Abstract
Coronavirus disease 2019 (COVID-19) has been categorized as evolving in overlapping phases. First, there is a viral phase that may well be asymptomatic or mild in the majority, perhaps 80% of patients. The pathophysiological mechanisms resulting in minimal disease in this initial phase are not well known. In the remaining 20% of cases, the disease may become severe and/or critical. In most patients of this latter group, there is a phase characterized by the hyperresponsiveness of the immune system. A third phase corresponds to a state of hypercoagulability. Finally, in the fourth stage organ injury and failure occur. Appearance of autoinflammatory/autoimmune phenomena in patients with COVID-19 calls attention for the development of new strategies for the management of life-threatening conditions in critically ill patients. Antiphospholipid syndrome, autoimmune cytopenia, Guillain-Barré syndrome and Kawasaki disease have each been reported in patients with COVID-19. Here we present a scoping review of the relevant immunological findings in COVID-19 as well as the current reports about autoinflammatory/autoimmune conditions associated with the disease. These observations have crucial therapeutic implications since immunomodulatory drugs are at present the most likely best candidates for COVID-19 therapy. Clinicians should be aware of these conditions in patients with COVID-19, and these observations should be considered in the current development of vaccines.
Keywords: SARS-CoV-2, COVID-19, Autoimmunity, Antiphospholipid syndrome, Cytopenia, Guillain-Barré syndrome, Kawasaki disease, Cytokine storm syndrome, Vaccines
Highlights
-
•
Autoimmune and autoinflammatory conditions may be triggered by SARS-CoV-2.
-
•
Bystander activation and molecular mimicry could explain the appearance of these conditions.
-
•
In severe and critical patients, a cytokine storm syndrome (CSS) and a hypercoagulable state occur and may overlap.
-
•
CSS may promote the appearance of autoimmune and autoinflammatory-like conditions.
-
•
These observations should be considered in the current development of vaccines.
Abbreviations
- ACE2
Angiotensin converting enzyme 2
- ADs
Autoimmune diseases
- AOSD
Adult-onset Still's disease
- APS
Antiphospholipid syndrome
- ARDS
Acute respiratory distress syndrome
- CAR
Chimeric antigen receptor
- COVID-19
Coronavirus disease 2019
- CP
Convalescent plasma
- CSS
Cytokine storm syndrome
- Ct
Cycle threshold
- CXCL
Chemokine (C-X-C motif) ligand
- DCs
Dendritic cells
- ECs
Endothelial cells
- GBS
Guillain-Barré syndrome
- G-CSF
Granulocyte colony-stimulating factor
- GM-CSF
Granulocyte macrophage colony-stimulating factor
- HLH
Hemophagocytic lymphohistiocytosis
- HS
Healthy subjects
- ICU
Intensive care unit
- IFN
Interferon
- IgG
Immunoglobulin G
- IgM
Immunoglobulin M
- IL
Interleukin
- IL-1RA
Interleukin-1 receptor antagonist
- IP10
IFN-γ inducible protein
- IVIG
Intravenous immunoglobulin
- KD
Kawasaki disease
- LDH
Lactate dehydrogenase
- MAS
Macrophage activation syndrome
- MCP-1
Monocyte chemotactic protein 1
- MCP-3
Monocyte chemotactic protein 3
- MERS
Middle East respiratory syndrome
- MIP
Macrophage inflammatory proteins
- MIS-C
Multisystem inflammatory syndrome in children
- NAbs
Neutralizing antibodies
- NK
Natural killer
- NLRP3
Nod-like receptor 3
- PaCO2
Partial pressure of carbon dioxide
- PaO2
Partial pressure of oxygen
- PCR
Polymerase chain reaction
- PCT
Procalcitonin
- pDCs
Plasmacytoid DCs
- PIMS
Pediatric multisystem inflammatory syndrome
- RA
Rheumatoid arthritis
- RBD
Receptor binding domain
- RNA
Ribonucleic acid
- S
Spike protein
- SARS-CoV-2
Severe Acute Respiratory Syndrome-Coronavirus-2
- S-IgG
anti-S-IgG
- SLE
Systemic lupus erythematosus
- SS
Sjögren's syndrome
- Th
T helper
- TLR
Toll-like-receptor
- TMPRSS2
Transmembrane serine protease 2
- TNF
Tumoral necrosis factor
- WHO
World health organization
1. Introduction
In December 2019, there were the earliest reported clusters of patients with pneumonia of unknown origin epidemiologically linked to exposure at a seafood and wet animal market in Wuhan (Hubei Province, China) [1]. The cause of this pneumonia was rapidly identified as a new β-coronavirus, named Severe Acute Respiratory Syndrome-Coronavirus-2 (SARS-CoV-2). In January 2020, the World Health Organization (WHO) officially coined the term coronavirus disease 2019 (COVID-19) which rapidly became a pandemic worldwide. As of June 9th, 2020, over 7,1 million cases of COVID-19 have been confirmed globally (coronavirus.jhu.edu/map.html), with a 3–7% mortality rate that largely occurs in the 20% of the cases that develop severe disease, defined as patients with bilateral interstitial pneumonia [2]. In these cases, respiratory failure resembling acute respiratory distress syndrome (ARDS) is considered the leading cause of mortality [3].
From a pathogenesis standpoint, viral infections generally trigger a vigorous immune response that is crucial for viral clearance, with a cascade of events involving both the innate and adaptive immune arms in most of the cases. As COVID-19 is a new emerging disease, little is known about the immunological changes that occur in the infected human host, but several reports have been published describing the immunological alterations in patients with this condition. These range from a maladaptive immune response and abnormal cytokine/chemokine production, to hyperactivation of T cells and increased number of activated monocytes, macrophages and neutrophils, which may ultimately be associated with COVID-19 outcome [[4], [5], [6], [7], [8]].
It seems that COVID-19 shares a similar inflammatory immune response with autoinflammatory and autoimmune conditions. Viruses not only share immune responses with autoimmune diseases (ADs), but they can break immunological tolerance by a variety of mechanisms that include molecular mimicry, bystander activation and epitope spreading [[9], [10], [11]]. Some examples of viruses linked to autoimmunity and autoinflammation include enteric viruses for type I diabetes [12], hepatitis C virus for cryoglobulinemic vasculitis and Sjögren's-like syndrome [13,14], influenza viruses for acute disseminated encephalomyelitis [15], and herpesviruses for systemic lupus erythematosus (SLE), rheumatoid arthritis (RA) and adult-onset Still's disease (AOSD) [[16], [17], [18]]. Nowadays, it has been already linked SARS-CoV-2 with Guillain-Barré syndrome (GBS) [[19], [20], [21], [22], [23], [24], [25], [26], [27], [28]], autoimmune hemolytic anemia, immune thrombocytopenic purpura [[29], [30], [31], [32], [33], [34], [35]], and Kawasaki disease (KD) [[36], [37], [38], [39], [40], [41], [42]]. In addition, the study of critically ill patients with COVID-19 has drawn attention to an increased risk of thrombotic events which appeared to be associated with the presence of antiphospholipid antibodies [[43], [44], [45], [46]]. In children, the appearance of clinical manifestations resembling KD has drawn attention to a new phenotype of autoimmunity [38], and recent estimates suggest a 30-fold increased incidence of this disease during the pandemic [39]. Moreover, the immunomodulatory therapy widely used for ADs, its being used for COVID-19 [[47], [48], [49]]. Herein, a scoping review on current advances in the immunopathogenesis of disease as well as the auto-inflammatory and autoimmune conditions observed during SARS-CoV-2 infection is presented.
2. Auto-inflammatory versus autoimmune diseases
Immunological diseases are generally classified into three major groups: autoinflammatory, autoimmune and “mixed pattern diseases” [50]. Auto-inflammatory and ADs have several features in common since they are both systemic inflammatory diseases involving the muscle-skeletal system and characterized by a hyperactivation of the immune response in genetically predisposed individuals. However, there are some differences between the two groups. In autoinflammatory diseases the innate immune cells directly cause damage whereas in ADs the innate immune system activates the adaptive immune responses which are ultimately responsible for tissue inflammation [51].
From a clinical standpoint, ADs predominantly affect women and are characterized by the activation of T cells (cellular mediated responses) or B cells (i.e., presence of autoantibodies), or both, leading to pathology while the autoinflammatory diseases are invariably seronegative and manifest fever as the most common symptom. Periodic fever, Behçet disease, gout and AOSD are paradigmatic for autoinflammatory diseases while SLE, RA and SS are major examples of ADs. A third intermediate group, named “mixed pattern diseases”, comprises several conditions which do not completely fit with either classification; this is the case for example of spondyloarthropathies, such as ankylosing spondylitis and psoriatic arthritis, and inflammatory bowel diseases such as Crohn's disease [52]. Increasing knowledge of these diseases has revealed that the two major groups are in fact interconnected with the “mixed pattern diseases” representing a bridge between them as a continuum model from autoinflammation to autoimmunity [53]. Indeed, it is well known that the innate and adaptive immune system are strongly interconnected and crucial points of connection are represented by Toll-like receptors (TLR), IL-1β and inflammasome activation [[54], [55], [56]].
A paradigmatic example in this sense is given by the activation of severe psoriasis flares after upper respiratory tract infections. Patients affected by chronic plaque psoriasis, a bona fide AD, may develop acute flares clinically resembling the generalized pustular psoriasis, a clinical entity with molecular features of autoinflammatory syndromes (i.e., deficiency of IL-36 receptor antagonist) [57]. Interestingly, in these patients, rhinoviruses and coronaviruses have been the infectious agents most frequently identified as triggering factors for both autoimmune- (plaque psoriasis) and autoinflammation- (pustular psoriasis) linked forms of the psoriatic disease [57]. The pathogenetic differences between auto-inflammation and autoimmunity are mirrored by the different therapeutic approaches, particularly approaches that involve the use of biologic agents.
3. Genetic predisposition to disease
Environmental, epigenetic, and genetic factors influence autoinflammatory and ADs. The inner characteristics of a population may influence the development of such conditions when they are exposed to an infection. This explains why, despite the association of certain pathogens with a specific disease, there is still a considerable group of healthy individuals who, after exposure to a microorganism, does not develop the disease [58].
Both HLA and non-HLA polymorphisms are associated with inflammatory and ADs. Polymorphisms in HLA class I and HLA class II molecules affect which amino acids are in the peptide-binding groove and thus their binding specificity. In general, foreign antigens presented by class I molecules are derived from intracellular infections caused by viruses, or from proteins synthesized in the cytosol [58]. A small study in a Han population suggested that HLA-C*07:29 and B*15:27 alleles may be associated with COVID‐19 [59]. The first genome-wide association study in a European population, published as a preliminary report, showed that carriers of ABO A-positive group were at a 45% increased for respiratory failure, while individuals with blood group O were at a 35% decreased risk for respiratory failure [60]. In addition, a cluster of genes that could be relevant to the development of severe COVID-19 was identified on chromosome 3p21. One of these genes, SLC6A20, encodes a transporter protein that interacts with angiotensin-converting enzyme 2 (ACE2), the SARS-CoV-2 receptor required for cell entry [60].
Host-pathogen interactions are vital to the understanding of infectious disease, as well as its treatment and prevention. In this sense, considering the genetic variation at the genome of SARS-CoV-2 (www.gisaid.org) will contribute to the knowledge of SARS-CoV-2 pathogenesis [61].
4. SARS-CoV-2 infection and the innate immune response
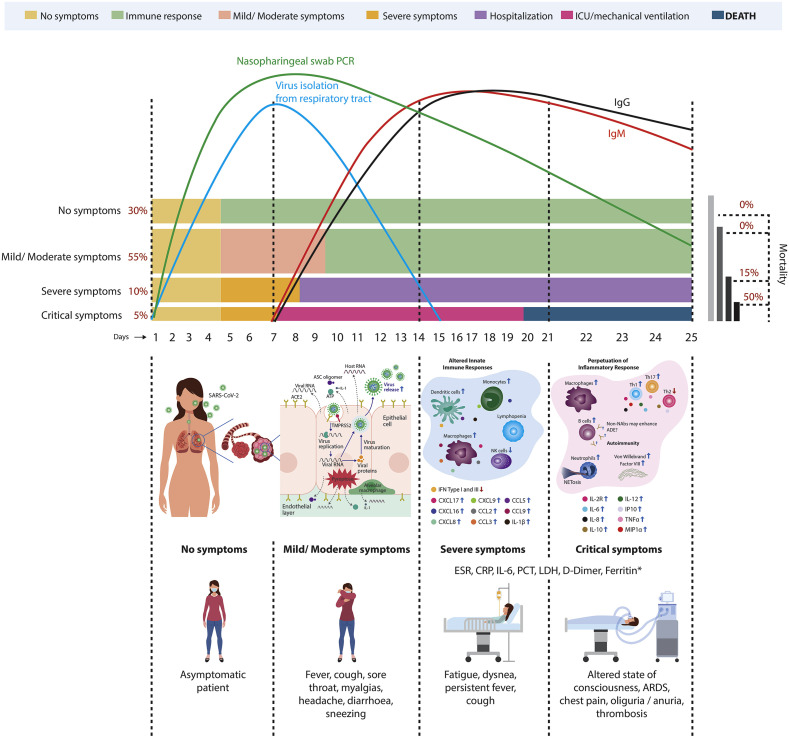
Lineage B coronaviruses, which include SARS-CoV and SARS-CoV-2 interact with the host receptor ACE2 for viral entry. The interaction is mediated by the receptor binding domain (RBD) region within the spike protein (S) of the virus, the latter common to all β coronaviruses. The ACE2 receptor is expressed in the lungs, small intestine, testis, kidneys, heart, thyroid, adipose tissue, brain, blood vessels, and muscle [[62], [63], [64], [65]]. After viral-receptor fusion in both SARS-CoV and SARS-CoV-2 infections, host proteases (such as transmembrane serine protease 2 (TMPRSS2) [66], endosomal cysteine proteases, cathepsin B and L) cleave the S protein of the virus leading to the release of the spike-fusion peptide which enables intracellular virus entry. Zang et al. [67], have shown that besides TMPRSS2, TMPRSS4 present in gut epithelial cells also contributes to enhance the localized tissue infectivity of SARS-CoV-2 (Fig. 1 ) [[4], [5], [6], [7], [8]].
Fig. 1.
Translational overview of COVID-19. Kinetics of IgM and IgG antibodies is shown. The dynamics of SARS-CoV-2 seroconversion is still under study as is the long-term immunity. Positivity of PCR for SARS-CoV-2 could last more than 25 days after the onset of disease. About 80% of patients develop no symptoms or present with a mild/moderate disease. Initially, infection of SARS-CoV-2 through the ACE2 receptor decreases the production of IFN type I and III, with a paradoxical increased secretion of chemokines which stimulate migration of innate immune cells to the lungs. This process takes place in the early stages of the disease. Then, migration of T and B cells, stimulated by chemokines, favors an increase of Th1/Th17 cytokines that perpetuate inflammation. Other cells such as neutrophils are thought to produce NETosis which may help to increase inflammation and produce the release of cryptic antigens leading to autoimmune phenomena. *All these markers are risk factors of progression of disease. Adapted from Ref. [[4], [5], [6], [7], [8]]. ACE2: Angiotensin-converting enzyme 2; ADE: Antibody-dependent enhancement; ARDS: Acute respiratory distress syndrome; ASC: Apoptosis-associated speck-like protein containing a CARD; CCL: Chemokine (C–C motif) ligand; CRP: C reactive protein; CXCL: chemokine (C-X-C motif) ligand; ESR: Erythrocyte sedimentation rate; ICU: Intensive care unit; Ig: Immunoglobulin; IFN: Interferon; IL: Interleukin; IP10: Interferon-inducible protein 10; LDH: lactate dehydrogenase; NAbs: Neutralizing antibodies; NK: Natural killer; PCR: Polymerase chain reaction; PCT: Procalcitonin; RNA: Ribonucleic acid; MIP1α: Macrophage inflammatory protein 1α; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; Th: T helper; TMPRSS2: Transmembrane protease serine 2; TNF: Tumoral necrosis factor.
Reduction in percentages of some innate immune cells may play an important role in COVID-19. Low eosinophil count has been suggested as a poor prognostic marker in COVID-19 patients, although the precise mechanisms remain unknown. Du et al. [68], described 85 fatal cases of COVID-19 with 81% of the patients having very low eosinophil count at admission. Additionally, other reports showed that low eosinophil count was less frequent in survivors of severe COVID-19 and in non–severe patients [69], than in a small cohort of recovering patients [70] confirming the potential association of low eosinophil count and poor prognosis.
Reduced percentages of circulating natural killer (NK) cells have also been reported [71], as well as an increased expression of their inhibitory receptor NKG2A that likely contributes to their reduced cytolytic activity [72]. Moreover, bronchoalveolar lavage fluid transcriptome from COVID-19 patients revealed an increase in dendritic cells (DCs) and activated neutrophils [73]. Higher percentages of inflammatory monocytes have been recorded in patients with severe lung pathology [74].
