Abstract
Background
Delays in the initiation of therapy among patients with early stage breast cancer (BC) can negatively affect outcomes. Patients treated with neoadjuvant systemic chemotherapy (NSC) usually display tumors with high‐risk features. Considering these high‐risk characteristics and the evidence supporting adverse outcomes associated with delays in adjuvant chemotherapy initiation, we sought to determine whether a delay in NSC initiation is associated with overall survival (OS).
Methods
We identified patients diagnosed between January 1995 and December 2015 with invasive primary BC (stage I–III) who received NSC at MD Anderson Cancer Center. Patients were categorized according to their time from BC diagnosis to NSC (in days) into three subgroups: 0‐30, 31–60, and ≥61 days. Primary endpoint was OS. Descriptive statistics and Cox's proportional hazard models were used.
Results
A total of 5,137 patients were included. Median follow‐up was 6.5 years. The 5‐year OS estimates according to time to NSC were 87%, 85%, and 83% in patients who received NSC within 0–30, 31–60, and ≥61 days after diagnosis, respectively (p = .006). In multivariable analysis, compared with time to NSC of 0–30 days, delayed NSC ≥61 days was associated with an increased risk of death (31–60 days: hazard ratio [HR] = 1.05 [95% confidence interval (CI) 0.92–1.19]; ≥61 days, HR = 1.28 [95% CI 1.06–1.54]). In stratified analyses, the association between delay in NSC initiation and increased risk of death was statistically significant for patients with stage I and II BC (31–60 days: HR = 1.22 [95% CI 1.02–1.47]; ≥61 days, HR = 1.41 [95% CI 1.07–1.86]) and among patients with HER2‐positive tumors ( ≥61 days, HR = 1.86 [95% CI 1.21–2.86]).
Conclusion
A delay in NSC initiation of more than 61 days after BC diagnosis was associated with an increased risk of death. Early initiation of NSC should be a priority; multidisciplinary teams must focus on coordination of care and patient‐centered, timely treatment planning and delivery.
Implications for Practice
The results of this study showed that a delay in neoadjuvant systemic chemotherapy initiation of more than 61 days after breast cancer diagnosis is associated with an increased risk of death; therefore, efforts must focus on early initiation of therapy, which should be a priority. Multidisciplinary teams must enhance coordination of care and patient‐centered, timely treatment planning and delivery.
Keywords: Locoregional breast cancer, Neoadjuvant chemotherapy, Time to systemic therapy
Short abstract
Considering the high‐risk characteristics of many patients treated with neoadjuvant systemic chemotherapy (NSC), among other factors, it is clinically relevant to determine whether delays in NSC administration are associated with poor outcomes. This article evaluates the relationship between time from breast cancer diagnosis to NSC and overall survival.
Introduction
Breast cancer (BC) mortality has considerably decreased over the last decades [1]. Undeniably, advances in systemic treatment substantially contributed to this improvement [2]. Adjuvant chemotherapy reduces breast cancer mortality and risk of recurrence, with substantial benefit for a large proportion of patients [3].
Delays in therapy, from surgery to systemic treatment, can profoundly impact survival outcomes in BC patients. Data from the Surveillance, Epidemiology, and End Results–Medicare database and the National Cancer Database demonstrated that greater time to surgery was associated with lower overall and disease‐specific survival [4]. Furthermore, delays in time to adjuvant chemotherapy initiation have an impact in survival outcomes. In a previously reported study by our group, a delay of more than 60 days in adjuvant chemotherapy initiation after definitive breast surgery was associated with a detriment in overall survival (OS) [5]. Other retrospective studies and pooled analyses have also linked a delay in adjuvant chemotherapy initiation to worse prognosis in early stage BC [6, 7, 8, 9]. In a large population‐based study using data from the California Cancer Registry [10], we demonstrated that individuals who initiated adjuvant chemotherapy ≥91 days from surgery experienced worse OS and breast cancer–specific survival.
Patients treated with neoadjuvant systemic chemotherapy (NSC) usually display tumors with high‐risk features, in terms of both biology and disease volume [11, 12, 13]. Although initially NSC was used mainly in the treatment of inoperable or locally advanced tumors, today its use has increased significantly. NSC is associated with increased rates of breast conserving surgery (BCS), allows for an in vivo assessment of tumor response, and provides important prognostic information [14, 15, 16, 17].
Considering the high‐risk features characteristic of many patients treated with NSC, the evidence supporting the adverse outcomes associated with delays in the administration of adjuvant chemotherapy, and the complexities associated with coordination of care by the multidisciplinary team, it is clinically relevant to determine whether delays in NSC administration are associated with poor outcomes. In this retrospective study, we sought to evaluate the relationship between time from BC diagnosis to NSC and OS. We hypothesized that a longer time between breast cancer diagnosis and NSC initiation would be associated with worse OS.
Subjects, Materials, and Methods
Patient Population
We performed a retrospective review of the Breast Medical Oncology Institutional database at the University of Texas MD Anderson Cancer Center (MDACC). We identified 8,797 women diagnosed with primary invasive BC (stage I–III) between January 1995 and December 2015 who received NSC at our institution. We excluded 30 patients with duplicate records and 3,008 patients who received neoadjuvant chemotherapy outside MDACC. Out of the 5,759 patients who received neoadjuvant chemotherapy at MDACC, we excluded 133 patients with HER2‐negative BC who received trastuzumab‐based chemotherapy. We excluded another 42 patients whose time interval from diagnosis to NST was greater than 6 months because that is a deviation from normal practice. We excluded 375 patients with inflammatory breast cancer and 72 patients who had received nonstandard chemotherapeutic agents. All patients received an anthracycline‐based and/or a taxane‐based chemotherapy. Once anti‐HER2 therapy was incorporated to the standard treatment of early stage BC after 2005, patients with HER2‐positive tumors received trastuzumab‐based chemotherapy. The final analysis included 5,137 patients.
