houses stem cells, produce CCL8. This pattern of chemokine expression is essential for recruiting LC precursors and DCs to sites of minor trauma, while keeping them away from the follicle stem cell niche, presumably to protect the protect the stem cells from immune-mediated damage (Nagao et al., 2012). By contrast, barrier disruption caused by genetic ablation of adherens junction proteins from bulge stem cells and their suprabasal “inner” bulge cells uniquely induces CCL2 production by the inner bulge that induces corralling of immune cells, in particular DCs, around the bulge (Lay et al., 2018).
Figure 1: Mutual crosstalk between epidermal and immune cells during homeostasis and tissue repair.
Hair follicles (HFs) serve as control towers that recruit and position skin-resident immune cells. HFs produce CCL20 to guide Langerhans cells (LCs) entry into epidermis and to position resident-memory T cells (TRMs), including regulatory T cell (Tregs), and innate lymphoid cells (ILCs). HFs also provide cytokines that TRMs and ILCs depend on for maintenance. Integrins αvβ6 and αvβ8 expressed on keratinocytes activate TGF-β that is required for epidermal retention of LCs and TRMs. Immune cells also send vital signals to orchestrate homeostatic maintenance of hair cycle and sebaceous glands and direct tissue-repair. Tregs express the Notch ligand, Jagged 1 (JAG1), and promote hair regeneration. TNF-producing macrophages, which are recruited by HF-derived CCL2, activate the hair regeneration program. In the natural cycle, resident macrophages produce stem cell activating Wnts to induce hair cycling. ILCs regulate sebaceous gland function to tune skin microbial equilibrium. FGF9 produced by dermal γδ T cells amplifies WNT signaling to promote wound-induced hair neogenesis. Epithelialization in wounds is mediated by immune cell-derived factors including epidermal growth factor receptor (EGFR) ligands and fibroblast growth factors (FGFs).
The CCL20-CCR6 axis is also operative in the lymphoid lineage. Neonatal migration of skin regulatory T cells (Tregs) takes place in conjunction with the hair follicle morphogenesis. CCL20 expression in upper hair follicles and is enhanced by skin-commensal bacteria and augments the frequency of Tregs in the skin at this developmental stage (Scharschmidt et al., 2017). Another important factor dictating the positioning of immune cells is their own pattern of chemokine receptor expression. This is perhaps best illustrated by cutaneous ILCs, which can be functionally discerned based on their expression of chemokine receptors CCR6 and CCR10 (Yang et al., 2016) (Kobayashi et al., 2019). CCR10 broadly directs skin-homing, while CCR6 guides ILCs to localize in close apposition of the sebaceous glands. CCR6+ epidermal and dermal ILCs regulate sebaceous gland size, modulating the composition and amount of sebum and influencing the composition of skin microbiota (Kobayashi et al., 2019).
Collectively, these studies showcase the tight compartmentalization of epithelial immune responses in the skin and in particular, underscore the follicular epitheliums’ heightened ability to attract immune cells in times of duress. Whether distinct epithelial populations (for example, follicular versus interfollicular) are developmentally specified to differentially recruit immune cells or tuned by their local milieu remains an open question. However, such regional segregation of immune-epithelial responses may be important for limiting tissue-wide dissemination of inflammation. When unchecked these same properties may potentiate inflammatory diseases by aberrant production of chemokines. The CCL20-CCR6 axis may thus be clinically relevant target, as effector lymphocytes in psoriatic skin are reported to express CCR6 (Hedrick et al., 2009; Mabuchi et al., 2013; Singh et al., 2016). The spectrum of chemokine receptor expression on immune cells and the unique chemokine profile from the epithelium provoked by distinct environmental triggers, demands a more nuanced characterization of chemokine-chemokine receptor network that dictates immune cell positioning during homeostasis and inflammation.
Keratinocytes promote tissue-residency of immune cells
In addition to recruiting immune cells to the epithelial interface, keratinocyte also regulate their survival and retention in the tissue (Figure 1). One cell type that is particularly reliant on epithelial derived factors for sustenance is the TRM. In accordance with their name, TRMs are long-lived cells that form after resolution of infection and remain in skin to confer protective immunity (Gebhardt et al., 2009; Jiang et al., 2012) (Gaide et al., 2015). TRMs not only accumulate at the site of primary inflammation, but they also distribute widely to distant, previously uninvolved skin to enable broader host protection against secondary challenge (Jiang et al., 2012). Members of the common gamma chain (γc) family of cytokines, IL-7 and IL-15 in particular, are required for the generation and/or maintenance of TRMs in peripheral tissues (Adachi et al., 2015; Mackay et al., 2013). In the skin, these key TRMs survival factors are supplied by keratinocytes, particularly by those in the infundibulum and isthmus (Adachi et al., 2015). Consistently, Skin TRMs and Tregs are enriched in the upper hair follicles and depend on IL-7 and IL-15 signaling (Adachi et al., 2015; Gratz et al., 2013)(Mackay et al., 2013) (Seneschal et al., 2012). Specific ablation of IL-7 from hair follicles leads to the loss of both CD4+ and CD8+ TRMs, while the lack of IL-15 results in the specific depletion of CD8+ TRMs, highlighting the unique and redundant functions of each of these cytokines.
The generation of a malignant lymphoma mouse model in which cancerous CD4+ lymphoma cells exhibit epidermotropism, a classic feature of human cutaneous T cell lymphoma, revealed the requirement for epidermal IL-7 in the epidermal persistence of lymphoma cells (Adachi et al., 2015). Thus, interrupting the supply of survival factors may be one way of halting cutaneous T cell neoplasms, but may come at the expense of host protection. Similarly, IL-15 plays a key role in the pathology of vitiligo, an autoimmune disease in which cytotoxic T cells aberrantly target melanocytes and cause skin depigmentation. (Richmond et al., 2018). Disrupting IL-15 signaling by targeting CD122, a subunit of IL-15 receptor on TRMs, leads to the depletion of melanocyte antigen-specific CD8+ TRMs from the skin and reverses disease phenotype in a mouse model of vitiligo. These studies highlight the keratinocyte-immune communication circuit as attractive therapeutic targets in inflammatory skin diseases and cutaneous T cell lymphoma.
Keratinocytes are also a vital source of survival signals for cells of the innate immune network. Colony stimulating factor 1 (CSF1) receptor signaling and its ligand CSF1 support macrophage development and maintenance. Similar to other tissue macrophages, LCs rely on CSF1 receptor signaling. However, rather than CSF1, LCs rely on another keratinocyte-derived CSF1 receptor ligand, IL-34, for their maintenance in the epidermal compartment (Greter et al., 2012; Wang et al., 2012). Loss of IL-34 specifically targets epidermal LCs while leaving dermal macrophage subsets unperturbed. The teleological reason for the divergent expression of CSF1 and IL-34 in the dermis and epidermis, respectively, is unclear. One enticing possibility is that the distinct spatial expression of CSFR ligands may control the functional specification of macrophage subsets in different skin layers. Indeed, despite the equivalent ability these two CSFR ligands to generate macrophages in vitro, the downstream functional responses of macrophages generated by IL-34 and CSF1 are significantly different (Boulakirba et al., 2018).
TGF-β signaling is essential for the retention of immune cells in epidermis and exerts its effects by enhancing the expression of integrins and other tissue tethering factors. Retention of LCs in the skin is critically dependent upon TGF-β signaling (Bobr et al., 2012). Inducible LC specific ablation of TGF-βRI or TGF-βRII results in mass-migration of LCs to regional lymph nodes without altering their inflammatory profile. TGF-β is secreted in its latent form, which requires activation by integrins αvβ6 and αvβ8 to exert biological activity (Munger et al., 1999). Mice that lack either of these integrins or their activities recapitulate the phenotypes of TGF-β null mice, including the absence of LCs (Aluwihare et al., 2009; Yang et al., 2007). Integrins αvβ6 and αvβ8 are each expressed by interfollicular and follicular keratinocytes, respectively, and transactivate latent TGF-β to maintain TGF-β signaling in LCs. Upon perturbation by UVB irradiation, keratinocytes downregulate the integrins, leading to disrupted TGF-β signaling that prompt LCs to emigrate from the epidermis (Mohammed et al., 2016). Genetic ablation or antibody-mediated blocking of integrins also results in loss of CD8+ TRMs. TGF-β is also necessary for the epidermal persistence of CD8+ TRMs and mediates its effects by upregulating integrin, alpha E (CD103) which engages E-cadherin expressed by adjacent keratinocytes (Mackay et al., 2013). CD103 interactions may also be relevant in cancer, as tumor cells are known to upregulate E-cadherin. Indeed, TRMs are directly linked to a better survival outcome of solid tumors of epithelial origin (Amsen et al., 2018). The enrichment of the TRM signature, particularly the expression of CD103 is predictive of longer survival in lung cancer (Ganesan et al., 2017). The importance of CD103 expression on TRMs is highlighted by a recent report that CD103-deficient CD8+ TRM to fail to suppress melanoma development (Malik et al., 2017) and that CD8+ TRMs maintain melanoma-immune equilibrium to suppress the outgrowth of lesions (Park et al., 2018). Thus, the TGF-β-CD103 axis is key to epidermal residency of immune cells that play vital roles in immune surveillance during homeostasis and in skin cancer
Immune cells facilitate hair growth by regulating follicle stem cell activation
Hair follicles undergo natural bouts of rest and regeneration. Transition through these phases is controlled by hair follicle stem cells (HFSCs) that reside in the bulge region of the hair follicle. Taking cues from their microenvironment or “niche”, HFSCs transition from a quiescent to an active state and back. (Hsu et al., 2014). In particular, HFSCs are largely regulated by suppressive BMP/FGF and activating Wnts emanating from various sources in the niche microenvironment. (Hsu et al., 2014; Plikus et al., 2008). Injury to the hair follicle, by hair plucking or depilation, also induces hair cycling to enable repair and grow hair, providing an additional shield following damage (Chen et al., 2015).
Immune cells have emerged as essential members of the HFSC niche where they provide vital signals to orchestrate hair cycling under homeostatic and stress conditions (Naik et al., 2018) (Figure 1). Skin-resident Tregs are found in close proximity to hair follicle stem cell area (bulge), and the absence of Tregs results in the delay of hair growth following chemical depilation. Tregs express the Notch ligand, Jagged 1, to presumably stimulate HFSC proliferation and promote hair growth (Ali et al., 2017). Supernatants from cultured Tregs preferentially promote HFSCs proliferation in vitro, when compared to supernatant from cultured naive T cells, further supporting a role for Tregs in stem cell proliferation (Lay et al., 2018). In vivo, whether the Tregs directly influence HFSC behavior or indirectly by modulating accessory cells requires further investigation. Additionally, the contribution of Tregs to natural hair cycling versus depilation or damage induced HFSC activation also demands attention. In the natural hair cycle, macrophages accumulate around the bulge throughout the resting phase, peaking before the activation of hair growth. These perifollicular macrophages secrete activating Wnts to facilitate HFSC activation (Castellana et al., 2014).
