Abstract
Background:
Guidelines for the care of women undergoing pelvic radiation therapy (RT) recommend vaginal dilator therapy (VDT) to prevent radiation-induced vaginal stenosis (VS), however, no standard protocol exists. This review seeks to update our current state of knowledge concerning VS and VDT in radiation oncology.
Methods:
A comprehensive literature review (1972–2017) was conducted using search terms “vaginal stenosis,” “radiation” and “vaginal dilator.” Information was organized by key concepts including VS definition, time course, pathophysiology, risk factors and interventions.
Results:
VS is a well described consequence of pelvic RT, with early manifestations as well as late changes evolving over several years. Strong risk factors for VS include RT dose and volume of vagina irradiated. Resultant vaginal changes can interfere with sexual function and correlational studies support the use of preventive VDT. The complexity of factors which drive non-compliance with VDT is well recognized. There is no prospective data to guide optimal duration of VDT and the consistency with which radiation oncologists monitor VS and manage its consequences is unknown.
Conclusions:
This review provides information concerning VS definition, pathophysiology and risk factors and identifies domains of VDT practice which are under-studied. Prospective efforts to monitor and measure outcomes of patients who are prescribed VDT are needed to guide practice.
INTRODUCTION
Women who undergo vaginal or pelvic radiotherapy (RT) are at risk for developing vaginal stenosis (VS). The reported incidence of VS resulting from RT varies, with an overall range of 2.5% to 88% [1–7], depending on patient, treatment, and post-treatment factors, including cancer type, hysterectomy status, RT dose, volume of vagina treated, use of brachytherapy alone versus pelvic RT, and use of dilators and hormones. VS detection and reporting also depend on subjective versus objective measurement and whether the definition incorporates interference with sexual function. In recent literature, crude estimates of VS (any grade) measured within the first 3 years post-RT among exclusive endometrial, cervical, and anal cancer populations are approximately 50%, 60%, and 80%, respectively [7–9].
The clinical impact of VS is multi-dimensional. If VS develops, scarring can limit post-treatment surveillance of the vagina and cervix and pelvic examination may be uncomfortable or painful [Figure 1]. Psychologically, pain with physical exams or sexual intercourse may trigger distressing or even traumatic memories for a patient of her cancer and treatments. Further, patients who report VS or impaired vaginal elasticity are more likely to have vaginal symptoms, dyspareunia, and sexual dysfunction [10–13].
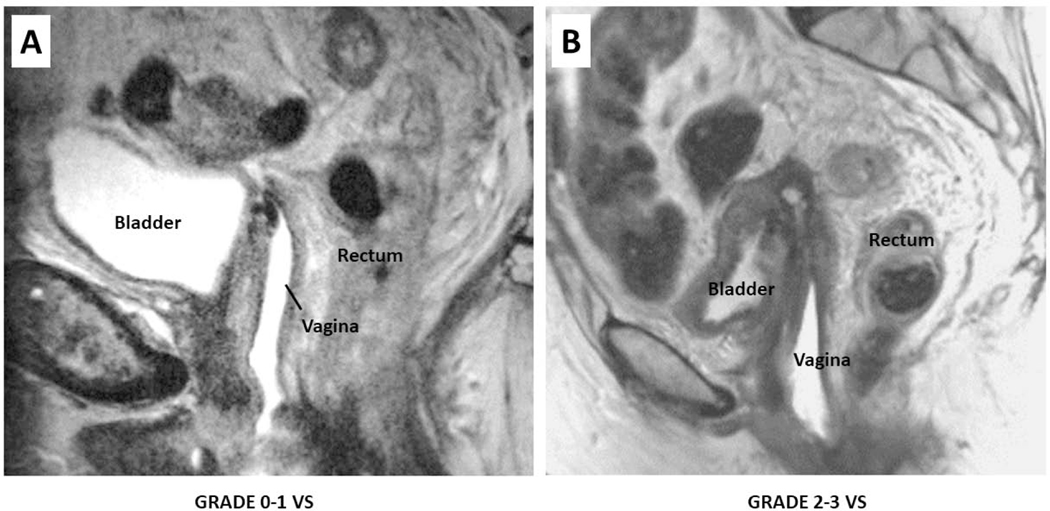
Figure 1:
MRI pelvis T2 sagittal images from a post-menopausal female with locally advanced cervical cancer pre- (A) and post- (B) definitive brachytherapy. She was not sexually active and she was compliant with dilators for only 6 months after completing her treatments. Her pelvic exam at 3 years post radiotherapy was remarkable for atrophic changes, an agglutinated vagina, and a cervix which could not be identified on visualization. On palpation, the parametria were not infiltrated and the rectovaginal septum was supple. Her pre-brachytherapy image with vaginal gel delineating the walls of the vagina is shown on the left (A). Her post treatment image with the same sequence performed 3 years later, on the right (B), shows uterine atrophy and closure of the upper 2/3 of the vaginal wall (black arrows) - a clear depiction of the grade 3 vaginal stenosis described by physical exam- and no evidence of disease.
Vaginal dilator therapy (VDT) is commonly recommended following a course of pelvic RT to prevent VS. Guidelines have proposed strategies to promote and standardize VDT education, methods of provision and technique [2]. A majority of radiation oncologists (RO) agree that they share the responsibility to manage vaginal and sexual health concerns after pelvic radiotherapy, yet indicate they need further education and practical training [13]. A recent survey by Kachnic et al highlighted confusion among U.S. practicing ROs regarding risk factors for VS and a lack of training in the measurement of VS or the management of sexual function [14].