Endpoints and Statistical Analysis
Patients were categorized according to their time from BC pathologic diagnosis to NSC (in days) into one of the three subgroups: 0‐30, 31–60, and ≥61 days. Patient clinical, pathologic, and treatment characteristics were evaluated. Patient characteristics included age at diagnosis, self‐reported race/ethnicity, body mass index, and type and number of comorbidities (osteoporosis, hypertension, dyslipidemia, heart disease, diabetes, pulmonary disease, kidney, liver, or neurologic disease). Pathologic characteristics included nuclear grade, breast cancer histology, lymphovascular invasion (LVI), and tumor subtype categorized as hormone receptor positive (estrogen receptor [ER] positive and/or progesterone receptor [PR] positive), HER2 positive (regardless of ER and PR status), and triple negative. Cancer stage was determined by clinical evaluation, in addition to dedicated breast and axillary imaging. Surgery was classified either as BCS or mastectomy. Chemotherapy was assorted as anthracycline plus taxane‐based, anthracycline‐based, or trastuzumab‐based. Patients with HER2‐positive tumors were categorized according to trastuzumab administration.
Univariate relationships between time from diagnosis to NSC intervals and baseline characteristics were evaluated using the chi‐square test. In a multivariable logistic regression model, we evaluated the association between baseline characteristics and initiation of NSC ≥61 days after diagnosis; results are expressed in odds ratios and 95% confidence intervals (CIs). The primary outcome was OS, measured from the date of BC histopathological diagnosis to date of death for any cause. Five‐year OS was calculated using the Kaplan‐Meier product limit method according to time interval categories. Groups were compared using the log‐rank statistic. Cox proportional hazards models were fit to determine the association between time to NSC (as a categorical and as a continuous variable) and survival after adjustment for other potential confounders. Variables in the model included age, race/ethnicity, number of comorbidities, body mass index, clinical stage at presentation, grade, histology, LVI, and subtype.
In order to address potential residual confounding, we implemented a Cox proportional hazards models including propensity score adjustment. The propensity score was based on the multivariable logistic regression model to determine factors associated with receipt of NSC ≥61 days. Furthermore, we evaluated the time to NSC as a continuous variable in the Cox proportional hazards model; results were expressed in hazard ratios (HRs) and 95% CIs. Values of p ≤ .05 were considered statistically significant; all tests were two‐sided and were not adjusted for multiple comparisons. Statistical analyses were carried out using SAS version 9.3 (SAS Institute Inc., Cary, NC) and R (version 3.5.1). This study underwent evaluation and approval by MDACC Institutional Review Board; it was considered exempt for consent given the code of regulations.
Results
A total of 5,137 patients were included; median follow‐up was 6.5 years. A total of 2,225 patients (43.3%) received NSC within 30 days of diagnosis, 2,231 (43.4%) within 31–60 days, and 681 (13.3%) ≥61 days after diagnosis. Median time to NSC was 34 days; Figure 1 illustrates time to NSC intervals according to year of diagnosis. Patient, tumor, and treatment characteristics stratified by time to NSC are shown in Table 1. A longer time to NSC was observed among older and obese patients, non‐Hispanic blacks, Hispanics, those with two or more comorbidities, and patients with a hormone receptor–positive subtype. In multivariable analysis (Table 2), NSC administration ≥61 days after diagnosis was associated with ethnicity other than white, obesity, and having two or more comorbidities.
Figure 1.
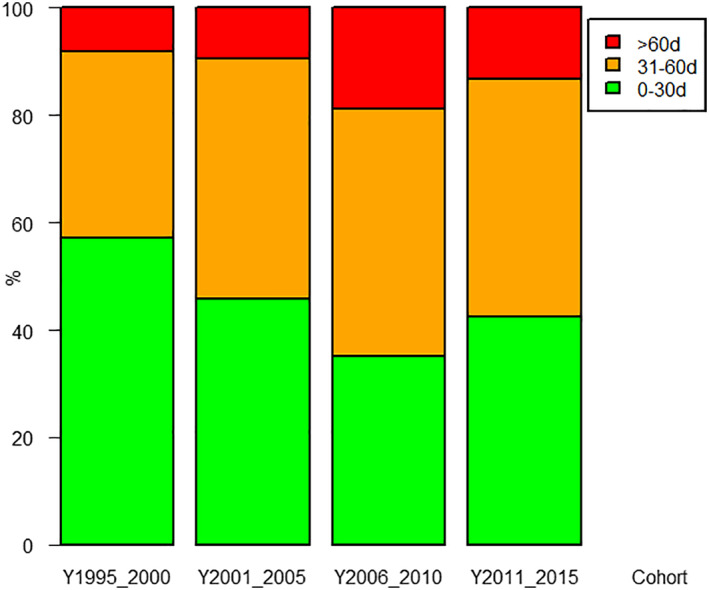
Time to neoadjuvant systemic chemotherapy according to year of diagnosis.
Table 1.