Follicular damage induced by hair-plucking leads to CCL2 production from hair follicle keratinocytes, to recruit TNF-producing macrophages that in turn activate the hair cycle. TNF spreads the news of this damage to adjacent undamaged follicles creating a gradient of activation around the injury (Chen et al., 2015). Precisely how TNF-induced activation in hair follicle stems cells occurs is unclear. However, the production of TNF following plucking is associated with AKT/β-catenin signaling in HFSCs (Wang et al., 2017), which could be either direct TNF signaling into HFSC or, more likely indirectly, via a niche cell which in turn produces activating Wnts to stimulate follicle SCs. Similar mechanisms involving signaling of immune cells into HFSC niche cells to augment Wnt production are involved in de novo generation of follicles. Following re-epithelialization of large wounds, the epidermis can generate novel hair follicles, a process termed follicle neogenesis. Hair follicle neogenesis is dependent on Wnt signals produced by fibroblasts (Ito et al., 2007). Dermal γδ T cells produce FGF9 to amplify Wnt signaling in the newly generated tissue and promote wound-induced hair neogenesis (Gay et al., 2013).
Immune-hair follicle interactions can also be deleterious as they underlie follicular damage. Indeed, genome-wide association studies for alopecia areata (AA), a prominent hair loss disorder, have revealed a number of susceptibility loci in immune modulatory genes (Petukhova et al., 2010). Disease pathology in AA is primarily mediated by cytotoxic T cells, which infiltrate the growing hair follicles and induce its premature regression, leading to the loss of hair (Pratt et al., 2017). Targeting CD8+ T cells by inhibition of IL-15 or the downstream JAK signaling can curb disease and promote hair growth (Xing et al., 2014) (Mackay-Wiggan et al., 2016). Detailed mechanisms of precisely how inflammatory T cells cause follicular damage and the triggers involved warrant further exploration.
To avoid adverse immune activity, the follicle has evolved a number of immune evasive mechanisms including upregulation of immune-dampening factors and reduced expression of immune-activating molecules (Paus and Bertolini, 2013). For instance, Agudo and colleagues recently uncovered that HFSCs down regulate expression of MHC class I to evade detection by T cells (Agudo et al., 2018). Thus, a complex and context specific volleying between the hair follicle and immune cells regulates the growth dynamics of this epidermal appendage, extending the role of the immune system in the skin beyond host-defense, to the regulation of tissue architecture.
Keratinocytes are sensors and immune sentinels
In addition to their contributions in forming physical boundaries of our body, epithelial cells in barrier organs such as the skin, lung and gut, are essential for calibrating the magnitude and type of local immune response. These cells express a range of sensors that recognize microbial elements as well as sterile stressors and relay this information by producing secondary mediators (cytokines, chemokines, growth factors) to alert the host to a state of emergency.
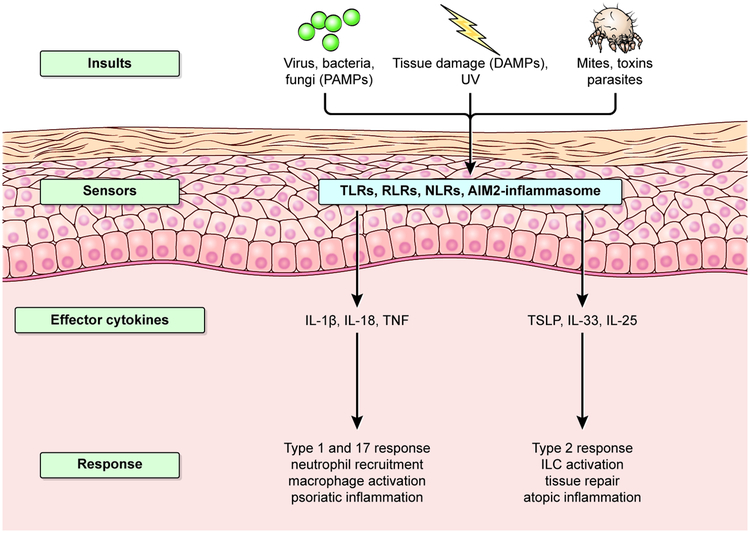
Keratinocytes are equipped with a sophisticated network of sensors that recognize insults through pattern recognition receptors (PRRs) such as Toll-like receptors (TLRs), Rig-like receptors (RLRs), NOD-like receptors (NLRs) and inflammasomes. These machineries enable sensing of foreign or microbial agents (Pathogen-associated molecular patterns: PAMPs) and tissue damage (Damage-associated molecular pattern: DAMPs) (Figure 2). Engagement of TLRs, RLRs and NLRs leads to the activation of NF-κB and/or mitogen-activated protein kinases (MAPKs) (Kawai and Akira, 2009). TLRs sense cell surface and endosomal PAMPs, whereas RLRs sense cytosolic PAMPs, and the engagement of NLR activate inflammasomes. These events ultimately lead to the production of proinflammatory chemokines and cytokines such as IL-1β, IL-18 and TNF that activate host immunity, including type 1 and 17 adaptive immune responses that protect against infections.
Figure 2: Perturbations to keratinocytes trigger and harness immune responses.
Keratinocytes sense microorganisms and other environmental stressors through Toll-like receptors (TLRs), Rig-like receptors (RLRs), NOD-like receptors (NLRs) and AIM2-inflammasomes and transmit the signals to immune cells. Upon infection by intracellular pathogens such as bacteria and viruses, keratinocytes produce IL-1β and TNF to initiate type 1 and type 17 immune responses, which further leads to the recruitment of neutrophils and activation of macrophages. Extracellular parasites, toxins and tissue damage promote type 2 immune responses via epithelial cell-derived alarmins TSLP, IL-33 and IL-25. The immunological pathways that are poised against infectious agents, when dysregulated, play central roles in inflammatory skin diseases such as psoriasis and atopic dermatitis.
Another innate sensing pathway that has a dominant role in skin immunity is IL-1R which signals via myeloid differentiation factor 88 (MyD88) family of adapter proteins. IL-1R plays a vital role in damage-induced responses that accompany barrier disruption. This signaling pathway synergizes with TLRs to amplify innate immune response against epidermis-penetrating infectious agents. In S. aureus skin infection models in mice, MyD88-deficient mice have an impairment in bacterial clearance due to altered neutrophil recruitment (Miller et al., 2006). The induction of IL-17A producing T cells, which are essential for defense against S. aureus, is dependent upon TLR2 and IL-1R signaling (Cho et al., 2010). Importantly, S. aureus-induced skin inflammation is abrogated by specific-depletion of MyD88 in keratinocytes (Nakagawa et al., 2017) or T cells (Liu et al., 2017). MyD88 signals have unique roles in these distinct cellular compartments. In cutaneous T cells, it promotes type 17 responses (Naik et al., 2012), while in keratinocytes, it activates a program of innate immunity (Cataisson et al., 2012). There is also a downstream crosstalk between MyD88 induced pathways in T cells and keratinocytes. Type 17 immune responses enhance keratinocyte production of antimicrobial peptides, such as beta defensin 2 and S100A9, thereby promoting antimicrobial host defense (Liang et al., 2006) (Miller and Cho, 2011)(Naik et al., 2015).
The unique involvement of TLRs in pathogenesis of skin diseases can be highlighted in contact hypersensitivity (CHS) responses, mouse models for allergic contact dermatitis. The contact allergen Ni2+ binds to human, but not mouse TLR4, and thus mice do not get sensitized to Ni2+. However, when TLR4−/− mice are genetically engineered to express human TLR4, they exhibit Ni2+-induced CHS (Schmidt et al., 2010). Metal allergy to Cr2+, another common contact allergen, involves both TLRs and inflammasomes. TLR-signaling mediates an initial priming phase to induce pro-IL-1β synthesis and the subsequent activation of the inflammasome component NLRP3 mediated by reactive oxygen species leads to the processing of pro-IL-1β into active forms (Adam et al., 2017).
Because keratinocytes secrete cytokines that can trigger cascades of inflammation, there has been regained interest in the role of keratinocytes in the pathogenesis of inflammatory skin diseases. Psoriasis is a chronic inflammatory skin disease that is mediated by several proinflammatory cytokines including IL-1β, TNF and IL-6 and type 17 cytokines (Dainichi et al., 2018; Nestle et al., 2009). While immune cell-derived cytokines are undoubtedly crucial for pathogenesis, it has been reported that dysregulated signaling pathways in keratinocytes alone can induce psoriatic inflammation. Keratinocyte-specific dysregulation of signaling pathways including NF-kB, AP-1/JUN, STAT3/SOCS3 and CARD14 results in the development of psoriatic dermatitis in mice, supporting a role for keratinocytes in initiating or amplifying proinflammatory cascades in psoriasis (Kumari et al., 2013; Pasparakis et al., 2002; Sano et al., 2005; Wang et al., 2018; Zenz et al., 2005). Conversely, deletion of TRAF6, a molecule downstream of NF-kB, from the epidermis leads to reduced production of type 17 cytokines and attenuated phenotype in the imiquimod-induced mouse model of psoriasis (Matsumoto et al., 2018). Studies in murine models of psoriasis have revealed that keratinocytes are a source of the p19 subunit of IL-23, IL-24, and IL-25 (IL-17E) that may initiate and/or potentiate inflammation and disease pathology (Kumari et al., 2013; Li et al., 2018; Xu et al., 2018). Additional studies are required to elucidate the relative contribution of each of these keratinocyte-derived cytokines to psoriasis, particularly in the therapeutic context.
In addition to sensing exogenous agents and immune mediators, keratinocytes also sense DAMPs, such as self-DNA, via a slew of cytoplasmic sensors such as AIM2s (Dombrowski et al., 2011). This sensing is amplified when self-DNA interacts with keratinocyte-derived DAMPs such as LL-37. The active form of cathelicidin, LL37, interacts with self-DNA to activate plasmacytoid DCs through TLR9, which results in the production of type l interferons that promotes Th17 cell activation (Lande et al., 2007) and tissue repair (Gregorio et al., 2010). LL37 also influences AIM2-inflammasome formation and IL-1β processing and secretion by keratinocytes (Dombrowski et al., 2011). Understanding how a dysregulation of nucleic acid sensing pathways may be a tipping point for breaking tolerance to self-DNA could provide unique insights into the initiation of autoinflammation. The roles of other DAMPs, such as HMGB1 and S100 proteins during skin inflammation in vivo still remain incompletely understood.
Keratinocytes boost type 2 immunity
In addition to the release of proinflammatory cytokines that enhance type 1 and type 17 immunity, epithelial cells produce type 2 cytokines which initiate immune responses against parasitic infections, contact allergens, mites, toxins, and promote tissue repair (Iwasaki and Medzhitov, 2015) (Figure 2). When unchecked, keratinocyte-triggered type 2 immune responses can drive pathology in atopic dermatitis, which was long regarded as solely an IgE-mediated allergic response. Accumulating evidence has now shifted this long-held view to support the roles for barrier disruption and shifts in the composition of microbiota (dysbiosis) as upstream drivers of atopic inflammation. In this regard, keratinocyte-derived alarmins, such as TSLP, IL-33, and IL-25 have been studied as prototypic cytokines that boost type 2 innate and adaptive immune responses.
