Abstract
Background
Muscular weakness and/or muscle wasting is recognized as a key medical problem in critically ill patients on intensive care units (ICUs) worldwide.
Methods and Results
Intensive care unit‐acquired weakness (ICUAW) results from various diseases leading to critical illness and is observed in about 40% [1080/2686 patients, 95% confidence interval (CI): 38–42%] of mixed (medical–surgical) ICU patients. Muscle strength at ICU discharge is directly associated with mortality 5 years after discharge [hazard ratio 0.946, 95% CI: 0.928–0.968 per point increase in Medical Research Council (MRC) scores, P = 0.001]. ICUAW serves as umbrella term for the subgroups ‘critical illness myopathy’, ‘critical illness polyneuropathy’, and ‘critical illness polyneuromyopathy’, the latter distinguished using electrophysiology and/or biopsy studies. Diagnosing, studying, and developing treatments for ICUAW among the critically ill seems challenging due to the acuity and severity of the underlying heterogeneous diseases. Ventilator‐induced diaphragmatic dysfunction occurs in up to 80% (n = 32/40) of ICUAW patients after mechanical ventilation and mostly results from distinct muscular pathologies, disuse, underlying critical illness, and/or effects imposed directly by mechanical ventilation. Swallowing disorders/dysphagia likely represent an additional (local) neuromuscular dysfunction/ICUAW sequelae and presents in 10.3% (n = 96/933) of mixed medical–surgical ICU survivors, with 60.4% (n = 58/96) of patients remaining dysphagia positive until hospital discharge. Key independent risk factors for dysphagia following mechanical ventilation are baseline neurological disease [odds ratio (OR) 4.45, 95% CI: 2.74–7.24, P < 0.01], emergency admission (OR 2.04, 95% CI: 1.15–3.59, P < 0.01), days on mechanical ventilation (OR 1.19, 95% CI: 1.06–1.34, P < 0.01), days on renal replacement therapy (OR 1.1, 95% CI: 1–1.23, P = 0.03), and disease severity (Acute Physiology and Chronic Health Evaluation II score within first 24 h; OR 1.03, 95% CI: 0.99–1.07, P < 0.01). Dysphagia positivity independently predicts 28‐day and 90‐day mortality (90‐day univariate hazard ratio: 3.74; 95% CI, 2.01–6.95; P < 0.001) and is associated with a 9.2% excess (all‐cause) mortality rate.
Conclusions
Neuromuscular weakness and muscle wasting is observed in many survivors of critical illness. ICUAW, ventilator‐induced diaphragmatic dysfunction, and dysphagia are associated with complicated and prolonged ICU stay, impaired weaning from mechanical ventilation, impeded rehabilitative measures, and a considerable impact on morbidity and mortality is noted. Future research strategies should further explore underlying pathomechanisms and lead to development of causal treatment strategies.
Introduction
Reduced muscular force, as in intensive care unit (ICU)‐acquired weakness (ICUAW), is observed in many, if not most, survivors of critical illness following various critical diseases. 1 , 2 , 3 , 4 , 5 Clinical consequences of muscular weakness include, for example, impaired mobilization, prolonged bed rest, and extended ICU and/or hospital length of stay. This may induce a ‘vicious cycle’ of (secondary) complications and necessitates repeated and/or intensified medical therapy, which may again result in increased morbidity and mortality. 2 , 3 , 4 , 5 , 6 Further, recent data indicate a considerable impact of muscular weakness and muscle wasting on quality of life in the years following critical illness. 7 , 8 , 9 Importantly, it should be noted that muscular weakness and/or muscle wasting imposes an important burden not only on affected individuals, but also on health care systems. 1 , 2 , 3 , 4 , 7 , 10 In an effort to improve the care for patients with ICUAW, consensus guidelines on the diagnosis 11 and research agenda 12 were recently published.
Here, we summarize the available data on muscular weakness and muscle wasting in critically ill patients in the light of current and future perspectives. Further, we will provide an outlook on other clinically relevant neuromuscular dysfunctions [including ventilator‐induced diaphragmatic dysfunction (VIDD) and dysphagia] and will discuss potential overlap.
Clinical presentation and nomenclature of muscular weakness in the critically ill
Intensive care unit‐acquired weakness
A bedside diagnosis of symmetric muscular weakness and decreased muscular tone, typically of the lower limbs, in patients with critical illness should raise the potential differential diagnosis of ICUAW 2 , 3 , 4 and is observed in at least 40% [1080/2686 patients, 95% confidence interval (CI): 38–42%] of ICU patients. 13 Lack of compliance (e.g. caused by impaired communication or need for sedation), fluid shifts confounding physiologic testing and diagnostic imaging studies, and rapid onset/progression of the underlying disease often delay establishing of the diagnosis ICUAW. Neurological examination may indicate decreased or absent deep tendon reflexes, which are not pathognomic. 14 Respiratory function may be impaired and present clinically as failure to wean from mechanical ventilation and potential overlap to VIDD should be considered in respective cases. 15
A round table conference in 2009 14 proposed new definitions and ICUAW is now typically understood as the clinical ‘umbrella term’, which embraces the following subgroups: critical illness polyneuropathy (CIP), critical illness myopathy (CIM), and their combination—critical illness polyneuromyopathy (CIPM). Importantly, for differentiation between respective subtypes, electrophysiological studies [e.g. nerve conduction studies, electromyography (EMG)] and muscle biopsies are required (also refer to Figure 1).
Figure 1.
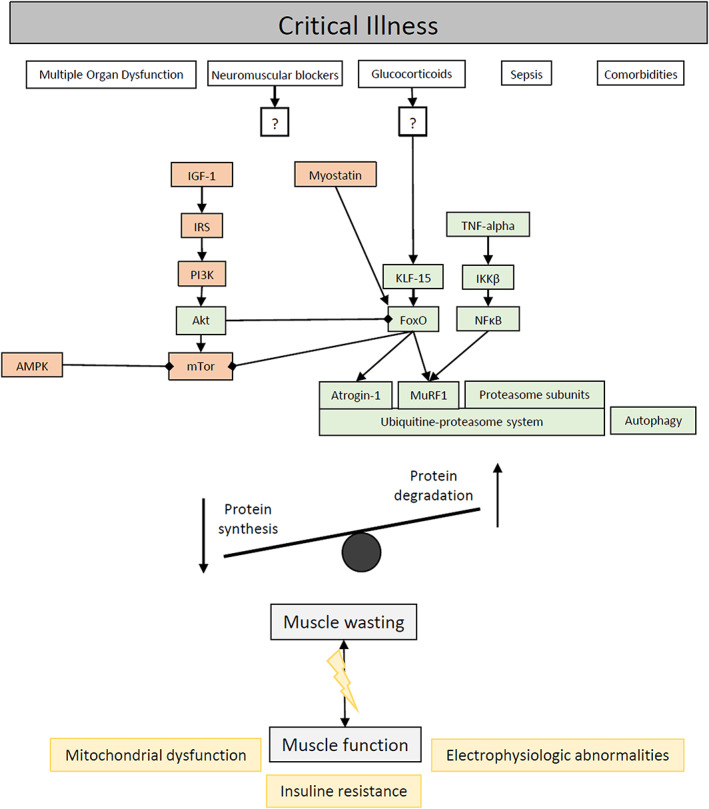
Risk factors and molecular mechanisms for muscle atrophy and muscle dysfunction in critically ill patients. Akt, Akt protein kinase B; AMPK, AMP‐activated protein kinase; IGF‐1, insulin growth factor‐1; IKKβ, inhibitor of nuclear factor kappa‐B kinase subunit beta; IRS, insulin receptor substrate; KLF‐15, Krüppel‐like factor‐15; mTor, mammalian target of rapamycin; MuRF1, muscle‐specific ring finger 1; NFκB, nuclear factor kappa‐light‐chain‐enhancer of activated B cells; PI3K, phosphoinositide 3‐kinase; TNF‐alpha, tumor necrosis factor alpha.
Critical illness polyneuropathy
Critical illness polyneuropathy is an acute and acquired polyneuropathy characterized by length‐dependent axonal damage. 2 , 3 , 4 , 16 Typically, thick myelinated sensory and motor fibres are affected, predominantly determining the clinical phenotype. For a definite diagnosis of CIP, published diagnostic criteria demand that patients fulfil the diagnostic criteria of ICUAW and show (in addition) typical electrophysiological evidence of an axonal motor and sensory polyneuropathy in the absence of a neuromuscular transmission deficit. 17 If muscle strength assessment is not available, the diagnosis of probable CIP can be based on electrophysiological findings only. A nerve biopsy is not mandatory for the respective diagnosis. Some reports from investigations of skin biopsies demonstrate that small fibres (i.e. sympathetic fibres and C‐fibres) can also undergo degeneration in CIP patients 18 , 19 and may explain why patients with CIP sometimes show typical symptoms of small fibre neuropathy in the subacute and chronic phase following critical illness. Currently, it is unknown whether occurrence of CIP and acute small fibre neuropathy is linked and/or whether these two entities share common pathophysiological mechanisms.
Critical illness myopathy
Critical illness myopathy is an acute and acquired primary myopathy. 20 , 21 , 22 , 23 , 24 Definite diagnosis of CIM is based on a multimodal approach. 17 Comparable with CIP, the diagnostic criteria of ICUAW have to be fulfilled. Additionally, electrophysiological studies consisting of nerve conduction studies and needle EMG are required. A neuromuscular transmission deficit needs to be ruled out using repetitive nerve stimulation. Ultimately, a muscle biopsy showing primary myopathy with myosin loss (and potential muscle cell necrosis) is needed for definite diagnosis; otherwise, only ‘probable’ CIM can be established. CIM often co‐exists with CIP, and this is referred to as CIPM. Differential diagnosis can be challenging and may require extensive electrophysiological investigation.
