Abstract
Elderly patients undergoing cardiac surgery are at an increased risk of adverse postoperative outcomes. Frailty, a state of decreased physiological reserve, is highly prevalent among elderly patients. Despite being associated with adverse surgical outcomes, no universally accepted definition or measurement tool for frailty exists. Moreover, regardless of all the recommendations, a routine perioperative frailty assessment is often ignored. In addition to complications, frailty increases the burden to the healthcare system, which is of particular concern in Southeast Asia due to its socioeconomically disadvantaged and resource limited settings. This narrative review focuses to develop clinical practice plans for perioperative frailty assessment in the context of a cardiac surgical setting.
Keywords: Cardiac surgery, frailty, geriatric, perioperative, risk stratification
INTRODUCTION
With the advancements in modern medicine, life expectancy in Southeast Asia has increased dramatically during the 20th century. The elderly population in low to middle-income countries has been predicted to increase more than three times compared to higher-income countries between 2010 and 2050.[1,2] Due to an increase in the aging demographic, surgeons are more likely to operate on these patients with up to 30% of invasive procedures performed during the final year of life.[3] This becomes important in the setting of cardiac surgery, where elderly patients with extensive valvular or coronary artery disease[4] are more likely to require urgent or emergent procedures. Progressive aging is associated with frailty, a syndrome of cumulative decline in physiological systems predisposing vulnerable patients to adverse outcomes. Accordingly, there is a need to better understand frailty in the context of perioperative care for older cardiac surgical patients.
Frailty and progressive aging
Age has historically been used to predict adverse surgical outcomes. It is intuitive that other factors being equal, a patient who is 26 years old will likely fare better than one who is 76 years old. The aging process involves several changes in body composition, such as increased fat mass, the loss of skeletal muscle (sarcopenia), dysregulated inflammation, and decreased bone mineral density (osteopenia/osteoporosis).[3] However, surgical outcomes vary greatly among older patients of similar ages. These outcomes can be explained by the concept of frailty, which highlights the discrepancies between a patient's chronological and physiological age.[4] Frailty—traditionally a geriatric assessment to identify disabilities and weaknesses in patients—much more accurately predicts clinical morbidity, mortality, delirium, and loss of independence after surgical procedures when compared to age alone.[5,6]
Frailty definition and models
Though no universal definition of frailty exists, it is generally considered to be a state of vulnerability and diminished physiologic reserve in response to external stressors.[7] Physical strength and endurance are significant components of frailty, but frailty is a multidimensional syndrome that takes into account other factors of daily living such as emotional state and cognitive function. There are two major physiological models of frailty based on deficit accumulation and phenotype. The deficit accumulation model defines frailty as an increasing number of accumulated medical, functional, and social deficits over time.[8,9] The phenotype model defines frailty as an age-related physiological state of weakness, exhaustion, slowness, and body composition.[9,10]
Disadvantages of existing risk prediction scores
The most commonly used risk prediction models, such as the Society of Thoracic Surgeons (STS) risk score and the European System for Cardiac Operative Risk Evaluation (EuroSCORE), often underestimate the perioperative risk for older patients and major surgeries.[11] These scores are generally accurate at the population level, but often fail at an individual level because they do not take nondemographic factors like patient frailty into account.[6]
Inadequate perioperative assessment of frailty
Preoperative frailty assessment and prehabilitation practices are recommended by the official Guidelines for Perioperative Care in Cardiac Surgery[12] and the Society of Perioperative Assessment and Quality Improvement (SPAQI).[13] Despite these recommendations, frailty is not being routinely assessed before surgery. In a 2018 national survey, greater than 80 percent of anesthesiologists stated that they “rarely or never” routinely assess for frailty.[5] There are many possible reasons for the lack of routine frailty assessment during the perioperative period. Firstly, there is a lack of consensus among different frailty instruments which have varying sensitivity and specificity. Secondly, the gold standard for frailty assessment involves a Comprehensive Geriatric Assessment (CGA), a multidisciplinary diagnostic and treatment process that identifies medical, psychosocial, and functional limitations of a frail older person.[14] Since the CGA generally requires a series of visits with a geriatric specialist, this form of frailty assessment is not always feasible before cardiac surgery, especially in urgent situations. Thirdly, the majority of elderly patients in developing countries reside in rural areas and encounter obstacles before accessing the healthcare system such as lower health literacy, lack of supportive infrastructure, and fewer options for dedicated specialty care when compared to their counterparts in major metropolitan areas.[15,16]
Journal selection and extraction
For the purpose of this narrative review, we specifically looked to identify the modifiable risk factors affecting the postsurgical outcomes in the frail/elderly undergoing cardiac surgery and to establish investigational modalities translatable to a resource-limited setting. In order to achieve this, a literature search was conducted on databases such as PUBMED, Cochrane, Embase, and Google scholar. In conjunction to a database search, we also performed targeted searches on data published by prominent researchers in the field to include all relevant information available on frailty. Grey literature was reviewed for statistical data and data from South East Asian Region. To ensure that the database search was all encompassing, we defined our key words, Boolean terms, and inclusion and exclusion criteria. For instance, the scientific papers included were published between 2009 and 2019, elderly was defined as age 60 years and above, and papers published in language other than English were excluded.