TSLP was cloned from mouse thymic stromal cells and was initially studied in the context of lymphocyte development and differentiation (Sims et al., 2000). Its potent biological effects in type 2 immunity were unveiled when TSLP was found to be highly expressed by keratinocytes from patients with atopic dermatitis (Soumelis et al., 2002). TSLP activates CD11c+ DCs and induces production of the chemokines CCL17 and CCL22. TSLP-treated DCs effectively prime CD4+ naive T cells to produce the type 2 cytokines, IL-5 and IL-13 (Soumelis et al., 2002).
Keratinocyte-specific overexpression or deletion of TSLP in mice illustrates the importance of keratinocyte-derived TSLP as a prominent driving force of type 2 immune responses and atopic inflammation in skin (Leyva-Castillo et al., 2013; Yoo et al., 2005). TSLP promotes the activation of a variety of type 2 immune cells including Th2, basophils and mast cells (Ziegler and Artis, 2010). In a vitamin D3 analogue (MC903)-induced mouse model of atopic inflammation, the expansion of ILC2 and atopic inflammation is dependent on TSLP, suggesting the crucial role of TSLP-ILC2 axis in atopic dermatitis (Kim et al., 2013). It is of interest to note that keratinocyte-derived TSLP acts directly on a subset of sensory neurons to trigger itch behaviors (Wilson et al., 2013). Concomitant with the induction of type 2 immunity is the promotion of a Treg programs by TSLP (Kashiwagi et al., 2017). Topical MC903 application has the remarkable capacity to expand systemic Treg cell levels, underscoring the multifaceted mechanisms by which TSLP exerts its effects.
Epithelia-derived IL-25 and IL-33 are also potent activators of ILC2 that contribute to the expulsion of parasitic worms in the gut (Huang et al., 2015; Moro et al., 2010). This axis appears to also be active in the skin, where IL-25 and IL-33 signaling is essential for ILC2 expansion during MC903-induced atopic inflammation (Salimi et al., 2013) and the forced expression of IL-33 in keratinocytes induces atopic inflammation with ILC2 expansion (Imai et al., 2013). Taken together, these studies establish that TSLP, IL-25, and IL-33 are keratinocyte-derived immune signals that activate resident innate immune cells, particularly ILC2, consequently leading the recruitment of other type 2 effectors. It is noteworthy that transcriptional profiling of ILC2s from different organs reveals dependency of ILC2 on distinct tissue-derived signals (Ricardo-Gonzalez et al., 2018). It was recently reported that a subset of skin ILC2s can also be activated by IL-18, a cytokine that was previously associated with type 1 immunity. In this regard, ILCs, which are currently classified by their cytokine profiles, may exhibit distinct characteristics that are shaped by the tissues and even the micro niches of the tissues they reside in. Future studies examining keratinocyte-specific sensing mechanisms of allergens, toxins and noxious agents are necessary to understand formation of type 2 immunity in the skin epithelium.
Immune cell communication with neurons
In addition to epithelium-immune cell crosstalk, neuroimmune communications are emerging as instructive dialogues for informing skin immunity. A number of sensory neurons, extending from the dorsal root ganglion, innervate the epidermis and hair follicles to detect the presence of noxious or harmful stimuli and provide mechanosensation (Abraira and Ginty, 2013; Dubin and Patapoutian, 2010). Sensory neurons that express the transient receptor potential cation channel subfamily V (TRPV), detect potentially damaging stimuli and also interact with dermal DCs in type 17 skin inflammation such as psoriatic inflammation and Candida albicans infection (Kashem et al., 2015; Riol-Blanco et al., 2014). In this setting, sensory neurons drive IL-23 production from dermal DCs and promote the expansion of IL-17-producing T cells. This neuron-DC interaction is mediated by neuropeptide calcitonin gene-related peptide (CGRP) that sensory neurons produce in response to infections (Kashem et al., 2015). Many questions remain. What other neuropeptides are involved in neuron-mediated activation of DCs? How do neurons interface with other tissue resident immune cells such as ILCs, which are known to form neuroimmune circuitry after exposure to parasite infections or allergens (Veiga-Fernandes and Artis, 2018)? S. aureus skin infections also induces pain sensation through nociceptors, presumably as an alarm to inform the host of its compromised positions (Chiu et al., 2013). Thus, bidirectional regulation of the immune and nervous components of the skin serve not only to boost immunity, but also may alter host behavior to limit the spread of invading pathogens.
Recent studies on neuroimmune communications have provided new insights into the mechanisms of itch, which serves as a physiological response to expel noxious substances. However, in disease settings such as atopic dermatitis, it causes significant impairment in the quality of life in patients. Sensory neurons express receptors for the type 2 cytokines IL-4 and IL-13, engagement of which activates itch-sensory pathways via TRP channel-dependent calcium influx (Oetjen et al., 2017). In the MC903-induced atopic inflammation model, signaling through IL-4Rα and JAK1 in neurons is required for inducing both itch behavior and dermatitis. Lymphocyte-derived IL-31 is also known as a pruritogenic or itch-inducing cytokine, and its receptor is expressed broadly on epithelial cells, immune cells and neurons. Analysis of mice overexpressing IL-31 showed the link of IL-31 with type 2 immune-mediated inflammation and itch (Dillon et al., 2004) (Cevikbas et al., 2014). While IL-31 is dispensable for mounting CHS responses (Takamori et al., 2018), it is important to note that human allergic contact dermatitis, which CHS is a model for, is an entity distinct from atopic dermatitis. These data emphasize the central role of neuroimmune communication in type 2 immunity-mediated itch and dermatitis, further providing a foundation for novel therapeutic strategies for chronic itch. Indeed, targeting IL-4R, IL-31R, and JAKs represent breakthrough therapies for atopic dermatitis.
Cross regulation of host immunity and the microbiota in health and disease
Advances in genomic sequencing technology have enabled a sophisticated and in-depth phylogenetic analysis of microbes residing on skin surface and in its appendages (Grice et al., 2009). These seminal studies revealed that the skin houses one of the most diverse communities of microbes across various heterogeneous niches (Byrd et al., 2018; Costello et al., 2009; Grice et al., 2009). Research in the past decade has fundamentally advanced our understanding of how the skin’s microbes influence local immunity and provided a huge momentum in barrier-surface immunology in mice and humans, and in health and disease.
No longer considered passive bystanders, skin resident microbes actively engage and tune host immunity to boost barrier function (Figure 3). Mice raised in germ-free conditions display a marked reduction in skin effector T cells and an enrichment of Tregs (Naik et al., 2012). Germ-free mice also display transcriptional differences in a wide range of genes associated with epidermal differentiation, TLRs, chemokines and cytokines (Meisel et al., 2018). Notably, the generation of gnotobiotic mice that are colonized with a single human skin-commensal species, S. epidermidis, rescues the balance of effector T cells and Tregs, in a manner that is dependent on the IL-1 and MyD88 pathway, but independent of the gut microbiota (Naik et al., 2012).
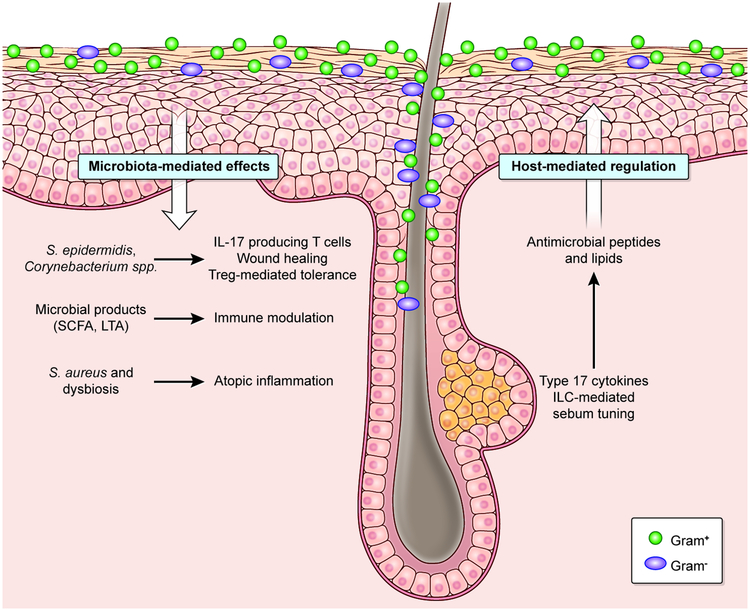
Figure 3: Dialogue between the microbiota and immune cells in skin.
Resident Commensal bacteria are required for tuning type 17 responses and promoting wound healing in the skin., Commensal derived the short-chain fatty acid (SCFA) and lipoteichoic acid (LTA) modulate immunity. Tregs generated against commensal bacteria early in life enable symbiosis with the host. The immune cells bolster the epidermal barrier by by enhancing the production of antimicrobial peptides and lipids to limit bacterial penetration and community dysbiosis. This effect is regulated by immune derived -type 17 cytokines and ILC-mediated regulation of sebum. The dialogue between the host and microbiota is discordant in inflammatory skin diseases such as atopic dermatitis, wherein dysbiosis, predominated by Staphylococcus aureus, drives atopic skin inflammation.
We now understand that specific microbes engage unique immune programs in the skin, and also induce the formation of antigen-specific T effectors. Topical association of specific pathogen free mice (SPF) with a human commensal S. epidermidis isolate induces a unique population of IL-17 production by CD8+ T cells (Tc17) (Naik et al., 2015). These cells are highly reminiscent of Tc17s from human skin and non-human primate skin. Cross-presentation by CD103+ DCs is required for the induction of antigen-specific Tc17s, and IL-1 from CD11b+ DCs supports IL-17 production (Naik et al., 2015). The induction of IL-17-producing CD8+ T cells (Tc17) is specific to the colonization of certain strains of S. epidermidis and is not observed with other skin-associated bacterial species including Staphylococcus xylosus, Cutibacterium (Propionibacterium) acnes, and S. aureus. Importantly, S. epidermidis-specific Tc17s provide heterologous protection against the yeast pathogen, C. albicans, by boosting the expression of keratinocyte antimicrobial peptides. Although the molecular details of how and why a commensal bacterium elicits a Tc17 response remain elusive, one possibility is the enrichment of lytic phages in certain strain of bacteria (for example S. epidermidis) that engage this particular facet of the skin’s immune repertoire.
Studies have begun to elucidate how the epithelial barrier senses species-level differences to harness distinct immune responses. Lipoteichoic acid (LTA), a cell wall component of gram-positive cocci, suppresses TLR3-mediated inflammation during tissue injury by binding to TLR2, thereby inducing TRAF1, a negative regulator of TLR-mediated NF-kB signaling (Lai et al., 2009). Although it is unclear if TLR2 and TLR3 exert their effects on similar or different cells in the skin, these findings underscore the utility of commensal byproducts for modulating immune responses. Members of Corynebacterium species promote the expansion of IL-17-producing γδ T cells through mycolic acid, a component of the cell envelope (Ridaura et al., 2018). In human skin, C. acnes produce short chain fatty acids (SCFAs) in anaerobic conditions with a lipid substrate for fermentation, and SCFA-mediated histone deacetylase inhibition modulates inflammatory response of epidermal keratinocytes to TLR ligands (Sanford et al., 2016). Investigating how commensal-derived signals including bacterial elements and their metabolites prime skin immunity, and the precise receptor-ligand interactions involved, require further exploration.
