Abstract
The present study was aimed to exploit the haloarchaeon Haloferax alexandrinus GUSF-1 (KF796625) for the presence of biomolecules possessing antioxidant activity. The culture produced a bright orange pigment when grown aerobically in nutrient rich medium with 25% crude solar salt. Biomolecules from cell-free supernatant and from the cells of the culture were individually extracted through the assistance of solvents of different polarities, such as ethanol, methanol and hexane, and monitored for scavenging of stable free radicals. Each of the extracts showed varying capacities to scavenge DPPH•(20, 31, and 80% DPPH• RSA; 160.19, 248.29 and 640.76 AAE µg g−1 of cells) at 1 mg mL−1. The extracellular ethanolic extract was polysaccharide in nature, equivalent to 47 µg mL−1 of glucose when assayed with the phenol-sulfuric acid method. The Fourier Transform-Infra Red spectroscopy confirmed the characteristic glycosidic peaks between 2000 and 1000 cm−1. Similarly, the glycerol diether moiety separated from hydroxylated methanolysates through thin-layer chromatography scavenged free radicals (10.47% DPPH• RSA; 80.03 AAE µg g−1 of cells). Further, the hexanolic extract exhibited spectral characteristics of red carotenoids and resolved into distinct compounds when separated by thin-layer chromatography using different developing systems. All separated compounds were positive for the DPPH• reaction (13–30% DPPH• RSA; 100–240 AAE µg g−1). Chemical profiling of the hexanolic extract using the high resolution-liquid chromatography–mass spectroscopy–diode array detector analysis confirmed the presence of different carbon length isoprenoids; C30: tetrahydrosqualene, C40: 3-hydroxyechinenone, astaxanthin, canthaxanthin, lycopene, phytofluene, phytoene and C50: bisanhydrobacterioruberin, monoanhydrobacterioruberin, bacterioruberin and haloxanthin. Thus, we conclude that the synergistic actions of all these components contribute to the antioxidant activity of the culture and that the antioxidant activity of the exopolysaccharide, glycerol dither moiety, tetrahydrosqualene, haloxanthin and 3-hydroxyechinenone is recorded as the first report for Haloferax alexandrinus GUSF-1 (KF796625). Therefore, recommended for use in microbial industrial biotechnology.
Supplementary Information
The online version contains supplementary material available at 10.1007/s13205-020-02584-9.
Keywords: Carotenoids, DPPH•, Exopolysaccharides, Glycerol diether moiety, Haloarchaea, HR-LC/MS–DAD
Introduction
Free radicals from atoms, molecules, or ions with highly unstable unpaired electrons are reported to be produced by oxidative reactions from biota and the environment (Lu et al. 2010). The balance between free radicals and oxidative stress caused by them in a living system is maintained by antioxidants (Wan et al. 2016; Alvares and Furtado 2018). Nutraceuticals, cosmetics, and food industries seek natural antioxidants, because they harbor phenolic components that scavenge free radicals (Utkina et al. 2004; Alvares and Furtado 2018). The antioxidant potential of marine halophilic eubacterial bionts isolated from several dominant marine invertebrates (sponges, corals, and bivalves) and haloarchaeal bionts are reported by (Velho Pereira and Furtado 2014; Velho Pereira et al. 2015), but their presence in Haloarchaeal cultures from solar salt is relatively less explored.
Reports of multiple biotechnological potentials in Haloarchaea, which belong to the third domain of life, are available (Singh and Singh 2017). Besides, they are known to degrade organic pollutants (Raghavan and Furtado 2000), mineralize manganese (Naik and Furtado 2019), produce extremozymes (Gaonkar and Furtado 2018; Malik and Furtado 2019), and synthesize nanoparticles (Patil et al. 2014).
Haloferax alexandrinus GUSF-1 (KF796625) retrieved from solar salt of Goa, India was selected because it biosynthesized silver nanoparticles (Patil et al. 2014), produced rhodochrosite (Naik and Furtado 2019) and its ability to scavenge free radicals (Alvares and Furtado 2018), all of significance, in the pharmaceutical and biotechnological industry.
This present study, therefore, is a record of the: (1) extraction of the biomolecules from the cells of Haloferax alexandrinus GUSF-1 (KF796625) assisted by the use of solvents such as ethanol, methanol and hexane; (2) the ability of the extracts to scavenge stable DPPH• free radicals and (3) the use of TLC, UV–visible, FTIR spectroscopy and HR-LC/MS–DAD for separation, identification and chemical profiling of the biomolecules responsible for the antioxidant activity of Haloferax alexandrinus GUSF-1 (KF796625).
Materials and methods
Chemicals and reagents
All chemicals and reagents used were of analytical grade. 1, 1-diphenyl-2-picrylhydrazyl (DPPH•) was acquired from Sigma-Aldrich and prepared in methanol (purity 97%)/hexane (purity > 95%) as required. Thin-layer chromatography (TLC) sheets (silica gel GF254 plates) used in this study were from Merck, Darmstadt, Germany.
Growth of Haloferax alexandrinus GUSF-1 (KF796625)
Haloferax alexandrinus GUSF-1 (GenBank accession number KF796625), a haloarchaeon, isolated from natural solar salt by Sequeira (1992), was used in this study. Cells were cultured in 5 L of Tryptone Yeast Extract containing 25% crude solar salt (NTYE) (Raghavan and Furtado 2000) at 42 °C and 150 rpm for 6 days (REMI CIS-24 PLUS, India), resulting in a wet cell mass of 107 g. Separation of cell mass from culture broth was achieved using a high-speed centrifuge at 12,857×g and 4 °C (Biofuge-Heraeus Stratos). The supernatant obtained was passed through a 0.2 μm membrane filter (MF-Millipore™) to give the cell-free supernatant (CFS).
Preparation of ethanolic extract from CFS, hydroxylated methanolysate and hexanolic extract
Cell-free supernatant was subjected to extraction by the Del Gallo and Haegi (1990) method using 96% cold ethanol. After addition, the mixture was kept standing overnight at -20 °C and centrifuged. The pellet obtained was dispersed in sterile distilled water and checked for the presence of total carbohydrate using the phenol–sulfuric acid method, according to Dubois et al. (1956) with glucose as the standard.
Hydroxylated methanolysate of cells was prepared by the Ross et al. (1981) method. One gram of cells was mixed with methanol:toluene:concentrated H2SO4 (3:3:1 v/v), heated at 50 °C in a water bath for 18 h, and extraction of the hydroxylated methanolysate was accomplished by adding hexane.
To the remaining cells, methanol was added in minimum light exposure and kept overnight at 4 °C. The cell debris were then separated by centrifuging to obtain a clear cell-free methanolic extract of cells. Components from this were fractionated with hexane and distilled water according to the method of Asker et al. (2002) to obtain the hexanolic extract. The ethanolic extract, hydroxylated methanolysates and the phase fractionated hexanolic extract were concentrated under N2 gas, wrapped in aluminium foil to avoid light exposure and stored at −20 °C.
Detection of free radical scavenging activity (RSA) of the ethanolic extract, hydroxylated methanolysate and hexanolic extract
Individual extracts at 1 mg mL−1 were checked for RSA by the method of Alvares and Furtado (2018). Each was mixed with 1 mL of 0.2 mM DPPH• reagent (prepared in hexane or methanol as required; Supplementary Fig. 1) kept in the dark at room temperature (28 ± 2 °C), for 30 min and the absorbance checked at 517 nm, against hexane/methanol as the reference solution.
The % radical scavenging activity was calculated using Eq. 1;
| 1 |
where AB is the absorbance of 0.2 mM DPPH• in the respective solvent (hexane or methanol). AS is the absorbance of the reaction mixture (DPPH• + extract). Ascorbic acid was used as the standard (R2 = 0.9835) and results were also expressed as Ascorbic Acid Equivalent (AAE) µg g−1 of cells. The individual reaction mixtures were then scanned between 190 and 800 nm after 30 min of incubation.
Characterization of ethanolic extract, hydroxylated methanolysate and hexanolic extract
UV–Vis spectroscopy
The ethanolic and the fractionated hexanolic extracts were individually scanned between 190 and 800 nm using a UV–visible spectrophotometer (UV-1601, Shimadzu, Kyoto, Japan) and against distilled water/hexane as the reference solution, as required.