Given the importance of frailty assessment and the aging demographics in Southeast Asia, this article aims to make narrative recommendations regarding preoperative, intraoperative, and postoperative frailty interventions with regards to cardiac surgery in resource limited settings [Table 1]. Whenever possible, we aim to identify interventions which are cheap and reliable, taking into account the unique considerations of the Indian healthcare system.
Table 1.
Frailty assessment and intervention during perioperative period - A stepwise approach (original diagram)
| STEP 1: Preoperative period | |
|---|---|
| Chair rise | 30-second chair rise for leg strength and endurance |
| Sitting in chair crossing arms on shoulders | |
| Number of times the patient can stand to a full upright position and sit down | |
| Gait speed | Small “acceleration” and “deceleration” zone flanking the actual testing zone |
| Timer started at the beginning and stopped at the end of the testing zone | |
| Gait speed >5 seconds for 4 meters (<0.85 m/s) increases risk of frailty | |
| Hand grip strength | Measured using a hydraulic dynamometer for isometric arm strength |
| Frail scale | Five-question verbal assessment of fatigue, resistance, aerobic capacity, illnesses and loss of weight |
| Categorized into three groups: robust (score=0), prefrail (1-2), and frail (3-5) | |
| Nutrition | Malnutrition when one of following is present |
| Weight loss more than 10 to 15% within 6 months | |
| Body mass index less than 18 kg/m2 | |
| Subjective global assessment of grade C | |
| Serum albumin less than 30 g/L | |
| STEP 2: Intraoperative period | |
| Lung protective strategies | |
| Minimal use of anesthetic medications potentially inappropriate for older adults | |
| Decreased intraoperative blood pressure variation | |
| Electroencephalogram (EEG)-guided anesthesia depth monitoring | |
| STEP 3: Postoperative period | |
| Help | Hospital Elder Life Program |
| Comprehensive and multidisciplinary patient-care program | |
| Provides optimal care for older persons in the hospital | |
| Pain | Optimal perioperative pain control |
| Minimal opioids | |
| Other | Chest physiotherapy |
| Early ambulation | |
| Nutrition | |
| Discharge planning | |
Frailty assessment and intervention during perioperative period A stepwise approach
Proper frailty assessment can risk-stratify patients during surgery and help predict their post-operative outcomes. When employed intelligently, clinicians can better facilitate discussions around surgical/anesthesia informed consent, especially in regards to their odds of functional decline after surgery and related activities of daily living. It can also help to identify the patients who are most appropriate for targeted monitoring or mobility protocols.
Since a CGA is usually not feasible in a busy preoperative holding area or clinic, the question remains: which tests are most appropriate to detect frailty? Though not an exhaustive list, we highlight 4 possibilities—chair rise, gait speed, handgrip, and FRAIL scale—that may have utility in Southeast Asia. Then, we describe intraoperative and postoperative factors to consider in the perioperative management of elderly and/or frail patients [Figure 1].
Figure 1.