Detailed mechanisms on how host immunity regulates commensal bacteria in the steady state are now being revealed (Figure 3). Physical barriers including hair and stratum corneum, and chemical barrier such as antimicrobial peptides and lipids, restrict the overgrowth of bacteria on skin surface and prevent their invasion into the viable layers (Chen et al., 2018; Gallo and Hooper, 2012). This epithelium-mediated control of bacterial commensalism is collaboratively adjusted by tissue-resident innate and adaptive immune cells. Rag-deficient mice lacking an adaptive immunity display a shift in the composition of skin-surface microbes and translocation of surface bacteria to skin draining lymph nodes (Shen et al., 2014). Similarly, patients with immunodeficiencies have significant shifts in composition of their skin commensals (Oh et al., 2013).
ILCs are strategically positioned in a distinct epidermal compartment to monitor and tune the microbiota. Hair follicles guide CCR6+ epidermal ILCs to localize in close proximity of the sebaceous glands. There, ILCs produce TNF receptor ligands that regulate sebocyte proliferation by repressing Notch signaling. As a result, the production of antimicrobial free fatty acids is suppressed, enabling the commensalism of gram-positive cocci. In the absence of ILCs, the sebaceous glands undergo hyperplasia with increased production of free fatty acids, resulting in the restriction of gram-positive cocci commensalism and the relative abundance of anaerobic bacteria. By contrast, absence of lymphocytes leads to increased commensalism of gram-positive cocci (Kobayashi et al., 2019). In addition to the classical view of immunity as merely an anti-pathogen system, these studies highlight the homeostatic role of barrier immune cells in regulating microbial equilibrium by exerting their influence on the tissue’s parenchyma.
Suppressing untoward immune responses against commensals is necessary to ensure stable host-microbe symbiosis. Among the variety of mechanisms that promote immune tolerance against commensal microbes, Tregs appear to play an important role in skin. Interestingly, immune tolerance to skin commensals occurs within a specific window of time during organismal development in an antigen-specific manner. Colonization of S. epidermidis induces the expansion of commensal-specific Tregs in neonatal mice but not in adult mice (Scharschmidt et al., 2015). In the gut, ILCs limit pathological adaptive immune responses against intestinal commensal-bacteria. This regulatory function is mediated via MHC class II (MHC II) expressed by ILCs that induce the cell death of commensal-specific CD4+ T cells (Hepworth et al., 2013). Although MHC ll expression on CCR10+ skin ILCs has been reported (Yang et al., 2016), whether this MHC ll-mediated regulation of commensal-specific CD4+ T cells is operative in skin has yet to be determined.
Given crucial roles of the microbiota in tuning the skin’s immune system, it is not surprising that the imbalance of resident microbiota, termed dysbiosis, has emerged as a pivotal driver of inflammatory skin diseases. Atopic dermatitis is a chronic inflammatory disease with underlying barrier disruption resulting in eczematous dermatitis and relentless itch. When left uncontrolled, it may lead to the sequential onset of asthma and food allergies, known as the “atopic march” (Bieber, 2008). Enrichment of S. aureus in active atopic dermatitis flares has been observed clinically for decades (Leyden et al., 1974). Cutting-edge 16S rRNA and whole genome sequencing of longitudinal analysis of microbes from atopic dermatitis patients revealed that the dysbiosis that was predominated by S. aureus and in part, S. epidermidis, during active disease (Byrd et al., 2017; Kong et al., 2012). However, whether S. aureus is a key driver of disease pathogenesis or is merely a result of chronic inflammation is still actively debated. Mice lacking the transmembrane proteinase, a disintegrin and metalloproteinase 17 (ADAM17) in epidermis, spontaneously develop eczematous dermatitis with features that mimic human atopic dermatitis. Skin inflammation is preceded by dysbiosis that is predominated by S. aureus and Corynebacterium spp., in relatively low-health status housing conditions. Antibiotics that target the dysbiotic flora results in regained microbial diversity and extinguishes skin inflammation. Whereas inoculation of each dysbiotic flora revealed S. aureus to be the main driver of skin inflammation, a Corynebacterium species induced prominent T helper 2 responses (Kobayashi et al., 2015). This observation not only indicates that S. aureus is a critical for eczema formation, but also suggests that a dysbiotic flora drives Th2 responses that may lead to the atopic march. Several possible mechanisms by which S. aureus drive atopic inflammation have been reported. S. aureus-derived molecules δ-toxin (Nakamura et al., 2013) and phenol soluble modulin-ɑ (Liu et al., 2017) (Nakagawa et al., 2017) induce skin inflammation directly via cytotoxic effects or indirectly through the stimulation of immune cells. S. aureus can also directly activate sensory neurons independent of immune cells, in part via bacteria-derived molecules, suggesting a direct link between S. aureus and itch sensation (Chiu et al., 2013).
The characteristics of S. aureus that colonize atopic skin warrants further exploration since the ability of S. aureus to induce immune responses is strain-dependent (Byrd et al., 2017). Furthermore, clinical evidence for the efficacy of antimicrobial treatments is still controversial, and whether or not a specific targeting of S. aureus enables disease control is under active investigation. Decreased abundance of certain bacterial species in atopic skin might also contribute to the susceptibility to S. aureus colonization and disease activity. It is possible that the restoration of such commensals can mitigate dysbiosis-mediated inflammation. Recently, topical colonization of coagulase-negative Staphylococcus (Nakatsuji et al., 2017) and Gram-negative bacteria (Myles et al., 2018) have been reported to inhibit S. aureus colonization or to decrease disease severity in human atopic dermatitis. Collectively, these studies open the door for microbiota-based therapeutic approaches in atopic dermatitis.
Immune cells orchestrate tissue repair
Tissue repair is a complex biological process that involves a cooperative action of a variety of immune and non-immune cells. Epithelialization, fibrosis, and angiogenesis are primarily mediated by epithelial and stromal cells, which occur in coordination with tissue-resident immune cells (Figure 1).
γδ T cells are enriched at barrier sites and constitute a unique lymphoid population that govern epithelial homeostasis and tissue repairs. Skin lacking γδ T cells shows pronounced delays in wound healing with increased apoptosis of keratinocytes. This is likely related to the deprivation of growth factors, including FGFs and IGF produced by γδ T cells (Jameson et al., 2002; Sharp et al., 2005). Specific lack of Vγ5Vδ1+ epidermal γδ T cells is sufficient to impair re-epithelialization after wounding, lending support for importance of interaction between keratinocyte and γδ T cell in tissue repair (Keyes et al., 2016). Interestingly, keratinocyte-T cell communication is impaired in aged skin, leading to decline of wound repair. This aging-related impaired crosstalk is attributed to defects of Skint family members in aged keratinocytes, and the expression of Skints is controlled by STAT3 signaling, which is enhanced by IL-6 (Keyes et al., 2016). These findings support a previous report that keratinocyte-specific defects of STAT3 leads to delays in wound-healing (Sano et al., 1999) and reveal an epithelial-immune cell interactive machinery, which involves both intrinsic and extrinsic factors, during wound healing process. Although human skin does not contain an analogous population of Vγ5Vδ1+ epidermal γδ T cells, it is important to note that cutaneous T cells isolated from human skin similarly produce epithelial growth factors upon wounding (Toulon et al., 2009).
γδ T cells are not the only immune cell subset important for promoting tissue repair. ILCs and LC can produce EGFR ligands, which are important regulators for keratinocyte proliferation and differentiation, these skin-resident immune cells can differentially contribute to the maintenance of epithelial barrier maintenance and repair (Rak et al., 2016; Shipman et al., 2018). Tregs facilitate repair in the lung and muscle by producing the EGFR ligand amphiregulin (Arpaia et al., 2015; Burzyn et al., 2013). In the skin, EGFR signaling on Tregs also seems to be important in wound repair, underscoring the complex nature of EGFR circuits in keratinocyte-immune cell crosstalk (Nosbaum et al., 2016). This crosstalk is not limited to interactions between host cells and also involves our microbial partners. Commensal bacteria influence Tc17 cells to promote tissue repair. Colonization of S. epidermidis induces Tc17 cells in a non-classical MHC class I-restricted manner. The transcriptome of commensal-specific skin Tc17s is enriched for immunoregulatory and tissue-repair signature, including genes of EGFR ligands and FGFs, and accelerate wound healing (Linehan et al., 2018). Together with the previous report that a bacterial product of S. epidermidis, lipoteichoic acid (LTA), promotes wound healing by mitigating inflammation through binding to TLR2 (Lai et al., 2009), this finding highlights the microbe-mediated tuning of immune functions in tissue repair.
Macrophages have also been long-recognized as a resident-immune cell that regulate tissue repair by interacting with stromal cell populations. Tissue-resident macrophages produce a wide range of growth factors and immune mediators to promote tissue repair. Macrophages that are activated by type 2 cytokines such as IL-4 and IL-13 produce MMPs, PDGF and TGF-β, all of which are closely involved in extracellular matrix regulation and fibroblast growth during wound healing (Gieseck et al., 2018). In skin wounds, IL-4Rα signaling in macrophages is critical to control collagen fibril assembly, which is likely mediated by macrophage-producing Relm-α (Knipper et al., 2015). In contrast to dendritic cell subsets, characteristics and functions of skin-resident macrophages remains ill-defined. Initially appreciated for their phagocytic activity, recent research is beginning to highlight the critical roles of macrophages in tissue development, homeostasis and metabolism (Okabe and Medzhitov, 2016). In addition, given that macrophages modulate extracellular matrix synthesis and degradation, they are thought as key immune cells in tissue fibrosis (Gieseck et al., 2018) and promote the functional heterogeneity of fibroblasts that enables dermal healing (Shook et al., 2018). A more in-depth characterization of the skin’s macrophage subsets and the establishment of better tools to target specific subsets of macrophages in skin will vastly improve our understanding of the pathological fibrotic conditions driven by macrophage-fibroblast circuits, such as chronic cutaneous graft-versus-host-disease and scleroderma.
Immune memory is a fundamental mechanism that effectively mounts host resistance to reinfections. Classical immune memory has been attributed to clonal expansion of memory T and B cells, which confers long-term antigen-specific protection. The view that immune memory is an exclusive feature of adaptive immune cells has clearly changed with the emergence of the concept on trained memory that is established through epigenetic reprogramming (Netea et al., 2016). Training by inflammatory or microbial signals of innate immune cells such as macrophages and NK cells enhances non-specific responses against secondary challenges. More recently, skin epithelial stem cells were shown to retain the memory previous inflammation at the level of chromatin (Naik et al., 2017). Acute psoriasis-like inflammation induced via topical application of imiquimod, a TLR7 agonist, results in enhanced wound healing months after resolution of initial stimuli. Resident immune cells are dispensable to boost repair in the inflammation-experienced skin, which is mediated by prolonged memory in epithelial stem cells. Inflammation-trained epithelial stem cells maintaining accessibility at key stress response genes, which are more rapidly activated by subsequent injury and re-epithelialize wounds at a faster rate. Human chronic allergic rhinitis resulting from type 2 inflammation has recently been shown to effect basal epithelial stem cells by increasing Wnt/β-catenin signaling in the airways (Ordovas-Montanes et al., 2018). Basal stem cells from inflamed airways are more responsive to secondary inflammatory cues in vitro, even after prolonged periods of rest, than control basal cells, indicating the retention of functional inflammatory memory. The principle of immune memory in epithelial cells represents an exciting advance in our understanding of barrier tissue homeostasis, which may further help dissect the mechanisms of chronic or recurring inflammation in skin diseases. In this regard, it would be of interest to determine the longevity of epithelial memory and, from a therapeutic standpoint, explore mechanisms that could reset or “erase” the memory.