Patient and tumor characteristics according to time from diagnosis to neoadjuvant systemic chemotherapy
| Characteristics | All patients (n = 5,137) n (%) | Time to NSC 0–30 days (n = 2,225) n (%) | Time to NSC 31–60 days (n = 2,231) n (%) | Time to NSC ≥61 days (n = 681) n (%) | p value |
|---|---|---|---|---|---|
| Age | |||||
| Median (range) | 50 (19, 87) | 49 | 50 | 51 | |
| Age ≤50 | 2,449 (47.7) | 1,120 (50.3) | 1,017 (45.6) | 312 (45.8) | .004 |
| Age >50 | 2,688 (52.3) | 1,105 (49.7) | 1,214 (54.4) | 369 (54.2) | |
| Race | |||||
| Non‐Hispanic white | 3,188 (62.1) | 1,561 (70.2) | 1,344 (60.2) | 283 (41.6) | <.001 |
| Non‐Hispanic black | 781 (15.2) | 204 (9.2) | 382 (17.1) | 195 (28.6) | |
| Hispanic | 783 (15.2) | 293 (13.2) | 337 (15.1) | 153 (22.5) | |
| Other | 385 (7.5) | 167 (7.5) | 168 (7.5) | 50 (7.3) | |
| Body mass index | |||||
| Normal | 1,638 (31.9) | 810 (36.4) | 659 (29.5) | 169 (24.8) | <.001 |
| Overweight | 1,541 (30) | 681 (30.6) | 675 (30.3) | 185 (27.2) | |
| Obese | 1,861 (36.2) | 690 (31) | 857 (38.4) | 314 (46.1) | |
| Unknown | 97 (1.9) | 44 (2) | 40 (1.8) | 13 (1.9) | |
| Clinical stage | |||||
| I or II | 3,279 (63.8) | 1,429 (64.2) | 1,438 (64.5) | 412 (60.5) | .15 |
| III | 1,858 (36.2) | 796 (35.8) | 793 (35.5) | 269 (39.5) | |
| Nuclear grade | |||||
| I or II | 1,744 (33.9) | 712 (32) | 793 (35.5) | 239 (35.1) | .012 |
| III | 3,178 (61.9) | 1,434 (64.4) | 1,331 (59.7) | 413 (60.6) | |
| Unknown | 215 (4.2) | 79 (3.6) | 107 (4.8) | 29 (4.3) | |
| Histology | |||||
| Ductal | 4,438 (86.4) | 1,921 (86.3) | 1,929 (86.5) | 588 (86.3) | .99 |
| Other | 699 (13.6) | 304 (13.7) | 302 (13.5) | 93 (13.7) | |
| Lymphovascular invasion | |||||
| Negative | 3,888 (75.7) | 1,676 (75.3) | 1,693 (75.9) | 519 (76.2) | .28 |
| Positive | 1,177 (22.9) | 526 (23.6) | 499 (22.4) | 152 (22.3) | |
| Unknown | 72 (1.4) | 23 (1) | 39 (1.7) | 10 (1.5) | |
| Breast cancer subtype | |||||
| HmR positive | 2,379 (46.3) | 968 (43.5) | 1,066 (47.8) | 345 (50.7) | <.001 |
| Her2neu positive | 1,210 (23.6) | 537 (24.1) | 516 (23.1) | 157 (23.1) | |
| Triple negative | 1,097 (21.4) | 480 (21.6) | 474 (21.2) | 143 (21) | |
| Unknown | 451 (8.8) | 240 (10.8) | 175 (7.8) | 36 (5.3) | |
| Comorbidity | |||||
| 0 | 903 (17.6) | 454 (20.4) | 349 (15.6) | 100 (14.7) | <.001 |
| 1 | 1,539 (30) | 678 (30.5) | 663 (29.7) | 198 (29.1) | |
| 2+ | 1,244 (24.2) | 494 (22.2) | 559 (25.1) | 191 (28) | |
| Unknown | 1,451 (28.2) | 599 (26.9) | 660 (29.6) | 192 (28.2) | |
| Chemotherapy | |||||
| Anthracycline | 90 (1.8) | 32 (1.4) | 41 (1.8) | 17 (2.5) | .27 |
| Anthracycline + taxanes | 3,851 (75) | 1,683 (75.6) | 1,674 (75) | 494 (72.5) | |
| Other | 277 (5.4) | 129 (5.8) | 109 (4.9) | 39 (5.7) | |
| Trastuzumab | 919 (17.9) | 381 (17.1) | 407 (18.2) | 131 (19.2) |
Abbreviations: Her2neu, human epidermal growth factor receptor 2; HmR, hormone receptor; NSC, neoadjuvant systemic chemotherapy.
Table 2.
Multivariable logistic regression model for receiving neoadjuvant systemic chemotherapy ≥61 days
| Characteristics | Odds ratio (95% CI) | p value |
|---|---|---|
| Age | ||
| Age ≤50 | 1 | |
| Age >50 | 1.08 (0.91–1.29) | .38 |
| Race | ||
| Non‐Hispanic white | 1 | |
| Non‐Hispanic black | 3.28 (2.65–4.05) | <.001 |
| Hispanic | 2.56 (2.05–3.18) | <.001 |
| Other | 1.63 (1.18–2.26) | .003 |
| Body mass index | ||
| Normal | 1 | |
| Overweight | 1.04 (0.83–1.31) | .72 |
| Obese | 1.36 (1.1–1.69) | .004 |
| Clinical stage | ||
| I/II | 1 | |
| III | 1.1 (0.92–1.31) | .28 |
| Nuclear grade | ||
| I/II | 1 | |
| III | 0.89 (0.73–1.08) | .23 |
| Histology | ||
| Ductal | 1 | |
| Other | 1.05 (0.81–1.35) | .73 |
| Lymphovascular invasion | ||
| Negative | 1 | |
| Positive | 0.97 (0.79–1.19) | .76 |
| Breast cancer subtype | ||
| HmR positive | 1 | |
| Her2neu positive | 0.9 (0.72–1.12) | .35 |
| Triple negative | 0.83 (0.66–1.04) | .11 |
| Comorbidity | ||
| 0 | 1 | |
| 1 | 1.16 (0.89–1.52) | .26 |
| 2+ | 1.39 (1.05–1.83) | .02 |
Abbreviations: CI, confidence interval; Her2neu, human epidermal growth factor receptor 2; HmR, hormone receptor.