In 2017, an interdisciplinary writing group formed at the annual meeting of the Scientific Network on Female Sexual Health and Cancer identified a need to synthesize our knowledge concerning VS and VDT. To address this, several members undertook an independent effort to perform a comprehensive literature review, tabulate the available evidence, and summarize key concepts. Our major findings are presented here along with discussion of opportunities for future research and clinical improvement.
METHODS
A literature search of PubMed with the search terms “Vaginal Stenosis AND Radiation” and “Vaginal Dilator AND Radiation” resulted in 80 unique citations published between 1972–2017. Data were summarized and reviewed by two of the authors (S.D. and D.D.J.). Fourteen references described surgical intervention studies for the correction of VS, 8 were interventional dilator studies, 10 were review articles, and 4 were practice surveys. Most remaining references were observational outcomes studies of cancer patients, including 19 cervical cancer patient studies, 12 uterine, 1 both cervical and uterine, 3 anal/rectal, 1 vulvar, and 1 study of sarcoma patients. 7 were case reports. Level 1 evidence is lacking on this topic, and thus observational and retrospective studies were included in the present review.
RESULTS
Question 1: What is Vaginal Stenosis?
The term VS is commonly defined as narrowing of the vaginal canal resulting from a build-up of scar tissue [15]. Although VS is most often reported as a consequence of ionizing radiation to the vaginal canal, it is not specific to RT and can also occur due to vulvovaginal atrophy resulting from hypo-estrogenism due to other causes [16] or conditions which lead to repeated and progressive inflammation [17]. In the gynecologic (GYN) cancer literature, the term “VS” has been used to encompass a variety of responses in the vaginal tissue including (1) shortening of the vaginal canal; (2) narrowing of the canal (fibrotic changes); (3) an inability to open a vaginal speculum due to loss of elasticity and atrophy of the mucosa; and (4) pain with exam/intercourse [1,10,18]. The Common Terminology Criteria for Adverse Events (CTCAE) has been used most widely in the modern era for reporting VS [7–9, 19–21] and is displayed along with other measures of VS in Table 1.
Table 1:
Physician and Patient Measures of Vaginal Stenosis
| VS Measurement Scale | Physician or patient graded | Detail |
|---|---|---|
| Vaginal sound method to measure length [SR1] | Physician | The vaginal sound used in this protocol was a modified plastic vaginal dilator 15cm long and 2.85 cm in diameter calibrated in centimeters, and used to measure vaginal length Measurements are recorded from the vaginal apex to the posterior fourchette using firm pressure. |
| Chassagne grading [SR2] | Physician | Grade 1: vaginal narrowing/shortening less than or equal to half of original dimension and/or mild dyspareunia Grade 2: vaginal narrowing/shortening greater than half of original dimension and/or moderate dyspareunia |
| LENT-SOMA scales [SR3] | Physician and Patient | Objective (length, obliteration, frequency of intercourse), subjective (dryness, painful intercourse, decreased desire, decreased satisfaction), management (use of lubricants, hormone replacement, and frequency of VDT), and analytic (evaluate quality of life and stenosis) |
| Modified Diche score [SR4] | Physician | Separate scores for bleeding, discharge, erythema, ulceration, telangiectasia, VS and pallor of mucosa based on physician observation, without reference to sexual activity or dyspareunia |
| CTCAE v4.0 | Physician | Grade 1: asymptomatic; mild vaginal narrowing/shortening Grade 2: vaginal narrowing/shortening not interfering with physical exam Grade 3: vaginal narrowing/shortening interfering with use of tampons, sexual activity, or physical exam |
| EORTC endometrial module (patient-reported) [SR5] | Patient | Rate on a scale of 1 to 4, (only if they have been sexually active in the past 4 weeks), “Has your vagina felt short and / or tight?” |
Abbreviations: SR= Supplementary Reference (supplementary references can be found in Appendix 1) LENT-SOMA = Late effects normal tissue - subjective, objective, management, analytic; VDT = vaginal dilator therapy; CTCAE= Common Terminology Criteria for Adverse Events; EORTC = European Organization for Research and Treatment of Cancer
VS is widely considered a subacute to “late effect” of radiotherapy occurring in weeks or months after radiotherapy is completed, increasing in incidence over time, and resulting in permanent vaginal changes [22–23]. Conversely, vaginal shortening, in some studies, has been characterized as an “early effect” resulting from increased adhesiveness of the vaginal epithelial mucosa rather than a late connective tissue reaction in the deeper parts of the vaginal wall [24]. The biologic mechanism of VS appears to have both direct as well as indirect causes and is likely both an early and late phenomenon. The direct effects may include denuding of the epithelium due to RT, which can lead to adhesion formation, which can cause early onset of vaginal shortening [18,25] [Figure 2]. The secondary effects are related to repair mechanisms in the irradiated tissue, or an “indirect response.” These effects include circumferential fibrosis associated with hyalinization, disorganized elastosis, and collagenation in the extra-cellular matrix and can lead to thickening of the vaginal wall and subsequent narrowing of the vaginal canal [25, 26] [Figure 3].
Figure 2:
MRI pelvis T2 sagittal images with vaginal gel illustrating vaginal shortening secondary to adhesion formation following definitive chemoradiation and brachytherapy in 2 different post-menopausal patients treated for stage II vaginal cancer. Moderate changes occluding the upper ½ of the vagina on the right (B) compared to mild-to-no changes on the left (A).
Figure 3:
Sagittal (A) and coronal (B) T2-weighted MRI images of an irradiated vagina with vaginal gel in a pre-menopausal female who was NED 7 months following chemoradiation and vaginal brachytherapy boost to the upper half of the vaginal canal. The arrows point to thickening due to irradiation in the upper ½ of the vaginal wall, with resulting narrowing of the vaginal apex.