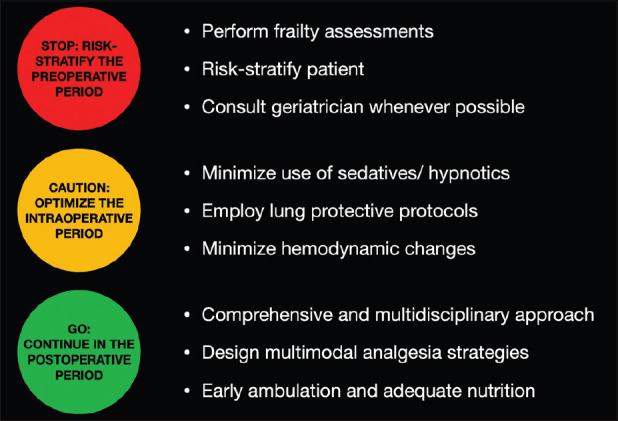
Infographic regarding frailty in the cardiac surgical setting (original diagram)
Step 1: Preoperative Interventions
-
Chair Rise: The 30-second chair rise test is a simple yet effective way in which to indirectly test leg strength and endurance. The patient sits down in a chair and crosses their arms to place their hands on the opposite shoulders. Without using their hands and keeping their feet flat on the floor, one can measure how many times the patient can stand to a full upright position and sit down in 30 seconds. These numbers can then be compared to a validated cohort for particular age ranges[17]
A recent prospective cohort study found that low chair rise scores had the highest sensitivity for predicting the outcome of delayed functional recovery after Surgical Aortic Valve Replacement (SAVR) and persistent functional impairment after Transcatheter Aortic Valve Replacement (TAVR), especially when combined with a CGA.[18] Furthermore, objective measures of lower extremity performance such as the chair rise test increase the likelihood of detecting impaired mobility (restricted mobility/disability) when compared to self-reported mobility restriction or disability.[19] The chair rise test can be employed quickly and does not require specialized personnel or equipment, conferring advantages for its use as a frailty screening instrument
Gait Speed: The gait speed test is another single-item test which involves timing an individual while they walk at a steady pace for a set distance, usually around 5 meters. The patient is often given a small “acceleration” and “deceleration” zone flanking the actual testing zone. One starts the timer at the beginning of the testing zone and ends the timer at the end of the testing zone; generally, a gait speed greater than 5 seconds for 4 meters (<0.85 m/s) is associated with an increased risk of frailty.[20] Since walking speed is related to a complex interplay of functions such as aerobic capacity,[21] proprioception,[22] and vision,[23] further testing or interventions are then warranted to ascertain the underlying physiological process. A multicenter prospective cohort of elderly cardiac surgical patients found that slow gait speed (less than 5 meters in 6 seconds) was an independent predictor of a composite end point of major morbidity or mortality after cardiac surgery[24]
Hand Grip: Hand grip strength (HGS) is a basic measure in the determination of musculoskeletal function and frailty. Generally, HGS is measured using a hydraulic dynamometer to obtain an objective measure of isometric arm strength, which can then be exported to a local laptop or smartphone. After normalizing for body weight, Chung et al showed if a patient's hand grip strength (measured by a dynamometer in kg) was lower than 25% of their total body weight, these patients were associated with lower survival in a cohort of patients with advanced heart failure before ventricular assist device implementation (75.0% vs 92.9% at 6 months).[25] This effect persisted for up to 3 years after left ventricular assist device (LVAD) placement. Multicomponent frailty instruments offer better risk discrimination than single-component ones.[19] For example, Sündermann et al. found that a composite metric (CAF score) of grip strength, walking speed, balance, chair rise, picking up a pen, and removing a jacket accurately predicted 1-year mortality and correlated with the length of intensive care unit stay.[26] Obtaining a measure of hand grip strength requires specialized equipment, meaning it is more difficult to obtain in the immediate preoperative setting
-
FRAIL Scale: Enhanced Recovery After Surgery (ERAS) Guidelines for Perioperative Care in Cardiac Surgery[12] recommend general frailty assessment and preoperative graded exercise regimens, but do not endorse any particular scale or timeframe in which these interventions should occur. A wide array of frailty scores exist, many of which require invasive lab values that may not be routinely assessed before cardiac surgery and have varying sensitivity and specificity for surgical outcomes
The FRAIL scale is a short five-question verbal assessment of fatigue, resistance, aerobic capacity, illnesses and loss of weight. The scale categorizes patients into three groups: robust (score = 0), prefrail (score = 1-2), and frail (score = 3-5). After adjustment by age, gender, and comorbidity, a statistically significant association was found between frailty and length of stay, post-operative complications, and discharge disposition to home versus rehabilitation in geriatric hip fracture patients.[27] The benefit of the FRAIL scale is that it is a short screen which is quickly operationalized into any pre-operative clinic or evaluation. In Southeast Asia, where the surgical volume is larger and the appointment time is shorter, these differences add up over time and make it more likely that a frailty assessment will be used in a meaningful way by clinicians
