Abstract
MicroRNA sequencing (miRNA-seq) was performed in the present study to investigate miRNA expression profiles in infarcted brain areas following focal cerebral ischemia induced by middle cerebral artery occlusion in rats. In total, 20 miRNAs were identified to be upregulated and 17 to be downregulated in the infarct area. The expression levels of six differentially expressed miRNAs (DEmiRs), miR-211-5p, miR-183-5p, miR-10b-3p, miR-182, miR-217-5p and miR-96-5p, were examined by reverse transcription-quantitative polymerase chain reaction. Subsequently, a miRNA-mRNA network was constructed. Gene Ontology and Kyoto Encyclopedia of Genes and Genomes analyses were performed to investigate the functions of the mRNAs targeted by these DEmiRs. The present study aimed to investigate the association between miRNAs and cerebral ischemia to provide potential insight into the molecular mechanisms underlying ischemic stroke.
Keywords: focal cerebral ischemia injury, microRNA sequencing, microRNA, middle cerebral artery occlusion
Introduction
Ischemic stroke, one of the leading causes of mortality and disability, poses a public health concern worldwide. Over the past two decades, accumulating evidence has demonstrated the involvement of endothelial dysfunction, neuroinflammation and oxidative stress in the development of ischemic stroke (1,2); however, the association between microRNAs (miRNAs) and stroke remains unclear.
miRNAs are small noncoding RNA molecules of 18–23 nucleotides in length. miRNAs are important regulators of physiological and pathological processes, and have been reported to influence various diseases (3,4). miRNA networks have been observed to be involved in various processes including the regeneration of central and peripheral nervous lesions, axonal regeneration, neural cell differentiation and chronic pain (5–7). Previous studies demonstrated that miRNAs may be used as biomarkers and therapeutic targets to treat certain neurological diseases. Additionally, previous studies demonstrated that miRNAs are involved in ischemic stroke (8–18). miRNA-182 (miR-182) may regulate the expression levels of clock circadian regulator in the pineal gland of neonatal rats following hypoxic-ischemic brain injury (16). Chang and Lai (17) demonstrated that miR-96 may be used as a biomarker for ischemia-reperfusion injured flaps. A previous study identified that increased expression of miR-211 may increase the risk of stroke by inhibiting angiopoietin-1, and that mutations in the miR-211 binding site in the 3′ untranslated region (3′-UTR) of angiopoietin-1 may decrease the risk of stroke (18).
In the present study, miRNA sequencing (miRNA-seq) was performed to identify miRNA expression profiles in infarcted brain areas of rats following focal cerebral ischemia injury induced by middle cerebral artery occlusion (MCAO). Gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses were performed to demonstrate the functions of the mRNAs targeted by differentially expressed miRNAs (DEmiRs). Additionally, an miRNA-mRNA network was constructed. The present study aimed to investigate the association between miRNAs and cerebral ischemia.
Materials and methods
Rat MCAO model
In total, 20 specific pathogen free male Sprague-Dawley rats (age, 7 weeks; weight, 200±20 g) were obtained from The Experimental Animal Center of Anhui Medical University (Hefei, China). All rats were allowed free access to food and water. Animals were housed individually at 18–22°C with 40–60% humidity and under a 12-h light/dark cycle. Rats were randomly divided into the control (sham surgery) and MCAO groups (n=10 in each group). A focal cerebral ischemia rat model induced by MCAO was established as previously described (19,20). All surgical procedures were performed under anesthesia to minimize animal suffering, according to the National Institutes of Health (NIH) Guide for Animal Care and Welfare (NIH publication no. 86-23) (21). Anesthesia was achieved by intravenous administration of sodium pentobarbital at a concentration of 40 mg/kg. The present study was approved by The Animal Experiments Ethics Committee of The Anhui University of Chinese Medicine (Hefei, China).
miRNA-seq and bioinformatics
Total RNA was extracted from infarcted areas of three rats following MCAO and from corresponding regions of the brain of three control rats with the mirVana™ miRNA Isolation kit (cat no. AM1561; Thermo Fisher Scientific, Inc., Waltham, MA, USA). RNA concentration and purity were measured using a spectrophotometer (ND-2000; NanoDrop Technologies; Thermo Fisher Scientific, Inc., Wilmington, DE, USA), and RNA integrity was assessed with an Agilent 2100 Bioanalyzer (Agilent Technologies, Inc., Santa Clara, CA, USA). A small RNA (sRNA) library (length, ~21 nucleotides) was generated using the NEBNext® Multiplex Small RNA Library Prep Set for Illumina® (Set 2; New England BioLabs, Inc., Ipswich, MA, USA) according to the manufacturer's protocols. Following quality control using an Agilent 2100 Bioanalyzer and Qubit® 2.0 fluorometer (Invitrogen; Thermo Fisher Scientific, Inc.), sRNA-seq was performed with a HiSeq 2000 Sequencing System (Illumina Inc., San Diego, CA, USA). Prior to analyzing the sequencing data, data quality was assessed and filtered with FASTX software (v.0.0.13; http://hannonlab.cshl.edu/fastx_toolkit/index.html). Subsequently, Bowtie (version 0.7.10) was used to map clean reads to the rat genome assembly Rnor_6.0 (ftp://ftp.ensembl.org/pub/release-83/fasta/rattus_norvegicus/dna/Rattus_norvegicus.Rnor_6.0.dna.toplevel.fa) and miRBase (version 21) was used for annotation of the miRNAs (22). miRNA counts were standardized to transcripts per million reads (23), and the differential expression of miRNAs was analyzed using edgeR (version 3.8.5) (24). Volcano plot analysis was performed using R Software (version 5.50; Mathsoft Engineering and Education, Inc.). Hierarchical clustering was performed using Cluster3.0 (http://bonsai.hgc.jp/~mdehoon/software/cluster/software.htm) with euclidean distance function and Java TreeView (Oracle Corporation; http://sourceforge.net/projects/jtreeview/files/). DEmiRs targets were predicted using miRanda (version 3.3a) (25). The miRNA-seq experiments were performed in triplicate.