Several studies have shown a torrent of pro-inflammatory cytokines (Table 1 ) [[74], [75], [76], [77], [78], [79], [80], [81], [82], [83], [84], [85]]. It is quite possible that much like SARS-CoV and Middle East Respiratory Syndrome (MERS)-CoV, SARS-CoV-2 could induce a delayed type I IFN response with a loss of viral control during the early stage of infection [86]. Blanco-Melo et al. [76], showed in animal models and select patients with COVID-19 that reduced type I and III IFN are associated with subsequent failure in the control of virus replication. This phenomenon was observed at initial stages, i.e., the first 7 days post-infection with a paradoxical increased production of chemokines such as CXCL17, CXCL16, CXCL9, CXCL8. These chemokines may enhance migration of additional innate cells, such as monocytes, macrophages and neutrophils. Decreased production of type I and III IFN is maintained during the late stages of disease (14 days post infection), accompanied by increased production of pro-inflammatory cytokines such as IL-6, IL-8, and TNF-α.
Table 1.
Cytokines and chemokines in severe COVID-19 patients.
| Cytokine/Chemokine | Main Source | Effects | Patients | Reference |
|---|---|---|---|---|
| IL-6 | CD4+ T cells, CD14+ CD16+ monocytes ND |
High risk of inflammatory cytokine storm caused by monocytes migration to the lung Impairment of cytotoxic activity |
High levels in ICU patients High levels in ICU patients |
[74] [75] |
| IFN-γ/GM-CSF | CD4+/CD8+T cells | Pathogenic Th1 cells play a role in hyper-inflammatory responses | Co-expressing IFN-γ and GM-CSF only in ICU patients | [74] |
| CXCL10/CXCL9/CCL2/IL1RA/ | ND | T cell stimulation and myeloid recruiting chemokines | A subset of COVID-19 patients have 6–8 times more CXCL10 concentration than controls | [76,85] |
| Several cytokines* | ND | Viral load and lung injury | High levels in severe patients | [77] |
| IL-8 | ND | Together with IL-6 and TNF-α prediction of disease severity and death | High levels in hospitalized patients | [78] |
| TNF-α | ND | Inducing T cell loss in severe patients | High levels in ICU patients | [79] |
| IL-10 | ND | IL-10 negatively regulating T cell survival or proliferation | High levels in ICU patients | [80] |
| IL-2 | ND | T cell activation | High levels in severe patients | [81] |
| IP-10/MCP-3 | ND | Disease deterioration and fatal outcome | High levels in severe patients | [82] |
| IL-1RA/RANTES | ND | Impaired immune homeostasis and increased viral load | High levels in critical patients | [83] |
| RANTES | ND | Predictor of mild disease | High levels in early stage of the infection | [84] |
| Eotaxin | ND | Decreased eosinophil recruitment | Low levels in COVID-19 patients | [85] |
IL: Interleukin; IFN-γ: Interferon gamma; GM-CSF: Granulocyte macrophage colony-stimulating factor; TNF-α: Tumor necrosis factor alpha; IP-10: Interferon gamma-induced protein 10; MCP-3: Monocyte chemotactic protein-3; IL-1RA: Interleukin-1 receptor antagonist; Th1: T helper 1 cells; ICU: Intensive care unit; Several cytokines*: M-CSF, IL-10, IFN-α2, IL-17, IL-4, IP-10, IL-7, IL-1RA, G-CSF, IL-12, IFN-γ, IL-1α, IL-2, hepatocyte growth factor, and platelet derived growth factor-BB.
Other cells involved in COVID-19 disease are the pulmonary endothelial cells (ECs). These cells may be associated with the development of pulmonary complications such as ARDS due to the modification of vessel barrier integrity, promotion of the pro-coagulative state, and dysregulation of inflammatory cells infiltration secondary to vascular inflammation (i.e., endothelialitis). ECs have an important role in ARDS and multi-organ failure, therefore therapeutic strategies related to these cells should be studied [87]. In fact, van de Veerdonk et al. [88], have proposed that pulmonary edema in COVID-19 is caused by activation of bradykinin 1-2 receptors on ECs. Therefore, inhibiting kallikrein activity could prevent ARDS.
5. SARS-CoV-2 infection and the adaptive immune response
Lymphopenia can be caused by viral infections and is commonly found in autoinflammatory and ADs [89]. In physiological conditions, T cells undergo homeostatic proliferation following episodes of lymphopenia induced by infectious agents, to restore their population in the peripheral blood [90], and this proliferative capacity correlates with their avidity for self-antigens. Several hypothetical mechanisms could explain lymphopenia in COVID-19 patients: 1) lymphocytes express ACE2 and these die due to viral infection, 2) virus directly destroy lymphatic organs that includes destruction of lymphoid cells, 3) the generation of cytokine storm may lead to lymphocyte apoptosis or block of lymphopoiesis [91], 4) metabolic alterations induced by the virus infection leads to the generation of molecules that result in lymphoid cell depletion [92] and finally, 5) lymphopenia could also reflect differences in lymphocyte homing into tissues out of the peripheral blood.
Studies on the changes in lymphocyte subsets demonstrated reduced percentages of CD4+ and CD8+ T cells as well as B cells in peripheral blood, especially in patients with severe disease [71]. A lymphopenic state could lead to failure in the maintenance of peripheral tolerance that leads to the activation of effector T cells with autoimmune potential. This mechanism of loss of self-tolerance highlights the paradoxical association between lymphopenia and autoimmunity [93]. Such a scenario is supported by the speculation forwarded previously that a temporary lymphopenia induced by viral infections might trigger autoimmunity [94].
After infection occurs, CD4+ T cells are activated in secondary lymphoid organs followed by their migration into the inflamed tissue. After pathogen clearance, the majority of these cells undergo apoptosis but a small population remains, becoming long lived memory cells, able to respond more effectively and rapidly during a subsequent infection [95]. This phenomenon is known as immunological memory and can be a double-edge sword. When these memory cells are formed against self-antigens, in fact, they are implicated in autoimmune flare ups [96].
According to preliminary published data, COVID-19 critical patients express decreased percentages of T helper memory and regulatory T cells in the blood compared to patients with less severe disease [71]. These findings raise unsolved questions. Is the loss of these cell subsets due to their selective trafficking to the lung, or is it because they are specifically targeted by the virus? More detailed studies about the phenotype of T cells in COVID-19 patients showed that both, CD4+ and CD8+ T cells express higher levels of the T cell activation markers (e.g., CD38 or CD44) [97]. Of interest, CD44 is an adhesion molecule incriminated in the pathogenesis of a variety of ADs, and has been proposed as a biomarker of disease and disease activity for SLE [98].
Evaluation of patients at the intensive care unit (ICU) disclosed an increase of OX40 expressing CD4+ T cells, and CD137 expressing CD8+ T cells [74]. OX40 is known to promote T cell cytokine production [99] while CD137, a member of the TNF receptor family, is a potent costimulatory molecule for activated T cells preferentially involving cytotoxic CD8+ T cells and CD8 memory cells [100]. Additional phenotypic studies showed an increased concentration of highly pro-inflammatory CCR6+ Th17 cells and a reduction in the number of CD28+ cytotoxic suppressor T and regulatory T cells [71,101]. It is possible that a finding of sustained decrease in the CD8+ T cell subset (lack of viral clearance by secretion of perforin, granzymes, and IFN-γ) could serve as an independent predictor of COVID-19 severity [102]. Reduction of regulatory T cells and over activation of T cells, manifested by increase of Th17 and high cytotoxicity of CD8+ T cells are described in several autoinflammatory/ADs and the Th17 subset is known to be highly pro-inflammatory and is implicated in the pathogenesis of multiple autoinflammatory/autoimmune conditions. The balance between these two cell populations is critical for health [103,104].
Two less obvious pathways that are altered in COVID-19 include IL-7 and p53. First, an increased expression of IL-7 has been reported in peripheral blood during COVID-19. IL-7 is a hematopoietic growth factor secreted by stromal cells in the bone marrow and DCs, which stimulates the differentiation and proliferation of all cells of the lymphoid lineage. Increased IL-7 in severe COVID-19 disease may represent an attempt to counteract SARS-CoV-2-induced lymphopenia. Second, Xiong et al. [105], found upregulation of pathways associated with apoptosis, autophagy, and p53 in peripheral blood mononuclear cells of COVID-19 patients. Some studies reported that lymphopenia might be related to mortality [106]. Lymphopenia was also found in MERS patients. MERS-CoV can directly infect human T cells and induce their apoptosis without a requirement for virus replication [107]. SARS-CoV-2 can similarly infect T cells, but not replicate within them. However whether this virus induces T cell apoptosis needs further investigation [108]. Furthermore, in COVID-19 a higher expression of PD1+Tim3+ T cells and an increased expression of NKG2A by CD8+ T cells have been observed, indicating that SARS-CoV-2 virus induces T cell exhaustion in COVID-19 patients [72,79].
Concerning B cells, a case report from Australia first described the kinetics of antibody responses in COVID-19 [109]. Immunoglobulin M (IgM) and immunoglobulin G (IgG) antibodies that bound SARS-CoV-2 virus were detected in blood before symptomatic recovery. These immunological changes persisted for at least 7 days following full resolution of symptoms [109]. In a small report, sera from 5 COVID-19 patients were able to neutralize SARS-CoV-2 in vitro suggesting a possible disease-suppressive humoral response [110]. A more recent study investigated the diagnostic potential of serological ELISA-based tests in 85 patients with confirmed COVID-19 and 24 suspected patients. This study showed that IgM and IgG antibodies against SARS-CoV-2 can be detected as early as day 4 post infection with readily detectable IgG antibodies by day 11 and ongoing post infection. These serological tests thus appear to be reasonably sensitive and have a good specificity for COVID-19 diagnosis. The IgG seropositive rate decreased at 28 days after disease onset, however the sample numbers tested (n = 7) were small and the results await confirmation [111].
Long et al. [7], by using a magnetic chemiluminescence enzyme immunoassay reported that seroconversion for IgG and IgM may occur simultaneously or sequentially. In their sample, 12.2% of the patients reached a plateau in IgG titer within 7 days of symptom onset [7]. More recently, Xu et al. [112], by using the same method, confirmed this result which suggest that “the value of IgM as an early marker for the acute phase of SARS-COV-2 infection might not be on par with that in other viral infection diagnostics”. The lack of similar methods and standardization precludes comparability among the studies.
Fafi-Kremer et al. [113], carried out a serological study in patients with mild SARS-CoV-2 infection, asserting that 80% of the population present subclinical or mild COVID-19. The authors showed that 13 days post-disease onset, 99% of the patients had antibodies against SARS-CoV-2-S protein and 97% of the patients had neutralizing antibodies one month after disease onset. Interestingly, neutralization capacity correlated with antibody levels [114,115]. Although recovered patients from COVID-19 without hospitalization and mild disease have low levels of NAbs, they do have potent antiviral activity and persist up to 40 days after symptoms onset [113,116]. The identification of anti-SARS-CoV-2 antibodies in recovered patients, which can bind and neutralize the virus, supports the use of convalescent plasma (CP), as a therapeutic approach of artificial passive immunity [113,117]. Up to now, there are several reports that confirm that NAbs against RBD can interrupt the interaction between SARS-CoV-2 and ACE2, thus controlling the infection [115,118].
Even though anti-viral neutralizing antibodies are important for the viral clearance, some authors are questioning if the presence of a premature humoral response against SARS-CoV-2 might be harmful. Previous studies in SARS-CoV animal models showed that antibodies against the spike protein (anti-S-IgG) shares the capability to cause severe lung injury by interfering with the inflammatory response, by promoting pro-inflammatory monocyte/macrophages accumulation and their release of the pro-inflammatory cytokine IL-8 [119]. Based on these observations, it has been speculated that SARS-CoV-2 shares a similar inflammatory response, with antibody-mediated lung damage through skewing of macrophages or through complement activation and antibody-dependent cell-mediated cytotoxicity mechanisms [120].
Although the safety of CP based therapies is still under investigation, recent and strong evidence suggest that this therapy is safe and could offer high rates of efficacy in COVID-19 (i.e., reduction of viral load, increase in neutralizing antibodies and improvement of clinical status) [121]. A study of 5000 patients showed that less than 1% of patients presented major adverse events such as transfusion-associated circulatory overload, transfusion-related acute lung injury, and/or severe allergic transfusion reactions [122]. In addition, the adverse events mechanisms associated with CP infusion suggest that they are similar to intravenous immunoglobulins (IVIG), which include anti-idiotype reactions, neutralization of complement and inflammatory cytokines, and reduction of migration of T and B cells, as well as macrophages [121].
6. Cytokine storm syndrome (CSS)
Several studies have now documented the presence of significantly high plasma levels of cytokines and chemokines in patients with COVID-19. Such chemokines recruit lymphocytes and leukocytes to sites of infection and include IL-1β, IFNγ, IFNγ-inducible protein (IP10), and monocyte chemotactic protein (MCP)-1 each of which are elevated in COVID-19 compared to healthy subjects (HS), suggesting an activated Th1 cell response (Fig. 1). In patients requiring ICU admission, higher concentrations of G-CSF (granulocyte colony-stimulation factor), IP10, MCP-1, macrophage inflammatory proteins (MIP)-1α, IL-2, IL-7, IL-10 and TNFα, have been detected compared to patients not requiring ICU admission. In the same study, IL-6 plasma levels were significantly higher in ICU patients compared to HS but not compared to non-ICU patients [123].
Several cytokines are involved in Th17-type responses in COVID-19. IL-1β and TNFα promote Th17 responses. Along with Th1, Th17-type response contributes to high levels of pro-inflammatory cytokines in the context of a CSS [124].
CSS are a group of disorders representing a variety of inflammatory etiologies with the final common result of overwhelming systemic inflammation, hemodynamic instability, multiple organ dysfunction, and potentially death. CSS is not a disease itself, but rather the common end point of different initial insults: infectious, autoimmune/inflammatory, and iatrogenic [125]. The hemophagocytic syndromes hemophagocytic lymphohistiocytosis (HLH) and macrophage activation syndrome (MAS) represent two clinically similar CSS with an unknown degree of overlap and pathophysiology. The clinical presentations of all CSS can be strikingly similar, creating diagnostic uncertainty [125].
In a report from 150 patients with mild symptoms or severe disease, IL-6 was found to be significantly higher in the latter group and possibly predictive of mortality [3]. In some patients, increased levels of IL-10 and IL-4 have also been reported, suggesting that a Th2 response is also initiated (Table 1) [126]. This line of evidence suggests that the most severe subgroup of patients might experience a CSS, elsewhere defined as a systemic inflammatory response that can be triggered by a variety of factors such as infections and certain drugs. CSS is characterized by a variety of symptoms including both mild, flu-like symptoms and severe life-threatening manifestations such as ARDS, because of the released of high levels of pro-inflammatory cytokines (Fig. 2 ).
Fig. 2.
Clinical manifestations in the cytokine storm syndrome. This condition is considered as a common end point of different initial insults: infectious, autoimmune/inflammatory, and iatrogenic. Patients with CSS exhibit a plethora of signs and symptoms that compromise several systems. These manifestations resemble those encountered in patients with COVID-19. CSS: Cytokine storm syndrome; CRP: C reactive protein; ESR: Erythrocyte sedimentation rate; PaO2: Partial pressure of oxygen in arterial blood; PaCO2: Partial pressure of carbon dioxide.
In the last few years, this condition has become of great interest especially among oncologists, since it represents an important adverse event following chimeric antigen receptor (CAR)-T cell immunotherapy for cancer [127]. The understanding of CSS pathophysiology is largely incomplete, but a model has been proposed in which the target cell lysis induces the T cell activation and cytokine release (Fig. 3 ) [128], while innate immune cells are also activated with further cytokine production.
Fig. 3.
Cytokine network in the cytokine storm syndrome. IFNγ has been recognized as a common mediator of inflammation in CSS, especially in MAS. The production of IFNγ is stimulated by IL-1β, IL-18, and IL-33, which are associated with inflammasome response and play a critical role in inflammation via NK and T cells. IL-6 has been suggested as the central role of CSS. In addition, high levels of IL-10 are thought to be an unsuccessful attempt to compensate inflammation induced by the inflammasome activation. Adapted from Ref. [128]. CSS: Cytokine storm syndrome; IFN: Interferon; IL: Interleukin; MAS: Macrophage activation syndrome; NK: Natural killer; TNF: Tumoral necrosis factor.
IL-6 seems to play a dominant role in CSS, since high levels of IL-6 can activate the coagulation pathway and vascular endothelial cells and inhibit myocardial function [129]. Indeed, elevated IL-6 levels are observed in patients with CSS and important beneficial effects of IL-6 blockade by tocilizumab (an anti-IL-6 receptor antibody) are achieved in patients with CSS induced by CAR-T cell therapy [129]. Tocilizumab, which is widely used to treat RA and giant-cell arteritis, was first used in China in 21 COVID-19 patients in critical conditions with remarkable improvements [130]. Since this first report, IL-6 blockade strategy has been applied to treat other COVID-19 patients, including Italian patients in different areas of the country, with promising preliminary results.