TLC analysis
Components in the hydroxylated methanolysate were chromatographed on four different TLC sheets and resolved separately in the developing system consisting of petroleum ether (60–80 °C):diethyl ether (85:15, v/v). The first chromatogram was placed in an iodine chamber to check for colorless compounds (Kushwaha et al. 1974). The second was sprayed with 10% dodecaphosphomolybdic acid and heated at 150 °C for 15 min (Ross et al. 1981). Detection of free radical scavenging activity was carried out by spraying the third chromatogram with 0.2 mM DPPH• and incubating in the dark. The fourth chromatogram was left untreated.
Similarly, components in the hexanolic extract were chromatographed and resolved using different developing systems: acetone:petroleum ether (60–80 °C) (20:80, v/v), methanol:chloroform (7:93, v/v) (Asker et al. 2002), acetone:hexane (50:50, v/v) (Ronnekleiv and Liaaen-Jensen 1995), and n-heptane:benzene (90:10, v/v) (Tornabene et al. 1969), respectively.
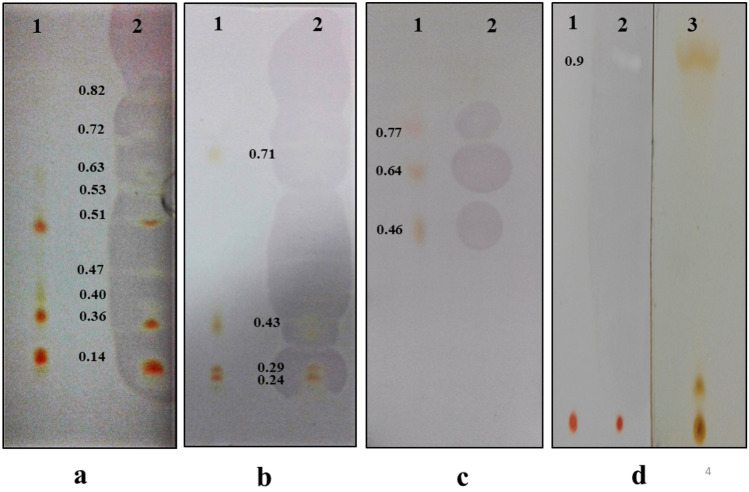
In each case, sets of three different TLC sheets were used. In each set, spotting on a single chromatogram was done at a distance of 1.5 cm apart. After resolving in respective developing system, all the chromatograms were first air-dried at room temperature in the dark. Visible compounds, if any that were separated, were noted. The chromatogram developed in n-heptane:benzene (90:10, v/v) was visualized with iodine vapors. In each case, the detection of free radical scavenging activity was carried out by spraying the second set of chromatograms with 0.2 mM DPPH•. The third set was left untreated. The retention factor, Rf, of the resolved spots that decolorized the purple DPPH• was recorded and compared with that reported in literature using identical developing systems.
Select spots from individual chromatograms that were positive for the DPPH• activity, were scrapped off from the respective resolved untreated chromatogram. Under minimum light, each compound was individually eluted out in the respective solvent, concentrated, and weighed. Each elute was checked for their RSA and UV–Vis spectral characteristics.
Quantification of the purified colored compound was carried out using Eq. 2 (Kushwaha et al. 1974) and as reported by Raghavan and Furtado (2005):
| 2 |
where A is the absorbance at λmax, Y= volume in mL (in this case, 1 mL cuvette was used). E1% = is the extinction coefficient specific for each separated compounds (Britton 1995). C50 carotenoids—2450 at λ490 (Kushwaha et al. 1974); Canthaxanthin—2092 at λ476 (Warren and Weedon 1958); 3-hydroxyechinenone—2100 at λ476 (An et al. 1989).
Fourier Transform Infrared (FTIR) spectroscopy
The ethanolic extract, the compound resolved on silica plate at Rf 0.2 from the hydroxylated methanolysate and hexanolic extract were individually mixed with KBr to form a pellet. Samples were then analyzed in the 4000 and 400 cm−1 spectral region at a resolution of 4.0 cm−1 using the FTIR spectrometer (Shimadzu IR Prestige-21).
High resolution-liquid chromatography–mass spectroscopy–diode array detector (HR-LC/MS–DAD) analysis
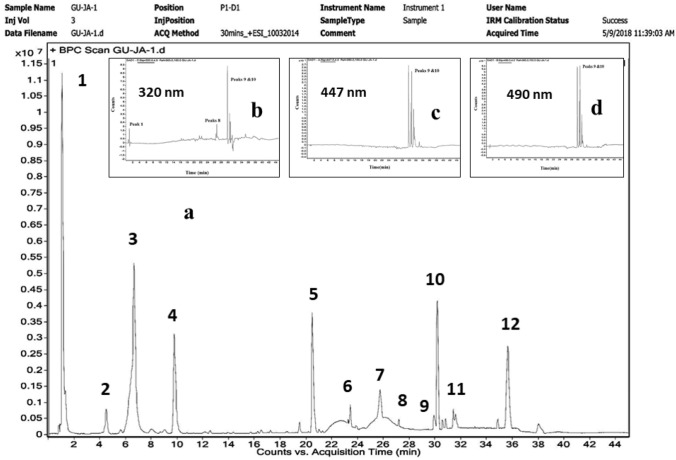
Chemical profiling of the constituents in hexanolic extract was achieved using a HR-LC/MS–DAD instrument; Agilent 1290 Infinity UHPLC system, 1260 Infinity Nano HPLC with Chipcube series (Agilent Technologies, USA) and a hypersil gold 3 microns 100 × 2.1 mm C-18 reverse-phase column from Agilent Technologies. The ESI interface consisted of a binary pump, an autosampler and 6550 i funnel QTOF-LC/MS. The two mobile phases used were: (A) 0.1% (v/v) formic acid in water and (B) 70% acetonitrile in 30% (v/v) methanol. The flow rate was adjusted for 0.3 mL/min at 25 °C. The UV–Vis spectrum was obtained between 190 and 600 nm and the chromatograms were processed at 320, 447 and 490 nm, respectively. Separated compounds were identified by comparing the obtained m/z values with the Agilent Mass Hunter Qualitative Analysis Software version (B.05.00), Japanese carotenoid database, Pubchem and earlier reported literature.
Results and discussion
Cells of Haloferax alexandrinus GUSF-1 (KF796625)
Haloferax alexandrinus GUSF-1 (KF796625) produced a bright orange pigment when grown aerobically in 25% NTYE broth and a cell mass of 107 g was recovered from the fully grown culture (Supplementary Fig. 2). The colonies of the culture have been reported to scavenge free radicals by the agar growth method in a previous study (Alvares and Furtado 2018).
Characterization of ethanolic extract
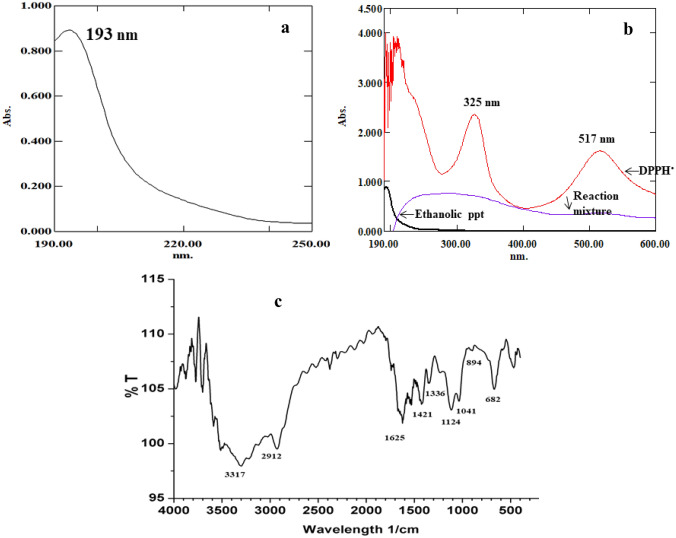
Ethanolic extracts from the cell-free supernatant resulted in a white precipitate soluble in distilled water and exhibited a value of 20 ± 0.4% DPPH• RSA (160.19 AAE µg g−1 of cells). The UV spectrum of this precipitate showed a single peak at 193 nm (Fig. 1a). The spectrum of the reaction mixture revealed that both peaks of the pure DPPH• radical seen at 517 and 325 nm were abolished along with the single peak at 193 nm during the decolorization process suggesting its involvement (Fig. 1b). In the FTIR spectrum, absorption peaks were seen between 3900 and 500 cm−1 (Fig. 1c). Characteristic peaks were also seen at 3317 cm−1 for the OH group, at 2912 cm−1 indicating the presence of the C-H group and at 1625 cm−1 for the carbonyl group. A broad stretch at 1041 cm−1 followed by an intense peak at 1124 cm−1 is indicative of the C–OH side groups and the C–O–C glycosidic band vibrations (Zhang et al. 2011). The ethanolic extract tested positive for carbohydrates with 47 mg mL−1 equivalent of glucose, hence, polysaccharide in nature (Supplementary Fig. 3).