Vaginal atrophy secondary to radiation-induced, medical or surgical castration is believed to facilitate both direct vaginal epithelial changes and indirect changes in extra-cellular connective tissues, though the biologic time course and specific effects of RT on the estrogen receptors within the vaginal mucosa are not well characterized [1,27].
Question 2: When does VS develop?
Gynecologic cancer literature spanning several decades lacks consensus on the time course of VS: some studies report early changes during [28], or within a few weeks to months after treatment [29–30], and other studies describe pathologic progression over years [31, 32]. This variation likely depends on the duration of observation, with recent literature highlighting the need for actuarial rather than crude reporting, as the incidence of VS increases as a function of time [8]. A summary of select publications that have detailed a time course to VS is presented in Table 2. Taken together, these studies indicate that acute effects such as vaginal shortening may become apparent very quickly in the early weeks-to-months following RT and these changes may continue to progress more slowly over the ensuing months-to-years.
Table 2:
Studies of RT Patients with Published Time Course to VS (According to Time Frame)
| Reference | Cancer Type | Number of Patients | Treatment | Outcome | Time Frame | Detail |
|---|---|---|---|---|---|---|
| Katz, 2001 [28] | CC + EC | 41 | EBRT + VB | Maximal extension of the vaginal cylinder above the superior border of the symphysis pubis at each insertion as measured on a 2D radiograph | Acute, Within 2 months | The median interval between brachytherapy treatments was 18 days (range, 11–52 days). The difference between the mean vaginal measurement at the first and second insertion (2.27 ± 1.14 cm vs. 1.72 ± 1.14 cm) was highly statistically significant (P < 0.0001). |
| Rakhra, 2016 [29] | EC | 381 | VB alone versus EBRT + VB | Median cylinder size | Acute, Within 2 months | In patients receiving VB alone, median cylinder size was 3 cm; after pelvic RT, it was 2.5 cm. The authors suggest that this difference resulted from changes and narrowing in the vaginal canal which occurred during the course of pelvic RT |
| Hartman, 1972 [1] | Locally advanced CC | 221 | EBRT + ICRT | Obliteration of vaginal canal | Sub-Acute, Within 6 months | One half of the patients acquired their major degree of stenosis of the vagina at 6 weeks, while one third had the maximum amount of obliteration at 3 months. The remainder had complete obliteration within 5 to 8 months. |
| Brand, 2006 [30] | Locally advanced CC | 188 | EBRT + ICRT | Partial or complete occlusion of vaginal canal | Sub-Acute, Within 12 months | VS of any grade was noted at a mean of 9.6 months and median of 7.5 months (range, 26 days–5.6 years) |
| Park, 2015 [21] | Early stage EC | 101 | VB alone | VS by CTCAE v4.0 | Sub-Acute, Within 12 months | The mean time to recorded VS of any grade was 6 months |
| Bruner, 1993 [18] | CC + EC | 90 | EBRT + VB or VB alone | Vaginal length measured with sound | Late, Within 24 months | Vaginal length continued to decrease up to 2 years after brachytherapy |
| Bahng, 2012 [7] | Early stage EC | 100 | VB alone | VS by CTCAE v4.0 | Late, Within 24 months | Median time interval from the date of brachytherapy completion to the reported date of grade 1–3 vaginal mucosal toxicities was 24 months (range, 3 months to 5 years) |
| Onsrud, 2001 [31] | Early stage EC | 217 | VB alone | Chassagne Grading Scale | Late, Within 24 months | The cumulative frequency of late vaginal reactions increased significantly within the first two years, and appeared to level off between years 2 and 3 |
| Kirchheiner, 2016 [8] | Locally advanced CC | 630 | Chemo/EBRT + ICRT | VS by CTCAE v3.0 | Late, Within 24–36 months | Prevalence rates for VS grade1 and grade2 increased continuously during the first 2–3 years of follow-up |
| Weiss, 1999 [32] | Early stage EC | 159 | Post-op EBRT+VB | Chassagne grading scale | Late, Within 36 months | VS was recorded to occur between 6 and 50 months after treatment, with the majority of cases noted within the first 12 months |
| Sorbe, 1990 [24] | Early stage EC | 404 | Post-op VB | Vaginal length measured with sound | Late, Within 36 months | Most (88%) VS occurred within the first 12 months. After 36 months, practically no further shrinkage took place. |
| Gondi, 2012 [19] | Locally advanced CC | 480 | EBRT + ICRT | VS by CTCAE v4.0 | Late, Within 48 months | VS risk of 42% at 5 years, with a steep, continual increase in VS from end of RT up to year 4 |
Abbreviations: CC= cervical cancer; EC= endometrial cancer; EBRT= external beam radiotherapy; VB= vaginal brachytherapy; ICRT= intra-cavitary radiotherapy; VS= vaginal stenosis; CTCAE= Common Terminology Criteria for Adverse Events
Late effects may continue up to 20 years after RT [33]; nevertheless, it appears that after 5 years, the likelihood of developing worsening or new VS is very small [19]. The two largest observational studies with the longest follow up include a report by Gondi et al. of 480 cervical cancer patients [19], and a report by Sorbe et al. of 404 early stage endometrial cancer patients [24]. Among the cervical cancer patients, the estimated continuous risk of VS beyond year 5 was 2.3% [19]. Among the endometrial cancer patients, 88% of the vaginal shortening occurred in the first 4 years post-RT [24]. The time frame of VS in other GYN and gastrointestinal cancers is sparsely studied.
Question 3: Which treatment factors affect the risk of VS?
