Abstract
Pseudoaneurysm rupture of the gastroduodenal artery (GDA) is life-threatening and can present as an acute upper gastrointestinal haemorrhage. Here, we present a case of upper gastrointestinal haemorrhage arising from a ruptured GDA pseudoaneurysm. A 56-year-old woman presented acutely with haematemesis. She reported ongoing upper epigastric pain for a few weeks. Laboratory evaluation revealed severe microcytic hypochromic anaemia (haemoglobin, 69 g/L; normal, 120–140 g/L) and a mildly raised serum amylase level. Upper gastrointestinal endoscopy revealed dark blood collection between the rugae of the distal stomach. An abdominal CT scan detected a homogeneously enhancing rounded lesion arising from the GDA adjacent to the second part of the duodenum. The median arcuate ligament was causing stenosis of the coeliac axis origin. The diagnosis of haematemesis secondary to a ruptured GDA pseudoaneurysm was confirmed by mesenteric angiography, and aneurysmal embolisation was done. The haemoglobin level stabilised after aneurysmal embolisation.
Keywords: GI bleeding, medical education, radiology, interventional radiology
Background
Pseudoaneurysm rupture of the gastroduodenal artery (GDA) is a seriously fatal condition and is considered a rare cause of upper gastrointestinal haemorrhage. Given its rarity, these lesions usually pass silently undiagnosed until they manifest as a potential life-threatening haemorrhage. As a consequence, the diagnosis requires a high level of suspicion as prompt intervention is life-saving.
Case presentation
An otherwise well 56-year-old woman presented acutely with haematemesis. She reported ongoing non-specific intermittent upper epigastric pain in the previous few weeks. Her medical background was significant for moderate, intermittent alcohol consumption since her 30s. Apart from mild epigastric tenderness, clinical examination was unremarkable. Laboratory evaluation revealed a severe microcytic hypochromic anaemia (haemoglobin level of 89 g/L (normal range, 120–140 g/L) that dropped to 69 g/L within 2 hours of admission) and a mildly raised serum amylase level. Liver function tests and coagulation indices (prothrombin time, activated partial thromboplastin time and international normalised ratio) were unremarkable.
Investigations
Upper gastrointestinal endoscopy revealed dark blood collection between the rugae of the distal stomach (figure 1). No oesophageal varices, oesophageal tears or peptic ulcerations were present. A portovenous abdominal CT scan depicted a calcified and atrophic pancreas consistent with chronic pancreatitis (figure 2). No radiological features of chronic liver disease or liver cirrhosis were seen. In particular, no radiological evidence of portosystemic shunting was present. On mesenteric artery evaluation, a homogeneously enhancing rounded lesion arising from the GDA was seen. This lesion was noted to be in very close proximity to the pancreas and the second part of the duodenum (figure 3A, B). The median arcuate ligament was causing stenosis of the coeliac axis origin and leading to a poststenotic dilatation (figure 4A, B).
Figure 1.
Upper gastrointestinal endoscopy. Note the dark blood within the distal gastric rugae (red arrows). No peptic ulceration or oesophageal varices were noted. The white material (yellow arrows) represents food particles that were washable during the procedure.
Figure 2.
Contrast-enhanced CT scan of the abdomen. The axial slice shows an atrophic pancreas with multiple calcified foci (red arrow) consistent with chronic pancreatitis. Mild peripancreatic fat stranding is seen, suggesting ongoing inflammation. A, aorta; K, kidney; L, liver; S, spleen.