Reverse transcription-quantitative polymerase chain reaction (RT-qPCR)
The expression levels of six DEmiRs that were randomly selected from the list of 37 DEmiRs, including miR-211-5p, miR-183-5p, miR-10b-3p, miR-182, miR-217-5p and miR-96-5p, were assessed by RT-qPCR. cDNA was synthesized using the Mir-X™ miRNA First-Strand Synthesis kit (Clontech Laboratories, Inc., Mountainview, CA, USA) and the reaction was performed at 37°C for 1 h followed by an incubation at 85°C for 5 min. qPCRs were performed using the Mir-X™ miRNA RT-qPCR SYBR kit (Clontech Laboratories, Inc.) in triplicate. The forward primers used were the following: miR-211-5p, TTCCCTTTGTCATCCTTTGCCT; miR-183-5p, TATGGCACTGGTAGAATTCACT; miR-10b-3p, ACAGATTCGATTCTAGGGGAA; miR-182, TTTGGCAATGGTAGAACTCACACCG; miR-217-5p, TACTGCATCAGGAACTGACTGG; miR-96-5p, TTTGGCACTAGCACATTTTTGCT. The reverse primer was provided by Mir-X™ miRNA First-Strand Synthesis kit. The forward and reverse primers for U6 were provided in the Mir-X™ miRNA First-Strand Synthesis kit (Clontech Laboratories, Inc.). qPCRs were performed using the following thermocycling conditions: Initial denaturation at 95°C for 30 sec, followed by 40 cycles at 95°C for 105 sec and 60°C for 20 sec.
U6 was used as an internal reference. The sequences of mature miRNAs were used as miRNA-specific 5′ primers. The results were calculated using the 2−ΔΔCq method (26).
Functional analysis of DEmiRs
The mRNAs targeted by the DEmiRs were predicted with miRanda. GO and KEGG (27–29) data enrichment analyses were performed using the mRNAs targeted by the DEmiRs to examine the biological functions, ‘biological processes’, ‘cellular components’, ‘molecular functions’ and signaling pathways enriched.
miRNA-mRNA network
The association between all DEmiRs and their targets was investigated, and the expression levels of the aforementioned six DEmiRs were examined by RT-qPCR. Subsequently, the miRNA-mRNA network was constructed using Cytoscape software (version 2.8.3; http://cytoscape.github.io/).
Hematoxylin and eosin (HE) staining
Animal tissues were fixed with 4% paraformaldehyde for 24 h at room temperature. Samples were embedded in paraffin and cut into 4-µm-thick sections. Subsequently, the sections were incubated with hematoxylin for 5 min at room temperature and with eosin for 2 min at room temperature. Stained sections were visualized using a light microscope (magnification, ×200).
Statistical analysis
Data are presented as the mean ± standard error of the mean. Data were analyzed using Student's t-test. P<0.05 was considered to indicate a statistically significant difference. DEmiRs were identified using the following thresholds: P<0.05; log2 |fold change|>1. Data were analyzed by SPSS (version 22.0) and presented as the mean ± standard error of the mean.
Results
HE staining
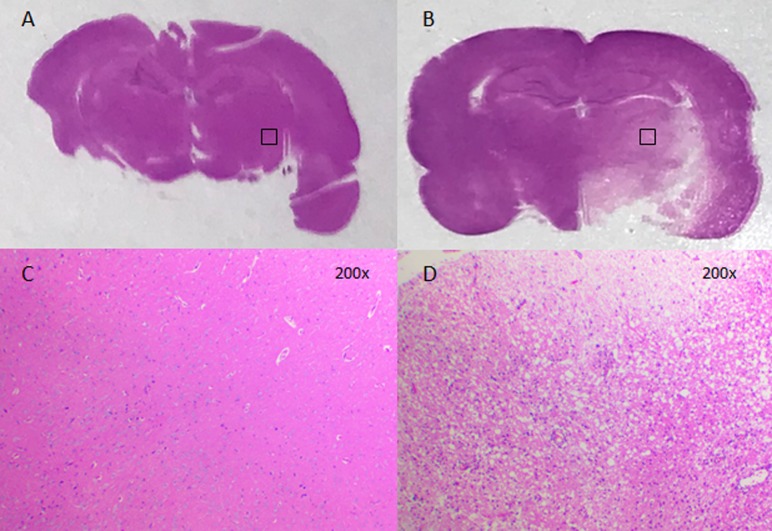
HE staining results suggested that the control group presented healthy tissues, whereas MCAO models presented an infarcted region in the striatum (Fig. 1A and B, respectively). In the lesion area, a large number of cells in the brain were observed to be necrotic following MCAO compared with the control, based on the presence of nuclear fragmentation and vacuolation (Fig. 1C and D).
Figure 1.
MCAO-induced rat cerebral injuries, as detected by hematoxylin and eosin staining. (A) Section of the brain of a rat in the control group. (B) Section of the brain of a rat following MCAO. Magnification of the area corresponding to the injury region in the (C) control group and in the (D) MCAO group. MCAO, middle cerebral artery occlusion.
Identification of DEmiRs in focal cerebral ischemia induced by MCAO
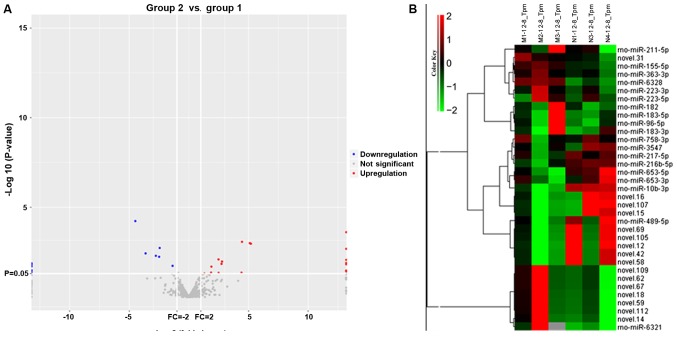
miRNA-seq was performed to identify the differential expression of miRNAs in the infarcted areas. In total, 37 DEmiRs were identified in the infarcted region, including 20 upregulated and 17 downregulated DEmiRs (n=3, P<0.05, log2 |fold change|>1). Volcano plot analysis and hierarchical clustering analysis of DEmiRs are presented in Fig. 2A and B, respectively.