Conclusion
We have discussed recent developments in skin immunity studied in the context of health and disease, which, in aggregate, highlight that the skin immune system is maintained through tissue-dependent mechanisms that involve an intricate crosstalk between the parenchyma and immune cells. In particular, skin epithelia send signals that organize trafficking and positioning of immune cells and determine the fate of immune responses. In turn, immune cells ensure barrier homeostasis. Host classical and non-classical immunity is empowered by the microbiota, and the immune cells, in coordination with the epithelia, keep the microbes in check to maintain microbial equilibrium, forming a sophisticated barrier system that ensure stable symbiosis. This symbiotic relationship is altered in individuals with genetic conditions with impaired epidermal barriers, resulting in dysbiosis that drives chronic inflammation. Atopic dermatitis is prototypic of such condition, but future studies may reveal other diseases in which failure to establish healthy symbiosis during developmental and post-developmental processes is involved. Epithelia, neurons, and immune cells form coordinated units where they communicate to formulate productive immune responses. Destabilization of these dialogues is tied to the development of chronic inflammatory skin diseases. Studies utilizing cutting-edge techniques such as single-cell genomic and metagenomic analyses, and the integration of immunology, microbiology, and structural biology have helped us open the door to a new realm of research in tissue immunity. Further exploration the tissue-immune dialogue will not only deepen our understanding of inflammatory diseases, but also unearth the importance of this crosstalk for tissue repair and regeneration.
Kobayashi, Naik, and Nagao discuss recent findings on skin immunity that highlight that the skin immune system is maintained through intricate crosstalk between the parenchyma, immune cells and microbiota, all of which are crucial components for boosting healthy skin function and driving pathology in disease.
Acknowledgement
We apologize to our colleagues in immunology and skin biology whose work we could not cite due to space constraints. This work was supported by the Division of Intramural Research of NIAMS and by grants from NIAID (1K22AI135099–01 S.N.) and the Damon Runyon Cancer Foundation (DFS-30–18 S.N.)
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Declaration of Interests
The authors declare no competing interests.
References
- Abraira Victoria E., and Ginty David D. (2013). The Sensory Neurons of Touch. Neuron 79, 618–639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adachi T, Kobayashi T, Sugihara E, Yamada T, Ikuta K, Pittaluga S, Saya H, Amagai M, and Nagao K (2015). Hair follicle-derived IL-7 and IL-15 mediate skin-resident memory T cell homeostasis and lymphoma. Nat. Med 21, 1272–1279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adam C, Wohlfarth J, Haussmann M, Sennefelder H, Rodin A, Maler M, Martin SF, Goebeler M, and Schmidt M (2017). Allergy-Inducing Chromium Compounds Trigger Potent Innate Immune Stimulation Via ROS-Dependent Inflammasome Activation. J. Invest. Dermatol 137, 367–376. [DOI] [PubMed] [Google Scholar]
- Agudo J, Park ES, Rose SA, Alibo E, Sweeney R, Dhainaut M, Kobayashi KS, Sachidanandam R, Baccarini A, Merad M, and Brown BD (2018). Quiescent Tissue Stem Cells Evade Immune Surveillance. Immunity 48, 271–285.e275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ali N, Zirak B, Rodriguez RS, Pauli ML, Truong HA, Lai K, Ahn R, Corbin K, Lowe MM, Scharschmidt TC, et al. (2017). Regulatory T Cells in Skin Facilitate Epithelial Stem Cell Differentiation. Cell 169, 1119–1129.e1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aluwihare P, Mu Z, Zhao Z, Yu D, Weinreb PH, Horan GS, Violette SM, and Munger JS (2009). Mice that lack activity of alphavbeta6- and alphavbeta8-integrins reproduce the abnormalities of Tgfb1- and Tgfb3-null mice. J. Cell Sci 122, 227–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amsen D, van Gisbergen K, Hombrink P, and van Lier RAW (2018). Tissue-resident memory T cells at the center of immunity to solid tumors. Nat. Immunol 19, 538–546. [DOI] [PubMed] [Google Scholar]
- Arpaia N, Green JA, Moltedo B, Arvey A, Hemmers S, Yuan S, Treuting PM, and Rudensky AY (2015). A Distinct Function of Regulatory T Cells in Tissue Protection. Cell 162, 1078–1089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bieber T (2008). Atopic dermatitis. N. Engl. J. Med 358, 1483–1494. [DOI] [PubMed] [Google Scholar]
- Bobr A, Igyarto BZ, Haley KM, Li MO, Flavell RA, and Kaplan DH (2012). Autocrine/paracrine TGF-beta1 inhibits Langerhans cell migration. Proc. Natl. Acad. Sci. U. S. A 109, 10492–10497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boulakirba S, Pfeifer A, Mhaidly R, Obba S, Goulard M, Schmitt T, Chaintreuil P, Calleja A, Furstoss N, Orange F, et al. (2018). IL-34 and CSF-1 display an equivalent macrophage differentiation ability but a different polarization potential. Sci. Rep 8, 256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burzyn D, Kuswanto W, Kolodin D, Shadrach JL, Cerletti M, Jang Y, Sefik E, Tan TG, Wagers AJ, Benoist C, and Mathis D (2013). A special population of regulatory T cells potentiates muscle repair. Cell 155, 1282–1295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Byrd AL, Belkaid Y, and Segre JA (2018). The human skin microbiome. Nat. Rev. Microbiol 16, 143–155. [DOI] [PubMed] [Google Scholar]
- Byrd AL, Deming C, Cassidy SKB, Harrison OJ, Ng WI, Conlan S, Belkaid Y, Segre JA, and Kong HH (2017). Staphylococcus aureus and Staphylococcus epidermidis strain diversity underlying pediatric atopic dermatitis. Sci. Transl. Med 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castellana D, Paus R, and Perez-Moreno M (2014). Macrophages contribute to the cyclic activation of adult hair follicle stem cells. PLoS Biol 12, e1002002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cataisson C, Salcedo R, Hakim S, Moffitt BA, Wright L, Yi M, Stephens R, Dai RM, Lyakh L, Schenten D, et al. (2012). IL-1R-MyD88 signaling in keratinocyte transformation and carcinogenesis. J. Exp. Med 209, 1689–1702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cevikbas F, Wang X, Akiyama T, Kempkes C, Savinko T, Antal A, Kukova G, Buhl T, Ikoma A, Buddenkotte J, et al. (2014). A sensory neuron-expressed IL-31 receptor mediates T helper cell-dependent itch: Involvement of TRPV1 and TRPA1. J. Allergy Clin. Immunol 133, 448–460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen CC, Wang L, Plikus MV, Jiang TX, Murray PJ, Ramos R, Guerrero-Juarez CF, Hughes MW, Lee OK, Shi S, et al. (2015). Organ-level quorum sensing directs regeneration in hair stem cell populations. Cell 161, 277–290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen YE, Fischbach MA, and Belkaid Y (2018). Skin microbiota-host interactions. Nature 553, 427–436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiu IM, Heesters BA, Ghasemlou N, Von Hehn CA, Zhao F, Tran J, Wainger B, Strominger A, Muralidharan S, Horswill AR, et al. (2013). Bacteria activate sensory neurons that modulate pain and inflammation. Nature 501, 52–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho JS, Pietras EM, Garcia NC, Ramos RI, Farzam DM, Monroe HR, Magorien JE, Blauvelt A, Kolls JK, Cheung AL, et al. (2010). IL-17 is essential for host defense against cutaneous Staphylococcus aureus infection in mice. J. Clin. Invest 120, 1762–1773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christoph T, Muller-Rover S, Audring H, Tobin DJ, Hermes B, Cotsarelis G, Ruckert R, and Paus R (2000). The human hair follicle immune system: cellular composition and immune privilege. Br. J. Dermatol 142, 862–873. [DOI] [PubMed] [Google Scholar]
- Clark RA (2015). Resident memory T cells in human health and disease. Sci. Transl. Med 7, 269rv261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collin M, McGovern N, and Haniffa M (2013). Human dendritic cell subsets. Immunology 140, 22–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Constantinides MG, Linehan JL, Sen S, Shaik J, Roy S, LeGrand JL, Bouladoux N, Adams EJ, and Belkaid Y (2017). Mucosal-associated invariant T cells respond to cutaneous microbiota. The Journal of Immunology 198, 218.215–218.215.27895177 [Google Scholar]
- Costello EK, Lauber CL, Hamady M, Fierer N, Gordon JI, and Knight R (2009). Bacterial community variation in human body habitats across space and time. Science 326, 1694–1697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dainichi T, Kitoh A, Otsuka A, Nakajima S, Nomura T, Kaplan DH, and Kabashima K (2018). The epithelial immune microenvironment (EIME) in atopic dermatitis and psoriasis. Nat. Immunol 19, 1286–1298. [DOI] [PubMed] [Google Scholar]
- Dillon SR, Sprecher C, Hammond A, Bilsborough J, Rosenfeld-Franklin M, Presnell SR, Haugen HS, Maurer M, Harder B, Johnston J, et al. (2004). Interleukin 31, a cytokine produced by activated T cells, induces dermatitis in mice. Nat. Immunol 5, 752–760. [DOI] [PubMed] [Google Scholar]
- Doebel T, Voisin B, and Nagao K (2017). Langerhans Cells - The Macrophage in Dendritic Cell Clothing. Trends Immunol 38, 817–828. [DOI] [PubMed] [Google Scholar]