Nutrition: Tissue injury induced by surgical trauma activates the sympathetic nervous system and the immune-hypothalamic-pituitary-adrenal axis.[28] In a recent narrative review, Gillis et al. summarize the important role that perioperative nutrition can play in modulating the surgical stress response.[29] Severe malnutrition is defined by the European Society for Clinical Nutrition and Metabolism (ESPEN) when one of the following is present: weight loss more than 10 to 15% within 6 months; body mass index less than 18 kg/m2; subjective global assessment of nutritional status falling under grade C (severely malnourished); or serum albumin less than 30 g/L (with no evidence of hepatic or renal dysfunction).[30] While intensive nutrition support for 7 to 10 days preoperatively has limited evidence for influencing morbidity and mortality in noncardiac surgical patients, it is unclear how effective these interventions may be in undernourished patients or in the cardiac surgical population in particular.[12] Strong randomized trials investigating the clinical significance of nutrition therapy in cardiac surgical patients are sparse. However, the American Society for Clinical Nutrition and Metabolism (ASPEN) suggest iatrogenic malnutrition is common and recommend the initiation of enteral nutrition within 24 h after surgery and a supplementation of 25–30 kcal and 1.5–2.5 g of protein per day and kilogram ideal body weight for critically ill patients.[31]
After frailty has been diagnosed through nutritional, cognitive, or physical/physiological screening tests, the next question for clinicians is how to move forward. Regardless, a recent narrative review[32] suggests that once preoperative screening identifies frailty and potential underlying causal contributors, the anesthesiologist should attempt to optimize that particular component before surgery. For example, if the problem involves nutrition, the anesthesiologist should ensure adequate protein and iron supplementation immediately after surgery; conversely, if the problem involves physical frailty, the patient could be referred to an exercise prehabilitation program. Here, logistical issues with patients admitted only 12–24 hours before surgery become challenging and must be balanced against the additional risk of adverse outcomes in the frail patient.
Step 2 Intraoperative interventions
While frailty tests/scales act as screening tools and predictors for postoperative complications, no consensus exists on how to decrease the risk of cardiovascular surgery in frail patients by modifying intraoperative management. In a recent narrative review, Graham et al. suggest applying geriatric principles of anesthetic care to the management of frail older adults.[33] Some of these principles include employing lung protective strategies in the operating room and minimizing the usage of anesthetic medications on the Beers Criteria of potentially inappropriate medications for older adults, such as long-acting benzodiazepines, diphenhydramine, scopolamine, and promethazine.[34]
The cardiac autonomic nervous system is likely to be impaired in frail patients as compared to nonfrail older adults, as indicated by preoperative blood pressure complexity, reduced heart rate variability, and intraoperative hemodynamics[35,36] For example, a single-center retrospective study of 1223 noncardiac surgical patients found an association of frailty with decreased intraoperative blood pressure variation (as measured by episodes of absolute change >15% in fractional mean arterial pressure) and that the relationship between frailty and 30-day mortality was partially mediated by these episodes of autonomic dysregulation.[37]
It is possible that using intraoperative anesthetic depth monitoring may decrease the amount of anesthetic required and therefore allow for more hemodynamic stability. Electroencephalogram (EEG) monitored anesthetic practice often relies on processed EEG scores, with numbers given from zero to one hundred to infer the depth of unconsciousness. Lower numbers refer to known deleterious patterns on the raw EEG waveform, such as burst suppression, a deep state of anesthetic-induced coma that has been associated with increased likelihood of perioperative neurocognitive disorders. However, elderly and/or frail patients display age-dependent changes in thalamocortical function reflected in increased burst suppression and increased risk of delirium after cardiac surgery.[38,39] While evidence is currently inconclusive for anesthetic depth monitoring and prevention of delirium, large studies have found a 30-day mortality benefit for the avoidance of burst suppression.[40] As more EEG-machines enter the market and cheaper open-source options improve, raw waveform-based neuromonitoring during cardiac surgery in high risk or frail patients may provide benefit.