Fig. 1.
Characterization of ethanolic extract from Haloferax alexandrinus GUSF-1 (KF796625). a UV spectrum showing a single peak at 193 nm, b DPPH• decolorization of ethanolic extract monitored by the spectrophotometer; which shows that both the peaks of DPPH• (325 and 517 nm) are abolished by the ethanolic extract, indicating the free radical scavenging capacity, c FTIR spectrum revealing the characteristic glycosidic peaks between 2000 and 1000 cm−1
Little is reported about exopolysaccharide (EPS) production from haloarchaea with Antón et al. (1988), first reporting the production of EPS by the Haloferax mediterranei (ATCC 33500). Antioxidant activity of EPS has been reported in the Haloterrigena turkmenica by Squillaci et al. (2016), at a value three times lower than our activity, although assayed at the same concentration.
Characterization of hydroxylated methanolysate
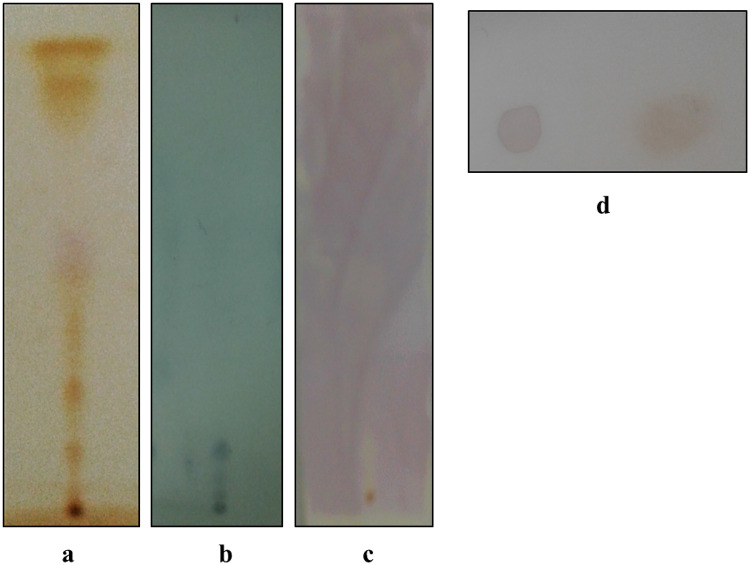
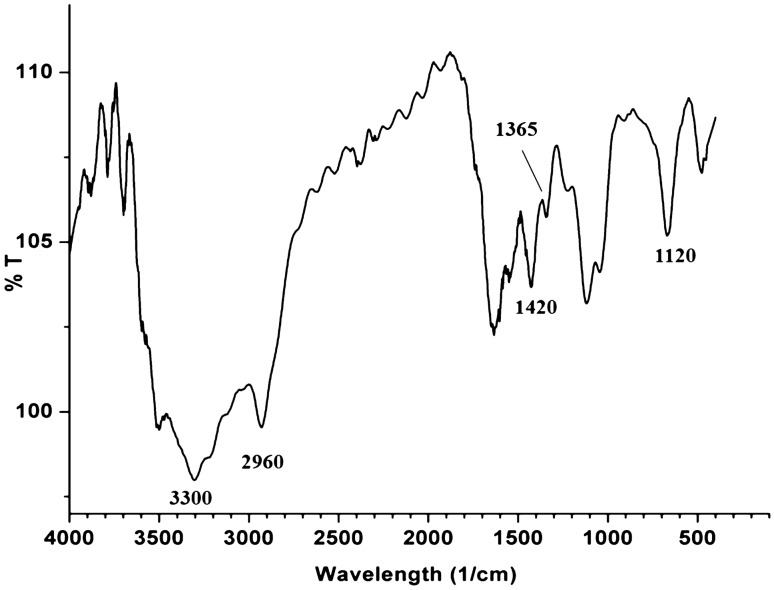
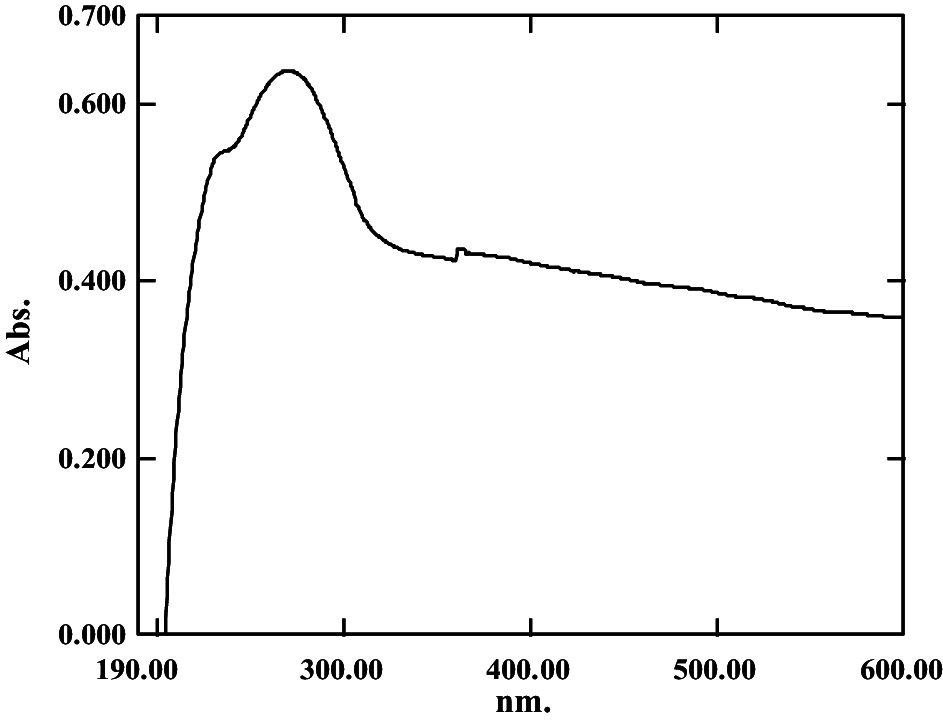
The hydroxylated methanolysate extract was pale yellow and of oily consistency and gave a value of 31 ± 0.24% DPPH• RSA (248.29 AAE µg g−1 of cells). The chromatogram revealed the presence of 6 spots in iodine chamber at Rf 0.2, 0.3, 0.48, 0.59, 0.78 and 0.89, respectively (Fig. 2a). On spraying with 10% dodecaphosphomolybdic acid, a blue spot was visualized at Rf 0.2 (Fig. 2b). Interestingly, the compound at Rf 0.2 decolorized DPPH• on the chromatogram and on the spot test (Fig. 2c, d) and the RSA of the compound after elution in hexane, gave a value of 10.47 ± 0.21% DPPH• RSA (80.03 ± 0.32 AAE µg g−1 of cells) (Table 1). These results also confirmed our earlier report (Alvares and Furtado 2018). The UV spectrum exhibited absorption maxima at 256, 263, and 270 nm (Table 1). Notable in the FTIR spectra was the complete absence of ester absorption peaks between 1730 and 1750 cm−1. However, the spectra did show strong absorption bands indicative of long-chain groups (2960 cm−1), OH groups (3300 cm−1), ether C–O–C groups (1120 cm−1) and the isopropyl units between 1365 and 1420 (Fig. 3). This spectrum matched with that reported for the purified glycerol diether moiety from Halobacterium cutirubrum NCMB 763 confirming the identity as glycerol diether moiety (GDEM), a compound that is unique to haloarchaea and which distinguishes it from the eubacterial domain (Ross et al. 1981,1985). Furthermore, the result was also consistent with reports by Braganca and Furtado (2009) and Gaonkar and Furtado (2018). Though GDEM is reported to protect Archaea and haloarchaea against hydrolysis of their membrane phospholipids, their role as scavengers of DPPH• observed by us in this study is the first record.
Fig. 2.
Characterization of hydroxylated methanolysate from cells of Haloferax alexandrinus GUSF-1 (KF796625): The thin-layer chromatogram was resolved in petroleum ether (40–60 °C): diethyl ether (85:15,v/v) and developed with a iodine vapor, b 10% dodecaphosphomolybdic acid in absolute ethanol and heated at 150 °C, c decolorization of hexanolic DPPH• solution by compound at Rf 0.2 and d DPPH• spot assay
Table 1.