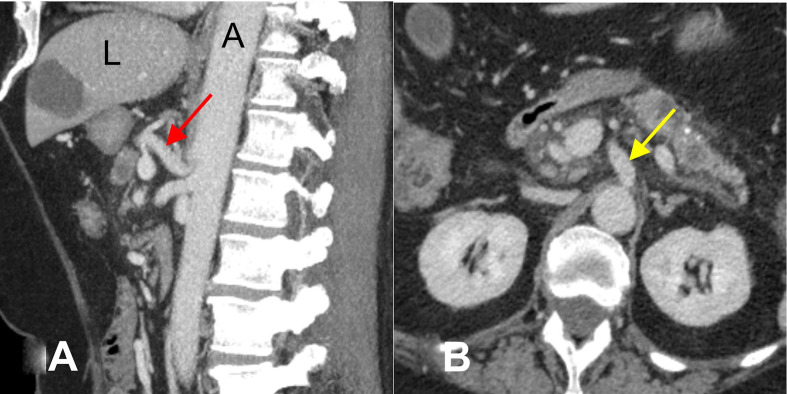
Figure 3.
Contrast-enhanced CT scan of the abdomen. (A) Coronal and (B) axial images showing a well-defined homogeneously enhancing 2 cm×1.5 cm lesion representing the gastroduodenal artery pseudoaneurysm (red arrow) encased by the pancreas (yellow arrow) and very close to the second part of the duodenum (blue arrow). H, heart; K, kidney; L, liver; S, stomach.
Figure 4.
Contrast-enhanced CT scan of the abdomen. (A) Sagittal image showing the origin of the coeliac trunk (red arrow). An incidental liver cyst is noted. (B) Note the stenosed origin of the coeliac trunk in the axial image secondary to external compression by the median arcuate ligament and the associated poststenotic dilatation (yellow arrow). A, aorta; L, liver.
Differential diagnosis
Common causes of upper gastrointestinal haemorrhage include peptic ulcer disease, oesophageal tears (Mallory-Weiss tears) and oesophageal varices. Although the background of chronic alcohol consumption and clinical presentation can favour any of these entities, upper gastrointestinal endoscopy excluded these conditions. Furthermore, abdominal CT imaging did not detect any features of chronic liver disease or portal hypertension, making these diagnoses unlikely. Moreover, CT imaging depicted the presence of GDA aneurysm that was in close proximity to the pancreas. Successful coiling of the aneurysm led to stabilisation of haemoglobin level with no further episodes of haematemesis.
Treatment
The diagnosis of an upper gastrointestinal haemorrhage secondary to a dual-aetiology (alcohol-induced chronic pancreatitis and external compression of the coeliac trunk by the median arcuate ligament) ruptured GDA pseudoaneurysm was made. The aneurysm was confirmed on mesenteric angiography and embolised successfully (figures 5, 6A, B and 7A-C). The haemoglobin level stabilised after embolisation and transfusion. Colonoscopy carried out to exclude other causes of iron deficiency anaemia and melaena was unremarkable.
Figure 5.
Volume-rendered three-dimensional image showing the visceral vascular anatomy. (A) Normally, the coeliac axis (CA, thick solid arrow) trifurcates into the splenic artery (SA, long arrows), common hepatic artery (CHA, arrowhead) and left gastric artery (LGA, open black arrow). The CHA bifurcates into the gastroduodenal artery (GDA, open white arrow) and the proper hepatic artery (PHA). The PHA bifurcates into the right (small arrow) and left hepatic arteries (curved arrow). (B) Anatomical variation in the origin of the hepatic artery. The right hepatic artery (arrowheads) originates from the superior mesenteric artery (black arrow). Note the CA trifurcating into the LGA (open black arrow), SA (long arrow) and CHA. The CHA bifurcates into the left hepatic artery (curved arrow) and GDA (open white arrow). Sometimes, the whole CHA originates from the SMA (see figure 6). Images courtesy of Dr Nilgün Özbülbül.34
Figure 6.
Direct catheter mesenteric angiography. (A) Coeliac trunk angiogram. Note the contrast ‘blush’ indicating the gastroduodenal artery pseudoaneurysm (red arrow). (B) Superior mesenteric artery angiogram. Interestingly, the common hepatic artery (red arrow) is displayed, and the anatomical variant originates from the superior mesenteric artery (yellow arrow) instead of adopting a normal origin from the coeliac trunk (see figure 5).
Figure 7.