Figure 2.
RNA-seq data corresponding to the DEmiRNAs between the MCAO model and control groups. (A) Hierarchical cluster of DEmiRNAs in the control vs. model groups. Green in the heat map represents downregulation and red represents upregulation. The average signals of the DEmiRNAs in each group were clustered using a Euclidean distance function. miRNAs exhibiting similar expression patterns are clustered together. n=3 in each group. (B) Volcano plots of transformed P-values (log10) against log2fold changes corresponding to transcripts per million reads in the two conditions examined. Blue and red dots represent differentially expressed miRNAs, whereas grey dots represent miRNAs that are not differentially expressed. RNA-seq, RNA sequencing; miRNA, microRNA; DEmiRNAs, differentially expressed microRNAs.
Differential expression levels of DEmiRs assessed by RT-qPCR analysis
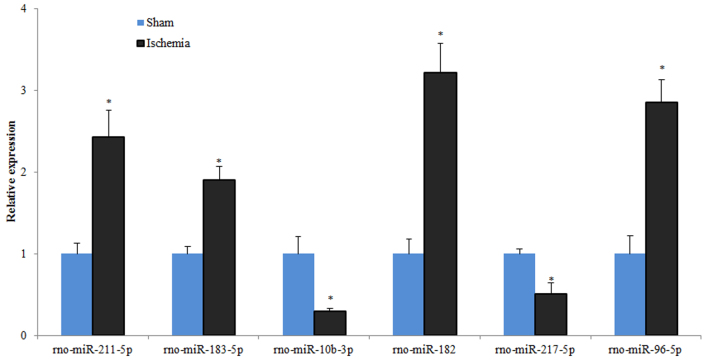
In order to confirm the sequencing results and to avoid the systematic bias caused by targeted selection, RT-qPCR analysis was performed. Therefore, six DEmiRs were randomly selected, including miR-211-5p, miR-183-5p, miR-10b-3p, miR-182, miR-217-5p and miR-96-5p, and their expression levels were assessed using RT-qPCR. miR-10b-3p and miR-217-5p were significantly downregulated, whereas miR-211-5p, miR-183-5p, miR-182 and miR-96-5p were upregulated in the infarcted area of MCAO rats compared with the control (Fig. 3). These findings were consistent with the results of miRNA-seq.
Figure 3.
Levels of expression of six miRs assessed by reverse transcription-quantitative polymerase chain reaction. n=3 in each group. *P<0.05 vs. sham control. miR, microRNA.
GO and KEGG analyses of the mRNAs targeted by the DEmiRs
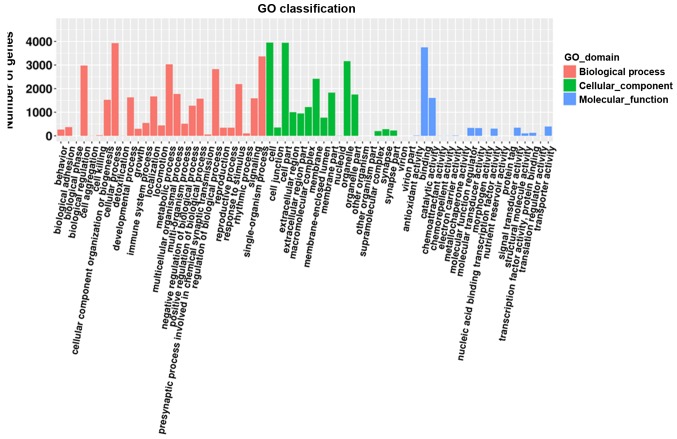
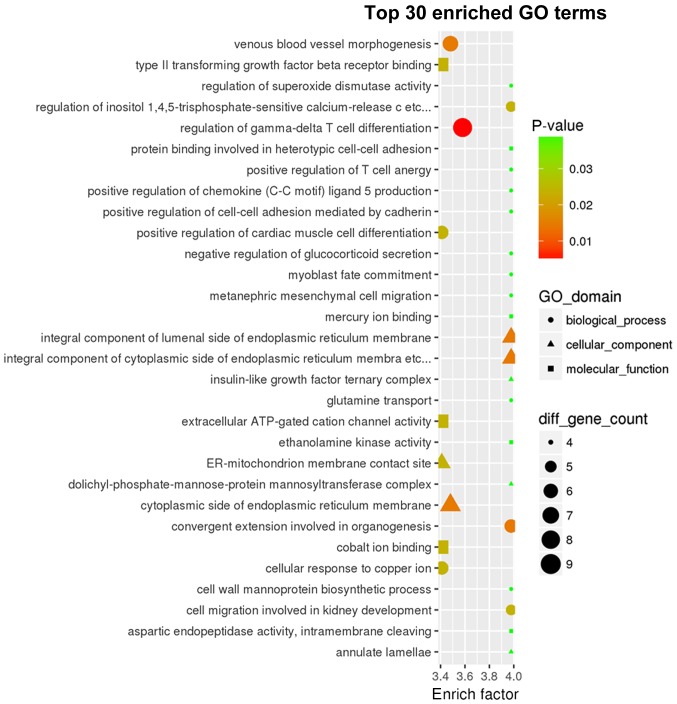
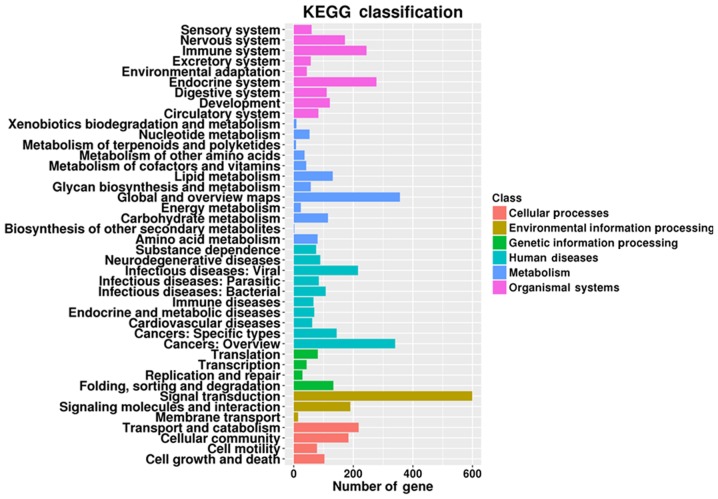
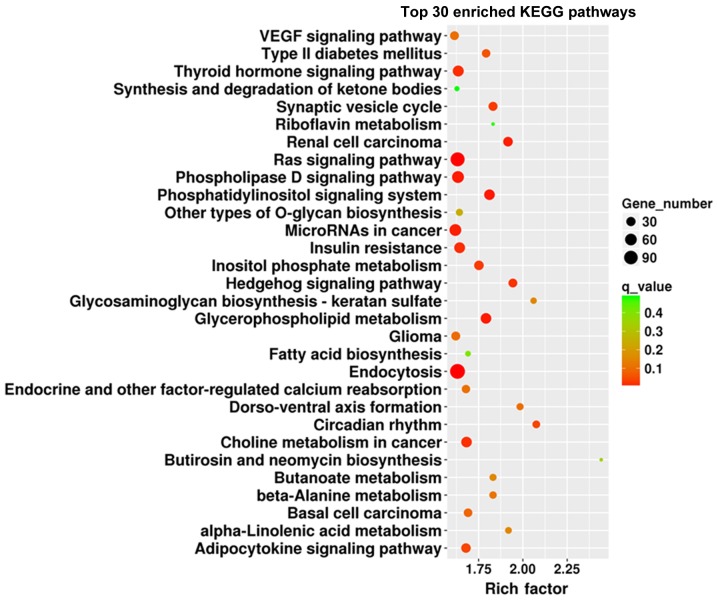