Step 3 Postoperative interventions
There are a multitude of ways to improve the care of frail patients in the postoperative setting. One of the thoroughly studied mechanisms is the Hospital Elder Life Program (HELP), a comprehensive and multidisciplinary patient-care program that provides optimal care for older persons in the hospital.[41] In addition to targeted and interdisciplinary geriatric assessment, the HELP volunteer model provides a structured mechanism to deliver daily orientation, early mobilization, feeding assistance, and a nonpharmacological sleep protocol in preventing delirium.[42] While there is a small upfront cost to implement the HELP program, the program saves approximately $831 USD per person-year in hospital costs once implemented primarily through prevention of delirium and falls (in addition to potential litigation fees).[43]
Frail patients are at high risk for other postoperative complications such as increased pain levels, pulmonary complications, and nutritional deficits. Recent combined recommendations from the American College of Surgeons and the American Geriatrics Society suggest ways to optimize the perioperative period for frail and geriatric patients.[44] Deliriogenic medications such as amitriptyline and benzodiazepines should be minimized with regional anesthesia and opioid-sparing analgesia techniques employed to improve pain control. Chest physiotherapy and lung-protective ventilation can be used to help prevent alveolar lung injury. Early functional rehabilitation should be prioritized with a combination of early patient ambulation (using physical therapy whenever possible), universal fall precautions to avoid injuries, and regular repositioning in order to prevent pressure sores. Since frail patients tend to have lower levels of muscle mass, it is imperative that a balanced diet with nutritional supplementation be resumed as quickly as possible following cardiac surgery.
Discharge planning for frail patients should include the consultation of a dietician for continuation of a long-term nutrition plan. Frail patients should ideally transition into skilled healthcare facilities. However, it is imperative to discuss the potential financial and social strains that may occur with family and caregivers during postoperative recovery and reintegration into normal activities of daily living. Low health literacy and poor communication from healthcare providers can result in adverse events for frail patients, so realistic expectations should be set with the opportunity to answer follow-up questions over the phone or during a later clinic appointment.
CONCLUSION
For many surgeons and anesthesiologists, patient frailty is the “elephant in the operating room”; it is easy to spot but is often ignored.[45] There are multiple limitations to using subjective or intuitive measures of detecting frailty. These limitations underscore the importance of using one or more instruments to screen for frailty in the preoperative period. Screening for frailty is especially important in developing countries such as India, where resource limitations and preexisting obstacles for frail patients predispose them to worse surgical outcomes.
Given an increasing number of elderly and frail patients undergoing cardiac surgery in India and the anesthesiologist's role in optimizing the perioperative period, it is crucial that frailty assessment be operationalized with buy-in from hospital administration. The optimization of patient frailty ideally includes lifestyle changes such as improved nutritional status and strength training in the primary care setting, with robust multidisciplinary follow-up in the postoperative months that follow. In Southeast Asia, family members and social connections often aid in the comprehensive care of frail individuals.[46] Evidence and tools exist for institutions to make a significant impact on the lives of patients; it is up to anesthesiologists to lead the charge.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Suzman R, Beard J. World Health Organization & US National Institute of Aging. Global Health and Aging. 2011:4. [Google Scholar]