A phase II clinical trial, investigating the efficacy and tolerability of tocilizumab in patients with COVID-19 pneumonia, has been completed and results await data analyses [131]. Given the condition of hyperinflammation found in these patients, selective inhibition of other pro-inflammatory cytokines such as IL-1β signaling by therapeutics such as anakinra or canakinumab has also been suggested [132]. Giamarellos-Bourboulis et al. [133], published an interesting study indicating that patients with COVID-19 and severe respiratory failure may present with a condition similar to MAS, driven by IL-1β, or to an immune dysregulation, driven by IL-6. These data provide support and the rationale for clinical trials of anakinra or tocilizumab, respectively, as therapeutic strategies for COVID-19 patients experiencing CSS.
In addition, the CSS may trigger the activation of auto-reactive T and B cells in autoimmune susceptible individuals, a phenomenon known as bystander activation which occurs when CD8+ T, CD4+ T, or B cells are activated in an antigen-independent manner [11]. Viral infections such as cytomegalovirus, Epstein-Barr virus, and hepatitis B virus are examples of infectious agents that appear to recognize viral epitopes that mimic self-epitopes, and trigger bystander activation via this mechanism [11]. Although there is no evidence regarding this issue in COVID-19, the disproportionate inflammatory response associated with SARS-CoV-2 may explain the appearance of autoimmune phenomena in the end-stages of the disease, including neurological and coagulopathy manifestations (Fig. 2).
Some patients with prior autoimmune and autoinflammatory conditions infected with SARS-CoV-2 have shown better outcomes may be due to baseline immunomodulatory medications. Some reports have shown that patients with ADs with prior treatment with hydroxychloroquine, TNFα antagonists [134], Anakinra [135], or Tocilizumab [136], may develop a mildest SARS-CoV-2 infection. These phenomena could be associated with a better control to COVID-19, and it may suggest that these medications, used chronically, decrease the severity of this infection. However, further analyses are warranted in which other factors associated with COVID-19 severity are included.
In contrast, those patients treated with Rituximab [137], or Secukinumab appeared to have worst outcome defined by a higher rate of ICU admissions [138]. Those patients treated with Rituximab and Secukinumab exhibited high concentrations of IL-6, suggesting that these medications failed to modulate IL-6 which has been strongly associated with mortality in COVID-19 [138]. In addition, CD20 blockers could impair B cells function hindering the production of NAbs against SARS-CoV-2 [138].
Since patients with ADs are prone to infections [58], a major concern is the possibility of a high rate of infection of SARS-CoV-2 in them. Emmi et al. [139], found an estimated frequency of 0.22% (0.01–1.21%) SARS-CoV-2 infections in their cohort of Italian patients with ADs which was comparable to that observed in the general population. In another study in patients with autoimmune liver conditions (i.e., autoimmune hepatitis and primary biliary cholangitis), authors did not find an increased frequency of cases of SARS-CoV-2 [140].
These results should be evaluated with caution. First, health systems worldwide have recommended that those patients with chronic conditions including ADs must avoid physical contact. These recommendations could influence the risk of SARS-CoV-2 infection and may bias the estimate of its prevalence in ADs. Second, as discussed above, patients with autoimmune and autoinflammatory conditions could exhibit a mild COVID-19. This scenario may influence the rate of clinical consultation by these patients, since most of infected individuals only visit clinical settings in situations of a severe disease or flares. Therefore, the questions about the risk and prevalence of COVID-19 in patients with inflammatory and ADs remains open.
7. Macrophage activation syndrome versus autoimmunity
MAS is seen most frequently in children with systemic juvenile idiopathic arthritis and in its adult equivalent, AOSD. However, it is increasingly reported in other rheumatic disease of childhood, including pediatric SLE, KD, juvenile dermatomyositis, and antiphospholipid syndrome (APS) [141]. Episodes of MAS appear to be most commonly triggered by infections, particularly viral infections, or during periods of high disease activity including the period of disease onset [141].
The immune response to viruses depends on the sophisticated balance and the coordinated function of several cell types and cytokines, encompassing both the innate and adaptive arms of the immune system. If the immune response is not successful in eliminating the virus, it continues to stimulate the inflammatory response with the potential to lead to the development of immunologically-mediated disorders [142]. As previously reported, patients with COVID-19 express higher levels of pro-inflammatory cytokines, considered the major contributors of lung damage. Moreover, this cytokine profile in COVID-19 patients is responsible for the hyperinflammatory syndrome with unremitting fever, cytopenia, hyperferritinemia, disseminated intravascular coagulation and multiorgan failure, including ARDS [143]. As mentioned, this syndrome is often associated with auto-inflammatory conditions and ADs, and can be triggered by infections [144]. The pathophysiology of MAS is still not completely elucidated, but a defect in lymphoid cell cytolytic activity has been hypothesized. A combination of genetic predisposition and a pro-inflammatory milieu, including the presence of IL-6 and IL-1β, may decrease cytolytic functions of NK cells and CD8+ T cells, with the consequent inability to lyse active antigen presenting cells or infected cells [75]. This results in an inflammatory cytokine storm leading to macrophage activation, hemophagocytosis and multi-organ failure [145]. It is likely that in genetically predisposed individuals, SARS-CoV-2 infection may trigger the occurrence of such a phenomenon. As mentioned, both a cytokine storm and a reduced cytolytic function of NK cells and CD8+ T cells have been reported in COVID-19 patients [72].
With regards to the role of specific cytokines, IL-1β and IL-6 are known to suppress regulatory T cells functions allowing unchecked adaptive autoimmune response [146,147]. These cytokines are also involved in the pathogenesis of several autoinflammatory and ADs, such as AOSD [148] and RA [149]. Moreover, IL-6 induces natural regulatory T cells to acquire characteristics of the Th17 cells in a TGF-β-dependent manner, with resultant enhanced pro-inflammatory activity [150]. These above-mentioned cytokines represent a Th1 response, notably pro-inflammatory, while cytokines such as IL-4 or IL-10 represent a Th2 response. Th2 cells initially have been described as anti-inflammatory, but a number of reports established a role for Th2 cells in tissue inflammation. Of note, several autoimmune conditions are Th2-driven (e.g., SLE) [151]. The promising results obtained by IL-6R blockers suggest that a Th1 response is predominant in the immunopathology of COVID-19 [130].
In summary, it is tempting to speculate that a pathophysiological model resembling AOSD might characterize COVID-19. In AOSD, an environmental trigger, possibly an infectious agent, leads to the induction of danger signals, activating a dysregulated Nod-like receptor 3 (NLRP3) inflammasome, which promotes release of IL-1β and IL-18 and Th1 polarization, with subsequent TNFα and IL-6 production. Contextually, the virus triggers activation of TLR7 and Th17 proliferation together with neutrophil recruitment [152]. TLR7 is mainly expressed by plasmacytoid DCs (pDCs), B cells, and to a lesser extent by macrophages. The TLR-7-MyD88 pathway is overexpressed in pDCs of AOSD compared to healthy individuals [153], and changes in the NLRP3 inflammasome could be involved in AOSD pathophysiology. However, no polymorphism in the NLRP3 gene have so far been identified, thus a reduced threshold of activation or a deregulation of the inflammasome have been hypothesized [154]. A similar condition may explain the devastating inflammatory response in the most severe COVID-19 patients. Moreover, IL-6 stimulates the liver synthesis of ferritin and IL-18 triggers NK cell-mediated IFNγ production, which promotes macrophage activation with possible onset of MAS (Fig. 3) [128,155].
8. Gender differences in COVID-19
Gender differences are recognized in many diseases and are generally manifest by changes in the clinical course, symptoms, therapy-response and clinical outcome. These differences are more striking for ADs [156]. It is well known that there is a very high gender bias towards females for systemic ADs such as SLE and SS (9:1 female/male ratio) while a few other immunological diseases, such as ankylosing spondylitis, are more prevalent in males [157].
Gender however not only influences the prevalence of these conditions but also the severity of symptoms and the degree of disability. Indeed, some studies describe SLE as a more severe disease in men compared to women [158], while women affected by axial spondylarthritis tend to have a greater disease activity and a lower quality of life compared to men [159]. The background for these gender-related differences is not completely elucidated but seems to be the result of a complex interaction between sex hormones, (epi-)genetics, and the composition of gut microbiota [160], all of which have immunological consequences. Overall, the female immune system has a higher reactivity with enhanced ability to produce antibodies, a stronger ability to mount a type I IFN response and also an increased antigen presenting activity by monocytes, while men are more susceptible to infections, with an increased inflammatory response to infectious pathogens [161]. Interestingly, estrogen was found to exert inhibitory effects on IL-6 production [162,163], suggesting it can directly hamper the COVID-19 related cytokine storm in females.
Some of these characteristics may be also responsible for the gender disparities reported for COVID-19 severe cases. In both China and Italy, the rate of infection among males and females was similar, but the death rate among males was much higher compared to females [164]. Indeed, a study from China reported that while men and women have the same prevalence for SARS-CoV-2 infection, men are more at risk for worse outcomes and death, independent of age [165]. The precise mechanism(s) that lead to such disparate outcomes remain to be defined although underlying health status and comorbidities (i.e., cardiac disease, obesity, among others) have been opined as contributory factors.
9. SARS-CoV-2 reinfection
The risk of reinfection is currently being analyzed in patients with a history of COVID-19. Animal models have been useful to clarify the risk of reinfection of different coronaviruses. A ferret model of reinfection was used. In acute SARS-CoV infection, the innate immune response was mainly led by the production of IFN. The symptomatic ferret previously reinfected or vaccinated lacked IFN antiviral response. This indicates that a likely modification in the initial response of IFN could influence future reinfections [166]. However, in a follow-up study in Rhesus macaques previously infected with SARS-CoV-2, no cases of reinfection were documented [167]. The presence of viral replication in the nasopharynx and anus was evaluated on different days, without finding viral activity. On the other hand, investigations in African green monkeys described the immune response associated with elimination of SARS-CoV and the persistence of lung inflammation, even after having eliminated the virus [168]. In this sense, after a period of reinfection, the viral clearance was faster; however, the lung inflammation lasted a few more days, so it is not ruled out that the persistence of a long-term “protective” humoral immune response may be associated with previous disease severity.
Although possible cases of reinfection in humans have been suggested, the presence of a second SARS-CoV-2 positive PCR does not necessarily mean viral activity and infectious capacity. It should be noted that PCR-based diagnosis does not reveal the presence of live and replicating viruses, merely the presence of viral RNA. Therefore, a positive PCR test should be evaluated within this context [169]. After the SARS-CoV infection disease, a longitudinal study was carried out in 176 patients previously infected, in order to evaluate the duration of antibodies. The authors showed that the levels of IgG antibodies were maintained for 2 years, and after 3 years after disease onset, these antibodies were drastically reduced [170]. Despite this, it is relevant to consider other factors associated with the durability of protective antibodies against infection, including age, and the presence of previous infections. Antibodies to coronaviruses are higher in older compared with younger adults and binding antibodies are more sensitive than neutralizing antibodies in identifying coronavirus‐associated illnesses [171].
The Korean Centers for Disease Control and Prevention carried out an epidemiological analysis of 285 positive cases and 790 contacts. The “re-positive cases” were renamed as “re-detected PCR” because neutralizing antibodies and negative viral cultures were documented [172]. Therefore, there is a low risk of reinfection. In addition, the immune memory response against SARS-CoV-2, caused by cross-reactivity upon previous exposure to other Human-CoV virus must be considered [171].
Detection of viral RNA after recovery does not necessarily indicate infectivity. The infectivity of the virus depends on the presence of the complete virus, not only on its RNA. Therefore, prolonged positive results may reflect only the lack of complete removal of nucleic acid from tissues [173]. Bullar et al. [174], showed that there was no growth in the samples with cycle threshold value (Ct) > 24 or symptom onset to analyze > 8 days. Therefore, the Ct indicated by the RT-qPCR test should be used as a reliable tool, in terms of predicting infectivity, and to define the maximum transmission risk period. These findings require further tests of cell infectivity in culture to demonstrate that RT-PCR positivity persists significantly beyond infectivity.
10. Autoimmunity in COVID-19
Potential mechanisms explaining the link between autoimmunity and COVID-19 include molecular mimicry and bystander activation [10,11]. The former occurs when similarities between foreign and self-peptides favor an activation of autoreactive T or B cells by foreign-derived peptides in a susceptible individual [10]. The SARS-CoV-2 proteome was described as sharing three sequences of six amino acids (GSQASS, LNEVAK, and SAAEAS) with three proteins, namely DAB1, AIFM, and SURF1 which are present in the brainstem respiratory pacemaker, also known as the pre-Bötzinger complex [175]. The authors suggested that these partial but not complete similarities might account for an autoimmune mediated respiratory central depression. Kanduc et al. [176], found that SARS-CoV-2 shared pentapeptides with pulmonary surfactant and related proteins that may account for autoimmune directed pulmonary damage. Moreover, a recent report has shown that SARS-CoV-2 spike protein antibody and tissue proteins such as transglutaminase 3, transglutaminase 2, ENA, myelin basic protein, mitochondria, nuclear antigen, α-myosin, thyroid peroxidase, collagen, claudin 5 + 6, and S100B have strong immune cross-reactions [177]. This may suggest that autoimmunity via molecular mimicry in susceptible individuals is likely and could explain some autoimmune-like manifestations encountered in COVID-19 patients.
10.1. Antiphospholipid syndrome and autoimmune cytopenia
Throughout the SARS-CoV-2 pandemic, autoimmune phenomena, mainly hematological ADs, have been documented (Table 2 ) [[19], [20], [21], [22], [23], [24], [25], [26], [27], [28], [29], [30], [31], [32], [33], [34], [35],[37], [38], [39],[41], [42], [43], [44], [45], [46]]. Among the most relevant is the case of a 65-year-old woman with a history of autoimmune hypothyroidism (i.e., Hashimoto's thyroiditis), who was diagnosed with COVID-19. On day 4 of admission, the patient presented purpuric lesions in the lower extremities, associated with epistaxis and, subsequently, subarachnoid hemorrhage with a platelet count of 16.000. Although no anti-platelet antibodies were documented, patients positively responded to IVIG [29]. In addition, a case series of 7 patients who developed autoimmune hemolytic anemia during the COVID-19 infection, specifically during the cytokine storm phase, was described [30]. The presence of a positive, direct anti-globulin test, anti-erythrocyte antibodies (warm antibodies in 4 cases), and cold agglutinins in 3 cases support the link between coronavirus infection and autoimmunity [30]. A case of simultaneous presentation of COVID-19 and autoimmune hemolytic anemia was reported. This disease should be considered in patients with COVID-19 and severe anemia [31]. Another case of immune thrombocytopenia was reported in a 41-year-old male patient with COVID-19, who debuted with nasal bleeding, and purpuric rash. Patient presented a mild COVID-19, as well as the thrombocytopenia. Interestingly, patient was managed with IVIG presenting an adequate clinical response [32].
Table 2.