The incidence and degree of VS appear to be related to host and treatment factors [1, 7–9, 18, 20–21, 24, 26–34]. Evidence is strongest for the relationship between VS and RT dose and the length and portion of vagina treated.
RT Dose:
Studies of endometrial cancer brachytherapy have shown that a higher dose at the vaginal surface is associated with a higher risk of VS. In 1990, a prospective observational study by Sorbe et al. of 404 women with stage I endometrial cancer who received vaginal cuff brachytherapy (dose was prescribed at depth 10mm from cylinder surface) found that a dose 9.0Gy per fraction, as compared to 6, 5, or 4.5 Gy per fraction, was positively associated with vaginal shortening. After 5 years, the vaginal shortening was 79% vs 60%, 50%, 31%, respectively [24]. A 2005 study by the same group, randomized 290 Swedish patients with early stage endometrial cancer to receive 2.5Gy versus 5.0Gy × 6 fractions, to a vaginal depth of 5mm. Comparing vaginal length after 5 years to baseline, there was no difference in the 2.5Gy group; but a highly significant difference occurred for the 5Gy group, which lost 25% of vaginal length on average [34]. Other observational studies of vaginal brachytherapy after hysterectomy have found a similar dose effect [21, 31, 35].
In the definitive treatment of cervical cancer, with uterus intact, there appears to be a direct relationship between higher vaginal dose and RT-associated vaginal toxicity [8,36]. The largest published study, including 630 cervical cancer patients from the EMBRACE cohort, showed that the recto-vaginal reference point dose significantly correlates with VS. With a median follow-up time of 24 months, the probability to develop symptomatic VS was 16% with recto-vaginal reference point dose of 55Gy, 20% with 65Gy, 27% with 75Gy, 34% with 85Gy and 43% with 95Gy [8].
For gastro-intestinal cancers, a single report of 54 anal and rectal patients found that severe VS was lower when the mean vaginal dose was <43Gy [20].
Vaginal Volume or Vaginal Portion Irradiated:
For both cervical and endometrial cancer patients, the risk of VS appears to be associated with a higher volume of treated vagina. Among 630 cervical cancer patients in the EMBRACE study, the 2-year actuarial estimate for symptomatic VS was 21%, and the degree of tumor extension into the vagina was a significant risk factor [8]. Several observational studies of early stage endometrial cancer patients who received brachytherapy found that treatment of longer length of the vaginal canal was associated with a higher risk of VS [21, 7, 24]. Outcome studies detailing the effects of vaginal volume/portion irradiated for vaginal, vulvar, and anal cancers are lacking; however, these patients have a high risk of VS and vaginal morbidity due to the exposure of the lower portion of the vagina, which has a poorer tolerance to RT than the upper or middle portions [37].
Question 4: Does preventive vaginal dilator therapy mitigate the risk of VS or improve sexual function?
The purpose of VDT in the early response phases post-RT is to separate and prevent the formation of early adhesions between the walls of the mucosa. The secondary purpose of the dilator is to counteract the late effects in the submucosa including elastosis and circumferential fibrosis of the vaginal canal by stretching the vaginal tissue and promoting epithelial cell growth. Several studies have found significant associations between preventive dilation use and lower risk of VS following RT [Table 3]. Nevertheless, a systematic Cochrane review on VDT and RT states that cause and effect cannot be deduced from observational/correlational studies [38]. Women with better vaginal function may be more likely to comply with dilation therapy instructions compared to those with increased atrophy or stricture. Further, the independent effect of vaginal dilators as opposed to use of vaginal gels, moisturizers or hormonal therapies [39–41] has not yet been fully elucidated, and there are no randomized trial data. Although there have been few singular reports of dehiscence, ulceration, or fistula related to VDT, such events are exceedingly rare with modern treatment, proper patient VDT education, and counseling [41]. It is commonly accepted that VDT safely delays or prevents VS and is considered “best practice” in an international guidelines statement [2].
Table 3:
Summary of Observational Studies Assessing the Relationship between Dilator Use and Vaginal Condition or Toxicity (Chronologic Order).
| Study | Cancer Type | Patient number | Study Design | Outcome Measure | Result |
|---|---|---|---|---|---|
| Decruze, 1999 [SR6] | Endometrial and cervical cancers | 70 | Observational, retrospective | Vaginal stenosis, not otherwise specified | 11% versus 57% of patients who did use versus did not use a custom-made vaginal stent had stenosis at follow-up |
| Velaskar, 2007 [SR7] | Locally advanced cervical cancer | 89 | Observational, retrospective | Vaginal length | Vaginal length increased from 6cm post RT to 9cm following vaginal dilation for 4 months |
| Bahng, 2012 [7] | Endometrial cancer | 100 | Observational, retrospective | CTCAE, Vaginal toxicity | Use of dilator at least 2–3 times per week was significantly associated with lower risk of vaginal mucosal toxicity on multivariate analysis (p<0.05) |
| Gondi, 2012 [19] | Locally advanced cervical cancer | 374 | Observational, retrospective | CTCAE, vaginal toxicity | Dilator compliance was defined as use 2 or more times per week for the first 2 years following treatment, with at least monthly use thereafter. At 3 years, probability of grade 3 late vaginal toxicity correlated with vaginal dilator compliance (p<0.05): High compliance: 15.6% Moderate compliance: 41.6% Poor compliance:44.9% |
| Law, 2015 [SR8] | GI and Gyn cancers | 109 | Observational, prospective | Patient’s ability to maintain her pre-RT vaginal dilator size | Compliance with dilators at 6 months was associated with increased likelihood of being able to insert pre-RT dilator at 12 months (p<0.05). |
| Vagal, 2017 [SR9] | Locally advanced cervical cancer | 183 | Observational, retrospective | Vaginal dimensions as measured with medium size vaginal dilator insertion | Those who underwent vaginal dilator therapy both maintained vaginal patency and also had significantly increased vaginal length from ~8cm to ~10cm |
| Stahl, 2018 [41] | Endometrial cancer | 243 | Observational, retrospective | CTCAE, vaginal stenosis | On multivariate analysis, lack of consistent dilator use (2 or more times per week for minimum of 1 year) was an independent predictor of higher VS (p<0.05). At 15 months, incidence of VS (grade ≥1) differed according to degree of VDT compliance (p<0.05): High compliance: 21.4% Moderate compliance: 33.5% Poor compliance:38.8% |
Abbreviations: SR= Supplementary Reference (supplementary references can be found in Appendix 1)
Prospective evidence linking routine dilator use to improved sexual function has been mixed. In two randomized studies in which vaginal dilation was encouraged, there was no improvement observed in womens’ sexual function scores [42,43]. In contrast, a recent nurse-led interventional study of 20 GYN cancer patients showed that an intervention targeting sexual recovery and VDT led to improved sexual function [44]; cause and effect related specifically to VDT could not be proven. Despite mixed evidence, experienced practitioners recognize VDT as an important tool along with vaginal lubricants, moisturizers, and hormonal therapies in maintaining vaginal and sexual function after cancer treatment [2, 45, 46].