- Doehl JSP, Bright Z, Dey S, Davies H, Magson J, Brown N, Romano A, Dalton JE, Pinto AI, Pitchford JW, and Kaye PM (2017). Skin parasite landscape determines host infectiousness in visceral leishmaniasis. Nature communications 8, 57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dombrowski Y, Peric M, Koglin S, Kammerbauer C, Goss C, Anz D, Simanski M, Glaser R, Harder J, Hornung V, et al. (2011). Cytosolic DNA triggers inflammasome activation in keratinocytes in psoriatic lesions. Sci. Transl. Med 3, 82ra38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driskell RR, Lichtenberger BM, Hoste E, Kretzschmar K, Simons BD, Charalambous M, Ferron SR, Herault Y, Pavlovic G, Ferguson-Smith AC, and Watt FM (2013). Distinct fibroblast lineages determine dermal architecture in skin development and repair. Nature 504, 277–281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dubin AE, and Patapoutian A (2010). Nociceptors: the sensors of the pain pathway. J. Clin. Invest 120, 3760–3772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaide O, Emerson RO, Jiang X, Gulati N, Nizza S, Desmarais C, Robins H, Krueger JG, Clark RA, and Kupper TS (2015). Common clonal origin of central and resident memory T cells following skin immunization. Nat. Med 21, 647–653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallo RL, and Hooper LV (2012). Epithelial antimicrobial defence of the skin and intestine. Nat. Rev. Immunol 12, 503–516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganesan AP, Clarke J, Wood O, Garrido-Martin EM, Chee SJ, Mellows T, Samaniego-Castruita D, Singh D, Seumois G, Alzetani A, et al. (2017). Tissue-resident memory features are linked to the magnitude of cytotoxic T cell responses in human lung cancer. Nat. Immunol 18, 940–950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gay D, Kwon O, Zhang Z, Spata M, Plikus MV, Holler PD, Ito M, Yang Z, Treffeisen E, Kim CD, et al. (2013). Fgf9 from dermal gammadelta T cells induces hair follicle neogenesis after wounding. Nat. Med 19, 916–923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gebhardt T, Wakim LM, Eidsmo L, Reading PC, Heath WR, and Carbone FR (2009). Memory T cells in nonlymphoid tissue that provide enhanced local immunity during infection with herpes simplex virus. Nat. Immunol 10, 524–530. [DOI] [PubMed] [Google Scholar]
- Gieseck RL 3rd, Wilson MS, and Wynn TA (2018). Type 2 immunity in tissue repair and fibrosis. Nat. Rev. Immunol 18, 62–76. [DOI] [PubMed] [Google Scholar]
- Gratz IK, Truong HA, Yang SH, Maurano MM, Lee K, Abbas AK, and Rosenblum MD (2013). Cutting Edge: memory regulatory t cells require IL-7 and not IL-2 for their maintenance in peripheral tissues. J. Immunol 190, 4483–4487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gregorio J, Meller S, Conrad C, Di Nardo A, Homey B, Lauerma A, Arai N, Gallo RL, Digiovanni J, and Gilliet M (2010). Plasmacytoid dendritic cells sense skin injury and promote wound healing through type I interferons. J. Exp. Med 207, 2921–2930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greter M, Lelios I, Pelczar P, Hoeffel G, Price J, Leboeuf M, Kundig TM, Frei K, Ginhoux F, Merad M, and Becher B (2012). Stroma-derived interleukin-34 controls the development and maintenance of langerhans cells and the maintenance of microglia. Immunity 37, 1050–1060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grice EA, Kong HH, Conlan S, Deming CB, Davis J, Young AC, Bouffard GG, Blakesley RW, Murray PR, Green ED, et al. (2009). Topographical and temporal diversity of the human skin microbiome. Science 324, 1190–1192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gudjonsson JE, Johnston A, Dyson M, Valdimarsson H, and Elder JT (2007). Mouse models of psoriasis. J. Invest. Dermatol 127, 1292–1308. [DOI] [PubMed] [Google Scholar]
- Guerrero-Juarez CF, Dedhia PH, Jin S, Ruiz-Vega R, Ma D, Liu Y, Yamaga K, Shestova O, Gay DL, Yang Z, et al. (2019). Single-cell analysis reveals fibroblast heterogeneity and myeloid-derived adipocyte progenitors in murine skin wounds. Nature communications 10, 650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guerrero-Juarez CF, and Plikus MV (2018). Emerging nonmetabolic functions of skin fat. Nat. Rev. Endocrinol 14, 163–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hedrick MN, Lonsdorf AS, Shirakawa AK, Richard Lee CC, Liao F, Singh SP, Zhang HH, Grinberg A, Love PE, Hwang ST, and Farber JM (2009). CCR6 is required for IL-23-induced psoriasis-like inflammation in mice. J. Clin. Invest 119, 2317–2329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hepworth MR, Monticelli LA, Fung TC, Ziegler CG, Grunberg S, Sinha R, Mantegazza AR, Ma HL, Crawford A, Angelosanto JM, et al. (2013). Innate lymphoid cells regulate CD4+ T-cell responses to intestinal commensal bacteria. Nature 498, 113–117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu YC, Li L, and Fuchs E (2014). Emerging interactions between skin stem cells and their niches. Nat. Med 20, 847–856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang Y, Guo L, Qiu J, Chen X, Hu-Li J, Siebenlist U, Williamson PR, Urban JF Jr., and Paul WE (2015). IL-25-responsive, lineage-negative KLRG1(hi) cells are multipotential ‘inflammatory’ type 2 innate lymphoid cells. Nat. Immunol 16, 161–169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imai Y, Yasuda K, Sakaguchi Y, Haneda T, Mizutani H, Yoshimoto T, Nakanishi K, and Yamanishi K (2013). Skin-specific expression of IL-33 activates group 2 innate lymphoid cells and elicits atopic dermatitis-like inflammation in mice. Proc. Natl. Acad. Sci. U. S. A 110, 13921–13926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito M, Yang Z, Andl T, Cui C, Kim N, Millar SE, and Cotsarelis G (2007). Wnt-dependent de novo hair follicle regeneration in adult mouse skin after wounding. Nature 447, 316–320. [DOI] [PubMed] [Google Scholar]
- Iwasaki A, and Medzhitov R (2015). Control of adaptive immunity by the innate immune system. Nat. Immunol 16, 343–353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jameson J, Ugarte K, Chen N, Yachi P, Fuchs E, Boismenu R, and Havran WL (2002). A role for skin gammadelta T cells in wound repair. Science 296, 747–749. [DOI] [PubMed] [Google Scholar]
- Jiang X, Clark RA, Liu L, Wagers AJ, Fuhlbrigge RC, and Kupper TS (2012). Skin infection generates non-migratory memory CD8+ T(RM) cells providing global skin immunity. Nature 483, 227–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kabashima K, Honda T, Ginhoux F, and Egawa G (2019). The immunological anatomy of the skin. Nat. Rev. Immunol 19, 19–30. [DOI] [PubMed] [Google Scholar]
- Kashem SW, Riedl MS, Yao C, Honda CN, Vulchanova L, and Kaplan DH (2015). Nociceptive Sensory Fibers Drive Interleukin-23 Production from CD301b+ Dermal Dendritic Cells and Drive Protective Cutaneous Immunity. Immunity 43, 515–526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kashiwagi M, Hosoi J, Lai JF, Brissette J, Ziegler SF, Morgan BA, and Georgopoulos K (2017). Direct control of regulatory T cells by keratinocytes. Nat. Immunol 18, 334–343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawai T, and Akira S (2009). The roles of TLRs, RLRs and NLRs in pathogen recognition. Int. Immunol 21, 317–337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keyes BE, Liu S, Asare A, Naik S, Levorse J, Polak L, Lu CP, Nikolova M, Pasolli HA, and Fuchs E (2016). Impaired Epidermal to Dendritic T Cell Signaling Slows Wound Repair in Aged Skin. Cell 167, 1323–1338.e1314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim BS, Siracusa MC, Saenz SA, Noti M, Monticelli LA, Sonnenberg GF, Hepworth MR, Van Voorhees AS, Comeau MR, and Artis D (2013). TSLP elicits IL-33-independent innate lymphoid cell responses to promote skin inflammation. Sci. Transl. Med 5, 170ra116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knipper JA, Willenborg S, Brinckmann J, Bloch W, Maass T, Wagener R, Krieg T, Sutherland T, Munitz A, Rothenberg ME, et al. (2015). Interleukin-4 Receptor alpha Signaling in Myeloid Cells Controls Collagen Fibril Assembly in Skin Repair. Immunity 43, 803–816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi T, Glatz M, Horiuchi K, Kawasaki H, Akiyama H, Kaplan DH, Kong HH, Amagai M, and Nagao K (2015). Dysbiosis and Staphylococcus aureus Colonization Drives Inflammation in Atopic Dermatitis. Immunity 42, 756–766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi T, Voisin B, Kim DY, Kennedy EA, Jo JH, Shih HY, Truong A, Doebel T, Sakamoto K, Cui CY, et al. (2019). Homeostatic Control of Sebaceous Glands by Innate Lymphoid Cells Regulates Commensal Bacteria Equilibrium. Cell 176, 982–997.e916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kong HH, Oh J, Deming C, Conlan S, Grice EA, Beatson MA, Nomicos E, Polley EC, Komarow HD, Murray PR, et al. (2012). Temporal shifts in the skin microbiome associated with disease flares and treatment in children with atopic dermatitis. Genome Res 22, 850–859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumari S, Bonnet MC, Ulvmar MH, Wolk K, Karagianni N, Witte E, Uthoff-Hachenberg C, Renauld JC, Kollias G, Toftgard R, et al. (2013). Tumor necrosis factor receptor signaling in keratinocytes triggers interleukin-24-dependent psoriasis-like skin inflammation in mice. Immunity 39, 899–911. [DOI] [PubMed] [Google Scholar]
- Lai Y, Di Nardo A, Nakatsuji T, Leichtle A, Yang Y, Cogen AL, Wu ZR, Hooper LV, Schmidt RR, von Aulock S, et al. (2009). Commensal bacteria regulate Toll-like receptor 3-dependent inflammation after skin injury. Nat. Med 15, 1377–1382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lande R, Gregorio J, Facchinetti V, Chatterjee B, Wang YH, Homey B, Cao W, Wang YH, Su B, Nestle FO, et al. (2007). Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature 449, 564–569. [DOI] [PubMed] [Google Scholar]
- Lay K, Yuan S, Gur-Cohen S, Miao Y, Han T, Naik S, Pasolli HA, Larsen SB, and Fuchs E (2018). Stem cells repurpose proliferation to contain a breach in their niche barrier. Elife 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leyden JJ, Marples RR, and Kligman AM (1974). Staphylococcus aureus in the lesions of atopic dermatitis. Br. J. Dermatol 90, 525–530. [DOI] [PubMed] [Google Scholar]
- Leyva-Castillo JM, Hener P, Jiang H, and Li M (2013). TSLP produced by keratinocytes promotes allergen sensitization through skin and thereby triggers atopic march in mice. J. Invest. Dermatol 133, 154–163. [DOI] [PubMed] [Google Scholar]
- Li H, Yao Q, Mariscal AG, Wu X, Hulse J, Pedersen E, Helin K, Waisman A, Vinkel C, Thomsen SF, et al. (2018). Epigenetic control of IL-23 expression in keratinocytes is important for chronic skin inflammation. Nature communications 9, 1420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang SC, Tan XY, Luxenberg DP, Karim R, Dunussi-Joannopoulos K, Collins M, and Fouser LA (2006). Interleukin (IL)-22 and IL-17 are coexpressed by Th17 cells and cooperatively enhance expression of antimicrobial peptides. J. Exp. Med 203, 2271–2279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linehan JL, Harrison OJ, Han SJ, Byrd AL, Vujkovic-Cvijin I, Villarino AV, Sen SK, Shaik J, Smelkinson M, Tamoutounour S, et al. (2018). Non-classical Immunity Controls Microbiota Impact on Skin Immunity and Tissue Repair. Cell 172, 784–796.e718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu H, Archer NK, Dillen CA, Wang Y, Ashbaugh AG, Ortines RV, Kao T, Lee SK, Cai SS, Miller RJ, et al. (2017). Staphylococcus aureus Epicutaneous Exposure Drives Skin Inflammation via IL-36-Mediated T Cell Responses. Cell Host Microbe 22, 653–666.e655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mabuchi T, Singh TP, Takekoshi T, Jia GF, Wu X, Kao MC, Weiss I, Farber JM, and Hwang ST (2013). CCR6 is required for epidermal trafficking of gammadelta-T cells in an IL-23-induced model of psoriasiform dermatitis. J. Invest. Dermatol 133, 164–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mackay LK, Rahimpour A, Ma JZ, Collins N, Stock AT, Hafon ML, Vega-Ramos J, Lauzurica P, Mueller SN, Stefanovic T, et al. (2013). The developmental pathway for CD103(+)CD8+ tissue-resident memory T cells of skin. Nat. Immunol 14, 1294–1301. [DOI] [PubMed] [Google Scholar]