- 2.Shetty P. Grey matter: Ageing in developing countries. Lancet. 2012;379:1285–7. doi: 10.1016/s0140-6736(12)60541-8. [DOI] [PubMed] [Google Scholar]
- 3.Panayi AC, Orkaby AR, Sakthivel D, Endo Y, Varon D, Roh D, et al. Impact of frailty on outcomes in surgical patients: A systematic review and meta-analysis. Am J Surg. 2019;218:393–400. doi: 10.1016/j.amjsurg.2018.11.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Afilalo J, Mottillo S, Eisenberg MJ, Alexander K, Noiseux N, Perrault L, et al. Addition of frailty and disability to cardiac surgery risk scores identifies elderly patients at high risk of mortality or major morbidity. Circ Cardiovasc Qual Outcomes. 2012;5:222–8. doi: 10.1161/CIRCOUTCOMES.111.963157. [DOI] [PubMed] [Google Scholar]
- 5.Bentov I, Kaplan SJ, Pham TN, Reed MJ. Frailty assessment: From clinical to radiological tools. Br J Anaesth. 2019;123:37–50. doi: 10.1016/j.bja.2019.03.034. [DOI] [PubMed] [Google Scholar]
- 6.Furukawa H, Tanemoto K. Frailty in cardiothoracic surgery: Systematic review of the literature. J Thorac Cardiovasc Surg. 2015;63:425–33. doi: 10.1007/s11748-015-0553-8. [DOI] [PubMed] [Google Scholar]
- 7.Robinson TN, Eiseman B, Wallace JI, Church S, McFann K, Pfister S, et al. Redefining geriatric preoperative assessment using frailty, disability and co-morbidity. Am Surg Assoc. 2009;127:93–9. doi: 10.1097/SLA.0b013e3181b45598. [DOI] [PubMed] [Google Scholar]
- 8.Cooper Z, Rogers SO, Ngo L, Guess J, Schmitt E, Jones RN, et al. Comparison of Frailty Measures as Predictors of Outcomes After Orthopedic Surgery. J Am Geriatr Soc. 2016;64:2464–71. doi: 10.1111/jgs.14387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Robinson TN, Walston JD, Brummel NE, Deiner S, Brown C, Kennedy M, et al. Frailty for surgeons: Review of a National Institute on Aging Conference on Frailty for Specialists. J Am Coll Surg. 2015;221:1083–92. doi: 10.1016/j.jamcollsurg.2015.08.428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hall DE, Arya S, Schmid KK, Blaser C, Carlson M, Bailey T, et al. Development and initial validation of the risk analysis index for measuring frailty in surgical populations. JAMA Surg. 2017;152:175–82. doi: 10.1001/jamasurg.2016.4202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Guida P, Mastro F, Scrascia G, Whitlock R, Paparella D. Performance of the European system for cardiac operative risk evaluation II: A meta-analysis of 22 studies involving 145,592 cardiac surgery procedures. J Thorac Cardiovasc Surg. 2014;148:3049–57. doi: 10.1016/j.jtcvs.2014.07.039. [DOI] [PubMed] [Google Scholar]
- 12.Engelman DT, Ben Ali W, Williams JB, Perrault LP, Reddy VS, Arora RC, et al. Guidelines for perioperative care in cardiac surgery: Enhanced recovery after surgery society recommendations. JAMA Surg. 2019;154:755–66. doi: 10.1001/jamasurg.2019.1153. [DOI] [PubMed] [Google Scholar]
- 13.Alvarez-Nebreda ML, Bentov N, Urman RD, Setia S, Huang JC, Pfeifer K, et al. Recommendations for preoperative management of frailty from the Society for Perioperative Assessment and Quality Improvement (SPAQI) J Clin Anesth. 2018;47:33–42. doi: 10.1016/j.jclinane.2018.02.011. [DOI] [PubMed] [Google Scholar]
- 14.Ellis G, Whitehead MA, Robinson D, O'Neill D, Langhorne P. Comprehensive geriatric assessment for older adults admitted to hospital: Meta-analysis of randomised controlled trials. BMJ. 2011:343. doi: 10.1136/bmj.d6553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shaheen M, Puri S, Tandon N. An overview of frailty in elderly. Indian J Geront. 2016;12:58–65. [Google Scholar]
- 16.Kumar S. Frailty index- assessment tools in elderly: Feasibility in India. AGEMS. 2017;4:45–9. [Google Scholar]
- 17.30-Second Chair Stand Assessment. STEADI: Stopping Elderly Accidents, Deaths, and Injuries. [Last accessed on 2020 Jan 19]. Published 2017. Available from: https://www.cdc.gov/steadi/pdf/STEADI-Assessment-30Sec-508.pdf .