Mesenteric embolisation. (A) Initial coiling of the afferent ‘backdoor’ feeding pancreaticoduodenal artery (red arrow). (B) Embolisation of gastroduodenal artery pseudoaneurysm with multiple small coils (red arrow). Note that a small amount of contrast is still seen within the gastroduodenal artery and the pseudoaneurysm (yellow arrows). (C) Further coiling of the feeding gastroduodenal artery (red arrow). Note that no contrast is seen extending into the pseudoaneurysm, indicating successful embolisation.
Outcome and follow-up
The patient was reviewed in the outpatient clinic 2 weeks after discharge. No further episode of haematemesis was reported. The haemoglobin level remained within target range. The patient was advised to avoid alcohol consumption and lead a healthy lifestyle.
Discussion
Visceral artery aneurysms are extremely rare, with a reported incidence of 0.01%–0.2% in the general population.1 Within such a rare entity, GDA aneurysms are one of the least common pathologies documented in the literature, representing only 1.5% of all visceral aneurysms.2 3 The pathogenesis of gastroduodenal pseudoaneurysms is multifactorial. The most common cause is vascular wall damage caused by leakage of proteolytic enzymes in up to one-tenth of patients with pancreatitis.4 5 The time interval to the development of this complication is variable and can range from days to years after the acute episode.6 Increased retrograde vascular blood flow secondary to coeliac axis stenosis with subsequent alteration in the normal haemodynamic pattern has been well described as an aetiological factor.7–9 In up to 44% of cases, such stenosis can be a result of external compression caused by median arcuate ligament.10 Other causes of mesenteric aneurysm formation include trauma, iatrogenic hepatobiliary injury and atherosclerosis.11 In the presented case, upper gastrointestinal endoscopy excluded common causes such as peptic ulcer disease, Mallory-Weiss tears, and oesophageal varices as underlying culprits although the background of chronic alcohol consumption was favourable. The findings of the CT scan suggested that the external compression of the coeliac axis and alcohol-induced chronic pancreatitis have interplayed in the pathogenesis of the pseudoaneurysm.
A literature review extending from 1956 to 2011 depicted gastrointestinal haemorrhage secondary aneurysmal rupture as the major mode of presentation in 52% of cases, whereas abdominal pain was documented in 46%.3 Pseudoaneurysmal rupture can take several clinical forms. An aneurysmal rupture into the duodenum may present with haematemesis, melaena and haemodynamic instability.11 Here, the mortality rate can be as high as 21%.12–14 Rupture can also occur in the retroperitoneal or intraperitoneal space with an associated mortality of 19%.14–16 Furthermore, haemorrhage in the pancreatic duct leading to haemosuccus pancreaticus and in the common bile duct leading to anaemia and jaundice has also been described.17 Our case presented with abdominal pain and haematemesis. With the lack of evidence to support the common culprits such as peptic ulceration and oesophageal varices and in conjunction with the angiographic findings, pseudoaneurysmal rupture was pinned down as the unifying diagnosis. Here, we postulate that the upper gastrointestinal haemorrhage is secondary to the rupture of the aneurysm into the pancreatic duct from which blood managed to track into the duodenum and then upstream to accumulate in the stomach and downstream, leading to melaena. The presence of blood in the distal stomach on gastroscopy without underlying peptic ulceration and the very close proximity of the pseudoaneurysm to the pancreas on CT support this theory. Furthermore, the lack of evidence to support duodenal perforation on endoscopy and CT excluded aneurysmal bleeding directly into the duodenum. Hyperacute haemorrhage is isodense on CT, and when scanning is carried out early enough, it may be difficult to detect blood on imaging.