Autoimmune diseases in COVID-19.
| COVID-19 associated autoimmune disease | Country | Number of patients | Gender | Comorbidities | Treatment for COVID-19 | Treatment for concurrent autoimmune diseases | References |
|---|---|---|---|---|---|---|---|
| Neurological syndromes | |||||||
| Guillain-Barré syndrome | Italy | 1 | Male | Hypertension, abdominal aortic aneurysm Lung cancer treated |
Oxygen via nonrebreather mask Lopinavir + Ritonavir Hydroxychloroquine |
IVIG | [19] |
| Guillain-Barré syndrome | Italy | 5 | Unknown | Unknown | Unknown | IVIG Plasma exchange |
[20] |
| Guillain-Barré syndrome | USA | 1 | Male | Unknown | Hydroxychloroquine | IVIG | [21] |
| Guillain-Barré syndrome | Switzerland | 1 | Male | Unknown | Unknown | IVIG | [22] |
| Guillain-Barré syndrome | Germany | 1 | Female | Unknown | Unknown | IVIG | [23] |
| Guillain-Barré syndrome | Spain | 1 | Male | Unknown | Lopinavir + Ritonavir Hydroxychloroquine |
Steroids | [24] |
| Guillain-Barré syndrome | France | 1 | Male | Diabetes mellitus type 2 | Cefotaxime + Azithromycin + Hydroxychloroquine | IVIG | [25] |
| Guillain-Barré syndrome | Spain | 2 | Male | Asthma (1) | Unknown | IVIG | [26] |
| Guillain-Barré syndrome | Italy | 1 | Female | Unknown | Lopinavir + Ritonavir Hydroxychloroquine |
IVIG | [27] |
| Ophthalmoparesis from cranial nerve palsy | USA | 2 | Female (1) Male (1) |
Hypertension | Hydroxychloroquine | IVIG | [28] |
| Hematological syndromes | |||||||
| Immune Thrombocytopenic Purpura | France | 1 | Female | Hypertension | Intravenous amoxicillin–clavulanic acid, low-molecular-weight heparin, and oxygen | IVIG | [29] |
| Immune Thrombocytopenic Purpura | Turkey | 1 | Male | Unknown | Favipiravir | IVIG | [32] |
| Immune Thrombocytopenic Purpura | United Kingdom | 3 | Male (1) Female (2) |
Atrial fibrillation, Ischemic heart disease Chronic kidney disease |
Unknown | IVIG tranexamic IVIG acid |
[33] |
| Immune Thrombocytopenic Purpura | Netherlands | 3 | Female (1) Male (2) |
Neuroendocrine tumor of the small bowel Hypertension Diabetes mellitus |
Unknown | Platelet transfusion IVIG Dexamethasone |
[34] |
| Immune Thrombocytopenic Purpura | China | 1 | Male | Unknown | Unknown | Interferon-α Umifenovir |
[35] |
| Autoimmune hemolytic anemia | France/Belgium | 7 | Female (3) Male (4) |
Hypertension (5) Chronic renal Failure (3) Atrial fibrillation (1) Cirrhosis (1) Obesity (2) Diabetes (3) Hypercholesterolemia (2) Chronic obstructive Bronchopneumopathy (1) Cardiopathy (1) |
Intervention: 19 | Steroids (5) Rituximab (2) RBC infusion (2) |
[30] |
| Autoimmune hemolytic anemia | USA | 1 | Female (1) | Unknown | Hydroxychloroquine | IVIG Steroids RBC infusion |
[31] |
| Antiphospholipid syndrome | China | 3 | Female (1) Male (2) |
Hypertension (3) Diabetes (1) Stroke (2) Coronary artery disease (1) Emphysema (1) Nasopharyngeal carcinoma (1) |
Oseltamivir Intravenous immunoglobulin Antibiotics Ribavirin, Rosuvastatin |
Anticoagulation | [43] |
| Latent Antiphospholipid syndrome | France | 25 | Unknown | Unknown | Unknown | Anticoagulation | [44] |
| Latent Antiphospholipid syndrome | United Kingdom | 31 | Female (7) Male (24) |
Unknown | Unknown | Anticoagulation | [45] |
| Latent Antiphospholipid syndrome | France | 150 | Female (28) Male (122) |
Unknown | Unknown | Anticoagulation | [46] |
| Vasculitis | |||||||
| Kawasaki Disease | United Kingdom | 8 | Female (3) Male (5) |
None | IVIG Antibiotics |
IVIG Hydroxicortisone Infliximab |
[38] |
| Kawasaki Disease | USA | 1 | Male | None | None | IVIG Aspirin |
|
| Kawasaki Disease | USA | 1 | Female | None | None | IVIG | [37] |
| Kawasaki Disease | France | 21 | Female (12) Male (9) |
Unknown | Antibiotics | Steroids IVIG Aspirin |
[42] |
| Kawasaki Disease | France | 35 | Female (17) Male (18) |
Asthma Lupus Overweight |
Anticoagulation Mechanical ventilation |
Steroids Inotropic support Interleukin 1 receptor antagonist IVIG |
[41] |
| Kawasaki Disease | Italy | 10 | Female (3) Male (7) |
None | IVIG | IVIG Aspirin Methylprednisolone |
[39] |
RBC: Red blood cells infusion; IVIG: Intravenous immunoglobulins; COVID-19: Coronavirus disease 2019.
A high risk of coagulopathy has been observed in patients with COVID -19. Interestingly, some tested positive for antiphospholipid antibodies indicating an APS-like condition. Zhang et al. [43], described 3 patients diagnosed with COVID-19 who had comorbidities and were critically ill. One of the patients had bilateral lower limb ischemia, ischemia in 2 fingers on the left hand, and multiple strokes. Thrombocytopenia and prolonged clotting times were documented with positive antiphospholipid antibodies (i.e., IgA and IgG anti-cardiolipin and anti-β2-glycoprotein antibodies). The other two patients presented a similar clinical course associated with antiphospholipid antibodies. Lupus anticoagulant was negative in all three patients [43].
Helms et al. [46], evaluated 150 patients managed at the ICU by ARDS associated with COVID-19. Up to sixty-four thrombotic events were observed, including pulmonary embolism, deep vein thrombosis, cerebral ischemic attack, mesenteric ischemia and others. In addition, the presence of D-dimer and fibrinogen were strongly elevated. On the other hand, coagulation factors such as von Willebrand factor, von Willebrand factor antigen and coagulation factor VIII were altered. Regarding the immunological profile, most of the patients had a positive lupus anticoagulant. When compared with Non-COVID-19 ARDS patients, the former COVID-19 ARDS patients presented a greater number of thrombotic events. It is highlighted that this high number of patients presented these thrombotic events despite anticoagulant management. These phenomena could be secondary to high levels of fibrinogen, associated with inflammation. However, an autoimmune origin associated with antiphospholipid antibodies deserves to be considered.
Moreover, Harzallah et al. [44], documented 56 patients diagnosed with COVID-19, of whom 25 were positive for lupus anticoagulant and 5 for anti-cardiolipin or anti-β2-glycoprotein. Along the same line, Bowles et al. [45], described the presence of lupus anticoagulant in 31 patients, of whom 2 developed thrombotic events. Although such antibodies could be present in acute infections, their association with thrombotic events is rare. Thus, the role of these autoantibodies in thrombo-embolic manifestations during severe SARS-CoV-2 infections should not be underestimated. As has been made evident in patients with catastrophic APS, autoantibody positivity is not strictly necessary for diagnosis since it may be present in clots from patients with ongoing thrombosis [178]. Therefore, it is plausible to hypothesize that some critically ill patients with thrombotic events could be negative for these autoantibodies. Therefore, antiphospholipid antibodies, including lupus anticoagulant, should be routinely evaluated in patients with COVID-19 as a risk marker of thrombotic events.
Raucci et al. [179], have shown the potential association of IL-17A in patients with COVID-19 that manifested thrombotic and vascular events. A recent classification for dermatological manifestations included livedo reticularis and necrosis as the fifth most common dermatological expression in COVID-19, presenting in about 6% of the patients, supporting the notion of an APS-like phenotype in these subjects [180].
10.2. Kawasaki disease
KD is an acute, self-limiting vasculitis, which mainly affects boys <5 years. The main symptoms include, fever, conjunctivitis, erythema in oral mucosa, cervical lymphadenopathy, and polymorphic rash (Fig. 4 ) [181]. KD may also involve cardiovascular, gastrointestinal, pulmonary, neurological, genitourinary and musculoskeletal systems [182]. An early innate immune response has been described, associated with a high number of neutrophils, and release of TNF, IL-1, and IL-6 [183]. In the first week, after the onset of fever, the presence of regulatory T cells and pro-inflammatory CD4+ T cells, and CD8+ T cells have been observed [184].
Fig. 4.
Clinical differences between classic KD (left) and KD associated with SARS-CoV-2 (right). Cardiac involvement is a critical clinical feature in patients with KD or Kawasaki-like conditions associated with SARS-CoV-2, also referred as pediatric inflammatory syndrome temporally related to COVID-19. Adapted from Refs. [[36], [37], [38], [39]].
Although the etiology of the disease is not clear, the main trigger seems to be infectious [185]. There has been a notable increase in the number of KD cases during the pandemic [38,39], suggesting an association between SARS-CoV-2 and this condition. To date, children continue to be the most affected. Additionally, main cases of incomplete KD have been described, together with a high rate of cardiac complications (Fig. 4) [[36], [37], [38], [39], [40], [41], [42]]. As reviewed by Son [186], “this condition is referred to variously as the pediatric multisystem inflammatory syndrome temporally associated with covid-19 (PIMS), the multisystem inflammatory syndrome in children and adolescents temporally related to COVID-19, and the multisystem inflammatory syndrome in children (MIS-C) associated with COVID-19”.
Riphagen et al. [38], reported an increase in the number of cases of children with hyperinflammatory shock with clinical characteristics resembling KD. They were 8 previously healthy children with fever, peripheral edema, gastrointestinal symptoms, conjunctivitis, progressing to vasoplegic refractory shock, requiring hemodynamic support. The presence of polyserositis and elevation of acute phase reactants was also described. In relation to cardiac compromise, an elevation of cardiac enzymes was found, and in one patient, echocardiographic changes as echo-bright coronary arteries on day 1 and then coronary aneurysm at day 7 were described. Two of the children were positive for SARS-CoV-2 (1 post-mortem), and four had an epidemiological link with relatives diagnosed with COVID-19.
Belhadjer et al. [41], described 35 children with acute heart failure and SARS-CoV-2. Of these, several patients showed clinical signs suggestive of atypical KD. On the other hand, Toubiana et al. [42], analyzed 21 cases of children with COVI-19 associated with KD. Half of patients exhibited cardiac involvement due to pericardial effusion, myocarditis, and cardiac arrhythmias, which reinforces the high number of patients with cardiac compromise.
A separate study in Italy, showed a 30-fold increased incidence of KD. Children exhibit a higher rate of cardiac involvement and features of MAS than those patients with similar manifestations prior to the beginning of this pandemic [39]. Jones et al. [37], described a previously healthy 6-month-old patient in whom the medical examination documented polymorphous rash, conjunctivitis, and changes in the oral mucosa after two days of fever together with a positive RT-PCR test for SARS-CoV-2. Additionally, Rivera-Figueroa et al. [40], reported the case of a 5-year-old boy with 8 days of fever, presenting with erythematous lips, non-exudative conjunctivitis, bilateral cervical lymphadenopathy, diarrhea, abdominal pain and dysuria were observed. The increase rate of KD in times of pandemic is striking. Beside the relationship between SARS-CoV-2 infection and CSS mentioned above, the mechanisms and risk factors by which KD in children with COVID-19 is increasing remains to be elucidated.
10.3. Guillain-Barré syndrome
GBS is an acute inflammatory immune-mediated polyradiculoneuropathy presenting typically with tingling, progressive weakness, autonomic dysfunction and pain. Immune injury specifically takes place at the myelin sheath and related Schwann-cell components in acute inflammatory demyelinating polyneuropathy, whereas in acute motor axonal neuropathy, the membranes on the nerve axon (the axolemma) are the primary target for immune-related injury [187].
The association between SARS-CoV-2 and autoimmune neurological diseases such as GBS has become relevant (Table 2). Alberti et al. [19], reported the case of a 71-year-old male patient who was admitted to the hospital secondary to paresthesia, progressive weakness, areflexia, and protein elevation in cerebrospinal fluid, fulfilling criteria for GBS diagnosis. Weeks before the diagnosis, the patient had experienced fever, and during hospitalization, the patient presented with dyspnea and severe hypoxia. Chest tomography showed multiple bilateral ground glass opacities and consolidations, all typical of COVID-19 pneumonia. However, confirmation of SARS-CoV-2 was not done.
Five additional cases of GBS following SARS-CoV-2 infection were reported by Toscano et al. [20]. The patients showed neurological symptoms between 5 and 10 days after onset of the viral disease. Four of them were successfully treated with IVIG and one received plasma exchange. However, a woman with COVID-19 developed a severe form of GBS that did not respond to IVIG [27]. This may suggest that select susceptible patients infected with SARS-CoV-2 may develop peripheral neurological diseases as reported for other viruses such as Zika, Dengue, or Cytomegalovirus [187]. At present, the GBS variant mainly associated with SARS-CoV-2, is the demyelinating one. The case reports presented in Table 2 indicate the possible causal relationship between SARS-CoV-2 infection and GBS through an autoimmune cross-reactivity mechanism.
Dinkin et al. [28], documented 2 patients with COVID-19 and ocular motor palsy. Miller Fisher syndrome and oculomotor nerve inflammation were suspected in one patient, while the second patient exhibited a sixth cranial nerve compromise. These cases support the association between COVID-19 and the appearance of inflammatory neuropathies similar to GBS. It has been observed that patients with lymphopenia, hyposmia and hypogeusia have a higher risk of developing these neurological manifestations [28]. Therefore, an autoimmune phenomenon triggered by COVID-19 should not be ruled out. Inflammation may compromise the integrity of the blood-brain barrier, and this facilitates the affectation of nerve structures [188]. On the other hand, macrophages expressing ACE2 receptors can prolong inflammation in the nervous tissue [189].
11. Concluding remarks
COVID-19 is a new disease that rapidly became a dominant global health issue in which scientific interdisciplinary and collaborative work has become more important than ever. Viruses are known to trigger several autoinflammatory and ADs, and many immunological abnormalities described so far in COVID-19 patients can be observed in auto-inflammatory/autoimmune conditions. Our current understanding of the COVID-19 pathogenic mechanisms is very limited. However, it is evident that the disease may evolve in four overlapping phases. An initial viral phase that may well be asymptomatic or mild in about 80% of patients. It is not known why such a large proportion of patients have mild or asymptomatic disease. Then host-virus interactions take place which dictate the outcome for the subsequent phases of the disease. The second phase corresponds to a hyperresponsiveness of the immune system. The third phase is characterized by a state of hypercoagulability. Lastly, in the fourth phase organ damage occurs and its related to the organ- and cell-specific expression of ACE2 receptor, the intensity of the inflammatory response (i.e., CSS) and the hypercoagulable state. Given this characterization, it is worth asking “is COVID-19 just an infectious disease or something else?” [190] or, are autoinflammatory and autoimmune conditions at the crossroad of COVID-19? It also raises the issue of whether other ADs may be caused by a coronavirus.
To date, there are no established evidence-based therapies for this new disease; however, the number of reports showing beneficial effects by immunomodulatory drugs is increasing. Some of these drugs possess both anti-viral and immunomodulatory effects [191]. Monoclonal antibodies (e.g., tocilizumab targeting IL-6R and anakinra targeting IL-1β) are currently studied in the treatment of COVID-19. Similarly, colchicine, an “old drug” used in auto-inflammatory disorders, is also under evaluation, suggesting that modulation and control of inflammation is crucial, and that there is a point during the course of the disease when immunosuppressive therapy maybe a prudent course for the therapy of this new pathological condition [192]. Clearly, we are not fully informed and therapeutic strategies remain in their infancy [193].
Current studies on SARS-CoV-2 vaccination should consider the topics we have raised here. As discussed above, bystander activation and molecular mimicry could be associated with the development of autoimmunity while antibody-dependent enhancement may be associated with severity of disease. In addition, personalized approaches including genotypification of risk genes and evaluation of risk factors for autoimmunity (i.e., familial autoimmunity) must be also considered in the current development of vaccines [194].
Funding
This work was supported by Universidad del Rosario (ABN-011).
Availability of data and materials
Not applicable.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Declaration of competing interest
Authors declare no competing interests.
Acknowledgements
Authors would like to thank colleagues from “PC-Covid-19 Group” for fruitful discussions, and Manuel Franco and Andres Cardona for comments on Fig. 1.