Muscle wasting in critical illness
In 1892, Sir William Osler reported a ‘rapid loss of flesh’ in patients with severe infections. 25 Today, it seems well established that muscle wasting constitutes a frequent complication in critical illness 10 and may be most prevalent in chronic critical illness (i.e. patients with prolonged ICU length of stay). However, it seems important to note that muscular wasting constitutes a separate disease entity. Whereas ICUAW is often associated with muscle wasting, muscle wasting does not per se imply the presence of a neuromuscular disorder. 3 Importantly, resulting muscle strength depends both on total muscle mass and force‐generating capacity (i.e. force per cross‐sectional area), which is, if reduced, a key feature in ICUAW but not necessarily in muscle wasting. 3 , 26
Additional clinical presentations of neuromuscular weakness on the intensive care unit
Ventilator‐induced diaphragmatic dysfunction
Ventilator‐induced diaphragmatic dysfunction is observed in up to 80% of ICUAW patients. 27 VIDD can be noted in ICU patients after prolonged controlled mechanical ventilation and is determined by a rapid loss in force‐generating diaphragmatic capacity that may also affect additional respiratory muscles (e.g. the intercostal musculature). 15 , 24 However, VIDD can be observed as early as after a few hours of mechanical ventilation in humans and VIDD intricates with, for example, self‐induced and sepsis‐associated diaphragmatic dysfunction. 15 As shown and discussed almost 20 years ago, 28 VIDD may not only be considered part of weaning failure when all other aetiologies are ruled out, but should also be considered as a cause of weaning difficulty intricated with others causes (e.g. fluid overload, atelectasis). Clinically, VIDD often presents as failure to wean from mechanical ventilation despite sustained clinical efforts, 15 and, in a first step, other underlying reasons (e.g. electrolyte abnormality or prolonged neuromuscular blockade) should be excluded. However, diaphragmatic activity can often not be assessed easily, which may hamper an early diagnosis. Clinical assessment of patients with suspected VIDD typically involves ultrasonic assessment of diaphragmatic motion (diaphragmatic dome excursion) upon spontaneous breathing trials and/or measurement of the diaphragmatic thickening fraction. 15 Further, diaphragmatic activity can be assessed by measurement of transdiaphragmatic pressures using an oesophageal probe, or via (invasive) analysis of diaphragmatic electrical activity 15 and phrenic nerve conduction studies. 29 , 30
Dysphagia
The clinical presentation of oropharyngeal dysphagia (OD) embraces drooling from the mouth, coughing following drinking/eating (and/or silent aspiration), and clinically obvious tracheal/pulmonary aspiration. 31 The neuromuscular process of swallowing is particularly complex, and exact underlying pathomechanisms leading to dysphagia in critical illness remain incompletely understood. 31 , 32 Recent data show that impaired swallowing can often be observed after mechanical ventilation in general ICU populations with up to one out of six patients with emergency admission likely affected. 33 At ICU discharge, 10.3% (n = 96/933) of mixed medical–surgical ICU survivors were reported to have confirmed dysphagia and 60.4% (n = 58/96) of affected patients remained dysphagia positive until hospital discharge. 33 Few studies are available on potential underlying risk factors in critically ill patients, and data show that risk factors for dysphagia and ICUAW may (at least partially) overlap. 34 In brief, proposed key independent risk factors for dysphagia following mechanical ventilation are baseline neurological disease [odds ratio (OR) 4.45, 95% CI: 2.74–7.24, P < 0.01], emergency admission (OR 2.04, 95% CI: 1.15–3.59, P < 0.01), days on mechanical ventilation (OR 1.19, 95% CI: 1.06–1.34, P < 0.01), days on renal replacement therapy (OR 1.1, 95% CI: 1–1.23, P = 0.03), and disease severity (Acute Physiology and Chronic Health Evaluation II score within first 24 h; OR 1.03, 95% CI: 0.99–1.07, P < 0.01). 34
Clinically, OD requires multidisciplinary efforts and should be systematically screened for in ICU patients at risk. 32 , 35 , 36 , 37 Reports demonstrate that awareness for dysphagia can be improved. 38 , 39 , 40 In patients with undetected OD, apparent or silent aspiration may prolong ICU/hospital stay, require more ICU resources (including financial resources), and may increase morbidity and mortality. 31 , 33 , 35 Further, dysphagia positivity was shown to independently predict 28‐day and 90‐day mortality (90‐day univariate hazard ratio: 3.74; 95% CI, 2.01–6.95; P < 0.001) and associated with a 9.2% excess (all‐cause) mortality rate. 33
Diagnostic approaches to generalized muscular weakness on the intensive care unit
Clinical
Peripheral muscular weakness should be quantified by use of the Medical Research Council (MRC) sum score (MRC‐SS). The MRC‐SS includes manual assessment of three functional muscle groups on both upper extremities (shoulder abduction, elbow flexion, and wrist extension) and lower extremities (hip flexion, knee extension, and ankle dorsiflexion). Muscle strength is quantified from 0 (no movement observed) to 5 (normal contraction against full resistance). ICUAW is diagnosed via an MRC‐SS < 48, which reflects an average MRC score of <4 (antigravity strength) and as it is a diagnosis of exclusion if no other plausible aetiology for the weakness other than critical illness itself is present. 3 , 11 , 41 , 42 Furthermore, dominant‐hand dynamometry results of <11 kg force for men and <7 kg force for women may be used to identify ICUAW in previously healthy individuals. 2 , 3 , 4
From a clinical perspective, a key challenge may be that the physical examination of critically ill patients on the ICU is often impeded, for example, by pre‐existing neuromuscular disease, reduced patient cooperativeness, partial sedation, prolonged neuromuscular blockade (which would typically involve cranial nerve‐innervated muscles), and/or presence of delirium, which may be particularly relevant for sensory testing as well as assessment of the MRC‐SS, and was thus shown to have a high interobserver variability. 43 , 44 , 45 Importantly, in individuals incapable of voluntary contraction, electrophysiological studies would be required for diagnosis.
However, clinicians have to bear in mind that the diagnostic criteria for ICUAW as well as the clinical findings in CIP and CIM are not specific. Respective muscular weakness can also be encountered in other diseases including Guillain–Barré syndrome, myasthenia gravis, myositis, and others. 46
Clinical findings in patients with CIP and CIM typically overlap. Deficits due to dysfunction of thick myelinated sensory nerve fibres can help to differentiate between these two entities and are only found in CIP and CIPM. In addition, one should bear in mind that deficits associated with small fibre dysfunction including temperature, pain, and sweat disturbance can be observed in CIP and CIPM. 18 Motor deficits are hallmarks of both CIP and CIM. In CIP, flaccid muscle paresis is typically symmetrical, most prominent in distal limb muscle groups, and affects the lower extremities more. Facial muscles are spared. Distal deep tendon reflexes are reduced or abolished. Clinical motor findings in CIM resemble findings in CIP and are typically symmetrical, flaccid with facial muscles spared. However, in contrast to CIP, proximal muscle groups are more often affected than distal muscles and deep tendon reflexes are reduced and only rarely abolished.
Electrophysiological testing
Typical electrophysiological studies in CIP and CIM consist of motor and sensory nerve conduction studies along with needle EMG. Repetitive stimulation of a motor nerve at 5 Hz is required to exclude neuromuscular transmission defects. In CIP, motor nerve conduction studies reveal reduced amplitudes of compound muscle action potentials (CMAPs) with normal distal motor latencies and normal nerve conduction velocities for stimulation of proximal nerve segments. F waves show normal latencies but are often missing. Sensory nerve conduction studies show reduced amplitudes of sensory action potentials or absent sensory action potentials. Sensory nerve conduction velocities are normal. Needle EMG typically shows signs of denervation with spontaneous activity and decreased recruitment.
In CIM, evoked CMAPs in motor nerve conduction studies are of low amplitudes (<80% of lower normal limit) with maybe increased duration due to a large variability in muscle fibre conduction velocities. Nerve conduction velocities and distal motor latencies are normal. Sensory nerve conduction studies are without pathological findings. Quantitative needle EMG reveals short duration and low amplitude polyphasic motor unit potentials with early or normal recruitment. Fibrillation potentials and positive sharp waves can be present. Comparison of CMAPs evoked by direct muscle stimulation and CMAPs elicited by stimulating the innervating nerve can help to distinguish between CIP from CIM. 47 Two prospective multicentre studies indicated that a simplified electrophysiological screening limited to a unilateral motor nerve conduction study of the peroneal nerve has a sensitivity/specificity of 100% and 85%, respectively, for detection of CIP and/or CIM. 48 , 49
Recently, in CIM and CIP, more complex electrophysiological techniques were applied to detect in vivo changes of muscle and nerve membrane potentials. Muscle membrane properties can be indirectly assessed by recording multifibre velocity recovery cycles. 50 In patients with probable CIM, it was inferred that muscle fibres were depolarized and/or that sodium channel inactivation was increased. 51 This is in line with earlier reports measuring absolute muscle membrane potential in critically ill patients, which showed prominent depolarization of muscle resting membrane potentials. 52 By repetitively recording muscle velocity recovery cycles in a porcine model of sepsis, it was shown that muscle membrane dysfunction can be observed within 6 h of experimental sepsis. 53 Furthermore, using the same model, development of membrane alterations correlated with applied vasopressor (norepinephrine) dose, thus likely indicating impaired microcirculation. 54 Nerve excitability testing is a non‐invasive approach to investigate the pathophysiology of peripheral nerve disorders by determining the electrical properties of the nerve membrane at the site of stimulation. In patients with established CIP, motor axons were shown depolarized. 55 Membrane depolarization was associated with raised extracellular potassium levels in patients with kidney dysfunction. Furthermore, voltage‐gated sodium channel dysfunction was shown as a characteristic feature of CIP. 21
Histology
A significant decrease in myocyte cross‐sectional area is evident as early as Day 5 after ICU admission and persists up to 6 months after ICU discharge. 56 , 57 The extent of muscle atrophy correlates with the severity of illness and ICU length of stay. 10 Data show that type II muscle fibres are mostly affected with an average rate of −4% per day during the early phase of critical illness, while fibre type distribution remains unaffected. 10 , 57 , 58 , 59 , 60 , 61 , 62 , 63 Signs for denervation atrophy (e.g. fibre type unspecific atrophy, fibre type grouping, target fibres, and atrophy of central nuclei) can be exclusively observed in CIP, but not CIM. 16 , 41 , 59 , 64 , 65 Even though muscle necrosis is described frequently and mostly in conjunction with macrophagocytosis, it should not be viewed as pathognomic for CIM/ICUAW. 10 , 60 , 66 , 67 Besides type II fibre atrophy, selective loss of myosin filaments is characteristic for CIM. 68 , 69 Few reports further indicate an accumulation of both interstitial tissue and fat. 59
Further, severe infections and sepsis are key risk factors for ICUAW. 3 Nonetheless, results regarding inflammatory muscular infiltrates of respective patients are conflicting. 57 , 67 The atrophy characteristics observed during ICUAW may overlap with histological changes induced by glucocorticoids and/or neuromuscular blockers; however, they are likely not the key trigger. 70 , 71 , 72 This seems underlined by the fact that corticosteroids were also shown to mediate partly protective effects if blood glucose levels are controlled. 73 Additionally, CIP not only manifests histologically in skeletal muscles, but also in nerve tissues, and while weakness is usually aggravated proximally, the histological manifestations can often be observed to a larger extent distally. 41 , 65 Characteristic findings include axonal degeneration and loss of myelinated fibres in peripheral motor and sensory nerves. 16 , 64 , 65 As mentioned, decreased intraepidermal nerve fibre density can also be observed in some CIP patients. 74 Further, central nervous system involvement (including chromatolysis of the anterior horn cells and loss of dorsal root ganglion) was reported in CIP. 65
Electron microscopy
The preferential loss of myosin filaments can be visualized through electron microscopy 58 , 59 , 68 , 69 , 75 (Figure 2). During the early phase of critical illness, a loss of myosin filaments with preserved ultrastructure of sarcomeres is typically observed, which is lost in later stages. 20 , 56 , 57 , 76 For survivors of critical illness, a regeneration of sarcomeric ultrastructure can be observed at about 6 months after ICU discharge. 56 Pathologic processes during critical illness affect both contractile filaments and mitochondria. In electron micrographs, swelling of mitochondria was observed early during critical illness, likely indicating mitochondrial dysfunction. 57 , 66 Moreover, reduced mitochondrial content and density is observed, which typically recovers until about 6 months after ICU discharge. 56
Figure 2.