Characterization and quantification of glycerol diether moiety extracted from cells of Haloferax alexandrinus GUSF-1 (KF796625) and separated using thin-layer chromatography
| Solvent system | Rf of compound with antioxidant activity | DPPH• reaction | Quantity obtained after TLC separation (mg 100 g−1 cells) | % DPPH• RSA; AAE µg g−1 of cells (mean ± SD) | UV–visible absorption spectrum | UV–visible absorption spectral peaks (nm) | Identification |
|---|---|---|---|---|---|---|---|
| Petroleum ether(60–80 °C): Diethyl ether (85:15, v/v) | 0.2 | + | 6.66 | 10.47 ± 0.21; 80.03 ± 0.32 |
|
256, 263, 270 | Glycerol diether moiety (GDEM) |
Fig. 3.
FTIR spectrum of glycerol diether moiety extracted from hydroxylated methanolysate from cells of Haloferax alexandrinus GUSF-1 (KF796625)
Characterization of hexanolic extract
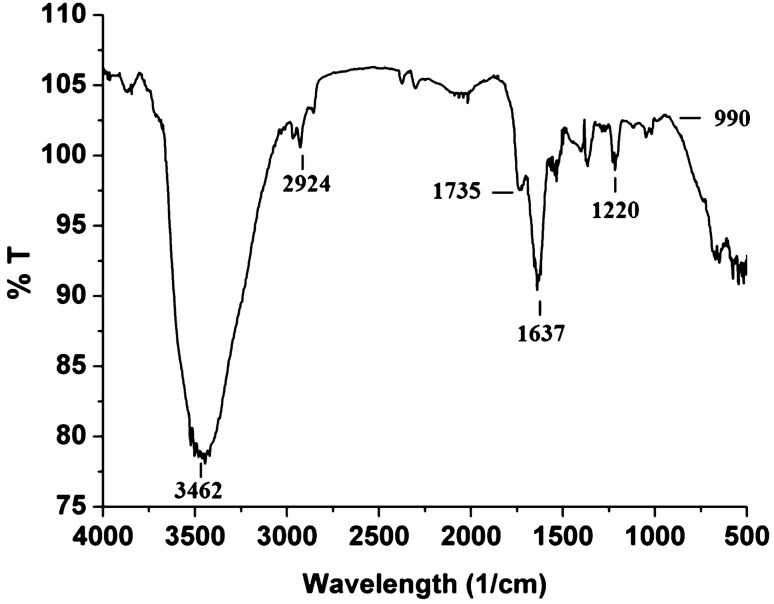
The hexanolic extract was bright orange, with absorption peaks both in the UV (190–300 nm) and the visible (300–700 nm) range (Fig. 4a, b). These peaks corroborated with red carotenoids and their derivatives, as reported earlier for the culture (Alvares and Furtado 2018) and also reported for Haloarchaea by Britton et al. (1995). The hexanolic extract decolorized the deep purple-colored stable DPPH• to straw yellow and showed a highly potent value of 80 ± 0.32% DPPH• RSA (640.76 AAE µg g−1) (Fig. 4c). The FTIR spectral analysis revealed intense absorption peaks at 3462 cm−1 and stretching vibrations were seen at 2924 cm−1, while absorption bands were observed at 1735, 1637, 1220, and 990 cm−1 (Fig. 5). The constituents of the hexanolic extract resolved by TLC were found to be all positive for the DPPH• reaction with values in the range of 13–30% DPPH• RSA; 100–240 AAE µg g−1. The TLC profiles, spectroscopic characteristics and quantification data obtained using the individual elutes' absorption coefficients are recorded in Fig. 6 and Tables 2 and 3.
Fig. 4.
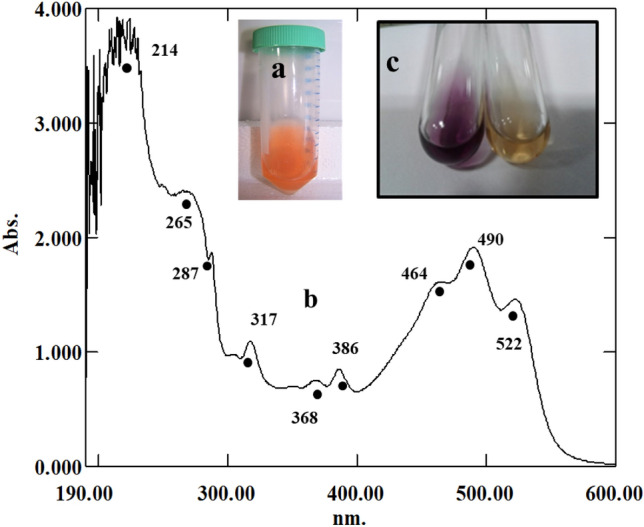
Decolorization of DPPH• by hexanolic extract from cells of Haloferax alexandrinus GUSF-1 (KF796625). a Bright orange hexanolic extract, b UV–Vis absorption spectrum of hexanolic extract with peaks in the UV and visible region and c the deep purple color of DPPH• (left) is decolorized by the hexanolic extract rendering it straw yellow (right), an indication of its potent free radical scavenging capacity
Fig. 5.
FTIR spectrum of hexanolic extract from cells of Haloferax alexandrinus GUSF-1 (KF796625)
Fig. 6.
Thin-layer chromatograms of hexanolic extract from cells of Haloferax alexandrinus GUSF-1 (KF796625) developed in a acetone:petroleum ether (20:80; v/v), b methanol:chloroform (7:93; v/v), and c acetone:hexane (50:50; v/v) and viewed as (1) visible spots and (2) after spraying with DPPH•. The hexanolic extract was also resolved in d n-heptane: benzene (90:10, v/v) and viewed as (1) visible spots, (2) after spraying with DPPH• and (3) exposed to iodine vapor
Table 2.
Separation of biomolecules from hexanolic extract of Haloferax alexandrinus GUSF-1 (KF796625) achieved using thin-layer chromatography and different developing systems
| Solvent system | Rf of separated carotenoids | DPPH• reaction |
|---|---|---|
| Acetone–petroleum ether (20: 80, v/v) | 0.82 | + |
| 0.72 | + | |
| 0.63 | + | |
| 0.53 | + | |
| 0.51 | + | |
| 0.47 | + | |
| 0.4 | + | |
| 0.36 | + | |
| 0.14 | + | |
| Methanol–chloroform (7:93, v/v) | 0.71 | + |
| 0.43 | + | |
| 0.29 | + | |
| 0.24 | + | |
| Acetone: hexane (50:50, v/v) | 0.71 | + |
| 0.58 | + | |
| 0.46 | + | |
| n-heptane: benzene (90:10, v/v) | 0.9 | + |
Table 3.