GO analysis was performed to identify the most enriched GO classes based on the number of genes involved in a specific pathway, and the 30 most enriched GO terms based on the corrected P-value of the target mRNAs regulated by the identified miRNAs (Figs. 4 and 5, respectively). The enriched GO terms included ‘regulation of γδdT-cell differentiation’, and ‘integral components of the cytoplasmic and luminal sides of the endoplasmic reticulum membrane’. In addition, enriched KEGG classes and pathways were identified using the genes targeted by the DEmiRs (Figs. 6 and 7, respectively). The genes targeted by the DEmiRs were identified to be involved in 42 subclasses of pathways included in six categories: ‘Human diseases’, ‘metabolism’, ‘genetic information processing’, ‘organismal systems’, ‘cellular processes’ and ‘environmental information processing’ (Fig. 6). ‘Ras signaling pathway’, ‘endocytosis’, ‘phospholipase D signaling pathway’, ‘microRNAs in cancer’ and ‘thyroid hormone signaling pathway’ were the most enriched KEGG pathways (Fig. 7).
Figure 4.
GO terms enriched in genes targeted by the differentially expressed microRNAs. GO, Gene Ontology.
Figure 5.
Top 30 most significant GO terms enriched in genes targeted by the differentially expressed microRNAs. GO, Gene Ontology.
Figure 6.
KEGG terms enriched in genes targeted by the differentially expressed microRNAs. KEGG, Kyoto Encyclopedia of Genes and Genomes.
Figure 7.
Top 30 KEGG pathways enriched in genes targeted by the differentially expressed microRNAs. KEGG, Kyoto Encyclopedia of Genes and Genomes.
Construction of the miRNA-mRNA network
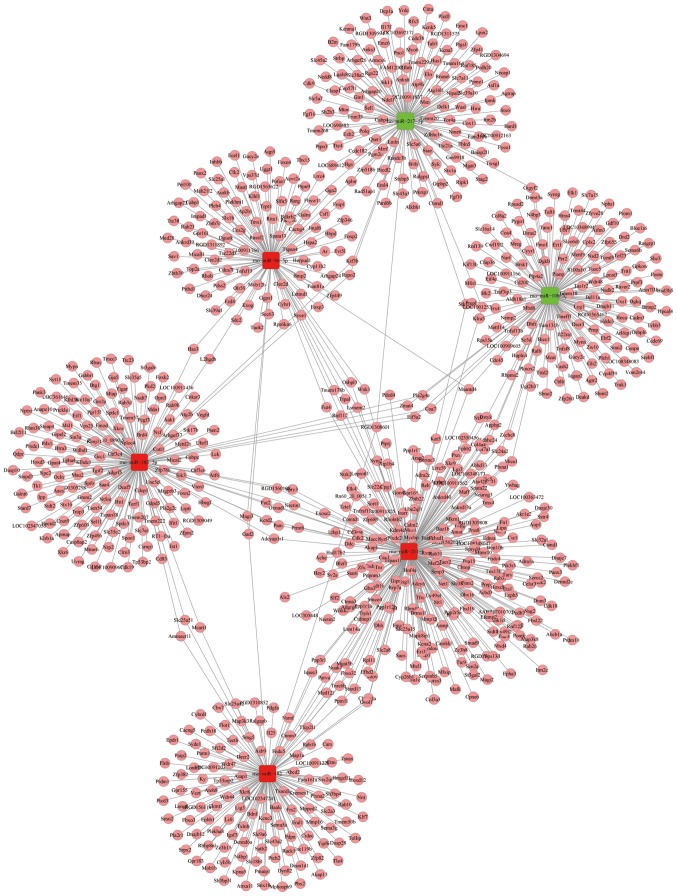
The mRNAs targeted by the DEmiRs were identified following miRanda analysis. Subsequently, an miRNA-mRNA network was constructed using all DEmiRs and their target mRNAs (data not shown). The miRNA-mRNA network generated using the six aforementioned DEmiRs was generated using Cytoscape (Fig. 8). Various miRNAs, possibly involved in ischemic injury, targeted numerous genes in the miRNA-mRNA network constructed. miRNAs regulating >100 genes may serve important roles in ischemic stroke (Table I). Additionally, target genes regulated by >10 DEmiRs are listed in Table II. Notably, receptor-type tyrosine-protein phosphatase F interacting protein α 1 (PPFIA1) was identified to be regulated by 24 miRNAs present in the constructed miRNA-mRNA network.