- Mackay-Wiggan J, Jabbari A, Nguyen N, Cerise JE, Clark C, Ulerio G, Furniss M, Vaughan R, Christiano AM, and Clynes R (2016). Oral ruxolitinib induces hair regrowth in patients with moderate-to-severe alopecia areata. JCI insight 1, e89790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malik BT, Byrne KT, Vella JL, Zhang P, Shabaneh TB, Steinberg SM, Molodtsov AK, Bowers JS, Angeles CV, Paulos CM, et al. (2017). Resident memory T cells in the skin mediate durable immunity to melanoma. Sci Immunol 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsumoto R, Dainichi T, Tsuchiya S, Nomura T, Kitoh A, Hayden MS, Ishii KJ, Tanaka M, Honda T, Egawa G, et al. (2018). Epithelial TRAF6 drives IL-17-mediated psoriatic inflammation. JCI insight 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meisel JS, Sfyroera G, Bartow-McKenney C, Gimblet C, Bugayev J, Horwinski J, Kim B, Brestoff JR, Tyldsley AS, Zheng Q, et al. (2018). Commensal microbiota modulate gene expression in the skin. Microbiome 6, 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menon GK (2002). New insights into skin structure: scratching the surface. Advanced drug delivery reviews 54 Suppl 1, S3–17. [DOI] [PubMed] [Google Scholar]
- Merad M, Ginhoux F, and Collin M (2008). Origin, homeostasis and function of Langerhans cells and other langerin-expressing dendritic cells. Nat. Rev. Immunol 8, 935–947. [DOI] [PubMed] [Google Scholar]
- Miller LS, and Cho JS (2011). Immunity against Staphylococcus aureus cutaneous infections. Nat. Rev. Immunol 11, 505–518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller LS, O’Connell RM, Gutierrez MA, Pietras EM, Shahangian A, Gross CE, Thirumala A, Cheung AL, Cheng G, and Modlin RL (2006). MyD88 mediates neutrophil recruitment initiated by IL-1R but not TLR2 activation in immunity against Staphylococcus aureus. Immunity 24, 79–91. [DOI] [PubMed] [Google Scholar]
- Mohammed J, Beura LK, Bobr A, Astry B, Chicoine B, Kashem SW, Welty NE, Igyarto BZ, Wijeyesinghe S, Thompson EA, et al. (2016). Stromal cells control the epithelial residence of DCs and memory T cells by regulated activation of TGF-beta. Nat. Immunol 17, 414–421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moro K, Yamada T, Tanabe M, Takeuchi T, Ikawa T, Kawamoto H, Furusawa J, Ohtani M, Fujii H, and Koyasu S (2010). Innate production of T(H)2 cytokines by adipose tissue-associated c-Kit(+)Sca-1(+) lymphoid cells. Nature 463, 540–544. [DOI] [PubMed] [Google Scholar]
- Mueller SN, and Mackay LK (2016). Tissue-resident memory T cells: local specialists in immune defence. Nat. Rev. Immunol 16, 79–89. [DOI] [PubMed] [Google Scholar]
- Munger JS, Huang X, Kawakatsu H, Griffiths MJ, Dalton SL, Wu J, Pittet JF, Kaminski N, Garat C, Matthay MA, et al. (1999). The integrin alpha v beta 6 binds and activates latent TGF beta 1: a mechanism for regulating pulmonary inflammation and fibrosis. Cell 96, 319–328. [DOI] [PubMed] [Google Scholar]
- Myles IA, Earland NJ, Anderson ED, Moore IN, Kieh MD, Williams KW, Saleem A, Fontecilla NM, Welch PA, Darnell DA, et al. (2018). First-in-human topical microbiome transplantation with Roseomonas mucosa for atopic dermatitis. JCI insight 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagao K, Kobayashi T, Moro K, Ohyama M, Adachi T, Kitashima DY, Ueha S, Horiuchi K, Tanizaki H, Kabashima K, et al. (2012). Stress-induced production of chemokines by hair follicles regulates the trafficking of dendritic cells in skin. Nat. Immunol 13, 744–752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naik S, Bouladoux N, Linehan JL, Han SJ, Harrison OJ, Wilhelm C, Conlan S, Himmelfarb S, Byrd AL, Deming C, et al. (2015). Commensal-dendritic-cell interaction specifies a unique protective skin immune signature. Nature 520, 104–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naik S, Bouladoux N, Wilhelm C, Molloy MJ, Salcedo R, Kastenmuller W, Deming C, Quinones M, Koo L, Conlan S, et al. (2012). Compartmentalized control of skin immunity by resident commensals. Science 337, 1115–1119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naik S, Larsen SB, Cowley CJ, and Fuchs E (2018). Two to Tango: Dialog between Immunity and Stem Cells in Health and Disease. Cell 175, 908–920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naik S, Larsen SB, Gomez NC, Alaverdyan K, Sendoel A, Yuan S, Polak L, Kulukian A, Chai S, and Fuchs E (2017). Inflammatory memory sensitizes skin epithelial stem cells to tissue damage. Nature 550, 475–480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakagawa S, Matsumoto M, Katayama Y, Oguma R, Wakabayashi S, Nygaard T, Saijo S, Inohara N, Otto M, Matsue H, et al. (2017). Staphylococcus aureus Virulent PSMalpha Peptides Induce Keratinocyte Alarmin Release to Orchestrate IL-17-Dependent Skin Inflammation. Cell Host Microbe 22, 667–677.e665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura Y, Oscherwitz J, Cease KB, Chan SM, Munoz-Planillo R, Hasegawa M, Villaruz AE, Cheung GY, McGavin MJ, Travers JB, et al. (2013). Staphylococcus delta-toxin induces allergic skin disease by activating mast cells. Nature 503, 397–401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakatsuji T, Chen TH, Narala S, Chun KA, Two AM, Yun T, Shafiq F, Kotol PF, Bouslimani A, Melnik AV, et al. (2017). Antimicrobials from human skin commensal bacteria protect against Staphylococcus aureus and are deficient in atopic dermatitis. Sci. Transl. Med 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nestle FO, Kaplan DH, and Barker J (2009). Psoriasis. N. Engl. J. Med 361, 496–509. [DOI] [PubMed] [Google Scholar]
- Netea MG, Joosten LA, Latz E, Mills KH, Natoli G, Stunnenberg HG, O’Neill LA, and Xavier RJ (2016). Trained immunity: A program of innate immune memory in health and disease. Science 352, aaf1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen MM, Witherden DA, and Havran WL (2017). gammadelta T cells in homeostasis and host defence of epithelial barrier tissues. Nat. Rev. Immunol 17, 733–745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nosbaum A, Prevel N, Truong HA, Mehta P, Ettinger M, Scharschmidt TC, Ali NH, Pauli ML, Abbas AK, and Rosenblum MD (2016). Cutting Edge: Regulatory T Cells Facilitate Cutaneous Wound Healing. J. Immunol 196, 2010–2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oetjen LK, Mack MR, Feng J, Whelan TM, Niu H, Guo CJ, Chen S, Trier AM, Xu AZ, Tripathi SV, et al. (2017). Sensory Neurons Co-opt Classical Immune Signaling Pathways to Mediate Chronic Itch. Cell 171, 217–228.e213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oh J, Freeman AF, Park M, Sokolic R, Candotti F, Holland SM, Segre JA, and Kong HH (2013). The altered landscape of the human skin microbiome in patients with primary immunodeficiencies. Genome Res 23, 2103–2114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okabe Y, and Medzhitov R (2016). Tissue biology perspective on macrophages. Nat. Immunol 17, 9–17. [DOI] [PubMed] [Google Scholar]
- Ordovas-Montanes J, Dwyer DF, Nyquist SK, Buchheit KM, Vukovic M, Deb C, Wadsworth MH 2nd, Hughes TK, Kazer SW, Yoshimoto E, et al. (2018). Allergic inflammatory memory in human respiratory epithelial progenitor cells. Nature 560, 649–654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otberg N, Richter H, Schaefer H, Blume-Peytavi U, Sterry W, and Lademann J (2004). Variations of hair follicle size and distribution in different body sites. J. Invest. Dermatol 122, 14–19. [DOI] [PubMed] [Google Scholar]
- Park SL, Buzzai A, Rautela J, Hor JL, Hochheiser K, Effern M, McBain N, Wagner T, Edwards J, McConville R, et al. (2018). Tissue-resident memory CD8(+) T cells promote melanoma-immune equilibrium in skin. Nature [DOI] [PubMed] [Google Scholar]
- Pasparakis M, Courtois G, Hafner M, Schmidt-Supprian M, Nenci A, Toksoy A, Krampert M, Goebeler M, Gillitzer R, Israel A, et al. (2002). TNF-mediated inflammatory skin disease in mice with epidermis-specific deletion of IKK2. Nature 417, 861–866. [DOI] [PubMed] [Google Scholar]
- Pasparakis M, Haase I, and Nestle FO (2014). Mechanisms regulating skin immunity and inflammation. Nat. Rev. Immunol 14, 289–301. [DOI] [PubMed] [Google Scholar]
- Paus R, and Bertolini M (2013). The role of hair follicle immune privilege collapse in alopecia areata: status and perspectives. J. Investig. Dermatol. Symp. Proc 16, S25–27. [DOI] [PubMed] [Google Scholar]
- Paus R, van der Veen C, Eichmuller S, Kopp T, Hagen E, Muller-Rover S, and Hofmann U (1998). Generation and cyclic remodeling of the hair follicle immune system in mice. J. Invest. Dermatol 111, 7–18. [DOI] [PubMed] [Google Scholar]