Visceral catheter angiography is the gold standard for aneurysmal evaluation and carries the highest image resolution in comparison with other imaging modalities.18–21 Selective angiography can influence both diagnosis and endovascular management and should be used in symptomatic patients where conventional cross-sectional imaging is suspicious for a pseudoaneurysm.5 22 Multidetector CT and magnetic resonance angiography can elegantly depict the pseudoaneurysm as a well-delineated rounded lesion originating from the GDA. High-attenuation or high-signal intensity thrombus may be seen within the aneurysmal sac on unenhanced CT scans and fat-suppressed T1-weighted MRIs. In the absence of thrombosis, the sac may fill with contrast material in contrast-enhanced studies.23 24 The diagnostic accuracy of CT imaging using dynamic techniques may be comparable with the accuracy of selective angiography and carries the advantage of being non-invasive and able to detect associated abnormalities such as features of acute pancreatitis.25 Doppler ultrasound may reveal turbulent arterial blood flow within or adjacent to a pseudocyst, raising the suspicion for an aneurysm.3 Plain X-ray of the abdomen is rarely helpful but may show shell-like calcifications in an atherosclerotic aneurysm or a calcified pancreas.26
Unlike other aneurysms, gastroduodenal pseudoaneurysms have a higher risk of rupture regardless of their morphology, making proactive treatment necessary irrespective of the aneurysmal size.27–29 Furthermore, there is an associated increased rupture risk when the pseudoaneurysm is associated with inflammatory, infective or connective tissue disease.30 Various therapeutic approaches can be adopted to manage pseudoaneurysms, including open surgery (aneurysmectomy, ligation and bypass grafting), laparoscopic surgery (mainly ligation) or endovascular therapy (embolisation and endografting).31 Endovascular therapy is effective for high-risk patients with multiple comorbidities and those with a history of abdominal surgery, for whom intraperitoneal adhesions are a concern. A recent systematic review and meta-analysis suggested that endovascular therapy is associated with shorter hospital stay and lower rates of cardiovascular complications.32 These results may justify the endovascular approach as a first-line treatment. Furthermore, following rupture, the benefit of such an approach is comparable to open surgery as an open repair is more complex and is associated with higher morbidity.33
Learning points.
Gastroduodenal artery pseudoaneurysm rupture is a serious and fatal complication that can affect patients with chronic pancreatitis.
Given their rarity, these lesions usually pass undiagnosed until picked up incidentally on imaging or manifest as a potential life-threatening haemorrhage.
To diagnose, a high level of suspicion is required, and prompt aneurysm embolisation is life-saving.
Visceral angiography is the gold standard for evaluation, and both invasive and non-invasive therapeutic options are available depending on the patient’s clinical circumstances.
Footnotes
Twitter: @mehsimabid
Contributors: GA is the main author who contributed to writing the whole article. MA contributed to writing the clinical presentation section and carried out patient consenting. SH provided interventional radiology input and image selection. SM is the supervising consultant.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent for publication: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Zuhaili B, Molnar RG, Malhotra NG. The endovascular management of a 3.5-cm gastroduodenal artery aneurysm presenting with gastritis and recurrent pancreatitis. Avicenna J Med 2017;7:130–2. 10.4103/ajm.AJM_3_17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lu M, Weiss C, Fishman EK, et al. Review of visceral aneurysms and pseudoaneurysms. J Comput Assist Tomogr 2015;39:1–6. 10.1097/RCT.0000000000000156 [DOI] [PubMed] [Google Scholar]