Question 5: What are strategies to improve compliance with VDT?
The low adherence rate of GYN cancer survivors to VDT recommendations is well-recognized and has been reported in multiple observational cohort studies [7, 41] with compliance in the early months post RT ranging from 20–70%, with precipitous decrease thereafter.
There have been a number of interventional studies examining methods to improve rates of compliance. Two small randomized controlled trials, with 32 and 42 patients respectively, demonstrated that a psycho-educational program can improve compliance compared with routine educational provisions among women with cervical or endometrial cancer [42, 43]. A more recent randomized controlled trial of 42 endometrial cancer patients conversely found no difference in adherence between an enhanced educational program with standard practice. The lack of difference between the arms was attributed to the small number of patients and a high attrition rate [47]. Additional prospective, nonrandomized, pilot studies have been done addressing psycho-educational interventions designed to improve adherence with VDT, with promising results [44].
Outside of the context of a clinical trial, interviews with patients have provided insightful qualitative data regarding factors which might facilitate VDT in routine practice. In one study, women noted that they were motivated when their doctor examined them and provided them with positive feedback; they were discouraged if their doctor did not talk about dilator use and if the pelvic exam was uncomfortable [48]. Some women may be more motivated to use a dilator if they understand its value for enabling pelvic examination and, therefore, cancer surveillance rather than solely as a strategy to maintain vaginal patency for sexual function [49]. Other motivating factors derived from interviews include patients’ feelings about having received adequate VS education, making it part of a routine, acceptance of VDT as an extension of her medical treatment, and perception of VDT as preventative care [50]. Additional simple strategies that increase patients’ physical comfort with VDT include lubricant use, routine vaginal moisturizer use 3–4 times per week to improve vaginal tissue quality, and use of smaller, flexible, or softer (rather than uncomfortable) dilators or vibrators [45, 48].
Question 6: Are radiation oncologists consistent in the way that they prescribe VDT and monitor patients?
In practice, it appears that ROs are fairly consistent regarding certain aspects of VDT education, provision, and technique [14]. In a large US survey of 233 practicing ROs by Kachnic et al, the most common responses regarding recommended VDT technique included initiation of use within 6 weeks of RT completion, frequency of 3 times per week, for 5–10 minutes per session, and for a duration of >12 months after RT completion [14]. However, other aspects of VDT protocols, which may be critical to women’s success and comfort with dilators, have not been ascertained. For example, guidelines from a group of international experts suggest that practitioners review the need for VDT on a “regular basis” with their patients and suggest discontinuation of VDT when “it is no longer required; e.g. when sexually active or experiencing no discomfort during vaginal examinations.” [2] These authors also suggest that if VS develops, physicians “should record vaginal toxicity using a recognized score.” It is unknown whether ROs – or other specialists such as gynecologists, or GYN oncologists responsible for post-radiation surveillance - consistently assess vaginal capacity pre-and post- VDT, measure and record VS during the follow-up period, or feel comfortable managing the resultant problems related to shortening, reduced elasticity, and dyspareunia. In one survey of US practicing ROs, specialists who treated primarily gastrointestinal malignancies were less likely than GYN specialists to perform routine GYN exam or refer for sexual counseling suggesting variance among ROs in monitoring [14]. Among 119 ROs surveyed in the Netherlands, 44% indicated a need for additional training in sexual counseling in RO practice [13]. Finally, in a survey of patients who had received pelvic RT, only 50% recalled having had a discussion with their RO provider concerning sexual health needs [SR10].
DISCUSSION
This review updates the state of knowledge about VS after RT and its management. Based on the literature review, we conclude that VS is a well described consequence of RT, with early manifestations as well as late changes evolving over several years. The pathophysiology of VS suggests that biologic changes continue to evolve for at least 4–5 years after treatment [19,24]. Established risk factors include dose and volume of vagina irradiated [8, 20–21, 24, 31, 33–37]. The CTCAE is the recommended scoring system for VS in contemporary studies and correlates with known risk factors [7–9, 19–21], though the scale is subjective and therefore inter-rater reliability may be low. RT-induced vaginal changes can interfere with sexual function [10–12] and correlational studies support the use of preventive VDT [7,19,21, SR6–9], though level I evidence is lacking [38]. There is widespread acceptance in the RO community concerning the efficacy of VDT to mitigate VS.