At the time of analysis 1,109 patients (21.6%) had died. Table 3 summarizes the unadjusted 5‐year OS for the complete cohort according to time to NSC categories. The 5‐year OS of the entire cohort was 86%. The 5‐year OS estimates were 87%, 85%, and 83% in patients who started NSC 0–30, 31–60, and ≥61 days after diagnosis, respectively (p = .006). Figure 2 illustrates OS Kaplan‐Meier curves for different NSC initiation time categories. In multivariable Cox proportional hazards model for overall survival (Table 4), after adjustment for potential confounders, we observed that compared with a time from diagnosis to NSC of 0–30 days, initiation ≥61 days was associated with a statistically significant increased risk of death (31–60 days: HR, 1.05; 95% CI, 0.90–1.19; ≥61 days: HR, 1.28; 95% CI, 1.06–1.54). Older age, non‐Hispanic black race, stage III disease, positive LVI, and triple‐negative BC (TNBC) subtype were also associated with increased risks of death. To address potential residual bias, we additionally adjusted for propensity score of receiving NSC ≥61 days in the multivariable Cox proportional hazards model, and results were similar (supplemental online Table 1). Compared with NSC initiation in 0–30 days, initiation of NSC ≥61 days was associated with a statistically significant increased risk of death (≥61 days: HR, 1.28; 95% CI, 1.06–1.54). Alternatively, we grouped patients into time from diagnosis to NSC 0–60 days versus ≥61 days, demonstrating that initiation of NSC ≥61 days was associated with a statistically significant increased risk of death (HR, 1.25; 95% CI, 1.05–1.48). Consistently, when evaluating time to NSC as a continuous variable, we observed that one day to NSC initiation was associated with a statistically significant increased risk of death (HR, 1.003; 95% CI, 1.001–1.006).
Table 3.
Five‐year overall survival estimates according to patient and tumor characteristics for all patients
| Characteristics | Patients, n | Events, n | 5‐year OS (95% CI) | p value |
|---|---|---|---|---|
| All | 5,137 | 1,109 | 0.86 (0.84–0.87) | |
| Time from dx to NSC | ||||
| 0–30 days | 2,225 | 478 | 0.87 (0.85–0.88) | |
| 31–60 days | 2,231 | 470 | 0.85 (0.84–0.87) | |
| ≥61 days | 681 | 161 | 0.83 (0.79–0.86) | .006 |
| Age | ||||
| Age ≤50 | 2,449 | 474 | 0.86 (0.85–0.88) | |
| Age >50 | 2,688 | 635 | 0.85 (0.83–0.86) | <.001 |
| Race | ||||
| Non‐Hispanic white | 3,188 | 718 | 0.86 (0.85–0.87) | |
| Non‐Hispanic black | 781 | 212 | 0.78 (0.75–0.81) | |
| Hispanic | 783 | 125 | 0.9 (0.87–0.92) | |
| Other | 385 | 54 | 0.88 (0.84–0.91) | <.001 |
| Body mass index | ||||
| Normal | 1,195 | 298 | 0.86 (0.84–0.88) | |
| Overweight | 1,153 | 294 | 0.86 (0.84–0.88) | |
| Obese | 1,244 | 354 | 0.83 (0.8–0.85) | .003 |
| Clinical stage | ||||
| I or II | 3,279 | 565 | 0.9 (0.89–0.91) | |
| III | 1,858 | 544 | 0.77 (0.75–0.79) | <.001 |
| Nuclear grade | ||||
| I or II | 1,744 | 342 | 0.91 (0.9–0.93) | |
| III | 3,178 | 737 | 0.82 (0.81–0.83) | <.001 |
| LVI | ||||
| Negative | 3,888 | 694 | 0.89 (0.88–0.9) | |
| Positive | 1,177 | 396 | 0.76 (0.73–0.78) | <.001 |
| Subtype | ||||
| HmR positive | 2,379 | 491 | 0.89 (0.87–0.9) | |
| Her2neu positive | 1,210 | 178 | 0.89 (0.87–0.91) | |
| Triple negative | 1,097 | 309 | 0.75 (0.72–0.78) | <.001 |
Abbreviations: CI, confidence interval; dx, diagnosis; Her2neu, human epidermal growth factor receptor 2; HmR, hormone receptor; LVI, lymphovascular invasion; NSC, neoadjuvant systemic chemotherapy; OS, overall survival.
Figure 2.
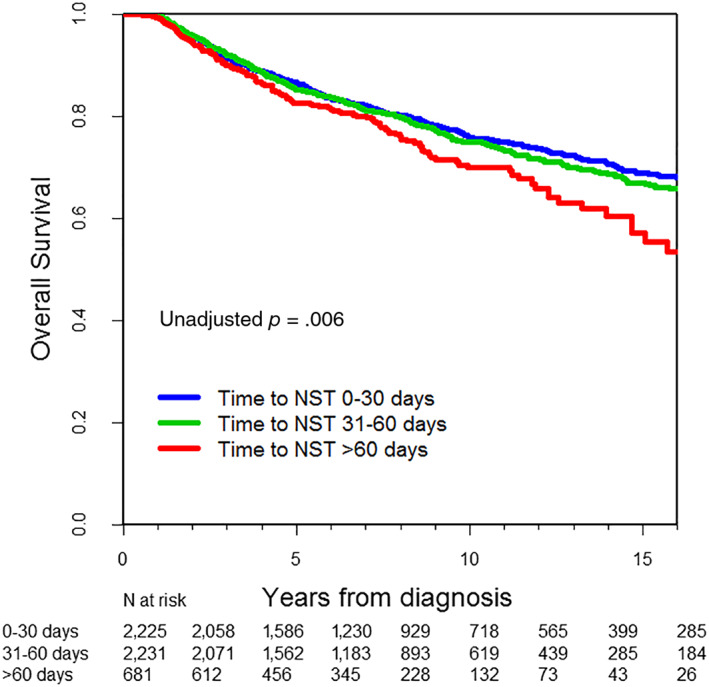
Kaplan‐Meier estimates of overall survival according to time to neoadjuvant systemic chemotherapy.