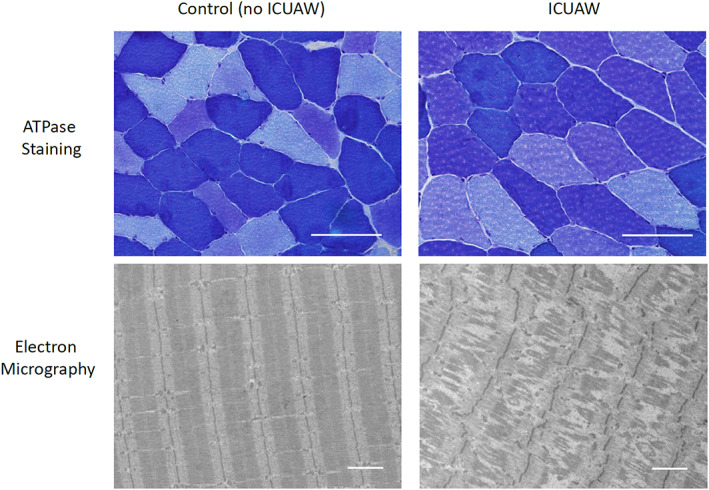
ATPase stained histologic sections and representative electron micrographs of critically ill patients with and without intensive care unit acquired weakness. Fiber types are differentiated by color (dark blue = I, intermediate blue = IIb, light blue = IIa). Scale bar indicates 100 μm (ATPase stained histologic sections) and 2 μm (electron micrographs).
The pathophysiology of muscular weakness in critical illness
Risk factors for intensive care unit‐acquired weakness
Data show that a high disease severity, number of both dysfunctional organs and of comorbidities, increases the risk for ICUAW. 23 , 41 , 77 , 78 , 79 , 80 , 81 , 82 , 83 On top of that, patients with bacteremia/sepsis seem at a particularly high risk. 3 , 77 , 79 , 80 , 81 , 83 , 84 Evidence suggests that persistent inflammation in patients with multiple organ dysfunction after acute pro‐inflammatory‐driven critical illness (e.g. sepsis, trauma) is strongly associated with end‐organ muscle inflammation, acute muscle wasting, and poor long‐term functional outcomes, 3 , 10 , 85 which may be of particular importance in chronic critical illness. 86
Although glucose levels and insulin therapy were discussed controversially, it appears that reduced serum glucose levels and higher insulin levels likely have protective effects. 23 , 82 , 84 Neuromuscular blockers were also proposed as risk factors for ICUAW. However, a recent meta‐analysis on studies with a low risk of bias identified that they likely do not increase the risk. 87 Corticosteroids were implicated early as a potential culprit in the development of ICUAW. While an association was shown by a recent meta‐analysis in the general ICU population, the findings did not extend to the subgroup of patients with sepsis. Some authors thus recommend to limit the use to low‐dose and short‐term usage in specific patient cohorts, but the discussion is overall still controversial 88 , 89 (please also refer to Figure 1).
Decreased protein synthesis
Intensive care unit‐acquired weakness‐associated muscle atrophy results partly from decreased protein synthesis. Fractional protein synthesis rates measured via leucine incorporation are depressed on the first day after ICU admission. 10 This is underlined by depressed mRNA expression levels for myosin heavy chains. 57 , 71 , 81 A major pathway involved in muscle protein synthesis is the IGF1–PI3K–Akt/PKB–mTOR pathway. 90 During critical illness, components of this pathway are considerably down‐regulated in patients with CIM, 91 while Akt is up‐regulated on both transcriptional and translational levels and phosphorylated forms are more abundant in muscle biopsy specimens, 91 which indicates that this pathway is only partially intact. Akt is physiologically able to suppress muscle protein degradation through phosphorylation of forkhead box protein (Fox)O. 92 , 93 , 94 Vice versa, FoxO can suppress muscle protein synthesis through mammalian target of rapamycin (mTOR) via sestrin and by upregulation of 4E‐BP1. 95 , 96 On a transcriptional level, we observed increased levels of mTOR and FoxO, but the mTOR increase changes to a decrease on a translational level, hinting towards a disrupted pathway at that point. 91 Furthermore, during sepsis, the eukaryotic initiation factor 4E (eIF4E) forms an inactive complex, which induces diminished translational activity. 22 AMPK (a potential factor to hamper protein synthesis) was not increased and its phosphorylated form decreased. 91 It thus seems that AMPK may therefore not be a major hindering factor for protein synthesis. Further investigations to determine exact mechanisms behind decreased protein syntheses are required.
Increased protein degradation
As outlined above, muscle atrophy is characteristic for ICUAW with a loss in myocyte cross‐sectional area accompanied by reduced protein content for slow and fast myosin on Days 5 and 15 after ICU admission. 57 The preferential myosin loss observed in histological staining and electron microscopy can also be observed on a protein level through a decreased myosin/actin ratio. 68 , 69 In fact, muscle protein degradation starts early during the course of the disease reflected by a shift in protein homeostasis towards breakdown on the first day after ICU admission, 10 which is mediated by two key systems: the ubiquitin–proteasome pathway and autophagy.
Ubiquitine–proteasome system
The ubiquitin–proteasome system plays a central role in ICUAW‐associated muscle atrophy. During early critical illness, FoxO1 and FoxO3 expression is induced. 57 , 91 While both factors were shown relevant for development of muscle atrophy, FoxO3 also directly induces atrogin‐1. 92 , 97 The induction of both members of the FoxO transcription factor family is reflected in the up‐regulation of muscle ring finger (MuRF)1 protein, atrogin‐1 mRNA expression, and respective proteins. 57 , 61 MuRF1 and atrogin‐1 are E3‐ligases that are considered to play key roles during muscle atrophy. 57 , 61 , 98 Moreover, mRNA expression of proteasome subunits and ubiquitination of respective proteins is increased 20 , 57 , 58 , 61 and increased 20S‐proteasome activity can be observed up to 6 months after ICU discharge. 56 MuRF1‐mediated muscle protein degradation was also shown to be activated by NFκB, which in turn is disinhibited through tumour necrosis factor (TNF)‐alpha via IKKβ. 99 Whereas TNF‐alpha plasma levels are typically increased at admission in critically ill patients, its mRNA expression in muscle biopsy specimens on ICU Days 5 or 15 is not increased. 100 , 101 However, it should be noted that muscle biopsy specimens only allow to evaluate one given point in time of critical illness and the expression of respective cytokines may undergo rapid changes over time. Thus, it cannot be conclusively said whether, for example, TNF‐alpha contributes to development of ICUAW‐associated muscle atrophy. 102 , 103 , 104
Glucocorticoids, whose involvement is still discussed controversially, are known to induce muscle atrophy via MuRF1 and atrogin‐1, which are again activated via KLF‐15 and FoxO. 98 , 105 , 106 Myostatin regulates skeletal muscle mass and its lack leads to muscle hypertrophy and hyperplasia. Myostatin overexpression promotes loss of muscle mass and cachexia via the ubiquitine–proteasome pathway along FoxO1, atrogin‐1, and MuRF1. 107 , 108 , 109 While induction of myostatin was suspected to be involved in ICUAW‐associated muscle atrophy, this could not be confirmed in critically ill patients. 10
Autophagy
Autophagy can be subdivided into three different mechanisms: (i) chaperon‐mediated autophagy, (ii) microautophagy, and (iii) macroautophagy (which is referred to in this passage). Autophagy is crucial for maintenance of muscle mass and integrity. 110 Thus, activation and suppression of autophagy need to be well balanced as its increased activity induces muscle atrophy, while its decreased activity was suspected to play an important role in some myopathies. 111 , 112 , 113
Critical illness can be considered a state of stress with increased damaged proteins and organelles due to, for example, oxidative damage. It was thus speculated that increased autophagy would be required for clearance of damaged proteins/organelles in an effort to maintain cellular integrity and function. Recent data show that during critical illness, insufficient activation of autophagy can be observed, which is reflected in vacuolization of both myofibers and central nuclei, as well as accumulation of p62 and ubiquitinated proteins. 114 It was subsequently shown that the extent to which autophagy could be activated in critically ill patients (as reflected by the LC3II:LC3I ratio) was protective in regard to development of muscle weakness. 81
Glucose metabolism
Hyperglycemia is common during critical illness and mostly results from both increased hepatic glucose liberation and reduced peripheral muscular glucose uptake due to insulin resistance. 115 , 116 , 117 Such insulin resistance is also reflected by a reduced insulin sensitivity index and a lack of muscular metabolic responses during hyperinsulinemic–euglycemic clamp. 91 Reduction of the insulin sensitivity index was shown pronounced in CIM patients when compared with non‐CIM patients. 91 Diminished metabolic responses to insulin are paralleled by a decreased relative mRNA expression of SLC2A4 and impaired translocation of glucose transporter (GLUT) 4 to the sarcolemmal membrane, both of which are aggravated in CIM as compared with non‐CIM patients. 91 Interestingly, the insulin receptor pathway involved in GLUT4 translocation appears intact up to the level of Akt phophorylation because an increase in phosphorylated Akt can be observed. Other downstream components of the insulin‐dependent GLUT4 translocation pathway (e.g. AS160, Rab protein) were not implicated in ICUAW so far. 118 Future investigations are necessary to elucidate the pathomechanisms behind the observed effects of insulin therapy for ICUAW.