It is unknown to what extent ROs monitor VDT adherence or manage symptoms of VS. There are likely differences across disciplines (GYN versus non-GYN) in the culture and practice of routine vaginal examination before/after RT, as well as comfort in measuring vaginal capacity and VS. It is likely that some practitioners rely on other GYN specialists to monitor the vaginal examination following pelvic RT and not perform GYN examination as part of their routine surveillance. A survey of RO practitioners to assess how they surveil their female patients who receive pelvic RT with respect to recognition, monitoring, and treatment of VS would be informative.
Without a standard in terms of how long patients need to use dilators, their monitoring may be a crucial component to determine what is feasible, effective or acceptable to a patient on an individual basis. While the literature would suggest that VS evolves over a time course of 3–5 years in endometrial and cervical cancer populations [19, 24], and correlational data would suggest that longer duration of VDT is better [19, 41], expert opinion suggests that a shorter course of VDT is commonly recommended. This gap may reflect philosophical differences, population differences or recognition among providers of the complexity of issues which drive non-compliance [SR11-SR12]. We agree conceptually with the suggestions for VDT monitoring laid out in the international expert guidelines previously cited [2], though a well-designed, prospective, randomized trial to guide the optimal duration of VDT should be a research priority. Certainly, all women undergoing pelvic RT should be counseled about the potential short and long term effects on vaginal and sexual function, as is routine for men undergoing pelvic radiation treatment. The best available study suggests half of women can recall no such counseling, which we find unacceptable.
It has been suggested that increased training opportunities for ROs to feel comfortable in managing the sexual sequelae of pelvic RT in their female patients are needed [13]. Towards this end, there are a number of recently published educational resources which have been written to help guide providers in assessing their oncologic patients’ sexual function and concerns [SR13] and in intervening for those who have negative sexual sequelae from their treatment [SR14]. Lastly, a review paper by Kirchheiner et al. provides a detailed visual description of vaginal examination findings post-RT, a topic which is not commonly taught during RO or GYN residency training [SR15] and this may be instructive for clinicians to classify the common changes to vaginal tissue which are seen post-RT including mucosal pallor, telangiectasia, fragility of the vaginal wall, ulceration, and adhesions/occlusion.
Our approach is limited in scope. Relevant topics not covered in the present study, but worthy of future examination, include psychologic factors that might affect VDT, special populations at high risk for VS, socioeconomic disparities relating to VDT use or monitoring, and relevance of VDT practices in lower resource settings. Although we attempted to include all relevant publications, studies lacking the key words used in our search may have been inadvertently missed. Also, given the lack of high quality evidence, no definite strong recommendations can be made for monitoring patients. We recognize that variations in practice exist.
In summary, it has been almost 50 years since vaginal dilators were first recommended as a way to minimize VS following pelvic RT [1]. Our review of the literature, as well as earlier reviews [45–46] conclude that we now have growing evidence on which to base VDT after pelvic RT [4,7–9, 19–21, SR6–9]. We also observe a convergence of international guidelines that support the routine practice of VDT [2, SR12]. Future work is needed to articulate disease site-specific guidelines and to address the need for pre- and post- treatment vaginal examinations for GYN and other non-GYN pelvic malignancies. Optimal duration of VDT can only be determined with follow-up of an individual patient over time, given the lack of consistency and more generalizable evidence on this topic. Finally, empirical evidence is needed to determine whether and over what duration dilator use results in improvements in sexual function and cancer-related outcomes.
Supplementary Material
Acknowledgments
The project did not receive funding. Dr. Lindau has no relevant conflicts of interest to disclose. Her effort on this study has been supported by philanthropic funding for WomanLab at the University of Chicago. Dr. Lindau discloses that she and her spouse own stocks and mutual funds, including some related to health care, that are managed by third parties.
Footnotes
Conflicts of Interest: The authors report no relevant conflicts of interest to disclose.
Disclaimer: The work performed by Dr. Jeffery on this manuscript was independent from her federal employment.
Supplementary Data
Supplementary data related to this article can be found in Appendix 1.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1-.Hartman P, Diddle AW. Vaginal stenosis following irradiation therapy for carcinoma of the cervix uteri. Cancer 1972;30:426–429. [DOI] [PubMed] [Google Scholar]
- 2-.International guidelines on vaginal dilation after pelvic radiotherapy. Produced by the International Clinical Guideline Group, chaired by Dr Tracie Miles, President, National Forum of Gynaecological Oncology Nurses, UK. https://owenmumford.com/us/wpcontent/uploads/sites/3/2014/11/Dilator-Best-Practice-Guidelines.pdf [Google Scholar]