Purification and quantification of the antioxidant compounds separated from the hexanolic extract of Haloferax alexandrinus GUSF-1 (KF796625)
| Solvent system |
Rf of separated compounds with antioxidant activity |
Quantity obtained after TLC separation (mg 100 g−1 cells) | % DPPH• RSA; AAE µg g−1 of cells (mean ± SD) | UV–visible absorption spectrum of pure compound | UV–visible absorption spectral peaks (nm) | Quantification of the purified compound using molar extinction coefficient (µg/g of cellular protein) (Kushwaha et al. 1974) | Identification |
|---|---|---|---|---|---|---|---|
| Acetone–petroleum ether (20:80, v/v) | 0.14 | 8 | 30 ± 0.04; 240.02 ± 0.5 |
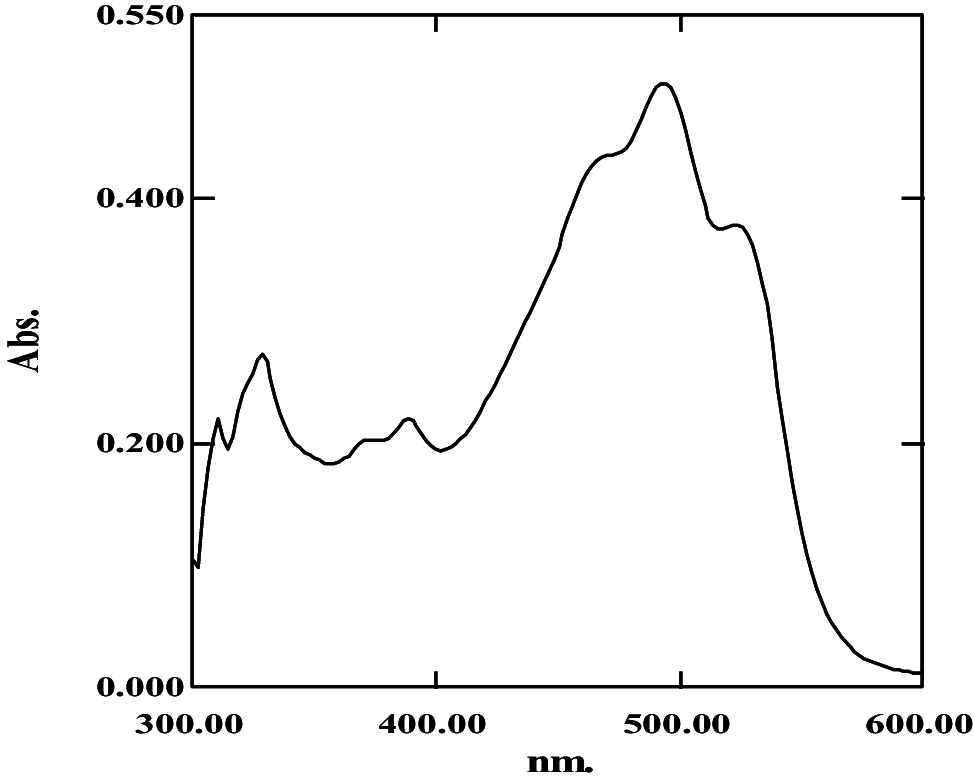
|
368, 387, 463, 492, 525 | 2 | Bacterioruberin |
| 0.36 | 5.6 | 17.51 ± 0.1; 140.0 ± 0.21 |
|
369, 388, 458 | 1.009 | 3-hydroxyechinenone | |
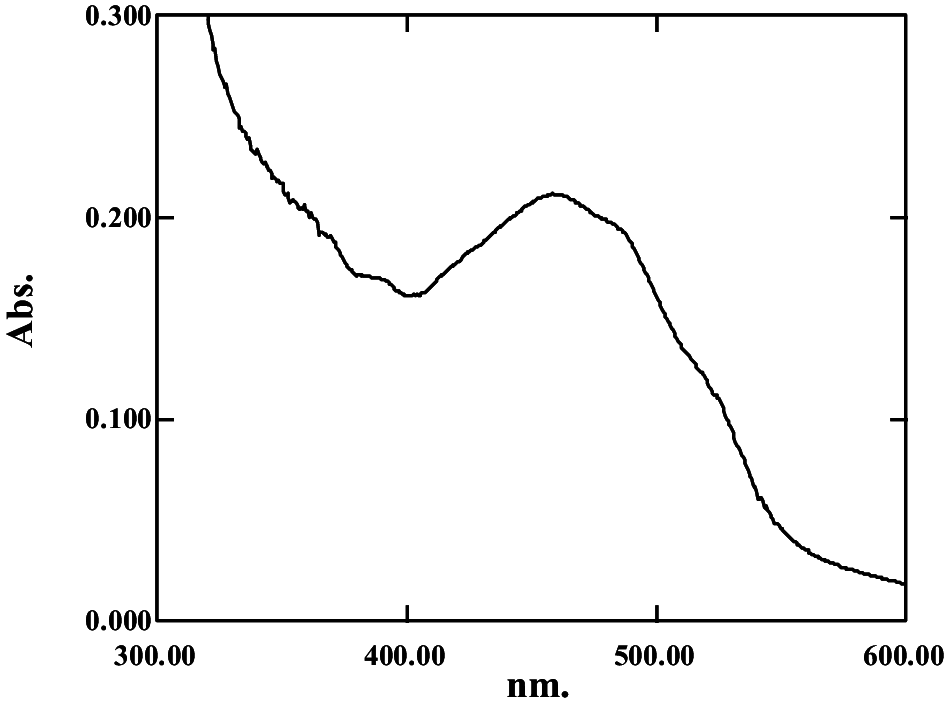
| 0.53 | 2.93 | 14.56 ± 0.18; 110.6 ± 0.36 |
|
374, 476 | 2.151 | Canthaxanthin | |
| Methanol–chloroform (7:93, v/v) | 0.24 | 8.53 | 28 ± 0.12; 220.4 ± 0.06 |
|
368, 386, 463, 492, 525 | 2 | Bacterioruberin |