Channelopathy
An early feature of CIM is a non‐excitable muscle membrane determined via a direct muscle stimulation CMAP of <3 mV. 119 Abnormal excitability of muscle membranes might thus result from decreased voltage‐dependent sodium channel availability due to inactivation, 51 which could be due to circulating factors (e.g. endotoxins). 51 , 120 In agreement, motor neuron sodium channel inactivation was observed in CIP. 21 Moreover, axonal membrane depolarization is observed during CIP, which is likely caused by increased extracellular potassium levels and/or hypoxia. 55
Therapeutic strategies in neuromuscular weakness of intensive care unit patients
General strategies: preventive concepts and early recognition
Evidence‐based (causal) therapies for ICUAW, CIP/CIM/CIPM, VIDD, and/ or swallowing disorders are currently unavailable, which underlines the importance of preventive strategies and/or strategies for early recognition of patients at risk. This may (partly) allow for avoidance of risk factors and may support the prevention of worsening ICUAW. In this regard, prevention and aggressive treatment of severe infections/sepsis and associated shock states should be aimed for. 1 , 2 , 3 Prevention of risk factors further embraces avoidance of prolonged bed rest, ‘over’‐analogosedation, and/or neuromuscular blockade whenever possible. In summary, preventive strategies include minimization of risk factors as well as sedation/neuromuscular blockade. Development of novel preventive and/or therapeutic strategies in affected patients may also be challenged by the acuteness and severity of the underlying (rather heterogeneous) diseases.
Early mobilization
On the ICU, multidisciplinary efforts should aim to reduce the duration of immobilization by early start of physical therapy (e.g. with in‐bed cycling 121 ). Although direct evidence is sparse, ICU length of stay, for example, can likely be reduced by such early mobilization concepts. Further, few data demonstrate that early physical therapy may improve muscular strength and ICU outcomes. 122 Despite obvious clinical benefits of early mobilization, however, in a recent Cochrane review, 123 there was insufficient evidence to prove that physiotherapeutic measures would shorten the time of rehabilitation from critical illness. Despite this, to prevent or treat VIDD, patients should be weaned from controlled mechanical ventilation as early as possible and adapted to spontaneous breathing capacity. 15 With regard to electrostimulation of the diaphragm, or of the swallowing apparatus, no direct evidence is currently available. Large‐scale clinical trials (e.g. on neuromuscular dysphagia) are currently performed. 124 In peripheral muscle groups, neuromuscular electrical stimulation was shown to preserve muscle mass and prevent muscle atrophy in critically ill patients. It was nevertheless not able to diminish the functional decline observed in ICUAW. 125 , 126
Nutritional interventions
Data indicate that an intensive insulin therapy (that would target blood glucose levels to 80–110 mg/dL) might reduce the incidence of ICUAW. Importantly, however, such intensive insulin therapy was shown to increase the number of hypoglycemic episodes and may actually worsen clinical outcomes in general ICU cohorts. 127 , 128 , 129 Thus, although euglycemia (treatment of ICU patients using insulin at blood glucose levels > 180 mg/dL) should likely be generally aimed for, an intensive insulin therapy is not advised. Further, a catabolic state is typically observed in (the early phase of) critical illness and better understanding of metabolic changes could theoretically provide novel therapeutic avenues for the treatment of ICU patients. 130 Previously, early parenteral nutrition was proposed to counteract critical illness‐induced catabolism, but recent data show that the early catabolic phase of critical illness can likely not be averted by artificial nutrition and that this may actually lead to adverse clinical outcomes, including higher ICUAW incidence. 10 Thus, although nutritional interventions appear appealing, direct evidence is missing warranting further research.
Intensive care unit‐acquired weakness‐associated complications
Prolonged bed rest in critically ill patients typically increases the risk for additional comorbidities. This includes venous thrombosis, pressure ulcers, atelectasis, and mood disorders including anxiety and depression. Prophylaxis for thrombosis and pressure ulcers, as well as symptomatic therapy of complications, is advised.
Outcome assessment
Muscle strength: Medical Research Council score
Manual muscle strength testing via the MRC score is the current guideline recommendation for diagnosing ICUAW. 11 Multiple studies showed a high interrater reliability of the MRC‐SS 6 , 43 , 44 , 45 , 131 , 132 with differences between scores of 4 and 5 contributing to most between‐rater incongruences. 43 Data on the need for periodical rater training are lacking. Further, muscle strength at ICU discharge is directly associated with mortality 5 years after discharge (hazard ratio 0.946, 95% CI: 0.928–0.968 per point increase in the MRC score, P = 0.001). 133
Hand grip strength
In line with data on manual muscle testing, hand grip strength has an acceptable interrater reliability in ICUAW. 43 , 132 , 134 Its sensitivity and specificity for ICUAW are above 80% for the cutoffs <11 kg (in men) and <7 kg (in women). 6
Muscle function: the 6‐min walking distance test
The 6‐min walking distance test (6MWD) measures muscle function and correlates both with physical function and health‐related quality of life. 135 , 136 , 137 Besides good convergent validity, the 6MWD has acceptable discriminant validity as it does not correlate with mental health‐related quality of life. 137 As changes over time may be considered more important than absolute values, the 6MWD may especially be useful during (clinical) follow‐up. 137 Limitations embrace the fact that repetitive testing and track length impact on the absolute 6MWD 135 and that many ICU patients would be unable to perform the test.
Short form health questionnaire (SF)‐36
The SF‐36 is commonly used for evaluation of health‐related quality of life after critical illness. It has sufficient reliability and validity in survivors of critical illness. 138 Nevertheless, data (specifically in ICUAW) are scarce and of low quality. 139
Long‐term disability
Investigations on long‐term outcomes from critical illness differ largely regarding the assessment tools applied, 140 resulting in considerable data heterogeneity. This limits data comparability and standardized outcome data sets are currently awaited. 141 , 142 , 143 Nevertheless, although physical function is regarded highly important after critical illness, the actual physical status and generalized fatigue may be key reasons to prevent follow‐up assessment(s) and return to work. 144 , 145 , 146 In summary, available data regarding physical performance are of poor to fair quality and further research is warranted. 139
Economic burden
Intensive care unit‐acquired weakness has a major economic impact. ICUAW was shown to prolong mechanical ventilation and ICU and hospital length of stay, and it prevents physical recovery (i.e. timely rehabilitation). 8 , 9 , 147 Despite major individual limitations, this also has considerable impact on public health care systems and on society.
Short‐term/initial hospitalization
Health care resources are largely affected by ICUAW. Early data from 1996 show that patients with neuromuscular weakness have significantly increased overall treatment costs. 148 Hermans et al. further demonstrate significantly higher treatment costs on the ICU, whereas post‐ICU (ward) costs were rather comparable with patients without ICUAW. 147 Both investigations matched patients and controls for ICUAW risk factors.
Post‐discharge/long‐term
Patients surviving critical illness face serious economic challenges most likely due to the ‘post‐intensive care syndrome’. Only about two thirds of patients that were working before critical illness return to work within a 12‐month post‐discharge period. Further, the cumulative loss in annual earnings is about 60% of previous income. 149 Five years after ICU discharge, only about 77% of patients returned to work. 8 Return to work is often hampered by depression, post‐traumatic stress, persisting muscle weakness and fatigue, and cognitive disability. 145 The negative economic impact is considerable with half of affected families making major lifestyle adjustments in order to provide care. 150 Further, after discharge of patients with ICUAW, the economic burden for society is considerable and patients are readmitted to hospitals more frequently. 145 , 151 However, data on the specific economic impact of ICUAW post‐discharge are scarce and further research is required.
Conclusions and future perspectives
In conclusion, loss of muscle mass and/or presence of muscle weakness represents a key medical challenge that affects a large number of patients on the ICU. Survivors of (prolonged) critical illness are often affected by the ‘post‐intensive care syndrome’, which is mainly characterized by long‐term physical and mental disability.
Despite the ‘classic’ presentation of symmetric peripheral weakness of limb muscles as in ICUAW, diaphragmatic muscular dysfunction and other neuromuscular dysfunctions including swallowing disorders were of major importance regarding morbidity and mortality of affected ICU patients. Importantly, evidence‐based causal therapies for ICUAW, CIP/CIM/CIPM, VIDD, and dysphagia are currently not available, which underlines the paramount importance of preventive and early diagnostic measures. Such measures include systematic screening (and thus identification) of patients at risk, avoidance of deep sedation/prolonged neuromuscular blockade, reducing length of mechanical ventilation whenever possible, promotion of early mobilization, as well as metabolic and nutritional control.
It seems that key to a better understanding of neuromuscular dysfunctions in critical illness is that there is considerable overlap regarding underlying pathomechanisms, and this understanding may open new research avenues. In the future, thorough studies investigating underlying pathomechanisms and innovative therapeutical approaches are highly warranted for critically ill patients with muscular weakness and muscle wasting.