Figure 8.
miRNA-mRNA network constructed using six differentially expressed miRNAs that were confirmed by reverse transcription-quantitative polymerase chain reaction. Red indicates upregulated miRNAs and green indicates downregulated miRNAs. Pink elliptical nodes represent mRNAs.
Table I.
Differentially expressed miRNAs regulating genes with degrees >100.
| A, Upregulated miRNAs | |||||
|---|---|---|---|---|---|
| miRNA ID | MCAO group, TPM | Control group, TPM | log2 fold change | P-value | Degree |
| rno-miR-6328 | 3.342325 | 0.50154693 | 2.736396 | 0.014408 | 791 |
| rno-miR-6321 | 12.46518 | 0.00000000 | ∞ | 0.000246 | 403 |
| rno-miR-211-5p | 106.0241 | 18.9583318 | 2.483488 | 0.008072 | 284 |
| rno-miR-183-5p | 1,293.749 | 37.5922773 | 5.104978 | 0.001003 | 174 |
| rno-miR-182 | 1,084.191 | 49.5397822 | 4.451888 | 0.000846 | 150 |
| rno-miR-96-5p | 176.9584 | 4.8116489 | 5.200735 | 0.001077 | 120 |
| B, Downregulated miRNAs | |||||
| miRNA ID | MCAO group, TPM | Control group, TPM | log2 fold change | P-value | Degree |
| rno-miR-653-3p | 0.727656 | 9.03609878 | −3.63437 | 0.003726 | 461 |
| rno-miR-3547 | 6.158764 | 34.6958647 | −2.49405 | 0.005801 | 306 |
| rno-miR-10b-3p | 0.00000000 | 0.37617954 | 0.00000000 | 0.034244 | 166 |
| rno-miR-217-5p | 2.659196 | 14.5212507 | −2.4491 | 0.001864 | 140 |
TPM, transcripts per million reads; miRNA/miR microRNA. Degree refers to the mean number of miRNAs connected.
Table II.
Genes regulated by miRNAs with degrees >10.
| Gene symbol | Ensembl gene ID | Number of targeting DEmiRNAs | Gene name |
|---|---|---|---|
| PPFIA1 | ENSRNOG00000020857 | 24 | PTPRF interacting protein α 1 |
| SLC7A1 | ENSRNOG00000000924 | 12 | Solute carrier family 7 member 1 |
| NTRK2 | ENSRNOG00000018839 | 11 | Neurotrophic receptor tyrosine kinase 2 |
| KDM4B | ENSRNOG00000049221 | 11 | Lysine demethylase 4B |
| ST3GAL1 | ENSRNOG00000008209 | 10 | ST3 β-galactoside alpha-2,3-sialyltransferase 1 |
| TBC1D2 | ENSRNOG00000023348 | 10 | TBC1 domain family member 2 |
| MCF2L | ENSRNOG00000028426 | 10 | MCF.2 cell line derived transforming sequence like |
| VPS37C | ENSRNOG00000046439 | 10 | VPS37C subunit of ESCRT-I |
| TAS1R2 | ENSRNOG00000061876 | 10 | Taste 1 receptor member 2 |
For specific genes, degree refers to the mean number of miRNAs connected.
Discussion
miRNAs are important regulators of physiological and pathological processes, including ischemic and hypoxic damage (8–18). The present study aimed to identify miRNAs involved in ischemic injury and to examine their association with MCAO-induced stroke. The present results suggested that 20 miRNAs were upregulated and 17 were downregulated in brain infarcted areas. Notably, recent studies demonstrated that various miRNAs identified in the present study may be involved in multiple biological processes. For example, miR-96, miR-182 and miR-183 were identified to be associated with cochlear cell fate and homeostasis in a mouse model (30). Additionally, the expression levels of miRNA-183 were used as a predictor of recurrence and prognosis in patients with colorectal cancer (31). Furthermore, miR-182 may inhibit smooth muscle cell dedifferentiation via fibroblast growth factor 9 and platelet-derived growth factor receptor β signaling (32). A previous study identified that anti-miR182 may be used as a therapeutic strategy to treat high-grade serous ovarian carcinoma (33). miR-182 was identified to regulate the expression of clock circadian regulator in the pineal gland of neonatal rats following hypoxic-ischemic brain injury (16). miR-182 was observed to activate the RAS/mitogen-activated protein kinases signaling pathway by inhibiting RAS p21 protein activator 1 and sprouty-related EVH1 domain containing 1 in human oral cavity squamous cell carcinoma (34). Chang and Lai (17) demonstrated that miR-96 may be used as a biomarker of ischemia-reperfusion injury in skin tissues. A recent study observed that miR-96 may represent a potential anticancer therapeutic target to treat glioblastoma (35). miR-96 was previously observed to serve as an oncosuppressor gene by targeting NUAK family kinase 1 in pancreatic cancer (36). Additionally, a previous study identified that an increased expression of miR-211 may increase the risk of stroke by inhibiting angiopoietin-1, but the risk may be reduced in response to mutations in the 3′-UTR of angiopoietin-1 (18). miR-10b was identified to promote the migration and invasion of human cancer cells by increasing the expression levels of Jun proto-oncogene, AP-1 transcription factor subunit via neurofibromin 1 and ras homolog family member C (37). Similarly, the expression levels of miR10a/b were observed to be promoted by transforming growth factor β and may increase migration of human glioma cells by targeting phosphatase and tensin homolog (38). Furthermore, a previous study observed that miR-217 may inhibit osteogenic differentiation by regulating runt related transcription factor 2 (39), and miR-217 may inhibit the invasion and proliferation of gastric cancer and osteosarcoma cells (40,41). In the present study, the DEmiRs exhibiting the highest number of associations in the miRNA-mRNA network were miR-6328, miR-653-3p, miR-6321 and miR-3547. The role of miR-6328, miR-653-3p, miR-6321 and miR-3547 in the development of stroke remains unclear.