- Petukhova L, Duvic M, Hordinsky M, Norris D, Price V, Shimomura Y, Kim H, Singh P, Lee A, Chen WV, et al. (2010). Genome-wide association study in alopecia areata implicates both innate and adaptive immunity. Nature 466, 113–117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plikus MV, Mayer JA, de la Cruz D, Baker RE, Maini PK, Maxson R, and Chuong CM (2008). Cyclic dermal BMP signalling regulates stem cell activation during hair regeneration. Nature 451, 340–344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pratt CH, King LE Jr., Messenger AG, Christiano AM, and Sundberg JP (2017). Alopecia areata. Nat Rev Dis Primers 3, 17011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rak GD, Osborne LC, Siracusa MC, Kim BS, Wang K, Bayat A, Artis D, and Volk SW (2016). IL-33-Dependent Group 2 Innate Lymphoid Cells Promote Cutaneous Wound Healing. J. Invest. Dermatol 136, 487–496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ricardo-Gonzalez RR, Van Dyken SJ, Schneider C, Lee J, Nussbaum JC, Liang HE, Vaka D, Eckalbar WL, Molofsky AB, Erle DJ, and Locksley RM (2018). Tissue signals imprint ILC2 identity with anticipatory function. Nat. Immunol 19, 1093–1099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richmond JM, Strassner JP, Zapata L Jr., Garg M, Riding RL, Refat MA, Fan X, Azzolino V, Tovar-Garza A, Tsurushita N, et al. (2018). Antibody blockade of IL-15 signaling has the potential to durably reverse vitiligo. Sci. Transl. Med 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ridaura VK, Bouladoux N, Claesen J, Chen YE, Byrd AL, Constantinides MG, Merrill ED, Tamoutounour S, Fischbach MA, and Belkaid Y (2018). Contextual control of skin immunity and inflammation by Corynebacterium. J. Exp. Med 215, 785–799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riol-Blanco L, Ordovas-Montanes J, Perro M, Naval E, Thiriot A, Alvarez D, Paust S, Wood JN, and von Andrian UH (2014). Nociceptive sensory neurons drive interleukin-23-mediated psoriasiform skin inflammation. Nature 510, 157–161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robertson K, and Rees JL (2010). Variation in epidermal morphology in human skin at different body sites as measured by reflectance confocal microscopy. Acta Derm. Venereol 90, 368–373. [DOI] [PubMed] [Google Scholar]
- Roediger B, Kyle R, Yip KH, Sumaria N, Guy TV, Kim BS, Mitchell AJ, Tay SS, Jain R, Forbes-Blom E, et al. (2013). Cutaneous immunosurveillance and regulation of inflammation by group 2 innate lymphoid cells. Nat. Immunol 14, 564–573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salimi M, Barlow JL, Saunders SP, Xue L, Gutowska-Owsiak D, Wang X, Huang LC, Johnson D, Scanlon ST, McKenzie AN, et al. (2013). A role for IL-25 and IL-33-driven type-2 innate lymphoid cells in atopic dermatitis. J. Exp. Med 210, 2939–2950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanford JA, Zhang LJ, Williams MR, Gangoiti JA, Huang CM, and Gallo RL (2016). Inhibition of HDAC8 and HDAC9 by microbial short-chain fatty acids breaks immune tolerance of the epidermis to TLR ligands. Sci Immunol 1. [DOI] [PubMed] [Google Scholar]
- Sano S, Chan KS, Carbajal S, Clifford J, Peavey M, Kiguchi K, Itami S, Nickoloff BJ, and DiGiovanni J (2005). Stat3 links activated keratinocytes and immunocytes required for development of psoriasis in a novel transgenic mouse model. Nat. Med 11, 43–49. [DOI] [PubMed] [Google Scholar]
- Sano S, Itami S, Takeda K, Tarutani M, Yamaguchi Y, Miura H, Yoshikawa K, Akira S, and Takeda J (1999). Keratinocyte-specific ablation of Stat3 exhibits impaired skin remodeling, but does not affect skin morphogenesis. EMBO J 18, 4657–4668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scharschmidt TC, Vasquez KS, Pauli ML, Leitner EG, Chu K, Truong HA, Lowe MM, Sanchez Rodriguez R, Ali N, Laszik ZG, et al. (2017). Commensal Microbes and Hair Follicle Morphogenesis Coordinately Drive Treg Migration into Neonatal Skin. Cell Host Microbe 21, 467–477.e465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scharschmidt TC, Vasquez KS, Truong HA, Gearty SV, Pauli ML, Nosbaum A, Gratz IK, Otto M, Moon JJ, Liese J, et al. (2015). A Wave of Regulatory T Cells into Neonatal Skin Mediates Tolerance to Commensal Microbes. Immunity 43, 1011–1021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schenkel JM, and Masopust D (2014). Tissue-resident memory T cells. Immunity 41, 886–897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt M, Raghavan B, Muller V, Vogl T, Fejer G, Tchaptchet S, Keck S, Kalis C, Nielsen PJ, Galanos C, et al. (2010). Crucial role for human Toll-like receptor 4 in the development of contact allergy to nickel. Nat. Immunol 11, 814–819. [DOI] [PubMed] [Google Scholar]
- Seneschal J, Clark RA, Gehad A, Baecher-Allan CM, and Kupper TS (2012). Human epidermal Langerhans cells maintain immune homeostasis in skin by activating skin resident regulatory T cells. Immunity 36, 873–884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharp LL, Jameson JM, Cauvi G, and Havran WL (2005). Dendritic epidermal T cells regulate skin homeostasis through local production of insulin-like growth factor 1. Nat. Immunol 6, 73–79. [DOI] [PubMed] [Google Scholar]
- Shen W, Li W, Hixon JA, Bouladoux N, Belkaid Y, Dzutzev A, and Durum SK (2014). Adaptive immunity to murine skin commensals. Proc. Natl. Acad. Sci. U. S. A 111, E2977–2986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shipman WD, Chyou S, Ramanathan A, Izmirly PM, Sharma S, Pannellini T, Dasoveanu DC, Qing X, Magro CM, Granstein RD, et al. (2018). A protective Langerhans cell-keratinocyte axis that is dysfunctional in photosensitivity. Sci. Transl. Med 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shook BA, Wasko RR, Rivera-Gonzalez GC, Salazar-Gatzimas E, Lopez-Giraldez F, Dash BC, Munoz-Rojas AR, Aultman KD, Zwick RK, Lei V, et al. (2018). Myofibroblast proliferation and heterogeneity are supported by macrophages during skin repair. Science 362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sims JE, Williams DE, Morrissey PJ, Garka K, Foxworthe D, Price V, Friend SL, Farr A, Bedell MA, Jenkins NA, et al. (2000). Molecular cloning and biological characterization of a novel murine lymphoid growth factor. J. Exp. Med 192, 671–680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh TP, Zhang HH, Borek I, Wolf P, Hedrick MN, Singh SP, Kelsall BL, Clausen BE, and Farber JM (2016). Monocyte-derived inflammatory Langerhans cells and dermal dendritic cells mediate psoriasis-like inflammation. Nature communications 7, 13581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soumelis V, Reche PA, Kanzler H, Yuan W, Edward G, Homey B, Gilliet M, Ho S, Antonenko S, Lauerma A, et al. (2002). Human epithelial cells trigger dendritic cell mediated allergic inflammation by producing TSLP. Nat. Immunol 3, 673–680. [DOI] [PubMed] [Google Scholar]
- Takamori A, Nambu A, Sato K, Yamaguchi S, Matsuda K, Numata T, Sugawara T, Yoshizaki T, Arae K, Morita H, et al. (2018). IL-31 is crucial for induction of pruritus, but not inflammation, in contact hypersensitivity. Sci. Rep 8, 6639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tong PL, Roediger B, Kolesnikoff N, Biro M, Tay SS, Jain R, Shaw LE, Grimbaldeston MA, and Weninger W (2015). The skin immune atlas: three-dimensional analysis of cutaneous leukocyte subsets by multiphoton microscopy. J. Invest. Dermatol 135, 84–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toulon A, Breton L, Taylor KR, Tenenhaus M, Bhavsar D, Lanigan C, Rudolph R, Jameson J, and Havran WL (2009). A role for human skin-resident T cells in wound healing. J. Exp. Med 206, 743–750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Veiga-Fernandes H, and Artis D (2018). Neuronal-immune system cross-talk in homeostasis. Science 359, 1465–1466. [DOI] [PubMed] [Google Scholar]
- Vivier E, van de Pavert SA, Cooper MD, and Belz GT (2016). The evolution of innate lymphoid cells. Nat. Immunol 17, 790–794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang M, Zhang S, Zheng G, Huang J, Songyang Z, Zhao X, and Lin X (2018). Gain-of-Function Mutation of Card14 Leads to Spontaneous Psoriasis-like Skin Inflammation through Enhanced Keratinocyte Response to IL-17A. Immunity 49, 66–79.e65. [DOI] [PubMed] [Google Scholar]
- Wang X, Chen H, Tian R, Zhang Y, Drutskaya MS, Wang C, Ge J, Fan Z, Kong D, Wang X, et al. (2017). Macrophages induce AKT/beta-catenin-dependent Lgr5(+) stem cell activation and hair follicle regeneration through TNF. Nature communications 8, 14091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Szretter KJ, Vermi W, Gilfillan S, Rossini C, Cella M, Barrow AD, Diamond MS, and Colonna M (2012). IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia. Nat. Immunol 13, 753–760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson SR, The L, Batia LM, Beattie K, Katibah GE, McClain SP, Pellegrino M, Estandian DM, and Bautista DM (2013). The epithelial cell-derived atopic dermatitis cytokine TSLP activates neurons to induce itch. Cell 155, 285–295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xing L, Dai Z, Jabbari A, Cerise JE, Higgins CA, Gong W, de Jong A, Harel S, DeStefano GM, Rothman L, et al. (2014). Alopecia areata is driven by cytotoxic T lymphocytes and is reversed by JAK inhibition. Nat. Med 20, 1043–1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu M, Lu H, Lee YH, Wu Y, Liu K, Shi Y, An H, Zhang J, Wang X, Lai Y, and Dong C (2018). An Interleukin-25-Mediated Autoregulatory Circuit in Keratinocytes Plays a Pivotal Role in Psoriatic Skin Inflammation. Immunity 48, 787–798.e784. [DOI] [PubMed] [Google Scholar]
- Yang J, Hu S, Zhao L, Kaplan DH, Perdew GH, and Xiong N (2016). Selective programming of CCR10(+) innate lymphoid cells in skin-draining lymph nodes for cutaneous homeostatic regulation. Nat. Immunol 17, 48–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Z, Mu Z, Dabovic B, Jurukovski V, Yu D, Sung J, Xiong X, and Munger JS (2007). Absence of integrin-mediated TGFbeta1 activation in vivo recapitulates the phenotype of TGFbeta1-null mice. J. Cell Biol 176, 787–793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoo J, Omori M, Gyarmati D, Zhou B, Aye T, Brewer A, Comeau MR, Campbell DJ, and Ziegler SF (2005). Spontaneous atopic dermatitis in mice expressing an inducible thymic stromal lymphopoietin transgene specifically in the skin. J. Exp. Med 202, 541–549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zenz R, Eferl R, Kenner L, Florin L, Hummerich L, Mehic D, Scheuch H, Angel P, Tschachler E, and Wagner EF (2005). Psoriasis-like skin disease and arthritis caused by inducible epidermal deletion of Jun proteins. Nature 437, 369–375. [DOI] [PubMed] [Google Scholar]
- Zhang LJ, Guerrero-Juarez CF, Hata T, Bapat SP, Ramos R, Plikus MV, and Gallo RL (2015). Innate immunity. Dermal adipocytes protect against invasive Staphylococcus aureus skin infection. Science 347, 67–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegler SF, and Artis D (2010). Sensing the outside world: TSLP regulates barrier immunity. Nat. Immunol 11, 289–293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zwick RK, Guerrero-Juarez CF, Horsley V, and Plikus MV (2018). Anatomical, Physiological, and Functional Diversity of Adipose Tissue. Cell Metab 27, 68–83. [DOI] [PMC free article] [PubMed] [Google Scholar]