Conflict of interest
J.C.S. reports grants from Orion Pharma, Abbott Nutrition International, B. Braun Medical AG, CSEM AG, Edwards Lifesciences Services GmbH, Kenta Biotech Ltd, Maquet Critical Care AB, Omnicare Clinical Research AG, Nestle, Pierre Fabre Pharma AG, Pfizer, Bard Medica S.A., Abbott AG, Anandic Medical Systems, Pan Gas AG Healthcare, Bracco, Hamilton Medical AG, Fresenius Kabi, Getinge Group Maquet AG, Dräger AG, Teleflex Medical GmbH, Glaxo Smith Kline, Merck Sharp and Dohme AG, Eli Lilly and Company, Baxter, Astellas, Astra Zeneca, CSL Behring, Novartis, Covidien, Phagenesis, and Nycomed outside the submitted work. The money went into departmental funds. No personal financial gain applied. All other authors declare no conflict of interest.
Acknowledgement
The authors of this manuscript certify that they comply with the ethical guidelines for authorship and publishing in the Journal of Cachexia, Sarcopenia and Muscle. 152
Schefold J. C., Wollersheim T., Grunow J. J., Luedi M. M., Z'Graggen W. J., and Weber‐Carstens S. (2020) Muscular weakness and muscle wasting in the critically ill, Journal of Cachexia, Sarcopenia and Muscle, 11, 1399–1412, 10.1002/jcsm.12620
References
- 1. Kramer CL. Intensive care unit‐acquired weakness. Neurol Clin 2017;35:723–736. [DOI] [PubMed] [Google Scholar]
- 2. Jolley SE, Bunnell AE, Hough CL. ICU‐acquired weakness. Chest 2016;150:1129–1140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Schefold JC, Bierbrauer J, Weber‐Carstens S. Intensive care unit‐acquired weakness (ICUAW) and muscle wasting in critically ill patients with severe sepsis and septic shock. J Cachexia Sarcopenia Muscle 2010;1:147–157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Hermans G, Van den Berghe G. Clinical review: intensive care unit acquired weakness. Crit Care 2015;19:274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Friedrich O, Reid MB, Van den Berghe G, Vanhorebeek I, Hermans G, Rich MM, et al. The sick and the weak: neuropathies/myopathies in the critically ill. Physiol Rev 2015;95:1025–1109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Ali NA, O'Brien JM Jr, Hoffmann SP, Phillips G, Garland A, Finley JC, et al. Acquired weakness, handgrip strength, and mortality in critically ill patients. Am J Respir Crit Care Med 2008;178:261–268. [DOI] [PubMed] [Google Scholar]
- 7. Zorowitz RD. ICU‐acquired weakness: a rehabilitation perspective of diagnosis, treatment, and functional management. Chest 2016;150:966–971. [DOI] [PubMed] [Google Scholar]
- 8. Herridge MS, Tansey CM, Matte A, Tomlinson G, Diaz‐Granados N, Cooper A, et al. Functional disability 5 years after acute respiratory distress syndrome. N Engl J Med 2011;364:1293–1304. [DOI] [PubMed] [Google Scholar]
- 9. Van Aerde N, Meersseman P, Debaveye Y, Wilmer A, Gunst J, Casaer MP, et al. Five‐year impact of ICU‐acquired neuromuscular complications: a prospective, observational study. Intensive Care Med 2020;1–10. [DOI] [PubMed] [Google Scholar]
- 10. Puthucheary ZA, Rawal J, McPhail M, Connolly B, Ratnayake G, Chan P, et al. Acute skeletal muscle wasting in critical illness. JAMA 2013;310:1591–1600. [DOI] [PubMed] [Google Scholar]
- 11. Fan E, Cheek F, Chlan L, Gosselink R, Hart N, Herridge MS, et al. An official American Thoracic Society Clinical Practice guideline: the diagnosis of intensive care unit‐acquired weakness in adults. Am J Respir Crit Care Med 2014;190:1437–1446. [DOI] [PubMed] [Google Scholar]
- 12. Latronico N, Herridge M, Hopkins RO, Angus D, Hart N, Hermans G, et al. The ICM research agenda on intensive care unit‐acquired weakness. Intensive Care Med 2017;43:1270–1281. [DOI] [PubMed] [Google Scholar]
- 13. Appleton RT, Kinsella J, Quasim T. The incidence of intensive care unit‐acquired weakness syndromes: a systematic review. J Intensive Care Soc 2015;16:126–136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Stevens RD, Marshall SA, Cornblath DR, Hoke A, Needham DM, de Jonghe B, et al. A framework for diagnosing and classifying intensive care unit‐acquired weakness. Crit Care Med 2009;37:S299–S308. [DOI] [PubMed] [Google Scholar]
- 15. Berger D, Bloechlinger S, von Haehling S, Doehner W, Takala J, Z'Graggen WJ, et al. Dysfunction of respiratory muscles in critically ill patients on the intensive care unit. J Cachexia Sarcopenia Muscle 2016;7:403–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Latronico N, Fenzi F, Recupero D, Guarneri B, Tomelleri G, Tonin P, et al. Critical illness myopathy and neuropathy. Lancet 1996;347:1579–1582. [DOI] [PubMed] [Google Scholar]
- 17. Latronico N, Bolton CF. Critical illness polyneuropathy and myopathy: a major cause of muscle weakness and paralysis. Lancet Neurol 2011;10:931–941. [DOI] [PubMed] [Google Scholar]
- 18. Latronico N, Filosto M, Fagoni N, Gheza L, Guarneri B, Todeschini A, et al. Small nerve fiber pathology in critical illness. PLoS ONE 2013;8:e75696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Skorna M, Kopacik R, Vlckova E, Adamova B, Kostalova M, Bednarik J. Small‐nerve‐fiber pathology in critical illness documented by serial skin biopsies. Muscle Nerve 2015;52:28–33. [DOI] [PubMed] [Google Scholar]
- 20. Jaber S, Petrof BJ, Jung B, Chanques G, Berthet JP, Rabuel C, et al. Rapidly progressive diaphragmatic weakness and injury during mechanical ventilation in humans. Am J Respir Crit Care Med 2011;183:364–371. [DOI] [PubMed] [Google Scholar]
- 21. Koch S, Bierbrauer J, Haas K, Wolter S, Grosskreutz J, Luft FC, et al. Critical illness polyneuropathy in ICU patients is related to reduced motor nerve excitability caused by reduced sodium permeability. Intensive Care Med Exp 2016;4:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Lang CH, Frost RA. Sepsis‐induced suppression of skeletal muscle translation initiation mediated by tumor necrosis factor alpha. Metabolism 2007;56:49–57. [DOI] [PubMed] [Google Scholar]
- 23. Van den Berghe G, Schoonheydt K, Becx P, Bruyninckx F, Wouters PJ. Insulin therapy protects the central and peripheral nervous system of intensive care patients. Neurology 2005;64:1348–1353. [DOI] [PubMed] [Google Scholar]
- 24. Larsson L, Friedrich O. Critical illness myopathy (CIM) and ventilator‐induced diaphragm muscle dysfunction (VIDD): acquired myopathies affecting contractile proteins. Compr Physiol 2016;7:105–112. [DOI] [PubMed] [Google Scholar]
- 25. Osler SW. The principles and practice of medicine, designed for the use of practitioners and students of medicine, 1st ed. Edinburgh; London: Young J. Putland; 1892. [Google Scholar]
- 26. Callahan LA, Supinski GS. Sepsis‐induced myopathy. Crit Care Med 2009;37:S354–S367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Jung B, Moury PH, Mahul M, de Jong A, Galia F, Prades A, et al. Diaphragmatic dysfunction in patients with ICU‐acquired weakness and its impact on extubation failure. Intensive Care Med 2016;42:853–861. [DOI] [PubMed] [Google Scholar]
- 28. Vassilakopoulos T, Roussos C, Zakynthinos S. Weaning from mechanical ventilation. J Crit Care 1999;14:39–62. [DOI] [PubMed] [Google Scholar]
- 29. Ackermann KA, Brander L, Tuchscherer D, Schroder R, Jakob SM, Takala J, et al. Esophageal versus surface recording of diaphragm compound muscle action potential. Muscle Nerve 2015;51:598–600. [DOI] [PubMed] [Google Scholar]
- 30. Tuchscherer D, Z'Graggen WJ, Passath C, Takala J, Sinderby C, Brander L. Neurally adjusted ventilatory assist in patients with critical illness‐associated polyneuromyopathy. Intensive Care Med 2011;37:1951–1961. [DOI] [PubMed] [Google Scholar]
- 31. Zuercher P, Moret CS, Dziewas R, Schefold JC. Dysphagia in the intensive care unit: epidemiology, mechanisms, and clinical management. Crit Care 2019;23:103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Zuercher P, Dziewas R, Schefold JC. Dysphagia in the intensive care unit: a (multidisciplinary) call to action. Intensive Care Med 2020;46:554–556. [DOI] [PubMed] [Google Scholar]
- 33. Schefold JC, Berger D, Zurcher P, Lensch M, Perren A, Jakob SM, et al. Dysphagia in mechanically ventilated ICU patients (DYnAMICS): a prospective observational trial. Crit Care Med 2017;45:2061–2069. [DOI] [PubMed] [Google Scholar]
- 34. Zuercher P, Schenk NV, Moret C, Berger D, Abegglen R, Schefold JC. Risk factors for dysphagia in ICU patients after invasive mechanical ventilation. Chest 2020. [DOI] [PubMed] [Google Scholar]
- 35. Perren A, Zurcher P, Schefold JC. Clinical approaches to assess post‐extubation dysphagia (PED) in the critically ill. Dysphagia 2019;34:475–486. [DOI] [PubMed] [Google Scholar]
- 36. Zuercher P, Dziewas R, Schefold JC. Dysphagia in critical care: focus on therapeutical interventions. Intensive Care Med 2020. [DOI] [PubMed] [Google Scholar]
- 37. Brodsky MB, Pandian V, Needham DM. Post‐extubation dysphagia: a problem needing multidisciplinary efforts. Intensive Care Med 2020;46:93–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Zuercher P, Moret C, Schefold JC. Dysphagia in the intensive care unit in Switzerland (DICE)—results of a national survey on the current standard of care. Swiss Med Wkly 2019;149:w20111. [DOI] [PubMed] [Google Scholar]
- 39. van Snippenburg W, Kroner A, Flim M, Hofhuis J, Buise M, Hemler R, et al. Awareness and management of dysphagia in Dutch intensive care units: a nationwide survey. Dysphagia 2019;34:220–228. [DOI] [PubMed] [Google Scholar]
- 40. Marian T, Dunser M, Citerio G, Kokofer A, Dziewas R. Are intensive care physicians aware of dysphagia? The MAD (ICU) survey results. Intensive Care Med 2018;44:973–975. [DOI] [PubMed] [Google Scholar]