Abbreviation: NST, neoadjuvant systemic therapy.
Table 4.
Multivariable Cox's proportional hazards model for overall survival for all patients (n = 5,137)
| Overall survival, hazard ratio (95% CI) | p value | |
|---|---|---|
| Time to NSC: 31–60 days vs. 0–30 days | 1.05 (0.92–1.19) | .47 |
| Time to NSC: ≥61 days vs. 0–30 days | 1.28 (1.06–1.54) | .009 |
| Age at diagnosis: ≥50 vs. <50 | 1.39 (1.22–1.57) | <.001 |
| Race: NH black vs. NH white | 1.18 (1.00–1.39) | .049 |
| Race: Hispanic vs. NH white | 0.75 (0.62–0.92) | .005 |
| Race: Other vs. NH white | 0.83 (0.63–1.10) | .19 |
| Clinic stage: III vs. I–II | 1.89 (1.67–2.13) | <.001 |
| Grade: 3 vs. 1–2 | 1.12 (0.97–1.30) | .12 |
| Histology: other vs. ductal | 1.00 (0.83–1.19) | .96 |
| Lymphovascular invasion: yes vs. no | 1.84 (1.62–2.09) | <.001 |
| Subtype: HER2+ vs. HmR+ | 0.75 (0.62–0.89) | .001 |
| Subtype: triple negative vs. HmR+ | 1.65 (1.41–1.93) | <.001 |
Other variables in the model included comorbidities and body mass index.
Abbreviations: CI, confidence interval; HmR, hormone receptor; NSC, neoadjuvant systemic chemotherapy; NH, non‐Hispanic.
Table 5 illustrates 5‐year survival estimates by patient and clinical characteristics according to BC stage and subtype. In the univariate subgroup analyses, the detrimental effect of delays in time to NSC was seen for patients with stage I and II (p = .04) and stage III (p = .05). Subgroup analysis according to BC subtype revealed that among 1,210 patients with HER2‐positive BC, the unadjusted 5‐year OS was 92%, 90%, and 82% for time to NSC intervals of 0–30, 31–60, and ≥61 days, respectively (p = .011). As for the 919 patients with HER2‐positve tumors who received trastuzumab‐based chemotherapy, the unadjusted 5‐year OS estimates were 94%, 92%, and 85% for time to NSC intervals of 0–30, 31–60, and ≥61 days, respectively (p = .02). Subgroup analyses among patients with hormone receptor–positive BC and TNBC revealed no significant impact of time to NSC with survival.
Table 5.
Five‐year survival estimates according to breast cancer subtype and stage
| Subtype or stage | Patients, n | Events, n | 5‐year overall survival (95% CI) | p value |
|---|---|---|---|---|
| Hormone receptor positive | ||||
| Time to NSC | ||||
| 0–30 days | 968 | 198 | 0.89 (0.87–0.91) | |
| 31–60 days | 1066 | 223 | 0.89 (0.86–0.9) | |
| ≥61 days | 345 | 70 | 0.87 (0.82–0.9) | .21 |
| HER2 positive | ||||
| Time to NSC | ||||
| 0–30 days | 537 | 73 | 0.92 (0.88–0.94) | |
| 31–60 days | 516 | 72 | 0.9 (0.86–0.92) | |
| ≥61 days | 157 | 33 | 0.82 (0.75–0.88) | .011 |
| HER2 positive with trastuzumab | ||||
| Time to NSC | ||||
| 0–30 days | 396 | 26 | 0.94 (0.9–0.96) | |
| 31–60 days | 418 | 39 | 0.92 (0.89–0.94) | |
| ≥61 days | 133 | 20 | 0.85 (0.77–0.9) | .02 |
| HER2 positive without trastuzumab | ||||
| Time to NSC | ||||
| 0–30 days | 141 | 47 | 0.87 (0.8–0.91) | |
| 31–60 days | 98 | 33 | 0.8 (0.7–0.87) | |
| ≥61 days | 24 | 13 | 0.7 (0.47–0.84) | .027 |
| Triple negative | ||||
| Time to NSC | ||||
| 0–30 days | 480 | 137 | 0.75 (0.71–0.79) | |
| 31–60 days | 474 | 126 | 0.75 (0.71–0.79) | |
| ≥61 days | 143 | 46 | 0.75 (0.66–0.81) | .35 |
| Stage I–II | ||||
| Time to NSC | ||||
| 0–30 days | 1429 | 238 | 0.91 (0.89–0.92) | |
| 31–60 days | 1438 | 255 | 0.89 (0.88–0.91) | |
| ≥61 days | 412 | 72 | 0.9 (0.87–0.93) | .04 |
| Stage III | ||||
| Time to NSC | ||||
| 0–30 days | 796 | 240 | 0.79 (0.76–0.82) | |
| 31–60 days | 793 | 215 | 0.78 (0.75–0.81) | |
| ≥61 days | 269 | 89 | 0.71 (0.65–0.77) | .05 |
Abbreviations: CI, confidence interval; NSC, neoadjuvant systemic chemotherapy.