| 0.29 | 5.06 | 23.24 ± 0.20; 180.6 ± 0.19 |
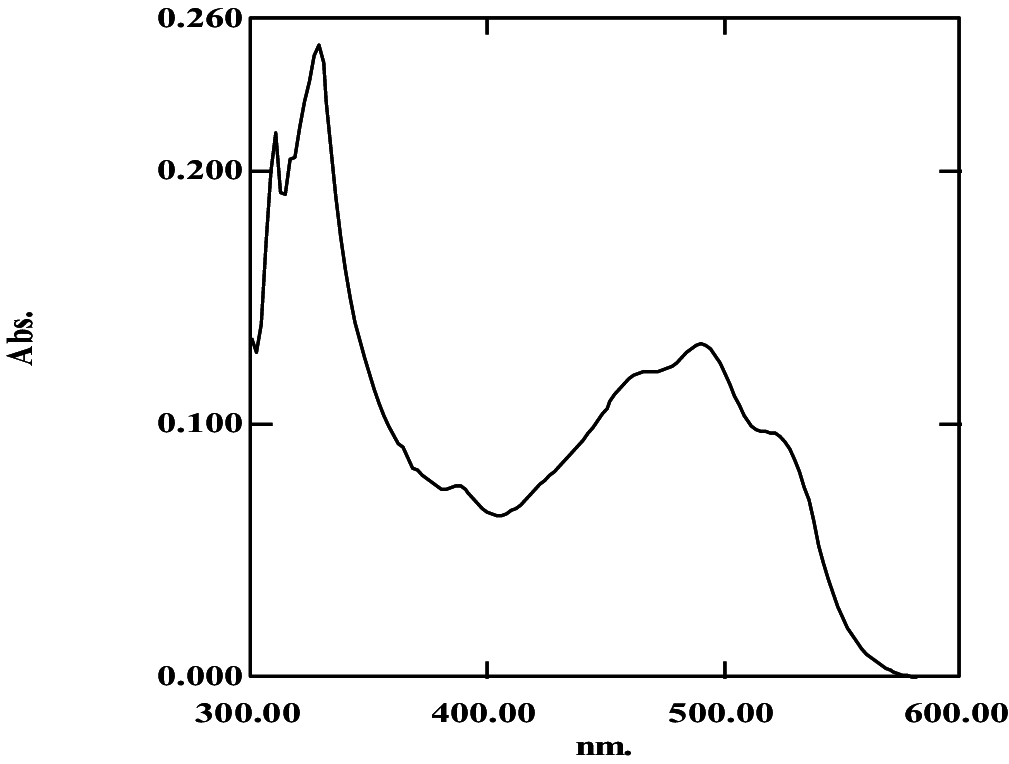
|
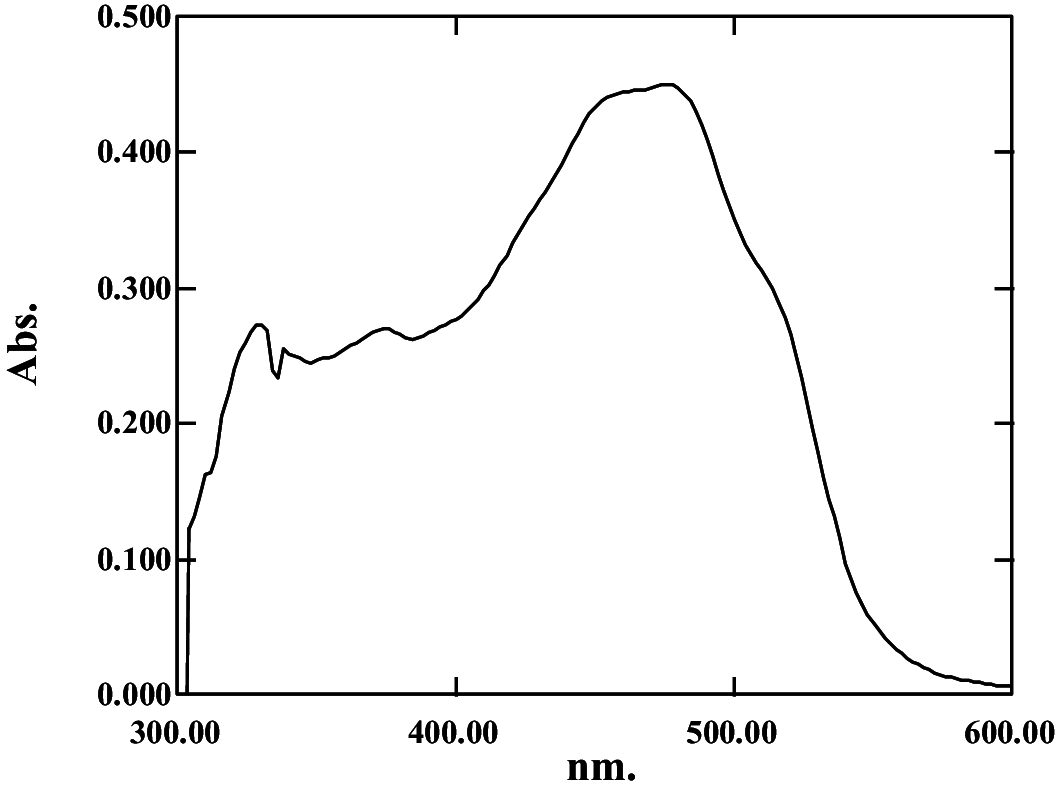
369, 389, 464, 490, 522 | 0.36 | Bisanhydrobacterioruberin | |
| 0.43 | 4.53 | 18.49 ± 0.09; 140.8 ± 0.01 |
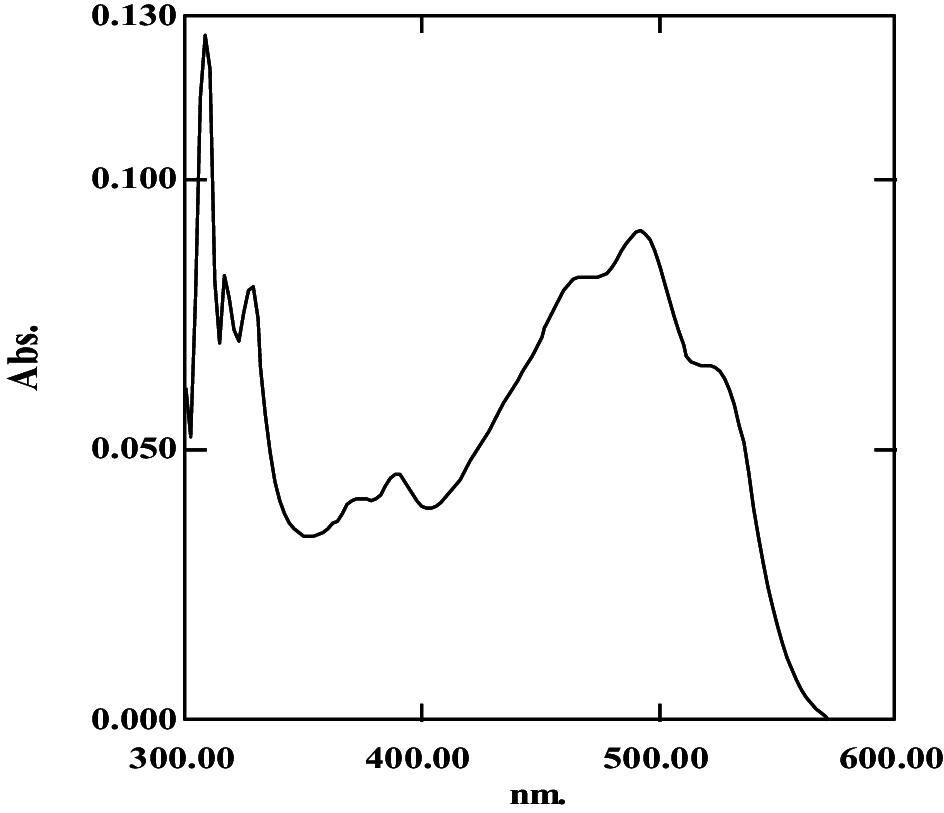
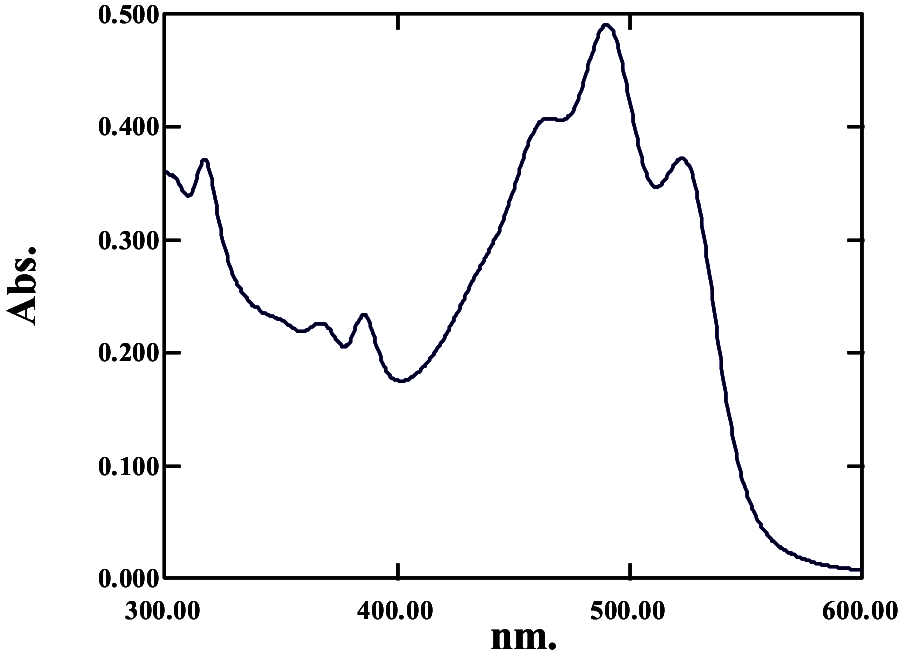
|
368, 387, 464, 489, 522 | 0.534 | Monoanhydrobacterioruberin | |
| Acetone:hexane (50:50, v/v) | 0.46 | 3.73 | 13.26 ± 0.07; 100.6 ± 0.14 |
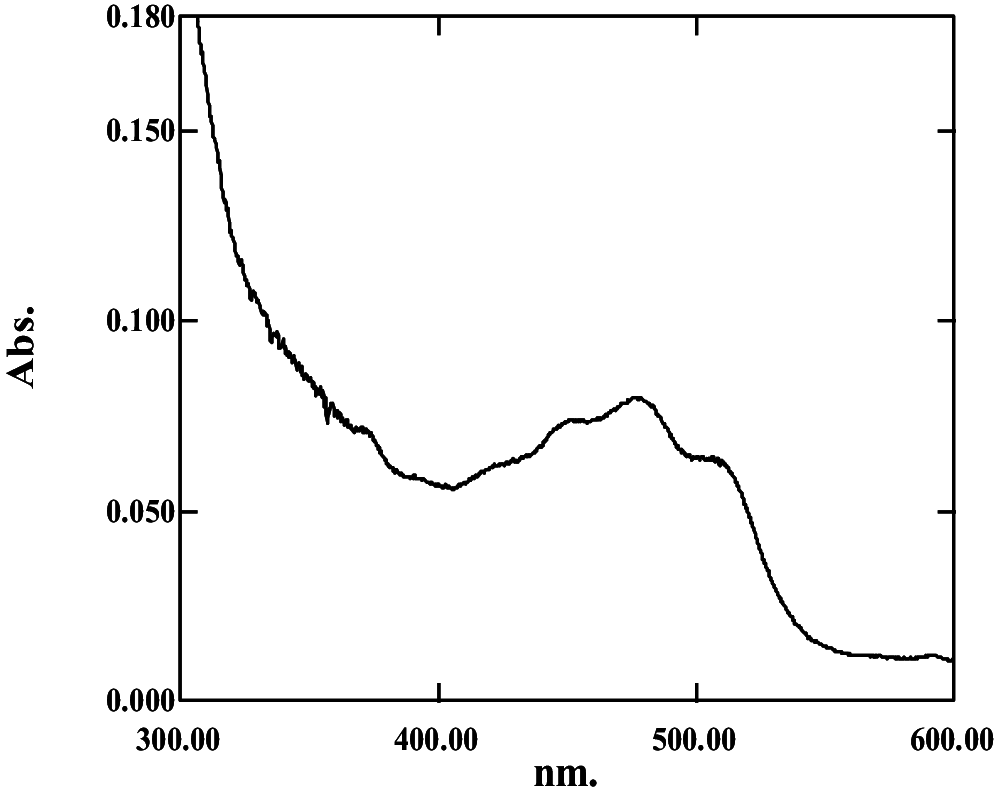
|
377, 451, 478, 512 | 0.3151 | Haloxanthin |
| n-Heptane:benzene (90:10, v/v) | 0.9 | 8 | 26.04 ± 0.12; 200.85 ± 0.1 | – | – | – | Tetrahydrosqualene |
This separation attained by TLC supported and confirmed the presence of canthaxanthin, 3-hydroxyechinenone, bacterioruberin, bisanhydrobacterioruberin, monoanhydrobacterioruberin, haloxanthin and tetrahydrosqualene. These results are in good agreement and consistent with that reported for Haloferax alexandrinus Strain TMT (Asker et al. 2002), Haloferax volcanii culture (Ronnekleiv et al. 1995) and in Halobacterium cutirubrum as described by Tornabene et al (1969).