- 41. De Jonghe B, Sharshar T, Lefaucheur JP, Authier FJ, Durand‐Zaleski I, Boussarsar M, et al. Paresis acquired in the intensive care unit: a prospective multicenter study. JAMA 2002;288:2859–2867. [DOI] [PubMed] [Google Scholar]
- 42. Kress JP, Hall JB. ICU‐acquired weakness and recovery from critical illness. N Engl J Med 2014;371:287–288. [DOI] [PubMed] [Google Scholar]
- 43. Hermans G, Clerckx B, Vanhullebusch T, Segers J, Vanpee G, Robbeets C, et al. Interobserver agreement of Medical Research Council sum‐score and handgrip strength in the intensive care unit. Muscle Nerve 2012;45:18–25. [DOI] [PubMed] [Google Scholar]
- 44. Hough CL, Lieu BK, Caldwell ES. Manual muscle strength testing of critically ill patients: feasibility and interobserver agreement. Crit Care 2011;15:R43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Connolly BA, Jones GD, Curtis AA, Murphy PB, Douiri A, Hopkinson NS, et al. Clinical predictive value of manual muscle strength testing during critical illness: an observational cohort study. Crit Care 2013;17:R229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Howard RS, Tan SV, Z'Graggen WJ. Weakness on the intensive care unit. Pract Neurol 2008;8:280–295. [DOI] [PubMed] [Google Scholar]
- 47. Rich MM, Bird SJ, Raps EC, McCluskey LF, Teener JW. Direct muscle stimulation in acute quadriplegic myopathy. Muscle Nerve 1997;20:665–673. [DOI] [PubMed] [Google Scholar]
- 48. Latronico N, Bertolini G, Guarneri B, Botteri M, Peli E, Andreoletti S, et al. Simplified electrophysiological evaluation of peripheral nerves in critically ill patients: the Italian multi‐centre CRIMYNE study. Crit Care 2007;11:R11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Latronico N, Nattino G, Guarneri B, Fagoni N, Amantini A, Bertolini G, et al. Validation of the peroneal nerve test to diagnose critical illness polyneuropathy and myopathy in the intensive care unit: the multicentre Italian CRIMYNE‐2 diagnostic accuracy study. F1000Research 2014;3:127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Z'Graggen WJ, Bostock H. Velocity recovery cycles of human muscle action potentials and their sensitivity to ischemia. Muscle Nerve 2009;39:616–626. [DOI] [PubMed] [Google Scholar]
- 51. Z'Graggen WJ, Brander L, Tuchscherer D, Scheidegger O, Takala J, Bostock H. Muscle membrane dysfunction in critical illness myopathy assessed by velocity recovery cycles. Clin Neurophysiol 2011;122:834–841. [DOI] [PubMed] [Google Scholar]
- 52. Cunningham JN Jr, Carter NW, Rector FC Jr, Seldin DW. Resting transmembrane potential difference of skeletal muscle in normal subjects and severely ill patients. J Clin Invest 1971;50:49–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Ackermann KA, Bostock H, Brander L, Schroder R, Djafarzadeh S, Tuchscherer D, et al. Early changes of muscle membrane properties in porcine faecal peritonitis. Crit Care 2014;18:484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Boerio D, Correa TD, Jakob SM, Ackermann KA, Bostock H, Z'Graggen WJ. Muscle membrane properties in a pig sepsis model: effect of norepinephrine. Muscle Nerve 2018;57:808–813. [DOI] [PubMed] [Google Scholar]
- 55. Z'Graggen WJ, Lin CS, Howard RS, Beale RJ, Bostock H. Nerve excitability changes in critical illness polyneuropathy. Brain 2006;129:2461–2470. [DOI] [PubMed] [Google Scholar]
- 56. Dos Santos C, Hussain SN, Mathur S, Picard M, Herridge M, Correa J, et al. Mechanisms of chronic muscle wasting and dysfunction after an intensive care unit stay. A pilot study. Am J Respir Crit Care Med 2016;194:821–830. [DOI] [PubMed] [Google Scholar]
- 57. Wollersheim T, Woehlecke J, Krebs M, Hamati J, Lodka D, Luther‐Schroeder A, et al. Dynamics of myosin degradation in intensive care unit‐acquired weakness during severe critical illness. Intensive Care Med 2014;40:528–538. [DOI] [PubMed] [Google Scholar]
- 58. Helliwell TR, Wilkinson A, Griffiths RD, McClelland P, Palmer TE, Bone JM. Muscle fibre atrophy in critically ill patients is associated with the loss of myosin filaments and the presence of lysosomal enzymes and ubiquitin. Neuropathol Appl Neurobiol 1998;24:507–517. [DOI] [PubMed] [Google Scholar]
- 59. Sander HW, Golden M, Danon MJ. Quadriplegic areflexic ICU illness: selective thick filament loss and normal nerve histology. Muscle Nerve 2002;26:499–505. [DOI] [PubMed] [Google Scholar]
- 60. Gutmann L, Blumenthal D, Gutmann L, Schochet SS. Acute type II myofiber atrophy in critical illness. Neurology 1996;46:819–821. [DOI] [PubMed] [Google Scholar]
- 61. Hooijman PE, Beishuizen A, Witt CC, de Waard MC, Girbes AR, Spoelstra‐de Man AM, et al. Diaphragm muscle fiber weakness and ubiquitin‐proteasome activation in critically ill patients. Am J Respir Crit Care Med 2015;191:1126–1138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Bierbrauer J, Koch S, Olbricht C, Hamati J, Lodka D, Schneider J, et al. Early type II fiber atrophy in intensive care unit patients with nonexcitable muscle membrane. Crit Care Med 2012;40:647–650. [DOI] [PubMed] [Google Scholar]
- 63. Helliwell TR, Coakley JH, Wagenmakers AJ, Griffiths RD, Campbell IT, Green CJ, et al. Necrotizing myopathy in critically‐ill patients. J Pathol 1991;164:307–314. [DOI] [PubMed] [Google Scholar]
- 64. Bolton CF, Gilbert JJ, Hahn AF, Sibbald WJ. Polyneuropathy in critically ill patients. J Neurol Neurosurg Psychiatry 1984;47:1223–1231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Zochodne DW, Bolton CF, Wells GA, Gilbert JJ, Hahn AF, Brown JD, et al. Critical illness polyneuropathy. A complication of sepsis and multiple organ failure. Brain 1987;110:819–841. [DOI] [PubMed] [Google Scholar]
- 66. Coakley JH, Nagendran K, Honavar M, Hinds CJ. Preliminary observations on the neuromuscular abnormalities in patients with organ failure and sepsis. Intensive Care Med 1993;19:323–328. [DOI] [PubMed] [Google Scholar]
- 67. De Letter MA, van Doorn PA, Savelkoul HF, Laman JD, Schmitz PI, Op de Coul AA, et al. Critical illness polyneuropathy and myopathy (CIPNM): evidence for local immune activation by cytokine‐expression in the muscle tissue. J Neuroimmunol 2000;106:206–213. [DOI] [PubMed] [Google Scholar]
- 68. Stibler H, Edstrom L, Ahlbeck K, Remahl S, Ansved T. Electrophoretic determination of the myosin/actin ratio in the diagnosis of critical illness myopathy. Intensive Care Med 2003;29:1515–1527. [DOI] [PubMed] [Google Scholar]
- 69. Ahlbeck K, Fredriksson K, Rooyackers O, Maback G, Remahl S, Ansved T, et al. Signs of critical illness polyneuropathy and myopathy can be seen early in the ICU course. Acta Anaesthesiol Scand 2009;53:717–723. [DOI] [PubMed] [Google Scholar]
- 70. Danon MJ, Carpenter S. Myopathy with thick filament (myosin) loss following prolonged paralysis with vecuronium during steroid treatment. Muscle Nerve 1991;14:1131–1139. [DOI] [PubMed] [Google Scholar]
- 71. Derde S, Hermans G, Derese I, Guiza F, Hedstrom Y, Wouters PJ, et al. Muscle atrophy and preferential loss of myosin in prolonged critically ill patients. Crit Care Med 2012;40:79–89. [DOI] [PubMed] [Google Scholar]
- 72. Khaleeli AA, Edwards RH, Gohil K, McPhail G, Rennie MJ, Round J, et al. Corticosteroid myopathy: a clinical and pathological study. Clin Endocrinol (Oxf) 1983;18:155–166. [DOI] [PubMed] [Google Scholar]
- 73. Hermans G, Wilmer A, Meersseman W, Milants I, Wouters PJ, Bobbaers H, et al. Impact of intensive insulin therapy on neuromuscular complications and ventilator dependency in the medical intensive care unit. Am J Respir Crit Care Med 2007;175:480–489. [DOI] [PubMed] [Google Scholar]
- 74. Axer H, Grimm A, Pausch C, Teschner U, Zinke J, Eisenach S, et al. The impairment of small nerve fibers in severe sepsis and septic shock. Crit Care 2016;20:64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Rathke TWT, Krebs M, Schülke M, Weber‐Carstens S. Critically ill patients with icu‐acquired weakness show reduced density of myosin in electron microscope early after onset of critical illness. Intensive Care Med Exp 2015;3:A44. [Google Scholar]
- 76. Leung TW, Wong KS, Hui AC, To KF, Lai ST, Ng WF, et al. Myopathic changes associated with severe acute respiratory syndrome: a postmortem case series. Arch Neurol 2005;62:1113–1117. [DOI] [PubMed] [Google Scholar]
- 77. Weber‐Carstens S, Deja M, Koch S, Spranger J, Bubser F, Wernecke KD, et al. Risk factors in critical illness myopathy during the early course of critical illness: a prospective observational study. Crit Care 2010;14:R119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Nanas S, Kritikos K, Angelopoulos E, Siafaka A, Tsikriki S, Poriazi M, et al. Predisposing factors for critical illness polyneuromyopathy in a multidisciplinary intensive care unit. Acta Neurol Scand 2008;118:175–181. [DOI] [PubMed] [Google Scholar]
- 79. Brunello AG, Haenggi M, Wigger O, Porta F, Takala J, Jakob SM. Usefulness of a clinical diagnosis of ICU‐acquired paresis to predict outcome in patients with SIRS and acute respiratory failure. Intensive Care Med 2010;36:66–74. [DOI] [PubMed] [Google Scholar]
- 80. de Letter MA, Schmitz PI, Visser LH, Verheul FA, Schellens RL, Op de Coul DA, et al. Risk factors for the development of polyneuropathy and myopathy in critically ill patients. Crit Care Med 2001;29:2281–2286. [DOI] [PubMed] [Google Scholar]
- 81. Hermans G, Casaer MP, Clerckx B, Guiza F, Vanhullebusch T, Derde S, et al. Effect of tolerating macronutrient deficit on the development of intensive‐care unit acquired weakness: a subanalysis of the EPaNIC trial. Lancet Respir Med 2013;1:621–629. [DOI] [PubMed] [Google Scholar]
- 82. Patel BK, Pohlman AS, Hall JB, Kress JP. Impact of early mobilization on glycemic control and ICU‐acquired weakness in critically ill patients who are mechanically ventilated. Chest 2014;146:583–589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Bednarik J, Vondracek P, Dusek L, Moravcova E, Cundrle I. Risk factors for critical illness polyneuromyopathy. J Neurol 2005;252:343–351. [DOI] [PubMed] [Google Scholar]