References
- 1.Zhu N., Zhang D., Wang W., Li X., Yang B., Song J., Zhao X., Huang B., Shi W., Lu R., Niu P., Zhan F., Ma X., Wang D., Xu W., Wu G., Gao G.F., Tan W. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.WHO . WHO; 2020. Coronavirus Disease 2019 (COVID-19)situation Report – 52. [Google Scholar]
- 3.Ruan Q., Yang K., Wang W., Jiang L., Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46:846–848. doi: 10.1007/s00134-020-05991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zuo Y., Yalavarthi S., Shi H., Gockman K., Zuo M., Madison J.A., Blair C., Weber A., Barnes B.J., Egeblad M., Woods R.J., Kanthi Y., Knight J.S. Neutrophil extracellular traps (NETs) as markers of disease severity in COVID-19. MedRxiv. 2020 doi: 10.1101/2020.04.09.20059626. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sethuraman N., Jeremiah S.S., Ryo A. Interpreting diagnostic tests for SARS-CoV-2. J. Am. Med. Assoc. 2020;323(22):2249–2251. doi: 10.1001/jama.2020.8259. In press. [DOI] [PubMed] [Google Scholar]
- 6.Tay M.Z., Poh C.M., Rénia L., MacAry P.A., Ng L.F.P. The trinity of COVID-19: immunity, inflammation and intervention. Nat. Rev. Immunol. 2020;20:363–374. doi: 10.1038/s41577-020-0311-8. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Long Q.-X., Liu B.-Z., Deng H.-J., Wu G.-C., Deng K., Chen Y.-K., Liao P., Qiu J.-F., Lin Y., Cai X.-F., Wang D.-Q., Hu Y., Ren J.-H., Tang N., Xu Y.-Y., Yu L.-H., Mo Z., Gong F., Zhang X.-L., Tian W.-G., Hu L., Zhang X.-X., Xiang J.-L., Du H.-X., Liu H.-W., Lang C.-H., Luo X.-H., Wu S.-B., Cui X.-P., Zhou Z., Zhu M.-M., Wang J., Xue C.-J., Li X.-F., Wang L., Li Z.-J., Wang K., Niu C.-C., Yang Q.-J., Tang X.-J., Zhang Y., Liu X.-M., Li J.-J., Zhang D.-C., Zhang F., Liu P., Yuan J., Li Q., Hu J.-L., Chen J., Huang A.-L. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat. Med. 2020 doi: 10.1038/s41591-020-0897-1. In press. [DOI] [PubMed] [Google Scholar]
- 8.Du S.Q., Yuan W. Mathematical modeling of interaction between innate and adaptive immune responses in COVID‐19 and implications for viral pathogenesis. J. Med. Virol. 2020 doi: 10.1002/jmv.25866. In press jmv.25866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Smatti M.K., Cyprian F.S., Nasrallah G.K., Al Thani A.A., Almishal R.O., Yassine H.M. Viruses and autoimmunity: a review on the potential interaction and molecular mechanisms. Viruses. 2019;11:762. doi: 10.3390/v11080762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rojas M., Restrepo-Jiménez P., Monsalve D.M., Pacheco Y., Acosta-Ampudia Y., Ramírez-Santana C., Leung P.S.C., Ansari A.A., Gershwin M.E., Anaya J.-M. Molecular mimicry and autoimmunity. J. Autoimmun. 2018;95:100–123. doi: 10.1016/j.jaut.2018.10.012. [DOI] [PubMed] [Google Scholar]
- 11.Pacheco Y., Acosta-Ampudia Y., Monsalve D.M., Chang C., Gershwin M.E., Anaya J.-M. Bystander activation and autoimmunity. J. Autoimmun. 2019;103:102301. doi: 10.1016/j.jaut.2019.06.012. [DOI] [PubMed] [Google Scholar]
- 12.Filippi C.M., von Herrath M.G. Viral trigger for type 1 diabetes: pros and cons. Diabetes. 2008;57:2863–2871. doi: 10.2337/db07-1023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cacoub P., Terrier B., Saadoun D. Hepatitis C virus-induced vasculitis: therapeutic options. Ann. Rheum. Dis. 2014;73:24–30. doi: 10.1136/annrheumdis-2013-203883. [DOI] [PubMed] [Google Scholar]
- 14.Ramos-Casals M., Loustaud-Ratti V., De Vita S., Zeher M., Bosch J.-A., Toussirot E., Medina F., Rosas J., Anaya J.-M., Font J. Sjögren syndrome associated with hepatitis C virus: a multicenter analysis of 137 cases. Medicine (Baltim.) 2005;84:81–89. doi: 10.1097/01.md.0000157397.30055.c9. [DOI] [PubMed] [Google Scholar]
- 15.Sanderson N.S.R., Zimmermann M., Eilinger L., Gubser C., Schaeren-Wiemers N., Lindberg R.L.P., Dougan S.K., Ploegh H.L., Kappos L., Derfuss T. Cocapture of cognate and bystander antigens can activate autoreactive B cells. Proc. Natl. Acad. Sci. Unit. States Am. 2017;114:734–739. doi: 10.1073/pnas.1614472114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chen J., Zhang H., Chen P., Lin Q., Zhu X., Zhang L., Xue X. Correlation between systemic lupus erythematosus and cytomegalovirus infection detected by different methods. Clin. Rheumatol. 2015;34:691–698. doi: 10.1007/s10067-015-2868-3. [DOI] [PubMed] [Google Scholar]
- 17.Dostál C., Newkirk M.M., Duffy K.N.W., Palečková A., Bošák V., Černá M., Žd’árský E., Zvárová J. Herpes viruses in multicase families with rheumatoid arthritis and systemic lupus erythematosus. Ann. N. Y. Acad. Sci. 1997;815:334–337. doi: 10.1111/j.1749-6632.1997.tb52078.x. [DOI] [PubMed] [Google Scholar]
- 18.Wouters J.M.G.W., van der Veen J., van de Putte L.B., de Rooij D.J. Adult onset Still's disease and viral infections. Ann. Rheum. Dis. 1988;47:764–767. doi: 10.1136/ard.47.9.764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Alberti P., Beretta S., Piatti M., Karantzoulis A., Piatti M.L., Santoro P., Viganò M., Giovannelli G., Pirro F., Montisano D.A., Appollonio I., Ferrarese C. Guillain-Barré syndrome related to COVID-19 infection. Neurol. Neuroimmunol. Neuroinflammation. 2020;7:e741. doi: 10.1212/NXI.0000000000000741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Toscano G., Palmerini F., Ravaglia S., Ruiz L., Invernizzi P., Cuzzoni M.G., Franciotta D., Baldanti F., Daturi R., Postorino P., Cavallini A., Micieli G. Guillain–barré syndrome associated with SARS-CoV-2. N. Engl. J. Med. 2020 doi: 10.1056/NEJMc2009191. In pressNEJMc2009191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Virani A., Rabold E., Hanson T., Haag A., Elrufay R., Cheema T., Balaan M., Bhanot N. Guillain-Barré Syndrome associated with SARS-CoV-2 infection. IDCases. 2020;20 doi: 10.1016/j.idcr.2020.e00771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Coen M., Jeanson G., Culebras Almeida L.A., Hübers A., Stierlin F., Najjar I., Ongaro M., Moulin K., Makrygianni M., Leemann B., Kronig I., Bertrand J., Reny J.-L., Schibler M., Serratrice J. Guillain-Barré syndrome as a complication of SARS-CoV-2 infection. Brain Behav. Immun. 2020 doi: 10.1016/j.bbi.2020.04.074. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Scheidl E., Canseco D.D., Hadji‐Naumov A., Bereznai B. Guillain‐Barre syndrome during SARS‐CoV ‐2 pandemic: a case report and review of recent literature. J. Peripher. Nerv. Syst. 2020 doi: 10.1111/jns.12382. In pressjns.12382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Juliao Caamaño D.S., Alonso Beato R. Facial diplegia, a possible atypical variant of Guillain-Barré Syndrome as a rare neurological complication of SARS-CoV-2. J. Clin. Neurosci. 2020 doi: 10.1016/j.jocn.2020.05.016. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Arnaud S., Budowski C., Ng Wing Tin S., Degos B. Post SARS-CoV-2 guillain-barré syndrome. Clin. Neurophysiol. 2020;131:1652–1654. doi: 10.1016/j.clinph.2020.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gutiérrez-Ortiz C., Méndez A., Rodrigo-Rey S., San Pedro-Murillo E., Bermejo-Guerrero L., Gordo-Mañas R., de Aragón-Gómez F., Benito-León J. Miller Fisher Syndrome and polyneuritis cranialis in COVID-19. Neurology. 2020 doi: 10.1212/WNL.0000000000009619. In press. [DOI] [PubMed] [Google Scholar]
- 27.Ottaviani D., Boso F., Tranquillini E., Gapeni I., Pedrotti G., Cozzio S., Guarrera G.M., Giometto B. Early Guillain-Barré syndrome in coronavirus disease 2019 (COVID-19): a case report from an Italian COVID-hospital. Neurol. Sci. 2020;41(6):1351–1354. doi: 10.1007/s10072-020-04449-8. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dinkin M., Gao V., Kahan J., Bobker S., Simonetto M., Wechsler P., Harpe J., Greer C., Mints G., Salama G., Tsiouris A.J., Leifer D. COVID-19 presenting with ophthalmoparesis from cranial nerve palsy. Neurology. 2020 doi: 10.1212/WNL.0000000000009700. [DOI] [PubMed] [Google Scholar]
- 29.Zulfiqar A.-A., Lorenzo-Villalba N., Hassler P., Andrès E. Immune thrombocytopenic purpura in a patient with covid-19. N. Engl. J. Med. 2020;382:e43. doi: 10.1056/NEJMc2010472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lazarian G., Quinquenel A., Bellal M., Siavellis J., Jacquy C., Re D., Merabet F., Mekinian A., Braun T., Damaj G., Delmer A., Cymbalista F. Autoimmune hemolytic anemia associated with Covid‐19 infection. Br. J. Haematol. 2020 doi: 10.1111/bjh.16794. In pressbjh.16794 In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lopez C., Kim J., Pandey A., Huang T., DeLoughery T.G. Simultaneous onset of COVID‐19 and autoimmune haemolytic anaemia. Br. J. Haematol. 2020 doi: 10.1111/bjh.16786. In pressbjh.16786 In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Murt A., Eskazan A.E., Yılmaz U., Ozkan T., Ar M.C. COVID‐19 presenting with immune thrombocytopenia: a case report and review of the literature. J. Med. Virol. 2020:26138. doi: 10.1002/jmv.26138. jmv. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ahmed M.Z., Khakwani M., Venkatadasari I., Horgan C., Giles H., Jobanputra S., Lokare A., Ewing J., Paneesha S., Murthy V. Thrombocytopenia as an initial manifestation of COVID‐19; case series and literature review. Br. J. Haematol. 2020:16769. doi: 10.1111/bjh.16769. bjh. [DOI] [PubMed] [Google Scholar]
- 34.Bomhof G., Mutsaers P.G.N.J., Leebeek F.W.G., Te Boekhorst P.A.W., Hofland J., Croles F.N., Jansen A.J.G. COVID-19-associated immune thrombocytopenia. Br. J. Haematol. 2020 doi: 10.1111/bjh.16850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chen W., Yang B., Li Z., Wang P., Chen Y., Zhou H. Sudden severe thrombocytopenia in a patient in the recovery stage of COVID-19. Lancet Haematol. 2020 doi: 10.1016/S2352-3026(20)30175-7. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nathan N., Prevost B., Corvol H. Atypical presentation of COVID-19 in young infants. Lancet. 2020;395:1481. doi: 10.1016/S0140-6736(20)30980-6. In this issue. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jones V.G., Mills M., Suarez D., Hogan C.A., Yeh D., Bradley Segal J., Nguyen E.L., Barsh G.R., Maskatia S., Mathew R. COVID-19 and Kawasaki disease: novel virus and novel case. Hosp. Pediatr. 2020 doi: 10.1542/hpeds.2020-0123. In press. [DOI] [PubMed] [Google Scholar]
- 38.Riphagen S., Gomez X., Gonzalez-Martinez C., Wilkinson N., Theocharis P. Hyperinflammatory shock in children during COVID-19 pandemic. Lancet. 2020;395:1607–1608. doi: 10.1016/S0140-6736(20)31094-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Verdoni L., Mazza A., Gervasoni A., Martelli L., Ruggeri M., Ciuffreda M., Bonanomi E., D’Antiga L. An outbreak of severe Kawasaki-like disease at the Italian epicentre of the SARS-CoV-2 epidemic: an observational cohort study. Lancet. 2020;395(10239):1771–1778. doi: 10.1016/S0140-6736(20)31103-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rivera-Figueroa E.I., Santos R., Simpson S., Garg P. Indian Pediatr; 2020. Incomplete Kawasaki Disease in a Child with Covid-19. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Belhadjer Z., Méot M., Bajolle F., Khraiche D., Legendre A., Abakka S., Auriau J., Grimaud M., Oualha M., Beghetti M., Wacker J., Ovaert C., Hascoet S., Selegny M., Malekzadeh-Milani S., Maltret A., Bosser G., Giroux N., Bonnemains L., Bordet J., Di Filippo S., Mauran P., Falcon-Eicher S., Thambo J.-B., Lefort B., Moceri P., Houyel L., Renolleau S., Bonnet D. Acute heart failure in multisystem inflammatory syndrome in children (MIS-C) in the context of global SARS-CoV-2 pandemic. Circulation. 2020 doi: 10.1161/CIRCULATIONAHA.120.048360. CIRCULATIONAHA.120.048360. [DOI] [PubMed] [Google Scholar]
- 42.Toubiana J., Poirault C., Corsia A., Bajolle F., Fourgeaud J., Angoulvant F., Debray A., Basmaci R., Salvador E., Biscardi S., Frange P., Chalumeau M., Casanova J.-L., Cohen J.F., Allali S. Kawasaki-like multisystem inflammatory syndrome in children during the covid-19 pandemic in Paris, France: prospective observational study. BMJ. 2020;369 doi: 10.1136/bmj.m2094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhang Y., Xiao M., Zhang S., Xia P., Cao W., Jiang W., Chen H., Ding X., Zhao H., Zhang H., Wang C., Zhao J., Sun X., Tian R., Wu W., Wu D., Ma J., Chen Y., Zhang D., Xie J., Yan X., Zhou X., Liu Z., Wang J., Du B., Qin Y., Gao P., Qin X., Xu Y., Zhang W., Li T., Zhang F., Zhao Y., Li Y., Zhang S. Coagulopathy and antiphospholipid antibodies in patients with covid-19. N. Engl. J. Med. 2020;382:e38. doi: 10.1056/NEJMc2007575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Harzallah I., Debliquis A., Drénou B. Lupus anticoagulant is frequent in patients with Covid‐19. J. Thromb. Haemostasis. 2020 doi: 10.1111/jth.14867. In pressjth.14867 In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bowles L., Platton S., Yartey N., Dave M., Lee K., Hart D.P., MacDonald V., Green L., Sivapalaratnam S., Pasi K.J., MacCallum P. Lupus anticoagulant and abnormal coagulation tests in patients with covid-19. N. Engl. J. Med. 2020 doi: 10.1056/NEJMc2013656. In pressNEJMc2013656 In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Helms J., Tacquard C., Severac F., Leonard-Lorant I., Ohana M., Delabranche X., Merdji H., Clere-Jehl R., Schenck M., Fagot Gandet F., Fafi-Kremer S., Castelain V., Schneider F., Grunebaum L., Anglés-Cano E., Sattler L., Mertes P.-M., Meziani F. High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intensive Care Med. 2020;46(6):1089–1098. doi: 10.1007/s00134-020-06062-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Luo P., Liu Y., Qiu L., Liu X., Liu D., Li J. Tocilizumab treatment in COVID-19: a single center experience. J. Med. Virol. 2020 doi: 10.1002/jmv.25801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ahn D.-G., Shin H.-J., Kim M.-H., Lee S., Kim H.-S., Myoung J., Kim B.-T., Kim S.-J. Current status of epidemiology, diagnosis, therapeutics, and vaccines for novel coronavirus disease 2019 (COVID-19) J. Microbiol. Biotechnol. 2020;30:313–324. doi: 10.4014/jmb.2003.03011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cantini F., Niccoli L., Matarrese D., Nicastri E., Stobbione P., Goletti D. Baricitinib therapy in COVID-19: a pilot study on safety and clinical impact. J. Infect. 2020 doi: 10.1016/j.jinf.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.McGonagle D., McDermott M.F. A proposed classification of the immunological diseases. PLoS Med. 2006;3:e297. doi: 10.1371/journal.pmed.0030297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Doria A., Zen M., Bettio S., Gatto M., Bassi N., Nalotto L., Ghirardello A., Iaccarino L., Punzi L. Autoinflammation and autoimmunity: bridging the divide. Autoimmun. Rev. 2012;12:22–30. doi: 10.1016/j.autrev.2012.07.018. [DOI] [PubMed] [Google Scholar]
- 52.Generali E., Bose T., Selmi C., Voncken J.W., Damoiseaux J.G.M.C. Nature versus nurture in the spectrum of rheumatic diseases: classification of spondyloarthritis as autoimmune or autoinflammatory. Autoimmun. Rev. 2018;17:935–941. doi: 10.1016/j.autrev.2018.04.002. [DOI] [PubMed] [Google Scholar]