Chemical profiling of hexanolic extract
The chemical profile of hexanolic extract by HR-LC/MS–DAD showed 12 peaks, which, in the increasing order of their retention time, eluted a total of 17 different compounds, with different m/z values (Fig. 7a). The diode array detector (DAD) revealed that peaks between Rt 29.943 and 31.68 absorbed at 320, 447, and 490 nm (Fig. 7b–d). These compounds were therefore, confirmed as red carotenoids. A combination of the DAD analyses (UV–Vis), the Agilent Mass Hunter software, the Japanese carotenoid database, and PubChem was used to identify the different compounds according to their m/z values. Compounds thus identified were canthaxanthin, tetrahydrosqualene, 3-hydroxyechinenone, phytoene, astaxanthin, bisanhydrobacterioruberin, monoanhydrobacterioruberin, bacterioruberin, lycopene, haloxanthin, and phytofluene, whose mass spectral data are presented in Table 4.
Fig. 7.
Chemical profile of hexanolic extract from cells of Haloferax alexandrinus GUSF-1 (KF796625). a HR-LC/MS–DAD chromatogram of hexanolic extract revealing a total of 12 peaks labeled according to the increasing retention times (Rt). Identification of the separated compounds was achieved by comparing the m/z values obtained with the Agilent Mass Hunter Qualitative Analysis Software version (B.05.00), Japanese carotenoid database, Pubchem, and reported literature. Diode Array Detector response at b 320, c 447 and d 490 nm
Table 4.
Mass spectral analysis of the hexanolic extract of Haloferax alexandrinus GUSF-1 (KF796625)
| Peak no | Retention time a | (m/z)a | za | Iona | Abundancea (conc) | Massa | Molecular formulab | Identificationb |
|---|---|---|---|---|---|---|---|---|
| 1 | 1.121 | 202.17 | 1 | (M + H) + | 1.12 × 107 | 201.17 | C11H23NO2 | Unidentified |
| 2 | 4.528 | 302.19 | 1 | (M + H) + | 0.07 × 107 | 301.18 | C16H23N5O | Unidentified |
| 3 | 6.823 | 565.3 | 1 | (M + H) + | 0.51 × 107 | 564.3 | C40H52O2 | Canthaxanthin |
| 4 | 9.801 | 430.24 | 1 | (M + H) + | 0.31 × 107 | 429.23 | C22H31N5O4 | Unidentified |
| 5 | 20.523 | 415.20 | 1 | (M + H) + | 0.37 × 107 | 414.20 | C30H54 | Tetrahydrosqualene |
| 6 | 23.457 | 301.13 | 1 | (M + Na) + | 0.02 × 107 | 278.15 | C16H22O4 | Unidentified |
| 7 | 25.471 | 568.45 | 1 | (M + H) + | 0.03 × 107 | 567.45 | C40H56O2 | 3-hydroxyechinenone |
| 595.38 | 1 | (M + Na) + | 0.08 × 107 | 572.39 | C40H60O2 | Unidentified | ||
| 546.40 | 1 | (M + H) + | 0.1 × 104 | 545.39 | C40H64 | Phytoene | ||
| 597.4 | 1 | (M + H) + | 2 × 104 | 596.6 | C40H52O4 | Astaxanthin | ||
| 8 | 27.226 | 337.23 | 1 | (M + Na) + | 0.04 × 107 | 314.24 | C18H34O4 | Unidentified |
| 9 | 29.943 | 705.55 | 1 | (M + H) + | 0.3 × 104 | 704.55 | C50H72O2 | Bisanhydrobacterioruberin |
| 723.57 | 1 | (M + H) + | 0.9 × 104 | 722.55 | C50H74O3 | Monoanhydrobacterioruberin | ||
| 741.58 | 1 | (M + H) + | 0.03 × 107 | 740.57 | C50H76O4 | Bacterioruberin | ||
| 10 | 30.209 | 537.4 | 1 | (M + H) + | 1 × 103 | 536 | C40H56 | Lycopene |
| 11 | 31.68 | 739.56 | 1 | (M + H) + | 0.07 × 107 | 738.55 | C50H74O4 | Haloxanthin |
| 12 | 35.658 | 543.2 | 1 | (M + H) + | 2.1 × 103 | 542.2 | C40H62 | Phytofluene |
aThe hexanolic extract was chromatographed on a C-18 reverse-phase column; Hypersil gold 3 micron 100 × 2.1 mm column. Peaks are labeled based on increasing retention times (Rt) that they are eluted out
bIdentified using the support system; Agilent Mass Hunter software, the Japanese carotenoid database, and Pubchem
Tetrahydrosqualene (C30 isoprenoid) attributes to stabilize the membrane structure in Halobacterium cutirubrum as reported by Tornabene et al. (1969) and as observed in this study, is recorded for its first-time role as a free radical scavenger (antioxidant). The C40 isoprenoids detected were 3-hydroxyechinenone, astaxanthin, canthaxanthin, lycopene, phytofluene and phytoene. Although all of these are reported in Haloferax alexandrinus strain TMT (Asker et al. 2002), but their biological property and role is not characterized. The antioxidant activity for astaxanthin, canthaxanthin, and lycopene is a known feature, but we demonstrated and recorded, for the first time, the free radical scavenging activity of 3-hydroxyechinenone. Yang et al. (2015) delineated phytofluene and phytoene as precursors in the biosynthetic pathway of C50 carotenoids in Haloarchaea. Phytoene detected in very low concentrations than the other chemical constituents in this study is consistent with that reported by Rodrigo-Baños et al. (2015).
The C50 isoprenoids identified were bacterioruberin along with its precursors; monoanhydrobacterioruberin and bisanhydrobacterioruberin. All of these decolorized DPPH•, confirming their antioxidant activity. Involvements of these carotenoids in free radical scavenging activity in a whole-cell extract of Haloterrigena turkmenica are known and reports are available (Squillaci et al. 2017). A dodecane C50 carotenoid, haloxanthin with a peroxide group detected in the hexanolic extract, has been identified in Haloferax volcanii (Ronnekleiv et al. 1995), but not in Haloferax alexandrinus GUSF-1 (KF796625) and its role in scavenging free radicals as observed in this study, is a novel report.
Conclusively, the present study is the first report of the antioxidant activity of exopolysaccharide, glycerol diether moiety, tetrahydrosqualene, haloxanthin, and 3-hydroxyechinenone of Haloferax alexandrinus GUSF-1 (KF796625). The synergistic actions of all of these components contribute to the antioxidant activity of the culture. The minimum aseptic conditions required to obtain cell mass possessing such high antioxidant ability gives it an economic advantage over other eubacterial counterparts for large-scale production. Therefore, the haloarchaeon, Haloferax alexandrinus GUSF-1(KF796625) is recommended for use in microbial industrial biotechnology.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The authors wish to thank IIT-Bombay, India, for the HR-LC/MS–DAD analysis and Department of Chemistry, Goa University, India, for the FTIR analysis.
Author contributions
Author IJ Furtado designed the experiments, analyzed the observations, and formulated the MS along with JJ Alvares who alone carried out all the experimental benchwork.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Haloferax alexandrinus GUSF-1 (GenBank accession number KF796625). Gen bank: http://www.ncbi.nlm.nih.gov.
Contributor Information
Jyothi Judith Alvares, Email: alvaresjv@gmail.com, Email: micro.jyothi@unigoa.ac.in.
Irene Jeronimo Furtado, Email: ijfurtado@unigoa.ac.in.