- 84. van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, et al. Intensive insulin therapy in critically ill patients. N Engl J Med 2001;345:1359–1367. [DOI] [PubMed] [Google Scholar]
- 85. Gardner AK, Ghita GL, Wang Z, Ozrazgat‐Baslanti T, Raymond SL, Mankowski RT, et al. The development of chronic critical illness determines physical function, quality of life, and long‐term survival among early survivors of sepsis in surgical ICUs. Crit Care Med 2019;47:566–573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Stortz JA, Mira JC, Raymond SL, Loftus TJ, Ozrazgat‐Baslanti T, Wang Z, et al. Benchmarking clinical outcomes and the immunocatabolic phenotype of chronic critical illness after sepsis in surgical intensive care unit patients. J Trauma Acute Care Surgery 2018;84:342–349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Price DR, Mikkelsen ME, Umscheid CA, Armstrong EJ. Neuromuscular blocking agents and neuromuscular dysfunction acquired in critical illness: a systematic review and meta‐analysis. Crit Care Med 2016;44:2070–2078. [DOI] [PubMed] [Google Scholar]
- 88. Yang T, Li Z, Jiang L, Wang Y, Xi X. Risk factors for intensive care unit‐acquired weakness: a systematic review and meta‐analysis. Acta Neurol Scand 2018;138:104–114. [DOI] [PubMed] [Google Scholar]
- 89. Z'Graggen WJ, Schefold JC. Critical illness myopathy: glucocorticoids revisited? Acta physiologica 2019;225:e13205. [DOI] [PubMed] [Google Scholar]
- 90. Schiaffino S, Dyar KA, Ciciliot S, Blaauw B, Sandri M. Mechanisms regulating skeletal muscle growth and atrophy. FEBS J 2013;280:4294–4314. [DOI] [PubMed] [Google Scholar]
- 91. Weber‐Carstens S, Schneider J, Wollersheim T, Assmann A, Bierbrauer J, Marg A, et al. Critical illness myopathy and GLUT4: significance of insulin and muscle contraction. Am J Respir Crit Care Med 2013;187:387–396. [DOI] [PubMed] [Google Scholar]
- 92. Sandri M, Sandri C, Gilbert A, Skurk C, Calabria E, Picard A, et al. FoxO transcription factors induce the atrophy‐related ubiquitin ligase atrogin‐1 and cause skeletal muscle atrophy. Cell 2004;117:399–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Stitt TN, Drujan D, Clarke BA, Panaro F, Timofeyva Y, Kline WO, et al. The IGF‐1/PI3K/Akt pathway prevents expression of muscle atrophy‐induced ubiquitin ligases by inhibiting FOXO transcription factors. Mol Cell 2004;14:395–403. [DOI] [PubMed] [Google Scholar]
- 94. Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS, et al. Akt promotes cell survival by phosphorylating and inhibiting a forkhead transcription factor. Cell 1999;96:857–868. [DOI] [PubMed] [Google Scholar]
- 95. Lee JH, Budanov AV, Park EJ, Birse R, Kim TE, Perkins GA, et al. Sestrin as a feedback inhibitor of TOR that prevents age‐related pathologies. Science 2010;327:1223–1228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Demontis F, Perrimon N. FOXO/4E‐BP signaling in Drosophila muscles regulates organism‐wide proteostasis during aging. Cell 2010;143:813–825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Kamei Y, Miura S, Suzuki M, Kai Y, Mizukami J, Taniguchi T, et al. Skeletal muscle FoxO1 (FKHR) transgenic mice have less skeletal muscle mass, down‐regulated type I (slow twitch/red muscle) fiber genes, and impaired glycemic control. J Biol Chem 2004;279:41114–41123. [DOI] [PubMed] [Google Scholar]
- 98. Bodine SC, Latres E, Baumhueter S, Lai VK, Nunez L, Clarke BA, et al. Identification of ubiquitin ligases required for skeletal muscle atrophy. Science 2001;294:1704–1708. [DOI] [PubMed] [Google Scholar]
- 99. Cai D, Frantz JD, Tawa NE Jr, Melendez PA, Oh BC, Lidov HG, et al. IKKbeta/NF‐kappaB activation causes severe muscle wasting in mice. Cell 2004;119:285–298. [DOI] [PubMed] [Google Scholar]
- 100. Langhans C, Weber‐Carstens S, Schmidt F, Hamati J, Kny M, Zhu X, et al. Inflammation‐induced acute phase response in skeletal muscle and critical illness myopathy. PLoS ONE 2014;9:e92048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Friedland JS, Porter JC, Daryanani S, Bland JM, Screaton NJ, Vesely MJ, et al. Plasma proinflammatory cytokine concentrations, Acute Physiology and Chronic Health Evaluation (APACHE) III scores and survival in patients in an intensive care unit. Crit Care Med 1996;24:1775–1781. [DOI] [PubMed] [Google Scholar]
- 102. Majetschak M, Krehmeier U, Bardenheuer M, Denz C, Quintel M, Voggenreiter G, et al. Extracellular ubiquitin inhibits the TNF‐alpha response to endotoxin in peripheral blood mononuclear cells and regulates endotoxin hyporesponsiveness in critical illness. Blood 2003;101:1882–1890. [DOI] [PubMed] [Google Scholar]
- 103. Majetschak M, Borgermann J, Waydhas C, Obertacke U, Nast‐Kolb D, Schade FU. Whole blood tumor necrosis factor‐alpha production and its relation to systemic concentrations of interleukin 4, interleukin 10, and transforming growth factor‐beta1 in multiply injured blunt trauma victims. Crit Care Med 2000;28:1847–1853. [DOI] [PubMed] [Google Scholar]
- 104. Oliver JC, Bland LA, Oettinger CW, Arduino MJ, McAllister SK, Aguero SM, et al. Cytokine kinetics in an in vitro whole blood model following an endotoxin challenge. Lymphokine Cytokine Res 1993;12:115–120. [PubMed] [Google Scholar]
- 105. Clarke BA, Drujan D, Willis MS, Murphy LO, Corpina RA, Burova E, et al. The E3 Ligase MuRF1 degrades myosin heavy chain protein in dexamethasone‐treated skeletal muscle. Cell Metab 2007;6:376–385. [DOI] [PubMed] [Google Scholar]
- 106. Shimizu N, Yoshikawa N, Ito N, Maruyama T, Suzuki Y, Takeda S, et al. Crosstalk between glucocorticoid receptor and nutritional sensor mTOR in skeletal muscle. Cell Metab 2011;13:170–182. [DOI] [PubMed] [Google Scholar]
- 107. McPherron AC, Lawler AM, Lee SJ. Regulation of skeletal muscle mass in mice by a new TGF‐beta superfamily member. Nature 1997;387:83–90. [DOI] [PubMed] [Google Scholar]
- 108. Zimmers TA, Davies MV, Koniaris LG, Haynes P, Esquela AF, Tomkinson KN, et al. Induction of cachexia in mice by systemically administered myostatin. Science 2002;296:1486–1488. [DOI] [PubMed] [Google Scholar]
- 109. McFarlane C, Plummer E, Thomas M, Hennebry A, Ashby M, Ling N, et al. Myostatin induces cachexia by activating the ubiquitin proteolytic system through an NF‐kappaB‐independent, FoxO1‐dependent mechanism. J Cell Physiol 2006;209:501–514. [DOI] [PubMed] [Google Scholar]
- 110. Masiero E, Agatea L, Mammucari C, Blaauw B, Loro E, Komatsu M, et al. Autophagy is required to maintain muscle mass. Cell Metab 2009;10:507–515. [DOI] [PubMed] [Google Scholar]
- 111. Mammucari C, Milan G, Romanello V, Masiero E, Rudolf R, Del Piccolo P, et al. FoxO3 controls autophagy in skeletal muscle in vivo. Cell Metab 2007;6:458–471. [DOI] [PubMed] [Google Scholar]
- 112. Zhao J, Brault JJ, Schild A, Cao P, Sandri M, Schiaffino S, et al. FoxO3 coordinately activates protein degradation by the autophagic/lysosomal and proteasomal pathways in atrophying muscle cells. Cell Metab 2007;6:472–483. [DOI] [PubMed] [Google Scholar]
- 113. Levine B, Kroemer G. Autophagy in the pathogenesis of disease. Cell 2008;132:27–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Vanhorebeek I, Gunst J, Derde S, Derese I, Boussemaere M, Guiza F, et al. Insufficient activation of autophagy allows cellular damage to accumulate in critically ill patients. J Clin Endocrinol Metab 2011;96:E633–E645. [DOI] [PubMed] [Google Scholar]
- 115. Mizock BA. Alterations in fuel metabolism in critical illness: hyperglycaemia. Best Pract Res Clin Endocrinol Metab 2001;15:533–551. [DOI] [PubMed] [Google Scholar]
- 116. Baron AD, Brechtel G, Wallace P, Edelman SV. Rates and tissue sites of non‐insulin‐ and insulin‐mediated glucose uptake in humans. Am J Physiol 1988;255:E769–E774. [DOI] [PubMed] [Google Scholar]
- 117. Zauner A, Nimmerrichter P, Anderwald C, Bischof M, Schiefermeier M, Ratheiser K, et al. Severity of insulin resistance in critically ill medical patients. Metabolism 2007;56:1–5. [DOI] [PubMed] [Google Scholar]
- 118. Huang S, Czech MP. The GLUT4 glucose transporter. Cell Metab 2007;5:237–252. [DOI] [PubMed] [Google Scholar]
- 119. Weber‐Carstens S, Koch S, Spuler S, Spies CD, Bubser F, Wernecke KD, et al. Nonexcitable muscle membrane predicts intensive care unit‐acquired paresis in mechanically ventilated, sedated patients. Crit Care Med 2009;37:2632–2637. [DOI] [PubMed] [Google Scholar]
- 120. Haeseler G, Foadi N, Wiegand E, Ahrens J, Krampfl K, Dengler R, et al. Endotoxin reduces availability of voltage‐gated human skeletal muscle sodium channels at depolarized membrane potentials. Crit Care Med 2008;36:1239–1247. [DOI] [PubMed] [Google Scholar]
- 121. Burtin C, Clerckx B, Robbeets C, Ferdinande P, Langer D, Troosters T, et al. Early exercise in critically ill patients enhances short‐term functional recovery. Crit Care Med 2009;37:2499–2505. [DOI] [PubMed] [Google Scholar]
- 122. Morris PE, Goad A, Thompson C, Taylor K, Harry B, Passmore L, et al. Early intensive care unit mobility therapy in the treatment of acute respiratory failure. Crit Care Med 2008;36:2238–2243. [DOI] [PubMed] [Google Scholar]
- 123. Connolly B, Salisbury L, O'Neill B, Geneen L, Douiri A, Grocott MP, et al. Exercise rehabilitation following intensive care unit discharge for recovery from critical illness: executive summary of a Cochrane Collaboration systematic review. J Cachexia Sarcopenia Muscle 2016;7:520–526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. https://clinicaltrials.gov/ct2/show/NCT03840395: (assessed Feb 18, 2020).