The miRNA-mRNA network proposed in the present study revealed that PPFIA1 may be regulated by 24 DEmiRs. PPFIA1 is a gene encoding liprin-α1 protein, which is involved in cell adhesion, integrin expression and cell-matrix interaction (42). Previous studies identified that liprin-α protein may be associated with synapse maturation and synaptic vesicular transport (43,44). PPFIA1 may serve as a tumor suppressor and regulates cell motility by interacting with inhibitor of growth family member 4 (45). A whole-genome association study identified that PPFIA1 may serve a role in acute lung injury induced by severe trauma (46). A previous study reported that solute carrier family 7 member 1 gene expression was required in insulin-stimulated L-arginine transport and was associated with human umbilical vein relaxation (47). It was observed that lysine demethylase 4B may be involved in mitochondrial-mediated apoptosis in colorectal cancer (48,49). miR-17 may target and downregulate the expression level of TBC1 domain family member 2, inhibiting cellular trafficking in HeLa cells (50).
In the present study, miRNA-seq was performed to identify the miRNAs exhibiting differential expression in infarcted brain areas following MCAO; however, the principal limitations of the present study include the use of only one software for target prediction, miRanda, which may lead to false positives. Further studies using a high number of prediction models are required to understand the molecular mechanism underlying the role of miRNAs in ischemic stroke following MCAO. Although the molecular mechanisms underlying the identified DEmiRs were not examined in the present study, our results suggested that they may be involved in the development of ischemic stroke. Therefore, further studies investigating the function of the miRNAs identified in the present study are required. In the future, the present research group aim to investigate the molecular mechanism underlying miR-211 function in ischemic stroke. The present data enrichment analysis performed on the genes targeted by the DEmiRs suggested that miR-211 may regulate cell proliferation and apoptosis via the cyclic guanosine monophosphate-dependent protein kinase (cGMP-PKG) signaling pathway, since the analysis of the target genes revealed that phosphoinositide-3-kinase regulatory subunit 5 was a predicted target of miR-211, which was demonstrated to regulate apoptosis via the cGMP-PKG pathway (51,52). Collectively, the present study investigated the association between miRNAs and cerebral ischemia and provided insight into the molecular mechanisms underlying stroke.
Acknowledgements
The authors would like to thank Mr. Qiang Fan (Ao-Ji Bio-tech Co., Ltd., Shanghai, China) for his help with data analysis.
Funding
The present study was financially supported by The National Key Research and Development Plan (grant nos. 2017YFC1701600 and 2017YFC1701601), National Natural Science Foundation of China (grant no. 81473387), Anhui Provincial Natural Science Foundation of China (grant no. 1508085QH191) and The Key Project of the National Science Fund of Anhui Province (grant nos. KJ2017A282 and KJ2017A284).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
XD and DP conceived and designed the study. XD and JG performed the experiments. QB, LX, JW and LW analyzed the data and drafted the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The present study was approved by The Animal Experiments Ethics Committee of The Anhui University of Chinese Medicine (Hefei, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Ceulemans AG, Zgavc T, Kooijman R, Hachimi-Idrissi S, Sarre S, Michotte Y. The dual role of the neuroinflammatory response after ischemic stroke: modulatory effects of hypothermia. J Neuroinflammation. 2010;7:74. doi: 10.1186/1742-2094-7-74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gürsoy-Ozdemir Y, Can A, Dalkara T. Reperfusion-induced oxidative/nitrative injury to neurovascular unit after focal cerebral ischemia. Stroke. 2004;35:1449–1453. doi: 10.1161/01.STR.0000126044.83777.f4. [DOI] [PubMed] [Google Scholar]
- 3.Mendell JT, Olson EN. MicroRNAs in stress signaling and human disease. Cell. 2012;148:1172–1187. doi: 10.1016/j.cell.2012.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ranganathan K, Sivasankar V. MicroRNAs-Biology and clinical applications. J Oral Maxillofac Pathol. 2014;18:229–234. doi: 10.4103/0973-029X.140762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ghibaudi M, Boido M, Vercelli A. Functional integration of complex miRNA networks in central and peripheral lesion and axonal regeneration. Prog Neurobiol. 2017;158:69–93. doi: 10.1016/j.pneurobio.2017.07.005. [DOI] [PubMed] [Google Scholar]
- 6.Lau P, Hudson LD. MicroRNAs in neural cell differentiation. Brain Res. 2010;1338:14–19. doi: 10.1016/j.brainres.2010.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.López-González MJ, Landry M, Favereaux A. MicroRNA and chronic pain: From mechanisms to therapeutic potential. Pharmacol Ther. 2017;180:1–15. doi: 10.1016/j.pharmthera.2017.06.001. [DOI] [PubMed] [Google Scholar]
- 8.Koutsis G, Siasos G, Spengos K. The emerging role of microRNA in stroke. Curr Top Med Chem. 2013;13:1573–1588. doi: 10.2174/15680266113139990106. [DOI] [PubMed] [Google Scholar]
- 9.Liu C, Peng Z, Zhang N, Yu L, Han S, Li D, Li J. Identification of differentially expressed microRNAs and their PKC-isoform specific gene network prediction during hypoxic pre-conditioning and focal cerebral ischemia of mice. J Neurochem. 2012;120:830–841. doi: 10.1111/j.1471-4159.2011.07624.x. [DOI] [PubMed] [Google Scholar]