- 53.Caso F., Costa L., Nucera V., Barilaro G., Masala I.F., Talotta R., Caso P., Scarpa R., Sarzi-Puttini P., Atzeni F. From autoinflammation to autoimmunity: old and recent findings. Clin. Rheumatol. 2018;37:2305–2321. doi: 10.1007/s10067-018-4209-9. [DOI] [PubMed] [Google Scholar]
- 54.Ghiringhelli F., Apetoh L., Tesniere A., Aymeric L., Ma Y., Ortiz C., Vermaelen K., Panaretakis T., Mignot G., Ullrich E., Perfettini J.-L., Schlemmer F., Tasdemir E., Uhl M., Génin P., Civas A., Ryffel B., Kanellopoulos J., Tschopp J., André F., Lidereau R., McLaughlin N.M., Haynes N.M., Smyth M.J., Kroemer G., Zitvogel L. Activation of the NLRP3 inflammasome in dendritic cells induces IL-1β–dependent adaptive immunity against tumors. Nat. Med. 2009;15:1170–1178. doi: 10.1038/nm.2028. [DOI] [PubMed] [Google Scholar]
- 55.Kahlenberg J.M., Thacker S.G., Berthier C.C., Cohen C.D., Kretzler M., Kaplan M.J. Inflammasome activation of IL-18 results in endothelial progenitor cell dysfunction in systemic lupus erythematosus. J. Immunol. 2011;187:6143–6156. doi: 10.4049/jimmunol.1101284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Rawlings D.J., Schwartz M.A., Jackson S.W., Meyer-Bahlburg A. Integration of B cell responses through Toll-like receptors and antigen receptors. Nat. Rev. Immunol. 2012;12:282–294. doi: 10.1038/nri3190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sbidian E., Madrange M., Viguier M., Salmona M., Duchatelet S., Hovnanian A., Smahi A., Le Goff J., Bachelez H. Respiratory virus infection triggers acute psoriasis flares across different clinical subtypes and genetic backgrounds. Br. J. Dermatol. 2019;181:1304–1306. doi: 10.1111/bjd.18203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Anaya J.-M., Shoenfeld Y., Rojas-Villarraga A., Levy R.A., Cervera R., editors. Autoimmunity: from Bench to Bedside. El Rosario University Press; Bogota (Colombia): 2013. [PubMed] [Google Scholar]
- 59.Wang W., Zhang W., Zhang J., He J., Zhu F. Distribution of HLA allele frequencies in 82 Chinese individuals with coronavirus disease-2019 (COVID-19) HLA. 2020 doi: 10.1111/tan.13941. In presstan.13941 In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ellinghaus D., Degenhardt F., Bujanda L., Buti M., Albillos A., Invernizzi P., Fernandez J., Prati D., Baselli G., Asselta R., Grimsrud M.M., Milani C., Aziz F., Kassens J., May S., Wendorff M., Wienbrandt L., Uellendahl-Werth F., Zheng T., Yi X., de Pablo R., Chercoles A.G., Palom A., Garcia-Fernandez A.-E., Rodriguez-Frias F., Zanella A., Bandera A., Protti A., Aghemo A., de Nalda A., Biondi A., Caballero-Garralda A., Gori A., Tanck A., Latiano A., Fracanzani A.L., Peschuck A., Julia A., Pesenti A., Voza A., Jimenez D., Mateos B., Jimenez B.N., Quereda C., Angelini C., Cea C., Solier A., Pestana D., Sandoval E., Paraboschi E.M., Navas E., Ceriotti F., Martinelli-Boneschi F., Peyvandi F., Blasi F., Tellez L., Blanco-Grau A., Grasselli G., Costantino G., Cardamone G., Foti G., Aneli S., Kurihara H., ElAbd H., My I., Martin J., Erdmann J., Ferrusquia-Acosta J., Garcia-Etxebarria K., Izquierdo-Sanchez L., Bettini L.R., Terranova L., Moreira L., Santoro L., Scudeller L., Mesonero F., Roade L., Schaefer M., Carrabba M., Riveiro Barciela M. del M., Basso M.E.F., Valsecchi M.G., Hernandez-Tejero M., Acosta-Herrera M., D\textquoterightAngio M., Baldini M., Cazzaniga M., Schulzky M., Cecconi M., Wittig M., Ciccarelli M., Rodriguez-Gandia M., Bocciolone M., Miozzo M., Braun N., Martinez N., Palmieri O., Faverio P., Preatoni P., Bonfanti P., Omodei P., Tentorio P., Castro P., Rodrigues P.M., Blandino Ortiz A., Roca R.F., Gualtierotti R., Nieto R., Badalamenti S., Marsal S., Matullo G., Pelusi S., Monzani V., Wesse T., Pumarola T., Rimoldi V., Bosari S., Albrecht W., Peter W., Gomez M.R., D\textquoterightAmato M., Duga S., Banales J.M., Hov J.R., Folseraas T., Valenti L., Franke A., Karlsen T.H. The ABO blood group locus and a chromosome 3 gene cluster associate with SARS-CoV-2 respiratory failure in an Italian-Spanish genome-wide association analysis. MedRxiv. 2020 doi: 10.1101/2020.05.31.20114991. [DOI] [Google Scholar]
- 61.Lokman S.M., Rasheduzzaman M.D., Salauddin A., Barua R., Tanzina A.Y., Rumi M.H., Hossain M.D.I., Siddiki A.M.A.M.Z., Mannan A., Hasan M.D.M. Exploring the genomic and proteomic variations of SARS-CoV-2 spike glycoprotein: a computational biology approach. Infect. Genet. Evol. 2020:104389. doi: 10.1016/j.meegid.2020.104389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Letko M., Marzi A., Munster V. Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat. Microbiol. 2020;5:562–569. doi: 10.1038/s41564-020-0688-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Meng-Yuan L., Lin L., Yue Z., Xiao-Sheng W. Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infect. Disease Poverty. 2020;9:45. doi: 10.1186/s40249-020-00662-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Puelles V.G., Lütgehetmann M., Lindenmeyer M.T., Sperhake J.P., Wong M.N., Allweiss L., Chilla S., Heinemann A., Wanner N., Liu S., Braun F., Lu S., Pfefferle S., Schröder A.S., Edler C., Gross O., Glatzel M., Wichmann D., Wiech T., Kluge S., Pueschel K., Aepfelbacher M., Huber T.B. Multiorgan and renal tropism of SARS-CoV-2. N. Engl. J. Med. 2020 doi: 10.1056/NEJMc2011400. In pressNEJMc2011400 In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Sungnak W., Huang N., Bécavin C., Berg M., Queen R., Litvinukova M., Talavera-López C., Maatz H., Reichart D., Sampaziotis F., Worlock K.B., Yoshida M., Barnes J.L. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nat. Med. 2020;26:681–687. doi: 10.1038/s41591-020-0868-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.-H., Nitsche A., Müller M.A., Drosten C., Pöhlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zang R., Gomez Castro M.F., McCune B.T., Zeng Q., Rothlauf P.W., Sonnek N.M., Liu Z., Brulois K.F., Wang X., Greenberg H.B., Diamond M.S., Ciorba M.A., Whelan S.P.J., Ding S. TMPRSS2 and TMPRSS4 promote SARS-CoV-2 infection of human small intestinal enterocytes. Sci. Immunol. 2020;5 doi: 10.1126/sciimmunol.abc3582. eabc3582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Du Y., Tu L., Zhu P., Mu M., Wang R., Yang P., Wang X., Hu C., Ping R., Hu P., Li T., Cao F., Chang C., Hu Q., Jin Y., Xu G. Clinical features of 85 fatal cases of COVID-19 from wuhan: a retrospective observational study. Am. J. Respir. Crit. Care Med. 2020;201(11):1372–1379. doi: 10.1164/rccm.202003-0543OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zhang J., Dong X., Cao Y., Yuan Y., Yang Y., Yan Y., Akdis C.A., Gao Y. 2020. Clinical Characteristics of 140 Patients Infected with SARS‐CoV‐2 in Wuhan. China, Allergy. In pressall.14238. [DOI] [PubMed] [Google Scholar]
- 70.Liu F., Xu A., Zhang Y., Xuan W., Yan T., Pan K., Yu W., Zhang J. Patients of COVID-19 may benefit from sustained Lopinavir-combined regimen and the increase of Eosinophil may predict the outcome of COVID-19 progression. Int. J. Infect. Dis. 2020;95:183–191. doi: 10.1016/j.ijid.2020.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Qin C., Zhou L., Hu Z., Zhang S., Yang S., Tao Y., Xie C., Ma K., Shang K., Wang W., Tian D.-S. Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in wuhan, China. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa248. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zheng M., Gao Y., Wang G., Song G., Liu S., Sun D., Xu Y., Tian Z. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell. Mol. Immunol. 2020;17:533–535. doi: 10.1038/s41423-020-0402-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zhou Z., Ren L., Zhang L., Zhong J., Xiao Y., Jia Z., Guo L., Yang J., Wang C., Jiang S., Yang D., Zhang G., Li H., Chen F., Xu Y., Chen M., Gao Z., Yang J., Dong J., Liu B., Zhang X., Wang W., He K., Jin Q., Li M., Wang J. Heightened innate immune responses in the respiratory tract of COVID-19 patients. Cell Host Microbe. 2020;27:883–890. doi: 10.1016/j.chom.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhou Y., Fu B., Zheng X., Wang D., Zhao C., Qi Y., Sun R., Tian Z., Xu X., Wei H. Pathogenic T cells and inflammatory monocytes incite inflammatory storm in severe COVID-19 patients. Natl. Sci. Rev. 2020 doi: 10.1093/nsr/nwaa041. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Mazzoni A., Salvati L., Maggi L., Capone M., Vanni A., Spinicci M., Mencarini J., Caporale R., Peruzzi B., Antonelli A., Trotta M., Zammarchi L., Ciani L., Gori L., Lazzeri C., Matucci A., Vultaggio A., Rossi O., Almerigogna F., Parronchi P., Fontanari P., Lavorini F., Peris A., Rossolini G.M., Bartoloni A., Romagnani S., Liotta F., Annunziato F., Cosmi L. Impaired immune cell cytotoxicity in severe COVID-19 is IL-6 dependent. J. Clin. Invest. 2020 doi: 10.1172/JCI138554. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Blanco-Melo D., Nilsson-Payant B.E., Liu W.-C., Uhl S., Hoagland D., Møller R., Jordan T.X., Oishi K., Panis M., Sachs D., Wang T.T., Schwartz R.E., Lim J.K., Albrecht R.A., TenOever B.R. Imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell. 2020;181(5):1036–1045.e9. doi: 10.1016/j.cell.2020.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Liu Y., Zhang C., Huang F., Yang Y., Wang F., Yuan J., Zhang Z., Qin Y., Li X., Zhao D., Li S., Tan S., Wang Z., Li J., Shen C., Li J., Peng L., Wu W., Cao M., Xing L., Xu Z., Chen L., Zhou C., Liu W.J., Liu L., Jiang C. Elevated plasma level of selective cytokines in COVID-19 patients reflect viral load and lung injury. Natl. Sci. Rev. 2020 doi: 10.1093/nsr/nwaa037. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Del Valle D.M., Kim-schulze S., Hsin-hui H., Beckmann N.D., Nirenberg S., Wang B., Lavin Y., Swartz T., Madduri D., Stock A., Marron T., Xie H., Patel M.K., van Oekelen O., Rahman A., Kovatch P., Aberg J., Schadt E., Jagannath S., Mazumdar M., Charney A., Firpo-Betancourt A., Mendu D.R., Jhang J., Reich D., Sigel K., Cordon-Cardo C., Feldmann M., Parekh S., Merad M., Gnjatic S. An inflammatory cytokine signature helps predict COVID-19 severity and death. MedRxiv. 2020 doi: 10.1101/2020.05.28.20115758. 2020.05.28.20115758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Diao B., Wang C., Tan Y., Chen X., Liu Y., Ning L., Chen L., Li M., Liu Y., Wang G., Yuan Z., Feng Z., Zhang Y., Wu Y., Chen Y. Reduction and functional exhaustion of T cells in patients with coronavirus disease 2019 (COVID-19) Front. Immunol. 2020;11:1–7. doi: 10.3389/fimmu.2020.00827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Liu J., Li S., Liu J., Liang B., Wang X., Wang H., Li W., Tong Q., Yi J., Zhao L., Xiong L., Guo C., Tian J., Luo J., Yao J., Pang R., Shen H., Peng C., Liu T., Zhang Q., Wu J., Xu L., Lu S., Wang B., Weng Z., Han C., Zhu H., Zhou R., Zhou H., Chen X., Ye P., Zhu B., Wang L., Zhou W., He S., He Y., Jie S., Wei P., Zhang J., Lu Y., Wang W., Zhang L., Li L., Zhou F., Wang J., Dittmer U., Lu M., Hu Y., Yang D., Zheng X. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55:102763. doi: 10.1016/j.ebiom.2020.102763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Shi Y., Tan M., Chen X., Liu Y., Huang J., Ou J., Deng X. Immunopathological characteristics of coronavirus disease 2019 cases in Guangzhou, China. MedRxiv. 2020 doi: 10.1101/2020.03.12.20034736. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yang Y., Shen C., Li J., Yuan J., Yang M., Wang F., Li G., Li Y., Xing L., Peng L., Wei J., Cao M., Zheng H., Wu W., Zou R., Li D., Xu Z., Wang H., Zhang M., Zhang Z., Liu L., Liu Y. Exuberant elevation of IP-10, MCP-3 and IL-1ra during SARS-CoV-2 infection is associated with disease severity and fatal outcome. MedRxiv. 2020 doi: 10.1101/2020.03.02.20029975. In press. [DOI] [Google Scholar]
- 83.Patterson B.K., Seethamraju H., Dhody K., Corley M.J., Kazempour K., Lalezari J.P., Pang A.P.S., Sugai C., Francisco E.B., Pise A., Rodrigues H., Ryou M., Wu H.L., Webb G.M., Park B.S., Kelly S., Pourhassan N., Lelic A., Kdouh L., Herrera M., Hall E., Aklin E., Ndhlovu L., Sacha J.B. Disruption of the CCL5/RANTES-CCR5 pathway restores immune homeostasis and reduces plasma viral load in critical COVID-19. MedRxiv. 2020 doi: 10.1101/2020.05.02.20084673. In press. [DOI] [Google Scholar]
- 84.Zhao Y., Qin L., Zhang P., Li K., Liang L., Sun J., Xu B., Dai Y., Li X., Zhang C., Peng Y., Feng Y., Li A., Hu Z., Xiang H., Ogg G., Ho L.-P., McMichael A.J., Jin R., Knight J.C., Dong T., Zhang Y. Longitudinal COVID-19 profiling associates IL-1Ra and IL-10 with disease severity and RANTES with mild disease, JCI Insight. 2020. In press. [DOI] [PMC free article] [PubMed]
- 85.Mathew D., Giles J.R., Baxter A.E., Greenplate A.R., Wu J.E., Alanio C., Oldridge D.A., Kuri-Cervantes L., Pampena M.B., D\textquoterightAndrea K., Manne S., Chen Z., Huang Y.J., Reilly J.P., Weisman A.R., Ittner C.A.G., Kuthuru O., Dougherty J., Nzingha K., Han N., Kim J., Pattekar A., Goodwin E.C., Anderson E.M., Weirick M.E., Gouma S., Arevalo C.P., Bolton M.J., Chen F., Lacey S.F., Hensley S.E., Apostolidis S., Huang A.C., Vella L.A., Betts M.R., Meyer N.J., Wherry E.J. Deep immune profiling of COVID-19 patients reveals patient heterogeneity and distinct immunotypes with implications for therapeutic interventions. BioRxiv. 2020 doi: 10.1101/2020.05.20.106401. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Channappanavar R., Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin. Immunopathol. 2017;39:529–539. doi: 10.1007/s00281-017-0629-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Teuwen L.-A., Geldhof V., Pasut A., Carmeliet P. COVID-19: the vasculature unleashed. Nat. Rev. Immunol. 2020:1–3. doi: 10.1038/s41577-020-0343-0. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.van de Veerdonk F.L., Netea M.G., van Deuren M., van der Meer J.W., de Mast Q., Brüggemann R.J., van der Hoeven H. Kallikrein-kinin blockade in patients with COVID-19 to prevent acute respiratory distress syndrome. Elife. 2020;9 doi: 10.7554/eLife.57555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Schulze-Koops H. Lymphopenia and autoimmune diseases. Arthritis Res. Ther. 2004;6:178–180. doi: 10.1186/ar1208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Boyman O., Létourneau S., Krieg C., Sprent J. Homeostatic proliferation and survival of naïve and memory T cells. Eur. J. Immunol. 2009;39:2088–2094. doi: 10.1002/eji.200939444. [DOI] [PubMed] [Google Scholar]