- 3.Habib N, Hassan S, Abdou R, et al. Gastroduodenal artery aneurysm, diagnosis, clinical presentation and management: a Concise review. Ann Surg Innov Res 2013;7:4. 10.1186/1750-1164-7-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bergert H, Hinterseher I, Kersting S, et al. Management and outcome of hemorrhage due to arterial pseudoaneurysms in pancreatitis. Surgery 2005;137:323–8. 10.1016/j.surg.2004.10.009 [DOI] [PubMed] [Google Scholar]
- 5.Boudghène F, L'Herminé C, Bigot JM. Arterial complications of pancreatitis: diagnostic and therapeutic aspects in 104 cases. J Vasc Interv Radiol 1993;4:551–8. 10.1016/S1051-0443(93)71920-X [DOI] [PubMed] [Google Scholar]
- 6.Sahni VA, Mortelé KJ. The bloody pancreas: MDCT and MRI features of hypervascular and hemorrhagic pancreatic conditions. AJR Am J Roentgenol 2009;192:923–35. 10.2214/AJR.08.1602 [DOI] [PubMed] [Google Scholar]
- 7.Vogler C, Faiss J, Krause FJ, et al. [Aneurysm of the gastroduodenal artery with aplasia of the celiac trunk]. Chirurg 1991;62:503–4. [PubMed] [Google Scholar]
- 8.Gouny P, Fukui S, Aymard A, et al. Aneurysm of the gastroduodenal artery associated with stenosis of the superior mesenteric artery. Ann Vasc Surg 1994;8:281–4. 10.1007/BF02018176 [DOI] [PubMed] [Google Scholar]
- 9.Schweizer W, Gries NC, Maddern G, et al. Splenic infarction complicating ligation of a gastroduodenal artery aneurysm. Dig Surg 1999;16:236–7. 10.1159/000018714 [DOI] [PubMed] [Google Scholar]
- 10.Bonardelli S, Spampinato B, Ravanelli M, et al. The role of emergency presentation and revascularization in aneurysms of the peripancreatic arteries secondary to celiac trunk or superior mesenteric artery occlusion. J Vasc Surg 2020;72:46S–55. 10.1016/j.jvs.2019.11.051 [DOI] [PubMed] [Google Scholar]
- 11.Skok PSA. Acute hemorrhage into the peritoneal cavity -a complication of chronic pancreatitis with pseudocyst : a case report from clinical practice. [PubMed]
- 12.Kueper MA, Ludescher B, Koenigsrainer I, et al. Successful coil embolization of a ruptured gastroduodenal artery aneurysm. Vasc Endovascular Surg 2007;41:568–71. 10.1177/1538574407305461 [DOI] [PubMed] [Google Scholar]
- 13.Rowsell C, Moore TL, Streutker CJ. Aneurysm of the gastroduodenal artery presenting as a bleeding duodenal ulcer. Clin Gastroenterol Hepatol 2006;4:A28. 10.1016/j.cgh.2006.03.009 [DOI] [PubMed] [Google Scholar]
- 14.Moore E, Matthews MR, Minion DJ, et al. Surgical management of peripancreatic arterial aneurysms. J Vasc Surg 2004;40:247–53. 10.1016/j.jvs.2004.03.045 [DOI] [PubMed] [Google Scholar]
- 15.Eckhauser FE, Stanley JC, Zelenock GB, et al. Gastroduodenal and pancreaticoduodenal artery aneurysms: a complication of pancreatitis causing spontaneous gastrointestinal hemorrhage. Surgery 1980;88:335–44. [PubMed] [Google Scholar]
- 16.Harris K, Chalhoub M, Koirala A. Gastroduodenal artery aneurysm rupture in hospitalized patients: an overlooked diagnosis. World J Gastrointest Surg 2010;2:291–4. 10.4240/wjgs.v2.i9.291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fodor M, Fodor L, Ciuce C. Gastroduodenal artery pseudoaneurysm ruptured in the common bile duct. Acta Chir Belg 2010;110:103–5. 10.1080/00015458.2010.11680579 [DOI] [PubMed] [Google Scholar]