- 10.Rink C, Khanna S. MicroRNA in ischemic stroke etiology and pathology. Physiol Genomics. 2011;43:521–528. doi: 10.1152/physiolgenomics.00158.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vinciguerra A, Formisano L, Cerullo P, Guida N, Cuomo O, Esposito A, Di Renzo G, Annunziato L, Pignataro G. MicroRNA-103-1 selectively downregulates brain NCX1 and its inhibition by anti-miRNA ameliorates stroke damage and neurological deficits. Mol Ther. 2014;22:1829–1838. doi: 10.1038/mt.2014.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dharap A, Bowen K, Place R, Li LC, Vemuganti R. Transient focal ischemia induces extensive temporal changes in rat cerebral microRNAome. J Cereb Blood Flow Metab. 2009;29:675–687. doi: 10.1038/jcbfm.2008.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Buller B, Liu X, Wang X, Zhang RL, Zhang L, Hozeska-Solgot A, Chopp M, Zhang ZG. MicroRNA-21 protects neurons from ischemic death. FEBS J. 2010;277:4299–4307. doi: 10.1111/j.1742-4658.2010.07818.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sepramaniam S, Tan JR, Tan KS, DeSilva DA, Tavintharan S, Woon FP, Wang CW, Yong FL, Karolina DS, Kaur P, et al. Circulating microRNAs as biomarkers of acute stroke. Int J Mol Sci. 2014;15:1418–1432. doi: 10.3390/ijms15011418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liu C, Zhao L, Han S, Li J, Li D. Identification and functional analysis of microRNAs in mice following focal cerebral ischemia injury. Int J Mol Sci. 2015;16:24302–24318. doi: 10.3390/ijms161024302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ding X, Sun B, Huang J, Xu L, Pan J, Fang C, Tao Y, Hu S, Li R, Han X, et al. The role of miR-182 in regulating pineal CLOCK expression after hypoxia-ischemia brain injury in neonatal rats. Neurosci Lett. 2015;591:75–80. doi: 10.1016/j.neulet.2015.02.026. [DOI] [PubMed] [Google Scholar]
- 17.Chang KP, Lai CS. Micro-RNA profiling as biomarkers in flap ischemia-reperfusion injury. Microsurgery. 2012;32:642–648. doi: 10.1002/micr.22046. [DOI] [PubMed] [Google Scholar]
- 18.Chen J, Yang T, Yu H, Sun K, Shi Y, Song W, Bai Y, Wang X, Lou K, Song Y, et al. A functional variant in the 3′-UTR of angiopoietin-1 might reduce stroke risk by interfering with the binding efficiency of microRNA 211. Hum Mol Genet. 2010;19:2524–2533. doi: 10.1093/hmg/ddq131. [DOI] [PubMed] [Google Scholar]
- 19.Duan X, Gan J, Xu F, Li L, Han L, Peng C, Bao Q, Xiao L, Peng D. RNA sequencing for gene expression profiles in a rat model of middle cerebral artery occlusion. Biomed Res Int. 2018;2018:2465481. doi: 10.1155/2018/2465481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bu X, Zhang N, Yang X, Liu Y, Du J, Liang J, Xu Q, Li J. Proteomic analysis of cPKCβII-interacting proteins involved in HPC-induced neuroprotection against cerebral ischemia of mice. J Neurochem. 2011;117:346–356. doi: 10.1111/j.1471-4159.2011.07209.x. [DOI] [PubMed] [Google Scholar]
- 21.Office of Laboratory Animal Welfare. https://grants.nih.gov/grants/olaw/references/dc96-1.htm
- 22.Kozomara A, Griffiths-Jones S. miRBase: Annotating high confidence microRNAs using deep sequencing data. Nucleic Acids Res. 2014;42:D68–D73. doi: 10.1093/nar/gkt1181. Database Issue. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mortazavi A, Williams BA, McCue K, Schaeffer L, Wold B. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods. 2008;5:621–628. doi: 10.1038/nmeth.1226. [DOI] [PubMed] [Google Scholar]
- 24.Robinson MD, McCarthy DJ, Smyth GK. edgeR: A bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics. 2010;26:139–140. doi: 10.1093/bioinformatics/btp616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Betel D, Wilson M, Gabow A, Marks DS, Sander C. The microRNA.org resource: Targets and expression. Nucleic Acids Res. 2008;36:D149–D153. doi: 10.1093/nar/gkm995. Database Issue. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 27.Yu G, Wang LG, Han Y, He QY. clusterProfiler: An R package for comparing biological themes among gene clusters. OMICS. 2012;16:284–287. doi: 10.1089/omi.2011.0118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gene Ontology Consortium, Blake JA, Dolan M, Drabkin H, Hill DP, Li N, Sitnikov D, Bridges S, Burgess S, Buza T, et al. Gene ontology annotations and resources. Nucleic Acids Res. 2013;41:D530–D535. doi: 10.1093/nar/gks1050. Database Issue. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kanehisa M, Goto S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000;28:27–30. doi: 10.1093/nar/28.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Weston MD, Tarang S, Pierce ML, Pyakurel U, Rocha-Sanchez SMR, McGee J, Walsh EJ, Soukup G. A mouse model of miR-96, miR-182 and miR-183 misexpression implicates miRNAs in cochlear cell fate and homeostasis. Sci Rep. 2018;8:3569. doi: 10.1038/s41598-018-21811-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chen Y, Song W. Wnt/catenin β1/microRNA 183 predicts recurrence and prognosis of patients with colorectal cancer. Oncol Lett. 2018;15:4451–4456. doi: 10.3892/ol.2018.7886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dong N, Wang W, Tian J, Xie Z, Lv B, Dai J, Jiang R, Huang D, Fang S, Tian J, et al. MicroRNA-182 prevents vascular smooth muscle cell dedifferentiation via FGF9/PDGFRβ signaling. Int J Mol Med. 2017;39:791–798. doi: 10.3892/ijmm.2017.2905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Xu X, Ayub B, Liu Z, Serna VA, Qiang W, Liu