Supplemental online Table 2 shows the multivariable model for OS and time from diagnosis to NSC according to subtype and stage. In the stratified multivariable models, among patients with HER2‐positive BC, a time to NSC of ≥61 days was associated with increased risk of death (31–60 days: HR, 1.08; 95% CI, 0.77–1.50; ≥61 days: HR, 1.86; 95% CI, 1.21–2.86). Among patients with HER2‐positive BC treated with trastuzumab, the impact of a delay in NSC of ≥61 days was of greater magnitude (HR, 2.23; 95% CI, 1.20–4.12). Among patients with stage I or II disease, delayed administration of NSC of 31–61 days and ≥61 days was associated with increased risk of death (31–60 days: HR, 1.22; 95% CI, 1.02–1.47; ≥61 days: HR, 1.41; 95% CI, 1.07–1.86) compared with patients who started NSC within the first 30 days after diagnosis. The HR estimates among patients with stage III BC or TNBC suggested an association between treatment delay and increased risk; however, no statistical significance was reached.
Discussion
In this large retrospective cohort, we observed that time to NSC initiation after BC diagnosis significantly influenced survival outcomes. Compared with a time interval of ≤30 days, a time from BC diagnosis to NSC initiation of ≥61 days was clearly associated with a detrimental effect in OS. In subgroup analyses, the association between a delay in NSC initiation and an increased risk of death was evident across all stages and among those with HER2‐positive BC, regardless of trastuzumab administration. The magnitude of the detrimental effect of a delayed time to NSC seemed to be greater among trastuzumab‐treated patients, likely highlighting the importance of early administration of highly effective treatments. Our findings are of clinical relevance and reinforce the importance of early initiation of NSC.
In recent years it has become evident that timely initiation of systemic chemotherapy is crucial. We and others have shown that a delay in adjuvant chemotherapy initiation is associated with worse OS among patients with early stage BC [5, 6, 7, 8, 9, 10]. A recent meta‐analysis including 12 studies evaluating the impact of time to adjuvant chemotherapy showed that a 4‐week delay in treatment initiation was associated with significantly worse OS (HR, 1.13; 95% CI, 1.08–1.19) and disease‐free survival (HR, 1.14; 95% CI, 1.05–1.24) [18]. These data and the increased use of NSC make the evaluation of the relationship between time to NSC and survival a priority.
Most studies evaluating the impact of time to treatment among patients receiving NSC evaluated time from completion of NSC to breast definitive surgery, and data evaluating time to NSC are particularly limited [19, 20]. Our group previously evaluated the outcomes associated between time from NSC completion to surgery according to different time intervals (<4 weeks, 4–6 weeks, or ≥6 weeks). There were no differences in relapse‐free survival (RFS); however, a sensitivity analysis suggested worse OS in the small number of patients who underwent surgery >8 weeks after completion of NSC [19]. In contrast, a small retrospective study by Omarini et al. reported that among 319 patients who completed NSC, those undergoing surgery 21 days or more after completion of NSC had worse RFS and OS [20].
The German Breast Group evaluated the relationship between the time interval from diagnosis to NSC and the time from last chemotherapy administration to breast definitive surgery and outcomes. Among 9,127 clinical trial participants, time to NSC was not associated with pathologic complete response. Furthermore, a delay in time to NSC was not associated with a deleterious impact in relapse or death. Although the complete data form this analysis have not been published, the time to NSC cutoffs evaluated were ≤28 or >28 days, likely explaining the difference in results when compared with our study, in which a deleterious effect was seen among patients who initiated NSC 61 days or more after diagnosis. In addition, their overall median time to NSC was 23 days compared with 34 in our study [21]. A small retrospective study (n = 720) by Sebai et al. evaluated the impact of the time interval between BC diagnosis to NSC initiation and the time interval between NSC to surgery. The authors could not establish a relationship between time to NSC and survival; given the small number of events in each cohort, no definitive conclusion was reached [22].
The present study represents the first original data that pointedly demonstrate the detrimental effect associated with a delay in NSC administration. Our data are valuable for our patients and for the multidisciplinary team, highlighting the importance of timely coordination of care. We demonstrate that delays in initiation of highly effective chemotherapy are detrimental. It is likely that delays in the initiation of highly effective treatment are associated with progression of micrometastatic disease and even the possibility of increase in resistant clones. In our previous work evaluating time to adjuvant chemotherapy, we observed that the detrimental effect of delays in adjuvant chemotherapy initiation were of greater magnitude among patients with HER2‐positive tumors and those with TNBC. In the current study, although delays in NSC initiation were associated in the entire cohort in our multivariable model, in stratified analyses we only observed detrimental outcomes among HER2‐positive patients. We did not observe the same phenomenon among patients with triple‐negative tumors. In patients with this aggressive phenotype, it is likely that for those with chemoresistant tumors, time to NSC administration is irrelevant in the setting of ineffective therapy. In addition, it is also possible that given the small number of patients and events in this category (143 and 46, respectively), statistical significance could not be reached.
The use of NSC has increased, and today, chemotherapy as an initial treatment strategy is common even among patients with operable tumors. Furthermore, it is likely that even more patients will be treated with NSC given recent data demonstrating the advantage of tailoring adjuvant treatment based on the presence of residual disease at the time of definitive breast surgery [23, 24], making our data relevant for current and future practice. Administration of NSC requires coordinated, complex, multidisciplinary care. In our cohort, the majority of patients initiated NSC within 60 days after diagnosis. We observed that the proportion of patients who had access to an early initiation of NSC within the first 30 days after diagnosis did not change over time, being relatively stable between years 2000 and 2015, reinforcing the complexity involved in multidisciplinary team coordination. We must note, however, that the proportion of patients receiving chemotherapy ≥31 days from the time of initial diagnosis increased in the time interval of 2006 to 2010 compared with previous cohorts. Although the exact causes of such increase are not clear, it is likely that practice changes associated with this time frame, including the introduction of HER2 determination, contributed.