- 91.Maeda K., Baba Y., Nagai Y., Miyazaki K., Malykhin A., Nakamura K., Kincade P.W., Sakaguchi N., Coggeshall K.M. IL-6 blocks a discrete early step in lymphopoiesis. Blood. 2005;106:879–885. doi: 10.1182/blood-2005-02-0456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Tan L., Wang Q., Zhang D., Ding J., Huang Q., Tang Y.-Q., Wang Q., Miao H. Lymphopenia predicts disease severity of COVID-19: a descriptive and predictive study. Signal Transduct. Target. Theroy. 2020;5:33. doi: 10.1038/s41392-020-0148-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Khoruts A., Fraser J.M. A causal link between lymphopenia and autoimmunity. Immunol. Lett. 2005;98:23–31. doi: 10.1016/j.imlet.2004.10.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Theofilopoulos A.N., Dummer W., Kono D.H. T cell homeostasis and systemic autoimmunity. J. Clin. Invest. 2001;108:335–340. doi: 10.1172/JCI200112173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Gray J.I., Westerhof L.M., MacLeod M.K.L. The roles of resident, central and effector memory CD4 T-cells in protective immunity following infection or vaccination. Immunology. 2018;154:574–581. doi: 10.1111/imm.12929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Devarajan P., Chen Z. Autoimmune effector memory T cells: the bad and the good. Immunol. Res. 2013;57:12–22. doi: 10.1007/s12026-013-8448-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Braun J., Loyal L., Frentsch M., Wendisch D., Georg P., Kurth F., Hippenstiel S., Dingeldey M., Kruse B., Fauchere F., Baysal E., Mangold M., Henze L., Lauster R., Mall M., Beyer K., Roehmel J., Schmitz J., Miltenyi S., Mueller M.A., Witzenrath M., Suttorp N., Kern F., Reimer U., Wenschuh H., Drosten C., Corman V.M., Giesecke-Thiel C., Sander L.-E., Thiel A. Presence of SARS-CoV-2 reactive T cells in COVID-19 patients and healthy donors. MedRxiv. 2020 doi: 10.1101/2020.04.17.20061440. [DOI] [PubMed] [Google Scholar]
- 98.Novelli L., Barbati C., Ceccarelli F., Perricone C., Spinelli F.R., Alessandri C., Valesini G., Perricone R., Conti F. CD44v3 and CD44v6 isoforms on T cells are able to discriminate different disease activity degrees and phenotypes in systemic lupus erythematosus patients. Lupus. 2019;28:621–628. doi: 10.1177/0961203319838063. [DOI] [PubMed] [Google Scholar]
- 99.Croft M., So T., Duan W., Soroosh P. The significance of OX40 and OX40L to T-cell biology and immune disease. Immunol. Rev. 2009;229:173–191. doi: 10.1111/j.1600-065X.2009.00766.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Zhu Y., Zhu G., Luo L., Flies A.S., Chen L. CD137 stimulation delivers an antigen-independent growth signal for T lymphocytes with memory phenotype. Blood. 2007;109:4882–4889. doi: 10.1182/blood-2006-10-043463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C., Liu S., Zhao P., Liu H., Zhu L., Tai Y., Bai C., Gao T., Song J., Xia P., Dong J., Zhao J., Wang F.-S. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020;8:420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Wang F., Nie J., Wang H., Zhao Q., Xiong Y., Deng L., Song S., Ma Z., Mo P., Zhang Y. Characteristics of peripheral lymphocyte subset alteration in COVID-19 pneumonia. J. Infect. Dis. 2020;221:1762–1769. doi: 10.1016/S1074-7613(02)00338-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Liblau R.S., Wong F.S., Mars L.T., Santamaria P. Autoreactive CD8 T cells in organ-specific autoimmunity. Immunity. 2002;17:1–6. doi: 10.1016/S1074-7613(02)00338-2. [DOI] [PubMed] [Google Scholar]
- 104.Knochelmann H.M., Dwyer C.J., Bailey S.R., Amaya S.M., Elston D.M., Mazza-McCrann J.M., Paulos C.M. When worlds collide: Th17 and Treg cells in cancer and autoimmunity. Cell. Mol. Immunol. 2018;15:458–469. doi: 10.1038/s41423-018-0004-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Xiong Y., Liu Y., Cao L., Wang D., Guo M., Jiang A., Guo D., Hu W., Yang J., Tang Z., Wu H., Lin Y., Zhang M., Zhang Q., Shi M., Liu Y., Zhou Y., Lan K., Chen Y. Transcriptomic characteristics of bronchoalveolar lavage fluid and peripheral blood mononuclear cells in COVID-19 patients, Emerg. Microb. Infect. 2020;9:761–770. doi: 10.1080/22221751.2020.1747363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Zeng Q., Li Y., Huang G., Wu W., Dong S., Xu Y. Mortality of COVID-19 is associated with cellular immune function compared to immune function in Chinese han population. MedRxiv. 2020 doi: 10.1101/2020.03.08.20031229. In press. [DOI] [Google Scholar]
- 107.Chu H., Zhou J., Wong B.H.-Y., Li C., Chan J.F.-W., Cheng Z.-S., Yang D., Wang D., Lee A.C.-Y., Li C., Yeung M.-L., Cai J.-P., Chan I.H.-Y., Ho W.-K., To K.K.-W., Zheng B.-J., Yao Y., Qin C., Yuen K.-Y. Middle East respiratory syndrome coronavirus efficiently infects human primary T lymphocytes and activates the extrinsic and intrinsic apoptosis pathways. J. Infect. Dis. 2016;213:904–914. doi: 10.1093/infdis/jiv380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Wang X., Xu W., Hu G., Xia S., Sun Z., Liu Z., Xie Y., Zhang R., Jiang S., Lu L. SARS-CoV-2 infects T lymphocytes through its spike protein-mediated membrane fusion. Cell. Mol. Immunol. 2020:2–4. doi: 10.1038/s41423-020-0424-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Thevarajan I., Nguyen T.H.O., Koutsakos M., Druce J., Caly L., van de Sandt C.E., Jia X., Nicholson S., Catton M., Cowie B., Tong S.Y.C., Lewin S.R., Kedzierska K. Breadth of concomitant immune responses prior to patient recovery: a case report of non-severe COVID-19. Nat. Med. 2020;26:453–455. doi: 10.1038/s41591-020-0819-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Zhou P., Yang X.-L., Wang X.-G., Hu B., Zhang L., Zhang W., Si H.-R., Zhu Y., Li B., Huang C.-L., Chen H.-D., Chen J., Luo Y., Guo H., Jiang R.-D., Liu M.-Q., Chen Y., Shen X.-R., Wang X., Zheng X.-S., Zhao K., Chen Q.-J., Deng F., Liu L.-L., Yan B., Zhan F.-X., Wang Y.-Y., Xiao G.-F., Shi Z.-L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Xiang F., Wang X., He X., Peng Z., Yang B., Zhang J., Zhou Q., Ye H., Ma Y., Li H., Wei X., Cai P., Ma W.-L. Antibody detection and dynamic characteristics in patients with COVID-19. Clin. Infect. Dis. 2020:1–23. doi: 10.1093/cid/ciaa461. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Xu X., Sun J., Nie S., Li H., Kong Y., Liang M., Hou J., Huang X., Li D., Ma T., Peng J., Gao S., Shao Y., Zhu H., Lau J.Y.-N., Wang G., Xie C., Jiang L., Huang A., Yang Z., Zhang K., Hou F.F. Seroprevalence of immunoglobulin M and G antibodies against SARS-CoV-2 in China. Nat. Med. 2020 doi: 10.1038/s41591-020-0949-6. [DOI] [PubMed] [Google Scholar]
- 113.Fafi-Kremer S., Bruel T., Madec Y., Grant R., Tondeur L., Grzelak L., Staropoli I., Anna F., Souque P., Mutter C., Collongues N., Bolle A., Velay A., Lefebvre N., Mielcarek M., Meyer N., Rey D., Charneau P., Hoen B., De Seze J., Schwartz O., Fontanet A. Serologic responses to SARS-CoV-2 infection among hospital staff with mild disease in eastern France. MedRxiv. 2020 doi: 10.1101/2020.05.19.20101832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.GeurtsvanKessel C.H., Okba N.M.A., Igloi Z., Embregts C.W.E., Laksono B.M., Leijten L., Rahamat-Langendoen J., van den Akker J.P.C., van Kampen J.J.A., van der Eijk A.A., van Binnendijk R.S., Haagmans B., Koopmans M. Towards the next phase: evaluation of serological assays for diagnostics and exposure assessment. MedRxiv. 2020 doi: 10.1101/2020.04.23.20077156. In press. [DOI] [Google Scholar]
- 115.Suthar M.S., Zimmerman M.G., Kauffman R.C., Mantus G., Linderman S.L., Hudson W.H., Vanderheiden A., Nyhoff L., Davis C.W., Adekunle S., Affer M., Sherman M., Reynolds S., Verkerke H.P., Alter D.N., Guarner J., Bryksin J., Horwath M., Arthur C.M., Saakadze N., Smith G.H., Edupuganti S., Scherer E.M., Hellmeister K., Cheng A., Morales J.A., Neish A.S., Stowell S.R., Frank F., Ortlund E., Anderson E., Menachery V.D., Rouphael N., Mehta A., Stephens D.S., Ahmed R., Roback J.D., Wrammert J. Rapid generation of neutralizing antibody responses in COVID-19 patients. Cell Rep. Med. 2020 doi: 10.1016/j.xcrm.2020.100040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Robbiani D.F., Gaebler C., Muecksch F., Lorenzi J.C.C., Wang Z., Cho A., Agudelo M., Barnes C.O., Gazumyan A., Finkin S., Hagglof T., Oliveira T.Y., Viant C., Hurley A., Hoffmann H.-H., Millard K.G., Kost R.G., Cipolla M., Gordon K., Bianchini F., Chen S.T., Ramos V., Patel R., Dizon J., Shimeliovich I., Mendoza P., Hartweger H., Nogueira L., Pack M., Horowitz J., Schmidt F., Weisblum Y., Michailidis E., Ashbrook A.W., Waltari E., Pak J.E., Huey-Tubman K.E., Koranda N., Hoffman P.R., West A.P., Rice C.M., Hatziioannou T., Bjorkman P.J., Bieniasz P.D., Caskey M., Nussenzweig M.C. Convergent antibody responses to SARS-CoV-2 infection in convalescent individuals. BioRxiv. 2020 doi: 10.1101/2020.05.13.092619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Bloch E.M., Shoham S., Casadevall A., Sachais B.S., Shaz B., Winters J.L., van Buskirk C., Grossman B.J., Joyner M., Henderson J.P., Pekosz A., Lau B., Wesolowski A., Katz L., Shan H., Auwaerter P.G., Thomas D., Sullivan D.J., Paneth N., Gehrie E., Spitalnik S., Hod E., Pollack L., Nicholson W.T., Pirofski L.-A., Bailey J.A., Tobian A.A. Deployment of convalescent plasma for the prevention and treatment of COVID-19. J. Clin. Invest. 2020;130(6):2757–2765. doi: 10.1172/JCI138745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Seydoux E., Homad L.J., MacCamy A.J., Parks K.R., Hurlburt N.K., Jennewein M.F., Akins N.R., Stuart A.B., Wan Y.-H., Feng J., Whaley R.E., Singh S., Boeckh M., Cohen K.W., McElrath M.J., Englund J.A., Chu H.Y., Pancera M., McGuire A.T., Stamatatos L. Analysis of a SARS-CoV-2 infected individual reveals development of potent neutralizing antibodies to distinct epitopes with limited somatic mutation. Immunity. 2020 doi: 10.1016/j.immuni.2020.06.001. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Liu L., Wei Q., Lin Q., Fang J., Wang H., Kwok H., Tang H., Nishiura K., Peng J., Tan Z., Wu T., Cheung K.-W., Chan K.-H., Alvarez X., Qin C., Lackner A., Perlman S., Yuen K.-Y., Chen Z. Anti–spike IgG causes severe acute lung injury by skewing macrophage responses during acute SARS-CoV infection. JCI Insight. 2019;4 doi: 10.1172/jci.insight.123158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Fu Y., Cheng Y., Wu Y. Understanding SARS-CoV-2-mediated inflammatory responses: from mechanisms to potential therapeutic tools. Virol. Sin. 2020 doi: 10.1007/s12250-020-00207-4. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Rojas M., Rodríguez Y., Monsalve D.M., Acosta-Ampudia Y., Camacho B., Gallo J.E., Rojas-Villarraga A., Ramírez-Santana C., Díaz-Coronado J.C., Manrique R., Mantilla R.D., Shoenfeld Y., Anaya J.-M. Convalescent plasma in Covid-19: possible mechanisms of action. Autoimmun. Rev. 2020;19(7):102554. doi: 10.1016/j.autrev.2020.102554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Joyner M.J., Wright R.S., Fairweather D., Senefeld J., Bruno K., Klassen S., Carter R., Klompas A., Wiggins C., Shepherd J.R.A., Rea R., Whelan E., Clayburn A., Spiegel M., Johnson P., Lesser E., Baker S., Larson K., Ripoll Sanz J., Andersen K., Hodge D., Kunze K., Buras M., Vogt M., Herasevich V., Dennis J., Regimbal R., Bauer P., Blair J., van Buskirk C., Winters J., Stubbs J., Paneth N., Casadevall A. Early safety indicators of COVID-19 convalescent plasma in 5,000 patients. J. Clin. Invest. 2020 doi: 10.1172/JCI140200. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Wu D., Yang X.O. TH17 responses in cytokine storm of COVID-19: an emerging target of JAK2 inhibitor Fedratinib. J. Microbiol. Immunol. Infect. 2020;53(3):368–370. doi: 10.1016/j.jmii.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Behrens E.M., Koretzky G.A. Review: cytokine storm syndrome: looking toward the precision medicine era. Arthritis Rheum. 2017;69:1135–1143. doi: 10.1002/art.40071. [DOI] [PubMed] [Google Scholar]
- 126.Wang Z., Yang B., Li Q., Wen L., Zhang R. Clinical features of 69 cases with coronavirus disease 2019 in wuhan, China. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa272. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Shimabukuro-Vornhagen A., Gödel P., Subklewe M., Stemmler H.J., Schlößer H.A., Schlaak M., Kochanek M., Böll B., von Bergwelt-Baildon M.S. Cytokine release syndrome. J. Immunother. Canc. 2018;6:56. doi: 10.1186/s40425-018-0343-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Behrens E.M. Cytokines in cytokine storm syndrome. In: Cron R.Q., Behrens E.M., editors. Cytokine Storm Syndr. Springer International Publishing; Cham: 2019. pp. 197–207. [DOI] [Google Scholar]
- 129.Tanaka T., Narazaki M., Kishimoto T. Immunotherapeutic implications of IL-6 blockade for cytokine storm. Immunotherapy. 2016;8:959–970. doi: 10.2217/imt-2016-0020. [DOI] [PubMed] [Google Scholar]
- 130.Xu X., Han M., Li T., Sun W., Wang D., Fu B., Zhou Y., Zheng X., Yang Y., Li X., Zhang X., Pan A., Wei H. Effective treatment of severe COVID-19 patients with tocilizumab. Proc. Natl. Acad. Sci. Unit. States Am. 2020;117(20):10970–10975. doi: 10.1073/pnas.2005615117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Perrone F., Ascierto P., Marata A.M., Parrella R., Popoli P., Salvarani C., Piccirillo M.C., Atripaldi L., Cascella M., Costantini M., Dolci G., Facciolongo N., Fragranza F., Massari M., Montesarchio V., Mussini C., Negri A., Tumori N., Pascale F.G. 2020. Protocol Title : " Multicenter Study on the Efficacy and Tolerability of Tocilizumab in the Treatment of Patients with COVID-19 Pneumonia; pp. 1–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Giamarellos-Bourboulis E.J., Netea M.G., Rovina N., Akinosoglou K., Antoniadou A., Antonakos N., Damoraki G., Gkavogianni T., Adami M.-E., Katsaounou P., Ntaganou M., Kyriakopoulou M., Dimopoulos G., Koutsodimitropoulos I., Velissaris D., Koufargyris P., Karageorgos A., Katrini K., Lekakis V., Lupse M., Kotsaki A., Renieris G., Theodoulou D., Panou V., Koukaki E., Koulouris N., Gogos C., Koutsoukou A. Complex immune dysregulation in COVID-19 patients with severe respiratory failure. Cell Host Microbe. 2020;27(10):992–1000.e3. doi: 10.1016/j.chom.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Duret P.-M., Sebbag E., Mallick A., Gravier S., Spielmann L., Messer L. Recovery from COVID-19 in a patient with spondyloarthritis treated with TNF-alpha inhibitor etanercept. Ann. Rheum. Dis. 2020 doi: 10.1136/annrheumdis-2020-217362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Moutsopoulos H.M. Anti-inflammatory therapy may ameliorate the clinical picture of COVID-19. Ann. Rheum. Dis. 2020 doi: 10.1136/annrheumdis-2020-217562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Mihai C., Dobrota R., Schröder M., Garaiman A., Jordan S., Becker M.O., Maurer B., Distler O. COVID-19 in a patient with systemic sclerosis treated with tocilizumab for SSc-ILD. Ann. Rheum. Dis. 2020;79:668–669. doi: 10.1136/annrheumdis-2020-217442. [DOI] [PubMed] [Google Scholar]
- 137.Guilpain P., Le Bihan C., Foulongne V., Taourel P., Pansu N., Maria A.T.J., Jung B., Larcher R., Klouche K., Le Moing V. Rituximab for granulomatosis with polyangiitis in the pandemic of covid-19: lessons from a case with severe pneumonia. Ann. Rheum. Dis. 2020 doi: 10.1136/annrheumdis-2020-217549. [DOI] [PubMed] [Google Scholar]