Y, Hernando E, Zabludoff S, Kurita T, Kong B, Wei JJ. Anti-miR182 reduces ovarian cancer burden, invasion, and metastasis: An in vivo study in orthotopic xenografts of nude mice. Mol Cancer Ther. 2014;13:1729–1739. doi: 10.1158/1535-7163.MCT-13-0982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wang J, Wang W, Li J, Wu L, Song M, Meng Q. miR182 activates the Ras-MEK-ERK pathway in human oral cavity squamous cell carcinoma by suppressing RASA1 and SPRED1. Onco Targets Ther. 2017;10:667–679. doi: 10.2147/OTT.S121864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Feng S, Yao J, Zhang Z, Zhang Y, Zhang Z, Liu J, Tan W, Sun C, Chen L, Yu X. miR96 inhibits EMT by targeting AEG1 in glioblastoma cancer cells. Mol Med Rep. 2018;17:2964–2972. doi: 10.3892/mmr.2017.8227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Huang X, Lv W, Zhang JH, Lu DL. miR96 functions as a tumor suppressor gene by targeting NUAK1 in pancreatic cancer. Int J Mol Med. 2014;34:1599–1605. doi: 10.3892/ijmm.2014.1940. [DOI] [PubMed] [Google Scholar]
- 37.Knirsh R, Ben-Dror I, Modai S, Shomron N, Vardimon L. MicroRNA 10b promotes abnormal expression of the proto-oncogene c-Jun in metastatic breast cancer cells. Oncotarget. 2016;7:59932–59944. doi: 10.18632/oncotarget.11000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Liu S, Sun J, Lan Q. TGF-β-induced miR10a/b expression promotes human glioma cell migration by targeting PTEN. Mol Med Rep. 2013;8:1741–1746. doi: 10.3892/mmr.2013.1709. [DOI] [PubMed] [Google Scholar]
- 39.Zhu YL, Wang S, Ding DG, Xu L, Zhu HT. miR-217 inhibits osteogenic differentiation of rat bone marrow-derived mesenchymal stem cells by binding to Runx2. Mol Med Rep. 2017;15:3271–3277. doi: 10.3892/mmr.2017.6349. [DOI] [PubMed] [Google Scholar]
- 40.Sun B, Yang M, Li M, Wang F. The microRNA-217 functions as a tumor suppressor and is frequently downregulated in human osteosarcoma. Biomed Pharmacother. 2015;71:58–63. doi: 10.1016/j.biopha.2015.04.008. [DOI] [PubMed] [Google Scholar]
- 41.Wang H, Dong X, Gu X, Qin R, Jia H, Gao J. The MicroRNA-217 functions as a potential tumor suppressor in gastric cancer by targeting GPC5. PLoS One. 2015;10:e0125474. doi: 10.1371/journal.pone.0125474. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 42.Pehkonen H, von Nandelstadh P, Karhemo PR, Lepikhova T, Grenman R, Lehti K, Monni O. Liprin-α1 is a regulator of vimentin intermediate filament network in the cancer cell adhesion machinery. Sci Rep. 2016;6:24486. doi: 10.1038/srep24486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Spangler SA, Hoogenraad CC. Liprin-alpha proteins: Scaffold molecules for synapse maturation. Biochem Soc Trans. 2007;35:1278–1282. doi: 10.1042/BST0351278. [DOI] [PubMed] [Google Scholar]
- 44.Miller KE, DeProto J, Kaufmann N, Patel BN, Duckworth A, Van Vactor D. Direct observation demonstrates that Liprin-alpha is required for trafficking of synaptic vesicles. Curr Biol. 2005;15:684–689. doi: 10.1016/j.cub.2005.02.061. [DOI] [PubMed] [Google Scholar]
- 45.Shen JC, Unoki M, Ythier D, Duperray A, Varticovski L, Kumamoto K, Pedeux R, Harris CC. Inhibitor of growth 4 suppresses cell spreading and cell migration by interacting with a novel binding partner, liprin alpha1. Cancer Res. 2007;67:2552–2558. doi: 10.1158/0008-5472.CAN-06-3870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Christie JD, Wurfel MM, Feng R, O'Keefe GE, Bradfield J, Ware LB, Christiani DC, Calfee CS, Cohen MJ, Matthay M, et al. Genome wide association identifies PPFIA1 as a candidate gene for acute lung injury risk following major trauma. PLoS One. 2012;7:e28268. doi: 10.1371/journal.pone.0028268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.González M, Gallardo V, Rodriguez N, Salomón C, Westermeier F, Guzmán-Gutiérrez E, Abarzúa F, Leiva A, Casanello P, Sobrevia L. Insulin-stimulated L-arginine transport requires SLC7A1 gene expression and is associated with human umbilical vein relaxation. J Cell Physiol. 2011;226:2916–2924. doi: 10.1002/jcp.22635. [DOI] [PubMed] [Google Scholar]
- 48.Deng WW, Hu Q, Liu ZR, Chen QH, Wang WX, Zhang HG, Zhang Q, Huang YL, Zhang XK. KDM4B promotes DNA damage response via STAT3 signaling and is a target of CREB in colorectal cancer cells. Mol Cell Biochem. 2018;449:81–90. doi: 10.1007/s11010-018-3345-5. [DOI] [PubMed] [Google Scholar]
- 49.Li H, Yang X, Wang G, Li X, Tao D, Hu J, Luo X. KDM4B plays an important role in mitochondrial apoptosis by upregulating HAX1 expression in colorectal cancer. Oncotarget. 2016;7:57866–57877. doi: 10.18632/oncotarget.11077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Serva A, Knapp B, Tsai YT, Claas C, Lisauskas T, Matula P, Harder N, Kaderali L, Rohr K, Erfle H, et al. miR-17-5p regulates endocytic trafficking through targeting TBC1D2/Armus. PLoS One. 2012;7:e52555. doi: 10.1371/journal.pone.0052555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Burley DS, Ferdinandy P, Baxter GF. Cyclic GMP and protein kinase-G in myocardial ischaemia-reperfusion: Opportunities and obstacles for survival signaling. Br J Pharmacol. 2007;152:855–869. doi: 10.1038/sj.bjp.0707409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Nagai-Kusuhara A, Nakamura M, Mukuno H, Kanamori A, Negi A, Seigel GM. cAMP-responsive element binding protein mediates a cGMP/protein kinase G-dependent anti-apoptotic signal induced by nitric oxide in retinal neuro-glial progenitor cells. Exp Eye Res. 2007;84:152–162. doi: 10.1016/j.exer.2006.09.010. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.