- 18.Kim DH, Afilalo J, Shi SM, Popma J, Khabbaz K, Laham R, et al. Evaluation of changes in functional status in the year after aortic valve replacement. JAMA Intern Med. 2019;179:383–91. doi: 10.1001/jamainternmed.2018.6738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kim DH, Kim CA, Placide S, Lipsitz LA, Marcantonio ER. Preoperative frailty assessment and outcomes at 6 months or later in older adults undergoing cardiac surgical procedures: A systematic review. Ann Intern Med. 2016;165:650–60. doi: 10.7326/M16-0652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Middleton A, Fritz SL, Lusardi M. Walking speed: The functional vital sign? J Aging Phys Act. 2015;23:314–22. doi: 10.1123/japa.2013-0236. doi: 10.1123/japa.2013-0236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fiser WM, Hays NP, Rogers SC, Kajkenova O, Williams AE, Evans CM, et al. Energetics of walking in elderly people: Factors related to gait speed. J Gerontol A Biol Sci Med Sci. 2010;65:1332–7. doi: 10.1093/gerona/glq137. [DOI] [PubMed] [Google Scholar]
- 22.Park YH, Kim YM, Lee BH. An ankle proprioceptive control program improves balance, gait ability of chronic stroke patients. J Phys Ther Sci. 2013;25:1321–4. doi: 10.1589/jpts.25.1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Aartolahti E, Hakkinen A, Lonnroos E, Kautiainen H, Sulkava R, Hartikainen S, et al. Relationship between functional vision and balance and mobility performance in community-dwelling older adults. Aging Clin Exp Res. 2013;25:545–52. doi: 10.1007/s40520-013-0120-z. [DOI] [PubMed] [Google Scholar]
- 24.Afilalo J, Eisenberg MJ, Morin J-F, Bergman H, Monette J, Noiseux N, et al. Gait speed as an incremental predictor of mortality and major morbidity in elderly patients undergoing cardiac surgery. J Am Coll Cardiol. 2010;56:1668–76. doi: 10.1016/j.jacc.2010.06.039. [DOI] [PubMed] [Google Scholar]
- 25.Chung CJ, Wu C, Jones M, Kato TS, Dam TT, Givens R, et al. Reduced handgrip strength as a marker of frailty predicts clinical outcomes in patients with heart failure undergoing ventricular assist device placement. J Cardiac Fail. 2014;20:310–5. doi: 10.1016/j.cardfail.2014.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sündermann S, Dademasch A, Praetorius J, Kempfert J, Dewey T, Falk V, et al. Comprehensive assessment of frailty for elderly high-risk patients undergoing cardiac surgery. Eur J Cardiothorac Surg. 2011;39:33–7. doi: 10.1016/j.ejcts.2010.04.013. [DOI] [PubMed] [Google Scholar]
- 27.Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir. 2017;18:1082–6. doi: 10.1016/j.jamda.2017.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kyrou I, Tsigos C. Stress hormones: Physiological stress and regulation of metabolism. Curr Opin Pharmacol. 2009;9:787–93. doi: 10.1016/j.coph.2009.08.007. [DOI] [PubMed] [Google Scholar]
- 29.Gillis C, Carli F. Promoting perioperative metabolic and nutritional care. Anesthesiology. 2015;123:1455–72. doi: 10.1097/ALN.0000000000000795. [DOI] [PubMed] [Google Scholar]
- 30.Braga M, Ljungqvist O, Soeters P, Fearon K, Weimann A, Bozzetti F. ESPEN guidelines on parenteral nutrition: Surgery. Clin Nutr. 2009;28:378–86. doi: 10.1016/j.clnu.2009.04.002. [DOI] [PubMed] [Google Scholar]
- 31.Hill A, Nesterova E, Lomivorotov V, Efremov S, Goetzenich S, Benstoem C, et al. Current evidence about nutrition support in cardiac surgery patients—What do we know? Nutrients. 2018;10:597. doi: 10.3390/nu10050597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.McClave SA, Taylor BE, Martindale RG, Warren MM, Johnson DR, Braunschweig C, et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (ASPEN) JPEN J Parenter Enter Nutr. 2016;40:159–211. doi: 10.1177/0148607115621863. [DOI] [PubMed] [Google Scholar]