Another important finding from our study was the observation that obesity, African American race, and Hispanic ethnicity were characteristics associated with longer time between diagnosis and NSC initiation. Disparities in socioeconomic factors, such as poverty, nonwhite race, and lack of a scholarship degree have been described as contributing factors associated with treatment delays and poor prognosis [25, 26]. A previous study demonstrated that black and Hispanic women have double the risk, compared with white women, of failing to receive necessary adjuvant treatments [27].
We acknowledge the limitations associated with the retrospective nature of our study. Although we cannot exclude residual confounding, however, we were able to incorporate important clinical variables in our multivariable model and additionally implement propensity score adjustment. Furthermore, our cohort is very large, and our patients were evaluated and treated in a homogenous way by the same multidisciplinary team. Although prospective data will be ideal to answer this important question, a prospective randomized trial will likely never be conducted because of obvious ethical aspects.
Conclusion
In summary, this large retrospective study pointedly demonstrates a detriment effect in OS among patients with BC who delay NSC initiation more than 61 days. Among patients being considered for NSC, who usually display high clinical and pathologic risk features, all efforts should be made by the multidisciplinary team to ensure a timely initiation of cytotoxic chemotherapy. As the care of patients with breast cancer increases in complexity, we must remember that the management of our patients should be guided by a cohesive and synchronic team of cancer specialists, who should be united in a multidisciplinary group in order to formulate and execute treatment recommendations in an organized and efficient manner.
Author Contributions
Conception/design: Debora de Melo Gagliato, Xiudong Lei, Sharon H. Giordano, Vicente Valero, Carlos H. Barcenas, Gabriel N. Hortobagyi, Mariana Chavez‐MacGregor
Provision of study material or patients: Debora de Melo Gagliato, Xiudong Lei, Sharon H. Giordano, Vicente Valero, Carlos H. Barcenas, Gabriel N. Hortobagyi, Mariana Chavez‐MacGregor
Collection and/or assembly of data: Debora de Melo Gagliato, Xiudong Lei, Sharon H. Giordano, Vicente Valero, Carlos H. Barcenas, Gabriel N. Hortobagyi, Mariana Chavez‐MacGregor
Data analysis and interpretation: Debora de Melo Gagliato, Xiudong Lei, Sharon H. Giordano, Vicente Valero, Carlos H. Barcenas, Gabriel N. Hortobagyi, Mariana Chavez‐MacGregor
Manuscript writing: Debora de Melo Gagliato, Xiudong Lei, Sharon H. Giordano, Vicente Valero, Carlos H. Barcenas, Gabriel N. Hortobagyi, Mariana Chavez‐MacGregor
Final approval of manuscript: Debora de Melo Gagliato, Xiudong Lei, Sharon H. Giordano, Vicente Valero, Carlos H. Barcenas, Gabriel N. Hortobagyi, Mariana Chavez‐MacGregor
Disclosures
The authors indicated no financial relationships.
Supporting information
See http://www.TheOncologist.com for supplemental material available online.
Table S1. Multivariable Cox proportional hazards model for OS including propensity score adjustment
Table S2: Adjusted hazard ratio for Overall Survival according to time from diagnosis to NSC. Subgroup analysis according to tumor subtype and stage.
Acknowledgments
This study was supported by a cancer center support grant from the National Cancer Institute to the University of Texas MD Anderson Cancer Center (NIH/NCI P30CA016672), Susan G. Komen SAC150061, CPRIT‐CERCIT 2.0 RP 160674, and the Conquer Cancer Foundation.
Disclosures of potential conflicts of interest may be found at the end of this article.
No part of this article may be reproduced, stored, or transmitted in any form or for any means without the prior permission in writing from the copyright holder. For information on purchasing reprints contact Commercialreprints@wiley.com. For permission information contact permissions@wiley.com.
Footnotes
For Further Reading: Ramon Colomer, Cristina Saura, Pedro Sánchez‐Rovira et al. Neoadjuvant Management of Early Breast Cancer: A Clinical and Investigational Position Statement. The Oncologist 2019;24:603–611.
Implications for Practice: Neoadjuvant treatment should be strongly considered as a therapeutic option for localized breast cancer and is a powerful tool for understanding breast cancer biology and investigating new treatment approaches.