- 3-.Khor TH, Tuan JK, Hee SW, et al. Radical radiotherapy with high-dose-rate brachytherapy for uterine cervix cancer long-term results. Australas Radiol 2007;51:570–577. [DOI] [PubMed] [Google Scholar]
- 4-.Fraunholz IB, Schopohl B, Böttcher HD Management of radiation injuries of vulva and vagina. Strahlenther Onkol 1998;174 Suppl 3:90–2. [PubMed] [Google Scholar]
- 5-.Nori D, Merimsky O, Batata M, et al. Postoperative high dose-rate intravaginal brachytherapy combined with external irradiation for early stage endometrial cancer: a long-term follow-up. Int J Radiat Oncol Biol Phys 1994;30:831–837. [DOI] [PubMed] [Google Scholar]
- 6-.Souhami L, Melo JA, Pareja G. The treatment of stage III carcinoma of the uterine cervix with telecobalt irradiation. Gynecol Oncol 1987;28:262–267. [DOI] [PubMed] [Google Scholar]
- 7-.Bahng AY, Dagan A, Bruner DW, et al. Determination of prognostic factors for vaginal mucosal toxicity associated with intravaginal high-dose rate brachytherapy in patients with endometrial cancer. Int J Radiat Oncol Biol Phys 2012;82:667–673. [DOI] [PubMed] [Google Scholar]
- 8-.Kirchheiner K, Nout RA, Lindegaard JC, et al. Dose-effect relationship and risk factors for vaginal stenosis after definitive radio(chemo)therapy with image-guided brachytherapy for locally advanced cervical cancer in the EMBRACE study. Radiother Oncol 2016;118:160–166. [DOI] [PubMed] [Google Scholar]
- 9-.Mirabeau-Beale K, Hong TS, Niemierko A, et al. Clinical and treatment factors associated with vaginal stenosis after definitive chemoradiation for anal canal cancer. Pract Radiat Oncol 2015;5:e113–e118. [DOI] [PubMed] [Google Scholar]
- 10-.Bergmark K, Avall-Lundqvist E, Dickman PW, et al. Vaginal changes and sexuality in women with a history of cervical cancer. New England Journal of Medicine 1999;340:1383–1389. [DOI] [PubMed] [Google Scholar]
- 11-.Kollberg KS, Waldenström AC, Bergmark K, et al. Reduced vaginal elasticity, reduced lubrication, and deep and superficial dyspareunia in irradiated gynecological cancer survivors, Acta Oncologica 2015;54:772–779. [DOI] [PubMed] [Google Scholar]
- 12-.Jensen PT, Groenvold M, Klee MC, et al. Longitudinal study of sexual function and vaginal changes after radiotherapy for cervical cancer. Int J Radiat Oncol Biol Phys 2003;56: 937–949. [DOI] [PubMed] [Google Scholar]
- 13-.Krouwel EM, Nicolai MP, van der Wielen GJ, et al. Sexual concerns after (pelvic) radiotherapy: is there any role for the radiation oncologist? J Sex Med 2015;12:1927–1939. [DOI] [PubMed] [Google Scholar]
- 14-.Kachnic LA, Bruner DW, Quereshi MM et al. Perceptions and practices regarding women’s vaginal health following radiation therapy: a survey of radiation oncologists practicing in the USA. Pract Radiat Oncol 2017;7:356–363. [DOI] [PubMed] [Google Scholar]
- 15-.medical-dictionary. thefreedictionary.com/vaginal+stenosis
- 16-.Mac Bride MB, Rhodes DJ, Shuster LT. Vulvovaginal atrophy. Mayo Clin Proc 2010;85:87–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17-.Mehdi I, Al Bahrani BJ, Al Lawati TM, et al. Breast cancer in a patient with Kindler’s syndrome. J Pak Med Assoc. 2017;67:1283–1286. [PubMed] [Google Scholar]
- 18-.Bruner DW, Lanciano R, Keegan M, et al. Vaginal stenosis and sexual function following intracavitary radiation for the treatment of cervical and endometrial cancer. Int J Radiat Oncol Biol Phys 1993;27:825–830. [DOI] [PubMed] [Google Scholar]
- 19-.Gondi V, Bentzen SM, Sklenar KL, et al. Severe late toxicities following concomitant chemoradiotherapy compared to radiotherapy alone in cervical cancer: an inter-era analysis. Int J Radiat Oncol Biol Phys 2012;84:973–982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20-.Son CH, Law E, Oh JH et al. Dosimetric predictors of radiation-induced vaginal stenosis after pelvic radiation therapy for rectal and anal cancer. Int J Radiat Oncol Biol Phys 2015; 92:548–554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21-.Park HS, Ratner ES, Lucarelli L, et al. Predictors of vaginal stenosis after intravaginal high-dose-rate brachytherapy for endometrial carcinoma. Brachytherapy 2015;14:464–470. [DOI] [PubMed] [Google Scholar]
- 22-.Yoshida K, Yamazaki H, Nakamura S, et al. Longitudinal analysis of late vaginal mucosal reactions after high-dose-rate brachytherapy in patients with gynecological cancer. Anticancer Research 2014;34: 4433–4438. [PubMed] [Google Scholar]
- 23-.Grigsby PW, Russell A, Bruner D, et al. Late injury of cancer therapy on the female reproductive tract. Int J Radiat Oncol Biol Phys 1995;31:1281–1299. [DOI] [PubMed] [Google Scholar]
- 24-.Sorbe BG, Smeds AC Postoperative vaginal irradiation with high dose rate afterloading technique in endometrial carcinoma stage I. Int J Radiat Oncol Biol Phys 1990;18:305–314. [DOI] [PubMed] [Google Scholar]
- 25-.Abitbol M, Davenport J. The irradiated vagina. Obstet. Gynecol 1974;44:249–256. [PubMed] [Google Scholar]
- 26-.Hofsjo A, Bohn-Starke N, Blomgren B, et al. Radiotherapy-induced vaginal fibrosis in cervical cancer survivors. Acta Oncologica 2017;56:661–666. [DOI] [PubMed] [Google Scholar]
- 27-.Pitkin RM, VanVoorhis LW. Postirradiation vaginitis: an evaluation of prophylaxis with topical estrogen. Therapeutic Radiology 1971;99:417–421. [DOI] [PubMed] [Google Scholar]