- 18.Jaffe TA, Nelson RC, Johnson GA, et al. Optimization of multiplanar reformations from isotropic data sets acquired with 16-detector row helical CT scanner. Radiology 2006;238:292–9. 10.1148/radiol.2381050404 [DOI] [PubMed] [Google Scholar]
- 19.Lin CT, Chiang CW, Hsieh HC. Extrasplenic pseudoaneurysm. The role of color flow Doppler ultrasound in diagnosis. Jpn Heart J 1999;40:365–8. 10.1536/jhj.40.365 [DOI] [PubMed] [Google Scholar]
- 20.Derchi LE, Biggi E, Cicio GR, et al. Aneurysms of the splenic artery: noninvasive diagnosis by pulsed Doppler sonography. J Ultrasound Med 1984;3:41–4. 10.7863/jum.1984.3.1.41 [DOI] [PubMed] [Google Scholar]
- 21.Pilleul F, Beuf O. Diagnosis of splanchnic artery aneurysms and pseudoaneurysms, with special reference to contrast enhanced 3D magnetic resonance angiography: a review. Acta Radiol 2004;45:702–8. 10.1080/02841850410001358 [DOI] [PubMed] [Google Scholar]
- 22.Yeh TS, Jan YY, Jeng LB, et al. Massive extra-enteric gastrointestinal hemorrhage secondary to splanchnic artery aneurysms. Hepatogastroenterology 1997;44:1152–6. [PubMed] [Google Scholar]
- 23.Deb P, Sahni H, Bhatoe HS. Cystic angiomatous meningioma in the cerebellopontine angle mimicking hemangioblastoma. J Cancer Res Ther 2010;6:560–3. 10.4103/0973-1482.77074 [DOI] [PubMed] [Google Scholar]
- 24.Mortelé KJ, Mergo PJ, Taylor HM, et al. Peripancreatic vascular abnormalities complicating acute pancreatitis: contrast-enhanced helical CT findings. Eur J Radiol 2004;52:67–72. 10.1016/j.ejrad.2003.10.006 [DOI] [PubMed] [Google Scholar]
- 25.Pérez C, Llauger J, Pallardó Y, et al. Radiologic diagnosis of pseudoaneurysms complicating pancreatitis. Eur J Radiol 1993;16:102–6. 10.1016/0720-048X(93)90005-8 [DOI] [PubMed] [Google Scholar]
- 26.Deterling RA. Aneurysm of the visceral arteries. J Cardiovasc Surg 1971;12:309–22. [PubMed] [Google Scholar]
- 27.Erben Y, Brownstein AJ, Rajaee S, et al. Natural history and management of splanchnic artery aneurysms in a single tertiary referral center. J Vasc Surg 2018;68:1079–87. 10.1016/j.jvs.2017.12.057 [DOI] [PubMed] [Google Scholar]
- 28.Björck M, Koelemay M, Acosta S, et al. Editor's Choice - Management of the Diseases of Mesenteric Arteries and Veins: Clinical Practice Guidelines of the European Society of Vascular Surgery (ESVS). Eur J Vasc Endovasc Surg 2017;53:460–510. 10.1016/j.ejvs.2017.01.010 [DOI] [PubMed] [Google Scholar]
- 29.Corey MR, Ergul EA, Cambria RP, et al. The natural history of splanchnic artery aneurysms and outcomes after operative intervention. J Vasc Surg 2016;63:949–57. 10.1016/j.jvs.2015.10.066 [DOI] [PubMed] [Google Scholar]
- 30.Hosn MA, Xu J, Sharafuddin M, et al. Visceral artery aneurysms: decision making and treatment options in the new era of minimally invasive and endovascular surgery. Int J Angiol 2019;28:11–16. 10.1055/s-0038-1676958 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Obara H, Kentaro M, Inoue M, et al. Current management strategies for visceral artery aneurysms: an overview. Surg Today 2020;50:38–49. 10.1007/s00595-019-01898-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Barrionuevo P, Malas MB, Nejim B, et al. A systematic review and meta-analysis of the management of visceral artery aneurysms. J Vasc Surg 2019;70:1694–9. 10.1016/j.jvs.2019.02.024 [DOI] [PubMed] [Google Scholar]
- 33.Laganà D, Carrafiello G, Mangini M, et al. Multimodal approach to endovascular treatment of visceral artery aneurysms and pseudoaneurysms. Eur J Radiol 2006;59:104–11. 10.1016/j.ejrad.2006.02.004 [DOI] [PubMed] [Google Scholar]
- 34.Ozbülbül NI. Ct angiography of the celiac trunk: anatomy, variants and pathologic findings. Diagn Interv Radiol 2011;17:150–7. 10.4261/1305-3825.DIR.3283-10.1 [DOI] [PubMed] [Google Scholar]