References
- 1. Guo F, Kuo YF, Shih YCT et al. Trends in breast cancer mortality by stage at diagnosis among young women in the United States. Cancer 2018;124:3500–3509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Welch HG, Prorok PC, O'Malley AJ et al. Breast‐cancer tumor size, overdiagnosis, and mammography screening effectiveness. N Engl J Med 2016;375:1438–1447. [DOI] [PubMed] [Google Scholar]
- 3. Early Breast Cancer Trialists’ Collaborative Group ; Peto R, Davies C, Godwin J et al. Comparisons between different polychemotherapy regimens for early breast cancer: Meta‐analyses of long‐term outcome among 100,000 women in 123 randomised trials. Lancet 2012;379:432–444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Bleicher RJ, Ruth K, Sigurdson ER et al. Time to surgery and breast cancer survival in the United States. JAMA Oncol 2016;2:330–339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Gagliato Dde M, Gonzalez‐Angulo AM, Lei X et al. Clinical impact of delaying initiation of adjuvant chemotherapy in patients with breast cancer. J Clin Oncol 2014;32:735–744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Colleoni M, Bonetti M, Coates AS et al. Early start of adjuvant chemotherapy may improve treatment outcome for premenopausal breast cancer patients with tumors not expressing estrogen receptors. The International Breast Cancer Study Group. J Clin Oncol 2000;18:584–590. [DOI] [PubMed] [Google Scholar]
- 7. Lohrisch C, Paltiel C, Gelmon K et al. Impact on survival of time from definitive surgery to initiation of adjuvant chemotherapy for early‐stage breast cancer. J Clin Oncol 2006;24:4888–4894. [DOI] [PubMed] [Google Scholar]
- 8. McLaughlin JM, Anderson RT, Ferketich AK et al. Effect on survival of longer intervals between confirmed diagnosis and treatment initiation among low‐income women with breast cancer. J Clin Oncol 2012;30:4493–4500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Abdel‐Rahman O. Impact of timeliness of adjuvant chemotherapy and radiotherapy on the outcomes of breast cancer; a pooled analysis of three clinical trials. Breast 2018;38:175–180. [DOI] [PubMed] [Google Scholar]
- 10. Chavez‐MacGregor M, Clarke CA, Lichtensztajn DY et al. Delayed initiation of adjuvant chemotherapy among patients with breast cancer. JAMA Oncol 2016;2:322–329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Graham PJ, Brar MS, Foster T et al. Neoadjuvant chemotherapy for breast cancer, is practice changing? A population‐based review of current surgical trends. Ann Surg Oncol 2015;22:3376–3382. [DOI] [PubMed] [Google Scholar]
- 12. Mougalian SS, Soulos PR, Killelea BK et al. Use of neoadjuvant chemotherapy for patients with stage I to III breast cancer in the United States. Cancer 2015;121:2544–2552. [DOI] [PubMed] [Google Scholar]
- 13. Onitilo AA, Onesti JK, Single RM et al. Utilization of neoadjuvant chemotherapy varies in the treatment of women with invasive breast cancer. PLoS One 2013;8:e84535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Early Breast Cancer Trialists’ Collaborative Group . Long‐term outcomes for neoadjuvant versus adjuvant chemotherapy in early breast cancer: Meta‐analysis of individual patient data from ten randomised trials. Lancet Oncol 2018;19:27–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Boughey JC, Suman VJ, Mittendorf EA et al. Sentinel lymph node surgery after neoadjuvant chemotherapy in patients with node‐positive breast cancer: The ACOSOG Z1071 (Alliance) clinical trial. JAMA 2013;310:1455–1461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Mamtani A, Barrio AV, King TA et al. How often does neoadjuvant chemotherapy avoid axillary dissection in patients with histologically confirmed nodal metastases? Results of a prospective study. Ann Surg Oncol 2016;23:3467–3474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Hayes DF, Schott AF. Neoadjuvant chemotherapy: What are the benefits for the patient and for the investigator? J Natl Cancer Inst Monogr 2015;2015:36–39. [DOI] [PubMed] [Google Scholar]
- 18. Zhan QH, Fu JQ, Fu FM et al. Survival and time to initiation of adjuvant chemotherapy among breast cancer patients: A systematic review and meta‐analysis. Oncotarget 2018;9:2739–2751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Sanford RA, Lei X, Barcenas CH et al. Impact of time from completion of neoadjuvant chemotherapy to surgery on survival outcomes in breast cancer patients. Ann Surg Oncol 2016;23:1515–1521. [DOI] [PubMed] [Google Scholar]
- 20. Omarini C, Guaitoli G, Noventa S et al. Impact of time to surgery after neoadjuvant chemotherapy in operable breast cancer patients. Eur J Surg Oncol 2017;43:613–618. [DOI] [PubMed] [Google Scholar]
- 21. Loibl S, Werutsky G, Nekljudova V et al. Impact in delay of starting chemotherapy and surgery on pCR and survival in breast cancer: A pooled analysis of individual patient data from six prospectively randomized neoadjuvant trials. J Clin Oncol 2017;35(suppl 15):571a.27870569 [Google Scholar]
- 22. Sebai ME, Psoter KJ, Gilmore RC et al. Survival outcomes of neoadjuvant chemotherapy timing start in relation to date of diagnosis and surgery in cases of breast cancer. J Am Coll Surg 2017;225(suppl 1):S25–S26. [Google Scholar]
- 23. Masuda N, Lee SJ, Ohtani S et al. Adjuvant capecitabine for breast cancer after preoperative chemotherapy. N Engl J Med 2017;376:2147–2159. [DOI] [PubMed] [Google Scholar]
- 24. von Minckwitz G, Huang CS, Mano MS et al. Trastuzumab emtansine for residual invasive HER2‐positive breast cancer. N Engl J Med 2019;380:617–628. [DOI] [PubMed] [Google Scholar]
- 25. Ward E, Jemal A, Cokkinides V et al. Cancer disparities by race/ethnicity and socioeconomic status. CA Cancer J Clin 2004;54:78–93. [DOI] [PubMed] [Google Scholar]
- 26. Albain KS, Unger JM, Crowley JJ et al. Racial disparities in cancer survival among randomized clinical trials patients of the Southwest Oncology Group. J Natl Cancer Inst 2009;101:984–992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Bickell NA, Wang JJ, Oluwole S et al. Missed opportunities: Racial disparities in adjuvant breast cancer treatment. J Clin Oncol 2006;24:1357–1362. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
See http://www.TheOncologist.com for supplemental material available online.
Table S1. Multivariable Cox proportional hazards model for OS including propensity score adjustment
Table S2: Adjusted hazard ratio for Overall Survival according to time from diagnosis to NSC. Subgroup analysis according to tumor subtype and stage.