References
- An GH, Schuman DB, Johnson EA. Isolation of Phaffia rhodozyma mutants with increased astaxanthin content. Appl Environ Microbiol. 1989;55(1):116–124. doi: 10.1128/AEM.55.1.116-124.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alvares JJ, Furtado IJ. Extremely halophilic Archaea and Eubacteria are responsible for free radical scavenging activity of solar salts of Goa-India. Global J Biosci Biotechnol. 2018;7(2):242–254. [Google Scholar]
- Antón J, Meseguer I, Rodríguez-Valera F. Production of an extracellular polysaccharide by Haloferax mediterranei. Appl Environ Microbiol. 1988;54(10):2381–2386. doi: 10.1128/AEM.54.10.2381-2386.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asker D, Awad T, Ohta Y. Lipids of Haloferax alexandrinus Strain TMT: an extremely halophilic canthaxanthin producing archaeon. J Biosci Bioeng. 2002;93(1):37–43. doi: 10.1016/S1389-1723(02)80051-2. [DOI] [PubMed] [Google Scholar]
- Braganca JM, Furtado I. Isolation and characterisation of haloarchaea from low- salinity coastal sediments and waters of Goa. Curr Sci. 2009;96:1182–1184. [Google Scholar]
- Britton G. UV/visible spectroscopy. In: Britton G, Liaaen-Jensen S, Pfander H, editors. Carotenoids. Switzerland: Basel; 1995. pp. 13–62. [Google Scholar]
- Del Gallo M, Haegi A. Characterization and quantification of exocellular polysaccharides in Azospirillum brasilense and Azospirillum lipoferum. Symbiosis. 1990;9:155–161. [Google Scholar]
- Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F. Colorimetric method for determination of sugars and related substances. Anal Chem. 1956;28:350–356. doi: 10.1021/ac60111a017. [DOI] [Google Scholar]
- Gaonkar SK, Furtado IJ. Isolation and culturing of protease and lipase producing Halococcus agarilyticus GUGFAWS-3 from marine Haliclona sp. inhabiting the rocky intertidal region of Anjuna in Goa, India. Ann Microbiol. 2018;68:851–861. doi: 10.1007/s13213-018-1391-6. [DOI] [Google Scholar]
- Kushwaha SC, Gochnauer MB, Kushner DJ, Kates M. Pigments and isoprenoid compounds in extremely and moderately halophilic bacteria. Can J Microbiol. 1974;20:241–245. doi: 10.1139/m74-038. [DOI] [PubMed] [Google Scholar]
- Lu JM, Lin PH, Yao Q, Chen C. Chemical and molecular mechanisms of antioxidants: experimental approaches and model systems. J Cell Mod Med. 2010;14:840–860. doi: 10.1111/j.1582-4934.2009.00897.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malik AD, Furtado IJ (2019) Haloferax sulfurifontis GUMFAZ2 producing xylanase‐free cellulase retrieved from Haliclona sp. inhabiting rocky shore of Anjuna, Goa‐India. J Basic Microbiol 59:692–700 [DOI] [PubMed]
- Naik S, Furtado I. Formation of Rhodochrosite by Haloferax alexandrinus GUSF-1. J Clust Sci. 2019;30:1435–1441. doi: 10.1007/s10876-019-01586-9. [DOI] [Google Scholar]
- Patil S, Fernandes J, Tangsali RB, Furtado I. Exploitation of Haloferax alexandrinus for biogenic synthesis of silver nanoparticles antagonistic to human and lower mammalian pathogens. J Clust Sci. 2014;25:423–433. doi: 10.1007/s10876-013-0621-0. [DOI] [Google Scholar]
- Raghavan TM, Furtado I. Tolerance of an estuarine halophilic archaebacterium to crude oil and constituent hydrocarbons. Bull Environ Contam Toxicol. 2000;65:725–731. doi: 10.1007/s0012800183. [DOI] [PubMed] [Google Scholar]
- Raghavan TM, Furtado I. Expression of carotenoid pigments of haloarchaeal cultures exposed to aniline. Environ Toxicol. 2005;20:165–169. doi: 10.1002/tox.20091. [DOI] [PubMed] [Google Scholar]
- Rodrigo-Baños M, Garbayo I, Vílchez C, Bonete MJ, Martínez-Espinosa RM. Carotenoids from Haloarchaea and their potential in biotechnology. Mar Drugs. 2015;13:5508–5532. doi: 10.3390/md13095508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ronnekleiv M, Liaaen-Jensen S. Bacterial carotenoids. 53, C50-carotenoids 23; Carotenoids of Haloferax volcanii versus other halophilic bacteria. Biochem Syst Ecol. 1995;23:627–734. doi: 10.1016/0305-1978(95)00047-X. [DOI] [Google Scholar]
- Ross HNM, Collins MD, Tindall BJ, Grant WD. A rapid procedure for the detection of archaebacterial lipids in halophilic bacteria. J Gen Microbiol. 1981;123:75–80. [Google Scholar]
- Ross HNM, Grant WD, Harris JE. Lipids in archaebacterial taxonomy. In: Goodfellow M, Minnikin DE, editors. Chemical methods in bacterial systematics. Society for Applied Bacteriology Technical Series 20. London: Academic Press; 1985. pp. 289–300. [Google Scholar]
- Sequeira F (1992) Microbiological study of salt pans of Goa. M.Sc. Dissertation, Goa University. Goa, India
- Singh A, Singh AK. Haloarchaea: worth exploring for their biotechnological potential. Biotechnol Lett. 2017;39:1793–1800. doi: 10.1007/s10529-017-2434-y. [DOI] [PubMed] [Google Scholar]
- Squillaci G, Finamore R, Diana P, Restaino OF, Schiraldi C, Arbucci S, Ionata E, La Cara F, Morana A. Production and properties of an exopolysaccharide synthesized by the extreme halophilic archaeon Haloterrigena turkmenica. Appl Microbiol Biotechnol. 2016;100:613–623. doi: 10.1007/s00253-015-6991-5. [DOI] [PubMed] [Google Scholar]
- Squillaci G, Parrella R, Carbone V, Minasi P, La Cara F, Morana A. Carotenoids from the extreme halophilic archaeon Haloterrigena turkmenica: identification and antioxidant activity. Extremophiles. 2017;21:933–945. doi: 10.1007/s00792-017-0954-y. [DOI] [PubMed] [Google Scholar]
- Tornabene TG, Kates M, Gelpi E, Oro J. Occurrence of squalene, di and tetrahydrosqualenes, and vitamin MK8 in an extremely halophilic bacterium, Halobacterium cutirubrum. J Lipid Res. 1969;10:294–303. doi: 10.1016/S0022-2275(20)43087-1. [DOI] [PubMed] [Google Scholar]
- Utkina NK, Makarchenko AE, Shchelokova OV, Virovaya MV. Antioxidant activity of phenolic metabolites from marine sponges. Chem Nat Comp. 2004;403:373–377. doi: 10.1023/B:CONC.0000048251.58246.22. [DOI] [Google Scholar]
- Velho Pereira S, Furtado I (2014) Retrieval of euryhaline eubacterial and haloarchaeal bionts from nine different benthic sponges: reflection of the bacteriological health of waters of Mandapam, India. Indian J Mar Sci 43:773–783
- Velho-Pereira S, Parvatkar P, Furtado I. Evaluation of antioxidant producing potential of halophilic bacterial bionts from marine invertebrates. Indian J Pharm Sci. 2015;77:183–189. doi: 10.4103/0250-474X.156572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan MY, Dong G, Yang BQ, Feng H. Identification and characterization of a novel antioxidant peptide from feather keratin hydrolysate. Biotechnol Lett. 2016;38:643–649. doi: 10.1007/s10529-015-2016-9. [DOI] [PubMed] [Google Scholar]
- Warren CK, Weedon BCL (1958) Carotenoids and related compounds. Part VII. Synthesis of canthaxanthin and echinenone. J Chem Soc 3986–3993
- Yang Y, Yatsunami R, Ando A, Miyoko N, Fukui T, Takaichi S, Nakamura S. Complete biosynthetic pathway of the C50 carotenoid bacterioruberin from lycopene in the extremely halophilic archaeon Haloarcula japonica. J Bacteriol. 2015;197:1614–1623. doi: 10.1128/JB.02523-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Lu X, Fu Z, Wang Z, Zhang J. Sulfated modification of a polysaccharide obtained from fresh persimmon (Diospyros kaki L.) fruit and antioxidant activities of the sulfated derivatives. Food Chem. 2011;127:1084–1090. doi: 10.1016/j.foodchem.2011.01.100. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.