- 138.Sharmeen S., Elghawy A., Zarlasht F., Yao Q.P. COVID-19 in rheumatic disease patients on immunosuppressive agents. Semin. Arthritis Rheum. 2020 doi: 10.1016/j.semarthrit.2020.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Emmi G., Bettiol A., Mattioli I., Silvestri E., Di Scala G., Urban M.L., Vaglio A., Prisco D. SARS-CoV-2 infection among patients with systemic autoimmune diseases. Autoimmun. Rev. 2020;19:102575. doi: 10.1016/j.autrev.2020.102575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Rigamonti C., Cittone M.G., De Benedittis C., Rizzi E., Casciaro G.F., Bellan M., Sainaghi P.P., Pirisi M. Rates of symptomatic SARS-CoV-2 infection in patients with autoimmune liver diseases in Northern Italy: a telemedicine study. Clin. Gastroenterol. Hepatol. 2020 doi: 10.1016/j.cgh.2020.05.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Schulert G.S., Grom A.A. Macrophage activation syndrome and cytokine-directed therapies. Best Pract. Res. Clin. Rheumatol. 2014;28:277–292. doi: 10.1016/j.berh.2014.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Braciale T.J., Hahn Y.S. Immunity to viruses. Immunol. Rev. 2013;255:5–12. doi: 10.1111/imr.12109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Ramos-Casals M., Brito-Zerón P., López-Guillermo A., Khamashta M.A., Bosch X. Adult haemophagocytic syndrome. Lancet. 2014;383:1503–1516. doi: 10.1016/S0140-6736(13)61048-X. [DOI] [PubMed] [Google Scholar]
- 144.Deane S., Selmi C., Teuber S.S., Gershwin M.E. Macrophage activation syndrome in autoimmune disease. Int. Arch. Allergy Immunol. 2010;153:109–120. doi: 10.1159/000312628. [DOI] [PubMed] [Google Scholar]
- 145.Crayne C.B., Albeituni S., Nichols K.E., Cron R.Q. The immunology of macrophage activation syndrome. Front. Immunol. 2019;10 doi: 10.3389/fimmu.2019.00119. 10:119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Bin Dhuban K., Bartolucci S., D'Hennezel E., Piccirillo C.A. Signaling through gp130 compromises suppressive function in human FOXP3+ regulatory T cells. Front. Immunol. 2019;10:1–13. doi: 10.3389/fimmu.2019.01532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.O'Sullivan B.J., Thomas H.E., Pai S., Santamaria P., Iwakura Y., Steptoe R.J., Kay T.W.H., Thomas R. IL-1β breaks tolerance through expansion of CD25 + effector T cells. J. Immunol. 2006;176:7278–7287. doi: 10.4049/jimmunol.176.12.7278. [DOI] [PubMed] [Google Scholar]
- 148.Pouchot J., Arlet J.-B. Biological treatment in adult-onset Still's disease. Best Pract. Res. Clin. Rheumatol. 2012;26:477–487. doi: 10.1016/j.berh.2012.07.002. [DOI] [PubMed] [Google Scholar]
- 149.Singh J.A., Beg S., Lopez-Olivo M.A. Tocilizumab for rheumatoid arthritis. In: Singh J.A., editor. Cochrane Database Syst. Rev. John Wiley & Sons, Ltd; Chichester, UK: 2010. [DOI] [PubMed] [Google Scholar]
- 150.Zheng S.G., Wang J., Horwitz D.A. Cutting edge: foxp3 + CD4 + CD25 + regulatory T cells induced by IL-2 and TGF-β are resistant to Th17 conversion by IL-6. J. Immunol. 2008;180:7112–7116. doi: 10.4049/jimmunol.180.11.7112. [DOI] [PubMed] [Google Scholar]
- 151.Davis L.S., Hutcheson J., Mohan C. The role of cytokines in the pathogenesis and treatment of systemic lupus erythematosus. J. Interferon Cytokine Res. 2011;31:781–789. doi: 10.1089/jir.2011.0047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Li S., Zheng S., Tang S., Pan Y., Zhang S., Fang H., Qiao J. Autoinflammatory pathogenesis and targeted therapy for adult-onset Still's disease. Clin. Rev. Allergy Immunol. 2020;58:71–81. doi: 10.1007/s12016-019-08747-8. [DOI] [PubMed] [Google Scholar]
- 153.Chen D.-Y., Lin C.-C., Chen Y.-M., Lan J.-L., Hung W.-T., Chen H.-H., Lai K.-L., Hsieh C.-W. Involvement of TLR7 MyD88-dependent signaling pathway in the pathogenesis of adult-onset Still's disease. Arthritis Res. Ther. 2013;15:R39. doi: 10.1186/ar4193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Antoniou K.M., Margaritopoulos G.A., Giannarakis I., Choulaki C., Fountoulakis N., Siafakas N.M., Sidiropoulos P. Adult onset Still's disease: a case report with a rare clinical manifestation and pathophysiological correlations. Case Rep. Med. 2013:1–4. doi: 10.1155/2013/981232. 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Gerfaud-Valentin M., Jamilloux Y., Iwaz J., Sève P. Adult-onset Still's disease. Autoimmun. Rev. 2014;13:708–722. doi: 10.1016/j.autrev.2014.01.058. [DOI] [PubMed] [Google Scholar]
- 156.Baggio G. Gender Medicine: a interdisciplinary approach to medicine. G. Ital. Med. Lav. Ergon. 2017;39:196–198. [PubMed] [Google Scholar]
- 157.Angela Tincani L.A., Chiara Gerardi Maria, Fredi Micaela. Gender differences in rheumatology and the point of view of the Italian Society for Rheumatology (SIR) Italy J. Gender-Specific Med. 2018;4:e79–e82. doi: 10.1723/3013.30121. [DOI] [Google Scholar]
- 158.Crosslin K.L., Wiginton K.L. Sex differences in disease severity among patients with systemic lupus erythematosus. Gend. Med. 2011;8:365–371. doi: 10.1016/j.genm.2011.10.003. [DOI] [PubMed] [Google Scholar]
- 159.Roussou E., Sultana S. Spondyloarthritis in women: differences in disease onset, clinical presentation, and Bath Ankylosing Spondylitis Disease Activity and Functional indices (BASDAI and BASFI) between men and women with spondyloarthritides. Clin. Rheumatol. 2011;30:121–127. doi: 10.1007/s10067-010-1581-5. [DOI] [PubMed] [Google Scholar]
- 160.Quintero O.L., Amador-Patarroyo M.J., Montoya-Ortiz G., Rojas-Villarraga A., Anaya J.-M. Autoimmune disease and gender: plausible mechanisms for the female predominance of autoimmunity. J. Autoimmun. 2012;38:J109–J119. doi: 10.1016/j.jaut.2011.10.003. [DOI] [PubMed] [Google Scholar]
- 161.Ortona E., Delunardo F., Maselli A., Pierdominici M., Malorni W. Sex hormones and gender disparity in immunity and autoimmunity. Italy J. Gender-Specifi c Med. 2015;1:45–50. doi: 10.1723/2188.23636. [DOI] [Google Scholar]
- 162.Naugler W.E., Sakurai T., Kim S., Maeda S., Kim K., Elsharkawy A.M., Karin M. Gender disparity in liver cancer due to sex differences in MyD88-dependent IL-6 production. Science. 2007;317(80-):121–124. doi: 10.1126/science.1140485. [DOI] [PubMed] [Google Scholar]
- 163.Zhang Z., Qin P., Deng Y., Ma Z., Guo H., Guo H., Hou Y., Wang S., Zou W., Sun Y., Ma Y., Hou W. The novel estrogenic receptor GPR30 alleviates ischemic injury by inhibiting TLR4-mediated microglial inflammation. J. Neuroinflammation. 2018;15:206. doi: 10.1186/s12974-018-1246-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Anna Ruggieri M.C.G. Gender differences in COVID-19: some open questions. Italy. J. Gender-Specific Med. 2020;6:49–50. [Google Scholar]
- 165.Jin J.-M., Bai P., He W., Wu F., Liu X.-F., Han D.-M., Liu S., Yang J.-K. Gender differences in patients with COVID-19: focus on severity and mortality. Front. Public. Health. 2020;8:152. doi: 10.3389/fpubh.2020.00152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Cameron M.J., Kelvin A.A., Leon A.J., Cameron C.M., Ran L., Xu L., Chu Y.-K., Danesh A., Fang Y., Li Q., Anderson A., Couch R.C., Paquette S.G., Fomukong N.G., Kistner O., Lauchart M., Rowe T., Harrod K.S., Jonsson C.B., Kelvin D.J. Lack of innate Interferon responses during SARS coronavirus infection in a vaccination and reinfection ferret model. PloS One. 2012;7 doi: 10.1371/journal.pone.0045842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Bao L., Deng W., Gao H., Xiao C., Liu J., Xue J., Lv Q., Liu J., Yu P., Xu Y., Qi F., Qu Y., Li F., Xiang Z., Yu H., Gong S., Liu M., Wang G., Wang S., Song Z., Zhao W., Han Y., Zhao L., Liu X., Wei Q., Qin C. Reinfection could not occur in SARS-CoV-2 infected rhesus macaques. BioRxiv. 2020 doi: 10.1101/2020.03.13.990226. In press. [DOI] [Google Scholar]
- 168.Clay C., Donart N., Fomukong N., Knight J.B., Lei W., Price L., Hahn F., Van Westrienen J., Harrod K.S. Primary severe acute respiratory syndrome coronavirus infection limits replication but not lung inflammation upon homologous rechallenge. J. Virol. 2012;86:4234–4244. doi: 10.1128/JVI.06791-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Chen D., Xu W., Lei Z., Huang Z., Liu J., Gao Z., Peng L. Recurrence of positive SARS-CoV-2 RNA in COVID-19: a case report. Int. J. Infect. Dis. 2020;93:297–299. doi: 10.1016/j.ijid.2020.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Wu L.-P., Wang N.-C., Chang Y.-H., Tian X.-Y., Na D.-Y., Zhang L.-Y., Zheng L., Lan T., Wang L.-F., Liang G.-D. Duration of antibody responses after severe acute respiratory syndrome. Emerg. Infect. Dis. 2007;13:1562–1564. doi: 10.3201/eid1310.070576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Gorse G.J., Donovan M.M., Patel G.B. Antibodies to coronaviruses are higher in older compared with younger adults and binding antibodies are more sensitive than neutralizing antibodies in identifying coronavirus‐associated illnesses. J. Med. Virol. 2020;92:512–517. doi: 10.1002/jmv.25715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.CDC Korea . 2020. Findings from Investigation and Analysis of Re-positive Cases. [Google Scholar]
- 173.Atkinson B., Petersen E. SARS-CoV-2 shedding and infectivity. Lancet. 2020;395:1339–1340. doi: 10.1016/S0140-6736(20)30868-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Bullard J., Dust K., Funk D., Strong J.E., Alexander D., Garnett L., Boodman C., Bello A., Hedley A., Schiffman Z., Doan K., Bastien N., Li Y., Van Caeseele P.G., Poliquin G. Predicting infectious SARS-CoV-2 from diagnostic samples. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa638. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Lucchese G., Flöel A. Molecular mimicry between SARS-CoV-2 and respiratory pacemaker neurons. Autoimmun. Rev. 2020;19(7):102556. doi: 10.1016/j.autrev.2020.102556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Kanduc D., Shoenfeld Y. On the molecular determinants of the SARS-CoV-2 attack. Clin. Immunol. 2020;215:108426. doi: 10.1016/j.clim.2020.108426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Vojdani A., Kharrazian D. Potential antigenic cross-reactivity between SARS-CoV-2 and human tissue with a possible link to an increase in autoimmune diseases. Clin. Immunol. 2020;217:108480. doi: 10.1016/j.clim.2020.108480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Aguiar C.L., Erkan D. Catastrophic antiphospholipid syndrome: how to diagnose a rare but highly fatal disease. Ther. Adv. Musculoskelet. Disease. 2013;5:305–314. doi: 10.1177/1759720X13502919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Raucci F., Mansour A.A., Casillo G.M., Saviano A., Caso F., Scarpa R., Mascolo N., Iqbal A.J., Maione F. Interleukin-17A (IL-17A), a key molecule of innate and adaptive immunity, and its potential involvement in COVID-19-related thrombotic and vascular mechanisms. Autoimmun. Rev. 2020;19(7):102572. doi: 10.1016/j.autrev.2020.102572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Galván Casas C., Català A., Carretero Hernández G., Rodríguez‐Jiménez P., Fernández Nieto D., Rodríguez‐Villa Lario A., Navarro Fernández I., Ruiz‐Villaverde R., Falkenhain D., Llamas Velasco M., García‐Gavín J., Baniandrés O., González‐Cruz C., Morillas‐Lahuerta V., Cubiró X., Figueras Nart I., Selda‐Enriquez G., Romaní J., Fustà‐Novell X., Melian‐Olivera A., Roncero Riesco M., Burgos‐Blasco P., Sola Ortigosa J., Feito Rodriguez M., García‐Doval I. Classification of the cutaneous manifestations of COVID‐19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br. J. Dermatol. 2020 doi: 10.1111/bjd.19163. In pressbjd.19163 In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Shamsizadeh A., Ziaei Kajbaf T., Razavi M., Cheraghian B. Clinical and epidemiological characteristics of Kawasaki disease. Jundishapur J. Microbiol. 2014;7 doi: 10.5812/jjm.11014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Fernandez-Cooke E., Barrios Tascón A., Sánchez-Manubens J., Antón J., Grasa Lozano C.D., Aracil Santos J., Villalobos Pinto E., Clemente Garulo D., Mercader Rodríguez B., Bustillo Alonso M., Nuñez Cuadros E., Navarro Gómez M.L., Domínguez-Rodríguez S., Calvo C. Epidemiological and clinical features of Kawasaki disease in Spain over 5 years and risk factors for aneurysm development. (2011-2016): KAWA-RACE study group. PloS One. 2019;14 doi: 10.1371/journal.pone.0215665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Matsubara T., Ichiyama T., Furukawa S. Immunological profile of peripheral blood lymphocytes and monocytes/macrophages in Kawasaki disease. Clin. Exp. Immunol. 2005;141:381–387. doi: 10.1111/j.1365-2249.2005.02821.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Franco A., Shimizu C., Tremoulet A.H., Burns J.C. Memory T-cells and characterization of peripheral T-cell clones in acute Kawasaki disease. Autoimmunity. 2010;43:317–324. doi: 10.3109/08916930903405891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Rowley A.H., Baker S.C., Shulman S.T., Rand K.H., Tretiakova M.S., Perlman E.J., Garcia F.L., Tajuddin N.F., Fox L.M., Huang J.H., Ralphe J.C., Takahashi K., Flatow J., Lin S., Kalelkar M.B., Soriano B., Orenstein J.M. Ultrastructural, immunofluorescence, and RNA evidence support the hypothesis of a “new” virus associated with Kawasaki disease. J. Infect. Dis. 2011;203:1021–1030. doi: 10.1093/infdis/jiq136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Son M.B.F. Pediatric inflammatory syndrome temporally related to covid-19. BMJ. 2020;369:m2123. doi: 10.1136/bmj.m2123. [DOI] [PubMed] [Google Scholar]
- 187.Rodríguez Y., Rojas M., Pacheco Y., Acosta-Ampudia Y., Ramírez-Santana C., Monsalve D.M., Gershwin M.E., Anaya J.-M. Guillain–Barré syndrome, transverse myelitis and infectious diseases. Cell. Mol. Immunol. 2018;15:547–562. doi: 10.1038/cmi.2017.142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Baig A.M., Khaleeq A., Ali U., Syeda H. Evidence of the COVID-19 virus targeting the CNS: tissue distribution, host–virus interaction, and proposed neurotropic mechanisms. ACS Chem. Neurosci. 2020;11:995–998. doi: 10.1021/acschemneuro.0c00122. [DOI] [PubMed] [Google Scholar]
- 189.Costello F., Dalakas M.C. Cranial neuropathies and COVID-19: neurotropism and autoimmunity. Neurology. 2020 doi: 10.1212/WNL.0000000000009921. In press10.1212/WNL.0000000000009921. [DOI] [PubMed] [Google Scholar]
- 190.Ferro F B.S., Elefante E., Baldini C., Bartoloni E., Puxeddu I., Talarico R., Mosca M. COVID-19: the new challenge for rheumatologists. Clin. Exp. Rheumatol. 2020;38(3):373–382. [PubMed] [Google Scholar]
- 191.Touret F., de Lamballerie X. Of chloroquine and COVID-19. Antivir. Res. 2020;177:104762. doi: 10.1016/j.antiviral.2020.104762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Sarzi-Puttini P., Giorgi V., Sirotti S., Marotto D., Ardizzone S., Rizzardini G., Antinori S. COVID-19, cytokines and immunosuppression: what can we learn from severe acute respiratory syndrome? Clin. Exp. Rheumatol. 2020;38:337–342. G.M. [PubMed] [Google Scholar]
- 193.Ceribelli A., Motta F., De Santis M., Ansari A.A., Ridgway W.M., Gershwin M.E., Selmi C. Recommendations for coronavirus infection in rheumatic diseases treated with biologic therapy. J. Autoimmun. 2020;109:102442. doi: 10.1016/j.jaut.2020.102442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Castiblanco J., Anaya J.-M. Genetics and vaccines in the era of personalized medicine. Curr. Genom. 2015;16:47–59. doi: 10.2174/1389202916666141223220551. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.