- 33.Graham A, Brown CH. Frailty, aging, and cardiovascular surgery. Anesth Analg. 2017;124:1053–60. doi: 10.1213/ANE.0000000000001560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.By the 2019 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2019 Updated AGS Beers Criteria® for Potentially Inappropriate Medication Use in Older Adults: 2019 AGS Beers Criteria Expert Panel®. J Am Geriatr Soc. 2019;67:674–94. doi: 10.1111/jgs.15767. [DOI] [PubMed] [Google Scholar]
- 35.Parvaneh S, Howe CL, Toosizadeh N, Honarvar B, Slepian M, Fain M, et al. Regulation of cardiac autonomic nervous system control across frailty statuses: A systematic review. Gerontology. 2015;62:3–15. doi: 10.1159/000431285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rangasamy V, Henriques TS, Xu X, Subramaniam B. Preoperative blood pressure complexity indices as a marker for frailty in patients undergoing cardiac surgery. J Cardiothorac Vasc Anesth. 2020;34:616–21. doi: 10.1053/j.jvca.2019.09.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.James LA, Levin MA, Lin H-M, Deiner SG. Association of preoperative frailty with intraoperative hemodynamic instability and postoperative mortality. Anesth Analg. 2019;128:1279–85. doi: 10.1213/ANE.0000000000004085. [DOI] [PubMed] [Google Scholar]
- 38.Ni K, Cooter M, Gupta DK, Thomas J, Hopkins T, Miller T, et al. Paradox of age: Older patients receive higher age-adjusted minimum alveolar concentration fractions of volatile anaesthetics yet display higher bispectral index values. Br J Anaesth. 2019;123:288–97. doi: 10.1016/j.bja.2019.05.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nomura Y, Nakano M, Bush B, Tian J, Yamaguchi A, Walston J, et al. Observational study examining the association of baseline frailty and postcardiac surgery delirium and cognitive change. Anesth Analg. 2019;129:507–14. doi: 10.1213/ANE.0000000000003967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wildes TS, Mickle AM, Ben Abdallah A, Maybrier H, Oberhaus J, Budelier TP, et al. Effect of electroencephalography-guided anesthetic administration on postoperative delirium among older adults undergoing major surgery: The ENGAGES randomized clinical trial. JAMA. 2019;321:473–83. doi: 10.1001/jama.2018.22005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Inouye SK, Bogardus ST, Charpentier PA, Leo-Summers L, Acampora Denise, Holford TR, et al. A multicomponent intervention to prevent delirium in hospitalized older patients. NEJM. 1999;340:669–76. doi: 10.1056/NEJM199903043400901. [DOI] [PubMed] [Google Scholar]
- 42.Reuben DB, Inouye SK, Bogardus ST, Baker DI, Leo-Summers L, Cooney LM. Models of geriatric practice; The hospital elder life program: A model of care to prevent cognitive and functional decline in older hospitalized patients. J Am Geriatr Soc. 2000;48:1697–706. doi: 10.1111/j.1532-5415.2000.tb03885.x. [DOI] [PubMed] [Google Scholar]
- 43.Rizzo JA, Bogardus ST Jr, Leo-Summers L, Williams CS, Acampora D, Inouye SK. Multicomponent targeted intervention to prevent delirium in hospitalized older patients: What is the economic value? Medical Care. 2001;39:740–52. doi: 10.1097/00005650-200107000-00010. [DOI] [PubMed] [Google Scholar]
- 44.Mohanty S, Rosenthal RA, Russell MM, Neuman MD, Ko CY, Esnaola NF. Optimal perioperative management of the geriatric patient: A best practices guideline from the American College of Surgeons NSQIP and the American Geriatrics Society. J Am Coll Surg. 2016;222:930–47. doi: 10.1016/j.jamcollsurg.2015.12.026. [DOI] [PubMed] [Google Scholar]
- 45.Hubbard RE, Story DA. Patient frailty: The elephant in the operating room. Anaesthesia. 2014;69:26–34. doi: 10.1111/anae.12490. [DOI] [PubMed] [Google Scholar]
- 46.Chatterjee P. Health and Wellbeing in Late Life: Perspectives and Narratives from India. Singapore: Springer; 2019. [Google Scholar]