- 28-.Katz A, Njuguna E, Rakowsky E, et al. Early development of vaginal shortening during radiation therapy for endometrial or cervical cancer. Int J Gynecol Cancer 2001;11:234–235. [DOI] [PubMed] [Google Scholar]
- 29-.Rakhra SS, Weaver C, Donnelly ED, et al. The effect of pelvic radiotherapy on vaginal brachytherapy cylinder diameter: Implications for optimal treatment order. Brachytherapy 2016;15:549–553. [DOI] [PubMed] [Google Scholar]
- 30-.Brand AH, Bull CA, Cakir B Vaginal Stenosis in patients treated with radiotherapy for carcinoma of the cervix. Int J Gynecol Cancer 2006;16:288–293. [DOI] [PubMed] [Google Scholar]
- 31-.Onsrud M, Strickert T, Marthinsen ABL. Late reactions after postoperative high-dose-rate intravaginal brachytherapy for endometrial cancer: a comparison of standardized and individualized target volumes. Int J Radiat Oncol Biol Phys 2001;49:749–755. [DOI] [PubMed] [Google Scholar]
- 32-.Weiss E, Hirnly P, Arnold-Bofinger H, et al. Therapeutic outcome and relation of acute and late side effects in the adjuvant radiotherapy of endometrial carcinoma stage I and II. Radiotherapy and Oncology 1999;53:37–44. [DOI] [PubMed] [Google Scholar]
- 33-.Eifel PJ, Levenback C, Wharton JT, et al. Time course and incidence of late complications in patients treated with radiation therapy for FIGO stage IB carcinoma of the uterine cervix. Int J Radiat Oncol Biol Phys 1995; 32:1289–1300. [DOI] [PubMed] [Google Scholar]
- 34-.Sorbe B, Straumits A, Karlsson L. intravaginal high-dose-rate brachytherapy for stage I endometrial cancer: a randomized study of two dose-per-fraction levels. Int J Radiat Oncol Biol Phys 2005;62:1385–1389. [DOI] [PubMed] [Google Scholar]
- 35-.Singh R, Chopra S, Engineer R, et al. Dose-volume correlation of cumulative vaginal doses and late toxicity after adjuvant external radiation and brachytherapy for cervical cancer. Brachytherapy 2017;16:855–861. [DOI] [PubMed] [Google Scholar]
- 36-.Susko M, Craciunescu O, Meltsner S, et al. Vaginal dose is associated with toxicity in image guided tandem ring or ovoid-based brachytherapy. Int J Radiat Oncol Biol Phys 2016;94:1099–1105. [DOI] [PubMed] [Google Scholar]
- 37-.Hintz BL, Kagan AR, Chan P, et al. Radiation tolerance of the vaginal mucosa. Int J Radiat Oncol Biol Phys 1980;6:711–716. [DOI] [PubMed] [Google Scholar]
- 38-.Miles T, Johnson N. Vaginal dilator therapy for women receiving pelvic radiotherapy. Cochrane Database of Systematic Reviews 2014;, Issue 9. Art. No.: CD007291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39-.Galuppi A, Perrne AM, Macchia ML, et al. Local a-tocopherol for acute and short-term vaginal toxicity prevention in patients treated with radiotherapy for gynecologic tumors. Int J Gynecol Cancer 2011;21: 1708–1711. [DOI] [PubMed] [Google Scholar]
- 40-.Laliscia C, Delishaj D, Fabrini MG, et al. Acute and late vaginal toxicity after adjuvant high-dose-rate vaginal brachytherapy in patients with intermediate risk endometrial cancer: is local therapy with hyaluronic acid of clinical benefit? J Contemp Brachytherapy 2016;8:512–517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41-.Stahl JM, Qian JM, Tien CJ, et al. Extended duration of dilator use beyond 1 year may reduce vaginal stenosis after intravaginal high-dose rate brachytherapy. Support Care Cancer 2019; 27:1425–1433. [DOI] [PubMed] [Google Scholar]
- 42.Jeffries SA, Robinson JW, Craighead PS, Keats MR. An effective group psychoeducational intervention for improving compliance with vaginal dilation; a randomized trial. International Journal of Radiation Biology and Physics 2006; 6:404–411. [DOI] [PubMed] [Google Scholar]
- 43.Robinson JW, Faris PD, Scott CB. Psychoeducational group increases vaginal dilation for younger women and reduces sexual fears for women of all ages with gynecological carcinoma treated with radiotherapy. International Journal of Radiation Oncology Biology and Physics 1999;.44: 497–506. [DOI] [PubMed] [Google Scholar]
- 44.Bakker RM, Mens JWM, de Groot HE, et al. A nurse-led sexual rehabilitation intervention after radiotherapy for gynecological cancer. Support Care Cancer 2017; 25:729–737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Huffman LB, Hartenbach EM, Carter J, et al. Maintaining sexual health throughout gynecologiccancer survivorship: a comprehensive review and clinical guide. Gynecol Oncol 2016; 140:359–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Morris L, Do V, Chard J, et al. Radiation-induced vaginal stenosis: current perspectives. Int J Womens Health 2017;2:273–279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hanlon A, Small W Jr, Strauss J, et al. Dilator use after vaginal brachytherapy for endometrial cancer: A randomized feasibility and adherence study. Cancer Nurs. 2017. April 21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bakker RM, Vermeer WM, Creutzberg CL, et al. Qualitative accounts of patients’ determinants ofvaginal dilator use after pelvic radiotherapy. J Sex Med 2015;12:764–773. [DOI] [PubMed] [Google Scholar]
- 49.Friedman LC, Abdallah R, Schluchter M et al. Adherence to vaginal dilation following high dose rate brachytherapy for endometrial cancer. Int J Rad Oncol Biol Phys 2011: 80;751–757. [DOI] [PubMed] [Google Scholar]
- 50.Bonner C, Nattress K, Anderson C, et al. Chore or priority? Barriers and facilitators affecting dilatoruse after pelvic radiotherapy for gynaecological cancer. Support Care Cancer 2012; 20:2305–2313. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.