- 125. Wollersheim T, Grunow JJ, Carbon NM, Haas K, Malleike J, Ramme SF, et al. Muscle wasting and function after muscle activation and early protocol‐based physiotherapy: an explorative trial. J Cachexia Sarcopenia Muscle 2019;10:734–747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Fossat G, Baudin F, Courtes L, Bobet S, Dupont A, Bretagnol A, et al. Effect of in‐bed leg cycling and electrical stimulation of the quadriceps on global muscle strength in critically ill adults: a randomized clinical trial. JAMA 2018;320:368–378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Hermans G, De Jonghe B, Bruyninckx F, Van den Berghe G. Interventions for preventing critical illness polyneuropathy and critical illness myopathy. Cochrane Database Syst Rev 2014;1:CD006832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Investigators N‐SS , Finfer S, Chittock DR, Su SY, Blair D, Foster D, et al. Intensive versus conventional glucose control in critically ill patients. N Engl J Med 2009;360:1283–1297. [DOI] [PubMed] [Google Scholar]
- 129. Griesdale DE, de Souza RJ, van Dam RM, Heyland DK, Cook DJ, Malhotra A, et al. Intensive insulin therapy and mortality among critically ill patients: a meta‐analysis including NICE‐SUGAR study data. CMAJ 2009;180:821–827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. van Gassel RJJ, Baggerman MR, van de Poll MCG. Metabolic aspects of muscle wasting during critical illness. Curr Opin Clin Nutr Metab Care 2020;23:96–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Fan E, Ciesla ND, Truong AD, Bhoopathi V, Zeger SL, Needham DM. Inter‐rater reliability of manual muscle strength testing in ICU survivors and simulated patients. Intensive Care Med 2010;36:1038–1043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Parry SM, Berney S, Granger CL, Dunlop DL, Murphy L, El‐Ansary D, et al. A new two‐tier strength assessment approach to the diagnosis of weakness in intensive care: an observational study. Crit Care 2015;19:52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Van Aerde N, Meersseman P, Debaveye Y, Wilmer A, Gunst J, Casaer MP, et al. Five‐year impact of ICU‐acquired neuromuscular complications: a prospective, observational study. Intensive Care Med 2020;46:1184–1193. [DOI] [PubMed] [Google Scholar]
- 134. Vanpee G, Segers J, Van Mechelen H, Wouters P, Van den Berghe G, Hermans G, et al. The interobserver agreement of handheld dynamometry for muscle strength assessment in critically ill patients. Crit Care Med 2011;39:1929–1934. [DOI] [PubMed] [Google Scholar]
- 135. Alison JA, Kenny P, King MT, McKinley S, Aitken LM, Leslie GD, et al. Repeatability of the six‐minute walk test and relation to physical function in survivors of a critical illness. Phys Ther 2012;92:1556–1563. [DOI] [PubMed] [Google Scholar]
- 136. Denehy L, Nordon‐Craft A, Edbrooke L, Malone D, Berney S, Schenkman M, et al. Outcome measures report different aspects of patient function three months following critical care. Intensive Care Med 2014;40:1862–1869. [DOI] [PubMed] [Google Scholar]
- 137. Chan KS, Pfoh ER, Denehy L, Elliott D, Holland AE, Dinglas VD, et al. Construct validity and minimal important difference of 6‐minute walk distance in survivors of acute respiratory failure. Chest 2015;147:1316–1326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Chrispin PS, Scotton H, Rogers J, Lloyd D, Ridley SA. Short Form 36 in the intensive care unit: assessment of acceptability, reliability and validity of the questionnaire. Anaesthesia 1997;52:15–23. [DOI] [PubMed] [Google Scholar]
- 139. Robinson KA, Davis WE, Dinglas VD, Mendez‐Tellez PA, Rabiee A, Sukrithan V, et al. A systematic review finds limited data on measurement properties of instruments measuring outcomes in adult intensive care unit survivors. J Clin Epidemiol 2017;82:37–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Turnbull AE, Rabiee A, Davis WE, Nasser MF, Venna VR, Lolitha R, et al. Outcome measurement in ICU survivorship research from 1970 to 2013: a scoping review of 425 publications. Crit Care Med 2016;44:1267–1277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Connolly B, Denehy L, Hart N, Pattison N, Williamson P, Blackwood B. Physical rehabilitation core outcomes in critical illness (PRACTICE): protocol for development of a core outcome set. Trials 2018;19:294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Dinglas VD, Chessare CM, Davis WE, Parker A, Friedman LA, Colantuoni E, et al. Perspectives of survivors, families and researchers on key outcomes for research in acute respiratory failure. Thorax 2018;73:7–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Needham DM, Sepulveda KA, Dinglas VD, Chessare CM, Friedman LA, Bingham CO 3rd, et al. Core outcome measures for clinical research in acute respiratory failure survivors. An International Modified Delphi Consensus Study. Am J Respir Crit Care Med 2017;196:1122–1130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Herridge MS, Cheung AM, Tansey CM, Matte‐Martyn A, Diaz‐Granados N, Al‐Saidi F, et al. One‐year outcomes in survivors of the acute respiratory distress syndrome. N Engl J Med 2003;348:683–693. [DOI] [PubMed] [Google Scholar]
- 145. Cheung AM, Tansey CM, Tomlinson G, Diaz‐Granados N, Matte A, Barr A, et al. Two‐year outcomes, health care use, and costs of survivors of acute respiratory distress syndrome. Am J Respir Crit Care Med 2006;174:538–544. [DOI] [PubMed] [Google Scholar]
- 146. Abshire M, Dinglas VD, Cajita MI, Eakin MN, Needham DM, Himmelfarb CD. Participant retention practices in longitudinal clinical research studies with high retention rates. BMC Med Res Methodol 2017;17:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Hermans G, Van Mechelen H, Clerckx B, Vanhullebusch T, Mesotten D, Wilmer A, et al. Acute outcomes and 1‐year mortality of intensive care unit‐acquired weakness. A cohort study and propensity‐matched analysis. Am J Respir Crit Care Med 2014;190:410–420. [DOI] [PubMed] [Google Scholar]
- 148. Rudis MI, Guslits BJ, Peterson EL, Hathaway SJ, Angus E, Beis S, et al. Economic impact of prolonged motor weakness complicating neuromuscular blockade in the intensive care unit. Crit Care Med 1996;24:1749–1756. [DOI] [PubMed] [Google Scholar]
- 149. Kamdar BB, Huang M, Dinglas VD, Colantuoni E, von Wachter TM, Hopkins RO, et al. Joblessness and lost earnings after acute respiratory distress syndrome in a 1‐year national multicenter study. Am J Respir Crit Care Med 2017;196:1012–1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Griffiths J, Hatch RA, Bishop J, Morgan K, Jenkinson C, Cuthbertson BH, et al. An exploration of social and economic outcome and associated health‐related quality of life after critical illness in general intensive care unit survivors: a 12‐month follow‐up study. Crit Care 2013;17:R100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Ruhl AP, Lord RK, Panek JA, Colantuoni E, Sepulveda KA, Chong A, et al. Health care resource use and costs of two‐year survivors of acute lung injury. An observational cohort study. Ann Am Thorac Soc 2015;12:392–401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. von Haehling S, Morley JE, Coats AJS, Anker SD. Ethical guidelines for publishing in the Journal of Cachexia, Sarcopenia and Muscle: update 2019. J Cachexia Sarcopenia Muscle 2019;10:1143–1145. [DOI] [PMC free article] [PubMed] [Google Scholar]


